Abstract
Background
Herpes simplex virus 1 (HSV-1), an important human pathogen, is capable of latent infection in neurons and productive (lytic) infection in other tissue cells. Once infected with HSV-1, the immune system of the organism cannot eliminate the virus and carries it lifelong. HSV-1 possesses approximately 150 kb of double-stranded linear genomic DNA and can encode at least 70 proteins and 37 mature microRNAs (miRNAs) derived from 18 precursor miRNAs (pre-miRNAs).
Summary
These HSV-1-encoded miRNAs are widely involved in multiple processes in the life cycle of the virus and the host cell, including viral latent and lytic infection, as well as host cell immune signaling, proliferation, and apoptosis.
Key Message
In this review, we focused primarily on recent advances in HSV-1-encoded miRNA expression, function, and mechanism, which may provide new research ideas and feasible research methods systemically and comprehensively.
Keywords: Herpes simplex virus 1, MicroRNAs, Mechanisms and functions
Introduction
Herpes simplex virus 1 (HSV-1) belongs to the order Herpesvirales, the family Herperviridae, the subfamily Alphaherpesvirinae, the genus Simplexvirus, and the species of human Alphaherpesvirus. According to World Health Organization evaluation, more than 3.7 billion people are infected with HSV-1 worldwide (http://www.who.int). Humans are the sole natural host of HSV-1, although it can infect various experimental animals, such as mice, tree shrews, and rhesus macaques, under experimental conditions [1, 2]. Although there are different types of candidate vaccines under development against HSV-1 infection at different stages of research and development or clinical trials, there are still no commercial HSV-1 vaccines on the market [3, 4]. FDA-approved antiviral drugs, such as acyclovir, famciclovir, and valacyclovir, can relieve clinical symptoms but cannot cure the diseases caused by HSV-1 infection.
HSV-1 has two significantly different lifestyles throughout its life cycle: alternate latent and lytic infections. The dominant feature of latent infection is that the lytic viral genes persist in a transcriptionally silenced state, whereas only latency-associated transcripts (LATs) are transcribed abundantly within sensory neurons in vivo [5–7]. By contrast, lytic infection is characterized by a sequential gene expression pattern in which so-called immediate-early (IE) or alpha genes, early (E) or beta genes, and later (L) or gamma genes are transcribed [8, 9]. In the primary infection, asymptomatic infection accounts for the majority except for symptoms including painful blisters or open sores called ulcers in or about infected oral-lip parts; in the latent stage of infection, the virus remains within neurons of sensory ganglia, such as the trigeminal ganglion (TG), and can be reactivated to reach primary infected parts along sensory nerve fibers to initiate a new round of infection.
MiRNAs, approximately 22 nucleotides (nt) in length endogenous single-stranded nonprotein-coding regulatory RNA molecules, modulate multiple biological processes, including development, metabolism, immunity, cellular physiology, and disease pathology at posttranscriptional levels in plants, animals, humans, and viruses [10–13]. In the past few decades, significant advances in the field of miRNA research have been made in both eukaryotes and viruses. The number of reported miRNAs exceeds 5,000 in humans (miRBase release v. 22.1, 2018) and 400 in viruses [14], and the functions of some of these have been partially elucidated. Although the discovery of virus-encoded miRNAs was later than that of plants and animals, rapid advances in studies on viral miRNA expression, function, and mechanism of action have been achieved since the discovery of the first virus-encoded miRNAs in Epstein-Barr virus (EBV)-infected B cells in 2004 [15]. Since then, more than 200 mature viral miRNAs have been discovered in different groups of herpesviruses [16–24].
Both viral and cellular miRNAs use the RNA silencing machinery of the host cell to suppress viral or/and cellular gene expression by either translational repression, destabilization, or, more rarely, messenger RNA (mRNA) cleavage of the target via binding to complementary sites, typically located in the 3′-untranslated regions (UTRs) [25]. Although viral miRNA biogenesis and functional targeting are fully dependent on the host’s molecular miRNA maturation and silencing machinery, to date, no viral proteins have been identified that directly contribute to either miRNA biogenesis or targeting mechanisms [13].
As one of the most successful human pathogens, HSV-1 has developed various strategies to manipulate cellular physiology to favor its production, including the use of viral miRNAs, which mediate the interplay of virus and host. In this review, we will summarize the expression, biological function, pleiotropic roles, and challenges to our understanding of HSV-1 miRNAs.
Discovery of HSV-1-Encoded miRNAs
The first identified miRNAs were derived from lin-4 transcripts, which have antisense complementarity to 3′-UTR of lin-14 messenger RNA and regulate lin-14 translation via an antisense RNA-RNA interaction in Caenorhabditis elegans (C. elegans) in 1993 [26]. Subsequently, another gene, lin-28, was shown to be regulated by the lin-4 RNA with the same way in C. elegans [27]. As a landmark discovery in 2,000, one C. elegans miRNA, let-7, a 21-nucleotide RNA, regulated multiple heterochronic genes, such as lin-14, lin-28, lin-41, lin-42, and daf-12, within the mRNA 3′-UTR via an antisense RNA-RNA interaction, and it was 100% conserved within the genomes of mice and humans [28, 29]. In 2004, five miRNAs (miR-BHRF1-1, 2, 3, miR-BART1, and miR-BART2) encoded by EBV were first identified by Northern blotting [15].
Based on the stem-loop structure characters of precursor miRNAs (pre-miRNAs) filtered following some criteria, a computer algorithm was applied to predict hairpin-structured precursors in the HSV-1 strain 17 genome (NC_001806); Pfeffer et al. [16] discovered seven unique miRNA precursor predictions, four of which were registered as miR-H4, miR-H8, miR-H11, and miR-H14 in miRBase (Table 1). Using computational prediction, Cui et al. [19] screened the HSV-1 strain 17 complete genomes (NC_001806) for sequences that adopted an extended stem-loop structure and were predicted to encode 13 miRNA precursors, four of which, including miR-H1, miR-H5, miR-H12, and miR-H13, were later registered in miRBase. To identify HSV-1 LAT-derived miRNAs, Umbach et al. constructed a LAT expression plasmid, pcDNA3/LAT, in which a heterologous promoter drives transcription of an approximate 10.8 kb HSV-1 genomic fragment containing the entire 8.3 kb LAT, then transfected this plasmid into HEK-293T cells and isolated total RNA to prepare complementary DNAs for 454 sequencing. Six HSV-1 miRNA sequences were obtained, derived from four HSV-1 miRNA precursor hairpins, of which miR-H2, miR-H3, and miR-H6 were novel viral miRNAs and can be detected by Northern blotting and reverse transcript-polymerase chain reaction (RT-PCR) both in HSV-1 lytic-infected Vero cells and mice latently infected TG [30]. Identification of novel miR-H7, encoded within the second exon of the HSV-1 LAT, used deep sequencing in human latently infected TG and determined its expression in latently and productively infected cells and tissue via RT-PCR [20]. Although the miR-H8 was first detected by sequencing, its prediction, named with pre-miRNA genome location, was made in Pfeffer Lab in 2005 [16]. To more completely define the various miRNA species expressed by HSV-1 during productive and latent infection, Jurak et al. [31] applied massively parallel sequencing of small RNA libraries to identify the viral miRNA expression in the sample of productive infection in HEK-293 cells with HSV-1 strain KOS at a multiplicity of infection (MOI) of 5 for 18 h and latent infection in mice TG, and they were able to identify 16 miRNAs expressed by HSV-1, including all previously known species and several previously unidentified virus-encoded miRNAs, such as miR-H15, miR-H16, miR-H17, and miR-H18. Munsun et al. [32] claimed that six novel HSV-1-derived miRNA candidates were identified in early infected HEK-293 cells of HSV-1 KOS strain using small RNA high-throughput sequencing approach and then validated by Northern blotting. With the exception of miR-92944, which is registered in miRBase under the name miR-H26, it is not clear why the remaining five miRNA candidates are not registered in miRBase. Wu et al. [33] scanned the whole genome sequence of HSV-1 strain F using RNA structure 3.2 to predict the secondary structures of the miRNA precursors constrained with certain criteria; they identified a viral miRNA, miR-H27, which was located in a noncoding region within the ICP0 gene and verified by Northern blotting and RT-PCR in human fetal fibroblast (KMB-17 strain) cells infected with HSV-1 strain F. Han et al. [34] identified two previously unreported viral miRNAs, miR-H28 and miR-H29, in HEK-293T and HEp-2 cells infected with HSV-1 strain F. The miR-H28 sequence is antisense to and maps close to the N-terminal domain of unique long 4 (UL4). Additionally, miR-H29 maps in the same sense as the coding sequence of UL30; however, neither molecule is registered in miRBase.
Table 1.
