Abstract
Newborn mice of several inbred strains develop few or no tumors following inoculation with highly tumorigenic strains of polyomavirus. Here we show that such resistant strains can be divided into two groups based on the responses of adult mice to radiation followed by virus inoculation. Most strains show a radiation-sensitive form of resistance (Rr-s) and develop tumors following radiation and virus challenge. This type of resistance has previously been recognized as immunological, based on T-cell responses against virus-encoded neoantigen(s) expressed in tumor cells. Other strains exhibit a radiation-resistant form of resistance (Rr-r) and fail to develop tumors when treated in the same manner. Three additional properties of Rr-r mice distinguish them from Rr-s mice: (i) survival of newborns following inoculation with a highly virulent and usually lethal strain of virus, (ii) resistance to virus spread in newborns inoculated with either tumorigenic or virulent virus strains, and (iii) dominant or semidominant transmission of resistance in crosses with a highly susceptible strain. The Rr-r phenotype reflects a constitutive nonimmunological type of resistance that is targeted to the virus and blocks its dissemination.
Studies of the genetics of the mouse in relation to infections by the oncogenic murine polyomavirus promise to reveal multiple factors with diverse modes of action. Variations in susceptibility to tumor induction by polyomavirus among inbred mouse strains have been noted (8, 11, 14; data not shown). Much of this variation reflects differences in the abilities of different strains of mice to generate virus-specific anti-tumor immune responses, resulting in variable overall tumor frequencies. In addition, some strains exhibit resistance to tumor development in specific target tissues while being susceptible at other sites, suggesting the existence of nonimmunological mechanism(s). The host genetic background can influence events as diverse as the early events of virus replication and spread as a necessary prelude to the induction of a broad array of tumors and late events affecting the likelihood of metastases of specific tumor types. The identity and mechanisms of action of most of these genes remain unknown. Here we describe two contrasting forms of host resistance, one that prevents tumor induction by targeting the virus and preventing its spread and one that acts downstream of virus spread, targeting incipient tumors for immunological rejection.
A survey of over 30 inbred mouse strains has uncovered several strains which show a total or near-total absence of tumors or development of a restricted spectrum of tumor types following inoculation of newborn animals with highly tumorigenic strains of virus. These resistant strains can be divided into two groups based on the responses of adult mice to ionizing radiation followed by virus inoculation. The use of radiation is necessary because newborn mice of even the most susceptible strains are known to acquire resistance as adults (16). Maturation of immune mechanisms is generally regarded as the basis for this acquisition of resistance, although other mechanisms may also be involved.
Most resistant strains show a radiation-sensitive form of resistance, designated Rr-s, indicated by the development of tumors in a high proportion of treated animals. Earlier studies have established the Rr-s phenotype as the norm of host resistance, mediated by cellular immune responses to polyomavirus-induced tumors (1, 15, 16, 24). MHC (major histocompatibility complex) type is a major determinant of this kind of resistance (11). The present study reveals a second form of host resistance, based on the observation that mice of certain strains fail to develop tumors following radiation and virus challenge. This radiation-resistant form of resistance, designated Rr-r, operates through a nonimmunological mechanism that blocks virus dissemination in the intact host.
MATERIALS AND METHODS
C3H/BiDa mice were obtained initially from Clarence Reeder (National Cancer Institute, Fredrick Cancer Research and Development Center, Fredrick, Md.). C57BR/cdJ, MA/MyJ, CE/J, and C57BL/6J mice were obtained from the Jackson Laboratory, Bar Harbor, Maine. All mice were maintained in our virus-free breeding colony. F1 crosses were performed reciprocally.
Mice younger than 24 h of age were inoculated intraperitoneally with 0.05 ml of A2, A2+ (10), or LID (2, 3) polyomavirus containing approximately 2 × 105 to 1 × 106 PFU. Four- to six-week-old mice received between 500 and 900 rads of γ radiation in a Gammacell 40 (Atomic Energy of Canada, Ltd., Ottawa) and 24 h later were inoculated intraperitoneally with virus. Mice were inspected twice weekly and necropsied when moribund or at 6 to 12 months of age. Tumors and tissues were fixed in Bouins’ solution and embedded in paraffin, and sections were stained with Harris’ hematoxylin and eosin as previously described (5, 13). All gross tumors other than those of bone were confirmed by microscopic examination.
