Abstract
Background
Perianal abscess is common. Traditionally, postoperative perianal abscess cavities are managed with internal wound packing, a practice not supported by evidence. The aim of this randomized clinical trial (RCT) was to assess if non-packing is less painful and if it is associated with adverse outcomes.
Methods
The Postoperative Packing of Perianal Abscess Cavities (PPAC2) trial was a multicentre, RCT (two-group parallel design) of adult participants admitted to an NHS hospital for incision and drainage of a primary perianal abscess. Participants were randomized 1:1 (via an online system) to receive continued postoperative wound packing or non-packing. Blinded data were collected via symptom diaries, telephone, and clinics over 6 months. The objective was to determine whether non-packing of perianal abscess cavities is less painful than packing, without an increase in perianal fistula or abscess recurrence. The primary outcome was pain (mean maximum pain score on a 100-point visual analogue scale).
Results
Between February 2018 and March 2020, 433 participants (mean age 42 years) were randomized across 50 sites. Two hundred and thirteen participants allocated to packing reported higher pain scores than 220 allocated to non-packing (38.2 versus 28.2, mean difference 9.9; P < 0.0001). The occurrence of fistula-in-ano was low in both groups: 32/213 (15 per cent) in the packing group and 24/220 (11 per cent) in the non-packing group (OR 0.69, 95 per cent c.i. 0.39 to 1.22; P = 0.20). The proportion of patients with abscess recurrence was also low: 13/223 (6 per cent) in the non-packing group and 7/213 (3 per cent) in the packing group (OR 1.85, 95 per cent c.i. 0.72 to 4.73; P = 0.20).
Conclusion
Avoiding abscess cavity packing is less painful without a negative morbidity risk.
Registration number
ISRCTN93273484 (https://www.isrctn.com/ISRCTN93273484).
Registration number
The PPAC2 randomized clinical trial investigated the role of continued internal wound packing after incision and drainage of acute perianal abscess, a common general surgical condition. Across 50 NHS sites, 433 participants were recruited and randomized to receive packing or no packing, and were followed up for 6 months. No packing was found to be associated with significantly less pain, and no significant increase in fistula-in-ano or abscess recurrence.
Introduction
Perianal abscess is a common acute general surgical condition1. Ninety per cent are cryptoglandular in origin and caused by blockage and infection of the anal glands within the intersphincteric space of the anal canal2,3. Inflammatory bowel disease is a further important cause. Infection can spread downwards to the perianal skin, alongside the rectum (ischiorectal), above the pelvic floor (supralevator), or between the internal and external sphincters (intersphincteric)4. Other risk factors include smoking (twofold risk) and diabetes mellitus. Perianal abscess is twice as common in men as women, and the mean age at presentation is 40 years1,5,6.
The management of acute perianal sepsis has remained largely unchanged for over 50 years. Surgical abscess incision and drainage under general anaesthesia is followed by continued internal wound dressing (packing) of the resultant wound cavity in the community until the wound has healed. Pack changes are required multiple times a week for several weeks7. Packing is an accepted treatment7. Surgical dogma is that packing reduces the rate of perianal fistula formation and abscess recurrence, but there is no evidence to support this. Perianal fistulae (epithelial-lined tract between the perianal skin and anal canal or rectum) are sequelae of perianal abscesses, causing pain, purulent discharge, and almost invariably require surgical treatment, which may be complex. The majority of patients presenting with a fistula have a clear history of a previous abscess8. Fistula incidence after perianal abscess is variably reported; 5 to 37 per cent from small case series3,9, 27 per cent from our pilot data7, and 15.5 per cent from a large population-based study1. There is limited specific guidance on the postoperative management of the abscess cavity in existing national guidelines10,11. To date, two underpowered trials with only 64 participants investigating the role of packing in this setting have been reported12,13.
This group previously reported a pilot observational and feasibility study of 141 patients7. Results showed that wound packing is painful with a two- to three-fold increase in patient reported pain scores (visual analogue score (VAS)) during and after dressing changes7. On average (mean), each patient underwent 13 pack changes in the first 3 weeks of treatment (range 0 to 21). Patient-reported outcomes are an increasingly important measure of the quality of surgical care. Pain has been found to significantly impact on overall patient experience. Notably, moderate or severe postoperative pain, which occurs more frequently than a postoperative complication, has been shown to be strongly associated with a lower likelihood of the patient being satisfied after surgery. A patient with severe pain is nearly twice as likely to be unsatisfied or regret surgery than a patient who experienced a complication14. The objective of the Postoperative Packing of Perianal Abscess Cavities (PPAC2) trial was to determine whether non-packing of perianal abscess cavities is less painful than packing, without an increase in perianal fistula and abscess recurrence.
Methods
Study design and participants
PPAC2 was a multicentre randomized clinical trial (standard two-group parallel design) performed across 50 NHS Acute Hospital Trusts in England, Wales, and Scotland between February 2018 and February 2020. Patients aged 18 years or over presenting with their first perianal abscess of cryptoglandular origin requiring an incision and drainage operation were eligible for inclusion. Exclusion criteria were perianal fistula, Fournier’s gangrene, and horseshoe abscess. All potential participants received an information leaflet. The trial was approved by Greater Manchester West Research Ethics committee (17/NW/0529). The trial was pre-registered with ClinicalTrials.gov in October 2017 (ClinicalTrials.gov Identifier: NCT03315169) with an analysis plan.
