Abstract
Snakebite envenoming represents a major health problem in tropical and subtropical countries. Considering the elevated number of accidents and high morbidity and mortality rates, the World Health Organization reclassified this disease to category A of neglected diseases. In Latin America, Bothrops genus snakes are mainly responsible for snakebites in humans, whose pathophysiology is characterized by local and systemic inflammatory and degradative processes, triggering prothrombotic and hemorrhagic events, which lead to various complications, organ damage, tissue loss, amputations, and death. The activation of the multicellular blood system, hemostatic alterations, and activation of the inflammatory response are all well-documented in Bothrops envenomings. However, the interface between inflammation and coagulation is still a neglected issue in the toxinology field. Thromboinflammatory pathways can play a significant role in some of the major complications of snakebite envenoming, such as stroke, venous thromboembolism, and acute kidney injury. In addition to exacerbating inflammation and cell interactions that trigger vaso-occlusion, ischemia–reperfusion processes, and, eventually, organic damage and necrosis. In this review, we discuss the role of inflammatory pathways in modulating coagulation and inducing platelet and leukocyte activation, as well as the inflammatory production mediators and induction of innate immune responses, among other mechanisms that are altered by Bothrops venoms.
Keywords: complement, hemostasis, inflammation, neutrophil, platelet, snake venom, thromboinflammation
1. Introduction
Snakebite envenoming remains the main neglected disease in tropical and subtropical countries due to morbidity and mortality high rates worldwide [1,2]. In Latin America and the Caribbean, at least 137,000 snakebites and 3400 deaths occur annually [3], and Bothrops represents the major snake genus that causes snakebite envenoming, sometimes resulting in tissue loss/limb amputations and permanent disability in humans [4]. Currently, 36 species of Bothrops have been cataloged, and the species display great variability, mainly regarding their color and size patterns, as well as the compositions and biological activities of their venoms [5,6,7]. Bothrops asper is a major snake that is responsible for human envenoming in Central America, while in South America B. atrox, B. erythromelas, B. jararaca, B. jararacussu, and B. moojeni are mainly responsible for snakebite envenoming in the region [8].
Bothrops venoms are mostly composed of zinc-dependent metalloproteases (SVMPs), phospholipases A2 (PLA2s), and serine proteases (SVSPs), which in situations of human envenoming, trigger a wide spectrum of pathophysiological manifestations [7,9,10,11,12,13,14,15], such as pain, edema, blistering, myonecrosis, vascular injury, ischemia, necrosis, blood incoagulability, oxidative stress, bleeding, among others [16,17,18]. Although the wide variability in the venom proteome of Bothrops is recognized, this pattern of local and systemic clinical manifestations is replicated among envenomed patients by species of this genus, which suggests the presence of a shared mechanism of action, which can lead to complications such as intracranial hemorrhage, acute kidney injury, compartment syndromes, and amputations [8,19,20,21,22,23,24,25].
The coagulation disorders observed in Bothrops snakebites are induced by hemostatically active components, such as thrombin-like, anti-, and procoagulant toxins, which generate a consumption coagulopathy, and result in bleeding and thrombotic events [26]. In addition, thrombocytopenia is also observed among cases of Bothrops envenoming [27,28,29]. Although the toxins characterization from these venoms that act on platelets is wide, the involvement of platelets in the pathogenesis of Bothrops envenoming has received less attention, neglecting the role of these cells in other phenomena such as inflammation [30].
The in-depth knowledge of the differences between the coagulation cascade and multicellular systems in Bothrops envenoming makes it feasible to understand the isolated role of these systems in the pathology, as well as the complete pathophysiological scenario. While the inflammation caused by Bothrops venoms is characterized by pain, edema formation, erythema, and cellular infiltrate, the hemostasis, platelets, leukocytes, complement system, and inflammation are tightly interlinked processes, and platelets are often the cell type that binds these processes together [16,30,31]. In this context, the term “thromboinflammation” has been used to describe the activation of the cascading blood systems, as well as the activation of the multicellular blood system [32].
In the current study “thromboinflammation” in Bothrops envenoming was considered to be the activation of integrated processes, which includes classical coagulation, clot formation, and immune responses elicited during Bothrops envenoming [30,33,34,35]. Thus, the thromboinflammation process was recognized as a broad-spectrum effect on envenoming. Furthermore, we hope that this theme becomes an umbrella that considers thrombus formation, coagulation system activation, and innate and adaptive immunity as an integrated harmful process that contributes to the amplification of the pathophysiological effects triggered by the Bothrops venoms. Moreover, studies on the mechanisms of thromboinflammation in snakebites are crucial to better management of the victims.
2. Overview of the Toxic Effects of Bothrops Venoms
2.1. Local Effects
The local effects of Bothrops envenoming are considered one of the most evidenced events due to the tissue damage caused, and the peripherally limited antivenom response. Among the mechanisms involved, the inflammatory response represents a major issue that occurs during local pathophysiology [16,36,37]. The venom toxins are capable of triggering inflammation by three primary mechanisms: (i) the direct recognition of venom components (commonly known as a venom-associated molecular pattern—VAMPs) by leukocyte receptors, such as Toll-like receptors, which induce activation and mediators production; (ii) an indirect inflammatory response that is induced by damage-associated molecular patterns (DAMPs), such as cellular and extracellular matrix degradation products as a result of the venom toxin’s tissue damage; (iii) the direct activation of complement system mediators by the toxins [38,39,40].
The major clinical local manifestations are associated with inflammation cardinal signs (Figure 1). Edema is the most common inflammatory sign in Bothrops envenoming, which can progress to a compartment syndrome that results in disabilities to the patient [19,21]. Edema is a consequence of the leukocyte infiltration and production of mediators that signal the inflammatory response and co-option of endogenous signaling pathways, which act as positive feedback; this aggravates the inflammatory response, according to Bickler [41]. Blister formation and epithelial damage are manifestations observed in patients related to severe envenomations, as they increase the chance of infection and necrosis [42,43]. This event is caused by the action of SVMPs on extracellular matrix components and epithelial tissue, inducing the separation of skin layers (dermis–epidermis) [44,45,46]. In all cases, a rich source of damage-associated molecular patterns (DAMPs), immunomodulators, matrix metalloproteinase-9 (MMP-9), platelet degranulation, and blood coagulation were observed [42,44,47]. Pain is also commonly reported in Bothrops envenoming. Its presence is mainly associated with the production of neurogenic and non-neurogenic compounds at the bite site, which occurs through the direct and/or indirect action of Bothrops venom toxins, in addition to the activation of non-neural cells that contribute to peripheral sensitization [16]. These compounds interact with their respective receptors, altering the nociceptive signal transduction. Some of these components have already been identified, including substance P, nitric oxide, cytokines, histamine, 5-hydroxytryptamine, bradykinin, lipid-derived mediators (prostaglandins, leukotrienes, and PAF), as well as the participation of leukocyte cells produced during the inflammatory response [16,48].
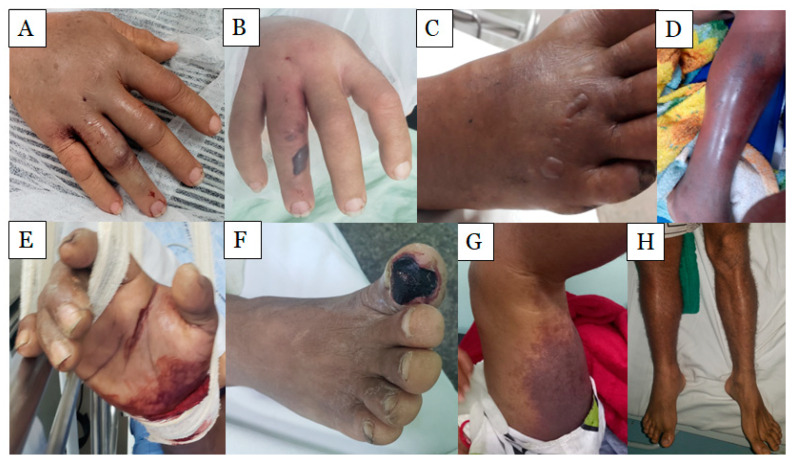
Figure 1.
Local clinical manifestations of Bothrops atrox snakebite envenoming. (A) Edema on the right hand; (B) blisters on the right hand; (C) blisters on the right foot; (D) edema on the right leg; (E) right wrist bleeding (bite site in the joint of the hand with the arm); (F) necrosis in the right hallux; (G): ecchymosis on the entire left thigh, distant from the bite site; (H): right limb swelling. Photos were taken by Lisele Brasileiro.
Another target of the action of bothropic venom toxins includes the body’s vascular system. Arteries, veins, and other vessels of the circulatory system change in permeability and integrity, mainly due to the action of SVMPs that act on the vascular basement membrane, which consist mainly of type IV collagen, laminin, nidogen/entactin, and perlecan; this results in the formation of hemorrhagic lesions (ecchymosis and petechiae) [45,46,49,50]. Another mechanism that seems to contribute to local hemorrhage in bothropic envenoming is blood incoagulability. The venom toxins catalyze the activation of clotting factor zymogens and integrin precursors or receptors, also leading to endogenous thrombin formation, which can consequently result in disseminated intravascular coagulation followed by consumption coagulopathy [51]. However, the relationship between this phenomenon and local hemorrhage is not well understood and deserves attention. In B. atrox envenoming, patients with only local and not systemic bleeding showed similar levels of hemostatic factors [29].
Additionally, skeletal muscle tissue is another target of bothropic venom toxins action that iinduces muscle degeneration and myonecrosis [36,52], which can lead to temporary and chronic sequelae, and in more severe cases, amputation. However, severe rhabdomyolysis is not common in Bothrops envenoming as it is in Crotalus envenoming. Muscle damage is caused by the action of myotoxins, which act by altering the influx of [Ca]2+ ions, consequently inducing cell death [53,54]. SVMPS may also damage the local blood flow, resulting in ischemia and secondary myonecrosis [49].
2.2. Systemic Effects
Bothrops envenoming could present systemic effects, such as coagulopathy or incoagulability, platelet consumption, a hemorrhage, which lead to death if it is not promptly managed (Figure 2) [28,55,56,57,58]. Blood incoagulability is the most predominant systemic effect in Bothrops envenoming [28]. Bothrops venoms induce a high tendency to bleeding at the bite site, in the gums, and in vital organs) [28,55,56,57,58]. In addition, Bothrops envenoming presents thrombocytopenia, probably caused by the action of toxins in inhibiting or activating platelets, as well as forming an active surface for the coagulation cascade, which leads to increased bleeding [59].
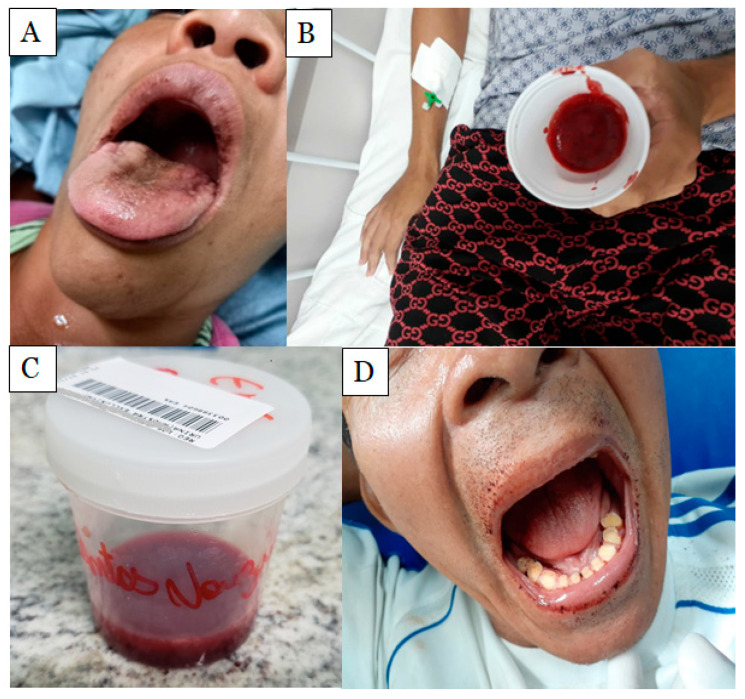
Figure 2.
Clinical systemic manifestations of Bothrops atrox snakebite envenoming. (A) Patient after hematemesis; (B) hemoptysis in a male patient; (C) macroscopic hematuria; (D) gum bleeding. Photos were taken by Lisele Brasileiro.
Systemic myotoxicity is characterized by increases in serum myoglobin concentration and creatine kinase (CK) activity, hyperkalemia, and also acute kidney injury; this is mainly due to myoglobin toxicity in the renal tubules that can cause death [8,36]. The venom also causes oxidative stress, triggering an increase in lipid peroxidation, catalase, and glutathione-S-transferase activity in the liver, as well as plasma levels of aspartate aminotransferase (AST) and alanine aminotransferase (ALT), indicating hepatotoxicity [54]. In addition, pulmonary changes—respiratory failure/acute lung edema—have been reported as systemic complications associated with death in cases of B. atrox snakebite envenoming [17,55]. However, renal alterations are the complication that causes greatest concern in snakebites, among them acute glomerulonephritis, acute tubular necrosis, and acute renal injury (ARI). the latter is related to cases of lethality from snakebite envenoming [60]. The pathogenesis of ARI is still not fully understood. However, it is known that kidney injuries can be produced by the isolated or combined action of different ischemic and/or nephrotoxic mechanisms triggered by the biological activities of venoms in the body [8]. Studies with isolated kidneys have shown that venoms from different Bothrops species alter renal function parameters that vary according to the venom composition [8,61,62,63].
3. Bothrops Snakebite as a Prothrombotic State
Current evidence about the prothrombotic state of Bothrops envenoming is mainly based on experimental (animal studies), clinical, and laboratory data, as well as case reports [26]. B. asper venom has a high procoagulant potential [64]; B. jararaca and B. atrox venoms contain toxins with thrombin-like activity, which catalyze the interaction of fibrinogen with fibrin [65,66,67]. Bothrops venoms also have prothrombin activators; the first known being berythractivase from B. erythromelas venom, a non-hemorrhagic class P-III metalloproteinase [68].
Procoagulant toxins from B. atrox venom activate factors II, X, and V, and increase the procoagulant activity of factor VIII, which, as a result, leads to the generation of intravascular thrombin that has also been reported [69,70,71]. Another clotting factor activated by isolated components of B. atrox venom is FXIII [71]. In addition, the fibrinogenolytic components of B. atrox venom, the poison may also contribute to fibrinogen degradation and coagulopathies [72]. B. jararaca venom also fibrinogenolytic activity [66], whereas B. atrox, B. jararaca, and B. neuwiedi venoms were able to degrade fibrin [73,74,75].
Snake venom thrombin-like enzymes (SVTLEs) belong to a class of serine proteases that can cause blood clotting in vitro, a characteristic exhibited by several snake venoms. Leucurobin from B. leucurus venom induces the release of fibrinopeptide A and traces of fibrinopeptide B [76]; SPBA from B. alternatus venom and BM-IIB34 kDa + BM-IIB32 kDa from B. moojeni venom induce plasma coagulation, and have fibrinogenolytic activity [77], as well as pictobin from B. pictus and barnettobin from B. barnetti venom, which coagulate plasma, but also fibrinogen, releasing fibrinopeptide A [78,79]. Moojase from B. moojeni venom also induces clotting of platelet-poor plasma and fibrinogen solutions in a dose-dependent manner, indicating thrombin-like properties due to proteolysis of human fibrinogen Aα chains, followed by late degradation of Bβ chains [80].
However, some Bothrops venoms induce systemic multifocal thrombotic complications that are triggered by a striking feature caused by B. caribbaeus and B. lanceolatus venoms; the mechanisms involved in these vascular obstructions still need to be clarified [23,81,82]. Interestingly, thrombocytopenia, minimally reduced prothrombin with normal activated partial thromboplastin time (APTT), and elevated fibrinogen concentration, are typical findings of victims with thrombosis after a bite by B. caribbaeus and B. lanceolatus, indicating the involvement of platelets and fibrinogen with this condition [23,81,82]. The victims of snakebites by B. atrox with systemic hemorrhage show a reduction in the levels of factor V, II, fibrinogen, plasminogen, and alpha 2-antiplasmin in plasma, while tissue factor and FDP levels are elevated [28,29]. Consumption coagulopathy resulting from the action of B. atrox venom may increase the risk of systemic bleeding seen in envenomation [28]. Moreover, in B. jararaca envenoming, about 56% of the patients had incoagulable blood; of these, 70% had systemic bleeding without systemic bleeding, and 18% had local bleeding alone (without local bleeding) [57].
