Abstract
Purpose
Even though there isn't enough clinical evidence to demonstrate that robot-assisted radical cystectomy (RARC) is preferable to open radical cystectomy (ORC), RARC has become a widely used alternative. We performed the present study of RARC vs ORC with a focus on oncologic, pathological, perioperative, and complication-related outcomes and health-related quality of life (QOL).
Methods
We conducted a literature review up to August 2022. The search included PubMed, EMBASE and Cochrane controlled trials register databases. We classified the studies according to version 2 of the Cochrane risk-of-bias tool for randomized trials (RoB 2). The data was assessed by Review Manager 5.4.0.
Results
8 RCTs comparing 1024 patients were analyzed in our study. RARC was related to lower estimated blood loss (weighted mean difference (WMD): -328.2; 95% CI -463.49—-192.92; p < 0.00001), lower blood transfusion rates (OR: 0.45; 95% CI 0.32 – 0.65; p < 0.0001) but longer operation time (WMD: 84.21; 95% CI 46.20 -121.72; p < 0.0001). And we found no significant difference in terms of positive surgical margins (P = 0.97), lymph node yield (P = 0.30) and length of stay (P = 0.99). Moreover, no significant difference was found between the two groups in terms of survival outcomes, pathological outcomes, postoperative complication outcomes and health-related QOL.
Conclusion
Based on the present evidence, we demonstrated that RARC and ORC have similar cancer control results. RARC is related to less blood loss and lower transfusion rate. We found no difference in postoperative complications and health-related QOL between robotic and open approaches. RARC procedures could be used as an alternate treatment for bladder cancer patients. Additional RCTs with long-term follow-up are needed to validate this observation.
Keywords: Bladder cancer, Robot-assisted radical cystectomy, Open radical cystectomy, Randomized controlled trial, Meta-analysis
Introduction
With more than half a million new cases annually, bladder cancer has become one of the ten most prevalent kinds of cancer in the world [1, 2]. Open radical cystectomy (ORC) is still the advised surgical procedure for patients with muscle-invasive bladder cancer and those with very high-risk non-muscle-invasive bladder cancer [3]. The value of ORC, however, has been limited because of the drawbacks of a high morbidity and mortality, such as urinary tract infection, urinary leak, renal failure, ileus and thromboembolic complications. More than 60% of patients receiving ORC undoubtedly have at least one perioperative problem, and 20% have a high-grade complication after their procedure [4, 5]. Radical cystectomy has been performed using minimally invasive surgical techniques over the past two decades [6]. Menon et al. reported the first robot-assisted radical cystectomy (RARC) twenty years ago [7], which has become common technology around the world, especially in many high-volume locations. Initially, a tiny incision was made in the abdominal wall to execute urinary diversion by extracorporeal procedure. The development of intracorporeal urinary diversion (ICUD) has, however, been made possible by technological adjustments made to RARC. [8]. From 2005 to 2016, the use of ICUD grew from 9 to 97% of urinary diversion surgeries [9].
Several studies have indicated that compared with ORC, RARC has fewer perioperative complications, shorter length of stay and lower estimated blood loss [10–12]. Nevertheless, very few studies have revealed the long-term oncological results, and the majority of investigations are retrospective. Randomized controlled trials (RCTs) remain crucial for establishing how RARC and ORC compare to one another. Recently, a sizable quantity of fresh data from RCTs emerged that included long-term oncological outcomes, adding new evidence to the theme [13–16].
To assess the oncologic, pathological, perioperative, postoperative complications outcomes as well as health-related quality of life (QOL), for RCTs comparing RARC with ORC, we carried out this meta-analysis to update the current evidence base.
Materials and methods
The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) Statement [17] was followed when conducting this study. As no primary personal information will be gathered, no extra ethical approval is necessary.
Literature search
The research was registered on PROSPERO (CRD42023396105). Two reviewers independently searched MEDLINE (2009 to August 2022), EMBASE (1995 to August 2022), and the Cochrane Controlled Trials Register databases for appropriate RCTs comparing RARC with ORC. Moreover, references from retrieved studies were searched. The following keywords were applied, such as “bladder cancer”, “cystectomy”, “robot”, “robotic”, “da Vinci”, “ORC”, “RARC”, and “randomized controlled trials”.
Inclusion criteria and selection of studies
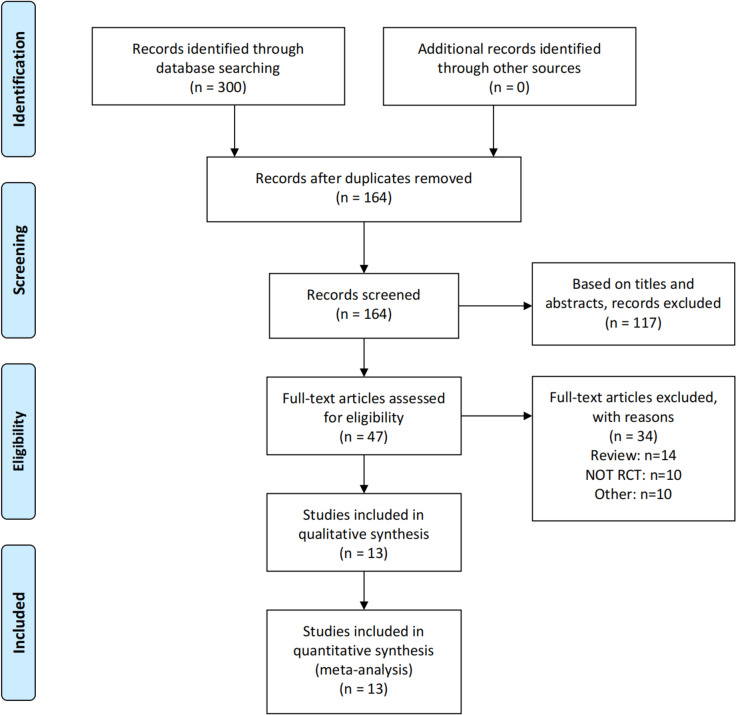
Studies were included if they conformed to the following criteria: (1) comparison of ORC with RARC; (2) the study provided analyzable data of interest: overall survival (OS), recurrence-free survival (RFS), oncologic efficacy (positive margin status [PSM], lymph node yield, sites of recurrence) and perioperative outcomes (estimated blood loss [EBL], blood transfusion rates, operating room time [ORT], length of stay [LOS]), postoperative complications and health-related QOL assessment; (3) The article's whole text was accessible. We included either the more current or higher-quality patient-cohort article when there were equivalent papers. If the same group performed numerous experiments on a similar set of participants, we included each study. A flowchart of the study selection process is presented in Fig. 1.
Fig. 1.
A flow diagram of the study selection. RCT: randomized controlled trial
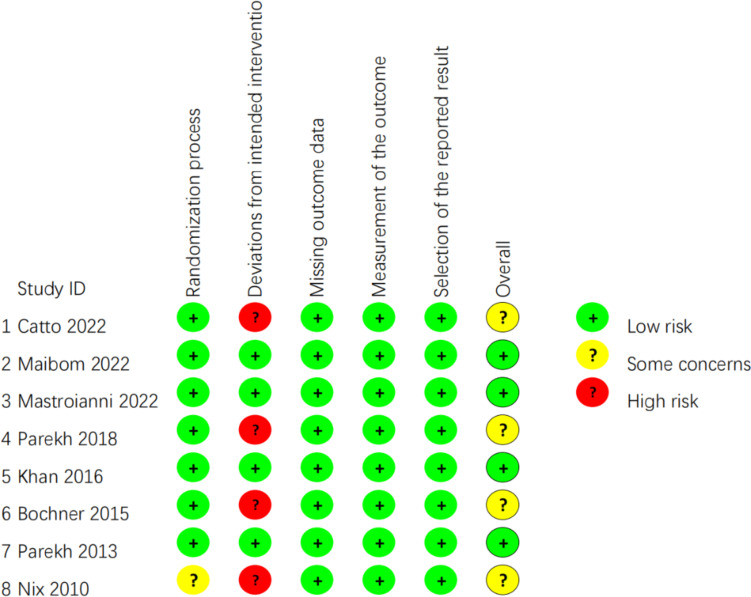
Risk of bias assessment
Using version 2 of the Cochrane risk-of-bias tool for randomized trials (RoB 2) [18], we classified the included studies. These studies were classified into three degrees: low risk of bias, some concerns, or high risk of bias. The writers came to an agreement on certain points where they disagreed.
