Abstract
Primary cilia are microtubule-based sensory and signaling organelles on the surfaces of most eukaryotic cells. Despite their early description by microscopy studies, islet cilia had not been examined in the functional context until recent decades. In pancreatic islets as in other tissues, primary cilia facilitate crucial developmental and signaling pathways in response to extracellular stimuli. Many human developmental and genetic disorders are associated with ciliary dysfunction, some manifesting as obesity and diabetes. Understanding the basis for metabolic diseases in human ciliopathies has been aided by close examination of cilia action in pancreatic islets at cellular and molecular levels. In this article, we review the evidence for ciliary expression on islet cells, known roles of cilia in pancreas development and islet hormone secretion, and summarize metabolic manifestations of human ciliopathy syndromes. We discuss emerging data on primary cilia regulation of islet cell signaling and the structural basis of cilia-mediated cell crosstalk, and offer our interpretation on the role of cilia in glucose homeostasis and human diseases.
Keywords: Cilia, Ciliopathies, Insulin, Insulin-secreting cells, Islets of Langerhans
KEY FIGURE
INTRODUCTION
Primary cilia are antenna-like organelles projecting from the cellular membrane. These tubulin-based structures are highly conserved across the species and found on almost every cell in the human body [1]. Cilia in the form of flagella on swimming protozoa were discovered in the 1670s by the great Dutch microscopist, Van Leeuwenhoek, father of microbiology [2]. It would take another 200 years for the discovery of primary cilia on mammalian cells in 1898 [3]. Unlike motile cilia whose role in human genetic diseases was established early on [4], the function of primary cilia had not been elucidated until these past two decades [5]. Since then, exploding interest in cilia have brought this once forgotten organelle to the forefront of cell biology and human disease research. Through recent advances in proteomics, genetics, and microscopy, we are starting to piece together aspects of primary cilia function through the characterization of receptors, ion channels and signaling molecules that are uniquely enriched and interactive in primary cilia [6-9]. At this point, primary cilia are recognized as important signaling hubs for cellular development, differentiation, and function, rather than vestigial organelles [8,10-12].
As cilia are highly conserved and present on most cells of vertebrate organisms, they play a central role in normal development and also in disease. In humans, inherited motile and primary cilia disorders are collectively termed ciliopathies and involve nearly all major organs [1]. Most early studies on cilia function had been limited to motile cilia [4,13], namely the complex motile ciliopathy syndrome known as primary ciliary dyskinesia which includes Kartagener’s syndrome (also known as immotile cilia syndrome) characterized by a triad of situs inversus, chronic sinusitis, and bronchiectasis resulting from cilia dysmotility [4]. The year 2000 saw the seminal discovery of hypomorphic mutation in Tg737, or intraflagellar transport 88 (IFT88), an essential gene for cilia assembly, where Tg737orpk mutant mice displayed the phenotype of autosomal recessive polycystic kidney disease [5] and prompted further investigation of the role of primary cilia in human disease. To date, multiple human genetic diseases have been linked to primary cilia dysfunction including autosomal dominant/recessive polycystic kidney disease, Bardet-Biedl syndrome (BBS), Alström syndrome, Meckel-Gruber syndrome, and Joubert syndrome [14-19]. The kidney, liver, brain, and retina are typically the organs most affected by primary ciliopathy and have been the traditional focus of ciliary studies [20-23]. Meanwhile, the association between cilia and energy metabolism found in BBS has sparked investigations into ciliary function in metabolic disorders such as obesity and diabetes [19]. In this review, we will summarize current knowledge about primary cilium structure, expression, and function in pancreatic islets, one of the regulatory centers of glucose and energy homeostasis in the body.
REVISITING THE STRUCTURE OF PRIMARY CILIA
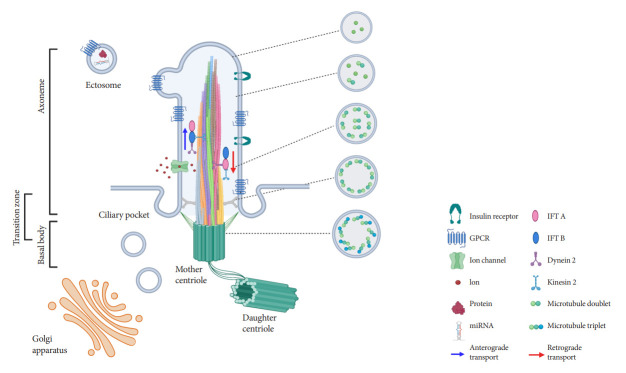
Ultrastructural examination of primary cilia cross-sections by electron microscopy typically reveals a cylindrical axoneme composed of nine outer microtubule doublets with no central pairs, dubbed as the “9+0” configuration, in contrast to the motile cilia “9+2” paradigm where a central microtubule pair and associated structural motifs confer motility to the axoneme. However, recent findings by serial selection- or cryoelectron tomography have challenged this dichotomous classification, revealing that primary cilia have a dynamic structure that does not always conform to “9+0” expectations [24-26]. The axoneme of the primary cilia starts with “9+0” microtubules at the base, but not all microtubules have equal length, so they decrease in number as the axoneme grows distally (Fig. 1) [24,25]. In cultured kidney cells, primary cilia have been seen to adopt a “7+2” microtubule configuration immediately after leaving the basal body and become “3+0” toward the tip [24]. In addition to microtubule length and number, the arrangement of microtubules also changes from base to tip, reducing their pairing from triplet to doublet to singlet [24,25]. Along with these extra-pancreatic observations, several pioneering studies in pancreatic β-cell primary cilia have reported microtubule patterns including “8+2,” “7+2,” “3+0,” or “2+0” [27-29]. It appears that, while most primary cilia start as 9+0 at the base, they soon become something else, and the cross-sectional structure differs depending on where you look. Thus, these non-“9+0” arrangements, once regarded as atypical manifestations, may in fact reflect the normal evolution of primary cilia structure from base to tip.
Fig. 1.
Structure of primary cilia in pancreatic β-cells. The axoneme is depicted as a helical bundle of microtubule filaments that decrease in number and diameter from base to tip, with evolving microtubule configurations from 9+0 to non-9+0, as shown by ultrastructural studies. Intraflagellar transport (IFT) trains move cargo bi-directionally along the length of the axoneme and are powered by motor proteins dynein and kinesin. The ciliary membrane is rich with G-protein coupled receptors (GPCRs) and other signaling proteins, as well as ion channels. Extracellular vesicles in the form of ectosomes may be released from primary cilia, containing bioactive materials such as protein and microRNA (miRNA).
The fact that most primary cilia diameter follows a gradual tapering from base to tip provides several clues that may explain the flexural bending properties, e.g., to extracellular flow and suggests greater complexity in the structure of primary cilia than previously assumed. Primary cilia are dynamic organelles that constantly undergo remodeling and whose functions depend on a busy intra-ciliary transport system [30,31]. Since at least half of the axoneme consists of microtubule singlets according to the recent findings [24,25], it raises questions about the current paradigm of a clear division between anterograde and retrograde IFT complexes on B- and A-tubules, and how bi-directional cargo trafficking might be accomplished on single microtubule filaments. Around 1,000 proteins have been identified in cilia-basal body complex, many of which are still of unknown function [32]. How these proteins are implicated in cilia assembly and maintenance, and how the intra-ciliary transport system functions in non-binary structures remain to be clarified. In addition to the evolving classification system based on cilia structure, there are also blurring distinctions of ciliary protein functions inside and outside the cilium. Growing evidence supports cilia function outside its membranebased structure through the release of extracellular vesicles (EVs) (Fig. 1), which serve to transmit long-range signals between cells [24,25,33-37]. EVs derived from primary cilia are heterogeneous and show signal-dependent ectocytosis [34,37]. The physiologic relevance of ciliary EVs in intercellular communication and function is entirely unstudied in pancreatic islets and therefore represents an opportunity for future investigation.
PRIMARY CILIA IN PANCREAS AND ISLETS
Ciliated cells in mouse and human pancreas
Cytoplasmic projections between the intercellular spaces were observed in β-cells of rabbits and guinea pigs in 1957 [38], and a year later these structures were identified as primary cilia in mouse β-cells, convincingly shown as a defined filamentous axoneme connected to the basal body and centrioles [39]. In early reports, islet cilia were only described in β-cells and not in other islet cells such as α-cells, ductal cells, or acinar cells. Subsequent studies have since demonstrated the presence of primary cilia in α-, δ-, and pancreatic polypeptide (PP) cells in mouse and rat pancreatic islets by electron and light microscopy (Table 1) [16,27-29,40-44]. In humans, primary cilia in the pancreas were first reported in β-cell tumor cells in 1964 [45]. So far, in human islets, only β- and α-cells have been documented to have primary cilia [42,45], whereas in the exocrine pancreas, primary cilia have been identified on ductal and centroacinar cells in both human and mouse [16,27,29,43]. RNA sequencing studies have demonstrated robust expression of cilia-related genes across cell types in mouse and human pancreas and islets, whose expressions are dynamically modulated by metabolic conditions such as hyperglycemia and diabetes mellitus [46-49]. These observations confirm near-ubiquitous expression of primary cilia in pancreatic tissues across species and implicate adaptability of cilia function in physiology and disease states.
Table 1.
