Abstract
Background
Pre-eclampsia can lead to maternal and neonatal complications and is a common cause of maternal mortality worldwide. This review has examined the effect of micronutrient supplementation interventions in women identified as having a greater risk of developing pre-eclampsia.
Methods
A systematic review was performed using the PRISMA guidelines. The electronic databases MEDLINE, EMBASE and the Cochrane Central Register of Controlled trials were searched for relevant literature and eligible studies identified according to a pre-specified criteria. A meta-analysis of randomised controlled trials (RCTs) was conducted to examine the effect of micronutrient supplementation on pre-eclampsia in high-risk women.
Results
Twenty RCTs were identified and supplementation included vitamin C and E (n = 7), calcium (n = 5), vitamin D (n = 3), folic acid (n = 2), magnesium (n = 1) and multiple micronutrients (n = 2). Sample size and recruitment time point varied across studies and a variety of predictive factors were used to identify participants, with a previous history of pre-eclampsia being the most common. No studies utilised a validated prediction model. There was a reduction in pre-eclampsia with calcium (risk difference, −0.15 (−0.27, −0.03, I2 = 83.4%)), and vitamin D (risk difference, −0.09 (−0.17, −0.02, I2 = 0.0%)) supplementation.
Conclusion
Our findings show a lower rate of pre-eclampsia with calcium and vitamin D, however, conclusions were limited by small sample sizes, methodological variability and heterogeneity between studies. Further higher quality, large-scale RCTs of calcium and vitamin D are warranted. Exploration of interventions at different time points before and during pregnancy as well as those which utilise prediction modelling methodology, would provide greater insight into the efficacy of micronutrient supplementation intervention in the prevention of pre-eclampsia in high-risk women.
Subject terms: Nutrition, Risk factors, Hypertension
Introduction
Pre-eclampsia is a hypertensive disorder of pregnancy associated with a high risk of maternal, fetal and neonatal morbidity [1]. Pre-eclampsia has been defined as high blood pressure after 20 weeks’ gestation associated with one or more of the following: proteinuria, multisystemic maternal organ dysfunction or placental dysfunction [2, 3]. Pre-eclampsia affects around 2–8% of pregnancies globally, with approximately 10–15% of direct maternal deaths being attributed to pre-eclampsia and eclampsia [4].
The pathophysiology of pre-eclampsia is not fully understood and this disorder presents as a clinical syndrome with a wide spectrum [5]. Early onset pre-eclampsia is generally considered as a defect in placentation whilst late onset pre-eclampsia is more often attributed to a range of interacting factors including normal placental senescence and a genetic predisposition to cardiovascular and metabolic disease [5]. Poor placental function has repeatedly been associated with oxidative stress [6].
Several systematic reviews have assessed the effects of single and multiple micronutrients on the risk of developing pre-eclampsia. High dose calcium supplementation has been shown to be effective in reducing pre-eclampsia, particularly in women with low dietary calcium intake, but with limited evidence on the effects of low dose supplementation [7]. A recent umbrella review [8] reported that vitamin D supplementation reduced pre-eclampsia, while reporting limited or no effect of iron, folic acid supplementation or of the antioxidants, vitamin C and/or E. Despite magnesium being utilised in the treatment of pre-eclampsia and eclampsia, previous reviews have not been able to establish an effect of magnesium supplementation [9]. Similarly, many reviews [10, 11] report no established effect of zinc supplementation. There is increasing interest in the role of multiple micronutrient supplements (MMS) and their potential benefit for pregnant women, particularly in low-income countries where more than one micronutrient deficiency may co-exist. A meta-analysis of 28 RCTs [12] reported that despite evidence from observational cohort studies reporting a reduction in the risk of pre-eclampsia following MMS, there was a lack of effect from RCTs.
Interventions may be better targeted to women with more specific risk for adverse pregnancy outcomes. Several studies have used prediction modelling to identify those women more likely to develop pre-eclampsia [13–15]. An externally validated model from The Avon Longitudinal Study of Parents and Children cohort used routinely collected data to predict pre-eclampsia risk in a 12,996 women. The study combined maternal early pregnancy characteristics (including initial mean arterial pressure [MAP]) with repeatedly measured MAP collected from 20–36 weeks’ gestation. The authors found that blood pressure recorded from 28 weeks’ gestation improved the model’s identification of women who would go on to develop pre-eclampsia with an area under the curve of 0.81 and 0.83 in the validated cohort [13]. Other cohorts have frequently combined clinical risk factors with biomarkers and imaging techniques such as uterine artery Doppler ultrasound recorded in the first trimester [16]. Multivariable prediction models such as this have often demonstrated better performance with predicting early-onset pre-eclampsia [17]. The application of predictive modelling in the context of preventative micronutrient interventions in pre-eclampsia may offer insight into effectiveness of predictive factors and algorithms in clinical practice.
To the best of our knowledge, there has been no review of studies which have utilised prediction tools to stratify interventions intended to reduce pre-eclampsia. There is a need to evaluate interventions that utilise micronutrient supplementation in women who have been identified as high-risk for pre-eclampsia. Moreover, there is a paucity of data on the effects of pre-pregnancy micronutrient interventions on the development of pre-eclampsia. As healthcare aims to move towards primary prevention, it is important to assess supplementation interventions prior to pregnancy on the development of pre-eclampsia. Finally, few reviews report the effect of micronutrient supplementation in women with differing severity of pre-eclampsia (mild, severe and superimposed) who have been identified as high risk, which could allow more tailored and personalised preventative approaches in the future.
The overall aim of this review was to assess RCTs of micronutrient supplementation (single and multiple micronutrients) either pre-pregnancy and/or during pregnancy to prevent pre-eclampsia in women identified as high risk. An additional aim included examining the effect of intervention on different severities of pre-eclampsia in higher-risk women.
Methods
This systematic review was registered in the PROSPERO database (CRD42021240941) and conducted according to the PRISMA guidelines [18].
Inclusion and exclusion criteria
The PICOS (population, intervention, comparison, outcomes and study design) framework described in Table 1 was used to develop the inclusion and exclusion criteria. Studies were eligible if they met the following criteria: (1) RCTs evaluating single or multiple micronutrient supplementation before and/or during pregnancy compared with a control arm (no supplementation, placebo, dose difference or alternative micronutrient supplementation intervention) with a primary or secondary outcome of any classification of pre-eclampsia; (2) reproductive aged women between 18 and 50 years who intended to become pregnant or were pregnant at any gestation; (3) women identified as high risk of developing pre-eclampsia using a defined eligibility criteria at study entry. Studies meeting the following criteria were excluded: (1) observational and non-randomised studies; (2) abstracts, reviews, letters, comments and editorials; (3) women with existing pre-eclampsia; (4) women aged less than 18 years or more than 50 years; (5) studies not published in English.
Table 1.
PICOS framework summary.
| P – Population | Women between the ages of 18 to 50 who were planning to become pregnant or were pregnant at any gestation, and at high-risk for pre-eclampsia identified using a defined eligibility criteria at study entry |
| I – Intervention | Interventions included micronutrient supplementation, in isolation or as multiple micronutrients. This included, but was not limited to calcium, vitamin D, folic acid, iron, zinc and magnesium |
| C – Comparison | No supplementation, placebo, different dose or alternative micronutrient supplementation intervention |
| O – Outcome | The main outcome was the development of pre-eclampsia. Secondary outcomes included gestational hypertension, eclampsia, diastolic and systolic blood pressure, HELLP syndrome, premature rupture of membranes, placental abruption, preterm birth, low birthweight, birth weight centile, small for gestational age (SGA), caesarean section, miscarriage, Apgar scores and maternal death |
| S – Study type | Randomised controlled trials |
Primary and secondary outcomes
The primary outcome of this review was the development of pre-eclampsia of any classification including mild, severe and superimposed pre-eclampsia, defined by any diagnostic criteria ranging from the use of systolic (SBP) and diastolic blood pressures (DBP), urinary protein measurements and other relevant clinical indicators such as liver enzymes and platelet count. Trials that reported definitions for severe pre-eclampsia generally defined this with the same diagnostic criteria, however with higher thresholds for blood pressure and urinary protein measurement. The secondary outcomes included gestational hypertension, eclampsia, diastolic and systolic blood pressure, HELLP syndrome, premature rupture of membranes, placental abruption, preterm birth, low birthweight, birth weight centile, small for gestational age (SGA), caesarean section, miscarriage, Apgar scores and maternal death, of which 7 of these secondary outcomes have been identified as part of the recommended core outcome set for pre-eclampsia for future studies [19]. Data were extracted on secondary outcomes from studies when available.
Literature search and study selection
The electronic databases MEDLINE, EMBASE and the Cochrane Central Register of Controlled trials were searched by two reviewers (SG, DDALS) on 14th April 2021. Search strategies are shown in Supplementary Information 1. Results of the search strategy were imported into EndNote for removal of duplicates and the remaining articles were imported into Rayyan for title and abstract screening [20]. If eligibility could not be determined by the title and abstract, full-text articles were screened. Any disagreement was resolved by a third reviewer (ACF). Additional studies were examined for eligibility through hand searching of reference lists.
Data extraction
Two reviewers (SG, DDALS) carried out the data extraction in duplicate and any disagreements were resolved by discussion or by consultation with another author to achieve a consensus opinion (ACF and KVD). A data extraction template was developed which included: title, authors, publication data, trial periods, study design, country of study, aim of study, sample size, characteristics of participants, inclusion and exclusion criteria, period of intervention (preconception and/or pregnancy), type and dose of intervention and clinical outcomes.
Data synthesis
The interventions and outcomes were assessed for suitability for data pooling to perform a meta-analysis. The analysis focused on assessing the effect of micronutrient interventions (single or multiple micronutrients) on the development of pre-eclampsia, the primary outcome, in high-risk populations. Micronutrient interventions included calcium, vitamin D, vitamin C and E, folic acid, magnesium and MMS in women defined as high risk of developing any classification of pre-eclampsia including severe pre-eclampsia, as previously defined. Summaries of exposure effect for each intervention were provided using a risk difference, calculated using Stata, version 16. The risk differences were calculated using a random effects model and the I2 statistic was used to assess heterogeneity amongst studies, with a threshold of >50% indicating significant heterogeneity. When meta-analysis was not possible due to too few studies, a narrative synthesis was performed.
Risk of bias (quality) assessment
The Cochrane Risk of Bias tool for randomised trials (RoB 2) [21] was used to assess the quality of each study included. The domains assessed include randomisation selection (selection bias), allocation concealment (selection bias), follow-up of participant from recruitment to termination of study and dropout (attrition bias) and other potential sources of bias. Disagreement between reviewers was resolved by discussion, with the overall risk of each study being deemed as either “low risk of bias,” “some concerns” or “high risk of bias.”
Results
The electronic database search resulted in 8922 articles. Following removal of duplicates, a total of 7237 articles were screened for eligibility using titles and abstracts. Full-text screening was conducted on 168 articles. Major reasons for exclusion included: publication type (e.g. commentary articles or protocols of clinical trials), ineligible outcome (e.g. studies that did not include pre-eclampsia in the results), ineligible population (e.g. low risk women, adolescents), studies which were not published in English, ineligible study design (e.g. review and observational studies), incorrect type of intervention (e.g. pharmacological intervention) and retraction of trials. Twenty articles [22–41] met the inclusion criteria and were included in this review. The study identification and selection process are summarised in Fig. 1.
Fig. 1. PRISMA Flow Diagram.
