Abstract
Background
Chronic fatigue syndrome (CFS) is a disabling illness of unknown aetiology. Disruption of gut microbiota may play a role in several neurological disorders. In this study, the effect of faecal microbiota transplantation (FMT) on fatigue severity and health-related quality of life (HRQOL) in patients with CFS was evaluated.
Methods
Randomized, placebo-controlled pilot trial. Patients and researchers were blinded to treatment assignment. 11 patients with CFS (10 female and 1 male, mean age 42.2 years and mean duration of CFS 6.3 years) were randomly assigned to receive either FMT from a universal donor (n = 5) or autologous FMT (n = 6) via colonoscopy. Patients’ HRQOL was assessed by using visual analog scale (VAS) and self-reporting questionnaires Modified Fatigue Impact Scale (MFIS), 15D and EQ-5D-3L. Patients’ HRQOL was evaluated at baseline, and 1 and 6 months after the FMT.
Results
The baseline VAS scores in the FMT and placebo groups were 62.4 and 76.0 (p = 0.29). 1-month scores were 60.0 and 73.7 and 6-months scores 72.8 and 69.5, respectively. Total MFIS scores in the FMT and placebo groups were 59.6 and 61.0 at the baseline (p = 0.80), 53.5 and 62.0 at 1 month and 58.6 and 56.2 at 6 months. Compared to the baseline scores, differences at 1 and 6 months were statistically insignificant both in VAS and in MFIS. The 15D and EQ-5D-3L profiles did not change after the FMT or placebo. FMT-related adverse events were not reported.
Conclusion
FMT was safe but did not relieve symptoms or improve the HRQOL of patients with CFS. Small number of study subjects limits the generalizability of these results.
Trial Registration ClinicalTrials.gov Identifier NCT04158427, https://register.clinicaltrials.gov, date of registration 08/08/2019.
Keywords: Chronic fatigue syndrome, Health-related quality of life, Faecal microbiota transplantation
Background
Chronic fatigue syndrome (CFS) is a complex and disabling illness of unknown etiology and a heterogenous constellation of symptoms [1]. Notably, these include profound and persistent fatigue, post-exertional malaise, neurocognitive impairment, autonomic dysfunction, recurrent flu-like symptoms and gastrointestinal (GI) disturbances [2]. Health related quality of life (HRQOL) among CFS patients has been found to be markedly lower than in the general population [3]. The underlying pathomechanism of CFS has not been established, and no diagnostic tests exist. Instead, diagnosis relies on symptom-specific criteria to identify cases of CFS after all relevant differential diagnoses have been excluded [4]. Up to 75% of CFS cases report an infection-like episode preceding the onset of their illness [5].
The human microbiota is an extensive community comprising collectively over 10,000 different microbial species including bacteria, viruses and archaea inhabiting various anatomical regions, especially gastrointestinal tract [6]. Naturally occurring symbiotic or commensal microbiota have co-evolved with the human host and have shown beneficial host interactions including involvement in mediating physiological processes necessary for metabolic and immune function, digestion and nutrition [5, 7, 8]. Composition of microbiota is distinct for each person. Each body region itself contains substantial amount of diversity, particularly the gut. Multiple factors affect this composition. Both internal factors, such as the genetic background of the host, and external environmental and lifestyle factors can also greatly influence the microbiota [8]. Disruption of the integrity or equilibrium of these intricate microbial networks has been implicated in numerous pathological conditions or exacerbation of disease [8, 9].
There is increasing agreement that the disruption of the gut microbiota impacts brain development, neurological outcomes, and disorders, resulting in long-term behavioral changes [10]. Hence, gut dysbiosis is reported to be associated with several neurological disorders, including Alzheimer’s [11], Parkinson’s [12], Huntington’s disease [13], and multiple sclerosis [14]. Evidence for immunological aberrations in CFS suggests that the underlying pathomechanism may be due to enteric dysbiosis [10]. The proposed mechanism describes an alteration in the mucosal barrier function of the gut, which subsequently becomes hyperpermeable and allows increased translocation of commensal bacteria and their components into the bloodstream, potentially triggering a systemic chronic inflammatory immune response [15].
Faecal microbiota transplantation (FMT) is an emerging treatment method to renew dysbiotic gut microbiota. It has well established efficacy and acknowledged position in the treatment of recurrent Clostridioides difficile infection (rCDI) [16]. The good results in rCDI have encouraged the science community to explore FMT also in many non-communicable gut dysbiosis associated diseases including several neurological conditions [10].