Data extraction
We extracted the required data: (1) the first author’s name and the published time of the article; (2) the region of each RCT; (3) study design; (4) treatment and sample size; (5) age, sex, body mass index (BMI) and American Society of Anesthesiologists (ASA) score; (6) tumor stage and urinary diversion type; (7) overall survival (OS) rate, recurrence-free survival (RFS) rate and sites of recurrence; (8) PSM and lymph node yield; (9) blood transfusion rates, ORT, EBL and LOS; (10) the date of postoperative complications and health-related QOL assessment.
Statistical analysis
This study used Review Manager Version 5.4 (Cochrane Collaboration, London, UK) to evaluate comparable data. Continuous results were estimated using weighted mean difference (WMD) with 95% confidence intervals (CIs). Besides, the odds ratio (OR) was used to estimate dichotomous variables. We used 95% CIs to express all outcomes, and statistical significance was set as p < 0.05. The degree of heterogeneity was calculated by Cochrane’s Q test and I2 test. If there was significant inconsistency (I2 > 50% or p < 0.10), we chose a random effect model for meta-analysis; if not, a fixed effect model was applied.
Results
Study characteristics
Overall, 300 potentially relevant articles were found by our initial literature search; 136 duplicates were removed. Furthermore, 117 and 34 publications were excluded after evaluating the title/abstract and reading the full-text, respectively. And 13 studies (8 RCTs) [4, 14–16, 19–27] involving 1024 participants were studied in the meta-analysis. The basic features of the included RCTs are listed in Table 1.
Table 1.
Characteristics of the included studies
| Study | Region | Group | Number of patients | Age, years (median or mean) |
Sex (M/F) | BMI (mean or median) |
ASA score (mean or median) | Tumor stage (T1 or lower) /T2/T3/T4 |
Diversion type ileal conduit/Neobladder/ continent cutaneous | Rate of neobladder (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Catto et al., 2022 [14] | UK | RARC/ORC | 161/156 | 69.3/68.7 | 128/33; 122/34 | NA | NA | 71/30/NA/NA 70/34/NA/NA | 142/NA/NA; 140/NA/NA | NA |
| Vejlgaard et al., 2022 [20] | Denmark | RARC/ORC | 25/25 | 70/67 | 18/7; 20/5 | 27.3/26.9 | 2.12/2 | 9/13/3/NA 6/15/4/NA | 25/0/0; 25/0/0 | 0/0 |
| Maibom et al., 2022 [16] | Denmark | RARC/ORC | 25/25 | 70/67 | 18/7; 20/5 | 27.3/26.9 | 2.12/2 | 9/13/3/NA 6/15/4/NA | 25/0/0; 25/0/0 | 0/0 |
| Mastroianni et al., 2022 [15] | Italy | RARC/ORC | 58/58 | 64/66 | 44/14; 40/18 | 26/26 | 2.19/2.07 | 11/45/2/0; 12/44/2/0 | NA/46/0; NA/42/NA | 79/72 |
| Khan et al., 2020 [21] | UK | RARC/ORC | 20/20 | 68/68 | 17/3; 18/2 | 27.50/26.99 | 1.85/1.85 | 11/3/6/0; 14/0/4/2 | 18/2/0; 17/3/0 | 10/15 |
| Venkatramani et al., 2020 [22] | USA | RARC/ORC | 150/152 | 70/67 | 126/24; 128/24 | 27.8/28.2 | NA | 48/82/6/4; 51/81/16/4 | 113/36/1; 122/30/0 | 24/20 |
| Bochner et al., 2018 [23] | USA | RARC/ORC | 60/58 | 66/65 | 51/9; 42/16 | 27.9/29 | 2.73/2.84 | 35/8/12/5; 32/7/15/4 | 27/33/0; 23/32/3 | 55/55 |
| Parekh et al., 2018 [4] | USA | RARC/ORC | 150/152 | 70/67 | 126/24; 128/24 | 27.8/28.2 | NA | 48/82/6/4; 51/81/16/4 | 113/36/1; 122/30/0 | 24/20 |
| Khan et al., 2016 [24] | UK | RARC/ORC | 20/20 | 68.6/66.6 | 17/3; 18/2 | 27.5/27.4 | 1.85/1.85 | 11/3/6/0; 14/0/4/2 | 18/2/0; 17/3/0 | 10/15 |
| Bochner et al., 2015 [25] | USA | RARC/ORC | 60/58 | 66/65 | 51/9; 42/16 | 27.9/29 | 2.73/2.84 | 35/8/12/5; 32/7/15/4 | 27/33/0; 23/32/3 | 55/55 |
| Messer et al., 2014 [26] | USA | RARC/ORC | 20/20 | 69.5/64.5 | 18/2; 16/4 | 27.6/28.3 | 3.0/3.0 | 7/3/3/7; 12/1/2/5 | 19/1/0; 18/2/0 | 5/10 |
| Parekh et al., 2013 [27] | USA | RARC/ORC | 20/20 | 69.5/64.5 | 18/2; 16/4 | 27.6/28.3 | 3.0/3.0 | 7/3/3/7; 12/1/2/5 | 19/1/0; 18/2/0 | 5/10 |
| Nix et al., 2010 [19] | USA | RARC/ORC | 21/20 | 67.4/69.2 | 14/7; 17/3 | 27.5/28.4 | 2.71/2.70 | 6/12/3/0; 5/14/1/0 | 14/7/0; 14/6/0 | 33/30 |
BMI Body mass index, ASA American Society of Anesthesiologists physical status classification, RARC Robot-assisted radical cystectomy, ORC Open radical cystectomy, NA Not available
Risk of bias
Each study included in the meta‑analysis was RCT. According to RoB 2 [18], the majority of the listed RCTs were categorized as “low risk of bias” or “some concerns”. The bias of quality assessment is shown in Fig. 2.
Fig. 2.
Quality assessment of each study
Oncologic outcomes
The oncologic outcomes evaluated in this meta-analysis included overall survival, recurrence-free survival and recurrence patterns.
