Abstract
Background
Due to the high prevalence of resistance to NNRTI-based ART since 2018, consolidated recommendations from the WHO have indicated dolutegravir as the preferred drug of choice for HIV treatment globally. There is a paucity of resistance outcome data from HIV-1 non-B subtypes circulating across West Africa.
Aims
We characterized the mutational profiles of persons living with HIV from a cross-sectional cohort in North-East Nigeria failing a dolutegravir-based ART regimen.
Methods
WGS of plasma samples collected from 61 HIV-1-infected participants following virological failure of dolutegravir-based ART were sequenced using the Illumina platform. Sequencing was successfully completed for samples from 55 participants. Following quality control, 33 full genomes were analysed from participants with a median age of 40 years and median time on ART of 9 years. HIV-1 subtyping was performed using SNAPPy.
Results
Most participants had mutational profiles reflective of exposure to previous first- and second-line ART regimens comprised NRTIs and NNRTIs. More than half of participants had one or more drug resistance-associated mutations (DRMs) affecting susceptibility to NRTIs (17/33; 52%) and NNRTIs (24/33; 73%). Almost a quarter of participants (8/33; 24.4%) had one or more DRMs affecting tenofovir susceptibility. Only one participant, infected with HIV-1 subtype G, had evidence of DRMs affecting dolutegravir susceptibility—this was characterized by the T66A, G118R, E138K and R263K mutations.
Conclusions
This study found a low prevalence of resistance to dolutegravir; the data are therefore supportive of the continual rollout of dolutegravir as the primary first-line regimen for ART-naive participants and the preferred switch to second-line ART across the region. However, population-level, longer-term data collection on dolutegravir outcomes are required to further guide implementation and policy action across the region.
Introduction
Due to the increasing prevalence of pre-treatment NNRTI drug resistance,1–3 the WHO has (since 2018) recommended the use of dolutegravir as the preferred ART drug of choice for both newly diagnosed and individuals transitioning from previous regimens.4 This decision was supported by numerous safety, potency, tolerability and cost-effective characteristics of dolutegravir.5 Since 2019, more than 50 countries across sub-Saharan Africa (SSA) have rolled out (or have plans to roll out) dolutegravir as part of standard treatment. The continuing rollout is aided by the availability of a low-cost, generic fixed-dose co-formulation of tenofovir disoproxil fumarate, lamivudine and dolutegravir, called TLD.6
Dolutegravir-based ART has been commercialized and distributed in Nigeria since late 2019. As of 2020, national Nigerian treatment guidelines recommend the transition to dolutegravir-based ART in both virally suppressed and unsuppressed participants.7 However, due to economic and other factors, there is no routine virological or resistance testing in Nigeria. Therefore, the majority of HIV-1-infected participants transitioned to dolutegravir-based regimens without prior viral load (VL) or resistance testing. Data from the ADVANCE and NAMSAL clinical trials,8,9 which recruited ART-naive participants exclusively in SSA, showed no evidence of emergence of drug resistance-associated mutations (DRMs) amongst participants on dolutegravir-based ART. Data on treatment outcomes of ART-experienced participants transitioning to TLD are limited, though some literature is beginning to emerge.10 Despite this, there are almost no data available from West Africa.
Given the high prevalence of ART resistance amongst treatment-experienced, HIV-1 participants following virological failure whilst on previous first- and second-line ART regimens,11 data on resistance outcomes following long-term use of dolutegravir are highly valuable. In this study, we present resistance outcomes and drug-resistance genotypes using next-generation HIV-1 sequencing, from participants in a small, Nigerian, treatment-experienced cohort failing dolutegravir-based ART following rollout.
Methods
Study population and design
This was a cross-sectional study performed at the University of Maiduguri Teaching Hospital, Borno State, Nigeria, between January and June 2021. Assessing virological and resistance outcomes in this region of Nigeria has been challenging due to ongoing and long-term armed conflict caused by the Boko Haram insurgency, which has turned the area into a conflict zone. Inclusion criteria for this study was as follows: participants with virological failure (two consecutive HIV-1 RNA VL >1000 copies/mL) whilst following a dolutegravir-based ART regimen for >6 months, ≥18 years of age, attending routine clinic visits and able to provide informed consent for participation in the study. All participants included in this study provided informed consent prior to sample and data collection. Available demographic data including age, gender, ART regimen, duration on ART and current CD4 count were collected from clinical files and recorded in Microsoft Excel (Office 365, Microsoft, Redmond, USA).
Laboratory methods
Plasma was separated from whole venous blood in EDTA within 2 h of collection and stored immediately at −80°C. Plasma VL testing and CD4+ counts were performed at the Defence Reference Laboratory, Asokoro Abuja using the COBAS AmpliPrep/COBAS TaqMan HIV type 1 (HIV1) v2.0 test (Roche Diagnostics, Basel, Switzerland). WGS was performed retrospectively using the ve-SEQ-HIV12 protocol on 61 plasma samples with VL > 1000 copies/mL and stored prior to confirmation of VL.
Briefly, total RNA was extracted from plasma samples, washed in ethanol, and eluted using the NUCLISENS easyMAG system (bioMérieux). Libraries were prepared using the SMARTer Stranded Total RNA-Seq Kits v2 (Clontech, Takara Bio) according to the manufacturer’s protocol. Total RNA was denatured, and reverse-transcribed to cDNA and a total of 500 ng of pooled libraries were hybridized to custom HIV-specific biotinylated 120-mer oligonucleotides (xGen Lockdown Probes, Integrated DNA Technologies). Captured libraries were then amplified by PCR to produce a final pool for sequencing with an Illumina MiSeq (San Diego, CA, USA) to produce up to 300-nucletoide paired-end reads.
