Abstract
PURPOSE:
The purpose of the study is to evaluate the safety and outcomes of corneal collagen cross-linking (CXL) and different CXL protocols in progressive keratoconus (PK) population at short and long-term.
MATERIALS AND METHODS:
A systematic review and meta-analysis was conducted. A total of eight literature databases were searched (up to February 15, 2022). Randomized controlled trials (RCTs) comparing CXL versus placebo/control or comparing different CXL protocols in the PK population were included. The primary objective was assessment of outcomes of CXL versus placebo and comparison of different CXL protocols in terms of maximum keratometry (Kmax) or Kmax change from baseline (Δ), spherical equivalent, best corrected visual acuity (BCVA), and central corneal thickness (CCT) in both at short term (6 months) and long term (1st, 2nd, and 3rd year or more). The secondary objective was comparative evaluation of safety. For the meta-analysis, the RevMan5.3 software was used.
RESULTS:
A total of 48 RCTs were included. Compared to control, CXL was associated with improvement in Δ Kmax at 1 year (4 RCTs, mean difference [MD], −1.78 [−2.71, −0.86], P = 0.0002) and 2 and 3 years (1 RCT); ΔBCVA at 1 year (7 RCTs, −0.10 [−0.14, −0.06], P < 0.00001); and Δ CCT at 1 year (2 RCTs) and 3 years (1 RCT). Compared to conventional CXL (C-CXL), deterioration in Δ Kmax, ΔBCVA and endothelial cell density was seen at long term in the transepithelial CXL (TE-CXL, chemical enhancer). Up to 2 years, there was no difference between TE-CXL using iontophoresis (T-ionto) and C-CXL. At 2 and 4 years, C-CXL performed better compared to accelerated CXL (A-CXL) in terms of improving Kmax. Although CCT was higher in the A-CXL arm at 2 years, there was no difference at 4 years. While exploring heterogeneity among studies, selection of control eye (fellow eye of the same patient vs. eye of different patient) and baseline difference in Kmax were important sources of heterogeneity.
CONCLUSION:
CXL outperforms placebo/control in terms of enhancing Kmax and CCT, as well as slowing disease progression over time (till 3 years). T-ionto protocol, on the other hand, performed similarly to C-CXL protocol up to 2 years.
Keywords: Collagen cross-linking, cornea, cross-linking, keratoconus, progressive keratoconus
Introduction
Keratoconus is a bilateral, noninflammatory progressive corneal ectasia,[1] resulting from alteration of corneal collagen fibrils,[1] manifesting as increased corneal curvature and irregular astigmatism.[2,3] In 2003, the Dresden protocol of riboflavin-assisted corneal collagen cross-linking (CXL) [Also known as conventional-CXL (C-CXL)] was introduced as treatment modality in keratoconus.[4] Owing to the epithelial debridement and prolonged ultraviolet (UV) A exposure-related side effects,[5-8] this protocol was later modified by subsequent authors and newer protocols came up, e.g., transepithelial CXL (TE-CXL),[9] iontophoresis-assisted TE-CXL (T-ionto),[9] accelerated CXL (A-CXL),[9] epithelium-disruption CXL,[9] corneal-reshaping and cross-linking (CRXL),[10] photorefractive intrastromal cross-linking (PiXL),[11] CXL combined with intracorneal ring segment (ICRS), Athens protocol,[12,13] and Cretan protocol. Although there are a few meta-analyses that have evaluated safety and efficacy of CXL in keratoconus, only few have addressed the same in a definite “progressive keratoconus (PK)” population. Among the meta-analyses, on analyzing the safety and efficacy of CXL in the “PK” population, only two were meta-analyses of RCTs; these are studies of Kobashi et al., 2017[14] (studies included up to December 2015) and Li et al.[15] (studies included up to September 31, 2014), whereas other meta-analyses also included observational studies. To date, there has not been a single meta-analysis available in the literature which evaluated the comparative safety and efficacy of different CXL protocols with evidence derived solely from RCTs. Furthermore, in most of the earlier meta-analysis, the pooled estimate of efficacy/safety was derived by combining data of different time points (combining short-term data [data of 3–6 months] with long-term data [data of 1 year or more]). However, looking into the dynamic nature of the disease (a time-dependent progressive condition) and collagen turnover rates of cornea,[2] simply pooling the results of different time points to give a pooled estimate is not justified. In this regard, we conducted this meta-analysis in which the evidence was derived solely from RCT data. The aim of this meta-analysis was to evaluate the effectiveness and safety of CXL, as well as to compare the safety and outcomes of different CXL protocols in the “PK” population over time (both at short term and long term).
The primary objective was to compare outcomes of CXL versus placebo and to compare the efficacy of different CXL protocols in terms of corneal topography (maximum keratometry, i.e., Kmax), refraction (spherical equivalent, i.e., SE), vision (best-corrected visual acuity, i.e., BCVA), and pachymetry (central corneal thickness, i.e., CCT) both at short term (6 months) and at long term (at 1 year, 2 years, 3 years, or more). The secondary objective was comparative evaluation of safety parameters (endothelial cell density [ECD] and adverse events). The null hypothesis for the meta-analysis is that there is no difference in safety and outcomes between CXL and control/sham as well as different CXL protocols in patients with PK at both short and long term.
Materials and Methods
This meta-analysis was performed in accordance with the reporting standards laid down by the “Cochrane” group,[16] and “PRISMA guidelines”[17] were also followed while reporting (PROSPERO registration CRD42021258032).
Inclusion criteria
Published RCTs (parallel-group design) comparing CXL versus placebo/sham/control or comparing different CXL protocols
Patients (≥14 years) with PK. PK is diagnosed if there is presence of any one of the following criteria: 1.0 D increase in steepest SimK, 1.0 D increase in refractive astigmatism, and 0.1-mm decrease in the back optic zone radius of the best-fitting contact lens in the last 1 year[18]
Studies reporting any or all of the efficacy and safety measures Kmax, SE, BCVA, CCT, ECD, and comparative adverse event profile.
Exclusion criteria
Nonrandomized studies, observational studies, case reports
Studies in which diagnosis of PK is not clear or not as per definition adopted in the study.
Different protocols of collagen cross-linking
For the study, details of different protocols are seen in eTable 1.
eTable 1.
Detail description of different corneal collagen cross-linking protocol used in the systematic review and meta-analysis
| CXL protocol | Salient features of the protocol |
|---|---|
| Conventional epi-off protocol (C-CXL) | First corneal epithelium is removed (7-9 mm), then isotonic riboflavin solution is applied over the area for 30 min, then cornea eradiated with UV-A (370 nm) wavelength, power 3 mW/cm2 or 5.4 J/cm2 surface radiation for 30 min (riboflavin drop applies throughout the eradiation period every 5 min)[1] |
| TE-CXL (TE-CXL) | In this procedure, corneal epithelium is not removed instead various modalities have been used to enhance riboflavin penetration through intact epithelium, e.g., TE-CXL using chemical enhancer (banzalkonium chloride, ethylenediamine tetraacetic acid, etc.), TE-CXL using T-Ionto/small electrical current through the tight junction of corneal epithelium (T-Ionto-CXL),TE-CXL using epithelium disruption (performing mechanical disruption of corneal epithelium e.g., Daya epithelial disruptor)[1] |
| A-CXL (A-CXL) | Utilizing “Bunsen-Roscoe Law of photochemical reciprocity” which states “Same photochemical effect can be achieved by reducing the irradiation interval but keeping the total energy level constant by a corresponding increase in the irradiation intensity,” i.e., high energy UV-A dose (up to 30 mW/cm2, total energy limit is kept constant at 5.4 J/cm2) applied for short duration[1] |
| CRXL | After application of riboflavin, a semi-scleral rigid contact lens is sutured over cornea which followed UV-A irradiation. During the UV-A eradiation, riboflavin drop is applied under contact lens every 5 min using a blunt cannula[2] |
| PiXL | Customized topography-guided photorefractive intrastromal cross-linking with asymmetrical treatment zones and variable treatment energies used in this technique. 9 mm of central epithelium is removed, 0.1% dextran-free riboflavin is applied every 2 min when and 1s on/1s off-pulsed 370 nm UV-A irradiation of 30 mW/cm2 applied (total duration 10 min)[3] |
| CXL along with ICRS | ICRS implanted in the mid-peripheral deep stroma to reduce central corneal curvature/regularizing and decreasing pathologic corneal steepening (e.g., Intacs , Ferrara Ring) which is combined with CXL procedure to stop the disease progression.[4] The optimal timing for ICRS implantation remains varies from author to author, e.g., simultaneous ICRS with CXL, CXL followed by ICRS, or ICRS followed by CXL, etc.[4] |
| Athens protocol | This protocol involves topography-guided transepithelial partial PRK followed by CXL.[5-10] |
| Cretan protocol | This protocol uses epithelium removal with t-PTK[11] |
CRXL=Corneal reshaping and crosslinking, PiXL=Photorefractive intrastromal cross-linking, ICRS=Intracorneal ring segment, CXL=Corneal collagen cross-linking, TE-CXL=Transepithelial-CXL, C-CXL=Conventional-CXL, A-CXL=Accelerated-CXL, T-Ionto=Iontophoresis, PRK=Photorefractive keratectomy, t-PTK=Transepithelial phototherapeutic keratectomy, UV-A=Ultra violet A
Database search
Two reviewers (HK and AB) independently searched PubMed, EMBASE, OVID, CINHAL, Web of Science, Scopus, Nature, and Cochrane-CENTRAL up to February 15, 2022, using Medical Subject Heading terms and the keywords “cross linking,” “cornea,” “collagen cross-linking,” “progressive keratoconus,” and “keratoconus” without language restriction. Only published studies were included for our study purpose.
Screening of relevant articles
“EndNote” was used to extract data extraction files after searching databases, and duplicates were removed. The file was then uploaded in Rayyan QCRI, a web-based app for systematic review. The title and abstract of all the studies were screened to identify relevant articles. Full text of relevant studies was further evaluated using predefined inclusion/exclusion criteria by HK and AB. In case of discrepancy, PS and BM were consulted, and the issue was resolved. [Detailed database search is presented in eTable 2].
eTable 2.
Detailed search strategy
| Key word | Search stratagy |
|---|---|
| Key word | ("corneal collagen cross-linking"):ti,ab,kw OR ("corneal collagen crosslinking"):ti,ab,kw OR ("corneal crosslinking"):ti,ab,kw OR ("collagen crosslink*"):ti,ab,kwOR ("collagen crosslinking"):ti,ab,kw (Word variations have been searched) ("collagen crosslinkage"):ti,ab,kw OR ("corneal crosslinkage"):ti,ab,kw OR ("corneal cross-linking"):ti,ab,kw OR ("C3-R Corneal cross-linking"):ti,ab,kw OR ("C3R corneal cross-linking"):ti,ab,kw (Word variations have been searched) ("Holcomb C3-R cross linking"):ti,ab,kw OR (CXL):ti,ab,kw OR (CCL):ti,ab,kw OR (C3R):ti,ab,kw OR ("corneal CXL"):ti,ab,kw (Word variations have been searched) #1 OR #2 OR #3 ("progressive keratoconus"):ti,ab,kw OR ("corneal ectasia"):ti,ab,kw OR ("conical cornea"):ti,ab,kw OR ("keratoconus"):ti,ab,kw (Word variations have been searched) MeSH descriptor: [Keratoconus] explode all trees #5 OR #6 #4 AND #7 Limit in trials |
CXL=Corneal collagen cross-linking, CCL=Central corneal thickness
Data extraction (selection and coding)
Data extraction was done independently by PS and AB. Data were entered into Review Manager (RevMan) [Version 5.0. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2008. (RevMan 5.4.1)] Software by PS after independent verification by BM, HK, and FH.
