Abstract
CYP19A1, also called aromatase, is a key enzyme for converting androgens to estrogens of estrogen synthesis. Elevated serum estrogen and high expression levels of estrogen-related proteins are found in cholangiocarcinoma (CCA; bile duct cancer). However, the expression of CYP19A1 in relation to estrogen-related proteins, including estrogen receptors (ERα, ERβ, and GPR30) and an estrogen response protein (TFF1), has never been explored in CCA. In this study, we investigated the expressions of CYP19A1 and estrogen-related proteins in CCA tissues (n = 74; 51 males and 23 females) using immunohistochemistry. The results showed that CYP19A1 was overexpressed in CCA cells compared with that in normal bile duct cells in the adjacent tissues. High expression of CYP19A1 was correlated with the metastatic status of the patients. High CYP19A1 expression was also positively correlated with GPR30 expression. Correlation between high CYP19A1 expression in the tumor tissues and shorter survival time was more prominent in male than in female CCA patients. To elucidate further, the effect of CYP19A1 knockdown on a CCA cell line was examined using a specific siRNA. When CYP19A1 gene expression was suppressed, migration and proliferation activities of CCA cells were significantly reduced. Moreover, the cell proliferation of high CYP19A1-expressing KKU-213 cells was more profoundly suppressed by CYP19A1 inhibitors (exemestane and letrozole) than low CYP19A1-expressing KKU-100 cells. Thus, CYP19A1 promotes CCA progression with aggressive clinical outcomes via increased migration and proliferation activities of cancer cells. CYP19A1 can be a potential chemotherapeutic target for CCA, especially in male patients.
Electronic supplementary material
The online version of this article (10.1007/s12672-018-0349-2) contains supplementary material, which is available to authorized users.
Keywords: Cholangiocarcinoma, CYP19A1, Aromatase, Estrogen, Estrogen receptors, Tumor progression
Introduction
Cholangiocarcinoma (CCA) is a liver cancer originating from bile duct epithelium and is the second most common primary hepatic malignancy worldwide [1]. Thailand has still the highest incidence and mortality rates of CCA in the world, especially in the Northeast of Thailand, where the incidence of CCA reaches up to 87.7 per 100,000 in males and 36.3 per 100,000 in females [2]. The high incidence of CCA in this region is associated with chronic inflammation of the biliary tract caused by liver fluke (Opisthorchis viverrini) infection, which is also highly prevalent in this area [3]. CCA is known as a “silent killer” because of its relatively silent clinical progression, which renders it difficult to diagnose at its early stages [4]. Therefore, the study on the molecular mechanisms of CCA development and progression is needed for identifying potential prognostic markers and therapeutic targets in CCA.
Estrogen is a group of sex steroid hormones and plays crucial roles in both reproductive and non-reproductive systems. The action of estrogen is mediated through estrogen receptor (ER) signaling pathways [5]. Increased estrogen levels and the effect of estrogen on tumor progression have been reported in breast, lung, endometrium, liver, bladder, and bile duct cancers [6–14]. In CCA, estrogen stimulates cell proliferation, cell invasion, and angiogenesis through ERs by inducing the expressions of pAKT, pERK1/2, trefoil factor 1 (TFF1), EGFR/MAPK, vascular endothelial growth factor (VEGF), and E26 transformation-specific variant 4 (ETV4) [12–17]. Male CCA patients have higher serum estrogen levels than age- and sex-matched controls, and high serum estrogen level was significantly correlated with poor survival of the patients [12]. These findings strongly suggest that estrogen induces CCA progression and is associated with aggressive clinical outcomes. Thus, estrogen and estrogen-related molecules may be potential molecular targets for diagnosis and treatment of CCA.
Cytochrome P450 family 19 subfamily A member 1 (CYP19A1; also called aromatase), a member of cytochrome P450 superfamily, is a key enzyme in estrogen biosynthesis, catalyzing the aromatization of aromatizable androgens (androstenedione and testosterone) to estrogens [18]. The overexpression of CYP19A1 is involved in progression of estrogen-related cancers such as breast, lung, and bladder cancers [8, 19, 20]. For example, in non-small cell lung cancer, high expression of CYP19A1 in the tumor tissues was correlated with higher estrogen levels in tumor tissues, higher cell proliferation rates, and poor prognosis of the patients [8, 21]. CYP19A1 expression has been shown to play an important role in the development and progression of ER-positive breast cancer [20]. Conversely, suppression of CYP19A1 reduced cell proliferation, migration, and invasion and increased apoptosis of non-small cell lung cancer cells [22, 23]. Thus, upregulation of CYP19A1 induces tumor progression of estrogen-related cancers.
Biliary obstruction and reduced estrogen turnover rates have been reported to be associated with the elevation of serum estrogen levels in CCA patients [12, 24, 25]. We further hypothesized that an estrogen-producing enzyme (CYP19A1) is upregulated in ER-positive CCA cells and could be a possible cause of elevated serum estrogen levels of CCA patients. To confirm this hypothesis, the expression patterns of CYP19A1 and estrogen-related proteins including estrogen receptors (ERα, ERβ, and GPR30) and an estrogen response protein (TFF1) in CCA tissues were determined using immunohistochemistry. In addition, the correlations between the protein expressions and the clinicopathological features (i.e., age, sex, histological types, metastatic status, and the survival rates of CCA patients) were analyzed. To further validate the importance of CYP19A1 in proliferation and metastatic activities of CCA cell lines, silencing CYP19A1 gene was performed by treatment of CCA cells with either a specific siRNA or CYP19A1 inhibitors (exemestane and letrozole). The overall results show that overexpression of CYP19A1 plays important roles in proliferation and migration of ER-positive CCA cells and can be used to determine the prognosis of CCA patients.
Materials and Methods
Human CCA Tissues
Paraffin-embedded human CCA tissues (n = 74; 51 males and 23 females) were obtained from the Cholangiocarcinoma Research Institute, Khon Kaen University, Thailand.
Immunohistochemistry
Immunohistochemical staining was performed to determine the expressions and localizations of all targeted proteins (i.e., CYP19A1, ERα, ERβ, GPR30, and TFF1) in human CCA tissues. The sections (5 μm thick) of paraffin-embedded human CCA tissues were deparaffinized in xylene and rehydrated with descending series of ethanol. For the heat-induced antigen retrieval, the sections were immersed in 10 mM sodium citrate buffer (pH 6) and heat-treated in a pressure cooker for 5 min. Endogenous peroxidase activity was blocked by 0.3% (v/v) hydrogen peroxide in phosphate-buffered saline (PBS) at room temperature for 30 min. Then, the sections were incubated for 30 min with 10% skim milk in PBS to block non-specific binding and were incubated with a primary antibody for 1 h at room temperature and then 4 °C overnight. After washing, the sections were incubated with peroxidase-conjugated Envision™ secondary antibody (Dako, Denmark) for 1 h at room temperature. Peroxidase activity was detected using a DAB (3, 3′-diaminobenzidine tetrahydrochloride) substrate kit (Vector Laboratories, Inc., USA) for 5 min, followed by nuclear counterstaining with hematoxylin. Tissue sections were dehydrated with the ascending ethanol series, cleared in xylene, and mounted with mounting solution.