CSF patients tend to have severely impaired HRQOL and they often remain without sufficient treatment options. Thus, we aimed to investigate the efficacy of reshaping the gut microbiota in CSF patients via FMT to improve HRQOL.
Methods
The trial was approved by the ethical committee of Tampere University Hospital (identifier R18006) and written informed consent was obtained from all study participants.
Aim of the study: to evaluate the effect of FMT on HRQOL of patients with CFS.
Patients
Altogether 128 patients with chronic fatigue were initially evaluated by Tampere University Hospital (TaUH) multidisciplinary specialist team between Jan 1st 2016 and May 1st 2019. TaUH is a tertiary-care hospital providing care for 540,000 inhabitants in the region. In all patients, a standardized protocol was applied to determine the cause of symptoms. Psychiatric assessment consisted of clinical evaluation by a psychiatrist. Patient-reported Beck’s Depression Inventory (BDI), Alcohol Use Disorders Identification Test (AUDIT) questionnaires and neuropsychological test were utilized. A neurologist ruled out neurological diseases based on the patient’s history and clinical condition. Nocturnal polysomnography was conducted to rule out sleep-related disorders. A specialist in internal medicine clarified patient’s medical history and a medical examination was conducted. Pre-determined laboratory tests were acquired to rule out diseases and medical conditions, which could cause fatigue (Table 1), and additional tests were obtained on the clinician’s decision.
Table 1.
List of laboratory tests included in the evaluation of fatigue patients
| Complete blood count |
|---|
| Erythrocyte sedimentation rate |
| Sodium, potassium |
| Creatinine |
| Thyroid-stimulating hormone, thyroxine |
| Cortisol |
| Testosterone (males) |
| Ferritin, transferrin saturation |
| Calcium |
| Vitamin B-12, Vitamin D |
| Blood fasting glucose, HbA1c |
| Alanine transaminase (ALT), alkaline phosphatase (ALP) |
| Antinuclear antibodies (ANA) |
| Creatine kinase (CK) |
| Ammonium ion |
| Blood gases |
| Epstein-Barr virus antibody test |
| Cytomegalovirus antibody test |
| Borrelia antibody test |
| HI-virus antigen and antibody test |
| Urinalysis |
| Electrocardigram (ECG) |
In diagnosing CFS, the Institute of Medicine (IOM) criteria were applied [17]. The three required main symptoms were (1) a substantial reduction or impairment in the ability to engage in pre-illness levels of activity; (2) post-exertional malaise and (3) unrefreshing sleep. Moreover, at least one of two additional manifestations (cognitive impairment and orthostatic intolerance) was necessitated.
Of 128 patients, 29 patients were diagnosed with exclusively CFS and no other fatigue causing disease or condition and they were asked to enroll into the study. 16 patients refused to participate. The main reason was intense fatigue and inability to conduct the FMT procedure.13 patients agreed and provided signed written informed consent after being fully informed about the study protocol. Two patients withdrew their informed consent before randomization; one patient due to pregnancy and in one patient fatigue was too severe to accomplish the FMT. Eventually, 11 participants were enrolled into the study.
Intervention
The 11 participants were randomly assigned in a double-blind fashion in a 1:1 ratio to receive either FMT or placebo. Randomization and preparation of transplants was performed by a study nurse. Patients in the FMT group received faecal transplantation from an exclusively tested universal donor [18]. Transplants were stored in a stool bank at – 80 °C and thawed immediately before colonoscopy [19]. The placebo group received a freshly prepared, autologous transplantation, i.e. a transplant prepared from patients own faeces. All patients were treated via a single colonoscopy with 30 g of faecal material administered into the caecum. To ensure blinding, both groups provided their stool for the preparation of placebo, and if the patient was randomized to the FMT group, the stool sample was discarded. Bowel preparation was performed using polyethylene glycol solution. No pre-treatment with antibiotics was given. The patients were not given any restrictions regarding nutrition or medications during the follow-up.
Outcome
The prospectively defined main outcome measure was the change in fatigue scores assessed using visual analog scale and several validated self-reporting questionnaires. Patients’ HRQOL was evaluated at baseline, and 1 and 6 months after FMT.