Randomization and masking
Participants were randomized postoperatively prior to hospital discharge to reduce bias through modification of the operative procedure (preoperative recruitment was allowed). Participants were randomly allocated on a 1:1 basis (using the participating site as a stratification factor), via the online Treatment Allocation RanDomIsation System (TARDIS) to receive current standard wound care with internal packing by nurses in the community (control packing arm) or to receive no packing (simple external dressings only). Participants were recruited and randomized by surgeons (trainees and consultants) and research nurses.
It was not feasible to blind the participants, site enrolment personnel, or community nurses as different information, dressings, and patient advice were given to the packing and no-packing groups. The clinical assessor (consultant or appropriately trained trainee surgeon) of wound healing, perianal fistula, and abscess recurrence remained blind to study allocation. The wound dressing was removed by a different member of the research team to ensure blinding of the assessor.
Procedures
All participants had standard intraoperative wound packing (for haemostasis), removed within the first postoperative 24 hours. In the packing group, the frequency, type of packing dressing, and duration of packing was left to the discretion of the treating nurses, as is current standard practice. In the no-packing group, the use of external absorbent dressings or pads was again left to the discretion of the nurses and the participants, in keeping with the pragmatic design.
Baseline data, including demographics, operative data, health-related quality of life (HRQoL) score (EQ-5DTM; EuroQoL Group, Rotterdam, the Netherlands), and postoperative metronidazole use, were recorded. Participants were discharged home with a symptom diary. Daily acute pain assessments (0–100 mm point VAS) of worst daily pain (where 0 is no pain and 100 is worst pain imaginable), and pain before, during, and after dressing changes (for up to 21 days) were recorded. HRQoL score, return to work, participant satisfaction with wound management (5-point scale of strongly disagree, disagree, neutral, agree, strongly agree), and participant global assessments (3-point scale of poor, fair, and good) were also recorded. Participants were contacted by telephone on day seven to record return-to-work data and to encourage data entry. Participants were reviewed in clinic at 4, 8, and 26 weeks to assess for fistula, abscess recurrence, and wound healing. Wound healing was defined as complete epithelialization of the wound. The 8-week appointment was replaced with a telephone call if the wound had healed at 4 weeks. Fistula and abscess recurrence were serious adverse events (SAEs) and were reviewed and reported even when patients were lost to standard trial follow-up.
Outcomes
The primary outcome was mean maximum pain score over the first 10 days after incision and drainage surgery (mean of worst daily wound-related pain scores measured on a 100 mm VAS where 0 is no pain, and 100 is worst pain imaginable). Pain was selected as the primary outcome measure for this trial after it was highlighted as the key outcome for patients during patient and public involvement (focus groups) in the PPAC feasibility study7. Secondary outcomes included pain before, during, and after dressing changes; postoperative perianal fistula and abscess recurrence (observed during a clinic appointment or recorded as a SAE); bleeding (requiring transfusion or return to theatre); and rate of wound healing (at 4, 8, and 26 weeks). Further patient-reported secondary endpoints included HRQoL (EQ-5D), return to work, participant satisfaction with wound care, and participant global assessment of care.
Sample size
Sample size calculation was based on the primary outcome mean maximum pain intensity score over the first 10 days after surgery. Pilot data from the PPAC study demonstrated a mean (s.d.) maximum pain intensity score over days 1 to 5 of 40 (27) mm (VAS). Considering a clinically relevant difference in mean maximum pain intensity to be a reduction in severity by 20 per cent15,16 (an absolute reduction of 8 mm) and using a two-sided alpha level of 0.05, a sample size of 526 patients (263 in each arm) was required to obtain 90 per cent power (inclusive of a 10 per cent dropout rate). The sample size calculation was adjusted at interim analysis in November 2019. Standard deviation from trial data was less than predicted at 21.5 mm. Sample size was recalculated using this reduction from the assumed s.d. and an updated dropout rate. A sample size of 432 patients was required to provide 85 per cent power to detect a difference in mean maximum pain score of 20 per cent (allowing for the reported missing primary outcome data).
Statistical analysis
Statistical analysis was performed by the North West Surgical Trials Centre. Continuous data were summarized as means (s.d.) and medians (i.q.r.). Categorical data were assessed as frequency counts and associated percentages. Analyses were performed on an intention-to-treat principle retaining all participants in their randomized groups, irrespective of any protocol violations. A number of sensitivity analyses including a per-protocol analysis were conducted. The primary outcome was defined as the mean maximum pain score over 10 days following the incision and drainage surgery. Only participants who returned at least three pain measurements were included in the analysis. Missing data were handled by investigating the pattern of missingness between treatment groups and other patient characteristics. Multivariable logistic regression was performed to identify factors associated with missing primary endpoint data. The analysis of the primary outcome between groups was performed using a Wilcox rank-sum test. A P value of 0.05 was used to determine statistical significance. All analyses were performed using Stata (version 15; StataCorp, College Station, TX, USA).