Whole mouse section hybridization was performed as previously described (6, 7). Briefly, mice inoculated at birth were sacrificed at 10 days and frozen in Tissue-Tek embedding medium (OCT 4583). Parasagittal sections were transferred to nitrocellulose membranes and hybridized to 35S-labelled nick-translated polyomavirus DNA, and the hybridization signal was visualized by autoradiography.
Tests for innate immunity (NK cells and antiviral interferons) were carried out as described previously (25; see text).
RESULTS
Patterns of susceptibility and resistance to tumor induction by polyomavirus.
Three patterns of host responses are apparent based on comparisons of tumor development in newborn versus irradiated adult mice (Table 1) as follows:
TABLE 1.
Tumor induction in newborn mice and in adult irradiated mice of different strainsa
| Mouse strain | No. of mice with tumors/ total no. of mice
|
|
|---|---|---|
| Newborn | Adult irradiated | |
| Susceptible (S) | ||
| C3H/BiDa | 63/64b | 9/9 |
| Resistant (R) | ||
| Radiation sensitive (Rr-s) | ||
| C57BR/cdJ | 1/41b | 17/17b |
| MOLD/Rk | 0/25 | 6/7 |
| Radiation resistant (Rr-r) | ||
| MA/MyJ | 0/28 | 0/11 |
| CE/J | 10/28c | 0/10 |
Newborn mice (<24 h old) were inoculated intraperitoneally with 2 × 105 to 5 × 105 PFUs of A2 or A2+ wild-type virus strains. Adult mice between 2 and 3 months of age were γ irradiated with 500 to 900 rads. The dose was adjusted within this range so that it was slightly under the 50% lethal dose for each strain: 650 to 800 rads (C3H/BiDa); 800 to 900 rads (C57BR/cdJ); 650 rads (MOLD/RkJ); 650 to 900 rads (MA/MyJ); 500 to 650 rads (CE/J). Mice were inoculated intraperitoneally 24 h later with the A2 virus at 2 × 105 to 5 × 105 PFU/animal. Development of tumors was followed by weekly gross examinations; tumors were confirmed at necropsy both visually and microscopically.
See text.
(i) Susceptible strains (S).
C3H/BiDa is one of several inbred strains known to be highly susceptible to the oncogenic effects of polyomavirus. When inoculated within 24 h of birth with an appropriate strain of virus, these mice develop multiple tumors in frequencies approaching 100% within a period of 3 to 4 months (5, 13). Susceptibility in these strains is based in large part on a common MHC background (H2k) coupled with an endogenous superantigen encoded by a particular mouse mammary tumor provirus (Mtv-7 sag). In the H-2k background, the Mtv-7 superantigen effectively deletes precursors of polyomavirus-specific cytotoxic T lymphocytes, rendering these mice unable to reject polyomavirus-induced tumors (17, 18).
Interestingly, mice of these highly susceptible strains develop resistance as adults. In the present study, for example, none of 16 adult C3H/BiDa mice developed tumors when inoculated with doses of virus that were the same or higher than those given to newborns. This resistance can be overcome by sublethal doses of radiation prior to virus inoculation. Radiation-sensitive resistance in adult mice suggests that immune mechanisms are lacking or not fully developed in the newborn.
(ii) Radiation-sensitive resistant strains (Rr-s).
Many strains of mice show inoculated as newborns partial or complete resistance to tumor induction. Earlier studies have shown that normally resistant strains such as C57BL/6J can be rendered susceptible following immune suppression by a variety of mechanisms, such as radiation or neonatal thymectomy or administration of antilymphocyte serum (1, 15, 16, 24).