In this study, the following primary antibodies were used: 10 μg/mL rabbit polyclonal anti-CYP19A1 antibody (Sigma-Aldrich, USA), 24.15 μg/mL rabbit monoclonal anti-ERα antibody (Abcam, UK), 2 μg/mL rabbit polyclonal anti-ERβ antibody (Abcam, UK), 1.33 μg/mL rabbit polyclonal anti-GPR30 antibody (Abcam, UK), and 1 μg/mL mouse monoclonal anti-TFF1 antibody (Sigma-Aldrich, USA).
The stained sections were reviewed under a light microscope. The DAB-positive staining was seen as brown to dark-brown color. Cell nuclei were appeared as blue-purple colors. The immunohistochemistry score (IHC score) was calculated by the multiplying of the intensities of DAB staining results (scored as 0 = negative, 1 = weak, 2 = modulate, and 3 = strong) and the frequency of the staining results (scored as 0 = none, + 1 = 1–25%, + 2 = 26–50%, + 3 = 51–75%, and + 4 = > 75%) [26]. Thus, the IHC score ranged from 0 to 12. In this study, the IHC score was used to discriminate high (IHC score > 4) and low (IHC score ≤ 4) expression patterns.
Cell Lines
Two distinct human intrahepatic CCA cell lines, KKU-100 and KKU-213, were established from primary tumors of CCA patients in the Northeast of Thailand and obtained from the Cholangiocarcinoma Research Institute, Khon Kaen University, Thailand. Immortalized human cholangiocyte cell line (MMNK1) was originally obtained from Okayama University, Japan [27], and maintained also in the Cholangiocarcinoma Research Institute, Khon Kaen University. All cell lines were cultured in complete medium which is Ham’s F-12 (Gibco®, Life technologies™, USA) supplemented with 10% heat-inactivated fetal bovine serum, 100 U/mL penicillin, and 100 mg/mL streptomycin (Life technologies™, USA). Cell lines were incubated in a humidified incubator with 5% CO2 and 95% relative humidity at 37 °C.
Small Interfering RNA Against CYP19A1
Specific siRNA for CYP19A1 (siCYP19A1) was designed by GE Healthcare Dharmacon (ON-TARGETplus SMARTpool Human CYP19A1 siRNA, GE Healthcare Dharmacon Inc., USA). The cells were transfected with siCYP19A1 using Lipofectamine RNAiMAX® (Thermo Fisher Scientific, USA) in a 6-well plate with 105 cells for 72-h transfection. The cells treated with scramble siRNA (ON-TARGETplus Non-targeting Control siRNA, GE Healthcare Dharmacon Inc., USA) were used as controls (scramble). Cells were harvested using trypsinization for RNA extraction, immunocytochemical analysis, cell migration assay, and cell viability assay.
RNA Extraction and Real-time PCR
Total RNA was isolated from cell pellets with Trizol® reagent (Invitrogen, USA) according to the manufacturer’s protocol. The quality of RNA was assessed with a NanoDrop ND-2000 spectrophotometer (NanoDrop Technologies, USA). Then, 2 μg of total RNA was converted to cDNA using High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, USA) as described in the manufacturer’s protocol. The expression levels of CYP19A1, BIRC5, and β-actin mRNA were measured using TaqMan gene expression assay with TaqMan probes (Hs00903411_m1 CYP19A1, Hs04194392_s1 BIRC5, and Hs99999903_m1 β-actin). The PCR was performed in an ABI-7500 real-time PCR system (Applied Biosystems, USA). Relative mRNA expression was analyzed with a cycle threshold (Ct) in the linear range of amplification using β-actin as an internal control.
Immunocytochemistry
Each cell lines (5.0 × 104 cells/well) were placed in a 48-well plate and incubated for 24 h. The attached cells were fixed with 10% paraformaldehyde in PBS for 30 min at room temperature. After washing, cells were incubated with 0.2% (v/v) Triton-X100 solution for 2 min. The cells were incubated with 0.3% (v/v) hydrogen peroxide in PBS for 30 min to block endogenous hydrogen peroxide activity and subsequently incubated with 3% (w/v) BSA in PBS for 30 min to block non-specific binding. The cells were incubated with each primary antibody: 10 μg/mL rabbit polyclonal anti-CYP19A1 antibody (Sigma-Aldrich, USA), 24.15 μg/mL rabbit monoclonal anti-ERα antibody (Abcam, UK), 2 μg/mL rabbit polyclonal anti-ERβ antibody (Abcam, UK), or 1.33 μg/mL rabbit polyclonal anti-GPR30 antibody (Abcam, UK) at 4 °C overnight. After extensive washing, the cells were incubated with peroxidase-conjugated secondary antibody. The peroxidase activity was detected as brown reaction product using a DAB substrate kit. After dehydration, the stained cells were reviewed under an inverted microscope.
Cell Migration Assay
The cell migration assay was performed using a Boyden transwell chamber consisting of a membrane filter insert with 8-μm pore size in a 24-well plate (Corning, USA). In brief, 72 h after knockdown treatment with siCYP19A1 or scramble (control), 4 × 104 cells were placed into the insert upper chamber with serum-free medium. In the lower chamber, complete medium was added. Then, the chambers were incubated for 12 or 18 h. Non-migrating cells in the upper chamber were removed. Migrated cells that attached at the undersurface of the filter were fixed with absolute methanol for 1 h and stained with hematoxylin overnight. The membrane was allowed to dry and the number of migrated cells were quantified and analyzed under a light microscope.
Cell Viability Assay
MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay was used to investigate CCA cell proliferation. Cells in each well of a 96-well flat-bottom microtiter plate (in triplicate) were incubated with 0.5 mg/mL MTT solution in a complete medium at 37 °C for 4 h in the dark. After washing, formazan crystal was solubilized by adding 100 μL DMSO and the plate was shaken using an orbital shaker for 10 min in the dark. The absorbance was measured at 540 nm using a microplate reader (Tecan Sunrise, Switzerland).
Estrogen Treatment
After 72 h of siCYP19A1 transfection, KKU-213 cells were reseeded and treated with or without 1 nM of 17β-estradiol (Sigma-Aldrich, USA) in the cell culture conditions. Cell migration was assessed by Boyden transwell chamber assay and cell viability was analyzed by MTT assay.
CYP19A1 Inhibitor Treatment
Exemestane (Pfizer Inc., USA) and letrozole (Novartis, Switzerland) were dissolved in dimethyl sulfoxide (DMSO) and pre-diluted in the complete medium before use. The final concentration of DMSO was adjusted to 0.5% in all conditions. The cell lines were treated with various concentrations of exemestane or letrozole for 48 h. After treatment, the cells were subjected to MTT assay.
Statistical Analysis
Statistical analysis was performed using SPSS Statistics software version 19.0 (IBM Cooperation, USA). The Kaplan-Meier estimate with log-rank test was used for survival analysis. The correlations between protein expression patterns in CCA tissues and clinicopathological data were analyzed using Pearson’s chi-square test. The correlations among protein expressions were analyzed by Pearson correlation. The results of mRNA expression, cell migration assay, and cell proliferation assay were analyzed using Student’s t test. P value < 0.05 is considered statistically significant.