Prediction or validation of HSV-1 miRNAs registered in miRBase (v22.1)
| Pre-miRNAs | Prediction/identification | Validation | Location (orientation) | Reference |
|---|---|---|---|---|
| miR-H4 | Computer algorithm | ND | 125,877–125,957 (+) 417–497 (−) | [15] |
| miR-H8 | Computer algorithm | ND | 123,803–123,876 (+) 2,498–2,571 (−) | [15] |
| miR-H11 | Computer algorithm | ND | 6,241–62,540 (+) 6,241–62,540 (−) | [15] |
| miR-H14 | Computer algorithm | ND | 4,466–4,554 (+) 121,820–121,908 (−) | [15] |
| miR-H1 | Computational prediction | Northern blotting | 118,317–118,402 (+) 7,972–8,057 (−) | [18] |
| miR-H5 | Computational prediction | ND | 126,720–126,799 (+) 151,398–151,477 (−) | [18] |
| miR-H12 | Computational prediction | ND | 146,150–146,237 (+) 131,960–132,047 (−) | [18] |
| miR-H13 | Computational prediction | ND | 131,971–132,034 (+) 146,163–146,226 (−) | [18] |
| miR-H2 | 454 sequencing | Northern blotting RT-PCR | 121,827–121,901 (+) 4,473–4,547 (−) | [29] |
| miR-H3 | 454 sequencing | RT-PCR | 125,729–125,809 (+) 565–645 (−) | [29] |
| miR-H6 | 454 sequencing | Northern blotting RT-PCR | 7,972–8,057 (+) 118,317–118,402 (−) | [29] |
| miR-H7 | Deep sequencing | RT-PCR | 123,410–123,492 (+) 2,882–2,964 (−) | [19] |
| miR-H15 | Parallel sequencing | ND | 7,072–7,173 (+) 119,201–119,302 (−) | [30] |
| miR-H16 | Parallel sequencing | ND | 69,020–69,119 (+) | [30] |
| miR-H17 | Parallel sequencing | ND | 150,560–150,654 (+) 127,543–127,637 (−) | [30] |
| miR-H18 | Parallel sequencing | ND | 131,836–131,930 (+) 146,267–146,361 (−) | [30] |
| miR-H26a | High-throughput sequencing | Northern blotting RT-PCR | 92,911–92,973 (+) | [31] |
| miR-H27 | RNA structure 3.2 | Northern blotting RT-PCR | 2,883–2,965 (+) 123,409–123,491 (−) | [32] |
| miR-H28* | Deep sequencing | RT-PCR | 12,387–12,533 (+) | [33] |
| miR-H29* | Deep sequencing | RT-PCR | 64,902–65,076 (+) | [33] |
| sncRNA1* | Computer-driven predictions | Northern blotting Small RNA clone | 119,756–119,817 (+) 6,557–6,618 (−) | [35] |
| sncRNA2* | Computer-driven predictions | Northern blotting Small RNA clone | 120,146–12,0181 (+) 6,193–6,228 (−) | [35] |
ND, not detected in reported papers.
*These miRNAs were not registered in miRBase.
aDesignated as miR-92944 in the original paper.
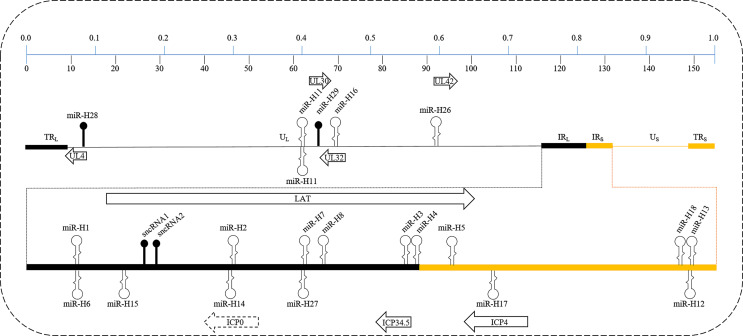
Peng et al. identified two small RNAs within the first 1.5 kb of the HSV-1-encoded LAT in human neuroblastoma cells (SK-N-SH) infected with dLAT2903R, a LAT null mutant (dLAT2903) with a rescued McKrae strain containing a restored [35], at an MOI of 5 for 16 h. Although these small RNAs possess extensive secondary structure and a stem-loop structure, bands migrating near 23 bases were undetected, suggesting that these small RNAs are not true miRNAs [36]. Bachmann et al. [37] reported that two novel small HSV-1-encoded RNAs, termed HVR1 and HVR2, with the length of 255 and 154 nt, respectively, were immediately synthesized in infected Vero cells with HSV-1 strain Lennette at an MOI of 5 as early as 4 hpi; however, they did not provide more evidence of sequence and genome location. Figure 1 and Table 1, respectively, summarize the location and discovery of HSV-1-encoded miRNAs.
Fig. 1.
Schematic of the HSV-1 strain 17 genome (NC_001806) expanded to display the details of internal repeat unique sequences of long (IRL) and internal repeat unique sequences of short (IRS) loci. The upper and lower panels indicate the percentage of the coordinated genome and exact kilobase nucleotides, respectively. The locations of pre-miRNAs in the viral genome are shown as hairpins with relative sizes; pre-miRNAs shown above the line are located in the plus strand, which are transcribed from left to right (in the same direction as LAT), whereas those below the line are located in the minus strand, which are transcribed from right to left (in the opposite direction as LAT). All pre-miRNAs have stem-loop structures except for miR-H28, miR-H29, sncRNA1, and sncRAN2, which are indicated by the icons of black solid balls. Additionally, pre-miR-H11 comprises a 130 bp perfectly inverted repeat sequence. TRL, terminal repeat sequence of long; TRS, terminal repeat sequence of short; UL, unique long; US, unique short.
Expression Profiles of Selected HSV-1-Encoded miRNAs during Lytic Infection, as well as Latent Infection and Reactivation
Compared with the expression model of HSV-1-encoded protein genes, which were typically divided into three waves and designated IE (alpha genes), early (beta genes), and later (gamma genes), the expression profiles of viral miRNAs presented obvious differences in lytic (productive) infection, as well as latent infection and reactivation. Several studies have been reported using different models to explore the expression profiles of HSV-1-encoded miRNAs.
Whether in SY5Y cells at 6 and 18 hpi, Hela cells at 18 hpi, or Vero cells at 24 hpi, productively infected with HSV-1 strain KOS at an MOI of 10, HSV-1 miR-H1, miR-H2, miR-H4, and miR-H6 showed higher expression levels than miR-H3 and miR-H5 [20]. Jurak et al. [31] applied the construction of a small RNA sequencing library from productive infection of HEK-293 and mice TG sample to discover that reads for miR-H1 and miR-H6 were the most abundant in the HSV-1 productive infection sample, whereas, in the latent samples, they were greatly outnumbered by reads for miR-H2, miR-H3, and miR-H4. Kramer et al. [38] performed a stem-loop qRT-PCR analysis of RNA from Vero cells infected with HSV-1 strain KOS for times of 2–18 hpi; they discovered that miR-H1, miR-H4-3p, and miR-H6 could be detected as early as 2 or 4 hpi. Furthermore, miR-H1 and miR-H6, approximately 300 and 4,000 molecules per cell, respectively, accumulated throughout the infection and were substantially more abundant than the other HSV-1 miRNAs, whereas miR-H2, miR-H4-3p, miR-H4-5p, and miR-H5 accumulated to approximately 30 molecules per cell, and miR-H3 to less than one molecule per cell [38]. By Northern blotting, Pan et al. discovered that six HSV-1 miRNAs (H1, H2, H3, H4, H5, and H6) exhibited high pre-miRNA/miRNA ratios during lytic infection in HEK-293T and Neuro-2A cells, as well as in acutely infected mouse TG. By contrast, considerably lower ratios were observed in latently infected mouse TG. This mechanism may benefit the virus by reducing the expression of repressive miRNAs during lytic infection while permitting elevated expression during latency [39].
Umbach et al. reported that deep sequencing of human TG recovered all five HSV-1 miRNAs previously documented in latently infected murine neurons (H2, H3, H4, H5, and H6) as well as two new LAT-derived miRNAs (H7 and H8) despite the low or undetectable copy number of miR-H5 in the two samples, whereas the HSV-1 miR-H1, previously detected only in cells undergoing productive HSV-1 replication, was not recovered in this analysis of latently infected human TG [19, 20, 30, 40]. Held et al. characterized the expression of five HSV-1-encoded miRNAs (H2-3p, H3, H4-3p, H4-5p, H5, and H6) in three human TG via qPCR, and the results indicated that all assessed miRNAs were present in the latently infected TG, except for miR-H3 in one subject. Meanwhile, they analyzed the expression of viral miRNAs in single neurons and discovered that HSV-1 miRNAs were only expressed in LAT+ neurons and not in LAT− neurons; contrary to whole-tissue RNA, on the single-cell level, miR-H6 showed an obvious difference in expression in LAT+ versus LAT− neurons [41]. By performing small RNA library sequencing, Cokaric Brdovcak et al. [42] detected only five miRNAs (miR-H2, miR-H3, miR-H4, miR-H6, and miR-H7) in latent infection of human TG, with miR-H2-3p and miR-H4-3p being the most abundant. Jurak et al. applied cell culture models of quiescent or latent infection; using deep sequencing, they analyzed the expression of HSV-1 miRNAs in two models of human fibroblasts (HFFF2 and HFF) and one rat neuron (SCG). In all cases, the expression patterns differed from those in productively infected cells, with the rat neuron pattern most closely resembling that found in latently infected human or mouse ganglia in vivo [43].
In the TG-reactivated mouse model, the amounts of miR-H3, miR-H5, and miR-H6 decreased by 30- to 100-fold between 5 and 9 h after excision; this decrease in miRNA accumulation is blocked by RNA or protein synthesis inhibitors [44]. In TG of mice inoculated by the corneal route with a 105 plaque-forming unit in each eye, the copy numbers of miRNAs (miR-H3, miR-H5, and miR-H6) increased with time and reached peak levels 9–14 d after inoculation, with amounts detected at 28 d after infection being approximately 10-fold lower than those quantified on days 9–14 [44].
Du et al. examined the synthesis and accumulation patterns of 18 HSV-1 miRNAs in productive, latent, and reactivated infection models. They reported that H2-3p, H3-3p, H4-3p, H5-3p, H6-3p, and H7-5p accumulated in ganglia-harboring latent virus; H8-5p, H15, H17, H18, H26, and H27 accumulated during reactivation; H1-5p, H2-3p, H3-3p, H5-3p, H6-3p, H7-5p, H13, H16, H26, and H27 were found to accumulate above background levels in productively infected cells [45]. The disparity in miRNA accumulation in cell culture and during reactivation may reflect differences in the regulation patterns of viral gene expression during productive infection and reactivation from the latent state.