An important event that is associated with systemic thrombotic complications is the occurrence of cerebral vessel occlusion. Although it is less common than hemorrhagic stroke, ischemic stroke has been described following B. atrox, B. caribbaeus, and B. lanceolatus snakebites [23,81,82,83]. The evidence of neurological symptoms occurred from 24 h up to 4 days after the snakebites, and all patients received antivenom therapy; the authors suggested that delayed time to treatment or the effectiveness of antivenom could be a factor that resulted in vascular cerebral thrombus formation. These events were directly associated with venom-induced coagulation disturbances (thrombocytopenia and clotting parameters alterations). Inflammatory parameters such as leukocytes/neutrophils and C-reactive protein were also elevated in some cases. Diagnosis of the cerebral infarction evaluated by imaging exams showed that the cerebral artery (anterior, posterior, or middle segments) and basilar artery were the main arteries with thrombosis, with infarcts occurring in cortical and occipital territories. Death and permanent disabilities were observed as a possible thrombotic stroke consequence in bitten patients [23,81,82].
As a consequence of the overall alterations in hemostasis, Bothrops venoms are responsible for inducing thrombotic events (Figure 3). Thrombotic microangiopathy (TMA) is an important complication observed in cases of Bothrops accidents and preclinical studies [24,84,85,86]. The TMA is characterized by erythrocyte destruction due to small-vessel damage and microthrombi deposition, resulting in thrombotic events that can lead to acute kidney injury as the predominant end-organ damage. Clinical–laboratory evidence associates this with microangiopathic hemolytic anemia, which is characterized by red blood cell fragments (schistocytes) on the peripheral blood film, delayed thrombocytopenia, and increased levels of bilirubin and lactate dehydrogenase [87]. Although there is an association of TMA with the coagulopathy induced by experimental Bothrops envenomation [88,89,90], the etiology of TMA remains unclear. Studies have claimed that the TMA observed in Bothrops accidents is associated with hemolytic uremic syndrome, due to its tendency for renal end-organ damage; however, there is no evidence that inflammatory mediators C3, C4, or ADAM13 were altered in the patients [24,84,85].
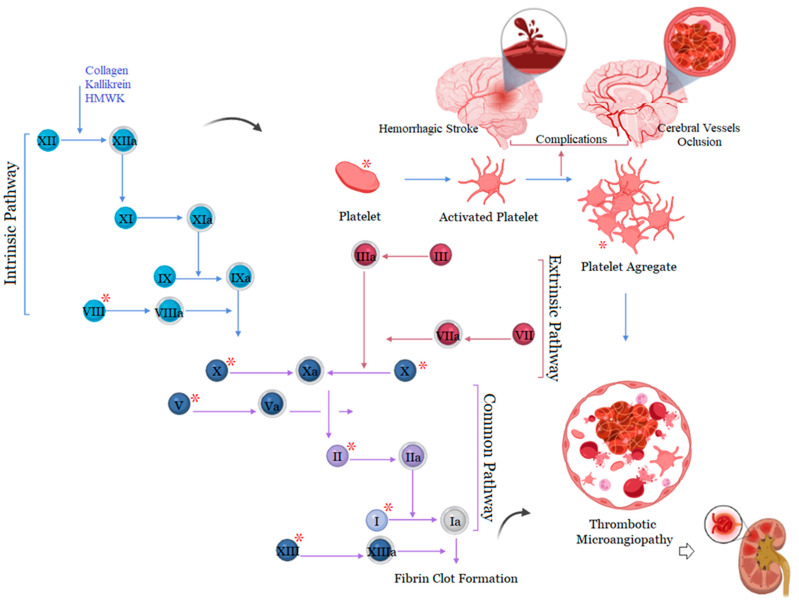
Figure 3.
Components of the hemostatic system targeted by toxins and several consequences of general changes in hemostasis during Bothrops snakebite envenoming. * Components of the hemostatic system that are affected by Bothrops toxins. Created with BioRender.com, accessed on 30 September 2022 by Joeliton S. Cavalcante.
4. Complement System Mediators’ Activation by Bothrops Venoms
The complement activity is another important pathway of the pro-inflammatory cascade that is responsible for vasodilation, chemotaxis, and leukocyte activation [91]. Bothrops venoms can activate the complement cascade, generating large amounts of anaphylatoxins, such as C3a, C4a, and C5a, which are considered to be the bridge between innate and adaptive immunity. Alternative pathway activity is completely inhibited by the venoms of B. atrox, B. cotiara, B. moojeni, B. itapeningae, B. paradoi, B. hyoprorus, B. insularis, B. bilienatus, B. brazili, B. jararaca, and B. marajoensis. However, this effect is not observed for the venoms of B. fonsecai, B. taeniata, B. alternatus, B. leucurus, B. erythromelas, B. jararacussu, and B. neuwiedi, which do not affect the alternative pathway (Figure 4) [92].
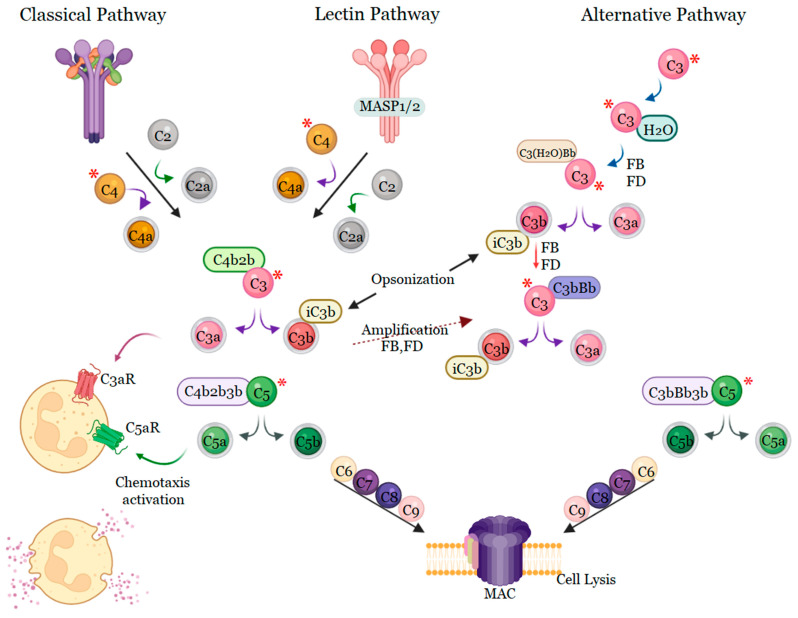
Figure 4.
Mechanisms of activation of the complement system by Bothrops venoms. Bothrops venoms activate the complement system via classical pathways, some also via lectins or the alternative pathway. All Bothrops venoms cleave C3 and C4, resulting in the synthesis of anaphylotoxins C3a, C4a, and C5a, and the terminal complement complex causing activation of chemotaxis. * Components of the complement system that are affected by Bothrops venoms. Created with BioRender.com, accessed on 30 September 2022 by Joeliton S. Cavalcante.
Delafonatine et al. [93] found that the B. lanceolatus venom, a Martinique native species, potentially activates the complement system and dose-dependently reduces the lytic activity of the alternative pathway. Bothrops venom also activates the classical complement pathway by cleaving the C1 inhibitor by proteases of this venom, interrupting the control of complement system activation [92]. The classical, lectin, and alternative pathways converge when an additional C3b protein associates with C2bC4b or C3bBb, which creates a C5 convertase that converts C5 to C5a and C5b. B. lanceolatus venom induces a significant production of C5a that is capable of inducing an influx of calcium in leukocytes. However, inhibition occurred in the presence of 1,10-phenanthroline, suggesting that C5 cleavage and consequent C5a release occur by the action of metalloproteases present in the venom [93]. C5b recruits C6 followed by C7, C8, and various C9 proteins to insert into the target cell membrane to form the MAC complex, also called the terminal complement complex, a pore in the membrane to induce cell lysis [94,95]. In addition to the formation of the MAC complex, the complement system has other functionalities, including opsonization, NETose, and the production of the so-called anaphylatoxins (C3a and C5a) [94,96,97].
5. Role of Neutrophils
Neutrophils are the first line of defense of the innate immune system [98], of which the ability of Bothrops venoms to stimulate these cells is a striking feature of great relevance in the context of inflammation. Neutrophils are present in myonecrotic and hemorrhagic areas, or even in the inflammatory infiltrate [31]; once activated, they produce pro-inflammatory cytokines, phagocytose, and release extracellular neutrophil traps (NETs), acting on the repair process, or potentially causing tissue damage [99,100,101,102,103,104,105]. The effects of Bothrops venoms on neutrophils have been explored, generating a large volume of information over the decades [31]. In vivo studies have shown that in inflammatory events, venoms can induce the migration of polymorphonuclear neutrophils to the envenoming sites of B. alternatus [106,107], B. asper [108,109], B. atrox [110], B. erythromelas [106], B. jararaca [109,111], B. jararacussu [112], B. lanceolatus [113], B. taeniata, and B. bilineata [114].
Neutrophilia and high concentrations of NETs in circulating blood, and thrombocytopenia, are described as an indicator of severity and a poor clinical outcome in thromboinflammatory diseases [115,116]; thus, they may be indicators for thromboinflammation and predictors of a poor clinical outcome during envenoming. It is known that patients with severe tissue damage caused by B. atrox venom have an immunological profile that is polarized to the Th1 response, and present a more intense local immune response with high levels of IL-1β, IL-6, TNF-α, MIP-1 (CXCL-1), and MIP-2 (CXCL-2) [43,110]. However, the Th1 response caused by B. atrox venom is regulated by neutrophils and the myeloid differentiation factor 88 (MyD88) pathway [40]. In addition, neutrophilia and thrombocytopenia of varying intensities have been reported in Bothrops snakebite envenoming, but more intense changes have been reported in cases that had tissue loss and/or limb amputations [19,21,22].
6. Cytokines and Their Role in Inflammation: An Overview
The cytokine storm is a systemic inflammatory syndrome characterized by the emergence of multiple disorders in the regulation of the immune response [117,118,119,120]. During the NETosis process, neutrophils can amplify the production of cytokines, while activated macrophages secrete large amounts of cytokines, which can cause organ damage. On the other hand, NK cells have their cytolytic function attenuated during the cytokine storm, hindering the process of inflammation [121].
Although diverse, only some subtypes of T cells are also implicated in and/or influenced by the cytokine storm. Thus, the ability of cytotoxic T lymphocytes (CTLs) to kill damaged and/or infected cells is impaired, which causes prolonged T cell activation that culminates in cascades of tissue damage at the site of inflammation [122]. A complex and interconnected network of cell types, signaling pathways, and cytokines is involved in cytokine storm disorders. Important crosstalk refers to the participation of plasma proteins, such as complement proteins and other inflammatory mediators, which contribute to the genesis and amplification of cellular responses, and provide feedback on cytokine signaling. In this way, cytokines can induce an increase in the production of complement proteins, which in turn can increase or inhibit the production of cytokines, which can cause collateral damage if excessive.
The inflammatory process caused by Bothrops venoms involves leukocytes infiltration, mainly polymorphonuclear and/or mononuclear cells at the site of injury, and the involvement of other resident cells that produce and release cytokines in response to Bothrops snake venom (Table 1) [123]. Bothrops venom toxins are also capable of inducing cytokine synthesis. Cytokine release in the kidney increased by the action of Asp-49 PLA2 (IL-10) and Lys-49 PLA2 (TNF-α, IL-1β, IL-10) from B. pauloensis venom [62], while levels of TNF-α, IL-1β, and IL-10 increased in isolated kidneys perfused with B. alternatus venom [124]. BJ-PLA2-I, a PLA2 Asp49 from B. jararaca venom, was found to induce increases in IL-6 and IL-1β in the inflammatory exudate of mice [125]. The injection of myotoxic PLA2s (Lys-49 and Asp-49) and B. asper venom SVMP also promoted increases in the concentrations of IL-1, IL-6, and TNF-α in the peritoneal exudate of mice [126,127]; however, intramuscularly, these toxins induced increases in IL-6 and IL-1β in muscle tissue, but not in TNF-α [118].
Table 1.
Some examples of the effects of Bothrops snake venoms in the synthesis and release of cytokines in different experimental models.
| Venom | Model | Cytokines | Reference |
|---|---|---|---|
| B. alternatus | mice peritoneal macrophages | TNFα, IL1, IL12 and IL6 | [107] |
| B. alternatus | Raw 264.7 cells | TNFα, IL1, IL12 and IL6 | [107] |
| B. asper | peritoneal cavity of mice | IL-6 and TNF-a | [126] |
| BaP1 from B. asper venom | macrophages in vitro | TNF-a | [128] |
| B. atrox | peritoneal cavity of mice | TNF-a, IL-6, IL-12p70, IL-10 and CCL-2 | [129] |
| B. erythromelas | mice splenocytes | IFN-γ, IL-6, IL-10 and NO | [130] |
| B. jararacussu | footpad samples | TNF-α and IL-1β | [112] |
| B. lanceolatus | human keratinocytes—HaCaT | IL-8, MCP-1, RANTES | [131] |
| B. lanceolatus | endothelial vascular cells—EAhy926 | IL-8, MCP-1, RANTES e IL-6 | [131] |
| B. moojeni | spinal cord | IL-10 | [132] |
| B. moojeni | footpad samples | IL-6, IL-10 and TNF-α | [132] |
Furthermore, soluble levels of chemokines (CXCL-8, CCL-5, CXCL-10, and CCL-2) and cytokines (IL-6, IL-1,0, and IL-2) are higher in victims of B. atrox bites, with some tissue complication, and can more significant based on the severity of the damage. In addition, patients with more severe complications were found to present a profile of individuals with a high production of the molecules CXCL-9, IL-6, and IL-10, while the group with severe complications presented the profile of a high production of molecules CXCL-8, CXCL-9, CCL-2, CXCL-10, IL-6, IL-1β, IL-10 and IL-2, a profile that is opposite to that observed in healthy individuals [43]. The Bothrops snakebite inflammatory response was shown to be responsible for modulating early adverse reactions (EARs) associated with antivenom therapy. Soares and colleagues [133] observed that patients who presented increased levels of CXCL-8 and IL-2 were less susceptible to developing EARs, suggesting the involvement of both chemokines/cytokines during the onset of antivenom adverse reactions. Patients with a cytokine storm have high-degree fevers in severe cases. Fever can be induced by interleukin-1, interleukin-6, or TNF, through different mechanisms; more severe cases can develop kidney failure, liver damage, and other outcomes (Figure 5). The specific markers are restricted to the dosage of circulating cytokines, and the laboratory findings from cytokine storms are variable and influenced by the cause, although nonspecific markers of inflammation, such as C-reactive protein (CRP), show an increase that is correlated with severity [122,134,135].
Figure 5.