Results
In total, 433 participants were recruited between 1 February 2018 and 1 February 2020 across 50 acute hospital NHS trusts in the UK. Of those randomized, 213/433 (49 per cent) were allocated to packing, and 220/433 (51 per cent) to no packing. Eighteen (8.0 per cent) in the packing group and 18 (8.1 per cent) in the no-packing group did not receive the allocated intervention (Fig. 1). The reasons for treatment arm crossover (crossover from packing to non-packing) were as follows: five found packing too painful; the cavity was too small to pack in three; three patients chose not to receive packing; three were crossed over in error; and four participants received a combination of packing and no packing. The reasons for crossover from no packing to packing are as follows: three participants chose to pack for fluid control/toileting, four made a medical decision to pack; eight participants chose to pack; and three were crossed over in error.
Fig. 1.
CONSORT diagram
Twenty-seven randomized participants were then found to not meet the inclusion criteria and therefore did not proceed in the trial (11/213 (5.0 per cent) of the packing group and 16/220 (7.3 per cent) of the no-packing group). There were 17 other major protocol deviations (9/213 in the packing group and 8/220 in the non-packing group). Follow-up was completed in June 2020, 6 months after the last participant was recruited. Baseline questionnaires and operative data were available for all participants. Baseline demographics were well matched (Table 1).
Table 1.
Patient demographics
| Variable | Overall (n = 433) | Packing group (n = 213) | No-packing group (n = 220) |
|---|---|---|---|
| Median (i.q.r.) age (years) | 42 (31–52) | 42 (14) | 42 (15) |
| Median (i.q.r.) height (cm) | 173 (166–180) | 172 (11) | 174 (10) |
| Median (i.q.r.) weight (kg) | 85 (70–96) | 84 (23) | 86 (21) |
| Smoking status | |||
| Never | 150 (35) | 75 (35) | 75 (34) |
| Current smoker | 178 (41) | 91 (43) | 87 (40) |
| Ex-smoker | 97 (22) | 42 (20) | 55 (25) |
| Missing | 8 (2) | 5 (2) | 3 (1) |
| Sex | |||
| Female | 144 (33) | 77 (36) | 67 (30) |
| Male | 289 (67) | 136 (64) | 153 (70) |
| Missing | 0 (0) | 0 (0) | 0 (0) |
| Diabetes | |||
| No | 359 (83) | 177 (83) | 182 (83) |
| Yes | 62 (14) | 28 (13) | 34 (15) |
| Missing | 12 (3) | 8 (4) | 4 (2) |
| Anticoagulants or immunosuppressants | |||
| No | 389 (90) | 186 (87) | 203 (92) |
| Yes | 39 (9) | 22 (10) | 17 (8) |
| Missing | 5 (1) | 5 (2) | 0 (0) |
In total, 98/213 (46 per cent) participants in the packing group and 107/220 (49 per cent) in the non-packing group completed the trial as per protocol. A similar loss to follow-up rate occurred in both arms (packing group: 65/207 (31 per cent); no-packing group: 67/220 (30 per cent)).
Missing data
Missing primary outcome data occurred for 87/213 (41 per cent) patients in the packing group and for 75/220 (34 per cent) in the no-packing group (162 patients in total (37 per cent)) (Table S1). There was no significant difference in the likelihood of missing data between treatment groups (OR 1.21, 95 per cent c.i. 0.80 to 1.83; P = 0.372 (Table S2)). Data on the key secondary outcomes of perianal fistula and abscess recurrence were available for 100 per cent of participants. These data were collected as SAEs from the participant’s clinical notes, even when the participant was lost to routine trial follow-up.
Primary outcome: mean maximum pain score over the first 10 postoperative days
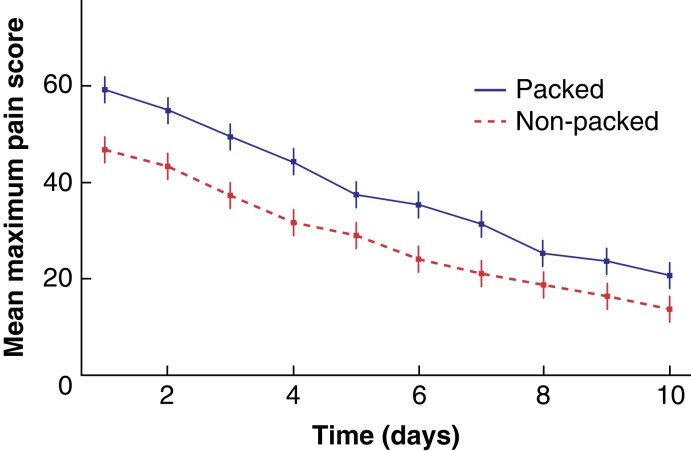
Mean maximum pain over the first 10 postoperative days was significantly lower in the no-packing group: the mean difference was 9.9 (95 per cent c.i. 4.84 to 15.05; P < 0.001), which was a 26.2 per cent reduction in pain in the no-packing arm versus the packing arm. Pain profiles over the 10 days following the incision and drainage operation are included in Fig. 2 and Table S3.
