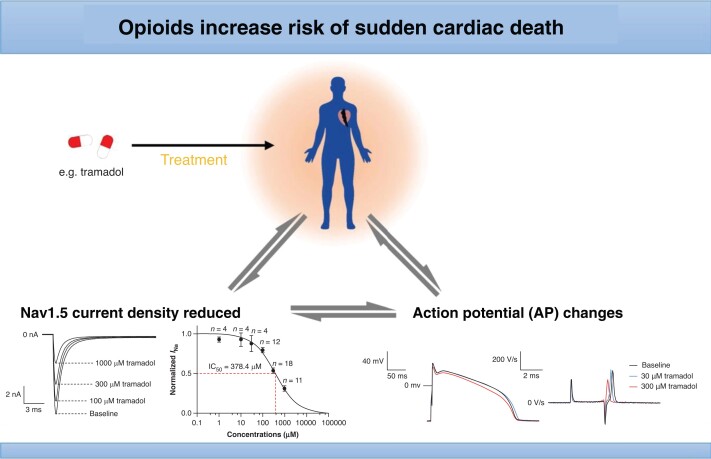
Abstract
Aims
Opioids are associated with increased risk of sudden cardiac death. This may be due to their effects on the cardiac sodium channel (Nav1.5) current. In the present study, we aim to establish whether tramadol, fentanyl, or codeine affects Nav1.5 current.
Methods and results
Using whole-cell patch-clamp methodology, we studied the effects of tramadol, fentanyl, and codeine on currents of human Nav1.5 channels stably expressed in HEK293 cells and on action potential (AP) properties of freshly isolated rabbit ventricular cardiomyocytes. In fully available Nav1.5 channels (holding potential −120 mV), tramadol exhibited inhibitory effects on Nav1.5 current in a concentration-dependent manner with an IC50 of 378.5 ± 33.2 µm. In addition, tramadol caused a hyperpolarizing shift of voltage-gated (in)activation and a delay in recovery from inactivation. These blocking effects occurred at lower concentrations in partially inactivated Nav1.5 channels: during partial fast inactivation (close-to-physiological holding potential −90 mV), IC50 of Nav1.5 block was 4.5 ± 1.1 μm, while it was 16 ± 4.8 μm during partial slow inactivation. The tramadol-induced changes on Nav1.5 properties were reflected by a reduction in AP upstroke velocity in a frequency-dependent manner. Fentanyl and codeine had no effect on Nav1.5 current, even when tested at lethal concentrations.
Conclusion
Tramadol reduces Nav1.5 currents, in particular, at close-to-physiological membrane potentials. Fentanyl and codeine have no effects on Nav1.5 current.
Keywords: Sudden cardiac death, Tramadol, Action potential, Nav1.5 current, Overdose, Risk association
Graphical Abstract
Graphical Abstract.
What’s new?
Opioids associated with an increased risk of sudden cardiac death due to life-threatening cardiac arrhythmias:
Tramadol reduced current of Nav1.5 channels in a fully available state in a dose-dependent manner and caused gating changes at lethal concentrations.
These effects were mirrored by reductions in the action potential upstroke velocity and reduction in action potential amplitude.
Fentanyl and codeine had no effects on Nav1.5 channels in a fully available state at lethal concentrations.
Translational Perspective.
Opioid use is increasing, along with death rates associated with it. This may be due to cardiovascular effects of these drugs. Use of various opioids is associated with an increased risk of sudden cardiac death (SCD) in the general population. This may be due to the ability of opioids to induce life-threatening cardiac arrhythmias by impacting on the cardiac ion channels that underlie cardiac electrophysiology. We sought to establish the cellular electrophysiological effects of the commonly used opioids tramadol, fentanyl, and codeine on currents of the human cardiac sodium channel Nav1.5 and repolarizing ion channels. We found that tramadol, but not fentanyl or codeine, reduced Nav1.5 currents and action potential upstroke velocity in a dose-dependent and rate-dependent manner at clinically relevant concentrations, but exhibited no significant effects on action potential duration. These findings are of potential clinical relevance, because they may help define individuals who are at elevated risk of tramadol-induced SCD such as individuals whose Nav1.5 current reserve is reduced because of inherited factors (genetic Nav1.5 variants) and/or acquired conditions (comorbidities such as ischemic heart disease). Identification of elevated risk may be the basis for personalized treatment of these individuals (lowering of tramadol dosages or avoidance of its use altogether) that aims at preventing drug-induced SCD.
Introduction
Opioids are increasingly prescribed for the treatment of severe and chronic pain.1 The incidence of opioid use disorders due to long-term exposure to opioids is increasing, along with death rates associated with unintentional opioid overdoses.2–4 This may be due to cardiovascular effects of these drugs.2,5 For instance, we demonstrated that use of opioids—in particular tramadol, oxycodone, codeine, fentanyl, morphine, and methadone (the six most widely prescribed opioids in The Netherlands)6—is associated with an increased risk of sudden cardiac death (SCD) in the general population.7,8 This may be due to the ability of these drugs to induce life-threatening cardiac arrhythmias5 by impacting on the cardiac ion channels whose concerted activity underlies the cardiac action potential (AP).9
Two potentially relevant ion channel targets for opioids are the cardiac sodium channel, Nav1.5 (which is important for AP initiation and propagation),10 and the hERG potassium channel (crucial for sarcolemma repolarization and AP termination).11 Increased SCD risk of opioids (in particular oxycodone, codeine, fentanyl, and morphine) is mostly ascribed to hERG block12 and is reflected by QT prolongation of the electrocardiogram (ECG).13,14 The possibility that opioids block Nav1.5 channels is less recognized and studied.7,15,16 Yet, Nav1.5 dysfunction may also increase SCD risk.17,18 Of note, drug-induced Nav1.5 current block and increased SCD risk were not only reported for drugs whose therapeutic effect is based on this effect (Vaughan-Williams class I antiarrhythmic drugs)16 but also for drugs used for noncardiac disease for which Nav1.5 block is an undesired off-target effect. This is increasingly recognized for a growing number of noncardiac drugs.7 At present, this group contains tricyclic antidepressants19,20 and antiepileptic drugs.21 We hypothesize that this group may also contain opioids and opioid agonists. Indeed, various opioids, including morphine, U-50, 488H, oxycodone, methadone, loperamide, and buprenorphine, inhibit Nav1.5 channels and/or cardiac AP upstroke velocities.22–27 Currently, the effects of tramadol, codeine, and fentanyl on Nav1.5 current are unknown. However, tramadol and fentanyl block neuronal (Nav1.2 and Nav1.7) voltage-gated sodium channels26,28 and because the Nav1.5 isoform is highly homologous with neuronal sodium channels,29 it is likely that these opioids also reduce Nav1.5 current. The aim of the present study was to establish whether tramadol, codeine, or fentanyl reduces Nav1.5 current and affects the morphology of APs. To this end, we carried out patch-clamp experiments on human embryonic kidney 293 (HEK293) cells stably expressing human Nav1.5 channels and determined the effects of the drugs on the APs of freshly isolated rabbit left ventricular cardiomyocytes.