Visual Analogue Scale (VAS): VAS measures fatigue intensity and consists of a 100 mm line, with two end points representing 0 (no fatigue) and 100 (extreme fatigue).
Modified Fatigue Impact Scale (MFIS): This instrument provides an assessment of the effects of fatigue in terms of physical, cognitive, and psychosocial functioning consisting of 21 items. Individual scores are generated by calculating the sum of specific sets of items.
15D: the 15D is a 15-dimensional instrument for measuring HRQOL among adult subjects. In each dimension, the 15D has five severity levels (1 = no problems, 5 = extreme problems/unable).
EQ-5D-3L: EQ-5D-3L consists of a descriptive system, which contains five items that each represent a health dimension, including mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. In each dimension, the EQ-5D-3L has three severity levels (no, some, extreme problems/unable).
Statistical analysis
Data are expressed as mean ± standard deviation (SD) unless otherwise stated. Patients’ characteristics were compared using the t-test for continuous variables.
Results
Of 11 patients, 10 were female. Patients’ mean age was 42.2 (± 6.7) years and the mean duration of CFS symptoms at randomization was 6.3 (± 2.5) years. After randomization into FMT or placebo, patients’ age or duration of CFS did not differ statistically significantly (Table 2). In all study subjects, colonoscopy was conducted without complications and no FMT-related adverse events were reported.
Table 2.
Patient characteristics
| FMT group (n = 5) | Placebo group (n = 6) | p value | |
|---|---|---|---|
| Female | 80% | 100% | |
| Mean age at randomization (years) (range) | 42,0 (32–49) | 42,4 (29–52) | 0,92 |
| Duration on CFS (years) (range) | 5,8 (5,0–6,7) | 6,6 (3,0–11,3) | 0,62 |
The average baseline VAS scores in the FMT and placebo groups were 62.4 (± 22.4) and 76.0 (± 3.6) (p = 0.29), respectively. Compared to the baseline scores, statistically significant changes at 1 or 6 months after the FMT/placebo were not found (Table 3.)
Table 3.
VAS scores in the FMT and placebo groups
| Baseline | 1 Month after the FMT | 6 Months after the FMT | p value (Baseline vs. 1 Month) | p value (Baseline vs. 6 Months) | |
|---|---|---|---|---|---|
| FMT group (n = 5) | 62.4 (22.4) | 60.0 (21.8) | 72.8 (13.6) | 0.88 | 0.45 |
| Placebo group (n = 6) | 76.0 (3.6) | 73.7 (4.9) | 69.5 (9.7) | 0.41 | 0.20 |