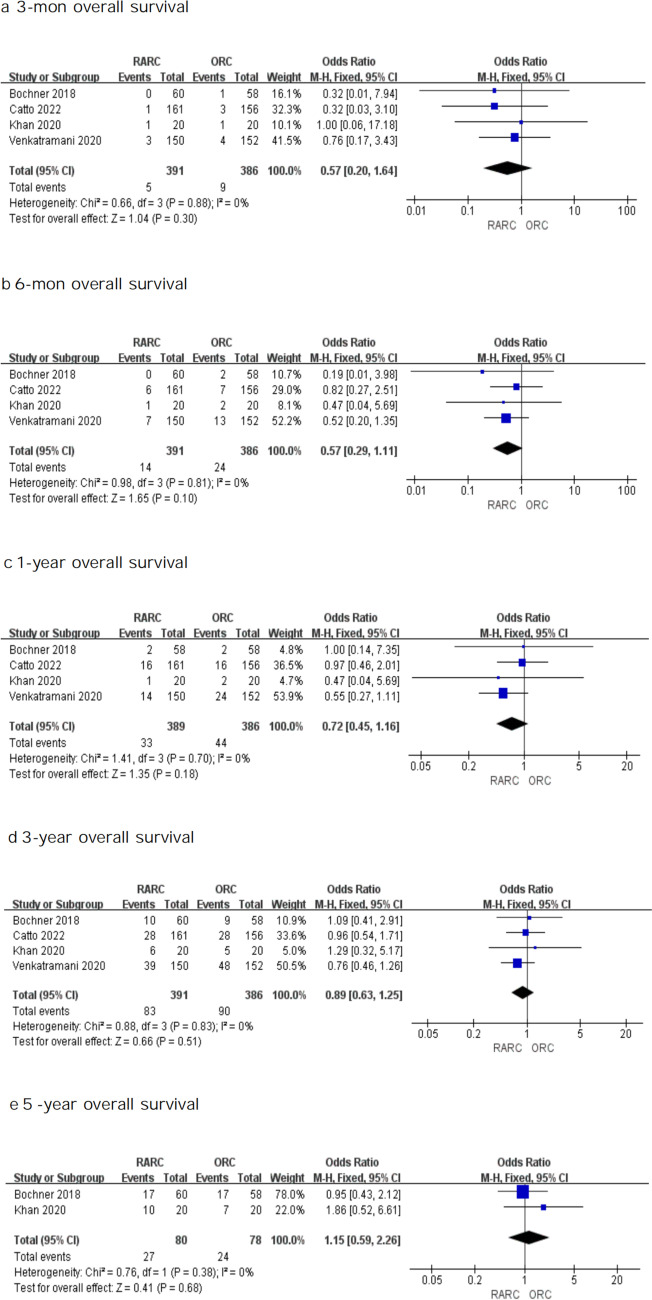
Overall survival
Four RCTs assessed OS between RARC and ORC. As seen in Fig. 3, the OS analysis in bladder cancer suggested no significant difference in survival rates between the two approaches at 3-mon (OR: 0.57; 95% CI 0.20 – 1.64; P = 0.30), 6-mon (OR: 0.57; 95% CI 0.29 – 1.11; P = 0.10), 1-year (OR: 0.72; 95% CI 0.45 – 1.16; P = 0.18), 3-year (OR: 0.89, 95% CI 0.63—1.25; P = 0.51) and 5-year (OR: 1.15, 95% CI 0.59—2.26; P = 0.68) follow-up. This result indicated that when compared with the ORC, the RARC group had equivalent effectiveness when it came to overall survival.
Fig. 3.
Forest plots showing (a) 3-month overall survival, (b) 6-month overall survival, (c) 1-year overall survival, (d) 3-year overall survival, and (e) 5-year overall survival in RARC group and ORC group. MH mantel–haenszel, CI confidence interval, df degrees of freedom
Recurrence-free survival
In order to determine recurrence-free survival, four RCTs were examined. There was little significant difference between the two approaches for the 3-month (OR: 0.43, 95% CI 0.17—1.10; P = 0.08), 6-mon (OR: 0.58, 95% CI 0.32—1.03; P = 0.06), 1-year (OR: 0.89, 95% CI 0.60—1.33; P = 0.58), 3-year (OR: 0.97, 95% CI 0.70—1.34; P = 0.86) and 5-year (OR: 0.82, 95% CI 0.43—1.56; P = 0.54) recurrence-free survival as shown in Fig. 4, which indicated that individuals who experienced RARC or ORC had similar recurrence-free survival.
Fig. 4.
Forest plots showing (a) 3-month recurrence-free survival, (b) 6-month recurrence-free survival, (c) 1-year recurrence-free survival, (d) 3-year recurrence-free survival, and (e) 5-year recurrence-free survival in RARC group and ORC group. MH mantel–haenszel, CI confidence interval, df degrees of freedom
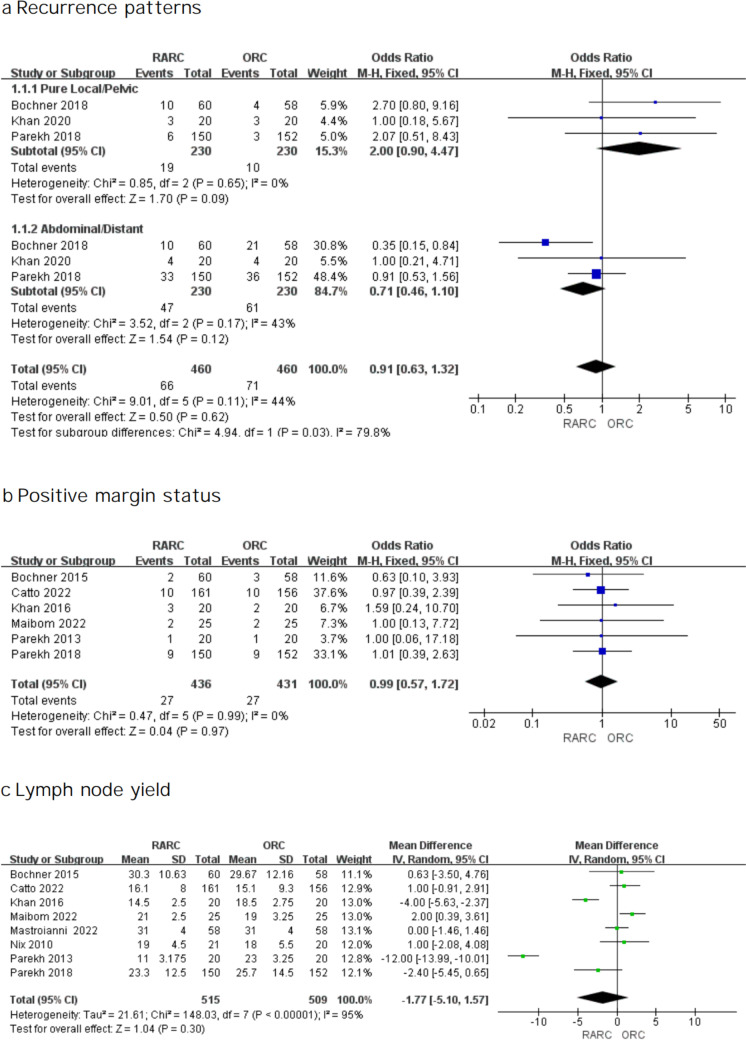
Recurrence patterns
Three RCTs were analyzed in terms of reported local (pelvic) and abdominal/distant recurrence data. Among the studies, no high risk of heterogeneity was found, so a fixed effects model was selected. As shown in Fig. 5a, we found neither RARC nor ORC was related to a substantially greater chance of a local or abdominal/distant recurrence. Nevertheless, regarding recurrence patterns, a significant difference was found between the two approaches (test for subgroup differences, P = 0.03).
Fig. 5.
Forest plots showing (a) recurrence patterns, (b) positive margin status, and (c) lymph node yield
Pathological outcomes
Six investigations including 867 participants reported PSM rates. The heterogeneity test suggested I2 = 0%, so we used a fixed effect model. No difference in PSM was found (OR: 0.99; 95% CI 0.57—1.72; P = 0.97 Fig. 5b). Likewise, no significant difference was found in terms of lymph node yield (WMD: -1.77; 95% CI -5.10 -1.57; P = 0.30 Fig. 5c).