Methods
HEK293 cell culture
We used a HEK293 cell line with stable human Nav1.5 channel expression.30 The HEK293 cells were cultured in DMEM with Glutamax (Gibco) supplemented with 10% FBS (Biowest), l-glutamine (Gibco), penicillin–streptomycin (Gibco), and Zeocin (of 200 µg/mL, Invitrogen) in a 5% CO2 incubator (Shel Lab) at 37°C. The cells were passaged every 3–4 days at 70% confluency in 25 mL flasks by using 0.25% trypsin (Gibco) treatments of around 2 min. On the day of the patch-clamp measurements, cells were trypsinized, stored at room temperature, and used within 3 h.
Rabbit ventricular cardiomyocyte preparation
Male New Zealand white rabbits (3.0–3.5 kg) were anesthetized by a combination of ketamine (intramuscular 100 mg) and xylazine (intramuscular 20 mg), heparinized (Heparin LEO 5000 IU), and killed by an injection of pentobarbital (240 mg). The hearts were excised and transported to the laboratory in cold (4°C) Tyrode’s solution containing (in mm) NaCl 128, KCl 4.7, CaCl2 1.5, MgCl2 0.6, NaHCO3 27, Na2HPO4 0.4, and glucose 11 (pH 7.4) by equilibration with 95% O2 and 5% CO2. Subsequently, the hearts were mounted on a Langendorff perfusion apparatus and left ventricular midmyocardial cardiomyocytes were isolated by enzymatic dissociation from the most apical part of the left midmyocardial ventricular free wall as described previously.31 Animal procedures were performed in accordance with governmental and institutional guidelines for animal use in research and were approved by the Animal Experimental Committee of Amsterdam UMC, The Netherlands.
Patch-clamp recording
We applied the whole-cell configuration of the patch-clamp technique using an Axopatch 200B amplifier (Molecular Devices, San Jose, CA, USA). Borosilicate glass patch pipettes (GC100F-10; Harvard Apparatus, UK) were pulled using a custom microelectrode puller and had a resistance of 2–3 MΩ after filling with the pipette solutions (for compositions, see below). All signals were low-pass filtered (5 kHz) and digitized at 33 kHz. Series resistance was compensated by ≥80%. Custom software (Scope, kindly provided by J.G. Zegers, and MacDAQ, kindly provided by A.C.G. van Ginneken) was used to record and analyze Nav1.5 currents and APs.
Sodium current measurements
Nav1.5 current was measured in single HEK293 cells using the ruptured patch-clamp technique at room temperature. The pipette solution contained (in mm) NaF 10, CsCl 10, CsF 110, EGTA 11, CaCl2 1.0, MgCl2 1.0, Na2ATP 2.0, and HEPES 10 (pH adjusted to 7.2 with CsOH). The bath solution contained (in mm) NaCl 20, CsCl 120, CaCl2 1.8, MgCl2 1.0, glucose 5.5, and HEPES 5.0 (pH adjusted to 7.4 with CsOH).30 Nav1.5 current was measured in response to depolarizing voltage steps from a holding potential of −120 mV (cycle length of 5 s). At −120 mV, all Nav1.5 channels are fully available for activation, and we named this the ‘fully available’ state of the channel. Nav1.5 current was defined as the difference between peak and steady-state current. The dose–response curves were fitted by the Hill equation: Y = 1/[(1 + (IC50/X)n)], where Y is the current normalized to baseline condition, IC50 is the dose required for 50% current block, and n is the Hill coefficient.
Nav1.5 (in)activation current was measured with a double-pulse protocol (see inserted protocols in figures). During the first depolarizing pulses (P1), Nav1.5 current activates and the currents analyzed here are used to determine current–voltage (I–V) relationships and the voltage dependency of activation. The second pulse (P2) is used to determine the voltage dependency of inactivation. The I–V relationships were corrected for driving force, normalized to maximum peak current, and fitted to a Boltzmann distribution curve. Voltage dependence of activation and inactivation curves was fitted with Boltzmann function: I/Imax = A/{1.0 + exp[(V1/2 − V)/k]}, where V1/2 is the midpoint of channel (in)activation and k is the slope factor of the (in)activation curve. The rate of recovery from inactivation was measured with a double-pulse protocol with two depolarizing steps (P1 and P2) from −120 to −20 mV and a variable interpulse interval (see inserted protocols in figures). Currents measured during P2 were normalized to currents measured during P1. Recovery from inactivation curves were fitted by a double exponential function: , where τf and τs are the fast and slow time constants of recovery from inactivation, and Af and As are the fractions of fast and slow recovery from inactivation. Use-dependent block was determined by application of 30 activating pulses with a duration of 200 ms from −120 to −20 mV at a frequency of 4 Hz (see inserted protocols in figures). Nav1.5 currents were normalized to the current of the first pulse. In addition to testing the drug effects on fully available Nav1.5 channels, we also tested drug effects on Nav1.5 channels that were partially inactivated. Effects on Nav1.5 current in partially fast-inactivated state were tested by applying a 1000 ms prepulse at −90 mV, followed by a 50 ms test pulse at −40 mV (see inserted protocols in figures). Effects on Nav1.5 current in partially slow-inactivated state were tested by applying a 1000 ms prepulse at −40 mV, followed by a 20 ms pulse at −120 mV allowing recovery from fast inactivation, and finally a test pulse to −40 mV (see inserted protocols in figures).
Action potential measurement
Action potentials were measured in single rabbit ventricular cardiomyocytes at 36°C using the amphotericin-perforated patch-clamp technique. Cells were superfused with modified Tyrode’s solution containing (in mm) NaCl 140, KCl 5.4, CaCl2 1.8, MgCl2 1.0, glucose 5.5, and HEPES 5.0 (pH adjusted to 7.4 with NaOH). Pipette solution contained (in mm) K-gluconate 125, KCl 20, NaCl 5.0, Amphotericin-B 0.44, and HEPES 10 (pH adjusted to 7.2 with KOH). Action potentials were evoked at 1–3 Hz using square 3-ms current pulses through the patch pipette, and potentials were corrected for the calculated liquid junction potential.32 We analyzed resting membrane potential (RMP), AP amplitude (APA), maximal AP upstroke velocity (dV/dtmax), and AP duration at 20, 50, or 90% repolarization (APD20, APD50, and APD90, respectively). Action potential parameters from 10 consecutive APs were averaged.