Although the expression profile of HSV-1 miRNAs has been studied systematically and intensively, conclusions with a high degree of consistency have not yet been obtained. The characteristics of dynamic expression changes may be closely related to the type of infected cells, the mode of infection, and the detection methods. Understanding the expression profile of miRNAs encoded by viruses may provide temporal coordinates for studying their function. We have summarized the details of expression profiles of HSV-1 miRNAs in Table 2.
Table 2.
Models used to characterize the expression profiles of HSV-1 miRNAs
| miRNAs | Productive | Latent | Reactivated | Reference | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| model | Virus strain | detection | model | virus strain | detection | model | virus strain | detection | ||
| miR-H1–miR-H6 | Vero | KOS | Northern blotting | ND | ND | ND | ND | ND | ND | [18] |
| SY5Y HeLa | KOS | qRT-PCR | Human TG | ND | Sequencing | ND | ND | ND | [19] | |
| HEK-293 | KOS | Sequencing | Mice TG | KOS | Sequencing | ND | ND | ND | [30] | |
| Vero | KOS | qRT-PCR | Mice TG | KOS | qRT-PCR | ND | ND | ND | [37] | |
| miR-H2–miR-H6 | ND | ND | ND | Human TG | ND | qRT-PCR | ND | ND | ND | [40] |
| miR-H3 miR-H5 miR-H6 | Mice TG | F | qRT-PCR | ND | ND | ND | Mice TG | F | qRT-PCR | [43] |
| miR-H1–miR-H6 | ND | ND | ND | HFFF2 HFF SCG | In1374 KOS Patton | Sequencing | ND | ND | ND | [42] |
| miR-H1–miR-H8 miR-H13 miR-H15–miR-H18 miR-H26–miR-H27 | Vero | F | qRT-PCR | Mice TG | F | qRT-PCR | Mice TG | F | qRT-PCR | [44] |
| miR-H2–miR-H4 miR-H6–miR-H7 | ND | ND | ND | Human TG | ND | Sequencing | ND | ND | ND | [41] |
| miR-H1–miR-H6 | HEK-293T Neuro-2A | KOS | Northern blotting | Mice TG | KOS | Northern blotting | Mice TG | KOS | Northern blotting | [38] |
HSV-1-Encoded miRNAs Regulated Host Innate Immunity
Innate immunity is considered the first-line defense against viral infection, which comprises various innate immune cells, such as granulocytes, natural killer (NK) cells, dendritic cells, and macrophages. Many of these cell types use conserved pattern recognition receptors to detect pathogen-associated molecular patterns on various viral pathogens. Upon detection of viral infection, innate immunity cells make use of pattern recognition receptors, including toll-like receptors, retinoic acid-inducible gene-I (RIG-I), NOD-like receptors, and DNA sensors, that recognize pathogen-associated molecular patterns and activate signaling pathway induction of IFN production, which in turn induces the expression of a large number of interferon-stimulated genes to inhibit viral replication [46]. Intrinsic immunity refers to cells constitutively expressing antiviral factors without activation of viral infection that can act as potent inhibitors of viral replication [47, 48]. During the long-term co-evolution with the host, HSV-1 adopts various mechanisms to modulate the host’s antiviral innate and intrinsic immunity, especially HSV-1-encoded multiple miRNAs entailed this process [49].
HSV-1 miR-H1-5p, a gene locus located in the LAT promoter region, is expressed abundantly during lytic infection. Applying bioinformatics prediction and dual-luciferase validation, miR-H1-5p was identified to target SORT1, which is involved in endocytic and intracellular trafficking pathways. Phagocytic uptake and multiple secreted cytokines with important roles in innate immune responses were significantly attenuated in miR-H1-5p-transfected primary human macrophages, suggesting a role for miR-H1-5p in immune regulation [50]. HSV-1 miR-H1-5p also directly targeted alpha-thalassemia/mental retardation syndrome X-linked (ATRX) protein, a component of intrinsic immunity factor nuclear domain 10. HSV-1 miR-H1-5p mimics can deplete the endogenous ATRX protein and repress the expression of a luciferase reporter linked to the 3′-UTR of ATRX mRNA in HEK-293 cells [51]. MiR-H1-5p has also been shown to be able to directly target the immune-related genes LIFR and TGFBR1, as well as the autophagy and antiviral-related gene ATG16L1; miR-H1-5p overexpression can attenuate viral entry and infection in human oral keratinocytes, but lack of detectable miR-H1-mediated cell death and antiviral response (e.g., IFN-α/β) [52]. These findings suggest that miR-H1-5p is an immune-associated HSV-1 miRNA that modulates host cell physiologic processes and the viral life cycle through multiple targets.
Using dual-luciferase reporter assay and bioinformatics, Duan et al. screened and identified Asp-Glu-Ala-Asp (DEAD)-box helicase 41 (DDX41), a cytosolic DNA sensor, which was directly targeted by HSV-1-encoded miR-H2-3p. The transfection of miR-H2-3p mimics inhibited DDX41 expression at mRNA and protein levels, as well as IFN-β and myxoma resistance protein I (MxI) expression induced by HSV-1 infection in human acute monocytic leukemia (THP-1) cells, and promoted viral replication and its gene transcription, whereas transfection of miR-H2-3p inhibitor showed opposite effects [53]. This finding indicated that HSV-1 attenuated the cytosolic DNA-sensing pathway to enhance virus replication via viral miRNAs in cultured cells.
Enk et al. applied fluorescence-activated cell sorting to analyze the effects of HSV-1-encoded 27 miRNAs on the expression of five NKG2D ligands (MICA, MICB, and ULBP1–3) in the B-cell line, BJAB. The result indicated that miR-H8 significantly reduced the expression level of two NKG2D ligands, ULBP2 and ULBP3, in transfected BJAB cells using lentiviral vector-expressed artificial hairpin of miR-H8. Next, they utilized the RNA hybridization prediction program, RNAhybrid, to predict that PIGT, a component of glycosylphosphatidylinositol transamidase (GPTI) complex, was directly targeted in the 3′-UTR by miR-H8-5p, and the prediction was validated using a dual-luciferase assay. Overexpression of miR-H8-5p reduces NK-dependent killing, and PIGT affects tetherin expression. By taking these means, HSV-1 decreases recognition and elimination by NK cells and enhances viral spread [54].
HSV-1 miR-H28 was identified using deep sequencing in late productively infected HEK-293T and HEp-2 cells with HSV-1 strain F. Ectopic expression of miR-H28 mimics in human cells before infection reduced viral mRNA and protein accumulation, plaque sizes, and viral yields at low MOI [34]. Further experiments revealed that miR-H28 induced the synthesis of IFN-γ in infected HEK-293T and HEp-2 cells, as well as HEp-2 cells transfected with miR-H28. HSV-1 replication was affected by IFN-γ exposure before infection but not during or after infection [55]. These experimental results may give us a hint that HSV-1 miRNAs can trigger innate immunity at the right moment to restrict the spread of the virus from infected to uninfected cells to benefit its life cycle.
HSV-1-Encoded miRNAs Regulated Viral Lytic Replication, Latency, and Reactivation
Whether it is the acute or latent infection in neurons of the sensory ganglia, reactivation from latency by explant or stress induction, or lytic infection in cultured cells, these related processes and mechanisms have been extensively studied. Accumulating evidence suggests that virally encoded miRNAs are more or less involved in regulating these events.
HSV-1 miR-H2-3p was identified to target viral ICP0 and regulate virus replication for the first time; for obvious reasons, ICP0, a viral IE transcriptional activator, was thought to play a key role in productive HSV-1 replication and reactivation from latency, and it is an inhibitor of innate immune responses [30, 56–61]. Co-transfection with wt or mutant ICP0 and either a wt or mutant miR-H2-3p expression plasmid into HEK-293T cells revealed that miR-H2-3p inhibited ICP0 protein expression but not mRNA [30]. To test the effect of endogenous miR-H2-3p on the expression of endogenous ICP0 in infected HEK-293 cells, a 17syn+ mutant lacking miR-H2-3p without disrupting ICP0 encoding sequence infection of HEK-293 cells revealed a significant and specific increase in the level of ICP0 mRNA and protein expression compared with wt virus strain. Furthermore, consistent results can be obtained by wt HSV-1-infected HEK-293 cells transfected with miRNA mimics encoding miR-H2-3p [62]. Mutational inactivation of miR-H2 resulted in a significantly lower level of viral replication at 24 and 72 h in Neuro-2A cells infected at a low MOI; however, in Neuro-2A cells infected at a high MOI or in NIH 3T3 cells infected at either a high or a low MOI, the levels of replication of the wt and mutant viruses were comparable [62]. An McK-ΔH2, in which miR-H2 has been disrupted without altering the predicted amino acid sequence of ICP0, produced increased amounts of ICP0. Although the replication of McK-ΔH2 was similar to that of its wt McKrae parental virus in rabbit skin cells and mouse eyes, significantly more reactivation was observed with McK-ΔH2 at earlier times [63]. Similar findings were obtained with a dLAT-ΔH2, a LAT-negative mutant, in which miR-H2 is disrupted in dLAT2903 without altering the predicted amino acid sequence of the overlapping ICP0 open reading frame [64]. These outcomes indicated that miR-H2 plays a role in modulating HSV-1 reactivation. Using methods similar to previous ones, another team demonstrated that engineered mutations disrupted miR-H2 without affecting ICP0 in HSV-1 strain KOS, showing wild-type levels of infectious virus production and no increase in ICP0 expression in lytically infected cells, and the mutant was not significantly different from wt virus in terms of infectious virus production in the mouse TG during acute infection or the rate of reactivation from explanted latently infected ganglia [65]. The cell type and viral strain type dependence of miR-H2-3p on ICP0 and viral replication regulation reflects the possibility that other molecules are involved. Recombinant HSV-1 17mH3 and 17dH4, which were mutated in the seed region without affecting the underlying coding sequence of ICP34.5 and simply deleted the noncoding sequence of ICP34.5, respectively, resulted in significant enhancement of viral replication at 24 h in Neuro-2A cells infected at a low MOI. By contrast, in NIH 3T3 cells, the viral 17mH3 and 17dH4 mutants replicated at levels comparable with that of WT HSV-1 [62]. The effects of miR-H3-3p and miR-H4-3p on viral replication are cell-type-dependent. Although miR-H3 and miR-H4 are located antisense to mature ICP34.5 mRNA, only miR-H4-3p targets ICP34.5, which is considered an immunomodulatory protein [62, 66–68].