Perioperative outcomes
Intraoperative outcomes: operative time, EBL and blood transfusion rate
Operative time
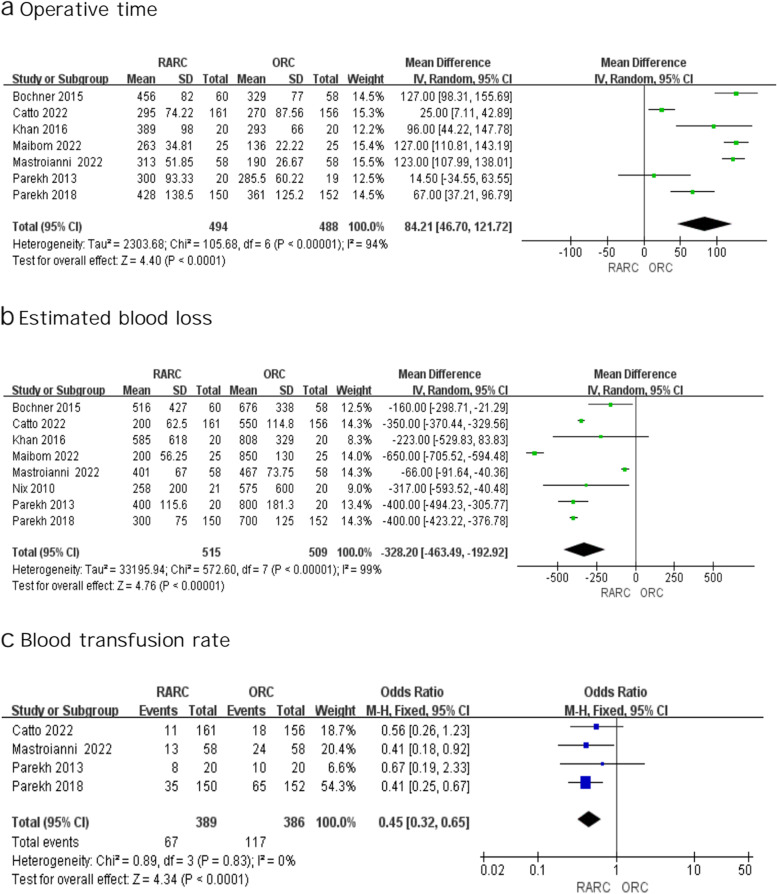
High heterogeneity was identified between studies (I2 = 94%), so we conducted the analysis using a random effect model. As shown in Fig. 6a, the forest plots indicated that RARC was related to longer operative time (WMD: 84.21; 95% CI 46.70 -121.72; p < 0.0001 Fig. 6a).
Fig. 6.
Forest plots showing (a) operative time, (b) estimated blood loss, and (c) blood transfusion rate
Estimated blood loss
Eight investigations showed the EBL [4, 14–16, 19, 24, 25, 27]. A random effect model was applied according to the findings of the heterogeneity test (I2 = 99%). Data from 1024 patients revealed that the EBL of RARC group was much lower than that of the ORC group. (WMD: -328.2; 95% CI -463.49—-192.92; p < 0.00001 Fig. 6b).
Blood transfusion rate
No high risk of heterogeneity was found, so we used a fixed effects model. Blood transfusion rates were found to be lower in RARC (OR: 0.45; 95% CI 0.32 – 0.65; p < 0.0001 Fig. 6c).
Analysis of postoperative complications
We evaluated postoperative complications within 30 days and 90 days following surgery. As for the postoperative complication grades of RARC and ORC within 30 days after surgery, the analysis did not identify any significant difference: any Clavien–Dindo grade (OR: 0.71; 95% CI 0.39 – 1.29; P = 0.26 Fig. 7a), Clavien–Dindo grade I–II (OR: 1.17; 95% CI 0.54 – 2.55; P = 0.69 Fig. 7b), Clavien–Dindo grade III–IV (OR: 1.09; 95% CI 0.48 – 2.46; P = 0.84 Fig. 7c). Likewise, we observed no significant difference regarding postoperative complication within 90 days between both groups: any Clavien–Dindo grade (OR: 0.84; 95% CI 0.63 – 1.12; P = 0.24 Fig. 7d), Clavien–Dindo grade I–II (OR: 0.91; 95% CI 0.68 – 1.21; P = 0.50 Fig. 7e), Clavien–Dindo grade III–IV (OR: 0.92; 95% CI 0.66 –1.29; P = 0.64 Fig. 7f).
Fig. 7.
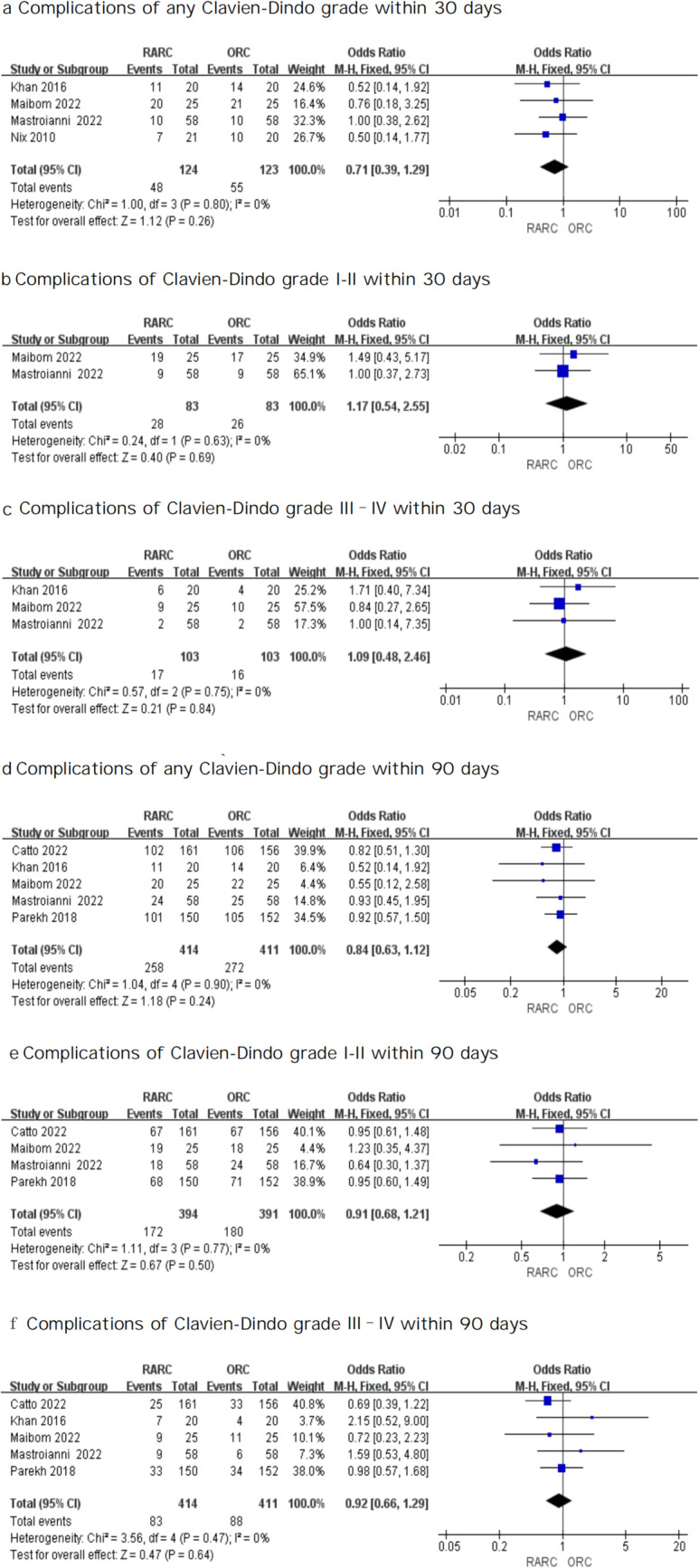
Forest plots showing (a) complications of any Clavien-Dindo grade within 30 days, (b) complications of Clavien-Dindo grade I-II within 30 days, (c) complications of Clavien-Dindo grade III–IV within 30 days, (d) complications of any Clavien-Dindo grade within 90 days, (e) complications of Clavien-Dindo grade I-II within 90 days, and (f) complications of Clavien-Dindo grade III–IV within 90 days
Length of stay
We adopted a random effects model because of high heterogeneity (I2 = 98%). Evaluation of combined data from seven trials found no significant difference in LOS between the two groups (WMD: -0.01; 95% CI -1.25—1.24; P = 0.99 Fig. 8).