Bioinformatics analysis
FastQ files were trimmed of all sequencing adapters, and mapped iteratively to the best available reference sequence, from a curated alignment of 3000 HIV-1 genomes downloaded from the Los Alamos HIV sequence database, using SHIVER.13,14 BAM files were quality controlled by determining the read depth at each position of the HIV-1 genome. Sequences with a depth of <50 reads were excluded from further analysis. Resistance genotyping was performed using an in-house script that utilizes the Stanford HIV drug resistance algorithm (v9.1) but is adapted to calculate the prevalence of DRMs from all available sequencing data rather than just consensus-level mutations. The script calls mutations with an absolute minimum read count of 50 reads, and minimum frequency of 2%, 10% and 20%. Haplotypes were reconstructed using CliqueSNV v2.0.315 using the -fdf extended, -m snv-illumina and -tf 0.05 flags. Phylogenies were inferred with IQ-TREE2 v2.2.215 using a GTR + F + I + R4 model with 1000 rapid bootstraps. Inference of transmission was made with phyloscanner v1.8.113 using overlapping windows of 150 bp across the whole genome. Phylogenies were rooted on an HIV-1 subtype G reference sequence, downloaded from the curated HIV-1 alignment from the Los Alamos National HIV Database. HIV-1 subtyping was performed using SNAPPy v1.0.0.16 Prediction of co-receptor usage was made using TROPHIX (prediction of HIV-1 tropism; http://sourceforge.net/projects/trophix/).
Ethics
The study was approved by the University of Maiduguri Teaching Hospital Ethics Committee (UMTH/REC/21/714). All participants provided written informed consent.
Results
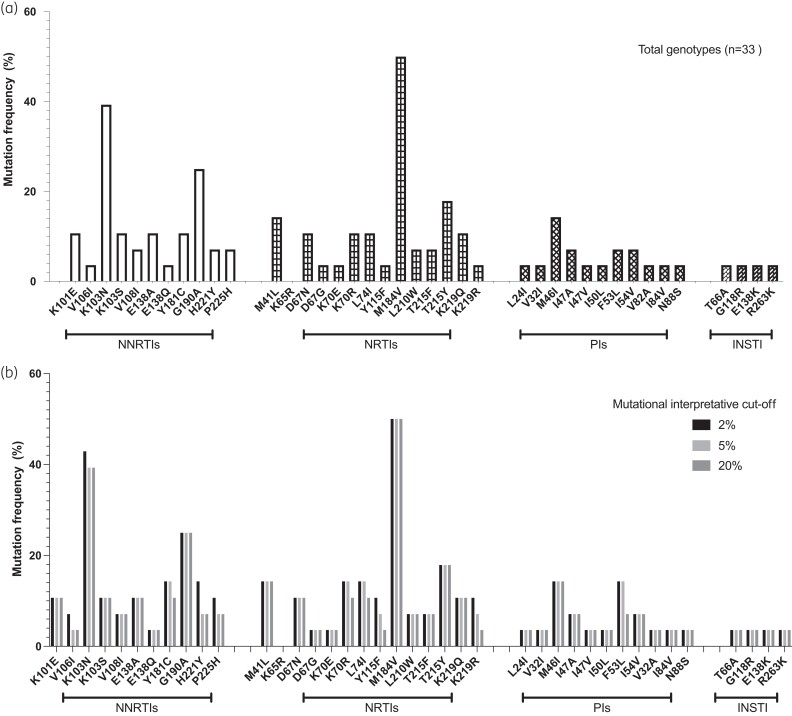
Virological assessment revealed that of 4263 HIV-1-positive participants on dolutegravir-based ART for ≥6 months, 452 (11%) had a detectable VL (>400 copies/mL), yielding a population-level suppression rate of 89%. Amongst those participants with a detectable VL, 281 had a VL of >1000 copies/mL; plasma collection was successful in 61 participants. Following quality control of the WGS, due to poor coverage and depth across most of the genome (Figure 1a and b, available as Supplementary data at JAC Online), 28 samples were excluded from further analysis. This resulted in a population of 33 participants with a fully intact whole genome, from which the prevalence of DRMs and minority variants were calculated.
This population (Table 1) was a largely ART-experienced cohort, with almost two-thirds switching from PI-based second-line ART, to dolutegravir-based ART. The median duration of ART was 9 years, with a median CD4+ count of 200 cells/mm3, and a median VL of 4.1 log10 copies/mL (range 3.0–5.2). Only two participants were initiating first-line dolutegravir-based regimens; the remainder were switched to dolutegravir as their second-line regimen. We did not observe from clinical records, any previous exposure to an INSTI in our study population.
Table 1.
Characteristics of study participants with successful WGS and genotypinga
| Characteristic | |
|---|---|
| Participants | |
| Total number of participants | 33 |
| Female, n (%) | 20 (61) |
| Age, years, median (IQR) | 40 (35–48) |
| CD4+ count, cells/mm3, median (IQR) | 200 (300–467) |
| Current ART regimen | |
| TDF + 3TC + DTG, n (%) | 33 (100) |
| Time on DTG, years, median (IQR) | 1.8 (1.4–1.9) |
| Time on ART, years, median (IQR) | 9.3 (5.8–15.0) |
| Switching to DTG, n (%) | 31 (94) |
| Starting DTG, n (%) | 2 (6) |
| ART regimen prior to DTG, n(%) | |
| TDF + 3TC + LPV/r | 17 (52) |
| TDF + 3TC + ATV/r | 4 (12) |
| ZDV + 3TC + EFV | 8 (24) |
| ABC + 3TC + EFV | 2 (6) |
| No prior ART | 2 (6) |
| HIV-1 subtype, n (%)b | |
| G | 13 (39) |
| G/A1 | 5 (15) |
| CRF02_AG | 3 (9) |
| A (A1) | 2 (6) |
| CRF02_AG/G | 2 (6) |
| 02AG/A1 | 2 (6) |
| Others | 6 (18) |
TDF, tenofovir disoproxil fumarate; 3TC, lamivudine; DTG, dolutegravir; LPV, lopinavir; r ritonavir; ATV, atazanavir; ZDV, zidovudine; EFV, efavirenz; ABC, abacavir.
