Abstract
The microRNAs (miRNAs) have been suggested as a tumor suppressor in recent years. miR-15b was reported to exert an anti-oncogenic role in the proliferation, migration, and invasion of diverse tumor cells. However, the mechanisms underlying miR-15b-mediated biology of glioblastoma are still unclear. In the present study, the expression of miR-15b was down-regulated in glioblastoma tumor tissues and U87 and U251 cells, but insulin-like growth factor receptor 1 (IGF1R) expression became up-regulated in these tumor tissues and cells (all p < 0.001). Furthermore, IGF1R expression was inversely associated with miR-15b expression. Notably, patients with lower miR-15b expression have a much shorter survival period compared with high expression (log-rank test p = 0.045). In vitro data demonstrated that miR-15b mimics inhibited the proliferation, cell cycle arrest, and invasion of U87 and U251 cells. Besides, we validated IGF1R as a direct target of miR-15b using dual luciferase assays, and IGF1R plasmids partially abrogated miR-15b mimics inhibited cell proliferation. In vivo, miR-15b mimics indeed repressed cell proliferation in mouse xenograft model. In conclusion, our study demonstrated that miR-15b inhibits the progression of glioblastoma cells through targeting IGF1R, and miR-15b can be recommended as a tumor suppressor in the progression of glioblastoma.
Keywords: miR-15b, IGF1R, Glioblastoma
Introduction
Glioblastoma acts as a common one of primary cerebral gliomas in the central neural system, and the 5-year overall survival rate is still unsatisfying which is less than 3% [1–3]. Glioblastoma is featured as the malignant infiltration, invasiveness, and growth [4–8]. In spite of the utility of chemotherapy, radiotherapy, and operation, the overall survival rate of patients with glioblastoma is not satisfying, which is defined into less than 2 years. So, it is essential to hunt for some novel effective therapy to treat glioblastoma patients.
MicroRNAs (miRNAs) act as a kind of small and single-stranded non-coding RNAs, which can bind to the complementary parts in the 3′-non-coding translation regions (3′-UTR) of target genes to mediate the repression of translation or degradation of miRNA [9–11]. Emerging reports revealed that miRNAs play a crucial role in cell growth, metabolism, differentiation, invasiveness, and motility [12–15]. It should be noted that miRNAs could act as tumor suppressors or oncogenes. Altered miRNA expression is related to many tumor types including gastric cancer, liver cancer, breast cancer, and bladder cancer [16]. The miR-15 family includes six highly conserved members, miR-15a, miR-15b, miR-16-1, miR-16-2, miR-195, and miR-497, which are clustered on three different chromosomes. The miR-15a cluster was originally reported as the target of 13q14 deletions or down-regulation in chronic lymphocytic leukemia [17, 18]. Specifically, miR-15a and miR-16 directly regulate insulin-like growth factor receptor 1 (IGF1R), which is an anti-apoptotic oncogene, and hence act as tumor suppressors by inducing apoptosis.
In the present study, our team identified that the expression of miR-15b was significantly down-regulated in both glioblastoma tissues and cells (U87 and U251). Furthermore, low miR-15b expression was related to short survival time in glioblastoma patients. In vitro assays further demonstrated that miR-15b overexpression affected cell proliferation and invasiveness by inhibition of the expression of IGF1R in the glioblastoma.
Methods and Materials
Ethics Statement
The use of all tissue specimens was approved by the Hospital Ethics Committee of Weifang Yidu Central Hospital. The entire participant provides their written informed consent to participate in the study.
Patient Samples and Cell Lines
Human glioblastoma tissues and non-tumor tissues (more than 3 cm away from the cancer tissues) were derived from Weifang Yidu Central Hospital. All patients did not receive radiotherapy or chemotherapy prior to the operation. These tissues were kept in liquid nitrogen for use. In addition, human glioblastoma cell lines including U87, U373, U251, and A172 and normal human astrocytes (NHAs) were obtained from ATCC, and then cells were cultured in DMEM medium supplemented with fetal bovine serum (FBS) in a humidified atmosphere of 5% CO2 at 37 °C. Before our study, informed consent was obtained from these glioblastoma patients, and this clinical item was approved by the ethical commitment of Weifang Yidu Central Hospital, and our study complied with the Declaration of Helsinki.
Cell Transfection
miR-15b mimics (sense: 5′-UAGCAGCACAUCAUGGUUUACA-3′, antisense: 5′-UAAACAUGAUGUGCUGCUGUU-3′), control miRNAs (10 nmol/L), IGF1R plasmids, and control vector were all obtained from Ambion (Ambion, USA). Then, cell transfection was carried out using Lipofectamine 2000 (Life Technologies, USA) according to manufacturer’s instructions.
