Abstract
Background
In patients with axial spondyloarthritis (axSpA) or psoriatic arthritis (PsA), concomitant depression might have a negative impact on the course of disease and treatment outcomes. The aims of this analysis are to determine the prevalence of depressive symptoms in axSpA and PsA patients in a real-world cohort study and to identify sociodemographic and clinical associated factors for moderate or severe depressive symptoms in both diseases.
Methods
Patients from the RABBIT-SpA cohort with an axSpA or PsA diagnosis and a valid WHO-5 Well-Being Index score at baseline were included. A descriptive analysis of baseline and outcome parameters by category of depressive symptoms was performed and factors associated with the presence of depressive symptoms (moderate or severe) were examined in a logistic regression.
Results
Two thousand four hundred seventy patients (1,245 axSpA; 1,225 PsA) were included in the analysis. In both diagnoses, the proportion of patients with moderate depressive symptoms was 8% and 21% with severe symptoms.
Patients with moderate or severe depressive symptoms were less likely to engage in sports than those with no or mild depressive symptoms, had more comorbidities and higher scores for disease activity, functional limitations, fatigue, and pain and took more analgesics.
In axSpA, patients with a higher disease activity, a greater functional impairment and more severe fatigue were more likely to experience depressive symptoms, while patients with more years in education and engaging in sports for at least 1 h/week were less likely to experience depressive symptoms. PsA patients with a greater functional impairment and more severe fatigue were more likely to experience depressive symptoms while those engaging in sports for at least 1 h/week were less likely to experience depressive symptoms.
Conclusion
We confirmed a high prevalence of depressive symptoms in both PsA and axSpA. Factors negatively associated with the presence of depressive symptoms were fatigue, not engaging in sports, and greater functional limitations. Depressive symptoms may affect the perception of disease activity / severity by patients. Thus, depressive symptoms are an important condition in axSpA and PsA that should be considered when evaluating disease activity and treatment outcomes.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13075-023-03127-2.
Keywords: Observational study, Spondyloarthritis, Psoriatic arthritis, Depression
Background
Axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) are chronic inflammatory diseases that result in a high burden of disease and reduced function and quality of life [1, 2]. AxSpA is characterised by chronic inflammatory back pain as its main symptom, often starting in the third decade of life. The most common peripheral manifestations are arthritis and enthesitis. Extra-articular manifestations can occur, such as acute anterior uveitis, psoriasis or inflammatory bowel disease [2]. PsA is a multifaceted disease that affects up to 30% of psoriasis patients [1]. Commonly, musculoskeletal symptoms start after years of psoriatic skin manifestations but, in approximately 15% of patients, both can appear at the same time or arthritis can start before any skin symptoms. Those affected suffer from painful inflammation of the joints or spine as well as pathological changes in the skin and nails.
Chronic pain and depression often occur together, causing a greater burden of disease than either condition alone; pain can lead to depression, and depression can increase the perception of physical pain [3]. In both axSpA and PsA, the relevant scores used to assess treatment success and to guide therapeutic decisions are either informed by patient reported outcome measures (PROMs), such as the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) or Bath Ankylosing Spondylitis Functional Index (BASFI) for axSpA or are composite scores containing PROMs (Ankylosing Spondylitis Disease Activity Score c-reactive protein (ASDAS-CRP) for axSpA and Disease Activity in Psoriatic Arthritis (DAPSA) for PsA). Thus, recognizing an existing depression in chronic pain patients and treating both conditions is of great importance.
A high prevalence of depressive symptoms has been shown for axSpA [4–6] and PsA patients, though for the latter, data is still comparatively rare [7, 8]. While data is still scarce, there are some studies suggesting that depressive symptoms independently, negatively influence treatment outcomes in rheumatoid arthritis [9], axSpA [10] and PsA [11].
The aims of this analysis are to 1) determine the prevalence of depressive symptoms in axSpA and PsA patients in the RABBIT-SpA cohort study, 2) identify sociodemographic and clinical differences between patients with no or mild depressive symptoms and those with moderate or severe symptoms, 3) analyse associated factors of depressive symptoms in axSpA and PsA patients.
Methods
Patients
Rabbit-SpA is an observational, longitudinal cohort study, started by the German Rheumatism Research Centre in 2017. The main aim of the study is to examine long-term safety and effectiveness of biologic and targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) in patients with a diagnosis of either axSpA or PsA. Participating rheumatology clinics or practices all over Germany enrol patients with at least one prior failure of a systemic therapy. Patients receiving a new b/tsDMARD are included in the treatment group, while patients receiving a conventional systemic treatment are included in the control group. Patients of both groups were included in this analysis.
Documentation is web-based, however, patients can also fill in paper questionnaires in the study centres. Both physicians and patients fill in case report forms (CRFs) at baseline, after 3 months and 6 months and every 6 months thereafter, for a maximum follow-up time of 10 years.
The physician CRF contains data on onset of first symptoms, first contact with a rheumatologist and date of first diagnosis. Information on comorbidities and extra-articular manifestations is collected as well as current and past antirheumatic therapy, both medical and non-medical. Disease activity is measured using a variety of instruments, including established composite scores of disease activity, clinical parameters such as laboratory values and imaging results. The patient questionnaire contains sociodemographic information and a number of PROMs (for example, BASDAI and BASFI for axSpA and the Dermatology Life Quality Index (DLQI) and Health Assessment Questionnaire (HAQ) for PsA). A more detailed description of the study can be found elsewhere [12, 13].
All patients recruited since the beginning of the study (May 2017) until database closure on March 1st, 2022 were included in the present analysis if they had a baseline physician’s CRF and a baseline patient’s CRF with a complete WHO-5 Well-Being Index (WHO-5).
Definition of variables
Depressive symptoms
Depressive symptoms were measured with the 5-item WHO-5, which is frequently used as a screening tool for depression [14]. It consists of 5 questions assessing subjective psychological well-being in the last 14 days, on a scale of 0 (never) to 5 (all of the time):
1: ‘I have felt cheerful and in good spirits’, 2: ‘I have felt calm and relaxed’, 3: ‘I have felt active and vigorous’, 4: ‘I woke up feeling fresh and rested’ and 5: ‘My daily life has been filled with things that interest me’.
The scores of the single items are added and multiplied by 4 to obtain a WHO-5 percentage score between 0 and 100. Established cut-off values [5, 15–17] were used to separate the score into the following four groups: Severe depressive symptoms indicated by values < 13, values between 13 and 28 suggest moderate depressive symptoms, values between 29 and 50 mild depressive symptoms, while values above 50 indicate good well-being and no depressive symptoms.