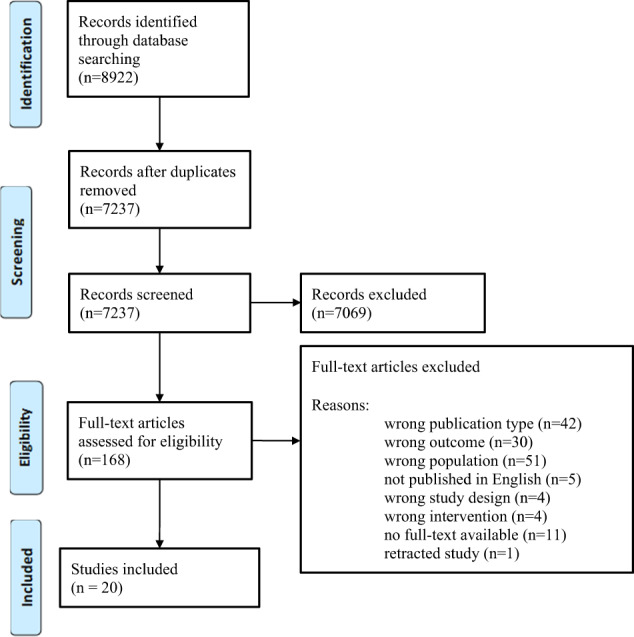
Flow diagram showing the study selection process.
Characteristics of included studies
This review included RCTs published between 1991 to 2021. A summary of these RCTs is shown in Table 2. The characteristics of studies including women identified as high-risk for pre-eclampsia are shown in Table 3. Six studies [23, 24, 29, 30, 32, 38] were conducted in Iran, three studies [31, 33, 39] in the United States, two studies [25, 40] in the United Kingdom, two studies [26, 34] in Brazil, one study in each of Colombia [27], India [28], Mexico [35] and China [36]. Three studies were multicentre and were conducted in South Africa, Zimbabwe and Argentina [22]; India, Peru, South America and Vietnam [37]; Argentina, Australia, Canada, Jamaica and the United Kingdom [41]. Fifteen studies were carried out in low- and middle-income countries [22–24, 26–30, 32, 34–38, 41]. The lowest sample size was 50 [28] and the largest sample size 2464 [41]. The studies aimed to assess the efficacy of either the supplementation of calcium, vitamin D, vitamin C in combination with vitamin E, magnesium, folic acid or MMS in reducing the incidence of pre-eclampsia in women classified at high-risk of pre-eclampsia at study entry. Of the 20 studies, two targeted women before pregnancy [18, 32]. The earliest time a pregnancy intervention was introduced was at 8 weeks’ gestation [41] with most of the studies continuing the micronutrient supplementation until delivery.
Table 2.
Summary of included studies.
| Reference | Publication date | Period of intervention | Country of study | Micronutrient supplementation intervention | Screening for pre-eclampsia | Gestational age during intervention |
|---|---|---|---|---|---|---|
| Hofmeyr et al. | 2019 | Prepregnancy into pregnancy | South Africa, Zimbabwe, Argentina | Calcium | Previous pre-eclampsia | From prepregnancy to 20 weeks’ gestation |
| Baba Dizavandy et al. | 1998 | Pregnancy | Iran | Calcium | Positive roll-over test | From 28–32 weeks gestation to delivery |
| Herrera et al. | 1998 | Pregnancy | Colombia | Calcium and linoleic acid | Positive roll-over test and high mean arterial pressure | From 28–32 weeks’ gestation to delivery |
| Niromanesh et al. | 2001 | Pregnancy | Iran | Calcium | Positive roll-over test and at least one risk factor for pre-eclampsia | Until delivery |
| Sanchez-Ramos et al. | 1994 | Pregnancy | USA | Calcium | Positive angiotensin sensitivity test | Not reported |
| Behjat et al. | 2017 | Pregnancy | Iran | Vitamin D3 | Previous pre-eclampsia | Until 36 weeks’ gestation |
| Karamali et al. | 2015 | Pregnancy | Iran | Vitamin D3 | Uterine artery doppler | From 20 weeks’ to 32 weeks’ gestation |
| Samimi et al. | 2015 | Pregnancy | Iran | Vitamin D3 and calcium | Laboratory tests including free ß-human chorionic gonadotrophin, inhibin α dimeric, unconjugated oestriol and maternal serum α-foetoprotein and haemodynamic assessment of uterine artery Doppler waveform at 16–20 weeks of gestation | From 20 weeks’ to 32 weeks’ gestation |
| Chappell et al. | 1999 | Pregnancy | UK | Vitamin C and vitamin E | Uterine artery doppler and previous pre-eclampsia | From 16–22 weeks’ gestation (depending on prior history of pre-eclampsia) to delivery |
| Poston et al. | 2006 | Pregnancy | UK and Holland | Vitamin C and vitamin E | One or more risk factors for pre-eclampsia (previous pre-eclampsia, delivery <37 weeks, diagnosis of HELLP in previous pregnancy, essential hypertension requiring medication currently or previously, maternal diastolic blood pressure of ≥90 mmHg before 20 weeks’ gestation in the current pregnancy, type 1 or type 2 diabetes requiring insulin or oral hypoglycaemic therapy before the pregnancy, antiphospholipid syndrome, chronic renal disease, multiple pregnancy, abnormal uterine artery doppler waveform, primiparity with BMI at first antenatal appointment of ≥30) | From 14–21 weeks’ gestation to delivery |
| Spinatto et al. | 2007 | Pregnancy | Brazil | Vitamin C and vitamin E | Previous pre-eclampsia in most recent pregnancy that progressed beyond 20 weeks’ gestation | From 12–19 weeks’ gestation to delivery |
| Kalpdev et al. | 2010 | Pregnancy | India | Vitamin C and vitamin E | Essential hypertension | From 16–22 weeks’ gestation to delivery |
| Beazley et al. | 2005 | Pregnancy | USA | Vitamin C and vitamin E | Previous pre-eclampsia, chronic hypertension, insulin-requiring diabetes or multiple gestation | From 14–20 weeks’ gestation |
| Vadillo-Ortega et al. | 2011 | Pregnancy | Mexico | Vitamin C and vitamin E | Previous pre-eclampsia or pre-eclampsia in first-degree relative | From 14–32 weeks’ gestation to delivery |
| Villar et al. | 2009 | Pregnancy | India, Peru, South Africa, Vietnam | Vitamin C and vitamin E | High risk for pre-eclampsia (chronic hypertension, renal disease, pre-eclampsia-eclampsia in the pregnancy preceding the index pregnancy requiring delivery before 37 weeks’ gestation, HELLP syndrome in any previous pregnancy, pregestational diabetes, primiparous with a BMI ≥ 30, history of preterm delivery, abnormal uterine artery Doppler waveforms and women with antiphospholipid syndrome) | 14–22 weeks’ gestation to delivery |
| Zheng et al. | 2020 | Prepregnancy into pregnancy | China | Folic acid | Previous pre-eclampsia | Preconception to delivery |
| Wen et al. | 2018 | Pregnancy | Argentina, Australia, Canada, Jamaica and UK | Folic acid | At least one risk factor for pre-eclampsia (pre-existing hypertension, prepregnancy type 1 or type 2 diabetes, twin pregnancy, previous pre-eclampsia or BMI ≥ 35) | From 8–16 weeks’ gestation to delivery |
| De Araujo et al. | 2020 | Pregnancy | Brazil | Magnesium | ≥1 risk factor for preterm birth or adverse perinatal outcomes in a prior pregnancy (i.e. preterm delivery <37 weeks, still birth at 201/7 weeks, placental abruption, pre-eclampsia or eclampsia, a live-born infant with SGA birthweight <3rd percentile or liveborn infant with birthweight <2500 g) or in current pregnancy (i.e. nulliparity, chronic hypertension, type 1 or 2 diabetes mellitus, maternal age >35 years, pre-pregnancy BMI > 30 or smoking) | From 12–20 weeks’ gestation to delivery |
| Azami et al. | 2017 | Pregnancy | Iran | MMS | At least one risk factor for pre-eclampsia (including chronic vascular disease, hydatidiform mole, multiparity, diabetes mellitus, thyroid disease, chronic hypertension, nulliparity, history of pre-eclampsia, maternal age >35 years, kidney disease, collagen vascular disease, antiphospholipid antibody syndrome, family history of pre-eclampsia, history of thrombophilia and BMI > 25) | From 20 weeks’ gestation to delivery |
| Parrish et al. | 2013 | Pregnancy | US | MMS | Previous pre-eclampsia, prior eclampsia, history of chronic hypertension, diabetes mellitus, connective tissue disease or inherited/acquired thrombophilia | Any time up to 12 weeks’ gestation until delivery |
Table 3.
Characteristics of included studies.
