Abstract
Klotho (Kl) is considered an antiaging gene, mainly for the inhibition of the insulin-like growth factor-1 signaling. Kl exists as full-length transmembrane, which acts as co-receptor for fibroblast growth factor receptor, and in soluble forms (sKl). The sKl may exert pleiotropic effects on organs and tissues by regulating several pathways involved in the pathogenesis of diseases associated with oxidative and inflammatory state. In diabetic Patients, serum levels of Kl are significantly decreased compared to healthy subjects, and are related to duration of diabetes. In diabetic retinopathy (DR), one of the most common microvascular complications of type 2 diabetes, serum Kl levels are negatively correlated with progression of the disease. A lot of evidences showed that Kl regulates several mechanisms involved in maintaining homeostasis and functions of retinal cells, including phagocytosis, calcium signaling, secretion of vascular endothelial growth factor A (VEGF-A), maintenance of redox status, and melanin biosynthesis. Experimental data have been shown that Kl exerts positive effects on several mechanisms involved in onset and progression of DR. In particular, treatment with Kl: (1) Prevents apoptosis induced by oxidative stress in human retinal endothelial cells and in retinal pigment epithelium (RPE) cells; (2) reduces secretion of VEGF-A by RPE cells; and (3) decreases subretinal fibrosis and preserves autophagic activity. Therefore, Kl may become a novel biomarker and a good candidate for the treatment of DR.
Keywords: Klotho, Diabetic retinopathy, Retinal pigment epithelium, Vascular endothelial growth factor A, Epithelial to mesenchimal transition, Ocular neo-vascularization
Core Tip: In diabetic Patients, serum levels of Klotho (Kl) are significantly decreased compared to healthy subjects. Moreover, serum Kl levels are negatively correlated with worsening of diabetic retinopathy (DR). Several evidence suggests that retina homeostasis may be affected by altered expression of membrane Kl, as well by reduced levels of soluble Kl. In this review we focused on the role of Kl in DR, highlighting the importance of Kl in maintaining retinal homeostasis and its positive effects on several mechanisms involved in DR onset and progression. Therefore, Kl could be a novel biomarker and a good candidate for the treatment of DR.
INTRODUCTION
Klotho
The name Klotho (Kl) derives from that of the youngest of the Three Fates who spins the thread of human life[1]. Indeed, it is considered an antiaging gene, since phenotypes of mice with mutation in this gene are similar to those of patients with premature-ageing syndromes. Kl shares sequence similarity with members of the glycosidase family 1 and it has been reported to function as a novel β-glucuronidase[2,3]. It encodes for 3 proteins: α-Kl, β-Kl and Kl-related protein (Klrp)[4]. β-Kl is mainly expressed in liver and adipose tissue and is involved in metabolic processes[4]; whereas Klrp is a cytosolic β-glucocerebrosidase[5]. α-Kl, generally simply referred as Kl, is a type I single-pass transmembrane glycoprotein mainly expressed in the kidneys, liver, brain, and at lower level in the pituitary, skeletal muscle, urinary bladder, pancreas, testis, ovary, colon, thyroid gland, placenta and vascular tissue[1]. Both the intracellular and the transmembrane domains of α-Kl are very short, whereas the extracellular domain is longer and contains two repeated sequences (KL1 and KL2)[4,6]. After association with fibroblast growth factor receptors (FGFRs), the full-length transmembrane Kl (mKl) acts as coreceptor for the bone-derived phosphaturic hormone FGF23, thus taking part to phosphate excretion and calcium homeostasis by regulating the expression and activity of the calcium channel transient receptor potential vanilloid 5 (TRPV5)[7]. Besides mKl, there are 2 isoforms of α-Kl: A shed soluble form (sKl), which derives from the cleavage of the extracellular domain of Kl from the cell surface by the metalloproteinases ADAM10 and ADAM17, and a secreted form that is produced by alternative splicing of Kl mRNA[4]. The shed soluble form of Kl seems to be dominant on both the secreted and the membrane forms in humans[8]. It has been proposed that the soluble forms of Kl function as a hormone[9]. Moreover, since circulating levels of sKl increase following exercise training, it has been also hypothesized that Kl may be related to the antiaging effects of physical activity[10]. The sKl has pleiotropic effects on a lot of organs and tissues, thus regulating several pathways[8]. Indeed, after the release in blood, urine and cerebrospinal fluid, sKl exerts biological effects involved in preservation of endothelial integrity and permeability, and affect intracellular signaling pathways including those related to insulin, insulin-like growth factor-1 (IGF-1), PI3K, NF-kB, p53/p21, cAMP, protein kinase C and Wnt[8,11-13]. In particular, a lot of evidence demonstrated that the anti-ageing effects of sKl have been associated with the inhibition of IGF-1 signaling and its downstream actions especially by enhancing resistance to oxidative stress[14,15]. Indeed, inhibition of the IGF-1 signaling by sKl results in increased production of antioxidant enzymes[16]. Therefore, activity of sKl may regulate several pathways involved in the pathogenesis of diseases associated with oxidative and inflammatory state.
It is not yet clear whether intracellular signaling of circulating Kl is mediated by a membrane receptor. Recent hypothesis suggests that sKL may act as a circulating co-receptor for membrane-bound FGFRs, thus allowing the interaction with FGF23 and regulating FGFR-mediated signaling also in cells lacking the full length form of Kl[17]. Moreover, it has been demonstrated that sKl is able to bind membrane lipid rafts, alter their organization, and affect caveolae-mediated TRPV5 endocytosis[18], suggesting that the intracellular signaling of sKl may occur at the level of caveoale.
Kl AND DIABETES
In diabetic patients, serum levels of Kl have been found significantly decreased compared with those of healthy subjects[19]. In addition, the amount of sKl is related to duration of diabetes and is negatively correlated to HbA1c. Kidneys are considered the main source of sKl[17], and are also the principal organ involved in the clearance of sKl from the circulation into the urine, thus playing a dual role in the homeostasis of Kl[9]. Therefore, altered kidney function may affect the systemic effects of Kl. Consequently, the anti-aging effects of Kl have been extensively investigated in kidneys, reporting that increased levels of Kl inhibit the progression of various kidney diseases[20,21]. In animal models of diabetes, Kl counteracts podocytic and glomerular albumin permeability induced by hyperglycemia[22], and prevents epithelial-mesenchymal transition (EMT) in diabetic kidneys[21]. Interestingly, expression of Kl has been found decreased in the renal cortices of mice with diabetes[22]. Moreover, Typiak et al[23] showed that decreased levels of membrane-bound Kl are associated to increased shedding of Kl, to higher levels in serum of diabetic rats and a to reduced urinary excretion[23]. In diabetic patients, the amount of soluble Kl is reduced in the early stage of chronic kidney disease (CKD), but increased with disease progression and the decrease of glomerular filtration rate[24]. A recent meta-analysis of data on sKl amount in patients with diabetic nephropathy (DN) confirms that levels of sKl are further lowered in the early stage of DN[25], suggesting that Kl might be considered as an early biomarker of DN[23,26]. However, although levels of sKl still remain lower in patients with DN, they seem to increase during the worsening of diabetic CKD probably linked to the decline in glomerular filtration rate that leads to reduced urinary excretion of Kl[23,27].
Expression of Kl has been detected also in mouse pancreatic islets and in beta-cell line[28,29]. It has been showed that Kl is involved in regulation of glucose-induced insulin secretion, probably, through regulation of TRPV2 expression[28,29]. Indeed, overexpression of Kl increases both insulin secretion and plasma membrane levels of TRPV2; whereas silencing of Kl negatively affects plasma membrane levels of TRPV2, glucose-induced calcium entry and insulin secretion[28]. Moreover, treatment with α- or β-Kl protects human beta-cells by cytokine-induced apoptosis and improved insulin secretion[30,31].
