Abstract
Non-alcoholic fatty liver disease (NAFLD) represents a major public health concern and is associated with a substantial global burden of liver-related and cardiovascular-related morbidity and mortality. High total energy intake coupled with unhealthy consumption of ultra-processed foods and saturated fats have long been regarded as major dietary drivers of NAFLD. However, there is an accumulating body of evidence demonstrating that the timing of energy intake across a the day is also an important determinant of individual risk for NAFLD and associated metabolic conditions. This review summarises the available observational and epidemiological data describing associations between eating patterns and metabolic disease, including the negative effects of irregular meal patterns, skipping breakfast and night-time eating on liver health. We suggest that that these harmful behaviours deserve greater consideration in the risk stratification and management of patients with NAFLD particularly in a 24-hour society with continuous availability of food and with up to 20% of the population now engaged in shiftwork with mistimed eating patterns. We also draw on studies reporting the liver-specific impact of Ramadan, which represents a unique real-world opportunity to explore the physiological impact of fasting. By highlighting data from preclinical and pilot human studies, we present a further biological rationale for manipulating timing of energy intake to improve metabolic health and discuss how this may be mediated through restoration of natural circadian rhythms. Lastly, we comprehensively review the landscape of human trials of intermittent fasting and time-restricted eating in metabolic disease and offer a look to the future about how these dietary strategies may benefit patients with NAFLD and non-alcoholic steatohepatitis.
Keywords: NONALCOHOLIC STEATOHEPATITIS, ENERGY METABOLISM, LIVER METABOLISM, DIABETES MELLITUS, OBESITY
Key messages
Observational data show that irregular meal patterns, skipping breakfast and night-time eating are associated with an increased risk of non-alcoholic fatty liver disease (NAFLD) and related metabolic conditions.
Distribution of total daily energy intake away from the end of the day may improve metabolic health.
Complete fasting between dawn and dusk during Ramadan is associated with weight loss, reduced insulin resistance and improved liver biochemistry.
Intermittent fasting protocols can lead to>5% wt loss, reduced hepatic steatosis and improved lipid profiles in patients with NAFLD and appear superior to standard dietary and weight loss advice.
Compared with continuous energy restriction, time-restricted eating (TRE) leads to similar reductions in body weight and intra-hepatic triglyceride but may be better tolerated and is associated with greater improvements in glycaemic control.
TRE can reprogramme circadian outputs across multiple tissues leading to synchronisation of behaviour and physiology across a 24-hour cycle.
TRE has emerged as a promising strategy to mitigate the adverse metabolic phenotype associated with circadian misalignment induced by night-shift working.
As the field continues to advance, it is likely that an increasing number of society consensus statements acknowledge the value of modifying timing of calorie intake as a potential strategy for the prevention and treatment of NAFLD and non-alcoholic steatohepatitis.
Introduction
Non-alcoholic fatty liver disease (NAFLD) represents a major public health concern affecting approximately one-quarter of the global adult population and is closely associated with the epidemic of type 2 diabetes (T2D) and obesity.1 While the majority of individuals living with NAFLD have isolated steatosis (non-alcoholic fatty liver), a proportion will develop non-alcoholic steatohepatitis (NASH), which predisposes to cirrhosis, primary liver cancer and both liver-related and cardiovascular-related mortality.2 3 From a pathophysiological perspective, NAFLD is a heterogeneous condition involving the complex interplay between immune cells, inflammatory mediators and metabolic target tissues, including adipose and skeletal muscle.4 These multiple converging pathogenic pathways have made drug discovery challenging and as yet, no therapeutic agents for NAFLD/NASH have progressed through late-phase trials and into licensing. The mainstay of treatment therefore continues to centre on lifestyle intervention and weight loss, which has traditionally been achieved through decreasing total calorie intake and modifications to dietary macronutrient composition.5 This has typically focused on limiting consumption of fructose, ultra-processed foods and saturated fats and/or prescribing a balanced Mediterranean diet, which has been shown to improve liver biochemistry and hepatic steatosis.5–7 However, there is also accumulating evidence that the timing of energy intake across the day may play an important role in determining an individual’s risk for NAFLD, including missing breakfast, irregular meal patterns and night-time eating. These behaviours are becoming increasingly important to consider in a modern 24-hour society where there is continuous availability of food, light at night, prevalent shiftwork and disrupted sleep/wake patterns all of which promote irregular eating patterns and an extended fed period.
The link between temporal eating habits and human disease is likely to be mediated through disruption to circadian rhythms. The circadian clock network is coordinated by a central clock in the suprachiasmatic nucleus (SCN) of the hypothalamus, which communicates via neuroendocrine signals with a number of peripheral clocks, including the liver. At a cellular level, all clocks are governed by the same transcriptional–translational feedback loop involving factors, including CLOCK, BMAL1, PER and CRY, leading to periodic regulation of multiple facets of metabolism, including glucose uptake, gluconeogenesis, lipogenesis and bile acid synthesis.8 9 While the central pacemaker in the SCN is predominantly influenced by the light–dark cycle, the liver clock is exquisitely sensitive to feeding patterns. Indeed, the circadian phase of the liver can be directly influenced by feeding and this is independent of both the SCN and light–dark signalling.10 11 There is now a wealth of rodent and human data linking circadian misalignment with metabolic dysfunction,12 13 including feeding exclusively during the resting phase which markedly shifts the liver clock, resulting in multiple features of the metabolic syndrome.14 15 Collectively, these findings have led to much interest in the use of fasting and time-restricted feeding/eating as a preventative and treatment measure for metabolic conditions, including NAFLD.
In this review, we summarise observational and epidemiological studies, which establish a link between timing of food intake and NAFLD. By drawing on data from preclinical and pilot human studies, we then present the biological rationale for manipulating energy timing to help improve metabolic health and discuss how this may be mediated through restoration of natural circadian rhythms. Lastly, we review the landscape of human trials exploring intermittent fasting (IF) and time-restricted eating (TRE) in metabolic disease and offer a look to the future as to how these dietary strategies may benefit patients with NAFLD and NASH.
Timing of food intake as a risk factor for NAFLD and metabolic dysfunction
Irregular eating patterns
The timing of food consumption, in addition to total caloric intake and dietary macronutrient composition, is an important determinant of an individual’s predisposition to weight gain and metabolic disease (figure 1). Erratic and irregular patterns of food consumption throughout the day have been consistently shown to be deleterious.16–18 For example, in a population-based study of 3607 Swedish adults, irregular meal schedules (eg, eating outside of breakfast, lunch and dinner) were associated with metabolic syndrome, insulin resistance and elevated serum concentrations of γ-glutamyl transferase (γGT).17 Similarly, National Health and Nutrition Examination Survey data showed that eating more than 5 times per day was positively associated with body mass index (BMI) and central adiposity.19 These findings were corroborated in a randomised trial which showed that irregular meal patterns (variable meal frequency of 3–9 meals/day) was associated with higher post-prandial insulin concentrations, elevated low-density lipoprotein (LDL) cholesterol and insulin resistance compared with regular meal consumption (6 eating events/day).20 Interestingly, despite a majority of free-living individuals believing they adopt a traditional three-meal structure to calorie intake, in reality this is rarely the case.21 22 For example, by using smartphone image capture technology to record each time food was consumed across a 3-week period in 156 healthy adults in the USA, Gill and Panda demonstrated a distinct lack of meal clustering.21 Indeed, the number of discrete eating events ranged from 4 to 15 in the bottom and top deciles, respectively. Furthermore, the median interval between meals (defined as intake lasting >15 min) was 3 hours, with 25% of all meals occurring within 1.5 hours of each other and only 25% of the meals occurring after >6 hour of fasting.21 Similar findings were replicated in an Indian cohort where more than 50% of participants spread their caloric intake across >15 hours in a day.22 These non-structured eating patterns, when entrenched at a younger age, may also predict future metabolic disease with irregular eating habits in adolescents being associated with prevalent metabolic syndrome up to 25 years later.23
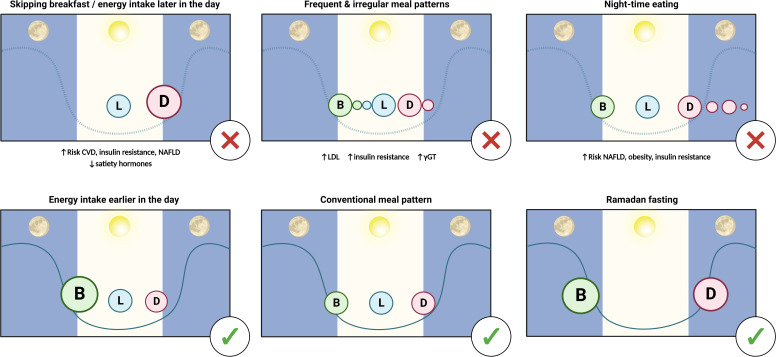
Figure 1.
Timing of food intake as a risk factor for NAFLD and metabolic dysfunction. Timing of energy intake across the day–night cycle is an important determinant of individual risk for NAFLD. Skipping breakfast, chaotic meal patterns and night-time eating are all associated with metabolic dysfunction and disruption to natural circadian rhythms (symbolised by blue line). Conversely, shifting energy intake toward the beginning of the day, standardised meal patterns and fasting during Ramadan are all associated with benefits to metabolic and liver health. γGT, γ-glutamyl transferase; B, breakfast; CVD, cardiovascular disease; D, dinner; L, lunch; LDL, low-density lipoprotein cholesterol; NAFLD, non-alcoholic fatty liver disease. Figure made with biorender.com.
Skipping breakfast
The traditional mantra that breakfast is the ‘most important meal of the day’ has been the subject of much investigation. Observational studies have shown that skipping breakfast may predispose to a range of adverse health outcomes, including obesity,24 early onset atherosclerosis,25 cardiovascular disease (CVD),26 metabolic syndrome,27 insulin resistance28 and NAFLD.29 30 In a US population-based study, skipping breakfast was also associated with a higher risk of long-term cardiovascular and cerebrovascular mortality in patients with metabolic-dysfunction associated liver disease defined by hepatic steatosis on ultrasound alongside a BMI ≥23 kg/m2, evidence of metabolic dysregulation or T2D.31 Despite this, up to 30% of western populations are known to regularly omit breakfast from their daily routine.32 Several mechanisms have been proposed to explain the potential pathogenic role of skipping breakfast. First, the satiating properties of food are known to decline over the course of the day, and, therefore, eating breakfast may serve to minimise subsequent calorie intake and protect against weight gain.33 This association appears to be macronutrient specific whereby high morning carbohydrate intake is coupled with subsequent reductions in carbohydrate intake throughout the rest of the day, and likewise with protein and fat consumption.34 These findings are likely underpinned by distinct endocrine profiles, with satiety hormones, including peptide YY (PYY) and glucagon-like peptide 1 (GLP-1) found to be elevated during the midday period in those eating compared with those skipping breakfast.35 These observations are partly tempered by results of small, mostly unblinded randomised trials, which show widely inconsistent results regarding the impact of breakfast on weight and energy balance.36 However, post-prandial glycaemic control after lunch is known to be more robust once breakfast has been consumed.37 38 Using stable isotope tracer techniques, this ‘second meal phenomenon’ appears to result from insulin secretion after breakfast suppressing plasma non-esterified fatty acids (NEFA), thereby improving skeletal muscle insulin sensitivity and facilitating greater glycogen storage after lunch.39
Alongside these metabolic pathways, there may also be a circadian component to the benefits derived from eating breakfast.40 A single daily morning meal was found to advance the circadian phase in core body temperature and heart rate by 1 hour compared with a single evening meal.41 Similarly, a second study showed that skipping breakfast for 6 days delayed diurnal rhythms in body temperature by approximately 45 min.42 In addition, breakfast feeding in infants is associated with a shift in sleep–wake cycle toward an earlier chronotype with studies suggesting that morning consumption of tryptophan, the main precursor of melatonin, may improve overall sleep quality and trigger earlier sleep-onset toward the end of the day.43 44 Taken together, this data suggest that breakfast may act as important external cue or ‘zeitgeber’ for entrainment of circadian rhythm serving to ensure healthy synchronisation of behaviour and physiology across the day–night cycle.