Independent variables
Independent variables were selected a priori, based on findings from the literature [2, 18, 19]. Sex, disease duration (years since diagnosis), obesity (BMI > = 30), arthritis (only in axSpA patients, 0–44 joints, dichotomized to yes (1 or more affected joints) vs. no), and enthesitis (0–16 affected enthesial points, dichotomized to yes (1 or more affected points) vs. no) were physician reported. Data on comorbidities was also collected from the rheumatologist, using a checklist of comorbidities relevant to axSpA and PsA, including for example cardiovascular diseases but also depression. The checked comorbidities were counted and the result categorized (‘0–2 comorbidities’ vs. ‘3 or more comorbidities’). Education (dichotomized to > = 10y vs. < 10y) and engaging in sports (‘How often do you engage in sport?’ with possible answers ‘more than 4 h a week’, ‘2 to 4 h a week’,’1 to 2 h a week’, ‘less than one hour a week’ and ‘do not engage in sport’) were collected in the patient CRF. Disease activity was measured with the instruments recommended for the respective diagnosis (ASDAS (CRP) for axSpA, DAPSA for PsA) by an international task force [20]. Functional impairment was measured by BASFI in axSpA and HAQ in PsA. In axSpA patients, fatigue was measured with the first BASDAI question: ‘How would you describe the overall level of fatigue / tiredness you have experienced [in the last 7 days]?’ on a scale from 0 ‘no fatigue/tiredness’ to 10 ‘very severe fatigue/tiredness’. In PsA patients, the second question of the Psoriatic Arthritis Impact of Disease 12-item questionnaire was used: ‘How severe was your tiredness / fatigue due to your psoriatic arthritis in the last week?’ on a scale from 0 ‘no tiredness’ to 10 ‘complete exhaustion’. Also in PsA patients, body surface area (BSA, physician-reported, 0–100%) was used to measure the extent of skin involvement and DLQI (patient reported) to measure the impact of skin involvement on quality of life.
Statistical analysis
Except for c-reactive protein (CRP) and disease duration, the proportion of missing values for analysis parameters was between 0 and 3%. For CRP, there were 15% missing values in axSpA patients and 11% in PsA patients. The ASDAS (CRP) score used in axSpA patients also had 15% missing values. For disease duration it was 7% and 9%, respectively. Therefore, data were imputed multiply (10 times), using fitted regression models [21]. Combined estimates were obtained by combining imputations.
For both diagnoses descriptive statistics as means (± standard deviation) and percentages were used to compare patients with ‘severe depressive symptoms’, ‘moderate depressive symptoms’ and ‘no or mild depressive symptoms’.
To identify sociodemographic or clinical factors that are associated with depressive symptoms, moderate and severe depressive symptoms (WHO-5 ≤ 28) were combined and compared to patients with no or mild depressive symptoms. For these two groups, a multivariate logistic regression was used to determine associations between independent variables (sex, education, comorbidities, engaging in sports, disease activity, physical function, and fatigue) and depressive symptoms. Adjusted odds ratios (OR) are given with 95% confidence intervals (95% CI).
Separate regression models were fitted for axSpA and PsA.
Calculations were carried out with the software package SAS, version 9.4 (SAS Institute, Cary, USA) and R, version 3.5.3 (R Foundation for Statistical Computing, Vienna, Austria).
Results
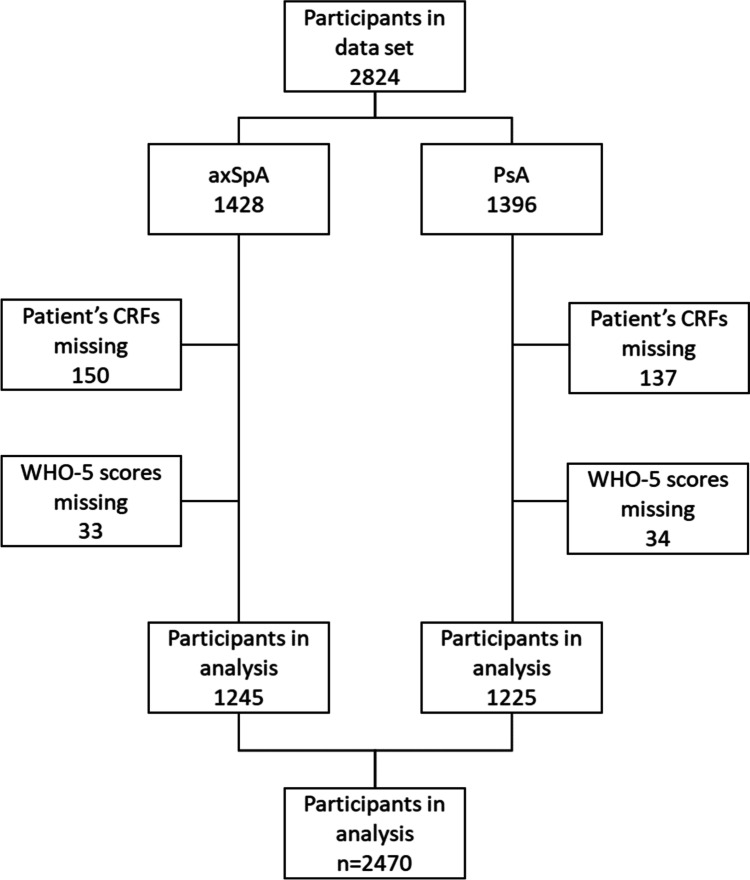
A total of 2470 patients were included in the analysis, 1245 with axSpA and 1225 with PsA. Patients were excluded if they had not submitted a patient’s questionnaire or if the WHO-5 score was missing or incomplete (see Fig. 1 for a detailed breakdown of patient selection).
Fig. 1.
Patient selection chart
Comparison of sociodemographic and clinical characteristics of axSpA and PsA patients
As demonstrated in Table 1, PsA patients tended to be older and more often female than axSpA patients. A higher proportion were obese or had three or more comorbidities. Physician- and patient-reported disease activity was similar in both diagnoses. The distribution of WHO-5 scores was also similar across patients with axSpA and PsA diagnoses. Among PsA patients, 8.2% had severe and 20.9% had moderate depressive symptoms compared to 8.2% and 21.5%, respectively, among axSpA patients. See Table 1 for patient characteristics stratified by diagnosis.
Table 1.
Characteristics of patients by diagnosis
| axSpA | PsA | Total | |
|---|---|---|---|
| n | 1245 | 1225 | 2470 |
| Age, years, mean (SD) | 44.5 ± 13 | 52.1 ± 12.3 | 48.3 ± 13.2 |
| Sex, female, n (%) | 561 (45) | 719 (59) | 1280 (52) |
| Obesity (BMI ≥ 30), n (%) | 316 (26) | 458 (38) | 774 (32) |
| Naive to biologic therapy, n (%) | 636 (61) | 594 (57) | 1230 (59) |
| Comorbidities, 3 or more, n (%) | 241 (19) | 382 (31) | 623 (25) |
| Physician global disease activity, NRS 0–10, mean (SD) | 5.4 ± 1.9 | 5.1 ± 1.9 | 5.3 ± 1.9 |
| Patient global disease activity, NRS 0–10, mean (SD) | 5.8 ± 2.4 | 5.7 ± 2.4 | 5.8 ± 2.4 |
| WHO-5 score, mean (SD) | 44 ± 21.4 | 45.8 ± 22.8 | 44.9 ± 22.1 |
| WHO-5 score: Severe depressive symptoms, n (%) | 102 (8) | 101 (8) | 203 (8) |
| WHO-5 score: Moderate depressive symptoms, n (%) | 268 (22) | 256 (21) | 524 (21) |
| WHO-5 score: No or mild depressive symptoms, n (%) | 875 (70) | 868 (71) | 1743 (71) |
NRS Numerical rating scale, WHO-5 WHO-5 Well-Being Index
Comparison of characteristics by category of depressive symptoms in axSpA patients
Characteristics of axSpA patients, stratified by the three WHO-5 categories, are shown in Table 2. Patients with moderate as well as those with severe depressive symptoms were more often female than those with no or mild depressive symptoms, they were more often obese and fewer of them were currently employed. Patients with moderate or severe depressive symptoms had a slightly shorter disease duration, were less often naive to biologic therapy at the time of enrolment and a higher proportion of these patients were taking systemic corticosteroids and both non-opioid and opioid pain medication. They had a higher number of comorbidities and more often a depression as a comorbidity. With higher levels of depressive symptoms, all measures of disease activity and functional impairment, fatigue, pain and sleep disturbance worsened. Patients with moderate or severe depressive symptoms were more often current smokers, had a lower level of education and a lower proportion of them were engaging in sports for at least 1 h/week.