Ciliated cells in the mouse and human pancreas
| Variable | Function | Mouse | Human | Reference | |
|---|---|---|---|---|---|
| Endocrine | |||||
| Islets | |||||
| α | Glucagon | + | + | [29,40-42] | |
| β | Insulin | + | + | [29,40-44] | |
| δ | Somatostatin | + | Unknown | [29,40,41] | |
| PP | Pancreatic polypeptide | + | Unknown | [40] | |
| ε | Ghrelin | Unknown | Unknown | - | |
| Enterochromaffin cell | Serotonin, motilin, substance P, histamine, and kinins | Unknown | Unknown | - | |
| G-cell | Gastrin | Unknown | Unknown | - | |
| Exocrine | |||||
| Acinar cell | Digestive enzymes such as trypsin, chymotrypsin, amylase, and carboxypeptidase | – | – | [16,29,43] | |
| Centroacinar cell | Mucin, bicarbonates | + | + | [16,27,29,43] | |
| Ductal epithelial cell | Mucin, bicarbonates | + | + | [16,28,29,43] | |
PP, pancreatic polypeptide.
Orientation of primary cilia in β-cells
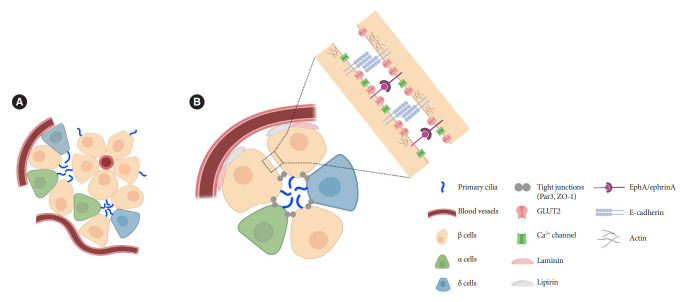
Islets are enclosed organoids containing endocrine cells arranged in a three-dimensional sphere, where cellular polarity and orientation are neither fixed nor fully understood. Emerging data suggest that rodent β-cells are polarized with regional specializations, such that there is lateral localization of glucose transporters and basal distribution of insulin granule [50-52]. The precise location of primary cilia on β-cells has been a challenge to define. Limited data by electron microscopy have shown conflicting results, with the cilium protruding either from the lateral surfaces of β-cells into the canalicular lumen or towards the vasculature [39,53]. It was unclear whether cilia orientation was conserved, or indeed mattered, until a finding in 2009 revealed that the pancreas-specific liver kinase B1 knockout mice exhibited altered β-cell polarity and ciliary orientation relative to blood vessels, showing for the first time that cilia orientation was regulatable in islet cells and may be linked to key β-cell functions such as cell size control and insulin secretion [54]. However, the issue remained that β-cell functional domains themselves were not well-characterized, thus there lacked reference points by which to define the spatial location of cilia and their polarity and orientation. Later studies contributed clarity to this issue by defining functional edge domains of the β-cell in mouse and human islets, showing consistent polarity determinants using three-dimensional confocal and serial electron microscopy [55-57]. The three distinct domains of pancreatic β-cells include the apical domain away from blood vessels, the lateral domain adjacent to neighboring β-cells, and the basal domain in contact with blood vessels (Fig. 2) [55]. In focused ion beam-scanning electron microscopy (FIB-SEM), β-cell primary cilia are found to project toward the extracellular apical lumen (Fig. 2) and along the apical tight junction (as marked by zonula occludens-1) while being away from the vasculature [55]. These findings together with earlier electron microscopy studies helped establish the current consensus view which is that islet cilia protrude into the canalicular lumen at intercellular junctions in the islet interior [53]. Compared to renal or vascular endothelial cells, in which the apical orientation of primary cilia is required for their role to detect flow or shear stress [58-60], the functional implication of apical-lateral positioned primary cilia in β-cells remains to be clarified. One potential explanation is that the apical orientation of primary cilia may enhance β-cell interaction with neighboring cells and potentiate autocrine and paracrine signaling [55]. Recently, we demonstrated that primary cilia not only have sensory but also motile function [49] which speculatively would better enable three-dimensional sampling of external cues. It would be of interest to examine ciliary motility as determined by its subcellular location and what varying degrees of freedom are afforded by its surrounding physical space.
Fig. 2.
Orientation of primary cilia in pancreatic islet cells. (A) Primary cilia in islets tend to be located opposite the vascular apogee. Neighboring islet endocrine cells organize their cilia in shared interstitial spaces that would potentially allow cilia-cilia interactions and paracrine signal detection. (B) Putative model based on published studies showing preferential cilia positioning in the apical zone, near the lateral domain that mediates cell-cell adhesion and signaling between adjacent cells. In our unpublished studies, cilia can also be seen projecting from the lateral surface and traversing the narrow space between adjacent cells. GLUT2, glucose transporter 2; ZO-1, zonula occludens-1; EphA, epoxide hydrolase A.
Motility of primary cilia in pancreatic islets
Traditionally, cilia have been classified as motile and non-motile cilia according to their microtubule structure (“9+2” vs. “9+0”), as the central microtubule pair is thought to be essential for active movement. However, exceptions to this rule exist, as both non-“9+2” motile cilia and “9+2” immotile cilia have been described, thus the “9+2” structure appears neither necessary nor sufficient for ciliary motility. Nodal cilia in the developing embryo, for example, have a “9+0” axonemal structure and are famous for their rotational movement that drives morphogen flow, which serves in L–R axis determination [61- 65]. Meanwhile, the “9+2” kinocilia in auditory hair cells are non-motile and represent one of the heterogeneous cilia forms in the organ of Corti [66]. In any given tissue or organ system, it is likely that neither the “9+0” nor “9+2” form is absolute, and there may exist both inter-cilia as well as intra-cilia differences, since the microtubule structure of even a single primary cilium can change from base to tip [24,25]. Thus, primary cilia may require a more permissive classification, one that includes the possibility of motility in addition to their predominant sensory function.
While primary cilia have been reported to move passively, in response to extracellular fluid flow [58,67] or to tugging by intracellular actin forces [68], active motility by primary cilia had not been demonstrated until recently. Two studies in human pancreatic islets have reported that islet cells express motile cilia genes and protein complexes that were once thought to only exist in classic motile cilia [48,49]. These include the core axonemal dyneins dynein axonemal intermediate chain 1 (DNAI1), dynein axonemal heavy cahin 5 (DNAH5), dynein axonemal light chain 1 (DNAL1), as well as central pair-associated proteins sperm flagellar 2 (SPEF2) and kinesin family member 9 (KIF9), with their ciliary localization confirmed by light and electron microscopy in human islet cells [49]. These motile components mediate active movement of primary cilia via adenosine triphosphate hydrolysis, a key energy-generating reaction in living cells [49]. Functionally, β-cell cilia movement occurs in response to extracellular glucose and is coupled to glucose-stimulated events including Ca2+ influx and insulin secretion [49]. Cilia motility genes are dynamically expressed in islets, with enrichment in pancreatic endocrine cells compared to non-endocrine cells, and greater expression in diabetic than healthy islets [49]. Consistent with these findings, gene expression network analysis in human pancreatic islets showed that cilia-related genes in α- and β-cells, including motility genes, are upregulated in individuals with type 2 diabetes mellitus, and that modulation of these genes led to changes in glucoseinduced insulin secretion [48]. Taken together, these results reveal dynamic cilia expression in health and disease and implicate a new functionality of primary cilia that bridges the sensory and motile divide, as well as a regulatory role for cilia motility in β-cell insulin secretion. Future studies might elaborate how cilia motility works in orchestrating β-cell coupling within islets as well as in mediating heterotypic cell communications via paracrine or juxtracrine signaling.
Role of primary cilia in pancreas development
Primary cilia are dynamic organelles whose assembly and disassembly are linked to the cell cycle and differentiation status of the cell [69-71]. The role of primary cilia in pancreas development has been studied using genetic mouse models targeting ciliogenesis or cilia function (Table 2) [16,40-43,54,72-81]. The global Ift88 homozygous mutant mice present with absent or shortened pancreatic cilia, which in the pancreas exhibits as reduced pancreas mass with collagen deposition, progressive acinar cell loss, and ductal hyperplasia with pancreatic cyst formation [16]. These exocrine defects are consistent with phenotypes seen in knockout mouse models of ciliary protein Chibby1 or pancreas-specific kinesin family member 3A (Kif3a), an essential gene for cilia formation [43,72]. Proposed mechanisms for these pancreatic morphological changes include defective exocytosis of zymogen granules, intrapancreatic activation of digestive enzymes such as carboxypeptidase, and activation of transforming growth factor β and mitogen-activated protein kinase kinase/extracellular signal-regulating kinase pathways [41,43,72]. Similar pancreatic phenotype of the whole-body Ift88 mutant mice were also observed in mice harboring mutations in the polycystin 2 (Pkd2) and inversin genes, which mediate cilia function rather than cilia assembly [16]. These results indicate that both accurate assembly and function of primary cilia are required for proper pancreatic tissue organization. Of interest, induction of Kif3a loss-of-function at 4 weeks of age rather than prenatally failed to elicit a significant pancreatic phenotype, suggesting that there may be a critical period which requires normal cilia for pancreas development [72].
Table 2.