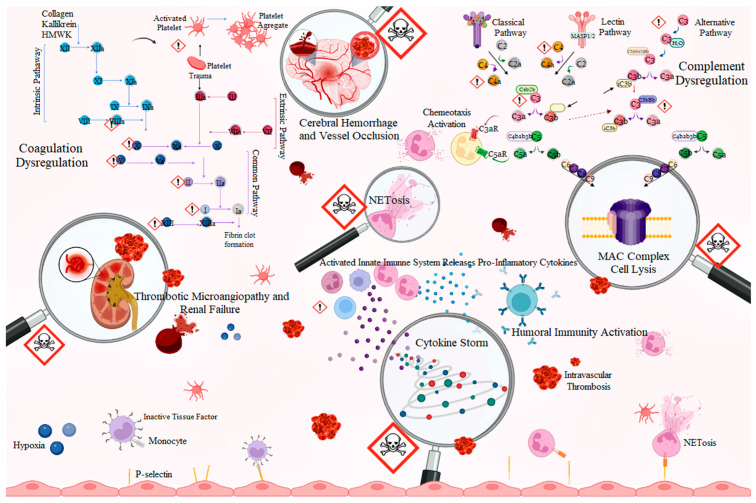
Thromboinflammatory pathways in Bothrops envenomation. The direct action of toxins on components of the hemostatic and complement system leads to hemostatic and inflammatory changes in Bothrops envenomation, which can result in clinical complications responsible for cases of tissue loss and death. Bothrops venoms act directly on clotting factors, causing the activation of the cascade that culminates in the formation of fibrin. During fibrin formation, clotting factors are consumed, resulting in disseminated intravascular coagulation. The generated fibrin induces the formation of intravascular thrombin, and plays a critical role in thrombotic microangiopathy through the lysis of erythrocytes that collide with it, which can lead to acute renal failure. The activation of the complement system modulated by the cleavage of key factors by the action of venoms amplifies the toxicity of the venom through chemotaxis and the activation of leukocytes, especially neutrophils. Neutrophils can form NETs that damage local tissue which has already been affected by the venom entering the body. In addition, the activation of the complement system culminates in the formation of the membrane attack complex that potentiates local tissue damage. Endothelial activation by platelets, leukocytes, and ischemia–reperfusion events result in the expression of adhesion molecules, including P-selectin, which recruits leukocytes and, in turn, red blood cells to blood vessel walls. The recruitment of leukocytes, platelets, and red blood cells to the vascular wall, together with the clotting processes, extracellular neutrophil trap components, and the formation of heterocellular aggregates between platelets, leukocytes, and red blood cells, with the subsequent entrapment of red blood cells; this results in the cerebral vaso-occlusive processes. In other cases, due to thrombocytopenia, cerebral hemorrhage occurs. Exclamation points represent targets of toxins. Skulls represent clinical complications associated with the effects of venom. Loupes indicate the need to study the consequences of clinical complications that are poorly understood. Created with BioRender.com, accessed on 4 June 2023 by Joeliton S. Cavalcante.
7. Platelets and Bothrops Venoms
Platelets are enucleated cells that are essential for hemostasis and predominant cellular elements in the thromboinflammation process [136]. After an injury, platelets perform a wide variety of functions, which include adhesion to endothelial and subendothelial structures, followed by activation and aggregation, constituting the initial hemostasis response [137]. Once platelets become aggregated, the coagulation cascade is triggered by either intrinsic or extrinsic pathways, leading to the activation of prothrombin to thrombin which converts fibrinogen molecules to fibrin [137]. Thrombin is considered to be a central mediator of thromboinflammation; it activates platelets by cleaving protease-activated receptors (PARs), leading to thrombus formation [138]. This mediator affects the components of the vasculature through the cleavage of components of the coagulation, complement, and fibrinolytic systems, as well as the activation of endothelial cells, leukocyte migration, macrophage activation, expression of vascular endothelial growth factor in smooth muscle cells and others (reviewed in [139]). In addition to PARS, a wide variety of receptors are found on the platelet surface (classified as integrins, selectins, and receptors of the immunoglobulin type) that allow platelet multifunctionality in hemostasis, inflammation, thromboinflammation, and resolution [140,141,142,143]. The engagement of platelet receptors allows platelets to act as contact elements between complement, coagulation, and contact systems [144]. In addition, these functionally multifaceted cells also have the ability to establish interactions with leukocytes in inflammation [136,142].
Platelet–complement interaction is mediated by several complement factors, including a form of C3 and other complement receptors such as C3aR and C5aR, and complement binding evokes an activation response in platelets. When activated, platelets can also activate the complement cascade, resulting in its amplification [142,145]. In addition, platelets, through the lectin pathway, affect the coagulation system by activating MASP-1, which can exert thrombin-like activity [146,147]. However, this interaction represents highly complex and multifactorial crosstalk, which is beyond the role of C3, C3aR, and C5aR. To illustrate, P-selectin is a platelet activator of the alternative complement pathway [148], of receptors binding to complement components such as C1q [149], the activation of C4 [150], the deposition of C3b and C5b-9 on activated platelets, of complement activation by platelets without thrombin production, and reduced platelet C3 activation in the absence of C1 or Factor B [150], representing other examples by which platelets can trigger the complement system.
Platelet–leukocyte interaction is mediated by P-selectin expressed on the surface of activated platelets, which can interact with the PSGL-1 selectin receptor expressed on leukocytes. At the site of inflammation, platelets aid leukocyte emigration by capturing leukocytes at specific extravasation sites, thus facilitating tissue infiltration in a PSGL-1/P-selectin-dependent manner [151]. Neutrophils actively search for activated platelets to engage in a PSGL-1-mediated signaling event [152]. In addition, activated platelets express the integrin GpIIb/IIIa, enabling binding to fibrinogen, leading to crosslinking with neutrophils mediated by their surface integrin receptor Mac-1 that can directly interact with the platelet receptor GpIbα, and thus regulating thrombosis [153]. Other platelet–leukocyte interactions are also known to modulate prothrombotic and proinflammatory pathways [142].
The role of platelets in inflammatory responses modulated by Bothrops venoms has been reported [30,154]. Despite toxins from Bothrops venom that exist with the ability to disrupt platelet activation, the entire ability of the venom to induce this process has been seldom explored. B. jararaca venom activates platelets, causing platelet sequestration and pulmonary thrombin development composed of platelet aggregates and dense fibrin sheaths, in addition to fibrin in the kidneys. Within this framework, it is important to highlight that the combination of pro-inflammatory and thrombotic scenarios can lead to organ failure due to excessive platelet activation, coagulation, and fibrin deposition in the microvasculature [155]. PLA2 isolated from Bothrops venoms has already been cataloged with the potential to inhibit platelet aggregation. BmooTX-I [156] and BmooPLA2 [157] isolated from B. moojeni venom, BthA-I-PLA2 from B. jararacussu [158], BJ-PLA2 from B. jararaca [159], BE-I-PLA2 from B. erythromelas [68], and BpPLA2-TXI of B. pauloensis [160] represent some examples. However, also noteworthy is the ability of bothrotropstoxin-II (BthTX-II), an Asp49 PLA2 isolated from B. jararacussu venom [161], to induce platelet aggregation. When approaching Bothrops SVMPs, an interesting case to present is jararhagin. Jararhagin is a P-III SVMPS from B. jararaca venom that inhibits platelet aggregation induced by ristocetin and collagen [56]. In addition, jararhagin dramatically reduces α2β1 integrin on the surface of platelets [162]. Alternagin from B. alternatus [163] also inhibits collagen-induced platelet aggregation, and barnettlysin-I from B. barnetti venom causes platelet inhibition induced by ristocetin and collagen [164]. In addition to PLA2 and SVMPs, other toxins from Bothrops venoms have already been isolated, and their roles on platelets have already been explored (Table 2).
Table 2.
Effects of toxins in Bothrops snake venoms on platelets.
| Protein Family | Protein Name | Species | Inhibition (−)/Activation (+) of Aggregation | References |
|---|---|---|---|---|
| PLA2 | Bothropstoxin-II | B. jararacussu | + | [161] |
| BmooPLA2 | B. moojeni | − | [157] | |
| BJ-PLA2 | B. jararaca | − | [136] | |
| BthA-I- PLA2 | B. jararacussu | − | [158] | |
| BE-I-PLA2 | B. erythromelas | − | [68] | |
| BpPLA2-TXI | B. pauloensis | − | [160] | |
| BmooTX-I | B. moojeni | − | [156] | |
| Braziliase-I and II | B. brazili | − | [165] | |
| BJ-PLA2-I | B. jararaca | − | [125] | |
| BaltPLA2 | B. alternatus | − | [166] | |
| SVSP | BpirSP27 | B. pirajai | + | [167] |
| BpirSP41 B | B. pirajai | + | [167] | |
| TLBm | B. marajoensis | + | [168] | |
| PA-BJ | B. jararaca | + | [169] | |
| Thrombocytin | B. jararaca | + | [169] | |
| Cerastotin | B. jararaca | + | [170] | |
| Bothrombin | B. jararaca | + | [171] | |
| Moojase | B. moojeni | + | [80] | |
| SVMP | BmooMPα-II | B. moojeni | − | [172] |
| Jararhagina | B. jararaca | − | [173] | |
| BmooPAi | B. moojeni | − | [174] | |
| Bar-I | B. barnetti | − | [164] | |
| Batroxrhagin | B. atrox | − | [74] | |
| BaltDC | B. alternatus | − | [175] | |
| Moojenactivase | B. moojeni | + | [176] | |
| Atroxlysin-III | B. atrox | − | [177] | |
| r-colombistatins 2,3,4 | B. colombiensis | − | [178] | |
| LAAOs | BpirLAAO-I | B. pirajai | + | [179] |
| Bp-LAAO | B. pauloensis | + | [180] | |
| BmooLAAO-I | B. moojeni | + | [181] | |
| Bl-LAAO | B. leucurus | − | [182] | |
| CTL | Botrocetin | B. jararaca | + | [183] |
| Baltetin | B. alternatus | − | [184] |
The platelet counts of patients in less severe cases appear to rapidly normalize after administration of the antivenom. The increase in platelet count after antivenom treatment may be related to the production of interleukin-6, a potent promoter of thrombopoiesis and megakaryocytopoiesis [27,185,186,187]. This has already been reported in experimental models using the venom of B. asper, B. jararaca [188], and in human envenomations by Bothrops from Costa Rica and Brazil [189,190].
Victims of B. atrox, before antivenom therapy, have a platelet count that correlates poorly with tissue factor, factor II, factor V, D-dimer, plasminogen activity, and a moderate correlation with fibrinogen and FDP. In addition, platelet counts are lower in patients with systemic bleeding [28,29]. Thus, the combination of thrombocytopenia and changes in clotting factor levels increases the risk of bleeding, suggesting crosstalk between these systems. Thrombocytopenia of varying intensity is found in victims of B. jararaca bites, but whether or not thrombocytopenia is associated with spontaneous systemic bleeding depends on the venom involved [27]. Victims of B. atrox, prior to antivenom therapy, have a platelet count that correlates poorly with tissue factor, factor II, factor V, D-dimer, plasminogen activity, and a moderate correlation with fibrinogen and FDP. In addition, platelet counts are lower in patients with systemic bleeding [29]. Thus, the combination of thrombocytopenia and changes in clotting factor levels increases the risk of bleeding, suggesting crosstalk between these systems.
8. Snake Venom-Induced Coagulation/Inflammation Cross-Talk and Participation of Tissue Factor
It is well known that both inflammation and the hemostatic system are highly integrated, and the unbalance of these events is responsible for several pathological conditions. An inflammatory response can trigger coagulation (and vice versa) [191]. The independent mechanisms of the inflammatory response and coagulation disturbance induced by Bothrops venoms are well known; however, their association still is a growing issue in the toxinology field. A recent study on B. atrox snakebite patients from the Brazilian Amazon has evaluated the inflammation/coagulation cross-talk [192]. By comparing inflammatory mediators’ profiles and fibrinogen consumption, the authors reported that the cytokines/chemokines CXCL-8, CXCL-9, CCL-2, and IL-6 are directly affected by fibrinogen levels, and increased levels of CCL-5 and decreased levels of IFN-γ were observed in patients with hypofibrinogenemia. The study was the first report on the inflammatory response and coagulation mutual relationship involving B. atrox snakebite patients, leading the way for other studies [192].
The interface of the immune response and hemostasis consists of a wide range of reactions characterized by the involvement of cellular events and blood mediators. Among the molecules, tissue factor (coagulation factor III) is considered to be a hallmark in the inflammation/coagulation cross-talk. Tissue factor (TF) is a transmembrane protein constitutively expressed in perivascular cells where it forms a hemostatic barrier. However, upon inflammatory stimulus, TF is expressed mostly by endothelial cells and monocytes within the intravascular compartment, and interacts with circulating factor VII to generate the extrinsic tenase complex, thus activating FX and triggering blood clotting [193,194]. Considering snake venoms, pre-clinical and clinical studies have shown the involvement of Bothrops venoms and toxins are capable to induce the expression of TF. The only clinical study showing the involvement of TF was conducted with patients following B. atrox snakebites [29]. The study showed an increase in plasma levels of TF antigen, which presented correlations with hemostatic components such as coagulation factors, platelets, and the fibrinolysis system. The authors also demonstrated that patients with systemic bleeding presented higher levels of TF compared to those without bleeding, and that TF levels also significantly increased in patients with moderate/severe edema when compared to those with mild edema. These data suggest the expression of intravascular TF, as well as extravascular TF released by local trauma and damage caused by envenomation [29]. Similar findings were also observed by Yamashita and colleagues [195], using an experimental approach, showing that both local (subcutaneous) and systemic (intravenous) administration of B. jararaca venom in mice increased plasma TF activity, which was confirmed by increased skin (local) and lung (systemic) TF expression [195].
In mechanisms of venom-induced TF participation, SVMPs have been found to play an important role. Yamashita and colleagues [195] observed that SVMPs are the major class of toxins within B. jararaca to induce TF activity in mice. In vitro, berythractivase and moojenactivase, both PIII SVMPs isolated from B. erythromelas and B. moojeni, respectively, were capable to induce TF expression on vascular cells. Berythractivase, a prothrombin activator, induced TF activity and gene expression in endothelial cells [196]. Moojenactivase, a prothrombin and factor X activator, was capable of inducing TF expression of peripheral blood mononuclear cells (PBMC), in both in vivo and in vitro, which led these cells to adopt pro-coagulant behavior [89,176]. Concerning the inflammation/coagulation interface, berythractivase was also found to up-regulate endothelial ICAM-1 gene expression, as well as nitric oxide (NO) generation, prostaglandin I2 (PGI2) and interleukin-8 (IL-8) release [197]. As for moojenactivase, the toxin was capable of inducing TF production/activity along with inflammatory mediators TNF-α, CXCL-8, and CCL-2 in PBMC in vitro, and leukocyte, IL-6, and TNF-α in mice [89,176]. Aside from SVMPs, Cezarette and colleagues [197] demonstrated that BjcuL, a galactoside-binding lectin, induced TF production/activity mediated by its pro-inflammatory activity. BjcuL was capable of interacting and activating Toll-like receptor 4 (TLR4) to induce IL-1β, TNF- α, and IL-6 expression, as well as TF in monocytes in vitro. When the cells were preincubated with galactose, TLR4, and NFκB antagonist/inhibitor, not only were inflammatory mediators mitigated, but also TF [198].
Considering the studies described above, the inflammatory response induced by Bothrops venoms is also responsible for triggering procoagulant behavior. Aside from the direct activation of hemostatic events by venom toxins, the inflammation acts as a potentiating factor to improve coagulation disturbances during snakebites.
9. Thromboinflammation and Snakebite
Thromboinflammation has been used to refer to the activation of the cascade systems present in the blood, as well as the activation of its multicellular system [32]. However, thromboinflammatory pathways can exacerbate inflammation and cellular interactions, which can lead to vaso-occlusion, ischemia/reperfusion, and eventually irreversible organ damage [199]. The cascade and multicellular blood systems are involved in the immediate response to snake venoms, and although classically seen as independent systems, their interrelationship has been associated with the amplification of venom toxicity [33,34,35]. The characterization of Bothrops envenoming as a prothrombotic, inflammatory state with multiple blood cell activation is supported by extensive experimental, clinical, and laboratory data, as well as case reports. From an experimental point of view, several components of the hemostatic system have already been characterized as direct and/or indirect targets of Bothrops venoms and/or their toxins. The same can be said for the complement system, whose different venoms can cleave key effectors that cause complement activation via the classical, alternative, or lectin pathways. Although there are some inconsistencies between the results, possibly reflecting differences in the composition of the venoms or, for example, issues related to the method, processing, and analysis of samples, it can be safely stated that Bothrops envenoming is a condition in which the hemostasis-complement-blood cells are affected.
However, the complex interaction between hemostasis and activation of innate immunity makes it difficult to precisely define the relative contribution of each of these two processes to the pathogenesis of different complications, possibly explaining the absence of a direct association between the classic biomarkers of hemostasis activation and the risk or severity of some of these clinical manifestations [200]. Therefore, the crossing of inflammation and thrombosis is very well exemplified during snakebite envenoming, due to the presence of a wide variety of characterized proteins that can activate the innate immune system and/or hemostasis. Evidence supports the cross-referencing of inflammation with hemostasis: (i) studies in animal models report disturbances of hemostasis; despite some heterogeneity within the model and within the venom, a less effective hemostatic system is associated with an increase in hemorrhagic manifestations; (ii) it has been demonstrated that components of the coagulation system, such as platelets, also signal via immunological pathways; (iii) there are examples of toxins and venoms whose mechanisms disturb the local hemostatic balance and induce inflammation; and (iv) studies show that leukocytes are not only found at the site of envenomation, but also in arterial and venous thrombi.