Results
Expression of CYP19A1 in CCA Tissues
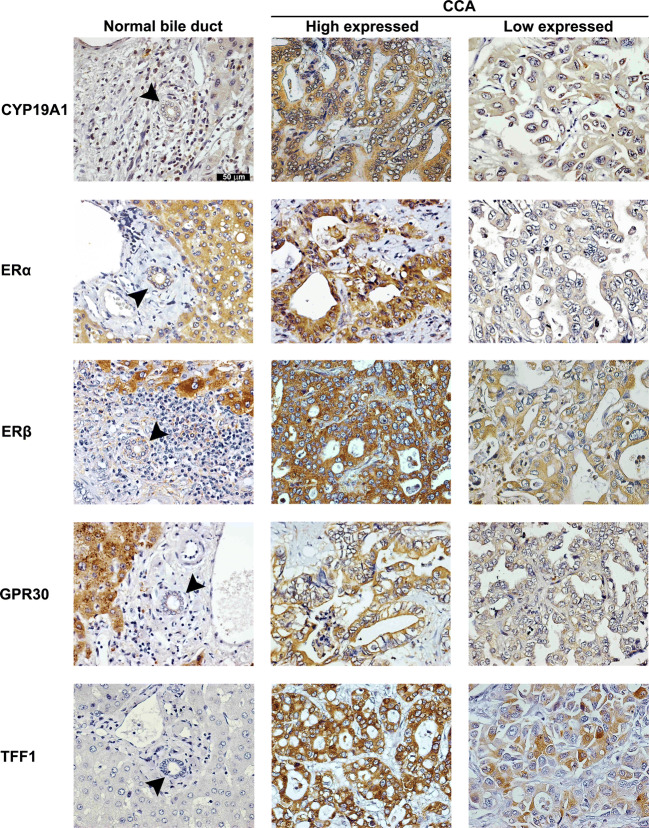
Immunohistochemical staining of CCA tissues demonstrated that CYP19A1 was strongly expressed in the cytoplasm of cancer cells but it was slightly expressed in the nucleus (Fig. 1). CYP19A1 was also slightly expressed in the cytoplasm of hepatocytes. The degrees of CYP19A1 expression determined by immunohistochemistry were significantly higher in the CCA cells compared with those in the normal bile duct epithelial cells located at the adjacent area (P < 0.001; graph not shown). More than half of the CCA tissues examined (45/74, 61%) showed high expression of CYP19A1.
Fig. 1.
The expressions of CYP19A1, ERα, ERβ, GPR30, and TFF1 in normal bile duct of the tumor adjacent area and CCA tissues (n = 74; original magnification × 200). Arrow heads indicate normal bile ducts. Positive staining immunoreactivities were represented with brown color. Blue color represented the nucleus
Expression of Estrogen Receptors in CCA Tissues
The expression patterns of three estrogen receptors, ERα, ERβ, and GPR30, in CCA tissues were shown in Fig. 1. While ERα and ERβ were seen as the cytoplasmic and nuclear distribution patterns in cancer cells, GPR30 was located at the cytoplasm and the cell membrane of cancer cells. All CCA tissues showed positive staining of ERα, ERβ, and/or GPR30 (100%, 74/74). Significantly positive correlation was observed between the expression patterns of ERα and ERβ (r = 0.263 and P = 0.024; Pearson correlation analysis). Also, the expression levels of all three estrogen receptors in immunohistochemistry were significantly higher in CCA cells than in the normal bile duct epithelial cells in the tumor adjacent tissues (P < 0.001; graph not shown). High expression levels of ERα, ERβ, and GPR30 in IHC score were observed in 78% (58/74), 50% (37/74), and 38% (28/74), respectively, of the CCA patients. All estrogen receptors were also highly expressed in the cytoplasm of hepatocytes. In spite of the high expression patterns of ERα, ERβ, and GPR30 in CCA tissues, their expression levels were not correlated with clinicopathological data such as age, sex, metastasis status, and CCA histology types.
Expression of TFF1, as a Representative of Estrogen-Responsive Proteins, in CCA Tissues
As a representative of estrogen-responsive proteins [15], the expression of TFF1 in both CCA cells and adjacent normal bile duct cells was examined using immunohistochemical staining. As shown in Fig. 1. TFF1 protein was highly expressed in the cytoplasm of CCA cells, whereas it was negative in normal bile duct epithelial cells. Among 74 CCA cases, 39 cases (53%) have high TFF1 expression pattern, although it was not correlated with clinicopathological data including age, sex, and CCA histology types. Moreover, TFF1 expression was significantly correlated with ERβ expression (r = 0.244 and P = 0.036; Pearson correlation).
Correlations of CYP19A1 with Clinicopathological Data and Estrogen-Related Proteins
Table 1 shows the correlations between CYP19A1 expression patterns and clinicopathological data as well as estrogen-related protein expression patterns. CYP19A1 expression patterns were not correlated with the sex and age of CCA patients. The expression of CYP19A1 in CCA tissues was significantly correlated with metastatic status (r = 0.449 and P < 0.001; Pearson correlation) and GPR30 expression (r = 0.284 and P = 0.014; Pearson correlation).
Table 1.