Distribution of energy intake across the day
Beyond the benefits of simply consuming breakfast, several lines of evidence suggest that distributing total daily energy intake away from the end of the day may further improve metabolic health. For example, a Spanish population-based cohort followed up over 3.5 years demonstrated a dose–response relationship between higher percentage energy intake at lunch and lower risk of weight gain.45 Similarly, a cross-sectional US analysis found that consuming >33% of total energy at dinner was associated with a higher likelihood of obesity.46 A second Spanish cohort also examined the relationship between energy distribution and incident metabolic dysfunction. Compared with the lowest sex-specific quartile of energy intake at dinner, the OR for developing metabolic syndrome ranged from 1.71 to 2.57 between the second and fourth quartiles, respectively.47 This was replicated in a large cohort of 1245 non-diabetic, non-obese Italian adults followed up over 6 years, in whom a high baseline calorie intake during dinner was associated with an elevated risk of going on to develop obesity and metabolic syndrome. Lastly, further analysis in a Japanese cohort also demonstrated that a large dinner or eating just before bedtime was associated with incident NAFLD defined using non-invasive scores for steatosis, even after controlling for total daily energy intake.48 49 These observations may be related to the known rhythmicity of insulin sensitivity which can be up to 33% higher in the morning compared with the evening, even in healthy individuals.50 51 As a result, the ability to appropriately store and metabolise dietary glucose and lipid intake is progressively impaired throughout the day. Furthermore, overweight individuals randomised to consume a large breakfast were also found to have greater weight loss, lower levels of ghrelin and higher daily satiety scores compared with those receiving an isocaloric large dinner.52
The adverse metabolic implications of eating later in the day are further reinforced by the damaging impact of night-time eating and snacking. These behaviours have been associated with a variety of chronic disease states, including NAFLD, obesity, metabolic syndrome and CVD.49 53 54 Significantly higher values of plasma glucose, insulin and triglycerides are recorded after a meal eaten at night compared with an identical meal eaten during the day.55 56 This metabolic phenotype is reminiscent of that observed in shift-workers and jet-lagged individuals who are forced to consume food out-of-sync with the usual pattern of day and night.57 At the extreme end of the spectrum are patients with night eating syndrome (NES), which is characterised by evening hyperphagia, nocturnal waking for food and morning anorexia.58 These patients demonstrate significant changes in the timing and amplitude of various circadian markers, including eating patterns, cortisol, ghrelin and insulin, despite retaining regular sleep–wake cycling. NES, therefore, represents a good example of how eating at night can lead to dissociation between central and peripheral timing mechanisms59 with resultant metabolic dysfunction.60 Finally, circadian preference toward eveningness (late/evening chronotype) is associated with a delay in meal timing, breakfast skipping and excessive calorie intake at night,61 which may contribute to the higher prevalence of metabolic disease and obesity observed in this group.62 63 Indeed, evening chronotype may also correlate with increased severity of NASH in obese individuals, as determined by scores incorporating liver biochemistry, homeostatic model assessment for insulin resistance (HOMA-IR), waist-to-hip ratio and circulating triglycerides.64 65
Diurnal fasting: insights from Ramadan
The annual practice of fasting during Ramadan is observed by many of the >1.6 billion Muslim population worldwide and requires complete abstinence from food and drink between dawn and dusk for an entire lunar month. While calorie restriction is not mandated, intake of food does become less frequent and exclusively nocturnal. Ramadan, therefore, constitutes a major shift away from routine eating habits and represents a unique real-world opportunity to explore the physiological impact of fasting and energy distribution. The behavioural implications of Ramadan are wide ranging and heterogeneous, with effects on sleep,66 67 exercise68 and dietary composition69 all being reported. This has led to variable results relating to weight and metabolism across both healthy and diseased populations. Nonetheless, several meta-analyses have shown that Ramadan fasting is generally associated with weight loss,69 70 reduced total fat mass70 and improvements in cardiometabolic risk factors, including lipid profiles,71 blood pressure71 and glycaemic parameters.72 73 Ramadan has also been shown to improve fasting glucose and HOMA-IR specifically in patients with NAFLD in parallel with weight loss and reductions in inflammatory cytokines (eg, interleukin 6 (IL-6) and C-reactive protein (CRP)).74 A meta-analysis of 20 studies in predominantly healthy individuals has also identified modest but significant improvements in aspartate transaminase (AST), γGT, alkaline phosphatase and bilirubin.75 Several other studies have pointed towards improved markers of inflammation and oxidative stress following Ramadan fasting; however, whether these improvements occur independently of weight loss has been difficult to decipher.76–78
The weight loss experienced during Ramadan tends to be greatest in those who are already overweight and the majority return to their baseline weight within 5 weeks after resuming standard eating habits.70 79 Moreover, there is contradictory evidence regarding the effect of Ramadan on total calorie consumption with separate studies showing reduced, unchanged and increased total daily energy intake.69 However, pooled analysis does suggest that energy intake tends to remain consistent before and during the fasting period.69 The fact that weight loss and improved metabolic health often occur despite an isocaloric diet suggests that fasting may trigger protective mechanisms separate from simple energy restriction. An additional point of discrepancy is that Ramadan leads to health advantage in spite of exclusive night-time eating, which has been shown to be detrimental in other contexts as previously discussed. This shift in eating patterns is mirrored by changes in the profiles of satiety hormones, including leptin, which has an important role in the regulation of energy metabolism and food intake. Ramadan has been associated with blunting of usual nocturnal elevations in leptin as well as a delay in peak overnight concentrations, reflecting the transition to later evening meal consumption after sunset.80 81 However, there is currently no consistent evidence that specific hormonal profiles before, during or after dietary modification can be used as a biomarker to predict future weight regain.82 In addition, the eating patterns of Ramadan are associated with profound temporal alterations in other circadian biomarkers, including melatonin, cortisol, testosterone, thyroid-stimulating hormone, prolactin and insulin, which can persist up to 1 month after fasting.83 The sleep window is also delayed and shortened by approximately 1 hour. Therefore, despite Ramadan in many ways, exemplifying an unnatural pattern of eating, there appears to be something about the act of fasting or limiting the eating window that ultimately leads to benefit. Indeed, Ramadan fasting has been associated with upregulated key regulatory proteins involved in glucose and lipid metabolism, circadian clock function and DNA repair.84 This has led to much investigation regarding the pleiotropic effects of IF and TRF.
Intermittent fasting and time-restricted eating
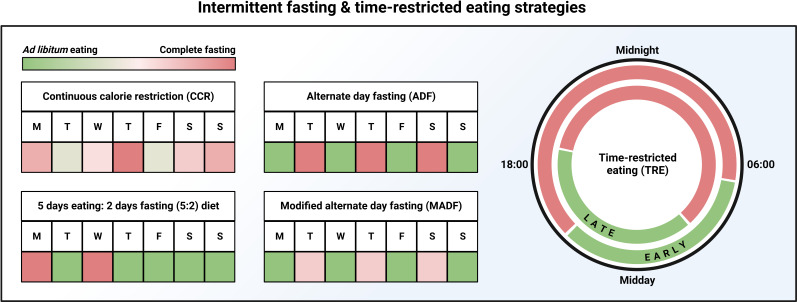
In light of observational studies linking dietary patterns with chronic disease, there has been considerable interest over the last two decades in manipulating eating schedules in order to optimise metabolic health. This has revolved around two major intervention strategies: periodic fasting or IF, where caloric intake is severely constrained for short periods and TRE (or TRF in mice), which limits the daily eating window with fasting for the remainder of the day (figure 2). The two most commonly employed IF strategies in humans comprise the so-called 5:2 diet and the alternate day fasting (ADF) regimen. In the 5:2 diet, fasting occurs on 2 non-consecutive days a week with no formal energy restrictions imposed on the remaining 5 days. ADF involves a continuous alternating pattern of eating and fasting days. Modified ADF allows for some energy consumption on fasting days, usually up to 25% of normal caloric intake, whereas traditional ADF typically includes complete fasting every other day. The protective effects of fasting are well recognised across species, including simple organisms, which have evolved to avoid age-dependent damage in energy poor environments.85 Prokaryotes, nematodes, yeasts and mice have all been shown to have an extended lifespan when exposed to fasted or nutrient-scarce conditions85 86 (figure 3). The molecular mechanisms governing this response in mammals are highly conserved with coordinated reprogramming of numerous metabolic and stress resistance pathways. This includes increased DNA repair, mitochondrial biogenesis, autophagy, expression of antioxidants and downregulation of inflammatory pathways.87 Another fundamental adaptive response to fasting is the metabolic switch from glucose to fatty acids and ketone bodies as a source of fuel. Not only is this more energy efficient but ketone bodies also act as potent signalling molecules triggering downstream protective pathways, including those implicated in metabolic dysfunction, NAFLD and NASH (eg, via proliferator-activated receptor γ coactivator 1α and fibroblast growth factor 21).88 89 In addition to the positive effects of fasting, TRE may also be beneficial through entraining the circadian clock and synchronising metabolic pathways with fixed periods of feeding.90 91
Figure 2.
IF and TRE strategies. Commonly employed IF and TRE strategies. Figure made with biorender.com. IF, Intermittent fasting; TRE, time-restricted eating.
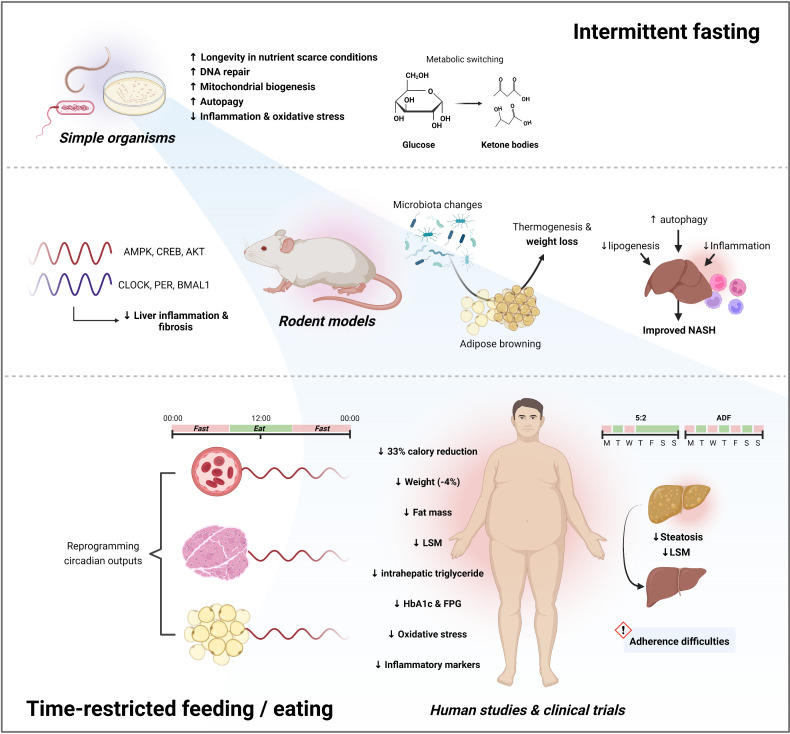
Figure 3.
Mechanistic basis for improved metabolic outcomes following fasting and TRF/TRE. The benefits of fasting (right) and TRF/TRE (left) are well conserved across simple organisms, rodent models and humans. Fasting has pleiotropic effects, including anti-inflammatory properties, weight loss through adipose browning and exerts a beneficial impact as a result of metabolic switching from glucose to fatty acids and ketones as a source of fuel. Time-restricted calorie intake introduces additional benefits by aligning circadian rhythms across multiple tissue types. 5:2, 5 days eating 2 days fasting; ADF, alternate day fasting; FPG, fasting plasma glucose; HbA1c, glycated haemoglobin; LSM, liver stiffness measurement; NASH, non-alcoholic steatohepatitis; TRE, time-restricted eating; TRF, time-restricted feeding. Figure made with biorender.com.