Preparation of opioids
Tramadol, fentanyl, and codeine (purity ≥98%), purchased from Sigma-Aldrich, were freshly dissolved in the bath solution to the desired concentration just before use. Nav1.5 current was measured at baseline conditions (no drug) and after 5–8 min wash-in of these drugs at various concentrations that are close to the lethal concentrations of these drugs.33–35 Tramadol was measured at 1, 10, 30, 100, 300, or 1000 µm. Fentanyl was measured at 1 µm. Codeine was measured at 100 µm.
Statistical analysis
Values are presented as mean ± SEM. Curve fitting and statistics was performed using Prism8 GraphPad (GraphPad Software, LLC, USA). One-way ANOVA or two-way ANOVA was used to assess the statistical significance of the differences among multiple groups. One-way repeated measures (RMs) ANOVA followed by pairwise comparison using the Holm–Sidak multiple comparisons test or one-way RM ANOVA on ranks (Friedman test) followed by Dunn’s multiple comparison test for post hoc analyses was used when data were not normally distributed. Differences between two groups were tested using paired Student’s t-tests or two-way RMs ANOVA followed by pairwise comparison using the Holm–Sidak multiple comparisons test. Details on normalization are given in Methods or in the figure legends. P < 0.05 was considered to be statistically significant.
Results
Effects on Nav1.5 current amplitude in HEK293 cells
We first studied whether high (lethal) concentrations of tramadol, fentanyl, or codeine impact Nav1.5 current amplitude in HEK293 cells by applying single 100 ms depolarizing pulses from −120 to −40 mV (Figure 1A–C). We found that 1 µm fentanyl and 100 µm codeine had no effect on fully available Nav1.5 current amplitude (Figure1B and C). In contrast, 1000 µm tramadol reduced Nav1.5 current amplitude by 69 ± 3.7% (n = 11) (Figure 1A). We therefore conducted additional studies with tramadol to assess its effects on Nav1.5 biophysical properties in more detail.
Figure 1.
Effects of opioids on current densities of Nav1.5 channels expressed in HEK293 cells. (A–C) Typical Nav1.5 current elicited by application of 100 ms depolarizing pulses from −120 to −40 mV (left panels) and average current amplitudes (right panels) in the absence or presence of 1000 µm tramadol (A), 1 µm fentanyl (B), and 100 µm codeine (C). Data are expressed as the mean ± SEM. *P < 0.05, tramadol vs. baseline (paired Student’s t-tests).
Effects of tramadol on current amplitude and gating properties of Nav1.5 channels in HEK293 cells
Using a similar depolarizing step as used in Figure 1, we studied the concentration dependency of tramadol-induced block (Figure 2A) of fully available Nav1.5 current and found that the IC50 of current reduction was 378.5 ± 33.2 µm. Subsequently, we tested if 300 μm tramadol, a concentration close to the IC50, caused changes in gating properties of Nav1.5. Figure 2B shows typical Nav1.5 currents under baseline condition and in the presence of 300 μm tramadol measured over a wide range of depolarizing voltages (for protocol, see Figure 2B, inset). Figure 2C shows the average I–V relationships and indicates that tramadol significantly decreased Nav1.5 current in the voltage range from −60 to 30 mV, e.g. by 56.4 ± 4.5% at −40 mV (n = 6, P < 0.05). Tramadol induced a hyperpolarizing shift in voltage dependency of both activation and inactivation (Figure 2D). The average V1/2 of channel activation was at −54.6 ± 1.2 mV in the absence and −61.3 ± 1.6 mV in the presence of tramadol (n = 6, P < 0.05). The average V1/2 of channel inactivation were −96.0 ± 2.2 mV (baseline) and −105.2 ± 2.7 mV (tramadol), respectively (n = 6, P < 0.05, Table 1). The slope of the activation curve, k, was significantly changed after the application of tramadol from 5.9 ± 0.4 to 5.2 ± 0.3 mV (n = 6, P < 0.05, Table 1). To study the rate-dependent effects of tramadol, we applied double-pulse protocols as shown in Figure2E and F. We found that tramadol delayed recovery from steady-state inactivation (Figure 2E and Table 1) with τf and τs significantly changed from 10.9 ± 2.3 to 34.5 ± 8.0 ms, and from 293.6 ± 70.4 to 800.1 ± 139.3 ms, respectively (n = 5, P < 0.05, Table 1). Consistent with this observation, the reduction in Nav1.5 current density at rising pulse numbers increased more in the presence of tramadol, e.g. by 81.7 ± 3.6% at the 30th pulse (n = 9, P < 0.05) (Figure 2F). Time constants of current inactivation at −20 mV were fitted with biexponential fits and were not significantly affected by 300 µm tramadol (data not shown).
Figure 2.
Effects of tramadol on amplitude and gating kinetics of Nav1.5 currents expressed in HEK293 cells. (A) Typical Nav1.5 currents under baseline conditions and in the presence of 100–1000 μm tramadol; examples (left panel) and concentration dependency (right panel) of Nav1.5 current inhibition. The solid line is a Hill fit to the average data. Values are normalized to the values measured under baseline conditions. Numbers near symbols indicated the number of cells (n) measured at the given concentrations. Inset: voltage clamp protocol used. (B) Typical Nav1.5 currents under baseline conditions and in the presence of 300 μm tramadol. Inset: voltage clamp protocol used to measure current–voltage (I–V) relationships and the voltage dependency of (in)activation. Cycle length was 5 s. (C) The Nav1.5 I–V relationships before and after the application of 300 μm tramadol. Nav1.5 current was normalized to the maximal peak amplitude under baseline conditions, but peak current was set to −1 to retain the well-known inward direction of the sodium current. (D) Effects of tramadol on the voltage dependency of Nav1.5 current (in)activation. Solid lines are Boltzmann fits to the average data. (E) Recovery from inactivation of Nav1.5 current in the absence or presence of 300 µm tramadol measured with a double-pulse protocol with variable interpulse intervals (inset). (F) Use dependency under baseline conditions and in the presence of tramadol measured during a train of 30 depolarizing pulses with an interpulse interval of 50 ms (inset). *P < 0.05, tramadol vs. baseline (two-way RM ANOVA).
Table 1.
Cardiac sodium current properties in the absence or presence of 300 µm tramadol
| Baseline | Tramadol | ||
|---|---|---|---|
| Activation | V 1/2 (mV) | −54.6 ± 1.2 (n = 6) | −61.3 ± 1.6 (n = 6)* |
| k (mV) | 5.9 ± 0.4 (n = 6) | 5.2 ± 0.3 (n = 6)* | |
| Inactivation | V 1/2 (mV) | −96.0 ± 2.2 (n = 6) | −105.2 ± 2.7 (n = 6)* |
| k (mV) | 6.6 ± 0.4 (n = 6) | 7.5 ± 0.6 (n = 6) | |
| Recovery from inactivation | τ f (ms) | 10.91 ± 2.28 (n = 5) | 34.52 ± 7.93 (n = 5)* |
| τ s (ms) | 293.6 ± 70.35 (n = 5) | 800.1 ± 139.3 (n = 5)* | |
| A s/(As + Af) (ms) | 0.29 ± 0.03 (n = 5) | 0.47 ± 0.02 (n = 5)* |
Data are expressed as the mean ± SEM, and n indicates the number of cells.