| Reference | Publication date | Period of intervention | Study design | Sample size | Study period | Country of study | Study aim |
|---|---|---|---|---|---|---|---|
| Hofmeyr et al. | 2019 | Prepregnancy into pregnancy | Multicentre, parallel arm, double-blind, randomised, placebo-controlled trial | N = 1355 | 2011–2016 | South Africa, Zimbabwe, Argentina | To test whether calcium supplementation before and in early pregnancy (up to 20 weeks’ gestation) prevents the development of pre-eclampsia |
| Baba Dizavandy et al. | 1998 | Pregnancy | Double-blind randomised trial | N = 143 | Not reported | Iran | To determine the effect of calcium supplementation in the incidence of hypertensive disorders of pregnancy (gestational hypertension and pre-eclampsia) in nulliparous and high risk women |
| Herrera et al. | 1998 | Pregnancy | Randomised double-blind placebo-controlled trial | N = 89 | 1995–1996 | Colombia | To determine the effect of low doses of linoleic acid and calcium on prostaglandin (PG) levels and the prevention of pre-eclampsia |
| Niromanesh et al. | 2001 | Pregnancy | Double-blind placebo randomised controlled trial | N = 30 | Not reported | Iran | To study the effect of calcium supplementation on reduction of pre-eclampsia in Iranian women at high risk of pre-eclampsia |
| Sanchez-Ramos et al. | 1994 | Pregnancy | Randomised, double-blind, placebo-controlled clinical trial | N = 67 | Not reported | USA | To evaluate the efficacy of oral supplemental calcium in reducing the incidence of pregnancy-induced hypertension (gestational hypertension or pre-eclampsia) in angiotensin sensitive nulliparas |
| Behjat et al. | 2017 | Pregnancy | Randomised double-blinded controlled clinical trial | N = 142 | Not reported | Iran | To evaluate if vitamin D supplementation prevents pre-eclampsia in women with history of pre-eclampsia |
| Karamali et al. | 2015 | Pregnancy | Randomised double-blind placebo-controlled trial | N = 60 | 2014 | Iran | To assess the beneficial effects of high-dose (cholecalciferol) vitamin D supplementation on metabolic profiles and pregnancy outcomes among pregnant women at risk for pre-eclampsia |
| Samimi et al. | 2015 | Pregnancy | Prospective, double-blind, placebo-controlled randomised trial | N = 60 | 2014–2015 | Iran | To examine the effects of vitamin D plus calcium administration on metabolic profiles and pregnancy outcomes among women at risk for pre-eclampsia |
| Chappell et al. | 1999 | Pregnancy | Randomised controlled trial | N = 283 | Not reported | UK | To assess the effect of supplementation with vitamin C and E in women at increased risk of the disorder on plasma markers of vascular endothelial activation and placental insufficiency and the occurrence of pre-eclampsia |
| Poston et al. | 2006 | Pregnancy | Randomised controlled trial | N = 2404 | 2003–2005 | UK and Holland | To assess whether supplementation with vitamin C and vitamin E prevents pre-eclampsia in women at increased risk |
| Spinatto et al. | 2007 | Pregnancy | Randomised controlled trial | N = 739 | 2003–2006 | Brazil | To evaluate whether antioxidant supplementation will reduce the incidence of pre-eclampsia among patients at increased risk |
| Kalpdev et al. | 2010 | Pregnancy | Randomised controlled trial | N = 50 | 2005–2007 | India | To investigate whether vitamin C and vitamin E prophylaxis will reduce the incidence of superimposed pre-eclampsia in chronic hypertensive women |
| Beazley et al. | 2005 | Pregnancy | Double-blind randomised controlled trial | N = 109 | Not reported | USA | To determine the effect of supplemental antioxidant vitamins C and E on the rate of pre-eclampsia in high-risk pregnant women |
| Vadillo-Ortega et al. | 2011 | Pregnancy | Randomised controlled trial | N = 628 | 2001–2005 | Mexico | To test the hypothesis that deficiency in L-arginine and antioxidant supplementation would reduce the development of pre-eclampsia in a population at high risk |
| Villar et al. | 2009 | Pregnancy | Multi-centre, randomised, double-blind controlled trial | N = 1365 | 2004–2006 | India, Peru, South Africa, Vietnam | To determine if vitamin C and E supplementation in high-risk pregnant women with low nutritional status reduces pre-eclampsia |
| Zheng et al. | 2020 | Prepregnancy into pregnancy | Randomised controlled trial | N = 1576 | Not reported | China | To investigate whether supplementation with high doses of folic acid would reduce the subsequent development of pre-eclampsia and its adverse outcomes |
| Wen et al. | 2018 | Pregnancy | Randomised, phase III, double-blind, international multi-centre clinical trial | N = 2464 | 2011–2015 | Argentina, Australia, Canada, Jamaica and UK | To determine the efficacy of high dose folic acid supplementation for prevention of pre-eclampsia in women with at least one risk factor: pre-existing hypertension, prepregnancy diabetes (type 1 or 2), twin pregnancy, pre-eclampsia in a previous pregnancy, or body mass index ≥35 |
| De Araujo et al. | 2020 | Pregnancy | Randomised double-blinded controlled clinical trial | N = 911 | 2014–2017 | Brazil | To evaluate magnesium citrate to prevent adverse perinatal and maternal outcomes among women at higher risk |
| Azami et al. | 2017 | Pregnancy | Randomised controlled trial | N = 100 | Not reported | Iran | To investigate the effect of multimineral-vitamin D supplements (calcium, magnesium, zinc and vitamin D) and vitamins (C + E) in the prevention of pre-eclampsia |
| Parrish et al. | 2013 | Pregnancy | Randomised, placebo-controlled double-blind trial | N = 684 | 2004–2011 | US | To evaluate if phytonutrient supplementation initiated in the first trimester of pregnancy and continued throughout gestation prevents pre-eclampsia |
Predictive factors of pre-eclampsia
The studies used a variety of strategies to identify women at risk of developing pre-eclampsia. The number of variables used ranged from one to 15 and included a range of factors from maternal history to clinical biomarkers (Table 4). Thirteen studies [22, 24–26, 31, 34–41] considered a history of pre-eclampsia in a prior pregnancy, with two [35, 38] of these also including family history of pre-eclampsia. Seven studies [26, 31, 37–41] required participants to have at least one risk factor of pre-eclampsia such as chronic hypertension, type 1 or 2 diabetes mellitus, multiple gestation, or history of preterm birth. Five studies [25, 29, 32, 37, 40] used uterine artery doppler waveforms to select participants, either in isolation or within a combination of other maternal characteristics. Three studies [23, 27, 30] included a positive rollover test, while only one study [33] utilised a positive angiotensin sensitivity test. This test was performed in women at 24–28 weeks’ gestation, by infusing increasing doses of angiotensin II every 5 min until the normal cut-off value (12 ng/kg/min) was reached or the DBP increased to 20 mmHg above the baseline before reaching this cut-off value (the effective pressor dose). If a participant’s effective pressor dose was achieved with a rate of less than 12 ng/kg/min, they were deemed to have a positive angiotensin sensitivity test and were then randomised. Three studies [25, 27, 30] required participants to have a combination of predictive factors, for example, both an abnormal two-stage uterine-artery doppler analysis and a previous history of pre-eclampsia [25]. None reported using a validated prediction model to identify women at high-risk of developing pre-eclampsia.
Table 4.
Screening at study entry and micronutrient supplementation interventions in included studies.
| Reference | Screening for pre-eclampsia | Intervention | Gestational age during intervention |
|---|---|---|---|
| Hofmeyr et al. | Women with previous pre-eclampsia and were intending to become pregnant again |
I = 500 mg of calcium carbonate daily from prepregnancy until 20 weeks’ gestation C = Placebo |
From prepregnancy to 20 weeks’ gestation |
| Baba Dizavandy et al. | Nulliparous women with singleton pregnancies, 24 weeks of gestation, blood pressure <140/90 mmHg, a positive roll-over test and hypocalcuria at 28–32 weeks of pregnancy |
I = 2000 mg of calcium gluconate daily C = Placebo |
From 28–32 weeks gestation to delivery |
| Herrera et al. | First pregnancy, gestation between 28–32 weeks, biopsychosocial risk score of 3 or more, positive roll-over test and high mean arterial pressure |
I = 50 mg of linoleic acid and 600 mg of calcium daily C = Placebo |
From 28–32 weeks’ gestation to delivery |
| Niromanesh et al. | High-risk for pre-eclampsia (identified as having positive results on rollover test and at least one risk factor for pre-eclampsia), gestational age between 28–32 weeks, blood pressure <140/90 mmHg |
I = 2000 mg of calcium daily C = Placebo |
Until delivery |
| Sanchez-Ramos et al. | Normotensive nulliparas at 20–24 weeks’ gestation at increased risk of developing pregnancy-induced hypertension (through positive angiotensin sensitivity test) |
I = 2000 mg of calcium daily C = Placebo |
Not reported |
| Behjat et al. | Pre-eclampsia in previous pregnancy, serum 25-hydroxy vitamin D levels >25 ng/ml |
I = 50000 IU of vitamin D3 every 2 weeks C = Placebo |
Until 36 weeks’ gestation |
| Karamali et al. | Pregnant women primigravida, age of 18–40 and risk of pre-eclampsia (identified through uterine artery Doppler) |
I = 50000 IU of vitamin D3 every 14 days C = Placebo |
From 20 weeks’ to 32 weeks’ gestation |
| Samimi et al. | Primigravida women, aged 18–40 years old, at risk for pre-eclampsia (indicated by laboratory tests including free ß-human chorionic gonadotrophin, inhibin α dimeric, unconjugated oestriol and maternal serum α-foetoprotein and haemodynamic assessment of uterine artery Doppler waveform at 16–20 weeks of gestation) |
I = 50000IU of vitamin D3 every 2 weeks and 1000 mg calcium daily C = Placebo |
From 20 weeks’ to 32 weeks’ gestation |
| Chappell et al. | Women with abnormal two-stage uterine-artery doppler analysis weeks and previous history of pre-eclampsia |
I = 1000 mg of vitamin C and 400 IU of vitamin E daily C = Placebo |
From 16–22 weeks’ gestation (depending on prior history of pre-eclampsia) to delivery |
| Poston et al. | Gestational age 14–21 weeks 6 days and one or more risk factors for pre-eclampsia including pre-eclampsia in the pregnancy preceding the index pregnancy, requiring delivery before 37 completed weeks’ gestation, diagnosis of HELLP syndrome in any previous pregnancy at any stage of gestation, essential hypertension requiring medication currently or previously, maternal diastolic blood pressure of ≥90 mmHg before 20 weeks’ gestation in the current pregnancy, type 1 or type 2 diabetes requiring insulin or oral hypoglycaemic therapy before the pregnancy, antiphospholipid syndrome, chronic renal disease, multiple pregnancy, abnormal uterine artery doppler waveform, primiparity with BMI at first antenatal appointment of ≥30 |
I = 1000 mg of vitamin C and 400IU vitamin E daily C = Placebo |
From 14–21 weeks’ gestation to delivery |
| Spinatto et al. | Pregnant women between 12 and 19 6/7 weeks gestation, non-proteinuric chronic hypertension or with a prior history of preeclampsia in their most recent pregnancy that progressed beyond 20 weeks gestation |
I = 1000 mg of vitamin C and 400IU of vitamin E daily C = Placebo |
From 12–19 weeks’ gestation to delivery |
| Kalpdev et al. | Essential hypertension, singleton pregnancy, gestational age 16 to 22 weeks |
I = 1000 mg of vitamin C and 400IU of vitamin E daily C = No supplementation |
From 16–22 weeks’ gestation to delivery |
| Beazley et al. | Pregnancy at 14–20 weeks and 6 days with a history of previous pre-eclampsia, chronic hypertension, insulin-requiring diabetes or multiple gestation |
I = 1000 mg of vitamin C and 400IU of vitamin E daily C = Placebo |
From 14–20 weeks’ gestation |
| Vadillo-Ortega et al. | Increased risk of pre-eclampsia (history of pre-eclampsia or pre-eclampsia in a first degree relative) |
I = 6.6 g of L-arginine + 500 mg of vitamin C + 400IU of vitamin E daily I2 = 500 mg of vitamin C + 400IU of vitamin E daily C = Placebo |
From 14–32 weeks’ gestation to delivery |
| Villar et al. | Pregnant women 14–22 weeks of gestation and with high risk for pre-eclampsia (chronic hypertension, renal disease, pre-eclampsia-eclampsia in the pregnancy preceding the index pregnancy requiring delivery before 37 weeks’ gestation, HELLP syndrome in any previous pregnancy, pregestational diabetes, primiparous with a BMI ≥ 30, history of preterm delivery, abnormal uterine artery Doppler waveforms and women with antiphospholipid syndrome) |
I = 1000 mg of vitamin C and 400IU of vitamin E daily C = Placebo |
14–22 weeks’ gestation to delivery |
| Zheng et al. | Previous pre-eclampsia, planning pregnancy, aged over 18 years, daily folic acid intake before randomization <1.1 mg |
C = Low dose folic acid (0.4 mg) daily I = High dose folic acid (4 mg) daily |
Preconception to delivery |
| Wen et al. | Pregnant women between 8–16 completed weeks of gestation and at least one risk factor for pre-eclampsia including pre-existing hypertension, prepregnancy diabetes (type 1 or 2), twin pregnancy, pre-eclampsia in a previous pregnancy or BMI ≥ 35 |
I = 4 mg of folic acid daily C = Placebo |
From 8–16 weeks’ gestation to delivery |
| De Araujo et al. | Women aged 18 to 45 years, 12 to 20 weeks of gestation, singleton pregnancy and ≥1 risk factor for preterm birth or adverse perinatal outcomes in a prior pregnancy (i.e. preterm delivery <37 weeks, still birth at 201/7 weeks, placental abruption, pre-eclampsia or eclampsia, a live-born infant with SGA birthweight <3rd percentile or liveborn infant with birthweight <2500 g) or in current pregnancy (i.e. nulliparity, chronic hypertension, type 1 or 2 diabetes mellitus, maternal age >35 years, pre-pregnancy BMI > 30 or smoking) |
I = 300 mg of magnesium citrate daily C = Placebo |
From 12–20 weeks’ gestation to delivery |
| Azami et al. | Women with at least one risk factor for pre-eclampsia (including chronic vascular disease, hydatidiform mole, multiparity, diabetes mellitus, thyroid disease, chronic hypertension, nulliparity, history of pre-eclampsia, maternal age >35 years, kidney disease, collagen vascular disease, antiphospholipid antibody syndrome, family history of pre-eclampsia, history of thrombophilia and BMI > 25) |
I1 = Ferrous sulfate tablet + one multimineral vitamin D tablet containing 800 mg of calcium, 8 mg of zinc and 400IU of vitamin D3 daily I2 = Ferrous sulfate tablet + 250 mg of vitamin C and 55 mg of vitamin E daily C = Ferrous sulfate tablet daily |
From 20 weeks’ gestation to delivery |
| Parrish et al. |
For low risk group: nulliparous or multiparous women, singleton gestation and no evidence of systemic vascular disease For high risk group: multiparous patients with singleton gestation and a prior history of preeclampsia (prior eclampsia, prior mild or severe preeclampsia, prior HELLP) or nulliparous/multiparous patients with singleton gestation with a documented history of chronic hypertension, diabetes mellitus, connective tissue disease or inherited/acquired thrombophilia |
I = Phytonutrients (7.5 mg beta-carotene, 234 mg vitamin C, 30 mg vitamin E, 420 mg folate and 60 mg calcium - mix through a concentrate of blended fruit and vegetable juice powder) taken twice daily until delivery C = Placebo |
Any time up to 12 weeks’ gestation until delivery |
Micronutrient interventions
The type of intervention varied between studies (Table 4). The majority evaluated the effect of single micronutrients including calcium [22, 23, 27, 30, 33], combined vitamin C and E [25, 28, 34, 35, 37, 39, 40], vitamin D [24, 29, 32], folic acid [36, 41] and magnesium [26]. Two studies evaluated the effect of MMS which included multimineral-vitamin D supplements (calcium, magnesium, zinc and vitamin D) compared to vitamins C and E [38] and phytonutrient supplementation [31].
