Abstract
The transposon Tn21 and a group of closely related transposons (the Tn21 family) are involved in the global dissemination of antibiotic resistance determinants in gram-negative facultative bacteria. The molecular basis for their involvement is carriage by the Tn21 family of a mobile DNA element (the integron) encoding a site-specific system for the acquisition of multiple antibiotic resistance genes. The paradigm example, Tn21, also carries genes for its own transposition and a mercury resistance (mer) operon. We have compiled the entire 19,671-bp sequence of Tn21 and assessed the possible origins and functions of the genes it contains. Our assessment adds molecular detail to previous models of the evolution of Tn21 and is consistent with the insertion of the integron In2 into an ancestral Tn501-like mer transposon. Codon usage analysis indicates distinct host origins for the ancestral mer operon, the integron, and the gene cassette and two insertion sequences which lie within the integron. The sole gene of unknown function in the integron, orf5, resembles a puromycin-modifying enzyme from an antibiotic producing bacterium. A possible seventh gene in the mer operon (merE), perhaps with a role in Hg(II) transport, lies in the junction between the integron and the mer operon. Analysis of the region interrupted by insertion of the integron suggests that the putative transposition regulator, tnpM, is the C-terminal vestige of a tyrosine kinase sensor present in the ancestral mer transposon. The extensive dissemination of the Tn21 family may have resulted from the fortuitous association of a genetic element for accumulating multiple antibiotic resistances (the integron) with one conferring resistance to a toxic metal at a time when clinical, agricultural, and industrial practices were rapidly increasing the exposure to both types of selective agents. The compendium offered here will provide a reference point for ongoing observations of related elements in multiply resistant strains emerging worldwide.
With the rich insights provided by the burgeoning information on entire prokaryotic genome sequences, it is important to recall that a significant percentage of the DNA in natural isolates of many eubacteria is in the form of extrachromosomal elements called plasmids. Many of these elements are transferable even very widely across species and genus boundaries. In addition to genes for their own maintenance and transfer, these ubiquitous genetic elements frequently carry embedded mobile genetic elements called transposons, which are capable of moving from one carrier replicon to another.
Much of what we understand about DNA replication and recombination in general has been learned through the study of such plasmids and transposons in the eubacteria. Although they are major agents of the horizontal gene exchange which complicates simple phylogenetic analyses of whole bacterial genome sequences, there are no compendia of the complete sequences of the genomes of these peripatetic members of the “floating genome.” We have compiled and annotated sequence information for one of the first recognized, largest, and most widely distributed examples of this group, the multiply composite transposon Tn21.
This analysis has been stimulated by the demonstrated involvement of the Tn21-family in the global dissemination of antibiotic resistance determinants (16–18) and consequent interest in the processes involved in the accumulation and exchange of resistance loci by plasmids and transposons. Horizontal and vertical transfer of antibiotic resistance genes in members of the family Enterobacteriaceae and Pseudomonas is mostly due to large broad-host-range plasmids and to the transposons they carry and share with other replicons (17). Many transposons encoding multiple antibiotic resistances in gram-negative members of the Enterobacteriaceae belong to the Tn21 subgroup of the Tn3 family of transposable elements (29, 63). Transposon Tn21 and many of its closest relatives carry within them a potentially independently mobile DNA element called an integron. The integron encodes a RecA-independent, site-specific integration system that is responsible for the acquisition of multiple small mobile elements called gene cassettes that encode antibiotic resistance genes (35, 60, 74, 104, 109). Tn21 also carries the genes involved in its own transposition (tnp) and a mercury resistance (mer) operon (29, 98).
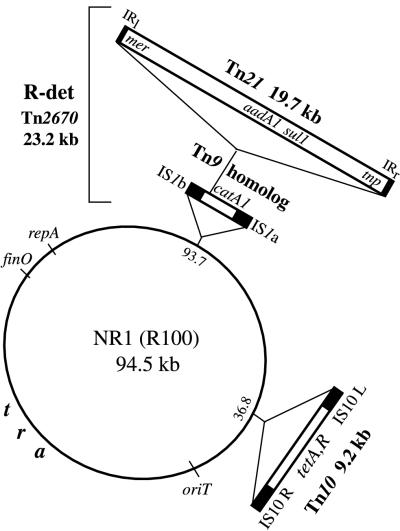
Transposon Tn21 itself is carried by plasmid NR1 (R100), which was originally isolated from Shigella flexneri in Japan in the late 1950s (70). NR1 is a 94.5-kb, self-transmissible, multiple-antibiotic resistance plasmid (Fig. 1) and is the archetype of a large collection of similar R-plasmids that have been discovered worldwide (18, 116). NR1, which belongs to the FII incompatibility group of R-plasmids, was initially considered to have two components: a resistance transfer factor (RTF), which carries the genes for self-transmissibility (tra) and autonomous replication (rep), and a resistance determinant (R-det), which contains a composite Tn9-like transposon, carrying the catA1 (catI) gene, and transposon Tn21 (120). The R-det component is bound by direct repeats (IS1a and IS1b) and is itself transposable as Tn2670 (37, 43). Tn21 is an independent transposon within the R-det and is bounded by inverted repeats IRl and IRr (19, 28, 127). Located in the RTF region of NR1 is transposon Tn10, which carries tetR (tet repressor) and tetA (confers tetracycline resistance) and is bounded by IS10R and IS10L (46). The other major functional loci within the RTF are the origin of conjugative transfer (oriT) gene and the transfer (tra) genes.
FIG. 1.
NR1, an IncFII self-transmissible, multiple-antibiotic resistance R plasmid. Inserts indicate mobile genetic elements.
Recently, the last section of the Tn21 transposon was sequenced (5), revealing that Tn21 itself includes four discrete mobile elements: a class 1 integron, the aadA1 gene cassette contained in the integron, and two insertion sequences, IS1326 and IS1353 (Table 1 and Fig. 2). We have assembled five overlapping GenBank sequences (accession no. X04891, M10791, X12870, U42226, and J01730 [Table 2]) and updated the annotation of the assembled sequence of 19,671 bp (GenBank accession no. AF071413 [Tables 3 to 5]). Due to the growing interest in integrons, we present the Tn21 sequence with the transposition genes on the left and the mer genes on the right, since this allows the components to be viewed in the orientation familiar to those studying the integron itself.
TABLE 1.
Transposon Tn21 and the mobile elements it contains
| Element | Coordinates | Size (bp) |
|---|---|---|
| Tn21 | 1–19671 | 19,671 |
| In2 | 4040a–15038 | 10,999 |
| aadA1 cassetteb | 5395–6250 | 856 |
| IS1326 | 8276–10719, 12333–12360c | 2,470 |
| IS1353 | 10720–12332 | 1,612 |
Duplication of 5 bp due to insertion of In2; coordinates 4035 to 4039 and 15039 to 15543.
The cassette is located within In2 but is independently mobile.
The sequence of IS1326 is interrupted by the insertion of IS1353, which creates a 2-bp target site duplication.
FIG. 2.
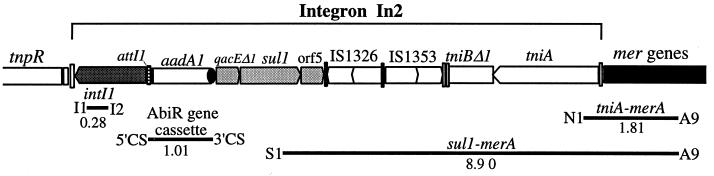
Symbols for the transposition (tnp) region, the integron, and the mer operon of transposon Tn21 (GenBank accession no. AF071413). Vertical bars indicate flanking inverted repeats (IR) of transposons and insertion sequences (IS). The tnp region consists of genes for the transposase (tnpA), the resolvase (tnpR), the putative transposition regulator (tnpM), and the resolution site (res) for Tn21. The 5′-CS of the integron includes the integrase gene (intI1) and the attI1 insertion site. The aadA1 gene cassette contains the aminoglycoside adenylyltransferase gene and the 59-be. The arrow indicates the direction of transcription. The 3′-CS includes genes encoding resistance to quaternary ammonium compound disinfectants (qacEΔ1) and sulfonamide resistance (sul1), an ORF (orf5) of unknown function, and two insertion sequences (IS1353 is inserted into IS1326). The tni (transposition of the integron) gene region has suffered deletion in Tn21, and only tniA and a portion of tniB remain. The mercury resistance (mer) operon consists of the regulatory genes merR and merD and the structural genes merT, merP, merC, and merA. There are two unknown reading frames, urf1 (also called merE) and urf2, downstream of merD. urf2M is a hypothetical gene defined in this work, which may have existed before integron insertion.
TABLE 2.
Sequenced regions comprising Tn21 (GenBank accession no. AF071413)
| Region | Coordinates | Reference(s) | Original GenBank accession no. |
|---|---|---|---|
| tnpA | 1–3056 | 115 | X04891 |
| tnpR, tnpM | 3057–4105 | 42 | M10791 |
| tnpR, <tnpMb | 22 | K01726a | |
| intI1, aadA1, (qacEΔ1, sul1) | 4106–7539 | 109 | X12870 |
| intI1, <aadA1 | 63 | M33633a | |
| qacEΔ1, sul1, orf5, istB, istA, orfA, orfB, tniBΔ1, tniA | 7540–14933 | 5, 33, 104 | U42226 |
| orf2, merE, D, A, C, P, T, R | 14934–19671 | 7, 65, 66 | J01730 |
| <merD, A, C, P, T, R | 1 | K03089a |
Not used in Tn21 assembly AF071413 but partial duplications of other regions used.
< indicates a partial gene sequence.
TABLE 3.
Features of the tnp transposition region of Tn21
| Feature | Coordinatesa |
|---|---|
| Tn21 IRtnp | 1–38 |
| tnpA gene | (34–3000) |
| tnpA −10 | (3063–3068) |
| tnpA −35 | (3086–3091) |
| tnpR gene | (3003–3563) |
| tnpR −10 | (3591–3596) |
| tnpR −35 | (3614–3618) |
| resIII | (3576–3605) |
| resII | (3610–3652) |
| resI | (3668–3704) |
| Recombination crossover point | (3684–3685) |
| tnpM gene | (3689–4039) |
| tnpM −10b | (4086–4091) |
| tnpM −35b | (4109–4114) |
| Alternate tnpM −10b | (4094–4099) |
| Alternate tnpM −35b | (4115–4120) |
Parentheses indicate that the ORF or control site is on the complementary strand in the sequence compiled in GenBank accession no. AF071413.
There are two possible −10 and −35 promoter regions for tnpM; however, neither conform well to the consensus sequence for ς−70 RNA polymerase. Both lie within the left end of In2.
TABLE 5.
Features of the In2 region of Tn21
| Feature | Coordinatesa | ||
|---|---|---|---|
| In2 boundaries | 4040–15038 | ||
| 5′ conserved segment | 4040–5394 | ||
| IRi | 4040–4064 | ||
| intI1 gene | (4242–5255) | ||
| Pint −10 | (5285–5290) | ||
| Pint −35 | (5308–5313) | ||
| Pc (P1) −35 | 5140–5145 | ||
| Pc (P1) −10 | 5163–5168 | ||
| P2 −35 | 5259–5264 | ||
| 3G insertion that creates P2 | 5278–5280 | ||
| P2 −10 | 5282–5287 | ||
| attI1 site | 5362–5400 | ||
| Core site, GTTRRRY gene cassette | 5394–5400 | ||
| aadA1 gene cassette | 5395–6250 | ||
| 5′ portion of 59-be | 5395–5400 | ||
| aadA1 gene | 5416–6195 | ||
| alternate start, aadA1 gene | 5404 | ||
| 78% identical to merO | 5491–5508 | ||
| 3′ portion of 59-be | 6197–6250 | ||
| Inverse core site, RYYYAAC | 6197–6203 | ||
| Central IR in 59-be | 6209–6225 | ||
| Central IR in 59-be | 6229–6243 | ||
| Core site, GTTRRRY | 6250–6256 | ||
| 3′ conserved segment | 6251–8275 | ||
| qacEΔ1 gene | 6359–6706 | ||
| Sequence diverges from qacE | 6650–6651 | ||
| sul1 gene | 6700–7539 | ||
| Potential 59-be | 7534–7567 | ||
| orf5 gene | 7667–8167 | ||
| IS1326 boundaries | 8276–12360 | ||
| IS1326 IR | 8276–8301 | ||
| istB gene | (8343–9128) | ||
| istA gene | (9115–10638) | ||
| istAB −10 | (10711–10716) | ||
| IS1353 boundaries | 10720–12332 | ||
| IS1353 IR | 10720–10731 | ||
| orfAB −35 | 10738–10743 | ||
| orfAB −10 | 10762–10767 | ||
| orfA gene | 10791–11429 | ||
| Frame shift | 11396–11402 | ||
| Stem-loop | 11407–11462 | ||
| orfB gene | 11390–12304 | ||
| IS1353 IR | 12320–12332 | ||
| IS1326 IR | 12335–12360 | ||
| istAB −35 | (12348–12353) | ||
| tni region | (12355–14990) | ||
| tniBΔ1 gene | (12355–13215) | ||
| tniA gene | (13218–14933) | ||
| tniAB −10 | (14963–14968) | ||
| tniAB −35 | (14985–14990) | ||
| IRt | 15014–15038 | ||
| 5-bp direct repeat of target | 15039–15043 |
Parentheses indicate that the ORF or control site is on the complementary strand in the sequence compiled in GenBank accession no. AF071413.