Complement activation and primary hemostasis can be linked to the moment of initiation, as a direct interaction of vWF and C1q was recently described [201]. The formation of platelet–leukocyte aggregates is highly relevant to cardiopulmonary bypass, which is mediated by C5a [202,203]. The formation of platelet–leukocyte aggregates, which is considered an important event in several contexts of thromboinflammation, is mediated by the alternative complement pathway and, specifically, by its regulator properdin; this suggests that the complement system may play a role in thrombocytopenia [204,205]. The reproduction of this phenomenon by the venom of other species, the mechanisms by which Bothrops venoms induce thrombocytopenia, and the possible relationship of the complement system on platelets, need elucidation. Focusing on the pathophysiological and translational relevance of a complement–platelet interaction hypothesis, it is noteworthy that complement activation is relevant in several inflammatory conditions associated with injury. Moreover, although platelets have been suggested as a key mediator of thromboinflammation in envenomation, complement activation can also be considered of equal relevance, mainly because of the evidence that has shown platelet activation by the complement system, as well as the ability of the complement pathway to be modulated by platelets.
10. Conclusions
The inflammatory response and coagulation disorders are considered hallmark mechanisms associated with local and systemic effects during Bothrops snakebite. However, the association of both events still is a recent issue in toxinology, although this is well established in other diseases. The crosstalk is responsible for potentiating both inflammatory and hemostatic alterations, enhancing prothrombotic conditions associated with thrombotic microangiopathy and tissue ischemia. Therefore, studies on the crosstalk between hemostasis/inflammation and thromboinflammatory events should be encouraged, seeking to highlight new mechanisms to understand their role in the pathophysiology of envenomation. The search for thromboinflammatory markers with predictive or prognostic roles can provide support to improve the clinical condition. Additionally, the progress made in better understanding the coagulation/inflammation interface enlightens the prospects to novel therapeutical approaches associated with antivenom therapy. Finally, we believe that this new aspect may elucidate previously unexplored paths in the complex pathology triggered by snake venoms and toxins, and may allow the discovery of new therapeutic targets and procedures to address the mortality and morbidity of envenomation.
Author Contributions
Conceptualization, J.S.C.; investigation, J.S.C., D.E.G.d.A., N.A.S.-F., M.A.S., A.d.A.B., L.B., P.L.A., S.S.O., W.M.M. and R.S.F.J.; writing—original draft preparation, J.S.C., P.L.A. and S.S.O.; writing—review and editing, N.A.S.-F., M.A.S., J.A.G.S., W.M.M. and R.S.F.J. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
This study was approved by the ethics committee of the Fundação de Medicina Tropical Heitor Vieira Dourado, Manaus, Western Brazilian Amazonia (FMT-HVD) (CAAE: 31535420.1.0000.0005).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
W.M.M. was funded by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq productivity scholarship), Fundação de Amparo à Pesquisa do Estado do Amazonas (PRÓ-ESTADO, call 011/2021—PCGP/FAPEAM, call 010/2021—CT&I ÁREAS PRIORITÁRIAS, call 003/2022-PRODOC/FAPEAM, and POSGRAD), and by the Ministry of Health, Brazil (proposal no. 733781/19-035). Rui Seabra Ferreira Júnior (RSFJr) is a CNPq PQ1D research fellow No. 301608/2022-9. The APC was funded by FAPESP Proc 2021/11936-3 [R.S.F.Jr.].
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Chippaux J.-P. Snakebite Envenomation Turns Again into a Neglected Tropical Disease! J. Venom. Anim. Toxins Incl. Trop. Dis. 2017;23:38. doi: 10.1186/s40409-017-0127-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Minghui R., Malecela M.N., Cooke E., Abela-Ridder B. WHO’s Snakebite Envenoming Strategy for Prevention and Control. Lancet Glob. Health. 2019;7:e837–e838. doi: 10.1016/S2214-109X(19)30225-6. [DOI] [PubMed] [Google Scholar]
- 3.Gutiérrez J.M., Calvete J.J., Habib A.G., Harrison R.A., Williams D.J., Warrell D.A. Snakebite Envenoming. Nat. Rev. Dis. Prim. 2017;3:17063. doi: 10.1038/nrdp.2017.63. [DOI] [PubMed] [Google Scholar]
- 4.Chippaux J.-P. Incidence and Mortality Due to Snakebite in the Americas. PLoS Negl. Trop. Dis. 2017;11:e0005662. doi: 10.1371/journal.pntd.0005662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Alencar L.R.V., Quental T.B., Grazziotin F.G., Alfaro M.L., Martins M., Venzon M., Zaher H. Diversification in Vipers: Phylogenetic Relationships, Time of Divergence and Shifts in Speciation Rates. Mol. Phylogenet. Evol. 2016;105:50–62. doi: 10.1016/j.ympev.2016.07.029. [DOI] [PubMed] [Google Scholar]
- 6.Carrasco P.A., Venegas P.J., Chaparro J.C., Scrocchi G.J. Nomenclatural Instability in the Venomous Snakes of the Bothrops Complex: Implications in Toxinology and Public Health. Toxicon. 2016;119:122–128. doi: 10.1016/j.toxicon.2016.05.014. [DOI] [PubMed] [Google Scholar]
- 7.Ferraz C.R., Arrahman A., Xie C., Casewell N.R., Lewis R.J., Kool J., Cardoso F.C. Multifunctional Toxins in Snake Venoms and Therapeutic Implications: From Pain to Hemorrhage and Necrosis. Front. Ecol. Evol. 2019;7:218. doi: 10.3389/fevo.2019.00218. [DOI] [Google Scholar]
- 8.Albuquerque P.L.M.M., Paiva J.H.H.G.L., Martins A.M.C., Meneses G.C., Silva Júnior G.B., Buckley N., Daher E.D.F. Clinical Assessment and Pathophysiology of Bothrops Venom-Related Acute Kidney Injury: A Scoping Review. J. Venom. Anim. Toxins Incl. Trop. Dis. 2020;26:e20190076. doi: 10.1590/1678-9199-jvatitd-2019-0076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Núñez V., Cid P., Sanz L., de La Torre P., Angulo Y., Lomonte B., Gutiérrez J.M., Calvete J.J. Snake Venomics and Antivenomics of Bothrops Atrox Venoms from Colombia and the Amazon Regions of Brazil, Perú and Ecuador Suggest the Occurrence of Geographic Variation of Venom Phenotype by a Trend towards Paedomorphism. J Proteom. 2009;73:57–78. doi: 10.1016/j.jprot.2009.07.013. [DOI] [PubMed] [Google Scholar]
- 10.Sousa L.F., Nicolau C.A., Peixoto P.S., Bernardoni J.L., Oliveira S.S., Portes-Junior J.A., Mourão R.H.V., Lima-dos-Santos I., Sano-Martins I.S., Chalkidis H.M., et al. Comparison of Phylogeny, Venom Composition and Neutralization by Antivenom in Diverse Species of Bothrops Complex. PLoS Negl. Trop Dis. 2013;7:e2442. doi: 10.1371/journal.pntd.0002442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Simizo A., Kitano E.S., Sant’Anna S.S., Grego K.F., Tanaka-Azevedo A.M., Tashima A.K. Comparative Gender Peptidomics of Bothrops Atrox Venoms: Are There Differences between Them? J. Venom. Anim. Toxins Incl. Trop. Dis. 2020;26:e20200055. doi: 10.1590/1678-9199-jvatitd-2020-0055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hatakeyama D.M., Tasima L.J., Bravo-Tobar C.A., Serino-Silva C., Tashima A.K., Rodrigues C.F., Aguiar W.D., Galizio N.D., Lima E.O., Kavazoi V.K., et al. Venom Complexity of Bothrops Atrox (Common Lancehead) Siblings. J. Venom. Anim. Toxins Incl. Trop. Dis. 2020;26:e20200018. doi: 10.1590/1678-9199-jvatitd-2020-0018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sanz L., Pérez A., Quesada-Bernat S., Diniz-Sousa R., Calderón L.A., Soares A.M., Calvete J.J., Caldeira C.A.S. Venomics and Antivenomics of the Poorly Studied Brazil’s Lancehead, Bothrops Brazili (Hoge, 1954), from the Brazilian State of Pará. J. Venom. Anim. Toxins Incl. Trop. Dis. 2020;26:e20190103. doi: 10.1590/1678-9199-jvatitd-2019-0103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pereira L.D., Messias E.A., Sorroche B.P., Oliveira A.D., Arantes L.M., Carvalho A.C., Tanaka-Azevedo A.M., Grego K.F., Carvalho A.L., Melendez M.E. In-Depth Transcriptome Reveals the Potential Biotechnological Application of Bothrops Jararaca Venom Gland. J. Venom. Anim. Toxins Incl. Trop. Dis. 2020;26:e20190058. doi: 10.1590/1678-9199-jvatitd-2019-0058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tasoulis T., Isbister G. A Review and Database of Snake Venom Proteomes. Toxins. 2017;9:290. doi: 10.3390/toxins9090290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.dos Santos Cavalcante J., Júnior F.A., Jorge R.J., Almeida C. Pain Modulated by Bothrops Snake Venoms: Mechanisms of Nociceptive Signaling and Therapeutic Perspectives. Toxicon. 2021;201:105–114. doi: 10.1016/j.toxicon.2021.08.016. [DOI] [PubMed] [Google Scholar]
- 17.da Silva Souza A., Sachett J.D., Alcântara J.A., Freire M., Alecrim M.D., de Lima Ferreira L.C., Fan H.W., de Souza Sampaio V., Monteiro W.M. Snakebites as Cause of Deaths in the Western Brazilian Amazon: Why and Who Dies? Deaths from Snakebites in the Amazon. Toxicon. 2018;145:15–24. doi: 10.1016/j.toxicon.2018.02.041. [DOI] [PubMed] [Google Scholar]
- 18.Nascimento da Costa T., Mota-da-Silva A., Colombini M., Moura-da-Silva A.M., Medeiros de Souza R., Monteiro W.M., Bernarde P.S. Relationship between Snake Size and Clinical, Epidemiological and Laboratory Aspects of Bothrops Atrox Snakebites in the Western Brazilian Amazon. Toxicon. 2020;186:160–167. doi: 10.1016/j.toxicon.2020.08.010. [DOI] [PubMed] [Google Scholar]
- 19.Luciano P.M., Silva G.E.B., Azevedo-Marques M.M. Acidente Botrópico Fatal. Med. (Ribeirão Preto) 2009;42:61–65. doi: 10.11606/issn.2176-7262.v42i1p61-65. [DOI] [Google Scholar]
- 20.Schulz R.S., Sousa Queiroz P.E., Bastos M.C., Miranda E.A., Jesus H.S., Gatis S.M.P. Treatment Of Wound Due To Ophidic Accident: Clinical Case. [(accessed on 4 June 2023)];CuidArte Enferm. 2016 10:172–179. Available online: http://www.webfipa.net/facfipa/ner/sumarios/cuidarte/2016v2/172-179.pdf. [Google Scholar]
- 21.Abero Valle L., da Fonseca Reis Silva D., Henrique Magalhães P., Aloísio Mattos P., Almeida Leal J. Bilateral Amputation of Inferior Extremities Due to Serious Bothrops Accident: A Case Report. [(accessed on 4 June 2023)];Arq. Med. Hosp. Fac. Cienc. Med. Santa Casa São Paulo. 2008 53:81–84. Available online: https://arquivosmedicos.fcmsantacasasp.edu.br/index.php/AMSCSP/article/view/404/456. [Google Scholar]
- 22.Graciano A.R., Naves De Carvalho K.C. Compartimental Syndrome Associated To Snake Bite Of The Bothrops Gender: Case Report. [(accessed on 4 June 2023)];Rev. Pesq. Saúde. 2017 18:54–56. Available online: https://periodicoseletronicos.ufma.br/index.php/revistahuufma/article/view/7881/4876. [Google Scholar]
- 23.Cirille S., Chatot-Henry D., Didier M., Moravie V., Thomas L., Numeric P., Bucher B. Multiple Cerebral Infarctions Following a Snakebite by Bothrops Caribbaeus. Am. J. Trop. Med. Hyg. 2002;67:287–288. doi: 10.4269/ajtmh.2002.67.287. [DOI] [PubMed] [Google Scholar]
- 24.Malaque C.M.S., Duayer I.F., Santoro M.L. Acute Kidney Injury Induced by Thrombotic Microangiopathy in Two Cases of Bothrops Envenomation. Clin. Toxicol. 2019;57:213–216. doi: 10.1080/15563650.2018.1510129. [DOI] [PubMed] [Google Scholar]
- 25.Pucca M.B., Franco M.V.S., Medeiros J.M., Oliveira I.S., Ahmadi S., Cerni F.A., Zottich U., Bassoli B.K., Monteiro W.M., Laustsen A.H. Chronic Kidney Failure Following Lancehead Bite Envenoming: A Clinical Report from the Amazon Region. J. Venom. Anim. Toxins Incl. Trop. Dis. 2020;26:e20200083. doi: 10.1590/1678-9199-jvatitd-2020-0083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Larréché S., Chippaux J.-P., Chevillard L., Mathé S., Résière D., Siguret V., Mégarbane B. Bleeding and Thrombosis: Insights into Pathophysiology of Bothrops Venom-Related Hemostasis Disorders. Int. J. Mol. Sci. 2021;22:9643. doi: 10.3390/ijms22179643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Santoro M.L., Sano-Martins I.S., Fan H.W., Cardoso J.L.C., Theakston R.D.G., Warrell D.A. Haematological Evaluation of Patients Bitten by the Jararaca, Bothrops Jararaca, in Brazil. Toxicon. 2008;51:1440–1448. doi: 10.1016/j.toxicon.2008.03.018. [DOI] [PubMed] [Google Scholar]
- 28.Oliveira S.S., Alves E.C., Santos A.S., Pereira J.P.T., Sarraff L.K.S., Nascimento E.F., De-Brito-Sousa J.D., Sampaio V.S., Lacerda M.V.G., Sachett J.A.G., et al. Factors Associated with Systemic Bleeding in Bothrops Envenomation in a Tertiary Hospital in the Brazilian Amazon. Toxins. 2019;11:22. doi: 10.3390/toxins11010022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Oliveira S.S., Alves E.C., Santos A.S., Nascimento E.F., Pereira J.P.T., Silva I.M., Sachett J.A.G., Sarraff L.K.S., Freitas-de-Sousa L.A., Colombini M., et al. Bleeding Disorders in Bothrops Atrox Envenomations in the Brazilian Amazon: Participation of Hemostatic Factors and the Impact of Tissue Factor. Toxins. 2020;12:554. doi: 10.3390/toxins12090554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Teixeira C., Fernandes C.M., Leiguez E., Chudzinski-Tavassi A.M. Inflammation Induced by Platelet-Activating Viperid Snake Venoms: Perspectives on Thromboinflammation. Front. Immunol. 2019;10:2082. doi: 10.3389/fimmu.2019.02082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zuliani J.P., Soares A.M., Gutiérrez J.M. Polymorphonuclear Neutrophil Leukocytes in Snakebite Envenoming. Toxicon. 2020;187:188–197. doi: 10.1016/j.toxicon.2020.09.006. [DOI] [PubMed] [Google Scholar]