Table 2.
Characteristics of axSpA patients by depressive symptoms
| Parameter | No or mild depressive symptoms N = 875 |
Moderate depressive symptoms N = 268 |
Severe depressive symptoms N = 102 |
Total N = 1245 |
|---|---|---|---|---|
| Age, years, mean (SD) | 44.5 ± 13.2 | 43.9 ± 12.7 | 45.7 ± 11.8 | 44.5 ± 13 |
| Sex, female, n (%) | 384 (44) | 128 (48) | 49 (48) | 561 (45) |
| Disease duration, years, mean (SD) | 7.1 ± 9.3 | 6.5 ± 8.7 | 6.4 ± 8.1 | 6.9 ± 9.1 |
| School, ≥ 10 years, n (%) | 723 (84) | 203 (78) | 71 (70) | 997 (81) |
| Currently employed, n (%) | 688 (82) | 184 (75) | 71 (76) | 943 (80) |
| Current smoking, n (%) | 318 (37) | 116 (44) | 47 (48) | 481 (40) |
| Engaging in sports, ≥ 1 h/week, n (%) | 500 (57) | 122 (46) | 30 (29) | 652 (53) |
| Obesity (BMI ≥ 30), n (%) | 207 (24) | 80 (30) | 29 (28) | 316 (26) |
| Comorbidities, 3 or more, n (%) | 151 (17) | 59 (22) | 31 (30) | 241 (19) |
| Depression as known comorbidity, n (%) | 42 (5) | 27 (10) | 18 (18) | 87 (7) |
| Of those, depression treated, n (%) | 23 (55) | 17 (63) | 13 (72) | 53 (61) |
| Fibromyalgia as comorbidity, n (%) | 17 (2) | 7 (3) | 6 (6) | 30 (2) |
| Naive to biologic therapy, n (%) | 462 (63) | 128 (56) | 46 (49) | 636 (61) |
| Systemic glucocorticoids, n (%) | 142 (16) | 57 (22) | 21 (21) | 220 (18) |
| Non-opioid analgesics, n (%) | 84 (10) | 39 (15) | 18 (19) | 141 (12) |
| Opioids, n (%) | 39 (5) | 21 (8) | 11 (11) | 71 (6) |
| HLA-B27 positive, n (%) | 626 (76) | 186 (74) | 67 (68) | 879 (75) |
| ASAS criteria fulfilled, n (%) | 685 (78) | 214 (80) | 78 (76) | 977 (78) |
| CRP ≥ 5 mg/l, n (%) | 387 (52) | 125 (55) | 57 (63) | 569 (54) |
| Arthritis, joint count (0–44), mean (SD) | 0.9 ± 2.4 | 1.7 ± 4.1 | 0.6 ± 1.7 | 1.1 ± 2.8 |
| Enthesitis, number of sites (0–16), mean (SD) | 0.4 ± 1.3 | 0.6 ± 1.6 | 0.5 ± 1.4 | 0.5 ± 1.4 |
| ASDAS (CRP), mean (SD) | 2.6 ± 1 | 3.3 ± 0.9 | 3.7 ± 0.9 | 2.9 ± 1 |
| Physician global disease activity, NRS 0–10, mean (SD) | 5.2 ± 2 | 5.9 ± 1.6 | 6.2 ± 1.7 | 5.4 ± 1.9 |
| BASDAI, mean (SD) | 4.1 ± 1.8 | 5.8 ± 1.6 | 6.7 ± 1.5 | 4.7 ± 2 |
| BASFI, mean (SD) | 3.2 ± 2.2 | 5 ± 2.2 | 6.2 ± 2.1 | 3.8 ± 2.4 |
| ASAS-HI Score (0–17), mean (SD) | 5.7 ± 3.2 | 9.2 ± 3.1 | 11.2 ± 2.9 | 6.9 ± 3.7 |
| ASAS-HI: good/very good (0–5), n (%) | 404 (47) | 29 (11) | 3 (3) | 436 (36) |
| ASAS-HI: moderate (6–12), n (%) | 438 (51) | 167 (65) | 57 (57) | 662 (54) |
| ASAS-HI: poor/very poor (> = 12), n (%) | 22 (3) | 61 (24) | 40 (40) | 123 (10) |
| Patient global disease activity, NRS 0–10, mean (SD) | 5.2 ± 2.3 | 7 ± 1.8 | 7.6 ± 2 | 5.8 ± 2.4 |
| Patient: Pain, NRS 0–10, mean (SD) | 5.1 ± 2.3 | 6.8 ± 1.9 | 7.5 ± 1.8 | 5.6 ± 2.3 |
| Patient: fatigue, NRS 0–10 mean (SD) | 4.8 ± 2.3 | 6.9 ± 1.8 | 7.5 ± 1.8 | 5.5 ± 2 |
| Patient: Sleep disturbance, NRS 0–10, mean (SD) | 4.7 ± 2.9 | 6.9 ± 2.4 | 7.7 ± 2.4 | 5.4 ± 3 |
| Patient: Current work ability, NRS 0–10 (higher is better), mean (SD) | 5.6 ± 2.3 | 3.8 ± 2.5 | 3.1 ± 2.6 | 5.1 ± 2.6 |
ASAS criteria ASAS Criteria for Axial Spondyloarthritis, ASAS-HI Assessment of SpondyloArthritis International Society Health Index, ASDAS (CRP) Ankylosing Spondylitis Disease Activity Score c-reactive protein, axSpA Axial spondyloarthritis, BASDAI Bath Ankylosing Spondylitis Disease Activity Index, BASFI Bath Ankylosing Spondylitis Functional Index, CRP C-reactive protein, HLA-B27 Human leukocyte antigen B27, NRS Numerical rating scale
Comparison of characteristics by category of depressive symptoms in PsA patients
PsA patients in all three groups compared were of similar age (Table 3). Patients with moderate or severe depressive symptoms were more often female, fewer of them had at least 10 years of school education or were currently employed. Among patients with severe depressive symptoms, the percentages of obese patients and those taking pain medications were substantially higher than in the two other groups, and this was even more pronounced than in axSpA. Unlike axSpA patients, PsA patients with severe depressive symptoms had the longest disease duration. In patients with moderate or severe depressive symptoms, more had at least three comorbidities. The proportion of patients with documented depression or fibromyalgia was higher in the groups of patients with moderate or severe depressive symptoms. Just as in axSpA patients, measures of disease activity and functional impairment, fatigue, pain and sleep disturbance were all worse in those with more severe depressive symptoms compared to patients with no or mild depressive symptoms.