TRANSPOSITION COMPONENTS
Tn3-Like Transposable Elements—Subtype Tn21
Tn3-like transposable elements are characterized by flanking inverted repeats of about 38 bp and have two genes, tnpA and tnpR, which encode a transposase and resolvase, respectively (29, 95). The tnpR gene and the res site (the site at which the resolvase acts) are located just upstream of tnpA. In the Tn21 transposon subgroup, the tnpA and tnpR genes are transcribed in the same direction. The TnpA proteins of this transposon family are at least 70% homologous to each other, as are their TnpR proteins (29). Transposases from this family share, as do many other transposases, a DD(35)E motif, which may form part of the catalytic center (125).
Tn21, a Class II Replicative Transposon
Transposition of transposon Tn21 is carried out by the transposase, TnpA (29). The sites of insertion, defined by a 5-bp duplication of target DNA, are usually AT rich but have no other obvious consensus sequence (77). Replicative transposition of Tn21 involves specific recognition of and binding to the terminal inverted-repeat (IR) elements of Tn21 by TnpA, which then mediates joining of the donor and recipient replicons (77). This cointegrate intermediate contains a copy of Tn21 and is resolved by the action of the tnpR gene product, a resolvase which acts at a specific site (res) adjacent to the tnpR gene (85).
The resolvase catalyzes site-specific recombination between supercoiled DNA and two directly repeated copies of the transposon DNA (102). Resolvase subunits bind to the three subsites (I, II, and III) of res within both recombination loci (100). These protein-DNA assemblies at each res site then interact with each other to form a synaptic complex (101) in which the resolvase separates the cointegrate to give a new recombinant replicon with one copy of the transposon and the original donor replicon, which retains its copy. In the Tn21 subgroup of transposons, the transposase-IR interactions and the resolvases are interchangeable; e.g., Tn21 tnpA can act on the Tn501 IRs and Tn21 tnpR can act on the Tn501 res site (29).
tnp transposition module.
The positions of the important features of the tnp region of Tn21 are presented in Table 3, and those of the IRmer region are presented in Table 4. The tnp region begins with the left IR (designated IRtnp) and extends to include the res site. The right end of Tn21 (designated IRmer) lies beyond the end of the merR gene. These left and right Tn21 IRs are 38-bp imperfect repeats (three base differences). tnpA is the largest structural gene in the transposon, encoding a 988-amino-acid (aa) transposase which terminates 5 bp into IRtnp (115). An independent promoter for tnpA has been suggested (29, 42) to occur within the last 80 bp of the tnpR gene. tnpR is 561 bp and terminates 2 bp before the putative initiation codon of tnpA. The predicted promoter region for the tnpR gene (and possibly the tnpA gene) is located within the res site (22, 31). The 129-bp res site has three resolvase binding sites (I, II, and III) with the recombination crossover point located in resI (85).
TABLE 4.
Features of the mer region of Tn21
| Feature | Coordinatesa |
|---|---|
| Tn21 IRmer | 19634–19671 |
| merR gene | (19204–19638) |
| merTPCAD −35 | (19193–19198) |
| merTPCAD −10 | (19168–19173) |
| merR −10 | (19166–19171) |
| merR −35 | (19145–19150) |
| merT gene | (18782–19132) |
| merP gene | (18493–18768) |
| merC gene | (18035–18457) |
| merA gene | (16289–17983) |
| merD gene | (15909–16271) |
| merE gene | (15676–15912) |
| urf2 putative gene | (14972–15679) |
| urf2M putative geneb | (3689–4039) + (15044–15679) |
Parentheses indicate that the ORF or control site is on the complementary strand in the sequence compiled in GenBank accession no. AF071413.
Newly defined here (see the text).
Putative tnpM.
An adjacent putative gene of undefined function, tnpM (351 bp), was originally defined as starting at the junction of the tnp region and the integron IRi (leftward inverted repeat) and terminating within resI (42). The ribosomal binding site and the promoter −10 and −35 regions for this putative tnpM gene are located within the integron, in IRi and in the noncoding region downstream of the intI1 gene, respectively.
Although the roles of tnpA and tnpR have been defined, regulation of the tnp genes in Tn21 is less well understood. Studies by de la Cruz and Grinsted with Tn21 (19) and by Tanaka et al. with Tn2603 (112) showed that transposition and resolution functions were intact in deletion derivatives which left the tnpA, tnpR, and tnpM reading frames intact but disrupted the putative −35 promoter region of tnpM. These authors concluded that tnpM was not necessary for transposition. However, Hyde and Tu (42) proposed that the tnpM gene produces a protein which enhances Tn21 transposition and suppresses cointegrate resolution. The basis for their proposal was the finding that a strain carrying a nonsense mutation in the tnpM reading frame was less effective than its parent in effecting Tn21 transposition and resolution. Furthermore, they observed that when a multicopy derivative carrying only Tn21 tnpM was present, transposition of Tn21 increased by 2.5-fold and resolution decreased 22-fold. They also noted an open reading frame (ORF) in Tn501 (orf2) at the same relative location and orientation, whose product was 84.6% similar to the putative Tn21 TnpM protein (Fig. 3). These similarities led Hyde and Tu to examine whether transposition and resolution of Tn501 was susceptible to modulation by Tn21 tnpM. They found that with the Tn21 tnpM gene in trans, transposition of Tn501 also increased by 2.5-fold and resolution decreased by 1.5-fold. However, no TnpM protein has been demonstrated. Thus, at present, neither the exact nature of the control for TnpA and TnpR nor the role of the putative TnpM is understood. We offer here the possible alternative that the tnpM reading frame is actually derived from a longer ORF which was interrupted by the insertion of In2 (see below). These alternatives are not necessarily mutually exclusive.
FIG. 3.
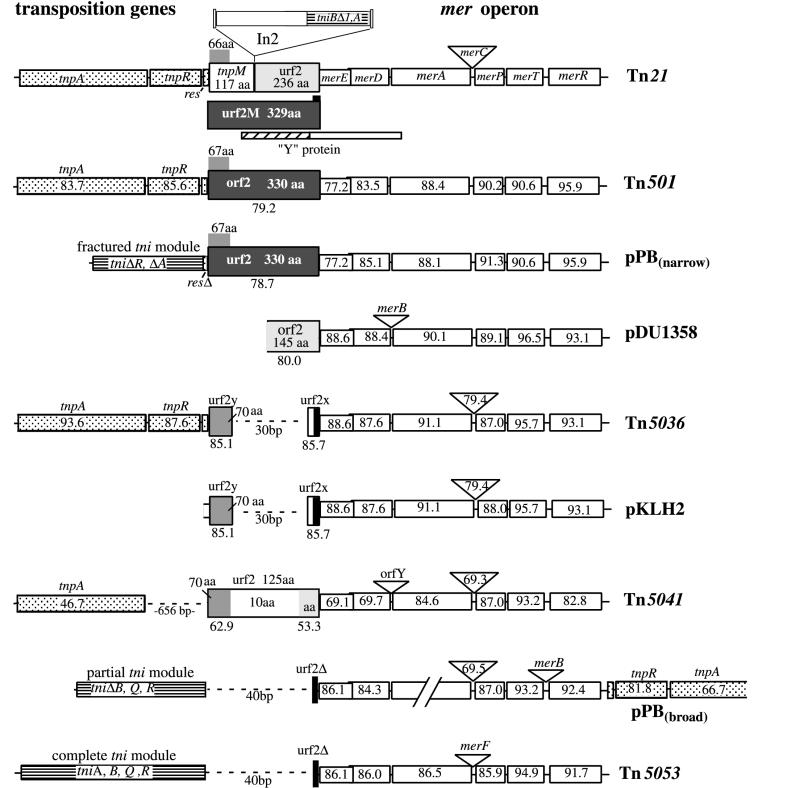
Defined or hypothetical proteins of Tn21, the closely related Tn501 and Tn5036, the distantly related transposons Tn5041 and Tn5053, and other mer operons in plasmids pDU1358, pKLH2, and pPB (narrow- and broad-spectrum mercury resistance operons). References are given in Table 6. Numbers in boxes and triangles representing mer or transposition genes are percent amino acid similarities to corresponding proteins of Tn21. Tall shaded boxes represent regions similar to putative Tn21 Urf2 (light gray), to the last 66 aa of putative TnpM (medium gray), or to hypothetical Tn21 Urf2M (dark gray). Black boxes represent small regions (5 to 7 aa) of similarity to the first 7 aa in Urf2M. Numbers below the tall shaded boxes are percent similarities to corresponding proteins in Tn21. Horizontal striped boxes represent tni genes, and spotted boxes represent tnp genes. The slashed box represents a region similar to Tn21 Urf2M and Y proteins. merA of the pPB broad-spectrum mer operon has not been sequenced.
MER OPERON
Mercury Resistance
Divalent mercuric ion, Hg(II), has a high affinity for thiol groups of proteins and for this reason is extremely toxic in biological systems. The current model of the mercury resistance (Hgr) mechanism of gram-negative bacteria posits that Hg(II) enters the periplasm and is bound to a pair of cysteine residues in MerP which transfer Hg(II) to cysteine residues in MerT or MerC. The latter move the ion across the cytoplasmic membrane in a series of ligand exchange reactions, resulting in the delivery of Hg(II) to the active site of the cytoplasmic flavin disulfide oxidoreductase, mercuric reductase (MerA) (98). Mercuric reductase catalyzes the reduction of Hg(II) to the highly volatile and less reactive monoatomic gas, Hg0, which diffuses from the cell and from the medium.
mer Genes
The mer region (Table 4) (1, 65, 66), flanked on its right by Tn21 IRmer and on its left by the rightward IR (designated IRt) of the integron, is sufficient to confer resistance to Hg(II). The mercury resistance operon of Tn21 contains two regulatory genes, merR and merD, and four structural genes, merT, merP, merC, and merA, encoding the NADH-dependent flavin oxidoreductase, mercuric reductase (MerA); two inner membrane proteins (MerT and MerC); and a periplasmic protein (MerP). Further genes may also play a role in mercury resistance.
Metalloregulator (merR).
The merR gene is at the opposite end of the transposon to the tnpA gene, and, like tnpA, merR terminates 5 bp into the IR bounding Tn21. The merR gene and the merTPCAD genes are transcribed divergently from a compact 71-bp promoter/operator (merOP) region in which their respective −10 RNA polymerase recognition hexamers overlap by 4 bp (39, 73). MerR binds a dyadic operator (merO) in the 19-bp spacer between the merTPCAD −35 and −10 promoter hexamers, and in the absence of Hg(II) it represses their expression. Since the dyadic binding site is also superimposed on the merR transcriptional start position, MerR represses its own synthesis (96). The metal binding domain of MerR, containing residues from Cys80 to Cys128, is sufficient to form a single, dimeric, trigonally coordinate, high-affinity Hg(II) binding site (126). When MerR binds Hg(II), an allosteric change in MerR allows transcription of the mer structural genes without changing the level of slightly leaky repression of MerR (86, 107). MerR also sequesters RNA polymerase at the merT promoter, forming a stable preinitiation complex (39). MerR contacts the α, β, and ς70 subunits of RNA polymerase both in the preinitiation and Hg(II)-activated promoter complexes (51).
Mercury transport genes (merT, merP, and merC).
The products of the first three structural genes of the mer operon are involved in uptake of Hg(II) into the cell. These small genes are tightly spaced, with the merT gene (351 bp) separated by only 13 bp from the merP gene (276 bp) and merP separated from the merC gene (423 bp) by 35 bp. Both MerT and MerC are inner membrane proteins (106) and are predicted to have three and four transmembrane-spanning α-helices, respectively. MerP is located in the periplasm (106) and is composed of four β-strands and two α-helices (103). Both cysteine residues (Cys33 and Cys36) in MerP are required for Hg(II) binding in vitro (90), and this is one of only two proteins of the operon whose structure is presently known (103). MerP is believed to transfer Hg(II) to the amino-terminal cysteine pair (Cys24 and Cys25) of MerT (36, 67). How Hg(II) is then transferred inside the cell to MerA is unknown. The MerP sequence constitutes the index example of the HMA motif found in several prokaryotic metal transporters (62, 72, 97) and in the human Cu transporters, MNK and WND (8).