Seventeen studies compared the micronutrient supplementation intervention to a placebo, whilst one study compared the intervention to standard clinic protocol [28]. One study compared the effects of different dosages of micronutrient supplementation [36] another study made comparisons between MMS and vitamin C and E supplementation [38].
Calcium supplementation
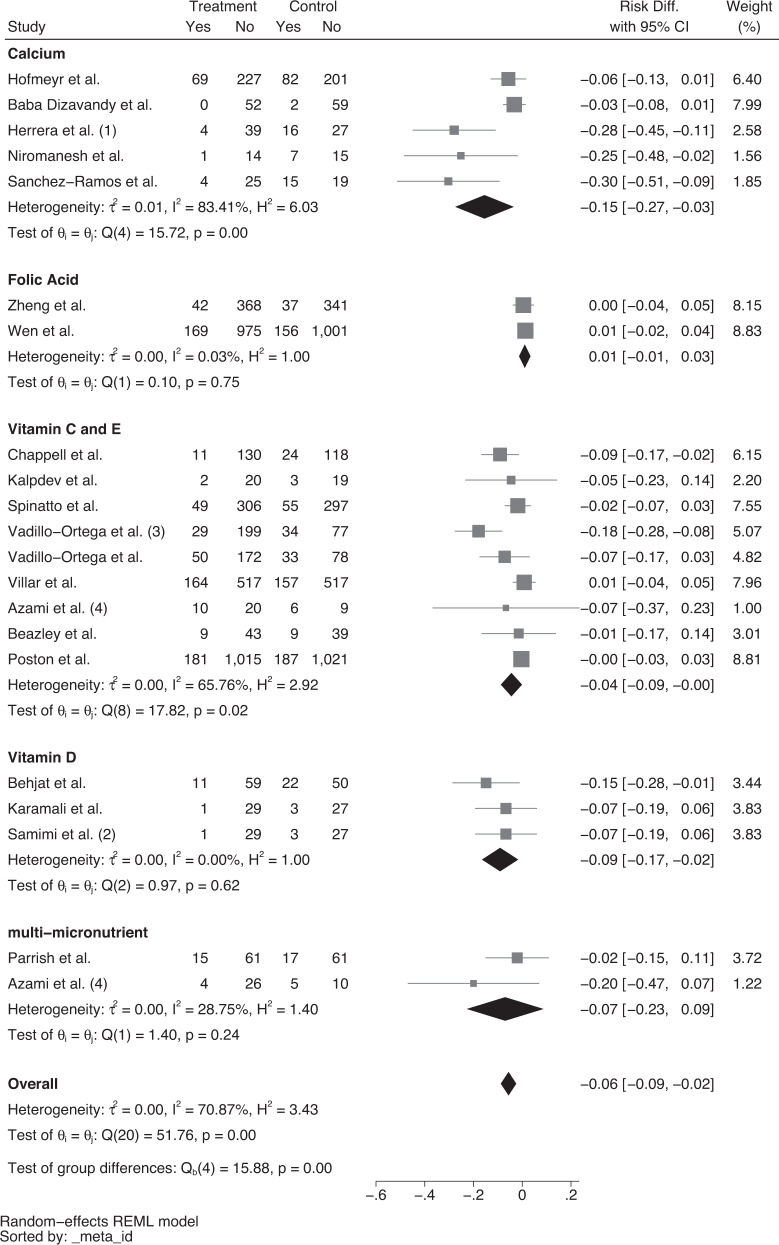
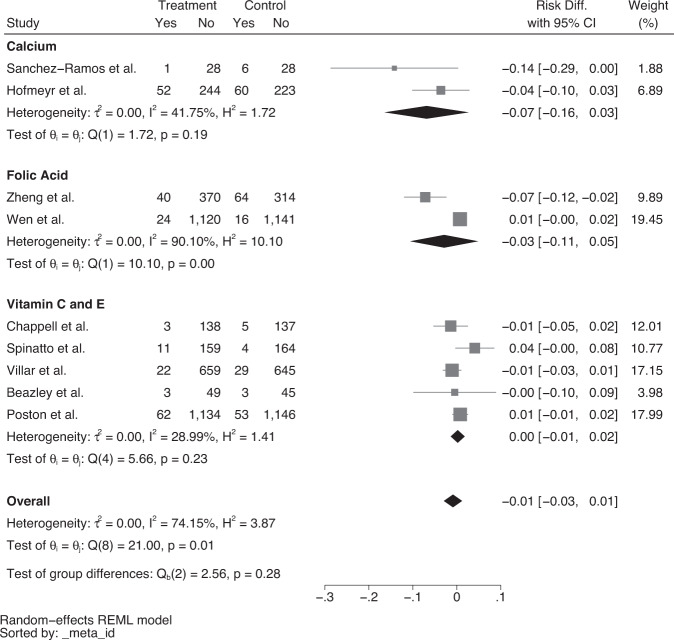
Three calcium interventions used a high dose between 1500 to 2000 mg daily [23, 30, 33]. Two studies [22, 27] used a lower dose of 500 and 600 mg calcium, respectively. Hofmeyr et al. started 500 mg of calcium supplementation before pregnancy until 20 weeks’ gestation whilst Herrera et al. used a 600 mg calcium supplement with 50 mg of linoleic acid between 28 to 32 weeks’ gestation until delivery [22, 27]. Similarly, Babadizavandy et al. utilised 2000 mg of calcium supplementation between 28 to 32 weeks’ gestation until delivery [23]. Both Niromanesh et al. and Sanchez-Ramos et al. also used 2000 mg of daily calcium supplementation. However, the period of the intervention was unclear [30, 33]. Of the 2 trials that reported data on compliance, Sanchez-Ramos et al. had a compliance rate of 91.1% while Hofmeyr et al. trial reported that approximately 50% of participants took at least 80% of their expected tablets. Random effects meta-analysis of five studies showed a lower rate of pre-eclampsia with calcium supplementation (risk difference = −0.15, 95% CI = −0.27 to −0.03), with significant heterogeneity among the studies (I2 = 83.41%, Fig. 2), however, there was no reduction in severe pre-eclampsia (Fig. 3).
Fig. 2. Forest plot of risk difference (95% CI) for overall pre-eclampsia, stratified by supplementation.
Forest plot assessing risk of overall pre-eclampsia with calcium, folic acid, vitamin C and E, vitamin D or multi-micronutrient supplementation. Key: (1) includes linoleic acid (2) includes calcium (3) includes L-arginine (4) all participants given ferrous sulfate tablets.
Fig. 3. Forest plot of risk difference (95% CI) for severe pre-eclampsia only, stratified by supplementation.
Forest plot assessing risk of severe pre-eclampsia with calcium, folic acid or vitamin C and E supplementation.
Vitamin D supplementation interventions
All three studies used a dose of 50,000 IU of vitamin D every 2 weeks, with 2 using this intervention in isolation [24, 29] and the third in combination with 1000 mg of calcium [32], with a reported compliance rate of 90–100% across all three trials. All 3 studies were conducted in the pregnancy period until 32 to 36 weeks’ gestation. There was a lower risk of pre-eclampsia with vitamin D supplementation in comparison to no supplementation (risk difference = −0.09, 95% CI = −0.17 to −0.02, I2 = 0.00%, Fig. 2), however there was no effect on severe pre-eclampsia (Fig. 3).
Vitamin C and E supplementation interventions
All 7 studies [25, 28, 34, 35, 37, 39, 40] used vitamin C and E together in combination with 6 studies using a dose of 1000 mg vitamin C and 400 IU vitamin E [25, 28, 34, 37, 39, 40]. The remaining study used a dose of 500 mg vitamin C and 400 IU vitamin E with or without L-arginine [35]. The majority of studies started the intervention in the second trimester of pregnancy, ranging from 14 to 24 weeks’ gestation, whilst Spinnato et al. initiated the intervention at the end of the first trimester [34]. Five trials reported a moderate to high level of compliance [25, 34, 35, 37, 40].
Random effects meta-analysis showed that the risk difference for pre-eclampsia was 4% for vitamin C and E, (risk difference = −0.04, 95% CI = −0.09 to 0.00) however, there was significant heterogeneity between the studies (I2 = 65.76%, Fig. 2). There was no effect on the rate of severe pre-eclampsia. Poston et al. reported no significant effect of vitamin C and E supplementation in the prevention of pre-eclampsia, however, infants born with a low birthweight were higher in the intervention group (I = 387 vs. C = 335, p = 0.023, RR = 1.15, 95% CI = 1.02–1.30).
Folic acid supplementation interventions
One study evaluated the effect of high dose folic acid (4 mg daily) compared to low dose folic acid (0.4 mg daily) from before pregnancy until delivery [36], whilst one study assessed the effect of the daily supplementation of 4 mg of folic acid from 8 to 16 weeks’ gestation until delivery [41]. Zheng et al. assessed homocysteine plasma levels and reported that levels were significantly lower in the high dose group which had a compliance rate of 74.1% compared to 73% in the low dose group. Among the high dose group, the incidence of pre-eclampsia was reduced when compliance was greater than 80% compared to 50%. Overall, there was no effect of folic acid supplementation on pre-eclampsia (Fig. 2) or severe pre-eclampsia (Fig. 3).
Magnesium supplementation interventions
De Araújo et al. assessed the effect of 300 mg of daily magnesium supplementation compared to a placebo from 12 to 20 weeks’ gestation until delivery and showed no significant impact on pre-eclampsia or severe pre-eclampsia [26]. Although adherence was defined in this trial, compliance rates were not reported.