Evidence for IF and TRE in preclinical models of NAFLD and NASH
Numerous studies have reported the positive effects of IF on total body weight, fat mass reduction and glucose homeostasis in rodent models91–96 (figure 3). For example, mice with diet-induced obesity have significant reductions in weight (~45%) when exposed to 4 weeks of ADF compared with ad libitum feeding, despite equivalent cumulative calorie intake.95 These changes are associated with greater insulin sensitivity, decreased hepatic triglyceride content and reduced serum alanine transaminase (ALT) concentrations. Very similar improvements in liver phenotype have also been observed following 16 weeks of a 2 day feeding and 1 day fasting (2:1) regimen in overweight mice.94 Adaptations within adipose tissue appear to be central to these metabolic benefits.94 95 97 For example, fasting has been shown to selectively stimulate beige fat development within white adipose tissue leading to increased energy expenditure and weight loss through non-shivering thermogenesis.95 This may be mediated through alterations to gut microbiota composition, with IF being associated with bacterial-derived elevations in acetate and lactate which subsequently drive adipose tissue browning through upregulated monocarboxylate transporter 1 expression.95 Furthermore, germ-free mice are resistant to fasting-induced browning, which is restored following microbiota-transplantation with subsequent improvements in metabolic homeostasis.95 These modifications to adipocyte physiology, however, have not clearly translated into humans in whom calorie restriction appears to improve metabolic health independent of adipose tissue browning.98 Fasting protocols may also directly improve liver histology in rodent models of NASH by downregulating hepatic inflammatory pathways, modifying lipogenic gene expression and increasing markers of autophagy.96 99–102 A single study also found that 48-hour fasting was associated with dampened hepatic stellate cell activation and reduced collagen deposition in an Abcb4-/- mouse model of chronic liver fibrosis.103
TRF, even with a high-fat diet (HFD), has also been shown to reverse or protect against obesity-related complications, including insulin resistance, weight gain, hepatic steatosis and systemic inflammation, in mice.104–106 Again, many of these changes occur independent of total calorie restriction. TRF also normalises HFD-related expression of genes involved in hepatic lipid synthesis.106 TRF for only 2 weeks can lead to acute reductions in hepatic lobular inflammation and endoplasmic reticulum stress, which precedes significant weight loss.107 In addition, in a rodent model of NASH, 6 weeks of TRF reduced histological inflammation to the same extent as combination therapy with obeticholic acid and semaglutide, with TRF also being associated with significant improvements in liver fibrosis (available as conference abstract form only).108 These benefits serve to highlight the physiological importance of aligning food habits, metabolic pathways and the circadian clock. Feed–fasting cycles drive daily rhythms in the activity of key regulators of nutrient homeostasis (eg, AMPK, CREB and AKT) which, in healthy conditions, run in parallel with autonomous circadian rhythms governed by clock transcription factors (eg, BMAL1, CLOCK, PER and CRY).109 TRF in obese mice has been shown to modify both these cyclical components by improving CREB, mTOR and AMPK pathway function and increase the expression of core clock components in the liver104 (figure 3). By increasing the circadian amplitude of adipocyte thermogenesis, restricting feeding during the active phase is able to protect mice from diet-induced obesity through enhanced energy expenditure.110 Furthermore, TRF is able to prevent the predictable evolution of obesity, metabolic syndrome, and NASH in mice with genetic or environmental clock distuption.111 112 These changes may be mediated by insulin and insulin-like growth factor 1 (IGF-1) production in response to food, which helps determine the phase and amplitude of circadian rhythms in vivo through increased PER synthesis.113 This action of insulin and IGF-1 is not restricted to any particular tissue, and facilitates widespread coordination of gene expression and behaviour with time of feeding. Lastly, although IF and TRF are often viewed separately, the mechanistic basis for these two dietary interventions are often linked and overlapping. For example, under calorie restricted conditions, mice begin to self-impose chronic cycles of 2-hour feeding and 22-hour fasting with accumulating benefits on overall lifespan.114 Taken together, all this evidence in rodent models points to IF and TRE as a promising treatment candidates for human metabolic disease and NAFLD.
Safety and efficacy of IF in patients with obesity and metabolic dysfunction
Clinical studies in healthy volunteers have shown that IF confers wide-ranging benefits on markers of metabolic health and appears safe and well tolerated even after protracted periods of up to 6 months.115 Despite the profound caloric restriction imposed on fasting days, participants do not appear to fully compensate by increasing caloric intake on feeding days.115 This accumulating energy deficit leads to weight loss and fat mass reductions. Weight independent benefits may also occur through the cardioprotective and immunomodulatory effects of circulating mediators such as ketone bodies and polyunsaturated fatty acids, which increase with fasting.115 116 Notably, despite considerable study heterogeneity, absolute calorie restriction (0% calories on fasting days) is rarely reported in the literature with most protocols preferring to employ 25% of daily calorie requirements on fasting days.
A number of randomised controlled trials (RCTs) have specifically explored the benefits of IF in the context of obesity and metabolic disease. These studies have consistently shown IF to be associated with an energy deficit and weight loss ranging from 3% to 8% over an intervention period of 8–12 weeks.117 However, in head-to-head trials, the degree of weight loss is typically equivalent to that observed with continuous calorie restriction (CCR). This is substantiated through network meta-analysis of 24 studies showing that IF regimens result in a similar degree of weight loss to CCR (mean difference: −0.26 kg over a period of 2–26 weeks).118 There is no clear evidence to suggest the relative superiority of either 5:2 or ADF regimens.118 Post-interventional increases in weight also appear comparable between IF and CCR, with one study showing that both groups regain ~30% of their accumulated weight loss within 6 months.119 Unlike pre-clinical studies, there is a lack of human trials which precisely match caloric intake between arms. This has made it difficult to establish whether IF contributes to weight loss and metabolic improvement independent of energy restriction. Despite variable results, IF studies in overweight patients have tended to show longitudinal improvements in glycaemic parameters including HOMA-IR, fasting glucose and insulin, although these changes are rarely superior to those observed with continuous calorie restriction (CRC).119–121 A single trial in patients with T2D found that both a 5:2 fasting regimen and CCR appear equally safe and effective in reducing glycated haemoglobin (HbA1c) over a 12-month period.122 Overall, meta-analysis of trials, including both healthy and disease populations, have shown that IF confers beneficial outcomes compared with baseline, in fasting plasma glucose, fasting insulin, HOMA-IR, as well as cardiovascular markers, including LDL-cholesterol, total cholesterol, triglycerides and blood pressure.123 In addition, IF may also improve systemic inflammation in overweight individuals with reduced plasma levels of amyloid A protein, IL-6, CRP,124 tumour necrosis factor (TNF)-α and interferon γ and increased adiponectin120 all being reported.
IF also appears safe with no increased frequency of gastrointestinal symptoms, fatigue, irritability or dizziness compared with standard calorie restriction.120 125 While there were early concerns that IF could perpetuate disordered eating behaviours (eg, binge eating, purging and negative body image perception), this has not been demonstrated in clinical trials.125 Caution should be exercised, however, when initiating IF or TRE in patients with T2D particularly in those on insulin or hypoglycaemic agents.126 IF has been associated with a small absolute increased risk of hypoglycaemic episodes, although this can be mitigated by clinician supervision, hypoglycaemia education and pre-emptive modifications to medication dosing.127 Adherence to IF does, however, remain challenging for some individuals with drop-out rates tending to be higher than those observed with CCR, ranging between 4% and 58%.121 128 129 Although the cognitive drivers of non-adherence are yet to be fully investigated, IF does require participants to precisely monitor energy intake on a regular basis which may lead to waning compliance. TRE may, therefore, be a more attractive dietary strategy for some individuals by allowing them to simply ‘watch the clock’ rather than meticulously calculate ongoing calorie consumption. This is demonstrated in meta-analysis of 24 RCTs where study retention rates used as a gross indicator of compliance were highest for TRE (94%), followed by 5:2 diet (88%) and ADF (85%).118
Clinical trials of IF in patients with NAFLD
Several studies have explored the beneficial effects of IF specifically in patients with NAFLD. Holmer et al performed an open-label randomised trial of a 5:2 fasting regimen compared with low-carbohydrate high-fat (LCHF) diet or standard of care (SoC) (routine lifestyle advice) in 74 patients diagnosed with NAFLD via imaging or FibroScan controlled attenuation parameter (CAP).128 Compared with SoC, the 5:2 and LCHF diets were both associated with greater absolute reductions in steatosis (5:2 diet: −6.1%, LCHF: −7.2%, SoC: −3.6%) and body weight (5:2 diet: −7.4 kg, LCHF −7.3 kg, SoC −2.5 kg). The 12-week 5:2 intervention also led to significant improvements in liver stiffness (−1.8 kPa), ALT (−17.6 U/L), HOMA-IR, HbA1c and LDL compared with baseline. Similarly, in a separate study, 8 weeks of ADF in patients with an established clinical diagnosis of NAFLD was associated with greater reductions in hepatic steatosis on ultrasound, improvements in hepatic sheer wave elastography and reduced ALT compared with ad libitum eating.130 Cai et al also demonstrated IF to be an effective and well-tolerated strategy for individuals with NAFLD, with ADF being associated with >5% wt loss and improvement in dyslipidaemia even after only 4 weeks.131 Incremental weight loss was subsequently observed at 12 weeks with concurrent reductions in fat mass superior to those randomised to control diets although there was no significant improvement in liver stiffness measurements (LSMs) compared with baseline.131 Notably, no trials of IF (or TRE) have been performed in overweight patients with cirrhosis. The relative risks and benefits of weight loss in this specific patient group has been subject of debate. While weight loss of 5%–10% in compensated cirrhosis has been shown to reduce liver disease progression and improve portal pressures,132 there have been residual concerns about the potential to precipitate sarcopenia and decompensation. Reassuringly, no decompensating events were reported in a randomised trial of semaglutide in compensated NASH cirrhosis despite 9% wt loss.133 Trials of IF in both elderly populations and in high-performance athletes have also not demonstrated any associations with reduced muscle mass or function.134 135 However, patients with decompensated cirrhosis do represent a particularly high risk group in whom weight loss interventions (including IF and TRE) should be implemented with caution alongside specialist multidisciplinary input, structured exercise and adequate protein intake in line with international societal practice guidelines.136 137 Lastly, the beneficial effect of night-time nutritional supplementation or an ‘evening snack’ on protein-energy balance has long been established in patients with cirrhosis, thus providing an additional reason for caution with the use of IF and TRE in this group outside of clinical trials.138
Safety and efficacy of TRE in patients with obesity and metabolic dysfunction
TRE involves limiting the window of energy intake to a specific number of hours per day with only water and zero-calorie beverages for the remainder of the day. During the eating window, individuals are usually permitted to eat ad libitum without any imposed calorie restriction. Most human trials have employed an eating window lasting between 6 hours and 10 hours. This duration has been shown to be safe, well tolerated and has tended to yield the greatest clinical benefit. Shorter windows (eg, 4 hours) are associated with an increased risk of minor adverse events (eg, headache, mood changes and nausea) without additional metabolic improvements139 and longer durations (≥12 hours) begin to imitate natural eating habits and have not shown any efficacy signal.140
Although TRE does not overtly attempt to minimise energy intake, by simply limiting the eating window to 6–10 hours, individuals naturally reduce their calorie consumption by as much as 566 kcal/day (−30%).139 This has consistently been associated with weight loss of approximately 3%–4% alongside corresponding reductions in total fat mass, BMI and waist circumference.141–144 Interestingly, however, the degree of weight loss with TRE appears to be smaller than that observed with equivalent calorie reductions achieved during standard CCR (−5% to 7%).121 129 145 These findings may be related to inaccurate food diary reporting in TRE trials or due to as yet unknown physiological mechanisms. Meta-analyses of RCTs has corroborated these observations showing that IF is likely to be the most effective dietary weight loss strategy, followed by CCR, and then TRE.118 Nonetheless, despite relatively modest weight reductions, TRE is associated with significant and often disproportionate improvements in cardiometabolic health. Indeed, several clinical studies have shown a beneficial impact on glucose regulation, oxidative stress and blood pressure independent of weight loss.146 147
As well as controlling the duration of eating and fasting periods, the timing of the daily eating window has been subject of increasing investigation. TRE can broadly be categorised along a spectrum between early TRE (eTRE) when the final meal is eaten mid-afternoon and late TRE (lTRE) when dinner is consumed in the evening. Several randomised trials have demonstrated superiority of early versus lTRE with regards to weight loss, glycaemic control, satiety hormone profiles (eg, reduced ghrelin and increased PYY) and hunger scores.148–151 Meta-analysis of studies has also indicated that eTRE is associated with the greatest improvements in insulin sensitivity.152 While most studies stipulate the period of energy consumption, some groups have allowed self-selection of the eating window with comparable improvements in weight and metabolic parameters.144 When participants are allowed to choose their preferred period of eating, the majority will select an early or intermediate timeframe where all eating is completed >2 hours before going to bed which is associated with improvements in fasting glucose.153 This re-enforces observational data showing that minimising energy intake in the evening is associated with improved metabolic health. Eating earlier in the day ensures that calorie intake occurs at the time of maximal insulin sensitivity, and also at the time at which the internal circadian clock is most sensitive to external inputs.