The merC gene is apparently not required for Hg(II) resistance, if MerP and MerT are present, since there are functional mer operons without merC, e.g., those found in transposons Tn501 and Tn5053 (Fig. 3) (98). Moreover, deleting merC from the Tn21 mer locus had no effect on Hg(II) resistance (36). However, the Thiobacillus mer loci do not have the merT or merP genes but have at least three distinct merC genes (Table 6) (40), and Thiobacillus MerC can function alone as a mercury transport system in Escherichia coli (52). Tn21 MerC binds Hg(II) (91), possibly via a heavy-metal consensus binding region similar to those found in MerP and in the amino-terminal region of MerA. The actual interactions of MerP, MerT, MerC, and MerA remain to be defined.
TABLE 6.
Sequences of mer loci from gram-negative bacteria
| Common name | Source and genes sequenced | Accession no. | References |
|---|---|---|---|
| Tn21 | Plasmid NR1 from Shigella flexneri merRTPCAD, urf1, urf2 | AF071413 | 7, 65, 66; this work |
| Tn5036 | Tn5036; plasmid pKLH256 from Enterobacter cloacae merRTPCAD, urf1 (E), urf2X, urf2Y | Y09025 | 124 |
| pMER610 | Plasmid pMER610 from Alcaligenes sp. merRTPCAD urf1 (E), urf2X, urf2Y | Y08993 | 124 |
| pKLH272 | Plasmid pKLH272 from Enterobacter agglomerans merRTPCAD, urf1 (E), urf2X, urf2Y | Y08992 | 124 |
| pKLH2 | Plasmid pKLH2 from Acinetobacter calcoaceticus merRTPCADE, urf2X, urf2Y | L04303 | 47 |
| pDU1358 | Plasmid pDU1358 from Serratia marcescens merRTPABD, orf1, orf2 | M24940, Z49200, M15049 | 27 |
| Tn5053 | Tn5053; Xanthomonas sp. strain W17 chromosome merRTPFADE, urf2 | L40585 | 49 |
| pPB | Plasmid pPB from Pseudomonas stutzeri merRTPAD, urf1, urf2 and merRBTPCAD, urf1, urf2Δ | U80214, U81032, U90263 | 84 |
| Tn501 | Plasmid pVS1, Tn501 in Pseudomonas aeruginosa merRTPAD, orf1, orf2 | Z00027 | 6, 65 |
| Tn5041 | Tn5041, Pseudomonas strain KHP41 merRTPCAYD, urf1, urf2 | X98999 | 124 |
| Thiobacillus | Thiobacillus ferrooxidans E-15 chromosome merR2, merC2, orf1, orf2, orf3, merC1, merR1 and urf, merC, merA | X57326D90110 | 44,45 |
Mercuric reductase gene (merA).
merA, which begins 51 bp on the 3′ side of merC, encodes a cytoplasmic, redox enzyme (564 aa) and is the largest gene in the mer operon. MerA belongs to the class I pyridine nucleotide-disulfide oxidoreductases and has sequence and structural homology to glutathione reductase and other disulfide oxidoreductases (93). The amino terminus of merA and of the merP gene (signal sequence excluded) are extensively homologous, indicating a fusion between ancestral genes for MerP and for glutathione reductase. MerA functions as an α2 homodimer, and two pairs of cysteine residues, a redox-active pair (Cys135 and Cys140) from one subunit of MerA and a carboxyl-terminal pair (Cys561 and Cys562) from the other subunit of MerA, form the catalytic site (21). MerA uses two hydrides from two NADPH molecules for the reduction of flavin adenine dinucleotide, which in turn reduces Hg(II) bound to the redox-active cysteines of the enzyme (89, 93). Studies by Miller et al. (64) have shown that pyridine nucleotide complexed dimers of the enzyme are asymmetric. Since the reduced enzyme EH2-NADPH complex is the reductant of Hg(II), these observations suggest that the enzyme may function asymmetrically during catalysis. Miller et al. (64) have proposed an alternating-sites model for the catalytic reduction of Hg(II), where both subunits of the dimer function in catalysis but the steps are staggered, allowing subunits to reverse roles after part of the reduction so that the enzyme can both bind Hg(II) very tightly and reduce it (64).
Secondary regulatory gene (merD).
The most promoter-distal experimentally defined gene of the mer operon, merD (363 bp), begins 17 bp downstream of merA (54). The MerD protein is made in very small amounts (54). The suggested role of MerD is that of a down-regulator of the mer operon which binds weakly to the merOP region; MerD null mutants show increased operon expression (69).
Putative merE (urf1).
Two other reading frames at the end of the mer operon in Tn21 are also frequently found in other mer operons (Fig. 3), but experimental definition for them is lacking. The first of these, which overlaps the 3′ end of merD by 4 bp, is a 237-bp ORF variously designated orf1 (6, 27), urf1 (7, 84, 124), urf (45), or merE (47, 49) (Table 6). Putative merE has a GTG start codon and an appropriately placed Shine-Dalgarno consensus sequence (AGGAGG) and encodes a predicted polypeptide of 78 aa. There is no evidence to indicate whether this ORF is transcribed or translated, and no null or nonsense mutations in it have been examined for their effect on Hg resistance. However, 10 highly similar (80.4% ± 8.6% as determined by GAP; Genetics Computer Group, Inc. [GCG], Madison, Wis.) putative MerE protein sequences (listed in Table 6) from other mer loci of gram-negative bacteria have been reported. Of the 10 putative merE genes, 9 lie in the same position and orientation as the urf1 in the Tn21 mer locus (Fig. 3) and the other is located 5′ to the merC gene of a Thiobacillus mer locus (Table 6) (45). The putative Tn21 merE is also 58.0% similar to merT (previously called ORF2) in the mer locus of a gram-positive bacterium, Bacillus sp. strain RC607 (114). These 12 sequences are the only homologs of Tn21 merE in the database.
Cysteine pairs are characteristically found in Mer proteins, and putative MerE has a cysteine pair (Cys28 and Cys30) which, with adjacent residues (LTCPCHL), is conserved in all of these putative MerE polypeptides. The predicted secondary structure of MerE would have two transmembrane helices with the cysteine pair positioned approximately in the middle of the first helix (87, 99). That the first cysteine pair is also found in the same predicted position in MerT, MerC, and MerF suggests that MerE may play a role in Hg(II) transport.
urf2 and the Hypothetical urf2M
Overlapping the 3′ end of merE by 4 bp is the second reading frame of unknown function, urf2 (708 bp), whose stop codon lies 67 bp inside the right end of the integron rightward boundary at IRt. However, if the integron and one of its flanking 5-bp direct repeats were removed, urf2 would fuse in frame with the putative tnpM gene to produce a 987-bp hypothetical gene which we designate Tn21 urf2M (Fig. 3). Its 328-aa predicted product, Urf2M, is 76.9% identical and 79.3% similar to the predicted 329-aa product of orf2 found in transposon Tn501. Transposon Tn501 is a Tn21 subgroup transposon with a gene organization similar to that of Tn21 but lacking both merC and the integron (29). Also, the predicted product of urf2 in the mer operon of plasmid pPB is 78.7% similar to Tn21 Urf2M. The predicted product of orf2 (an incomplete sequence) in the mer operon of plasmid pDU1358 is 80.0% similar to Tn21 Urf2M.
Further, part of Tn21 Urf2M (226 of 328 aa) (Fig. 3) is also about 62% similar to a group of hypothetical proteins which belong to the YEGE/YHDA/YHJK/YJCC family of proteins (4, 121). These putative proteins (referred to here as Y proteins) are characterized by transmembrane regions and tyrosine kinase domains as found in two-component signal regulators (41). The carboxyl termini of the Y proteins are 62.3% ± 1.0% similar (40.3% ± 3.5% identical) to Tn21 Urf2M beginning at aa 22 and ending 35 aa into the beginning of putative TnpM (a total of 226 contiguous residues). This similarity to the carboxyl termini of the Y proteins also occurs in Tn501 Orf2 and pPB Urf2.
These similarities suggest that before In2 insertion, a distinct insertion or deletion occurred, forming an ORF (encoding a 328-aa product) between the mer and tnp genes or within a gene at the 3′ end of an ancestral mer operon. Possible remnants of this event are the last 66 aa of Tn21 TnpM, the last 67 aa of Tn501 Orf2 and pPB Urf2, and the last 70 aa of Tn5041 Urf2, pKLH2 Urf2y, and Tn5036 Urf2y, which are conserved (including three conserved cysteine pairs) (Fig. 3). Ribosomal binding sites in front of possible translation start sites occur in the genes encoding each of the aforementioned 66-, 67-, and 70-aa predicted products, suggesting that a smaller gene may be present in the larger ORFs.
To assess the possible origin of Tn21 urf2M and Tn501 orf2, we compared codon usage patterns of Tn21 urf2M with those of the Tn21 and Tn501 merA and tnpA genes (Tables 7 and 8). The GCG program Correspond (24) compares two codon frequency tables (20) and yields the residuals statistic D2 (Table 7, footnote b), which, when greater than 3, indicates different codon usage patterns. D2 values of <3.0 are consistent with a common origin for the genes (24). We largely limited our analysis to sequences of >1,000 bp since we observed that D2 increased when the sequence was small.
TABLE 7.
Comparison of codon usage in tnpA, merA, and the tnp-mer junction of Tn21 family transposons and a distantly related transposon, Tn5041
| Genes compareda
|
D2b | |||
|---|---|---|---|---|
| Gene A | Length (bp) | Gene B | Length (bp) | |
| Tn21 tnpA | 2,967 | Tn501 tnpA | 2,967 | 0.62 |
| Tn5036 tnpA | 2,973 | 0.97 | ||
| Tn5041 tnpA | 2,988 | 2.55 | ||
| Tn21 merA | 1,695 | Tn501 merA | 1,686 | 0.41 |
| Tn5036 merA | 1,686 | 0.50 | ||
| Tn5041 merA | 1,680 | 2.72 | ||
| Tn21 urf2M | 987 | Tn21 merA | 1,695 | 1.00 |
| Tn21 tnpA | 2,967 | 3.21 | ||
| Tn501 orf2 | 990 | 1.31 | ||
| Tn501 merA | 1,686 | 0.98 | ||
| Tn501 tnpA | 2,967 | 3.61 | ||
Tn21, Tn501, and Tn5036 belong to the Tn21 family of transposons; Tn5041 is distantly related to them (124).
The D2 statistic, calculated with the GCG program Correspond, compares the pattern of codon usage generated by the GCG program CodonFrequency (20). D2 is the sum over all codons of the residual equation D2 = (frequencycodon table 1 − frequencycodon table 2)2. D2 increases as patterns of codon usage become more dissimilar. D2 values of <3.0 are consistent with a common origin for the genes (24). (The significance of D2 decreases with sequence sizes of 1,000 bp).
TABLE 8.
Comparative codon usage in Tn21
| Tn21 gene | Length (bp) |
D2a with respect to:
|
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| tnpA | intI1 | aadA1 | qacEΔ1 | sul1 | istAB | orfAB | tniA | urf2M | merA | ||
| tnpA | 2,967 | 0 | |||||||||
| intI1 | 1,014 | 1.28 | 0 | ||||||||
| aadA1 | 792 | 5.09 | 4.31 | 0 | |||||||
| qacEΔ1 | 348 | 10.50 | 7.98 | 5.83 | 0 | ||||||
| sul1 | 840 | 1.69 | 1.85 | 3.97 | 8.20 | 0 | |||||
| istAB | 2,310 | 4.30 | 4.50 | 1.89 | 5.41 | 3.53 | 0 | ||||
| orfAB | 1,554 | 3.25 | 2.82 | 1.64 | 5.08 | 2.52 | 1.75 | 0 | |||
| tniA | 1,716 | 0.62 | 0.88 | 4.08 | 9.36 | 1.10 | 4.05 | 2.67 | 0 | ||
| urf2M | 987 | 3.20 | 3.47 | 4.54 | 6.80 | 3.33 | 3.27 | 1.53 | 3.26 | 0 | |
| merA | 1,695 | 2.73 | 3.40 | 4.93 | 8.84 | 3.90 | 3.74 | 2.25 | 2.78 | 1.00 | 0 |
See Table 7, footnote b.