HSV-1 miR-H6 is located in the LAT promoter, lies complementary to miR-H1, and was first shown to target the HSV-1 transcription factor, ICP4, in co-transfected HEK-293T cells [30]. The subsequent experiment showed that transfection of miR-H6-3p mimics into HSV-1-infected human cornea epithelial cells decreases ICP4 and interleukin 6 (IL-6) expression and inhibits productive infection [69]. A recombinant virus (strain F) was constructed by expressing the miR-H6-5p sponge to produce the loss-of-function phenotype of miR-H6-3p. Infection with a recombinant virus in HEp-2 cells reduced viral protein levels and virus yield, but the establishment of latency and reactivation did not differ between the recombinant virus and wt HSV-1 [70]. Another recombinant virus lacking the sequences for miR-H1-5p and miR-H6-3p (termed 17dmiR-H1/H6) exhibited a modest increase in viral yields and a decreased and delayed reactivation phenotype than that of wt 17syn+. These results suggest that miR-H1/H6 plays a crucial role in facilitating efficient reactivation from latency [71].
A recombinant virus lacking miR-H8 (termed 17dmiR-H8) displayed only subtle reductions in viral yields in rabbit skin cells and a significant decrease in viral yields and DNA replication in Neuro-2A cells, whereas there was a moderate increase in DNA replication in differentiated human neuronal cells (Lund human mesencephalic cells), and no difference existed in viral yields in the Lund human mesencephalic cell or explant and adrenaline-induced reactivation [72]. These results indicate that miR-H8 is dispensable for the establishment of HSV-1 latency and reactivation and affects viral yields and viral DNA replication in cell culture in a cell type-specific manner.
A recombinant virus of HSV-1 lacking miR-H11 demonstrated a decrease in viral DNA and protein synthesis, as well as viral yields, by expressing VP55, a vaccinia virus protein that adds nontemplated adenosines specifically to the miRNAs and degrades those miRNAs [73]. These results indicate that HSV-1 miR-H11 may play a role in promoting productive viral infection by some unknown mechanism.
HSV-1 miR-H26 (named miR-92944 in the original article) is located in the sense orientation of the 5′-UTR of the UL42 gene. Viral mutants that do not produce miR-H26 exhibited significant reductions in viral titers in both single and multistep growth analyses and a fourfold reduction in plaque size [32]. This finding indicated that it significantly aided viral lytic replication in the early stages of infection in cell culture.
HSV-1 miR-H27, located in the LAT 3p exon and antisense to miR-H7, directly targeted Kelch-like 24 (KLHL24) mRNA 3′-UTR. Transfection of miR-H27-3p inhibitor decreases viral transcriptional efficiency and replication in HSV-1-infected Vero cells [33]. This discovery revealed that HSV-1-encoded miR-H27 aided its replication and proliferation by downregulating the expression of a cellular transcriptional repressor of viral genes (KLHL24).
Han et al. reported two HSV-1-encoded miRNAs, designated miR-H28 and miR-H29, in late productively infected HEK-293T and HEp-2 cells with HSV-1 strain F, both of which were absent in mouse TG in latency but accumulated in TG after reactivation. Ectopic expression of miRNA mimics in human cells before infection reduced the accumulation of viral mRNAs and proteins, plaque sizes, and viral yields at low MOI [34]. This could be a mechanism by which the virus reduces the size of primary or recurrent lesions and enhances person-to-person transmission via miRNA-mediated replication regulation.
Other Functions of HSV-1 miRNAs
Most HSV-1-encoded miRNAs participate in the regulation of host innate immune response as well as viral lytic and latent infection and reactivation, as previously described, whereas viral miRNAs can regulate different host or viral biological processes via targeting various targets. HSV-1 miR-H1-5p has been shown to target ubiquitin protein ligase E3 component n-recognin 1 (Ubr1), which is a RING-type E3 ubiquitin ligase of the Arg/N-end rule pathway that causes the degradation of proteins bearing “destabilizing” N-terminal residues, such as neurodegeneration-associated protein fragment β-Amyloid. Additionally, miR-H1-5p significantly repressed the expression and activity of Ubr1, resulting in β-Amyloid accumulation [74]. This result showed that miR-H1-5p might contribute to neurodegenerative pathogenesis.
Transforming growth factor-β1-SMAD (TGF-β1-SMAD) signaling regulates various biological processes, including cell growth, differentiation, matrix production, migration, and apoptosis [75]. Reportedly, HSV-1 infection suppressed TGF-β1 and SMAD3 expression in human cornea epithelial cells [76]. A recent study has demonstrated that miR-H2-3p binds to both SMAD mRNAs, whereas miR-H3-3p and miR-H4-5p only inhibit the expression of SMAD4 and SMAD3, respectively [77]. These HSV-1-encoded miRNAs may be involved in the regulation of TGF-β1-SMAD signaling. HSV-1-encoded miR-H4-5p has been shown to exert pro-proliferative functions by inhibiting its target gene, CDKN2A, and regulating CDKN2A-related PI3K-Akt pathways [78].
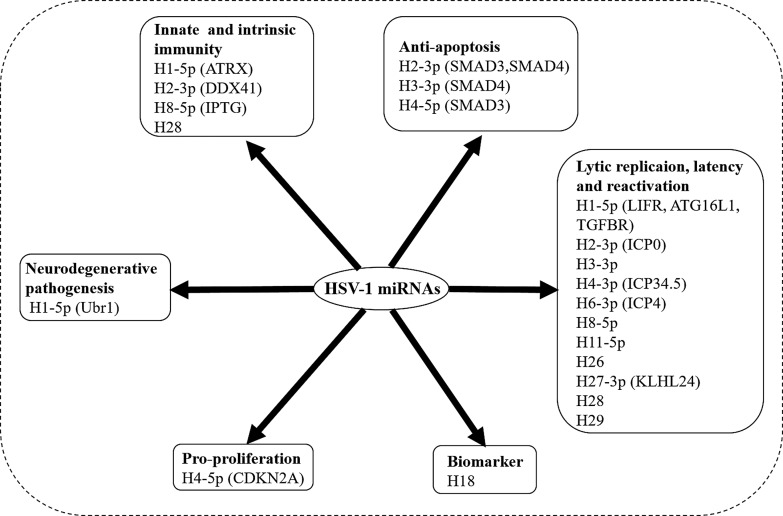
HSV-1-encoded miR-H18, a miRNA with uncharacterized functions, is expressed in urine samples from prostate cancer patients and may be applied as a diagnostic marker for prostate cancer in clinical practice [79]. It is worth mentioning that two small noncoding RNAs, sncRNA1 and sncRNA2 [36], located within the region of the first 1.5 kb of LAT, can inhibit HSV-1 productive infection, cooperate to inhibit host cell apoptosis [80], and cooperate with RIG-I to induce IFN-β promoter activity and promote cell survival [81], as well as promote herpesvirus entry mediator (TNFRSF14) [82] via activating the herpesvirus entry mediator promoter [83]. These results suggest that LAT can regulate virus replication and host cell survival differently from miRNAs. Table 3 and Figure 2, respectively, summarize the identified miRNA function and regulation.
Table 3.
Function of HSV-1-encoded miRNAs
| miRNAs | Targets | Validation method | Functions | Models | References |
|---|---|---|---|---|---|
| miR-H1-5p | ATRX | Western blotting Luciferase assay |
A component of ND, generated a repressive environment for viral gene expression | HEK-293 | [50] |
| SORT1 | Luciferase assay | Involvement in endocytic and intracellular trafficking pathways | HEK-293 | [49] | |
| Ubr1 | Western blotting Luciferase assay |
A RING-type E3 ubiquitin ligase of the Arg/N-end rule pathway | HEK-293 SHSY5Y |
[73] | |
| ND | ND | Inhibition of viral yields in vitro and increased reactivation from latency | RS Neuro-2A |
[70] | |
| LIFR | Luciferase assay | Inhibition of HSV-1 entry and infection in host cells | HOK | [51] | |
| ATG16L1 | |||||
| TGFBR | |||||
| miR-H2-3p | ICP0 | Northern blotting | A key IE HSV-1 transcriptional activator and inhibitor of innate immune responses | HEK-293T | [29] |
| Western blotting | |||||
| ICP0 | Luciferase assay Western blotting |
HEK-239 | [61] | ||
| Neuro-2A | |||||
| NIH3T3 | |||||
| ICP0 | Western blotting | RS | [62, 63] | ||
| ICP0 | Western blotting | HEK-293T Vero | [64] | ||
| Neuro-2A | |||||
| DDX41 | Luciferase assay | A cytosolic DNA sensor, involved in the induction of IFNs | THP-1 | [52] | |
| Western blotting | |||||
| SMAD3 | Luciferase assay | Involvement in TGF-β/SMAD signaling | LX-2 | [76] | |
| SMAD4 | HEK-293T | ||||
| miR-H3-3p | ND | ND | The regulation of HSV-1 replication in neuronal cells | HEK-239 | [61] |
| Neuro-2A | |||||
| NIH3T3 | |||||
| SMAD4 | Luciferase assay | Involvement in TGF-β/SMAD signaling | LX-2 | [76] | |
| HEK-293T | |||||
| miR-H4-3p | ICP34.5 | Luciferase assay Western blotting |
A neurovirulence protein that inhibits innate immune responses | HEK-239 | [61] |
| Neuro-2A | |||||
| NIH3T3 | |||||
| miR-H4-5p | CDKN2A | Luciferase assay Western blotting | Cyclin-dependent kinase inhibitor | SHSY5Y | [77] |
| SMAD3 | Luciferase assay | Involvement in TGF-β/SMAD signaling | LX-2 | [76] | |
| HEK-293T | |||||
| miR-H6-3p | ICP4 | RNAse protection analysis | A transcription factor required for the expression of most HSV-1 genes during productive infection | HEK-293T | [29] |
| Western blotting | |||||
| ICP4 | Western blotting | HCE | [68] | ||
| ND | ND | Inhibition of viral yields in vitro and increased reactivation from latency | Murine Rabbit |
[70] | |
| RS | |||||
| Neuro-2A | |||||
| miR-H6-5p | ND | ND | Promotion of viral protein levels and virus yield | HEp-2 | [69] |
| miR-H8-5p | PIGT | FACS | GPI anchoring pathway involved in NK cells activation | BJAB | [53] |
| Luciferase assay | |||||
| ND | ND | Regulation of viral yields and viral DNA replication in cell culture | Murine Rabbit |
[71] | |
| RS | |||||
| Neuro-2A LUHMES |
|||||
| miR-H11-5p | ND | ND | Promotion of viral DNA and protein synthesis and viral yields and spreading | HEp-2 | [72] |
| miR-H18 | ND | ND | As a diagnostic marker for prostate cancer | Biological fluids | [78] |
| miR-H26 | ND | ND | Promotion of viral titer in the early stage of lytic infection | HEK-293 | [31] |
| miR-H27-3p | KLHL24 | GFP intensity assay Western blotting |
A cellular transcriptional repressor of viral genes | Vero | [32] |
| miR-H28 | ND | ND | Inducing production of IFN-γ, inhibition of viral spread, and replication | HEK-293T HEp-2 | [33, 54] |
| miR-H29 | ND | ND | Inhibition of viral spread and replication | HEK-293T | [33] |
| HEp-2 |
RS, rabbit skin; HOK, human oral keratinocytes; FACS, fluorescence-activated cell sorting; HCE, human cornea epithelial; LUHMES, Lund human mesencephalic.