TRE can reprogramme circadian regulation of human metabolism
Several human studies have directly explored the impact of TRE on circadian markers. First, TRE has been associated with an increase in the amplitude of cortisol rhythms while melatonin levels, which are predominantly influenced through light–dark signalling, remain unaffected.154 Detailed metabolic phenotyping of obese male participants has also demonstrated marked alterations in 24-hour profiles of insulin, NEFA and triglycerides following 8 weeks of self-selected 10-hour TRE.154 These systemic changes seem to occur rapidly after dietary intervention, with a second study in overweight individuals showing similar shifts in circulating insulin, NEFA, gastric inhibitory peptide and GLP-1 after only 4 days of TRE.155 These fluctuations occur in parallel with increased whole blood expression of multiple clock genes and genes involved in longevity and autophagy (eg, SIRT1 and BDNF).148 These findings were corroborated in a further study in healthy volunteers, which also showed widespread enhancement of clock gene expression in peripheral blood, which was more pronounced with early compared with mid-day TRE.156 In addition to circulating biomarkers, TRE also has a direct impact on the rhythmic behaviour of metabolically active tissues known to be integral to the pathogenesis of NAFLD, including adipose and skeletal muscle. In subcutaneous adipose tissue, TRE has the remarkable ability to trigger the oscillation of hundreds of genes which are otherwise arrhythmic at baseline.154 This includes genes involved in adipogenesis, adipose browning and pathways mediating lipogenesis (eg, SREBF1) which has an important role in the sensitisation of adipose tissue to insulin.157 Similarly, short-term TRE in overweight men tended to advance the circadian phase of both circulating serum metabolites and genes controlling amino acid transport in skeletal muscle.158 These latter changes, however, occurred in the absence of alterations to muscle clock gene oscillations, suggesting that TRE may have tissue-specific effects on metabolic function independent of the autonomous clock machinery. Further experimental work in humans is required to decipher the role of feeding cues in reprogramming diurnal metabolism, including the differential impact on central and peripheral clocks. Several studies have also shown beneficial effects of TRE on sleep quality and duration further implicating the role of eating patterns in establishing circadian realignment.159 This concept is also demonstrated by accumulating data showing a benefit of TRE in shift-workers with circadian misalignment who tend to have longer eating windows and higher total energy intake.160 In a recent randomised trial, including 24-hour shift-working firefighters, TRE improved very-low-density lipoprotein (VLDL) particle size, quality of life and sleep disturbance, and led to reductions in HbA1c and blood pressure in those with elevated cardiovascular risk at baseline.161
Therapeutic potential of TRE in NAFLD
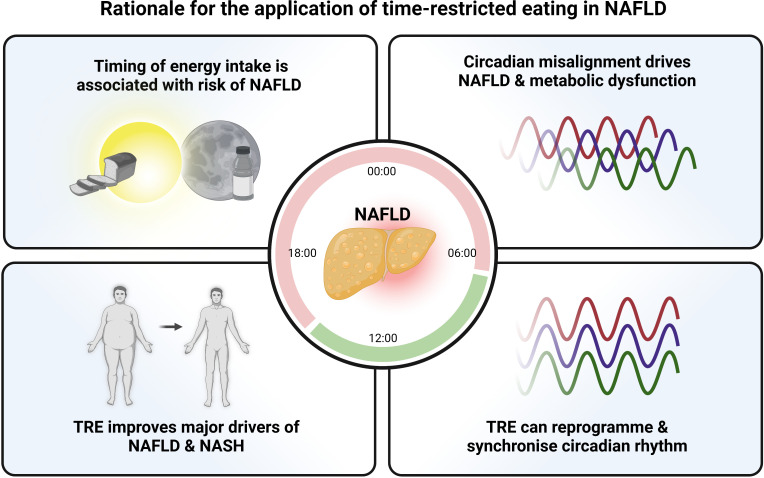
TRE research over the last two decades has largely revolved around single-arm, proof-of-concept longitudinal studies or small pilot trials over short durations with improvements in weight and cardiovascular markers as the most common primary outcomes. Nonetheless, the wide reproducibility of positive findings has allowed the field to expand exponentially with numerous large, more rigorous RCTs planned or underway across an expanding range of chronic health conditions.144 There is now clear rationale for further exploration of TRE specifically in NAFLD. The background evidence is compelling as discussed above; circadian misalignment is associated with hepatic metabolic dysfunction, chaotic dietary patterns predispose to steatosis and inflammation, TRE can reprogramme circadian outputs and has been shown to improve key drivers of NAFLD progression, including weight and insulin resistance (figure 4).
Figure 4.
Rationale for the application of TRE in NAFLD. Multiple strands of evidence point toward the therapeutic potential of TRE in NAFLD and NASH. Timing of energy intake, circadian rhythms and metabolic phenotype are all intrinsically linked. These interactions can be positively influenced by TRE which has been shown to restore the rhythmicity of metabolic pathways and is known to improve major drivers of liver disease progression, including obesity and insulin resistance. NAFLD, non-alcoholic fatty liver disease; NASH, non-alcoholic steatohepatitis; TRE, time-restricted eating. Figure made with biorender.com.
Some data do currently exist which report a benefit of TRE in patients with NAFLD. In a Chinese trial, Cai et al reported outcomes in 96 NAFLD patients with high LSM at baseline (>9.6 kPa) who were randomised to a self-selected 8 hour TRE window.131 Compared with control diet, TRE was well tolerated and associated with significant weight loss (−4.8%), decreased fat mass and improved triglycerides over 12 weeks. However, there was no change in LSM likely due to the relatively short intervention period and the study did not include assessments of steatosis, inflammation, liver biochemistry or circadian markers at baseline or follow-up. A second RCT in T2D showed that eating breakfast and lunch (between 06:00 and 16:00) over 12 weeks was more effective at reducing body weight, fasting plasma glucose and intrahepatic triglyceride (IHTG) than the same calorie restriction split across six small meals throughout the day. Lastly, a rigorous trial by Wei et al randomised 88 patients with obesity and NAFLD to receive either TRE with calorie restriction (08:00 and 16:00) or calorie restriction alone for 12 months. All participants consumed <1800 kcal/day for men and <1500 kcal/day for women and received regular telephone reminders and face-to-face dietician input. Both interventions resulted in substantial weight loss of >10% with marked reductions in both IHTG and LSM, emphasising the importance of achieving energy deficit in the management of obesity and NAFLD. While TRE significantly improved HOMA-IR compared with daily calorie restriction, it was not superior in reducing IHTG as the primary endpoint. This may be related to the study’s particularly stringent calorie targets masking relatively subtle benefits derived from differential food timing. Exploring whether TRE can achieve meaningful clinical benefits with less aggressive calorie restriction will help establish whether this strategy has wider real-world applicability outside of a clinical trial setting.
Two further RCTs in individuals without established NAFLD have reported a tendency towards improved liver biochemistry with TRE. Xie et al showed a modest but significant reduction in plasma AST in healthy volunteers randomised to eTRE (−3 U/L) compared with control diet, which occurred in parallel with reduced inflammatory mediators (eg, TNF-α and IL-8), improved insulin sensitivity, upregulated clock gene expression and enhanced gut microbial diversity.156 A second study of 10-hour TRE for 12 weeks in patients with metabolic syndrome was also associated with a 10% reduction in both AST and ALT, although this did not reach statistical significance (p=0.09).147 Further dedicated trials incorporating liver-specific outcomes are required in well-phenotyped patients with NAFLD/NASH in order to establish efficacy and decipher underlying mechanistic pathways contributing to improved liver and metabolic health. Several additional RCTs of TRE in patients with NAFLD are currently registered with clinicaltrials.gov. These report a range of target sample sizes reaching a maximum of 400 patients (NCT05579158) and have variable intervention time-frames between 6 weeks and 16 weeks. Primary outcomes in these active trials have predominantly focused on hepatic steatosis assessed by magnetic resonance techniques or CAP. Some groups also plan to report on hepatic de novo lipogenesis (NCT04997486) as well as markers of fibrogenesis, oxidative stress and changes in gut microbiota (NCT05220956). It remains unclear to what extent these trials will explore the effect of TRE on circadian rhythms, sleep, and diurnal patterns of liver metabolism and inflammation. Future studies will need to establish the longer-term efficacy and tolerability of these regimens in patients with NAFLD and directly compare different fasting regimens and calorie restriction thresholds.
Lastly, despite accumulating human data signalling the benefits of IF and TRE, these dietary interventions are currently under-represented in national and international clinical practice guidelines relating to metabolic disease, including NAFLD (table 1). This is likely to be partly related to the lack of currently available data from well-powered trials reporting on clinical outcomes or robust surrogate endpoints. There is also little acknowledgement in clinical guidelines that unhealthy timing of energy intake (eg, skipping breakfast and night-time eating) may represent discreet risk factors for metabolic disease. In the coming years, as the field continues to advance, it is likely that an increasing number of consensus statements acknowledge the value of modifying timing of calorie intake as a potential strategy for NAFLD prevention and treatment.
Table 1.
Reference to timing of energy intake in societal guidelines for NAFLD and metabolic disease
| Society clinical practice guideline | Patient group | Year | Dietary recommendations | Specific reference to timing of energy intake | Specific reference to IF and/or TRE |
| EASL—EASD—EASO162 | NAFLD | 2016 | ∙ Energy restriction ∙ Reduce processed and high-fructose foods ∙ Mediterranean diet |
– | – |
| AASLD163 | NAFLD | 2022 | ∙ Prescribe a diet leading to calorie deficit ∙ Encourage Mediterranean diet |
– | IF included as potential dietary strategy to achieve calorie deficit |
| AGA164 | NAFLD | 2021 | ∙ Hypocaloric diet ∙ Should follow the Mediterranean diet |
– | ∙ Potential benefits of IF and TRE but require an individualised approach before prescribing ∙ Additional studies are needed before IF and TRE can routinely be considered |
| APASL165 | MAFLD | 2020 | ∙ Energy restriction ∙ Reduce processed and high-fructose foods ∙ Mediterranean diet |
– | – |
| BASL and BSG166 | NAFLD | 2022 | ∙ Tailored dietary advice to achieve a calorie deficit ∙ Reduction of refined carbohydrates and processed foods ∙ Increased consumption of vegetables, lean protein and fish |
– | – |
| EASO167 | Obesity | 2015 | ∙ Individualised energy restriction diets ∙ Decrease energy density of foods and reduce ∙ Portion sizes ∙ Manage and reduce episodes of loss of control or binge eating |
∙ Avoid snacking between meals ∙ Do not skip breakfast ∙ Avoid eating at night |
– |
| ACC—AHA—The Obesity Society168 | Obesity | 2013 | ∙ Prescribe an individualised diet leading to calorie deficit (eg, low-carbohydrate, low-protein, Mediterranean, low-glycaemic load) | – | – |
| EASD and ADA168 | T2D | 2022 | ∙ Individually selected diet to achieve energy deficit ∙ Mediterranean and low carbohydrate diet |
– | ∙ IF likely to lead to similar improvements in glycaemic control to CCR ∙ Caution with insulin and sulphonylureas |
| ADA169 | Obesity in T2D | 2022 | ∙ Individualised nutritional recommendations to achieve energy deficit | – | – |
AASLD, American Associate for the Study of the Liver Diseases; ACC, American College of Cardiology; ADA, American Diabetes Association; AGA, American Gastroenterology Association; AHA, American Heart Association; APASL, Asian Pacific Association for the Study of the Liver; BASL, British Association for the Study of the Liver; BSG, British Society for Gastroenterology; CCR, continuous calorie restriction; EASD, European Association for the Study of Diabetes; EASL, European Association for the Study of the Liver; EASO, European Association for the Study of Obesity; IF, intermittent fasting; MAFLD, metabolic-dysfunction associated fatty liver disease; NAFLD, non-alcoholic fatty liver disease; T2D, type 2 diabetes; TRE, time-restricted eating.