Fig. 2.
Daily mean maximum pain scores (visual analogue scale) over the first 10 postoperative days
This pain reduction was consistent under all sensitivity analyses, including a per-protocol analysis, which removed all major protocol deviations, including treatment arm crossovers; the mean maximum pain score difference was 13.7 (95 per cent c.i. 8.5 to 18.9; P < 0.001) (Table 2). In the packing group 126/213 (59 per cent) of participants had analysable primary outcome data versus 145/220 (66 per cent) participants in the no-packing group.
Table 2.
Primary outcome sensitivity analyses
| Sensitivity analysis | Packing group (n = 213) | No-packing group (n = 220) | Difference in mean maximum pain scores (95% c.i.) | P | ||
|---|---|---|---|---|---|---|
| No. patients with available data | Mean maximum pain (s.e.) | No. patients with available data | Mean maximum pain (s.e.) | |||
| Stratified mixed model | 126 | 38.2 (1.89) | 145 | 28.2 (1.77) | 10.8 (5.55–16.06) | <0.001 |
| Per protocol | 108 | 38.6 (2.05) | 121 | 24.9 (1.66) | 13.7 (8.48–18.89) | <0.001 |
| Outlier | 126 | 38.2 (1.89) | 144 | 27.8 (1.72) | 10.4 (5.38–15.45) | <0.001 |
| Excluding patients prescribed metronidazole | 104 | 38.2 (1.99) | 123 | 28.5 (2.02) | 9.7 (4.09– 15.28) | 0.001 |
Stratified model: results from a linear model, including patient site as a main effect. Per protocol: analysis removing 42 patients who had a major protocol deviation (including treatment arm crossover) and provided sufficient data to define the primary endpoint. Outlier: analysis removing patients identified as having outlying data points. Excluding participants prescribed metronidazole: analysis removing 44 patients who received metronidazole (possible confounder) and provided sufficient data for the primary analysis.
Missing primary outcome data was equal between groups. Exploratory analysis on treatment arm, sex, age (less than 40 years, more than 40 years), and smoking status investigated the relationship between these covariants and the probability of observing missing data. No difference was detected between the treatment arms (χ2 P = 0.176) or sex (P = 0.564). Participants aged 40 and over were more likely to return the symptoms diary (less than 40 years of age 41 per cent missing, more than 40 years of age 32 per cent missing; close to statistical significance P = 0.051). Current smokers were much more likely to have missing pain diaries (never smoked: 26 per cent missing; current smoker: 50 per cent missing; ex-smoker: 26 per cent missing (P < 0.001)). Multiple strategies were employed to minimize loss to follow-up (Table S2).
Secondary outcomes: pain before, during, and after dressing change
Pain scores both during dressing change (mean difference 10.2, 95 per cent c.i. 4.1 to 16.3; P < 0.001) and after dressing change (mean difference 5.8, 95 per cent c.i. 0.1 to 11.5; P = 0.007) favoured the no-packing group (Table 3).
Table 3.
Secondary outcomes
| Outcome | Level | Packing group | No-packing group | P |
|---|---|---|---|---|
| Pain prior to dressing change | n (%) | 104 (49) | 113 (51) | |
| Mean (s.e.) | 30 (1.9) | 26 (2) | 0.05 | |
| Pain at dressing change | n (%) | 104 (49) | 112 (51) | |
| Mean (s.e.) | 42 (2.1) | 32 (2) | <0.001 | |
| Pain following dressing | n (%) | 104 (49) | 113 (51) | |
| Mean (s.e.) | 32 (1.9) | 26 (2.1) | 0.007 | |
| Healing rate (at 26 weeks) | n | 163 | 175 | |
| No | 51 | 37 | ||
| Yes | 112 | 138 | 0.05 | |
| Metronidazole prescribed | n (%) | 202 | 212 | |
| No | 166 (78) | 174 (79) | 1 | |
| Yes | 36 (17) | 38 (17) | ||
| Patient Global Assessment | n (%) | 125 | 141 | |
| Poor | 6 (5) | 1 (1) | ||
| Fair | 29 (23) | 20 (14) | ||
| Good | 68 (54) | 88 (62) | ||
| Excellent | 22 (18) | 32 (23) | 0.03 | |
| Patient satisfaction | n | 116 | 134 | |
| Strongly disagree | 3(3) | 2 (1) | ||
| Disagree | 6 (5) | 0 (0) | ||
| Neutral | 8 (7) | 6 (4) | ||
| Agree | 37 (32) | 53 (40) | ||
| Strongly agree | 62 (53) | 73(54) | ||
| Median (i.q.r.) | 5 (4–5) | 5(4–5) | 0.06 | |
| Quality of life | n | 123 | 139 | |
| Day 1 | 58 (1.90) | 58 (1.85) | 0.76 | |
| Day 7 | 73 (1.82) | 76 (1.80) | 0.19 | |
| Day 14 | 84 (1.43) | 83 (1.80) | 0.86 | |
| Day 21 | 85 (1.75) | 86 (1.86) | 0.52 | |
| Overall | 67 (1.26) | 68 (1.27) | 0.66 |
Secondary endpoints
Fistula was seen by 6 months of follow-up in 32/213 (15 per cent) of the packing group and 24/220 (11 per cent) of the non-packing group (OR 0.69, 95 per cent c.i. 0.39 to 1.22; P = 0.204). Despite no detectable statistically significant difference, the OR of 0.69 reflects a likelihood of lower fistula rate in the no-packing group. However, this study was not powered to detect non-inferiority. These data suggest that one perianal fistula would be avoided for every 25 patients not packed (number needed to treat (NNT) 25). Abscess recurrence was low; 7/213 (3 per cent) in the packing group and 13/229 (6 per cent) in the no-packing group, likely favouring the packing group but with wide imprecision around the confidence intervals, and no statistically significant difference detected (OR 1.85, 95 per cent c.i. 0.72 to 4.73; P = 0.200). Again, this study was not powered to detect non-inferiority; however, these data suggest that one additional abscess recurrence would occur for every 37 patients not packed (NNT 37).