Fig. 2.
Schematics of viral miRNAs in cellular pathway and viral life cycle. Regulation of miRNAs can be sorted by cellular pathways, including innate and intrinsic immunity, anti-apoptosis, pro-proliferation, neurodegenerative pathogenesis, and biomarker; and viral life cycles, including lytic replication, latency, and reactivation. If the target gene of corresponding miRNAs is identified, it is shown in brackets after it.
Conclusion
MiRNAs as regulatory factors for virus-encoded genes have several obvious advantages, including utilizing small genomic coding resources, targeting multiple genes, and lacking immunogenicity. However, because of their ability to fine-tune target genes at the posttranscriptional and/or translational levels, miRNAs cannot play a dominant role in biological processes and must instead collaborate with proteins to perform specific functions. Since the discovery of the first HSV-1-encoded miRNAs in 2005, there has been rapid progress in understanding their expression, functions, and mechanisms. Previously reported LAT functions, such as anti-apoptosis [80, 84–86] and immune modulation [87, 88], may be performed by LAT-derived miRNAs, as LAT-encoded miRNAs have been shown to perform these functions. Besides using self-encoded miRNAs, HSV-1 can use host cell infection-regulated or cell-specific miRNAs to regulate its own and/or host physiological processes to favor its survival, making up for the lack of self-encoded miRNAs [89–103]. Prediction and identification of the target genes of miRNAs is a crucial component of studies on the mechanisms of miRNAs, although some of these studies do not identify their direct targets. Bioinformatics prediction is optional but not optimal due to many false-positive results, which may bring great difficulties to further biochemical identification [104]. High-throughput and reliable biochemical techniques for target identification have been developed, such as ribonucleoprotein immunoprecipitation microarray (RIP-Chip) [105], high-throughput sequencing of RNAs isolated by cross-linking immunoprecipitation [106], and photoactivatable ribonucleoside-enhanced cross-linking and immunoprecipitation [107, 108], which was used to identify EBV, KSHV, and HCMV miRNA targets [109–116]. As a promising candidate for the clinical application of small molecule drugs, miRNAs can be developed into an anti-HSV-1 drug with an in-depth study of the precise mechanism of action. A study confirmed that miRNAs targeting ICP4 can inhibit HSV-1 replication [117], which may provide ideas for the development of specific antiviral drugs to become a long-term clinical reality. Furthermore, miRNAs have several advantages as molecular biomarkers [118, 119]. Hence, HSV-1 miRNA may be used as a potential diagnostic and prognostic marker for diseases.
Acknowledgments
The authors apologize to all the researchers whose work they cannot cite here owing to significant space constraints.
Conflict of Interest Statement
The authors declare that they have no competing interests.
Funding Sources
This work was supported by grants from the National Natural Sciences Foundation of China (NSFC, Grant No. 31670173), the Chinese Academy of Medical Sciences Initiative for Innovative Medicine (2016-I2M-1-019), the Major Science and Technology Projects of Yunnan Province (2017ZF020, 2017ZF006), the Scientific Research Fund Project of Yunnan Provincial Department of Education (2020J0152), and the Joint Special Project of Yunnan Provincial Department of Science and Technology – Kunming Medical University for Applied Basic Research (202001AY070001-176).
Author Contributions
Conceptualization: Yongzhong Duan and Le Sun; manuscript: Yongzhong Duan; funding acquisition: Yongzhong Duan and Qihan Li; supervision and approval: Qihan Li.
Funding Statement
This work was supported by grants from the National Natural Sciences Foundation of China (NSFC, Grant No. 31670173), the Chinese Academy of Medical Sciences Initiative for Innovative Medicine (2016-I2M-1-019), the Major Science and Technology Projects of Yunnan Province (2017ZF020, 2017ZF006), the Scientific Research Fund Project of Yunnan Provincial Department of Education (2020J0152), and the Joint Special Project of Yunnan Provincial Department of Science and Technology – Kunming Medical University for Applied Basic Research (202001AY070001-176).
References
- 1. Fan S, Cai H, Xu X, Feng M, Wang L, Liao Y, et al. The characteristics of herpes simplex virus type 1 infection in rhesus macaques and the associated pathological features. Viruses. 2017;9(2):26. 10.3390/v9020026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Li L, Li Z, Wang E, Yang R, Xiao Y, Han H, et al. Herpes simplex virus 1 infection of tree shrews differs from that of mice in the severity of acute infection and viral transcription in the peripheral nervous system. J Virol. 2016;90(2):790–804. 10.1128/JVI.02258-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Chentoufi AA, Dhanushkodi NR, Srivastava R, Prakash S, Coulon PGA, Zayou L, et al. Combinatorial herpes simplex vaccine strategies: from bedside to bench and back. Front Immunol. 2022;13:849515. 10.3389/fimmu.2022.849515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Xu X, Zhang Y, Li Q. Characteristics of herpes simplex virus infection and pathogenesis suggest a strategy for vaccine development. Rev Med Virol. 2019;29(4):e2054. 10.1002/rmv.2054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Bloom DC, Giordani NV, Kwiatkowski DL. Epigenetic regulation of latent HSV-1 gene expression. Biochim Biophys Acta. 2010;1799(3–4):246–56. 10.1016/j.bbagrm.2009.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Mehta A, Maggioncalda J, Bagasra O, Thikkavarapu S, Saikumari P, Valyi-Nagy T, et al. In situ DNA PCR and RNA hybridization detection of herpes simplex virus sequences in trigeminal ganglia of latently infected mice. Virology. 1995;206(1):633–40. 10.1016/s0042-6822(95)80080-8. [DOI] [PubMed] [Google Scholar]
- 7. Hill JM, Gebhardt BM, Wen R, Bouterie AM, Thompson HW, O’Callaghan RJ, et al. Quantitation of herpes simplex virus type 1 DNA and latency-associated transcripts in rabbit trigeminal ganglia demonstrates a stable reservoir of viral nucleic acids during latency. J Virol. 1996;70(5):3137–41. 10.1128/JVI.70.5.3137-3141.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Honess RW, Roizman B. Regulation of herpesvirus macromolecular synthesis. I. Cascade regulation of the synthesis of three groups of viral proteins. J Virol. 1974;14(1):8–19. 10.1128/JVI.14.1.8-19.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Pereira L, Wolff MH, Fenwick M, Roizman B. Regulation of herpesvirus macromolecular synthesis. V. Properties of alpha polypeptides made in HSV-1 and HSV-2 infected cells. Virology. 1977;77(2):733–49. 10.1016/0042-6822(77)90495-0. [DOI] [PubMed] [Google Scholar]
- 10. Ramalingam D, Ziegelbauer JM. Viral microRNAs target a gene network, inhibit STAT activation, and suppress interferon responses. Sci Rep. 2017;7:40813. 10.1038/srep40813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bartel DP. Metazoan MicroRNAs. Cell. 2018;173(1):20–51. 10.1016/j.cell.2018.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Gallaher AM, Das S, Xiao Z, Andresson T, Kieffer-Kwon P, Happel C, et al. Proteomic screening of human targets of viral microRNAs reveals functions associated with immune evasion and angiogenesis. PLoS Pathog. 2013;9(9):e1003584. 10.1371/journal.ppat.1003584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Boss IW, Renne R. Viral miRNAs and immune evasion. Biochim Biophys Acta. 2011;1809(11–12):708–14. 10.1016/j.bbagrm.2011.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Grundhoff A, Sullivan CS. Virus-encoded microRNAs. Virology. 2011;411(2):325–43. 10.1016/j.virol.2011.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Pfeffer S, Zavolan M, Grässer FA, Chien M, Russo JJ, Ju J, et al. Identification of virus-encoded microRNAs. Science. 2004;304(5671):734–6. 10.1126/science.1096781. [DOI] [PubMed] [Google Scholar]
- 16. Pfeffer S, Sewer A, Lagos-Quintana M, Sheridan R, Sander C, Grässer FA, et al. Identification of microRNAs of the herpesvirus family. Nat Methods. 2005;2(4):269–76. 10.1038/nmeth746. [DOI] [PubMed] [Google Scholar]
- 17. Dunn W, Trang P, Zhong Q, Yang E, van Belle C, Liu F. Human cytomegalovirus expresses novel microRNAs during productive viral infection. Cell Microbiol. 2005;7(11):1684–95. 10.1111/j.1462-5822.2005.00598.x. [DOI] [PubMed] [Google Scholar]