DIABETIC RETINOPATHY
Diabetic retinopathy (DR) is a common microvascular complications of type 2 diabetes and represents the primary cause of blindness in working age adults[32]. Actually, retinal neurodegenerative lesions may occur earlier than microvascular ones, therefore DR has been defined as a highly tissue-specific neurovascular complication of diabetes by the American Diabetes Association[33]. The early manifestations of DR involves damages to both microcirculation and retinal neurons and are associated with oxidative stress[34]. The resulting sustained proinflammatory environment, in turns, increases oxidative stress, due to the reduced levels of antioxidant enzymes in the retina. Photoreceptors and the retinal pigment epithelium (RPE) cells are highly susceptible to oxidative stress in the early stage of DR and their dysfunction lead to progression of retinal degeneration[34]. Furthermore, chronic inflammation causes vasoregression and alters vascular permeability, leading to formation of microaneurysms and exudates. Then, hypoxia and the release of proangiogenic factors, such as vascular endothelial growth factor A (VEGF-A), may promote pathological ocular neovascularization[34]. In the retina, VEGF-A is mainly produced by RPE cells, a monolayer of highly specialized cells located between the choroid and photoreceptors that forms the outer blood-retinal barrier[35]. Due to their localization, RPE cells may affect retinal homeostasis by altering the function and maintenance of both the photoreceptors and capillary endothelium[36]. Indeed, under normal condition, VEGF-A is released at low concentrations from the basal side of the RPE to maintain endothelial cell function[37]. However, under pathological condition, such as chronic hyperglycemia, secretion of VEGF-A increases leading to activation of endothelial cells and altered permeability of the choroidal vessels[37,38]. It is well known that dysfunction of RPE cells contributes to onset and progression of DR. Therefore, maintaining the function of RPE and controlling the levels of VEGF-A are of great importance in preventing worsening of DR to the proliferative state.
Kl AND RETINAL HOMEOSTASIS
It has been found that Kl is expressed in the human retina, optic nerve, and lens[39,40]. Several evidence showed that Kl regulates a lot of mechanisms involved in maintaining homeostasis and functions of retinal cells[39,41,42]. Firstly, Kl knockout mice display several morphological changes as compared to wild type mice: Decreased pigmentation of the RPE layer, large choroidal vessels, thinner and deformed basal membrane, and signs of degeneration in the outer segment of photoreceptors (POS)[41]. Proteomics analysis reveals that proteins involved in eye development, visual perception and mitochondrial function are downregulated in Kl knockout mice[42]. Accordingly, Kl knockout mice have reduced retinal function, with functional deficit comparable to those observed in IGF-1 knockout mice[39]. Considering that Kl knockout mice are hypoglycemic, it can be hypothesized that the effects observed in the retina may be attributable to increased sensitive to the insulin and IGF-1 signaling.
Kokkinaki et al[41] demonstrated that Kl is expressed in primary cultures of RPE cells, mainly in the cell membrane, and that its depletion compromises several important function of RPE cells[41]. Moreover, they demonstrated that treatment with recombinant Kl protein has protective effects on RPE function, including phagocytosis, VEGF-A secretion, oxidative stress response, and melanogenesis.
Phagocytosis of POS is of particular importance in maintaining visual function and the visual cycle. It has been shown that transfection of RPE cells with Kl siRNA significantly reduced phagocytosis[41], suggesting that Kl is involved in the regulation of this important function. Evidences that treatment of RPE cells with Kl significantly increased phagocytosis in RPE cells confirm this hypothesis[41]. POS phagocytosis is regulated by several factors, among them, the Ca2+ signaling and the expression of Mer Tyrosine Kinase (MerTK) seem to play an important role[43]. Rise in intracellular Calcium is required for maintaining POS phagocytosis rate[44-46]. It has been reported that secreted Kl may regulate calcium homeostasis by affecting activity of calcium channels, including TRPVs and the Ca2+ release-activated Ca2+ channel (CRAC)[28,47,48]. Interestingly, human RPE expresses both TRPV5 and CRAC, which regulate calcium entry in this cells[49,50]. However, Kokkinaki et al[41] showed that treatment of RPE cells with Kl did not increase intracellular Calcium concentration[41], suggesting that Kl increases phagocytosis through a mechanism independent to calcium. Internalization of POS requires the engagement of MerTK, a cell surface receptor member of the tyro/Axl/Mer family of receptor tyrosine kinase, therefore MerTK expression is critical for POS phagocytosis[43]. Interestingly, it has been demonstrated that Kl regulates phagocytosis by upregulating MerTK expression, indeed treatment of RPE cells with Kl induces intracellular signaling that leads to increased expression of MerTK and, consequently, improves phagocytosis efficiency[41].
VEGF-A is one of the main important pro-angiogenic factor and its excessive secretion is implicated in promoting the pathological neovascularization of the choroidal vasculature[51,52]. RPE cells are the major responsible of VEGF-A production in the retina. Treatment of the RPE cell line ARPE-19 with Kl significantly decreases VEGF-A secretion from both the apical and the basal sides[41]. Moreover, the presence of Kl inhibits the phosphorylation of VEGFR2 induced by VEGF-A, thus affecting intracellular signaling activated by VEGF-A.
Due to its extremely active metabolism, the retina is one of the organ with major request of oxygen, therefore it may be susceptible to overproduction of reactive oxygen species (ROS). Under normal conditions, ROS take part to the retinal physiological signaling, however, when generation of ROS exceeds the natural antioxidants defenses, oxidative stress may contribute to the pathogenesis of several retinal diseases, including DR. Experimental data demonstrate that Kl contributes to maintain the redox balance in the retina. Indeed, mRNA levels of Kl have been found significantly decreased in ARPE-19 cells treated with hydrogen peroxide (H2O2)[53]. Moreover, Kokkinaki et al[41] demonstrated that down-regulation of Kl expression leads to reduced expression of the anti-oxidant Superoxide dismutase 2 (SOD2) in RPE cells[41]. On the contrary, pretreatment with sKl prevented rise in ROS induced by H2O2 enhancing the antioxidant activities of ARPE-19[53], and decreased apoptosis induced by oxidative stress in human retinal endothelial cells[54].
Eye pigmentation is essential to maintain visual function. The RPE contribute to absorption of scattered light and to reduce retinal damage from ultraviolet light by forming a dark-brown pigmented wall[35,55]. Studies on models in which Kl expression has been down-regulated revealed that Kl is involved in regulation of genes encoding for melanin biosynthesis[41]. Indeed, pigmentation of eyes from Kl k/o mice was reduced and their RPE cells contained fewer melanin granules than normal RPE cells[41].
All these findings suggest that retina homeostasis may be affected by altered expression of Kl, as well altered levels of soluble Kl (Table 1).
Table 1.