Fig. 8.
Forest plots showing Length of stay
Postoperative health-related QOL outcomes
Although six RCTs reported postoperative health-related QOL (Table 2), different quality-of-life assessment tools were used. As a result, it might not be possible to conduct a pooled analysis of data. The FACT-VCI was adopted by Messer et al. [26] to assess changes in health-related QOL scores between the two approaches at baseline, 3, 6, 9 and 12 months following surgery in 40 patients. In order to compare health-related QOL at baseline, 3 and 6 months after surgery, Bochner et al. [25]and Parekh et al. [4] adopted the QLQ-C30 and FACT-VCI, respectively, while after a mean of 8 months after surgery, Khan et al. [24] completed the FACT-BI. Vejlgaard et al. [20] evaluated health-related QOL at baseline and 3 months after surgery using QLQ-C30. According to all research, health-related QOL of the two groups did not significantly differ from one another. Nevertheless, in a new trial, Catto et al. [14] used several measures in 317 participants to evaluate health-related QOL and concluded those who underwent robotic surgery showed better QOL vs open surgery at 5 weeks (difference in EQ-5D-5L scores, –0.07 [95% CI, –0.11 to –0.03]; P = 0.003) and less disability at 5 weeks (WHODAS 2.0 scores, 0.48 [95% CI, 0.15–0.73]; P = 0.003) and at 12 weeks (difference in WHODAS 2.0 scores, 0.38 [95% CI, 0.09–0.68]; P = 0.01). By 26 weeks, there was little difference in terms of QOL and disability scores.
Table 2.
Postoperative health-related QOL change
| Author | QOL scale | Arm | QOL score baseline | QOL score at 1 month | QOL score at 3 months | QOL score at 6 months | QOL score at 9 months | QOL score at 12 months |
QOL score overall |
|---|---|---|---|---|---|---|---|---|---|
| Catto 2022 [14] | EQ-5D-5L | RARC/ORC | 0.89/0.90 |
0.83/0.77 (P = 0.003) |
0.88/0.86 (P = 0.1) |
0.87/0.87 (P = 0.2) |
NR | NR | NR |
| QLQ-C30 | 87.55/87.74 |
76.46/68.66 (P < 0.001) |
86.06/81.96 (P = 0.01) |
87.14/84.86 (P = 0.17) |
NR | NR | NR | ||
| QLQ-BLM30 | NR | NR | NR | NR | NR | NR | NR | ||
| WHODAS-2 | 9.3/9.2 |
20.9/26.4 (P = 0.003) |
10.2/14.9 (P = 0.01) |
10.8/11.1 (P = 0.24) |
NR | NR | NR | ||
| Vejlgaard 2022 [20] | QLQ-C30 | RARC/ORC | 77/78 | NR |
84/77 (P = 0.47) |
NR | NR | NR | NR |
| Parekh 2018 [4] | FACT-VCI | RARC/ORC | 120.1/120.9 | NR | 122.8/125.2 | 126.0/127.5 | NR | NR | NR |
| Khan 2016 [24] | FACT-Bl | RARC/ORC | NR | NR | NR | NR | NR | NR | NR |
| Bochner 2015 [25] | QLQ-C30 | RARC/ORC | 78/75 | NR |
77/72 (P = 0.40) |
76/78 (P = 0.5) |
NR | NR | NR |
| Messer 2014 [26] | FACT-VCI | RARC/ORC | 119/135 | NR |
126.5/135.5 (P = 0.85) |
121.5/126 (P = 0.58) |
141.5/127.5 (P = 0.63) |
116/129 (P = 0.48) |
NR |
QOL Quality of life, RARC Robot-assisted radical cystectomy, ORC Open radical cystectomy, NA Not available
Discussion
Bladder cancer poses significant cumulative morbidity because of the advanced age and substantial smoking prevalence. Due to the intricacy of the procedure and the patients' intrinsic weakness, radical cystectomy—the recommended therapy for patients with very high-risk and muscle-invasive bladder cancer—is related to a significant prevalence of complications [1, 28]. Despite their greater cost and steeper learning curve, minimally invasive operations like RARC are being used in many areas of medicine [28, 29]. RARC has gained popularity in bladder cancer treatment because of its prospective benefits. According to reports, the percentage of cystectomies carried out with the RARC increased from 0.6% to 12.8% [30]. With a focus on the differences in results between RCTs, the purpose of the updated meta-analysis was to compare the most recent evidence on the differential influence of these two strategies on oncologic, perioperative, and health-related (QOL) outcomes as well as complication-related outcomes.
Here we report the largest RCT outcomes analysis between RARC and ORC, including 1024 patients from eight studies. As a malignant tumor, bladder cancer patients' long-term follow-up oncological outcomes are a major concern for surgeons. Our analysis of the existing literature showed equivalent oncologic results. Besides, we didn't observe a difference in recurrence patterns, OS or RFS in all RCTs, indicating that RARC is a safe procedure, which has long-term survival effectiveness for bladder cancer that is comparable to that of ORC.
These conclusions are based on hypothesis and could be accessed further in a meta-analysis of individual patient data according to properly considered and standard definitions of recurrence locations. PSM and lymph node yield have a significant effect on postoperative survival. We also came to a conclusion from our study that there were no significant differences in PSM and lymph node yield.
Consistent with the benefits of the robotic approach for other malignant neoplasms, we found that RARC was related to lower blood loss and lower transfusion rates at the expense of prolonged operative time in terms of perioperative safety [31]. RARC has significant advantages in controlling bleeding, which may be due to clearer operational vision, more elaborate manipulation and the use of hemostatic devices in robotic-assisted surgery. In addition, another major factor affecting bleeding is the pneumoperitoneum used in robot-assisted surgery. The increase of pressure is conducive to the occlusion of small blood vessels, reducing the amount of bleeding [32].
In terms of the complication rate within 30 and 90 days, we observed no difference between ORC and RARC. Fewer postoperative complications are a potential benefits of RARC [33–36]. Yet, the RARC's benefit may not always be apparent. Additionally, in the Clavien–Dindo subgroup analyses, no significance was found between the two groups regarding minor and major complications at 30 days and 90 days following surgery. Urinary tract infections are typical complications following radical cystectomy [37]. Urinary diversions, however, make it hard to prevent these consequences. Despite continual improvements in surgical methods, similar complications have happened on occasion. Thus, there remains a great need to study postoperative complications [38].
Urinary construction is a key factor in postoperative complications, which is a controversial subject, with intracorporeal and extracorporeal options to choose from. In this meta-analysis, there are only three RCTs performed ICUD method [14–16]. Hussein et al. [9] analyzed 2,125 patients with radical cystectomy and concluded that the postoperative complications of ICUD decreased over time. However, ICUD has intrinsic difficulties, such as a challenging learning curve, insufficient clinical experience of the surgeons, unreasonable operating times, and the possibility of undermining the quality of the uretero-ileal anastomoses outweighing its advantages [39, 40]. Further studies about the procedure of urinary construction are needed, and it is essential to improve the understanding and management of these serious and common adverse reactions.
