Rice blast, caused by Magnaporthe oryzae, is a serious disease in rice (Oryzae sativa L.) worldwide. Because the pathogen mutates rapidly, rice blast has become one of the major threats for global rice production (Wang and Valent, 2017). Using host resistance is the most effective method for blast control. However most rice resistance genes are race‐specific (Li et al., 2017). Discovering race non‐specific resistance genes will enhance blast resistance in rice cultivars.
Fusarium graminearum is a hemibiotrophic fungal pathogen for wheat Fusarium head blight (FHB) and shares a similar lifestyle with Magnaporthe oryzae for rice blast. Previously, we have cloned Fhb1 encoding a histidine‐rich calcium‐binding protein (TaHRC) from wheat for race non‐specific resistance to Fusarium species, which has been widely deployed in many wheat cultivars worldwide (Bai et al., 2018; Su et al., 2019). TaHRC serves as a susceptibility factor to promote FHB spread and loss‐of‐function mutation in TaHRC confers FHB resistance (Chen et al., 2022). HRC is highly conservative with the closest TaHRC homologue from rice (OsHRC), however, the functions of OsHRC in rice blast resistance remains to be investigated.
We cloned OsHRC (LOC_Os01g03060) from ‘Zhonghua11' (ZH11) and the OsHRC DNA sequence is 2390 bp with three exons and two introns. OsHRC encodes a putative 33.5 kDa protein of 285‐amino acids and shares 63.5%–64.5% amino acid identity with TaHRC from three wheat subgenomes (A, B and D). The TaHRC and OsHRC proteins were predicted to have similar biological features and both contain a histidine and arginine enriched region without any known functional domain except for a motif of a nuclear localization signal (NLS), indicating the conserved HRC function between rice and wheat. The OsHRC protein was localized to the nucleus in rice leaf protoplasts (Figure 1a). Transactivation activity assay showed that yeast transiently expressing BD‐OsHRC and BD‐TaHRC did not grow in a deficient medium (SD/‐Trp‐His) (Figure 1b), suggesting that both nuclear proteins do not have transactivational activity and share the same biological functions.
Figure 1.
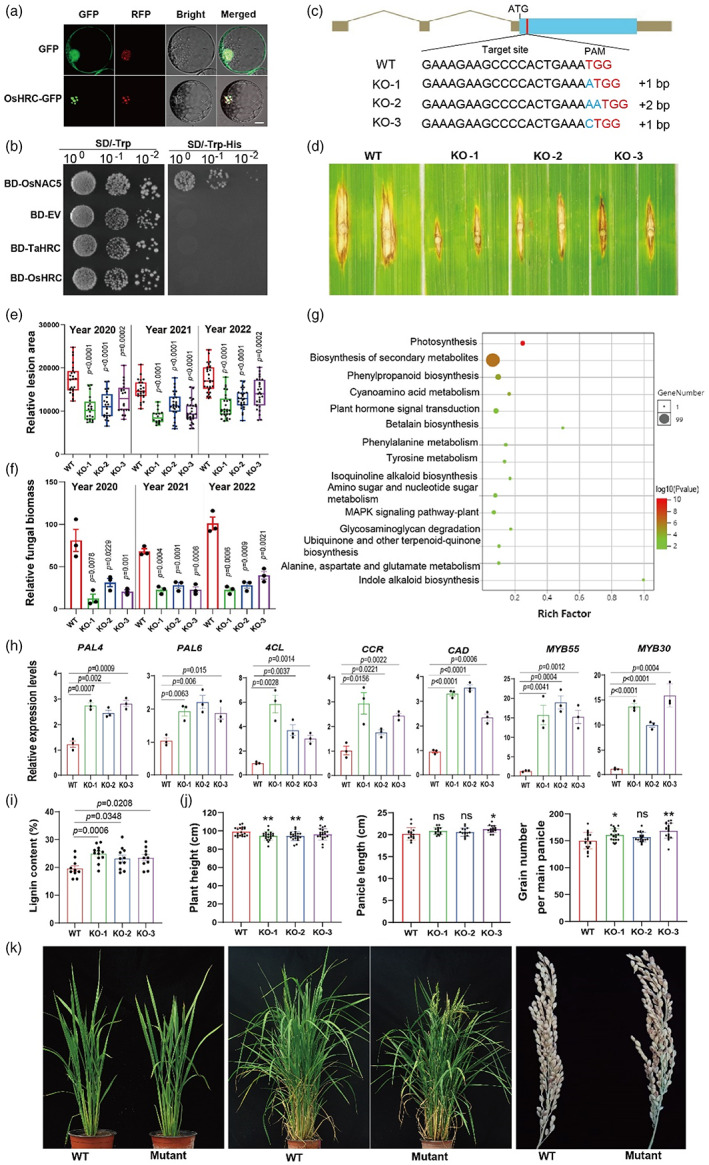