P < 0.05, tramadol vs. baseline (paired Student’s t-tests).
Effects of tramadol on density of partially inactivated Nav1.5 current in HEK293 cells
A previous study in neuronal sodium channels showed that tramadol induced more pronounced effects when these channels were in a partially inactivated state than when they were in a fully available state.28 To study whether this also occurs in Nav1.5 channels, we studied the concentration dependency of tramadol on Nav1.5 current of channels in a partially fast-inactivated or partially slow-inactivated state in HEK293 cells (Figure 3). The used voltage clamp protocols are shown in the left panels; the middle panels depict typical current traces. Figure 3A shows that tramadol blocked partially fast-inactivated Nav1.5 current in a concentration-dependent manner, with an IC50 of 4.5 ± 1.1 μm. Figure 3B shows that tramadol blocked partially slow-inactivated Nav1.5 current in a concentration-dependent manner, with an IC50 of 16 ± 4.8 μm.
Figure 3.
Effects of tramadol on amplitude of partially inactivated Nav1.5 current in HEK293 cells. (A and B) Block of fast-inactivated (A) and slow-inactivated (B) Nav1.5 current by tramadol in a concentration-dependent manner. Inset: voltage clamp protocol used to measure (left panel) and typical Nav1.5 currents under baseline conditions and in the presence of 1–100 μm tramadol (middle panel). We achieved partial fast inactivation (A) by applying a −90 mV prepulse and studied the effect of partial fast inactivation by applying the test pulse immediately following this prepulse. We achieved partial slow inactivation (B) by applying a −40 mV prepulse, during which Nav1.5 channels enter into both a fast-inactivated and a slow-inactivated state. This was followed by a brief (20 ms long) repolarizing pulse to −120 mV, which allows almost full recovery from fast inactivation, but not slow inactivation. Thus, the ensuing test pulse activates only Nav1.5 channels that have recovered from fast inactivation, but not Nav1.5 channels that are still in a slow inactivation state. Cycle length was 5 s. Solid lines are Hill fits to the average data. Values are normalized to the values measured under baseline conditions. Numbers near symbols indicated the number of cells (n) measured at the given concentrations.
Effects of tramadol on action potentials in rabbit ventricular cardiomyocytes
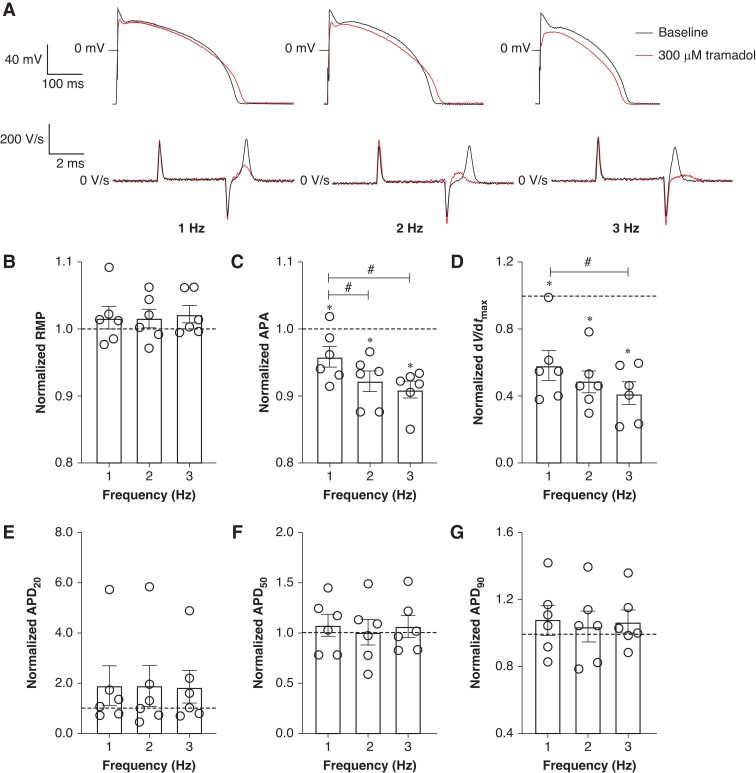
To verify the functional implication of the tramadol-induced effects on Nav1.5 current, we tested the effects of tramadol (10, 30, 100, and 300 μm) on APs of isolated rabbit left ventricular cardiomyocytes. Figure 4A shows typical APs elicited at 1 Hz under baseline conditions and in the presence of 30 and 300 µm tramadol; average AP parameters after application of 10–300 µm tramadol are summarized in Figure 4B–G. Tramadol reduced dV/dtmax in a concentration-dependent manner (Figure 4D), while other AP parameters (RMP, APA, APD20, APD50, and APD90) were not significantly changed (Figure4B, C, and E–G). Subsequently, we studied the effect of 300 µm tramadol on APs at faster stimulation frequencies. Figure 5A shows typical APs elicited at 1, 2, and 3 Hz under baseline conditions and in the presence of 300 µm tramadol; average AP parameters at 1–3 Hz are summarized in Figure 5B–G. Tramadol caused statistically significant decreases in APA and dV/dtmax in a frequency-dependent manner (Figure5C and D). For example, APA decreased by 4.1 ± 1.5% (from 119.2 ± 1.2 to 114.2 ± 1.0 mV, n = 6, P < 0.05) at 1 Hz and by 9.0 ± 1.3% (from 119 ± 2.3 to 108.3 ± 2.3 mV, n = 6, P < 0.05) at 3 Hz (Figure 5C). Similarly, dV/dtmax was significantly decreased at all rates, e.g. by 41.7 ± 9.0% (from 327.7 ± 44.3 to 192.2 ± 37.8 V/s, n = 6, P < 0.05) at 1 Hz and by 58.2 ± 6.7% (from 320.2 ± 51.7 to 136 ± 31.4 V/s, n = 6, P < 0.05) at 3 Hz (Figure 5D). Thus, the reductions in APA and dV/dtmax were larger at higher stimulation frequencies (n = 6, P < 0.05), consistent with the reduced rate of recovery from inactivation of Nav1.5 (Figure2E and F). Resting membrane potential, APD20, APD50, and APD90 at 1–3 Hz were not significantly affected by tramadol (Figure5B and E–G).
Figure 4.