Main effects of Klotho on retinal cells
|
Functions
|
Effects of Klotho depletion
|
Effects of treatment with Klotho
|
Type of cell
|
Ref.
|
| Phagocytosis | Reduced | Improved | RPE cells | [41,43] |
| Increased expression of Mertk | ||||
| VEGF-A | Decreased secretion | RPE cells | [41] | |
| Reduced signaling mediated by VEGFR2- and IGF-1R | ||||
| Redox balance | Increased oxidative stress | Restored | RPE cells | [41,53] |
| Prevention of ROS production | ||||
| Increased NRF2 expression and nuclear translocation | ||||
| Reduced expression of SOD2 | Restored expression of SOD2 and CAT | |||
| Pigmentation | Reduced | RPE cells | [41] | |
| Decreased melanin granules | ||||
| Mitochondrial function | Reduced biogenesis of mitochondria | Preserved | RPE cells | [53] |
| Autophagy | Improved | Retina | [42] | |
| Decreased activation of AMPK | ||||
| Reduced expression of SIRT1 | ||||
| EMT | Decreased expression of mesenchymal cell markers | RPE cells | [66] | |
| Apoptosis | Reduced | RPE and retinal endothelial cells | [42,53,54] | |
| Increased expression of Bcl-2 | ||||
| Decreased expression of Bax | ||||
| Decreased activity of Caspase-3 |
VEGF-A: Vascular endothelial growth factor A; IGF-1R: Insulin-like growth factor-1; RPE: Retinal pigment epithelium; ROS: Reactive oxygen species; AMPK: 5' adenosine monophosphate-activated protein kinase; SIRT1: Silent information regulator 1; EMT: Epithelial-mesenchymal transition; NRF2: Nuclear factor E2-related factor 2; SOD2: Superoxide dismutase 2; CAT: Catalase.
Kl AND DR
Levels of sKl has been found reduced in ocular pathologies characterized by inflammatory state[56-59], suggesting that the reduced levels of sKl may be a common feature in several ocular diseases. In particular, decreased levels of Kl may be associated with increased risk of onset and worsening of DR. Indeed, circulating levels of Kl are lower in diabetic subject with DR than in those without this complication[54,60]. Moreover, serum Kl levels are negatively correlated with progression of DR[54,60]. Following the onset of DR in diabetic patients reveals that patients with progression of retinopathy had lower levels of serum Kl as compared to those without[60]. In addition, Ji et al[54] found that levels of sKl are gradually reduced among patient with diabetes without DR, non-proliferative DR (PDR) and PDR, independently of DN[54]. Corcillo et al[60] hypothesize that a halving of circulating Kl levels may increase the risk of retinopathy progression by 44%[60]. On the other hand, the incidence of the functional “KL-VS” variant of the Kl gene, which is associated with higher longevity in humans, is lower in people with DR and is associated with reduced serum levels of inflammatory markers and pro-angiogenic factors, suggesting that this genotype may be protective against retinopathy incidence[61].
As reported in the previous section, several experimental models demonstrated that depletion of Kl negatively affects important function of retinal cells, including oxidative stress response, VEGF-A secretion, and phagocytosis, leading to activation of mechanisms that may contribute to onset and progression of DR. On the other hand, there are also several evidence that treatment with recombinant sKl or overexpression of Kl ameliorate retinal function.
Oxidative stress and inflammation have been causative associated with DR[62,63]. It has been reported that Kl exerts protective effects against oxidative stress in retinal cells[13,41,42,53]. Firstly, it has been observed that pretreatment with sKl prevents increment of ROS production in ARPE-19 cells exposed to H2O2[41,53]. In particular, Wen et al[53] demonstrated that sKl improves redox balance in H2O2-treated ARPE-19 cells by increasing expression and nuclear translocation of nuclear factor E2-related factor 2 (Nrf2), thus restoring glutathione peroxidase, SOD2 and catalase to the levels of untreated cells[53]. In addition, pretreatment with sKl prevents H2O2-induced apoptosis of ARPE-19 cells[42,53], by increasing expression of Bcl-2 and decreasing the activation of caspase-3[53].
It is well established that VEGF-A plays an important role in driving pathological neovascularization of the retina during DR, and that neovascularization due to severe hypoxia is a hallmark of PDR[34]. The expression of VEGF-A is regulated by hypoxia-inducible factor-1α (HIF-1α), which is a transcription factor involved in cellular response to hypoxia and hyperglycemia[64,65]. Interestingly, Kl levels have been found decreased in ARPE-19 cells exposed to hypoxia and in laser-induced CNV lesions in mice[66]. Xie et al[66] demonstrated that HIF-1α, besides directly increase VEGF-A transcription, may be responsible of down-regulation of Kl expression during hypoxia[66]. Indeed, HIF-1α activates p53, which, in turns, leads to the increased levels of miRNA34, that targets Kl thus reducing its expression[66]. Given that Kl is expressed in ocular tissues, it is possible that part of the sKl that acts in the eye derives by local shedding of mKl, therefore its contribution may be lost when expression of Kl is down-regulated. It has been reported that treatment with Kl reduces VEGF-A secretion from ARPE-19 cells[41]. In particular, Kl was able to decrease VEGF-A secretion by reducing phosphorylation of both IGF-1 receptor (IGF-1R) and VEGR2. The pathogenic role of IGF-1 in the development of PDR is still debated, several studies indicate that increased activation of IGF-1 signaling may contribute to retinal neovascularization, however a strong relationship between IGF-1 and the development of proliferative retinopathy has not been still clearly demonstrated[67-69]. Several studies reported that IGF-1R signaling is regulated by lipid raft integrity and interaction with caveolin-1[70-74]. In particular, down-regulation of caveolin-1 expression in RPE cells significantly reduces both basal and IGF-1-stimulated VEGF-A secretion[72]. These data together with the ability of Kl to modify the lipid organization within lipid rafts/caveolae[18] suggest that Kl may reduce the phosphorylation of IGF-1R by altering these microdomains. Hyperglycemia increases production and secretion of VEGF-A by Muller cells in the retina. In particular, Yu et al[75] demonstrated that hyperglycemia increases the production of VEGF-A in Muller glial cells through the activation of FGFR1[75]. It is well known that sKl acts as a co-receptor for FGFs at non-renal sites and activates protective pathways in several cell types[76,77]. Interestingly, screening the potential pathogenic genes associated with DR revealed that hyperglycemia increases the expression of FGF23[78], and of its membrane receptor FGFR1 on Muller glial cells[75,79]. Considering that absence of Kl may allow Kl-independent activation of FGFRs resulting in pathological cellular changes[17,76,77], and that Kl-independent action of FGF23 has been reported to contribute to endothelial dysfunction[17], these findings suggest that lower levels of Kl together with increased production of FGF23 may contribute to the onset of DR and to progression to PDR by increasing VEGF-A production.
Autophagy is a highly conserved lysosomal pathway for the turnover of cytoplasmic organelles and long-lived proteins that acts as an adaptive response to cellular stresses and regulates homeostasis, differentiation, development and survival in several cell types[80]. In retinal cells, autophagy plays an important role by participating to POS degradation, visual pigment recycling, and lipofuscin degradation[81-83]. Altered activation of autophagy has been found in experimental models of DR and in the retina of diabetic patients[84,85]. For instance, RPE cells exposed to high glucose concentration increase formation of autophagosome, suggesting that induction of autophagy is a cytoprotective response against high glucose (HG)[84,85]. However, the excessive activation of this mechanism may lead to its impairment as occur in retinal Muller cells, where the process of degradation cannot be completed due to the lysosomal dysfunction[85]. It has been reported that autophagic activity is reduced in DM mice and human renal proximal tubule cells exposed to HG[86]. Recent studies showed that Kl may act as a regulator of autophagy even in diabetic condition[87]. Specific expression of Kl significantly improves autophagy in both pancreatic beta cells and in renal tubule cells exposed to HG[29,86]. Moreover, Zou et al[21] showed that activation of 5' adenosine monophosphate-activated protein kinase (AMPK), a positive regulator of autophagy, is significantly decreased in the retina of Kl deficient mice as compared to that of WT mice[42]. Although there is no direct evidence, these finding suggest that Kl may affect autophagy also in retinal cells. A decreased activation of AMPK has been observed also in arterial endothelial cells of Kl deficient mice[88], confirming that AMPK is a crucial mediator of protective effects of Kl. Moreover, Kl deficient mice have also reduced activity of silent information regulator (SIRT) 1[88], another important player in autophagy[89]. Interestingly, the expression of SIRT1 is reduced in DR and intravitreal administration of SIRT1 reverses DR in a mouse model of type 2 diabetes[90]. These results suggest that regulation of SIRT1 may be another mechanism through which Kl improve DR.