Quantitative Real-Time PCR Assay
Total RNA was extracted from cells, and complementary DNA (cDNA) was synthesized from total RNA using the High-Capacity cDNA Reverse Transcription Kit from Thermo Fisher. Relative gene expression was determined by SYBR Green (Bio-Rad) incorporation using a Bio-Rad MyCycler as described previously. MicroRNA was extracted from cells using PureLink miRNA Isolation Kit (Thermo Fisher), and cDNA was synthesized by using miScript II RT Kit (Qiagen). Relative miRNA expression was determined by miScript SYBR Green PCR kit (Qiagen). In the present study, all primers are listed as follows: IGF1R, 5′-GGAGGCTGAATACCGCAAAGTC-3 (forward), 5′-AAAGACGAAGTTGGAGGCGCT-3′ (reverse); GAPDH, 5′-GGTGAAGGTCGGAGTCAACG-3′ (forward), 5′-CAAAGTTGTCATGGATGHACC-3′ (reverse); miR-15b, 5′-ATGAACTTTCTCTGTCTTGG-3′ (forward), 5′-TCACCG CCTCGGCTTGTCACA-3′ (reverse). The PCR steps were 95 °C for 5 min, followed by 40 cycles of denaturation at 95 °C for 15 s and annealing/elongation step at 60 °C for 30 s. The relative expression was analyzed by the 2−ΔΔCq method.
Bioinformatics Analysis
Potential miRNA targets were predicted and analyzed using three publicly available algorithms: PicTar, TargetScan, and miRanda. “Human” was selected as the species, and “miR-15b” was entered. The number of false-positive results was decreased by accepting only putative target genes that were predicted by at least two programs.
Dual Luciferase Activity Assay
As for dual luciferase activity assay, the 3′-UTR target site could be amplified using the PCR system, and then the luciferase reporter constructs were also amplified by PCR, which is the IGF1R 3′-UTR and carried a putative miR-15b-binding site. Cells were transfected with the reporter constructs and then transfected with miR-15b or control miRNAs using Lipofectamine 2000 (Life Technologies, USA). Reporter assays were carried out using the dual-luciferase assay system (Promega) according to the manufacturer’s instructions.
Western Blot Analysis
Protein was extracted with 1× RIPA lysis buffer (Upstate Biotechnology, Charlottesville, VA, USA), 1 mM PMSF (Sigma, St. Louis, MO, USA), and a protease inhibitor cocktail (Sigma) from cultured cells and tissues. The protein content was evaluated using the DC Protein Assay (Bio-Rad Laboratories, Munich, Germany) and using bovine serum albumin (BSA) as the standard. Equal amounts of total protein were boiled and separated on NuPAGE® 4–12% Bis-Tris Gel (Life Technologies, Carlsbad, CA, USA) and then transferred onto a PVDF membrane (Bio-Rad). The membranes were blocked by incubating in Tris-buffered saline and Tween-20 (TBST) containing 5% skim dry milk for 2 h before incubation with monoclonal antibody IGF1R at the dilution of 1:1000 and mouse anti-human GPADH monoclonal antibody (Cell Signaling Technology) at the dilution of 1:1000 at 4 °C overnight. Following washing, the membranes were subsequently probed with respective secondary antibodies (horseradish peroxidase-conjugated goat anti-rabbit or anti-mouse IgG (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) diluted to 1:10,000 for 1 h at room temperature. The protein band signals were detected by using an Amersham™ ECL™ Prime Western blotting detection reagent (GE Healthcare, UK) after rinsing membranes with TBST three times according to the manufacturer’s instructions. The strips were finally scanned with ChemiDoc™ XRS+ imaging System (Bio-Rad Laboratories). Quantification of band intensities was conducted via ImageJ software (National Institute of Health, Bethesda, MD, USA).
Cell Proliferation and Invasion Assays
CCK-8 assay (Kumamoto, Japan) was used to detect the cell growth status according to the manufacturer’s instruction. Cell proliferation rate was detected at 0, 24, 48, and 72 h following the treatment. As for cell invasion assay, cell invasion was determined using 24-well transwell chambers with 8-μm pore size polycarbonate membranes (Corning Incorporated, Corning, NY, USA). Transfected cells (2 × 105) were seeded on the top side of the membrane pre-coated with Matrigel (BD, Franklin Lakes, NJ, USA) in DMEM without serum. The lower chambers were filled with DMEM containing 10% FBS as a chemoattractant. After being incubated at 24 h, the non-invasive cells on the top side of the membrane were removed with a cotton swab, and invaded cells on the lower membrane surface were fixed in 20% methanol and then stained with 0.1% crystal violet. Invasion was quantified by counting cells in five randomly selected fields of view in each well under the invert microscope (Olympus, Tokyo, Japan).
Cell Cycle Assay
Cells were washed with PBS and fixed with 70% ethanol for at least 1 h. After extensive washing, the cells were suspended in Hank’s balanced salt solution (HBSS) containing 50 μg/mL PI and 50 μg/mL RNase A and incubated for 1 h at room temperature and analyzed by FACScan (Becton Dickinson, USA). Cell cycle analysis was analyzed by ModFit software. Experiments were performed in triplicate. Results were presented as percentage of cell in a particular phase.