The D2 values, 0.98 to 1.31, indicated that Tn21 merA, Tn501 merA, and Tn501 orf2 have similar codon usage patterns to Tn21 urf2M (Table 7). Higher D2 values (3.21 and 3.61) indicated that Tn21 tnpA and Tn501 tnpA have dissimilar codon usage patterns compared to Tn21 urf2M. Codon usage in each of the two ORFs which comprise the putative Tn21 urf2M (tnpM and urf2) shows that each is as distant from tnpA and as close to merA as is the putative composite gene; thus, by the D2 statistic, neither component gene is related to tnpA or to merA any differently than the putative fused gene. We also noted a surprisingly low D2 value of 1.13 between one of the genes which encodes the Y protein CY02B10.18C (Mycobacterium tuberculosis) and Tn21 urf2M. On the basis of these comparisons, Tn21 urf2M is more likely to have been associated longer with the mer genes than with the tnp genes, although its function in mercury resistance and its relationship to the Y proteins is unknown. An alternative hypothesis is that the origins of these ORFs cannot be discerned by codon usage comparisons because they are nonfunctional and therefore are freer to drift than is a functional gene.
IN2 INTEGRON
Integrons are a major vehicle for the spread of multiple-antibiotic resistance, and for class 1 integrons, this has been aided by the piggybacking of integrons on other transposons as in Tn21 and its relatives. Integrons provide the cassette-associated resistance genes with two advantages: a “built-in” expression system and ready mobility. Since the promoters located in the 5′-conserved sequence (5′-CS) of the integron (upstream of the inserted cassette) are functional in a wide range of bacteria, the integron can effect the coexpression of several cassette-associated antibiotic resistance genes (11). Cassette-associated resistance genes also harness several mechanisms for their dispersal. Not only are cassettes themselves mobile (10), but also the integrons into which they incorporate are transposable (5). Finally, integrons often occur in plasmids, many of which have broad host range, assisting interspecies spread (16, 81, 104). Integrons and gene cassettes have been reviewed elsewhere (34, 35, 80, 81).
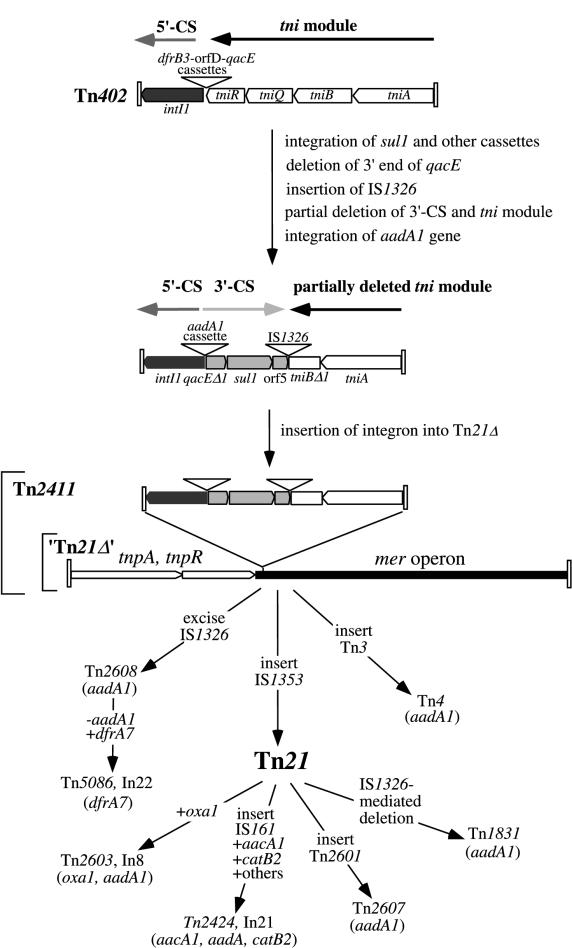
The integron found in Tn21 is a class 1 integron, designated In2 (104), that includes one integrated gene cassette, the aadA1 cassette. At 11 kb, In2 accounts for over half of the total length of Tn21. In2 is flanked by imperfect inverted repeats of 25 bp (IRi and IRj) and bounded by a 5-bp direct duplication of the target sequence (5, 7), indicating that it has transposed into an ancestral tnp-mer transposon to create Tn21. In2 does not include a complete set of transposition (tni) genes and cannot transpose itself (5). However, movement of In2 has occurred, presumably by using transposition proteins supplied in trans, because close relatives of In2 are found in other independent locations (5).
In2 is a composite element that contains a single cassette and two insertion sequences (ISs). The backbone of In2 consists of three distinct regions: 5′-CS, the 3′-conserved segment (3′-CS), and vestiges of the tni transposition gene module (Fig. 2). The 5′-CS and 3′-CS lie on either side of the cassette insertion point, which in In2 is occupied only by the aadA1 cassette. A remnant of the tni module is located to the right of two IS elements, IS1326 and IS1353. One or more deletions originating at the right boundary of IS1326 may have eliminated most of the tni genes. Similarly, a deletion originating at the left end of IS1326 has removed part of the 3′-CS (5). IS1353, which is located within IS1326, is presumed to have been acquired after the integron moved into Tn21, because close relatives of Tn21, e.g., Tn2411, that do not contain IS1353 are known (5, 28). The aadA1 cassette is also found in Tn2411 and may have been present in the In2 progenitor that inserted into the tnp-mer transposon. These six regions are described in detail below.
5′-Conserved Segment
The 5′-CS or intI module of class 1 integrons extends from the left IR, designated IRi, to the boundary of the first integrated cassette (Fig. 2). The 5′-CS includes three repeats at its left end that are predicted to bind the tni transposase (79) (described in more detail below): the intI1 gene, encoding the class 1 integrase; the promoter regions for the intI1 and aadA1 genes; and the bulk of the attI1 site, the IntI1-specific recombination site into which cassettes are inserted. IntI1 is a member of the integrase family of site-specific recombinases (14, 26, 74) and catalyzes recombination between various pairs of recombination sites (59-be [see below] × 59-be, 59-be × attI1, and attI1 × attI1) in experimental conduction assays (32, 38, 60, 61, 82, 105). IntI1 also catalyzes the integration of additional gene cassettes at attI1 (10) and both the excision and rearrangement of gene cassettes (12, 13). Experimentally, the attI1 site consists of at least 34 bp but no more than 64 bp of the 5′-CS and includes 6 to 10 bp at the first adjacent cassette (Fig. 4) (82), although a shorter length has recently been claimed (38). There are four IntI1 binding sites in attI1: two sites appear to consist of a simple integrase recombination site made up of a pair of inversely oriented IntI1-binding sites and two additional IntI1-binding sites (DR1 and DR2) that enhance the efficiency of the simple recombination site (Fig. 4) (14, 25).
FIG. 4.
In2 promoter and attI1 regions. The direction and position of transcription from the promoters Pc and P2 for the aadA1 gene and transcription from the promoter Pint for the intI1 gene are indicated by arrows with parentheses. Translation start sites of aadA1 and intI1 are indicated by the dashed arrows. The attI1 site includes a 64-bp region (sufficient for full recombination site activity for attI1). The numbering scheme relates to the number of base pairs to the left (negative) and to the right (positive) of the recombination crossover point (designated position 0), which is marked with a vertical arrow. A box surrounds the attI1-aadA1 recombination site. Integrase binding sites DR1 and DR2 (closely related to the 7-bp consensus sequence, GTTRRRY) and the “simple site” recognized by IntI1 are underscored with arrows. The numbers 5100 to 5500 indicate the location in the GenBank sequence AF071413.
Integrons also include a promoter, Pc (formerly known as Pant or P1), that directs transcription of the cassette-borne gene(s) (Fig. 4). The sequences of the 5′-CS of class 1 integrons found in several different locations are >99% identical. The most frequent differences between the sequences of the 5′-CS from different integrons occur within the −10 or −35 regions of the Pc promoter (104), giving rise to variants with different strengths (9, 11, 55); the variant of Pc in In2 is the weakest. However, the 5′-CS of In2 contains a unique insertion of three G residues (bases 5278 to 5280 [Table 5]; bold G’s in Fig. 4) that increases the spacing between a distinct set of potential −10 and −35 hexamers from 14 to 17 bp, thereby creating a second promoter, P2, which is more efficient than Pc (11, 55). P2 is a secondary promoter found only in In2 and close relatives. Constitutive transcripts from both Pc and P2 can be detected (11); there is no information on whether either is subject to regulation by other factors.
aadA1 Cassette
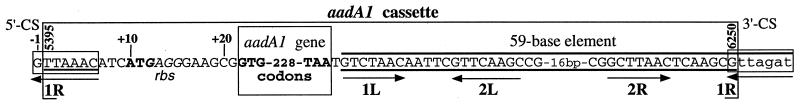
In2 includes a single gene cassette containing the aadA1 gene, which determines resistance to streptomycin and spectinomycin. The AAD(3") protein is an aminoglycoside adenylytransferase that inactivates these antibiotics by adding an adenyl or other nucleotidyl group. The cassette also contains an IntI1-specific recombination site, the aadA1 59-base element (59-be; also called attC) located downstream of the gene (Fig. 5). The aadA1 cassette thus has the features typical of gene cassettes, i.e., a gene and a 59-be. The aadA1 cassette was presumably incorporated into In2 by IntI1-mediated recombination between the attI1 site and the 59-be in a free circular aadA1 cassette. Experimental evidence for the free circular intermediate and for cassette integration has been reviewed extensively elsewhere (34, 35, 80).
FIG. 5.
The aadA1 cassette containing the aadA1 (aminoglycoside adenylyltransferase) gene and 59-be. The base pairs comprising the 59-be in its free circular form are indicated by the heavy line below them, and those comprising its integrated form are indicated by the heavy line above them. Small boxes surround the composite attI/aadA1 (left) and aadA1/qacE 59-be (right) recombination sites. The putative initiation codon of the aadA1 gene and its stop codon are in bold (large box). The putative ribosomal binding site (rbs) is marked in italics. Arrows indicate imperfect inverted repeat sequences. The numbering scheme is as described in the legend to Fig. 4.
The aadA1 cassette is organized compactly (Fig. 5). The first possible in-frame ATG codon for aadA1 is only 8 bp from the cassette boundary but is not preceded by a consensus ribosome binding site within the cassette. Alternatively, translation of aadA1 may initiate at a GTG codon, 12 bp downstream, which is preceded by a ribosome binding site. Translation of the aadA2 gene in a related cassette is initiated in this manner (3). The 59-be of the aadA1 cassette lies only 1 bp from the termination codon of the gene.
In the aadA1 cassette, the 59-be is actually 60 bp long and has features common to this family of recombination sites (13, 32, 105). Since the 59-be, of the free circular cassette form, is separated into two parts by the recombination crossover, the 59-be immediately downstream from aadA1 in In2 actually includes 54 bp of the aadA1 59-be (from the circular cassette form) and at least 6 bp (lowercase in Fig. 5) of the adjacent module, which is the 3′-CS of In2. This composite 59-be is called the aadA1/qacE 59-be. The terminal 6 bp of the aadA1 59-be in the circular cassette form lie at the beginning of the integrated aadA1 cassette and constitute part of the composite attI1/aadA1 recombination site of In2. The outer 25 bp of all 59-be are relatively highly conserved (13, 105). Within each 25-bp consensus region is a simple site configuration (1L-2L and 2R-1R [Fig. 5]), as found with other integrase recognition sites, and each simple site includes a pair of inversely oriented 7-bp core sites. Only 8 base of the composite core sites are completely conserved: three each in 1L and 1R (ACC and GTT) and one each in 2L and 2R (A and T) in all 59-be (105).
3′-Conserved Segment
In early studies of class 1 integrons, the 3′-CS was defined as a conserved region containing three ORFs (qacEΔ1, sul1, and orf5) located to the right of the different cassette arrays found in various integrons (Fig. 2) (104). More recent studies (5, 33) have revealed that the length of the 3′-CS region is variable and that some integrons have additional sequence (orf6 or portions of orf6) downstream of orf5. However, not all class 1 integrons have this region (Fig. 6); e.g., Tn402, also recently called Tn5090 (79), does not have a 3′-CS region.
FIG. 6.
Evolution of Tn21. Dark gray represents the 5′-CS, and light gray represents the 3′-CS. Cassette insertion is indicated by plus (+), and cassette excision is indicated by minus (−).
The qacE gene encodes an inner membrane protein with four transmembrane segments and determines resistance to quaternary ammonium compounds (antiseptics and disinfectants) by an efflux mechanism (71, 88). The last 66 bp of qacEΔ1 (encoding 115 aa) differs from the complete qacE gene (encoding 110 aa); the qacEΔ1 gene confers only marginal resistance to quaternary ammonium compounds (75). The sul1 gene (formerly sulI) encodes a sulfonamide-resistant dihydropteroate synthase (279 aa), which can replace the normal bacterial enzyme targeted by this group of drugs (111). The function of orf5 is not known; however, the predicted protein is 38% identical and 47% similar to a puromycin N-acetyltransferase (gi 1346911) of Streptomyces alboniger and is 36% identical and 50% similar to a putative acetyltransferase (gi 3449263) of Streptomyces coelicolor (121). Although in In2, transcription originating from Pc or P2 in the 5′-CS is likely to extend into the 3′-CS, affording expression at these three 3′-CS genes, a weak promoter that is sufficient for expression of sulfonamide resistance has also been identified upstream of the qacEΔ1 gene (30).