Further secondary endpoints are included in Table 3.
Wound healing (defined as complete epithelialization of the wound) was assessed repeatedly throughout the course of the study and further details are provided in Table S4. Over the 6-month follow-up period, a total of 112/163 (69 per cent) in the packing group and 138/175 (79 per cent) in the no-packing group were observed as healed (P = 0.046).
The use of metronidazole was similar between groups and was therefore unlikely to impact on the pain data.
Global assessment of care was reported as either good or excellent by 90/125 (72 per cent) and 120/141 (85 per cent) in the packing group and no-packing group, respectively (P = 0.033, Table 3). With respect to patient satisfaction with wound management: 99/116 (85 per cent) and 126/134 (94 per cent) of participants in the packing group and no-packing group, respectively, agreed or strongly agreed that they were satisfied with the way their wound was managed, and approached statistical significance (P = 0.062, Table 3).
Details on HRQoL for each component of the EQ-5DTM are included in Table S5. Quality of life was assessed on days 1, 7, 14, and 21. Both treatment groups showed a monotonic increase in the mean overall HRQoL. Comparisons between mean scores at each time point are included in Table S4 and showed no significant differences between the treatment groups.
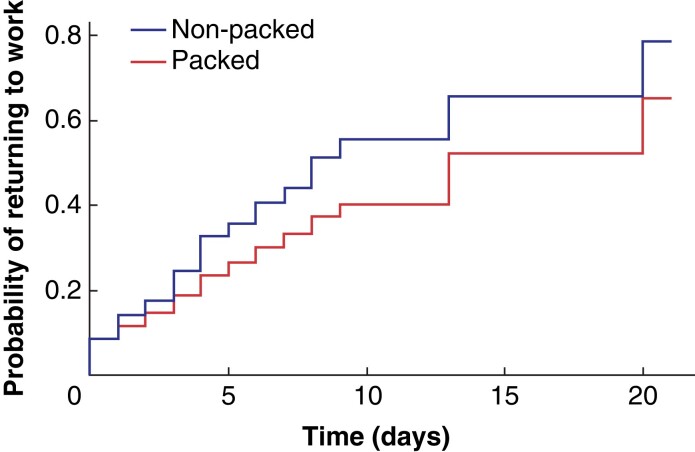
Return to work was investigated as a post-hoc secondary outcome. Sixty-five per cent of the packing group versus 80 per cent of the non-packing group had returned to work 21 days after their incision and drainage operation (Fig. 3).
Fig. 3.
Probability of returning to work over time after incision and drainage operation
Discussion
The PPAC2 trial has demonstrated that postoperative perianal abscess wound cavities managed without internal wound packing result in significantly less pain without significantly increasing adverse events of perianal fistula and abscess recurrence. Global satisfaction with care was greater in the no-packing group and a greater proportion of participants had returned to work by 21 days. The baseline demographics are consistent with those reported in a large population-based observational study6. One in six had diabetes mellitus, slightly higher than the 8 per cent reported1 in a series with fistula related to inflammatory bowel disease (a lower rate of concomitant diabetes is expected).
Pain in the no-packing group was significantly reduced compared with the packing group, quantified by the primary outcome of mean worst daily pain scores. Daily maximum pain was consistently lower in the non-packing group over the first 10 days. Additionally, pain was significantly higher in the packing group during dressing change, which persisted afterwards. This is consistent with data reported in our observational study7. Pain was identified by participants in the PPAC feasibility study, as the most important postoperative outcome7, and is now known to be a key measure of patient overall experience and satisfaction with their surgical care14.
The minimal clinically important difference (MCID) in pain resulting from the use of an analgesic intervention, pharmaceutical, or adjunct has been reported to be 20 per cent15,16 or 10 mm or greater17,18 on a VAS score. The PROSPECT working group, highlighting the lack of robust guidance, has designed methodology in order to develop international evidence-based recommendations for procedure-specific perioperative pain management18. This group has defined a MCID of 10 mm. The current study reports a difference of 9.9 mm, which can be rounded to 10 mm. The current study has also shown larger relative reductions in postoperative pain compared to other trials investigating pain in proctological surgery. The eTHoS trial compared stapled haemorrhoidopexy with traditional excisional surgery; the stapling procedure was less painful than traditional surgery (TS) with a 13 per cent reduction in worst pain on day 7 (10 mm, TS 7.9, SH 6.9, −1.04 effect size)19, a lesser effect size than reported here.