- 18. Grey F, Antoniewicz A, Allen E, Saugstad J, McShea A, Carrington JC, et al. Identification and characterization of human cytomegalovirus-encoded MicroRNAs. J Virol. 2005;79(18):12095–9. 10.1128/JVI.79.18.12095-12099.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Cui C, Griffiths A, Li G, Silva LM, Kramer MF, Gaasterland T, et al. Prediction and identification of herpes simplex virus 1-encoded microRNAs. J Virol. 2006;80(11):5499–508. 10.1128/JVI.00200-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Umbach JL, Nagel MA, Cohrs RJ, Gilden DH, Cullen BR. Analysis of human alphaherpesvirus microRNA expression in latently infected human trigeminal ganglia. J Virol. 2009;83(20):10677–83. 10.1128/JVI.01185-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Umbach JL, Wang K, Tang S, Krause PR, Mont EK, Cohen JI, et al. Identification of viral microRNAs expressed in human sacral ganglia latently infected with herpes simplex virus 2. J Virol. 2010;84(2):1189–92. 10.1128/JVI.01712-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Cai X, Lu S, Zhang Z, Gonzalez CM, Damania B, Cullen BR. Kaposi’s sarcoma-associated herpesvirus expresses an array of viral microRNAs in latently infected cells. Proc Natl Acad Sci U S A. 2005;102(15):5570–5. 10.1073/pnas.0408192102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Samols MA, Hu J, Skalsky RL, Renne R. Cloning and identification of a microRNA cluster within the latency-associated region of Kaposi's sarcoma-associated herpesvirus. J Virol. 2005;79(14):9301–5. 10.1128/JVI.79.14.9301-9305.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kim H, Iizasa H, Kanehiro Y, Fekadu S, Yoshiyama H. Herpesviral microRNAs in cellular metabolism and immune responses. Front Microbiol. 2017;8:1318. 10.3389/fmicb.2017.01318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136(2):215–33. 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993;75(5):843–54. 10.1016/0092-8674(93)90529-y. [DOI] [PubMed] [Google Scholar]
- 27. Moss EG, Lee RC, Ambros V. The cold shock domain protein LIN-28 controls developmental timing in C. elegans and is regulated by the lin-4 RNA. Cell. 1997;88(5):637–46. 10.1016/s0092-8674(00)81906-6. [DOI] [PubMed] [Google Scholar]
- 28. Pasquinelli AE, Reinhart BJ, Slack F, Martindale MQ, Kuroda MI, Maller B, et al. Conservation of the sequence and temporal expression of let-7 heterochronic regulatory RNA. Nature. 2000;408(6808):86–9. 10.1038/35040556. [DOI] [PubMed] [Google Scholar]
- 29. Reinhart BJ, Slack FJ, Basson M, Pasquinelli AE, Bettinger JC, Rougvie AE, et al. The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature. 2000;403(6772):901–6. 10.1038/35002607. [DOI] [PubMed] [Google Scholar]
- 30. Umbach JL, Kramer MF, Jurak I, Karnowski HW, Coen DM, Cullen BR. MicroRNAs expressed by herpes simplex virus 1 during latent infection regulate viral mRNAs. Nature. 2008;454(7205):780–3. 10.1038/nature07103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Jurak I, Kramer MF, Mellor JC, van Lint AL, Roth FP, Knipe DM, et al. Numerous conserved and divergent microRNAs expressed by herpes simplex viruses 1 and 2. J Virol. 2010;84(9):4659–72. 10.1128/JVI.02725-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Munson DJ, Burch AD. A novel miRNA produced during lytic HSV-1 infection is important for efficient replication in tissue culture. Arch Virol. 2012;157(9):1677–88. 10.1007/s00705-012-1345-4. [DOI] [PubMed] [Google Scholar]
- 33. Wu W, Guo Z, Zhang X, Guo L, Liu L, Liao Y, et al. A microRNA encoded by HSV-1 inhibits a cellular transcriptional repressor of viral immediate early and early genes. Sci China Life Sci. 2013;56(4):373–83. 10.1007/s11427-013-4458-4. [DOI] [PubMed] [Google Scholar]
- 34. Han Z, Liu X, Chen X, Zhou X, Du T, Roizman B, et al. miR-H28 and miR-H29 expressed late in productive infection are exported and restrict HSV-1 replication and spread in recipient cells. Proc Natl Acad Sci U S A. 2016;113(7):E894–901. 10.1073/pnas.1525674113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Perng GC, Dunkel EC, Geary PA, Slanina SM, Ghiasi H, Kaiwar R, et al. The latency-associated transcript gene of herpes simplex virus type 1 (HSV-1) is required for efficient in vivo spontaneous reactivation of HSV-1 from latency. J Virol. 1994;68(12):8045–55. 10.1128/JVI.68.12.8045-8055.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Peng W, Vitvitskaia O, Carpenter D, Wechsler SL, Jones C. Identification of two small RNAs within the first 1.5-kb of the herpes simplex virus type 1-encoded latency-associated transcript. J Neurovirol. 2008;14(1):41–52. 10.1080/13550280701793957. [DOI] [PubMed] [Google Scholar]
- 37. Bachmann M, Falke D, Preuhs J, Schröder HC, Pfeifer K, Müller WE. Occurrence of novel small RNAs with concomitant inhibition of host cellular U small nuclear RNA synthesis in Vero cells infected with herpes simplex virus type 1. J Gen Virol. 1986;67(Pt 12):2587–94. 10.1099/0022-1317-67-12-2587. [DOI] [PubMed] [Google Scholar]
- 38. Kramer MF, Jurak I, Pesola JM, Boissel S, Knipe DM, Coen DM. Herpes simplex virus 1 microRNAs expressed abundantly during latent infection are not essential for latency in mouse trigeminal ganglia. Virology. 2011;417(2):239–47. 10.1016/j.virol.2011.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Pan D, Li G, Morris-Love J, Qi S, Feng L, Mertens ME, et al. Herpes simplex virus 1 lytic infection blocks MicroRNA (miRNA) biogenesis at the stage of nuclear export of pre-miRNAs. mBio. 2019;10(1):e02856-18. 10.1128/mBio.02856-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Umbach JL, Cullen BR. The role of RNAi and microRNAs in animal virus replication and antiviral immunity. Genes Dev. 2009;23(10):1151–64. 10.1101/gad.1793309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Held K, Junker A, Dornmair K, Meinl E, Sinicina I, Brandt T, et al. Expression of herpes simplex virus 1-encoded microRNAs in human trigeminal ganglia and their relation to local T-cell infiltrates. J Virol. 2011;85(19):9680–5. 10.1128/JVI.00874-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Cokarić Brdovčak M, Zubković A, Ferenčić A, Šoša I, Stemberga V, Cuculić D, et al. Herpes simplex virus 1 miRNA sequence variations in latently infected human trigeminal ganglia. Virus Res. 2018;256:90–5. 10.1016/j.virusres.2018.08.002. [DOI] [PubMed] [Google Scholar]
- 43. Jurak I, Hackenberg M, Kim JY, Pesola JM, Everett RD, Preston CM, et al. Expression of herpes simplex virus 1 microRNAs in cell culture models of quiescent and latent infection. J Virol. 2014;88(4):2337–9. 10.1128/JVI.03486-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Du T, Zhou G, Roizman B. HSV-1 gene expression from reactivated ganglia is disordered and concurrent with suppression of latency-associated transcript and miRNAs. Proc Natl Acad Sci U S A. 2011;108(46):18820–4. 10.1073/pnas.1117203108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Du T, Han Z, Zhou G, Roizman B. Patterns of accumulation of miRNAs encoded by herpes simplex virus during productive infection, latency, and on reactivation. Proc Natl Acad Sci U S A. 2015;112(1):E49–55. 10.1073/pnas.1422657112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Wu J, Chen ZJ. Innate immune sensing and signaling of cytosolic nucleic acids. Annu Rev Immunol. 2014;32:461–88. 10.1146/annurev-immunol-032713-120156. [DOI] [PubMed] [Google Scholar]
- 47. Yan N, Chen ZJ. Intrinsic antiviral immunity. Nat Immunol. 2012;13(3):214–22. 10.1038/ni.2229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Bieniasz PD. Intrinsic immunity: a front-line defense against viral attack. Nat Immunol. 2004;5(11):1109–15. 10.1038/ni1125. [DOI] [PubMed] [Google Scholar]
- 49. Cullen BR. Viral and cellular messenger RNA targets of viral microRNAs. Nature. 2009;457(7228):421–5. 10.1038/nature07757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Naqvi AR, Shango J, Seal A, Shukla D, Nares S. Viral miRNAs alter host cell miRNA profiles and modulate innate immune responses. Front Immunol. 2018;9:433. 10.3389/fimmu.2018.00433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Jurak I, Silverstein LB, Sharma M, Coen DM. Herpes simplex virus is equipped with RNA- and protein-based mechanisms to repress expression of ATRX, an effector of intrinsic immunity. J Virol. 2012;86(18):10093–102. 10.1128/JVI.00930-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Naqvi AR, Seal A, Shango J, Brambila MF, Martinez G, Chapa G, et al. Herpesvirus-encoded microRNAs detected in human gingiva alter host cell transcriptome and regulate viral infection. Biochim Biophys Acta Gene Regul Mech. 2018;1861(5):497–508. 10.1016/j.bbagrm.2018.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Duan Y, Zeng J, Fan S, Liao Y, Feng M, Wang L, et al. Herpes simplex virus type 1-encoded miR-H2-3p manipulates cytosolic DNA-stimulated antiviral innate immune response by targeting DDX41. Viruses. 2019;11(8):756. 10.3390/v11080756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Enk J, Levi A, Weisblum Y, Yamin R, Charpak-Amikam Y, Wolf DG, et al. HSV1 MicroRNA modulation of GPI anchoring and downstream immune evasion. Cell Rep. 2016;17(4):949–56. 10.1016/j.celrep.2016.09.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Huang R, Wu J, Zhou X, Jiang H, Guoying Zhou G, Roizman B. Herpes simplex virus 1 MicroRNA miR-H28 exported to uninfected cells in exosomes restricts cell-to-cell virus spread by inducing gamma interferon mRNA. J Virol. 2019;93(21):e01005-19. 10.1128/JVI.01005-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Everett RD. ICP0, a regulator of herpes simplex virus during lytic and latent infection. Bioessays. 2000;22(8):761–70. . [DOI] [PubMed] [Google Scholar]