In Vivo Tumorigenesis Assay
The experiments involving animals were performed in accordance with the institutional guidelines for animal care and were approved by the University Committee for the Use and Care of Animals. Male BALB/c nude mice (4 weeks old and weighing about 20 g) were purchased from the Shanghai Laboratory Animal Center (Shanghai, China). miR-15b-overexpressing or control cells (5 × 106 per mouse, 3 mice per group) were subcutaneously injected into the right flanks of mice. Tumor size was measured every week using a caliper. At 28 days after cell injection, animals were sacrificed and xenograft tumors were excised and weighted. We detected the tumor length, width, and weight every 3 days. All mice in this study were euthanized by way of cervical traction at 25 days following tumor cell inoculation.
Statistical Analysis
Data in this study is expressed as the mean ± standard deviation (SD). The differences between groups were calculated using Student t test or one-way ANOVA. Survival analysis was assessed using the Kaplan-Meier curve. Spearman’s coefficient of correlation was analyzed to demonstrate the correlation. p < 0.05 was considered as statistically significant.
Results
The Expression Model of miR-15b and IGF1R in Glioblastoma Tissues
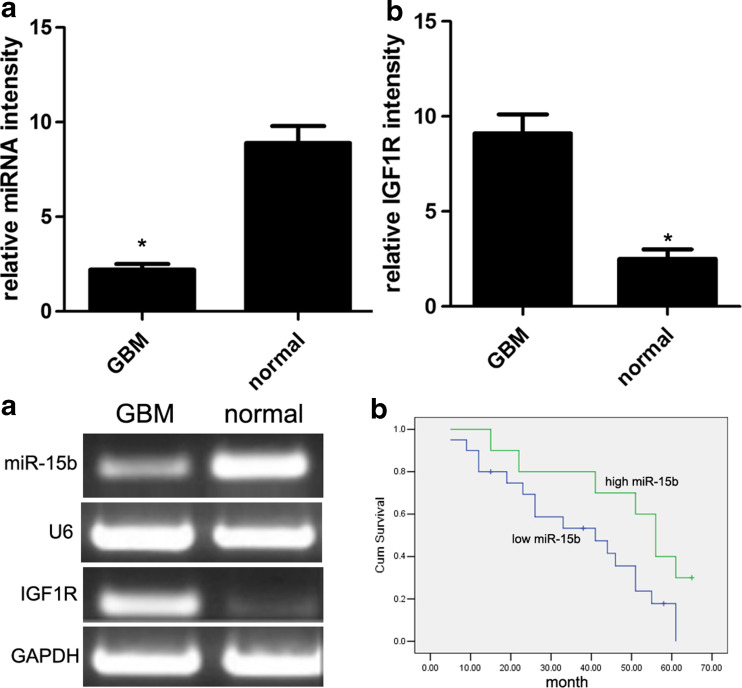
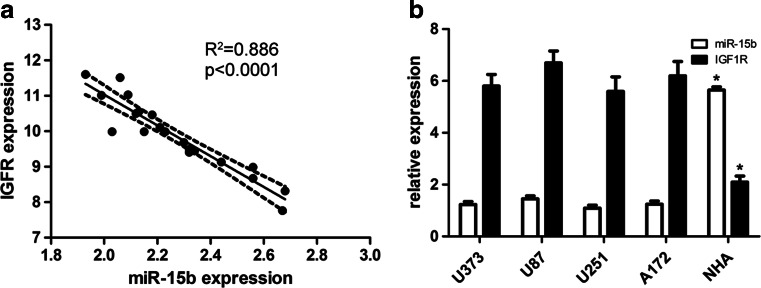
In order to elucidate the significance and role of miR-15b and IGF1R in the development of glioblastoma, the qRT-PCR assay was carried out to detect the expression model of miR-15b and IGF1R. qRT-PCR analysis showed that the expression of miR-15b was significantly decreased in tumor tissues as compared with the non-malignant counterparts (Fig. 1a), while the expression of IGF1R was significantly increased in tumor tissues as compared with the non-malignant counterparts (Fig. 1b, c). Overall, low miR-15b expression was observed in 30 cases of glioblastoma tissues, which were concomitant with high IGF1R expression. However, high miR-15b expression was detected in the rest of 10 cases of glioblastoma tissues, which were concomitant with low IGF1R expression. In addition, glioblastoma patients with low miR-15b expression had a lower overall survival rate than those with high miR-15b expression (log-rank test, p = 0.045) (Fig. 1d). Finally, we further characterized the correlation between IGF1R and miR-15b. According to Pearson’s correlation analysis, the expression of IGF1R was inversely associated with the expression of miR-15b (r 2 = −0.886; p < 0.001) (Fig. 2a).
Fig. 1.