Concentration dependency of effects of tramadol on action potentials (APs) of single rabbit ventricular cardiomyocytes. (A) Superimposed representative APs at 1 Hz under baseline conditions and in the presence of 30 and 300 µm tramadol. Inset: time derivatives of the AP upstrokes. (B–G) Average AP characteristics at 1 Hz of resting membrane potential (B), AP amplitude (C), maximal AP upstroke velocity (D), AP duration at 20% of repolarization (E), AP duration at 50% of repolarization (F), and AP duration at 90% of repolarization (G) in the absence or presence of 10, 30, 100, or 300 µm tramadol. Data are mean ± SEM. *P < 0.05, tramadol vs. baseline (paired t-test); #P < 0.05, drug effects observed with different concentrations (one-way RM ANOVA). APA, AP amplitude; APD20, AP duration at 20% of repolarization; APD50, AP duration at 50% of repolarization; APD90, AP duration at 90% of repolarization; dV/dtmax, maximal AP upstroke velocity; RMP, resting membrane potential.
Figure 5.
Frequency dependency of effect of 300 µm tramadol on APs of rabbit ventricular cardiomyocytes. (A) Superimposed representative APs at 1, 2, and 3 Hz under baseline conditions and in the presence of 300 µm tramadol. Inset: time derivatives of the AP upstrokes. (B–G) Average AP characteristics at 1, 2, and 3 Hz of RMP (B), APA (C), dV/dtmax (D), APD20 (E), APD50 (F), and APD90 (G). Action potential parameters are normalized to the AP parameters measured under baseline conditions in the same cell. Data are mean ± SEM. *P < 0.05, tramadol vs. baseline (paired t-test); #P < 0.05, drug effects observed with different frequencies (one-way RM ANOVA).
Discussion
Our main findings were as follows: (i) tramadol reduced current of Nav1.5 channels in a fully available state in a dose-dependent manner, while fentanyl and codeine had no effect even when tested at lethal concentrations; (ii) tramadol shifted the voltage dependency of (in)activation and slowed the recovery from inactivation of Nav1.5; (iii) tramadol caused block of Nav1.5 channels at lower concentrations when these channels were in a partially inactivated state than when they were in a fully available state; (iv) tramadol reduced dV/dtmax and APA in rabbit cardiomyocytes with a larger amount of reduction at fast pacing rates; and (v) tramadol did not affect AP repolarization.
Our results on Nav1.5 are consistent with previous findings on the effects of tramadol on neuronal sodium channels. Of note, the concentration of tramadol required for 50% inhibition of Nav1.5 currents when measured at a holding potential of −120 mV (IC50 378.5 ± 33.2 µm) was only mildly greater than the concentrations needed to obtain 50% block of TTX-sensitive neuronal sodium currents in rodent neuroblastoma ND7/23 cells (194 ± 9 µm)24 and of rat Nav1.2 in HEK293 cells (103 ± 8 µm).28 Very recently, Bok et al.36 also tested the effect of tramadol on Nav1.7 and Nav1.5 currents in HEK293 cells. They found that tramadol reduced Nav1.5 current, consistent with our findings, with an IC50 of 0.85 ± 0.04 mm at a −120 mV holding potential. We also used a holding potential of −120 mV, but we found an IC50 of 0.38 ± 0.03 mm, which is thus 2.2 times smaller than in the study of Bok et al.36 In both studies, the IC50 decreases upon more depolarized holding potentials indicating that the blocking effect of tramadol is increased in the presence of more inactivated Nav1.5 channels. This principle is also observed for Nav1.5 and Nav1.2 currents in HEK293 cells.28,36 In our study, the V1/2 of Nav1.5 inactivation is slightly more negative under control conditions compared to the study of Bok et al. As a result, we might have more inactivated Nav1.5 channels at −120 mV resulting in a more pronounced inhibition and a shift in IC50 to lower concentrations compared to the study of Bok et al.36 Moreover, the tramadol-induced negative shift of voltage-gated inactivation and the use-dependent block of Nav1.5 that we found agree with the findings of Bok et al.,36 and this was also observed for Nav1.2 current28 and Nav1.7.36 We found also a negative shift in the V1/2 of activation, and this was also found by Bok et al. for Nav1.7, but not for Nav1.5.36 While our findings are thus largely consistent with the study of Bok et al., our study offers significant advances beyond that study by also including other opioids and AP studies in cardiomyocytes. Action potential studies are of particular relevance because previous studies have suggested that tramadol may impact not only on cardiac depolarization and Nav1.5 current but also on cardiac repolarization and AP duration. Tramadol reduced dV/dtmax and APA in rabbit cardiomyocytes in a concentration-dependent and frequency-dependent manner, consistent with our measurement of Nav1.5 current in HEK293 cells. It has been reported that tramadol poisoning could induce tachycardia,37 and the use dependency of tramadol’s effects that we observed would exaggerate tramadol’s inhibitory action on Nav1.5.
This inhibitory action is in line with ECG changes such as QRS widening and Brugada syndrome (BrS) pattern, of patients with tramadol poisoning37–39 which are importantly due to reduced Nav1.5 channel function.40,41 We report that the IC50 of tramadol to block fully available Nav1.5 channels is 378.5 ± 33.2 µm. This is far above the therapeutic range (0.38–1.14 µm).42 When we analyzed tramadol’s effects on fully available Nav1.5 currents, we used a strongly hyperpolarized holding potential of −120 mV. While this method is routinely used for electrophysiological studies, it may hamper extrapolation to physiological conditions, where the RMP of cardiac myocytes is around −80 mV. In fact, at −80 mV, many Nav1.5 channels are in an inactivated state. Therefore, the effects of drugs, including tramadol, on partially inactivated Nav1.5 channels are clinically more relevant. When we analyzed tramadol’s effects on Nav1.5 channels that were partially in a fast-inactivated state (by utilizing a RMP of −90 mV), we found that the inhibition of Nav1.5 current was strongly enhanced. In fact, the IC50 on Nav1.5 channels that were in a partially fast-inactivated state was now 4.5 ± 1.1 μm, which is close to therapeutic concentrations (up to 1.14 µm). This effect was also documented by Bok et al.36 In the meantime, we also notice that this change is not so obvious in the AP measurements in which the cardiomyocytes have a RMP of around −80 mV. Our result showed that the tramadol-induced dV/dtmax reduction is concentration dependent, and tramadol-induced reduction of dV/dtmax was only present at 300 µm in comparison to the baseline condition. However, compared to 10 µm, tramadol could also reduce the dV/dtmax at 100 µm (Figure 4D) in rabbit ventricular cardiomyocytes. This may be due to the different experiment conditions, such as measurement temperature, the sodium and fluoride concentrations of the used solutions, or the different cell models.20,43–45 Even so, at clinically relevant high concentrations, tramadol-induced overdose toxicity still may be achieved in clinical cases. For instance, the tramadol concentrations in peripheral blood were reported from 1.6 mg/L (6.1 μm) to 15.1 mg/L (57.3 μm) in an overview report, which is close to our measured IC50 of Nav1.5 channels in a partially fast-inactivated or slow-inactivated state.46 In another report, tramadol fatal concentrations ranged from 0.03 to 134 mg/L (0.1–508.7 µm).34
While the effects of tramadol on Nav1.5 channels occurred at concentrations that are somewhat higher than therapeutic concentrations, a relevant consideration is that certain patient groups may have increased sensitivity to the Nav1.5 blocking effects of tramadol.37 This may stem from acquired comorbidities and/or from inherited susceptibility. A highly prevalent acquired condition is myocardial ischemia and infarction.47 The concept that these comorbidities may permit or facilitate the occurrence of fatal cardiac arrhythmia and SCD upon the use of Nav1.5 current blocking drugs was discovered in the Cardiac Arrhythmia Suppression Trial. In this trial, patients randomized to the class 1c antiarrhythmic drugs flecainide and encainide—potent Nav1.5 current blockers—suffered excess SCD rates compared to placebo-treated patients.16 Excess SCD rates occurred in patients with myocardial ischemia.48 Inherited susceptibility may stem from carrying variants in genes that encode subunits of the Nav1.5 channel, in particular variants in SCN5A, the gene that encodes its α-subunit.49,50 Loss-of-function mutations in this gene underlie the BrS51 and cardiac conduction disease,52 inherited cardiac arrhythmia syndromes associated with elevated SCD risk.