Table 3.
Characteristics of PsA patients by depressive symptoms
| Parameter | No or mild depressive symptoms N = 868 |
Moderate depressive symptoms N = 256 |
Severe depressive symptoms N = 101 |
Total N = 1225 |
|---|---|---|---|---|
| Age, years, mean (SD) | 52.3 ± 12.8 | 51.3 ± 10.9 | 52.8 ± 11.1 | 52.1 ± 12.3 |
| Sex, female, n (%) | 489 (56) | 158 (62) | 72 (71) | 719 (59) |
| Disease duration, years, mean (SD) | 6 ± 7.1 | 6.7 ± 8.3 | 7.8 ± 8.1 | 6.3 ± 7.4 |
| School, ≥ 10 years, n (%) | 680 (80) | 192 (76) | 75 (75) | 947 (79) |
| Currently employed, n (%) | 555 (67) | 155 (65) | 52 (54) | 762 (66) |
| Current smoking, n (%) | 230 (28) | 82 (33) | 37 (37) | 349 (30) |
| Engaging in sports, ≥ 1 h/week, n (%) | 386 (46) | 87 (35) | 29 (29) | 502 (42) |
| Obesity (BMI ≥ 30), n (%) | 304 (36) | 98 (39) | 56 (55) | 458 (38) |
| Comorbidities, 3 or more, n (%) | 247 (29) | 89 (35) | 46 (46) | 382 (31) |
| Depression as known comorbidity, n (%) | 64 (7) | 60 (23) | 25 (25) | 149 (12) |
| Of those, depression treated, n (%) | 44 (69) | 39 (65) | 19 (76) | 102 (68) |
| Fibromyalgia as comorbidity, n (%) | 30 (4) | 21 (8) | 11 (11) | 62 (5) |
| Naive to biologic therapy, n (%) | 442 (60) | 107 (50) | 45 (50) | 594 (57) |
| Systemic glucocorticoids, n (%) | 255 (30) | 92 (36) | 46 (46) | 393 (32) |
| Non-opioid analgesics, n (%) | 92 (12) | 36 (16) | 33 (34) | 161 (15) |
| Opioids, n (%) | 31 (4) | 21 (9) | 15 (16) | 67 (6) |
| CRP ≥ 5 mg/l, n (%) | 314 (41) | 104 (44) | 41 (45) | 459 (42) |
| Tender joint count (0–68), mean (SD) | 6.4 ± 7.7 | 7.2 ± 7.2 | 11.1 ± 10.7 | 6.9 ± 8.0 |
| Swollen joint count (0–66), mean (SD) | 3.0 ± 4.1 | 3.5 ± 4.7 | 4.7 ± 6.2 | 3.3 ± 4.4 |
| Enthesitis, number of sites (0–16), mean (SD) | 2.9 ± 2.5 | 3.6 ± 2.1 | 3.8 ± 3.2 | 3.2 ± 2.5 |
| Nail psoriasis, n (%) | 344 (40) | 103 (40) | 50 (50) | 497 (41) |
| Body surface area, mean (SD) | 8.3 ± 14.6 | 8.9 ± 16.2 | 10.2 ± 17.9 | 8.6 ± 15.2 |
| DAPSA, mean (SD) | 20.6 ± 12.5 | 24.8 ± 12.7 | 32.6 ± 15.9 | 22.5 ± 13.3 |
| Physician global disease activity, NRS 0–10, mean (SD) | 4.9 ± 1.8 | 5.5 ± 1.8 | 6.3 ± 1.6 | 5.1 ± 1.9 |
| Patient global disease activity, NRS 0–10, mean (SD) | 5.2 ± 2.3 | 6.6 ± 2.1 | 8 ± 2 | 5.7 ± 2.4 |
| Patient: DLQI, mean (SD) | 4.9 ± 5.5 | 7.2 ± 6.9 | 9.2 ± 8.2 | 5.8 ± 6.2 |
| Patient: HAQ, mean (SD) | 0.8 ± 0.6 | 1.2 ± 0.6 | 1.6 ± 0.6 | 0.9 ± 0.7 |
| Patient: Fatigue, NRS 0–10, mean (SD) | 4.4 ± 2.2 | 6.8 ± 2.0 | 8.1 ± 1.7 | 5.2 ± 2.7 |
| Patient: Pain, NRS 0–10, mean (SD) | 5 ± 2.3 | 6.4 ± 2.1 | 7.8 ± 1.7 | 5.5 ± 2.4 |
| Patient: Sleep disturbance, NRS 0–10, mean (SD) | 4.2 ± 2.9 | 6.3 ± 2.7 | 7.2 ± 2.7 | 4.9 ± 3.1 |
| Patient: Current work ability, NRS 0–10 (higher is better), mean (SD) | 5.6 ± 2.8 | 3.7 ± 2.7 | 3.5 ± 2.9 | 5 ± 2.9 |
CRP C-reactive protein, DAPSA Disease Activity in Psoriatic Arthritis, DLQI Dermatology Life Quality Index, HAQ Health Assessment Questionnaire, NRS Numerical rating scale
Discrepancy between physician-reported and patient-reported disease activity
In both axSpA and PsA patients, the mean values of physician and patient-reported global disease activity were remarkably similar in the group of patients with no or mild depressive symptoms (Fig. 2a and b). However, patients with moderate depressive symptoms rated themselves worse than their rheumatologist and in patients with severe depressive symptoms, that discrepancy was even greater.
Fig. 2.