Conclusion
Temporal eating patterns, liver homeostasis, circadian clock function and metabolic health are all intrinsically linked. Food intake is an extremely powerful external timing cue, which serves to synchronise rhythms of hepatic energy metabolism to fed-fasted cycles, independently of the light–dark cycle. As a result, certain non-physiological eating behaviours, including missing breakfast, irregular meal patterns and night-time snacking, are closely associated with an adverse metabolic and liver phenotype. However, this has presented an exciting opportunity for therapeutic intervention though manipulating the timing of calorie intake. IF protocols in humans have drawn on a wealth of preclinical data showing that multiple advantageous metabolic pathways are upregulated in nutrient scarce conditions. Furthermore, TRE may facilitate incremental benefits beyond fasting by resynchronising circadian rhythms across multiple metabolically active tissues involved in the pathogenesis of NAFLD. As a result, TRE has been shown in clinical trials to be a safe and effective way to reduce steatosis and improve multiple domains of the metabolic syndrome. Taken together, there is a strong rationale for further detailed exploration of the liver-specific benefits of TRE and this dietary strategy may emerge as a promising management option for patients with NAFLD and NASH.
Footnotes
Twitter: @tom_marjot
Contributors: TM was responsible for the review’s concept and design, performed the literature search and synthesis of the evidence, and wrote the original draft of the manuscript. All authors were involved in the critical revision of the work.
Funding: TM receives funding via a Wellcome Trust Clinical Research Training Fellowship (ref. 102176/B/13/Z). JWT receives funding through a programme grant from the Medical Research Council (MRC) (programme ref. MR/P011462/1). LH receives funding through a senior research fellowship from the British Heart Foundation (fellowship ref. FS/SBSRF/21/31013). DWR receives funding via NIHR Oxford Health Biomedical Research Centre (grant reference number: NIHR203316; MRC: MR/W019000/1 and MR/V034049/1). No funders were involved in the writing of the manuscript or the decision to submit for publication.
Competing interests: JWT has received personal fees outside of the submitted work for acting as an advisory board member for Pfizer and Poxel, and as a member of the data and safety monitoring committee for Novartis. TM has received educational honoraria from Falk, outside of the current work. There are no additional interests to declare.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
Not applicable.
References
- 1. Yip TC, Vilar‐Gomez E, Petta S, et al. Geographical similarity and differences in the burden and genetic predisposition of NAFLD. Hepatology 2023;77:1404–27. 10.1002/hep.32774 [DOI] [PubMed] [Google Scholar]
- 2. Sanyal AJ, Van Natta ML, Clark J, et al. Prospective study of outcomes in adults with Nonalcoholic fatty liver disease. N Engl J Med 2021;385:1559–69. 10.1056/NEJMoa2029349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Targher G, Byrne CD, Tilg H. NAFLD and increased risk of cardiovascular disease: clinical associations, pathophysiological mechanisms and pharmacological implications. Gut 2020;69:1691–705. 10.1136/gutjnl-2020-320622 [DOI] [PubMed] [Google Scholar]
- 4. Marjot T, Moolla A, Cobbold JF, et al. Nonalcoholic fatty liver disease in adults: Current concepts in etiology, outcomes, and management. Endocr Rev 2020;41:66–117. 10.1210/endrev/bnz009 [DOI] [PubMed] [Google Scholar]
- 5. Dufour J-F, Anstee QM, Bugianesi E, et al. Current therapies and new developments in NASH. Gut 2022;71:2123–34. 10.1136/gutjnl-2021-326874 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Fraser A, Abel R, Lawlor DA, et al. A modified Mediterranean diet is associated with the greatest reduction in alanine aminotransferase levels in obese type 2 diabetes patients: results of a quasi-randomised controlled trial. Diabetologia 2008;51:1616–22. 10.1007/s00125-008-1049-1 [DOI] [PubMed] [Google Scholar]
- 7. Yaskolka Meir A, Rinott E, Tsaban G, et al. Effect of green-Mediterranean diet on Intrahepatic fat: the DIRECT PLUS randomised controlled trial. Gut 2021;70:2085–95. 10.1136/gutjnl-2020-323106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Saran AR, Dave S, Zarrinpar A. Circadian rhythms in the pathogenesis and treatment of fatty liver disease. Gastroenterology 2020;158:1948–66. 10.1053/j.gastro.2020.01.050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Marjot T, Ray DW, Tomlinson JW. Is it time for Chronopharmacology in NASH Journal of Hepatology 2022;76:1215–24. 10.1016/j.jhep.2021.12.039 [DOI] [PubMed] [Google Scholar]
- 10. Stokkan K-A, Yamazaki S, Tei H, et al. Entrainment of the circadian clock in the liver by feeding. Science 2001;291:490–3. 10.1126/science.291.5503.490 [DOI] [PubMed] [Google Scholar]
- 11. Damiola F, Le Minh N, Preitner N, et al. Restricted feeding Uncouples circadian Oscillators in peripheral tissues from the central pacemaker in the Suprachiasmatic nucleus. Genes Dev 2000;14:2950–61. 10.1101/gad.183500 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Turek FW, Joshu C, Kohsaka A, et al. Obesity and metabolic syndrome in circadian clock mutant mice. Science 2005;308:1043–5. 10.1126/science.1108750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Kettner NM, Voicu H, Finegold MJ, et al. Circadian homeostasis of liver metabolism suppresses Hepatocarcinogenesis. Cancer Cell 2016;30:909. 10.1016/j.ccell.2016.10.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Mukherji A, Kobiita A, Chambon P. Shifting the feeding of mice to the rest phase creates metabolic alterations, which, on their own, shift the peripheral circadian clocks by 12 hours. Proc Natl Acad Sci USA 2015;112:E6683–90. 10.1073/pnas.1519735112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Asher G, Sassone-Corsi P. Time for food: the intimate interplay between nutrition, metabolism, and the circadian clock. Cell 2015;161:84–92. 10.1016/j.cell.2015.03.015 [DOI] [PubMed] [Google Scholar]
- 16. Pot GK, Almoosawi S, Stephen AM. Meal irregularity and Cardiometabolic consequences: results from observational and intervention studies. Proc Nutr Soc 2016;75:475–86. 10.1017/S0029665116000239 [DOI] [PubMed] [Google Scholar]
- 17. Sierra-Johnson J, Undén A-L, Linestrand M, et al. Eating meals irregularly: a novel environmental risk factor for the metabolic syndrome. Obesity 2008;16:1302–7. 10.1038/oby.2008.203 [DOI] [PubMed] [Google Scholar]
- 18. Howarth NC, Huang TT-K, Roberts SB, et al. Eating patterns and dietary composition in relation to BMI in younger and older adults. Int J Obes 2007;31:675–84. 10.1038/sj.ijo.0803456 [DOI] [PubMed] [Google Scholar]
- 19. Murakami K, Livingstone MBE. Eating frequency is positively associated with overweight and central obesity in US adults. J Nutr 2015;145:2715–24. 10.3945/jn.115.219808 [DOI] [PubMed] [Google Scholar]
- 20. Farshchi HR, Taylor MA, Macdonald IA. Beneficial metabolic effects of regular meal frequency on dietary Thermogenesis, insulin sensitivity, and fasting lipid profiles in healthy obese women. The American Journal of Clinical Nutrition 2005;81:16–24. 10.1093/ajcn/81.1.16 [DOI] [PubMed] [Google Scholar]
- 21. Gill S, Panda S. A Smartphone App reveals erratic diurnal eating patterns in humans that can be modulated for health benefits. Cell Metab 2015;22:789–98. 10.1016/j.cmet.2015.09.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Gupta NJ, Kumar V, Panda S. A camera-phone based study reveals erratic eating pattern and disrupted daily eating-fasting cycle among adults in India. PLoS One 2017;12:e0172852. 10.1371/journal.pone.0172852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Wennberg M, Gustafsson PE, Wennberg P, et al. Irregular eating of meals in adolescence and the metabolic syndrome in adulthood: results from a 27-year prospective cohort. Public Health Nutr 2016;19:667–73. 10.1017/S1368980015001445 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Okada C, Tabuchi T, Iso H. Association between skipping breakfast in parents and children and childhood overweight/obesity among children: a nationwide 10.5-year prospective study in Japan. Int J Obes (Lond) 2018;42:1724–32. 10.1038/s41366-018-0066-5 [DOI] [PubMed] [Google Scholar]
- 25. Uzhova I, Fuster V, Fernández-Ortiz A, et al. The importance of breakfast in Atherosclerosis disease: insights from the PESA study. J Am Coll Cardiol 2017;70:1833–42. 10.1016/j.jacc.2017.08.027 [DOI] [PubMed] [Google Scholar]
- 26. Kubota Y, Iso H, Sawada N, et al. Association of breakfast intake with incident stroke and coronary heart disease: the Japan public health center–based study. Stroke 2016;47:477–81. 10.1161/STROKEAHA.115.011350 [DOI] [PubMed] [Google Scholar]
- 27. Maki KC, Phillips-Eakley AK, Smith KN. The effects of breakfast consumption and composition on metabolic wellness with a focus on carbohydrate metabolism. Adv Nutr 2016;7:613S–21S. 10.3945/an.115.010314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Jakubowicz D, Wainstein J, Ahren B, et al. Fasting until noon triggers increased postprandial hyperglycemia and impaired insulin response after lunch and dinner in individuals with type 2 diabetes: a randomized clinical trial. Diabetes Care 2015;38:1820–6. 10.2337/dc15-0761 [DOI] [PubMed] [Google Scholar]
- 29. Han AL. Association between non-alcoholic fatty liver disease and dietary habits, stress, and health-related quality of life in Korean adults. Nutrients 2020;12:1555. 10.3390/nu12061555 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Tsuruta G, Tanaka N, Hongo M, et al. Nonalcoholic fatty liver disease in Japanese junior high school students: its prevalence and relationship to Lifestyle habits. J Gastroenterol 2010;45:666–72. 10.1007/s00535-009-0198-4 [DOI] [PubMed] [Google Scholar]
- 31. Xie J, Huang H, Chen Y, et al. Skipping breakfast is associated with an increased Long‐Term cardiovascular mortality in metabolic Dysfunction‐Associated fatty liver disease (MAFLD) but not MAFLD‐Free individuals. Aliment Pharmacol Ther 2022;55:212–24. 10.1111/apt.16727 Available: https://onlinelibrary.wiley.com/toc/13652036/55/2 [DOI] [PubMed] [Google Scholar]
- 32. St-Onge M-P, Ard J, Baskin ML, et al. Meal timing and frequency: implications for cardiovascular disease prevention: a scientific statement from the American heart Association. Circulation 2017;135:e96–121. 10.1161/CIR.0000000000000476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. de Castro JM. When, how much and what foods are eaten are related to total daily food intake. Br J Nutr 2009;102:1228–37. 10.1017/S0007114509371640 [DOI] [PubMed] [Google Scholar]
- 34. de Castro JM. The time of day and the proportions of Macronutrients eaten are related to total daily food intake. Br J Nutr 2007;98:1077–83. 10.1017/S0007114507754296 [DOI] [PubMed] [Google Scholar]
- 35. Forester SM, Widaman AM, Krishnan S, et al. A clear difference emerges in hormone patterns following a standard midday meal in young women who regularly eat or skip breakfast. J Nutr 2018;148:685–92. 10.1093/jn/nxy020 [DOI] [PubMed] [Google Scholar]