Data available from 33 participants following quality control.
Other subtypes comprising: CRF06_CPX (n = 1); CRF13_CPX-like (n = 1); CRF13_CPX/G (n = 1); CRF 11_CPX (n = 1); G/C (n = 1); G/J/A (n = 1).
Using the SNAPPy HIV-1 subtyping tool, ∼40% of viruses were assigned to subtype G, and 15% were G_A1 subtypes. The remaining viruses were recombinants, as expected from previous work (Table 1).17,18 Using a minimum threshold of 20%, NRTI, NNRTI, PI and INSTI DRMS occurred in 17 (52%), 24 (73%), 4 (12%) and 1 (3%) of samples, respectively (Figure 1a). Dual-class resistance occurred in 17 (50%) participants and tri-class mutations occurred in 5 (14.7%) participants, which is consistent with long-term exposure to lamivudine. The most prevalent NRTI mutation was M184V,19 though we also observed occurrence of L74I in two cases (6%); this mutation compensates for the fitness defect induced by M184V.20 The most prevalent NNRTI DRM was K103N, reflective of prior exposure to nevirapine and efavirenz in this cohort of treatment-experienced persons living with HIV (PLWH). The frequency of detected DRMs was similar across the different interpretive thresholds of 2%, 10% and 20% (Figure 1b).
Figure 1.
(a) Proportion of participants with DRMs using the Stanford algorithm. (b) Proportion of participants with DRMs using the Stanford algorithm, subdivided into interpretational cut-offs of 2%, 5% and 20%. Evidence suggests that minority variants may play a role in drug resistance.21
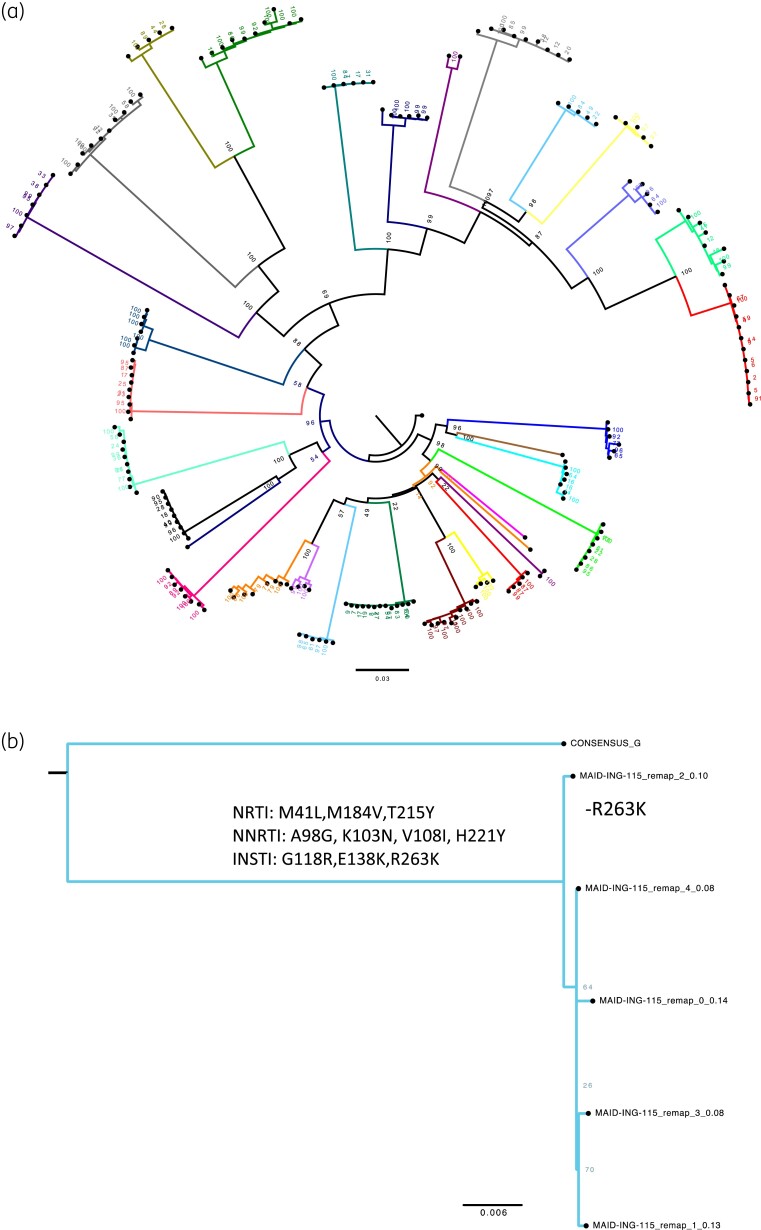
One participant, aged 18 years, known to be vertically infected, was found to have high-level resistance to NRTIs, NNRTIs and INSTIs, including almost complete resistance to the long-acting injectable cabotegravir. Mutations included inE138K, inG118R, inT66A, inR263K, rtH221Y, rtV108I, rtK103N, rtM184V, rtM41L, rtA98G and rtT215Y, all at frequencies >40%, with a mean read depth of 770×. This participant was established on dolutegravir for a median of 1.2 years and on ART for a median of 12 years. Data on other clinical parameters including nadir CD4+ counts were unavailable.
To identify within-host diversity and potential transmission of DRMs between participants, we reconstructed viral haplotypes (Figure 2a) for each participant, and individually for the cabotegravir-resistant participant (Figure 2b). Viral haplotypes were largely homogeneous within host, each containing highly similar DRMs within each of the reconstructed haplotypes for each participant. Following this, we investigated whether there was evidence of transmission or genetic linkage between participants in this cohort (Figure 2). However, no significant transmission or strong degree of genetic linkages were identified.