MMS interventions
The two studies [31, 38] that used multiple micronutrients differed according to composition. Azami et al. compared three groups of pregnant women who received a daily ferrous sulphate tablet with a multimineral-vitamin D tablet consisting of 800 mg calcium, 9 mg zinc and 400 IU vitamin D (Group A), a daily ferrous sulphate tablet with vitamin C and vitamin E (Group B) or a daily ferrous sulphate tablet alone (Group C) [38], while Parrish et al. assessed the effect of a combination of phytonutrients including 7.5 mg of beta-carotene, 234 mg of Vitamin C, 30 mg of vitamin E, 420 mg of folate and 60 mg of calcium, taken twice daily until delivery, compared to placebo [31]. There were no data reported on compliance.
Pooled estimate of the 2 studies showed that MMS was not associated with a reduction in overall pre-eclampsia (Fig. 2). Parrish et al. also showed no reduction in the rate of severe pre-eclampsia [31].
Diagnostic criteria for pre-eclampsia
The studies diagnosed and classified pre-eclampsia using primarily SBP and DBP, urinary protein measurements and other clinical indicators such as liver enzymes and platelet count (Table 5).
Table 5.
Diagnostic criteria and outcomes of included studies.
| Reference | Diagnostic criteria for pre-eclampsia | Pre-eclampsia | Classifications of pre-eclampsia | Other outcomes |
|---|---|---|---|---|
| Hofmeyr et al. |
Gestational hypertension (DBP > 90 mmHg on two occasions 4 h apart, or >110 mmHg once, or SBP > 140 mmHg on two occasions 4 h apart, or >160 mmHg once after 20 weeks’ gestation) and gestational proteinuria (2 r more on urine dipstick, or >300 mg/24 h or 500 mg/L or urinary protein:creatinine ratio >0.034 g/mmol after 20 weeks’ gestation) as diagnosed by the attending clinicians. Severe pre-eclampsia (proteinuria + severe DBP [>110 mmHg] or systolic [>160 mmHg] hypertension) |
I = 69/296 (23%) vs C = 82/283 (29%), [RR = 0·80 (95% CI 0·61–1·06)], p = 0.121 | I = 52/296 (18%) vs C = 60/283 (21%), [RR = 0.83 (95% CI 0.59–1.16)], p = 0.268 | Not significant |
| Baba Dizavandy et al. | Having both gestational hypertension (SBP increase ≥30 mmHg and DBP increase ≥15 mmHg on two occasions at least 6 h apart, or SBP ≥ 140 mmHg and DBP ≥ 90 mmHg after 24 weeks’ gestation in absence of proteinuria) and proteinuria (>0.3 g/L urine on at least two separate random urine specimens more than 6 h apart after 24 weeks’ gestation) | I = 0% vs C = 3.3% | Not reported |
Gestational hypertension: I = 11.4% vs C – 31.2%, p < 0.01 Pregnancy-induced hypertension: I = 11.4% vs C = 35.6%, p < 0.01 Serum calcium levels vs. SBP in C group: r = −0.28, p = 0.02 |
| Herrera et al. | Development of hypertension from 20 weeks’ gestation (≥140/90 mmHg where there is an increase of ≥20 mmHg in DBP compared to previous levels during pregnancy on at least two occasions 6 or more hours apart) and significant 24-hour proteinuria (>0.3 g/L) in the absence of a urinary tract infection | I = 4 (9.3%) vs C = 16 (37.2%), [RR = 0.25], p = 0.002 | Not reported |
DBP: I = 74.6 ± 11.2 mmHg vs C = 81.9 ± 11.9 mmHg, p = 0.001 Gestational age at birth: I = 39.3 ± 1.4 weeks vs C = 38.2 ± 2.3 weeks, p = 0.03 Caesarean: I = 10 (23.3%) vs C = 19 (44.2%), p = 0.04 Birth weight: I = 3180 ± 340 g vs C = 3056 ± 475 g, p = 0.03 PGE2 levels after 30 days: I = + 106% vs C = −33%, p = 0.02 |
| Niromanesh et al. | SBP of ≥140 mmHg (an increase of 30 mmHg) and DBP ≥ 90 mmHg (an increase of 15 mmHg) on two occasions at an interval of 4–6 h with proteinuria (1 + proteinuria on random sampling of urine, measured as sulfosalicylic acid) | I = 1/15 vs C = 7/15, p = 0.014 | Not reported |
Time of onset of pre-eclampsia: I = 37 weeks vs C = 34 weeks, p < 0.05 Time of onset of hypertension: I = 37 ± 2 weeks vs C = 34 ± 1.9 weeks, p < 0.040 Duration of pregnancy: I = 39.5 ± 0.8 weeks vs C = 37.7 ± 2.5 weeks, p < 0.05 Infant birth weight: I = 3316 ± 308 g vs C = 2764 ± 761 g, p < 0.05 |
| Sanchez-Ramos et al. | BP ≥ 140/90 mmHg measured twice at 4–6 h apart and significant proteinuria (≥1+ on dipstick or at least 300 mg/2 h) | I = 13.8% vs C = 44.1%, p = 0.01, [RR = 0.37, (95% CI 0.15–0.92)] |
Mild pre-eclampsia: I = 3/29 vs C = 9/34 Severe pre-eclampsia: I = 1/29 vs C = 6/34 |
Pregnancy-induced hypertension: I = 31.0% vs C = 64.7%, p = 0.01 [RR = 0.46, 95% CI 0.25–0.86] |
| Behjat et al. | BP ≥ 140/90 mmHg in sitting position with proteinuria of ≥1+ on urine dipstick |
I = 11 (15.7%) vs C = 22 (30.6%), p = 0.036 Risk in the control group was 1.94 times higher (95% CI 1.02–3.71) |
Not reported | Not reported |
| Karamali et al. | Not reported | I = 3.3% vs C = 10.0%, p = 0.30 | Not reported |
Serum vitamin D levels: I = 17.92 ± 2.88 ng/ml vs C = 0.27 ± 3.19 ng/ml, p = 0.001 Increase in insulin levels: I = 1.08 ± 6.90 µIU/ml vs C = 9.57 ± 10.32 µIU/ml, p < 0.001) |
| Samimi et al. | Not reported | I = 3.3% C = 10.0%, p = 0.30 | Not reported |
Change in DBP: I = −2.0 mmHg, SD 6.6 mmHg vs C = + 3.7 mmHg, SD 6.3 mmHg, p = 0.001 Change in SBP: I = −3.8 mmHg, SD 5.8 mmHg vs C = + 1.7 mmHg, SD = 8.7 mmHg, p = 0.006 Mean serum 25(OH) D concentration: I = 8.2 ng/mL, SD = 7.7 ng/mL vs C = + 0.1 ng/mL, SD = 3.2 ng/mL, p < 0.001 Change in FPG: I = −5.7 mg/dL, SD = 5.5 mg/dL vs C = −0.6 mg/dL, SD = 12.6 mg/dL, p = 0.04 Change in insulin: I = −0.28 µIU/ml, SD = 6.0 µIU/ml vs C = + 7.7 µIU/ml, SD = 9.0 µIU/ml, p < 0.001 Change in HOMA-IR: I = −0.8, SD = 1.3 vs C = + 1.6, SD = 2.2, p < 0.001 Change in HOMA-B: I = −8.2, SD = 25.8 vs C = + 32.6, SD = 41.3, p < 0.001 Change in QUICKI score: I = + 0.02, SD = 0.02 vs C = −0.02, SD – 0.02, p < 0.001 Change in Serum HDL cholesterol: I = + 4.6 mg/dL, SD = 8.3 mg/dL vs C = −2.9 mg/dL, SD = 7.7 mg/dL, p = 0.001 Change in plasma GSH concentration: I = + 23.4 µm, SD = 124.0 µm vs. C = −94.8 µm, SD = 130.2 µm, p = 0.001 |
| Chappell et al. |
Two recordings of DBP ≥ 90 mmHg at least 4 h apart (for severe pre-eclampsia, two recordings of DBP ≥ 110 mmHg at least 4 h apart or one reading of 120 mmHg) and proteinuria (excretion of 300 mg or more in 24 h or two readings of ≥2+ on midstream urine dipstick or catheter urine if 24-hour collection is not available) Superimposed pre-eclampsia was defined as the new development of proteinuria in women with chronic hypertension |
ITT I = 11/141 (8%) vs C = 24/142 (17%) [Adj OR = 0.39 (95% CI 0.17–0.90)], p = 0.02 |
Mild pre-eclampsia: ITT I = 5/141 vs C = 13/142 Severe pre-eclampsia: ITT I = 3/141 vs C = 5/142 Superimposed pre-eclampsia: ITT I = 3/141 vs C = 6/142 |
PAI-1:PAI-2 ratio: 21% reduction in I (85% CI 4–35), p 0.015 |
| Poston et al. | Gestational hypertension (two or more readings of DBP ≥ 90 mmHg taken at least 4 h and up to 168 h apart, occurring after 20 weeks’ gestation or up to 48 h in the early postnatal period, excluding labour) or severe gestational hypertension (same as above however with a DBP ≥ 110 mmHg on two or more occasions or a single reading of ≥120 mmHg), and proteinuria (excretion of 300 mg/24 h protein or two readings of ≥2+ on urine dipstick analysis) or severe proteinuria (excretion of ≥5000 mg/24 h) | I = 181 (15%) vs C = 187 (16%) [RR = 0·97, 95% CI 0·80–1·17], p = 0.754 | Severe pre-eclampsia: I = 62 (5%) vs C = 53 (4%) [RR = 1.17, 95% CI 0.82–1.68] |
Gestational hypertension: I = 84 (7%) vs. C = 55 (5%), [RR = 1.53, 95% CI 1.10–2.13] Low birth weight: I = 387 (28%) vs C = 335 (24%), p = 0.023 [RR = 1.15, 95% CI 1.02–1.30] Low birth weight in women with diabetes: I = 20% {n = 19) vs C = 10% (n = 6), risk ratio = 3.26 95% CI 1.36–7.84 |
| Spinatto et al. |
SBP ≥ 140 mmHg and DBP ≥ 90 mmHg or proteinuria (either 300 mg/24 hours or ≥2+ by dipstick on two or more occasions 4 h apart) Severe pre-eclampsia defined as severe hypertension (if ≥ 2 SBP values obtained 4 or more hours apart were 160 mmHg or if ≥2 DBP values were 110 mmHg) and proteinuria; urinary protein excretion 5 g/day with any degree of hypertension; hypertension complicated by pulmonary oedema or a low platelet count (<100,000/mL); or haemolysis, an elevated serum aspartate aminotransferase concentration (>70 units/L), and a low platelet count (HELLP syndrome). Superimposed pre-eclampsia was defined as hypertension + proteinuria (either 300 mg/24 h or ≥2+ by dipstick) in chronically hypertensive patients |
ITT I = 49/355 (13.8%) vs C = 55/352 (15.6%), [Adj RR = 0.87, (95.42% CI 0.61–1.25)], p = 0.43 | Severe pre-eclampsia in those without chronic hypertension: I = 11/170 (6.5%) vs C = 4/168 (2.4%), p = 0.11, [OR = 2.78, 95% CI 0.79–12.62] | Premature rupture of membranes: I = 10.6% vs C = 5.5%, p = 0.015 [RR = 1.89, 95% CI 1.11–3.23] |
| Kalpdev et al. | Superimposed pre-eclampsia was defined as the new onset proteinuria of ≥300 mg/24 h in hypertensive women with no proteinuria before 20 weeks’ gestation or a sudden increase in proteinuria or BP or platelet count of <100,000 mm3 in women with hypertension and proteinuria before 20 weeks’ gestation | N/A | Superimposed pre-eclampsia: I = 8% vs C = 12%, p = 1.000 | FRAP levels: I = 1168.95 ± 191.32 µM vs C = 835.93 ± 162.35 µM, p = 0.022 |
| Beazley et al. | Not reported | I = 17.3% vs C = 18.8% [RR = 0.92, 95% CI 0.4–2.13] |
Severe pre-eclampsia: I = 3/52 vs 3/48 Total cases of mild pre-eclampsia: 5 Total cases of superimposed pre-eclampsia: 7 |
Not significant |
| Vadillo-Ortega et al. |
Hypertension (SBP ≥ 140 mmHg, DBP ≥ 90 mmHg, or both) and proteinuria (>300 mg/24 h) after 20 weeks’ gestation in women known to be previously normotensive Severe pre-eclampsia defined as proteinuria >2.0 g/24 h, BP ≥ 160/110 mmHg or both |