Statistical analysis
The meta-analysis was carried out using RevMan5.3 software. As appropriate, pooled-effect estimates included mean difference (MD) for continuous data and risk ratio (RR) for dichotomous data. Pooled data were represented as MD/RR (95% confidence interval [CI]). In the entire study, “N” represented number of RCTs, “n” represents number of eyes in the study arm, and ∆ represents change from baseline. I2 was used as a measure of heterogeneity. In case of significant heterogeneity (I2 > 50%), random-effect model was used; otherwise, fixed-effect model was used for the analysis.[19] In case of high heterogeneity, control eye selection (fellow eye of the same patient vs. eye of different patient), baseline difference in Kmax between the treatment groups, and percentage of riboflavin and dextran used were all used to investigate high heterogeneity. P < 0.05 was considered criteria for statistical significance. The Cochrane risk-of-bias tool for RCTs was used to assess the risk of bias of the included RCTs.[20]
Exploration of heterogeneity
In case of high heterogeneity, it was explored using control eye selection (same patient vs. eye of different patient), baseline difference in Kmax between the treatment groups, and percentage of riboflavin and dextran used.
Risk of bias
AB and PS separately evaluated the risk of bias utilizing Cochrane risk-of-bias tool for RCTs.[20] Publication bias was not evaluated by funnel plot as there was less than 10 studies for any comparison.[21]
Results
Search results and characteristics of included studies
A total of 564 nonduplicate studies were screened using title and abstract, among which full-text evaluation was done for 76 relevant studies. Finally, 48 RCTs fulfilling predefined inclusion/exclusion criteria were included in our meta-analysis. The different comparisons in these RCTs were CXL versus control/placebo[2,18,22-36] (N = 17), TE-CXL (using chemical enhancer [CE]) versus C-CXL[6-8,37-43] (N = 10), T-ionto-CXL versus C-CXL[44-47] (N = 4), A-CXL versus C-CXL[48-57] (N = 10), PiXL versus A-CXL[11] (N = 1), CRXL versus C-CXL[10] (N = 1), and CXL versus CXL combined with other refractive procedure[58-62] (N = 5). PRISMA flowchart and details of included studies are shown in eFigure 1 (112.9KB, tif) and eTable 3 (1.7MB, tif) .
Overall, the quality of the included RCTs was good, but high risk of bias was seen in the domain of blinding of participants and personals (performance bias). Again, blinding of outcome assessment (detection bias) was unclear in few of the RCTs [eFigure 2 (58.3KB, tif) ].
Comparative outcome evaluation: Collagen cross-linking versus placebo/control
Short term
At 6 months, there was no significant difference in Kmax between the placebo/control arm and the CXL arm (2 RCTs,[2,25] n = 141 eyes in CXL, n = 142 eyes in control arm; MD − 1.87 [95% CI − 3.89, 0.16], I2 = 0%, fixed-effect model) [Table 1].
Table 1.
Comparative outcomes of different corneal collagen cross-linking protocols in patients with progressive keratoconus
| Comparison of different protocols | Parameter | Number of studies | Sample size | Model (fixed versus random effect) | MD/SMD (95% CI) | P | Heterogeneity | |||
|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||
| χ2/df | P | I2 (%) | ||||||||
|
| ||||||||||
| A | B | |||||||||
| CXL (A) versus control/placebo (B) | Kmax at 6 months[1,2] | 2 | 141 | 142 | Fixed | −1.87 (−3.89-0.16) | 0.07 | 0.31/1 | 0.58 | 0 |
| ∆Thinnest corneal thickness at 1 year | 2 | 71 | 63 | Random | −4.23 (−32.33-23.87) | 0.77 | 11.51/1 | 0.0007 | 91 | |
| TE-CXL (using chemical enhancer, A) versus C-CXL (B) | Kmax at 6 months[3,4] | 2 | 55 | 46 | Fixed | −0.83 (−3.04-1.38) | 0.46 | 0.06/1 | 0.80 | 0 |
| BCVA at 6 months[3-6] | 4 | 115 | 106 | Fixed | 0.02 (−0.01-0.04) | 0.13 | 2.81/3 | 0.42 | 0 | |
| CCT at 6 months[3-6] | 4 | 115 | 106 | Fixed | 3.47 (−1.76-8.70) | 0.19 | 3.53/3 | 0.32 | 15 | |
| SE at 6 months[3-6] | 4 | 75 | 66 | Fixed | −2.87 (−10.50-4.77) | 0.46 | 213.69/2 | <0.00001 | 99 | |
| SE at 1-year[3-5,7] | 4 | 105 | 96 | Random | 0.34 (−1.05-1.74) | 0.63 | 11/3 | 0.01 | 73 | |
| CCT at 1-year[3-5,7] | 4 | 105 | 96 | Fixed | 0.98 (−4.39-6.35) | 0.72 | 0.55/3 | 0.91 | 0 | |
| T-Ionto (enhanced using T-Ionto, A) versus C-CXL (B) | BCVA at 6 month[8,9] | 2 | 98 | 95 | Random | −0.03 (−0.12-0.05) | 0.43 | 2.44/1 | 0.12 | 59 |
| CCT at 6 month[8,9] | 2 | 98 | 95 | Fixed | −5.51 (−16.46-5.45) | 0.32 | 0.36/1 | 0.55 | 0 | |
| BCVA at 1-year[8,9] | 2 | 98 | 85 | Fixed | −0.02 (−0.07-0.03) | 0.90 | 0.6/1 | 0.51 | 0 | |
| CCT at 1-year[8,9] | 2 | 98 | 85 | Fixed | 0.75 (−11.09-12.59) | 0.94 | 0.01/1 | 0.94 | 0 | |
| BCVA at 2 year[8,10] | 2 | 98 | 85 | Fixed | 0.00 (−0.07-0.06) | 0.98 | 0.38/1 | 0.54 | 0 | |
| CCT at 2 year[8,10] | 2 | 98 | 85 | Fixed | −1.36 (−10.09-7.38) | 0.76 | 0.88/1 | 0.35 | 0 | |
| A-CXL (A) versus C-CXL | BCVA at 6 month[11-15] | 5 | 153 | 151 | Fixed | 0.01 (−0.04-0.06) | 0.76 | 7.18/4 | 0.13 | 44 |
| SE at 6 months[11,12,15] | 3 | 62 | 64 | Fixed | 0.05 (−0.98-1.08) | 0.93 | 1.11/2 | 0.58 | 0 | |
| Kmax at 6 months[11,13-16] | 5 | 153 | 151 | Random | 0.09 (−1.35-1.53) | 0.90 | 12.82/4 | 0.01 | 69 | |
| CCT at 6 months[14,16] | 2 | 43 | 40 | Fixed | 16.20 (4.20-28.20) | 0.008 | 0.53/1 | 0.047 | 0 | |
| ∆thinnest corneal thickness[11,17] | 2 | 46 | 51 | Random | 0.09 (−1.35-1.53) | 0.90 | 12.82/1 | 0.01 | 69 | |
| BCVA at 1 year[13,14,18] | 3 | 106 | 102 | Fixed | −0.03 (−0.08-0.03) | 0.33 | 0.37/2 | 0.83 | 0 | |
| CCT at 1 year[14,18] | 2 | 29 | 26 | Fixed | 14.22 (−0.99-29.43) | 0.07 | 0.61/1 | 0.44 | 0 | |
Kmax=Maximum keratometry, ECD=Endothelial cell density, BCVA=Best-corrected visual acuity, SE=Spherical equivalence, CCT=Central corneal thickness, MD=Mean difference, CI=Confidence interval, CXL=Corneal collagen cross-linking, TE-CXL=Transepithelial CXL, C-CXL=Conventional CXL, A-CXL=Accelerated CXL, T-Ionto=Iontophoresis, SMD=Standard mean difference P<0.005 indicate statistically significant difference. ∆Indicate change from baseline
Two RCTs provided 6-month data on BCVA[2,25] and SE;[2,25] however, owing to high heterogeneity, data were not pooled in the meta-analysis. At 6 months, Bhattacharyya et al.[2] reported significant improvement in BCVA (LogMar) and SE in the CXL arm. Although Hersh et al.[25] found a trend of improvement in BCVA (number of letters, 38.1 ± 12.1 in the CXL arm [n = 102] and 35 ± 13.8 in the control arm [n = 103]) and SE (−3.9 ± 4.3 in the CXL arm [n = 102] and −4.8 ± 4.9 in the control arm [n = 103]), they did not mention whether the difference between the two groups was statistically significant or not.
Regarding CCT, no data were available. Regarding change of thinnest corneal thickness, single RCT reported change of thinnest corneal thickness (∆thinnest corneal thickness) at 6 months. No difference was found between CXL versus control group at the end of 6 months (P > 0.05). No data on CCT were available.
Long term
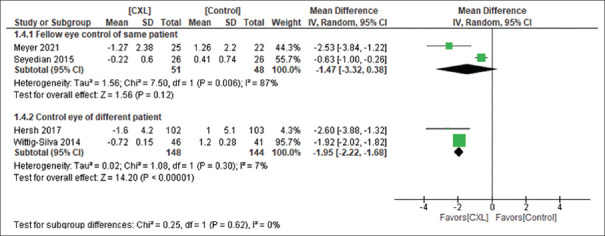
At 1 year, improvement was seen in terms of ∆Kmax (4 RCTs,[24,25,30,36] n = 199 in CXL and n = 192 in control, MD −1.78 [−2.71, −0.86], P = 0.0002, I2 = 94%, random-effect) [Figure 1] and ∆BCVA (7 RCTs,[18,22,24,28,30,35,36] n = 238 in CXL and n = 174 in the control arm, MD −0.10 [−0.14, −0.06], P < 0.00001, I2 = 45%, fixed-effect) in the CXL group as compared to control. However, no difference was seen between two groups in ∆SE (4 RCTs,[24,30,35,36] n = 107 in the CXL arm and N = 99 in control, MD 0.27 [−0.78, 1.33], P = 0.61, I2 = 80%, random-effect) [Figure 1].
Figure 1.
CXL versus control at 1 year in terms of Kmax, BCVA, SE and CCT. ∆Indicate change from baseline, Kmax = Maximum keratometry, BCVA = Best corrected visual acuity, SE = Spherical equivalence, CCT = Central corneal thickness, CXL = Corneal Collagen cross-linking
No difference was seen between two groups in change in thinnest corneal thickness (∆) at 1 year (2 RCTs,[24,36] n = 71 in CXL and n = 63 in the control arm, MD −4.23 [−32.33, 23.87], P = 0.77, I2 = 91%, random-effect) [Table 1]. Single RCT[30] reported no difference in ∆CCT (increase in CCT from baseline) between CXL versus control at 1 year (n = 26 in CXL and n = 61 in the control arm).
At 2 years (single RCT,[24] n = 44 in CXL, n = 31 in control), CXL arm continued to show benefits in terms of ∆Kmax (P < 0.01); however, no significant difference was observed in ∆CCT, ∆BCVA, and ∆SE.
At 3 years, a single RCT[24] (n = 41 in CXL, n = 27 in control) reported that the CXL arm had a significant improvement in ∆Kmax (P < 0.001) and ∆CCT (P < 0.013); however, no difference in ∆BCVA and ∆SE was seen.
Comparative outcome of different collagen cross-linking protocols
Transepithelial collagen cross-linking (chemical enhancer) versus conventional collagen cross-linking
Short term
At 6 months, there was no difference in Kmax between TE-CXL (CE) versus C-CXL in Kmax (2 RCTs,[8,37] n = 55 in TE-CXL, n = 46 in C-CXL; MD −0.83 [−3.04, 1.38], P = 0.46 I2 = 0%, fixed-effect), BCVA (4 RCTs,[8,37,41,42] n = 115 in TE-CXL, n = 106 in C-CXL, MD 0.02 [−0.01, 0.04], P = 0.13, I2 = 0%, fixed-effect), SE (3 RCTs,[8,37,41] n = 75 in TE-CXL, n = 66 in C-CXL, MD −2.87 [−10.50, 4.77], P = 0.46, I2 = 99%, random-effect), and CCT (4 RCTs,[8,37,41,42] n = 115 in TE-CXL, n = 106 in C-CXL, MD 3.47 [−1.76, 8.70], P = 0.19, I2 = 15%, fixed-effect) [Table 1].