Pancreatic defects in mouse models involved in ciliary genes
| Gene | Mutation type | Phenotype |
Reference | |
|---|---|---|---|---|
| Exocrine pancreas | Endocrine pancreas | |||
| Alms1 | Constitutive, homozygous (–/–) | Unaffected | Islet hyperplasia, hypertrophy, and cystic change | [74,81] |
| Partially degranulated β-cells | ||||
| Hyperinsulinemia and insulin resistance | ||||
| Impaired glucose tolerance, diabetes (>80% in foz/foz mouse) | ||||
| Bbs4 | Constitutive, homozygous (–/–) | Not examined | Normal islet morphology | [80] |
| Impaired glucose tolerance | ||||
| Chibby1 | Constitutive, homozygous (–/–) | Ductal dilation | Normal islet structure | [43] |
| Mucus accumulation and lipomatosis | ||||
| Disorganized acinar cells with zymogen granule accumulation | ||||
| Progressive loss of acinar cells with chronic pancreatitis | ||||
| Hnf6 | Constitutive, homozygous (–/–) | Pancreatic cysts with dysmorphic epithelial lining | Not examined | [77] |
| Ift88 (Tg737) | Constitutive, hypomorphic mutation (Tg737orpk) | Reduced pancreas mass | Normal islet architecture | [16] |
| Collagen deposition | Clustering of islets | |||
| Progressive acinar cell loss | Impaired glucose tolerance | |||
| Ductal hyperplasia with cyst formation | ||||
| Conditional (Pdx1-CreER), homozygous (–/–) | Not examined | ↓ β-Cell mass | [79] | |
| ↑ β-Cell apoptosis | ||||
| ↓ Glucose-stimulated insulin secretion | ||||
| Impaired glucose tolerance | ||||
| Conditional (Ins1-Cre), homozygous (–/–) | Not examined | ↓ β-Cell mass | [42] | |
| ↑ δ-Cell mass | ||||
| ↓ Insulin content and secretion | ||||
| ↑ Glucagon, somatostatin secretion | ||||
| impaired glucose tolerance | ||||
| Inv | Constitutive, homozygous (–/–) | Pancreatic cysts | Islet cell hyperplasia and disorganization | [76] |
| Vacuolization of the acinar cells | ||||
| Ductal dilation | ||||
| Kif3a | Conditional (Pdx1-Cre), homozygous (–/–) | Ductal dilation and cyst formation | Normal islet structure | [72] |
| Acinar-to-ductal metaplasia | Normal glucose tolerance | |||
| Progressive acinar cell loss | ||||
| Extensive fibrosis and lipomatosis | ||||
| Lkb1 | Conditional (Pdx1-CreER), homozygous (–/–) | Not examined | ↑ β-Cell size | [54] |
| Altered β-cell polarity (clustering of β-cell nuclei) | ||||
| Enhanced glucose tolerance | ||||
| Pkd1 | Constitutive, homozygous (–/–) | Pancreatic cyst | ↓ Islet number | [75] |
| Ductal dilation | ||||
| ↓ Acinar number | ||||
| Pkhd1 | Constitutive, homozygous (del2/del2) | Pancreatic cysts | Not examined | [78] |
| Ductal dilation and thickening | ||||
| Constitutive, homozygous (del4/del4) | Pancreatic cysts (10%) | Not examined | [73] | |
| Ductal dilation | ||||
| Periductal fibrosis | ||||
| Rfx3 | Constitutive, homozygous (–/–) | Unaffected | Small and disorganized islets | [40] |
| ↓ α-, β-, and ghrelin cell mass | ||||
| ↑ PP cell mass | ||||
| ↓ Insulin content/secretion, glucagon, and ghrelin | ||||
| ↑ PP | ||||
| Impaired glucose tolerance | ||||
ALMS, Alström syndrome 1; Bbs4, Bardet-Biedl syndrome 4; Hnf6, hepatocyte nuclear factor 6; Ift88, intraflagellar transport 88; orpk, oak ridge polycystic kidney; Pdx1, pancreatic and duodenal homebox 1; CreER, Cre-estrogen receptor; Ins1, insulin 1; Inv, inversin; Kif3a, kinesin family member 3A; Lkb1, liver kinase B1; Pkd1, polycystin 1; Pkhd1, polycystic kidney and hepatic disease 1; del, deletion; Rfx3, regulatory factor X3; PP, pancreatic polypeptide.
In contrast to in the exocrine pancreas, ciliary phenotypes in endocrine pancreas were less clear in the early studies, given conflicting results about their requirement in islet cell development and function [16,40-43,72]. The Chibby1- or pancreas-specific Kif3a-knockout mice exhibited grossly normal islet structure and apparently unperturbed endocrine cell differentiation, resulting in animals with normal glucose level or tolerance [43,72]. The hypomorphic Ift88 mutant mice showed comparable islet areas and β-cell maturity marker expression as in wild-type mice [16]. However, Ift88 hypomorph animals showed impaired glucose tolerance when challenged with high glucose, suggesting that primary cilia may be involved in the sensing or regulation of blood glucose levels [41]. Supporting this notion, whole-body deletion of regulatory factor X3 (Rfx3), a key ciliogenic gene, and β-cell specific deletion of Ift88 (β-cell specific cilia knockout [βCKO]) both pre- and post-natally produced reduced or absent cilia, which led to impaired glucose tolerance and decreased insulin secretion [40,42]. Both these cilia deletion models showed altered islet cell composition, suggesting cilia involvement in islet development. In Rfx3–/– mice, mRNA and/or protein expression of insulin, glucagon, and ghrelin decreased 10- to 150-fold while PP increased 15-fold [40]. β-Cell specific Ift88–/– mice showed reduced β-cell mass, insulin content and secretion, while δ-cell mass and somatostatin secretion were increased [42]. In another inducible β-cell Ift88–/– βICKO mice, β-cell mass was normal in the early period of cilia deletion but progressively decrease over time: normal 6 weeks after cilia deletion, to 10% decrease after 8 weeks, and over 80% decrease after 20 weeks [42,79]. These findings suggest that primary cilia play a requisite role in pancreatic endocrine cell differentiation as well as maintenance of islet cell mass. Consistently, cilia-dependent Gli/Hedgehog activation in mice causes β-cell dedifferentiation, providing a potential mechanism by which primary cilia regulate islet cell fate [82,83]. Further studies are needed to elucidate how primary cilia regulate the development of specific islet cell subsets and to identify ciliary signaling pathways linked to endocrine cell differentiation.
ROLE OF CILIA IN ISLET HORMONE RELEASE
Emerging consensus from both in vitro and in vivo studies show that primary cilium defects significantly impair β-cell function by reducing both insulin production and secretion [40,42,79,80]. In βCKO mice, deletion of β-cell Ift88 attenuated first-phase insulin secretion and delayed recovery to baseline secretion compared with wild type [42], indicating that that primary cilia regulate β-cell synchrony in their secretory response to glucose [42,84]. Similar results were reported in tamoxifen-inducible cilia knockout mice [42,79], demonstrating that the removal of cilia function in mature functional β-cells, rather than the developmental changes that result from cilia deletion, accounted for the β-cell phenotypes in these experimental models.
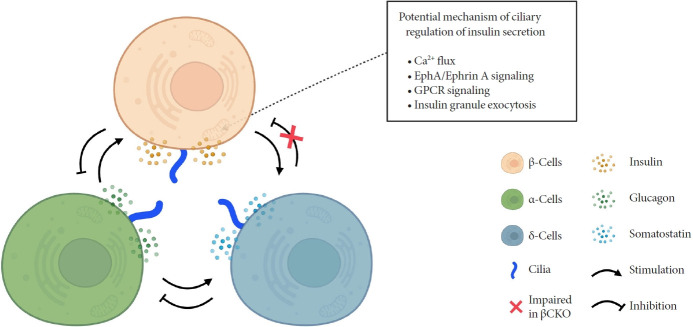
How do cilia regulate insulin secretion? Several key intracellular β-cell events may be under primary cilia control. First, primary cilia regulate glucose-stimulated Ca2+ entry into β-cells. Ift88 deletion and thus the absence of primary cilia in βCKO mouse islets abrogated the first-phase Ca2+ influx as well as second-phase Ca2+ oscillations [42], although it was unaddressed whether these observed effects on Ca2+ are mediated through directly ciliary control of β-cell membrane potential. Second, primary cilia modulate epoxide hydrolase A (EphA)-ephrin-A signaling, a juxtacrine communication pathway that controls insulin secretion in β-cells [85]. Cilia-ablated adult β-cells exhibit hyperphosphorylation of EphA due to impaired endosomal recycling, resulting in Rac family small GTPase 1 (Rac1) inhibition and decrease of insulin secretion [79]. Interestingly, Eph-ephrin signaling may partly account for the reciprocal regulation of cilia and the cytoskeletal network, as actin and microtubule polymerization impact ciliation, and primary cilia signaling through the planar cell polarity pathway may modulate F-actin density, a known mechanism for regulating insulin granule release [86]. There may be direct evidence that cilia loss-of-function causes impaired insulin granule exocytosis, thus accounting for the insulin secretory defect [80]. Bbs4- and oral-facial-digital syndrome type I (Ofd1)-deleted islets had decreased mRNA or protein level of membrane fusion soluble N-ethylmaleimide-sensitive factor attachment receptor (SNARE) proteins Syntaxin1A and synaptosomal associated protein 25 (SNAP-25), while overexpression of Syntaxin1A restored β-cell function comparable to that of the control group [80]. G-protein coupled receptor (GPCR) signaling through the β-cell primary cilium is another established regulator of insulin secretion, a subject that will be discussed in the next section.
Primary cilia of β-cells regulate insulin secretion via cell-intrinsic events as well as via cell communication pathways in response to neighboring islet cells. Direct evidence for primary cilia regulation of paracrine communication has been made in βCKO mouse islets. In the absence of β-cell cilia, δ-cells showed higher basal somatostatin secretion at low glucose and attenuated response to hyperglycemia, revealing the role of β-cell cilia in cross-regulating other endocrine cell types [42]. Inhibition of glucose-induced insulin secretion by somatostatin is impaired in absence of β-cell cilia, while augmentation of glucose-induced insulin secretion by glucagon is intact [42]. These results speak to the highly specialized sensing by the β-cell primary cilium, such that it may selectively mediate responsiveness to one paracrine hormone but not another, both present in high concentrations in the intra-islet space. In support of such specialization, β-cell cilia have been shown to express the somatostatin receptor 3 (SSTR3), as well as the insulin receptor (IR) under dynamic conditions [49,80], but todate there has been no report of ciliary localization of the glucagon receptor. Thus, along with juxtacrine signaling pathways that have been described in β-cell primary cilia, islet cells use their cilia to carry out multiple modes of communication including autocrine, juxtacrine, paracrine, and endocrine, spanning a diverse range of cellular signals. Taken together, β-cell primary cilia play a critical role in crosstalk with α- and δ-cells in maintaining glucose-mediated hormone secretion in the islets (Fig. 3). Given greater degree of heterotypic endocrine cell mingling in human islets than rodent islets [87,88], primary cilia likely play an even more complex role in regulating human islet hormone secretion and maintaining balanced α/β/δ-cell function.