Figure 2.
(a) Maximum-likelihood phylogeny of reconstructed haplotypes for all participants and (b) only the patient with cabotegravir resistance, with 1000 ultrafast bootstraps (indicated at each node). Mutations defining each haplotype are listed at the node. Haplotypes were homogeneous, with little diversity or changes in frequency of DRMs. This figure appears in colour in the online version of JAC and in black and white in the print version of JAC.
Two participants were identified as harbouring CXCR-4 co-receptor using viruses. One of these participants had DRMs associated with reduced susceptibility to NNRTIs, characterized by K103N and E138A. The other participant had DRMs reducing susceptibility to both NRTIs and NNRTIs, characterized by M184V and G190A. Neither participant had any PI or INSTI DRMs.
Discussion
Following the initial rollout of previous first-line, NNRTI-based ART regimens, DRMs were first observed within the first year of virological failure in ∼15%–35% of participants, characterized by resistance to lamivudine, tenofovir disoproxil fumarate and NNRTIs.22,23 Drug resistance is associated with increased mortality in hospitalized individuals in low-middle income (LMIC) settings.24 The second-generation INSTI dolutegravir has been systematically adopted and rolled out across SSA since 2018, though there are concerns regarding the emergence of DRMs following virological failure in these limited-monitoring settings. In a recent analysis of pooled evidence on virological and resistance outcomes following dolutegravir failure in SSA, there was an overall high rate of virological response to dolutegravir: 88.5% (95% CI: 73.8–97.8). Participants in these studies experienced an exceptionally low rate of virological failure,10 and there was minimal evidence of the emergence of DRMs during short observation periods. However, prolonged virological failure is expected to eventually lead to the selection of DRMs within viral quasispecies, as a result of intrahost evolution.21,25
It is worth noting that the presence of pre-existing DRMs, prior to switching to dolutegravir-based ART regimens may have a significant impact on both virological and resistance outcomes. Previous studies suggest that NNRTI mutations acquired before treatment can reduce the effectiveness of dolutegravir-based ART regimens.26 However, other studies conducted in various countries in SSA have reported similar virological and resistance outcomes in both ART-naive27,28 and ART-experienced participants29,30 (without evidence of DRMs), as well as in ART-experienced individuals with a history of NRTI mutations, particularly M184V/I.31–33 Nonetheless, it is important to acknowledge that resuppression can occur after a viral rebound, but DRMs accumulated during that rebound can lead to high-level resistance against second-line ART components.34
In this study, we conducted a cross-sectional analysis to determine the prevalence of DRMs, using WGS, in a small cohort of HIV-1-positive participants who were experiencing virological failure on dolutegravir-based ART regimens (defined as a VL > 1000 copies/mL). This study is particularly relevant as the majority of HIV-1 in Nigeria is subtype G or AG recombinants, rather than the commonly investigated subtype C from many other parts of SSA. Many of the viraemic participants were ART-experienced, having previously been on PI-based regimens before being switched to TLD. We attribute the high rate of virological failure amongst these participants to poor adherence. Despite this, we observed a low incidence of individuals with TDF or dolutegravir resistance despite experiencing viraemia on TLD. This is reassuring as it suggests that adherence counselling would likely result in resuppression of viraemic participants, similar to those rates observed with PI-based ART.35
Amongst the 33 participants with sequencing data of sufficient quality, the observed mutational landscape was reflective of exposure to previous first-line NNRTI-based regimens, with only a single participant having DRMs reducing the efficacy of dolutegravir or other PIs. This participant was vertically infected, likely at a young age, due to the length of ART treatment. The participant had a number of DRMs conferring high-level resistance to dolutegravir and other INSTIs, i.e. T66A, G118R, E138K (accessory) and R263K. The T66A mutation is non-polymorphic and primarily selected by elvitegravir and raltegravir, resulting in an ∼9-fold reduction in susceptibility to elvitegravir but minimal impact on other INSTIs. The E138K mutation has negligible effect on susceptibility to INSTIs; however, when found in combination with other DRMs, this results in further decreased susceptibility to dolutegravir.36 Further, the G118R and R263K mutations observed in this patient, conferring between 2- and 15-fold reductions to dolutegravir susceptibility,37,38 have also been observed in participants experiencing virological failure on INSTI-based regimens in non-B HIV subtypes39–42 and is known to reduce long terminal repeat DNA binding, replication capacity, infectivity and viral fitness.43 It is likely that the G118R mutation emerged first and led to the accumulation of other mutations including the E138K compensatory mutation, as G118R has been described as a dolutegravir-resistance pathway in non-B subtypes.37,44 Research on non-B subtype drug resistance is therefore important, especially as polymorphisms in the integrase gene may have a negative effect on impact on resistance outcomes to other INSTIs.45
We note that there was a complete absence of the NRTI mutation K65R, along with low-level resistance to tenofovir disoproxil fumarate. This phenomenon has also been described in other Nigerian cohorts.46,47 To ensure that this was a true finding, investigation of read coverage shows that there was sufficient depth to determine DRMs at this site, though there was a complete absence of it (Figure 2). We note the frequent presence of several thymidine analogue mutations (TAMs) alongside other NNRTI mutations likely reflects prior use of zidovudine, stavudine + NNRTI-based ART, prior to virological failure, with subsequent switch to tenofovir disoproxil fumarate-containing PI-based regimens. It should be noted that K65R is antagonistic to TAMs on the same genome,21,48 and that the occurrence of NRTI and PI mutations is relatively uncommon during viraemia on PI-based ART.49,50
Given the increasing use of dolutegravir in clinical practice, the most effective strategy for managing participants with persistent viraemia whilst on dolutegravir-based ART regimens remains uncertain. This is especially true in resource-limited settings such as Nigeria, where the capacity for drug resistance testing is limited.51 Despite this, this study has provided additional evidence to support favourable outcomes when providing long-term ART services in areas of conflict and civil unrest. However, several factors may increase the risk of dolutegravir resistance emerging in the region, including prolonged virological failure due to the lack of routine virological monitoring52,53 as well as suboptimal treatment adherence,54 which is an independent determinant of virological outcomes in these settings.