- 36. Sievert K, Hussain SM, Page MJ, et al. Effect of breakfast on weight and energy intake: systematic review and meta-analysis of randomised controlled trials. BMJ 2019:l42. 10.1136/bmj.l42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Jovanovic A, Gerrard J, Taylor R. The second-meal phenomenon in type 2 diabetes. Diabetes Care 2009;32:1199–201. 10.2337/dc08-2196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Meng H, Matthan NR, Ausman LM, et al. Effect of prior meal Macronutrient composition on postprandial Glycemic responses and Glycemic index and Glycemic load value determinations. The American Journal of Clinical Nutrition 2017;106:1246–56. 10.3945/ajcn.117.162727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Jovanovic A, Leverton E, Solanky B, et al. The second-meal phenomenon is associated with enhanced muscle Glycogen storage in humans. Clin Sci 2009;117:119–27. 10.1042/CS20080542 [DOI] [PubMed] [Google Scholar]
- 40. Lewis P, Oster H, Korf HW, et al. Food as a circadian time cue—evidence from human studies. Nat Rev Endocrinol 2020;16:213–23. 10.1038/s41574-020-0318-z [DOI] [PubMed] [Google Scholar]
- 41. Kräuchi K, Cajochen C, Werth E, et al. Alteration of internal circadian phase relationships after morning versus evening carbohydrate-rich meals in humans. J Biol Rhythms 2002;17:364–76. 10.1177/074873040201700409 [DOI] [PubMed] [Google Scholar]
- 42. Ogata H, Horie M, Kayaba M, et al. Skipping breakfast for 6 days delayed the circadian rhythm of the body temperature without altering clock gene expression in human leukocytes. Nutrients 2020;12:2797. 10.3390/nu12092797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Nakade M, Takeuchi H, Taniwaki N, et al. An integrated effect of protein intake at breakfast and morning exposure to sunlight on the circadian typology in Japanese infants aged 2–6 years. J Physiol Anthropol 2009;28:239–45. 10.2114/jpa2.28.239 [DOI] [PubMed] [Google Scholar]
- 44. Harada T, Hirotani M, Maeda M, et al. Correlation between breakfast Tryptophan content and Morningness–Eveningness in Japanese infants and students aged 0–15 Yrs. J Physiol Anthropol 2007;26:201–7. 10.2114/jpa2.26.201 [DOI] [PubMed] [Google Scholar]
- 45. Hermenegildo Y, López-García E, García-Esquinas E, et al. Distribution of energy intake throughout the day and weight gain: a population-based cohort study in Spain. Br J Nutr 2016;115:2003–10. 10.1017/S0007114516000891 [DOI] [PubMed] [Google Scholar]
- 46. Wang JB, Patterson RE, Ang A, et al. Timing of energy intake during the day is associated with the risk of obesity in adults. J Hum Nutr Diet 2014;27 Suppl 2:255–62. 10.1111/jhn.12141 [DOI] [PubMed] [Google Scholar]
- 47. Hermenegildo-López Y, Donat-Vargas C, Sandoval-Insausti H, et al. A higher intake of energy at dinner is associated with incident metabolic syndrome: A prospective cohort study in older adults. Nutrients 2021;13:3035. 10.3390/nu13093035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Bo S, Musso G, Beccuti G, et al. Consuming more of daily caloric intake at dinner predisposes to obesity. A 6-year population-based prospective cohort study. PLoS One 2014;9:e108467. 10.1371/journal.pone.0108467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Nishi T, Babazono A, Maeda T, et al. Effects of eating fast and eating before bedtime on the development of Nonalcoholic fatty liver disease. Popul Health Manag 2016;19:279–83. 10.1089/pop.2015.0088 [DOI] [PubMed] [Google Scholar]
- 50. Lee A, Ader M, Bray GA, et al. Diurnal variation in glucose tolerance: cyclic suppression of insulin action and insulin secretion in normal-weight, but not obese, subjects. Diabetes 1992;41:750–9. 10.2337/diab.41.6.750 [DOI] [PubMed] [Google Scholar]
- 51. Poggiogalle E, Jamshed H, Peterson CM. Circadian regulation of glucose, lipid, and energy metabolism in humans. Metabolism 2018;84:11–27.:S0026-0495(17)30329-3. 10.1016/j.metabol.2017.11.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Jakubowicz D, Barnea M, Wainstein J, et al. High caloric intake at breakfast vs. dinner Differentially influences weight loss of overweight and obese women. Obesity (Silver Spring) 2013;21:2504–12. 10.1002/oby.20460 [DOI] [PubMed] [Google Scholar]
- 53. Ha K, Song Y. Associations of meal timing and frequency with obesity and metabolic syndrome among Korean adults. Nutrients 2019;11:2437. 10.3390/nu11102437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Almoosawi S, Vingeliene S, Karagounis LG, et al. Chrono-nutrition: a review of current evidence from observational studies on global trends in time-of-day of energy intake and its association with obesity. Proc Nutr Soc 2016;75:487–500. 10.1017/S0029665116000306 [DOI] [PubMed] [Google Scholar]
- 55. Al-Naimi S, Hampton SM, Richard P, et al. Postprandial metabolic profiles following meals and snacks eaten during simulated night and day shift work. Chronobiol Int 2004;21:937–47. 10.1081/cbi-200037171 [DOI] [PubMed] [Google Scholar]
- 56. Takahashi M, Ozaki M, Kang M-I, et al. Effects of meal timing on postprandial glucose metabolism and blood metabolites in healthy adults. Nutrients 2018;10:1763. 10.3390/nu10111763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Vetter C, Dashti HS, Lane JM, et al. Night shift work, genetic risk, and type 2 diabetes in the UK Biobank. Diabetes Care 2018;41:762–9. 10.2337/dc17-1933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. STUNKARD AJ, GRACE WJ, WOLFF HG. The night-eating syndrome: a pattern of food intake among certain obese patients. Am J Med 1955;19:78–86. 10.1016/0002-9343(55)90276-x [DOI] [PubMed] [Google Scholar]
- 59. Goel N, Stunkard AJ, Rogers NL, et al. Circadian rhythm profiles in women with night eating syndrome. J Biol Rhythms 2009;24:85–94. 10.1177/0748730408328914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Akdevelioglu Y, Sahin TO, Yesildemir O. Sleep quality and its relationship with night eating syndrome, the risk of diabetes, and nutritional status among university students. Prog Nutr 2020;22:304–15. [Google Scholar]
- 61. Mazri FH, Manaf ZA, Shahar S, et al. The association between Chronotype and dietary pattern among adults: a Scoping review. IJERPH 2020;17:68. 10.3390/ijerph17010068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Maukonen M, Kanerva N, Partonen T, et al. Chronotype and energy intake timing in relation to changes in Anthropometrics: a 7-year follow-up study in adults. Chronobiol Int 2019;36:27–41. 10.1080/07420528.2018.1515772 [DOI] [PubMed] [Google Scholar]
- 63. Yu JH, Yun C-H, Ahn JH, et al. Evening Chronotype is associated with metabolic disorders and body composition in middle-aged adults. The Journal of Clinical Endocrinology & Metabolism 2015;100:1494–502. 10.1210/jc.2014-3754 [DOI] [PubMed] [Google Scholar]
- 64. Vetrani C, Barrea L, Verde L, et al. Evening Chronotype is associated with severe NAFLD in obesity. Int J Obes 2022;46:1638–43. 10.1038/s41366-022-01159-3 [DOI] [PubMed] [Google Scholar]
- 65. Younes R, Rosso C, Petta S, et al. Usefulness of the index of NASH–ION for the diagnosis of Steatohepatitis in patients with Non‐Alcoholic fatty liver: an external validation study. Liver Int 2018;38:715–23. 10.1111/liv.13612 [DOI] [PubMed] [Google Scholar]
- 66. Faris MA-IE, Jahrami HA, Alhayki FA, et al. Effect of diurnal fasting on sleep during Ramadan: a systematic review and meta-analysis. Sleep Breath 2020;24:771–82. 10.1007/s11325-019-01986-1 [DOI] [PubMed] [Google Scholar]
- 67. Reilly T, Waterhouse J. Altered sleep–wake cycles and food intake: the Ramadan model. Physiology & Behavior 2007;90:219–28. 10.1016/j.physbeh.2006.09.004 [DOI] [PubMed] [Google Scholar]
- 68. Lessan N, Saadane I, Alkaf B, et al. The effects of Ramadan fasting on activity and energy expenditure. The American Journal of Clinical Nutrition 2018;107:54–61. 10.1093/ajcn/nqx016 [DOI] [PubMed] [Google Scholar]
- 69. Sadeghirad B, Motaghipisheh S, Kolahdooz F, et al. Islamic fasting and weight loss: a systematic review and meta-analysis. Public Health Nutr 2014;17:396–406. 10.1017/S1368980012005046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Fernando HA, Zibellini J, Harris RA, et al. Effect of Ramadan fasting on weight and body composition in healthy non-athlete adults: a systematic review and meta-analysis. Nutrients 2019;11:478. 10.3390/nu11020478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Jahrami HA, Faris ME, I Janahi A, et al. Does four-week consecutive, dawn-to-sunset intermittent fasting during Ramadan affect Cardiometabolic risk factors in healthy adults? A systematic review, meta-analysis, and meta-regression. Nutr Metab Cardiovasc Dis 2021;31:2273–301. 10.1016/j.numecd.2021.05.002 [DOI] [PubMed] [Google Scholar]
- 72. Aydın N, Kul S, Karadağ G, et al. Effect of Ramadan fasting on Glycaemic parameters & body mass index in type II diabetic patients: A meta-analysis. Indian J Med Res 2019;150:546. 10.4103/ijmr.IJMR_1380_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Faris MA-I, Jahrami H, BaHammam A, et al. A systematic review, meta-analysis, and meta-regression of the impact of diurnal intermittent fasting during Ramadan on Glucometabolic markers in healthy subjects. Diabetes Research and Clinical Practice 2020;165:108226. 10.1016/j.diabres.2020.108226 [DOI] [PubMed] [Google Scholar]
- 74. Aliasghari F, Izadi A, Gargari BP, et al. The effects of Ramadan fasting on body composition, blood pressure, glucose metabolism, and markers of inflammation in NAFLD patients: an observational trial. Journal of the American College of Nutrition 2017;36:640–5. 10.1080/07315724.2017.1339644 [DOI] [PubMed] [Google Scholar]
- 75. Faris M, Jahrami H, Abdelrahim D, et al. The effects of Ramadan intermittent fasting on liver function in healthy adults: A systematic review, meta-analysis, and meta-regression. Diabetes Res Clin Pract 2021;178:108951. 10.1016/j.diabres.2021.108951 [DOI] [PubMed] [Google Scholar]
- 76. Faris MA-IE, Jahrami HA, Obaideen AA, et al. Impact of diurnal intermittent fasting during Ramadan on inflammatory and oxidative stress markers in healthy people: systematic review and meta-analysis. Journal of Nutrition & Intermediary Metabolism 2019;15:18–26. 10.1016/j.jnim.2018.11.005 [DOI] [Google Scholar]
- 77. Ünalacak M, Kara İH, Baltaci D, et al. Effects of Ramadan fasting on biochemical and hematological parameters and Cytokines in healthy and obese individuals. Metabolic Syndrome and Related Disorders 2011;9:157–61. 10.1089/met.2010.0084 [DOI] [PubMed] [Google Scholar]
- 78. Faris M-I, Kacimi S, Al-Kurd RA, et al. Intermittent fasting during Ramadan attenuates proinflammatory Cytokines and immune cells in healthy subjects. Nutr Res 2012;32:947–55. 10.1016/j.nutres.2012.06.021 [DOI] [PubMed] [Google Scholar]
- 79. Hajek P, Myers K, Dhanji A-R, et al. Weight change during and after Ramadan fasting. J Public Health (Bangkok) 2012;34:377–81. 10.1093/pubmed/fdr087 [DOI] [PubMed] [Google Scholar]
- 80. Gaeini Z, Mirmiran P, Bahadoran Z. Effects of Ramadan intermittent fasting on Leptin and adiponectin: A systematic review and meta-analysis. Hormones (Athens) 2021;20:237–46. 10.1007/s42000-021-00285-3 [DOI] [PubMed] [Google Scholar]
- 81. Bogdan A, Bouchareb B, Touitou Y. Response of circulating Leptin to Ramadan daytime fasting: a circadian study. Br J Nutr 2005;93:515–8. 10.1079/bjn20041380 [DOI] [PubMed] [Google Scholar]
- 82. Strohacker K, McCaffery JM, MacLean PS, et al. Adaptations of Leptin, Ghrelin or insulin during weight loss as predictors of weight regain: a review of current literature. Int J Obes 2014;38:388–96. 10.1038/ijo.2013.118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Bogdan A, Bouchareb B, Touitou Y. Ramadan fasting alters endocrine and Neuroendocrine circadian patterns. meal–time as a Synchronizer in humans Life Sciences 2001;68:1607–15. 10.1016/S0024-3205(01)00966-3 [DOI] [PubMed] [Google Scholar]