Characterization of OsHRC for resistance to rice blast isolate RB22. (a) Subcellular localization of OsHRC and TaHRC transiently expressed in rice protoplasts. GFP, green fluorescent protein. RFP, red fluorescent proteins, respectively. Scale bar = 5 μm. (b) Transactivation activity assay of OsHRC and TaHRC in yeast used a rice transcription factor OsNAC5 as a positive control. EV, empty vector. BD, a vector with the DNA binding domain activity. SD/‐Trp, synthetic dropout (SD) medium lacking tryptophan. SD/‐Trp‐His, a SD medium lacking tryptophan and histidine. (c) OsHRC gRNA target site showing mutated sequences in the three T1 lines generated from Zhonghua11. CRISPR/Cas9 and gRNA were cloned into a pCBSG032 vector and transformed into Zhonghua11 callus via Agrobacterium tumefaciens. Blue letters refer to inserted nucleotides after editing. ATG, start codon. Blue box, ORF. (d–f) Lesions caused by M. oryzae on the mutant lines KO‐1, KO‐2 and KO‐3 were significantly smaller with less fungal biomass than in non‐edited Zhonghua11 at 14 days post inoculation (DPI) with RB22. (g) The KEGG analysis of differentially expressed genes in WT and KO‐1 at 14 DPI. (h) Differential expression of the seven lignin‐related genes between three mutants and Zhonghua11 at 14 DPI detected by qRT‐PCR analysis. Data were normalized by OsActin. (i) Higher lignin content was observed in the leaves of three mutants than Zhonghua11 at 14 DPI. (j, k) The mutants showed shorter plants, slightly longer panicle and more grain per panicle compared to Zhonghua11. Values are means and standard errors with n = 3 (e,f,h), n ≥ 10 (i) and n ≥ 15 (j).
To verify the function of OsHRC on rice blast resistance, we designed a guide RNA to target a 20‐nt sequence in the 59 bp downstream of the translation start codon of OsHRC and conducted CRISPR/Cas9‐mediated gene editing to knock out (KO) OsHRC in ZH11. Among the five OsHRC KO mutants generated, three (KO‐1, KO‐2 and KO‐3) showed premature translation termination due to frameshifts, and were predicted to generate truncated proteins of 39, 236, and 39 amino acids, respectively (Figure 1c). The three mutants and ZH11 were inoculated with RB22, a highly virulent M. oryzae strain from China, 60 days after planting. All the three mutants showed significantly smaller lesions and less fungal biomass than their non‐edited controls at 14 days post inoculation (DPI) (Figure 1d–f). To examine whether the resistance is race non‐specific, the three mutants were inoculated with another virulant strain S005 and showed the same level of resistance as to RB22 (Figure S1a,b), demonstrating that the loss‐of‐function of OsHRC confers race non‐specific resistance to M. oryzae.