PDR is also characterized by formation of fibrous proliferative anterior membrane[91]. Subretinal fibrosis is mediated by EMT, a process that leads RPE cells to the acquisition of a mesenchymal phenotype[92]. Several evidence demonstrated that HG induce EMT in RPE[93,94]. It has been shown that Kl expression is down-regulated in models of induced fibrosis, suggesting a protective role of Kl[22,95,96]. In particular, the protective effects of Kl have been related to inhibition of the Wnt/β-catenin and the Egr-mediated signaling pathways. Recently, it has been reported that overexpression of Kl decreased the expression of mesenchymal cell markers induced by hypoxia in ARPE-19 cells[66]. Moreover, overexpression of Kl was able to reduce subretinal fibrosis in a mouse laser-induced CNV model[66]. Here, under hypoxic conditions, Kl was able to block the axis that through HIF-1α leads to the activation of p53 and promotes EMT in RPE cells, confirming that Kl may be useful in preventing EMT also in RPE cells.
Besides hyperglycemia, dyslipidemia is another important actor in the progression of DR[97,98]. Palmitic acid (PA) is involved in the onset of DR and may induce endothelial cell damage[98]. It has been demonstrated that Kl pretreatment significantly reduces apoptosis induced by PA in human retinal endothelial cells[54]. This effect implies the activation of the PI3K and subsequent phosphorylation of AKT[54]. Moreover, Kl affects expression of proteins involved in apoptosis leading to increased expression of the anti-apoptotic Bcl-2 and down-regulation of the pro-apoptotic Bax[54]. Consistent with these data, pretreatment with Kl reduced the apoptosis rate in ARPE-19 cells exposed to H2O2 by up-regulating Bcl-2 expression and decreasing levels of Bax[53]. In addition, Kl was able to prevent the decrease of mitochondrial membrane potential and the activation of Caspase-3 induced by H2O2[53].
CONCLUSION
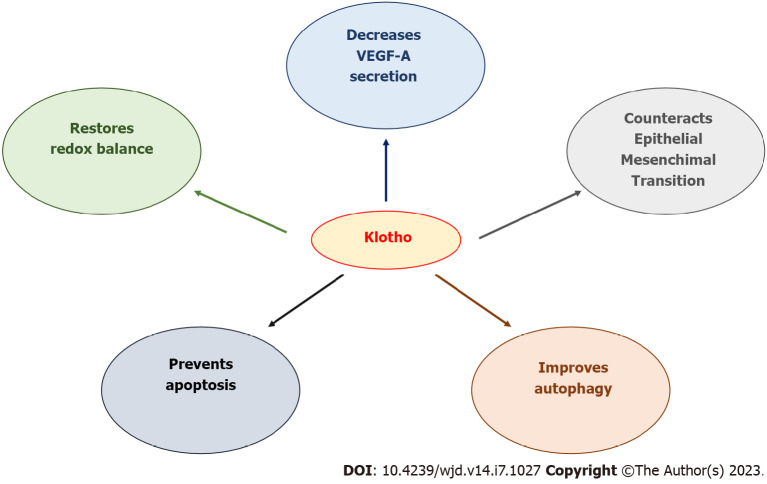
DR is a common complication of diabetes. The International Diabetes Federation estimated the global population with diabetes mellitus to be 463 million in 2019 and 700 million in 2045[99]. These data require the development of strategies able to prevents the onset and the progression of DR. To date, the first line treatment for PDR is intravitreal anti-VEGF therapy. However, it is not so successful for routine treatment of non-PDR[32,100]. Therefore, new molecules in development have been designed to target other pathways involved in pathogenesis of DR[101,102]. It has been demonstrated that Kl has protective effects in DN and that pathological mechanisms between DR and DN share similarities[19,29], suggesting that Kl may be a good candidate in counteracting DR. Experimental models targeting Kl have been shown to have positive effects on several mechanisms involved in DR onset and progression (Figure 1). Therefore, Kl may become a novel biomarker and a good candidate for the treatment of DR[60].
Figure 1.
Positive effects of Klotho in diabetic retinopathy. VEGF-A: Vascular endothelial growth factor A.
Footnotes
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Peer-review started: February 16, 2023
First decision: April 11, 2023
Article in press: May 22, 2023
Specialty type: Endocrinology and metabolism
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Cen LS, China; Liu Y, China; Long P, China; Mansour AM, Lebanon S-Editor: Fan JR L-Editor: A P-Editor: Fan JR
Contributor Information
Alessandra Puddu, Department of Internal Medicine and Medical Specialties, University of Genova, Genova 16132, Italy. alep100@hotmail.com.
Davide Carlo Maggi, Department of Internal Medicine and Medical Specialties, University of Genova, Genova 16132, Italy.