Correlations of CYP19A1 expression with estrogen-related protein expressions and clinicopathological features
| Value | CYP19A1 in CCA | ||
|---|---|---|---|
| Low (n = 29) | High (n = 45) | #P value | |
| Age | |||
| < 57 | 15 | 21 | 0.671 |
| ≥ 57 | 14 | 24 | |
| Sex | |||
| Male | 20 | 31 | 0.994 |
| Female | 9 | 14 | |
| Metastasis status | |||
| Non-metastasis | 21 | 12 | < 0.001 |
| Metastasis | 8 | 33 | |
| Histological type | |||
| Tubular | 12 | 27 | 0.117 |
| Papillary | 17 | 18 | |
| Estrogen-related protein | |||
| ERα | |||
| Low | 7 | 9 | 0.673 |
| High | 22 | 36 | |
| ERβ | |||
| Low | 18 | 19 | 0.096 |
| High | 11 | 26 | |
| GPR30 | |||
| Low | 23 | 23 | 0.015 |
| High | 6 | 22 | |
| TFF1 | |||
| Low | 17 | 18 | 0.117 |
| High | 12 | 27 | |
#P value was analyzed by Pearson’s chi-square test
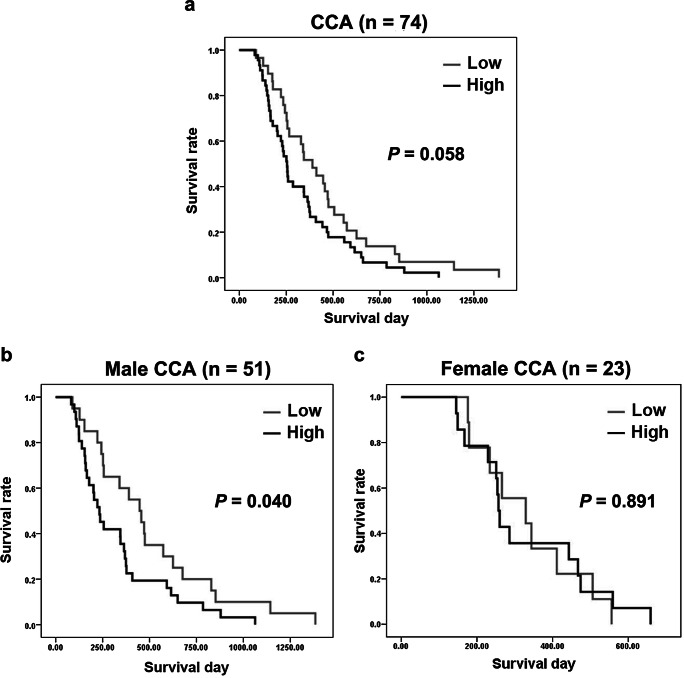
Correlations Between CYP19A1 Expression Levels and the Survival Rate of Patients
CCA patients with high CYP19A1 expression in their tumor tissues showed no correlation with poor prognosis (P = 0.058; log-rank test) as shown in Fig. 2a. When CCA patients were divided into males and females, male CCA patients showed high expression of CYP19A1 in tumor tissues, which was significantly correlated with poor prognosis (P = 0.040; log-rank test) as shown in Fig. 2b. The estimated median survival time of male CCA patients with high expression of CYP19A1 (233 days) in the cancer tissues was shorter than that of CCA patients with low CYP19A1 expression (447 days). In contrast, no correlation between CYP19A1 expression patterns in tumor tissues and the survival rates was observed in female CCA patients (Fig. 2c) which can be explained by higher circulating concentrations of estrogens in women than in men, although the sample size of females in this study is not being enough to draw any solid conclusion. These results suggested that CYP19A1 plays essential roles in CCA progression and its expression can be used as a CCA prognostic marker, especially for male CCA patients.
Fig. 2.
Kaplan-Meier analysis of CYP19A1 in a all human CCA tissues (n = 74), b male CCA tissues (n =51), and c female CCA tissues (n = 23)
Expressions of CYP19A1 and Estrogen Receptors in Immortal Cholangiocyte and CCA Cell Lines
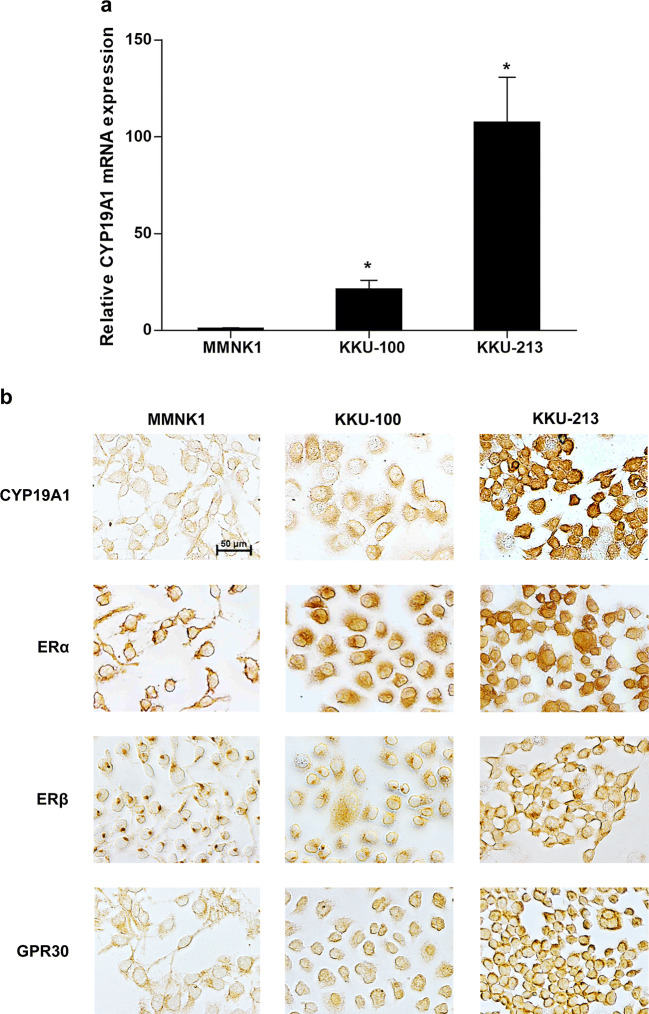
The mRNA and protein expression levels of CYP19A1 in two CCA cell lines and in an immortalized cholangiocyte cell line, MMNK1, were examined using real-time PCR and immunocytochemistry. CYP19A1 mRNA and protein levels were highly expressed in CCA cell lines (KKU-213 > KKU-100) compared to those in MMNK1 cell line (Fig. 3a, b). Likewise, ERα, ERβ, and GPR30 were also highly expressed in all CCA cells compared to those in MMNK1 cells, according to the immunocytochemistry results (Fig. 3b).
Fig. 3.
The expressions of CYP19A1, ERα, ERβ, and GPR30 in cholangiocyte (MMNK1) and CCA (KKU-100 and KKU-213) cell lines. a Relative CYP19A1 mRNA expression levels detected by real-time PCR. *P value < 0.05 compared to MMNK1. b CYP19A1, ERα, ERβ, and GPR30 protein expressions detected by immunocytochemistry. Positive staining results were represented in brown color
Effect of CYP19A1 Gene Silencing by siRNA on Cell Proliferation and Migration
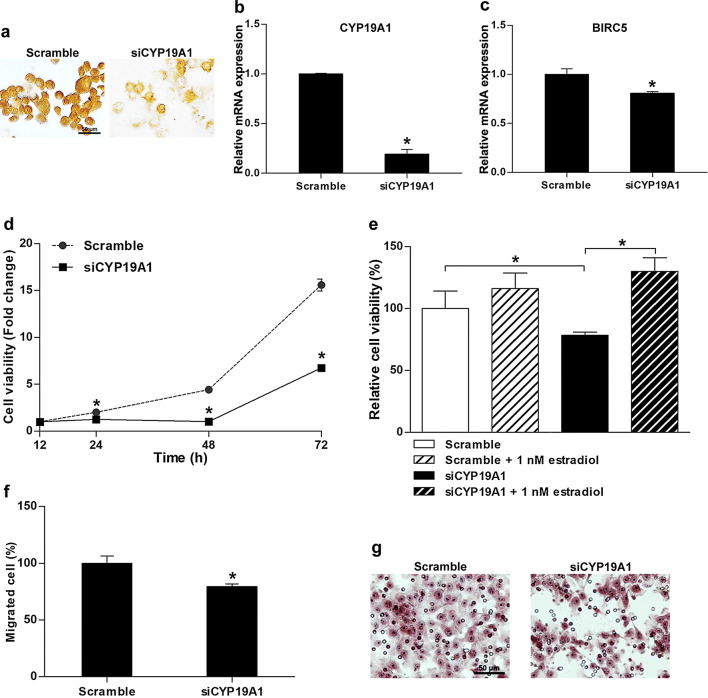
Effect of CYP19A1 suppression by siRNA was examined in KKU-213, a high-CYP19A1-expressing CCA cell line. After silencing by specific siRNA (siCYP19A1), both CYP19A1 protein and mRNA levels were decreased at 72 h after treatment when compared with those treated with the scramble (Fig. 4a, b). Moreover, after siCYP19A1 transfection, the expression of BIRC5, an anti-apoptotic gene, was found to be decreased (Fig. 4c). Low expression levels of BIRC could be related to a decrease in CCA cell proliferation rates at 24, 48, and 72 h after re-seeding (Fig. 4d). However, adding exogenous estradiol to the culture media could restore the reduction of proliferative activity of CYP19A1-knockdown-KKU213 cells (Fig. 4e). On the other hand, siCYP19A1 treatment significantly could reduce the migration activity of cancer cells at 12 h after re-seeding (Fig. 4f, g). However, the reduced migration activity was rescued by adding external estradiol, as evidence by an increase in the number of migrated cells in scramble- or siCYP19A1-treated cells as shown in Fig. S1 (see Supporting Information). Thus, our results confirm that CYP19A1 increases estrogen generation, which leads to the upregulation of the estrogen response and results in CCA progression with aggressive clinical outcomes.
