Abstract
Objective: To investigate the anti-cerebral ischemia-reperfusion injury (CIRI) effect and mechanism of Zhenlong Xingnao capsules based on Notch/NF-κB signaling pathway. Methods: The rat model of middle cerebral artery occlusion (MCAO) was established using the Longa suture occlusion method, and 70 rats were divided into sham-operated, model, low dose Zhenlong Xingnao capsule group (125 mg/kg Zhenlong Xingnao capsule solution) and high dose Zhenlong Xingnao capsule group (250 mg/kg Zhenlong Xingnao capsule solution), low dose Zhenlong Xingnao capsule + neurogenic site notch homologous protein 1 (Notch1) antibody (Jagged1 group, 125 mg/kg capsule solution + 25 mg/kg Jagged1 solution), high dose Zhenlong Xingnao capsule + Jagged1 group (250 mg/kg capsule solution + 25 mg/kg Jagged1 solution), and Jagged1 group (25 mg/kg Jagged1 solution). The learning and memory abilities (behavioral score, spontaneous movement, and rotarod test), neurological function score, inflammatory factors and oxidative stress levels [interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), malondialdehyde (MDA), glutathione peroxidase (GSH-Px), superoxide dismutase (SOD)] in hippocampal tissue, and Bcl-2, Bax, and Caspase-3 mRNA levels were measured by reverse transcription quantitative polymerase chain reaction, and Notch1/NF-κB signaling pathway-related protein expression was assessed by Western blot. Results: The low and high dose interventions of Zhenlong Xingnao capsules significantly improved the learning and memory abilities of MCAO rats, reduced the neurological impairment scores, improved the levels of IL-6, TNF-α, MDA, GSH-Px, SOD, and inhibited the expression levels of Notch1, p-NF-κB p65, and Hes-1 proteins. However, the protective effect of Zhenlong Xingnao capsules on neurons in rat brain tissue could be reduced after treatment with Jagged1. Conclusions: Zhenlong Xingnao capsules can promote neuronal repair during ischemia-reperfusion, and its mechanism may be related to inhibiting the activation of Notch/NF-κB signaling pathway and reducing inflammation and oxidative stress response.
Keywords: Zhenlong Xingnao capsules, ischemia-reperfusion injury, Notch, inflammation, oxidative stress
Introduction
Cerebral ischemia/reperfusion injury (CIRI) is a prevalent secondary injury in the pathogenesis of ischemic stroke [1], leading to an exacerbation of pathological processes such as oxidative stress, neuroinflammation, and neuronal apoptosis. As a result, patients experience impaired neurological function as well as cognitive, memory, and motor deficits [2]. The Notch/NF-κB signaling pathway is a highly evolutionarily conserved pathway, which plays a crucial role in a series of pathophysiological processes such as cell proliferation, differentiation, individual growth and development, and apoptosis [3-5]. Meanwhile, Notch1 signal axis is an important signal transduction pathway in oxidative stress response. Nrf2 contains leucine zipper protein, which plays a key role in cellular oxidative stress damage. Ren et al. [6] have shown that activation of Notch signaling pathway can reduce trophoblast apoptosis caused by oxidative stress, and its mechanism may be related to the cytoprotective effect by reducing cellular ROS levels, sFlt-1 and VEGF expression. Studies [7,8] have demonstrated that increased Notch1 expression after cerebral ischemia can activate NF-κB phosphorylation and nuclear translocation, enhance the release of pro-inflammatory cytokines, and aggravate the inflammatory response. Additionally, the downstream gene Hes1 increases the expression and accelerates the release of inflammatory factors, while Jagged1 can elevate Notch1 expression, thereby aggravating ischemic injury.
Zhenlong Xingnao capsules are a classic Chinese formula for the treatment of “white vein disease”, widely used in the treatment of hypertension, coronary heart disease, stroke, and other cardiovascular and cerebrovascular diseases with remarkable efficacy [9,10]. Yu et al. [10] revealed that Zhenlong Xingnao capsules combined with butylphthalide had significant clinical efficacy in the treatment of acute cerebral infarction, which can significantly improve the symptoms of neurological injury, reduce the degree of neurological deficits in patients, and inhibit the inflammatory response of the body. This combination demonstrated clinical safety and efficacy; however, the specific mechanism remains unclear. Therefore, in this study, a rat model of focal cerebral ischemia-reperfusion was established to investigate the effect of Zhenlong Xingnao capsules on ameliorating CIRI through the Notch/NF-κB signaling pathway, so as to elucidate the neuroprotective mechanism of Zhenlong Xingnao capsules.