Incidence of pre-eclampsia and eclampsia together: L-arginine + antioxidant vitamins (I) vs placebo (C) = lower incidence in I [χ2 = 19.41, p < 0.001, ARR 0.17, 0.12–0.21] No differences between antioxidants (I2) group vs placebo (C), p = 0.052 |
Not reported |
Preterm delivery: I = ARR = 0.53 [95% CI 0.33–0.84, χ2: p = 0.003], I2 = ARR = 0.44 [95% CI 0.28–0.70, χ2: p < 0.001] Side-effects: Significantly more side-effects in I than C: nausea (p = 0.019), symptoms of dyspepsia (p = 0.04), dizziness (p = 0.039), palpitations (p = 0.019) and headache (p = 0.09) 1-minute Apgar score: I vs C ARR = 0.44, 95% CI% 0.24–0.64, p = 0.000 I2 vs C ARR = 0.25, 95% CI = 0.04–0.47, p = 0.015 I vs I2 ARR = 0.18, 95% CI 0.01 = 0.36, p = 0.04 5-minute Apgar score: I vs C ARR = 0.21, 95% CI 0.10–0.33, p = 0.000 |
| Villar et al. |
De novo hypertension (≥2 readings of DBP ≥ 90 mmHg, taken 4 h apart or more, but <160 h apart, and occurring after 20 weeks’ gestation) and new-onset proteinuria (excretion of ≥300 mg/24 h or two readings of >1+ on dipstick if 24-h collection is not available) Severe pre-eclampsia defined as SBP ≥ 160 mmHg and/or DBP ≥ 110 mmHg on two occasions, at least 4 h but not more than 168 h apart, or if the first measurement was immediately followed by treatment with an antihypertensive, either of these scenarios being associated with proteinuria. |
I = 24.1% vs C = 23.3% [RR = 1.0, 95% CI 0.9–1.3] | Severe pre-eclampsia: I = 3.2% vs C = 4.3% [RR = 0.8, 95% CI 0.4–1.3] | Not significant |
| Zheng et al. |
BP ≥ 140/90 mmHg and proteinuria Severe pre-eclampsia diagnostic criteria not reported |
Overall pre-eclampsia: Low dose group = 37/378 (9.8%) vs High dose group = 42/410 (10.2%) [RR = 0.96, 95% CI 0.76–1.19)], p = 0.684 | Severe pre-eclampsia: Low dose group = 64/378 (16.9%) vs High dose group = 40/410 (9.8%) [RR = 1.69. 95% CI 0.55–4.80], p = 0.011 |
Severe gestational hypertension: Low dose group = Low dose group = 126/378 (33.3%) vs high dose group = 89/410 (21.7%), p = 0.021 [RR = 1.54, 95% CI = 0.67–3.70] Early onset of pre-eclampsia (<32 weeks): Low dose group = 73/378 (19.3%) vs high dose group = 52/4410 (12.7%), p = 0.039 [RR = 1.52, 95% CI 0.53–4.20] Apgar score <7 at 5 min: Low dose group = 5/378 (1.3%) vs high dose group = 3/410 (0.7%), p = 0.013 [RR = 1.85, 95% CI 0.44–4.32] |
| Wen et al. |
DBP ≥ 90 mmHg on two occasions 4 or more hours apart and proteinuria (≥2+ on dipstick, or urinary protein ≥300 mg/24 h, or random protein:creatinine ratio ≥30 mg protein/mmol) in women at ≥20 weeks’ gestation, or diagnosis of HELLP syndrome or superimposed pre-eclampsia (history of pre-existing hypertension diagnosed before pregnancy or before 20 weeks’ gestation with new proteinuria) Severe pre-eclampsia diagnostic criteria not reported |
I = 169/1144 (14.8%) vs C = 156/1157 (13.5%) [RR = 1.10, 95% CI 0.90–1.34] p = 0.37 | Severe pre-eclampsia: I = 21/1144 (1.84%) vs C = 16/1156 (1.38%) [RR = 1.52, 95% CI 0.81–2.84], p = 0.19 | Not significant |
| De Araujo et al. | Elevated SBP > 140 mmHg or DBP > 90 mmHg, with ≥2+ proteinuria on urine dipstick, and/or HELLP syndrome | I = 24 (5.9%) vs C = 20 (4.7%), [unadjusted RR = 1.24 (0.70 to 2.22)], [Adj OR = 1.25 (0.68 to 2.31)] | Not reported | Placental abruption: I = 9 (2.2%) vs C = 21 (5.0%), unadjusted RR = 0.44 (0.21–0.96), Adj OR = 0.43 (0.20–0.95) |
| Azami et al. | BP ≥ 140/90 after 20 weeks’ gestation and proteinuria ≥300 mg/24 hours or 1+ on urine dipstick |
Group A (I) = 13.3% vs Group C (C) = 36.7%, p = 0.03 Group B (I) = 33.3% vs Group C (C) = 36.7%, p = 0.50 Group A vs Group B, p = 0.063 |
Not reported | Neonatal complications (unspecified): Lower in Group A (p = 0.01) |
| Parrish et al. | Not reported |
High and low risk group: I = 15.9% vs C = 16.3%, p = 0.93 Low risk group: I = 10.7% vs C = 8.8%, p = 0.73, [RR = 1.22, 95% CI 0.40–3.77] High risk group: I = 19.7% vs C = 21.9%, p = 0.75, [RR = 0.91, 95% CI 0.49–1.68] |
Mild pre-eclampsia: Low risk group I = 8.9% vs C = 8.8%, p = 0.98 [RR = 1.02, 95% CI 0.31–3.32]; high risk group I = 2.6% vs C = 1.3%, p = 0.99 [RR = 1.03, 95% CI 0.07–16.1] Severe pre-eclampsia: Low risk group I = 0% vs C = 0%; high risk group I = 5.3% vs C = 3.9%, p = 0.67 [RR = 1.37, 95% CI 0.32–5.91] Superimposed pre-eclampsia: Low risk group I = 0% vs C = 0%; high risk group I = 11.8% vs C = 16.7%, p = 0.40 [RR = 0.71, 95% CI 0.32–1.56] |
Not significant |
RR relative risk, OR odds ratio, ARR absolute risk reduction, I intervention, C control, ITT intention to treat, Adj adjusted, SD standard deviation, BP blood pressure, DBP diastolic blood pressure, SBP systolic blood pressure’
Quality of included studies
The quality of the studies included in this review is shown in Supplementary Information 2. In total, 12 trials [22, 24, 25, 27, 29, 32–35, 37, 40, 41] were assessed as ‘low risk of bias,’ seven trials [23, 28, 30, 31, 36, 38, 39] as “high risk of bias” and one [26] was classified as “some concerns.” The main source of bias across the studies was either lack of information available on compliance and adherence in the study or non-adherence to the micronutrient supplementation intervention.
Discussion
This review aimed to evaluate the effect of micronutrient supplementation interventions on the development of pre-eclampsia in women identified as high risk. Our findings showed a lower rate of pre-eclampsia with calcium and vitamin D supplementation. There was no effect of micronutrient supplementation on severe pre-eclampsia. The review was limited by studies not adequately powered to detect a difference in pre-eclampsia and heterogeneity between studies was high.
Calcium supplementation has been previously reported to reduce the risk of pre-eclampsia [7] with the greatest effects observed in women with low calcium diets, however doses of calcium varied significantly across the trials, ranging from 500 mg to 2000 mg. The mechanism is largely unknown; however it is suggested that calcium lowers blood pressure [42] and may also reduce activation of the vascular endothelium [43]. Our findings support the use of calcium, in women with low dietary intake, to reduce pre-eclampsia in high-risk women, however, the studies had a small sample size and were under powered for the outcome [22]. The variance amongst these trials, ranging from different diagnostic criteria to the differences in sample size, highlights the need for more well-powered and larger-scale trials to establish when and how calcium supplementation can be the most beneficial to women identified as high risk of developing pre-eclampsia.
The vitamin D supplementation trials showed a slight reduction in pre-eclampsia, however, from studies with a small sample size. Previous reviews have reported on the lack of consistent evidence of benefit of vitamin D supplementation and its role in the prevention of pre-eclampsia, adding that inconsistencies in reporting the timing and duration of the intervention have not yet been adequately addressed [44]. Our findings in high-risk women support this, with some studies lacking clarity in reporting the intervention components [24] and time of gestation during the intervention, highlighting the need for further studies in higher risk women and better reporting in clinical trials.
There was a slightly lower rate of pre-eclampsia with antioxidants (vitamin C and E), however the confidence interval included zero and adverse outcomes such as low birthweight [40] were higher in the intervention arm. Previous reviews have reported no benefit of vitamin C and E supplementation on pre-eclampsia in ‘all-risk’ women, however there might be a protective effect in low- and middle-income countries [8]. This review provides some evidence that vitamin C and E supplementation may reduce pre-eclampsia in higher-risk women by 4%, including in low- and middle-income countries [35], however secondary outcomes were unfavourable, including a higher instance of SGA infants in the intervention arm. Oxidative stress is known to play a pivotal role in the manifestation of conditions such as pre-eclampsia [45], and alternative methods of reversing oxidative stress or poor antioxidant status may be worth investigating further in higher risk women.
This review found no evidence of benefit of folic acid supplementation on the development of pre-eclampsia. A recent 2018 review of observational studies [46] reported that folic acid was associated with lower risk of developing pre-eclampsia in pregnancy, perhaps being most effective in combination with multivitamins. However, RCTs evaluating the effect of folic acid on the development of pre-eclampsia are scarce, with conflicting data about the optimal dosages and whether it is best in isolation or combination with other micronutrients [46] and suggests a need for further research in women identified as high risk. Appropriately designed RCTs which encompass both dosage comparison of folic acid and stratify participants by high vs low risk are needed to elucidate the role of folic acid in the prevention of pre-eclampsia.
A limited number of studies addressed the impact of magnesium. Additionally, few studies assessed effects of MMS supplements with considerable variation in the combination of micronutrients used in each trial. A 2019 review reported improvements in preterm birth, low birth weight and SGA when supplemented with MMS together with iron and folic acid [47], however the effect of MMS on pre-eclampsia is still unclear. Higher-quality studies are needed to evaluate the potential benefits of MMS on the prevention of pre-eclampsia with a focus on finding the most effective combination of micronutrients. We did not retrieve any studies assessing the efficacy of zinc in this review.