This type of resistance is illustrated with two other highly resistant strains, C57BR/cdJ and MOLD/RkJ. The phenotype of these Rr-s mice is consistent with immunological resistance based not only on their radiation susceptibility as adults but also on the known interaction of MHC and endogenous superantigen as codeterminants of susceptibility in this system (17). Thus, C57BL/6J and MOLD/RkJ bear a type other than H-2k at the MHC locus, and C57BR/cdJ, though H-2k, lacks the Mtv-7 provirus (17). Although radiation overcomes the resistance in adult mice of both S and Rr-s strains, the tumor profiles are different. The adult irradiated C3H/BiDa (S) mice developed no fibrosarcomas but otherwise showed a broad range of tumors similar to the range seen with newborn mice. Adult irradiated C57BR/cdJ (Rr-s) mice all developed salivary gland tumors but showed much-reduced frequencies of other tumor types and none in either kidney or bone. The greater frequency of tumors in adult irradiated C3H/BiDa mice may reflect the presence of the dominant susceptibility gene Mtv 7-sag in this strain (17).
(iii) Radiation-resistant resistant strains (Rr-r).
Not all neonatally-resistant mice develop a radiation-sensitive form of resistance as adults as is clearly illustrated with mice of the MA/MyJ strain. Adult mice of this strain maintain complete resistance to tumor induction by polyomavirus, even when they are given immunosuppressive doses (up to 900 rads) of radiation.
CE/J mice show limited susceptibility as newborns but are nevertheless considered to have an Rr-r phenotype. The 10 of 28 neonatally infected CE/J mice that developed tumors developed only single fibrosarcomas after an unusually long latent period; no other tumors typical of the polyomavirus tumor profile were seen in these animals. Most importantly, CE/J mice showed complete resistance to tumor development as adults following irradiation and virus challenge.
MA/MyJ mice are resistant to the virulent LID virus strain.
LID is a virulent strain of polyomavirus which spreads rapidly in newborn mice, inducing widespread cytolytic damage, particularly in the kidney and brain (2, 3). Newborn mice were inoculated intraperitoneally with 106 PFU of the LID strain, as previously described (2). Following are data on the fractions of animals surviving up to 30 days. All 20 newborn C3H/BiDa mice inoculated with this virus died within 2 weeks. All of the mice of the Rr-s strains C57BR/cdJ (n = 20) and C57BL/6J (n = 21) were also killed by LID. Some of these mice survived for up to 3 weeks but uniformly succumbed, with the same pattern of virus spread and cytolytic damage as that seen in C3H/BiDa mice. The slightly extended average survival time of Rr-s compared to that of susceptible mice may reflect a degree of viral clearance by polyomavirus-specific cytotoxic T lymphocytes present in the former but absent in the latter strains (17, 18).
In contrast to Rr-s mice, 25 of 25 newborn MA/MyJ mice survived following inoculation with LID. Although none of these mice succumbed to the acute lytic effects of LID, about 60% developed tumors. Such tumors developed late and occurred only in bone and kidney. CE/J mice, unlike MA/MyJ mice, succumbed to LID infection as newborns.
The limited tumor profile induced in MA/MyJ mice by LID is similar to that induced in highly susceptible C3H/BiDa mice by the low tumor virus strain RA, which replicates poorly in these mice (5). A single amino acid substitution in the outer capsid protein VP1 underlies the important biological differences between high and low tumor virus strains (6, 9, 12). This difference also dictates the viruses’ ability to discriminate among different linkages of sialic acid on the cell surface and their ability to spread in the intact host (22, 23). The virulence determinant in LID likewise maps to a single amino acid substitution in VP1; in this case, the substitution results in a lower affinity of binding by the virus to its sialic acid receptor (2).
These results correlating pathogenicity of the virus to its interactions with cell receptors suggest that MA/MyJ mice may have a mechanism which curtails virus spread, acting perhaps at the level of virus-cell binding or possibly at the level of entry or release of virus from infected cells. Such a mechanism would be consistent with the findings noted above, viz., diminution of pathogenicity of both tumorigenic and virulent strains of polyomavirus in MA/MyJ mice. Since Rr-s mice succumb to disseminated lytic infection by LID, they would be expected to lack such a mechanism preventing virus spread.