Visual Analogue Scale (VAS) scores (mean) (± SD) in the FMT and the placebo groups. 0 = no fatigue, 100 = extreme fatigue. The difference in baseline scores between the groups was statistically insignificant (p = 0.29)
The MFIS scores are shown in the Table 4. There were not statistically significant differences in physical, cognitive, psychosocial or total scores between the groups. Neither the subscale scores nor the total scores changed after the FMT/placebo procedure.
Table 4.
MFIS scores in the FMT and placebo groups
| Baseline | 1 Month after the FMT | 6 Months after the FMT | p value (Baseline vs. 1 Month) | p value (Baseline vs. 6 Months) | |
|---|---|---|---|---|---|
| FMT group (n = 5) | |||||
| Physical subscale | 29.4 (2,4) | 26.2 (2.8) | 27,4 (4,6) | 0.08 | 0.42 |
| Cognitive subscale | 24.8 (7.5) | 23.0 (8.0) | 25.4 (8.9) | 0.74 | 0.91 |
| Psychosocial subscale | 5.4 (1.1) | 5.5 (1.3) | 5.8 (1,5) | 0.91 | 0.64 |
| Total | 59.6 (9.2) | 53.5 (9.9) | 58.6 (11.9) | 0.45 | 0.79 |
| Placebo group (n = 6) | |||||
| Physical subscale | 30.0 (6,2) | 29.5 (4.8) | 25.7 (7.9) | 0.88 | 0.32 |
| Cognitive subscale | 24.8 (4.5) | 25.8 (8.2) | 25.0 (11,1) | 0.80 | 0.97 |
| Psychosocial subscale | 6.2 (1.0) | 6.7 (1.2) | 5.5 (2.3) | 0.45 | 0.54 |
| Total | 61.0 (8.3) | 62.0 (9.8) | 56.2 (20.3) | 0.85 | 0.61 |
Modified Fatigue Impact Scale (MFIS) scores (mean) (± SD) in the FMT and the placebo groups. Differences in the baseline scores between the groups were statistically insignificant (p values: Physical subscale 0.83; Cognitive subscale 0.99; Psychosocial subscale 0.27 and Total 0.80)
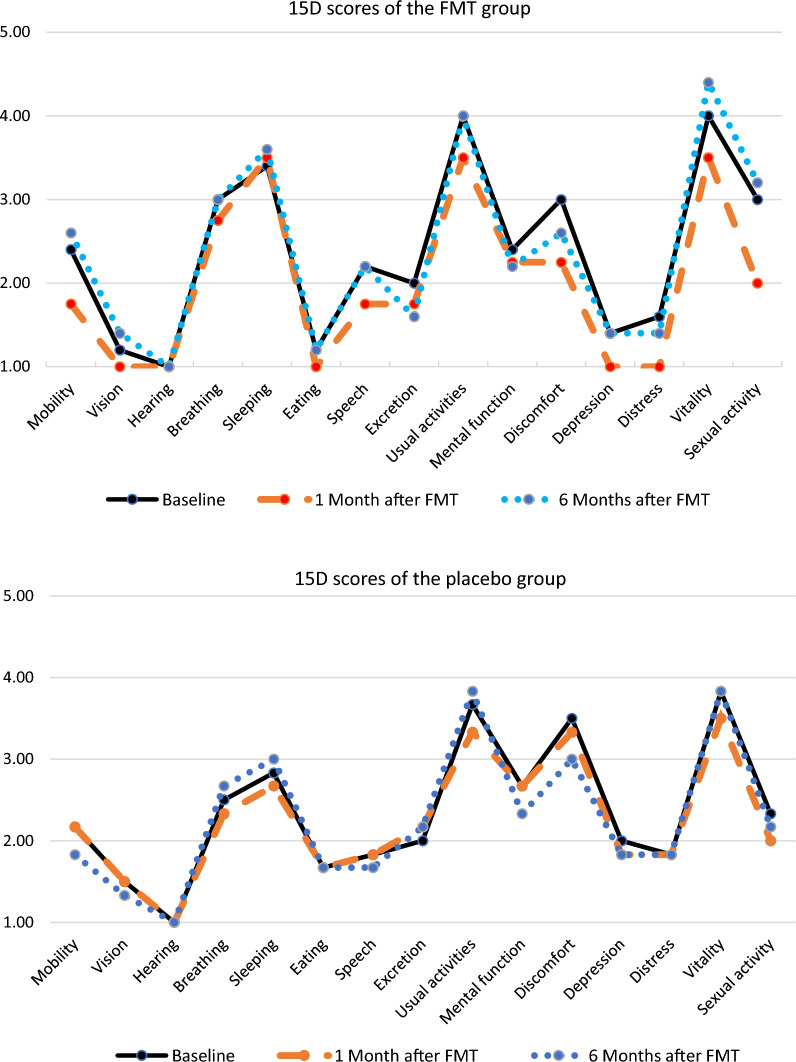
The 15D-profiles in the FMT and placebo groups are shown in the Fig. 1. In both groups, the highest scores reflecting severe reductions in HRQOL were found in dimensions assessing breathing, sleeping, usual activities, discomfort and vitality. After the FMT/placebo procedure, the profiles remained unchanged.
Fig. 1.
15D-scores in FMT and placebo groups. Severity levels in each dimension: 1 = no problems, 5 = extreme problems/unable. Compared to baseline scores, differences in 1 and 6 months scores are statistically non-significant
Table 5 shows EQ-5D-3L scores in the individual patients. There is some variety in the severity of symptoms between the subjects but there were no statistically significant changes in any dimension of single patient’s symptoms after the FMT/placebo. In the patient 3, 1 month data were not provided.
Table 5.