Fig. 3.
Schematic depicting primary cilia regulation of hormone secretion and islet cell crosstalk. Genetic deletion of β-cell cilia leads to reduced glucose-stimulated insulin secretion via suggested mechanisms depicted in the box. β-Cell inhibition by δ-cell-derived somatostatin is also blocked in the β-cell specific cilia knockout (βCKO) model, suggesting that primary cilia on β-cells mediates paracrine somatostatin signaling. Meanwhile, β-cell response to glucagon appears to remain intact in βCKO islets, suggesting that some but not all paracrine signals are regulated by the β-cell primary cilium. EphA, epoxide hydrolase A; GPCR, G-protein coupled receptor.
CILIA-DEPENDENT SIGNALING IN PANCREATIC β-CELLS
Primary cilia have vast signaling capacity thanks to its abundant expression of surface receptors, signal transduction proteins, ion channels, and membrane-bound enzymes [6-8]. The enrichment of these molecules within the confines of a small subcellular domain increases the density of signals, making the primary cilium a unique sensor for extracellular cues and intercellular communication [6]. Primary cilia possess a phospholipid composition distinct from that of the plasma membrane, abundant in phosphatidylinositol-4-phosphate (PI(4)P) but deficient in phosphatidylinositol-4,5-bisphosphate (PI(4,5) P2), and localization of phosphatidylinositol-3,4,5-triphosphate (PI(3,4,5)P3) and PI(4,5)P2 at the ciliary base [89,90]. Differential distribution of polyphosphoinositides plays a key role in ciliary protein trafficking and signaling [91]. For example, tubby-like protein 3 (TULP3), a crucial regulator for ciliary trafficking of ion channels and GPCRs, interacts with protein cargo in a PI(4,5)P2-dependent manner [91,92]. The facilitated TULP3-cargo interaction under PI(4,5)P2 in the plasma membrane is weakened in the ciliary membrane lacking PI(4,5)P2. This enables the transport and release of cargo proteins from the plasma membrane to the cilia. Enrichment of PI(3,4,5)P3 and PI(4,5)P2 at the ciliary base contributes to the integrity and barrier function of the transition zone, which controls the entry and exit of ciliary protein [93-95].
A growing number of signaling pathways are coordinated by primary cilia, including sonic hedgehog (Shh), Notch, wingless/Int (Wnt), GPCRs, receptor tyrosine kinases, transforming growth factor-β, and insulin growth factor [6-8]. These signaling pathways have been studied in model organisms such as Drosophila, zebrafish, Chlamydomonas, or Caenorhabditis elegans, while mammalian data comes mostly from in cancer cell lines and mouse embryonic cells [7,8]. Specific to pancreatic cells, one established cilia-dependent signaling pathway is Shh signaling, whose activation is inhibited by primary cilium [82]. Full Shh activation is achieved only under cilium ablation in transgenic mice with pancreas-specific GLI family zinc finger 2 (GLI2) overexpression, a transcriptional mediator of Shh pathway [82]. Insulin signaling pathway via IR is also cilia- and context-dependent. One study has demonstrated that ciliary localization of IR is required for insulin signaling in β-cells, implicating a possible role for cilia in insulin-mediated autocrine or paracrine signaling [80]. In cultured mouse and human β-cells, IR-A is recruited to cilia under insulin stimulation, while basal body/ciliary disruption by Ofd1 or Bbs4 deletion mimics the phenotype of the loss of IR activity in stimulated β-cells [80].
GPCRs represent the largest and most diverse family of signaling receptors in the human body, responding to a variety of external signals including proteins, amino acids, peptides, ions, lipids, nucleotides, photons, and odorants [96,97]. GPCRs are among the most targetable structures for pharmacological modulation as they are accessible on the cell surface and play important roles in human pathophysiology [98]. Many GPCRs are selectively targeted to cilia on various mammalian cells which may confer a spatiotemporal advantage in their signaling [99], as it has been demonstrated that cyclic adenosine monophosphate (cAMP) generation selectively in the cilium in zebrafish and mammalian cells inhibits Shh signaling [100]. Similarly, γ-aminobutyric acid (GABA) receptor B1, recently found on primary cilia of islet cells, exhibits subcellular and even sub-organelle regulation of cilia-dependent signaling [101]. Cilia regulate local Ca2+ influx through GABAB1 receptors while isolating themselves against changes in the cytosolic Ca2+ concentrations through Ca2+ extrusion, and interestingly GABA regulation of ciliary Ca2+ is accomplished through cyclic guanosine monophosphate (cGMP) rather than cAMP [101]. In terms of hormone regulation, two GPCRs, free fatty acid receptor 4 (FFAR4) and prostaglandin E receptor 4 (PTGER4), have highlighted the significance of the cilia-dependent signaling in pancreatic β-cells [9]. Ciliary localization by these GPCRs in β- and α-cells enables ciliary cAMP signaling, and their cognate agonists regulate insulin and glucagon secretion with potencies on par with glucagon-like peptide 1 receptor agonists (GLP-1RAs), one of the most effective insulin secretagogues in clinical use today [9]. There is an additive effect on glucose-induced insulin secretion when FFAR4 agonists are treated with GLP-1RAs [9], suggesting that ciliary GPCR modulation may be an orthogonal therapeutic target to complement existing treatment strategies in diabetes. The SSTR3 is also localized to cilia in pancreatic β-cells [102-104], though the relative contribution of this particular SSTR isoform to the total β-cell paracrine response to somatostatin awaits clarification. Taken together, future investigations in ciliary GPCR signaling in pancreatic β-cells are expected to provide insights into novel therapeutic targets for diabetes and other pancreatic diseases.
CILIARY DYSFUNCTION AND DIABETES
Inherited defects in ciliary genes manifest as genetic ciliopathies in humans. Of these, two representative entities, BBS and Alström syndrome, exhibit prominent metabolic phenotypes including early-onset obesity, insulin resistance, and type 2 diabetes mellitus [105,106]. However, unlike highly penetrant obesity phenotype in both syndromes, the incidence of type 2 diabetes mellitus is significantly higher in Alström syndrome, at 50% to 75% for all ages and up to 82% in those over 16 years of age, compared to less than 25% for BBS [105,106]. In addition, the development of type 2 diabetes mellitus in Alström syndrome appears independent of the onset or severity of obesity [106]. These disparities imply that distinct pathogenesis other than obesity, such as β-cell function or mass, may contribute to diabetes development in affected individuals.
A growing body of evidence from animal and human studies has implicated multiple ciliary proteins in the regulation of metabolism and energy homeostasis. Mouse models of ciliopathy have shown impaired insulin secretion, aberrant islet hormonal regulation, and loss of β-cells over time, with varying degrees of severity and onset timing [40,42,79,80]. Male Bbs4 knockout mice with basal body/ciliary defects exhibit glucose intolerance prior to the onset of obesity [80]. Meanwhile, Ift88 knockout βCKO showed no obesity phenotype but had strong deficits in β-cell insulin secretion and glucose imbalance [42]. Dynamic association of islet cilia with diabetes has been found in diabetes-prone New Zealand Obese (NZO) and diabetesresistant B6.V-Lepobob (B6-ob/ob) mice [47]. NZO islets present fewer cilia under low glucose conditions and exhibit no changes after carbohydrate feeding while well-ciliated B6-ob/ob islets disassemble cilia under high glucose conditions [47]. Islet cilia genes are differentially expressed in B6-ob/ob mice compared to NZO mice and are implicated in cell cycle regulation [47], suggesting that functional cilia dynamics in islets are linked to β-cell proliferation under high glucose conditions and implicating a role for cilia in β-cell mass compensation in type 2 diabetes mellitus.
A number of islet cilia genes become differentially expressed in humans with type 2 diabetes mellitus, consistent with those described in NZO mice [47]. Abnormal glucose metabolism and diabetes phenotypes are described in several human ciliopathy disorders other than BBS and Alström syndrome (Table 3) [105-121]. Individuals with autosomal dominant polycystic kidney disease present abnormal glucose tolerance and decreased insulin secretion [107]. Pericentrin is a pericentriolar molecule that forms a complex with intraflagellar transport and polycystin-2 and is required for primary cilia assembly [122]. A genetic defect in pericentrin (PCNT), a gene encoding the pericentrin, causes early-onset of type 2 diabetes mellitus at an average age of 15 years [108,109]. Increasing depth of transcriptome studies are expected to reveal new ciliary gene linkages to human diabetes [47,48], where the molecular mechanism of individual cilia gene polymorphisms await mechanistic studies and showing the link between genetic variants of ciliary genes and risk of diabetes in humans [123-126].
Table 3.