Tramadol was reported to block hERG current in NG108-15 neuronal cells (IC50 25 µm),53 and in neuronal APs modified by 4-aminopyridine (4-AP) in rat sciatic nerves (measured at 4 mm),54 and to prolong the duration of the QTc interval of the ECG,55 although the latter is not a consistent finding.56 We found that tramadol had no significant effect on APD20, APD50, or APD90. This suggests that tramadol may not reduce hERG current in cardiomyocytes or that our used concentrations of tramadol may have been insufficient to influence AP repolarization. An alternative explanation is that a reduction in outward hERG current is counterbalanced by reduced inward INa (this study and Bok et al.36) and l-type Ca2+ current,57 culminating in no net change in AP duration. We also found no significant effect of tramadol on RMP, suggesting that IK1 is largely unaltered. Because RMP was unchanged, we had no evidence that reduction in Nav1.5 channel availability contributed to the observed tramadol-induced reduction in dV/dtmax.58
We found no evidence that fentanyl or codeine blocked fully available Nav1.5 current, even when tested at fatal concentrations (1 and 100 µm, respectively), although fentanyl was shown to block neuronal sodium current with IC50 of 141 ± 6 µm in rat Nav1.2 current and 153.2 µm in rat cultured thalamic neurons.28,59 Our findings are consistent with the study of Tschirhart et al.60 who also found no effects of fentanyl (10 µm) on the sodium current of neonatal rat ventricular myocytes. Taken together, we found evidence that tramadol blocks Nav1.5 current at clinically relevant concentrations. It is possible that this effect contributes to the increased SCD incidence observed among users of these drugs. Conversely, we found no evidence that fentanyl or codeine blocks Nav1.5 current at clinically relevant concentrations.
Conclusion
In conclusion, we found that tramadol reduces Nav1.5 current by reducing its current amplitude and changing its gating properties; these effects are reflected in changes in AP properties. Fentanyl and codeine have no effects on fully available Nav1.5 current, even when tested at fatal concentrations.
Authors’ contributions
H.L.T. conceived and designed the study. L.J. and A.O.V. structured and designed the patch-clamp studies. L.J. carried out the patch-clamp experiments and statistical analysis of the patch-clamp data and drafted the first version of the manuscript. M.W.V. contributed to sample preparation and edited the manuscript. All authors contributed to manuscript revision and approved the final version.
Acknowledgements
The authors thank Shirley van Amersfoorth and Cees Schumacher for their excellent technical assistance.
Contributor Information
Lixia Jia, Department of Clinical and Experimental Cardiology, Heart Center, Amsterdam UMC, University of Amsterdam, Meibergdreef 9, Amsterdam 1105 AZ, The Netherlands.
Marieke W Veldkamp, Department of Clinical and Experimental Cardiology, Heart Center, Amsterdam UMC, University of Amsterdam, Meibergdreef 9, Amsterdam 1105 AZ, The Netherlands.
Arie O Verkerk, Department of Clinical and Experimental Cardiology, Heart Center, Amsterdam UMC, University of Amsterdam, Meibergdreef 9, Amsterdam 1105 AZ, The Netherlands; Department of Medical Biology, Amsterdam Cardiovascular Sciences, Amsterdam UMC, University of Amsterdam, Meibergdreef 9, Amsterdam 1105 AZ, The Netherlands.
Hanno L Tan, Department of Clinical and Experimental Cardiology, Heart Center, Amsterdam UMC, University of Amsterdam, Meibergdreef 9, Amsterdam 1105 AZ, The Netherlands; Netherlands Heart Institute, Moreelsepark 1, Utrecht 3511 EP, The Netherlands.
Funding
This work was supported by the European Union’s Horizon 2020 research and innovation program (grant number 733381); the European Cooperation in Science and Technology (grant number CA19137); the China Scholarship Council; and Netherlands Cardio Vascular Research Initiative (grant numbers CVON-2017-15 and CVON-2018-30).
Conflict of interest: None declared.
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
- 1. Singh JA, Cleveland JD. National US time-trends in opioid use disorder hospitalizations and associated healthcare utilization and mortality. PLoS One 2020;15:e0229174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Singleton JH, Abner EL, Akpunonu PD, Kucharska-Newton AM. Association of nonacute opioid use and cardiovascular diseases: a scoping review of the literature. J Am Heart Assoc 2021;10:e021260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Okie S. A flood of opioids, a rising tide of deaths. N Engl J Med 2010;363:1981–5. [DOI] [PubMed] [Google Scholar]
- 4. Centers for Disease Control and Prevention (CDC) . Vital signs: overdoses of prescription opioid pain relievers—United States, 1999–2008. MMWR Morb Mortal Wkly 2011;60:1487–92. [PubMed] [Google Scholar]
- 5. Pugsley MK. The diverse molecular mechanisms responsible for the actions of opioids on the cardiovascular system. Pharmacol Ther 2002;93:51–75. [DOI] [PubMed] [Google Scholar]
- 6. GIPdatabank.nl ZNG. https://www.gipdatabank.nl/databank? infotype=g&label=00-totaal&tabel=B_01-basis&geg=ddd&item=N03AF (26 July 2022, date last accessed).