No data were available from RCT regarding thinnest corneal thickness at 6 months.
Long-term
At 1 year, there was no difference in ∆Kmax (3 RCTs,[6-8] n = 79 in TE-CXL [CE], n = 72 in C-CXL, MD 1.01 [−0.31, 2.33], P = 0.13, I2 = 51%, random-effect), BCVA (4 RCTs,[7,8,37,42] n = 105 in TE-CXL, n = 96 in C-CXL, MD 0.05 [−0.03, 0.12], P = 0.59, I2 = 67%, random-effect), SE (4 RCTs,[7,8,37,42] n = 105 in TE-CXL, n = 96 in C-CXL, MD 0.34 [−01.05, 1.74], P = 0.63, I2 = 73%, random-effect), and CCT (4 RCTs,[7,8,37,42] n = 105 in TE-CXL, n = 96 in C-CXL, MD 0.98 [−4.39, 6.35], P = 0.91, I2 = 0%, fixed effect) [Table 1 and Figure 2].
Figure 2.
TE-CXL (CE) versus C-CXL protocol at 1 year in terms of ∆Kmax and BCVA. CE = Chemical enhancer, Kmax = Maximum keratometry, BCVA = Best-corrected visual acuity, CXL = Collagen cross-linking, TE-CXL = Transepithelial CXL, C-CXL = Conventional CXL
A single RCT compared TE-CXL versus C-CXL at 2 and 3 years[6] and it was found that ∆Kmax and ∆BCVA deteriorated significantly (P < 0.05) at 2 and 3 years in the TE-CXL (CE) group (indicating continued disease progression [deterioration of Kmax and BCVA]) compared to consistent improvement in the C-CXL group. Long-term (3-year) disease stabilization was achieved in the C-CXL arm in 100%, while only 45% of patients in the TE-CXL arm showed disease stabilization. In CCT,[6] there was no significant difference between the two protocols.
Transepithelial collagen cross-linking using iontophoresis versus conventional collagen cross-linking
Short term
At 6 months, no difference was found between T-ionto versus C-CXL in terms of BCVA (2 RCTs,[44,45] n = 98 in T-ionto-CXL, n = 85 in C-CXL, MD −0.03 [−0.12, 0.05], P = 0.9, I2 = 59%, random-effect model) and CCT (2 RCTs,[44,45] n = 98 in T-ionto-CXL, n = 85 in C-CXL, MD −5.51 [−16.46, 5.45], P = 0.32, I2 = 0%, fixed effect). A single RCT[47] (n = 22 eyes in CXL, n = 22 in control) found no differe0nce in Kmax (P = 0.72) and SE (P = 0.30). No data from RCT are available for thinnest corneal thickness.
Long term
At 1 year, there was no difference between T-ionto-CXL versus C-CXL in ∆Kmax (1 RCT,[44] ∆Kmax −0.52 ± 1.30 in T-ionto arm and −0.82 ± 1.20 in C-CXL arm, n = 22 in both arms, P = 0.53), BCVA (2 RCTs,[44,45] n = 98 in T-ionto-CXL, n = 85 in C-CXL, MD −0.02 [−0.07, 0.03], P = 0.9, I2 = 0%), and CCT (2 RCTs,[44,45] n = 98 in T-Ionto-CXL, n = 85 in C-CXL, MD 0.75 [−11.09, 12.59], P = 0.94, I2 = 0%) [Table 1]. No data from RCTs regarding thinnest corneal thickness were available.
At 2 years, there was no difference in Kmax (1 RCT,[46] n = 22 in T-ionto-CXL, n = 12 eyes in C-CXL, MD 0.5 [−2.67, 3.67], P = 0.76), SE (1 RCT,[46] n = 22 in T-ionto-CXL, n = 12 in C-CXL, MD 0.08 [−1.44, 1.28], P = 0.91), BCVA (2 RCTs,[45,46] n = 98 in T-ionto-CXL, n = 85 in C-CXL, MD 0.00 [−0.07, 0.06], I2 = 0%, P = 0.98), and CCT (2 RCTs,[45,46] n = 98 in T-ionto-CXL, n = 85 in C-CXL, MD −1.36 [−10.09, 7.38], I2 = 0%, P = 0.76) [Table 1].
Epithelium-disruption collagen cross-linking versus conventional collagen cross-linking
At 1 year (single RCT[38]), there was no difference between C-CXL (mechanical-removal) and epithelium-disruption CXL (using Daya epithelium disruptor) in Kmax and BCVA (P > 0.05), however, epithelium-disruption CXL was associated with significant improvement in SE (P < 0.05) and CCT (apex pachymetry, P < 0.05).
Accelerated collagen cross-linking versus conventional collagen cross-linking
Short term
At 6 months, there was no difference between A-CXL versus C-CXL in terms of Kmax (5 RCTs,[48,49,54,55,57] n = 153 in A-CXL, n = 151 in C-CXL, MD 0.09 [−1.35, 1.53], P = 0.90, I2 = 69%, random-effect), BCVA (5 RCTs,[48,49,51,54,57] n = 153 in A-CXL, n = 151 in C-CXL, MD 0.01 [−0.04, 0.06], P = 0.76, I2 = 44%, fixed-effect), and SE (3 RCTs,[51,54,57] n = 62 in A-CXL, n = 64 in C-CXL, MD 0.05 [−0.98, 1.08], P = 0.93, I2 = 0%, fixed-effect).
While pooling the pachymetry result, significant difference in terms of CCT (2 RCTs,[49,52] n = 43 in A-CXL, n = 40 in C-CXL, MD 16.20 [4.20, 28.20], P = 0.008, I2 = 0%, fixed-effect) and ∆ thinnest corneal thickness (2 RCTs,[50,54] n = 46 in A-CXL, n = 51 in C-CXL, MD 14.12 [0.42, 27.82], P = 0.03, I2 = 79%, random-effect) was seen at the end of 6 months favoring the A-CXL protocol over the C-CXL protocol [Table 1].
Long term
At 1 year, there was no difference between A-CXL versus C-CXL in Kmax (3 RCTs,[48,49,53] n = 106 in A-CXL, n = 102 in C-CXL, MD −0.67 [−2.24, 0.90], P = 0.41, I2 = 73%, random-effect) [Figure 3], SE (1 RCT,[53] n = 15 in A-CXL, n = 15 in C-CXL, MD 0.12 [−1.98, 2.22], P = 0.91, fixed-effect), BCVA (3 RCTs,[48,49,53] n = 106 in A-CXL, n = 102 in C-CXL, MD −0.03 [−0.08, 0.03], P = 0.33, I2 = 0%, fixed-effect), and CCT [2 RCTs,[49,53] n = 29 in A-CXL, n = 26 in C-CXL, MD 14.22 [−0.99, 29.43], P = 0.07, I2 = 0%) [Table 1]. Single RCT[50] reported that A-CXL has added advantage over C-CXL in terms of thinnest corneal thickness (∆) favoring A-CXL arm over the period of 1 year.
Figure 3.
A-CXL versus C-CXL protocol in terms of Kmax at 1 year. Kmax = Maximum keratometry, CXL = Collagen cross-linking, C-CXL = Conventional CXL, A-CXL = Accelerated CXL
At 2 years, a single RCT[56] (n = 32 in C-CXL, n = 27 in A-CXL group) observed disease progression (deterioration of Kmax by at least 1D) only in the A-CXL group (3 of 27 eyes, i.e., 11.1%), but no case of progression was noted in the C-CXL group indicating that, compared to A-CXL (9 mW/cm2), C-CXL was better in halting disease progression. In terms of BCVA (P = 0.351) and SE (P = 0.873),[56] no difference was seen between the two protocols. CCT and thinnest corneal thickness, on the other hand, were higher in the A-CXL group compared to C-CXL group (P = 0.004 and 0.006, respectively)[56] at 24 months.
C-CXL was associated with better anterior corneal flattening, i.e., improvement of Kmax (change in anterior 3 and 8 mm) at 4 years, compared to A-CXL (18 mW/cm2) (single RCT,[55] n = 27 in each arm, P < 0.05). However, no significant differences in CCT (P = 0.454), SE (P = 0.495), or BCVA (P = 0.537)[55] were found between groups.
Photorefractive intrastromal cross-linking versus collagen cross-linking
In a single RCT,[11] individualized customized PiXL (topography-guided) treatment centered on the ectatic zone was associated with significant improvement in Kmax (P < 0.05) and BCVA (P < 0.05), but no difference in SE (P = 0.03) was found at 6 and 12 months when compared to uniform 9 mm CXL (n = 25 in both arms).
Athens protocol and Cretan protocol versus conventional collagen cross-linking
Although many observational studies claim that the Athens protocol[13,63-67] and Cretan protocol[68] are superior to C-CXL, no RCT was available.
Corneal-reshaping and cross-linking versus conventional collagen cross-linking
Although a single RCT[10] documenting deeper densitometry in the C-CXL-arm compared to the CRXL arm (significant difference at 6 months, P < 0.001) is available, no data for our study parameters are available.
Collagen cross-linking in combination with other refractive surgery
Compared to C-CXL followed by ICRS, treatment with ICRS followed by CXL (mean interval between the two procedures: 7 months) was associated with significant improvement/benefit in BCVA and CCT (single RCT,[62] n = 24 in each group). When ICRS followed by CXL on the same day was compared with ICRS followed by C-CXL after 3 months (1 RCT,[61] n = 104 in same-day arm and n = 94 in 3-month arm), both protocols performed equally in terms of BCVA and Kmax at 6 months.
Compared to ICRS alone, ICRS followed by TE-CXL (CE) after 1 month showed significantly lower SE at 6 months (1 RCT,[59] n = 20 in each group, P = 0.004). However, when compared to ICRS alone, treatment with ICRS plus concurrent (same-day) A-CXL (single RCT,[60] n = 28 in ICRS, n = 19 in ICRS + A-CXL arm) did not result in additional benefit.
Secondary outcome (safety profile)
Endothelial cell density
There was no significant difference in ECD between CXL versus control/placebo at 6 months,[2] 1 year,[24,35] 2 years,[24,35] and 3 years;[24] T-ionto-CXL versus C-CXL[44,46,47] (∆ECD) at 6 months,[47] 1 year,[44] and 2 years;[46] A-CXL versus C-CXL at 6 months[48,50,51,57] and 1 year;[48,50,53,69] PiXL versus C-CXL at 6 months[11] and 1 year;[11] and TE-CXL (CE) versus C-CXL at 6 months.[8] However, compared to TE-CXL (CE), the C-CXL arm showed higher ECD at 1 year (2 RCTs,[7,8] MD −130 [−230 to −30], fixed effect, P = 0.01) [eTable 4].
eTable 4.