Disengagement from care is also a significant problem, although it is possible to re-engage with a substantial proportion of cases,55 highlighting the benefits that can be gained through outreach and tracing efforts. Given that INSTI-based long-acting injectables are being considered as pre-exposure prophylaxis,56 it is crucial to conduct further analyses of resistance across SSA over extended periods and to monitor newly diagnosed individuals for INSTI resistance.
This study was subject to limitations. Study sample size was small, with only 33 samples generating high-quality sequences for analysis despite sample collection from 61 participants. We speculate this could be due to human-related factors, which may include shipping and storage conditions. We did not collect data on adherence although the mutational landscape and profile of patients are consistent with prior treatment with NNRTI-based first-line regimens and with evidence of virological failure on dolutegravir. Nonetheless, given the paucity of data across this region, this study provides critical outcome and resistance data including highlighting the need for larger and more comprehensive population-level cohort studies to further understand the emerging trends and prevalence of DRMs in individuals failing dolutegravir-based ART in resource-limited settings.
Finally, given the potential for dolutegravir resistance to emerge due to suboptimal treatment adherence and prolonged virological failure, efforts to improve adherence and increase access to routine virological monitoring should be prioritized to minimize the emergence and spread of drug resistance. Further research is needed to evaluate the effectiveness of different strategies for managing individuals experiencing persistent viraemia on dolutegravir-based ART in resource-limited settings, particularly in the context of ongoing conflict and civil unrest.
Supplementary Material
Acknowledgements
We are thankful to the volunteers who participated in the study.
Contributor Information
Adam Abdullahi, Department of Medicine, Cambridge Institute of Therapeutic Immunology & Infectious Disease (CITIID), Cambridge, UK; Department of Medicine, University of Cambridge, Cambridge, UK; Institute of Human Virology Nigeria, Abuja, Nigeria.
Ibrahim Musa Kida, Department of Infectious Disease and Clinical Immunology, University of Maiduguri, Borno, Nigeria.
Umar Abdullahi Maina, Department of Veterinary Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Maiduguri, Borno, Nigeria.
Amina Husaini Ibrahim, Federal Medical Centre, Abuja, Nigeria.
James Mshelia, Department of Infectious Disease and Clinical Immunology, University of Maiduguri, Borno, Nigeria.
Haruna Wisso, Institute of Human Virology Nigeria, Abuja, Nigeria.
Abdullahi Adamu, Department of Veterinary Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Maiduguri, Borno, Nigeria.
James Ezenwa Onyemata, Institute of Human Virology Nigeria, Abuja, Nigeria.
Martin Edun, Institute of Human Virology Nigeria, Abuja, Nigeria.
Haruna Yusuph, Department of Infectious Disease and Clinical Immunology, University of Maiduguri, Borno, Nigeria.
Sani H Aliyu, Department of Microbiology, Addenbrooke’s Hospital, Cambridge University Hospitals NHS Foundation Trust, Cambridge, UK.
Man Charurat, Institute of Human Virology, University of Maryland School of Medicine, Baltimore, USA.
Alash’le Abimiku, Institute of Human Virology Nigeria, Abuja, Nigeria.
Lucie Abeler-Dorner, Nuffield Department of Medicine, Big Data Institute, University of Oxford, Oxford, UK.
Christophe Fraser, Nuffield Department of Medicine, Big Data Institute, University of Oxford, Oxford, UK.
David Bonsall, Nuffield Department of Medicine, Big Data Institute, University of Oxford, Oxford, UK.
Steven A Kemp, Department of Medicine, Cambridge Institute of Therapeutic Immunology & Infectious Disease (CITIID), Cambridge, UK; Department of Medicine, University of Cambridge, Cambridge, UK; Nuffield Department of Medicine, Big Data Institute, University of Oxford, Oxford, UK.
Ravindra K Gupta, Department of Medicine, Cambridge Institute of Therapeutic Immunology & Infectious Disease (CITIID), Cambridge, UK; Department of Medicine, University of Cambridge, Cambridge, UK; Africa Health Research Institute, Durban, South Africa.
PANGEA consortium:
Lucie Abeler-Dörner, Helen Ayles, David Bonsall, Rory Bowden, Vincent Calvez, Max Essex, Sarah Fidler, Christophe Fraser, Kate Grabowski, Tanya Golubchik, Ravindra Gupta, Richard Hayes, Joshua Herbeck, Joseph Kagaayi, Pontiano Kaleebu, Jairam Lingappa, Sikhulile Moyo, Vladimir Novitsky, Thumbi Ndung'u, Deenan Pillay, Thomas Quinn, Andrew Rambaut, Oliver Ratmann, Janet Seeley, Deogratius Ssemwanga, Frank Tanser, Maria Wawer, Myron Cohen, Tulio D'Oliveira, Ann Dennis, Max Essex, Sarah Fidler, Dan Frampton, Christophe Fraser, Tanya Golubchik, Richard Hayes, Josh Herbeck, Anne Hoppe, Pontiano Kaleebu, Paul Kellam, Cissy Kityo, Andrew Leigh-Brown, Jairam Lingappa, Vladimir Novitsky, Nick Paton, Deenan Pillay, Tom Quinn, Oliver Ratmann, Deogratius Ssemwanga, Frank Tanser, and Maria Wawer
Funding
A.A. is supported by an Africa Research Excellence Fund Research Development Fellowship (AREF-318-ABDUL-F-C0882) and Cambridge-Africa award. A.A. and S.A.K. are supported by the Bill and Melinda Gates Foundation via the Phylogenetics and Networks for Generalised Epidemics in Africa (PANGEA) (grant number OPP1175094). A.A and R.K.G were partially supported by NIH (R01AI147331-01). This study was also supported by the NIH/NIAID 5R01AI147331-02 grant to R.K.G.