Newborn MA/MyJ mice are highly resistant to virus spread.
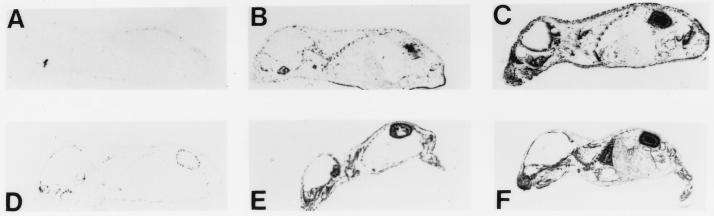
To test these predictions, newborn mice of each prototype strain were inoculated with either the high tumor (A2) or virulent (LID) strain of virus, and the degree of virus spread was determined by whole mouse section hybridization at 10 days (Fig. 1). The results show that MA/MyJ (Rr-r) mice are highly resistant to the spread of both virus strains (Fig. 1A and D). C57BR/cdJ (Rr-s) mice, on the other hand, are susceptible to disseminated infection by both viruses (Fig. 1B and E), showing degrees of spread only slightly less than those seen in C3H/BiDa (S) mice (Fig. 1C and F).
FIG. 1.
Whole mouse section hybridization. MA/MyJ (A and D), C57BR/cdJ (B and E), and C3H/BiDa (C and F) mice were inoculated at <1 day old with A2 (A to C) or LID (D to F) virus and sacrificed at 10 days. See text.
Immunoperoxidase staining for VP1 in kidney sections of these mice gave indications of virus spread consistent with the results of hybridization. MA/MyJ mice showed strong systemic resistance to virus spread relative to both C57BR/cdJ and C3H/BiDa mice, regardless of the route of virus inoculation (intraperitoneal, subcutaneous, or intravenous). Similar resistance to spread was also seen in irradiated newborn MA/MyJ mice, suggesting that the mechanism which blocks virus spread most likely underlies the radiation-resistant form of resistance shown by these mice to the pathological effects of the virus (data not shown).
Cell cultures derived from MA/MyJ mice are susceptible to lytic virus infection.
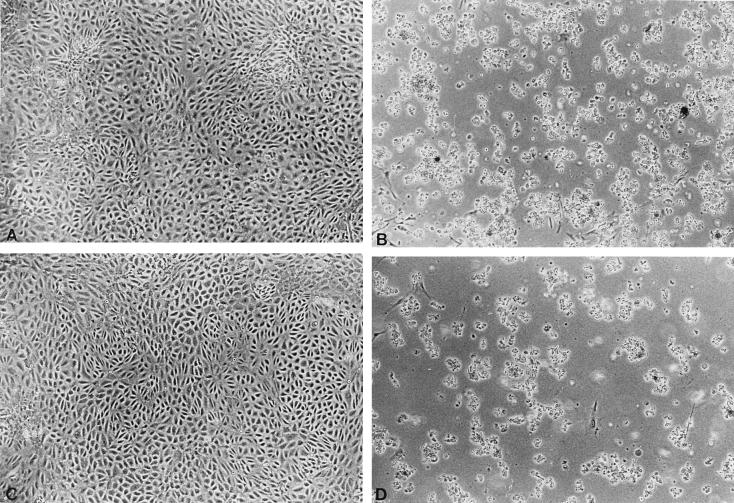
The resistance of MA/MyJ mice could be accounted for by a lack of or a reduced level of expression of functional virus receptors. This possibility was examined by a comparison of the results of infection of primary kidney cell cultures prepared from baby MA/MyJ and C3H/BiDa mice. Cultures were infected at a low multiplicity (<0.1 PFU/cell), so that multiple cycles of infection would be required to induce full cytopathic effects. As shown in Fig. 2, cultures from both mice developed extensive cytopathic changes typical of polyomavirus infection after 1 week. Thus, while kidneys of MA/MyJ mice are largely spared in vivo (Fig. 1), kidney cells in culture from these mice are readily killed by the virus. This result demonstrates that at the cellular level, MA/MyJ mice are capable of expressing functional virus receptors as well as other factors essential for productive viral infection.