- 32.Guo L., Rondina M.T. The Era of Thromboinflammation: Platelets Are Dynamic Sensors and Effector Cells During Infectious Diseases. Front. Immunol. 2019;10:2204. doi: 10.3389/fimmu.2019.02204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cavalcante J.S., Brito I.M., De Oliveira L.A., De Barros L.C., Almeida C., Rossini B.C., Sousa D.L., Alves R.S., Jorge R.J.B., Santos L.D. Experimental Bothrops Atrox Envenomation: Blood Plasma Proteome Effects after Local Tissue Damage and Perspectives on Thromboinflammation. Toxins. 2022;14:613. doi: 10.3390/toxins14090613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cavalcante J.S., Borges da Silva W.R.G., de Oliveira L.A., Brito I.M.C., Muller K.S., Vidal I.S.J., dos Santos L.D., Jorge R.J.B., Almeida C., de Lima Bicho C. Blood Plasma Proteome Alteration after Local Tissue Damage Induced by Bothrops Erythromelas Snake Venom in Mice. J. Proteom. 2022;269:104742. doi: 10.1016/j.jprot.2022.104742. [DOI] [PubMed] [Google Scholar]
- 35.dos Santos Cavalcante J., de Almeida C.A., Clasen M.A., da Silva E.L., de Barros L.C., Marinho A.D., Rossini B.C., Marino C.L., Carvalho P.C., Jorge R.J., et al. A Fingerprint of Plasma Proteome Alteration after Local Tissue Damage Induced by Bothrops Leucurus Snake Venom in Mice. J. Proteom. 2022;253:104464. doi: 10.1016/j.jprot.2021.104464. [DOI] [PubMed] [Google Scholar]
- 36.Gutiérrez J., Escalante T., Hernández R., Gastaldello S., Saravia-Otten P., Rucavado A. Why Is Skeletal Muscle Regeneration Impaired after Myonecrosis Induced by Viperid Snake Venoms? Toxins. 2018;10:182. doi: 10.3390/toxins10050182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gutiérrez J.M., Rucavado A., Escalante T., Herrera C., Fernández J., Lomonte B., Fox J.W. Unresolved Issues in the Understanding of the Pathogenesis of Local Tissue Damage Induced by Snake Venoms. Toxicon. 2018;148:123–131. doi: 10.1016/j.toxicon.2018.04.016. [DOI] [PubMed] [Google Scholar]
- 38.Rucavado A., Nicolau C., Escalante T., Kim J., Herrera C., Gutiérrez J., Fox J. Viperid Envenomation Wound Exudate Contributes to Increased Vascular Permeability via a DAMPs/TLR-4 Mediated Pathway. Toxins. 2016;8:349. doi: 10.3390/toxins8120349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Luchini L.S.G., Pidde G., Squaiella-Baptistão C.C., Tambourgi D.V. Complement System Inhibition Modulates the Pro-Inflammatory Effects of a Snake Venom Metalloproteinase. Front. Immunol. 2019;10:1137. doi: 10.3389/fimmu.2019.01137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Moreira V., Teixeira C., Borges da Silva H., D’Império Lima M.R., Dos-Santos M.C. The Role of TLR2 in the Acute Inflammatory Response Induced by Bothrops Atrox Snake Venom. Toxicon. 2016;118:121–128. doi: 10.1016/j.toxicon.2016.04.042. [DOI] [PubMed] [Google Scholar]
- 41.Bickler P.E. Amplification of Snake Venom Toxicity by Endogenous Signaling Pathways. Toxins. 2020;12:68. doi: 10.3390/toxins12020068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gimenes S.N.C., Sachett J.A.G., Colombini M., Freitas-de-Sousa L.A., Ibiapina H.N.S., Costa A.G., Santana M.F., Park J.-J., Sherman N.E., Ferreira L.C.L., et al. Observation of Bothrops Atrox Snake Envenoming Blister Formation from Five Patients: Pathophysiological Insights. Toxins. 2021;13:800. doi: 10.3390/toxins13110800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ibiapina H.N.S., Costa A.G., Sachett J.A.G., Silva I.M., Tarragô A.M., Neves J.C.F., Kerr M.W.A., Santana M.F., Martins-Filho O.A., Lacerda M.V.G., et al. An Immunological Stairway to Severe Tissue Complication Assembly in Bothrops Atrox Snakebites. Front. Immunol. 2019;10:1882. doi: 10.3389/fimmu.2019.01882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Escalante T., Rucavado A., Pinto A.F.M., Terra R.M.S., Gutiérrez J.M., Fox J.W. Wound Exudate as a Proteomic Window to Reveal Different Mechanisms of Tissue Damage by Snake Venom Toxins. J. Proteome Res. 2009;8:5120–5131. doi: 10.1021/pr900489m. [DOI] [PubMed] [Google Scholar]
- 45.Gutiérrez J., Escalante T., Rucavado A., Herrera C. Hemorrhage Caused by Snake Venom Metalloproteinases: A Journey of Discovery and Understanding. Toxins. 2016;8:93. doi: 10.3390/toxins8040093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gutiérrez J., Escalante T., Rucavado A., Herrera C., Fox J. A Comprehensive View of the Structural and Functional Alterations of Extracellular Matrix by Snake Venom Metalloproteinases (SVMPs): Novel Perspectives on the Pathophysiology of Envenoming. Toxins. 2016;8:304. doi: 10.3390/toxins8100304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Macêdo J.K.A., Joseph J.K., Menon J., Escalante T., Rucavado A., Gutiérrez J.M., Fox J.W. Proteomic Analysis of Human Blister Fluids Following Envenomation by Three Snake Species in India: Differential Markers for Venom Mechanisms of Action. Toxins. 2019;11:246. doi: 10.3390/toxins11050246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zambelli V., Picolo G., Fernandes C., Fontes M., Cury Y. Secreted Phospholipases A2 from Animal Venoms in Pain and Analgesia. Toxins. 2017;9:406. doi: 10.3390/toxins9120406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Baldo C., Jamora C., Yamanouye N., Zorn T.M., Moura-da-Silva A.M. Mechanisms of Vascular Damage by Hemorrhagic Snake Venom Metalloproteinases: Tissue Distribution and In Situ Hydrolysis. PLoS Negl. Trop. Dis. 2010;4:e727. doi: 10.1371/journal.pntd.0000727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Herrera C., Escalante T., Voisin M.-B., Rucavado A., Morazán D., Macêdo J.K.A., Calvete J.J., Sanz L., Nourshargh S., Gutiérrez J.M., et al. Tissue Localization and Extracellular Matrix Degradation by PI, PII and PIII Snake Venom Metalloproteinases: Clues on the Mechanisms of Venom-Induced Hemorrhage. PLoS Negl. Trop. Dis. 2015;9:e0003731. doi: 10.1371/journal.pntd.0003731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kini R., Koh C. Metalloproteases Affecting Blood Coagulation, Fibrinolysis and Platelet Aggregation from Snake Venoms: Definition and Nomenclature of Interaction Sites. Toxins. 2016;8:284. doi: 10.3390/toxins8100284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Santos Barreto G.N., de Oliveira S.S., Dos Anjos I.V., Chalkidis H.D., Mourão R.H., Moura-da-Silva A.M., Sano-Martins I.S., Gonçalves L.R. Experimental Bothrops Atrox Envenomation: Efficacy of Antivenom Therapy and the Combination of Bothrops Antivenom with Dexamethasone. PLoS Negl. Trop. Dis. 2017;11:e0005458. doi: 10.1371/journal.pntd.0005458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mora-Obando D., Fernández J., Montecucco C., Gutiérrez J.M., Lomonte B. Synergism between Basic Asp49 and Lys49 Phospholipase A2 Myotoxins of Viperid Snake Venom In Vitro and In Vivo. PLoS ONE. 2014;9:e109846. doi: 10.1371/journal.pone.0109846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sunitha K., Hemshekhar M., Thushara R.M., Santhosh M.S., Sundaram M.S., Kemparaju K., Girish K.S. Inflammation and Oxidative Stress in Viper Bite: An Insight within and Beyond. Toxicon. 2015;98:89–97. doi: 10.1016/j.toxicon.2015.02.014. [DOI] [PubMed] [Google Scholar]
- 55.Benvenuti L.A., França F.O.S., Barbaro K.C., Nunes J.R., Cardoso J.L.C. Pulmonary Haemorrhage Causing Rapid Death after Bothrops Jararacussu Snakebite: A Case Report. Toxicon. 2003;42:331–334. doi: 10.1016/S0041-0101(03)00167-3. [DOI] [PubMed] [Google Scholar]
- 56.Kamiguti A.S., Hay C.R.M., Theakston R.D.G., Zuzel M. Insights into the Mechanism of Haemorrhage Caused by Snake Venom Metalloproteinases. Toxicon. 1996;34:627–642. doi: 10.1016/0041-0101(96)00017-7. [DOI] [PubMed] [Google Scholar]
- 57.Kamiguti A.S., Cardoso J.L.C., Theakston R.D.G., Sano-Martins I.S., Hutton R.A., Rugman F.P., Warrell D.A., Hay C.R.M. Coagulopathy and Haemorrhage in Human Victims of Bothrops Jararaca Envenoming in Brazil. Toxicon. 1991;29:961–972. doi: 10.1016/0041-0101(91)90079-7. [DOI] [PubMed] [Google Scholar]
- 58.Silva de Oliveira S., Freitas-de-Sousa L.A., Alves E.C., de Lima Ferreira L.C., da Silva I.M., de Lacerda M.V.G., Fan H.W., Moura-da-Silva A.M., Monteiro W.M. Fatal Stroke after Bothrops Snakebite in the Amazonas State, Brazil: A Case Report. Toxicon. 2017;138:102–106. doi: 10.1016/j.toxicon.2017.08.021. [DOI] [PubMed] [Google Scholar]
- 59.de Queiroz M.R., de Sousa B.B., da Cunha Pereira D.F., Mamede C.C.N., Matias M.S., de Morais N.C.G., de Oliveira Costa J., de Oliveira F. The Role of Platelets in Hemostasis and the Effects of Snake Venom Toxins on Platelet Function. Toxicon. 2017;133:33–47. doi: 10.1016/j.toxicon.2017.04.013. [DOI] [PubMed] [Google Scholar]
- 60.Sitprija V. Animal Toxins and the Kidney. Nat. Clin. Pr. Nephrol. 2008;4:616–627. doi: 10.1038/ncpneph0941. [DOI] [PubMed] [Google Scholar]
- 61.Havt A., Toyama M.H., do Nascimento N.R.F., Toyama D.O., Nobre A.C.L., Martins A.M.C., Barbosa P.S.F., Novello J.C., Boschero A.C., Carneiro E.M., et al. A New C-Type Animal Lectin Isolated from Bothrops Pirajai Is Responsible for the Snake Venom Major Effects in the Isolated Kidney. Int. J. Biochem. Cell Biol. 2005;37:130–141. doi: 10.1016/j.biocel.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 62.Marinho A.D., de Moraes Silveira J.A., Chaves Filho A.J., Jorge A.R., Júnior F.A., Pereira V.B., de Aquino P.E., Pereira C.A., Evangelista J.S., Macedo D.S., et al. Bothrops Pauloensis Snake Venom-Derived Asp-49 and Lys-49 Phospholipases A2 Mediates Acute Kidney Injury by Oxidative Stress and Release of Inflammatory Cytokines. Toxicon. 2021;190:31–38. doi: 10.1016/j.toxicon.2020.12.004. [DOI] [PubMed] [Google Scholar]
- 63.Barbosa P.S.F., Havt A., Facó P.E.G., Sousa T.M., Bezerra I.S.A.M., Fonteles M.C., Toyama M.H., Marangoni S., Novello J.C., Monteiro H.S.A. Renal Toxicity of Bothrops Moojeni Snake Venom and Its Main Myotoxins. Toxicon. 2002;40:1427–1435. doi: 10.1016/S0041-0101(02)00156-3. [DOI] [PubMed] [Google Scholar]
- 64.Slagboom J., Mladić M., Xie C., Kazandjian T.D., Vonk F., Somsen G.W., Casewell N.R., Kool J. High Throughput Screening and Identification of Coagulopathic Snake Venom Proteins and Peptides Using Nanofractionation and Proteomics Approaches. PLoS Negl. Trop. Dis. 2020;14:e0007802. doi: 10.1371/journal.pntd.0007802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Stocker K., Barlow G.H. Methods in Enzymology. Academic Press; Cambridge, MA, USA: 1976. The Coagulant Enzyme from Bothrops Atrox Venom (Batroxobin) pp. 214–223. [DOI] [PubMed] [Google Scholar]
- 66.Antunes T.C., Yamashita K.M., Barbaro K.C., Saiki M., Santoro M.L. Comparative Analysis of Newborn and Adult Bothrops Jararaca Snake Venoms. Toxicon. 2010;56:1443–1458. doi: 10.1016/j.toxicon.2010.08.011. [DOI] [PubMed] [Google Scholar]
- 67.Monteiro W.M., Contreras-Bernal J.C., Bisneto P.F., Sachett J., Mendonça da Silva I., Lacerda M., Guimarães da Costa A., Val F., Brasileiro L., Sartim M.A., et al. Bothrops Atrox, the Most Important Snake Involved in Human Envenomings in the Amazon: How Venomics Contributes to the Knowledge of Snake Biology and Clinical Toxinology. Toxicon X. 2020;6:100037. doi: 10.1016/j.toxcx.2020.100037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.de Albuquerque Modesto J.C., Spencer P.J., Fritzen M., Valença R.C., Oliva M.L.V., da Silva M.B., Chudzinski-Tavassi A.M., Guarnieri M.C. BE-I-PLA2, a Novel Acidic Phospholipase A2 from Bothrops Erythromelas Venom: Isolation, Cloning and Characterization as Potent Anti-Platelet and Inductor of Prostaglandin I2 Release by Endothelial Cells. Biochem. Pharm. 2006;72:377–384. doi: 10.1016/j.bcp.2006.04.032. [DOI] [PubMed] [Google Scholar]
- 69.Hofmann H., Bon C. Blood Coagulation Induced by the Venom of Bothrops Atrox. 2. Identification, Purification, and Properties of Two Factor X Activators. Biochemistry. 1987;26:780–787. doi: 10.1021/bi00377a019. [DOI] [PubMed] [Google Scholar]
- 70.Rosing J., Govers-Riemslag J.W.P., Yukelson L., Tans G. Factor V Activation and Inactivation by Venom Proteases. Pathophysiol. Haemost. Thromb. 2001;31:241–246. doi: 10.1159/000048069. [DOI] [PubMed] [Google Scholar]
- 71.Kirby E.P., Niewiarowski S., Stocker K., Kettner C., Shaw E., Brudzynski T.M. Thrombocytin, a Serine Protease from Bothrops Atrox Venom. 1. Purification and Characterization of the Enzyme. Biochemistry. 1979;18:3564–3570. doi: 10.1021/bi00583a020. [DOI] [PubMed] [Google Scholar]
- 72.Cintra A.C.O., de Toni L.G.B., Sartim M.A., Franco J.J., Caetano R.C., Murakami M.T., Sampaio S.V. Batroxase, a New Metalloproteinase from B. Atrox Snake Venom with Strong Fibrinolytic Activity. Toxicon. 2012;60:70–82. doi: 10.1016/j.toxicon.2012.03.018. [DOI] [PubMed] [Google Scholar]
- 73.Baldo C., Tanjoni I., León I.R., Batista I.F.C., Della-Casa M.S., Clissa P.B., Weinlich R., Lopes-Ferreira M., Lebrun I., Amarante-Mendes G.P., et al. BnP1, a Novel P-I Metalloproteinase from Bothrops Neuwiedi Venom: Biological Effects Benchmarking Relatively to Jararhagin, a P-III SVMP. Toxicon. 2008;51:54–65. doi: 10.1016/j.toxicon.2007.08.005. [DOI] [PubMed] [Google Scholar]