HRQOL relates to the influence of illness and therapy on one's physical, mental, and social spheres as related to overall well-being [41], which is considered to be one of the important parameters after malignant tumor treatment [42, 43]. As several techniques were used to assess QOL, it was difficult to pool QOL data for our study. Five RCTs found no significant difference regarding health-related QOL. However, a new study comparing recovery after RARC with intracorporeal reconstruction vs ORC suggested that any statistically significant change at five weeks was in favour of robotic surgery in terms of postoperative health-related QOL outcomes [14]. This might be due to the change in urinary reconstruction and the large sample size in this study.
In this review, we also observed no significant difference in LOS, which reflect similar complication rates between the two procedures.
Compared with some existing meta-analyses [44–46], this study conducted the most comprehensive analysis of RCTs, both in terms of the number of RCTs included and the indicators analyzed. In addition to analyzing indicators such as PSM, EBL, blood transfer rates, ORT, LOS, etc. We also analyzed lymph node yield and health related QOL. We used more detailed methods to analyze survival indicators and postoperative complications, and conducted subgroup analysis of recurrence patterns. Nevertheless, several limitations cannot be avoided. First, a limited sample size of just 8 randomized controlled trials (1024 patients) raises concerns about the validity of our findings on effectiveness and safety. Next, the majority of the studies were not considered to use a blinding procedure for their participants, and thus there may be potential bias. The lack of information on postoperative complications is a disadvantage. Future research should follow criteria for evaluating and reporting of postoperative complications. Health-related QOL assessment tools differ significantly between eligible RCTs. In addition, most studies were performed at a single center, which is reflected in the results of the heterogeneity test of operative time and might reflect the experience of the individual surgeon. Furthermore, some included studies were carried out with enhanced recovery after surgery (ERAS) programmes. ERAS is a standardized, multimodal, and multidisciplinary scientific concept for perioperative management. ERAS aims to decrease intra-operative blood loss, decrease postoperative complications, and reduce recovery times. The main content of ERAS in urology includes admission assessment, preoperative preparation, intraoperative measures, and postoperative management. In particular, bleeding can be reduced by perioperative fluid management. Additionally, restrictive intraoperative hydration, along with norepinephrine administration, decreases intraoperative blood loss [47]. According to a study from the University of Sheffield [48], the use of the ERAS setting was related to less blood loss and quicker recovery time following radical cystectomy, which related to study heterogeneity. It should be noted our single center has been implementing ERAS since 2015, and a retrospective cohort study including 192 patients from our center shows that ERAS may successfully speed up patient rehabilitation and is associated with less blood loss and LOS [49]. Furthermore, we performed a meta-analysis about this theme, evaluating and confirming the effectiveness of ERAS in the perioperative outcomes of radical cystectomy[50].
Conclusion
In conclusion, RARC had similar oncological outcomes compared with ORC as shown in our systematic review and meta-analysis. RARC leads to less EBL and lower blood transfusion rates at the expense of prolonged operative time. RARC and ORC resulted in similar rates of postoperative complications and health-related QOL. Further well-designed RCTs are essential to confirm this conclusion.
Acknowledgements
Not applicable.
Abbreviations
- ROB2
The version 2 of the Cochrane risk-of-bias tool for randomized trials
- RARC
Robot‐assisted radical cystectomy
- ORC
Open radical cystectomy
- QOL
Quality of life
- ICUD
Intracorporeal urinary diversion
- RCTs
Randomized controlled trials
- PRISMA
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- OS
Overall survival
- RFS
Recurrence-free survival
- PSM
Positive margin status
- EBL
Estimated blood loss
- LOS
Length of stay
- WMD
Weighted mean difference
- CI
Confidence intervals
- OR
Odds ratio
- FACT-VCI
Functional Assessment of Cancer Therapy-Vanderbilt Cystectomy Index
- QLQ-C30
Quality of Life Questionnaire Core 30
- FACT-BI
the Functional Assessment of Cancer Therapy – Bladder
- EQ-5D-5L
The EuroQol-5 dimension-5 level
- WHODAS 2.0
WHO Disability Assessment Schedule 2.0
- ERAS
Enhanced recovery after surgery
- ASA
American Society of Anesthesiologists physical status classification
- BMI
Body mass index
Authors’ contributions
Hongquan Liu and Zhongbao Zhou: Data curation; Investigation; Formal analysis; Writing original draft. Huibao Yao: Data curation; Investigation; Methodology; Resources; Supervision; Visualization; Writing—original draft. Qiancheng Mao: Formal analysis; Investigation; Methodology; Validation; Visualization; Writing—review & editing. Yongli Chu: Formal analysis; Investigation; Methodology; Validation; Visualization; Writing—review & editing. Yuanshan Cui and Jitao Wu: Conceptualization; Funding acquisition; Methodology; Project administration; Writing—review & editing. All authors reviewed the manuscript.
Funding
This work was supported by Joint Fund of Shandong Natural Science Foundation (ZR2021LSW019), grants from the National Nature Science Foundation of China (Nos. 81870525) and the Taishan Scholars Program of Shandong Province (No. tsqn201909199).
Availability of data and materials
The datasets that support the findings of this study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
The research does not involve patients, so ethical approval was not necessary.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hongquan Liu and Zhongbao Zhou contributed equally to this work as co-first authors.
Contributor Information
Yuanshan Cui, Email: doctorcuiys@163.com.
Jitao Wu, Email: wjturology@163.com.