EQ-5D-3L scores in the FMT and placebo groups
| Mobility | Self-care | Usual activities | Pain/discomfort | Anxiety/depression | |
|---|---|---|---|---|---|
| FMT group | |||||
| Patient 1 | 1 1 1 | 1 1 1 | 2 2 2 | 2 1 2 | 1 1 1 |
| Patient 2 | 2 2 2 | 2 2 2 | 3 3 3 | 2 1 2 | 1 1 1 |
| Patient 3 | 3–3 | 2–2 | 3–3 | 3–3 | 2–1 |
| Patient 4 | 2 2 2 | 2 2 2 | 3 3 3 | 2 2 1 | 1 1 1 |
| Patient 5 | 2 2 2 | 1 1 1 | 3 2 3 | 2 2 2 | 1 1 1 |
| Placebo group | |||||
| Patient 6 | 2 2 2 | 2 2 2 | 2 2 2 | 3 2 3 | 2 2 2 |
| Patient 7 | 1 1 1 | 1 1 1 | 2 2 2 | 2 2 2 | 1 1 1 |
| Patient 8 | 2 2 1 | 2 1 1 | 3 3 2 | 3 3 2 | 2 2 2 |
| Patient 9 | 2 2 2 | 2 2 1 | 2 3 2 | 2 2 3 | 1 1 1 |
| Patient 10 | 2 2 2 | 1 1 1 | 2 3 2 | 2 2 3 | 2 2 2 |
| Patient 11 | 3 3 3 | 2 1 1 | 3 3 3 | 2 3 2 | 1 2 2 |
EQ-5D-3L profiles in individual patients. In each dimension, the first digit represents the severity of symptoms at baseline, the second and the third digits describe severity at 1 month and at 6 months after the FMT, respectively. 1 = no problems; 2 = some problems; 3 = extreme problems/unable. 1 month data of patient 3 missing
Discussion
To our knowledge, this is the first randomized study to evaluate the effect of FMT on the HRQOL of patients with CFS. In this study, the IOM criteria were utilized to diagnose CFS. Several other case definitions have emerged. In 1994, Fukuda et al. introduced recommendations for the clinical evaluation of fatigued persons with key symptoms including post-exertional malaise, neurocognitive deficits and unrefreshing sleep [20]. The Clinical Canadian Consensus criteria (CCC) in 2003 [21] required seven symptoms selecting a smaller group of patients than the definition by Fukuda et al. [22]. Compared to Fukuda et al. and CCC clinical case definitions, IOM criteria bring in a larger group of patients, even though core symptoms are identified [23]. In this study, however, patients with severe fatigue were systematically evaluated by multidisciplinary specialist team. Strict criteria were applied and of 128 patients with fatigue, only 29 were diagnosed with CFS. In these patients, other fatigue causing diseases and conditions had been excluded, and they comprised a homogenous group without significant comorbidities.
There is no objective test to measure fatigue or HRQOL, but by utilizing a patient-reported outcome measure, professionals are able to gather insights with direct relevance to the patient questioned. Several different methods are applied to assess quality of life and there is no consensus on the best technique for determining patient well-being. In certain patient populations, substantial variability among commonly utilized questionnaires has been reported and discrepancies have been particularly apparent as data have been evaluated at the individual level. To avoid bias, we decided to apply an array of HRQOL measures [24].
FMT is commonly administered by three modalities: colonoscopy, oral capsules and gastroscope or nasojejunal probe [25]. Oral capsule is well tolerated and perhaps the most acceptable way by participants, while colonoscopy requires bowel lavage and is time consuming. In a recent meta-analysis, FMT delivered via colonoscopy was superior compared to oral capsules in patients with irritable bowel syndrome [26]. In this study, colonoscopy was applied to maximize the likelihood of successful FMT. However, a remarkable proportion of CFS patients refused to participate in the study while they considered the colonoscopy procedure being too burdensome for them.
In previous studies, HRQOL of CFS patient has been remarkably lower compared to the general population. In this study, 10 patients out of 11 reported their baseline VAS fatigue score over 50 (scale 0–100). The average total MFIS score in the FMT and placebo groups were 59.6 and 61.0, respectively. Besharat and colleagues reported mean MFIS score 46.7 in CFS patients compared to 25.4 in healthy controls [27]. Compared to patients with relapsing–remitting multiple sclerosis, the total MFIS score, and particularly physical and cognitive subscale scores were higher in our patients [28]. Prak and colleagues recently reported average total MFIS score 35.1 in patients with primary Sjögren’s syndrome [29].