- 7. Eroglu TE, Blom MT, Souverein PC, de Boer A, Tan HL. Non-cardiac depolarization-blocking drugs are associated with increased risk of out-of-hospital cardiac arrest in the community. Pharmacoepidemiology 2022;1:64–75. [Google Scholar]
- 8. Eroglu TE, Barcella CA, Blom MT, Souverein PC, Mohr GH, Torp-Pedersen Cet al. Opioid use is associated with increased out-of-hospital cardiac arrest risk among 40 000-cases across two countries. Br J Clin Pharmacol 2022;88:2256–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. András V, Tomek J, Nagy N, Virág L, Passini E, Rodriguez Bet al. Cardiac transmembrane ion channels and action potentials: cellular physiology and arrhythmogenic behavior. Physiol Rev 2021;101:1083–176. [DOI] [PubMed] [Google Scholar]
- 10. Catterall WA, Goldin AL, Waxman SG. International Union of Pharmacology. XLVII. Nomenclature and structure-function relationships of voltage-gated sodium channels. Pharmacol Rev 2005;57:397–409. [DOI] [PubMed] [Google Scholar]
- 11. Antzelevitch C, Sun Z-Q, Zhang Z-Q, Yan G-X. Cellular and ionic mechanisms underlying erythromycin-induced long QT intervals and torsade de pointes. J Am Coll Cardiol 1996;28:1836–48. [DOI] [PubMed] [Google Scholar]
- 12. Katchman AN, McGroary KA, Kilborn MJ, Kornick CA, Manfredi PL, Woosley RLet al. Influence of opioid agonists on cardiac humanether-a-go-go-related gene K+ currents. J Pharmacol Exp Ther 2002;303:688–94. [DOI] [PubMed] [Google Scholar]
- 13. January CT, Gong Q, Zhou Z. Long QT syndrome: cellular basis and arrhythmia mechanism in LQT2. J Cardiovasc Electrophysiol 2000;11:1413–8. [DOI] [PubMed] [Google Scholar]
- 14. Behzadi M, Joukar S, Beik A. Opioids and cardiac arrhythmia: a literature review. Med Princ Pract 2018;27:401–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Amin AS, Asghari-Roodsari A, Tan HL. Cardiac sodium channelopathies. Pflugers Arch 2010;460:223–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cardiac Arrhythmia Suppression Trial (CAST) Investigators . Preliminary report: effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction. N Engl J Med 1989;321:406–12. [DOI] [PubMed] [Google Scholar]
- 17. Giudicessi JR, Ye D, Stutzman MJ, Zhou W, Tester DJ, Ackerman MJ. Prevalence and electrophysiological phenotype of rare SCN5A genetic variants identified in unexplained sudden cardiac arrest survivors. Europace 2020;22:622–31. [DOI] [PubMed] [Google Scholar]
- 18. Calloe K, Geryk M, Freude K, Treat JA, Vold VA, Frederiksen HRSet al. The G213D variant in Nav1. 5 alters sodium current and causes an arrhythmogenic phenotype resulting in a multifocal ectopic Purkinje-related premature contraction phenotype in human-induced pluripotent stem cell-derived cardiomyocytes. Europace 2022;24:2015–27. [DOI] [PubMed] [Google Scholar]
- 19. Eroglu TE, Barcella CA, Gerds TA, Kessing LV, Zylyftari N, Mohr GHet al. Risk of out-of-hospital cardiac arrest in antidepressant drug users. Br J Clin Pharmacol 2022;88:3162–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Plijter IS, Verkerk AO, Wilders R. The antidepressant paroxetine reduces the cardiac sodium current. Int J Mol Sci 2023;24:1904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Jia L, Eroglu TE, Wilders R, Verkerk AO, Tan HL. Carbamazepine increases the risk of sudden cardiac arrest by a reduction of the cardiac sodium current. Front Cell Dev Biol 2022;10:891996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Hung C, Tsai C, Su M. Opioid receptor independent effects of morphine on membrane currents in single cardiac myocytes. Br J Anaesth 1998;81:925–31. [DOI] [PubMed] [Google Scholar]
- 23. Meents JE, Juhasz K, Stölzle-Feix S, Peuckmann-Post V, Rolke R, Lampert A. The opioid oxycodone use-dependently inhibits the cardiac sodium channel NaV1. 5. Br J Pharmacol 2018;175:3007–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Schulze V, Stoetzer C, O'Reilly A, Eberhardt E, Foadi N, Ahrens Jet al. The opioid methadone induces a local anaesthetic-like inhibition of the cardiac Na+ channel, Nav1.5. Br J Pharmacol 2014;171:427–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kang J, Compton DR, Vaz RJ, Rampe D. Proarrhythmic mechanisms of the common anti-diarrheal medication loperamide: revelations from the opioid abuse epidemic. J Pharmacol Exp Ther 2016;389:1133–7. [DOI] [PubMed] [Google Scholar]
- 26. Leffler A, Frank G, Kistner K, Niedermirtl F, Koppert W, Reeh PWet al. Local anesthetic-like inhibition of voltage-gated Na+ channels by the partial μ-opioid receptor agonist buprenorphine. Anesthesiology 2012;116:1335–46. [DOI] [PubMed] [Google Scholar]
- 27. Alarcón S, Hernández J, Laorden M. Cardiac electrophysiological effects of U-50,488 H on guinea-pig papillary muscle. Neuropeptides 1993;24:313–6. [DOI] [PubMed] [Google Scholar]
- 28. Haeseler G, Foadi N, Ahrens J, Dengler R, Hecker H, Leuwer M. Tramadol, fentanyl and sufentanil but not morphine block voltage-operated sodium channels. Pain 2006;126:234–44. [DOI] [PubMed] [Google Scholar]
- 29. Fozzard HA, Hanck DA. Structure and function of voltage-dependent sodium channels: comparison of brain II and cardiac isoforms. Physiol Rev 1996;76:887–926. [DOI] [PubMed] [Google Scholar]
- 30. Portero V, Wilders R, Casini S, Charpentier F, Verkerk AO, Remme CA. KV4.3 expression modulates NaV1.5 sodium current. Front Physiol 2018;9:178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ruijter HMD, Verkerk AO, Coronel R. Incorporated fish oil fatty acids prevent action potential shortening induced by circulating fish oil fatty acids. Front Physiol 2010;1:149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Barry PH, Lynch JW. Liquid junction potentials and small cell effects in patch-clamp analysis. J Membr Biol 1991;121:101–17. [DOI] [PubMed] [Google Scholar]
- 33. Bailey DN, Shaw RF. Blood concentrations and clinical findings in nonfatal and fatal intoxications involving glutethimide and codeine. J Toxicol Clin Toxicol 1985;23:557–70. [DOI] [PubMed] [Google Scholar]
- 34. Clarot F, Goulle J, Vaz E, Proust B. Fatal overdoses of tramadol: is benzodiazepine a risk factor of lethality? Forensic Sci Int 2003;134:57–61. [DOI] [PubMed] [Google Scholar]