References
- 1.Kuro-o M, Matsumura Y, Aizawa H, Kawaguchi H, Suga T, Utsugi T, Ohyama Y, Kurabayashi M, Kaname T, Kume E, Iwasaki H, Iida A, Shiraki-Iida T, Nishikawa S, Nagai R, Nabeshima YI. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature. 1997;390:45–51. doi: 10.1038/36285. [DOI] [PubMed] [Google Scholar]
- 2.Hayashi Y, Okino N, Kakuta Y, Shikanai T, Tani M, Narimatsu H, Ito M. Klotho-related protein is a novel cytosolic neutral beta-glycosylceramidase. J Biol Chem. 2007;282:30889–30900. doi: 10.1074/jbc.M700832200. [DOI] [PubMed] [Google Scholar]
- 3.Tohyama O, Imura A, Iwano A, Freund JN, Henrissat B, Fujimori T, Nabeshima Y. Klotho is a novel beta-glucuronidase capable of hydrolyzing steroid beta-glucuronides. J Biol Chem. 2004;279:9777–9784. doi: 10.1074/jbc.M312392200. [DOI] [PubMed] [Google Scholar]
- 4.Xu Y, Sun Z. Molecular basis of Klotho: from gene to function in aging. Endocr Rev. 2015;36:174–193. doi: 10.1210/er.2013-1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hayashi Y, Ito M. Klotho-Related Protein KLrP: Structure and Functions. Vitam Horm. 2016;101:1–16. doi: 10.1016/bs.vh.2016.02.011. [DOI] [PubMed] [Google Scholar]
- 6.Dalton GD, Xie J, An SW, Huang CL. New Insights into the Mechanism of Action of Soluble Klotho. Front Endocrinol (Lausanne) 2017;8:323. doi: 10.3389/fendo.2017.00323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wolf MT, An SW, Nie M, Bal MS, Huang CL. Klotho up-regulates renal calcium channel transient receptor potential vanilloid 5 (TRPV5) by intra- and extracellular N-glycosylation-dependent mechanisms. J Biol Chem. 2014;289:35849–35857. doi: 10.1074/jbc.M114.616649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baranowska B, Kochanowski J. The metabolic, neuroprotective cardioprotective and antitumor effects of the Klotho protein. Neuro Endocrinol Lett. 2020;41:69–75. [PubMed] [Google Scholar]
- 9.Hu MC, Shi M, Zhang J, Addo T, Cho HJ, Barker SL, Ravikumar P, Gillings N, Bian A, Sidhu SS, Kuro-o M, Moe OW. Renal Production, Uptake, and Handling of Circulating αKlotho. J Am Soc Nephrol. 2016;27:79–90. doi: 10.1681/ASN.2014101030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Corrêa HL, Raab ATO, Araújo TM, Deus LA, Reis AL, Honorato FS, Rodrigues-Silva PL, Neves RVP, Brunetta HS, Mori MADS, Franco OL, Rosa TDS. A systematic review and meta-analysis demonstrating Klotho as an emerging exerkine. Sci Rep. 2022;12:17587. doi: 10.1038/s41598-022-22123-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Prud'homme GJ, Kurt M, Wang Q. Pathobiology of the Klotho Antiaging Protein and Therapeutic Considerations. Front Aging. 2022;3:931331. doi: 10.3389/fragi.2022.931331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wolf I, Levanon-Cohen S, Bose S, Ligumsky H, Sredni B, Kanety H, Kuro-o M, Karlan B, Kaufman B, Koeffler HP, Rubinek T. Klotho: a tumor suppressor and a modulator of the IGF-1 and FGF pathways in human breast cancer. Oncogene. 2008;27:7094–7105. doi: 10.1038/onc.2008.292. [DOI] [PubMed] [Google Scholar]
- 13.Wang Y, Kuro-o M, Sun Z. Klotho gene delivery suppresses Nox2 expression and attenuates oxidative stress in rat aortic smooth muscle cells via the cAMP-PKA pathway. Aging Cell. 2012;11:410–417. doi: 10.1111/j.1474-9726.2012.00796.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kurosu H, Yamamoto M, Clark JD, Pastor JV, Nandi A, Gurnani P, McGuinness OP, Chikuda H, Yamaguchi M, Kawaguchi H, Shimomura I, Takayama Y, Herz J, Kahn CR, Rosenblatt KP, Kuro-o M. Suppression of aging in mice by the hormone Klotho. Science. 2005;309:1829–1833. doi: 10.1126/science.1112766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Xie B, Zhou J, Shu G, Liu DC, Chen J, Yuan L. Restoration of klotho gene expression induces apoptosis and autophagy in gastric cancer cells: tumor suppressive role of klotho in gastric cancer. Cancer Cell Int. 2013;13:18. doi: 10.1186/1475-2867-13-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yamamoto M, Clark JD, Pastor JV, Gurnani P, Nandi A, Kurosu H, Miyoshi M, Ogawa Y, Castrillon DH, Rosenblatt KP, Kuro-o M. Regulation of oxidative stress by the anti-aging hormone klotho. J Biol Chem. 2005;280:38029–38034. doi: 10.1074/jbc.M509039200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Richter B, Faul C. FGF23 Actions on Target Tissues-With and Without Klotho. Front Endocrinol (Lausanne) 2018;9:189. doi: 10.3389/fendo.2018.00189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dalton G, An SW, Al-Juboori SI, Nischan N, Yoon J, Dobrinskikh E, Hilgemann DW, Xie J, Luby-Phelps K, Kohler JJ, Birnbaumer L, Huang CL. Soluble klotho binds monosialoganglioside to regulate membrane microdomains and growth factor signaling. Proc Natl Acad Sci U S A. 2017;114:752–757. doi: 10.1073/pnas.1620301114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang L, Liu T. Clinical implication of alterations in serum Klotho levels in patients with type 2 diabetes mellitus and its associated complications. J Diabetes Complications. 2018;32:922–930. doi: 10.1016/j.jdiacomp.2018.06.002. [DOI] [PubMed] [Google Scholar]
- 20.Xue J, Wang L, Sun Z, Xing C. Basic Research in Diabetic Nephropathy Health Care: A study of the Renoprotective Mechanism of Metformin. J Med Syst. 2019;43:266. doi: 10.1007/s10916-019-1412-4. [DOI] [PubMed] [Google Scholar]
- 21.Zou D, Wu W, He Y, Ma S, Gao J. The role of klotho in chronic kidney disease. BMC Nephrol. 2018;19:285. doi: 10.1186/s12882-018-1094-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li Y, Xue M, Hu F, Jia Y, Zheng Z, Yang Y, Liu X, Wang Y. Klotho prevents epithelial-mesenchymal transition through Egr-1 downregulation in diabetic kidney disease. BMJ Open Diabetes Res Care. 2021;9 doi: 10.1136/bmjdrc-2020-002038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Typiak M, Kulesza T, Rachubik P, Rogacka D, Audzeyenka I, Angielski S, Saleem MA, Piwkowska A. Role of Klotho in Hyperglycemia: Its Levels and Effects on Fibroblast Growth Factor Receptors, Glycolysis, and Glomerular Filtration. Int J Mol Sci. 2021;22 doi: 10.3390/ijms22157867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kacso IM, Bondor CI, Kacso G. Soluble serum Klotho in diabetic nephropathy: relationship to VEGF-A. Clin Biochem. 2012;45:1415–1420. doi: 10.1016/j.clinbiochem.2012.07.098. [DOI] [PubMed] [Google Scholar]