The 15D is a standardized measure of HRQOL that can be used both as a profile and single index score measure representing overall HRQOL. It generically assesses health status in terms of 15 dimensions not specific to any patient group or disease. The index score (range 0–1) is calculated by using a set of population-based utility weights [30]. Similarly, the EQ-5D-3L is a widely used, simple and generic questionnaire for use in clinical trials. The descriptive system evaluates five dimensions of health with three levels of severity providing altogether 243 potential health states. Like the 15D-profiles, each EQ-5D-3L health state can further be converted into a single numerical value ranging from 0 to 1. These profile-based index scores are generally used in the economic health studies with large number of patients. In this report, the profiles are preferred instead of index scores to provide a comprehensive perspective of patients’ symptoms.
Our main hypothesis was that gut dysbiosis might explain the pathomechanism of CFS and that gut microbiota intervention with FMT could relieve CFS-related symptoms. We exclusively selected the CFS patients from a large group of patients with fatigue caused by other reasons. The study subjects were randomly allocated in a double-blind fashion to the FMT or the placebo groups. Colonoscopy was applied to administer the FMT. In a previous study using the same FMT procedure without prior antibiotic pre-conditioning, one dose of FMT was found to alter the microbiota of irritable bowel syndrome patients to resemble that of the donor [31].
During the six-month follow up, the HRQOL was assessed twice, and four different self-reported questionnaires were utilized. In this study, we could not find improvement in the HRQOL scores after the single FMT from an universal donor. There were no statistically significant differences in the characteristics or the baseline HRQOL scores between the FMT and the placebo groups. Compared to the baseline VAS and MFIS scores, the differences in 1- and 6-months scores were statistically non-significant in both the FMT and the placebo groups. There was a slight, but statistically non-significant improvement in the 1-month MFIS physical subscale scores. However, such tendency was not present in VAS scores. The individual patient’s EQ-5D-3L profiles remained practically unaltered throughout the study. The 15D profiles in the FMT and the placebo groups did not change during the follow-up. The limited number of study subjects inhibits the generalizability of these results. There was only one male subject in the study. To exclude gender-based bias, we re-analyzed the data after having the male subject removed from the analysis. The results did not change: no statistically significant difference was found between the FMT and the placebo groups. This pilot study did not confirm nor exclude the possibility that FMT could have some effect on the HRQOL of patients with CFS. The pathomechanism of CFS remains unknown and gut microbiota may affect the pathophysiology of several neurological disorders. Further research and larger trials including also evaluation of changes in bacterial composition are needed to discover the underlying mechanisms of CFS.
Conclusion
In this pilot study, FMT was safe, but did not relieve symptoms or improve the HRQOL of patients with CFS. Small number of study subjects limits the generalizability of these results and further research is needed to assess the potential of FMT in CSF patients.
Abbreviations
- AUDIT
Alcohol use disorders identification test
- BDI
Beck’s depression inventory
- CCC
Clinical Canadian consensus criteria
- CFS
Chronic fatigue syndrome
- FMT
Faecal microbiota transplantation
- HRQOL
Health-related Quality of Life (HRQOL)
- IOM
Institute of medicine
- MFIS
Modified fatigue impact scale
- rCDI
Recurrent Clostridioides difficile infection
- SD
Standard deviation (SD)
- TAUH
Tampere university hospital
- VAS
Visual analog scale
Author contributions
TS designed the study protocol, analyzed and interpreted the patient data and was a major contributor in writing the manuscript. EJ made a substantial contribution to the design of the work, administered the FMT and was a contributor in writing the manuscript. RS and PL contributed to the study design concerning the FMT treatment and to the writing of the manuscript and substantively revised it. All authors read and approved the final manuscript.