To explore the molecular basis of OsHRC resistance to rice blast, RNA‐seq was conducted to identify differentially expressed genes (DEGs) between an OsHRC KO mutant and ZH11. At 14 DPI with RB22, a total of 1333 DEGs were detected at the fold‐change of 2 and false discovery rate (FDR) < 0.05 using the R package DESeq2. KEGG analysis indicated that the DEGs were mainly enriched in the pathways of photosynthesis, biosynthesis of secondary metabolites, and phenylpropanoid biosynthesis (Figure 1g). Phenylpropanoids play important roles in plant responses to biotic and abiotic stresses (Li et al., 2020). Quantitative RT‐PCR results demonstrated that the genes in the phenylpropanoid biosynthesis pathway including genes encoding two phenylalanine ammonia‐lyase (PAL6, Os04g0518400; PAL4, Os02g0627100), a 4‐coumarate‐CoA ligase (4CL, Os01g0901600), a cinnamyl‐CoA reductase (CCR, Os02g0180700) and a cinnamyl‐alcohol dehydrogenase (CAD, Os04g0229100) were significantly upregulated in the three OsHRC mutants compared with ZH11 (Figures 1hand S2a), consistent with Vanholme et al. (2019). In addition, the transcription factors, MYB30 and MYB55 involved in the regulation of the phenylpropanoid pathway (Li et al., 2020) were upregulated in the three OsHRC mutants (Figures 1h and S2a). Lignin is one of the most important phenylpropanoid metabolites, and its accumulation in leaf inhibits the penetration of M. oryzae (Li et al., 2020; Zhou et al., 2018). Since all the seven genes are pivotal for the lignin biosynthesis (Vanholme et al., 2019), we compared the lignin content between all mutants and the non‐edited control, and found that the leaves of three mutants contained significantly higher lignin content than the control at 14 DPI with the isolate RB22 (Figures 1i and S2b), suggesting that OsHRC‐mediated blast resistance is most likely due to the activation of the lignin synthesis pathway after M. oryzae infection.
HRC is a conserved gene in cereal crops and the loss‐of‐function mutation in HRC confers resistance to wheat FHB (Su et al., 2019) and maize ear rot without significant penalty on yield (Liu et al., 2022). To evaluate effects of OsHRC on major rice agronomic traits, we compared heading date, plant height, and grain size between the mutants and WT control in greenhouse experiments and found that the mutants were slightly shorter, but with larger spikes, more spikelets per spike, and earlier heading date than the control (Figure 1j,k). These data indicate that OsHRC mutants showed significant improvement in blast resistance without obvious yield reduction in greenhouses, therefore, can be used as a new source of blast resistance in rice breeding.
Funding
This work was supported by the National Key Research and Development Program of China (2022YFD1201502), Talent Funds of China Agricultural University (2021RC009) and US Wheat and Barley Scab Initiative.
Author contributions
ZS and GB designed the project and wrote the manuscript. ZS, YD, FZ, ZZ, XH, FS, JL performed experiments. All authors revised and approved the manuscript.
Conflicts of interest
The authors declare no conflicts of interest.
Supporting information
Data S1 Material and methods.
Figure S1 Comparison of OsHRC of rice for responses to the inoculation of M. oryzae isolate S005. (a) Lesions caused by M. oryzae on the non‐edited control (WT) and three knockout (KO) mutant lines (KO‐1, KO‐2 and KO‐3). (b) Relative lesion areas (means ± standard errors) in the three mutants were significantly smaller than in non‐edited control at 14 days post inoculation (DPI) with the M. oryzae isolate S005. (c) Relative fungal biomass (means ± standard errors) in the three mutants was significantly lower than in non‐edited ZH11 control at 14 DPI.
Figure S2 Comparisons of transcript levels of lignin‐related genes and lignin content in the leaves between ZH11 and the mutants after different time points of inoculation with the M. oryzae isolates RB22. (a) Comparisons of seven differentially expressed lignin‐related genes between three mutants and ZH11 (WT control) at 0, 5, 10 and 14 days post inoculation (dpi). Data were normalized using the OsActin gene control. Error bars represent the standard error (SE) of three mutants. (b) Comparisons of lignin content between the inoculated and uninoculated leaves of the three mutants and ZH11 (WT control) at 14 DPI with isolate RB22. CK represent uninoculated control. Treatment represents 14 days post inoculation with isolate RB22.