Comparison of endothelial cell density among different corneal collagen cross-linking protocols
| Comparison of different protocols | Endothelial cell density (cells/mm2) | Number of studies | Sample size | Study data (in case of single study data was not pooled and individual study data presented) | Mean difference (95% CI), model, P value (in case meta-analysis was conducted and data was pooled) | Heterogeneity parameters (χ2/df/P/I2) | |
|---|---|---|---|---|---|---|---|
|
| |||||||
| A | B | ||||||
| CXL (A)/control (B) | ECD 6 months[25] | 1 | 55 | 46 | Control: 2822±240, CXL: 2780±245, P=0.45 | NA | NA |
| ∆ECD 1 year[19] | 1 | 46 | 41 | Control: −65±49, CXL: 28±53, P=0.201 | NA | NA | |
| ∆ECD 2 years[19] | 1 | 44 | 31 | Control :17±57, CXL: 13±65, P=0.963 | NA | NA | |
| ∆ECD 3 years[19] | 1 | 41 | 27 | Control : −30±49, CXL: −35±50, P=0.941 | NA | NA | |
| TE-CXL (A)/C-CXL (B) | ECD 6 months[29,35] | 2 | 55 | 46 | NA | 72.05 (−20.67-164.77), fixed effect, 0.13 | 1.66/1/0.20/40% |
| ECD 1 year[34,35] | 2 | 45 | 36 | NA | −130 (−230-−30), fixed effect, 0.01 | 1/1/0.29/9% | |
| T-Ionto CXL (A)/C-CXL (B) | ECD 6 months[40] | 1 | 22 | 12 | T-Ionto: 2666±235, C-CXL: 2647±351, P>0.05 | NA | NA |
| ECD 1 year[40] | 1 | 22 | 12 | T-Ionto: 2675±311, C-CXL: 2658±321, P>0.05 | NA | NA | |
| ECD 24 months[39,42] | 2 | 98 | 85 | NA | 7.99 (−17-33), fixed effect, 0.54 | 0.00/1/0.99/0% | |
| A-CXL (A)/C-CXL (B) | ECD 6 months[44,45,50] | 3 | 126 | 125 | NA | 34 (−50-119), random effect, 0.43 | 4.7/2/0.1/57% |
| ∆ECD at 6 months[44,47] | 2 | 110 | 112 | NA | 19 (2.02-37), fixed effect, 0.03* | 0.25/2/0.62/0% | |
| ECD 1 years[44,48] | 2 | 92 | 91 | NA | −19 (−56-17), fixed effect, 0.31 | 0.79/1/0.38/0% | |
| ∆ECD at 1 year[44,47] | 2 | 110 | 112 | NA | 23 (5-41), fixed effect,0.009* | 0.35/1/0.56/0% | |
| PiXL (A)/CXL (B) | ∆ECD 6 months[3] | 1 | 25 | 25 | PiXL: 17±103, CXL: −12±101, P=0.42 | NA | NA |
| ∆ECD 1 year[3] | 1 | 25 | 25 | PiXL: −5±92, CXL: 6±66, P=0.45 | NA | NA | |
| CXL followed by ICRS (A)/ICRS followed by CXL (B) | ECD 6 months[53] | 1 | 24 | 24 | CXL followed by ICRS: 2559±133, ICRS followed by CXL: 2589±41, P<0.05* | NA | NA |
| CXL + ICRS (A)/riboflavin + ICRS (B) | ECD 2 years[54] | 1 | 19 | 20 | CXL + ICRS: 2687.4±270.4, riboflavin + ICRS: 2714.8±230.8, P>0.05 | NA | NA |
*P represents P value as presented in the original study. ∆Represents change from baseline, A and B represents respective protocols for that comparison. In case of single study, data was not pooled and represented as it is in the original study. In case of two or more studies, data was pooled and point estimate of mean difference was calculated. ECD=Endothelial cell density, CXL=Corneal collagen cross-linking, TE-CXL=Transepithelial-CXL, C-CXL=Conventional-CXL, A-CXL=Accelerated-CXL, T-Ionto=Iontophoresis, ICRS=Intra-corneal-ring-segment, PiXL=Photorefractive intra-stromal cross linking, CI=Confidence interval, NA=Not available
Adverse event
Compared to control/placebo, higher treatment-related adverse events (TAEs) were observed in CXL arm (mostly related to epithelium removal) with the most common being corneal haze.[25] Other TAEs were persistent epithelial defect/delayed healing, pain, striae,[25] corneal erosion, glare,[2] blurring of vision,[25] photophobia,[25] and contact lens intolerance [eTable 5]. In TAE, there was no significant difference between TE-CXL (CE) versus C-CXL,[8,37] T-ionto-CXL versus C-CXL,[44] A-CXL versus C-CXL, CRXL versus C-CXL, and PiXL versus C-CXL. In case of CXL combined with ICRS implantation, complications related to ICRS implantation such as extrusion of ICRS,[58,59,61] inflammation around ICRS necessitating removal,[58,61] glare,[61] and infectious keratitis[59] have been reported.
eTable 5.
Comparative treatment related adverse events in corneal collagen cross-linking versus control in patients with progressive keratoconus
| Adverse event (study reference) | Number of studies | Number of event/sample size | Model (fixed versus random effect) | RR (95% CI) | P | Heterogeneity | |||
|---|---|---|---|---|---|---|---|---|---|
|
|
|
||||||||
| CXL arm | Control arm | χ2/Df | P | I2 (%) | |||||
| Corneal haze[17,24,25, 13, 18] | 5 | 165/210 | 0/229 | Fixed | 75.07 (21.4-263.290) | <0.001 | 0.24/4 | 0.99 | 0 |
| Corneal scar[19,25] | 2 | 7/85 | 0/80 | Fixed | 7.7 (1.00-59.08) | 0.05 | 0.55/1 | 0.46 | 0 |
| Pain[24,25] | 2 | 32/141 | 0/141 | Fixed | 33 (4.59-237.36) | 0.0005 | 0.00/1 | 0.95 | 0 |
| Punctuate keratitis[24,25] | 2 | 26/141 | 0/141 | Random | 13.52 (0.67-274.84) | 0.001 | 2.05/1 | 0.15 | 51 |
| Persistent epithelial defect/delayed healing[24,25] | 2 | 24/141 | 0/141 | Fixed | 25 (3.42-182.62) | 0.002 | 1.92/1 | 0.17 | 48 |
| Striae[24] | 1 | 24/102 | 0/102 | NA | 49 (3.02-795.06) | 0.006 | NA | NA | NA |
| Corneal erosion[17] | 1 | 1/22 | 0/22 | Fixed | 3 (0.13-69.87) | 0.49 | NA | NA | NA |
| Contact lens intolerence[17] | 1 | 2/22 | 0/22 | NA | 5 (0.25-98.52) | 0.29 | NA | NA | NA |
| Glare[25] | 1 | 24/39 | 0/39 | NA | 49 (3.08-778) | 0.006 | NA | NA | NA |
| Blurring[24] | 1 | 16/102 | 0/102 | NA | 33 (2.01-542.78) | 0.01 | NA | NA | NA |
| Photophobia[24] | 1 | 11/102 | 0/102 | NA | 23 (1.37-385) | 0.03 | NA | NA | NA |
| Hyperemia[24] | 1 | 10/102 | 0/102 | NA | 21 (1.25-353) | 0.03 | NA | NA | NA |
| Irritation[24] | 1 | 10/102 | 0/102 | NA | 21 (1.25-353) | 0.03 | NA | NA | NA |
| Increased lacrimation[24] | 1 | 5/102 | 0/102 | NA | 11 (0.62-196) | 0.10 | NA | NA | NA |
| Dry eye[24] | 1 | 6/102 | 0/102 | NA | 13 (0.74-227) | 0.08 | NA | NA | NA |
| Corneal edema[19] | 1 | 1/46 | 0/41 | NA | 2.68 (0.11-64) | 0.54 | NA | NA | NA |
| Sub epithelial/sterile infiltrate[13,19] | 2 | 2/79 | 0/74 | Fixed | 2.84 (0.30-26.64) | 0.36 | 0.00/1 | 0.96 | 0 |
| Peripheral corneal vascularization[19] | 1 | 1/46 | 0/41 | NA | 2.68 (0.11-64) | 0.54 | NA | NA | NA |
| Anterior uveitis[13,19] | 2 | 2/79 | 0/74 | Fixed | 2.84 (0.30-26.64) | 0.36 | 0.00/1 | 0.96 | 0 |
CXL=Corneal collagen cross-linking, CI=Confidence interval, RR=Risk ratio, NA=Not available
Exploration of heterogeneity
In our study, in case of high heterogeneity (I2 > 50%), the cause of heterogeneity was explored on the basis of control eye selection, baseline intergroup difference in Kmax, and percentage of riboflavin and dextran used. A meta-regression analysis could not be performed because there were fewer than ten studies for each comparison.
Collagen cross-linking versus control
High heterogeneity was seen at 1 year in ∆Kmax and ∆SE. Selection of control eye could explain the high heterogeneity observed in both these cases [Figure 4 and eFigure 3 (115.9KB, tif) ].
Figure 4.
CXL versus control: ∆Kmax at 1 year subgroup analysis based on control eye selection (fellow eye of same patient vs. eye of different patient). Kmax = Maximum keratometry, CXL = Collagen cross-linking
Transepithelial collagen cross-linking (chemical enhancer) versus conventional collagen cross-linking
High heterogeneity was seen in BCVA and ∆Kmax at 1 year [Figure 2]. In the study by Rossi et al.,[7] there was significant difference in baseline uncorrected visual aquity (UCVA) and best corrected visual aquity (BCVA) between two groups. Hence, we conducted a sensitivity analysis by conducting the same analysis with excluding the study by Rossi et al.,[7] and it was found that the overall conclusion remained the same for BCVA, but the conclusion reversed in case of ∆Kmax, and interestingly, in both cases, heterogeneity decreased significantly (I2 = 0% in both) [eFigure 4a (162.8KB, tif) ]. Concentration of riboflavin or dextran or type of formulation of riboflavin (dextran or nondextran formulation) used could not explain the high heterogeneity observed in any of the cases [eFigure 4b (162.8KB, tif) ].
Accelerated collagen cross-linking versus conventional collagen cross-linking
High heterogeneity was seen in Kmax at 1 year and baseline intergroup-difference in Kmax (>1 D vs. <1 D) could explain the high heterogeneity observed [eFigure 5 (82.4KB, tif) ].
Quality of evidence using Grading of Recommendations Assessment, Development, and Evaluation approach
As the number of RCTs was less across many comparisons, quality of evidence was evaluated across few comparisons (CXL vs. control, A-CXL vs. C-CXL, and TE-CXL [CE] vs. C-CXL) for the primary outcome Kmax. As the studies were deficient in many parameters (allocation concealment, blinding), and the results were heterogeneous, the certainty of evidence ranged from low to very low. The details of the certainty of evidence[70] are shown in Table 2.
Table 2.
Evaluation of certainty of evidence using The Grading of Recommendations Assessment, Development, and Evaluation approach for few major comparisons against the outcome maximum keratometry
| Outcomes | Anticipated absolute effects (95% CI)* | Number of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
|---|---|---|---|---|---|
|
| |||||
| Risk with control | Risk with the intervention | ||||
| CXL versus control ∆Kmax at 1 year | The median ∆Kmax at 1 year in control was 1.1 | Intervention: CXL MD 1.78 lower (2.71 lower to 0.86 lower) | 391 (4 RCTs) | ⨁⨁◯◯ Lowa,b |
The evidence suggests CXL in PK reduces CXL versus control ∆Kmax at 1 year slightly |
| A-CXL versus C-CXL Kmax at 1 year | The median Kmax at 1 year in C-CXL was 50.2 | Intervention A-CXL MD 0.67 lower (2.24 lower to 0.9 higher) | 208 (3 RCTs) | ⨁◯◯◯Very lowc,d | The evidence is very uncertain about the effect of CXL in PK on A-CXL versus C-CXL Kmax at 1 year |
| TE-CXL (CE) versus C-CXL ∆Kmax at 1 year | The median ∆Kmax at 1 year in C-CXL was-1.5 | Intervention: TE-CXL (CE) MD 1.01 higher (0.31 lower to 2.33 higher) | 151 (3 RCTs) | ⨁⨁◯◯ Lowe |
The evidence suggests that CXL in PK does not increase TE-CXL (CE) versus C-CXL ∆Kmax at 1 year |
aDowngraded by one level for serious risk of bias due to some concerns in three out of four studies, bDowngraded by one level for serous inconsistency due to high I square value 94%, cDowngraded by one level serious risk of bias due to high risk of selection bias, dDowngraded by one level serious inconsistency I square value is 74%, eDowngraded by one level for serious risk of bias due to studies with some concerns on selection bias. CI=Confidence interval, CXL=Corneal collagen cross-linking, TE-CXL=Transepithelial CXL, C-CXL=Conventional CXL, A-CXL=Accelerated CXL, MD=Mean difference, CE=Chemical enhancer, RCTs=Randomized controlled trials, Kmax=Maximum keratometry, GRADE=The Grading of Recommendations Assessment, Development, and Evaluation, PK=Progressive keratoconus
Discussion
In our meta-analysis, the beneficial effects of CXL over placebo/control were not prominent at 6 months; however, CXL arm showed significant benefits at 1 year (in ∆Kmax, ∆BCVA, and ∆CCT), 2 years (∆Kmax), and 3 years (∆Kmax, ∆CCT) when compared to placebo/control. The improvement in Kmax seen at 1 year was maintained up to 3 years. Another improvement which became apparent at 3 years was improvement in ∆CCT.