Fig. 4.
Effect of CYP19A1 silencing on KKU-213 CCA cell proliferation and migration between cells transfected with siCYP19A1 or scramble. a Expression levels of CYP19A1 protein (brown staining) in cells treated with siCYP19A1 or scramble. b Relative CYP19A1 mRNA expression levels (mean ± SD) detected by real-time PCR. c Relative BIRC5 mRNA expression levels (mean ± SD) analyzed by real-time PCR. d Cell viability assays (represented as fold change ± SD relative to scrambled control) in cells treated with siCYP19A1 or scramble. e Effect of 17β-estradiol on cell viability, represented as relative percentage ± SD, analyzed using MTT assay at 48 h post treatment. f Effect of siCYP19A1 on the cell migration expressed as a percentage of migrated cells (mean ± SD) at 12 h post migration and g hematoxylin staining of migrated cells (red-purple color) at 12 h post migration. *P value < 0.05 compared to a scramble control (b–d, f). Scale bars are equal to 50 μm
Effects of CYP19A1 Inhibitors on Cell Viability of CCA Cell Lines
The effects of exemestane and letrozole on cell viability of two different CCA cell lines, i.e., CYP19A1-low-KKU-100 and CYP19A1-high-KKU-213, were investigated. The selected cell lines were treated with various concentrations of exemestane or letrozole for 48 h. The results showed that the calculated IC50 (mean ± SD) of CYP19A1 inhibitors for KKU-213 were 5.36 ± 2.68 μM of exemestane and 42.00 ± 16.35 μM of letrozole which are more sensitive than those of KKU-100 with IC50 76.42 ± 12.11 μM of exemestane and 125.59 ± 31.64 μM of letrozole (Fig. 5). These results suggest that both CYP19A1 inhibitors are more effective in suppressing cell viabilities of CYP19A1-high-KKU-213 cells than those of CYP19A1-low-KKU-100 cells.
Fig. 5.
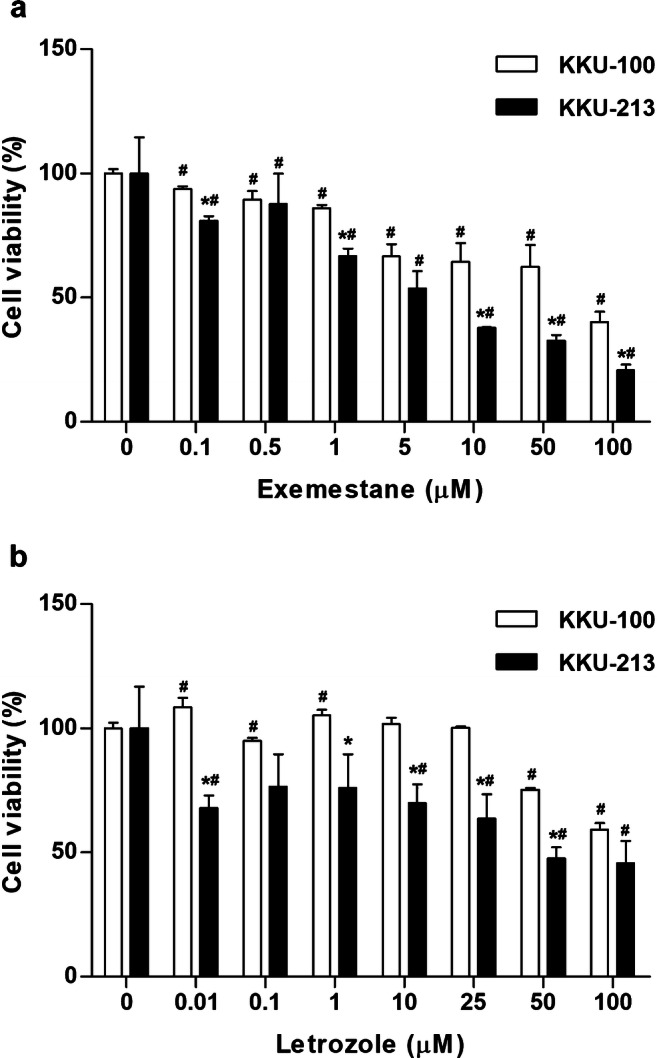
Effect of exemestane (a) and letrozole (b) on cell viability of CCA cell lines. The percentage of relative cell viabilities (mean ± SD) was investigated by MTT assay in KKU-100 and KKU-213 treated with various concentrations of exemestane or letrozole for 48 h. *P value < 0.05 compared to KKU-100 at the same concentrations. #P value < 0.05 compared to 0 μM of drug in each cell type
Discussion
Estrogen receptors and estrogen responsive proteins play important roles in CCA progression via induction of cell proliferation, cell invasion, and angiogenesis [12–14]. Moreover, serum estrogen levels of male CCA patients were significantly higher than in healthy male subjects [12]. The present results demonstrated that CYP19A1 was overexpressed in CCA cells compared with that in normal bile duct cells in the adjacent normal tissues. The majority of CCA patients have high expression levels of CYP19A1 and estrogen receptors (ERα, ERβ, and/or GPR30). A positive correlation between the expression levels of CYP19A1 and estrogen receptors was observed. These results suggest that overexpressed CYP19A1 may upregulate the estrogen production by converting androgen to estrogen resulting in high concentrations of estrogen in CCA cells. These processes promote the induction of estrogen responses of CCA cells and could be, at least in part, a possible cause of elevated serum estrogen levels of CCA patients.