- 57. Hagglund R, Roizman B. Role of ICP0 in the strategy of conquest of the host cell by herpes simplex virus 1. J Virol. 2004;78(5):2169–78. 10.1128/jvi.78.5.2169-2178.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Everett RD, Parsy ML, Orr A. Analysis of the functions of herpes simplex virus type 1 regulatory protein ICP0 that are critical for lytic infection and derepression of quiescent viral genomes. J Virol. 2009;83(10):4963–77. 10.1128/JVI.02593-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Rodríguez MC, Dybas JM, Hughes J, Weitzman MD, Boutell C. The HSV-1 ubiquitin ligase ICP0: modifying the cellular proteome to promote infection. Virus Res. 2020;285:198015. 10.1016/j.virusres.2020.198015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Cai W, Schaffer PA. Herpes simplex virus type 1 ICP0 regulates expression of immediate-early, early, and late genes in productively infected cells. J Virol. 1992;66(5):2904–15. 10.1128/JVI.66.5.2904-2915.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Cai W, Astor TL, Liptak LM, Cho C, Coen DM, Schaffer PA. The herpes simplex virus type 1 regulatory protein ICP0 enhances virus replication during acute infection and reactivation from latency. J Virol. 1993;67(12):7501–12. 10.1128/JVI.67.12.7501-7512.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Flores O, Nakayama S, Whisnant AW, Javanbakht H, Cullen BR, Bloom DC. Mutational inactivation of herpes simplex virus 1 microRNAs identifies viral mRNA targets and reveals phenotypic effects in culture. J Virol. 2013;87(12):6589–603. 10.1128/JVI.00504-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Jiang X, Brown D, Osorio N, Hsiang C, Li L, Chan L, et al. A herpes simplex virus type 1 mutant disrupted for microRNA H2 with increased neurovirulence and rate of reactivation. J Neurovirol. 2015;21(2):199–209. 10.1007/s13365-015-0319-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Jiang X, Brown D, Osorio N, Hsiang C, BenMohamed L, Wechsler SL. Increased neurovirulence and reactivation of the herpes simplex virus type 1 latency-associated transcript (LAT)-negative mutant dLAT2903 with a disrupted LAT miR-H2. J Neurovirol. 2016;22(1):38–49. 10.1007/s13365-015-0362-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Pan D, Pesola JM, Li G, McCarron S, Coen DM. Mutations inactivating herpes simplex virus 1 MicroRNA miR-H2 do not detectably increase ICP0 gene expression in infected cultured cells or mouse trigeminal ganglia. J Virol. 2017;91(2):e02001-16. 10.1128/JVI.02001-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. He B, Gross M, Roizman B. The gamma(1)34.5 protein of herpes simplex virus 1 complexes with protein phosphatase 1alpha to dephosphorylate the alpha subunit of the eukaryotic translation initiation factor 2 and preclude the shutoff of protein synthesis by double-stranded RNA-activated protein kinase. Proc Natl Acad Sci U S A. 1997;94(3):843–8. 10.1073/pnas.94.3.843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Pan S, Liu X, Ma Y, Cao Y, He B. Herpes simplex virus 1 γ(1)34.5 protein inhibits STING activation that restricts viral replication. J Virol. 2018;92(20):e01015-18. 10.1128/JVI.01015-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Orvedahl A, Alexander D, Tallóczy Z, Sun Q, Wei Y, Zhang W, et al. HSV-1 ICP34.5 confers neurovirulence by targeting the Beclin 1 autophagy protein. Cell Host Microbe. 2007;1(1):23–35. 10.1016/j.chom.2006.12.001. [DOI] [PubMed] [Google Scholar]
- 69. Duan F, Liao J, Huang Q, Nie Y, Wu K. HSV-1 miR-H6 inhibits HSV-1 replication and IL-6 expression in human corneal epithelial cells in vitro. Clin Dev Immunol. 2012;2012:192791. 10.1155/2012/192791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Huang R, Zhou X, Ren S, Liu X, Han Z, Zhou GG. Effect of loss-of-function of the Herpes simplex virus-1 microRNA H6-5p on virus replication. Virol Sin. 2019 Aug;34(4):386–96. 10.1007/s12250-019-00111-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Barrozo ER, Nakayama S, Singh P, Vanni EAH, Arvin AM, Neumann DM, et al. Deletion of herpes simplex virus 1 MicroRNAs miR-H1 and miR-H6 impairs reactivation. J Virol. 2020;94(15):e00639-20. 10.1128/JVI.00639-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Barrozo ER, Nakayama S, Singh P, Neumann DM, Bloom DC. Herpes simplex virus 1 MicroRNA miR-H8 is dispensable for latency and reactivation in vivo. J Virol. 2021;95(4):e02179-20. 10.1128/JVI.02179-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Zou W, Zhou X, Wang L, Zhou GG, Chen X. Degradation of herpes simplex virus-1 viral miRNA H11 by vaccinia virus protein VP55 attenuates viral replication. Front Microbiol. 2020;11:717. 10.3389/fmicb.2020.00717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Zheng K, Liu Q, Wang S, Ren Z, Kitazato K, Yang D, et al. HSV-1-encoded microRNA miR-H1 targets Ubr1 to promote accumulation of neurodegeneration-associated protein. Virus Genes. 2018;54(3):343–50. 10.1007/s11262-018-1551-6. [DOI] [PubMed] [Google Scholar]
- 75. Heldin CH, Miyazono K, ten Dijke P. TGF-beta signalling from cell membrane to nucleus through SMAD proteins. Nature. 1997;390(6659):465–71. 10.1038/37284. [DOI] [PubMed] [Google Scholar]
- 76. Nie Y, Cui D, Pan Z, Deng J, Huang Q, Wu K. HSV-1 infection suppresses TGF-beta1 and SMAD3 expression in human corneal epithelial cells. Mol Vis. 2008;14:1631–8. [PMC free article] [PubMed] [Google Scholar]
- 77. Shojaei Jeshvaghani Z, Arefian E, Asgharpour S, Soleimani M. Latency-associated transcript-derived MicroRNAs in herpes simplex virus type 1 target SMAD3 and SMAD4 in TGF-β/smad signaling pathway. Iran Biomed J. 2021;25(3):169–79. 10.29252/ibj.25.3.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Zhao H, Zhang C, Hou G, Song J. MicroRNA-H4-5p encoded by HSV-1 latency-associated transcript promotes cell proliferation, invasion and cell cycle progression via p16-mediated PI3K-Akt signaling pathway in SHSY5Y cells. Int J Clin Exp Med. 2015;8(5):7526–34. [PMC free article] [PubMed] [Google Scholar]
- 79. Yun SJ, Jeong P, Kang HW, Kim YH, Kim EA, Yan C, et al. Urinary MicroRNAs of prostate cancer: virus-encoded hsv1-miR-H18 and hsv2-miR-H9-5p could Be valuable diagnostic markers. Int Neurourol J. 2015;19(2):74–84. 10.5213/inj.2015.19.2.74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Shen W, Sa e Silva M, Jaber T, Vitvitskaia O, Li S, Henderson G, et al. Two small RNAs encoded within the first 1.5 kilobases of the herpes simplex virus type 1 latency-associated transcript can inhibit productive infection and cooperate to inhibit apoptosis. J Virol. 2009;83(18):9131–9. 10.1128/JVI.00871-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. da Silva LF, Jones C. Small non-coding RNAs encoded within the herpes simplex virus type 1 latency associated transcript (LAT) cooperate with the retinoic acid inducible gene I (RIG-I) to induce beta-interferon promoter activity and promote cell survival. Virus Res. 2013;175(2):101–9. 10.1016/j.virusres.2013.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Allen SJ, Rhode-Kurnow A, Mott KR, Jiang X, Carpenter D, Rodriguez-Barbosa JI, et al. Interactions between herpesvirus entry mediator (TNFRSF14) and latency-associated transcript during herpes simplex virus 1 latency. J Virol. 2014;88(4):1961–71. 10.1128/JVI.02467-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Tormanen K, Wang S, Matundan HH, Yu J, Jaggi U, Ghiasi H. Herpes simplex virus 1 small noncoding RNAs 1 and 2 activate the herpesvirus entry mediator promoter. J Virol. 2022;96(3):Jvi0198521. 10.1128/JVI.01985-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Carpenter D, Hsiang C, Brown DJ, Jin L, Osorio N, BenMohamed L, et al. Stable cell lines expressing high levels of the herpes simplex virus type 1 LAT are refractory to caspase 3 activation and DNA laddering following cold shock induced apoptosis. Virology. 2007;369(1):12–8. 10.1016/j.virol.2007.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Carpenter D, Hsiang C, Jiang X, Osorio N, BenMohamed L, Jones C, et al. The herpes simplex virus type 1 (HSV-1) latency-associated transcript (LAT) protects cells against cold-shock-induced apoptosis by maintaining phosphorylation of protein kinase B (AKT). J Neurovirol. 2015;21(5):568–75. 