- 25.Xin C, Sun X, Li Z, Gao T. Relationship of Soluble Klotho and Early Stage of Diabetic Nephropathy: A Systematic Review and Meta-Analysis. Front Endocrinol (Lausanne) 2022;13:902765. doi: 10.3389/fendo.2022.902765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Piwkowska A, Zdrojewski Ł, Heleniak Z, Dębska-Ślizień A. Novel Markers in Diabetic Kidney Disease-Current State and Perspectives. Diagnostics (Basel) 2022;12 doi: 10.3390/diagnostics12051205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang K, Mao Y, Lu M, Liu X, Sun Y, Li Z, Li Y, Ding Y, Zhang J, Hong J, Xu D. Association between serum Klotho levels and the prevalence of diabetes among adults in the United States. Front Endocrinol (Lausanne) 2022;13:1005553. doi: 10.3389/fendo.2022.1005553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lin Y, Sun Z. Antiaging gene Klotho enhances glucose-induced insulin secretion by up-regulating plasma membrane levels of TRPV2 in MIN6 β-cells. Endocrinology. 2012;153:3029–3039. doi: 10.1210/en.2012-1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lin Y, Sun Z. In vivo pancreatic β-cell-specific expression of antiaging gene Klotho: a novel approach for preserving β-cells in type 2 diabetes. Diabetes. 2015;64:1444–1458. doi: 10.2337/db14-0632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Son DO, Liu W, Li X, Prud'homme GJ, Wang Q. Combined effect of GABA and glucagon-like peptide-1 receptor agonist on cytokine-induced apoptosis in pancreatic β-cell line and isolated human islets. J Diabetes. 2019;11:563–572. doi: 10.1111/1753-0407.12881. [DOI] [PubMed] [Google Scholar]
- 31.Geng L, Liao B, Jin L, Yu J, Zhao X, Zhao Y, Zhong L, Wang B, Li J, Liu J, Yang JK, Jia W, Lian Q, Xu A. β-Klotho promotes glycolysis and glucose-stimulated insulin secretion via GP130. Nat Metab. 2022;4:608–626. doi: 10.1038/s42255-022-00572-2. [DOI] [PubMed] [Google Scholar]
- 32.Tan TE, Wong TY. Diabetic retinopathy: Looking forward to 2030. Front Endocrinol (Lausanne) 2022;13:1077669. doi: 10.3389/fendo.2022.1077669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Solomon SD, Chew E, Duh EJ, Sobrin L, Sun JK, VanderBeek BL, Wykoff CC, Gardner TW. Diabetic Retinopathy: A Position Statement by the American Diabetes Association. Diabetes Care. 2017;40:412–418. doi: 10.2337/dc16-2641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wong TY, Cheung CM, Larsen M, Sharma S, Simó R. Diabetic retinopathy. Nat Rev Dis Primers. 2016;2:16012. doi: 10.1038/nrdp.2016.12. [DOI] [PubMed] [Google Scholar]
- 35.Strauss O. The retinal pigment epithelium in visual function. Physiol Rev. 2005;85:845–881. doi: 10.1152/physrev.00021.2004. [DOI] [PubMed] [Google Scholar]
- 36.Ponnalagu M, Subramani M, Jayadev C, Shetty R, Das D. Retinal pigment epithelium-secretome: A diabetic retinopathy perspective. Cytokine. 2017;95:126–135. doi: 10.1016/j.cyto.2017.02.013. [DOI] [PubMed] [Google Scholar]
- 37.Kannan R, Zhang N, Sreekumar PG, Spee CK, Rodriguez A, Barron E, Hinton DR. Stimulation of apical and basolateral VEGF-A and VEGF-C secretion by oxidative stress in polarized retinal pigment epithelial cells. Mol Vis. 2006;12:1649–1659. [PubMed] [Google Scholar]
- 38.Takahashi H, Shibuya M. The vascular endothelial growth factor (VEGF)/VEGF receptor system and its role under physiological and pathological conditions. Clin Sci (Lond) 2005;109:227–241. doi: 10.1042/CS20040370. [DOI] [PubMed] [Google Scholar]
- 39.Reish NJ, Maltare A, McKeown AS, Laszczyk AM, Kraft TW, Gross AK, King GD. The age-regulating protein klotho is vital to sustain retinal function. Invest Ophthalmol Vis Sci. 2013;54:6675–6685. doi: 10.1167/iovs.13-12550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang Y, Wang L, Wu Z, Yu X, Du X, Li X. The Expressions of Klotho Family Genes in Human Ocular Tissues and in Anterior Lens Capsules of Age-Related Cataract. Curr Eye Res. 2017;42:871–875. doi: 10.1080/02713683.2016.1259421. [DOI] [PubMed] [Google Scholar]
- 41.Kokkinaki M, Abu-Asab M, Gunawardena N, Ahern G, Javidnia M, Young J, Golestaneh N. Klotho regulates retinal pigment epithelial functions and protects against oxidative stress. J Neurosci. 2013;33:16346–16359. doi: 10.1523/JNEUROSCI.0402-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhou S, Hum J, Taskintuna K, Olaya S, Steinman J, Ma J, Golestaneh N. The Anti-Aging Hormone Klotho Promotes Retinal Pigment Epithelium Cell Viability and Metabolism by Activating the AMPK/PGC-1α Pathway. Antioxidants (Basel) 2023;12 doi: 10.3390/antiox12020385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kwon W, Freeman SA. Phagocytosis by the Retinal Pigment Epithelium: Recognition, Resolution, Recycling. Front Immunol. 2020;11:604205. doi: 10.3389/fimmu.2020.604205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Karl MO, Kroeger W, Wimmers S, Milenkovic VM, Valtink M, Engelmann K, Strauss O. Endogenous Gas6 and Ca2+ -channel activation modulate phagocytosis by retinal pigment epithelium. Cell Signal. 2008;20:1159–1168. doi: 10.1016/j.cellsig.2008.02.005. [DOI] [PubMed] [Google Scholar]
- 45.Müller C, Más Gómez N, Ruth P, Strauss O. CaV1.3 L-type channels, maxiK Ca(2+)-dependent K(+) channels and bestrophin-1 regulate rhythmic photoreceptor outer segment phagocytosis by retinal pigment epithelial cells. Cell Signal. 2014;26:968–978. doi: 10.1016/j.cellsig.2013.12.021. [DOI] [PubMed] [Google Scholar]
- 46.Strauß O, Reichhart N, Gomez NM, Müller C. Contribution of Ion Channels in Calcium Signaling Regulating Phagocytosis: MaxiK, Cav1.3 and Bestrophin-1. Adv Exp Med Biol. 2016;854:739–744. doi: 10.1007/978-3-319-17121-0_98. [DOI] [PubMed] [Google Scholar]
- 47.Chang Q, Hoefs S, van der Kemp AW, Topala CN, Bindels RJ, Hoenderop JG. The beta-glucuronidase klotho hydrolyzes and activates the TRPV5 channel. Science. 2005;310:490–493. doi: 10.1126/science.1114245. [DOI] [PubMed] [Google Scholar]
- 48.Xuan NT, Hai NV. Changes in expression of klotho affect physiological processes, diseases, and cancer. Iran J Basic Med Sci. 2018;21:3–8. [PMC free article] [PubMed] [Google Scholar]
- 49.Cordeiro S, Strauss O. Expression of Orai genes and I(CRAC) activation in the human retinal pigment epithelium. Graefes Arch Clin Exp Ophthalmol. 2011;249:47–54. doi: 10.1007/s00417-010-1445-3. [DOI] [PubMed] [Google Scholar]
- 50.Kennedy BG, Torabi AJ, Kurzawa R, Echtenkamp SF, Mangini NJ. Expression of transient receptor potential vanilloid channels TRPV5 and TRPV6 in retinal pigment epithelium. Mol Vis. 2010;16:665–675. [PMC free article] [PubMed] [Google Scholar]
- 51.Kwak N, Okamoto N, Wood JM, Campochiaro PA. VEGF is major stimulator in model of choroidal neovascularization. Invest Ophthalmol Vis Sci. 2000;41:3158–3164. [PubMed] [Google Scholar]
- 52.Miller JW, Le Couter J, Strauss EC, Ferrara N. Vascular endothelial growth factor a in intraocular vascular disease. Ophthalmology. 2013;120:106–114. doi: 10.1016/j.ophtha.2012.07.038. [DOI] [PubMed] [Google Scholar]