Ciliopathies associated with human pancreas development and diabetes
| Ciliopathy | Gene | Inheritance | Affected ciliary structure | Clinical phenotype | T2DM prevalence | Reference |
|---|---|---|---|---|---|---|
| ADPKD | PKD1, PKD2 | AD | Axoneme | Renal and extra-renal cyst formation, loss of renal function, cardiovascular abnormalities, impaired glucose tolerance/insulin secretion | NA | [107,110-112] |
| Alström syndrome | ALMS1 | AR | Centrosome, basal body | Central obesity, T2DM, insulin resistance, hyperinsulinemia, hyperlipidemia, short stature, neurosensory deficits, hypogonadotropic hypogonadism, polycystic ovary syndrome, growth hormone deficiency, hypothyroidism, developmental delay, cardiomyopathy, progressive pulmonary, hepatic, and renal dysfunction | 50%–75% | [105,106,113] |
| Bardet-Biedl syndrome | BBS1-21, CEP164, SCLT1, SCAPER | AR | BBsome, basal body, chaperonin complex, centriole | Central obesity, T2DM, retinal rod-cone dystrophy, polydactyly, cognitive impairment, polycystic ovary syndrome, hypogonadism, genitourinary, renal, gastrointestinal, and dental abnormalities, congenital heart disease, developmental delay, ataxia, anosmia/hyposmia | <25% | [105,114-116] |
| MOPD II | PCNT | AR | Pericentrin | Growth retardation, severe insulin resistance, early-onset of T2DM, facial dysmorphism, bone and dental dysplasia, cerebrovascular abnormalities | 47% | [108,109] |
| Renal-hepatic-pancreatic dysplasia | NPHP3, NEK8/NPHP9 | AR | Axoneme, centrosome | Renal, hepatic, and pancreatic cyst, fibrosis, or dysplasia, congenital heart defects, hypoplastic lung, situs abnormalities | NA | [117,118] |
| OFD syndrome I | OFD1 | X-linked dominant | Basal body centriole, transition zone, axoneme | Abnormalities of the face, oral cavity, and digits, developmental and cognitive defects, pancreatic, renal, hepatic, and ovarian cysts | NA | [119] |
| VHL disease | VHL | AD | Axoneme | Pancreatic cyst (>70%) and neuroendocrine tumors, retinal or CNS hemangioblastomas, renal cysts, renal cell carcinoma, pheochromocytoma | NA | [120,121] |
T2DM, type 2 diabetes mellitus; ADPKD, autosomal dominant polycystic kidney disease; PKD1, polycystin 1; PKD2, polycystin 2; AD, autosomal dominant; NA, not available; ALMS1, Alstöm syndrome 1; AR, autosomal recessive; BBS1-21, Bardet-Biedl syndrome 21; CEP164, centrosomal protein 164; SCLT1, sodium channel and clathrin linker 1; SCAPER, S-phase cyclin A associated protein in the endoplasmic reticulum; MOPD II, microcephalic osteodysplastic primordial dwarfism type II; PCNT, pericentrin; NPHP3, nephrocystin 3; NEK8/NPHP9, NIMA related kinase 8/nephrocystin 9; OFD, oral-facial-digital; VHL, von Hippel-Lindau; CNS, central nervous system.
CONCLUSIONS
The view of primary cilia, once dismissed as rudimentary or “vestigial,” has been changing into one that is all-important for cellular communication and signal transduction. Primary cilia are now regarded as versatile sensors and interpreters of extracellular information, crucial for maintaining homeostasis and cell crosstalk. The accelerated pace of cilia research over the past two decades have led to important discoveries, also raised key questions about cilia action in pancreatic islets. Of particular relevance to islet β-cells, future studies should define the mechanism of nutrient sensing by cilia, how they convey intraislet signals to effect context-specific hormonal secretion, and how cell-to-cell communication might be mediated through cilia, thereby enhancing islet coordination as a unit. Our speculative opinion is that cilia may contribute to β-cell heterogeneity at multiple levels, not only morphologically but also functionally; and are likely controlled via ciliary length, number, motility, and trafficking of ciliary signaling proteins. An intriguing role for cilia in defining functional β-cell subsets was demonstrated in a recent study showing leader β-cells exhibit differential cilia gene expression [127]. Elucidating primary cilia roles in β-cell differentiation, especially in niche stem cell populations, might also inform treatment strategies targeting β-cell regeneration. Given the multi-faceted and dynamic roles that primary cilia play in cellular function and fate determination, future research at the junction of cilia and islet biology should generate new insights to treat human metabolic diseases including diabetes.
Acknowledgments
Graphics were created with BioRender.com.
Footnotes
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. NRF-2021R1C1C1013016) to Eun Young Lee; by the NIH (Grants DK115795A), National Institutes of Health grant DK115795A (Jing W. Hughes), National Institutes of Health grant DK127748 (Jing W. Hughes), Doris Duke Charitable Foundation grant DDFRCS (Jing W. Hughes), National Institutes of Health grant P30 DK 020579 (Washington University Diabetes Research Center) to Jing W. Hughes.
REFERENCES
- 1.Fliegauf M, Benzing T, Omran H. When cilia go bad: cilia defects and ciliopathies. Nat Rev Mol Cell Biol. 2007;8:880–93. doi: 10.1038/nrm2278. [DOI] [PubMed] [Google Scholar]
- 2.Van Leeuwenhoek A. Concerning little animals by him observed in rain-well-sea- and snow water: as also in water wherein pepper had lain infused. Phil Trans R Soc. 1677;12:821–31. [Google Scholar]
- 3.Zimmermann K. Beitrage zur Kenntnis einiger Drasen und Epithelien. Arch Mikrosk Anat. 1898;52:552–706. [Google Scholar]
- 4.Afzelius BA. A human syndrome caused by immotile cilia. Science. 1976;193:317–9. doi: 10.1126/science.1084576. [DOI] [PubMed] [Google Scholar]
- 5.Pazour GJ, Dickert BL, Vucica Y, Seeley ES, Rosenbaum JL, Witman GB, et al. Chlamydomonas IFT88 and its mouse homologue, polycystic kidney disease gene tg737, are required for assembly of cilia and flagella. J Cell Biol. 2000;151:709–18. doi: 10.1083/jcb.151.3.709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pala R, Alomari N, Nauli SM. Primary cilium-dependent signaling mechanisms. Int J Mol Sci. 2017;18:2272. doi: 10.3390/ijms18112272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nishimura Y, Kasahara K, Shiromizu T, Watanabe M, Inagaki M. Primary cilia as signaling hubs in health and disease. Adv Sci (Weinh) 2018;6:1801138. doi: 10.1002/advs.201801138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Anvarian Z, Mykytyn K, Mukhopadhyay S, Pedersen LB, Christensen ST. Cellular signalling by primary cilia in development, organ function and disease. Nat Rev Nephrol. 2019;15:199–219. doi: 10.1038/s41581-019-0116-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wu CT, Hilgendorf KI, Bevacqua RJ, Hang Y, Demeter J, Kim SK, et al. Discovery of ciliary G protein-coupled receptors regulating pancreatic islet insulin and glucagon secretion. Genes Dev. 2021;35:1243–55. doi: 10.1101/gad.348261.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yang DJ, Hong J, Kim KW. Hypothalamic primary cilium: a hub for metabolic homeostasis. Exp Mol Med. 2021;53:1109–15. doi: 10.1038/s12276-021-00644-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brewer KM, Brewer KK, Richardson NC, Berbari NF. Neuronal cilia in energy homeostasis. Front Cell Dev Biol. 2022;10:1082141. doi: 10.3389/fcell.2022.1082141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sun JS, Yang DJ, Kinyua AW, Yoon SG, Seong JK, Kim J, et al. Ventromedial hypothalamic primary cilia control energy and skeletal homeostasis. J Clin Invest. 2021;131:e138107. doi: 10.1172/JCI138107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bush A, Chodhari R, Collins N, Copeland F, Hall P, Harcourt J, et al. Primary ciliary dyskinesia: current state of the art. Arch Dis Child. 2007;92:1136–40. doi: 10.1136/adc.2006.096958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mujahid S, Hunt KF, Cheah YS, Forsythe E, Hazlehurst JM, Sparks K, et al. The endocrine and metabolic characteristics of a large Bardet-Biedl syndrome clinic population. J Clin Endocrinol Metab. 2018;103:1834–41. doi: 10.1210/jc.2017-01459. [DOI] [PubMed] [Google Scholar]
- 15.Collin GB, Marshall JD, Ikeda A, So WV, Russell-Eggitt I, Maffei P, et al. Mutations in ALMS1 cause obesity, type 2 diabetes and neurosensory degeneration in Alstrom syndrome. Nat Genet. 2002;31:74–8. doi: 10.1038/ng867. [DOI] [PubMed] [Google Scholar]
- 16.Cano DA, Murcia NS, Pazour GJ, Hebrok M. Orpk mouse model of polycystic kidney disease reveals essential role of primary cilia in pancreatic tissue organization. Development. 2004;131:3457–67. doi: 10.1242/dev.01189. [DOI] [PubMed] [Google Scholar]
- 17.Barker AR, Thomas R, Dawe HR. Meckel-Gruber syndrome and the role of primary cilia in kidney, skeleton, and central nervous system development. Organogenesis. 2014;10:96–107. doi: 10.4161/org.27375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fleming LR, Doherty DA, Parisi MA, Glass IA, Bryant J, Fischer R, et al. Prospective evaluation of kidney disease in Joubert syndrome. Clin J Am Soc Nephrol. 2017;12:1962–73. doi: 10.2215/CJN.05660517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ansley SJ, Badano JL, Blacque OE, Hill J, Hoskins BE, Leitch CC, et al. Basal body dysfunction is a likely cause of pleiotropic Bardet-Biedl syndrome. Nature. 2003;425:628–33. doi: 10.1038/nature02030. [DOI] [PubMed] [Google Scholar]