The expression of miR-15b and IGF1R was decreased in glioblastoma tissues. The expression of miR-15b (a) and IGF1R (b) in 40 cases of glioblastoma tissues and normal tissues was detected using qRT-PCR assay (c). The expression of miR-15b and IGF1R in 40 cases of glioblastoma tissues and normal tissues was detected using RT-PCR assay. The expression of miR-15b in glioblastoma tumor tissues was significantly lower than that in paired normal tissues. d The overall survival rate of glioblastoma patients with low miR-15b expression was much lower than that of glioblastoma patients with high miR-15b expression. *p < 0.001 vs. normal control
Fig. 2.
The expression of miR-15b and IGF1R was decreased in glioblastoma cell lines. a According to Pearson’s correlation analysis, the expression of IGF1R was inversely associated with the expression of miR-15b (r 2 = −0.886; p < 0.001). b The expression of miR-15b and IGF1R in glioblastoma cell lines including U87, U373, U251, and A172, and NHA cells was detected using qRT-PCR. *p < 0.001 vs normal NHA cells
The Expression Model of miR-15b and IGF1R in Glioblastoma Tissues and Cells
Next, we further detected the expression level of miR-15b and IGF1R in a panel of glioblastoma cell lines and found that the expression of miR-15b was also decreased, but the expression of IGF1R was increased in a panel of glioblastoma cell lines, including U87, U373, U251, and A172 cells as compared to that in NHA cells (Fig. 2b).
miR-15b Affects Glioblastoma Cell Proliferation and Cell Cycle
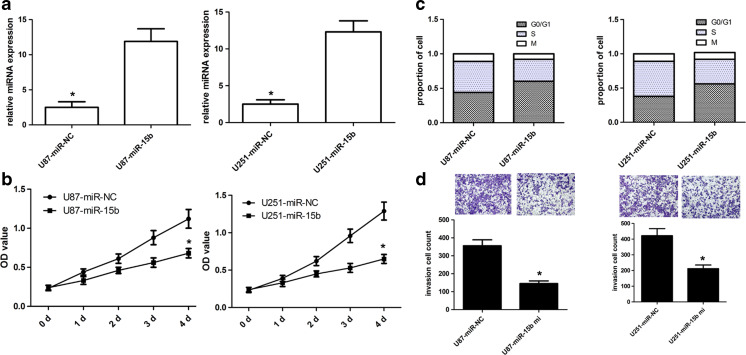
To elucidate the role of miR-15b in glioblastoma cell proliferation and cell cycle, miR-15b mimic or control miRNAs were transfected into U87 and U251 cells, and then the transfection efficiency of miR-15b was confirmed using qRT-PCR analysis (Fig. 3a). We found that ectopic expression of miR-15b affected the cell proliferation in U87 and U251 cells (Fig. 3b). Subsequently, we used flow cytometry to determine whether miR-15b was implicated in the cell cycle regulation of glioblastoma. miR-15b mimics in U87 and U251 cells dramatically increased the cell population at G0/G1 phase and decreased the cell population at S and G2/M phases (Fig. 3c). Collectively, these findings showed that miR-15b mimics could induce G0/G1 phase cell cycle arrest in glioblastoma cells.
Fig. 3.
miR-15b affected glioblastoma cell proliferation, cell cycle, and invasion. a The expression level of miR-15b in U87 and U251 cells was detected using qRT-PCR assay. b The cell proliferation capacity of U87 and U251 cells was detected using CCK-8 assay. c The ectopic miR-15b expression induced cell cycle arrest of U87 and U251 cells. d The ectopic miR-15b expression repressed cell invasion of U87 and U251 cells. *p < 0.001 vs control
miR-15b Affected Glioblastoma Cell Invasion
Because the miR-15b mimics were closely related to cell proliferation and cell cycle in human glioblastoma, we further explored the effect of miR-15b on cell invasion in U87 and U251 cells by transwell invasion assay. Our findings showed that ectopic miR-15b expression affected cell invasion in U87 and U251 cells (Fig. 3d).