Transparency declarations
None to declare.
Author contributions
Study conception, design and administration: A. Abdullahi, R.K.G. and I.M.K.; data collection: A. Abdullahi, I.M.K., A.H.I., J.M., A. Adamu, J.E.O., H.Y., M.E., S.H.A., A. Abdullahi, S.A.K. and R.K.G.; data analysis: A. Abdullahi, I.M.K., S.A.K., A. Abdullahi and R.K.G.; data interpretation: A. Abdullahi, S.A.K., A. Abdullahi and R.K.G.; manuscript preparation; A. Abdullahi wrote the first draft of the manuscript with the critical input of all co-authors. All authors reviewed the results and approved the final version of the manuscript.
Supplementary data
Figures S1 and S2 are available as Supplementary data at JAC Online.
References
- 1. Gupta RK, Jordan MR, Sultan BJ et al. Global trends in antiretroviral resistance in treatment-naive individuals with HIV after rollout of antiretroviral treatment in resource-limited settings: a global collaborative study and meta-regression analysis. Lancet 2012; 380: 1250–8. 10.1016/S0140-6736(12)61038-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Gupta RK, Gregson J, Parkin N et al. HIV-1 drug resistance before initiation or re-initiation of first-line antiretroviral therapy in low-income and middle-income countries: a systematic review and meta-regression analysis. Lancet Infect Dis 2018; 18: 346–55. 10.1016/S1473-3099(17)30702-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Chimukangara B, Lessells RJ, Rhee S-Y et al. Trends in pretreatment HIV-1 drug resistance in antiretroviral therapy-naive adults in South Africa, 2000–2016: a pooled sequence analysis. EClinicalMedicine 2019; 9: 26–34. 10.1016/j.eclinm.2019.03.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. WHO . Update of recommendations on first- and second-line antiretroviral regimens. 2019. https://apps.who.int/iris/bitstream/handle/10665/325892/WHO-CDS-HIV-19.15-eng.pdf?ua=1.
- 5. Jamieson L, Serenata C, Makhubele L et al. Cost and cost-effectiveness of dolutegravir-based antiretroviral regimens: an economic evaluation of a clinical trial. AIDS 2021; 35: S173–82. 10.1097/QAD.0000000000003068 [DOI] [PubMed] [Google Scholar]
- 6. Dorward J, Hamers RL. Dolutegravir in sub-Saharan Africa: context is crucial. Lancet HIV 2019; 6: e72–3. 10.1016/S2352-3018(18)30331-X [DOI] [PubMed] [Google Scholar]
- 7. Federal Ministry of Health Abuja Nigeria . National guideline for HIV prevention, treatment and care. 2020. https://drive.google.com/file/d/1s7fsW8fCx8eddftO_Ly9TjpbRTRwPqaL/view?usp=drive_link
- 8. Calmy A, Tovar Sanchez T, Kouanfack C et al. Dolutegravir-based and low-dose efavirenz-based regimen for the initial treatment of HIV-1 infection (NAMSAL): week 96 results from a two-group, multicentre, randomised, open label, phase 3 non-inferiority trial in Cameroon. Lancet HIV 2020; 7: e677–87. 10.1016/S2352-3018(20)30238-1 [DOI] [PubMed] [Google Scholar]
- 9. Venter WDF, Sokhela S, Simmons B et al. Dolutegravir with emtricitabine and tenofovir alafenamide or tenofovir disoproxil fumarate versus efavirenz, emtricitabine, and tenofovir disoproxil fumarate for initial treatment of HIV-1 infection (ADVANCE): week 96 results from a randomised, phase 3. Lancet HIV 2020; 7: e666–76. 10.1016/S2352-3018(20)30241-1 [DOI] [PubMed] [Google Scholar]
- 10. Semengue ENJ, Santoro MM, Ndze VN et al. HIV-1 integrase resistance associated mutations and the use of dolutegravir in sub-Saharan Africa: a systematic review and meta-analysis. PLOS Glob Public Heal 2022; 2: e0000826. 10.1371/journal.pgph.0000826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Stockdale AJ, Saunders MJ, Boyd MA et al. Effectiveness of protease inhibitor/nucleos(t)ide reverse transcriptase inhibitor–based second-line antiretroviral therapy for the treatment of human immunodeficiency virus type 1 infection in sub-Saharan Africa: a systematic review and meta-analysis. Clin Infect Dis 2017; 66: 1846–57. 10.1093/cid/cix1108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Bonsall D, Golubchik T, de Cesare M et al. A comprehensive genomics solution for HIV surveillance and clinical monitoring in low-income settings. J Clin Microbiol 2020; 58: e00382-20. 10.1128/JCM.00382-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Wymant C, Hall M, Ratmann O et al. PHYLOSCANNER: inferring transmission from within- and between-host pathogen genetic diversity. Mol Biol Evol 2018; 35: 719–33. 10.1093/molbev/msx304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Kuiken C, Korber B, Shafer RW. HIV sequence databases. AIDS Rev 2003; 5: 52–61. [PMC free article] [PubMed] [Google Scholar]
- 15. Minh BQ, Schmidt HA, Chernomor O et al. IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 2020; 37: 1530–4. 10.1093/molbev/msaa015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Araújo PMM, Martins JS, Osório NS. SNAPPy: a snakemake pipeline for scalable HIV-1 subtyping by phylogenetic pairing. Virus Evol 2019; 5: vez050. 10.1093/ve/vez050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. El Bouzidi K, Datir RP, Kwaghe V et al. Deep sequencing of HIV-1 reveals extensive subtype variation and drug resistance after failure of first-line antiretroviral regimens in Nigeria. J Antimicrob Chemother 2022; 77: 474–82. 10.1093/jac/dkab385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Chaplin B, Eisen G, Idoko J et al. Impact of HIV type 1 subtype on drug resistance mutations in Nigerian patients failing first-line therapy. AIDS Res Hum Retroviruses 2010; 27: 71–80. 10.1089/aid.2010.0050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Collier DA, Monit C, Gupta RK. The impact of HIV-1 drug escape on the global treatment landscape. Cell Host Microbe 2019; 26: 48–60. 10.1016/j.chom.2019.06.010 [DOI] [PubMed] [Google Scholar]