- 20.Engle SE, Bansal R, Antonellis PJ, Berbari NF. Cilia signaling and obesity. Semin Cell Dev Biol. 2021;110:43–50. doi: 10.1016/j.semcdb.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee CH, Kang GM, Kim MS. Mechanisms of weight control by primary cilia. Mol Cells. 2022;45:169–76. doi: 10.14348/molcells.2022.2046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang DJ, Tran LT, Yoon SG, Seong JK, Shin DM, Choi YH, et al. Primary cilia regulate adaptive responses to fasting. Metabolism. 2022;135:155273. doi: 10.1016/j.metabol.2022.155273. [DOI] [PubMed] [Google Scholar]
- 23.Pollara L, Sottile V, Valente EM. Patient-derived cellular models of primary ciliopathies. J Med Genet. 2022;59:517–27. doi: 10.1136/jmedgenet-2021-108315. [DOI] [PubMed] [Google Scholar]
- 24.Sun S, Fisher RL, Bowser SS, Pentecost BT, Sui H. Three-dimensional architecture of epithelial primary cilia. Proc Natl Acad Sci U S A. 2019;116:9370–9. doi: 10.1073/pnas.1821064116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kiesel P, Alvarez Viar G, Tsoy N, Maraspini R, Gorilak P, Varga V, et al. The molecular structure of mammalian primary cilia revealed by cryo-electron tomography. Nat Struct Mol Biol. 2020;27:1115–24. doi: 10.1038/s41594-020-0507-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xu CS, Pang S, Shtengel G, Muller A, Ritter AT, Hoffman HK, et al. An open-access volume electron microscopy atlas of whole cells and tissues. Nature. 2021;599:147–51. doi: 10.1038/s41586-021-03992-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Green WR. Abnormal cilia in human pancreas. Hum Pathol. 1980;11:686–7. doi: 10.1016/s0046-8177(80)80085-2. [DOI] [PubMed] [Google Scholar]
- 28.Bockman DE, Buchler M, Beger HG. Structure and function of specialized cilia in the exocrine pancreas. Int J Pancreatol. 1986;1:21–8. doi: 10.1007/BF02795236. [DOI] [PubMed] [Google Scholar]
- 29.Aughsteen AA. The ultrastructure of primary cilia in the endocrine and excretory duct cells of the pancreas of mice and rats. Eur J Morphol. 2001;39:277–83. doi: 10.1076/ejom.39.5.277.7380. [DOI] [PubMed] [Google Scholar]
- 30.Nachury MV, Mick DU. Establishing and regulating the composition of cilia for signal transduction. Nat Rev Mol Cell Biol. 2019;20:389–405. doi: 10.1038/s41580-019-0116-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang L, Dynlacht BD. The regulation of cilium assembly and disassembly in development and disease. Development. 2018;145:dev151407. doi: 10.1242/dev.151407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Heydeck W, Fievet L, Davis EE, Katsanis N. The complexity of the cilium: spatiotemporal diversity of an ancient organelle. Curr Opin Cell Biol. 2018;55:139–49. doi: 10.1016/j.ceb.2018.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hogan MC, Manganelli L, Woollard JR, Masyuk AI, Masyuk TV, Tammachote R, et al. Characterization of PKD protein-positive exosome-like vesicles. J Am Soc Nephrol. 2009;20:278–88. doi: 10.1681/ASN.2008060564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nager AR, Goldstein JS, Herranz-Perez V, Portran D, Ye F, Garcia-Verdugo JM, et al. An actin network dispatches ciliary GPCRs into extracellular vesicles to modulate signaling. Cell. 2017;168:252–63. doi: 10.1016/j.cell.2016.11.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wood CR, Rosenbaum JL. Ciliary ectosomes: transmissions from the cell’s antenna. Trends Cell Biol. 2015;25:276–85. doi: 10.1016/j.tcb.2014.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Volz AK, Frei A, Kretschmer V, de Jesus Domingues AM, Ketting RF, Ueffing M, et al. Bardet-Biedl syndrome proteins modulate the release of bioactive extracellular vesicles. Nat Commun. 2021;12:5671. doi: 10.1038/s41467-021-25929-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang J, Nikonorova IA, Silva M, Walsh JD, Tilton PE, Gu A, et al. Sensory cilia act as a specialized venue for regulated extracellular vesicle biogenesis and signaling. Curr Biol. 2021;31:3943–51. doi: 10.1016/j.cub.2021.06.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lacy PE. Electron microscopy of the normal islets of Langerhans: studies in the dog, rabbit, guinea pig and rat. Diabetes. 1957;6:498–507. doi: 10.2337/diab.6.6.498. [DOI] [PubMed] [Google Scholar]
- 39.Munger BL. A light and electron microscopic study of cellular differentiation in the pancreatic islets of the mouse. Am J Anat. 1958;103:275–311. doi: 10.1002/aja.1001030207. [DOI] [PubMed] [Google Scholar]
- 40.Ait-Lounis A, Baas D, Barras E, Benadiba C, Charollais A, Nlend Nlend R, et al. Novel function of the ciliogenic transcription factor RFX3 in development of the endocrine pancreas. Diabetes. 2007;56:950–9. doi: 10.2337/db06-1187. [DOI] [PubMed] [Google Scholar]
- 41.Zhang Q, Davenport JR, Croyle MJ, Haycraft CJ, Yoder BK. Disruption of IFT results in both exocrine and endocrine abnormalities in the pancreas of Tg737(orpk) mutant mice. Lab Invest. 2005;85:45–64. doi: 10.1038/labinvest.3700207. [DOI] [PubMed] [Google Scholar]
- 42.Hughes JW, Cho JH, Conway HE, DiGruccio MR, Ng XW, Roseman HF, et al. Primary cilia control glucose homeostasis via islet paracrine interactions. Proc Natl Acad Sci U S A. 2020;117:8912–23. doi: 10.1073/pnas.2001936117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cyge B, Voronina V, Hoque M, Kim EN, Hall J, Bailey-Lundberg JM, et al. Loss of the ciliary protein Chibby1 in mice leads to exocrine pancreatic degeneration and pancreatitis. Sci Rep. 2021;11:17220. doi: 10.1038/s41598-021-96597-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Phelps EA, Cianciaruso C, Santo-Domingo J, Pasquier M, Galliverti G, Piemonti L, et al. Advances in pancreatic islet monolayer culture on glass surfaces enable super-resolution microscopy and insights into beta cell ciliogenesis and proliferation. Sci Rep. 2017;7:45961. doi: 10.1038/srep45961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Greider MH, Elliott DW. Electron microscopy of human pancreatic tumors of islet cell origin. Am J Pathol. 1964;44:663–78. [PMC free article] [PubMed] [Google Scholar]
- 46.Hendley AM, Rao AA, Leonhardt L, Ashe S, Smith JA, Giacometti S, et al. Single-cell transcriptome analysis defines heterogeneity of the murine pancreatic ductal tree. Elife. 2021;10:e67776. doi: 10.7554/eLife.67776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kluth O, Stadion M, Gottmann P, Aga H, Jahnert M, Scherneck S, et al. Decreased expression of cilia genes in pancreatic islets as a risk factor for type 2 diabetes in mice and humans. Cell Rep. 2019;26:3027–36. doi: 10.1016/j.celrep.2019.02.056. [DOI] [PubMed] [Google Scholar]
- 48.Walker JT, Saunders DC, Rai V, Dai C, Orchard P, Hopkirk AL, et al. RFX6-mediated dysregulation defines human β cell dysfunction in early type 2 diabetes. bioRxiv. 2021 Dec 17; doi: 10.1101/2021.12.16.466282. [Preprint]. [DOI] [Google Scholar]
- 49.Cho JH, Li ZA, Zhu L, Muegge BD, Roseman HF, Lee EY, et al. Islet primary cilia motility controls insulin secretion. Sci Adv. 2022;8:eabq8486. doi: 10.1126/sciadv.abq8486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Orci L, Thorens B, Ravazzola M, Lodish HF. Localization of the pancreatic beta cell glucose transporter to specific plasma membrane domains. Science. 1989;245:295–7. doi: 10.1126/science.2665080. [DOI] [PubMed] [Google Scholar]
- 51.Tomita T. Immunocytochemical localization of glucose transporter-2 (GLUT-2) in pancreatic islets and islet cell tumors. Endocr Pathol. 1999;10:213–21. doi: 10.1007/BF02738882. [DOI] [PubMed] [Google Scholar]
- 52.Low JT, Zavortink M, Mitchell JM, Gan WJ, Do OH, Schwiening CJ, et al. Insulin secretion from beta cells in intact mouse islets is targeted towards the vasculature. Diabetologia. 2014;57:1655–63. doi: 10.1007/s00125-014-3252-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yamamoto M, Kataoka K. A comparative study on the intercellular canalicular system and intercellular junctions in the pancreatic islets of some rodents. Arch Histol Jpn. 1984;47:485–93. doi: 10.1679/aohc.47.485. [DOI] [PubMed] [Google Scholar]
- 54.Granot Z, Swisa A, Magenheim J, Stolovich-Rain M, Fujimoto W, Manduchi E, et al. LKB1 regulates pancreatic beta cell size, polarity, and function. Cell Metab. 2009;10:296–308. doi: 10.1016/j.cmet.2009.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Gan WJ, Zavortink M, Ludick C, Templin R, Webb R, Webb R, et al. Cell polarity defines three distinct domains in pancreatic β-cells. J Cell Sci. 2017;130:143–51. doi: 10.1242/jcs.185116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cottle L, Gan WJ, Gilroy I, Samra JS, Gill AJ, Loudovaris T, et al. Structural and functional polarisation of human pancreatic beta cells in islets from organ donors with and without type 2 diabetes. Diabetologia. 2021;64:618–29. doi: 10.1007/s00125-020-05345-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Geron E, Boura-Halfon S, Schejter ED, Shilo BZ. The edges of pancreatic islet β cells constitute adhesive and signaling microdomains. Cell Rep. 2015;10:317–25. doi: 10.1016/j.celrep.2014.12.031. [DOI] [PubMed] [Google Scholar]