IGF1R Was a Direct Target of miR-15b
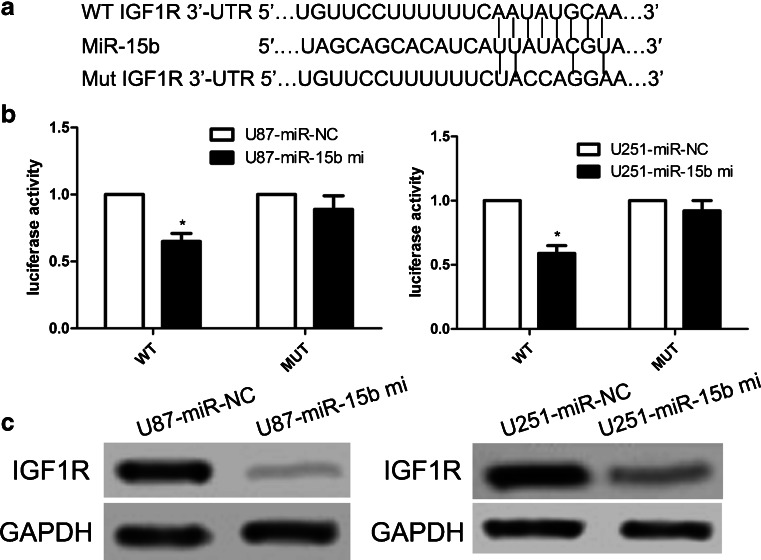
To figure out the molecular mechanism underlying cell proliferation and invasion inhibited by miR-15b, potential targets of miR-15b were predicted utilizing bioinformatics tools including miRanda, PicTar, and TargetScan [19]. Hundreds of different targets were predicted; of these genes, IGF1R was selected as a potential target of miR-15b since IGF1R is regarded as a protooncogene in various cancers. To further confirm whether IGF1R responds to miR-15b through direct 3′-UTR interaction in glioblastoma, we subcloned the IGF1R 3′-UTR wild type of the miR-15b-binding site into a luciferase reporter vector (Fig. 4a). Effect of miR-15b on the luciferase activity was detected by dual-luciferase assay. We found that overexpression of miR-15b reduced the luciferase activity of the wild-type 3′-UTR of IGF1R by 25% when compared with the mutated 3′-UTR of the IGF1R group (Fig. 4b). Importantly, ectopic expression of IGF1R also repressed the protein expression of IGF1R in U87 and U251 cells (Fig. 4c).
Fig. 4.
IGF1R is a direct target of miR-15b. a The WT and Mut of the 3′-UTR of IGF1R messenger RNA (mRNA) contains the binding sequences of miR-15b. b The wild-type and mutated 3′-UTR of IGF1R mRNA was transfected into a luciferase reporter vector, and then we detected the luciferase activity. Firefly luciferase values were normalized to Renilla luciferase activity and used as relative luciferase activity. c The protein expression of IGF1R in U87 and U251 cells was measured by western blot. *p < 0.001 vs control
Overexpression of IGF1R Partially Attenuated miR-15b-Inhibited Cell Growth
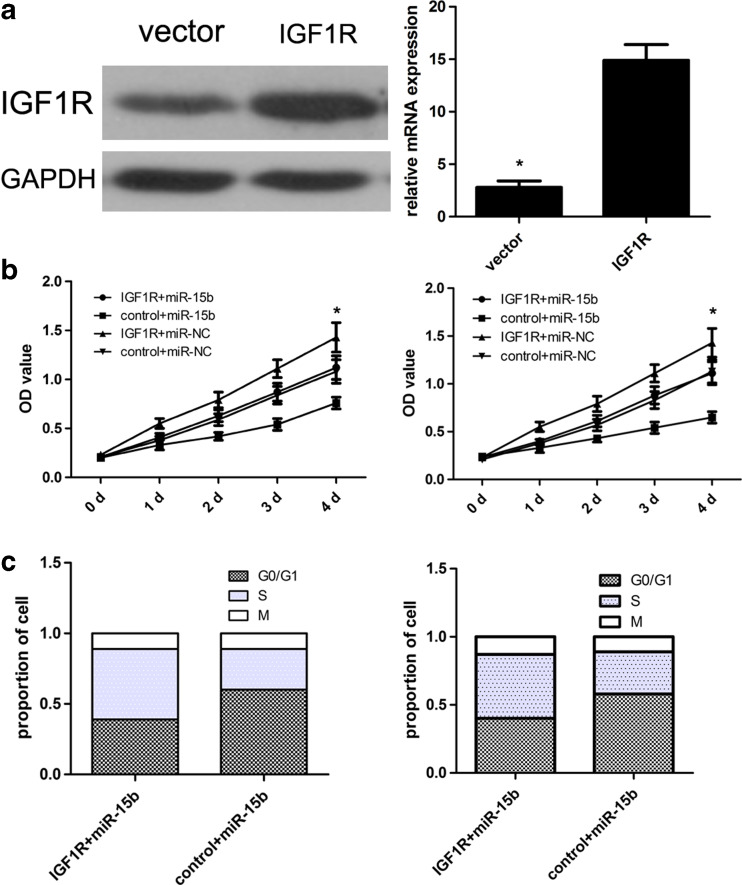
In this work, the transfection efficiency of the IGF1R vector was detected and confirmed using western blot analysis (Fig. 5a). Subsequently, CCK8 assay was conducted and results identified that ectopic IGF1R expression partially attenuated miR-15b-inhibited cell growth in U87 and U251 cells (Fig. 5b). At the same time, the miR-15b-inhibited cell cycle arrest was also partially restored due to transfection of the IGF1R vector (Fig. 5c).
Fig. 5.