Materials and methods
Experimental animals
Seventy specific pathogen-free (SPF) adult male Sprague-Dawley (SD) rats with body weight of 300±20 g were purchased from Hunan Slaughter Jingda Laboratory Animal Co. All rats were provided ad libitum access to water and housed indoor at 23±3°C with a relative humidity of 45-55%. The experiment was approved by the Medical Ethics Committee of Jiangxi University of Technology.
Model construction and drug treatment
The middle cerebral artery occlusion (MCAO) model was replicated according to the Longa suture occlusion method [11]. Rats were anesthetized with intraperitoneal injection of 2.5% pentobarbital at a dosage of 2 mL/kg. A median cervical incision was made with a length of 1.5 cm, and the right common carotid artery (CCA), external carotid artery (ECA), and internal carotid artery (ICA) were gently separated. The wire embolus was introduced through ECA and slowly pushed into ICA, and the head of the wire embolus was adjusted to block the origin of the middle cerebral artery (MCA), causing occlusive ischemia. After 2 hours, the wire plug was gently withdrawn, successfully inducing MCA reperfusion. In the sham-operated group, only the right CCA was isolated, without inserting wire plugs. The rats regained consciousness postoperatively after modeling.
After modeling, the rats were treated after 2 h of wakefulness. The rats in the low and high dose Zhenlong Xingnao capsule groups were given 125 and 250 mg/kg of Zhenlong Xingnao capsule (Jinha Tibetan Medicine Co., Ltd., lot no. 201901, specification: 0.3 g per capsule) solution respectively by gavage, once in the morning and once in the evening, for 7 days. The rats in the low and high dose Zhenlong Xingnao capsule + Jagged1 groups were given 125 and 250 mg/kg of the capsule solution respectively by gavage, once in the morning and once in the evening for 7 days, and after the rats were stabilized for 10 min, 25 mg/kg of Jagged1 solution (MCE, item no. HY-P1846A) was intraperitoneally injected once every other day for 7 days. The rats in the Jagged1 group were intraperitoneally injected with 25 mg/kg Jagged1 solution once every other day for 7 days. Rats in sham-operated and model groups were gavaged with equal amounts of normal saline.
Neurological impairment scores
On the 7th day after modeling, the neurological impairment scores of rats in each group were assessed according to the Longa 5-point deficit scale [11]. A score of 0 indicated normal neurological function and normal behavioral activity; a score of 1 indicated left forelimb flexion during the tail suspension test; a score of 2 indicated rotation of the body to the left while crawling; a score of 3 indicated leaning to the left during rest or movement; a score of 4 indicated decreased consciousness and lack of spontaneous limb movement; and a score of 5 indicated no response to stimuli or death. Higher neurological impairment scores indicated more severe behavioral impairments and neurological deficits of the rats.
Learning motor ability assessment
On the 7th day after modeling, the rats were placed in a quiet room for the open field test. The rats were placed in a 100 cm × 100 cm × 40 cm uncovered chamber with blackened walls and floor. The chamber was divided into 25 equal compartments, with the outer compartments along the walls and the central compartments comprising the remaining area. The rats were placed in a central compartment, and their activities were monitored with a camera, focusing on the total number of times the rats crossed the central compartment and stood upright on their hind legs within 3 min.
The rotarod test required the rat to stand and balance on a uniformly rotating rod, and the time the rat maintained on the rotating rod was recorded. Before starting all tests, the rats were placed in the testing room for 15 min to acclimate. The entire test was performed three times, with a 15-min interval between each test. The rats were trained for 3 days before the procedure to ensure that each rat could move on the apparatus for 3 min. The constant speed of the apparatus was set at 40 rotations per minute.
Morris water maze experiment
The Morris water maze experiment for rats was mainly conducted in a black circular pool of 200 cm × 60 cm, with a water temperature of 22±2°C. A 10 cm diameter platform was placed 2 cm below the water surface. An automatic image acquisition and processing system was connected, and the data were analyzed using the MT-200 water maze system. The experimental procedures were as follows.