- 53.Wen X, Li S, Zhang Y, Zhu L, Xi X, Zhang S, Li Y. Recombinant human klotho protects against hydrogen peroxide-mediated injury in human retinal pigment epithelial cells via the PI3K/Akt-Nrf2/HO-1 signaling pathway. Bioengineered. 2022;13:11767–11781. doi: 10.1080/21655979.2022.2071023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ji B, Wei H, Ding Y, Liang H, Yao L, Wang H, Qu H, Deng H. Protective potential of klotho protein on diabetic retinopathy: Evidence from clinical and in vitro studies. J Diabetes Investig. 2020;11:162–169. doi: 10.1111/jdi.13100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yang S, Zhou J, Li D. Functions and Diseases of the Retinal Pigment Epithelium. Front Pharmacol. 2021;12:727870. doi: 10.3389/fphar.2021.727870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ahoor MH, Ghorbanihaghjo A, Sorkhabi R, Kiavar A. Klotho and Endothelin-1 in Pseudoexfoliation Syndrome and Glaucoma. J Glaucoma. 2016;25:919–922. doi: 10.1097/IJG.0000000000000553. [DOI] [PubMed] [Google Scholar]
- 57.Ma Z, Liu J, Li J, Jiang H, Kong J. Klotho Levels are Decreased and Associated with Enhanced Oxidative Stress and Inflammation in the Aqueous Humor in Patients with Exudative Age-related Macular Degeneration. Ocul Immunol Inflamm. 2022;30:630–637. doi: 10.1080/09273948.2020.1828488. [DOI] [PubMed] [Google Scholar]
- 58.Tokuc EO, Yuksel N, Kır HM, Acar E. Evaluation of serum and aqueous humor klotho levels in pseudoexfoliation syndrome, pseudoexfoliation and primary open-angle glaucoma. Int Ophthalmol. 2021;41:2369–2375. doi: 10.1007/s10792-021-01790-5. [DOI] [PubMed] [Google Scholar]
- 59.Yamamoto K, Sato K, Yukita M, Yasuda M, Omodaka K, Ryu M, Fujita K, Nishiguchi KM, Machida S, Nakazawa T. The neuroprotective effect of latanoprost acts via klotho-mediated suppression of calpain activation after optic nerve transection. J Neurochem. 2017;140:495–508. doi: 10.1111/jnc.13902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Corcillo A, Fountoulakis N, Sohal A, Farrow F, Ayis S, Karalliedde J. Low levels of circulating anti-ageing hormone Klotho predict the onset and progression of diabetic retinopathy. Diab Vasc Dis Res. 2020;17:1479164120970901. doi: 10.1177/1479164120970901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Słomiński B, Ryba-Stanisławowska M, Skrzypkowska M, Myśliwska J, Myśliwiec M. The KL-VS polymorphism of KLOTHO gene is protective against retinopathy incidence in patients with type 1 diabetes. Biochim Biophys Acta Mol Basis Dis. 2018;1864:758–763. doi: 10.1016/j.bbadis.2017.12.015. [DOI] [PubMed] [Google Scholar]
- 62.Rübsam A, Parikh S, Fort PE. Role of Inflammation in Diabetic Retinopathy. Int J Mol Sci. 2018;19 doi: 10.3390/ijms19040942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Semeraro F, Cancarini A, dell'Omo R, Rezzola S, Romano MR, Costagliola C. Diabetic Retinopathy: Vascular and Inflammatory Disease. J Diabetes Res. 2015;2015:582060. doi: 10.1155/2015/582060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Chang ML, Chiu CJ, Shang F, Taylor A. High glucose activates ChREBP-mediated HIF-1α and VEGF expression in human RPE cells under normoxia. Adv Exp Med Biol. 2014;801:609–621. doi: 10.1007/978-1-4614-3209-8_77. [DOI] [PubMed] [Google Scholar]
- 65.Xiao Q, Zeng S, Ling S, Lv M. Up-regulation of HIF-1alpha and VEGF expression by elevated glucose concentration and hypoxia in cultured human retinal pigment epithelial cells. J Huazhong Univ Sci Technolog Med Sci. 2006;26:463–465. doi: 10.1007/s11596-006-0422-x. [DOI] [PubMed] [Google Scholar]
- 66.Xie L, Wang Y, Li Q, Ji X, Tu Y, Du S, Lou H, Zeng X, Zhu L, Zhang J, Zhu M. The HIF-1α/p53/miRNA-34a/Klotho axis in retinal pigment epithelial cells promotes subretinal fibrosis and exacerbates choroidal neovascularization. J Cell Mol Med. 2021;25:1700–1711. doi: 10.1111/jcmm.16272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Arroba AI, Campos-Caro A, Aguilar-Diosdado M, Valverde ÁM. IGF-1, Inflammation and Retinal Degeneration: A Close Network. Front Aging Neurosci. 2018;10:203. doi: 10.3389/fnagi.2018.00203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Raman P, Singal AK, Behl A. Effect of Insulin-Like Growth Factor-1 on Diabetic Retinopathy in Pubertal Age Patients With Type 1 Diabetes. Asia Pac J Ophthalmol (Phila) 2019;8:319–323. doi: 10.1097/APO.0000000000000250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wu TE, Chen HS. The role of growth hormone and IGF-1 in retinopathy: a prospective study of retinopathy in patients with acromegaly and impaired fasting glucose. Diabetol Metab Syndr. 2022;14:38. doi: 10.1186/s13098-022-00806-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hong S, Huo H, Xu J, Liao K. Insulin-like growth factor-1 receptor signaling in 3T3-L1 adipocyte differentiation requires lipid rafts but not caveolae. Cell Death Differ. 2004;11:714–723. doi: 10.1038/sj.cdd.4401405. [DOI] [PubMed] [Google Scholar]
- 71.Martins AS, Ordóñez JL, Amaral AT, Prins F, Floris G, Debiec-Rychter M, Hogendoorn PC, de Alava E. IGF1R signaling in Ewing sarcoma is shaped by clathrin-/caveolin-dependent endocytosis. PLoS One. 2011;6:e19846. doi: 10.1371/journal.pone.0019846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Puddu A, Sanguineti R, Maggi D. Caveolin-1 Down-Regulation Reduces VEGF-A Secretion Induced by IGF-1 in ARPE-19 Cells. Life (Basel) 2021;12 doi: 10.3390/life12010044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Salani B, Briatore L, Garibaldi S, Cordera R, Maggi D. Caveolin-1 down-regulation inhibits insulin-like growth factor-I receptor signal transduction in H9C2 rat cardiomyoblasts. Endocrinology. 2008;149:461–465. doi: 10.1210/en.2007-0312. [DOI] [PubMed] [Google Scholar]
- 74.Salani B, Passalacqua M, Maffioli S, Briatore L, Hamoudane M, Contini P, Cordera R, Maggi D. IGF-IR internalizes with Caveolin-1 and PTRF/Cavin in HaCat cells. PLoS One. 2010;5:e14157. doi: 10.1371/journal.pone.0014157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yu Y, Bao Z, Wang X, Gong W, Chen H, Guan H, Le Y, Su S, Chen K, Wang JM. The G-Protein-Coupled Chemoattractant Receptor Fpr2 Exacerbates High Glucose-Mediated Proinflammatory Responses of Müller Glial Cells. Front Immunol. 2017;8:1852. doi: 10.3389/fimmu.2017.01852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Han X, Cai C, Xiao Z, Quarles LD. FGF23 induced left ventricular hypertrophy mediated by FGFR4 signaling in the myocardium is attenuated by soluble Klotho in mice. J Mol Cell Cardiol. 2020;138:66–74. doi: 10.1016/j.yjmcc.2019.11.149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yanucil C, Kentrup D, Campos I, Czaya B, Heitman K, Westbrook D, Osis G, Grabner A, Wende AR, Vallejo J, Wacker MJ, Navarro-Garcia JA, Ruiz-Hurtado G, Zhang F, Song Y, Linhardt RJ, White K, Kapiloff MS, Faul C. Soluble α-klotho and heparin modulate the pathologic cardiac actions of fibroblast growth factor 23 in chronic kidney disease. Kidney Int. 2022;102:261–279. doi: 10.1016/j.kint.2022.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gu C, Lhamo T, Zou C, Zhou C, Su T, Draga D, Luo D, Zheng Z, Yin L, Qiu Q. Comprehensive analysis of angiogenesis-related genes and pathways in early diabetic retinopathy. BMC Med Genomics. 2020;13:142. doi: 10.1186/s12920-020-00799-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hueber A, Wiedemann P, Esser P, Heimann K. Basic fibroblast growth factor mRNA, bFGF peptide and FGF receptor in epiretinal membranes of intraocular proliferative disorders (PVR and PDR) Int Ophthalmol. 20:345–350. doi: 10.1007/BF00176889. [DOI] [PubMed] [Google Scholar]