- 74.Freitas-de-Sousa L.A., Amazonas D.R., Sousa L.F., Sant’Anna S.S., Nishiyama M.Y., Serrano S.M.T., Junqueira-de-Azevedo I.L.M., Chalkidis H.M., Moura-da-Silva A.M., Mourão R.H.V. Comparison of Venoms from Wild and Long-Term Captive Bothrops Atrox Snakes and Characterization of Batroxrhagin, the Predominant Class PIII Metalloproteinase from the Venom of This Species. Biochimie. 2015;118:60–70. doi: 10.1016/j.biochi.2015.08.006. [DOI] [PubMed] [Google Scholar]
- 75.Moretto Del-Rei T.H., Sousa L.F., Rocha M.M.T., Freitas-de-Sousa L.A., Travaglia-Cardoso S.R., Grego K., Sant’Anna S.S., Chalkidis H.M., Moura-da-Silva A.M. Functional Variability of Bothrops Atrox Venoms from Three Distinct Areas across the Brazilian Amazon and Consequences for Human Envenomings. Toxicon. 2019;164:61–70. doi: 10.1016/j.toxicon.2019.04.001. [DOI] [PubMed] [Google Scholar]
- 76.Magalhães A., Magalhães H.P.B., Richardson M., Gontijo S., Ferreira R.N., Almeida A.P., Sanchez E.F. Purification and Properties of a Coagulant Thrombin-like Enzyme from the Venom of Bothrops Leucurus. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2007;146:565–575. doi: 10.1016/j.cbpa.2005.12.033. [DOI] [PubMed] [Google Scholar]
- 77.Fernandes de Oliveira L.M., Ullah A., Masood R., Zelanis A., Spencer P.J., Serrano S.M.T., Arni R.K. Rapid Purification of Serine Proteinases from Bothrops Alternatus and Bothrops Moojeni Venoms. Toxicon. 2013;76:282–290. doi: 10.1016/j.toxicon.2013.10.016. [DOI] [PubMed] [Google Scholar]
- 78.Vivas-Ruiz D.E., Sandoval G.A., Mendoza J., Inga R.R., Gontijo S., Richardson M., Eble J.A., Yarleque A., Sanchez E.F. Coagulant Thrombin-like Enzyme (Barnettobin) from Bothrops Barnetti Venom: Molecular Sequence Analysis of Its CDNA and Biochemical Properties. Biochimie. 2013;95:1476–1486. doi: 10.1016/j.biochi.2013.03.015. [DOI] [PubMed] [Google Scholar]
- 79.Vivas-Ruiz D.E., Sandoval G.A., Gonzalez-Kozlova E., Zarria-Romero J., Lazo F., Rodríguez E., Magalhães H.P.B., Chávez-Olortegui C., Oliveira L.S., Alvarenga V.G., et al. Fibrinogen-Clotting Enzyme, Pictobin, from Bothrops Pictus Snake Venom. Structural and Functional Characterization. Int. J. Biol. Macromol. 2020;153:779–795. doi: 10.1016/j.ijbiomac.2020.03.055. [DOI] [PubMed] [Google Scholar]
- 80.Amorim F., Menaldo D., Carone S., Silva T., Sartim M., De Pauw E., Quinton L., Sampaio S. New Insights on Moojase, a Thrombin-Like Serine Protease from Bothrops Moojeni Snake Venom. Toxins. 2018;10:500. doi: 10.3390/toxins10120500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Cañas C.A. Brainstem Ischemic Stroke after to Bothrops Atrox Snakebite. Toxicon. 2016;120:124–127. doi: 10.1016/j.toxicon.2016.08.005. [DOI] [PubMed] [Google Scholar]
- 82.Ayeboua L., Plumelle Y., Smadja D., Merle H., Thomas L., Donnio A. Occipital infarction revealed by quadranopsia following snakebite by bothrops lanceolatus. Am. J. Trop. Med. Hyg. 2005;73:583–585. doi: 10.4269/ajtmh.2005.73.583. [DOI] [PubMed] [Google Scholar]
- 83.Thomas L., Chausson N., Uzan J., Kaidomar S., Vignes R., Plumelle Y., Bucher B., Smadja D. Thrombotic Stroke Following Snake Bites by the “Fer-de-Lance” Bothrops Lanceolatus in Martinique despite Antivenom Treatment: A Report of Three Recent Cases. Toxicon. 2006;48:23–28. doi: 10.1016/j.toxicon.2006.04.007. [DOI] [PubMed] [Google Scholar]
- 84.Bucaretchi F., Pimenta M.M.B., Borrasca-Fernandes C.F., Prado C.C., Capitani E.M., Hyslop S. Thrombotic Microangiopathy Following Bothrops Jararaca Snakebite: Case Report. Clin. Toxicol. 2019;57:294–299. doi: 10.1080/15563650.2018.1514621. [DOI] [PubMed] [Google Scholar]
- 85.Cañas C.A., Vecino M.J., Posso-Osorio I. Atypical Hemolytic Uremic Syndrome in a Patient With Bothrops Asper Envenomation. Wilderness Env. Med. 2022;33:109–115. doi: 10.1016/j.wem.2021.08.010. [DOI] [PubMed] [Google Scholar]
- 86.Mota S.M.B., Albuquerque P.L.M.M., Silva Júnior G.B.d., Daher E.D.F. Thrombotic Microangiopathy Due to Bothrops Erythromelas: A Case Report in Northeast Brazil. Rev. Inst. Med. Trop. Sao Paulo. 2020;62:e53. doi: 10.1590/s1678-9946202062053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Noutsos T., Currie B.J., Wijewickrama E.S., Isbister G.K. Snakebite Associated Thrombotic Microangiopathy and Recommendations for Clinical Practice. Toxins. 2022;14:57. doi: 10.3390/toxins14010057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Senise L.V., Yamashita K.M., Santoro M.L. Bothrops Jararaca Envenomation: Pathogenesis of Hemostatic Disturbances and Intravascular Hemolysis. Exp. Biol. Med. 2015;240:1528–1536. doi: 10.1177/1535370215590818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sartim M.A., Cezarette G.N., Jacob-Ferreira A.L., Frantz F.G., Faccioli L.H., Sampaio S.V. Disseminated Intravascular Coagulation Caused by Moojenactivase, a Procoagulant Snake Venom Metalloprotease. Int. J. Biol. Macromol. 2017;103:1077–1086. doi: 10.1016/j.ijbiomac.2017.05.146. [DOI] [PubMed] [Google Scholar]
- 90.Thomazini C.M., Sachetto A.T., de Albuquerque C.Z., de Moura Mattaraia V.G., de Oliveira A.K., Serrano S.M., Lebrun I., Barbaro K.C., Santoro M.L. Involvement of von Willebrand Factor and Botrocetin in the Thrombocytopenia Induced by Bothrops Jararaca Snake Venom. PLoS Negl. Trop. Dis. 2021;15:e0009715. doi: 10.1371/journal.pntd.0009715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Cedzyński M., Thielens N.M., Mollnes T.E., Vorup-Jensen T. Editorial: The Role of Complement in Health and Disease. Front. Immunol. 2019;10:1869. doi: 10.3389/fimmu.2019.01869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Pidde-Queiroz G., de Fátima Furtado M., Filgueiras C.F., Pessoa L.A., Spadafora-Ferreira M., Van den Berg C.W., Tambourgi D.V. Human Complement Activation and Anaphylatoxins Generation Induced by Snake Venom Toxins from Bothrops Genus. Mol. Immunol. 2010;47:2537–2544. doi: 10.1016/j.molimm.2010.07.003. [DOI] [PubMed] [Google Scholar]
- 93.Delafontaine M., Villas-Boas I.M., Pidde G., van den Berg C.W., Mathieu L., Blomet J., Tambourgi D.V. Venom from Bothrops Lanceolatus, a Snake Species Native to Martinique, Potently Activates the Complement System. J. Immunol. Res. 2018;2018:3462136. doi: 10.1155/2018/3462136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Berends E.T.M., Kuipers A., Ravesloot M.M., Urbanus R.T., Rooijakkers S.H.M. Bacteria under Stress by Complement and Coagulation. FEMS Microbiol. Rev. 2014;38:1146–1171. doi: 10.1111/1574-6976.12080. [DOI] [PubMed] [Google Scholar]
- 95.Morgan B.P. The Membrane Attack Complex as an Inflammatory Trigger. Immunobiology. 2016;221:747–751. doi: 10.1016/j.imbio.2015.04.006. [DOI] [PubMed] [Google Scholar]
- 96.Joiner K.A., Brown E.J., Frank M.M. Complement and Bacteria: Chemistry and Biology in Host Defense. Annu. Rev. Immunol. 1984;2:461–492. doi: 10.1146/annurev.iy.02.040184.002333. [DOI] [PubMed] [Google Scholar]
- 97.Palmer L.J., Damgaard C., Holmstrup P., Nielsen C.H. Influence of Complement on Neutrophil Extracellular Trap Release Induced by Bacteria. J. Periodontal Res. 2016;51:70–76. doi: 10.1111/jre.12284. [DOI] [PubMed] [Google Scholar]
- 98.Lehman H.K., Segal B.H. The Role of Neutrophils in Host Defense and Disease. J. Allergy Clin. Immunol. 2020;145:1535–1544. doi: 10.1016/j.jaci.2020.02.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Beavers W.N., Skaar E.P. Neutrophil-Generated Oxidative Stress and Protein Damage in Staphylococcus Aureus. Pathog. Dis. 2016;74:ftw060. doi: 10.1093/femspd/ftw060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Manda-Handzlik A., Demkow U. Neutrophils: The Role of Oxidative and Nitrosative Stress in Health and Disease. Pulm. Infect. 2015;857:51–60. doi: 10.1007/5584_2015_117. [DOI] [PubMed] [Google Scholar]
- 101.Mortaz E., Alipoor S.D., Adcock I.M., Mumby S., Koenderman L. Update on Neutrophil Function in Severe Inflammation. Front. Immunol. 2018;9:2171. doi: 10.3389/fimmu.2018.02171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Ravindran M., Khan M.A., Palaniyar N. Neutrophil Extracellular Trap Formation: Physiology, Pathology, and Pharmacology. Biomolecules. 2019;9:365. doi: 10.3390/biom9080365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Rosales C. Neutrophil: A Cell with Many Roles in Inflammation or Several Cell Types? Front. Physiol. 2018;9:113. doi: 10.3389/fphys.2018.00113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Vorobjeva N.V., Chernyak B.V. NETosis: Molecular Mechanisms, Role in Physiology and Pathology. Biochemistry. 2020;85:1178–1190. doi: 10.1134/S0006297920100065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Wang J. Neutrophils in Tissue Injury and Repair. Cell Tissue Res. 2018;371:531–539. doi: 10.1007/s00441-017-2785-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Flores C.A., Zappellini A., Prado-Franceschi J. Lipoxygenase-Derived Mediators May Be Involved in in Vivo Neutrophil Migration Induced by Bothrops Erythromelas and Bothrops Alternatus Venoms. Toxicon. 1993;31:1551–1559. doi: 10.1016/0041-0101(93)90339-K. [DOI] [PubMed] [Google Scholar]
- 107.Echeverría S., Leiguez E., Guijas C., do Nascimento N.G., Acosta O., Teixeira C., Leiva L.C., Rodríguez J.P. Evaluation of Pro-Inflammatory Events Induced by Bothrops Alternatus Snake Venom. Chem. Biol. Interact. 2018;281:24–31. doi: 10.1016/j.cbi.2017.12.022. [DOI] [PubMed] [Google Scholar]
- 108.Farsky S.H.P., Gonçalves L.R.C., Gutiérrez J.M., Correa A.P., Rucavado A., Gasque P., Tambourgi D.V. Bothrops Asper Snake Venom and Its Metalloproteinase BaP–1 Activate the Complement System. Role in Leucocyte Recruitment. Mediat. Inflamm. 2000;9:213–221. doi: 10.1080/09629350020025728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Zamuner S.R., Teixeira C.F.P. Cell Adhesion Molecules Involved in the Leukocyte Recruitment Induced by Venom of the Snake Bothrops Jararaca. Mediat. Inflamm. 2002;11:351–357. doi: 10.1080/0962935021000051548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Escocard R.D., Kanashiro M.M., Petretski J.H., Azevedo-Silva J., de Carvalho E.C., da Silva W.D., Kipnis T.L. Neutrophils Regulate the Expression of Cytokines, Chemokines and Nitric Oxide Synthase/Nitric Oxide in Mice Injected with Bothrops Atrox Venom. Immunobiology. 2006;211:37–46. doi: 10.1016/j.imbio.2005.08.003. [DOI] [PubMed] [Google Scholar]
- 111.Zamuner S.R., Gutiérrez J.M., Muscará M.N., Teixeira S.A., Teixeira C.F.P. Bothrops Asper and Bothrops Jararaca Snake Venoms Trigger Microbicidal Functions of Peritoneal Leukocytes in Vivo. Toxicon. 2001;39:1505–1513. doi: 10.1016/S0041-0101(01)00123-4. [DOI] [PubMed] [Google Scholar]
- 112.Wanderley C.W.S., Silva C.M.S., Wong D.V.T., Ximenes R.M., Morelo D.F.C., Cosker F., Aragão K.S., Fernandes C., Palheta-Júnior R.C., Havt A., et al. Bothrops Jararacussu Snake Venom-Induces a Local Inflammatory Response in a Prostanoid- and Neutrophil-Dependent Manner. Toxicon. 2014;90:134–147. doi: 10.1016/j.toxicon.2014.08.001. [DOI] [PubMed] [Google Scholar]
- 113.Arruda V.A., de Queiroz Guimarães A., Hyslop S., de Araújo P.M., Bon C., de Araújo A.L. de Bothrops Lanceolatus (Fer de Lance) Venom Stimulates Leukocyte Migration into the Peritoneal Cavity of Mice. Toxicon. 2003;41:99–107. doi: 10.1016/S0041-0101(02)00238-6. [DOI] [PubMed] [Google Scholar]
- 114.Porto B.N., Telli C.A., Dutra T.P., Alves L.S., Bozza M.T., Fin C.A., Thiesen F.V., Renner M.F. Biochemical and Biological Characterization of the Venoms of Bothriopsis Bilineata and Bothriopsis Taeniata (Serpentes: Viperidae) Toxicon. 2007;50:270–277. doi: 10.1016/j.toxicon.2007.03.020. [DOI] [PubMed] [Google Scholar]
- 115.Guan W., Ni Z., Hu Y., Liang W., Ou C., He J., Liu L., Shan H., Lei C., Hui D.S.C., et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Chaudhary R., Garg J., Houghton D.E., Murad M.H., Kondur A., Chaudhary R., Wysokinski W.E., McBane R.D. Thromboinflammatory Biomarkers in COVID-19: Systematic Review and Meta-Analysis of 17,052 Patients. Mayo Clin Proc. Innov. Qual. Outcomes. 2021;5:388–402. doi: 10.1016/j.mayocpiqo.2021.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Karki R., Kanneganti T.-D. The ‘Cytokine Storm’: Molecular Mechanisms and Therapeutic Prospects. Trends Immunol. 2021;42:681–705. doi: 10.1016/j.it.2021.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Wang J., Jiang M., Chen X., Montaner L.J. Cytokine Storm and Leukocyte Changes in Mild versus Severe SARS-CoV-2 Infection: Review of 3939 COVID-19 Patients in China and Emerging Pathogenesis and Therapy Concepts. J. Leukoc. Biol. 2020;108:17–41. doi: 10.1002/JLB.3COVR0520-272R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Sun X., Wang T., Cai D., Hu Z., Chen J., Liao H., Zhi L., Wei H., Zhang Z., Qiu Y., et al. Cytokine Storm Intervention in the Early Stages of COVID-19 Pneumonia. Cytokine Growth Factor Rev. 2020;53:38–42. doi: 10.1016/j.cytogfr.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Azkur A.K., Akdis M., Azkur D., Sokolowska M., Veen W., Brüggen M., O’Mahony L., Gao Y., Nadeau K., Akdis C.A. Immune Response to SARS-CoV-2 and Mechanisms of Immunopathological Changes in COVID-19. Allergy. 2020;75:1564–1581. doi: 10.1111/all.14364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Gillot C., Favresse J., Mullier F., Lecompte T., Dogné J.-M., Douxfils J. NETosis and the Immune System in COVID-19: Mechanisms and Potential Treatments. Front. Pharmacol. 2021;12:708302. doi: 10.3389/fphar.2021.708302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Fajgenbaum D.C., June C.H. Cytokine Storm. N. Engl. J. Med. 2020;383:2255–2273. doi: 10.1056/NEJMra2026131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Luna K., da Silva M., Pereira V. Clinical and Immunological Aspects of Envenomations by Bothrops Snakes. J. Venom. Anim. Toxins Incl. Trop. Dis. 2011;17:130–141. doi: 10.1590/S1678-91992011000200003. [DOI] [Google Scholar]