- 20. Gregson J, Rhee SY, Datir R et al. Human immunodeficiency virus-1 viral load is elevated in individuals with reverse-transcriptase mutation M184V/I during virological failure of first-line antiretroviral therapy and is associated with compensatory mutation L74I. J Infect Dis 2020; 222: 1108–16. 10.1093/infdis/jiz631 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Kemp SA, Charles OJ, Derache A et al. HIV-1 evolutionary dynamics under nonsuppressive antiretroviral therapy. mBio 2022; 13: e00269-22. 10.1128/mbio.00269-22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Gregson J, Kaleebu P, Marconi VC et al. Occult HIV-1 drug resistance to thymidine analogues following failure of first-line tenofovir combined with a cytosine analogue and nevirapine or efavirenz in sub Saharan Africa: a retrospective multi-centre cohort study. Lancet Infect Dis 2017; 17: 296–304. 10.1016/S1473-3099(16)30469-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. TenoRes Study Group . Global epidemiology of drug resistance after failure of WHO recommended first-line regimens for adult HIV-1 infection: a multicentre retrospective cohort study. Lancet Infect Dis 2016; 16: 565–75. 10.1016/S1473-3099(15)00536-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Gupta-Wright A, Fielding K, van Oosterhout JJ et al. Virological failure, HIV-1 drug resistance, and early mortality in adults admitted to hospital in Malawi: an observational cohort study. Lancet HIV 2020; 7: e620–8. 10.1016/S2352-3018(20)30172-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Datir R, Kemp S, El Bouzidi K et al. In vivo emergence of a novel protease inhibitor resistance signature in HIV-1 matrix. mBio 2020; 11: e02036-20. 10.1128/mBio.02036-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Siedner MJ, Moorhouse MA, Simmons B et al. Reduced efficacy of HIV-1 integrase inhibitors in patients with drug resistance mutations in reverse transcriptase. Nat Commun 2020; 11: 5922. 10.1038/s41467-020-19801-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Maggiolo F, Gulminetti R, Pagnucco L et al. Lamivudine/dolutegravir dual therapy in HIV-infected, virologically suppressed patients. BMC Infect Dis 2017; 17: 215. 10.1186/s12879-017-2311-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Joly V, Burdet C, Landman R et al. Dolutegravir and lamivudine maintenance therapy in HIV-1 virologically suppressed patients: results of the ANRS 167 trial (LAMIDOL). J Antimicrob Chemother 2019; 74: 739–45. 10.1093/jac/dky467 [DOI] [PubMed] [Google Scholar]
- 29. Taiwo BO, Marconi VC, Berzins B et al. Dolutegravir plus lamivudine maintains human immunodeficiency virus-1 suppression through week 48 in a pilot randomized trial. Clin Infect Dis 2018; 66: 1794–7. 10.1093/cid/cix1131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Cahn P, Madero JS, Arribas JR et al. Dolutegravir plus lamivudine versus dolutegravir plus tenofovir disoproxil fumarate and emtricitabine in antiretroviral-naive adults with HIV-1 infection (GEMINI-1 and GEMINI-2): week 48 results from two multicentre, double-blind, randomised, non-inferiority, phase 3 trials. Lancet 2019; 393: 143–55. 10.1016/S0140-6736(18)32462-0 [DOI] [PubMed] [Google Scholar]
- 31. Hidalgo-Tenorio C, Cortés LL, Gutiérrez A et al. DOLAMA study: effectiveness, safety and pharmacoeconomic analysis of dual therapy with dolutegravir and lamivudine in virologically suppressed HIV-1 patients. Medicine (Baltimore) 2019; 98: e16813. 10.1097/MD.0000000000016813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Jary A, Marcelin A-G, Charpentier C et al. M184V/I does not impact the efficacy of abacavir/lamivudine/dolutegravir use as switch therapy in virologically suppressed patients. J Antimicrob Chemother 2020; 75: 1290–3. 10.1093/jac/dkaa019 [DOI] [PubMed] [Google Scholar]
- 33. De Miguel R, Rial-Crestelo D, Dominguez-Dominguez L et al. Dolutegravir plus lamivudine for maintenance of HIV viral suppression in adults with and without historical resistance to lamivudine: 48-week results of a non-randomized, pilot clinical trial (ART-PRO). eBioMedicine 2020; 55: 102779. 10.1016/j.ebiom.2020.102779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Gupta RK, Goodall RL, Ranopa M et al. High rate of HIV resuppression after viral failure on first-line antiretroviral therapy in the absence of switch to second-line therapy. Clin Infect Dis 2014; 58: 1023–6. 10.1093/cid/cit933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Collier D, Iwuji C, Derache A et al. Virological outcomes of second-line protease inhibitor–based treatment for human immunodeficiency virus type 1 in a high-prevalence rural South African setting: a competing-risks prospective cohort analysis. Clin Infect Dis 2017; 64: 1006–16. 10.1093/cid/cix015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Mesplède T, Osman N, Wares M et al. Addition of E138K to R263K in HIV integrase increases resistance to dolutegravir, but fails to restore activity of the HIV integrase enzyme and viral replication capacity. J Antimicrob Chemother 2014; 69: 2733–40. 