- 35. Martin TL, Woodall KL, McLellan BA. Fentanyl-related deaths in Ontario, Canada: toxicological findings and circumstances of death in 112 cases (2002–2004). J Anal Toxicol 2006;30:603–10. [DOI] [PubMed] [Google Scholar]
- 36. Bok CS, Kim RE, Cho YY, Choi JS. Tramadol as a voltage-gated sodium channel blocker of peripheral sodium channels Nav1.7 and Nav1.5. Biomol Ther (Seoul) 2023;31:168–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Alizadeh Ghamsari A, Dadpour B, Najari F. Frequency of electrocardiographic abnormalities in tramadol poisoned patients; a brief report. Emerg (Tehran) 2016;4:151–4. [PMC free article] [PubMed] [Google Scholar]
- 38. Omraninava A, Mehdizade A, Karimi E, Ghabousian A. Potential impact of 3% hypertonic saline infusion on tramadol poisoning-induced electrocardiogram changes; a randomized clinical trial. Arch Acad Emerg Med 2022;10:e26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Cole JB, Sattiraju S, Bilden EF, Asinger RW, Bertog SC. Isolated tramadol overdose associated with Brugada ECG pattern. Pacing Clin Electrophysiol 2012;35:e219–21. [DOI] [PubMed] [Google Scholar]
- 40. Harmer A, Valentin JP, Pollard C. On the relationship between block of the cardiac Na+ channel and drug-induced prolongation of the QRS complex. Br J Pharmacol 2011;164:260–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Meregalli PG, Wilde AAM, Tan HL. Pathophysiological mechanisms of Brugada syndrome: depolarization disorder, repolarization disorder, or more? Cardiovasc Res 2005;67:367–78. [DOI] [PubMed] [Google Scholar]
- 42. Musshoff F, Madea B. Fatality due to ingestion of tramadol alone. Forensic Sci Int 2001;116:197–9. [DOI] [PubMed] [Google Scholar]
- 43. Ghovanloo M-R, Shuart NG, Mezeyova J, Dean RA, Ruben PC, Goodchild SJ. Inhibitory effects of cannabidiol on voltage-dependent sodium currents. J Biol Chem 2018;293:16546–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Sheets M, Hanck D, Fozzard H. Nonlinear relation between Vmax and INa in canine cardiac Purkinje cells. Circ Res 1988;63:386–98. [DOI] [PubMed] [Google Scholar]
- 45. Murray KT, Anno T, Bennett PB, Hondeghem LM. Voltage clamp of the cardiac sodium current at 37 degrees C in physiologic solutions. Biophys J 1990;57:607–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. De Decker K, Cordonnier J, Jacobs W, Coucke V, Schepens P, Jorens PG. Fatal intoxication due to tramadol alone: case report and review of the literature. Forensic Sci Int 2008;175:79–82. [DOI] [PubMed] [Google Scholar]
- 47. Echt DS, Liebson PR, Mitchell LB, Peters RW, Obias-Manno D, Barker AHet al. Mortality and morbidity in patients receiving encainide, flecainide, or placebo: the Cardiac Arrhythmia Suppression Trial. N Engl J Med 1991;324:781–8. [DOI] [PubMed] [Google Scholar]
- 48. Greenberg HM, Dwyer E, Hochman JS, Steinberg JS, Echt DS, Peters RW. Interaction of ischaemia and encainide/flecainide treatment: a proposed mechanism for the increased mortality in CAST I. Br Heart J 1995;74:631–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Rivaud MR, Marchal GA, Wolswinkel R, Jansen JA, van der Made I, Beekman Let al. Functional modulation of atrio-ventricular conduction by enhanced late sodium current and calcium-dependent mechanisms in Scn5a1798insD/+ mice. Europace 2020;22:1579–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. O’Reilly M, Sommerfeld LC, O’Shea C, Broadway-Stringer S, Andaleeb S, Reyat Jet al. Familial atrial fibrillation mutation M1875T-SCN5A increases early sodium current and dampens the effect of flecainide. Europace 2023;25:1152–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Amin AS, Reckman YJ, Arbelo E, Spanjaart AM, Postema PG, Tadros Ret al. SCN5A mutation type and topology are associated with the risk of ventricular arrhythmia by sodium channel blockers. Int J Cardiol 2018;266:128–32. [DOI] [PubMed] [Google Scholar]
- 52. Tan HL, Bink-Boelkens MT, Bezzina CR, Viswanathan PC, Beaufort-Krol G, van Tintelen PJet al. A sodium-channel mutation causes isolated cardiac conduction disease. Nature 2001;409:1043–7. [DOI] [PubMed] [Google Scholar]
- 53. Tsai T-Y, Tsai Y-C, Wu S-N, Liu Y-C. Tramadol-induced blockade of delayed rectifier potassium current in NG108-15 neuronal cells. Eur J Pain 2006;10:597–601. [DOI] [PubMed] [Google Scholar]
- 54. Mert T, Gunes Y, Guven M, Gunay I, Gocmen C. Differential effects of lidocaine and tramadol on modified nerve impulse by 4-aminopyridine in rats. Pharmacology 2003;69:68–73. [DOI] [PubMed] [Google Scholar]
- 55. Emamhadi M, Sanaei-Zadeh H, Nikniya M, Zamani N, Dart RC. Electrocardiographic manifestations of tramadol toxicity with special reference to their ability for prediction of seizures. Am J Emerg Med 2012;30:1481–5. [DOI] [PubMed] [Google Scholar]
- 56. Fanoe S, Jensen GB, Sjøgren P, Korsgaard MP, Grunnet M. Oxycodone is associated with dose-dependent QTc prolongation in patients and low-affinity inhibiting of hERG activity in vitro. Br J Clin Pharmacol 2009;67:172–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Medei E, Raimundo JM, Nascimento JHM, Trachez MM, Sudo RT, Zapata-Sudo G. Inhibition of L-type calcium current by tramadol and enantiomers in cardiac myocytes from rats. Arq Bras Cardiol 2011;97:324–31. [DOI] [PubMed] [Google Scholar]
- 58. Berecki G, Wilders R, De Jonge B, Van Ginneken AC, Verkerk AO. Re-evaluation of the action potential upstroke velocity as a measure of the Na+ current in cardiac myocytes at physiological conditions. PLoS One 2010;5:e15772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Hashimoto K, Amano T, Kasakura A, Uhl GR, Sora I, Sakai Net al. μ-Opioid receptor-independent fashion of the suppression of sodium currents by μ-opioid analgesics in thalamic neurons. Neurosci Lett 2009;453:62–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Tschirhart JN, Li W, Guo J, Zhang S. Blockade of the human ether a-go-go–related gene (hERG) potassium channel by fentanyl. Mol Pharmacol 2019;95:386–97. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data underlying this article will be shared on reasonable request to the corresponding author.