- 84. Mindikoglu AL, Abdulsada MM, Jain A, et al. Intermittent fasting from dawn to sunset for 30 consecutive days is associated with anticancer Proteomic signature and Upregulates key regulatory proteins of glucose and lipid metabolism, circadian clock, DNA repair, cytoskeleton remodeling, immune system and cognitive function in healthy subjects. J Proteomics 2020;217:103645. 10.1016/j.jprot.2020.103645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Longo VD, Mattson MP. Fasting: molecular mechanisms and clinical applications. Cell Metabolism 2014;19:181–92. 10.1016/j.cmet.2013.12.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Redman LM, Smith SR, Burton JH, et al. Metabolic slowing and reduced oxidative damage with sustained caloric restriction support the rate of living and oxidative damage theories of aging. Cell Metabolism 2018;27:805–815. 10.1016/j.cmet.2018.02.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. de Cabo R, Mattson MP. Effects of intermittent fasting on health, aging, and disease. N Engl J Med 2019;381:2541–51. 10.1056/NEJMra1905136 [DOI] [PubMed] [Google Scholar]
- 88. Di Francesco A, Di Germanio C, Bernier M, et al. A time to fast. Science 2018;362:770–5. 10.1126/science.aau2095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Gälman C, Lundåsen T, Kharitonenkov A, et al. The circulating metabolic regulator Fgf21 is induced by prolonged fasting and PPARα activation in man. Cell Metab 2008;8:169–74. 10.1016/j.cmet.2008.06.014 [DOI] [PubMed] [Google Scholar]
- 90. Charlot A, Hutt F, Sabatier E, et al. Beneficial effects of early time-restricted feeding on metabolic diseases: importance of Aligning food habits with the circadian clock. Nutrients 2021;13:1405. 10.3390/nu13051405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Kim YH, Lee JH, Yeung JL-H, et al. Thermogenesis-independent metabolic benefits conferred by Isocaloric intermittent fasting in OB/OB mice. Sci Rep 2019;9:1–10. 10.1038/s41598-019-39380-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Baumeier C, Kaiser D, Heeren J, et al. Caloric restriction and intermittent fasting alter hepatic lipid droplet proteome and diacylglycerol species and prevent diabetes in NZO mice. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 2015;1851:566–76. 10.1016/j.bbalip.2015.01.013 [DOI] [PubMed] [Google Scholar]
- 93. Goodrick CL, Ingram DK, Reynolds MA, et al. Effects of intermittent feeding upon body weight and LifeSpan in inbred mice: interaction of genotype and age. Mech Ageing Dev 1990;55:69–87. 10.1016/0047-6374(90)90107-q [DOI] [PubMed] [Google Scholar]
- 94. Kim K-H, Kim YH, Son JE, et al. Intermittent fasting promotes Adipose Thermogenesis and metabolic homeostasis via VEGF-mediated alternative activation of macrophage. Cell Res 2017;27:1309–26. 10.1038/cr.2017.126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Li G, Xie C, Lu S, et al. Intermittent fasting promotes white Adipose Browning and decreases obesity by shaping the gut Microbiota. Cell Metabolism 2017;26:672–685. 10.1016/j.cmet.2017.08.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. de Castro-de-Paiva P, Marinho T de S, Mandarim-de-Lacerda CA, et al. Intermittent fasting, high-intensity interval training, or a combination of both have beneficial effects in obese mice with Nonalcoholic fatty liver disease. J Nutr Biochem 2022;104:108997. 10.1016/j.jnutbio.2022.108997 [DOI] [PubMed] [Google Scholar]
- 97. Bartelt A, Heeren J. Adipose tissue Browning and metabolic health. Nat Rev Endocrinol 2014;10:24–36. 10.1038/nrendo.2013.204 [DOI] [PubMed] [Google Scholar]
- 98. Barquissau V, Léger B, Beuzelin D, et al. Caloric restriction and diet-induced weight loss do not induce Browning of human subcutaneous white Adipose tissue in women and men with obesity. Cell Rep 2018;22:1079–89. 10.1016/j.celrep.2017.12.102 [DOI] [PubMed] [Google Scholar]
- 99. Elsayed HRH, El-Nablaway M, Khattab BA, et al. Independent of calorie intake, short-term alternate-day fasting Alleviates NASH, with modulation of markers of Lipogenesis. J Histochem Cytochem 2021;69:575–96. 10.1369/00221554211041607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Yang W, Cao M, Mao X, et al. Alternate-day fasting protects the livers of mice against high-fat diet–induced inflammation associated with the suppression of toll-like receptor 4/nuclear factor ΚB signaling. Nutr Res 2016;36:586. 10.1016/j.nutres.2016.02.001 [DOI] [PubMed] [Google Scholar]
- 101. Chaudhary R, Liu B, Bensalem J, et al. Intermittent fasting activates markers of Autophagy in mouse liver, but not muscle from mouse or humans. Nutrition 2022;101:111662. 10.1016/j.nut.2022.111662 [DOI] [PubMed] [Google Scholar]
- 102. Marinho T de S, Ornellas F, Barbosa-da-Silva S, et al. Beneficial effects of intermittent fasting on steatosis and inflammation of the liver in mice Fed a high-fat or a high-fructose diet. Nutrition 2019;65:103–12. 10.1016/j.nut.2019.02.020 [DOI] [PubMed] [Google Scholar]
- 103. Sokolović A, van Roomen CPAA, Ottenhoff R, et al. Fasting reduces liver fibrosis in a mouse model for chronic cholangiopathies. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2013;1832:1482–91. 10.1016/j.bbadis.2013.05.012 [DOI] [PubMed] [Google Scholar]
- 104. Hatori M, Vollmers C, Zarrinpar A, et al. Time-restricted feeding without reducing caloric intake prevents metabolic diseases in mice Fed a high-fat diet. Cell Metab 2012;15:848–60. 10.1016/j.cmet.2012.04.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Chaix A, Zarrinpar A, Miu P, et al. Time-restricted feeding is a preventative and therapeutic intervention against diverse nutritional challenges. Cell Metabolism 2014;20:991–1005. 10.1016/j.cmet.2014.11.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Chung H, Chou W, Sears DD, et al. Time-restricted feeding improves insulin resistance and hepatic steatosis in a mouse model of postmenopausal obesity. Metabolism 2016;65:1743–54. 10.1016/j.metabol.2016.09.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Wilson RB, Zhang R, Chen YJ, et al. Two-week Isocaloric time-restricted feeding decreases liver inflammation without significant weight loss in obese mice with non-alcoholic fatty liver disease. IJMS 2020;21:9156. 10.3390/ijms21239156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Briand F, Grasset E, Breyner N, et al. 272-OR: weight loss with time-restricted feeding or treatment with Obeticholic acid/Semaglutide combination have a different impact on Nonalcoholic Steatohepatitis in diet-induced obese mice. Diabetes 2022;71.(Supplement_1) 10.2337/db22-272-OR [DOI] [Google Scholar]
- 109. Vollmers C, Gill S, DiTacchio L, et al. Time of feeding and the intrinsic circadian clock drive rhythms in hepatic gene expression. Proc Natl Acad Sci USA 2009;106:21453–8. 10.1073/pnas.0909591106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Hepler C, Weidemann BJ, Waldeck NJ, et al. Time-restricted feeding mitigates obesity through Adipocyte Thermogenesis. Science 2022;378:276–84. 10.1126/science.abl8007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Chaix A, Lin T, Le HD, et al. Time-restricted feeding prevents obesity and metabolic syndrome in mice lacking a circadian clock. Cell Metabolism 2019;29:303–319. 10.1016/j.cmet.2018.08.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Schilperoort M, van den Berg R, Dollé MET, et al. Time-restricted feeding improves adaptation to chronically alternating light-dark cycles. Sci Rep 2019;9:7874. 10.1038/s41598-019-44398-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Crosby P, Hamnett R, Putker M, et al. Insulin/IGF-1 drives PERIOD synthesis to Entrain circadian rhythms with feeding time. Cell 2019;177:896–909. 10.1016/j.cell.2019.02.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Acosta-Rodríguez V, Rijo-Ferreira F, Izumo M, et al. Circadian alignment of early onset caloric restriction promotes longevity in male C57Bl/6J mice. Science 2022;376:1192–202. 10.1126/science.abk0297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Stekovic S, Hofer SJ, Tripolt N, et al. Alternate day fasting improves physiological and molecular markers of aging in healthy, non-obese humans. Cell Metab 2019;30:462–76. 10.1016/j.cmet.2019.07.016 [DOI] [PubMed] [Google Scholar]
- 116. Puchalska P, Crawford PA. Multi-dimensional roles of ketone bodies in fuel metabolism, signaling, and Therapeutics. Cell Metab 2017;25:262–84. 10.1016/j.cmet.2016.12.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Varady KA, Cienfuegos S, Ezpeleta M, et al. Clinical application of intermittent fasting for weight loss: progress and future directions. Nat Rev Endocrinol 2022;18:309–21. 10.1038/s41574-022-00638-x [DOI] [PubMed] [Google Scholar]
- 118. Elortegui Pascual P, Rolands MR, Eldridge AL, et al. A Meta‐Analysis comparing the effectiveness of alternate day fasting, the 5: 2 diet, and Time‐Restricted eating for weight loss. Obesity 2023;31:9–21. 10.1002/oby.23568 Available: https://onlinelibrary.wiley.com/toc/1930739x/31/S1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Catenacci VA, Pan Z, Ostendorf D, et al. A randomized pilot study comparing Zero‐Calorie Alternate‐Day fasting to daily caloric restriction in adults with obesity. Obesity 2016;24:1874–83. 10.1002/oby.21581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Harvie MN, Pegington M, Mattson MP, et al. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: a randomized trial in young overweight women. Int J Obes 2011;35:714–27. 10.1038/ijo.2010.171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Trepanowski JF, Kroeger CM, Barnosky A, et al. Effects of alternate-day fasting or daily calorie restriction on body composition, fat distribution, and circulating Adipokines: secondary analysis of a randomized controlled trial. Clinical Nutrition 2018;37:1871–8. 10.1016/j.clnu.2017.11.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Carter S, Clifton PM, Keogh JB. Effect of intermittent compared with continuous energy restricted diet on Glycemic control in patients with type 2 diabetes: a randomized Noninferiority trial. JAMA Netw Open 2018;1:e180756. 10.1001/jamanetworkopen.2018.0756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Patikorn C, Roubal K, Veettil SK, et al. Intermittent fasting and obesity-related health outcomes: an umbrella review of meta-analyses of randomized clinical trials. JAMA Netw Open 2021;4:e2139558. 10.1001/jamanetworkopen.2021.39558 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Wang X, Yang Q, Liao Q, et al. Effects of intermittent fasting diets on plasma concentrations of inflammatory biomarkers: a systematic review and meta-analysis of randomized controlled trials. Nutrition 2020;79–80:110974. 10.1016/j.nut.2020.110974 [DOI] [PubMed] [Google Scholar]
- 125. Hoddy KK, Kroeger CM, Trepanowski JF, et al. Safety of alternate day fasting and effect on disordered eating behaviors. Nutr J 2015;14:1–3. 10.1186/s12937-015-0029-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Horne BD, Grajower MM, Anderson JL. Limited evidence for the health effects and safety of intermittent fasting among patients with type 2 diabetes. JAMA 2020;324:341. 10.1001/jama.2020.3908 [DOI] [PubMed] [Google Scholar]
- 127. Corley BT, Carroll RW, Hall RM, et al. Intermittent fasting in type 2 diabetes mellitus and the risk of Hypoglycaemia: a randomized controlled trial. Diabet Med 2018;35:588–94. 10.1111/dme.13595 Available: http://doi.wiley.com/10.1111/dme.2018.35.issue-5 [DOI] [PubMed] [Google Scholar]