The C-CXL protocol had substantial concerns with epithelium removal-related side effects. To overcome the same, many epithelium-on (transepithelial) protocols were developed, which used epithelial disruptors or different forms of enhancers (CEs [e.g., benzalkonium chloride, ethylene-diamine-tetra-acetic-acid] or iontophoresis) to increase riboflavin penetration. At 6 months and at 1 year, there was no difference in any of the study parameters when TE-CXL (CE) was compared to C-CXL except ECD. At 1 year, ECD was significantly higher in the C-CXL arm. In our meta-analysis, higher ECD has been seen in C-CXL protocol as compared to the transepithelial protocol which shows advantage of C-CXL protocol over the TE-CXL. Another recently published meta-analysis by Nath et al.[71] also reports similar results. The reason behind may be due to hypoosmolar riboflavin solution used in the TE-CXL protocol. Ocak et al.[72] in their study also reported lower level of ECD in the A-CXL group as compared to C-CXL group in case of thin cornea (<400 mm) and they hypothesized use of hypoosmolar riboflavin, which is used to increase the corneal thickness before UV application. Another study by Gu et al.[73] has also reported slight decrease in ECD after C-CXL with hypotonic riboflavin.[73] Wollensak et al.[74] in their animal studies in rabbit cornea have proved that CXL can cause significant endothelial cell necrosis and a complete loss of endothelial cells by using higher energy in rabbit corneas, with an average thickness of 400 μm. Hafezi et al. have recently published the protocol for keratoconus patients with sub-400 mm that is pachymetry guided but modified the duration of irradiation instead of the riboflavin frequency.[75]
At 2 and 3 years, TE-CXL (CE) arm showed continued progression (deterioration of Kmax and BCVA) with disease stabilization occurring in only 45% of patients at 3 -years, compared to 100% disease stabilization with C-CXL arm (single RCT).[6] There was no difference in efficacy between the T-ionto-CXL versus C-CXL protocol in both short term (6 months) and long term (1 year and 2 years). At 12 months, use of epithelial-disruption protocol was associated with significant benefit in terms of SE and CCT; however, no difference was found in Kmax and BCVA (single RCT).[38] Another meta-analysis of RCTs on corneal ectasia concluded that TE-CXL is inferior to epithelial-off CXL and disease progression was more in TE-CXL group; however, the TE-CXL was better in terms of safety.[71]
While comparing A-CXL versus C-CXL protocol, no difference was seen between the two protocols in any of the studied outcomes at 6 months and 1 year. Disease progression (progression of Kmax by more than 1 D) was seen in 11% in the A-CXL group at 2 years (single RCT[56]) but none in the C-CXL group.[56] On the other hand, CCT was higher in the A-CXL arm compared to C-CXL arm. At 4 years, C-CXL protocol continued to show benefit in terms of better anterior corneal flattening and Kmax; however, no difference in CCT was evident at this time point.[55] The higher efficacy found in C-CXL could be attributable to a larger cross-linking area, improved riboflavin penetration in the corneal stroma, and a longer exposure time.[76] However, we have limited data regarding comparative efficacy and safety of other protocols.
Among different combined protocols (CXL + other refractive procedure), ICRS followed by CXL (mean interval 7 months) demonstrated a benefit over its reverse protocol[62] in BCVA and CCT at 6 months. However, the optimal gap between the two procedures remains a knowledge gap, and further research is necessary.
In terms of safety, there was no significant difference in ECD between CXL and the placebo/control arm, but TAEs were more common after CXL with the majority of the side effects (postoperative discomfort, pain, and a decrease in visual parameters) was related to epithelium removal, but they were manageable with conservative measures.
While exploring heterogeneity among the study findings, it was found that selection of control eye (fellow eye of same patient vs. eye of different patient) and baseline difference in Kmax were important source of heterogeneity. Although a few observational studies[77,78] indicated that formulation (dextran versus hydroxypropyl methyl cellulose-based formulation) and concentration of riboflavin have a significant effect on CXL outcome, none were shown to be significant causes of heterogeneity in our meta-analysis.
Limitations
Only a few RCTs have looked at the long-term safety and effectiveness of CXL therapy. Again, the majority of RCTs did not provide data regarding posterior-corneal topography, which is an important indicator of keratoconus progression.[79]
Conclusion
CXL is superior to placebo/control in terms of better corneal-topography and CCT at long-term (till 3 years). At long term, the C-CXL protocol outperforms the TE-CXL (CE) and A-CXL protocols, with a decreased rate of disease progression. T-ionto protocol, on the other hand, performed similarly well as C-CXL protocol (up to 2 years). Other protocols (e.g., epithelial-disruption CXL, PiXL, CXL combined with other refractive surgery) may have some advantages, although there are only a few RCTs that compare them. To clearly characterize the comparative safety and results of these regimens, more RCTs are needed.
What was known?
Corneal CXL is effective in halting the disease progression on keratoconus population
Furthermore, in most previous meta-analyses, the pooled estimate of efficacy/safety was derived by combining data of different time points (combining short-term data [data of 3–6 months] with long-term data [data of 12 months or more]).
What this study adds?
This is the first meta-analysis which evaluated the comparative safety and efficacy of different CXL protocols using evidence derived solely from RCTs (44 RCTs) comparing the efficacy and safety of corneal CXL procedure in a well-defined PK population as compared to placebo/control both in long term and short term as well as a comparative efficacy and safety analysis of different CXL protocols
This is the first meta-analysis explaining the high heterogeneity among the included RCTs
Our meta-analysis reveals that CXL is associated with improvement in Kmax at long term (up to 3 years).Among different CXL protocols, C-CXL was better in halting disease progression at long term, while TE-CXL using iontophoresis was equieffective to C-CXL up to 2 years
Corneal CXL is safe and effective in halting disease progression in PK at long term (up to 3 years).
Access to data statement
PS and AB had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Financial support and sponsorship
Nil.
Conflicts of interest
The authors declare that there are no conflicts of interests of this paper.
PRISMA flow diagram of included randomized control trials
Characteristics of included randomized controlled trials
Risk of bias graph among included randomized control trials. “Cochrane Risk of Bias tool for randomized control trial” has been used for risk of bias assessment.[54] Six domains taken into account are: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other bias. In the risk of bias graph green color indicates low risk; yellow color indicates unclear; and red color indicates high risk of bias
CXL vs. control: ∆SE at 1 year subgroup analysis based on control eye selection (Fellow eye of same patient vs. eye of different patient). ∆Indicate change from baseline, CXL = Collagen cross-linking, SE = Spherical equivalence
(a) TE-CXL (CE) versus C-CXL: ∆Kmax at 1 year: Sensitivity analysis (excluding study by Rossi et al). (b) TE-CXL (CE) versus C-CXL: ∆Kmax at 1 year: Subgroup analysis based on different riboflavin formulation (dextran based riboflavin vs. no dextran riboflavin). CE = Chemical enhancer, Kmax = Maximum keratometry, CXL = Collagen cross-linking, TE-CXL = Transepithelial CXL, C-CXL = Conventional CXL
A-CXL versus C-CXL protocol: Kmax at 1 year: Subgroup analysis based on baseline Kmax difference (>1D vs. <1D). Kmax = Maximum keratometry, CXL = Collagen cross-linking, C-CXL = Conventional CXL, A-CXL = Accelerated CXL
Acknowledgments
The authors acknowledge Mr. Siris Kr Bhattacharyya and Mrs. Anima Bhattacharyya for their support.
Supplementary References
Subasinghe SK, Ogbuehi KC, Dias GJ. Current perspectives on corneal collagen crosslinking (CXL). Graefes Arch Clin Exp Ophthalmol 2018;256:1363-84.
Beckman Rehnman J, Behndig A, Hallberg P, Lindén C. Initial results from mechanical compression of the cornea during crosslinking for keratoconus. Acta Ophthalmol 2014;92:644-9.
Nordström M, Schiller M, Fredriksson A, Behndig A. Refractive improvements and safety with topography-guided corneal crosslinking for keratoconus: 1-year results. Br J Ophthalmol 2017;101:920-5.
Zhu AY, Jun AS, Soiberman US. Combined protocols for corneal collagen cross-linking with photorefractive surgery for refractive management of keratoconus: Update on Techniques and review of literature. Ophthalmol Ther 2019;8:15-31.
Krueger RR, Kanellopoulos AJ. Stability of simultaneous topography-guided photorefractive keratectomy and riboflavin/UVA cross-linking for progressive keratoconus: Case reports. J Refract Surg 2010;26: S827-32.
Kanellopoulos AJ. The impact of keratoconus treatment with the Athens Protocol (partial topography-guided photorefractive keratectomy combined with higher-fluence corneal collagen cross-linking) on quality of life: A long-term study. Clin Ophthalmol 2019;13:795-803.
Kanellopoulos AJ. Long term results of a prospective randomized bilateral eye comparison trial of higher fluence, shorter duration ultraviolet A radiation, and riboflavin collagen cross linking for progressive keratoconus. Clin Ophthalmol 2012;6:97-101.
Kanellopoulos AJ, Asimellis G. Comparison of Placido disc and Scheimpflug image-derived topography-guided excimer laser surface normalization combined with higher fluence CXL: The Athens protocol, in progressive keratoconus. Clin Ophthalmol 2013;7:1385-96.
Kanellopoulos AJ, Asimellis G. Keratoconus management: Long-term stability of topography-guided normalization combined with high-fluence CXL stabilization (the Athens protocol). J Refract Surg 2014;30:88-93.
Kontadakis GA, Kankariya VP, Tsoulnaras K, Pallikaris AI, Plaka A, Kymionis GD. Long-term comparison of simultaneous topography-guided photorefractive keratectomy followed by corneal cross-linking versus corneal cross-linking alone. Ophthalmology 2016;123:974-83.
Grentzelos MA, Liakopoulos DA, Siganos CS, Tsilimbaris MK, Pallikaris IG, Kymionis GD. Long-term comparison of combined t-PTK and CXL (Cretan Protocol) versus CXL with mechanical epithelial debridement for keratoconus. J Refract Surg 2019;35:650-5.
Meyer JJ, Jordan CA, Patel DV, Gokul A, Dean SJ, Ahmed S, et al. Five-year results of a prospective, randomised, contralateral eye trial of corneal crosslinking for keratoconus. Clin Exp Ophthalmol 2021;49:542-9.
Wittig-Silva C, Whiting M, Lamoureux E, Lindsay RG, Sullivan LJ, Snibson GR. A randomized controlled trial of corneal collagen cross-linking in progressive keratoconus: Preliminary results. J Refract Surg 2008;24: S720-5.
Greenstein SA, Fry KL, Hersh MJ, Hersh PS. Higher-order aberrations after corneal collagen crosslinking for keratoconus and corneal ectasia. J Cataract Refract Surg 2012;38:292-302.
Greenstein SA, Fry KL, Hersh PS. Corneal topography indices after corneal collagen crosslinking for keratoconus and corneal ectasia: One-year results. J Cataract Refract Surg 2011;37:1282-90.
Hersh PS, Greenstein SA, Fry KL. Corneal collagen crosslinking for keratoconus and corneal ectasia: One-year results. J Cataract Refract Surg 2011;37:149-60.
O’Brart DP, Chan E, Samaras K, Patel P, Shah SP. A randomised, prospective study to investigate the efficacy of riboflavin/ultraviolet A (370 nm) corneal collagen cross-linkage to halt the progression of keratoconus. Br J Ophthalmol 2011;95:1519-24.