Overexpression of IGF1R partially attenuated miR-15b-inhibited cell growth. a The protein expression level of IGF1R in U87 cells was detected using western blot. Then, the mRNA expression level of IGF1R in U87 cells was detected using qRT-PCR assay. b The U87 and U251 cell growth was detected using CCK-8 assay. Cells with miR-15b or miR-NC were transfected with IGF1R plasmids or vector control, respectively. c The cell cycle was detected using flow cytometry. *p < 0.001 vs control
miR-15b Affected U87-Engrafted Tumor Growth
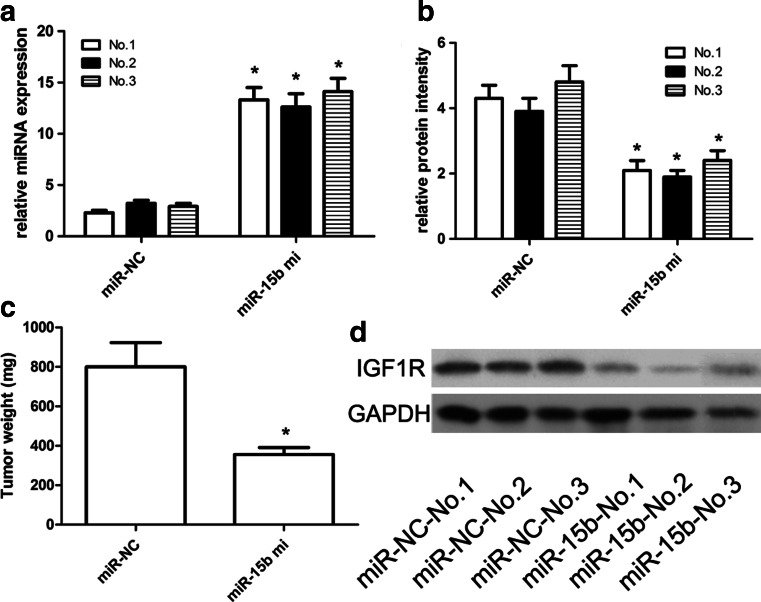
In the present study, the experimental animals receiving intra-tumor injection of miR-15b survived very well compared with control, and no toxic effect was found until xenograft tumors were excised and weighted. We demonstrated that miR-15b mimics could enhance the ectopic expression of miR-15b in the U87-engrafted tumors (Fig. 6a). According to in vivo data, miR-15b mimics could suppress the U87-engrafted tumor growth when compared with control oligonucleotide-treated tumors. Furthermore, the weight of miR-15b-treated U87-engrafted tumors was obviously lower than that of the control-treated U87-engrafted tumors. Administration of miR-15b mimics affected the U87-engrafted tumor growth when compared with the control oligonucleotide-treated tumors (Fig. 6c). At the same time, it should be noted that the expression of IGF1R was much lower in miR-15b-treated tumors than that in the control-treated U87-engrafted tumors (Fig. 6b, d).
Fig. 6.
miR-15b affected the U87-engrafted tumor growth. U87 cells stably transfected with miR-15b-expressing plasmids or empty vector were subcutaneously injected into nude mice (three nude mice for the experiment group, and three nude mice for the control group), and tumor volumes were measured every week. Tumors were resected and weighed at 4 weeks after cell injection. Each injection contained 40 ng of miR-15b mimic in 10 μL saline solution. Mice were stitched and reanimated under warm light. Control mice were injected with mimic control. a The expression of miR-15b was detected using qRT-PCR assay. b The expression of IGF1R mRNA was detected using qRT-PCR assay. c We identified that miR-15b mimics could repress the U87-engrafted tumor growth when compared with control-induced U87-engrafted tumors. The weight of miR-15b-treated tumors was obviously lower compared with control-treated U87-engrafted solid tumor mass. d The expression of IGF1R protein detected using western blot. *p < 0.001 vs control
Discussions
Recently, some studies reported that miR-15b plays an essential role in the development of cancers [16]. As reported, qRT-PCR and ELISA analysis demonstrated that miR-15b expression was significantly lower and IL-15 expression was significantly higher in all EMG, LMG, and thymoma cases compared to healthy controls. miR-15b overexpression could inhibit IL-15 expression in EAMG mice [17]. To date, through searching in bioinformatics databases, a highly conserved consequential pairing between target genes and miR-15b can be identified [17]. Subsequent dual luciferase assay further verified this match. In this work, our team identified that the expression of miR-15b was decreased in glioblastoma tissues in comparison with paired non-tumor tissues. Thirty cases of glioblastoma tissues had lower level of miR-15b when compared with paired non-tumor tissues. The overall survival rate of glioblastoma patients with low miR-15b expression was lower than in those with high miR-15b expression. In addition, the expression of miR-15b was also decreased in all glioblastoma cell lines. These findings were consistent with other studies of miR-15b [18].