In Tn402, the qacE gene is associated with a 59-be and hence is part of a mobile cassette (81), whereas qacEΔ1 is not mobile. The sul1 and adjacent sequence may also have originally been part of cassettes which integrated at attI1 and subsequently became immobilized by loss of their adjacent 59-be recombination sites (Fig. 6) (5, 104). Thus, the 3′-CS appears to contain vestiges of the earliest insertion events. The presence of the sul1 gene would explain the wide distribution of integrons containing this region, since sulfonamides were the first widely used antimicrobial agents.
tni Transposition Module
Of the class 1 integrons, where sufficient sequence is available, only Tn402 (Fig. 6) carries the full complement of four tni genes (5, 79), believed to encode transposition functions on the basis of their similarity to the tni genes of Tn5053 (Fig. 3) (48). The tniA, tniB, and tniQ genes of Tn5053 are required for transposition, and the fourth gene, tniR, encodes a resolvase which resolves the cointegrates as does TnpR of Tn21 (48). In In2, only one complete gene, tniA, and a truncated version of the tniB gene are present. All class 1 integrons, including Tn402, have complex ends (79). Both ends have three copies of a 19-bp sequence, the outermost of which overlaps the terminal 25-bp IRs. By analogy to Tn7, these three 19-bp repeats are believed to function as transposase binding sites, although the identity of the Tni protein involved is not known.
IS1326 Insertion Sequence
The insertion sequence IS1326 has terminal 26-bp IRs and includes two ORFs that overlap by 14 bp (5). These ORFs have been designated istA (1,524 bp) and istB (786 bp) due to the similarity of their predicted products to IstA and IstB, required for transposition in IS21 (83). IstA has a DD(45)E transposase motif and a helix-turn-helix motif, and IstB has a distinctive set of nucleoside triphosphate binding domains (5). These two genes have share a common promoter upstream of istA. However, in In2, IS1353 has inserted between the −10 and −35 promoter regions of IS1326, and thus istAB may not be expressed. IS1326 also occurs in other integrons, e.g., integrons In0 and In5 in plasmids pVS1 and pSCH884, respectively (5).
IS1353 Insertion Sequence
The acquisition of IS1353 by In2 appears to be a recent event that occurred after the movement of the In2 to its present location in Tn21 (5). IS1353 is a member of the IS3 family (5) and has the characteristic 5′-TG and CA-3′ at the outer ends of its terminal 12-bp IRs. IS1353 is flanked by a direct duplication of 2 bp of the target sequence. It contains two ORFs, orfA and orfB, transcribed in the opposite direction from istA and istB of IS1326. A potential promoter for orfA and orfB lies 24 bp upstream of the putative OrfA translational start. OrfB has a DD(35)E transposase motif similar to other transposases of the IS3 family (23). A potential frameshift motif, A6G, and a predicted mRNA stem-loop are present where the orfA and orfB reading frames overlap, suggesting that (like other IS3 relatives, IS911 and IS150), three polypeptides, OrfA, OrfB, and a fusion protein (OrfAB) that is the actual transposase, may be produced (78). The initiation codon of OrfB has yet to be determined; it may be at the ATT codon 3 bp upstream of the A6G motif (as seen in IS911) or at the ATG codon 6 bp downstream of the A6G motif.
TN21 EVOLUTION
Transposons are important for bacterial evolution and adaptation because they carry traits that can be useful for the host cell in varying environments; Tn21 provides its host cell with both antibiotic and mercury resistance determinants. The Tn21 subgroup of transposons is phylogenetically identified on the basis of tnp genes, res sites, and IR ends, but its members vary considerably in the number and types of additional genes (29, 58) they carry. Moreover, some transposons in this subgroup do not carry either a mer region or any resistance genes. The ubiquity and variety of elements in the Tn21 subgroup is the result of several recombination processes (29).
Two characteristics of the transposition machinery of the Tn21 subgroup of transposons foster their ready association with many different genetic loci. The first of these characteristics is that both the resolvase and the transposase have rather broad substrate specificities for the DNA sites which are their substrates. For example, the Tn21 TnpA transposase recognizes several different IR sequences and can also mediate one-ended transposition to generate novel joint molecules (29). In addition, the various TnpR resolvases act on each other’s res sites, yielding distinct rearrangements of these transposons (29). The second characteristic of the Tn21 subgroup which enhances their variety is the placement of the res site to one side of tnpA and tnpR (Fig. 2) rather than between them (as found in the Tn3 transposon family). This arrangement minimizes the segregation of tnpR and tnpA which might otherwise occur in aberrant resolution events and results in a relatively stable resolvase-transposase “module.” In addition to these properties of the Tn21 subgroup, two or more Tn21-like elements are often found in the same bacterial strain, typically on large conjugative plasmids, where host-mediated homologous recombination can also contribute to their reassortment (59).
Moreover, when an integron is present, two IntI1-mediated, RecA-independent, site-specific recombination processes occur: (i) insertion, rearrangement, or excision of antibiotic resistance gene cassettes (10, 12, 13) and/or (ii) formation and resolution of cointegrates. Both processes involve recombination between various pairs of specific recombination sites (59-be × 59-be, 59-be × attI1, and attI1 × attI1) (32, 38, 60, 61, 82, 105), thus enabling the rapid development of new arrays of antibiotic resistance genes (Fig. 6). Further variation results from the acquisition of insertion sequences, transposons, and integrons and subsequent deletions arising at their ends and mediated by them (5).
Model for Tn21 Evolution
Several transposons, considered to be very close relatives of Tn21, have nearly identical DNA sequences or restriction maps in the tnp and mer regions but differ in that they carry no or different antibiotic resistance genes. Tanaka et al. (112) first proposed that those Tn21-like transposons which confer multiple antibiotic resistance had descended from an ancestral mercury resistance transposon like Tn501 by successive insertions of antibiotic resistances and/or insertion sequences. Their restriction endonuclease analysis indicated similarity between the mer and the tnp regions of Tn2613, which lacks antibiotic resistance genes, and the corresponding regions of Tn2608, which has a 6.3-kb insertion between the mer and tnp regions and has also gained streptomycin and sulfonamide resistances. These observations suggested that Tn2613 might reasonably be considered the predecessor of Tn2608. However, Tn21 itself has a slightly different restriction map in the Tn21 IRmer and in the tnpM regions from those of both Tn2613 and Tn2608, suggesting that neither of these transposons was the immediate predecessor of Tn21.
A more recent model for the evolution of Tn21 and other family members (5) is based on restriction maps, heteroduplex analyses, and sequence data for a larger group of these transposons (19, 53, 63, 94, 110, 113, 117, 119, 122, 123). In this analysis, those considered members of the family are similar to Tn21 in carrying the same tnpRA transposition genes and an integron in the same position as In2. They may or may not carry the complete mer locus, and they also differ from Tn21 in either the identity or number of gene cassettes in the integron or by the acquisition or loss of other IS elements or transposons. For example, the original Tn21, isolated in Japan, includes a 1.6-kb region corresponding to IS1353 that is not present in related isolates from Germany (29), which suggests that Tn21 acquired IS1353 after the global spread of an ancestor such as Tn2411 that does not contain this IS (5).
Brown et al. (5) proposed that Tn2411 arose from a hypothetical ancestor called Tn21Δ, which lacks an integron but contains the mer operon (Fig. 6). In Tn21Δ, urf2M would be present (as seen in orf2 and urf2 adjacent to mer operons in Tn501 [6] and pPB [84], respectively [Fig. 3]). The ancestor of In2 (without IS1353) inserted into Tn21Δ, perhaps via a transposition event catalyzed in trans by another transposon with a complete set of tni transposition genes. The alternative, i.e., that the In2 ancestor might have been complete and later lost its tni genes by a deletion event occurring on or after insertion of IS1353, seems unlikely since integrons with very similar organization to In2 are found in other locations (5).
Subsequently, transposon Tn2411 became widely distributed, experiencing integration, excision, or exchange of gene cassettes, insertion of transposons or insertion sequences, and IS-mediated deletions or excisions (Fig. 6), which have given rise to the examples of this group described so far (19, 53, 63, 94, 110, 119, 122, 123) and probably many others.
Model for In2 Evolution
The In2 integron is believed to have been a highly evolved element (Fig. 6) before it integrated into Tn21Δ. Brown et al. (5) have proposed that the central components of In2 evolved from a progenitor, similar to Tn402, which had experienced several genetic events prior to insertion into Tn21Δ. These genetic events would include the creation of the 3′-CS via the integration of qacE, as well as sul1, and other putative cassettes and subsequent events to immobilize them. After the insertion of IS1326, this IS caused the partial deletion of the adjacent 3′-CS and tni module. The insertion of IS1353 occurred after the integron moved into Tn21Δ and is likely to be the latest event in the creation of both In2 and Tn21.
Codon Usage Comparisons
Tn21 can be viewed as an assemblage of genes captured by transposition genes: the tnp genes acquired the mer operon, and the tni genes acquired the integrase system. After these two independent events, the integron progenitor, itself an assemblage of genes captured by either the tni module or the integrase module, inserted into Tn21Δ, followed by insertion of IS1353 (Fig. 6). Some hints about the variety of sources of these many components is given by their codon usage patterns.
Codon usage comparisons of Tn21 genes point to significant diversity in their origins (Table 8). Codon usage comparisons of merA and urf2M reveal low D2 values with respect to each other but not with respect to other integron or tnp genes, with the exception of orfAB of IS1353. Codon usage in aadA1, as in the majority of genes found in integrated cassettes (2), is quite dissimilar to that in other genes within the integron, presumably reflecting diverse origins of the cassette-associated genes. In Tn21, the exception to this appears to be the low D2 values between aadA1 and istAB (IS1326) and orfAB (IS1353). High D2 values are also observed between qacEΔ1 (a former cassette) and all other Tn21 genes compared with it. Codon usage comparisons of genes within the integron revealed low D2 values between intI1, sul1, and tniA, suggesting that these genes originated from closely related microorganisms which are different from E. coli or Pseudomonas aeruginosa (2, 79). This observation supports the view that sul1 was integrated early in the evolution of class 1 integrons (104). Comparison of codon usage in Tn402 genes revealed similar low D2 values (0.81) between int and tniA as for those genes in Tn21 and high D2 (9.27 to 6.98) values between qacE and other genes within this integron (79). The low D2 values between most of the Tn21 genes and the orfAB genes in IS1353 suggest that perhaps IS1353 was acquired while Tn21 was in a host with codon utilization similar to that of the ancestral host of Tn21.
TN21 SAILS THE WORLD
The Tn21 family is widely distributed in both clinical and environmental isolates of gram-negative bacteria. All characterized examples of the Tn21 family have been found on large conjugative plasmids. Although bacterial conjugation was once believed to be restricted in host range, it now appears that this mechanism of transfer permits genetic exchange between many different bacterial genera in nature (16, 50).
Environmental Occurrences
Pearson et al. observed the widespread distribution of Tn21- and Tn501-related tnpA and tnpR sequences in soil bacteria from three sites (two mercury-polluted sites and one pristine site); 20 of 30 isolates hybridized to transposase and resolvase gene probes (76). They observed extensive recombination between different transposition genes and reassortment of the mer and the transposition gene regions. Further evidence for widespread distribution includes a recent report of the occurrence of Tn21-, Tn501-, and Tn3-related tnpA sequences in total bacterial DNA from the marine environment (15). The Tn21 tnpA sequence occurred once per 103 or 104 bacteria.
Clinical Occurrences
Apart from the above environmental isolates, Tn21 carriage of integrons and integrated cassette-associated resistance genes in gram-negative pathogens is also well documented (32, 53, 80). The Tn21 family encodes resistances against both older and newer antibiotics. Strains of bacteria resistant to streptomycin, an antibiotic rarely used medically in the past 30 years but still extensively used in animal husbandry, are frequently found (68). In a 1995 study, over half (53%) of 49 clinical isolates with transferable resistance to aminoglycosides carried integrons with aadA1 (25%) or aacA4 (75%) gene cassettes (92). In an earlier (1992) study, 807 unselected gram-negative clinical isolates were screened to determine the distribution of the integron and Tn21 transposition genes (128). A total of 19% of the isolates carried some part of the tnpA tnpR, or intI1 gene and 8% of the isolates had all of the genes, although linkage of tnp genes and integrons was not demonstrated. Besides complete Tn21-like transposons, a great variety of defective transposons were observed in many genera of bacteria (128).