- 128. Holmer M, Lindqvist C, Petersson S, et al. Treatment of NAFLD with intermittent calorie restriction or low-Carb high-fat diet–a randomised controlled trial. JHEP Reports 2021;3:100256. 10.1016/j.jhepr.2021.100256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Headland ML, Clifton PM, Keogh JB. Effect of intermittent compared to continuous energy restriction on weight loss and weight maintenance after 12 months in healthy overweight or obese adults. Int J Obes 2019;43:2028–36. 10.1038/s41366-018-0247-2 [DOI] [PubMed] [Google Scholar]
- 130. Johari MI, Yusoff K, Haron J, et al. A randomised controlled trial on the effectiveness and adherence of modified alternate-day calorie restriction in improving activity of non-alcoholic fatty liver disease. Sci Rep 2019;9:11232. 10.1038/s41598-019-47763-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Cai H, Qin Y-L, Shi Z-Y, et al. Effects of alternate-day fasting on body weight and Dyslipidaemia in patients with non-alcoholic fatty liver disease: a randomised controlled trial. BMC Gastroenterol 2019;19:1–8. 10.1186/s12876-019-1132-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Berzigotti A, Albillos A, Villanueva C, et al. Effects of an intensive Lifestyle intervention program on portal hypertension in patients with cirrhosis and obesity: the Sportdiet study. Hepatology 2017;65:1293–305. 10.1002/hep.28992 [DOI] [PubMed] [Google Scholar]
- 133. Loomba R, Abdelmalek MF, Armstrong MJ, et al. Semaglutide 2· 4 mg once weekly in patients with non-alcoholic Steatohepatitis-related cirrhosis: a randomised, placebo-controlled phase 2 trial. The Lancet Gastroenterology & Hepatology 2023;8:511–22. 10.1016/S2468-1253(23)00068-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Tinsley GM, Forsse JS, Butler NK, et al. Time-restricted feeding in young men performing resistance training: A randomized controlled trial. Eur J Sport Sci 2017;17:200–7. 10.1080/17461391.2016.1223173 [DOI] [PubMed] [Google Scholar]
- 135. Domaszewski P, Konieczny M, Pakosz P, et al. Effect of a six-week times restricted eating intervention on the body composition in early elderly men with overweight. Sci Rep 2022;12:9816. 10.1038/s41598-022-13904-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Lai JC, Tandon P, Bernal W, et al. Malnutrition, Frailty, and Sarcopenia in patients with cirrhosis: 2021 practice guidance by the American Association for the study of liver diseases. Hepatology 2021;74:1611–44. 10.1002/hep.32049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Merli M, Berzigotti A, Zelber-Sagi S, et al. EASL clinical practice guidelines on nutrition in chronic liver disease. Journal of Hepatology 2019;70:172–93. 10.1016/j.jhep.2018.06.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Plank LD, Gane EJ, Peng S, et al. Nocturnal nutritional supplementation improves total body protein status of patients with liver cirrhosis: a randomized 12‐Month trial. Hepatology 2008;48:557–66. 10.1002/hep.22367 [DOI] [PubMed] [Google Scholar]
- 139. Cienfuegos S, Gabel K, Kalam F, et al. Effects of 4-and 6-H time-restricted feeding on weight and Cardiometabolic health: a randomized controlled trial in adults with obesity. Cell Metab 2020;32:366–78. 10.1016/j.cmet.2020.06.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Phillips NE, Mareschal J, Schwab N, et al. The effects of time-restricted eating versus standard dietary advice on weight, metabolic health and the consumption of processed food: a pragmatic randomised controlled trial in community-based adults. Nutrients 2021;13:1042. 10.3390/nu13031042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Schroder JD, Falqueto H, Mânica A, et al. Effects of time-restricted feeding in weight loss, metabolic syndrome and cardiovascular risk in obese women. J Transl Med 2021;19:1–11. 10.1186/s12967-020-02687-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Pureza IROM, Melo ISV, Macena ML, et al. Acute effects of time-restricted feeding in low-income women with obesity placed on Hypoenergetic diets: randomized trial. Nutrition 2020;77:110796. 10.1016/j.nut.2020.110796 [DOI] [PubMed] [Google Scholar]
- 143. Li C, Xing C, Zhang J, et al. Eight-hour time-restricted feeding improves endocrine and metabolic profiles in women with Anovulatory Polycystic ovary syndrome. J Transl Med 2021;19:1–9. 10.1186/s12967-021-02817-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Manoogian ENC, Chow LS, Taub PR, et al. Time-restricted eating for the prevention and management of metabolic diseases. Endocr Rev 2022;43:405–36. 10.1210/endrev/bnab027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Conley M, Le Fevre L, Haywood C, et al. Is two days of intermittent energy restriction per week a feasible weight loss approach in obese males? A randomised pilot study. Nutr Diet 2018;75:65–72. 10.1111/1747-0080.12372 Available: http://doi.wiley.com/10.1111/ndi.2018.75.issue-1 [DOI] [PubMed] [Google Scholar]
- 146. Sutton EF, Beyl R, Early KS, et al. Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with Prediabetes. Cell Metab 2018;27:1212–1221.S1550-4131(18)30253-5. 10.1016/j.cmet.2018.04.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Wilkinson MJ, Manoogian ENC, Zadourian A, et al. Ten-hour time-restricted eating reduces weight, blood pressure, and Atherogenic lipids in patients with metabolic syndrome. Cell Metab 2020;31:92–104. 10.1016/j.cmet.2019.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148. Jamshed H, Beyl RA, Della Manna DL, et al. Early time-restricted feeding improves 24-hour glucose levels and affects markers of the circadian clock, aging, and Autophagy in humans. Nutrients 2019;11:1234. 10.3390/nu11061234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149. Zhang L-M, Liu Z, Wang J-Q, et al. Randomized controlled trial for time-restricted eating in overweight and obese young adults. IScience 2022;25:104870. 10.1016/j.isci.2022.104870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Hutchison AT, Regmi P, Manoogian ENC, et al. Time‐Restricted feeding improves glucose tolerance in men at risk for type 2 diabetes: a randomized crossover trial. Obesity (Silver Spring) 2019;27:724–32. 10.1002/oby.22449 [DOI] [PubMed] [Google Scholar]
- 151. Ravussin E, Beyl RA, Poggiogalle E, et al. Early time-restricted feeding reduces appetite and increases fat oxidation but does not affect energy expenditure in humans. Obesity (Silver Spring) 2019;27:1244–54. 10.1002/oby.22518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. Liu J, Yi P, Liu F. The effect of early time-restricted eating vs. later time-restricted eating on weight loss and metabolic health: A network meta-analysis of randomized controlled trials. J Clin Endocrinol Metab 2023;dgad036:dgad036. 10.1210/clinem/dgad036 [DOI] [PubMed] [Google Scholar]
- 153. Simon SL, Blankenship J, Manoogian ENC, et al. The impact of a self-selected time restricted eating intervention on eating patterns, sleep, and late-night eating in individuals with obesity. Front Nutr 2022;9:1007824. 10.3389/fnut.2022.1007824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Zhao L, Hutchison AT, Liu B, et al. Time–restricted eating alters the 24‐Hour profile of Adipose tissue Transcriptome in men with obesity. Obesity (Silver Spring) 2023;31 Suppl 1(Suppl 1):63–74. 10.1002/oby.23499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Parr EB, Devlin BL, Radford BE, et al. A delayed morning and earlier evening time-restricted feeding protocol for improving Glycemic control and dietary adherence in men with overweight/obesity: a randomized controlled trial. Nutrients 2020;12:505. 10.3390/nu12020505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156. Xie Z, Sun Y, Ye Y, et al. Randomized controlled trial for time-restricted eating in healthy volunteers without obesity. Nat Commun 2022;13:1003. 10.1038/s41467-022-28662-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Song Z, Xiaoli AM, Yang F. Regulation and metabolic significance of de novo Lipogenesis in Adipose tissues. Nutrients 2018;10:1383. 10.3390/nu10101383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. Lundell LS, Parr EB, Devlin BL, et al. Time-restricted feeding alters lipid and amino acid metabolite Rhythmicity without Perturbing clock gene expression. Nat Commun 2020;11:4643. 10.1038/s41467-020-18412-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Kesztyüs D, Fuchs M, Cermak P, et al. Associations of time-restricted eating with health-related quality of life and sleep in adults: a secondary analysis of two pre-post pilot studies. BMC Nutr 2020;6:76.:76. 10.1186/s40795-020-00402-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160. Marot LP, Balieiro lct, Lopes T d. VC, et al. n.d. Meal timing variability of rotating shift workers throughout a complete shift cycle and its effect on daily energy and Macronutrient intake: a field study [European journal of nutrition]. [DOI] [PubMed]
- 161. Manoogian ENC, Zadourian A, Lo HC, et al. Feasibility of time-restricted eating and impacts on Cardiometabolic health in 24-H shift workers: the healthy heroes randomized control trial. Cell Metabolism 2022;34:1442–1456. 10.1016/j.cmet.2022.08.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162. European Association for the Study of the Liver (EASL), European Association for the Study of Diabetes (EASD), European Association for the Study of Obesity (EASO) . EASL-EASD-EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease. J Hepatol 2016;64:1388–402. 10.1016/j.jhep.2015.11.004 [DOI] [PubMed] [Google Scholar]
- 163. Rinella ME, Neuschwander-Tetri BA, Siddiqui MS, et al. AASLD practice guidance on the clinical assessment and management of Nonalcoholic fatty liver disease. Hepatology 2023;77:1797–835. 10.1097/HEP.0000000000000323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. Younossi ZM, Corey KE, Lim JK. AGA clinical practice update on Lifestyle modification using diet and exercise to achieve weight loss in the management of Nonalcoholic fatty liver disease: expert review. Gastroenterology 2021;160:912–8. 10.1053/j.gastro.2020.11.051 [DOI] [PubMed] [Google Scholar]
- 165. Eslam M, Sarin SK, Wong VW-S, et al. The Asian Pacific Association for the study of the liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease. Hepatol Int 2020;14:889–919. 10.1007/s12072-020-10094-2 [DOI] [PubMed] [Google Scholar]
- 166. McPherson S, Armstrong MJ, Cobbold JF, et al. Quality standards for the management of non-alcoholic fatty liver disease (NAFLD): consensus recommendations from the British Association for the study of the liver and British society of Gastroenterology NAFLD special interest group. The Lancet Gastroenterology & Hepatology 2022;7:755–69. 10.1016/S2468-1253(22)00061-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167. Yumuk V, Tsigos C, Fried M, et al. European guidelines for obesity management in adults. Obes Facts 2015;8:402–24. 10.1159/000442721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168. Jensen M, Ryan D, Apovian C, et al. In: AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. 2013. [DOI] [PubMed] [Google Scholar]
- 169. Committee ADAPP. Obesity and weight management for the prevention and treatment of type 2 diabetes: standards of medical care in Diabetes—2022. Diabetes Care 2022;45(Suppl 1):S113–24. 10.2337/dc22-S008 [DOI] [PubMed] [Google Scholar]