References
- 1.Lenis AT, Lec PM, Chamie K, Mshs M. Bladder Cancer: A Review. JAMA. 2020;324:1980. doi: 10.1001/jama.2020.17598. [DOI] [PubMed] [Google Scholar]
- 2.Richters A, Aben KKH, Kiemeney LALM. The global burden of urinary bladder cancer: an update. World J Urol. 2020;38:1895–1904. doi: 10.1007/s00345-019-02984-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Alfred Witjes J, Lebret T, Compérat EM, Cowan NC, De Santis M, Bruins HM, et al. Updated 2016 EAU Guidelines on Muscle-invasive and Metastatic Bladder Cancer. Eur Urol. 2017;71:462–475. doi: 10.1016/j.eururo.2016.06.020. [DOI] [PubMed] [Google Scholar]
- 4.Parekh DJ, Reis IM, Castle EP, Gonzalgo ML, Woods ME, Svatek RS, et al. Robot-assisted radical cystectomy versus open radical cystectomy in patients with bladder cancer (RAZOR): an open-label, randomised, phase 3, non-inferiority trial. The Lancet. 2018;391:2525–2536. doi: 10.1016/S0140-6736(18)30996-6. [DOI] [PubMed] [Google Scholar]
- 5.Shabsigh A, Korets R, Vora KC, Brooks CM, Cronin AM, Savage C, et al. Defining Early Morbidity of Radical Cystectomy for Patients with Bladder Cancer Using a Standardized Reporting Methodology. Eur Urol. 2009;55:164–176. doi: 10.1016/j.eururo.2008.07.031. [DOI] [PubMed] [Google Scholar]
- 6.Falagario U, Veccia A, Weprin S, Albuquerque EV, Nahas WC, Carrieri G, et al. Robotic-assisted surgery for the treatment of urologic cancers: recent advances. Expert Rev Med Devices. 2020;17:579–590. doi: 10.1080/17434440.2020.1762487. [DOI] [PubMed] [Google Scholar]
- 7.Menon M, Hemal AK, Tewari A, Shrivastava A, Shoma AM, El-Tabey NA, et al. Nerve-sparing robot-assisted radical cystoprostatectomy and urinary diversion: NERVE-SPARING ROBOT-ASSISTED CYSTOPROSTATECTOMY and URINARY DIVERSION. BJU Int. 2003;92:232–236. doi: 10.1046/j.1464-410x.2003.04329.x. [DOI] [PubMed] [Google Scholar]
- 8.Fujimura T. Current status and future perspective of robot-assisted radical cystectomy for invasive bladder cancer. Int J Urol. 2019;26:1033–1042. doi: 10.1111/iju.14076. [DOI] [PubMed] [Google Scholar]
- 9.Hussein AA, May PR, Jing Z, Ahmed YE, Wijburg CJ, Canda AE, et al. Outcomes of Intracorporeal Urinary Diversion after Robot-Assisted Radical Cystectomy: Results from the International Robotic Cystectomy Consortium. J Urol. 2018;199:1302–1311. doi: 10.1016/j.juro.2017.12.045. [DOI] [PubMed] [Google Scholar]
- 10.Wang GJ, Barocas DA, Raman JD, Scherr DS. Robotic vs open radical cystectomy: prospective comparison of perioperative outcomes and pathological measures of early oncological efficacy. BJU Int. 2007;101:89–93. doi: 10.1111/j.1464-410X.2007.07212.x. [DOI] [PubMed] [Google Scholar]
- 11.Pruthi RS, Wallen EM. Robotic Assisted Laparoscopic Radical Cystoprostatectomy: Operative and Pathological Outcomes. J Urol. 2007;178:814–818. doi: 10.1016/j.juro.2007.05.040. [DOI] [PubMed] [Google Scholar]
- 12.Johar RS, Hayn MH, Stegemann AP, Ahmed K, Agarwal P, Balbay MD, et al. Complications After Robot-assisted Radical Cystectomy: Results from the International Robotic Cystectomy Consortium. Eur Urol. 2013;64:52–57. doi: 10.1016/j.eururo.2013.01.010. [DOI] [PubMed] [Google Scholar]
- 13.Satkunasivam R, Tallman CT, Taylor JM, Miles BJ, Klaassen Z, Wallis CJD. Robot-assisted Radical Cystectomy Versus Open Radical Cystectomy: A Meta-analysis of Oncologic, Perioperative, and Complication-related outcomes. European Urology Oncology. 2019;2:443–447. doi: 10.1016/j.euo.2018.10.008. [DOI] [PubMed] [Google Scholar]
- 14.Catto JWF, Khetrapal P, Ricciardi F, Ambler G, Williams NR, Al-Hammouri T, et al. Effect of Robot-Assisted Radical Cystectomy With Intracorporeal Urinary Diversion vs Open Radical Cystectomy on 90-Day Morbidity and Mortality Among Patients With Bladder Cancer: A Randomized Clinical Trial. JAMA. 2022;327:2092. doi: 10.1001/jama.2022.7393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mastroianni R, Ferriero M, Tuderti G, Anceschi U, Bove AM, Brassetti A, et al. Open Radical Cystectomy versus Robot-Assisted Radical Cystectomy with Intracorporeal Urinary Diversion: Early Outcomes of a Single-Center Randomized Controlled Trial. J Urol. 2022;207:982–992. doi: 10.1097/JU.0000000000002422. [DOI] [PubMed] [Google Scholar]
- 16.Maibom SL, Røder MA, Aasvang EK, Rohrsted M, Thind PO, Bagi P, et al. Open vs robot-assisted radical cystectomy (BORARC): a double-blinded, randomised feasibility study. BJU Int. 2022;130:102–113. doi: 10.1111/bju.15619. [DOI] [PubMed] [Google Scholar]
- 17.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int J Surg. 2021;88:105906. doi: 10.1016/j.ijsu.2021.105906. [DOI] [PubMed] [Google Scholar]
- 18.Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 19.Nix J, Smith A, Kurpad R, Nielsen ME, Wallen EM, Pruthi RS. Prospective Randomized Controlled Trial of Robotic versus Open Radical Cystectomy for Bladder Cancer: Perioperative and Pathologic Results. Eur Urol. 2010;57:196–201. doi: 10.1016/j.eururo.2009.10.024. [DOI] [PubMed] [Google Scholar]
- 20.Vejlgaard M, Maibom SL, Joensen UN, Thind PO, Rohrsted M, Aasvang EK, et al. Quality of life and secondary outcomes for open versus robot-assisted radical cystectomy: a double-blinded, randomised feasibility trial. World J Urol. 2022;40:1669–1677. doi: 10.1007/s00345-022-04029-9. [DOI] [PubMed] [Google Scholar]
- 21.Khan MS, Omar K, Ahmed K, Gan C, Van Hemelrijck M, Nair R, et al. Long-term Oncological Outcomes from an Early Phase Randomised Controlled Three-arm Trial of Open, Robotic, and Laparoscopic Radical Cystectomy (CORAL) Eur Urol. 2020;77:110–118. doi: 10.1016/j.eururo.2019.10.027. [DOI] [PubMed] [Google Scholar]
- 22.Venkatramani V, Reis IM, Castle EP, Gonzalgo ML, Woods ME, Svatek RS, et al. Predictors of Recurrence, and Progression-Free and Overall Survival following Open versus Robotic Radical Cystectomy: Analysis from the RAZOR Trial with a 3-Year Followup. J Urol. 2020;203:522–529. doi: 10.1097/JU.0000000000000565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bochner BH, Dalbagni G, Marzouk KH, Sjoberg DD, Lee J, Donat SM, et al. Randomized Trial Comparing Open Radical Cystectomy and Robot-assisted Laparoscopic Radical Cystectomy: Oncologic Outcomes. Eur Urol. 2018;74:465–471. doi: 10.1016/j.eururo.2018.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Khan MS, Gan C, Ahmed K, Ismail AF, Watkins J, Summers JA, et al. A Single-centre Early Phase Randomised Controlled Three-arm Trial of Open, Robotic, and Laparoscopic Radical Cystectomy (CORAL) Eur Urol. 2016;69:613–621. doi: 10.1016/j.eururo.2015.07.038. [DOI] [PubMed] [Google Scholar]
- 25.Bochner BH, Dalbagni G, Sjoberg DD, Silberstein J, Keren Paz GE, Donat SM, et al. Comparing Open Radical Cystectomy and Robot-assisted Laparoscopic Radical Cystectomy: A Randomized Clinical Trial. Eur Urol. 2015;67:1042–1050. doi: 10.1016/j.eururo.2014.11.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Messer JC, Punnen S, Fitzgerald J, Svatek R, Parekh DJ. Health-related quality of life from a prospective randomised clinical trial of robot-assisted laparoscopic vs open radical cystectomy: Health-related quality of life in trial of ORC vs RARC. BJU Int. 2014;114:896–902. doi: 10.1111/bju.12818. [DOI] [PubMed] [Google Scholar]