Wittig-Silva C, Chan E, Pollock G, Snibson GR. Localized changes in stromal reflectivity after corneal collagen cross-linking observed with different imaging techniques. J Refract Surg 2013;29:410-6.
Wittig-Silva C, Chan E, Islam FM, Wu T, Whiting M, Snibson GR. A randomized, controlled trial of corneal collagen cross-linking in progressive keratoconus: Three-year results. Ophthalmology 2014;121:812-21.
Hallahan KM, Rocha K, Roy AS, Randleman JB, Stulting RD, Dupps WJ Jr. Effects of corneal cross-linking on ocular response analyzer waveform-derived variables in keratoconus and postrefractive surgery ectasia. Eye Contact Lens 2014;40:339-44.
Seyedian MA, Aliakbari S, Miraftab M, Hashemi H, Asgari S, Khabazkhoob M. Corneal collagen cross-linking in the treatment of progressive keratoconus: A randomized controlled contralateral eye study. Middle East Afr J Ophthalmol 2015;22:340-5.
Sharma N, Suri K, Sehra SV, Titiyal JS, Sinha R, Tandon R, et al. Collagen cross-linking in keratoconus in Asian eyes: Visual, refractive and confocal microscopy outcomes in a prospective randomized controlled trial. Int Ophthalmol 2015;35:827-32.
Kim BZ, Jordan CA, McGhee CN, Patel DV. Natural history of corneal haze after corneal collagen crosslinking in keratoconus using Scheimpflug analysis. J Cataract Refract Surg 2016;42:1053-9.
Hersh PS, Stulting RD, Muller D, Durrie DS, Rajpal RK, United States Crosslinking Study Group. United States multicenter clinical trial of corneal collagen crosslinking for keratoconus treatment. Ophthalmology 2017;124:1259-70.
Bhattacharyya A, Sarma P, Das K, Agarwal B, Medhi J, Das Mohapatra SS. Safety and efficacy of riboflavin-assisted collagen cross-linking of cornea in progressive keratoconus patients: A prospective study in North East India. Indian J Pharmacol 2019;51:157-67.
Henriquez MA, Izquierdo L Jr, Bernilla C, Zakrzewski PA, Mannis M. Riboflavin/ultraviolet A corneal collagen cross-linking for the treatment of keratoconus: Visual outcomes and Scheimpflug analysis. Cornea 2011;30:281-6.
Toro-Giraldo L, Morales Flores N, Santana-Cruz O, Ramirez-Miranda A, Navas A, Olivo-Payne A, et al. Cool cross-linking: Riboflavin at 4°C for pain management after cross-linking for patients with keratoconus, a randomized clinical trial. Cornea 2020;40:1-4.
Rosenblat E, Hersh PS. Intraoperative corneal thickness change and clinical outcomes after corneal collagen crosslinking: Standard crosslinking versus hypotonic riboflavin. J Cataract Refract Surg 2016;42:596-605.
Nawaz S, Gupta S, Gogia V, Sasikala NK, Panda A. Trans-epithelial versus conventional corneal collagen crosslinking: A randomized trial in keratoconus. Oman J Ophthalmol 2015;8:9-13.
Al Zubi K, Albakar Y, Nasser R. Transepithelial versus epithelium off crosslinking for treating keratoconus among Jordanians. Open Ophthalmol J 2019;13:8-14.
Cifariello F, Minicucci M, Di Renzo F, Di Taranto D, Coclite G, Zaccaria S, et al. Epi-off versus Epi-on corneal collagen cross-linking in keratoconus patients: A comparative study through 2-year follow-up. J Ophthalmol 2018;2018:4947983.
Stojanovic A, Zhou W, Utheim TP. Corneal collagen cross-linking with and without epithelial removal: A contralateral study with 0.5% hypotonic riboflavin solution. Biomed Res Int 2014;2014:619398.
Al Fayez MF, Alfayez S, Alfayez Y. Transepithelial versus epithelium-off corneal collagen cross-linking for progressive keratoconus: A prospective randomized controlled trial. Cornea 2015;34 Suppl 10: S53-6.
Rossi S, Orrico A, Santamaria C, Romano V, De Rosa L, Simonelli F, et al. Standard versus trans-epithelial collagen cross-linking in keratoconus patients suitable for standard collagen cross-linking. Clin Ophthalmol 2015;9:503-9.
Soeters N, Wisse RP, Godefrooij DA, Imhof SM, Tahzib NG. Transepithelial versus epithelium-off corneal cross-linking for the treatment of progressive keratoconus: A randomized controlled trial. Am J Ophthalmol 2015;159:821-8.e3.
Zaheryani. Corneal Collagen Cross-linking Using Epithelium Disruptor Instrument in Progressive Keratoconus. Available from: http://www.jcurrophthalmol.org/article.asp?issn=2452-2325;year=2020;volume=32;issue=3;spage=256;epage=262;aulast=Zaheryani. [Last accessed on 2020 Nov 10].
Lai MJ, Greenstein SA, Gelles JD, Hersh PS. Corneal haze after transepithelial collagen cross-linking for keratoconus: A scheimpflug densitometry analysis. Cornea 2020;39:1117-21.
Hersh PS, Lai MJ, Gelles JD, Lesniak SP. Transepithelial corneal crosslinking for keratoconus. J Cataract Refract Surg 2018;44:313-22.
Bikbova G, Bikbov M. Standard corneal collagen crosslinking versus transepithelial iontophoresis-assisted corneal crosslinking, 24 months follow-up: Randomized control trial. Acta Ophthalmol 2016;94:e600-6.
Lombardo M, Serrao S, Raffa P, Rosati M, Lombardo G. Novel technique of transepithelial corneal cross-linking using iontophoresis in progressive keratoconus. J Ophthalmol 2016;2016:7472542.
Lombardo M, Giannini D, Lombardo G, Serrao S. Randomized controlled trial comparing transepithelial corneal cross-linking using iontophoresis with the Dresden protocol in progressive keratoconus. Ophthalmology 2017;124:804-12.
Lombardo M, Serrao S, Lombardo G, Schiano-Lomoriello D. Two-year outcomes of a randomized controlled trial of transepithelial corneal crosslinking with iontophoresis for keratoconus. J Cataract Refract Surg 2019;45:992-1000.
Sherif AM. Accelerated versus conventional corneal collagen cross-linking in the treatment of mild keratoconus: A comparative study. Clin Ophthalmol 2014;8:1435-40.
Hashemian H, Jabbarvand M, Khodaparast M, Ameli K. Evaluation of corneal changes after conventional versus accelerated corneal cross-linking: A randomized controlled trial. J Refract Surg 2014;30:837-42.
Hashemi H, Fotouhi A, Miraftab M, Bahrmandy H, Seyedian MA, Amanzadeh K, et al. Short-term comparison of accelerated and standard methods of corneal collagen crosslinking. J Cataract Refract Surg 2015;41:533-40.
Hashemi H, Miraftab M, Seyedian MA, Hafezi F, Bahrmandy H, Heidarian S, et al. Long-term results of an accelerated corneal cross-linking protocol (18 mW/cm2) for the treatment of progressive keratoconus. Am J Ophthalmol 2015;160:1164-70.e1.
Shetty R, Pahuja NK, Nuijts RM, Ajani A, Jayadev C, Sharma C, et al. Current protocols of corneal collagen cross-linking: Visual, refractive, and tomographic outcomes. Am J Ophthalmol 2015;160:243-9.
Sadoughi MM, Einollahi B, Baradaran-Rafii A, Roshandel D, Hasani H, Nazeri M. Accelerated versus conventional corneal collagen cross-linking in patients with keratoconus: An intrapatient comparative study. Int Ophthalmol 2018;38:67-74.
Choi M, Kim J, Kim EK, Seo KY, Kim TI. Comparison of the conventional Dresden protocol and accelerated protocol with higher ultraviolet intensity in corneal collagen cross-linking for keratoconus. Cornea 2017;36:523-9.
Razmjoo H, Peyman A, Rahimi A, Modrek HJ. Cornea collagen cross-linking for keratoconus: A comparison between accelerated and conventional methods. Adv Biomed Res 2017;6:10.
Hashemi H, Mohebbi M, Asgari S. Standard and accelerated corneal cross-linking long-term results: A randomized clinical trial. Eur J Ophthalmol 2020;30:650-7.
Uçakhan ÖÖ, Yeşiltaş YS. Comparative 2-year outcomes of conventional and accelerated corneal collagen crosslinking in progressive keratoconus. Int J Ophthalmol 2020;13:1223-30.
Coskunseven E, Jankov MR 2nd, Hafezi F, Atun S, Arslan E, Kymionis GD. Effect of treatment sequence in combined intrastromal corneal rings and corneal collagen crosslinking for keratoconus. J Cataract Refract Surg 2009;35:2084-91.
Renesto Ada C, Melo LA Jr, Sartori Mde F, Campos M. Sequential topical riboflavin with or without ultraviolet a radiation with delayed intracorneal ring segment insertion for keratoconus. Am J Ophthalmol 2012;153:982-93.e3.
Elsaftawy HS, Ahmed MH, Saif MY, Mousa R. Sequential intracorneal ring segment implantation and corneal transepithelial collagen cross-linking in keratoconus. Cornea 2015;34:1420-6.
Mohammadpour M, Masoumi A, Dehghan M, Hashemian MN, Karami SA, Mahmoudi A. MyoRing implantation with and without corneal collagen crosslinking for the management of keratoconus. J Ophthalmic Vis Res 2020;15:486-92.
Hersh PS, Issa R, Greenstein SA. Corneal crosslinking and intracorneal ring segments for keratoconus: A randomized study of concurrent versus sequential surgery. J Cataract Refract Surg 2019;45:830-9.
References
- 1.Krachmer JH, Feder RS, Belin MW. Keratoconus and related noninflammatory corneal thinning disorders. Surv Ophthalmol. 1984;28:293–322. doi: 10.1016/0039-6257(84)90094-8. [DOI] [PubMed] [Google Scholar]
- 2.Bhattacharyya A, Sarma P, Das K, Agarwal B, Medhi J, Das Mohapatra SS. Safety and efficacy of riboflavin-assisted collagen cross-linking of cornea in progressive keratoconus patients: A prospective study in North East India. Indian J Pharmacol. 2019;51:157–67. doi: 10.4103/ijp.IJP_75_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lang SJ, Messmer EM, Geerling G, Mackert MJ, Brunner T, Dollak S, et al. Prospective, randomized, double-blind trial to investigate the efficacy and safety of corneal cross-linking to halt the progression of keratoconus. BMC Ophthalmol. 2015;15:78. doi: 10.1186/s12886-015-0070-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-a-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135:620–7. doi: 10.1016/s0002-9394(02)02220-1. [DOI] [PubMed] [Google Scholar]
- 5.Nath S, Shen C, Koziarz A, Banfield L, Fava MA, Hodge WG. Transepithelial versus epithelium-off corneal collagen cross-linking for corneal ectasia: Protocol for a systematic review, meta-analysis and trial sequential analysis of randomised controlled trials. BMJ Open. 2019;9:e025728. doi: 10.1136/bmjopen-2018-025728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Al Fayez MF, Alfayez S, Alfayez Y. Transepithelial versus epithelium-off corneal collagen cross-linking for progressive keratoconus: A prospective randomized controlled trial. Cornea. 2015;34(Suppl 10):S53–6. doi: 10.1097/ICO.0000000000000547. [DOI] [PubMed] [Google Scholar]
- 7.Rossi S, Orrico A, Santamaria C, Romano V, De Rosa L, Simonelli F, et al. Standard versus trans-epithelial collagen cross-linking in keratoconus patients suitable for standard collagen cross-linking. Clin Ophthalmol Auckl NZ. 2015;9:503–9. doi: 10.2147/OPTH.S73991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Soeters N, Wisse RP, Godefrooij DA, Imhof SM, Tahzib NG. Transepithelial versus epithelium-off corneal cross-linking for the treatment of progressive keratoconus: A randomized controlled trial. Am J Ophthalmol. 2015;159:821–8.e3. doi: 10.1016/j.ajo.2015.02.005. [DOI] [PubMed] [Google Scholar]
- 9.Subasinghe SK, Ogbuehi KC, Dias GJ. Current perspectives on corneal collagen crosslinking (CXL) Graefes Arch Clin Exp Ophthalmol. 2018;256:1363–84. doi: 10.1007/s00417-018-3966-0. [DOI] [PubMed] [Google Scholar]
- 10.Beckman Rehnman J, Behndig A, Hallberg P, Lindén C. Initial results from mechanical compression of the cornea during crosslinking for keratoconus. Acta Ophthalmol. 2014;92:644–9. doi: 10.1111/aos.12380. [DOI] [PubMed] [Google Scholar]
- 11.Nordström M, Schiller M, Fredriksson A, Behndig A. Refractive improvements and safety with topography-guided corneal crosslinking for keratoconus:1-year results. Br J Ophthalmol. 2017;101:920–5. doi: 10.1136/bjophthalmol-2016-309210. [DOI] [PubMed] [Google Scholar]
- 12.Kanellopoulos AJ, Asimellis G. Epithelial remodeling after partial topography-guided normalization and high-fluence short-duration crosslinking (Athens protocol): Results up to 1 year. J Cataract Refract Surg. 2014;40:1597–602. doi: 10.1016/j.jcrs.2014.02.036. [DOI] [PubMed] [Google Scholar]
- 13.Kanellopoulos AJ. The impact of keratoconus treatment with the Athens Protocol (partial topography-guided photorefractive keratectomy combined with higher-fluence corneal collagen cross-linking) on quality of life: A long-term study. Clin Ophthalmol. 2019;13:795–803. doi: 10.2147/OPTH.S188519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kobashi H, Rong SS. Corneal collagen cross-linking for keratoconus: Systematic review. Biomed Res Int. 2017;2017:8145651. doi: 10.1155/2017/8145651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li J, Ji P, Lin X. Efficacy of corneal collagen cross-linking for treatment of keratoconus: A meta-analysis of randomized controlled trials. PLoS One. 2015;10:e0127079. doi: 10.1371/journal.pone.0127079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cochrane Handbook for Systematic Reviews of Interventions. Handbook. [Last accessed on 2020 Jun 28]. Available from:https://training.cochrane.org/handbook .