Specific classifications of pre-eclampsia were reported in 11 studies [22, 25, 28, 31, 33, 34, 36, 37, 39–41], including mild, severe and superimposed pre-eclampsia. There was no effect of micronutrient supplementation interventions on severe pre-eclampsia. Tailoring micronutrients and their dosages to predicted severity of pre-eclampsia may be more beneficial than more generalised preventative approaches, particularly in this stratified population as opposed to all-risk women. With a lack of reviews evaluating the effect of micronutrient supplementation interventions on different classifications of pre-eclampsia, future studies on pre-eclampsia and its various severities, along with more exploration of the associated predictive factors, could inform primary prevention strategies to improve maternal healthcare.
The studies in this review used a wide variety of predictive factors at study entry. Of the eight studies [24, 25, 27, 30, 33, 35, 36, 38] with positive findings, five [24, 25, 35, 36, 38] used a history of pre-eclampsia to identify high-risk women whilst two studies used a positive roll-over test in addition to another predictive factor [27, 30], with the remaining study using a positive angiotensin sensitivity test [33]. A history of pre-eclampsia was the most common factor used to select participants at study entry, with very few studies using a combination of more than one risk factor. Predictive factors have shown to be most effective through a combination of using maternal characteristics, biomarkers and other variables such as the uterine artery doppler [17]. For example, a new first-trimester screening algorithm combining MAP, uterine artery doppler and circulating levels of placental growth factor (PIGF) to predict preterm pre-eclampsia, has a true positive rate of 82%, almost double the rate of detection via the UK National Institute for Health and Care Excellence (NICE) guidelines which uses clinical factors alone [48]. Many trials in this review may have benefited from using prediction models, which utilise a combination of specific variables and may offer higher predictive ability [48]. With no study in this review using a validated prediction model to identify high-risk women and a lack of externally validated prediction algorithms, future studies assessing the effect of micronutrient supplementation interventions in women at high risk of developing pre-eclampsia could use validated prediction models to select participants, with more trials also exploring alternative combinations of predictive factors that accurately determine women who are at high risk of developing pre-eclampsia.
The trials included in this review also varied in other aspects of study design and reporting of findings. In addition to differences in the identification of high-risk women, there was variability in the eligibility criteria, with one trial including women with other co-morbidities, for example, kidney disease [38]. Furthermore, some trials reported the diagnostic criteria of severe pre-eclampsia, but several did not [31, 33, 36, 39, 41] while other trials did not report a diagnostic criterion for any classification of pre-eclampsia [29, 31, 32, 39]. There was a lack of reporting on statistical methodology, in particular the inclusion of confounding variables in the analysis. Several trials did not clearly state whether confounding variables had been adjusted for. In those that did, the most common confounding factors adjusted for were maternal age and BMI. Compliance across the trials ranged from moderate to high, with the majority of studies using tablet count while Zheng et al. used plasma homocysteine levels as a method of demonstrating the potential confounding effect of compliance on the outcome of pre-eclampsia. Data for compliance was not reported in several trials [23, 26–28, 30, 31, 38, 39] while there was also variation across trials in the frequency, dosing and timing of supplementation, while the gestational age at the time of intervention was often not reported.
Two trials [18, 32] commenced in the preconception period. Calcium is known to have a benefit in reducing the risk of pre-eclampsia from 20 weeks’ gestation onwards, however our findings suggest that calcium supplementation before this point may not necessarily be effective as is shown by Hofmeyr et al. [22]. On the other hand, Zheng et al. reported that supplementation of folic acid from preconception to delivery may effectively reduce risk of severe pre-eclampsia [36]. Micronutrient supplementation interventions may also be effective for different durations and at different time points, which could be investigated in future trials to further elucidate the effect of these micronutrients. However, caution is imperative as our findings show that some micronutrient interventions, notably vitamin C and E, are not entirely benign.
Strengths and limitations
This systematic review has several strengths. This study addressed whether micronutrient interventions are effective in reducing the development of pre-eclampsia in women identified as high risk for the condition. Previous reviews have focused on an unselected approach, often with no risk stratification. The identification of women with an increased risk of developing pre-eclampsia might enable targeted intervention in women most likely to benefit. This work complements previous findings that calcium and vitamin D are beneficial in reducing pre-eclampsia including in those identified as high risk, whilst highlighting the need for more large-scale well-powered studies with improved and more consistent reporting of interventions and findings. A comprehensive search strategy was used to screen for studies that targeted interventions at higher risk populations by using a pre-specified eligibility criteria. The findings from this review are important to inform the design of future RCTs to improve the data quality and clarify the effects of micronutrient supplementation, particularly in women at high risk for pre-eclampsia and the effect on different classifications of pre-eclampsia.
This review has limitations. It only included studies published in English which may have introduced publication bias. The high risk of bias in several studies in this review highlights the low-quality evidence in this research area and supports the need for more robust future trials. Additionally, many trials with significant results were not adequately powered to detect a difference in pre-eclampsia between treatment groups or may have overestimated the effect of the given intervention as a result of a small sample size. The analysis was limited in scope as pooling of data were not possible for all micronutrient interventions, in addition to a lack of adjustment for potential confounders. Furthermore, there was high methodological variability between studies particularly with calcium, vitamin D and vitamin C and E trials, limiting the consistency of data across these studies. One reason for this may be because of differences in the interventions themselves, as Herrera et al. did not investigate calcium alone but in combination with linoleic acid, whilst Samimi et al. investigated vitamin D given with calcium. Additionally, the gestational age at which interventions were administered varied significantly amongst trials. Finally, the risk factors used to screen for women at high risk of pre-eclampsia differed significantly between trials.
Recommendations for further research and practice
Our review highlighted the lack of studies of interventions targeting higher risk women in the preconception period. Only two studies [22, 36] in this review initiated the intervention in the preconception period. With symptoms of pre-eclampsia beginning from 20 weeks’ gestation, preconception and early pregnancy interventions require further exploration as they may provide greater insight into how to improve maternal and neonatal outcomes. We have reported a lower rate of pre-eclampsia with calcium and vitamin D, however, conclusions were limited by small sample sizes, methodological variability and heterogeneity between studies. Future studies of these two micronutrients are warranted, however they must be larger-scale and well-powered to allow more thorough and reliable conclusions to be drawn. Additionally, several studies [24, 25, 30, 38, 39] in this review did not clearly state the gestation of the participants at the beginning and end of the given micronutrient supplementation intervention, as well as failing to report on adherence and compliance. Better reporting of trials is required in future studies to ascertain the relationship between the effectiveness of the given micronutrient supplementation intervention and the timing with which it is administered. Further studies using externally validated prediction models that have demonstrated higher predictive performance such as models by Poon et al. [49] and Odibo et al. [50] with a detection rate of 91.7% and 80.0% respectively for pre-eclampsia requiring early delivery and use a variety of predictive factors such as chronic hypertension and PAPP-A may additionally clarify the effects of these micronutrients. Finally, future research is needed to determine the effect of micronutrient supplementation interventions on different classifications of pre-eclampsia, from mild to superimposed pre-eclampsia, to progress towards a more personalised and tailored approach in the primary prevention of pre-eclampsia.
Conclusion
This study showed a small effect of calcium and vitamin D in the prevention of pre-eclampsia in women who were identified as higher risk of developing the condition. The review was limited by the inclusion of studies with a small sample size. Significant heterogeneity between studies as well as methodological variability was evident. Further higher quality, large-scale RCTs of calcium and vitamin D, the use of prediction modelling, and particularly at different points of time before and during pregnancy, may be beneficial to assess the efficacy of micronutrient supplementation intervention in the prevention of pre-eclampsia in high-risk women.
Supplementary information
Author contributions
The research question and study design were formulated by ACF. Titles, abstracts and full text articles were independently screened by SG and DDALS. OQ contributed to data extraction and provided feedback on the systematic review process. Data extraction and analysis was carried out by SG, DDALS and KVD. The writing of the article was completed by SG, DDALS, KVD, LP, LM, SLW, JRF and ACF. ACF had overall responsibility for the manuscript.
Funding
ACF and LP are supported by Tommy’s charity. KVD is supported by the Medical Research Council (MR/V005839/1).
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Kathryn V. Dalrymple, Angela C. Flynn.
Supplementary information
The online version contains supplementary material available at 10.1038/s41430-022-01232-0.