Tn21 and Tn21-like transposons with integrons have also been observed in primate intestinal bacteria that were challenged with Hg(II) released from dental amalgams (108). There was a strong association of multiple-antibiotic resistance with two of the most abundant phylogenetic types of Hg resistance locus (locus 1 and locus 4) in these bacteria (58, 118). Using PCR, we have established (Fig. 7) that mer locus 1 carries a In2-like integron and that some examples of mer locus 4 carry a class 1 integron similar to In4 in Tn1696 (119). In both mer locus types, the adjacent integrons carry from one to four antibiotic resistance genes. Hg exposure experienced by the fecal flora of these animals led to enrichment for strains carrying Hg resistance loci closely linked to integron multiple-antibiotic resistant transposons.
FIG. 7.
PCR primers used to detect integron components and physical linkage between mer operons and class 1 integrons in gram-negative bacteria. Primers I1 and I2 are located in the intI1 gene, and intergenic primers, 5′-CS and 3′-CS, lie between the intI1 gene and the antibiotic resistance (AbiR) gene cassette and between the AbiR gene cassette and the qacEΔ1 gene, respectively (56). S1 and N1 primers are located in the sul1 and tniA genes of the integron, and primer A9 is located in the merA gene of the mer operon (57). Numbers under bars indicate the expected size (in kilobases) of the PCR products for Tn21.
The causes of the dissemination of the Tn21 family of transposons are presently unknown but may be due to the chance association of the integron and mer at a time when the rich ecosystem of the human intestinal microflora was experiencing increasing antibiotic usage coinciding with exposure to Hg due to its use in the treatment of syphilis and in amalgam dental restorations.
CONCLUDING REMARKS
Tn21 and its relatives are major agents in the dissemination of mercury resistance and antibiotic resistance genes in gram-negative bacteria. Understanding the molecular processes involved in the accumulation and exchange of resistance genes by these integron-carrying transposons will inform efforts to control the spread of these genes as well as others which will arise in response to newer antibiotics. The regulation of antibiotic resistance gene cassette movement and stability, the factors that influence the recombination processes in the transposon, and the question whether there are environmental selection pressures other than antibiotics that contribute to their spread are subjects of intense study in many laboratories worldwide. Like mariners of old, Tn21 has left its kin in many ports and picked up barnacles along the way. As others of its family come to light, we expect that there will be answers to many of the questions posed above. We encourage others working on similar complex transposable elements and plasmids to compile and deposit their sequences. Such data will be essential to understanding the still-evolving relationships between the ubiquitous, peripatetic “floating” genome and the more constant cellular chromosomes.
ACKNOWLEDGMENTS
We gratefully acknowledge the cheerful and competent assistance of Alice L. Watson for sequence entry into GenBank; GCG advice from Michael Weise; and comments on the manuscript by Nigel Brown, Paul H. Roy, Jim Shapiro, and an anonymous reviewer.
This project was partially supported by grants from NIH (GM28211), the Wallace Research Foundation, and the International Academy of Oral Medicine and Toxicology to A.O.S. and grants from the NHIMRC to R.M.H.
REFERENCES
- 1.Barrineau P, Gilbert M P, Jackson W J, Jones C S, Summers A O, Wisdom S. The DNA sequence of the mercury resistance operon of the IncFII plasmid NR1. J Mol Appl Genet. 1984;2:601–619. [PubMed] [Google Scholar]
- 2.Bissonnette L, Roy P H. Characterization of In0 of Pseudomonas aeruginosa plasmid pVS1, an ancestor of integrons of multiresistant plasmids and transposons of gram-negative bacteria. J Bacteriol. 1992;174:1248–1257. doi: 10.1128/jb.174.4.1248-1257.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bito A, Susani M. Revised analysis of aadA2 gene of plasmid pSA. Antimicrob Agents Chemother. 1994;38:1172–1175. doi: 10.1128/aac.38.5.1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Blattner F R, Plunkett G, Bloch C A, Perna N T, Burland V, Riley M, Collado Vides J, Glasner J D, Rode C K, Mayhew G F, Gregor J, Davis N W, Kirkpatrick H A, Goeden M A, Rose D J, Mau B, Shao Y. The complete genome sequence of Escherichia coli K-12. Science. 1997;277:1453–1474. doi: 10.1126/science.277.5331.1453. [DOI] [PubMed] [Google Scholar]
- 5.Brown H J, Stokes H W, Hall R M. The integrons In0 In2, and In5 are defective transposon derivatives. J Bacteriol. 1996;178:4429–4437. doi: 10.1128/jb.178.15.4429-4437.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brown N L, Ford S J, Pridmore R D, Fritzinger D C. Nucleotide sequence of a gene from the Pseudomonas transposon Tn501 encoding mercuric reductase. Biochemistry. 1983;22:4089–4095. doi: 10.1021/bi00286a015. [DOI] [PubMed] [Google Scholar]
- 7.Brown N L, Misra T K, Winnie J N, Schmidt A, Seiff M, Silver S. The nucleotide sequence of the mercuric resistance operons of plasmid R100 and transposon Tn501: further evidence for mer genes which enhance the activity of the mercuric ion detoxification system. Mol Gen Genet. 1986;202:143–151. doi: 10.1007/BF00330531. [DOI] [PubMed] [Google Scholar]
- 8.Bull P C, Cox D W. Wilson disease and Menkes disease: new handles on heavy-metal transport. Trends Genet. 1994;10:246–252. doi: 10.1016/0168-9525(94)90172-4. [DOI] [PubMed] [Google Scholar]
- 9.Bunny K L, Hall R M, Stokes H W. New mobile cassette containing an aminoglycoside resistance gene, aacA7, and a chloramphenicol resistance gene, catB3, in a integron in pBWH301. Antimicrob Agents Chemother. 1995;39:686–693. doi: 10.1128/AAC.39.3.686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Collis C M, Grammaticopoulos G, Briton J, Stokes H W, Hall R M. Site-specific insertion of gene cassettes into integrons. Mol Microbiol. 1993;9:41–52. doi: 10.1111/j.1365-2958.1993.tb01667.x. [DOI] [PubMed] [Google Scholar]
- 11.Collis C M, Hall R M. Expression of antibiotic resistance genes in the integrated cassettes of integrons. Antimicrob Agents Chemother. 1995;39:155–162. doi: 10.1128/aac.39.1.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Collis C M, Hall R M. Gene cassettes from the insert region of integrons are excised as covalently closed circles. Mol Microbiol. 1992;6:2875–2885. doi: 10.1111/j.1365-2958.1992.tb01467.x. [DOI] [PubMed] [Google Scholar]
- 13.Collis C M, Hall R M. Site-specific deletion and rearrangement of integron insert genes catalyzed by the integron DNA integrase. J Bacteriol. 1992;174:1574–1585. doi: 10.1128/jb.174.5.1574-1585.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Collis C M, Kim M-J, Stokes H W, Hall R M. Binding of the purified integron DNA integrase IntI1 to integron- and cassette-associated recombination sites. Mol Microbiol. 1998;29:477–490. doi: 10.1046/j.1365-2958.1998.00936.x. [DOI] [PubMed] [Google Scholar]
- 15.Dahlberg C, Hermansson M. Abundance of Tn3, Tn21, and Tn501 transposase (tnpA) sequences in bacterial community DNA from marine environments. Appl Environ Microbiol. 1995;61:3051–3056. doi: 10.1128/aem.61.8.3051-3056.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Davies J. Inactivation of antibiotics and the dissemination of resistance genes. Science. 1994;264:375–382. doi: 10.1126/science.8153624. [DOI] [PubMed] [Google Scholar]
- 17.Davies J E. Origins, acquisition and dissemination of antibiotic resistance determinants. In: Chadwick D J, Goode J, editors. Antibiotic resistance: origins, evolution, selection and spread. New York, N.Y: John Wiley & Sons, Inc.; 1997. pp. 15–27. [PubMed] [Google Scholar]
- 18.Davies J E, Rownd R. Transmissible multiple drug resistance in Enterobacteriaceae. Science. 1972;176:758–768. doi: 10.1126/science.176.4036.758. [DOI] [PubMed] [Google Scholar]
- 19.de la Cruz F, Grinsted J. Genetic and molecular characterization of Tn21, a multiple resistance transposon from R100.1. J Bacteriol. 1982;151:222–228. doi: 10.1128/jb.151.1.222-228.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Devereux J, Haeberli P, Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984;12:387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Distefano M D, Moore M J, Walsh C T. Active site of mercuric reductase resides at the subunit interface and requires Cys-135 and Cys-140 from one subunit and Cys-558 and Cys-559 from the adjacent subunit: evidence from in vivo and in vitro heterodimer formation. Biochemistry. 1990;29:2703–2713. doi: 10.1021/bi00463a013. [DOI] [PubMed] [Google Scholar]
- 22.Diver W P, Ginstead J, Fritzinger D C, Brown D C, Altenbuchner J, Rogowsky P, Schmitt R. DNA sequence of and complementation by tnpR genes of Tn21, Tn501, and Tn1721. Mol Gen Genet. 1983;191:189–193. doi: 10.1007/BF00334812. [DOI] [PubMed] [Google Scholar]
- 23.Fayet O, Ramond P, Polard P, Prère M, Chandler M. Functional similarities between retroviruses and the IS3 family of bacterial insertion sequences. Mol Microbiol. 1990;4:1771–1777. doi: 10.1111/j.1365-2958.1990.tb00555.x. [DOI] [PubMed] [Google Scholar]
- 24.Grantham R, Gautier C, Guoy M, Jacobzone M, Mercier R. Codon catalog usage is a genome strategy modulated for gene expressivity. Nucleic Acids Res. 1981;9:r43–r74. doi: 10.1093/nar/9.1.213-b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gravel A, Fournier B, Roy P H. DNA complexes obtained with the integron integrase IntI1 at the attI site. Nucleic Acids Res. 1998;26:4347–4355. doi: 10.1093/nar/26.19.4347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gravel A, Messier N, Roy P H. Point mutations in the integron integrase IntI1 that affect recombination and/or substrate recognition. J Bacteriol. 1998;180:5437–5442. doi: 10.1128/jb.180.20.5437-5442.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Griffin H G, Foster T J, Silver S, Misra T K. Cloning and DNA sequence of the mercuric and organomercurial-resistance determinates of plasmid pDU1358. Proc Natl Acad Sci USA. 1987;84:3112–3116. doi: 10.1073/pnas.84.10.3112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Grinsted J, Brown N. A Tn21 terminal sequence within Tn501: complementation of tnpA gene function and transposon evolution. Mol Gen Genet. 1984;197:497–502. doi: 10.1007/BF00329949. [DOI] [PubMed] [Google Scholar]
- 29.Grinsted J, de la Cruz F, Schmitt R. The Tn21 subgroup of bacterial transposable elements. Plasmid. 1990;24:163–189. doi: 10.1016/0147-619x(90)90001-s. [DOI] [PubMed] [Google Scholar]
- 30.Guerineau F, Brooks L, Mullineaux P. Expression of the sulfonamide resistance gene from plasmid R-46. Plasmid. 1990;23:35–41. doi: 10.1016/0147-619x(90)90042-b. [DOI] [PubMed] [Google Scholar]
- 31.Halford S, Jordan S L, Kirkbrid E A. The resolvase protein from the transposon Tn21. Mol Gen Genet. 1985;200:169–175. doi: 10.1007/BF00383331. [DOI] [PubMed] [Google Scholar]
- 32.Hall R M, Brookes D E, Stokes H W. Site-specific insertion of genes into integrons: role of the 59-base element and determination of the recombination cross-over point. Mol Microbiol. 1991;5:1941–1959. doi: 10.1111/j.1365-2958.1991.tb00817.x. [DOI] [PubMed] [Google Scholar]
- 33.Hall R M, Brown H J, Brookes D E, Stokes H W. Integrons found in different locations have identical 5′ ends but variable 3′ ends. J Bacteriol. 1994;174:6286–6294. doi: 10.1128/jb.176.20.6286-6294.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hall R M, Collis C M. Antibiotic resistance in gram-negative bacteria: the role of gene cassettes and integrons. Drug Resist Updates. 1998;1:109–119. doi: 10.1016/s1368-7646(98)80026-5. [DOI] [PubMed] [Google Scholar]