Spatial navigation test: This test was performed 3 days after the rat modeling, and the time the rat took to climb onto the platform was recorded. If the rat did not find the platform within 60 s, it was guided to the platform for 30 s, and the escape latency was recorded as 60 s. The experiment lasted for 3 days. The rats were trained 3 times a day for 60 s each time, and the average escape latency for each group was calculated to assess the ability of rats to remember spatial locations.
Spatial exploration: This test assessed the ability of rats to remember the spatial location of the platform after learning to find it. After 3 days of spatial navigation tests, the platform was removed, and the rats were placed into the water in quadrant 1 facing the wall. They were allowed to swim for 60 s. The number of times the animals passed the original platform position and the percentage of time spent in the target quadrant were recorded.
Observation of hippocampal neuronal injury by hematoxylin and eosin (HE) staining
The rats were anesthetized with an intraperitoneal injection of 3% pentobarbital sodium. Brain tissue was taken from the decapitated head and fixed with 4% paraformaldehyde. A 3 mm coronal section containing the hippocampus was taken from both the anterior and posterior parts of the brain. The tissue was dehydrated, cleared, and embedded in paraffin. Sections with a thickness of 4 μm were prepared, deparaffinized, and stained with HE (with the HE kit purchased from Beijing Solabao Technology Co., Ltd.) to observe hippocampal neuronal injury in rats.
Observation of cerebral infarction size in rats by TTC staining
At 7 days after treatment, the rats were anesthetized by intraperitoneal injection of 3% pentobarbital sodium. Brain tissue was taken from the decapitated head and frozen at -20°C for 15 min. The brain was cut into 4 pieces from 2 mm of the frontal end on ice, quickly placed in 2% TTC dye solution, incubated for 20 min at 37°C, and then fixed with 4% paraformaldehyde for 24 h. The infarcted area was white and the normal area was red, and the percentage of infarcted area in each group of brain tissue was calculated by ImageJ software with the following formula: percentage of infarcted area (%) = (section infarcted area × thickness)/(section area × thickness) × 100%.
Detection of inflammatory factors and oxidative stress indicators in hippocampal tissues using ELISA
After anesthetizing the rats, hippocampal tissues were isolated. The levels of interleukin 6 (IL-6), tumor necrosis factor α (TNF-α), malondialdehyde (MDA), and the activities of glutathione peroxidase (GSH-Px) and superoxide dismutase (SOD) in hippocampal tissues were detected using enzyme-linked immunosorbent assay (ELISA) kits (Wuhan Doctor Bio Co., Ltd.).
Detection of B-cell lymphoma-2 (Bcl-2), Bax, and Caspase-3 mRNA levels by RT-qPCR
Hippocampal tissues of rats in each group were extracted with Trizol lysate for total RNA isolation. RevertAidTM First Strand cDNA Synthesis Kit was used as a quantitative fluorescence template for cDNA synthesis. The primers were designed and synthesized by Takara Corporation, Japan, with the primer sequences as followings: Bcl-2: upstream sequence (5’→3’) GTCACAGAGGGGCTACGAGT, downstream sequence (5’→3’) CAGGCTGGAAGGAGAAGATG; Bax: upstream sequence (5’→3’) ATGGCTGGGGAGACACCTGA, downstream sequence (5’→3’) TGGGCGTCCCGAAGTAGGAA; Caspase-3: upstream sequence (5’→3’) ATGTCGATGCAGCTAACCTC, downstream sequence (5’→3’) AACTGCTCCTTTTGCTGTGA; GAPDH: upstream sequence (5’→3’) TGCTTCACCACCTTCTTGA, downstream sequence (5’→3’) TCACCATCTTCCAGGAGC.
Reaction system: 0.5 μL dNTPs + 5 μL 5 × Buffer + 0.3 μL Taq enzyme + 1.5 μL MgCl2 + 2 μL cDNA template + 1 μL of each upstream and downstream primer, with deionized water added to a total volume of 25 μL.
Reaction conditions: Pre-denaturation at 95°C for 5 min, denaturation at 95°C for 30 s, annealing at 62°C for 30 s, extension at 72°C for 30 s, with 40 cycles, followed by extension at 72°C for 10 min, and termination at 4°C for 5 min. The relative expression level of the target gene mRNA was calculated using the 2-ΔΔCT method.