FIG. 2.
Uninfected and infected kidney cultures from baby C3H/BiDa (A and B, respectively) and MA/MyJ (C and D, respectively) mice. Infected cultures are shown 7 days postinfection. See text.
Tests for innate immunity in S, Rr-s, and Rr-r mice.
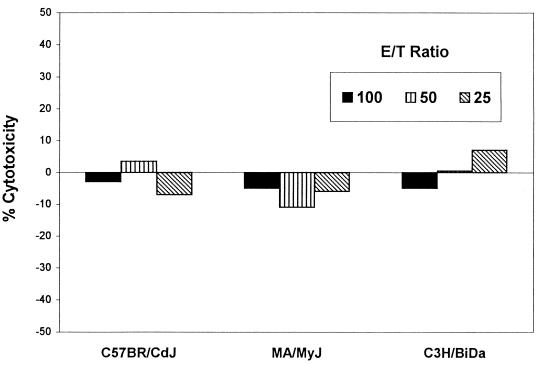
Several aspects of innate immunity are known to affect host resistance to viral infection. Among these, the production of NK cells is of possible relevance to the Rr-r phenotype, first because of the known relative resistance of NK cells to radiation, and second because NK cells are regulated in part by type 1 (antiviral) interferons. To test the possibility that resistance of MA/MyJ mice may be due to these aspects of innate immunity, levels of NK activity and antiviral interferons were measured and compared in young adult mice of each prototype strain. NK activity was measured in a standard cytotoxic assay with spleen-derived leukocytes and chromium-labeled YAC-1 target cells (25). No significant levels of NK activity were seen, and no differences among the three mouse strains were noted (Fig. 3).
FIG. 3.
Assay for NK cytotoxic activity. E/T, effector/target ratios with splenocyte suspensions from normal mouse spleens as effectors and chromium-51-labelled YAC-1 target cells. Incubations were overnight at 37°C. Percent cytotoxicity was calculated as described previously (25).
Sera from the same mice were tested for their ability to protect L929 cells from infection by vesicular stomatitis virus in a standard assay for antiviral interferons (25). Serum titers were found to be uniformly low. Those found for MA/MyJ mice, though somewhat higher than those for C57BR/cdJ, were no higher than those for susceptible C3H/BiDa mice (Table 2).
TABLE 2.
Test for interferons in sera from normal micea
| Strain (phenotype) | Serum titer
|
|
|---|---|---|
| Expt 1 | Expt 2 | |
| C3H/BiDa (S) | 4 | 16 |
| C57BR/cdJ (Rr-s) | <4 | <4 |
| MA/MyJ (Rr-r) | 4 | 16 |
Antiviral interferon levels were measured in sera from uninfected young adult mice as described previously (25). Briefly, L929 cells in microtiter wells were incubated for 2 h with serial twofold dilutions of sera and then challenged with vesicular stomatitis virus of a titer sufficient to cause complete cytopathic effect 48 h postinfection in untreated cultures. Serum titers given are the reciprocal of the highest dilution conferring observable protection against complete cytopathic effect by microscopic examination at 48 h.
Sera of normal mice were also tested in a hemagglutination inhibition assay with polyomavirus and guinea pig erythrocytes. The titers were insignificant or undetectable in all cases (data not shown). These results rule out the presence of natural virus inhibitors in sera of MA/MyJ mice as well as that of specific antiviral antibodies which would have been present if MA/MyJ mice in our colony were accidentally infected by and carrying polyomavirus. The resistance of MA/MyJ mice to polyomavirus infections thus does not appear to be due to high levels of innate immunity or to preexisting polyomavirus infection with development of antiviral humoral immunity.
Differences in genetic transmission of resistance by Rr-s and Rr-r mice.