- 58.Praetorius HA, Spring KR. Bending the MDCK cell primary cilium increases intracellular calcium. J Membr Biol. 2001;184:71–9. doi: 10.1007/s00232-001-0075-4. [DOI] [PubMed] [Google Scholar]
- 59.Nauli SM, Alenghat FJ, Luo Y, Williams E, Vassilev P, Li X, et al. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat Genet. 2003;33:129–37. doi: 10.1038/ng1076. [DOI] [PubMed] [Google Scholar]
- 60.Nauli SM, Kawanabe Y, Kaminski JJ, Pearce WJ, Ingber DE, Zhou J. Endothelial cilia are fluid shear sensors that regulate calcium signaling and nitric oxide production through polycystin-1. Circulation. 2008;117:1161–71. doi: 10.1161/CIRCULATIONAHA.107.710111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Nonaka S, Tanaka Y, Okada Y, Takeda S, Harada A, Kanai Y, et al. Randomization of left-right asymmetry due to loss of nodal cilia generating leftward flow of extraembryonic fluid in mice lacking KIF3B motor protein. Cell. 1998;95:829–37. doi: 10.1016/s0092-8674(00)81705-5. [DOI] [PubMed] [Google Scholar]
- 62.Takeda S, Yonekawa Y, Tanaka Y, Okada Y, Nonaka S, Hirokawa N. Left-right asymmetry and kinesin superfamily protein KIF3A: new insights in determination of laterality and mesoderm induction by kif3A-/- mice analysis. J Cell Biol. 1999;145:825–36. doi: 10.1083/jcb.145.4.825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Corbit KC, Aanstad P, Singla V, Norman AR, Stainier DY, Reiter JF. Vertebrate smoothened functions at the primary cilium. Nature. 2005;437:1018–21. doi: 10.1038/nature04117. [DOI] [PubMed] [Google Scholar]
- 64.Katoh TA, Omori T, Mizuno K, Sai X, Minegishi K, Ikawa Y, et al. Immotile cilia mechanically sense the direction of fluid flow for left-right determination. Science. 2023;379:66–71. doi: 10.1126/science.abq8148. [DOI] [PubMed] [Google Scholar]
- 65.Djenoune L, Mahamdeh M, Truong TV, Nguyen CT, Fraser SE, Brueckner M, et al. Cilia function as calcium-mediated mechanosensors that instruct left-right asymmetry. Science. 2023;379:71–8. doi: 10.1126/science.abq7317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sobkowicz HM, Slapnick SM, August BK. The kinocilium of auditory hair cells and evidence for its morphogenetic role during the regeneration of stereocilia and cuticular plates. J Neurocytol. 1995;24:633–53. doi: 10.1007/BF01179815. [DOI] [PubMed] [Google Scholar]
- 67.Schwartz EA, Leonard ML, Bizios R, Bowser SS. Analysis and modeling of the primary cilium bending response to fluid shear. Am J Physiol. 1997;272(1 Pt 2):F132–8. doi: 10.1152/ajprenal.1997.272.1.F132. [DOI] [PubMed] [Google Scholar]
- 68.Battle C, Ott CM, Burnette DT, Lippincott-Schwartz J, Schmidt CF. Intracellular and extracellular forces drive primary cilia movement. Proc Natl Acad Sci U S A. 2015;112:1410–5. doi: 10.1073/pnas.1421845112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kim S, Zaghloul NA, Bubenshchikova E, Oh EC, Rankin S, Katsanis N, et al. Nde1-mediated inhibition of ciliogenesis affects cell cycle re-entry. Nat Cell Biol. 2011;13:351–60. doi: 10.1038/ncb2183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Li A, Saito M, Chuang JZ, Tseng YY, Dedesma C, Tomizawa K, et al. Ciliary transition zone activation of phosphorylated Tctex-1 controls ciliary resorption, S-phase entry and fate of neural progenitors. Nat Cell Biol. 2011;13:402–11. doi: 10.1038/ncb2218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Jackson PK. Do cilia put brakes on the cell cycle? Nat Cell Biol. 2011;13:340–2. doi: 10.1038/ncb0411-340. [DOI] [PubMed] [Google Scholar]
- 72.Cano DA, Sekine S, Hebrok M. Primary cilia deletion in pancreatic epithelial cells results in cyst formation and pancreatitis. Gastroenterology. 2006;131:1856–69. doi: 10.1053/j.gastro.2006.10.050. [DOI] [PubMed] [Google Scholar]
- 73.Gallagher AR, Esquivel EL, Briere TS, Tian X, Mitobe M, Menezes LF, et al. Biliary and pancreatic dysgenesis in mice harboring a mutation in Pkhd1. Am J Pathol. 2008;172:417–29. doi: 10.2353/ajpath.2008.070381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Collin GB, Cyr E, Bronson R, Marshall JD, Gifford EJ, Hicks W, et al. Alms1-disrupted mice recapitulate human Alstrom syndrome. Hum Mol Genet. 2005;14:2323–33. doi: 10.1093/hmg/ddi235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lu W, Peissel B, Babakhanlou H, Pavlova A, Geng L, Fan X, et al. Perinatal lethality with kidney and pancreas defects in mice with a targetted Pkd1 mutation. Nat Genet. 1997;17:179–81. doi: 10.1038/ng1097-179. [DOI] [PubMed] [Google Scholar]
- 76.Morgan D, Turnpenny L, Goodship J, Dai W, Majumder K, Matthews L, et al. Inversin, PCNTft-right axis pathway, is partially deleted in the inv mouse. Nat Genet. 1998;20:149–56. doi: 10.1038/2450. [DOI] [PubMed] [Google Scholar]
- 77.Pierreux CE, Poll AV, Kemp CR, Clotman F, Maestro MA, Cordi S, et al. The transcription factor hepatocyte nuclear factor-6 controls the development of pancreatic ducts in the mouse. Gastroenterology. 2006;130:532–41. doi: 10.1053/j.gastro.2005.12.005. [DOI] [PubMed] [Google Scholar]
- 78.Woollard JR, Punyashtiti R, Richardson S, Masyuk TV, Whelan S, Huang BQ, et al. A mouse model of autosomal recessive polycystic kidney disease with biliary duct and proximal tubule dilatation. Kidney Int. 2007;72:328–36. doi: 10.1038/sj.ki.5002294. [DOI] [PubMed] [Google Scholar]
- 79.Volta F, Scerbo MJ, Seelig A, Wagner R, O’Brien N, Gerst F, et al. Glucose homeostasis is regulated by pancreatic β-cell cilia via endosomal EphA-processing. Nat Commun. 2019;10:5686. doi: 10.1038/s41467-019-12953-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gerdes JM, Christou-Savina S, Xiong Y, Moede T, Moruzzi N, Karlsson-Edlund P, et al. Ciliary dysfunction impairs beta-cell insulin secretion and promotes development of type 2 diabetes in rodents. Nat Commun. 2014;5:5308. doi: 10.1038/ncomms6308. [DOI] [PubMed] [Google Scholar]
- 81.Arsov T, Silva DG, O’Bryan MK, Sainsbury A, Lee NJ, Kennedy C, et al. Fat Aussie: a new Alstrom syndrome mouse showing a critical role for ALMS1 in obesity, diabetes, and spermatogenesis. Mol Endocrinol. 2006;20:1610–22. doi: 10.1210/me.2005-0494. [DOI] [PubMed] [Google Scholar]
- 82.Cervantes S, Lau J, Cano DA, Borromeo-Austin C, Hebrok M. Primary cilia regulate Gli/Hedgehog activation in pancreas. Proc Natl Acad Sci U S A. 2010;107:10109–14. doi: 10.1073/pnas.0909900107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Landsman L, Parent A, Hebrok M. Elevated Hedgehog/Gli signaling causes beta-cell dedifferentiation in mice. Proc Natl Acad Sci U S A. 2011;108:17010–5. doi: 10.1073/pnas.1105404108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Head WS, Orseth ML, Nunemaker CS, Satin LS, Piston DW, Benninger RK. Connexin-36 gap junctions regulate in vivo first- and second-phase insulin secretion dynamics and glucose tolerance in the conscious mouse. Diabetes. 2012;61:1700–7. doi: 10.2337/db11-1312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Konstantinova I, Nikolova G, Ohara-Imaizumi M, Meda P, Kucera T, Zarbalis K, et al. EphA-Ephrin-A-mediated beta cell communication regulates insulin secretion from pancreatic islets. Cell. 2007;129:359–70. doi: 10.1016/j.cell.2007.02.044. [DOI] [PubMed] [Google Scholar]
- 86.Kalwat MA, Thurmond DC. Signaling mechanisms of glucose-induced F-actin remodeling in pancreatic islet β cells. Exp Mol Med. 2013;45:e37. doi: 10.1038/emm.2013.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Cabrera O, Berman DM, Kenyon NS, Ricordi C, Berggren PO, Caicedo A. The unique cytoarchitecture of human pancreatic islets has implications for islet cell function. Proc Natl Acad Sci U S A. 2006;103:2334–9. doi: 10.1073/pnas.0510790103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Brissova M, Fowler MJ, Nicholson WE, Chu A, Hirshberg B, Harlan DM, et al. Assessment of human pancreatic islet architecture and composition by laser scanning confocal microscopy. J Histochem Cytochem. 2005;53:1087–97. doi: 10.1369/jhc.5C6684.2005. [DOI] [PubMed] [Google Scholar]