The loss of primary outcome data (although not of fistula or abscess recurrence data) is undeniably high. Despite this, the study has sufficient statistical precision to detect differences between the treatment groups. We believe the attrition and data loss is unlikely to have introduced significant bias and has had minimal impact on the strength of the trial findings. We are confident that this reflects the best possible data collection in this context, and that these data are robust. Strategies recommended by Mercieca-Bebber and colleagues to reduce missing patient-reported outcome data (PRO) included simplifying the PRO data set, using a measure acceptable to participants, reduced data and time points, and postal returns20. The loss of data may reflect the young working age population, data-entry fatigue, lack of motivation in absence of pain or other significant symptoms, the use of a pen-and-paper approach rather than electronic data entry, and also the nature of an acute, and possibly embarrassing, condition. Missing PRO data in clinical trials is a widely recognized issue21–23.
Perianal fistula was an important secondary outcome for this trial. Incidence of fistula up to 6 months of follow-up was similar between the packing group and the no-packing group. The OR reflects a likelihood of lower fistula rate in the non-packing group; however, this study was not powered to detect non-inferiority. The sample size needed to detect a true difference is 980, which was not achievable within the funding and time constraints of this study. These data suggest that one perianal fistula would be avoided for every 25 patients not packed (NNT 25). If this difference were true, reducing the rate of fistula would be a further important reason to avoid wound packing. Assessment of healing and fistula occurrence was made by individuals blinded to treatment allocation. The abscess recurrence rate was low—although no statistically significant difference between treatment groups was detected. These data suggest a likelihood of a higher rate of abscess recurrence rate in the non-packing group with one additional abscess recurrence for every 37 patients not packed (NNT 37). The sample size needed to detect a true difference is again higher than the funding and time constraints of this study allowed, at 900. Perianal fistula commonly requires further surgery and has greater long-term consequences for patients than an acute abscess recurrence. Arguably, a small increase in abscess recurrence risk would be a reasonable tradeoff for a reduction in fistula rate.
HRQoL was similar between the two groups. ‘Usual activities’ and ‘pain/discomfort’ were the most affected parameters, with 75 per cent of participants reported experiencing severe problems in their ‘usual activities’ at day 1. This dropped dramatically by day 7 and stabilized by day 14, with no further improvement beyond this point. Participants in both groups were generally back to usual activities within 2 weeks. It is now accepted that time to return to work is a key indicator of quality of life24. Although there was no identified difference in EQ-5DTM scores between the packing and no-packing groups, a greater proportion of participants in the no-packing group had returned to work at 21 days—a real-life measure of quality of life and functional status.
The main limitations of this trial are the loss of primary outcome data, and non-blinding of participants to their treatment. Non-blinding of participants may have led to bias in patient-reported outcomes. There was a small degree of non-compliance with treatment allocation. This trial was conducted in emergency general surgery units at 50 acute hospitals, including small general hospitals and large academic units, so the trial findings are widely applicable. The study has challenged the dogma of wound packing after incision and drainage of perianal abscess, and found it to be unwarranted.
Collaborators
A. Watson, M. Johnson, L. Hiller, E. Psarelli, L. Murray, A. Smith, S. Brown, B. Singh, C. Newby, O. Ali, A. Sukha, N. Blencowe, S. Narang, N. Reeves, G. Faulkner, S. Rajamanickam, J. Evans, S. Mangam, M. Harilingham, C. J. Smart, S. J. Ward, M. Bogdan, K. Amin, Z. Al-Khaddar, E. Davies, P. Patel, A. Stearns, I. Shaik, J. Hernon, A. Pal, M. Lewis, J. Barker, A. Gerrard, M. Abdel-Halim, P. Shuttleworth, M. J. Lee, A. B. P. Peckham-Cooper, A. G. Hague, C. Challand, C. Steele, N. Fearnhead, S. Van Laarhoven, R. Brady, F. Shaban, N. Wong, W. Ngu, G. Williams, R. Codd, D. Magowan, K. Leong, G. Williams, A. Torrance, B. Bharathan, N. Pawa, H. Kaur Sekhon, I. Singh, A. Alabi, D. Berry, V. Trompetas, J. L. Hughes, R. Lunevicius, R. Lunevicius, K. Mann, S. Dixon, T. Ingram, T. Gilbert, C. Brooks, G. Madzamba, A. Pullyblank, G. Dovell, L. Newton, N. Carter, P. May-Miller, S. Shaikh, R. Shearer, C.Macleod, C. Parnaby, A. Abdelmabod, L. Titu, T. Majeed, R. Hargest, J. Parker, C. Zabkiewicz, N. Reeves, F. Soliman, G. Gossedge, H. Selvachandran, M. Dilworth, D. Vimalachandran, H. Singh, H. Koh, J. Randall, S. Moug, A. Adeosun, G. Dennison, N. Curtis, N. Smart, S. Duff, M. Rahman, F. Wu.