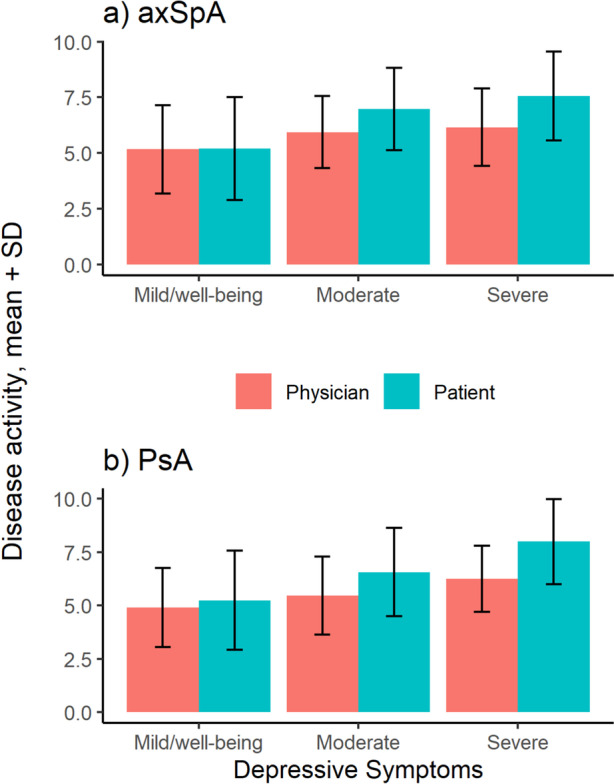
Physician-rated vs. patient-rated global disease activity by category of depressive symptoms a) axSpA; b) PsA. axSpA: Axial spondyloarthritis; PsA Psoriatic arthritis
Factors associated with the presence of depressive symptoms
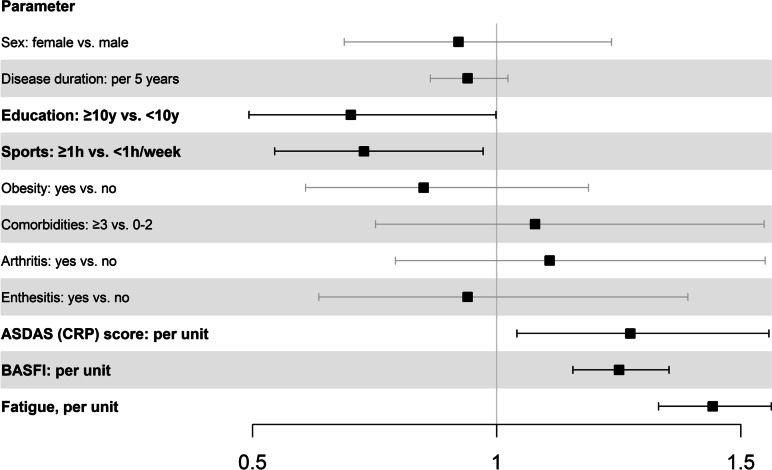
AxSpA patients with 10 or more years of education (OR = 0.70, 95% CI = 0.49–1.00) and patients who engaged in sports for at least 1 h a week (OR = 0.73, 95% CI = 0.54–0.97) were less likely to experience depressive symptoms. Patients with a higher disease activity as measured by ASDAS (CRP) (OR = 1.27, 95% CI = 1.04–1.56) and a higher functional impairment as measured by BASFI (OR = 1.25, 95% CI = 1.15–1.35) were more likely to experience depressive symptoms. Fatigue was most strongly associated with depressive symptoms (OR = 1.44, 95% CI = 1.33–1.56), see Fig. 3, Suppl. Table S1.
Fig. 3.
Adjusted OR (± 95% CI) of factors associated with the presence of symptoms suggestive of depression (WHO-5 score of ≤ 28) in axSpA patients: results from multivariable logistic regression. ASDAS (CRP): Ankylosing Spondylitis Disease Activity Score; BASFI: Bath Ankylosing Spondylitis Functional Index c-reactive protein
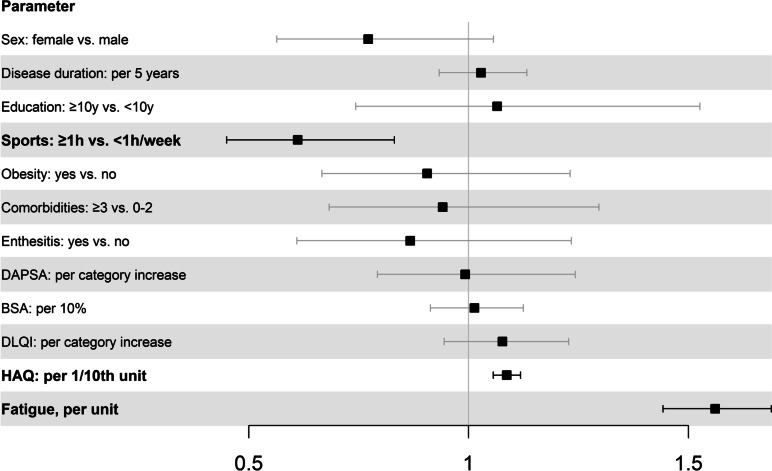
PsA patients who engaged in sports for at least 1 h a week (OR = 0.61, 95% CI = 0.45–0.83) were less likely to experience depressive symptoms. Patients with higher functional impairment as measured by HAQ (OR = 1.08, 95% CI = 1.06–1.12) were more likely to experience depressive symptoms. As in axSpA patients, the strongest association was with fatigue (OR = 1.56, 95% CI = 1.44–1.69), see Fig. 4, Suppl. Table S2.
Fig. 4.
Adjusted OR (± 95%CI) of factors associated with the presence of symptoms suggestive of depression (WHO-5 score of ≤ 28) in PsA patients: results from multivariable logistic regression. BSA: Body surface area; DAPSA: Disease Activity in Psoriatic Arthritis; DLQI: Dermatology Life Quality Index; HAQ: Health Assessment Questionnaire
Discussion
Depressive symptoms are common in axSpA as well as in PsA with a prevalence of almost 30% in our cohort. The multivariate analysis showed that depressive symptoms are associated with fatigue, not engaging in sports and increased functional impairment in both diagnoses. In axSpA patients, fewer years in education and higher disease activity measured by ASDAS (CRP) are also associated with depressive symptoms.
The WHO-5 has been shown to be a suitable screening tool for depression [16, 22] and has been validated in a number of populations and settings [17, 23–27]. A study comparing the WHO-5, Patient Health Questionnaire 9 (PHQ-9) and Hospital Anxiety and Depression Scale (HADS) for the purpose of screening for depressive disorders, using the Structured Clinical Interview for DSM-IV as the criterion standard, found all three to perform well [16].
A recent systematic review of 39 studies, measuring the prevalence of depression or depressive symptoms in axSpA patients, found a large heterogeneity with prevalence estimates ranging from 10 to 64%. This is due, at least in part, to the use of different instruments to measure depression and to the use of different cut off values [6]. A previous analysis by our group [5], also using the WHO-5, found a prevalence of moderate to severe depressive symptoms of 31% in axSpA patients in Germany, using claims data that were linked to a cross-sectional survey. This is very similar to the prevalence we found in our cohort of patients from a German disease register.
Similarly heterogeneous are the studies on the prevalence of depression in PsA patients, summarised by Zusman et al. [8]. The authors found 18 studies reporting on the prevalence of depression, with prevalences ranging from 9 to 70%. The pooled prevalence was 17% (95% CI 13 – 21%). Depression was assessed using the PHQ-9, the HADS with different cut-offs or a documented ICD-9 or ICD-10 code. The prevalence found in our analysis fits well into the described range.
In the aforementioned analysis of factors associated with an outcome of moderate to severe depressive symptoms (≤ 28 points in the WHO-5) in axSpA patients [5], the authors identified a higher disease activity (BASDAI), functional impairment (BASFI), a lower income, self-reported stress and lack of exercise, and younger age as factors associated with moderate-to-severe depressive symptoms. A systematic review identified eight studies that were examining the association between depression and disease activity or functional impairment. All studies reported significantly poorer BASDAI and BASFI values for patients with depression compared to those without [6]. Another analysis, using longitudinal data and general estimating equation models to examine associations between sociodemographic and disease-specific factors and well-being (measured with the Bath Ankylosing Spondylitis Patient Global Score), found reduced well-being to be associated with pain, fatigue and physical functioning but not sociodemographic factors like age, sex or educational level [28].