- 17.Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ. 2009;339:b2700. doi: 10.1136/bmj.b2700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hersh PS, Greenstein SA, Fry KL. Corneal collagen crosslinking for keratoconus and corneal ectasia: One-year results. J Cataract Refract Surg. 2011;37:149–60. doi: 10.1016/j.jcrs.2010.07.030. [DOI] [PubMed] [Google Scholar]
- 19.Sarma P, Kaur H, Kumar H, Mahendru D, Avti P, Bhattacharyya A, et al. Virological and clinical cure in COVID-19 patients treated with hydroxychloroquine: A systematic review and meta-analysis. J Med Virol. 2020;92:776–85. doi: 10.1002/jmv.25898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cochrane Handbook for Systematic Reviews of Interventions. [Last accessed on 2018 Dec 07]. Handbook Available from:https://www.cochrane.org/news/new-cochrane-handbook-systematic-reviews-interventions .
- 21.Maneeton N, Maneeton B, Putthisri S, Woottiluk P, Narkpongphun A, Srisurapanont M. Risperidone for children and adolescents with autism spectrum disorder: A systematic review. Neuropsychiatr Dis Treat. 2018;14:1811–20. doi: 10.2147/NDT.S151802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wittig-Silva C, Whiting M, Lamoureux E, Lindsay RG, Sullivan LJ, Snibson GR. A randomized controlled trial of corneal collagen cross-linking in progressive keratoconus: Preliminary results. J Refract Surg. 2008;24:S720–5. doi: 10.3928/1081597X-20080901-15. [DOI] [PubMed] [Google Scholar]
- 23.Hallahan KM, Rocha K, Roy AS, Randleman JB, Stulting RD, Dupps WJ., Jr Effects of corneal cross-linking on ocular response analyzer waveform-derived variables in keratoconus and postrefractive surgery ectasia. Eye Contact Lens. 2014;40:339–44. doi: 10.1097/ICL.0000000000000090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wittig-Silva C, Chan E, Islam FM, Wu T, Whiting M, Snibson GR. A randomized, controlled trial of corneal collagen cross-linking in progressive keratoconus: Three-year results. Ophthalmology. 2014;121:812–21. doi: 10.1016/j.ophtha.2013.10.028. [DOI] [PubMed] [Google Scholar]
- 25.Hersh PS, Stulting RD, Muller D, Durrie DS Rajpal RK United States Crosslinking Study Group. United States Multicenter Clinical Trial of Corneal Collagen Crosslinking for Keratoconus Treatment. Ophthalmology. 2017;124:1259–70. doi: 10.1016/j.ophtha.2017.03.052. [DOI] [PubMed] [Google Scholar]
- 26.Rosenblat E, Hersh PS. Intraoperative corneal thickness change and clinical outcomes after corneal collagen crosslinking: Standard crosslinking versus hypotonic riboflavin. J Cataract Refract Surg. 2016;42:596–605. doi: 10.1016/j.jcrs.2016.01.040. [DOI] [PubMed] [Google Scholar]
- 27.Greenstein SA, Fry KL, Hersh MJ, Hersh PS. Higher-order aberrations after corneal collagen crosslinking for keratoconus and corneal ectasia. J Cataract Refract Surg. 2012;38:292–302. doi: 10.1016/j.jcrs.2011.08.041. [DOI] [PubMed] [Google Scholar]
- 28.Greenstein SA, Fry KL, Hersh PS. Corneal topography indices after corneal collagen crosslinking for keratoconus and corneal ectasia: One-year results. J Cataract Refract Surg. 2011;37:1282–90. doi: 10.1016/j.jcrs.2011.01.029. [DOI] [PubMed] [Google Scholar]
- 29.O'Brart DP, Chan E, Samaras K, Patel P, Shah SP. A randomised, prospective study to investigate the efficacy of riboflavin/ultraviolet A (370 nm) corneal collagen cross-linkage to halt the progression of keratoconus. Br J Ophthalmol. 2011;95:1519–24. doi: 10.1136/bjo.2010.196493. [DOI] [PubMed] [Google Scholar]
- 30.Seyedian MA, Aliakbari S, Miraftab M, Hashemi H, Asgari S, Khabazkhoob M. Corneal collagen cross-linking in the treatment of progressive keratoconus: A randomized controlled contralateral eye study. Middle East Afr J Ophthalmol. 2015;22:340–5. doi: 10.4103/0974-9233.159755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wittig-Silva C, Chan E, Pollock G, Snibson GR. Localized changes in stromal reflectivity after corneal collagen cross-linking observed with different imaging techniques. J Refract Surg. 2013;29:410–6. doi: 10.3928/1081597X-20130410-02. [DOI] [PubMed] [Google Scholar]
- 32.Toro-Giraldo L, Morales Flores N, Santana-Cruz O, Ramirez-Miranda A, Navas A, Olivo-Payne A, et al. Cool crosslinking: Riboflavin at 4°C for pain management after crosslinking for keratoconus patients, a randomized clinical trial. Cornea. 2021;40:1–4. doi: 10.1097/ICO.0000000000002484. [DOI] [PubMed] [Google Scholar]
- 33.Sharma N, Suri K, Sehra SV, Titiyal JS, Sinha R, Tandon R, et al. Collagen cross-linking in keratoconus in Asian eyes: Visual, refractive and confocal microscopy outcomes in a prospective randomized controlled trial. Int Ophthalmol. 2015;35:827–32. doi: 10.1007/s10792-015-0054-x. [DOI] [PubMed] [Google Scholar]
- 34.Kim BZ, Jordan CA, McGhee CN, Patel DV. Natural history of corneal haze after corneal collagen crosslinking in keratoconus using Scheimpflug analysis. J Cataract Refract Surg. 2016;42:1053–9. doi: 10.1016/j.jcrs.2016.04.019. [DOI] [PubMed] [Google Scholar]
- 35.Henriquez MA, Izquierdo L, Jr, Bernilla C, Zakrzewski PA, Mannis M. Riboflavin/ultraviolet A corneal collagen cross-linking for the treatment of keratoconus: Visual outcomes and scheimpflug analysis. Cornea. 2011;30:281–6. doi: 10.1097/ICO.0b013e3181eeaea1. [DOI] [PubMed] [Google Scholar]
- 36.Meyer JJ, Jordan CA, Patel DV, Gokul A, Dean SJ, Ahmed S, et al. Five-year results of a prospective, randomised, contralateral eye trial of corneal crosslinking for keratoconus. Clin Exp Ophthalmol. 2021;49:542–9. doi: 10.1111/ceo.13959. [DOI] [PubMed] [Google Scholar]
- 37.Stojanovic A, Zhou W, Utheim TP. Corneal collagen cross-linking with and without epithelial removal: A contralateral study with 0.5% hypotonic riboflavin solution. Biomed Res Int. 2014;2014:619398. doi: 10.1155/2014/619398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zaheryani Corneal Collagen Cross-linking Using Epithelium Disruptor Instrument in Progressive Keratoconus. [Last accessed on 2020 Nov 10]. Available from:http://www.jcurrophthalmol.org/article.asp?issn=2452-2325;year=2020.volume=32;issue=3;spage=256;epage=262;aulast=Zaheryani . [DOI] [PMC free article] [PubMed]
- 39.Lai MJ, Greenstein SA, Gelles JD, Hersh PS. Corneal haze after transepithelial collagen cross-linking for keratoconus: A scheimpflug densitometry analysis. Cornea. 2020;39:1117–21. doi: 10.1097/ICO.0000000000002334. [DOI] [PubMed] [Google Scholar]
- 40.Hersh PS, Lai MJ, Gelles JD, Lesniak SP. Transepithelial corneal crosslinking for keratoconus. J Cataract Refract Surg. 2018;44:313–22. doi: 10.1016/j.jcrs.2017.12.022. [DOI] [PubMed] [Google Scholar]
- 41.Nawaz S, Gupta S, Gogia V, Sasikala NK, Panda A. Trans-epithelial versus conventional corneal collagen crosslinking: A randomized trial in keratoconus. Oman J Ophthalmol. 2015;8:9–13. doi: 10.4103/0974-620X.149855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Al Zubi K, Albakar Y, Nasser R. Transepithelial versus epithelium off crosslinking for treating keratoconus among Jordanians. Open Ophthalmol J. 2019;13:8–14. [Google Scholar]
- 43.Cifariello F, Minicucci M, Di Renzo F, Di Taranto D, Coclite G, Zaccaria S, et al. Epi-off versus Epi-on corneal collagen cross-linking in keratoconus patients: A comparative study through 2-year follow-up. J Ophthalmol. 2018;2018:4947983. doi: 10.1155/2018/4947983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lombardo M, Giannini D, Lombardo G, Serrao S. Randomized controlled trial comparing transepithelial corneal cross-linking using iontophoresis with the Dresden protocol in progressive keratoconus. Ophthalmology. 2017;124:804–12. doi: 10.1016/j.ophtha.2017.01.040. [DOI] [PubMed] [Google Scholar]
- 45.Bikbova G, Bikbov M. Standard corneal collagen crosslinking versus transepithelial iontophoresis-assisted corneal crosslinking, 24 months follow-up: Randomized control trial. Acta Ophthalmol. 2016;94:e600–6. doi: 10.1111/aos.13032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lombardo M, Serrao S, Lombardo G, Schiano-Lomoriello D. Two-year outcomes of a randomized controlled trial of transepithelial corneal crosslinking with iontophoresis for keratoconus. J Cataract Refract Surg. 2019;45:992–1000. doi: 10.1016/j.jcrs.2019.01.026. [DOI] [PubMed] [Google Scholar]
- 47.Lombardo M, Serrao S, Raffa P, Rosati M, Lombardo G. Novel technique of transepithelial corneal cross-linking using iontophoresis in progressive keratoconus. J Ophthalmol. 2016;2016:7472542. doi: 10.1155/2016/7472542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hashemian H, Jabbarvand M, Khodaparast M, Ameli K. Evaluation of corneal changes after conventional versus accelerated corneal cross-linking: A randomized controlled trial. J Refract Surg. 2014;30:837–42. doi: 10.3928/1081597X-20141117-02. [DOI] [PubMed] [Google Scholar]
- 49.Sherif AM. Accelerated versus conventional corneal collagen cross-linking in the treatment of mild keratoconus: A comparative study. Clin Ophthalmol. 2014;8:1435–40. doi: 10.2147/OPTH.S59840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shetty R, Pahuja NK, Nuijts RM, Ajani A, Jayadev C, Sharma C, et al. Current protocols of corneal collagen cross-linking: Visual, refractive, and tomographic outcomes. Am J Ophthalmol. 2015;160:243–9. doi: 10.1016/j.ajo.2015.05.019. [DOI] [PubMed] [Google Scholar]