Supplementary Material
Acknowledgements
Trial data presented at the Association of Coloproctology of Great Britain and Ireland congress June 2021; and the Association of Surgeons of Great Britain and Ireland congress May 2022.
Contributor Information
Katy Newton, Department of General Surgery, Manchester Royal Infirmary, Manchester University NHS Foundation Trust, UK.
Jo Dumville, School of Midwifery, Nursing and Social Work, University of Manchester, UK.
Michelle Briggs, School of Midwifery, Nursing and Social Work, University of Manchester, UK.
Jennifer Law, North West Research Collaborative, UK.
Julia Martin, North West Research Collaborative, UK.
Lyndsay Pearce, Department of General Surgery, Salford Royal NHS Foundation Trust, UK.
Cliona Kirwan, Department of Academic Surgery, Wythenshawe Hospital, Manchester University NHS Foundation Trust, UK.
Thomas Pinkney, Academic Department of Surgery, University of Birmingham, UK.
Alexander Needham, Liverpool Clinical Trials Unit, UK.
Richard Jackson, Liverpool Clinical Trials Unit, UK.
Simon Winn, Liverpool Clinical Trials Unit, UK.
Haley McCulloch, Liverpool Clinical Trials Unit, UK.
James Hill, Department of General Surgery, Manchester Royal Infirmary, Manchester University NHS Foundation Trust, UK.
PPAC2 Collaborators:
A Watson, M Johnson, L Hiller, E Psarelli, L Murray, A Smith, S Brown, B Singh, C Newby, O Ali, A Sukha, N Blencowe, S Narang, N Reeves, G Faulkner, S Rajamanickam, J Evans, S Mangam, M Harilingham, C J Smart, S J Ward, M Bogdan, K Amin, Z Al-Khaddar, E Davies, P Patel, A Stearns, I Shaik, J Hernon, A Pal, M Lewis, J Barker, A Gerrard, M Abdel-Halim, P Shuttleworth, M J Lee, A B P Peckham-Cooper, A G Hague, C Challand, C Steele, N Fearnhead, S Van Laarhoven, R Brady, F Shaban, N Wong, W Ngu, G Williams, R Codd, D Magowan, K Leong, G Williams, A Torrance, B Bharathan, N Pawa, H Kaur Sekhon, I Singh, A Alabi, D Berry, V Trompetas, J L Hughes, R Lunevicius, R Lunevicius, K Mann, S Dixon, T Ingram, T Gilbert, C Brooks, G Madzamba, A Pullyblank, G Dovell, L Newton, N Carter, P May-Miller, S Shaikh, R Shearer, C Macleod, C Parnaby, A Abdelmabod, L Titu, T Majeed, R Hargest, J Parker, C Zabkiewicz, N Reeves, F Soliman, G Gossedge, H Selvachandran, M Dilworth, D Vimalachandran, H Singh, H Koh, J Randall, S Moug, A Adeosun, G Dennison, N Curtis, N Smart, S Duff, M Rahman, and F Wu
Funding
This project is funded by the National Institute for Health and Care Research (NIHR) under its Research for Patient Benefit (RfPB) Programme (Grant Reference Number PB-PG-0815-20037). The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care. Trial was funded by the National Institute for Health and Care Research ‘Research for Patient Benefit’ programme (PB-PG: 0815-20037). NIHR Research for Patient Benefit reviewed, approved, and funded the study design (RfPB—PB-PG: 0815-20037 July 2016). The funder played no part in study design, data analysis, or writing of this report. All authors had full access to all trial data and had final responsibility for the decision to submit for publication.
Disclosure. The authors declare no conflict of interest.
Supplementary material
Supplementary material is available at BJS online.
Data Availability
The data is stored at the Liverpool Cancer Trials Unit as per the study's ethical approval, but is not available in a public repository.