In our analysis, not engaging in sports, disease activity (ASDAS (CRP)) and impaired functional capacity as well as fatigue were associated with depressive symptoms in axSpA patients, while education was a protective factor. We decided to use the composite score ASDAS (CRP) as disease activity measure, because it has emerged as the most appropriate instrument for disease activity [29].
Relatively few analyses of factors associated with depressive symptoms in PsA patients were found by us. One [30] showed abnormal physical function (Health Assessment Questionnaire Disability Index (HAQ-DI) ≥ 0.5) and elevated Interleukin-6 to be associated with depressive symptoms, another [31] identified DAPSA and HAQ-DI as being associated with depressive symptoms. In another study [32], patients who were employed were less likely to experience depressive symptoms while those who exhibited higher levels of fatigue were more likely to experience depressive symptoms. We identified not engaging in sports, impaired physical function and fatigue as factors associated with depressive symptoms. DAPSA, a recommended composite score to measure disease activity [20], was not associated with depressive symptoms.
Disease activity in axSpA, as measured by ASDAS (CRP), was associated with depressive symptoms. In contrast, there was no correlation between depressive symptoms and disease activity by DAPSA score in PsA patients. ASDAS (CRP) relies on PROMs (pain, morning stiffness and patient global disease activity) and CRP while DAPSA relies on tender and swollen joint count, CRP, and PROMs (patient global disease activity and pain). Hence, these composite scores measure disease activity very differently and the influence of PROMs on the total score result is much higher in the case of ASDAS (CRP). There is data suggesting that PROMs like fatigue and pain are closely related to mental health [33, 34] and therefore composite scores that rely mainly on PROMs are more influenced by mental health. An analysis from the Danish DANBIO Registry indicates that there may be a risk of overtreatment in rheumatoid arthritis patients with psychological problems or central sensitization where the disease activity calculation is based mainly on PROMs [35]. Another analysis from Portugal compared patients in remission to patients where only an insufficient patient global assessment prevented the achievement of remission [36]. The authors conclude that a modified treat to target approach, distinguishing between (objective) disease activity and the impact of disease on patients as a separate target might be beneficial and avoid unnecessary therapy escalations.
The association of fatigue with depressive symptoms is expected, as fatigue is an important characteristic of depression. Fatigue is a very common symptom in axSpA [37] and PsA [38]. As the first question of the BASDAI covers fatigue it is not surprising that the total BASDAI score is also influenced by depressive symptoms as shown previously [5].
The impact of depression or other mental health problems such as anxiety on the effectiveness of antirheumatic treatments has not been studied extensively yet. In a study from the Toronto PsA clinic, the probability of reaching minimal disease activity was reduced in the presence of depressive symptoms [11]. Similar results were found by Michelsen et al. for Rheumatoid arthritis and PsA [9]. In an analysis from the British Society for Rheumatology Biologics Register in Ankylosing Spondylitis (BSRBR-AS), the authors found a higher number of comorbidities and poor mental health to be negatively associated with treatment response, as were sociodemographic factors like fewer years in education and not working full-time [10]. In a follow-up analysis the authors found that a reduction of depressive symptoms from moderate/severe to ‘less than mild’ (0–7 points as measured by HADS) would improve treatment response by approximately 2 BASDAI units and reduce treatment discontinuation by a third [39]. Another analysis from BSRBR-AS showed symptoms of anxiety and depression to be independently associated with poorer treatment outcomes, both continuous disease parameters and binary response criteria.
Interestingly, we found an association between depressive symptoms and a discordance between physician and patient rated global disease activity. In patients with no or mild depressive symptoms, the mean values of patient and physician global disease activity were concordant. However, patients with moderate depressive symptoms rated their disease activity higher, and in patients with severe depressive symptoms, this discordance was even more pronounced. Discordance between physician and patient scores of global disease activity has been described before. In axSpA patients, Desthieux et al. [40] found the proportion of patients with a discordant disease activity assessment (defined as a difference of at least three points) to be between 26 and 29% from baseline to three years after baseline. Discordance was associated with spine pain and fatigue.
It has been shown that the discrepancy between patient- and physician- rated symptom severity is associated with depression [41]. Similar results were also found in psoriasis, with greater degree of discordance in patients with mental health disorders [42].
This discordance has implications for shared decision making in chronic conditions, which relies on similar assessment of disease states by patient and physician. Disagreement on disease activity and treatment success may lead to reduced patient satisfaction with treatment decisions and thus reduced adherence and treatment response. Physicians need to be aware of both comorbid depressive symptoms and the associated risk of discordant assessment of disease activity.
Another interesting finding in our analysis is the increasing intake of analgesics including opioids in axSpA and PsA patients associated with depressive symptoms. This goes along with higher levels of pain.
A limitation of the present analysis is the instrument to measure depressive symptoms, the WHO-5. This instrument was chosen because it is a validated screening tool for depression. It consists of only five questions and is therefore feasible in large longitudinal cohort studies with multiple research questions. The feasibility is evident from the very low number of missing values for the WHO-5 score. However, it is not a diagnostic instrument, therefore, our data needs to be interpreted carefully. However, we are confident that our results regarding depressive symptoms are robust and indicate a clinically relevant problem. Another limitation is that patients are included into our cohort at the start of a new systemic therapy, hence in a situation in which there is a need for treatment intensification. While it is a strength that this is the same for all patients, it can be expected that patients are at a low point and may exhibit more severe depressive symptoms than before or after inclusion. Another important limitation is the cross-sectional design of this analysis, therefore no causal relationships can be analysed.
Conclusion
In our study of patients starting a new systemic therapy, depressive symptoms are common in axSpA and PsA. Depressive symptoms are associated with fatigue, not engaging in sports, and limited physical function. We conclude that mental health needs to be taken into account when assessing a patient’s situation in a more holistic way. A multidisciplinary treatment approach including psychotherapeutic interventions may help to optimize the management of disease in axSpA and PsA patients. A better understanding of the influence of depressive symptoms on the effectiveness of the antirheumatic treatment is an important future research task.
Supplementary Information
Additional file 1: Table S1. Adjusted OR (±95% CI) of parameters associated with the presence of symptoms suggestive of depression (WHO-5 score of ≤28) in axSpA patients: results from multivariable logistic regression. Table S2. Adjusted OR (±95% CI) of parameters associated with the presence of symptoms suggestive of depression (WHO-5 score of ≤28) in PsA patients: results from multivariable logistic regression.
Acknowledgements
The authors acknowledge the invaluable contributions of all participating consultant rheumatologists and their patients. In particular, we would like to thank those rheumatologists who enrolled the highest numbers of patients: Kaufmann J, Schwarze I, Bohl-Bühler M, Feuchtenberger M, Eisterhus C, Kellner H, Klopsch T, Zinke S, Spengler L, Karberg K, Krüger K, Wernitzsch H, Marycz T, Liebhaber A, Stille C, Krause A, Müller B, Fricke-Wagner H, Kühne C, Dahmen G, Tony H-P, Burmester G, Baumann C, and Häckel B.