- 35.Hall R M, Collis C M. Mobile gene cassettes and integrons: capture and spread of genes by site-specific recombination. Mol Microbiol. 1995;15:593–600. doi: 10.1111/j.1365-2958.1995.tb02368.x. [DOI] [PubMed] [Google Scholar]
- 36.Hamlett N V, Landale E C, Davis B H, Summers A O. Roles of the Tn21 merT, merP, and merC gene products in mercury resistance and mercury binding. J Bacteriol. 1992;174:6377–6385. doi: 10.1128/jb.174.20.6377-6385.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hanni C, Meyer J, Iida S, Arber W. Occurrence and properties of composite transposon Tn2672: evolution of multiple drug resistance transposons. J Bacteriol. 1982;150:1266–1273. doi: 10.1128/jb.150.3.1266-1273.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hansson K, Skold O, Sundstrom L. Non-palindromic attI sites of integrons are capable of site-specific recombination with one another and with secondary sites. Mol Microbiol. 1997;26:441–453. doi: 10.1046/j.1365-2958.1997.5401964.x. [DOI] [PubMed] [Google Scholar]
- 39.Heltzel A, Lee I W, Totis P A, Summers A O. Activator-dependent preinduction binding of ς-70 RNA polymerase at the metal-regulated mer promoter. Biochemistry. 1990;29:9572–9584. doi: 10.1021/bi00493a011. [DOI] [PubMed] [Google Scholar]
- 40.Hobman J L, Brown N L. Bacterial mercury-resistance genes. In: Sigel A, Sigel H, editors. Metal ions in biological systems. New York, N.Y: Marcel Dekker, Inc.; 1997. pp. 527–568. [PubMed] [Google Scholar]
- 41.Hoch A H, Sander C. Two-component signal transduction. Washington, D.C: ASM Press; 1995. [Google Scholar]
- 42.Hyde D R, Tu C P D. tnpM: a novel regulatory gene that enhances Tn21 transposition and suppresses cointegrate resolution. Cell. 1985;42:629–638. doi: 10.1016/0092-8674(85)90120-5. [DOI] [PubMed] [Google Scholar]
- 43.Iida S, Hanni C, Echardi C, Arber W. Is the Is1 flanked R-determinate of the R plasmid NR1 a transposon? J Gen Microbiol. 1981;126:413–425. doi: 10.1099/00221287-126-2-413. [DOI] [PubMed] [Google Scholar]
- 44.Inoue C, Sugawara K, Kusano T. The merR regulatory gene in Thiobacillus ferrooxidans is spaced apart from the mer structural genes. Mol Microbiol. 1991;5:2707–2718. doi: 10.1111/j.1365-2958.1991.tb01979.x. [DOI] [PubMed] [Google Scholar]
- 45.Inoue C, Sugawara K, Kusano T, Kitagawa Y. Nucleotide sequence of the Thiobacillus ferrooxidans chromosomal gene encoding mercuric reductase. Gene. 1989;84:47–54. doi: 10.1016/0378-1119(89)90138-8. [DOI] [PubMed] [Google Scholar]
- 46.Jorgensen R A, Reznikoff W S. Organization of structural and regulatory genes that mediate tetracycline resistance in transposon Tn10. J Bacteriol. 1979;138:705–714. doi: 10.1128/jb.138.3.705-714.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kholodii G Y, Gorlenko O L, Lomovskaya S Z, Nikiforov V G. Molecular characterization of an aberrant mercury resistance transposable element from an environmental Acinetobacter strain. Plasmid. 1993;30:303–308. doi: 10.1006/plas.1993.1064. [DOI] [PubMed] [Google Scholar]
- 48.Kholodii G Y, Mindlin S Z, Bass I A, Yurieva O V, Minakhina S V, Nikiforov V G. Four genes, two ends, and a res region are involved in transposition of Tn5053: a paradigm for a novel family of transposons carrying either a mer operon or an integron. Mol Microbiol. 1995;17:1189–1200. doi: 10.1111/j.1365-2958.1995.mmi_17061189.x. [DOI] [PubMed] [Google Scholar]
- 49.Kholodii G Y, Yurieva O V, Lomovskaya O L, Gorlenko Z M, Mindlin S Z, Nikiforov V G. Tn5053, a mercury resistance transposon with integron ends. J Mol Biol. 1993;230:1103–1107. doi: 10.1006/jmbi.1993.1228. [DOI] [PubMed] [Google Scholar]
- 50.Kruse H, Sorum H. Transfer of multiple drug resistance plasmids between bacteria of diverse origins in natural microenvironments. Appl Environ Microbiol. 1994;60:4015–4021. doi: 10.1128/aem.60.11.4015-4021.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kulkarni R, Summers A O. MerR crosslinks to the α, β and ς70 subunits of RNA polymerase in the pre-initiation complex at the merTPCAD promoter. Biochemistry. 1999;38:3362–3368. doi: 10.1021/bi982814m. [DOI] [PubMed] [Google Scholar]
- 52.Kusano T, Ji G, Inoue C, Silver S. Constitutive synthesis of a transport function encoded by the Thiobacillus ferrooxidans merC gene cloned in Escherichia coli. J Bacteriol. 1990;172:2688–2692. doi: 10.1128/jb.172.5.2688-2692.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lafond M, Couture F, Vezina G, Levesque R C. Evolutionary perspectives on multiresistance β-lactamase transposons. J Bacteriol. 1989;171:6423–6429. doi: 10.1128/jb.171.12.6423-6429.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lee I W, Gambill B D, Summers A O. Translation of merD in Tn21. J Bacteriol. 1989;171:2222–2225. doi: 10.1128/jb.171.4.2222-2225.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Levesque C, Brassard S, Lapointe J, Roy P H. Diversity and relative strengths of tandem promoters for the antibiotic resistance genes of several promoters. Gene. 1994;142:49–54. doi: 10.1016/0378-1119(94)90353-0. [DOI] [PubMed] [Google Scholar]
- 56.Levesque C, Piche L, Larose C, Roy P H. PCR mapping of integrons reveals several novel combinations of resistance genes. Antimicrob Agents Chemother. 1995;39:185–191. doi: 10.1128/aac.39.1.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Liebert C A. Ph.D. dissertation. Athens: University of Georgia; 1999. [Google Scholar]
- 58.Liebert C A, Wireman J, Smith T, Summers A O. Phylogeny of mercury resistance (mer) operons of gram-negative bacteria isolated from the fecal flora of primates. Appl Environ Microbiol. 1997;63:1066–1076. doi: 10.1128/aem.63.3.1066-1076.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Lloyd R G, Low K B. Homologous recombination. In: Neidhardt F C, Curtiss III R, Ingraham J L, Lin E C C, Low K B, Magasanik B, Reznikoff W S, Riley M, Schaechter M, Umbarger H E, editors. Escherichia coli and Salmonella: cellular and molecular biology. 2nd ed. Washington, D.C: ASM Press; 1996. pp. 2236–2255. [Google Scholar]
- 60.Martinez E, de la Cruz F. Genetic elements involved in Tn21 site-specific integration, a novel mechanism for the dissemination of antibiotic resistance genes. EMBO J. 1990;9:1275–1281. doi: 10.1002/j.1460-2075.1990.tb08236.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Martinez E, de la Cruz F. Transposon Tn21 encodes a RecA-independent site-specific integration system. Mol Gen Genet. 1988;211:320–325. doi: 10.1007/BF00330610. [DOI] [PubMed] [Google Scholar]
- 62.Melchers K, Weitzenegger T, Buhmann A, Steinhilber W, Sachs G, Schafer K P. Cloning and membrane topology of a P type ATPase from Helicobacter pylori. J Biol Chem. 1996;271:446–457. doi: 10.1074/jbc.271.1.446. [DOI] [PubMed] [Google Scholar]
- 63.Mercier J, Lachapelle J, Couture F, Lafond M, Vezina G, Boissinot M, Levesque R C. Structural and functional characterization of tnpI, a recombinase locus in Tn21 and related β-lactamase transposons. J Bacteriol. 1990;172:3745–3757. doi: 10.1128/jb.172.7.3745-3757.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Miller S M, Massey V, Williams C H, Ballou D P, Walsh C T. Communication between the active sites in dimeric mercuric ion reductase: an alternating sites hypothesis for catalysis. Biochemistry. 1991;30:2600–2612. doi: 10.1021/bi00224a006. [DOI] [PubMed] [Google Scholar]
- 65.Misra T K, Brown N L, Fritzinger D C, Pridmore R D, Barnes W M, Haberstroh L, Silver S. The mercuric-ion resistance operons of plasmid R100 and transposon Tn501: the beginning of the operon including the regulatory region and the first two structural genes. Proc Natl Acad Sci USA. 1984;81:5975–5979. doi: 10.1073/pnas.81.19.5975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Misra T K, Brown N L, Haberstroh L, Schmidt A, Goddette D, Silver S. Mercuric reductase structural genes from plasmid R100 and transposon Tn501: functional domains of the enzyme. Gene. 1985;34:253–262. doi: 10.1016/0378-1119(85)90134-9. [DOI] [PubMed] [Google Scholar]
- 67.Morby A P, Hobman J L, Brown N L. The role of cysteine residues in the transport of mercuric ions by the Tn501 MerT and MerP mercury-resistance proteins. Mol Microbiol. 1995;17:25–35. doi: 10.1111/j.1365-2958.1995.mmi_17010025.x. [DOI] [PubMed] [Google Scholar]
- 68.Morell V. Antibiotic resistance: road of no return. Science. 1997;278:575–576. doi: 10.1126/science.278.5338.575b. [DOI] [PubMed] [Google Scholar]
- 69.Mukhopadhyay D, Yu H, Nucifora G, Misra T K. Purification and functional characterization of MerD: a coregulator of the mercury resistance operon in Gram-negative bacteria. J Biol Chem. 1991;266:18538–18542. [PubMed] [Google Scholar]
- 70.Nakaya R, Nakamura A, Murata Y. Resistance transfer agents in Shigella. Biochem Biophys Res Commun. 1960;3:654–659. doi: 10.1016/0006-291x(60)90081-4. [DOI] [PubMed] [Google Scholar]
- 71.Neyfakh A A, Bidnenko V E, Chen L B. Efflux-mediated multidrug resistance in Bacillus subtilis: similarities and dissimilarities with the mammalian system. Proc Natl Acad Sci USA. 1991;88:4781–4785. doi: 10.1073/pnas.88.11.4781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Odermatt A, Solioz M. Two trans-acting metalloregulatory proteins controlling expression of the copper-ATPases of Enterococcus hirae. J Biol Chem. 1995;270:4349–4354. doi: 10.1074/jbc.270.9.4349. [DOI] [PubMed] [Google Scholar]
- 73.O’Halloran T V, Frantz B, Shin M K, Ralston D M, Wright J G. The MerR heavy metal receptor mediates positive activation in a topologically novel transcription complex. Cell. 1989;56:119–129. doi: 10.1016/0092-8674(89)90990-2. [DOI] [PubMed] [Google Scholar]
- 74.Ouellette M, Bissonnette L, Roy P H. Precise insertion of antibiotic resistance determinants into Tn21-like transposons: nucleotide sequence of the OXA-1 β-lactamase gene. Proc Natl Acad Sci USA. 1987;84:7378–7382. doi: 10.1073/pnas.84.21.7378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Paulsen I T, Littlejohn T G, Radstrom P, Sundstrom L, Skold O, Swedberg G, Skurray R A. The 3′ conserved segment of integrons contains a gene associated with multidrug resistance to antiseptics and disinfectants. Antimicrob Agents Chemother. 1993;37:761–768. doi: 10.1128/aac.37.4.761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pearson A J, Bruce K D, Osborn A M, Ritchie D A, Strike P. Distribution of class II transposase and resolvase genes in soil bacteria and their association with mer genes. Appl Environ Microbiol. 1996;62:2961–2965. doi: 10.1128/aem.62.8.2961-2965.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Plasterk R H. Mechanisms of DNA transposition. In: Sherratt D J, editor. Mobile genetic elements. New York, N.Y: Oxford University Press; 1995. pp. 18–32. [Google Scholar]
- 78.Polard P, Prere M F, Chandler M, Fayet O. Programmed translational frameshifting and initiation at an AUU codon in gene expression of bacterial insertion sequence IS911. J Mol Biol. 1991;222:465–477. doi: 10.1016/0022-2836(91)90490-w. [DOI] [PubMed] [Google Scholar]
- 79.Radstrom P, Skold O, Swedberg G, Flensburg J, Roy P H, Sundstrom L. Transposon Tn5090 of the plasmid R751, which carries an integron is related to Tn7, Mu, and the retroelements. J Bacteriol. 1994;176:3257–3268. doi: 10.1128/jb.176.11.3257-3268.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Recchia G D, Hall R M. Gene cassettes: a new class of mobile element. Microbiology. 1995;141:3015–3027. doi: 10.1099/13500872-141-12-3015. [DOI] [PubMed] [Google Scholar]
- 81.Recchia G D, Hall R M. Origins of the mobile gene cassettes found in integrons. Trends Microbiol. 1997;5:389–394. doi: 10.1016/S0966-842X(97)01123-2. [DOI] [PubMed] [Google Scholar]