The resistance of Rr-s mice was previously found to be recessive in a cross between C57BR/Rr-r cdJ (Rr-s) and C3H/BiDa (S) (17, 19). In contrast, resistance conferred by the Rr-r trait is inherited in a dominant or codominant fashion. The dominance of Rr-r is evident with variable penetrance, depending on the assay. Under the conditions of our tests, the Rr-r trait shows incomplete penetrance in F1 animals, based on protection from tumor development, but complete penetrance in protecting the host from the acute lethality of the virulent LID strain.
Following is a comparison of tumor frequencies in neonatally infected F1 mice produced in crosses between C3H/BiDa, the common susceptible parent, and either C57BR/cdJ (Rr-s) or MA/MyJ (Rr-r), the resistant parent. Newborn F1 animals were inoculated intraperitoneally with the A2 strain of polyomavirus at 2 × 105 to 5 × 105 PFU/animal. F1s between C57BR/cdJ and C3H/BiDa were fully susceptible, with 95% (45 of 47) of the animals developing multiple tumors, with an average latency of 93 days (19). Less than 50% (14 of 32) of F1s whose resistant parent was MA/MyJ developed tumor(s). These F1 animals developed fewer total tumors and fewer tumor types than F1s whose resistant parent was C57BR/cdJ; no tumors in skin or thymus and only rare tumors in bone and kidney were seen. In addition, tumors developed after a considerably longer, 200-day latent period. When the virus dose was reduced, none of the (MA/MyJ × C3H/BiDa) F1 animals developed tumors. C57BR/cdJ × C3H/BiDa F1 mice, though not tested here, are expected to remain fully susceptible to low virus doses, based on the known mechanism of susceptibility operating in this cross (17) and on the fact that C3H/BiDa mice are susceptible to extremely low doses of virus (5).
In an assay based on the survival of newborn F1 mice inoculated with LID, the dominance of the Rr-r trait was complete. Newborn F1 animals (<24 h) were inoculated intraperitoneally with the LID strain of virus at approximately 2 × 105 to 5 × 105 PFU/animal. Survival was measured at 30 days. Virtually 100% (14 of 14) of F1 mice with the resistant parent MA/MyJ survived infection by LID. This result contrasts with 0% survival of F1s (0 of 20) produced by a cross between C57BR/cdJ and C3H/BiDa, two LID-susceptible strains.
DISCUSSION
The resistance of MA/MyJ mice to polyomavirus differs from that of C57BR/cdJ and other resistant strains which mount effective anti-polyomavirus tumor immune responses. Indeed, MA/MyJ mice carry both the MHC type (H-2k) and an endogenous mouse mammary tumor virus superantigen (Mtv-43 sag, with Vβ specificity identical to that of Mtv-7 sag [21]) predictive of a highly susceptible phenotype based on an inability to mount such antitumor responses (17). The fact that their resistance cannot be overcome by immunosuppressive doses of radiation further indicates a nonimmunological basis.
The Rr-r form of resistance exhibited by MA/MyJ mice contrasts with the Rr-s form of C57BR/cdJ in several additional ways. The resistance of MA/MyJ extends in dramatic fashion to newborns inoculated with the virulent LID strain of virus, which kills C57BR/cdJ and other Rr-s strains but not MA/MyJ. Virus replication and spread are sharply curtailed in MA/MyJ compared with those in C57BR/cdJ mice. In crosses with the highly susceptible C3H/BiDa strain, MA/MyJ transmits its resistance in a dominant or codominant manner, while that of C57BR/cdJ is completely recessive. MA/MyJ is unique among the strains we have examined so far in that it exhibits all of the above-mentioned properties. However, the CE/J mouse resembles MA/MyJ in its adult onset Rr-r phenotype, showing resistance to the induction of tumors following radiation as adults but limited susceptibility as newborns.
The Rr-r phenotype is based on a nonimmunological mechanism that blocks dissemination of the virus in vivo. Neither a high level of NK cells nor a high level of antiviral interferons accounts for this form of resistance. The failure of the virus to replicate and spread in vivo does not result from an inability of the host to express virus receptors, since kidney cultures from baby MA/MyJ mice are fully susceptible to infection. A simple deficiency of virus receptors in vivo would also be incompatible with the observed dominance of the Rr-r phenotype. A limited array of tumors arise in roughly half of MA/MyJ mice inoculated with LID as newborns, indicating that at least some cell types in these mice bear receptors for the virus and can be transformed in vivo. These results are consistent with a strong down-regulation of virus spread which spares the animal from the acute lethal effects of the virus while allowing enough dissemination for the induction of some tumors.