10.1007/s13365-015-0361-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Tormanen K, Allen S, Mott KR, Ghiasi H. The latency-associated transcript inhibits apoptosis via downregulation of components of the type I interferon pathway during latent herpes simplex virus 1 ocular infection. J Virol. 2019;93(10):e00103-19. 10.1128/JVI.00103-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Srivastava R, Dervillez X, Khan AA, Chentoufi AA, Chilukuri S, Shukr N, et al. The herpes simplex virus latency-associated transcript gene is associated with a broader repertoire of virus-specific exhausted CD8+ T cells retained within the trigeminal ganglia of latently infected HLA transgenic rabbits. J Virol. 2016;90(8):3913–28. 10.1128/JVI.02450-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Chentoufi AA, Dervillez X, Dasgupta G, Nguyen C, Kabbara KW, Jiang X, et al. The herpes simplex virus type 1 latency-associated transcript inhibits phenotypic and functional maturation of dendritic cells. Viral Immunol. 2012;25(3):204–15. 10.1089/vim.2011.0091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Ru J, Sun H, Fan H, Wang C, Li Y, Liu M, et al. MiR-23a facilitates the replication of HSV-1 through the suppression of interferon regulatory factor 1. PLoS One. 2014;9(12):e114021. 10.1371/journal.pone.0114021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Zhang Y, Dai J, Tang J, Zhou L, Zhou M. MicroRNA-649 promotes HSV-1 replication by directly targeting MALT1. J Med Virol. 2017;89(6):1069–79. 10.1002/jmv.24728. [DOI] [PubMed] [Google Scholar]
- 91. Zheng SQ, Li Y, Zhang Y, Li X, Tang H. MiR-101 regulates HSV-1 replication by targeting ATP5B. Antiviral Res. 2011;89(3):219–26. 10.1016/j.antiviral.2011.01.008. [DOI] [PubMed] [Google Scholar]
- 92. Wang X, Diao C, Yang X, Yang Z, Liu M, Li X, et al. ICP4-induced miR-101 attenuates HSV-1 replication. Sci Rep. 2016;6:23205. 10.1038/srep23205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Mulik S, Xu J, Reddy PBJ, Rajasagi NK, Gimenez F, Sharma S, et al. Role of miR-132 in angiogenesis after ocular infection with herpes simplex virus. Am J Pathol. 2012;181(2):525–34. 10.1016/j.ajpath.2012.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Lagos D, Pollara G, Henderson S, Gratrix F, Fabani M, Milne RSB, et al. miR-132 regulates antiviral innate immunity through suppression of the p300 transcriptional co-activator. Nat Cell Biol. 2010;12(5):513–9. 10.1038/ncb2054. [DOI] [PubMed] [Google Scholar]
- 95. Hill JM, Zhao Y, Clement C, Neumann DM, Lukiw WJ. HSV-1 infection of human brain cells induces miRNA-146a and Alzheimer-type inflammatory signaling. Neuroreport. 2009;20(16):1500–5. 10.1097/WNR.0b013e3283329c05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Xie Y, He S, Wang J. MicroRNA-373 facilitates HSV-1 replication through suppression of type I IFN response by targeting IRF1. Biomed Pharmacother. 2018;97:1409–16. 10.1016/j.biopha.2017.11.071. [DOI] [PubMed] [Google Scholar]
- 97. Bhela S, Mulik S, Gimenez F, Reddy PBJ, Richardson RL, Varanasi SK, et al. Role of miR-155 in the pathogenesis of herpetic stromal keratitis. Am J Pathol. 2015;185(4):1073–84. 10.1016/j.ajpath.2014.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Majer A, Caligiuri KA, Gale KK, Niu Y, Phillipson CS, Booth TF, et al. Induction of multiple miR-200/182 members in the brains of mice are associated with acute herpes simplex virus 1 encephalitis. PLoS One. 2017;12(1):e0169081. 10.1371/journal.pone.0169081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Lutz G, Jurak I, Kim ET, Kim JY, Hackenberg M, Leader A, et al. Viral ubiquitin ligase stimulates selective host MicroRNA expression by targeting ZEB transcriptional repressors. Viruses. 2017;9(8):210. 10.3390/v9080210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Sun B, Yang X, Hou F, Yu X, Wang Q, Oh HS, et al. Regulation of host and virus genes by neuronal miR-138 favours herpes simplex virus 1 latency. Nat Microbiol. 2021;6(5):682–96. 10.1038/s41564-020-00860-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Cokarić Brdovčak M, Zubković A, Jurak I. Herpes simplex virus 1 deregulation of host MicroRNAs. Noncoding RNA. 2018;4(4):36. 10.3390/ncrna4040036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Sharma N, Wang C, Kessler P, Sen GC. Herpes simplex virus 1 evades cellular antiviral response by inducing microRNA-24, which attenuates STING synthesis. PLoS Pathog. 2021;17(9):e1009950. 10.1371/journal.ppat.1009950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Venuti A, Musarra-Pizzo M, Pennisi R, Tankov S, Medici MA, Mastino A, et al. HSV-1\EGFP stimulates miR-146a expression in a NF-κB-dependent manner in monocytic THP-1 cells. Sci Rep. 2019;9(1):5157. 10.1038/s41598-019-41530-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Haecker I, Renne R. HITS-CLIP and PAR-CLIP advance viral miRNA targetome analysis. Crit Rev Eukaryot Gene Expr. 2014;24(2):101–16. 10.1615/critreveukaryotgeneexpr.2014006367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Keene JD, Komisarow JM, Friedersdorf MB. RIP-Chip: the isolation and identification of mRNAs, microRNAs and protein components of ribonucleoprotein complexes from cell extracts. Nat Protoc. 2006;1(1):302–7. 10.1038/nprot.2006.47. [DOI] [PubMed] [Google Scholar]
- 106. Chi SW, Zang JB, Mele A, Darnell RB. Argonaute HITS-CLIP decodes microRNA-mRNA interaction maps. Nature. 2009;460(7254):479–86. 10.1038/nature08170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Hafner M, Landthaler M, Burger L, Khorshid M, Hausser J, Berninger P, et al. PAR-CliP--a method to identify transcriptome-wide the binding sites of RNA binding proteins. J Vis Exp. 2010;41:2034. 10.3791/2034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Hafner M, Landthaler M, Burger L, Khorshid M, Hausser J, Berninger P, et al. Transcriptome-wide identification of RNA-binding protein and MicroRNA target sites by PAR-CLIP. Cell. 2010;141(1):129–41. 10.1016/j.cell.2010.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Malterer G, Dölken L, Haas J. The miRNA-targetome of KSHV and EBV in human B-cells. RNA Biol. 2011;8(1):30–4. 10.4161/rna.8.1.13745. [DOI] [PubMed] [Google Scholar]
- 110. Gottwein E, Corcoran DL, Mukherjee N, Skalsky RL, Hafner M, Nusbaum JD, et al. Viral microRNA targetome of KSHV-infected primary effusion lymphoma cell lines. Cell Host Microbe. 2011;10(5):515–26. 10.1016/j.chom.2011.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Haecker I, Gay LA, Yang Y, Hu J, Morse AM, McIntyre LM, et al. Ago HITS-CLIP expands understanding of Kaposi’s sarcoma-associated herpesvirus miRNA function in primary effusion lymphomas. PLoS Pathog. 2012;8(8):e1002884. 10.1371/journal.ppat.1002884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Riley KJ, Rabinowitz GS, Yario TA, Luna JM, Darnell RB, Steitz JA. EBV and human microRNAs co-target oncogenic and apoptotic viral and human genes during latency. EMBO J. 2012;31(9):2207–21. 10.1038/emboj.2012.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Skalsky RL, Corcoran DL, Gottwein E, Frank CL, Kang D, Hafner M, et al. The viral and cellular microRNA targetome in lymphoblastoid cell lines. PLoS Pathog. 2012;8(1):e1002484. 10.1371/journal.ppat.1002484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Skinner CM, Ivanov NS, Barr SA, Chen Y, Skalsky RL. An epstein-barr virus MicroRNA blocks interleukin-1 (IL-1) signaling by targeting IL-1 receptor 1. J Virol. 2017;91(21):e00530-17. 10.1128/JVI.00530-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Hook LM, Grey F, Grabski R, Tirabassi R, Doyle T, Hancock M, et al. Cytomegalovirus miRNAs target secretory pathway genes to facilitate formation of the virion assembly compartment and reduce cytokine secretion. Cell Host Microbe. 2014;15(3):363–73. 10.1016/j.chom.2014.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Bouvet M, Voigt S, Tagawa T, Albanese M, Chen YFA, Chen Y, et al. Multiple viral microRNAs regulate interferon release and signaling early during infection with epstein-barr virus. mBio. 2021;12(2):e03440-20. 10.1128/mBio.03440-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Wang L, Chen X, Zhou X, Roizman B, Zhou GG. miRNAs targeting ICP4 and delivered to susceptible cells in exosomes block HSV-1 replication in a dose-dependent manner. Mol Ther. 2018;26(4):1032–9. 10.1016/j.ymthe.2018.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Bhela S, Rouse BT. Are miRNAs critical determinants in herpes simplex virus pathogenesis? Microbes Infect. 2018;20(9–10):461–5. 10.1016/j.micinf.2017.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Louten J, Beach M, Palermino K, Weeks M, Holenstein G. MicroRNAs expressed during viral infection: biomarker potential and therapeutic considerations. Biomark Insights. 2015;10(Suppl 4):25–52. 10.4137/BMI.S29512. [DOI] [PMC free article] [PubMed] [Google Scholar]