Based on the function analysis, we found that the overexpression of miR-15b affected glioblastoma cell growth, cell cycle, and invasiveness in U87 and U251 cells. Consistent with our results, Zhang et al. reported that miR-15b and miR-152 attenuated the expression of NRP-2 and MMP-3 protein by binding to NRP-2 and MMP-3 transcripts, which further significantly decreased glioma cell invasiveness, and in vitro tube formation assay data showed that miR-15b reduced tube formation [18]. This preliminary study indicated that miR-15b and miR-152 deactivated the MEK-ERK pathway via NRP-2 and MMP-3 in glioma cells [18]. Mechanically, we demonstrated the IGF1R gene as a direct target of miR-15b in the development of glioblastoma. At the same time, the overexpression of the IGF1R gene partially attenuated the miR-15b-inhibited cell proliferation. Based on an in vivo study, the expression status of the IGF1R protein in glioblastoma tumor tissues was also significantly higher than that in paired non-tumor tissues. Besides, the expression of IGF1R was inversely associated with the expression of miR-15b. Notably, the overexpression of miR-15b repressed the development and growth of glioblastoma in a U87 xenograft nude mouse model. These results indicated that miR-15b played a crucial role in affecting the progression of glioblastoma probably by targeting IGF1R.
The IGF1R is a transmembrane receptor tyrosine kinase, closely related to the insulin receptor (InsR) that forms homodimers or heterodimerizes with InsR to recognize its ligands, IGF-1 and IGF-2 [19]. Upon ligand binding, IGF1R activates multiple downstream signaling cascades, the two most prominent being PI3K/AKT and RAS/RAF/MEK/ERK [20]. PI3K/AKT enhances cellular metabolism and protein synthesis via mTOR and enhances survival via BAD/Bcl2, p53, NF-κB, and FOXOs, whereas RAS/RAF/MEK/ERK activation generally results in increased cellular proliferation [21]. In our study, dual luciferase activity assay showed that overexpression of miR-15b repressed the luciferase activity of reporter vectors. Ectopic miR-15b expression affected the expression of IGF1R miRNA in both U87 and U251 cells. Most essentially, we demonstrated that the overexpression of IGF1R partially attenuated the miR-15b-inhibited cell growth. These results indicated that miR-15b inhibited glioblastoma cell growth, cell cycle progression, and invasion by targeting the expression of IGF1R.
Notably, in other studies, the expression of miR-15b was identified to be up-regulated in pancreatic cancer cell lines and tissues. Zhang et al. reported that overexpression of miR-15b enhanced the pancreatic cancer cell invasion, growth, and EMT via regulating the expression of SMURF2 [22]. These reports represent the diversity of miRNAs. miRNAs play a crucial role in the regulation of genes by binding to their potential target mRNAs and then affecting the relevant protein expressions [23–25].
In conclusion, this study showed that the expression of miR-15b was decreased in both glioblastoma tissues and cells. Moreover, low miR-15b expression was associated with short survival rate in glioblastoma patients. We also identified that miR-15b suppressed cell proliferation and invasion by targeting the expression of IGF1R in glioblastoma cells. Thus, we thought that miR-15b could function as a tumor suppressor in clinical treatment of glioblastoma patients.
Acknowledgements
This work was supported by some other members in our lab.
Compliance with Ethical Standards
The use of all tissue specimens was approved by the Hospital Ethics Committee of Weifang Yidu Central Hospital. The entire participant provides their written informed consent to participate in the study.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Zhao F, Li M, Kong L, Zhang G, Yu J. Delineation of radiation therapy target volumes for patients with postoperative glioblastoma: a review. Onco Targets Ther. 2016;9:3197–3204. doi: 10.2147/OTT.S104241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Neagu MR, Reardon DA. An update on the role of immunotherapy and vaccine strategies for primary brain tumors. Curr Treat Options in Oncol. 2015;16(11):54. doi: 10.1007/s11864-015-0371-3. [DOI] [PubMed] [Google Scholar]
- 3.Bradshaw A, Wickremsekera A, Tan ST, Peng L, Davis PF, Itinteang T. Cancer stem cell hierarchy in glioblastoma multiforme. Front Surg. 2016;3:21. doi: 10.3389/fsurg.2016.00021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Smith AW, Mehta MP, Wernicke AG. Neural stem cells, the subventricular zone and radiotherapy: implications for treating glioblastoma. J Neuro-Oncol. 2016;128(2):207–216. doi: 10.1007/s11060-016-2123-z. [DOI] [PubMed] [Google Scholar]