Expression in the intact host of some factor(s) that binds and diverts the virus from infecting target cells or acts intracellularly to prevent virus release could, in principle, account for the Rr-r phenotype. An efficient apoptotic response of MA/MyJ mice to viral infection, i.e., one that acts in some way to override the polyomavirus’s antiapoptotic mechanism (4, 20), would also be consistent with the present findings. Experimental approaches to identify the physiological and molecular bases of the Rr-r form of polyomavirus resistance are limited at present due to inability to model the resistance in culture. However, with the relatively rapid assay LID survival test and the clear dominance of the Rr-r trait, a genetic approach to identify the gene(s) involved should be possible.
ACKNOWLEDGMENTS
This work has been supported by Public Health Service grants R35-CA44343 and PO1-CA50661.
We acknowledge the expert technical assistance of Sherrie Witt.
REFERENCES
- 1.Allison A C, Law L W. Effects of antilymphocyte serum on virus oncogenisis. Proc Soc Exp Biol Med. 1968;127:207–212. doi: 10.3181/00379727-127-32657. [DOI] [PubMed] [Google Scholar]
- 2.Bauer P H, Bronson R T, Fung S C, Freund R, Stehle T, Harrison S C, Benjamin T L. Genetic and structural analysis of a virulence determinant in polyomavirus VP1. J Virol. 1995;69:7925–7931. doi: 10.1128/jvi.69.12.7925-7931.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bolen J B, Fisher S E, Chowdhury K, Shan T C, Williams J E, Dawe C J, Israel M A. A determinant of polyomavirus virulence enhances virus growth in cells of renal origin. J Virol. 1985;53:335–339. doi: 10.1128/jvi.53.1.335-339.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dahl J, Jurczak A, Cheng L A, Baker D C, Benjamin T L. Evidence for a role of phosphatidylinositol 3-kinase activation in blocking of apoptosis by polyoma virus middle T antigen. J Virol. 1998;72:3221–3226. doi: 10.1128/jvi.72.4.3221-3226.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dawe C J, Freund R, Mandel G, Ballmer-Hofer K, Talmage D A, Benjamin T L. Variations in polyoma virus genotype in relation to tumor induction in mice: characterization of wild type strains with widely differing tumor profiles. Am J Pathol. 1987;127:243–261. [PMC free article] [PubMed] [Google Scholar]
- 6.Dubensky T W, Freund R, Dawe C J, Benjamin T L. Polyomavirus replication in mice: influences of VP1 type and route of inoculation. J Virol. 1991;65:342–349. doi: 10.1128/jvi.65.1.342-349.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dubensky T W, Murphy F A, Villarreal L P. The detection of DNA and RNA virus genomes in the organ systems of whole mice: patterns of mouse organ infection by polyomavirus. J Virol. 1984;50:779–783. doi: 10.1128/jvi.50.3.779-783.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Eddy B E. Polyoma virus. In: Gard S, Hallauer C, Meyer K F, editors. Virology monographs. Vol. 7. New York, N.Y: Springer-Verlag; 1969. pp. 1–114. [Google Scholar]
- 9.Freund R, Calderone A, Dawe C J, Benjamin T L. Polyomavirus tumor induction in mice: effects of polymorphisms of VP1 and large T antigen. J Virol. 1990;65:335–341. doi: 10.1128/jvi.65.1.335-341.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Freund R, Dawe C J, Benjamin T L. Duplication of noncoding sequences in polyomavirus is required for the development of thymic tumors in mice. J Virol. 1988;62:3896–3899. doi: 10.1128/jvi.62.10.3896-3899.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Freund R, Dubensky T, Bronson R, Sotnikov R, Carroll J, Benjamin T. Polyoma tumorigenesis in mice: evidence for dominant resistance and dominant susceptibility genes of the host. Virology. 1992;191:724–731. doi: 10.1016/0042-6822(92)90248-n. [DOI] [PubMed] [Google Scholar]