- 124.Júnior F.N., Jorge A.R., Marinho A.D., Silveira J.D., Alves N.T., Costa P.H., Chaves-Filho A.J., Lima D.B., Sampaio T.L., de Morais G.B., et al. Bothrops Alternatus Snake Venom Induces Cytokine Expression and Oxidative Stress on Renal Function. Curr. Top. Med. Chem. 2019;19:2058–2068. doi: 10.2174/1568026619666190809100319. [DOI] [PubMed] [Google Scholar]
- 125.Cedro R.C.A., Menaldo D.L., Costa T.R., Zoccal K.F., Sartim M.A., Santos-Filho N.A., Faccioli L.H., Sampaio S.V. Cytotoxic and Inflammatory Potential of a Phospholipase A2 from Bothrops Jararaca Snake Venom. J. Venom. Anim. Toxins Incl. Trop. Dis. 2018;24:33. doi: 10.1186/s40409-018-0170-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Zuliani J.P., Fernandes C.M., Zamuner S.R., Gutiérrez J.M., Teixeira C.F.P. Inflammatory Events Induced by Lys-49 and Asp-49 Phospholipases A2 Isolated from Bothrops Asper Snake Venom: Role of Catalytic Activity. Toxicon. 2005;45:335–346. doi: 10.1016/j.toxicon.2004.11.004. [DOI] [PubMed] [Google Scholar]
- 127.Maria Fernandes C., Regina Zamuner S., Pavan Zuliani J., Rucavado A., Maria Gutiérrez J., de Fátima Pereira Teixeira C. Inflammatory Effects of BaP1 a Metalloproteinase Isolated from Bothrops Asper Snake Venom: Leukocyte Recruitment and Release of Cytokines. Toxicon. 2006;47:549–559. doi: 10.1016/j.toxicon.2006.01.009. [DOI] [PubMed] [Google Scholar]
- 128.Rucavado A., Escalante T., Teixeira C.F.P., Fernándes C.M., Díaz C., Gutiérrez J.M. Increments in Cytokines and Matrix Metalloproteinases in Skeletal Muscle after Injection of Tissue-Damaging Toxins from the Venom of the Snake Bothrops Asper. Mediat. Inflamm. 2002;11:121–128. doi: 10.1080/09629350220131980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Moreira V., Dos-Santos M.C., Nascimento N.G., da Silva H.B., Fernandes C.M., D’Império Lima M.R., Teixeira C. Local Inflammatory Events Induced by Bothrops Atrox Snake Venom and the Release of Distinct Classes of Inflammatory Mediators. Toxicon. 2012;60:12–20. doi: 10.1016/j.toxicon.2012.03.004. [DOI] [PubMed] [Google Scholar]
- 130.Luna K.P., Melo C.M., Pascoal V.P., Martins Filho O.A., Pereira V.R. Bothrops Erythromelas Snake Venom Induces a Proinflammatory Response in Mice Splenocytes. Int. J. Interferon Cytokine Mediat. Res. 2011;9:9–18. doi: 10.2147/IJICMR.S14331. [DOI] [Google Scholar]
- 131.Delafontaine M., Villas-Boas I., Mathieu L., Josset P., Blomet J., Tambourgi D. Enzymatic and Pro-Inflammatory Activities of Bothrops Lanceolatus Venom: Relevance for Envenomation. Toxins. 2017;9:244. doi: 10.3390/toxins9080244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Nadur-Andrade N., Dale C.S., Oliveira V.R., Toniolo E.F., Feliciano R.D., da Silva J.A., Jr., Zamuner S.R. Analgesic Effect of Photobiomodulation on Bothrops Moojeni Venom-Induced Hyperalgesia: A Mechanism Dependent on Neuronal Inhibition, Cytokines and Kinin Receptors Modulation. PLoS Negl. Trop. Dis. 2016;10:e0004998. doi: 10.1371/journal.pntd.0004998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Soares F.G.S., Ibiapina H.N., Sartim M.A., Mendonça-da-Silva I., Nascimento E.F., Ferreira L.C.L., Cerni F.A., Malheiro A., Pucca M.B., Wen F.H., et al. Lower Levels of CXCL-8 and IL-2 on Admission as Predictors of Early Adverse Reactions to Bothrops Antivenom in the Brazilian Amazon. Cytokine. 2022;152:155825. doi: 10.1016/j.cyto.2022.155825. [DOI] [PubMed] [Google Scholar]
- 134.Lee D.W., Gardner R., Porter D.L., Louis C.U., Ahmed N., Jensen M., Grupp S.A., Mackall C.L. Current Concepts in the Diagnosis and Management of Cytokine Release Syndrome. Blood. 2014;124:188–195. doi: 10.1182/blood-2014-05-552729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Templin C., Ghadri J.R., Diekmann J., Napp L.C., Bataiosu D.R., Jaguszewski M., Cammann V.L., Sarcon A., Geyer V., Neumann C.A., et al. Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N. Engl. J. Med. 2015;373:929–938. doi: 10.1056/NEJMoa1406761. [DOI] [PubMed] [Google Scholar]
- 136.Burkard P., Vögtle T., Nieswandt B. Platelets in Thrombo-Inflammation: Concepts, Mechanisms, and Therapeutic Strategies for Ischemic Stroke. Hamostaseologie. 2020;40:153–164. doi: 10.1055/a-1151-9519. [DOI] [PubMed] [Google Scholar]
- 137.Periayah M.H., Halim A.S., Zaharil A., Saad M. Mechanism Action of Platelets and Crucial Blood Coagulation Pathways in Hemostasis. Int. J. Hematol.-Oncol. Stem Cell Res. 2017;11:319. [PMC free article] [PubMed] [Google Scholar]
- 138.Andersen H., Greenberg D.L., Fujikawa K., Xu W., Chung D.W., Davie E.W. Protease-Activated Receptor 1 Is the Primary Mediator of Thrombin-Stimulated Platelet Procoagulant Activity. Proc. Natl. Acad. Sci. USA. 1999;96:11189–11193. doi: 10.1073/pnas.96.20.11189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Heuberger D.M., Schuepbach R.A. Protease-Activated Receptors (PARs): Mechanisms of Action and Potential Therapeutic Modulators in PAR-Driven Inflammatory Diseases. Thromb. J. 2019;17:4. doi: 10.1186/s12959-019-0194-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Clemetson K.J., Clemetson J.M. Platelets. Elsevier; Amsterdam, The Netherlands: 2019. Platelet Receptors; pp. 169–192. [Google Scholar]
- 141.Cameron-Vendrig A., Reheman A., Siraj M.A., Xu X.R., Wang Y., Lei X., Afroze T., Shikatani E., El-Mounayri O., Noyan H., et al. Glucagon-Like Peptide 1 Receptor Activation Attenuates Platelet Aggregation and Thrombosis. Diabetes. 2016;65:1714–1723. doi: 10.2337/db15-1141. [DOI] [PubMed] [Google Scholar]
- 142.Margraf A., Zarbock A. Platelets in Inflammation and Resolution. J. Immunol. 2019;203:2357–2367. doi: 10.4049/jimmunol.1900899. [DOI] [PubMed] [Google Scholar]
- 143.Mezger M., Nording H., Sauter R., Graf T., Heim C., von Bubnoff N., Ensminger S.M., Langer H.F. Platelets and Immune Responses During Thromboinflammation. Front. Immunol. 2019;10:1731. doi: 10.3389/fimmu.2019.01731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Xu X.R., Zhang D., Oswald B.E., Carrim N., Wang X., Hou Y., Zhang Q., Lavalle C., McKeown T., Marshall A.H., et al. Platelets Are Versatile Cells: New Discoveries in Hemostasis, Thrombosis, Immune Responses, Tumor Metastasis and Beyond. Crit. Rev. Clin. Lab. Sci. 2016;53:409–430. doi: 10.1080/10408363.2016.1200008. [DOI] [PubMed] [Google Scholar]
- 145.Arbesu I., Bucsaiova M., Fischer M.B., Mannhalter C. Platelet-Borne Complement Proteins and Their Role in Platelet-Bacteria Interactions. J. Thromb. Haemost. 2016;14:2241–2252. doi: 10.1111/jth.13495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Kozarcanin H., Lood C., Munthe-Fog L., Sandholm K., Hamad O.A., Bengtsson A.A., Skjoedt M.-O., Huber-Lang M., Garred P., Ekdahl K.N., et al. The Lectin Complement Pathway Serine Proteases (MASPs) Represent a Possible Crossroad between the Coagulation and Complement Systems in Thromboinflammation. J. Thromb. Haemost. 2016;14:531–545. doi: 10.1111/jth.13208. [DOI] [PubMed] [Google Scholar]
- 147.Hamad O.A., Ekdahl K.N., Nilsson P.H., Andersson J., Magotti P., Lambris J.D., Nilsson B. Complement Activation Triggered by Chondroitin Sulfate Released by Thrombin Receptor-Activated Platelets. J. Thromb. Haemost. 2008;6:1413–1421. doi: 10.1111/j.1538-7836.2008.03034.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.del Conde I., Crúz M.A., Zhang H., López J.A., Afshar-Kharghan V. Platelet Activation Leads to Activation and Propagation of the Complement System. J. Exp. Med. 2005;201:871–879. doi: 10.1084/jem.20041497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Peerschke E.I.B., Ghebrehiwet B. Platelet Receptors for the Complement Component C1q: Implications for Hemostasis and Thrombosis. Immunobiology. 1998;199:239–249. doi: 10.1016/S0171-2985(98)80030-2. [DOI] [PubMed] [Google Scholar]
- 150.Peerschke E.I.B., Yin W., Grigg S.E., Ghebrehiwet B. Blood Platelets Activate the Classical Pathway of Human Complement. J. Thromb. Haemost. 2006;4:2035–2042. doi: 10.1111/j.1538-7836.2006.02065.x. [DOI] [PubMed] [Google Scholar]
- 151.Zuchtriegel G., Uhl B., Puhr-Westerheide D., Pörnbacher M., Lauber K., Krombach F., Reichel C.A. Platelets Guide Leukocytes to Their Sites of Extravasation. PLoS Biol. 2016;14:e1002459. doi: 10.1371/journal.pbio.1002459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Sreeramkumar V., Adrover J.M., Ballesteros I., Cuartero M.I., Rossaint J., Bilbao I., Nácher M., Pitaval C., Radovanovic I., Fukui Y., et al. Neutrophils Scan for Activated Platelets to Initiate Inflammation. Science (1979) 2014;346:1234–1238. doi: 10.1126/science.1256478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Wang Y., Gao H., Shi C., Erhardt P.W., Pavlovsky A., Soloviev D.A., Bledzka K., Ustinov V., Zhu L., Qin J., et al. Leukocyte Integrin Mac-1 Regulates Thrombosis via Interaction with Platelet GPIbα. Nat. Commun. 2017;8:15559. doi: 10.1038/ncomms15559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Yamashita K.M., Nogueira T.O., Senise L.V., Cirillo M.C., Gonçalves L.R.C., Sano-Martins I.S., Giorgi R., Santoro M.L. Involvement of Circulating Platelets on the Hyperalgesic Response Evoked by Carrageenan and Bothrops Jararaca Snake Venom. J. Thromb. Haemost. 2011;9:2057–2066. doi: 10.1111/j.1538-7836.2011.04449.x. [DOI] [PubMed] [Google Scholar]
- 155.Santoro M., Sano-Martins I. Platelet Dysfunction during Bothrops Jararaca Snake Envenomation in Rabbits. Thromb. Haemost. 2004;92:369–383. doi: 10.1160/TH04-02-0120. [DOI] [PubMed] [Google Scholar]
- 156.Santos-Filho N.A., Silveira L.B., Oliveira C.Z., Bernardes C.P., Menaldo D.L., Fuly A.L., Arantes E.C., Sampaio S.V., Mamede C.C.N., Beletti M.E., et al. A New Acidic Myotoxic, Anti-Platelet and Prostaglandin I2 Inductor Phospholipase A2 Isolated from Bothrops Moojeni Snake Venom. Toxicon. 2008;52:908–917. doi: 10.1016/j.toxicon.2008.08.020. [DOI] [PubMed] [Google Scholar]
- 157.Silveira L.B., Marchi-Salvador D.P., Santos-Filho N.A., Silva F.P., Marcussi S., Fuly A.L., Nomizo A., da Silva S.L., Stábeli R.G., Arantes E.C., et al. Isolation and Expression of a Hypotensive and Anti-Platelet Acidic Phospholipase A2 from Bothrops Moojeni Snake Venom. J. Pharm. Biomed. Anal. 2013;73:35–43. doi: 10.1016/j.jpba.2012.04.008. [DOI] [PubMed] [Google Scholar]
- 158.Andrião-Escarso S.H., Soares A.M., Fontes M.R.M., Fuly A.L., Corrêa F.M.A., Rosa J.C., Greene L.J., Giglio J.R. Structural and Functional Characterization of an Acidic Platelet Aggregation Inhibitor and Hypotensive Phospholipase A2 from Bothrops jararacussu Snake Venom. Biochem.Pharm. 2002;64:723–732. doi: 10.1016/S0006-2952(02)01210-8. [DOI] [PubMed] [Google Scholar]
- 159.Serrano S.M.T., Reichl A.P., Mentele R., Auerswald E.A., Santoro M.L., Sampaio C.A.M., Camargo A.C.M., Assakura M.T. A Novel Phospholipase A2, BJ-PLA2, from the Venom of the Snake Bothrops Jararaca: Purification, Primary Structure Analysis, and Its Characterization as a Platelet-Aggregation-Inhibiting Factor. Arch. Biochem. Biophys. 1999;367:26–32. doi: 10.1006/abbi.1999.1230. [DOI] [PubMed] [Google Scholar]
- 160.Ferreira F., Gomes M., de Souza D., Gimenes S., Castanheira L., Borges M., Rodrigues R., Yoneyama K., Brandeburgo M., Rodrigues V. Molecular Cloning and Pharmacological Properties of an Acidic PLA2 from Bothrops pauloensis Snake Venom. Toxins. 2013;5:2403–2419. doi: 10.3390/toxins5122403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Fuly A.L., Soares A.M., Marcussi S., Giglio J.R., Guimarães J.A. Signal Transduction Pathways Involved in the Platelet Aggregation Induced by a D-49 Phospholipase A2 Isolated from Bothrops jararacussu Snake Venom. Biochimie. 2004;86:731–739. doi: 10.1016/j.biochi.2004.07.001. [DOI] [PubMed] [Google Scholar]
- 162.Deluca M., Ward C.M., Ohmori K., Andrews R.K., Berndt M.C. Jararhagin and Jaracetin: Novel Snake Venom Inhibitors of the Integrin Collagen Receptor, A2β1. Biochem. Biophys. Res. Commun. 1995;206:570–576. doi: 10.1006/bbrc.1995.1081. [DOI] [PubMed] [Google Scholar]
- 163.Souza D.H.F., Iemma M.R.C., Ferreira L.L., Faria J.P., Oliva M.L.V., Zingali R.B., Niewiarowski S., Selistre-de-Araujo H.S. The Disintegrin-like Domain of the Snake Venom Metalloprotease Alternagin Inhibits A2β1 Integrin-Mediated Cell Adhesion. Arch. Biochem. Biophys. 2000;384:341–350. doi: 10.1006/abbi.2000.2120. [DOI] [PubMed] [Google Scholar]
- 164.Sanchez E.F., Richardson M., Gremski L.H., Veiga S.S., Yarleque A., Niland S., Lima A.M., Estevao-Costa M.I., Eble J.A. A Novel Fibrinolytic Metalloproteinase, Barnettlysin-I from Bothrops Barnetti (Barnett´s Pitviper) Snake Venom with Anti-Platelet Properties. Biochim. Et Biophys. Acta (BBA)–Gen. Subj. 2016;1860:542–556. doi: 10.1016/j.bbagen.2015.12.021. [DOI] [PubMed] [Google Scholar]