10.1093/jac/dku199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Rhee S-Y, Grant PM, Tzou PL et al. A systematic review of the genetic mechanisms of dolutegravir resistance. J Antimicrob Chemother 2019; 74: 3135–49. 10.1093/jac/dkz256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Brenner BG, Thomas R, Blanco JL et al. Development of a G118R mutation in HIV-1 integrase following a switch to dolutegravir monotherapy leading to cross-resistance to integrase inhibitors. J Antimicrob Chemother 2016; 71: 1948–53. 10.1093/jac/dkw071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Ahmed N, Flavell S, Ferns B et al. Development of the R263K mutation to dolutegravir in an HIV-1 subtype D virus harboring 3 class-drug resistance. Open forum Infect Dis 2019; 6: ofy329. 10.1093/ofid/ofy329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Chimbetete C, Chirimuta L, Pascoe M et al. A case report of untreatable HIV infection in Harare, Zimbabwe. South Afr J HIV Med 2019; 20: 885. 10.4102/sajhivmed.v20i1.885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Ndashimye E, Avino M, Kyeyune F et al. Absence of HIV-1 drug resistance mutations supports the use of dolutegravir in Uganda. AIDS Res Hum Retroviruses 2018; 34: 404–14. 10.1089/aid.2017.0205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Mahomed K, Wallis CL, Dunn L et al. Case report: emergence of dolutegravir resistance in a patient on second-line antiretroviral therapy. South African J HIV Med 2020; 21: 1062. 10.4102/sajhivmed.v21i1.1062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Xiao MA, Cleyle J, Yoo S et al. The G118R plus R263K combination of integrase mutations associated with dolutegravir-based treatment failure reduces HIV-1 replicative capacity and integration. Antimicrob Agents Chemother 2023; 67: e0138622. 10.1128/aac.01386-22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Semengue ENJ, Armenia D, Inzaule S et al. Baseline integrase drug resistance mutations and conserved regions across HIV-1 clades in Cameroon: implications for transition to dolutegravir in resource-limited settings. J Antimicrob Chemother 2021; 76: 1277–85. 10.1093/jac/dkab004 [DOI] [PubMed] [Google Scholar]
- 45. El Bouzidi K, Kemp SA, Datir RP et al. High prevalence of integrase mutation L74I in West African HIV-1 subtypes prior to integrase inhibitor treatment. J Antimicrob Chemother 2020; 75: 1575–9. 10.1093/jac/dkaa033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Aliyu GG, Lawton JG, Mitchell AB et al. Prevalence of HIV drug resistance in Nigeria: results from a cross-sectional, population-based survey of Nigerian adults with unsuppressed viral load. AIDS 2023; 37: 333–9. 10.1097/QAD.0000000000003413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Hawkins CA, Chaplin B, Idoko J et al. Clinical and genotypic findings in HIV-infected patients with the K65R mutation failing first-line antiretroviral therapy in Nigeria. J Acquir Immune Defic Syndr 2009; 52: 228–34. 10.1097/QAI.0b013e3181b06125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Parikh UM, Barnas DC, Faruki H et al. Antagonism between the HIV-1 reverse-transcriptase mutation K65R and thymidine-analogue mutations at the genomic level. J Infect Dis 2006; 194: 651–60. 10.1086/505711 [DOI] [PubMed] [Google Scholar]
- 49. Hosseinipour MC, Gupta RK, Van Zyl G et al. Emergence of HIV drug resistance during first- and second-line antiretroviral therapy in resource-limited settings. J Infect Dis 2013; 207 Suppl 2: S49–56. 10.1093/infdis/jit107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Hill A, McBride A, Sawyer AW et al. Resistance at virological failure using boosted protease inhibitors versus nonnucleoside reverse transcriptase inhibitors as first-line antiretroviral therapy—implications for sustained efficacy of ART in resource-limited settings. J Infect Dis 2013; 207 Suppl 2: S78–84. 10.1093/infdis/jit112 [DOI] [PubMed] [Google Scholar]
- 51. da Silva J, Pals S, Chang J et al. Monitoring emerging human immunodeficiency virus drug resistance in sub-Saharan Africa in the era of dolutegravir. J Infect Dis 2022; 225: 364–6. 10.1093/infdis/jiab382 [DOI] [PubMed] [Google Scholar]
- 52. Villa G, Abdullahi A, Owusu D et al. Determining virological suppression and resuppression by point-of-care viral load testing in a HIV care setting in sub-Saharan Africa. EClinicalMedicine 2020; 18: 100231. 10.1016/j.eclinm.2019.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Gupta RK, Hill A, Sawyer AW et al. Virological monitoring and resistance to first-line highly active antiretroviral therapy in adults infected with HIV-1 treated under WHO guidelines: a systematic review and meta-analysis. Lancet Infect Dis 2009; 9: 409–17. 10.1016/S1473-3099(09)70136-7 [DOI] [PubMed] [Google Scholar]
- 54. Geretti AM, Abdullahi A, Fopoussi OM et al. An apparent paradox: resistance mutations in HIV-1 DNA predict improved virological responses to antiretroviral therapy. J Antimicrob Chemother 2019; 74: 3011–5. 10.1093/jac/dkz264 [DOI] [PubMed] [Google Scholar]
- 55. El Bouzidi K, Murtala-Ibrahim F, Kwaghe V et al. Disengagement from HIV care and failure of second-line therapy in Nigeria: a retrospective cohort study, 2005–2017. J Acquir Immune Defic Syndr 2022; 90: 88–96. 10.1097/QAI.M0000000000002918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. WHO . Guidelines on long-acting injectable cabotegravir for HIV prevention. 2022. https://www.who.int/publications/i/item/9789240054097.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.