- 27.Parekh DJ, Messer J, Fitzgerald J, Ercole B, Svatek R. Perioperative Outcomes and Oncologic Efficacy from a Pilot Prospective Randomized Clinical Trial of Open Versus Robotic Assisted Radical Cystectomy. J Urol. 2013;189:474–479. doi: 10.1016/j.juro.2012.09.077. [DOI] [PubMed] [Google Scholar]
- 28.Kimura S, Iwata T, Foerster B, Fossati N, Briganti A, Nasu Y, et al. Comparison of perioperative complications and health-related quality of life between robot-assisted and open radical cystectomy: A systematic review and meta-analysis. Int J Urol. 2019;26:760–774. doi: 10.1111/iju.14005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tang J-Q, Zhao Z, Liang Y, Liao G. Robotic-assisted versus open radical cystectomy in bladder cancer: A meta-analysis of four randomized controlled trails. Int J Med Robotics Comput Assist Surg. 2018;14:e1867. doi: 10.1002/rcs.1867. [DOI] [PubMed] [Google Scholar]
- 30.Leow JJ, Reese SW, Jiang W, Lipsitz SR, Bellmunt J, Trinh Q-D, et al. Propensity-Matched Comparison of Morbidity and Costs of Open and Robot-Assisted Radical Cystectomies: A Contemporary Population-Based Analysis in the United States. Eur Urol. 2014;66:569–576. doi: 10.1016/j.eururo.2014.01.029. [DOI] [PubMed] [Google Scholar]
- 31.Leow JJ, Chang SL, Meyer CP, Wang Y, Hanske J, Sammon JD, et al. Robot-assisted Versus Open Radical Prostatectomy: A Contemporary Analysis of an All-payer Discharge Database. Eur Urol. 2016;70:837–845. doi: 10.1016/j.eururo.2016.01.044. [DOI] [PubMed] [Google Scholar]
- 32.Farnham SB, Webster TM, Herrell SD, Smith JA. Intraoperative blood loss and transfusion requirements for robotic-assisted radical prostatectomy versus radical retropubic prostatectomy. Urology. 2006;67:360–363. doi: 10.1016/j.urology.2005.08.029. [DOI] [PubMed] [Google Scholar]
- 33.Nepple KG, Strope SA, Grubb RL, Kibel AS. Early oncologic outcomes of robotic vs. open radical cystectomy for urothelial cancer. Urologic Oncology. 2013;31:894–8. doi: 10.1016/j.urolonc.2011.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hanna N, Leow JJ, Sun M, Friedlander DF, Seisen T, Abdollah F, et al. Comparative effectiveness of robot-assisted vs. open radical cystectomy. Urologic Oncology. 2018;36(88):e1–88.e9. doi: 10.1016/j.urolonc.2017.09.018. [DOI] [PubMed] [Google Scholar]
- 35.Cacciamani GE, Winter M, Medina LG, Ashrafi AN, Miranda G, Tafuri A, et al. Radical cystectomy pentafecta: a proposal for standardisation of outcomes reporting following robot-assisted radical cystectomy. BJU Int. 2020;125:64–72. doi: 10.1111/bju.14861. [DOI] [PubMed] [Google Scholar]
- 36.Brassetti A, Tuderti G, Anceschi U, Ferriero M, Guaglianone S, Gallucci M, et al. Combined reporting of surgical quality, cancer control and functional outcomes of robot-assisted radical cystectomy with intracorporeal orthotopic neobladder into a novel trifecta. Minerva Urol Nefrol. 2019;71:590–596. doi: 10.23736/S0393-2249.19.03566-5. [DOI] [PubMed] [Google Scholar]
- 37.Lawrentschuk N, Colombo R, Hakenberg OW, Lerner SP, Månsson W, Sagalowsky A, et al. Prevention and Management of Complications Following Radical Cystectomy for Bladder Cancer. Eur Urol. 2010;57:983–1001. doi: 10.1016/j.eururo.2010.02.024. [DOI] [PubMed] [Google Scholar]
- 38.Djaladat H, Katebian B, Bazargani ST, Miranda G, Cai J, Schuckman AK, et al. 90-Day complication rate in patients undergoing radical cystectomy with enhanced recovery protocol: a prospective cohort study. World J Urol. 2017;35:907–911. doi: 10.1007/s00345-016-1950-z. [DOI] [PubMed] [Google Scholar]
- 39.Hosseini A, Ebbing J, Collins J. Clinical outcomes of robot-assisted radical cystectomy and continent urinary diversion. Scand J Urol. 2019;53:81–88. doi: 10.1080/21681805.2019.1598486. [DOI] [PubMed] [Google Scholar]
- 40.Kurpad R, Woods M, Pruthi R. Current Status of Robot-Assisted Radical Cystectomy and Intracorporeal Urinary Diversion. Curr Urol Rep. 2016;17:42. doi: 10.1007/s11934-016-0598-y. [DOI] [PubMed] [Google Scholar]
- 41.Karimi M, Brazier J. Health, Health-Related Quality of Life, and Quality of Life: What is the Difference? Pharmacoeconomics. 2016;34:645–649. doi: 10.1007/s40273-016-0389-9. [DOI] [PubMed] [Google Scholar]
- 42.Chhabra KR, Sacks GD, Dimick JB. Surgical Decision Making: Challenging Dogma and Incorporating Patient Preferences. JAMA. 2017;317:357. doi: 10.1001/jama.2016.18719. [DOI] [PubMed] [Google Scholar]
- 43.Mastroianni R, Tuderti G, Ferriero M, Anceschi U, Bove AM, Brassetti A, et al. Open vs robotic intracorporeal Padua ileal bladder: functional outcomes of a single-centre RCT. World J Urol. 2023;41:739–746. doi: 10.1007/s00345-023-04312-3. [DOI] [PubMed] [Google Scholar]
- 44.Fallara G, Di Maida F, Bravi CA, De Groote R, Piramide F, Turri F, et al. A systematic review and meta-analysis of robot-assisted vs. open radical cystectomy: where do we stand and future perspective. Minerva Urol Nephrol. 2023;75:134–43. doi: 10.23736/S2724-6051.23.05065-6. [DOI] [PubMed] [Google Scholar]
- 45.Fontanet S, Basile G, Baboudjian M, Gallioli A, Huguet J, Territo A, et al. Robot-assisted vs. open radical cystectomy: systematic review and meta-analysis of randomized controlled trials. Actas Urol Esp (Engl Ed). 2023;S2173–5786(23):00005–7. doi: 10.1016/j.acuroe.2023.01.003. [DOI] [PubMed] [Google Scholar]
- 46.Kowalewski K-F, Wieland VLS, Kriegmair MC, Uysal D, Sicker T, Stolzenburg J-U, et al. Robotic-assisted Versus Laparoscopic Versus Open Radical Cystectomy-A Systematic Review and Network Meta-analysis of Randomized Controlled Trials. Eur Urol Focus. 2022;S2405–4569(22):00285–291. doi: 10.1016/j.euf.2022.12.001. [DOI] [PubMed] [Google Scholar]
- 47.Azhar RA, Bochner B, Catto J, Goh AC, Kelly J, Patel HD, et al. Enhanced Recovery after Urological Surgery: A Contemporary Systematic Review of Outcomes, Key Elements, and Research Needs. Eur Urol. 2016;70:176–187. doi: 10.1016/j.eururo.2016.02.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pang KH, Groves R, Venugopal S, Noon AP, Catto JWF. Prospective Implementation of Enhanced Recovery After Surgery Protocols to Radical Cystectomy. Eur Urol. 2018;73:363–371. doi: 10.1016/j.eururo.2017.07.031. [DOI] [PubMed] [Google Scholar]
- 49.Wei C, Wan F, Zhao H, Ma J, Gao Z, Lin C. Application of enhanced recovery after surgery in patients undergoing radical cystectomy. J Int Med Res. 2018;46:5011–5018. doi: 10.1177/0300060518789035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang D, Sun K, Wang T, Wu G, Wang J, Cui Y, et al. Systematic Review and Meta-Analysis of the Efficacy and Safety of Enhanced Recovery After Surgery vs. Conventional Recovery After Surgery on Perioperative Outcomes of Radical Cystectomy. Front Oncol. 2020;10:541390. doi: 10.3389/fonc.2020.541390. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets that support the findings of this study are available from the corresponding author on reasonable request.