- 165.Sobrinho J.C., Kayano A.M., Simões-Silva R., Alfonso J.J., Gomez A.F., Gomez M.C.V., Zanchi F.B., Moura L.A., Souza V.R., Fuly A.L., et al. Anti-platelet aggregation activity of two novel acidic Asp49-phospholipases A2 from Bothrops brazili snake venom. Int. J. Biol. Macromol. 2018;107 Pt A:1014–1022. doi: 10.1016/j.ijbiomac.2017.09.069. [DOI] [PubMed] [Google Scholar]
- 166.Dias E.H.V., dos Santos Paschoal T., da Silva A.P., da Cunha Pereira D.F., de Sousa Simamoto B.B., Matias M.S., Santiago F.M., Rosa J.C., Soares A., Santos-Filho N.A., et al. BaltPLA2: A New Phospholipase A2 from Bothrops alternatus Snake Venom with Antiplatelet Aggregation Activity. Protein Pept. Lett. 2018;25:943–952. doi: 10.2174/0929866525666181004101622. [DOI] [PubMed] [Google Scholar]
- 167.Menaldo D.L., Bernardes C.P., Santos-Filho N.A., de Andrade Moura L., Fuly A.L., Arantes E.C., Sampaio S.V. Biochemical Characterization and Comparative Analysis of Two Distinct Serine Proteases from Bothrops pirajai Snake Venom. Biochimie. 2012;94:2545–2558. doi: 10.1016/j.biochi.2012.07.007. [DOI] [PubMed] [Google Scholar]
- 168.Vilca-Quispe A., Ponce-Soto L.A., Winck F.V., Marangoni S. Isolation and Characterization of a New Serine Protease with Thrombin-like Activity (TLBm) from the Venom of the Snake Bothrops marajoensis. Toxicon. 2010;55:745–753. doi: 10.1016/j.toxicon.2009.11.006. [DOI] [PubMed] [Google Scholar]
- 169.Santos B.F., Serrano S.M.T., Kuliopulos A., Niewiarowski S. Interaction of Viper Venom Serine Peptidases with Thrombin Receptors on Human Platelets. FEBS Lett. 2000;477:199–202. doi: 10.1016/S0014-5793(00)01803-2. [DOI] [PubMed] [Google Scholar]
- 170.Marrakchi N., Barbouche R., Guermazi S., Karoui H., Bon C., el Ayeb M. Cerastotin, a Serine Protease from Cerastes Cerastes Venom, with Platelet-Aggregating and Agglutinating Properties. Eur. J. Biochem. 1997;247:121–128. doi: 10.1111/j.1432-1033.1997.00121.x. [DOI] [PubMed] [Google Scholar]
- 171.Nishida S., Fujimura Y., Miura S., Yoshida E., Sugimoto M., Yoshioka A., Fukui H., Ozaki Y., Usami Y. Purification and Characterization of Bothrombin, a Fibrinogen-Clotting Serine Protease from the Venom of Bothrops Jararaca. Biochemistry. 1994;33:1843–1849. doi: 10.1021/bi00173a030. [DOI] [PubMed] [Google Scholar]
- 172.de Queiroz M.R., Mamede C.C.N., Fonseca K.C., de Morais N.C.G., de Sousa B.B., Santos-Filho N.A., Beletti M.E., Arantes E.C., Stanziola L., de Oliveira F. Rapid Purification of a New P-I Class Metalloproteinase from Bothrops Moojeni Venom with Antiplatelet Activity. Biomed.Res. Int. 2014;2014:352420. doi: 10.1155/2014/352420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Tanjoni I., Butera D., Bento L., Della-Casa M.S., Marques-Porto R., Takehara H.A., Gutiérrez J.M., Fernandes I., Moura-da-Silva A.M. Snake Venom Metalloproteinases: Structure/Function Relationships Studies Using Monoclonal Antibodies. Toxicon. 2003;42:801–808. doi: 10.1016/j.toxicon.2003.10.010. [DOI] [PubMed] [Google Scholar]
- 174.Sousa B.B., Mamede C.C., Matias M.S., Pereira D.F., Queiroz M.R., Dias E.H., Silva A.C., Dantas N.O., Costa J.D., Oliveira F.D. A New Platelet-Aggregation-Inhibiting Factor Isolated from Bothrops moojeni Snake Venom. Biomed. Res. Int. 2017;2017:4315832. doi: 10.1155/2017/4315832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Matias M.S., de Sousa B.B., da Cunha Pereira D.F., Dias E.H.V., Mamede C.C.N., de Queiroz M.R., Silva A.C.A., Dantas N.O., Soares A.M., de Oliveira Costa J., et al. BaltDC: Purification, Characterization and Infrared Spectroscopy of an Antiplatelet DC Protein Isolated from Bothrops Alternatus Snake Venom. J. Venom. Anim. Toxins Incl. Trop. Dis. 2017;23:36. doi: 10.1186/s40409-017-0126-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Sartim M.A., Costa T.R., Laure H.J., Espíndola M.S., Frantz F.G., Sorgi C.A., Cintra A.C.O., Arantes E.C., Faccioli L.H., Rosa J.C., et al. Moojenactivase, a Novel pro-Coagulant PIIId Metalloprotease Isolated from Bothrops moojeni Snake Venom, Activates Coagulation Factors II and X and Induces Tissue Factor up-Regulation in Leukocytes. Arch. Toxicol. 2016;90:1261–1278. doi: 10.1007/s00204-015-1533-6. [DOI] [PubMed] [Google Scholar]
- 177.Oliveira L.S., Estevão-Costa M.I., Alvarenga V.G., Vivas-Ruiz D.E., Yarleque A., Lima A.M., Cavaco A., Eble J.A., Sanchez E.F. Atroxlysin-III, A Metalloproteinase from the Venom of the Peruvian Pit Viper Snake Bothrops atrox (Jergón) Induces Glycoprotein VI Shedding and Impairs Platelet Function. Molecule. 2019;26:3489. doi: 10.3390/molecules24193489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Suntravat M., Helmke T.J., Atphaisit C., Cuevas E., Lucena S.E., Uzcátegui N.L., Sánchez E.E., Rodriguez-Acosta A. Expression, Purification, and Analysis of Three Recombinant ECD Disintegrins (r-Colombistatins) from P-III Class Snake Venom Metalloproteinases Affecting Platelet Aggregation and SK-MEL-28 Cell Adhesion. Toxicon. 2016;122:43–49. doi: 10.1016/j.toxicon.2016.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Izidoro L.F.M., Ribeiro M.C., Souza G.R.L., Sant’Ana C.D., Hamaguchi A., Homsi-Brandeburgo M.I., Goulart L.R., Beleboni R.O., Nomizo A., Sampaio S.V., et al. Biochemical and Functional Characterization of an L-Amino Acid Oxidase Isolated from Bothrops pirajai Snake Venom. Bioorg. Med. Chem. 2006;14:7034–7043. doi: 10.1016/j.bmc.2006.06.025. [DOI] [PubMed] [Google Scholar]
- 180.Rodrigues R.S., da Silva J.F., Boldrini França J., Fonseca F.P.P., Otaviano A.R., Henrique Silva F., Hamaguchi A., Magro A.J., Braz A.S.K., dos Santos J.I. Structural and Functional Properties of Bp-LAAO, a New l-Amino Acid Oxidase Isolated from Bothrops pauloensis Snake Venom. Biochimie. 2009;91:490–501. doi: 10.1016/j.biochi.2008.12.004. [DOI] [PubMed] [Google Scholar]
- 181.Stábeli R.G., Sant’Ana C.D., Ribeiro P.H., Costa T.R., Ticli F.K., Pires M.G., Nomizo A., Albuquerque S., Malta-Neto N.R., Marins M., et al. Cytotoxic L-Amino Acid Oxidase from Bothrops moojeni: Biochemical and Functional Characterization. Int. J. Biol.Macromol. 2007;41:132–140. doi: 10.1016/j.ijbiomac.2007.01.006. [DOI] [PubMed] [Google Scholar]
- 182.Naumann G.B., Silva L.F., Silva L., Faria G., Richardson M., Evangelista K., Kohlhoff M., Gontijo C.M.F., Navdaev A., de Rezende F.F., et al. Cytotoxicity and Inhibition of Platelet Aggregation Caused by an L-Amino Acid Oxidase from Bothrops leucurus Venom. Biochim. Et Biophys. Acta (BBA)–Gen. Subj. 2011;1810:683–694. doi: 10.1016/j.bbagen.2011.04.003. [DOI] [PubMed] [Google Scholar]
- 183.Read M., Smith S., Lamb M., Brinkhous K. Role of Botrocetin in Platelet Agglutination: Formation of an Activated Complex of Botrocetin and von Willebrand Factor. Blood. 1989;74:1031–1035. doi: 10.1182/blood.V74.3.1031.1031. [DOI] [PubMed] [Google Scholar]
- 184.da Cunha Pereira D.F., Ribeiro M.S., de Sousa Simamoto B.B., Dias E.H., de Oliveira Costa J., Santos-Filho N.A., Bordon K.D., Arantes E.C., Dantas N.O., Silva A.C., et al. Baltetin: A New C-Type Lectin-like Isolated from Bothrops alternatus Snake Venom Which Act as a Platelet Aggregation Inhibiting. J. Chromatogr. B. 2021;1173:122695. doi: 10.1016/j.jchromb.2021.122695. [DOI] [PubMed] [Google Scholar]
- 185.Ishibashi T., Kimura H., Shikama Y., Uchida T., Kariyone S., Hirano T., Kishimoto T., Takatsuki F., Akiyama Y. Interleukin-6 Is a Potent Thrombopoietic Factor in Vivo in Mice. Blood. 1989;74:1241–1244. doi: 10.1182/blood.V74.4.1241.1241. [DOI] [PubMed] [Google Scholar]
- 186.Asano S., Okano A., Ozawa K., Nakahata T., Ishibashi T., Koike K., Kimura H., Tanioka Y., Shibuya A., Hirano T. In Vivo Effects of Recombinant Human Interleukin-6 in Primates: Stimulated Production of Platelets. Blood. 1990;75:1602–1605. doi: 10.1182/blood.V75.8.1602.1602. [DOI] [PubMed] [Google Scholar]
- 187.Hill R.J., Warren M.K., Levin J. Stimulation of Thrombopoiesis in Mice by Human Recombinant Interleukin 6. J. Clin. Investig. 1990;85:1242–1247. doi: 10.1172/JCI114559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Petricevich V.L., Teixeira C.F.P., Tambourgi D.V., Gutiérrez J.M. Increments in Serum Cytokine and Nitric Oxide Levels in Mice Injected with Bothrops Asper and Bothrops Jararaca Snake Venoms. Toxicon. 2000;38:1253–1266. doi: 10.1016/S0041-0101(99)00227-5. [DOI] [PubMed] [Google Scholar]
- 189.Ávila-Agüero M.L., París M.M., Hu S., Peterson P.K., Gutiérrez J.M., Lomonte B., Faingezicht I. Systemic Cytokine Response in Children Bitten by Snakes in Costa Rica. Pediatr. Emerg. Care. 2001;17:425–429. doi: 10.1097/00006565-200112000-00006. [DOI] [PubMed] [Google Scholar]
- 190.Barraviera B., Lomonte B., Tarkowski A., Hanson L.Å., Meira D.A. acute-phase reactions, including cytokines, in patients bitten by Bothrops and Crotalus snakes in Brazil. J. Venom. Anim. Toxins. 1995;1:11–22. doi: 10.1590/S0104-79301995000100003. [DOI] [Google Scholar]
- 191.Foley J.H., Conway E.M. Cross Talk Pathways Between Coagulation and Inflammation. Circ. Res. 2016;118:1392–1408. doi: 10.1161/CIRCRESAHA.116.306853. [DOI] [PubMed] [Google Scholar]
- 192.Wellmann I.A.M., Ibiapina H.N.S., Sachett J.A.G., Sartim M.A., Silva I.M., Oliveira S.S., Tarragô A.M., Moura-da-Silva A.M., Lacerda M.V.G., Ferreira L.C., et al. Correlating Fibrinogen Consumption and Profiles of Inflammatory Molecules in Human Envenomation’s by Bothrops Atrox in the Brazilian Amazon. Front. Immunol. 2020;11:1874. doi: 10.3389/fimmu.2020.01874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Grover S.P., Mackman N. Tissue Factor. Arter. Thromb. Vasc. Biol. 2018;38:709–725. doi: 10.1161/ATVBAHA.117.309846. [DOI] [PubMed] [Google Scholar]
- 194.Zelaya H., Rothmeier A.S., Ruf W. Tissue Factor at the Crossroad of Coagulation and Cell Signaling. J. Thromb. Haemost. 2018;16:1941–1952. doi: 10.1111/jth.14246. [DOI] [PubMed] [Google Scholar]
- 195.Yamashita K.M., Alves A.F., Barbaro K.C., Santoro M.L. Bothrops Jararaca Venom Metalloproteinases Are Essential for Coagulopathy and Increase Plasma Tissue Factor Levels during Envenomation. PLoS Negl. Trop. Dis. 2014;8:e2814. doi: 10.1371/journal.pntd.0002814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Pereira A.L.M., Fritzen M., Faria F., Da Motta G., Chudzinski-Tavassi A.M. Releasing or Expression Modulating Mediator Involved in Hemostasis by Berythractivase and Jararhagin (SVMPs) Toxicon. 2006;47:788–796. doi: 10.1016/j.toxicon.2006.02.014. [DOI] [PubMed] [Google Scholar]
- 197.Silva M.B., Schattner M., Ramos C.R.R., Junqueira-De-Azevedo I.L.M., Guarnieri M.C., Lazzari M.A., Sampaio C.A.M., Pozner R.G., Ventura J.S., Ho P.L., et al. A Prothrombin Activator from Bothrops erythromelas (Jararaca-Da-Seca) Snake Venom: Characterization and Molecular Cloning. Biochem. J. 2003;369:129–139. doi: 10.1042/bj20020449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Cezarette G.N., Sartim M.A., Sampaio S.V. Inflammation and Coagulation Crosstalk Induced by BJcuL, a Galactose-Binding Lectin Isolated from Bothrops Jararacussu Snake Venom. Int. J. Biol. Macromol. 2020;144:296–304. doi: 10.1016/j.ijbiomac.2019.12.015. [DOI] [PubMed] [Google Scholar]
- 199.Conran N., De Paula E.V. Thromboinflammatory Mechanisms in Sickle Cell Disease–Challenging the Hemostatic Balance. Haematologica. 2020;105:2380–2390. doi: 10.3324/haematol.2019.239343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Cavalcante J.D., de Almeida D.E., Moraes M.S., Santos S.R., Pincinato P.M., Riciopo P.M., de Oliveira L.L., Monteiro W.M., Ferreira-Junior R.S. Challenges and Opportunities in Clinical Diagnostic Routine of Envenomation Using Blood Plasma Proteomics. Toxins. 2023;15:180. doi: 10.3390/toxins15030180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Kölm R., Schaller M., Roumenina L.T., Niemiec I., Kremer Hovinga J.A., Khanicheh E., Kaufmann B.A., Hopfer H., Trendelenburg M. Von Willebrand Factor Interacts with Surface-Bound C1q and Induces Platelet Rolling. J. Immunol. 2016;197:3669–3679. doi: 10.4049/jimmunol.1501876. [DOI] [PubMed] [Google Scholar]
- 202.Rinder C.S., Smith M.J., Rinder H.M., Cortright D.N., Brodbeck R.M., Krause J.E., Smith B.R. Leukocyte Effects of C5a-Receptor Blockade During Simulated Extracorporeal Circulation. Ann. Thorac. Surg. 2007;83:146–152. doi: 10.1016/j.athoracsur.2006.08.019. [DOI] [PubMed] [Google Scholar]
- 203.Lappegård K.T., Fung M., Bergseth G., Riesenfeld J., Mollnes T.E. Artificial Surface-Induced Cytokine Synthesis: Effect of Heparin Coating and Complement Inhibition. Ann. Thorac. Surg. 2004;78:38–44. doi: 10.1016/j.athoracsur.2004.02.005. [DOI] [PubMed] [Google Scholar]
- 204.Riedl M., Noone D.G., Khan M.A., Pluthero F.G., Kahr W.H.A., Palaniyar N., Licht C. Complement Activation Induces Neutrophil Adhesion and Neutrophil-Platelet Aggregate Formation on Vascular Endothelial Cells. Kidney Int. Rep. 2017;2:66–75. doi: 10.1016/j.ekir.2016.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Blatt A.Z., Pathan S., Ferreira V.P. Properdin: A Tightly Regulated Critical Inflammatory Modulator. Immunol. Rev. 2016;274:172–190. doi: 10.1111/imr.12466. [DOI] [PMC free article] [PubMed] [Google Scholar]