- 82.Recchia G D, Stokes H W, Hall R M. Characterization of specific and secondary recombination sites recognised by the integron DNA integrase. Nucleic Acids Res. 1994;22:2071–2078. doi: 10.1093/nar/22.11.2071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Reimmann C, Moore R, Little S, Savioz A, Willetts N S, Haas D. Genetic structure, function and regulation of the transposable element IS21. Mol Gen Genet. 1989;215:416–424. doi: 10.1007/BF00427038. [DOI] [PubMed] [Google Scholar]
- 84.Reniero D, Mozzon E, Galli E, Barbieri P. Two aberrant mercury resistance transposons in the Pseudomonas stutzeri plasmid pPB. Gene. 1998;208:37–42. doi: 10.1016/s0378-1119(97)00641-0. [DOI] [PubMed] [Google Scholar]
- 85.Rogowsky P, Halford S E, Schmitt R. Definition of three resolvase binding sites at the res loci of Tn21 and Tn1721. EMBO J. 1985;4:2135–2141. doi: 10.1002/j.1460-2075.1985.tb03904.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ross W, Park S-J, Summers A O. Genetic analysis of transcriptional activation and repression in the Tn21 mer operon. J Bacteriol. 1989;171:4009–4018. doi: 10.1128/jb.171.7.4009-4018.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rost B, Casadio R, Fariselli P, Sander C. Prediction of helical transmembrane segments at 95% accuracy. Protein Sci. 1995;4:521–533. doi: 10.1002/pro.5560040318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Rouch D A, Cram D S, DiBerardino D, Littlejohn T G, Skurray R A. Efflux-mediated antiseptic resistance gene qacA from Staphylococcus aureus: common ancestry with tetracycline- and sugar-transport proteins. Mol Microbiol. 1990;4:2051–2062. doi: 10.1111/j.1365-2958.1990.tb00565.x. [DOI] [PubMed] [Google Scholar]
- 89.Sahlman L, Lambeir A-M, Lindskog S, Dunford H B. The reaction between NADPH and mercuric reductase from Pseudomonas aeruginosa. J Biol Chem. 1984;259:12403–12408. [PubMed] [Google Scholar]
- 90.Sahlman L, Skarfstad E G. Mercuric ion binding abilities of MerP variants containing only one cysteine. Biochem Biophys Res Commun. 1993;196:583–588. doi: 10.1006/bbrc.1993.2289. [DOI] [PubMed] [Google Scholar]
- 91.Sahlman L, Wong W, Powlowski J. A mercuric ion uptake role for the integral inner membrane protein, MerC, involved in bacterial mercuric ion resistance. J Biol Chem. 1997;272:29518–29526. doi: 10.1074/jbc.272.47.29518. [DOI] [PubMed] [Google Scholar]
- 92.Sallen B, Rajoharison A, Desvarenne S, Mabilat C. Molecular epidemiology of integron-associated antibiotic-resistance genes in clinical isolates of Enterobacteriaceae. Microb Drug Resist. 1995;1:195–202. doi: 10.1089/mdr.1995.1.195. [DOI] [PubMed] [Google Scholar]
- 93.Schiering N, Kabsch W, Moore M J, Distefano M D, Walsh C T, Pai E F. Structure of the detoxification catalyst mercuric ion reductase from Bacillus sp. strain RC607. Nature. 1991;352:168–172. doi: 10.1038/352168a0. [DOI] [PubMed] [Google Scholar]
- 94.Schmidt F, Klopfer-Kaul I. Evolutionary relationships between Tn21-like elements and pBP201, a plasmid from Klebsiella pneumoniae mediating resistance to gentamycin and eight other drugs. Mol Gen Genet. 1984;197:109–119. doi: 10.1007/BF00327930. [DOI] [PubMed] [Google Scholar]
- 95.Sherratt D. Tn3 and other related transposable elements: Site-specific recombination and transposition. In: Berg D E, Howe M M, editors. Mobile DNA. Washington, D.C: American Society for Microbiology; 1989. pp. 163–184. [Google Scholar]
- 96.Shewchuk L M, Helmann J D, Ross W, Park S J, Summers A O, Walsh C T. Transcriptional switching by the MerR protein: activation and repression mutants implicate distinct DNA and mercury(II) binding domains. Biochemistry. 1989;28:2340–2344. doi: 10.1021/bi00431a053. [DOI] [PubMed] [Google Scholar]
- 97.Silver S, Nucifora G, Phung L T. Human Menkes X-chromosome disease and the staphylococcal cadmium-resistance ATPases: a remarkable similarity in protein sequences. Mol Microbiol. 1993;10:7–12. doi: 10.1111/j.1365-2958.1993.tb00898.x. [DOI] [PubMed] [Google Scholar]
- 98.Silver S, Phung L T. Bacterial heavy metal resistance: new surprises. Annu Rev Microbiol. 1996;50:753–789. doi: 10.1146/annurev.micro.50.1.753. [DOI] [PubMed] [Google Scholar]
- 99.Sonnhammer E L, von Heijne G, Krogh A. Proceedings of the Sixth International Conference on Intelligent Systems for Molecular Biology. 1998. A hidden Markov model for predicting transmembrane helices in protein sequences; pp. 175–182. [PubMed] [Google Scholar]
- 100.Soultanas P, Oram M, Halford S E. Site-specific recombination at res site containing DNA binding sequences for both Tn21 and Tn3 resolvases. J Mol Biol. 1995;245:208–218. doi: 10.1006/jmbi.1994.0017. [DOI] [PubMed] [Google Scholar]
- 101.Stark W M, Boocock M R. Topological selectivity in site-specific recombination. In: Sherratt D J, editor. Mobile genetic elements. New York, N.Y: Oxford University Press; 1995. pp. 101–125. [Google Scholar]
- 102.Stark W M, Boocock M R, Sherratt D J. Site-specific recombination by Tn3 resolvase. Trends Genet. 1989;5:304–309. doi: 10.1016/0168-9525(89)90113-3. [DOI] [PubMed] [Google Scholar]
- 103.Steel R A, Opella S J. Structures of the reduced and mercury-bound forms of MerP, the periplasmic protein from the bacterial mercury detoxification system. Biochemistry. 1997;36:6885–6895. doi: 10.1021/bi9631632. [DOI] [PubMed] [Google Scholar]
- 104.Stokes H W, Hall R M. A novel family of potentially mobile DNA elements encoding a site-specific gene-integration function: integrons. Mol Microbiol. 1989;3:1669–1683. doi: 10.1111/j.1365-2958.1989.tb00153.x. [DOI] [PubMed] [Google Scholar]
- 105.Stokes H W, O’Gorman D B, Recchia G D, Parekhian M, Hall R M. Structure and function of 59-base element recombination sites associated with mobile gene cassettes. Mol Microbiol. 1997;26:731–745. doi: 10.1046/j.1365-2958.1997.6091980.x. [DOI] [PubMed] [Google Scholar]
- 106.Summers A O. Organization, expression and evolution of genes for mercury resistance. Annu Rev Microbiol. 1986;40:607–634. doi: 10.1146/annurev.mi.40.100186.003135. [DOI] [PubMed] [Google Scholar]
- 107.Summers A O. Untwist and shout: a heavy metal-responsive transcriptional regulator. J Bacteriol. 1992;174:3097–3101. doi: 10.1128/jb.174.10.3097-3101.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Summers A O, Wireman J, Vimy M J, Lorscheider F L, Marshall B, Levy S B, Bennett S, Billard L. Mercury released from dental “silver” fillings provokes an increase in mercury- and antibiotic-resistant bacteria in the oral and intestinal flora of primates. Antimicrob Agents Chemother. 1993;37:825–834. doi: 10.1128/aac.37.4.825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Sundstrom L, Radstrom P, Swedberg G, Skold O. Site-specific recombination promotes linkage between trimethoprim and sulfonamide resistance genes. Sequence characterization of the dhfrV and sulI and a recombination active locus of Tn21. Mol Gen Genet. 1988;213:191–201. doi: 10.1007/BF00339581. [DOI] [PubMed] [Google Scholar]
- 110.Sundstrom L, Swedberg G, Skold O. Characterization of transposon Tn5086, carrying the site-specifically inserted gene dhfrVII mediating trimethoprim resistance. J Bacteriol. 1993;175:1796–1805. doi: 10.1128/jb.175.6.1796-1805.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Swedberg G. Organization of two sulfonamide resistance genes on plasmids of gram-negative bacteria. Antimicrob Agents Chemother. 1987;31:306–311. doi: 10.1128/aac.31.2.306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Tanaka M, Yamamoto T, Sawai T. Evolution of complex resistance transposons from an ancestral mercury transposon. J Bacteriol. 1983;153:1432–1438. doi: 10.1128/jb.153.3.1432-1438.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Tanaka M, Yamamoto T, Sawai T. Fine structure of transposition genes of Tn2603 and complementation of its tnpA and tnpR mutations by related transposons. Mol Gen Genet. 1983;191:442–450. doi: 10.1007/BF00425761. [DOI] [PubMed] [Google Scholar]
- 114.Wang Y, Moore M J, Levinson H S, Silver S, Walsh C T. Nucleotide sequence of a chromosomal mercury resistance determinant from a Bacillus sp. with broad-spectrum mercury resistance. J Bacteriol. 1989;171:83–92. doi: 10.1128/jb.171.1.83-92.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ward E, Grinsted J. The nucleotide sequence of the tnpA gene of Tn21. Nucleic Acids Res. 1987;15:1799–1806. doi: 10.1093/nar/15.4.1799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Watanabe T. Infective heredity of multiple drug resistance in bacteria. Bacteriol Rev. 1963;27:87–115. doi: 10.1128/br.27.1.87-115.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Wiedemann B, Meyer J F, Zuhlsdorf M T. Insertions of resistance genes into Tn21-like transposons. J Antimicrob Chemother. 1986;18(Suppl. C):85–92. doi: 10.1093/jac/18.supplement_c.85. [DOI] [PubMed] [Google Scholar]
- 118.Wireman J, Liebert C A, Smith C T, Summers A O. Association of mercury resistance and antibiotic resistance in the gram-negative fecal bacteria of primates. Appl Environ Microbiol. 1997;63:4494–4503. doi: 10.1128/aem.63.11.4494-4503.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Wohlleben W, Arnold W, Bissonnette L, Pelletier A, Tanguay A, Roy P H, Gamboa G C, Barry G F, Aubert E, Davies J, Kagan S A. On the evolution of Tn21-like multiresistance transposons: sequence analysis of the gene (aacC1) for gentamicin acetyltransferase-3-I(AAC(3)-I), another member of the Tn21-based expression cassette. Mol Gen Genet. 1989;217:202–208. doi: 10.1007/BF02464882. [DOI] [PubMed] [Google Scholar]
- 120.Womble D D, Rownd R H. Genetic and physical map of plasmid NR1: comparison with other IncFII antibiotic resistance plasmids. Microbiol Rev. 1988;52:433–451. doi: 10.1128/mr.52.4.433-451.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Worley K C, Wiese B A, Smith R F. BEAUTY: an enhanced BLAST-based search tool that integrates multiple biological information resources into sequence similarity search results. Genome Res. 1995;5:173–184. doi: 10.1101/gr.5.2.173. [DOI] [PubMed] [Google Scholar]
- 122.Yamamoto T, Tanaka M, Baba R, Yamagishi S. Physical and functional mapping of Tn2603, a transposon encoding ampicillin, streptomycin, sulfonamide, and mercury resistance. Mol Gen Genet. 1981;181:464–469. doi: 10.1007/BF00428737. [DOI] [PubMed] [Google Scholar]
- 123.Yoshifumi I, Watson J M, Haas D, Leisinger T. Genetic and molecular characterization of the Pseudomonas plasmid pVS1. Plasmid. 1984;11:206–220. doi: 10.1016/0147-619x(84)90027-1. [DOI] [PubMed] [Google Scholar]
- 124.Yurieva O, Kholodii G, Minakhin L, Gorlenko Z, Kalyaeva E, Mindlin S, Nikiforov V. Intercontinental spread of promiscuous mercury-resistance transposons in environmental bacteria. Mol Microbiol. 1997;24:321–329. doi: 10.1046/j.1365-2958.1997.3261688.x. [DOI] [PubMed] [Google Scholar]
- 125.Yurieva O, Nikiforov V. Catalytic center quest: comparison of transposases belonging to the Tn3 family reveals an invariant triad of acidic amino acid residues. Biochem Mol Biol Int. 1996;38:15–20. [PubMed] [Google Scholar]
- 126.Zeng Q, Stålhandske C, Anderson M C, Scott R, Summers A O. The core metal-recognition domain of MerR. Biochemistry. 1998;37:15885–15895. doi: 10.1021/bi9817562. [DOI] [PubMed] [Google Scholar]
- 127.Zheng Z X, Chandler M, Hipskind R, Clerget M, Caro L. Dissection of the r-determinants of the plasmid R100.1: the sequence at the extremes of Tn21. Nucleic Acids Res. 1982;9:6265–6278. doi: 10.1093/nar/9.23.6265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Zuhlsdorf M T, Wiedemann B. Tn21-specific structures in gram-negative bacteria from clinical isolates. Antimicrob Agents Chemother. 1992;36:1915–1921. doi: 10.1128/aac.36.9.1915. [DOI] [PMC free article] [PubMed] [Google Scholar]