CYP19A1 overexpression is involved in the progression of various types of estrogen-related cancers such as breast, lung, and bladder cancers [18–20, 22]. In the present study, overexpression of CYP19A1 was significantly correlated with metastatic status of CCA patients. CYP19A1 may promote CCA progression through induction of cell invasion and migration. This finding was further confirmed by the knockdown of CYP19A1 in CCA cell lines using specific siRNA. CYP19A1 silencing significantly reduced the cell migration activity of CCA cell lines. The reduction of cell migration activity was rescued by adding external estradiol to the cell culture media. Related to this, the expression of CYP19A1 in CCA tissues was not significantly correlated with poor prognosis in general. However, statistically significant correlation was observed in male CCA patients. These findings suggested that CYP19A1 is involved in CCA progression and is associated with aggressive clinical outcomes, especially in male CCA patients. Furthermore, CYP19A1 expression was positively correlated with GPR30 expression. GPR30 modulates both genomic transcriptional action and rapid non-genomic action of estrogen [28] and is involved in progression of estrogen-related cancers such as breast, endometrium, and ovarian cancers through the activations of EGFR/MAPK and PI3K/Akt signaling pathways [29–31]. Since the EGFR/MAPK and PI3K/Akt signaling pathways are implicated in the progression of CCA [16, 32, 33], the activation of GPR30 by estrogen is associated with CCA progression via modulation of EGFR/MAPK and PI3K/Akt signaling pathways.
Since overexpression of CYP19A1 has been reported in estrogen-related cancers and CYP19A1 inhibitors including anastrozole, letrozole, and exemestane are currently in clinical use for breast cancer treatment [34], exemestane is known to reduce cell viability of breast cancer cells via activation of mitochondrial-mediated apoptosis and induction of autophagy [35]. This compound also inhibited tumorigenic properties and caused morphological changes of non-small cell lung cancer cell lines [22, 23]. On the other hand, letrozole could reduce tumor size and induce apoptosis of ER-positive breast cancer cells both in vitro and in vivo [20]. Additionally, plasma estrogen levels of postmenopausal women with breast cancer were downregulated by CYP19A1 inhibitors [36, 37]. In the present study, two CYP19A1 inhibitors, exemestane and letrozole, could suppress cell proliferation of both high- and low-CYP19A1-expressing CCA cell lines. Drug sensitivities of the cell lines were related to the basal level of CYP19A1 expression. Similarly, silencing of CYP19A1 using specific siRNA reduced cell proliferation of CCA cells via downregulation of BIRC5 which is an anti-apoptotic gene. This result was consistent with BIRC5 being known as an estrogen-inducible gene in human ovarian and breast cancer cells [38, 39]. Moreover, BIRC5 expression was associated with development and poor prognosis of CCA [40, 41], suggesting its anti-apoptotic role in CCA. Interestingly, the reduction of cell proliferation by siCYP19A1 was rescued by the addition of exogenous estradiol. Therefore, in the case of CCA, it could be suggested that CYP19A1 promoted CCA cell proliferation and inhibited cell apoptosis via estrogen-related pathways.
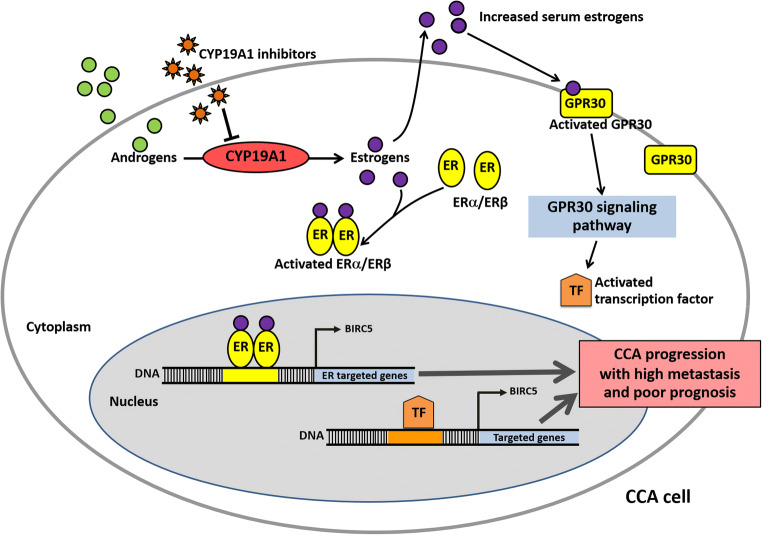
Based on the aforementioned findings, we propose possible mechanisms by which CYP19A1 and downstream estrogen-related proteins can cause CCA progression (Fig. 6). Briefly, the overexpression of CYP19A1 permits elevated conversion of androgen to estrogen, resulting in accumulation of estrogen in cancer cells and high serum estrogen levels. High estrogen levels can cause aberrant estrogen-ER interactions, which in turn can trigger the expressions of estrogen-responsive genes via activating nuclear estrogen receptor (ER) and/or membrane receptor (GPR30) signaling cascade and promoting ER/GPR30-dependent transcription factor changes in response to estrogen, leading to CCA progression with aggressive clinical outcomes. Therefore, CYP19A1 inhibitors could be applied for the targeted therapy of CCA.
Fig. 6.
Proposed mechanisms of CYP19A1 and estrogen-related proteins in CCA progression (ER estrogen receptor)
Electronic Supplementary Material
Effects of siCYP19A1 and 17β-estradiol on KKU-213 cell migratory activity. (a) Effect of siCYP19A1 on the cell migration expressed as a percentage of migrated cells (mean ± SD) at 18 h post migration and (b) hematoxylin staining of migrated cells (as shown in red-purple color) at 18 h post migration (PNG 134 kb)
Acknowledgements
We would like to acknowledge Prof. Yukifumi Nawa, for editing the manuscript via Publication Clinic KKU, Thailand.
Author Contributions
Designed research topic, drafted research grants, and analyzed data: RT. Drafted and wrote the manuscript: RT and WK. Performed experiments and analyzed data: WK, CS, PU, and NA. Analyzed clinical data of CCA patients: PU and NK. Critically reviewed the manuscript and provided input: NN and KS. All authors are involved in drafting the article critically for important intellectual content and have read and approved the final version of the manuscript.
Funding Information
The study was financially supported by the Cholangiocarcinoma Research Institute (CARI), Khon Kaen University, Thailand (LFCRC 005/2558) to RT and WK; Invitation Research Fund from Faculty of Medicine, Khon Kaen University, Thailand to WK and RT (IN60331); Khon Kaen University Research Fund to RT (611001); and Thailand Research Fund (RSA5980031) to RT.
Compliance with Ethical Standards
The protocols of sample collection and study plan were approved by the Ethics Committee for Human Research, Khon Kaen University (HE571283). All CCA subjects were provided with the approved informed consent.
Conflict of Interest
The authors declare that they have no conflict of interest.