References
- 1. Sahnan K, Askari A, Adegbola SO, Tozer PJ, Phillips RKS, Hart A et al. Natural history of anorectal sepsis. Br J Surg 2017;104:1857–1865 [DOI] [PubMed] [Google Scholar]
- 2. Eisenhammer S. The final evaluation and classification of the surgical treatment of the primary anorectal cryptoglandular intermuscular (intersphincteric) fistulous abscess and fistula. Dis Colon Rectum 1978;21:237–254 [DOI] [PubMed] [Google Scholar]
- 3. Hamadani A, Haigh PI, Liu ILA, Abbas MA. Who is at risk for developing chronic anal fistula or recurrent anal sepsis after initial perianal abscess? Dis Colon Rectum 2009;52:217–221 [DOI] [PubMed] [Google Scholar]
- 4. Parks AG, Gordon PH, Hardcastle JD. A classification of fistula-in-ano. Br J Surg 1976;63:1–12 [DOI] [PubMed] [Google Scholar]
- 5. Sainio P. Fistula-in-ano in a defined population. Incidence and epidemiological aspects. Ann Chir Gynaecol 1984;73:219–224 [PubMed] [Google Scholar]
- 6. Sahnan K, Askari A, Adegbola SO, Warusavitarne J, Lung PFC, Hart A et al. Persistent fistula after anorectal abscess drainage: local experience of 11 years. Dis Colon Rectum 2019;62:327–332 [DOI] [PubMed] [Google Scholar]
- 7. Pearce L, Newton K, Smith SR, Barrow P, Smith J, Hancock L et al. Multicentre observational study of outcomes after drainage of acute perianal abscess. Br J Surg 2016;103:1063–1068 [DOI] [PubMed] [Google Scholar]
- 8. Sugrue J, Nordenstam J, Abcarian H, Bartholomew A, Schwartz JL, Mellgren A et al. Pathogenesis and persistence of cryptoglandular anal fistula: a systematic review. Tech Coloproctol 2017;21:425–432 [DOI] [PubMed] [Google Scholar]
- 9. Vasilevsky CA, Gordon PH. The incidence of recurrent abscesses or fistula-in-ano following anorectal suppuration. Dis Colon Rectum 1984;27:126–130 [DOI] [PubMed] [Google Scholar]
- 10. Ommer A, Herold A, Berg E, Fürst A, Post S, Ruppert R et al. German S3 guidelines: anal abscess and fistula (second revised version). Langenbecks Arch Surg 2017;402:191–201 [DOI] [PubMed] [Google Scholar]
- 11. Williams G, Williams A, Tozer P, Phillips R, Ahmad A, Jayne D et al. The treatment of anal fistula: second ACPGBI Position Statement—2018. Color Dis 2018;20:5–31 [DOI] [PubMed] [Google Scholar]
- 12. Tonkin DM, Murphy E, Brooke-Smith M, Hollington P, Rieger N, Hockley S et al. Perianal abscess: a pilot study comparing packing with nonpacking of the abscess cavity. Dis Colon Rectum 2004;47:1510–1514 [DOI] [PubMed] [Google Scholar]
- 13. Perera AP, Howell AM, Sodergren MH, Farne H, Darzi A, Purkayastha S et al. A pilot randomised controlled trial evaluating postoperative packing of the perianal abscess. Langenbecks Arch Surg 2015;400:267–271 [DOI] [PubMed] [Google Scholar]
- 14. Berkowitz R, Vu J, Brummett C, Waljee J, Englesbe M, Howard R. The impact of complications and pain on patient satisfaction. Ann Surg 2021;273:1127–1134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Cepeda MS, Africano JM, Polo R, Alcala R, Carr DB. What decline in pain intensity is meaningful to patients with acute pain? Pain 2003;105:151–157. [DOI] [PubMed] [Google Scholar]
- 16. Farrar JT, Portenoy RK, Berlin JA, Kinman JL, Strom BL. Defining the clinically important difference in pain outcome measures. Pain 2000;88:287–294 [DOI] [PubMed] [Google Scholar]
- 17. Myles PS, Myles DB, Galagher W, Boyd D, Chew C, MacDonald N et al. Measuring acute postoperative pain using the visual analog scale: the minimal clinically important difference and patient acceptable symptom state. Br J Anaesth 2017;118:424–429 [DOI] [PubMed] [Google Scholar]
- 18. Joshi GP, Van de Velde M, Kehlet H, Pogatzki-Zahn E, Schug S, Bonnet F et al. Development of evidence-based recommendations for procedure-specific pain management: PROSPECT methodology. Anaesthesia 2019;74:1298–1304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Watson AJM, Hudson J, Wood J, Kilonzo M, Brown SR, Mcdonald A et al. Comparison of stapled haemorrhoidopexy with traditional excisional surgery for haemorrhoidal disease (eTHoS): a pragmatic, multicentre, randomised controlled trial. Lancet 2016;388:2375–2385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Mercieca-Bebber R, Palmer MJ, Brundage M, Calvert M, Stockler MR, King MT. Design, implementation and reporting strategies to reduce the instance and impact of missing patient-reported outcome (PRO) data: a systematic review BMJ Open 2016;6:e010938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Bylicki O, Gan HK, Joly F, Maillet D, You B, Péron J. Poor patient-reported outcomes reporting according to CONSORT guidelines in randomized clinical trials evaluating systemic cancer therapy. Ann Oncol 2015;26:231–237 [DOI] [PubMed] [Google Scholar]
- 22. Efficace F, Fayers P, Pusic A, Cemal Y, Yanagawa J, Jacobs M et al. Quality of patient-reported outcome reporting across cancer randomized controlled trials according to the CONSORT patient-reported outcome extension: a pooled analysis of 557 trials. Cancer 2015;121:3335–3342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Fielding S, Maclennan G, Cook JA, Ramsay CR. A review of RCTs in four medical journals to assess the use of imputation to overcome missing data in quality of life outcomes. Trials 2008;9:51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Peters S, Johnston V, Hines S, Ross M, Coppieters M. Prognostic factors for return-to-work following surgery for carpal tunnel syndrome: a systematic review. JBI Database of System Rev Implement Rep 2016;14:135–216 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data is stored at the Liverpool Cancer Trials Unit as per the study's ethical approval, but is not available in a public repository.