- 89.Nechipurenko IV. The enigmatic role of lipids in cilia signaling. Front Cell Dev Biol. 2020;8:777. doi: 10.3389/fcell.2020.00777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Conduit SE, Vanhaesebroeck B. Phosphoinositide lipids in primary cilia biology. Biochem J. 2020;477:3541–65. doi: 10.1042/BCJ20200277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Badgandi HB, Hwang SH, Shimada IS, Loriot E, Mukhopadhyay S. Tubby family proteins are adapters for ciliary trafficking of integral membrane proteins. J Cell Biol. 2017;216:743–60. doi: 10.1083/jcb.201607095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Mukhopadhyay S, Wen X, Chih B, Nelson CD, Lane WS, Scales SJ, et al. TULP3 bridges the IFT-A complex and membrane phosphoinositides to promote trafficking of G proteincoupled receptors into primary cilia. Genes Dev. 2010;24:2180–93. doi: 10.1101/gad.1966210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Dyson JM, Conduit SE, Feeney SJ, Hakim S, DiTommaso T, Fulcher AJ, et al. INPP5E regulates phosphoinositide-dependent cilia transition zone function. J Cell Biol. 2017;216:247–63. doi: 10.1083/jcb.201511055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Conduit SE, Ramaswamy V, Remke M, Watkins DN, Wainwright BJ, Taylor MD, et al. A compartmentalized phosphoinositide signaling axis at cilia is regulated by INPP5E to maintain cilia and promote sonic Hedgehog medulloblastoma. Oncogene. 2017;36:5969–84. doi: 10.1038/onc.2017.208. [DOI] [PubMed] [Google Scholar]
- 95.Reiter JF, Blacque OE, Leroux MR. The base of the cilium: roles for transition fibres and the transition zone in ciliary formation, maintenance and compartmentalization. EMBO Rep. 2012;13:608–18. doi: 10.1038/embor.2012.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Mykytyn K, Askwith C. G-protein-coupled receptor signaling in cilia. Cold Spring Harb Perspect Biol. 2017;9:a028183. doi: 10.1101/cshperspect.a028183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Schou KB, Pedersen LB, Christensen ST. Ins and outs of GPCR signaling in primary cilia. EMBO Rep. 2015;16:1099–113. doi: 10.15252/embr.201540530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Hauser AS, Attwood MM, Rask-Andersen M, Schioth HB, Gloriam DE. Trends in GPCR drug discovery: new agents, targets and indications. Nat Rev Drug Discov. 2017;16:829–42. doi: 10.1038/nrd.2017.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Hilgendorf KI, Johnson CT, Jackson PK. The primary cilium as a cellular receiver: organizing ciliary GPCR signaling. Curr Opin Cell Biol. 2016;39:84–92. doi: 10.1016/j.ceb.2016.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Truong ME, Bilekova S, Choksi SP, Li W, Bugaj LJ, Xu K, et al. Vertebrate cells differentially interpret ciliary and extraciliary cAMP. Cell. 2021;184:2911–26. doi: 10.1016/j.cell.2021.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Sanchez GM, Incedal TC, Prada J, O’Callaghan P, Dyachok O, Echeverry S, et al. The β-cell primary cilium is an autonomous Ca2+ compartment for paracrine GABA signaling. J Cell Biol. 2023;222:e202108101. doi: 10.1083/jcb.202108101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Iwanaga T, Miki T, Takahashi-Iwanaga H. Restricted expression of somatostatin receptor 3 to primary cilia in the pancreatic islets and adenohypophysis of mice. Biomed Res. 2011;32:73–81. doi: 10.2220/biomedres.32.73. [DOI] [PubMed] [Google Scholar]
- 103.O’Connor AK, Malarkey EB, Berbari NF, Croyle MJ, Haycraft CJ, Bell PD, et al. An inducible CiliaGFP mouse model for in vivo visualization and analysis of cilia in live tissue. Cilia. 2013;2:8. doi: 10.1186/2046-2530-2-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Li ZA, Cho JH, Woodhams LG, Hughes JW. Fluorescence imaging of beta cell primary cilia. Front Endocrinol (Lausanne) 2022;13:1004136. doi: 10.3389/fendo.2022.1004136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Girard D, Petrovsky N. Alstrom syndrome: insights into the pathogenesis of metabolic disorders. Nat Rev Endocrinol. 2011;7:77–88. doi: 10.1038/nrendo.2010.210. [DOI] [PubMed] [Google Scholar]
- 106.Marshall JD, Bronson RT, Collin GB, Nordstrom AD, Maffei P, Paisey RB, et al. New Alstrom syndrome phenotypes based on the evaluation of 182 cases. Arch Intern Med. 2005;165:675–83. doi: 10.1001/archinte.165.6.675. [DOI] [PubMed] [Google Scholar]
- 107.Pietrzak-Nowacka M, Safranow K, Byra E, Nowosiad M, Marchelek-Mysliwiec M, Ciechanowski K. Glucose metabolism parameters during an oral glucose tolerance test in patients with autosomal dominant polycystic kidney disease. Scand J Clin Lab Invest. 2010;70:561–7. doi: 10.3109/00365513.2010.527012. [DOI] [PubMed] [Google Scholar]
- 108.Rauch A, Thiel CT, Schindler D, Wick U, Crow YJ, Ekici AB, et al. Mutations in the pericentrin (PCNT) gene cause primordial dwarfism. Science. 2008;319:816–9. doi: 10.1126/science.1151174. [DOI] [PubMed] [Google Scholar]
- 109.Huang-Doran I, Bicknell LS, Finucane FM, Rocha N, Porter KM, Tung YC, et al. Genetic defects in human pericentrin are associated with severe insulin resistance and diabetes. Diabetes. 2011;60:925–35. doi: 10.2337/db10-1334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lee SH, Somlo S. Cyst growth, polycystins, and primary cilia in autosomal dominant polycystic kidney disease. Kidney Res Clin Pract. 2014;33:73–8. doi: 10.1016/j.krcp.2014.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kathem SH, Mohieldin AM, Nauli SM. The roles of primary cilia in polycystic kidney disease. AIMS Mol Sci. 2014;1:27–46. doi: 10.3934/molsci.2013.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Walker RV, Keynton JL, Grimes DT, Sreekumar V, Williams DJ, Esapa C, et al. Ciliary exclusion of polycystin-2 promotes kidney cystogenesis in an autosomal dominant polycystic kidney disease model. Nat Commun. 2019;10:4072. doi: 10.1038/s41467-019-12067-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Marshall JD, Maffei P, Collin GB, Naggert JK. Alstrom syndrome: genetics and clinical overview. Curr Genomics. 2011;12:225–35. doi: 10.2174/138920211795677912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Adam MP, Mirzaa GM, Pagon RA, Wallace SE, Bean LJH, Gripp KW, et al. Seattle: University of Washington, Seattle; 1993-2023. Gene Reviews. Chapter, Bardet-Biedl syndrome overview [cited 2023 Apr 6]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1363. [PubMed] [Google Scholar]
- 115.Forsythe E, Beales PL. Bardet-Biedl syndrome. Eur J Hum Genet. 2013;21:8–13. doi: 10.1038/ejhg.2012.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Suspitsin EN, Imyanitov EN. Bardet-Biedl syndrome. Mol Syndromol. 2016;7:62–71. doi: 10.1159/000445491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Bergmann C, Fliegauf M, Bruchle NO, Frank V, Olbrich H, Kirschner J, et al. Loss of nephrocystin-3 function can cause embryonic lethality, Meckel-Gruber-like syndrome, situs inversus, and renal-hepatic-pancreatic dysplasia. Am J Hum Genet. 2008;82:959–70. doi: 10.1016/j.ajhg.2008.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Frank V, Habbig S, Bartram MP, Eisenberger T, Veenstra-Knol HE, Decker C, et al. Mutations in NEK8 link multiple organ dysplasia with altered Hippo signalling and increased c-MYC expression. Hum Mol Genet. 2013;22:2177–85. doi: 10.1093/hmg/ddt070. [DOI] [PubMed] [Google Scholar]
- 119.Franco B, Thauvin-Robinet C. Update on oral-facial-digital syndromes (OFDS) Cilia. 2016;5:12. doi: 10.1186/s13630-016-0034-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Tirosh A, Sadowski SM, Linehan WM, Libutti SK, Patel D, Nilubol N, et al. Association of VHL genotype with pancreatic neuroendocrine tumor phenotype in patients with von Hippel-Lindau disease. JAMA Oncol. 2018;4:124–6. doi: 10.1001/jamaoncol.2017.3428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.van Asselt SJ, de Vries EG, van Dullemen HM, Brouwers AH, Walenkamp AM, Giles RH, et al. Pancreatic cyst development: insights from von Hippel-Lindau disease. Cilia. 2013;2:3. doi: 10.1186/2046-2530-2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Jurczyk A, Gromley A, Redick S, San Agustin J, Witman G, Pazour GJ, et al. Pericentrin forms a complex with intraflagellar transport proteins and polycystin-2 and is required for primary cilia assembly. J Cell Biol. 2004;166:637–43. doi: 10.1083/jcb.200405023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Censin JC, Nowak C, Cooper N, Bergsten P, Todd JA, Fall T. Childhood adiposity and risk of type 1 diabetes: a Mendelian randomization study. PLoS Med. 2017;14:e1002362. doi: 10.1371/journal.pmed.1002362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Grarup N, Moltke I, Andersen MK, Dalby M, Vitting-Seerup K, Kern T, et al. Loss-of-function variants in ADCY3 increase risk of obesity and type 2 diabetes. Nat Genet. 2018;50:172–4. doi: 10.1038/s41588-017-0022-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Saxena R, Hivert MF, Langenberg C, Tanaka T, Pankow JS, Vollenweider P, et al. Genetic variation in GIPR influences the glucose and insulin responses to an oral glucose challenge. Nat Genet. 2010;42:142–8. doi: 10.1038/ng.521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Dupuis J, Langenberg C, Prokopenko I, Saxena R, Soranzo N, Jackson AU, et al. New genetic loci implicated in fasting glucose homeostasis and their impact on type 2 diabetes risk. Nat Genet. 2010;42:105–16. doi: 10.1038/ng.520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Chabosseau P, Yong F, Delgadillo-Silva LF, Lee EY, Melhem R, Li S, et al. Molecular phenotyping of single pancreatic islet leader beta cells by “Flash-Seq”. Life Sci. 2023;316:121436. doi: 10.1016/j.lfs.2023.121436. [DOI] [PubMed] [Google Scholar]