Abbreviations
- ASAS criteria
ASAS Criteria for Axial Spondyloarthritis
- ASAS-HI
Assessment of SpondyloArthritis International Society Health Index
- ASDAS (CRP)
Ankylosing Spondylitis Disease Activity Score
- axSpA
Axial spondyloarthritis
- BASDAI
Bath Ankylosing Spondylitis Disease Activity Index
- BASFI
Bath Ankylosing Spondylitis Functional Index
- bDMARD
Biologic disease modifying anti-rheumatic drug
- BSA
Body surface area
- BSRBR-AS
British Society for Rheumatology Biologics Register in Ankylosing Spondylitis
- CRF
Case report form
- CRP
C-reactive protein
- DAPSA
Disease Activity in Psoriatic Arthritis
- DLQI
Dermatology Life Quality Index
- HADS
Hospital Anxiety and Depression Scale
- HAQ
Health Assessment Questionnaire
- HAQ-DI
Health Assessment Questionnaire Disability Index
- HLA-B27
Human leukocyte antigen B27
- PHQ-9
Patient Health Questionnaire 9
- PROM
Patient reported outcome measure
- PsA
Psoriatic arthritis
- tsDMARD
Targeted synthetic disease modifying anti-rheumatic drug
- WHO-5
WHO-5 Well-Being Index
Authors’ contributions
A.R., A.W., L.L., X.B., D.P., A.S., and A.C.R. were involved in study initiation, conception and design. D.P., X.B., S.Z., and C.S. substantially contributed to the acquisition of clinical data. A.R. and A.W. processed the data and performed the statistical analysis. A.R. and A.C.R wrote the article and all coauthors revised the manuscript critically for important intellectual content. A.R. and A.W. had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors agreed on the final content of the submitted manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. RABBIT-SpA is supported by a joint, unconditional grant from AbbVie, Amgen, Biogen, Celltrion, Hexal, Janssen-Cilag, Lilly, MSD, Novartis, Pfizer, UCB, and Viatris.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
Ethical approval was obtained from the Ethics Committee of the Charité – Universitätsmedizin Berlin. Participants have to consent to participate in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
10/11/2024
A Correction to this paper has been published: 10.1186/s13075-024-03411-9
References
- 1.Ritchlin CT, Colbert RA, Gladman DD. Psoriatic arthritis. N Engl J Med. 2017;376(10):957–70. [DOI] [PubMed] [Google Scholar]
- 2.Sieper J, Poddubnyy D. Axial spondyloarthritis. The Lancet. 2017;390(10089):73–84. [DOI] [PubMed] [Google Scholar]
- 3.Bair MJ, Robinson RL, Katon W, Kroenke K. Depression and pain comorbidity: a literature review. Arch Intern Med. 2003;163(20):2433–45. [DOI] [PubMed] [Google Scholar]
- 4.Park JYE, Howren AM, Zusman EZ, Esdaile JM, De Vera MA. The incidence of depression and anxiety in patients with ankylosing spondylitis: a systematic review and meta-analysis. BMC Rheumatology. 2020;4(1):12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Redeker I, Hoffmann F, Callhoff J, Haibel H, Sieper J, Zink A, et al. Determinants of psychological well-being in axial spondyloarthritis: an analysis based on linked claims and patient-reported survey data. Ann Rheum Dis. 2018;77(7):1017–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhao S, Thong D, Miller N, Duffield SJ, Hughes DM, Chadwick L, et al. The prevalence of depression in axial spondyloarthritis and its association with disease activity: a systematic review and meta-analysis. Arthritis Res Ther. 2018;20(1):140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kamalaraj N, El-Haddad C, Hay P, Pile K. Systematic review of depression and anxiety in psoriatic arthritis. Int J Rheum Dis. 2019;22(6):967–73. [DOI] [PubMed] [Google Scholar]
- 8.Zusman EZ, Howren AM, Park JYE, Dutz J, De Vera MA. Epidemiology of depression and anxiety in patients with psoriatic arthritis: A systematic review and meta-analysis. Semin Arthritis Rheum. 2020;50(6):1481–8. [DOI] [PubMed] [Google Scholar]
- 9.Michelsen B, Kristianslund EK, Sexton J, Hammer HB, Fagerli KM, Lie E, et al. Do depression and anxiety reduce the likelihood of remission in rheumatoid arthritis and psoriatic arthritis? Data from the prospective multicentre NOR-DMARD study. Ann Rheum Dis. 2017;76(11):1906–10. [DOI] [PubMed] [Google Scholar]
- 10.Macfarlane GJ, Pathan E, Jones GT, Dean LE. Predicting response to anti-TNFα therapy among patients with axial spondyloarthritis (axSpA): results from BSRBR-AS. Rheumatology. 2020;59(9):2481–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wong A, Ye JY, Cook RJ, Gladman DD, Chandran V. Depression and Anxiety Reduce the Probability of Achieving a State of Sustained Minimal Disease Activity in Patients With Psoriatic Arthritis. Arthritis Care Res. 2022;74(9):1430–4. [DOI] [PubMed] [Google Scholar]
- 12.Regierer AC, Weiß A, Baraliakos X, Zink A, Listing J, Strangfeld A. RABBIT-SpA: a new disease register for axial spondyloarthritis and psoriatic arthritis. Z Rheumatol. 2020;79(2):135–42. [DOI] [PubMed] [Google Scholar]
- 13.Lindner L, Weiß A, Reich A, Kindler S, Behrens F, Braun J, et al. Implementing an automated monitoring process in a digital, longitudinal observational cohort study. Arthritis Res Ther. 2021;23(1):1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Topp CW, Østergaard SD, Søndergaard S, Bech P. The WHO-5 Well-Being Index: A Systematic Review of the Literature. Psychother Psychosom. 2015;84(3):167–76. [DOI] [PubMed] [Google Scholar]
- 15.Acker J, Richter K, Piehl A, Herold J, Ficker JH, Niklewski G. Obstructive sleep apnea (OSA) and clinical depression-prevalence in a sleep center. Sleep Breath. 2017;21(2):311–8. [DOI] [PubMed] [Google Scholar]
- 16.Löwe B, Spitzer RL, Gräfe K, Kroenke K, Quenter A, Zipfel S, et al. Comparative validity of three screening questionnaires for DSM-IV depressive disorders and physicians’ diagnoses. J Affect Disord. 2004;78(2):131–40. [DOI] [PubMed] [Google Scholar]
- 17.Halliday JA, Hendrieckx C, Busija L, Browne JL, Nefs G, Pouwer F, et al. Validation of the WHO-5 as a first-step screening instrument for depression in adults with diabetes: Results from Diabetes MILES - Australia. Diabetes Res Clin Pract. 2017;132:27–35. [DOI] [PubMed] [Google Scholar]