References
- 1.Ito T, Sakurai-Yageta M, Goto A, Pairojkul C, Yongvanit P, Murakami Y. Genomic and transcriptional alterations of cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2014;21:380–387. doi: 10.1002/jhbp.67. [DOI] [PubMed] [Google Scholar]
- 2.Khuntikeo N, Chamadol N, Yongvanit P, Loilome W, Namwat N, Sithithaworn P, Andrews RH, Petney TN, Promthet S, Thinkhamrop K, Tawarungruang C, Thinkhamrop B, investigators C. Cohort profile: cholangiocarcinoma screening and care program (CASCAP) BMC Cancer. 2015;15:459. doi: 10.1186/s12885-015-1475-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sripa B, Pairojkul C. Cholangiocarcinoma: lessons from Thailand. Curr Opin Gastroenterol. 2008;24:349–356. doi: 10.1097/MOG.0b013e3282fbf9b3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sithithaworn P, Yongvanit P, Duenngai K, Kiatsopit N, Pairojkul C. Roles of liver fluke infection as risk factor for cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2014;21:301–308. doi: 10.1002/jhbp.62. [DOI] [PubMed] [Google Scholar]
- 5.Cui J, Shen Y, Li R. Estrogen synthesis and signaling pathways during aging: from periphery to brain. Trends Mol Med. 2013;19:197–209. doi: 10.1016/j.molmed.2012.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Russo J, Russo IH. The role of estrogen in the initiation of breast cancer. J Steroid Biochem Mol Biol. 2006;102:89–96. doi: 10.1016/j.jsbmb.2006.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chen GG, Zeng Q, Tse GM. Estrogen and its receptors in cancer. Med Res Rev. 2008;28:954–974. doi: 10.1002/med.20131. [DOI] [PubMed] [Google Scholar]
- 8.Niikawa H, Suzuki T, Miki Y, Suzuki S, Nagasaki S, Akahira J, Honma S, Evans DB, Hayashi S, Kondo T, Sasano H. Intratumoral estrogens and estrogen receptors in human non-small cell lung carcinoma. Clin Cancer Res. 2008;14:4417–4426. doi: 10.1158/1078-0432.CCR-07-1950. [DOI] [PubMed] [Google Scholar]
- 9.Stabile LP, Davis AL, Gubish CT, Hopkins TM, Luketich JD, Christie N, Finkelstein S, Siegfried JM. Human non-small cell lung tumors and cells derived from normal lung express both estrogen receptor alpha and beta and show biological responses to estrogen. Cancer Res. 2002;62:2141–2150. [PubMed] [Google Scholar]
- 10.Zhang Z, Zhou D, Lai Y, Liu Y, Tao X, Wang Q, Zhao G, Gu H, Liao H, Zhu Y, Xi X, Feng Y. Estrogen induces endometrial cancer cell proliferation and invasion by regulating the fat mass and obesity-associated gene via PI3K/AKT and MAPK signaling pathways. Cancer Lett. 2012;319:89–97. doi: 10.1016/j.canlet.2011.12.033. [DOI] [PubMed] [Google Scholar]
- 11.Shen SS, Smith CL, Hsieh JT, Yu J, Kim IY, Jian W, Sonpavde G, Ayala GE, Younes M, Lerner SP. Expression of estrogen receptors-alpha and -beta in bladder cancer cell lines and human bladder tumor tissue. Cancer. 2006;106:2610–2616. doi: 10.1002/cncr.21945. [DOI] [PubMed] [Google Scholar]
- 12.Hunsawong T, Singsuksawat E, In-chon N, Chawengrattanachot W, Thuwajit C, Sripa B, Paupairoj A, Chau-in S, Thuwajit P. Estrogen is increased in male cholangiocarcinoma patients’ serum and stimulates invasion in cholangiocarcinoma cell lines in vitro. J Cancer Res Clin Oncol. 2012;138:1311–1320. doi: 10.1007/s00432-012-1207-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mancino A, Mancino MG, Glaser SS, Alpini G, Bolognese A, Izzo L, Francis H, Onori P, Franchitto A, Ginanni-Corradini S, Gaudio E, Alvaro D. Estrogens stimulate the proliferation of human cholangiocarcinoma by inducing the expression and secretion of vascular endothelial growth factor. Dig Liver Dis. 2009;41:156–163. doi: 10.1016/j.dld.2008.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Alvaro D, Barbaro B, Franchitto A, Onori P, Glaser SS, Alpini G, Francis H, Marucci L, Sterpetti P, Ginanni-Corradini S, Onetti Muda A, Dostal DE, De Santis A, Attili AF, Benedetti A, Gaudio E. Estrogens and insulin-like growth factor 1 modulate neoplastic cell growth in human cholangiocarcinoma. Am J Pathol. 2006;169:877–888. doi: 10.2353/ajpath.2006.050464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Thuwajit P, Chawengrattanachot W, Thuwajit C, Sripa B, May FE, Westley BR, Tepsiri NN, Paupairoj A, Chau-In S. Increased TFF1 trefoil protein expression in Opisthorchis viverrini-associated cholangiocarcinoma is important for invasive promotion. Hepatol Res. 2007;37:295–304. doi: 10.1111/j.1872-034X.2007.00045.x. [DOI] [PubMed] [Google Scholar]
- 16.Kosriwong K, Menheniott TR, Giraud AS, Jearanaikoon P, Sripa B, Limpaiboon T. Trefoil factors: tumor progression markers and mitogens via EGFR/MAPK activation in cholangiocarcinoma. World J Gastroenterol. 2011;17:1631–1641. doi: 10.3748/wjg.v17.i12.1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Singsuksawat E, Thuwajit C, Charngkaew K, Thuwajit P. Increased ETV4 expression correlates with estrogen-enhanced proliferation and invasiveness of cholangiocarcinoma cells. Cancer Cell Int. 2018;18:25. doi: 10.1186/s12935-018-0525-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bulun SE, Chen D, Lu M, Zhao H, Cheng Y, Demura M, Yilmaz B, Martin R, Utsunomiya H, Thung S, Su E, Marsh E, Hakim A, Yin P, Ishikawa H, Amin S, Imir G, Gurates B, Attar E, Reierstad S, Innes J, Lin Z. Aromatase excess in cancers of breast, endometrium and ovary. J Steroid Biochem Mol Biol. 2007;106:81–96. doi: 10.1016/j.jsbmb.2007.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nguyen DP, O’Malley P, Al Hussein Al Awamlh B, Furrer MA, Mongan NP, Robinson BD, Wang GJ, Scherr DS. Association of aromatase with bladder cancer stage and long-term survival: new insights into the hormonal paradigm in bladder cancer. Clin Genitourin Cancer. 2016;15:256–262.e1. doi: 10.1016/j.clgc.2016.05.017. [DOI] [PubMed] [Google Scholar]
- 20.Mukhopadhyay KD, Liu Z, Bandyopadhyay A, Kirma NB, Tekmal RR, Wang S, Sun LZ. Aromatase expression increases the survival and malignancy of estrogen receptor positive breast cancer cells. PLoS One. 2015;10:e0121136. doi: 10.1371/journal.pone.0121136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Skjefstad K, Grindstad T, Khanehkenari MR, Richardsen E, Donnem T, Kilvaer T, Andersen S, Bremnes RM, Busund LT, Al-Saad S. Prognostic relevance of estrogen receptor alpha, beta and aromatase expression in non-small cell lung cancer. Steroids. 2016;113:5–13. doi: 10.1016/j.steroids.2016.05.008. [DOI] [PubMed] [Google Scholar]