Funding
Open access funding provided by Tampere University including Tampere University Hospital, Tampere University of Applied Sciences (TUNI). This study was supported by an academic grant from a Finnish governmental research fund and from Björkqvist fund.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
The trial was approved by the ethical committee of Tampere University Hospital (identifier R18006) and written informed consent was obtained from all study participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Panelli MC JTM advances in uncharted territories: diseases and disorders of unknown etiology. J Transl Med. 2017;15:192. doi: 10.1186/s12967-017-1293-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Carruthers BM, van de Sande MI, De Meirleir KL, Klimas NG, Broderick G, Mitchell T, et al. Myalgic encephalomyelitis: international consensus criteria. J Intern Med. 2011;270:327–338. doi: 10.1111/j.1365-2796.2011.02428.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Johnston SC, Brenu EW, Hardcastle SL, Huth TK, Staines DR, Marshall-Gradisnik SM. A comparison of health status in patients meeting alternative definitions for chronic fatigue syndrome/myalgic encephalomyelitis. Health Qual Life Outcomes. 2014;12:64. doi: 10.1186/1477-7525-12-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Du Preez S, Corbitt M, Cabanas H, Eaton N, Staines D, Marshall-Gradisnik S. A systematic review of enteric dysbiosis in chronic fatigue syndrome/myalgic encephalomyelitis. Syst Rev. 2018;7(1):241. doi: 10.1186/s13643-018-0909-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Choutka J, Jansari V, Hornig M, Iwasaki A. Unexplained post-acute infection syndromes. Nat Med. 2022;28(5):911–923. doi: 10.1038/s41591-022-01810-6. [DOI] [PubMed] [Google Scholar]
- 6.Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444(7122):1022–1023. doi: 10.1038/4441022a. [DOI] [PubMed] [Google Scholar]
- 7.Rutsch A, Kantsjö JB, Ronchi F. The gut-brain axis: how microbiota and host inflammasome influence brain physiology and pathology. Front Immunol. 2020;10(11):604179. doi: 10.3389/fimmu.2020.604179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liang D, Leung RK, Guan W, Au WW. Involvement of gut microbiome in human health and disease: brief overview, knowledge gaps and research opportunities. Gut Pathog. 2018;25(10):3. doi: 10.1186/s13099-018-0230-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.DeGruttola AK, Low D, Mizoguchi A, Mizoguchi E. Current understanding of dysbiosis in disease in human and animal models. Inflamm Bowel Dis. 2016;22(5):1137–1150. doi: 10.1097/MIB.0000000000000750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vendrik KEW, Ooijevaar RE, de Jong PRC, Laman JD, van Oosten BW, van Hilten JJ, et al. Fecal microbiota transplantation in neurological disorders. Front Cell Infect Microbiol. 2020;24(10):98. doi: 10.3389/fcimb.2020.00098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vogt NM, Kerby RL, Dill-McFarland KA, Harding SJ, Merluzzi AP, Johnson SC, et al. Gut microbiome alterations in Alzheimer‘s disease. Sci Rep. 2017;7:1–11. doi: 10.1038/s41598-017-13601-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Boertien JM, Murtomäki K, Pereira PAB, van der Zee S, Mertsalmi TH, Levo R, et al. Fecal microbiome alterations in treatment-naive de novo Parkinson's disease. NPJ Parkinsons Dis. 2022;8(1):129. doi: 10.1038/s41531-022-00395-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Du G, Dong W, Yang Q, Yu X, Ma J, Gu W, et al. Altered gut microbiota related to inflammatory responses in patients with huntington's disease. Front Immunol. 2021;11:603594. doi: 10.3389/fimmu.2020.603594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Engelenburg HJ, Lucassen PJ, Sarafian JT, Parker W, Laman JD. Multiple sclerosis and the microbiota: progress in understanding the contribution of the gut microbiome to disease. Evol Med Public Health. 2022;10(1):277–294. doi: 10.1093/emph/eoac009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maes M, Mihaylova I, Leunis JC. Increased serum IgA and IgM against LPS of enterobacteria in chronic fatigue syndrome (CFS): indication for the involvement of gram-negative enterobacteria in the etiology of CFS and for the presence of an increased gut-intestinal permeability. J Affect Disord. 2007;99:237–240. doi: 10.1016/j.jad.2006.08.021. [DOI] [PubMed] [Google Scholar]
- 16.Johnson S, Lavergne V, Skinner AM, Gonzales-Luna AJ, Garey KW, Kelly CP, et al. Clinical practice guideline by the infectious diseases society of America (IDSA) and society for healthcare epidemiology of America (SHEA): 2021 focused update guidelines on management of clostridioides difficile infection in adults. Clin Infect Dis. 2021;73(5):e1029–e1044. doi: 10.1093/cid/ciab549. [DOI] [PubMed] [Google Scholar]