- 80.Glick D, Barth S, Macleod KF. Autophagy: cellular and molecular mechanisms. J Pathol. 2010;221:3–12. doi: 10.1002/path.2697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lei L, Tzekov R, Li H, McDowell JH, Gao G, Smith WC, Tang S, Kaushal S. Inhibition or Stimulation of Autophagy Affects Early Formation of Lipofuscin-Like Autofluorescence in the Retinal Pigment Epithelium Cell. Int J Mol Sci. 2017;18 doi: 10.3390/ijms18040728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Mathew B, Chennakesavalu M, Sharma M, Torres LA, Stelman CR, Tran S, Patel R, Burg N, Salkovski M, Kadzielawa K, Seiler F, Aldrich LN, Roth S. Autophagy and post-ischemic conditioning in retinal ischemia. Autophagy. 2021;17:1479–1499. doi: 10.1080/15548627.2020.1767371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Villarejo-Zori B, Jiménez-Loygorri JI, Zapata-Muñoz J, Bell K, Boya P. New insights into the role of autophagy in retinal and eye diseases. Mol Aspects Med. 2021;82:101038. doi: 10.1016/j.mam.2021.101038. [DOI] [PubMed] [Google Scholar]
- 84.Dehdashtian E, Mehrzadi S, Yousefi B, Hosseinzadeh A, Reiter RJ, Safa M, Ghaznavi H, Naseripour M. Diabetic retinopathy pathogenesis and the ameliorating effects of melatonin; involvement of autophagy, inflammation and oxidative stress. Life Sci. 2018;193:20–33. doi: 10.1016/j.lfs.2017.12.001. [DOI] [PubMed] [Google Scholar]
- 85.Lopes de Faria JM, Duarte DA, Montemurro C, Papadimitriou A, Consonni SR, Lopes de Faria JB. Defective Autophagy in Diabetic Retinopathy. Invest Ophthalmol Vis Sci. 2016;57:4356–4366. doi: 10.1167/iovs.16-19197. [DOI] [PubMed] [Google Scholar]
- 86.Xue M, Yang F, Le Y, Yang Y, Wang B, Jia Y, Zheng Z, Xue Y. Klotho protects against diabetic kidney disease via AMPK- and ERK-mediated autophagy. Acta Diabetol. 2021;58:1413–1423. doi: 10.1007/s00592-021-01736-4. [DOI] [PubMed] [Google Scholar]
- 87.Zhou H, Pu S, Zhou H, Guo Y. Klotho as Potential Autophagy Regulator and Therapeutic Target. Front Pharmacol. 2021;12:755366. doi: 10.3389/fphar.2021.755366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Gao D, Zuo Z, Tian J, Ali Q, Lin Y, Lei H, Sun Z. Activation of SIRT1 Attenuates Klotho Deficiency-Induced Arterial Stiffness and Hypertension by Enhancing AMP-Activated Protein Kinase Activity. Hypertension. 2016;68:1191–1199. doi: 10.1161/HYPERTENSIONAHA.116.07709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kim JY, Mondaca-Ruff D, Singh S, Wang Y. SIRT1 and Autophagy: Implications in Endocrine Disorders. Front Endocrinol (Lausanne) 2022;13:930919. doi: 10.3389/fendo.2022.930919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Adu-Agyeiwaah Y, Vieira CP, Asare-Bediako B, Li Calzi S, DuPont M, Floyd J, Boye S, Chiodo V, Busik JV, Grant MB. Intravitreal Administration of AAV2-SIRT1 Reverses Diabetic Retinopathy in a Mouse Model of Type 2 Diabetes. Transl Vis Sci Technol. 2023;12:20. doi: 10.1167/tvst.12.4.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Nawaz IM, Rezzola S, Cancarini A, Russo A, Costagliola C, Semeraro F, Presta M. Human vitreous in proliferative diabetic retinopathy: Characterization and translational implications. Prog Retin Eye Res. 2019;72:100756. doi: 10.1016/j.preteyeres.2019.03.002. [DOI] [PubMed] [Google Scholar]
- 92.Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15:178–196. doi: 10.1038/nrm3758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Che D, Zhou T, Lan Y, Xie J, Gong H, Li C, Feng J, Hong H, Qi W, Ma C, Wu Q, Yang X, Gao G. High glucose-induced epithelial-mesenchymal transition contributes to the upregulation of fibrogenic factors in retinal pigment epithelial cells. Int J Mol Med. 2016;38:1815–1822. doi: 10.3892/ijmm.2016.2768. [DOI] [PubMed] [Google Scholar]
- 94.You ZP, Chen SS, Yang ZY, Li SR, Xiong F, Liu T, Fu SH. GEP100/ARF6 regulates VEGFR2 signaling to facilitate high-glucose-induced epithelial-mesenchymal transition and cell permeability in retinal pigment epithelial cells. Am J Physiol Cell Physiol. 2019;316:C782–C791. doi: 10.1152/ajpcell.00312.2018. [DOI] [PubMed] [Google Scholar]
- 95.Li X, Lu P, Shao XF, Jiang T, Liu F, Li G. Klotho Regulates Epithelial-to-Mesenchymal Transition In Vitro via Wnt/β-Catenin Pathway and Attenuates Chronic Allograft Dysfunction in a Rat Renal Transplant Model. Ann Transplant. 2021;26:e930066. doi: 10.12659/AOT.930066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yang Z, Zhan YW, Huang YY, Huang W, Zhan F, Lin SD. Regulation of epithelial mesenchymal transition by the renin-angiotensin system: a role for klotho in renal tubular epithelial cells. J Biol Regul Homeost Agents. 2020;34:57–67. doi: 10.23812/19-410-A-27. [DOI] [PubMed] [Google Scholar]
- 97.Kowluru RA, Mishra M, Kowluru A, Kumar B. Hyperlipidemia and the development of diabetic retinopathy: Comparison between type 1 and type 2 animal models. Metabolism. 2016;65:1570–1581. doi: 10.1016/j.metabol.2016.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Kumar B, Kowluru A, Kowluru RA. Lipotoxicity augments glucotoxicity-induced mitochondrial damage in the development of diabetic retinopathy. Invest Ophthalmol Vis Sci. 2015;56:2985–2992. doi: 10.1167/iovs.15-16466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Saeedi P, Petersohn I, Salpea P, Malanda B, Karuranga S, Unwin N, Colagiuri S, Guariguata L, Motala AA, Ogurtsova K, Shaw JE, Bright D, Williams R IDF Diabetes Atlas Committee. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract. 2019;157:107843. doi: 10.1016/j.diabres.2019.107843. [DOI] [PubMed] [Google Scholar]
- 100.Gonzalez-Cortes JH, Martinez-Pacheco VA, Gonzalez-Cantu JE, Bilgic A, de Ribot FM, Sudhalkar A, Mohamed-Hamsho J, Kodjikian L, Mathis T. Current Treatments and Innovations in Diabetic Retinopathy and Diabetic Macular Edema. Pharmaceutics. 2022;15 doi: 10.3390/pharmaceutics15010122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Xia HQ, Yang JR, Zhang KX, Dong RL, Yuan H, Wang YC, Zhou H, Li XM. Molecules related to diabetic retinopathy in the vitreous and involved pathways. Int J Ophthalmol. 2022;15:1180–1189. doi: 10.18240/ijo.2022.07.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Muniyandi A, Hartman GD, Song Y, Mijit M, Kelley MR, Corson TW. Beyond VEGF: targeting inflammation and other pathways for treatment of retinal disease. J Pharmacol Exp Ther. 2023;386:15–25. doi: 10.1124/jpet.122.001563. [DOI] [PMC free article] [PubMed] [Google Scholar]