- 22.Giannopoulou E, Siatis KE, Metsiou D, Kritikou I, Papachristou DJ, Kalofonou M, Koutras A, Athanassiou G, Kalofonos HP. The inhibition of aromatase alters the mechanical and rheological properties of non-small-cell lung cancer cell lines affecting cell migration. Biochim Biophys Acta. 2015;1853:328–337. doi: 10.1016/j.bbamcr.2014.11.016. [DOI] [PubMed] [Google Scholar]
- 23.Koutras A, Giannopoulou E, Kritikou I, Antonacopoulou A, Evans TR, Papavassiliou AG, Kalofonos H. Antiproliferative effect of exemestane in lung cancer cells. Mol Cancer. 2009;8:109. doi: 10.1186/1476-4598-8-109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Narasaka T, Moriya T, Endoh M, Suzuki T, Shizawa S, Mizokami Y, Matsuoka T, Sasano H. 17Beta-hydroxysteroid dehydrogenase type 2 and dehydroepiandrosterone sulfotransferase in the human liver. Endocr J. 2000;47:697–705. doi: 10.1507/endocrj.47.697. [DOI] [PubMed] [Google Scholar]
- 25.Liossi AK, Aroni KG, Kyrkou KA, Kittas C, Markaki SP. Immunohistochemical study of sex steroid hormones in primary liver cancer. Cancer Detect Prev. 1988;13:195–201. [PubMed] [Google Scholar]
- 26.Thanan R, Murata M, Ma N, Hammam O, Wishahi M, El Leithy T, Hiraku Y, Oikawa S, Kawanishi S. Nuclear localization of COX-2 in relation to the expression of stemness markers in urinary bladder cancer. Mediat Inflamm. 2012;2012:165879. doi: 10.1155/2012/165879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Maruyama M, Kobayashi N, Westerman KA, Sakaguchi M, Allain JE, Totsugawa T, Okitsu T, Fukazawa T, Weber A, Stolz DB, Leboulch P, Tanaka N. Establishment of a highly differentiated immortalized human cholangiocyte cell line with SV40T and hTERT. Transplantation. 2004;77:446–451. doi: 10.1097/01.TP.0000110292.73873.25. [DOI] [PubMed] [Google Scholar]
- 28.Wang D, Hu L, Zhang G, Zhang L, Chen C. G protein-coupled receptor 30 in tumor development. Endocrine. 2010;38:29–37. doi: 10.1007/s12020-010-9363-z. [DOI] [PubMed] [Google Scholar]
- 29.Filardo EJ. Epidermal growth factor receptor (EGFR) transactivation by estrogen via the G-protein-coupled receptor, GPR30: a novel signaling pathway with potential significance for breast cancer. J Steroid Biochem Mol Biol. 2002;80:231–238. doi: 10.1016/S0960-0760(01)00190-X. [DOI] [PubMed] [Google Scholar]
- 30.Fujiwara S, Terai Y, Kawaguchi H, Takai M, Yoo S, Tanaka Y, Tanaka T, Tsunetoh S, Sasaki H, Kanemura M, Tanabe A, Yamashita Y, Ohmichi M. GPR30 regulates the EGFR-Akt cascade and predicts lower survival in patients with ovarian cancer. J Ovarian Res. 2012;5:35. doi: 10.1186/1757-2215-5-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ge X, Guo R, Qiao Y, Zhang Y, Lei J, Wang X, Li L, Hu D. The G protein-coupled receptor GPR30 mediates the nontranscriptional effect of estrogen on the activation of PI3K/Akt pathway in endometrial cancer cells. Int J Gynecol Cancer. 2013;23:52–59. doi: 10.1097/IGC.0b013e31827912b8. [DOI] [PubMed] [Google Scholar]
- 32.Padthaisong S, Thanee M, Techasen A, Namwat N, Yongvanit P, Liwatthakun A, Hankla K, Sangkhamanon S, Loilome W. Nimotuzumab inhibits cholangiocarcinoma cell metastasis via suppression of the epithelial-mesenchymal transition process. Anticancer Res. 2017;37:3591–3597. doi: 10.21873/anticanres.11729. [DOI] [PubMed] [Google Scholar]
- 33.Yothaisong S, Dokduang H, Techasen A, Namwat N, Yongvanit P, Bhudhisawasdi V, Puapairoj A, Riggins GJ, Loilome W. Increased activation of PI3K/AKT signaling pathway is associated with cholangiocarcinoma metastasis and PI3K/mTOR inhibition presents a possible therapeutic strategy. Tumour Biol. 2013;34:3637–3648. doi: 10.1007/s13277-013-0945-2. [DOI] [PubMed] [Google Scholar]
- 34.Chumsri S. Clinical utilities of aromatase inhibitors in breast cancer. Int J Womens Health. 2015;7:493–499. doi: 10.2147/IJWH.S69907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Amaral C, Borges M, Melo S, da Silva ET, Correia-da-Silva G, Teixeira N. Apoptosis and autophagy in breast cancer cells following exemestane treatment. PLoS One. 2012;7:e42398. doi: 10.1371/journal.pone.0042398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Geisler J, King N, Anker G, Ornati G, Di Salle E, Lonning PE, Dowsett M. In vivo inhibition of aromatization by exemestane, a novel irreversible aromatase inhibitor, in postmenopausal breast cancer patients. Clin Cancer Res. 1998;4:2089–2093. [PubMed] [Google Scholar]
- 37.Geisler J, Haynes B, Anker G, Dowsett M, Lonning PE. Influence of letrozole and anastrozole on total body aromatization and plasma estrogen levels in postmenopausal breast cancer patients evaluated in a randomized, cross-over study. J Clin Oncol. 2002;20:751–757. doi: 10.1200/JCO.2002.20.3.751. [DOI] [PubMed] [Google Scholar]
- 38.Zhu J, Lu X, Hua KQ, Sun H, Yu YH, Feng YJ. Oestrogen receptor alpha mediates 17beta-estradiol enhancement of ovarian cancer cell motility through up-regulation of survivin expression. Arch Gynecol Obstet. 2012;286:729–737. doi: 10.1007/s00404-012-2368-5. [DOI] [PubMed] [Google Scholar]
- 39.Yu X, Zhang X, Dhakal IB, Beggs M, Kadlubar S, Luo D. Induction of cell proliferation and survival genes by estradiol-repressed microRNAs in breast cancer cells. BMC Cancer. 2012;12:29. doi: 10.1186/1471-2407-12-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Javle MM, Tan D, Yu J, LeVea CM, Li F, Kuvshinoff BW, Gibbs JF. Nuclear survivin expression predicts poor outcome in cholangiocarcinoma. Hepatogastroenterology. 2004;51:1653–1657. [PubMed] [Google Scholar]
- 41.Chang Q, Liu ZR, Wang DY, Kumar M, Chen YB, Qin RY. Survivin expression induced by doxorubicin in cholangiocarcinoma. World J Gastroenterol. 2004;10:415–418. doi: 10.3748/wjg.v10.i3.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Effects of siCYP19A1 and 17β-estradiol on KKU-213 cell migratory activity. (a) Effect of siCYP19A1 on the cell migration expressed as a percentage of migrated cells (mean ± SD) at 18 h post migration and (b) hematoxylin staining of migrated cells (as shown in red-purple color) at 18 h post migration (PNG 134 kb)