Detection of protein expression of Notch1/NF-κB signaling pathway using Western blot
Hippocampal tissues of rats were extracted after grinding and homogenizing. The protein concentration was determined using a bicinchoninic acid (BCA) protein quantification kit (Thermo Fisher Scientific Co., Ltd.). After quantification, loading buffer was added to the protein, mixed in an EP tube, and boiled in boiling water for 5 minutes to denature the protein. The polyvinylidene fluoride (PVDF) membrane was placed into primary antibody (CST, USA) dilution solution with a dilution ratio of 1:2000, and incubated overnight at 4°C. Then, goat anti-rabbit secondary antibody (1:1000) (CST, USA) was added, followed by incubation for 2 h at 37°C. The membrane was washed three times with TBST buffer and ECL luminescent solution was added dropwise. After 1 min of reaction, the membrane was placed on a gel imaging system. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal reference protein for Western blot analysis, and the corresponding grayscale values of each protein were analyzed using ImageJ software to calculate the relative protein expression. Relative protein expression = grayscale value of target protein/grayscale value of internal reference protein.
Statistical analysis
The Statistical Package for the Social Sciences (SPSS) 25.0 software was used to analyze the data, with measurement data expressed as mean ± standard deviation (x̅ ± sd). One-way analysis of variance (ANOVA) was used for the comparison of multiple samples, and the Student-Newman-Keuls (SNK)-q test was used for the comparison of two samples. P<0.05 was considered statistically significant.
Results
The effect of Zhenlong Xingnao capsules on the learning and motor ability of rats
Compared with the model group, the behavioral scores and spontaneous movements in the low and high dose groups of Zhenlong Xingnao capsules were significantly decreased, while the motor ability on the rotarod was significantly increased, and the difference was statistically significant (P<0.05). However, the effect of Zhenlong Xingnao capsules on learning and motor ability of rats was weakened after treatment with Jagged1 (P<0.05, Figure 1).
Figure 1.
Effect of Jenlong Awakening capsules on the learning motor ability of rats. It shows that the (A) behavioral scores and (B) spontaneous movement levels of rats in the low and high dose groups of Zhenlong Xingnao capsules were significantly reduced and (C) the level of the motor ability on the rotarod was significantly increased, but the treatment with Jagged1 could weaken the improvement effect of Zhenlong Xingnao capsules. Note: aindicates comparison with the sham-operated group, P<0.05; bindicates comparison with the model group, P<0.05; cindicates comparison with the low-dose group of Zhenlong Xingnao capsules, P<0.05; dindicates comparison with the high-dose group of Zhenlong Xingnao capsules, P<0.05.
Effects of Zhenlong Xingnao capsules on the learning and memory ability of rats
In the spatial navigation training, the escape latency time of rats in the low and high dose groups of Zhenlong Xingnao capsules was significantly decreased compared with the model group (P<0.05). In the spatial exploration experiment, the activity time and the number of crossing platforms in the original platform quadrant were significantly decreased in the model group (P<0.05), while the activity time and the number of crossing platforms in the target quadrant were significantly increased in the low and high dose groups of Zhenlong Xingnao capsules (P<0.01). However, the effect of Zhenlong Xingnao capsules on learning and memory ability of rats was weakened after treatment with Jagged1 (P<0.01, Table 1).
Table 1.
The effect of Zhenlong Xingnao capsules on the learning and memory ability of rats
| Group | Escape latency/S | Percentage of time spent in the target quadrant/% | Crossing the platform/times | ||
|---|---|---|---|---|---|
|
| |||||
| Day 1 | Day 2 | Day 3 | |||
| Sham surgery group | 40.84±2.72 | 20.35±1.58 | 13.73±1.66 | 51.84±3.73 | 5.22±0.21 |
| Model group | 58.72±1.69a | 45.88±1.92a | 36.95±1.55a | 22.09±0.91a | 0.72±0.31a |
| Low dose group | 46.47±2.55b | 35.74±3.11b | 30.75±1.22b | 39.18±2.88b | 4.58±0.37b |
| High Dose Group | 43.10±2.48b,c | 31.68±2.97b,c | 28.55±0.82b,c | 36.09±2.31b,c | 3.95±0.29b,c |
| low dose + Jagged1 group | 51.69±2.38c | 40.42±2.59c | 33.69±1.95c | 33.54±2.72c | 2.75±0.51c |
| High dose + Jagged1 group | 49.10±2.62d | 38.75±2.63d | 31.73±1.57d | 32.93±2.41d | 3.08±0.52d |
| Jagged1 group | 54.72±2.52c,d | 42.42±2.19c,d | 34.66±2.01c,d | 29.66±1.95c,d | 2.91±0.59c,d |
indicates comparison with the sham-operated group, P<0.05;
indicates comparison with the model group, P<0.05;
indicates comparison with the low-dose group, P<0.05;
indicates comparison with high-dose group, P<0.05.