Acknowledgement
Authors thank Dr. H. Fang from Chinese Academy of Agricultural Sciences (CAAS) for technical help with rice blast phenotyping and Prof. Y. Ning from CAAS and Prof. Y. Peng from China Agricultural University for providing M. oryzae isolates. USDA is an equal opportunity provider and employer.
Contributor Information
Guihua Bai, Email: guihua.bai@usda.gov.
Zhenqi Su, Email: zhenqisu@cau.edu.cn.
References
- Bai, G. , Su, Z. and Cai, J. (2018) Wheat resistance to Fusarium head blight. Can. J. Plant Pathol. 40, 336–346. [Google Scholar]
- Chen, H. , Su, Z. , Tian, B. , Hao, G. , Trick, H.N. and Bai, G. (2022) TaHRC suppresses the calcium‐mediated immune response and triggers wheat head blight susceptibility. Plant Physiol. 190, 1566–1569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, W. , Zhu, Z. , Chen, M. , Yin, J. , Yang, C. , Ran, L. , Cheng, M. et al. (2017) A natural allele of a transcription factor in rice confers broad–spectrum blast resistance. Cell 170, 114–126. [DOI] [PubMed] [Google Scholar]
- Li, W. , Wang, K. , Chen, M. et al. (2020) Sclerenchyma cell thickening through enhanced lignification induced by OsMYB30 prevents fungal penetration of rice leaves. New Phytol. 226, 1850–1863. [DOI] [PubMed] [Google Scholar]
- Liu, C. , Kong, M. , Zhu, J. , Qi, X. , Duan, C. and Xie, C. (2022) Engineering null mutants in ZmFER1 confers resistance to ear rot caused by Fusarium verticillioides in maize. Plant Biotechnol. J. 20, 2045–2047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su, Z. , Bernardo, A. , Tian, B. , Chen, H. , Wang, S. , Ma, H. , Cai, S. et al. (2019) A deletion mutation in TaHRC confers Fhb1 resistance to Fusarium head blight in wheat. Nat. Genet. 51, 1099–1105. [DOI] [PubMed] [Google Scholar]
- Vanholme, R. , De Meester, B. , Ralph, J. and Boerjan, W. (2019) Lignin biosynthesis and its integration into metabolism. Curr. Opin. Biotech. 56, 230–239. [DOI] [PubMed] [Google Scholar]
- Wang, G. and Valent, B. (2017) Durable resistance to rice blast. Science 355, 906–907. [DOI] [PubMed] [Google Scholar]
- Zhou, X. , Liao, H. , Chern, M. , Yin, J. , Chen, Y. , Wang, J. , Zhu, X. et al. (2018) Loss of function of a rice TPR‐domain RNA‐binding protein confers broad‐spectrum disease resistance. Proc. Natl. Acad. Sci. U. S. A. 115, 3174–3179. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data S1 Material and methods.
Figure S1 Comparison of OsHRC of rice for responses to the inoculation of M. oryzae isolate S005. (a) Lesions caused by M. oryzae on the non‐edited control (WT) and three knockout (KO) mutant lines (KO‐1, KO‐2 and KO‐3). (b) Relative lesion areas (means ± standard errors) in the three mutants were significantly smaller than in non‐edited control at 14 days post inoculation (DPI) with the M. oryzae isolate S005. (c) Relative fungal biomass (means ± standard errors) in the three mutants was significantly lower than in non‐edited ZH11 control at 14 DPI.
Figure S2 Comparisons of transcript levels of lignin‐related genes and lignin content in the leaves between ZH11 and the mutants after different time points of inoculation with the M. oryzae isolates RB22. (a) Comparisons of seven differentially expressed lignin‐related genes between three mutants and ZH11 (WT control) at 0, 5, 10 and 14 days post inoculation (dpi). Data were normalized using the OsActin gene control. Error bars represent the standard error (SE) of three mutants. (b) Comparisons of lignin content between the inoculated and uninoculated leaves of the three mutants and ZH11 (WT control) at 14 DPI with isolate RB22. CK represent uninoculated control. Treatment represents 14 days post inoculation with isolate RB22.