- 51.Hashemi H, Fotouhi A, Miraftab M, Bahrmandy H, Seyedian MA, Amanzadeh K, et al. Short-term comparison of accelerated and standard methods of corneal collagen crosslinking. J Cataract Refract Surg. 2015;41:533–40. doi: 10.1016/j.jcrs.2014.07.030. [DOI] [PubMed] [Google Scholar]
- 52.Hashemi H, Miraftab M, Seyedian MA, Hafezi F, Bahrmandy H, Heidarian S, et al. Long-term results of an accelerated corneal cross-linking protocol (18 mW/cm2) for the treatment of progressive keratoconus. Am J Ophthalmol. 2015;160:1164–70.e1. doi: 10.1016/j.ajo.2015.08.027. [DOI] [PubMed] [Google Scholar]
- 53.Sadoughi MM, Einollahi B, Baradaran-Rafii A, Roshandel D, Hasani H, Nazeri M. Accelerated versus conventional corneal collagen cross-linking in patients with keratoconus: An intrapatient comparative study. Int Ophthalmol. 2018;38:67–74. doi: 10.1007/s10792-016-0423-0. [DOI] [PubMed] [Google Scholar]
- 54.Choi M, Kim J, Kim EK, Seo KY, Kim TI. Comparison of the conventional dresden protocol and accelerated protocol with higher ultraviolet intensity in corneal collagen cross-linking for keratoconus. Cornea. 2017;36:523–9. doi: 10.1097/ICO.0000000000001165. [DOI] [PubMed] [Google Scholar]
- 55.Hashemi H, Mohebbi M, Asgari S. Standard and accelerated corneal cross-linking long-term results: A randomized clinical trial. Eur J Ophthalmol. 2020;30:650–7. doi: 10.1177/1120672119839927. [DOI] [PubMed] [Google Scholar]
- 56.Uçakhan ÖÖ, Yeşiltaş YS. Comparative 2-year outcomes of conventional and accelerated corneal collagen crosslinking in progressive keratoconus. Int J Ophthalmol. 2020;13:1223–30. doi: 10.18240/ijo.2020.08.07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Razmjoo H, Peyman A, Rahimi A, Modrek HJ. Cornea collagen cross-linking for keratoconus: A comparison between accelerated and conventional methods. Adv Biomed Res. 2017;6:10. doi: 10.4103/2277-9175.200785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Renesto Ada C, Melo LA, Jr, Sartori Mde F, Campos M. Sequential topical riboflavin with or without ultraviolet a radiation with delayed intracorneal ring segment insertion for keratoconus. Am J Ophthalmol. 2012;153:982–93.e3. doi: 10.1016/j.ajo.2011.10.014. [DOI] [PubMed] [Google Scholar]
- 59.Elsaftawy HS, Ahmed MH, Saif MY, Mousa R. Sequential intracorneal ring segment implantation and corneal transepithelial collagen cross-linking in keratoconus. Cornea. 2015;34:1420–6. doi: 10.1097/ICO.0000000000000600. [DOI] [PubMed] [Google Scholar]
- 60.Mohammadpour M, Masoumi A, Dehghan M, Hashemian MN, Karami SA, Mahmoudi A. MyoRing implantation with and without corneal collagen crosslinking for the management of keratoconus. J Ophthalmic Vis Res. 2020;15:486–92. doi: 10.18502/jovr.v15i4.7790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hersh PS, Issa R, Greenstein SA. Corneal crosslinking and intracorneal ring segments for keratoconus: A randomized study of concurrent versus sequential surgery. J Cataract Refract Surg. 2019;45:830–9. doi: 10.1016/j.jcrs.2019.01.020. [DOI] [PubMed] [Google Scholar]
- 62.Coskunseven E, Jankov MR, Hafezi F, Atun S, Arslan E, Kymionis GD. Effect of treatment sequence in combined intrastromal corneal rings and corneal collagen crosslinking for keratoconus. J Cataract Refract Surg. 2009;35:2084–91. doi: 10.1016/j.jcrs.2009.07.008. [DOI] [PubMed] [Google Scholar]
- 63.Krueger RR, Kanellopoulos AJ. Stability of simultaneous topography-guided photorefractive keratectomy and riboflavin/UVA cross-linking for progressive keratoconus: Case reports. J Refract Surg. 2010;26:S827–32. doi: 10.3928/1081597X-20100921-11. [DOI] [PubMed] [Google Scholar]
- 64.Kanellopoulos AJ. Long term results of a prospective randomized bilateral eye comparison trial of higher fluence, shorter duration ultraviolet A radiation, and riboflavin collagen cross linking for progressive keratoconus. Clin Ophthalmol. 2012;6:97–101. doi: 10.2147/OPTH.S27170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kanellopoulos AJ, Asimellis G. Comparison of placido disc and scheimpflug image-derived topography-guided excimer laser surface normalization combined with higher fluence CXL: The Athens protocol, in progressive keratoconus. Clin Ophthalmol. 2013;7:1385–96. doi: 10.2147/OPTH.S44745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kanellopoulos AJ, Asimellis G. Keratoconus management: Long-term stability of topography-guided normalization combined with high-fluence CXL stabilization (the Athens protocol) J Refract Surg. 2014;30:88–93. doi: 10.3928/1081597X-20140120-03. [DOI] [PubMed] [Google Scholar]
- 67.Kontadakis GA, Kankariya VP, Tsoulnaras K, Pallikaris AI, Plaka A, Kymionis GD. Long-term comparison of simultaneous topography-guided photorefractive keratectomy followed by corneal cross-linking versus corneal cross-linking alone. Ophthalmology. 2016;123:974–83. doi: 10.1016/j.ophtha.2016.01.010. [DOI] [PubMed] [Google Scholar]
- 68.Grentzelos MA, Liakopoulos DA, Siganos CS, Tsilimbaris MK, Pallikaris IG, Kymionis GD. Long-term comparison of combined t-PTK and CXL (Cretan protocol) versus CXL With mechanical epithelial debridement for keratoconus. J Refract Surg. 2019;35:650–5. doi: 10.3928/1081597X-20190917-01. [DOI] [PubMed] [Google Scholar]
- 69.Hagem AM, Thorsrud A, Sandvik GF, Drolsum L. Randomized study of collagen cross-linking with conventional versus accelerated UVA Irradiation using riboflavin with hydroxypropyl methylcellulose: Two-year results. Cornea. 2019;38:203–9. doi: 10.1097/ICO.0000000000001791. [DOI] [PubMed] [Google Scholar]
- 70.GRADEpro. [Last accessed on 2020 Aug 25]. Available from:https://gradepro.org/
- 71.Nath S, Shen C, Koziarz A, Banfield L, Nowrouzi-Kia B, Fava MA, et al. Transepithelial versus epithelium-off corneal collagen cross-linking for corneal ectasia: A systematic review and meta-analysis. Ophthalmology. 2021;128:1150–60. doi: 10.1016/j.ophtha.2020.12.023. [DOI] [PubMed] [Google Scholar]
- 72.Yurttaser Ocak S, Mangan MS. Endothelial cell loss after accelerated corneal crosslinking using pachymetry-guided hypo-osmolar riboflavin dosing in thin keratoconic corneas. J Cataract Refract Surg. 2021;47:1530–4. doi: 10.1097/j.jcrs.0000000000000686. [DOI] [PubMed] [Google Scholar]
- 73.Gu SF, Fan ZS, Wang LH, Tao XC, Zhang Y, Wang CQ, et al. Ashort-term study of corneal collagen cross-linking with hypo-osmolar riboflavin solution in keratoconic corneas. Int J Ophthalmol. 2015;8:94–7. doi: 10.3980/j.issn.2222-3959.2015.01.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Wollensak G, Spoerl E, Wilsch M, Seiler T. Endothelial cell damage after riboflavin-ultraviolet-A treatment in the rabbit. J Cataract Refract Surg. 2003;29:1786–90. doi: 10.1016/s0886-3350(03)00343-2. [DOI] [PubMed] [Google Scholar]
- 75.Hafezi F, Kling S, Gilardoni F, Hafezi N, Hillen M, Abrishamchi R, et al. Individualized corneal cross-linking with riboflavin and UV-A in ultrathin corneas: The Sub400 protocol. Am J Ophthalmol. 2021;224:133–42. doi: 10.1016/j.ajo.2020.12.011. [DOI] [PubMed] [Google Scholar]
- 76.Hammer A, Richoz O, Arba Mosquera S, Tabibian D, Hoogewoud F, Hafezi F. Corneal biomechanical properties at different corneal cross-linking (CXL) irradiances. Invest Ophthalmol Vis Sci. 2014;55:2881–4. doi: 10.1167/iovs.13-13748. [DOI] [PubMed] [Google Scholar]
- 77.Rapuano PB, Mathews PM, Florakis GJ, Trokel SL, Suh LH. Corneal collagen crosslinking in patients treated with dextran versus isotonic hydroxypropyl methylcellulose (HPMC) riboflavin solution: A retrospective analysis. Eye Vis (Lond) 2018;5:23. doi: 10.1186/s40662-018-0116-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Thorsrud A, Hagem AM, Sandvik GF, Drolsum L. Superior outcome of corneal collagen cross-linking using riboflavin with methylcellulose than riboflavin with dextran as the main supplement. Acta Ophthalmol (Copenh) 2019;97:415–21. doi: 10.1111/aos.13928. [DOI] [PubMed] [Google Scholar]
- 79.Sedaghat M, Bagheri M, Ghavami S, Bamdad S. Changes in corneal topography and biomechanical properties after collagen cross linking for keratoconus:1-year results. Middle East Afr J Ophthalmol. 2015;22:212–9. doi: 10.4103/0974-9233.151877. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
PRISMA flow diagram of included randomized control trials
Characteristics of included randomized controlled trials
Risk of bias graph among included randomized control trials. “Cochrane Risk of Bias tool for randomized control trial” has been used for risk of bias assessment.[54] Six domains taken into account are: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other bias. In the risk of bias graph green color indicates low risk; yellow color indicates unclear; and red color indicates high risk of bias
CXL vs. control: ∆SE at 1 year subgroup analysis based on control eye selection (Fellow eye of same patient vs. eye of different patient). ∆Indicate change from baseline, CXL = Collagen cross-linking, SE = Spherical equivalence
(a) TE-CXL (CE) versus C-CXL: ∆Kmax at 1 year: Sensitivity analysis (excluding study by Rossi et al). (b) TE-CXL (CE) versus C-CXL: ∆Kmax at 1 year: Subgroup analysis based on different riboflavin formulation (dextran based riboflavin vs. no dextran riboflavin). CE = Chemical enhancer, Kmax = Maximum keratometry, CXL = Collagen cross-linking, TE-CXL = Transepithelial CXL, C-CXL = Conventional CXL
A-CXL versus C-CXL protocol: Kmax at 1 year: Subgroup analysis based on baseline Kmax difference (>1D vs. <1D). Kmax = Maximum keratometry, CXL = Collagen cross-linking, C-CXL = Conventional CXL, A-CXL = Accelerated CXL