Effects of Zhenlong Xingnao capsules on neurological function score and cerebral infarction area in rats
Compared with the sham-operated group, the neurological impairment scores of rats in the model group were significantly increased (P<0.05). The neurological function scores and cerebral infarction area of rats in the low and high dose groups of Zhenlong Xingnao capsules were significantly lower than those in the model group (P<0.05). However, after treatment with Jagged1, the protective effect of Zhenlong Xingnao capsules on neurons of rats was weakened, and the area of cerebral infarction was increased (P<0.05, Figure 2; Table 2).
Figure 2.

Comparison of cerebral infarction volume in each group. Note: (A) Sham-operated group; (B) Model group; (C) High-dose group; (D) High-dose + Jagged1 group; (E) Jagged1 group.
Table 2.
The effect of Zhenlong Xingnao capsules on the neurological impairment scores of rats
| Group | Neurological impairment score | Cerebral infarction area |
|---|---|---|
| Sham-operated group | - | - |
| Model group | 2.13±0.72a | 29.53±2.09a |
| Low dose group | 1.56±0.69b | 8.15±0.79b |
| High Dose Group | 1.38±0.71b,c | 6.82±0.51b,c |
| Low dose + Jagged1 group | 1.83±0.63c | 15.09±1.22c |
| High dose + Jagged1 group | 1.74±0.72d | 13.22±0.81d |
| Jagged1 group | 2.03±0.88c,d | 27.69±1.66c,d |
indicates comparison with sham-operated group, P<0.05;
indicates comparison with model group, P<0.05;
indicates comparison with low-dose group, P<0.05;
indicates comparison with high-dose group, P<0.05.
Effect of Zhenlong Xingnao capsules on the neuronal damage of hippocampus in rats
HE staining results showed that the hippocampal neurons in the sham-operated group were structurally intact, densely arranged, with clear nuclei and uniform staining. In the model group, the structure of hippocampal neurons was damaged, the nuclei were deformed, shrunken and broken, the staining was deepened, the cells were loosely arranged, and cavities were formed. The damage of hippocampal neurons was significantly improved in the low and high dose groups (Figure 3).
Figure 3.
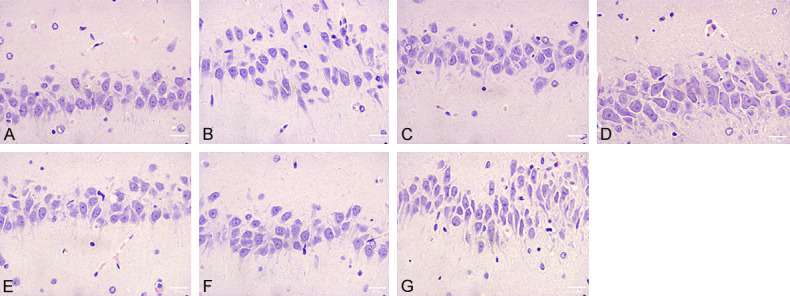
Hippocampal neuronal injury in rats (× 400). It shows that in the model group, the structure of hippocampal neurons was damaged, the nuclei were deformed, shrunken and broken, the staining was deepened, the cells were loosely arranged, and cavities were formed. The damage of hippocampal neurons was significantly improved in the low and high dose groups. Note: (A) Sham-operated group; (B) Model group; (C) Low-dose group; (D) High-dose group; (E) Low-dose + Jagged1 group; (F) High-dose + Jagged1 group; (G) Jagged1 group.