- 18.Coates LC, Soriano ER, Corp N, Bertheussen H, Callis Duffin K, Campanholo CB, et al. Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA): updated treatment recommendations for psoriatic arthritis 2021. Nat Rev Rheumatol. 2022;18(8):465–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Braun J, van den Berg R, Baraliakos X, Boehm H, Burgos-Vargas R, Collantes-Estevez E, et al. 2010 update of the ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann Rheum Dis. 2011;70(6):896–904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smolen JS, Schöls M, Braun J, Dougados M, FitzGerald O, Gladman DD, et al. Treating axial spondyloarthritis and peripheral spondyloarthritis, especially psoriatic arthritis, to target: 2017 update of recommendations by an international task force. Ann Rheum Dis. 2018;77(1):3–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.van Buuren S. Multiple imputation of discrete and continuous data by fully conditional specification. Stat Methods Med Res. 2007;16(3):219–42. [DOI] [PubMed] [Google Scholar]
- 22.Krieger T, Zimmermann J, Huffziger S, Ubl B, Diener C, Kuehner C, et al. Measuring depression with a well-being index: further evidence for the validity of the WHO Well-Being Index (WHO-5) as a measure of the severity of depression. J Affect Disord. 2014;156:240–4. [DOI] [PubMed] [Google Scholar]
- 23.Dadfar M, Momeni Safarabad N, Asgharnejad Farid AA, Nemati Shirzy M, Ghazie Pour Abarghouie F. Reliability, validity, and factorial structure of the World Health Organization-5 Well-Being Index (WHO-5) in Iranian psychiatric outpatients. Trends Psychiatry Psychother. 2018;40(2):79–84. [DOI] [PubMed] [Google Scholar]
- 24.Garland AF, Deyessa N, Desta M, Alem A, Zerihun T, Hall KG, et al. Use of the WHO’s Perceived Well-Being Index (WHO-5) as an efficient and potentially valid screen for depression in a low income country. Fam Syst Health. 2018;36(2):148–58. [DOI] [PubMed] [Google Scholar]
- 25.Cichoń E, Kiejna A, Kokoszka A, Gondek T, Rajba B, Lloyd CE, et al. Validation of the Polish version of WHO-5 as a screening instrument for depression in adults with diabetes. Diabetes Res Clin Pract. 2020;159:107970. [DOI] [PubMed] [Google Scholar]
- 26.Omani-Samani R, Maroufizadeh S, Almasi-Hashiani A, Sepidarkish M, Amini P. The WHO-5 Well-Being Index: A Validation Study in People with Infertility. Iran J Public Health. 2019;48(11):2058–64. [PMC free article] [PubMed] [Google Scholar]
- 27.de Wit M, Pouwer F, Gemke RJ, Delemarre-van de Waal HA, Snoek F. Validation of the WHO-5 Well-Being Index in adolescents with type 1 diabetes. Diabetes Care. 2007;30(8):2003–6. [DOI] [PubMed] [Google Scholar]
- 28.Hirano F, van der Heijde D, van Gaalen FA, Landewé RBM, Gaujoux-Viala C, Ramiro S. Determinants of the patient global assessment of well-being in early axial spondyloarthritis: 5-year longitudinal data from the DESIR cohort. Rheumatology. 2020;60(1):316–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ramiro S, Nikiphorou E, Sepriano A, Ortolan A, Webers C, Baraliakos X, et al. ASAS-EULAR recommendations for the management of axial spondyloarthritis: 2022 update. Ann Rheum Dis. 2023;82(1):19–34. [DOI] [PubMed] [Google Scholar]
- 30.De Lorenzis E, Natalello G, Bruno D, Tanti G, Magurano MR, Lucchetti D, et al. Psoriatic arthritis and depressive symptoms: does systemic inflammation play a role? Clin Rheumatol. 2021;40(5):1893–902. [DOI] [PubMed] [Google Scholar]
- 31.Lai TL, Au CK, Chung HY, Lau CS. Depression in psoriatic arthritis: Related to socio-demographics, comorbid loads or disease activity? Int J Rheum Dis. 2022;25(4):474–80. [DOI] [PubMed] [Google Scholar]
- 32.McDonough E, Ayearst R, Eder L, Chandran V, Rosen CF, Thavaneswaran A, et al. Depression and Anxiety in Psoriatic Disease: Prevalence and Associated Factors. J Rheumatol. 2014;41(5):887–96. [DOI] [PubMed] [Google Scholar]
- 33.Pollard LC, Choy EH, Gonzalez J, Khoshaba B, Scott DL. Fatigue in rheumatoid arthritis reflects pain, not disease activity. Rheumatology. 2006;45(7):885–9. [DOI] [PubMed] [Google Scholar]
- 34.Günaydin R, Göksel Karatepe A, Çeşmeli N, Kaya T. Fatigue in patients with ankylosing spondylitis: relationships with disease-specific variables, depression, and sleep disturbance. Clin Rheumatol. 2009;28(9):1045–51. [DOI] [PubMed] [Google Scholar]
- 35.Jensen Hansen IM, Asmussen Andreasen R, van Bui Hansen MN, Emamifar A. The reliability of disease activity score in 28 joints–C-reactive protein might be overestimated in a subgroup of rheumatoid arthritis patients, when the score is solely based on subjective parameters: a cross-sectional, exploratory study. JCR: J Clin Rheumatol. 2017;23(2):102–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ferreira RJO, Welsing PMJ, Jacobs JWG, Gossec L, Ndosi M, Machado PM, et al. Revisiting the use of remission criteria for rheumatoid arthritis by excluding patient global assessment: an individual meta-analysis of 5792 patients. Ann Rheum Dis. 2021;80(3):293–303. [DOI] [PubMed] [Google Scholar]
- 37.Bedaiwi M, Sari I, Thavaneswaran A, Ayearst R, Haroon N, Inman RD. Fatigue in Ankylosing Spondylitis and Nonradiographic Axial Spondyloarthritis: Analysis from a Longitudinal Observation Cohort. J Rheumatol. 2015;42(12):2354–60. [DOI] [PubMed] [Google Scholar]
- 38.Husted JA, Tom BD, Schentag CT, Farewell VT, Gladman DD. Occurrence and correlates of fatigue in psoriatic arthritis. Ann Rheum Dis. 2009;68(10):1553–8. [DOI] [PubMed] [Google Scholar]
- 39.Zhao SS, Jones GT, Hughes DM, Moots RJ, Goodson NJ. Depression and anxiety symptoms at TNF inhibitor initiation are associated with impaired treatment response in axial spondyloarthritis. Rheumatology. 2021;60(12):5734–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Desthieux C, Molto A, Granger B, Saraux A, Fautrel B, Gossec L. Patient-physician discordance in global assessment in early spondyloarthritis and its change over time: the DESIR cohort. Ann Rheum Dis. 2016;75(9):1661–6. [DOI] [PubMed] [Google Scholar]
- 41.Zastrow A, Faude V, Seyboth F, Niehoff D, Herzog W, Löwe B. Risk factors of symptom underestimation by physicians. J Psychosom Res. 2008;64(5):543–51. [DOI] [PubMed] [Google Scholar]
- 42.Carr E, Mahil SK, Brailean A, Dasandi T, Pink AE, Barker JN, et al. Association of Patient Mental Health Status With the Level of Agreement Between Patient and Physician Ratings of Psoriasis Severity. JAMA Dermatol. 2021;157(4):413–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Adjusted OR (±95% CI) of parameters associated with the presence of symptoms suggestive of depression (WHO-5 score of ≤28) in axSpA patients: results from multivariable logistic regression. Table S2. Adjusted OR (±95% CI) of parameters associated with the presence of symptoms suggestive of depression (WHO-5 score of ≤28) in PsA patients: results from multivariable logistic regression.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.