References
- 1.World Health Organization. WHO recommendations for Prevention and treatment of pre-eclampsia and eclampsia [Internet]. 2011 [cited 2021 Sep 2]. Available from: http://apps.who.int/iris/bitstream/handle/10665/44703/9789241548335_eng.pdf [PubMed]
- 2.NICE. Hypertension in pregnancy: diagnosis and management NICE guideline [Internet]. 2019. Available from: www.nice.org.uk/guidance/ng133
- 3.Mol BWJ, Roberts CT, Thangaratinam S, Magee LA, de Groot CJM, Hofmeyr GJ. Pre-eclampsia. Lancet. 2016;387:999–1011. doi: 10.1016/S0140-6736(15)00070-7. [DOI] [PubMed] [Google Scholar]
- 4.Rawlins B, Plotkin M, Rakotovao JP, Getachew A, Vaz M, Ricca J, et al. Screening and management of pre-eclampsia and eclampsia in antenatal and labor and delivery services: Findings from cross-sectional observation studies in six sub-Saharan African countries. BMC Pregnancy Childbirth. 2018;18:1–11. doi: 10.1186/s12884-018-1972-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Burton GJ, Redman CW, Roberts JM, Moffett A Pre-eclampsia: pathophysiology and clinical implications. Vol. 366, The BMJ. BMJ Publishing Group; 2019. [DOI] [PubMed]
- 6.Aouache R, Biquard L, Vaiman D, Miralles F. Oxidative stress in preeclampsia and placental diseases. Int J Mol Sci. 2018;19:1496. doi: 10.3390/ijms19051496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hofmeyr GJ, Lawrie TA, Atallah ÁN, Torloni MR. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst Rev. 2018;2018:1–123. doi: 10.1002/14651858.CD001059.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kinshella MLW, Omar S, Scherbinsky K, Vidler M, Magee LA, von Dadelszen P, et al. Effects of maternal nutritional supplements and dietary interventions on placental complications: an umbrella review, meta-analysis and evidence map. Nutrients. MDPI AG. 2021;13:1–29. doi: 10.3390/nu13020472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Makrides M, Crosby DD, Shepherd E, Crowther CA. Magnesium supplementation in pregnancy. Cochrane Database Syst Rev. 2014;2019:1–57. doi: 10.1002/14651858.CD000937.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wilson R, Grieger J, Bianco-Miotto T, Roberts C. Association between maternal zinc status, dietary zinc intake and pregnancy complications: a systematic review. Nutrients. 2016;8:1–28. doi: 10.3390/nu8100641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Oh C, Keats E, Bhutta Z. Vitamin and mineral supplementation during pregnancy on maternal, birth, child health and development outcomes in low- and middle-income countries: a systematic review and meta-analysis. Nutrients. 2020;12:1–30. doi: 10.3390/nu12020491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fu ZM, Ma ZZ, Liu GJ, Wang LL, Guo Y. Vitamins supplementation affects the onset of preeclampsia. J Formos Med Assoc. 2018;117:6–13. doi: 10.1016/j.jfma.2017.08.005. [DOI] [PubMed] [Google Scholar]
- 13.Macdonald-Wallis C, Silverwood RJ, de Stavola BL, Inskip H, Cooper C, Godfrey KM, et al. Antenatal blood pressure for prediction of pre-eclampsia, preterm birth, and small for gestational age babies: development and validation in two general population cohorts. BMJ. 2015;351:h5948–48. doi: 10.1136/bmj.h5948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kenny LC, Black MA, Poston L, Taylor R, Myers JE, Baker PN, et al. Early pregnancy prediction of preeclampsia in nulliparous women, combining clinical risk and biomarkers. Hypertension. 2014;64:644–52. doi: 10.1161/HYPERTENSIONAHA.114.03578. [DOI] [PubMed] [Google Scholar]
- 15.Kleinrouweler CE, Cheong-See FM, Collins GS, Kwee A, Thangaratinam S, Khan KS, et al. Prognostic models in obstetrics: available, but far from applicable. Am J Obstet Gynecol. 2016;214:79–90.e36. doi: 10.1016/j.ajog.2015.06.013. [DOI] [PubMed] [Google Scholar]
- 16.Onwudiwe N, Yu CKH, Poon LCY, Spiliopoulos I, Nicolaides KH. Prediction of pre-eclampsia by a combination of maternal history, uterine artery Doppler and mean arterial pressure. Ultrasound Obstet Gynecol. 2008;32:877–83. doi: 10.1002/uog.6124. [DOI] [PubMed] [Google Scholar]
- 17.Chappell LC, Cluver CA, Kingdom J, Tong S. Pre-eclampsia. The Lancet. Elsevier B.V. 2021;398:341–54. doi: 10.1016/S0140-6736(20)32335-7. [DOI] [PubMed] [Google Scholar]
- 18.Moher D, Liberati A, Tetzlaff J, Altman DG, Altman D, Antes G, et al. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009;6:1–6. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Duffy J, Cairns A, Richards‐Doran D, ’t Hooft J, Gale C, Brown M, et al. A core outcome set for pre‐eclampsia research: an international consensus development study. BJOG: Int J Obstet Gynaecol. 2020;127:1516–26. doi: 10.1111/1471-0528.16319. [DOI] [PubMed] [Google Scholar]
- 20.Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan—a web and mobile app for systematic reviews. Syst Rev. 2016;5:210. doi: 10.1186/s13643-016-0384-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. The BMJ. 2019;366:1–8. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 22.Hofmeyr GJ, Betrán AP, Singata-Madliki M, Cormick G, Munjanja SP, Fawcus S, et al. Prepregnancy and early pregnancy calcium supplementation among women at high risk of pre-eclampsia: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2019;393:330–9. doi: 10.1016/S0140-6736(18)31818-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Baba Dizavandy E, Seyyedi Alavi G, Cordi M. The effect of calcium supplementation in the prevention of hypertensive disorder of pregnancy in nulliparous women. Med J Islamic Repub Iran. 1998;12:11–4. [Google Scholar]
- 24.Behjat Sasan S, Zandvakili F, Soufizadeh N, Baybordi E. The effects of vitamin D supplement on prevention of recurrence of preeclampsia in pregnant women with a History of preeclampsia. Obstet Gynecol Intern. 2017;2017:1–5. doi: 10.1155/2017/8249264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chappell LC, Seed PT, Briley AL, Kelly FJ, Lee R, Hunt BJ, et al. Effect of antioxidants on the occurrence of pre-eclampsia in women at increased risk: a randomised trial. Lancet. 1999;354:810–6. doi: 10.1016/S0140-6736(99)80010-5. [DOI] [PubMed] [Google Scholar]
- 26.de Araújo CAL, Ray JG, Figueiroa JN, Alves JG. BRAzil magnesium (BRAMAG) trial: a double-masked randomized clinical trial of oral magnesium supplementation in pregnancy. BMC Pregnancy Childbirth. 2020;20:1–7. doi: 10.1186/s12884-020-02935-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Herrera MJA, Arevalo-Herrera M, Herrera S. Prevention of preeclampsia by linoleic acid and calcium supplementation: a randomized controlled trial. Obstet Gynecol. 1998;91:585–90. doi: 10.1016/s0029-7844(97)00711-4. [DOI] [PubMed] [Google Scholar]
- 28.Kalpdev A, Saha SC, Dhawan V. Vitamin C and E supplementation does not reduce the risk of superimposed PE in pregnancy. Hypertens Pregnancy. 2011;30:447–56. doi: 10.3109/10641955.2010.507840. [DOI] [PubMed] [Google Scholar]
- 29.Karamali M, Beihaghi E, Mohammadi A, Asemi Z. Effects of high-dose vitamin D supplementation on metabolic status and pregnancy outcomes in pregnant women at risk for pre-eclampsia. Hormone Metab Res. 2015;47:867–72. doi: 10.1055/s-0035-1548835. [DOI] [PubMed] [Google Scholar]
- 30.Niromanesh S, Laghaii S, Mosavi-Jarrahi A. Supplementary calcium in prevention of pre-eclampsia. Int J Gynecol Obstet. 2001;74:17–21. doi: 10.1016/S0020-7292(01)00374-5. [DOI] [PubMed] [Google Scholar]
- 31.Parrish MR, Martin JN, Lamarca BB, Ellis B, Parrish SA, Owens MY, et al. Randomized, placebo controlled, double blind trial evaluating early pregnancy phytonutrient supplementation in the prevention of preeclampsia. J Perinatol. 2013;33:593–9. doi: 10.1038/jp.2013.18. [DOI] [PubMed] [Google Scholar]
- 32.Samimi M, Kashi M, Foroozanfard F, Karamali M, Bahmani F, Asemi Z, et al. The effects of vitamin D plus calcium supplementation on metabolic profiles, biomarkers of inflammation, oxidative stress and pregnancy outcomes in pregnant women at risk for pre-eclampsia. J Hum Nutri Diet. 2016;29:505–15. doi: 10.1111/jhn.12339. [DOI] [PubMed] [Google Scholar]
- 33.Sanchez-Ramos L, Briones DK, Kaunitz AM, Delvalle GO, Gaudier FL, Walker CD. Prevention of pregnancy-induced hypertension by calcium supplementation in angiotensin II-sensitive patients. Obstet Gynecol [Internet]. 1994;84:349–53. [PubMed] [Google Scholar]
- 34.Spinnato JA, Freire S, Pinto E, Silva JL, Cunha Rudge MV, Martins-Costa S, Koch MA, et al. Antioxidant therapy to prevent preeclampsia. Obstet Gynecol. 2007;110:1311–8. doi: 10.1097/01.AOG.0000289576.43441.1f. [DOI] [PubMed] [Google Scholar]
- 35.Vadillo-Ortega F, Perichart-Perera O, Espino S, Avila-Vergara MA, Ibarra I, Ahued R, et al. Effect of supplementation during pregnancy with L-arginine and antioxidant vitamins in medical food on pre-eclampsia in high risk population: randomised controlled trial. BMJ. 2011;342:1–8. doi: 10.1136/bmj.d2901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zheng L, Huang J, Kong H, Wang F, Su Y, Xin H. The effect of folic acid throughout pregnancy among pregnant women at high risk of pre-eclampsia: a randomized clinical trial. Pregnancy Hypertens. 2020;19:253–8. doi: 10.1016/j.preghy.2020.01.005. [DOI] [PubMed] [Google Scholar]
- 37.Villar J, Purwar M, Merialdi M, Zavaleta N, thi Nhu Ngoc N, Anthony J, et al. World Health Organisation multicentre randomised trial of supplementation with vitamins C and E among pregnant women at high risk for pre-eclampsia in populations of low nutritional status from developing countries. BJOG: Int J Obstet Gynaecol. 2009;116:780–8. doi: 10.1111/j.1471-0528.2009.02158.x. [DOI] [PubMed] [Google Scholar]
- 38.Azami M, Azadi T, Sc M, Farhang S, Rahmati S, Pourtaghi K. The effects of multi mineral-vitamin D and vitamins (C+E) supplementation in the prevention of preeclampsia: an RCT. Int J Reprod BioMed. 2017;15:273–8. [PMC free article] [PubMed] [Google Scholar]
- 39.Beazley D, Ahokas R, Livingston J, Griggs M, Sibai BM. Vitamin C and E supplementation in women at high risk for preeclampsia: a double-blind, placebo-controlled trial. Am J Obstet Gynecol. 2005;192:520–1. doi: 10.1016/j.ajog.2004.09.005. [DOI] [PubMed] [Google Scholar]
- 40.Poston L, Briley A, Seed P, Kelly F, Shennan A. Vitamin C and vitamin E in pregnant women at risk for pre-eclampsia (VIP trial): randomised placebo-controlled trial. Lancet. 2006;367:1145–54. doi: 10.1016/S0140-6736(06)68433-X. [DOI] [PubMed] [Google Scholar]
- 41.Wen SW, White RR, Rybak N, Gaudet LM, Robson S, Hague W, et al. Effect of high dose folic acid supplementation in pregnancy on pre-eclampsia (FACT): double blind, phase III, randomised controlled, international, multicentre trial. BMJ. 2018;362:1–8. doi: 10.1136/bmj.k3478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hatton DC, Yue Q, McCarron DA. Mechanisms of calcium’s effects on blood pressure. Semin Nephrol. 1995;15:593–602. [PubMed] [Google Scholar]
- 43.DeSousa J, Tong M, Wei J, Chamley L, Stone P, Chen Q. The anti-inflammatory effect of calcium for preventing endothelial cell activation in preeclampsia. J Hum Hypertens. 2016;30:303–8. doi: 10.1038/jhh.2015.73. [DOI] [PubMed] [Google Scholar]
- 44.Purswani JM, Gala P, Dwarkanath P, Larkin HM, Kurpad A, Mehta S. The role of vitamin D in pre-eclampsia: a systematic review. BMC Pregnancy Childbirth. 2017;17:1–15. doi: 10.1186/s12884-017-1408-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Aouache R, Biquard L, Vaiman D, Miralles F. Oxidative stress in preeclampsia and placental diseases. Int J Mol Sci. 2018;19:1–29. doi: 10.3390/ijms19051496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Liu C, Liu C, Wang Q, Zhang Z. Supplementation of folic acid in pregnancy and the risk of preeclampsia and gestational hypertension: a meta-analysis. Archives Gynecol Obstet. 2018;298:697–704. doi: 10.1007/s00404-018-4823-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Keats EC, Haider BA, Tam E, Bhutta ZA. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst Rev. 2019;3:1–128. doi: 10.1002/14651858.CD004905.pub6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tan MY, Wright D, Syngelaki A, Akolekar R, Cicero S, Janga D, et al. Comparison of diagnostic accuracy of early screening for pre-eclampsia by NICE guidelines and a method combining maternal factors and biomarkers: results of SPREE. Ultrasound Obstet Gynecol. 2018;51:743–50. doi: 10.1002/uog.19039. [DOI] [PubMed] [Google Scholar]
- 49.Poon LCY, Stratieva V, Piras S, Piri S, Nicolaides KH. Hypertensive disorders in pregnancy: combined screening by uterine artery Doppler, blood pressure and serum PAPP-A at 11–13 weeks. Prenatal Diagnosis. 2010;30:216–23. doi: 10.1002/pd.2440. [DOI] [PubMed] [Google Scholar]
- 50.Odibo AO, Zhong Y, Goetzinger KR, Odibo L, Bick JL, Bower CR, et al. First-trimester placental protein 13, PAPP-A, uterine artery Doppler and maternal characteristics in the prediction of pre-eclampsia. Placenta. 2011;32:598–602. doi: 10.1016/j.placenta.2011.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.