Effect of Zhenlong Xingnao capsules on the inflammation and oxidative stress of hippocampal tissue in rats
Compared with the sham-operated group, the levels of IL-6, TNF-α and MDA in the hippocampal tissue of rats in the model group were significantly increased, while the activities of GSH-Px and SOD were significantly decreased, and the differences were statistically significant (P<0.05). Compared with the model group, the levels of IL-6, TNF-α and MDA were significantly decreased, while the activities of GSH-Px and SOD were significantly increased in the low and high dose groups of Zhenlong Xingnao capsules, and the differences were statistically significant (P<0.05). However, after treatment with Jagged1, the improvement effect of Zhenlong Xingnao capsules on inflammation and oxidative stress in hippocampal tissues of rats was weakened in each treatment group (P<0.05, Figure 4).
Figure 4.
Effect of Zenglong Xingnao capsules on the level of inflammation and oxidative stress in hippocampal tissue of rats. It shows that the levels of (A) IL-6, (B) TNF-α, (C) MDA, (D) GSH-Px, and (E) SOD activity in hippocampal tissue were significantly decreased and increased in the low and high dose groups, but the treatment with Jagged1 could weaken the ameliorative effect of Zhenlong Xingnao capsules. Note: aindicates comparison with the sham-operated group, P<0.05; bindicates comparison with the model group, P<0.05; cindicates comparison with the low-dose group, P<0.05; dindicates comparison with the high-dose group, P<0.05.
Effect of Zhenlong Xingnao capsules on the expression of apoptosis-related genes in hippocampal tissue of rats
Compared with the sham-operated group, the mRNA expressions of Bax and Caspase-3 in hippocampal tissue of rats in the model group were significantly increased, while the mRNA expression of Bcl-2 was significantly decreased (P<0.05). Compared with the model group, the mRNA expressions of Bax and Caspase-3 were significantly decreased, while the mRNA expression of Bcl-2 was significantly increased in the low and high dose groups of Zhenlong Xingnao capsules, and the differences were statistically significant (P<0.05). However, after treatment with Jagged1, the improvement effect of Zhenlong Xingnao capsules on apoptosis-related genes in rat hippocampal tissue was weakened in each treatment group (P<0.05) (Figure 5).
Figure 5.
Effect of Zenglong Xingnao capsules on the expression of apoptosis-related genes in hippocampal tissues of rats. It shows that the levels of (A) Bcl-2, (B) Bax, and (C) Caspase-3 in hippocampal tissues were significantly decreased and increased in the low and high dose groups, but the treatment with Jagged1 weakened the ameliorative effect of the capsules. Note: aindicates comparison with the sham-operated group, P<0.05; bindicates comparison with the model group, P<0.05; cindicates comparison with the low-dose group, P<0.05; dindicates comparison with the high-dose group, P<0.05.
Expression of Notch1/NF-κB signaling pathway-related proteins in hippocampal tissues of rats
Compared with the sham-operated group, the protein expression levels of Notch1, p-NF-κB p65, and Hes-1 in hippocampal tissues of the model group was significantly increased (P<0.05), but the difference of NF-κB p65 protein expression level was not statistically significant (P>0.05). Compared with the model group, the protein expression levels of Notch1, p-NF-κB p65, and Hes1 were significantly decreased in the low and high dose groups of Zhenlong Xingnao capsules (P<0.05), but the differences in NF-κB p65 protein expression levels were not statistically significant (P>0.05). However, after treatment with Jagged1, the improvement of Notch1/NF-κB signaling pathway-related proteins in hippocampal tissues of rats was weakened in each treatment group of Zhenlong Xingnao capsules (P<0.05) (Figure 6).
Figure 6.
Effect of Zenglong Xingnao capsules on the expression of Notch1/NF-κB signaling pathway-related proteins in hippocampal tissue of rats. (A) Western Blot images of expression of Notch1/NF-κB signaling pathway-related proteins. It shows that (B) Notch1, (D) p-NF-κB p65, and (E) Hes-1 protein expression levels in hippocampal tissues were significantly reduced in the low and high dose groups of Zhenlong Xingnao capsules, but the treatment with Jagged1 could weaken the improvement effect of Zhenlong Xingnao capsules; the difference of NF-κB p65 protein expression level in each group (C) was not statistically significant. Note: aindicates comparison with the sham-operated group, P<0.05; bindicates comparison with the model group, P<0.05; cindicates comparison with the low-dose group, P<0.05; dindicates comparison with the high-dose group, P<0.05.