- 17.Committee on the Diagnostic Criteria for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome; Board on the Health of Select Populations; Institute of Medicine. Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness. Washington (DC): National Academies Press (US); 2015. [PubMed]
- 18.Cammarota G, Ianiro G, Tilg H, Rajilić-Stojanović M, Kump P, Satokari R, et al. European consensus conference on faecal microbiota transplantation in clinical practice. Gut. 2017;66(4):569–580. doi: 10.1136/gutjnl-2016-313017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Satokari R, Mattila E, Kainulainen V, Arkkila PET. Simple faecal preparation and efficacy of frozen inoculum in faecal microbiota transplantation for recurrent Clostridium difficile infection–an observational cohort study. Aliment Pharmacol Ther. 2015;41(1):46–53. doi: 10.1111/apt.13009. [DOI] [PubMed] [Google Scholar]
- 20.Fukuda K, Straus SE, Hickie I, Sharpe MC, Dobbins JG, Komaroff A. The chronic fatigue syndrome: a comprehensive approach to its definition and study. International chronic fatigue syndrome study group. Ann Intern Med. 1994;121(12):953–959. doi: 10.7326/0003-4819-121-12-199412150-00009. [DOI] [PubMed] [Google Scholar]
- 21.Carruthers BM, Jain AK, De Meirleir KL, Peterson DL, Klimas NG, Lerner AM, et al. Myalgic encephalomyelitis/chronic fatigue syndrome: clinical working case definition, diagnostic and treatment protocols. J. Chronic Fatigue Syndr. 2003;11:7–116. http://www.mefmaction.com/images/stories/Medical/ME-CFS-Consensus-Document.pdf. Accessed 28 Mar 2023
- 22.Jason LA, Sunnquist M, Kot B, Brown A. Unintended consequences of not specifying exclusionary illnesses for systemic exertion intolerance disease. Diagnostics. 2015;5(2):272–286. doi: 10.3390/diagnostics5020272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jason LA, McManimen S, Sunnquist M, Newton JL, Strand EB. Clinical criteria versus a possible research case definition in chronic fatigue syndrome/myalgic encephalomyelitis. Fatigue. 2017;5(2):89–102. doi: 10.1080/21641846.2017.1299077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hornberger JC, Redelmeier DA, Petersen J. Variability among methods to assess patients' well-being and consequent effect on a cost-effectiveness analysis. J Clin Epidemiol. 1992;45(5):505–512. doi: 10.1016/0895-4356(92)90099-9. [DOI] [PubMed] [Google Scholar]
- 25.Allegretti JR, Mullish BH, Kelly C, Fischer M. The evolution of the use of faecal microbiota transplantation and emerging therapeutic indications. Lancet. 2019;394(10196):420–431. doi: 10.1016/S0140-6736(19)31266-8. [DOI] [PubMed] [Google Scholar]
- 26.Wu J, Lv L, Wang C. Efficacy of fecal microbiota transplantation in irritable bowel syndrome: a meta-analysis of randomized controlled trials. Front Cell Infect Microbiol. 2022;12:827395. doi: 10.3389/fcimb.2022.827395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Besharat MA, Behpajooh A, Poursharifi H, Zarani F. Personality and chronic fatigue syndrome: the role of the five-factor model. Asian J Psychiatr. 2011;4(1):55–59. doi: 10.1016/j.ajp.2010.12.001. [DOI] [PubMed] [Google Scholar]
- 28.Kargarfard M, Shariat A, Ingle L, Cleland JA, Kargarfard M. Randomized controlled trial to examine the impact of aquatic exercise training on functional capacity, balance, and perceptions of fatigue in female patients with multiple sclerosis. Arch Phys Med Rehabil. 2018;99(2):234–241. doi: 10.1016/j.apmr.2017.06.015. [DOI] [PubMed] [Google Scholar]
- 29.Prak RF, Arends S, Verstappen GM, van Zuiden G, Kroese FGM, Bootsma H, et al. Fatigue in primary Sjögren's syndrome is associated with an objective decline in physical performance, pain and depression. Clin Exp Rheumatol. 2022;40(12):2318–2328. doi: 10.55563/clinexprheumatol/70s6cs. [DOI] [PubMed] [Google Scholar]
- 30.Sintonen H, Pekurinen M. A fifteen-dimensional measure of health-related quality of life (15D) and its applications. In: Walker SR, Rosser RM, editors. Quality of life assessment: key issues in the 1990s. Dordrecht: Kluver Academic Publishers; 1993. pp. 185–195. [Google Scholar]
- 31.Lahtinen P, Jalanka J, Hartikainen A, Mattila E, Hillilä M, Punkkinen J, et al. Randomised clinical trial: faecal microbiota transplantation versus autologous placebo administered via colonoscopy in irritable bowel syndrome. Aliment Pharmacol Ther. 2020;51(12):1321–1331. doi: 10.1111/apt.15740. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.