- 5.Binder DC, Davis AA, Wainwright DA. Immunotherapy for cancer in the central nervous system: current and future directions. Oncoimmunology. 2015;5(2):e1082027. doi: 10.1080/2162402X.2015.1082027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Katakowski M, Chopp M. Exosomes as tools to suppress primary brain tumor. Cell Mol Neurobiol. 2016;36(3):343–352. doi: 10.1007/s10571-015-0280-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Krishna Priya S, Nagare RP, Sneha VS, Sidhanth C, Bindhya S, Manasa P, et al. Tumour angiogenesis—origin of blood vessels. Int J Cancer. 2016;139(4):729–735. doi: 10.1002/ijc.30067. [DOI] [PubMed] [Google Scholar]
- 8.Khosla D. Concurrent therapy to enhance radiotherapeutic outcomes in glioblastoma. Ann Transl Med. 2016;4(3):54. doi: 10.3978/j.issn.2305-5839.2016.01.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li X, Nie J, Mei Q, Han WD. MicroRNAs: novel immunotherapeutic targets in colorectal carcinoma. World J Gastroenterol. 2016;22(23):5317–5331. doi: 10.3748/wjg.v22.i23.5317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Diab M, Muqbil I, Mohammad RM, Azmi AS, Philip PA. The role of microRNAs in the diagnosis and treatment of pancreatic adenocarcinoma. J Clin Med. 2016;5(6):59. doi: 10.3390/jcm5060059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tsai MM, Wang CS, Tsai CY, Huang HW, Chi HC, Lin YH et al (2016) Potential diagnostic, prognostic and therapeutic targets of microRNAs in human gastric cancer. Int J Mol Sci 17(6). doi:10.3390/ijms17060945 [DOI] [PMC free article] [PubMed]
- 12.Kushlinskii NE, Fridman MV, Braga EA. Molecular mechanisms and microRNAs in osteosarcoma pathogenesis. Biochemistry (Mosc) 2016;81(4):315–328. doi: 10.1134/S0006297916040027. [DOI] [PubMed] [Google Scholar]
- 13.Dallaire A, Simard MJ. The implication of microRNAs and endo-siRNAs in animal germline and early development. Dev Biol. 2016;416(16):30098–30097. doi: 10.1016/j.ydbio.2016.06.007. [DOI] [PubMed] [Google Scholar]
- 14.Lu YC, Chang JT, Chan EC, Chao YK, Yeh TS, Chen JS, et al. miR-196, an emerging cancer biomarker for digestive tract cancers. J Cancer. 2016;7(6):650–655. doi: 10.7150/jca.13460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen WX, Ren LH, Shi RH. Implication of miRNAs for inflammatory bowel disease treatment: systematic review. World J Gastrointest Pathophysiol. 2014;5(2):63–70. doi: 10.4291/wjgp.v5.i2.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.MacLean JA, 2nd, King ML, Okuda H, Hayashi K. WNT7A regulation by miR-15b in ovarian cancer. PLoS One. 2016;11(5):e0156109. doi: 10.1371/journal.pone.0156109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shi L, Liu T, Zhang M, Guo Y, Song C, Song D, et al. miR-15b is downregulated in myasthenia gravis patients and directly regulates the expression of interleukin-15 (IL-15) in experimental myasthenia gravis mice. Med Sci Monit. 2015;21:1774–1780. doi: 10.12659/MSM.895272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zheng X, Chopp M, Lu Y, Buller B, Jiang F. MiR-15b and miR-152 reduce glioma cell invasion and angiogenesis via NRP-2 and MMP-3. Cancer Lett. 2013;329(2):146–154. doi: 10.1016/j.canlet.2012.10.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Solarek W, Czarnecka AM, Escudier B, Bielecka ZF, Lian F, Szczylik C. Insulin and IGFs in renal cancer risk and progression. Endocr Relat Cancer. 2015;22(5):R253–R264. doi: 10.1530/ERC-15-0135. [DOI] [PubMed] [Google Scholar]
- 20.Pollak M. The insulin and insulin-like growth factor receptor family in neoplasia: an update. Nat Rev Cancer. 2012;12(3):159–169. doi: 10.1038/nrc3215. [DOI] [PubMed] [Google Scholar]
- 21.Myal Y, Shiu RPC, Bhaumick B, Bala M. Receptor binding and growth-promoting activity of insulin-like growth factors in human breast cancer cells (T-47D) in culture. Cancer Res. 1984;44(12 Part 1):5486–5490. [PubMed] [Google Scholar]
- 22.Zhang WL, Zhang JH, Wu XZ, Yan T, Lv W. miR-15b promotes epithelial-mesenchymal transition by inhibiting SMURF2 in pancreatic cancer. Int J Oncol. 2015;47(3):1043–1053. doi: 10.3892/ijo.2015.3076. [DOI] [PubMed] [Google Scholar]
- 23.Li M, Tian L, Wang L, Yao H, Zhang J, Lu J, et al. Down-regulation of miR-129-5p inhibits growth and induces apoptosis in laryngeal squamous cell carcinoma by targeting APC. PLoS One. 2013;8:e77829. doi: 10.1371/journal.pone.0077829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li D, Xie X, Wang J, Bian Y, Li Q, Gao X, et al. MiR-486 regulates lactation and targets the PTEN gene in cow mammary glands. PLoS One. 2015;10:e0118284. doi: 10.1371/journal.pone.0118284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gattolliat CH, Uguen A, Pesson M, Trillet K, Simon B, Doucet L, et al. MicroRNA and targeted mRNA expression profiling analysis in human colorectal adenomas and adenocarcinomas. Eur J Cancer. 2015;51(3):409–420. doi: 10.1016/j.ejca.2014.12.007. [DOI] [PubMed] [Google Scholar]