- 12.Freund R, Garcea R L, Sahli R, Benjamin T L. A single-amino-acid substitution in polyomavirus VP1 correlates with plaque size and hemagglutination behavior. J Virol. 1991;65:350–355. doi: 10.1128/jvi.65.1.350-355.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Freund R, Sotnikov A, Bronson R T, Benjamin T L. Changes in frequency, morphology and behavior of tumors induced in mice by a polyoma virus mutant with a specifically altered oncogene. Am J Pathol. 1992;141:1409–1425. [PMC free article] [PubMed] [Google Scholar]
- 14.Gross L G. Oncogenic viruses. 3rd ed. Oxford, England: Pergamon Press; 1983. [Google Scholar]
- 15.Law L L. Immunologic responsiveness and the induction of experimental neoplasms. Cancer Res. 1966;26:1121–1132. [PubMed] [Google Scholar]
- 16.Law L L, Dawe C J. Influence of total body X-irradiation on tumor induction by parotid tumor agent in adult mice. Proc Soc Exp Biol Med. 1960;105:414–419. doi: 10.3181/00379727-105-26127. [DOI] [PubMed] [Google Scholar]
- 17.Lukacher A E, Ma Y, Carroll J P, Abromson-Leeman S R, Laning J C, Dorf M E, Benjamin T L. Susceptibility to tumors induced by polyoma virus is conferred by an endogenous mouse mammary tumor virus superantigen. J Exp Med. 1995;18:1683–1692. doi: 10.1084/jem.181.5.1683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lukacher A E, Wilson C S. Resistance to polyoma virus-induced tumors correlates with CTL recognition of an immunodominant H-2Dk-restricted epitope in the middle T protein. J Immunol. 1998;160:1724–1734. [PubMed] [Google Scholar]
- 19.Lukacher A L, Freund R, Carroll J P, Bronson R T, Benjamin T L. Pyvs: a dominantly acting gene in C3H/BiDa mice conferring susceptibility to tumor induction by polyoma virus. Virology. 1993;196:241–248. doi: 10.1006/viro.1993.1472. [DOI] [PubMed] [Google Scholar]
- 20.Meili R, Cron P, Hemmings B A, Ballmer-Hofer K. Protein kinase B/Akt is activated by polyomavirus middle-T antigen via a phosphatidylinositol 3-kinase-dependent mechanism. Oncogene. 1998;16:903–907. doi: 10.1038/sj.onc.1201605. [DOI] [PubMed] [Google Scholar]
- 21.Rudy C K, Kraus E, Palmer E, Huber B T. Mls-l-like superantigen in the MA/MyJ mouse is encoded by a new mammary tumor provirus that is distinct from Mtv-7. J Exp Med. 1992;175:1613–1621. doi: 10.1084/jem.175.6.1613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stehle T, Harrison S C. Crystal structures of polyomavirus in complex with straight-chain and branched-chain sialyloligosaccharide receptor fragments. Structure. 1996;4:183–194. doi: 10.1016/s0969-2126(96)00021-4. [DOI] [PubMed] [Google Scholar]
- 23.Stehle T, Yan Y, Benjamin T L, Harrison S C. The structure of murine polyomavirus complexed with an oligosaccharide receptor fragment. Nature. 1994;369:160–163. doi: 10.1038/369160a0. [DOI] [PubMed] [Google Scholar]
- 24.Ting R C, Law L W. The role of thymus in transplantation resistance induced by polyoma virus. J Natl Cancer Inst. 1965;34:521–527. [PubMed] [Google Scholar]
- 25.Welsh R M, Yang H, Bukowski J F. The role of interferon in the regulation of virus infections by cytotoxic lymphocytes. Bioessays. 1988;8:10–13. doi: 10.1002/bies.950080104. [DOI] [PubMed] [Google Scholar]