Discussion
Numerous studies [12-14] have highlighted the critical role of inflammation and oxidative stress responses in brain tissue in the development of brain damage, motor, and cognitive neurological abnormalities in patients with ischemic stroke. These responses can also serve as indirect indicators of the extent of brain damage in patients. In this study, the rat model of MCAO was successfully replicated by Longa suture occlusion method. The results demonstrated that the brain tissue of the model group was significantly damaged, which promoted the production of inflammatory factors and oxidative stress, confirming the success of the modeling.
Zhenlong Xingnao capsules were used as an intervention, and it was observed that different doses of drug interventions significantly improved the neurological impairment, reduced hippocampal neuron damage, and enhanced learning memory and motor abilities in rats. These findings suggest that Zhenlong Xingnao capsules have notable protective effects on CIRI in rats.
In previous researches [15,16], it has been shown that mitigating oxidative stress and inflammatory responses can considerably reduce neuronal apoptosis in patients with ischemic cerebral perfusion injury. The approach can also effectively decrease the neurological deficits in rats with CIRI. Our study revealed that Zhenlong Xingnao capsules intervention could significantly reduce the levels of IL-6, TNF-α, and MDA in hippocampal tissue, increase the activities of GSH-Px and SOD, and specifically change mRNA expression, decrease Bax and Caspase-3 and increase Bcl-2. These results indicated that Zhenlong Xingnao capsules could substantially ameliorate free radical damage, inflammatory damage, and apoptosis in CIRI.
Existing literature [3,17] has pointed out that the Notch1/NF-κB signaling pathway plays a pivotal role in ischemic stroke pathogenesis. During cerebral ischemia-reperfusion, the expression level of Notch1 in hippocampal tissue is significantly increased. Notch1 promotes the phosphorylation of NF-κB p65, leading to nuclear translocation, accelerated inflammatory cell infiltration, and increased release of inflammatory factors, which exacerbate neuronal damage during cerebral ischemia [18,19]. Hes1 is a downstream molecule of the Notch1 pathway, which may directly reflect Notch1 activation [20]. Our findings indicated that Zhenlong Xingnao capsules significantly reduced the expression levels of Notch1, p-NF-κB p65, and Hes1 proteins, suggesting that the capsules may protect against CIRI by inhibiting the Notch/NF-κB signaling pathway. To further verify whether Zhenlong Xingnao capsules act through this pathway, the Notch1 inhibitor Jagged1 was employed. The results revealed that after treatment with Jagged1, the improvement effect of Zhenlong Xingnao capsules on neurological function and hippocampal neuronal injury was weakened.
However, this study also has the following deficiencies. The study only evaluated the immediate effect of Notch/NF-κB signaling pathway, and in subsequent studies, the therapeutic observation period should be extended to further study the correlation between Zhenlong Xingnao capsules and oxidative stress. Meanwhile, the process of inflammatory injury and apoptosis implicates multiple signaling pathways. In future research, the protective effects of related signaling pathways on CIRI will be further explored. In addition, Zhenlong Xingnao capsules are a compound preparation, and its exact active ingredients have not been discovered, which should be explored and analyzed in the next research.
In conclusion, Zhenlong Xingnao capsules can markedly enhance neurological recovery and exert neuroprotective effects in rats with CIRI. The underlying mechanism may be related to the downregulation of Notch1 expression and the inhibition of Notch1/NF-κB signaling pathway activation.
Acknowledgements
This research is funded by Natural Science Foundation Project of Jiangxi Provincial Science and Technology Department (No. 20202BABL206160) and Science and Technology Project of Jiangxi Provincial Health Commission (No. 202311154).
Disclosure of conflict of interest
None.
References
- 1.Li Y, Yi M, Wang D, Zhang Q, Yang L, Yang C. LncRNA KCNQ1OT1 regulates endoplasmic reticulum stress to affect cerebral ischemia-reperfusion injury through targeting miR-30b/GRP78. Inflammation. 2020;43:2264–2275. doi: 10.1007/s10753-020-01295-w. [DOI] [PubMed] [Google Scholar]
- 2.Fangma Y, Zhou H, Shao C, Yu L, Yang J, Wan H, He Y. Hydroxysafflor yellow A and anhydrosafflor yellow B protect against cerebral ischemia/reperfusion injury by attenuating oxidative stress and apoptosis via the silent information regulator 1 signaling pathway. Front Pharmacol. 2021;12:739864. doi: 10.3389/fphar.2021.739864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hong Q, Ye J, Wang X, Zhang C. The mechanism of gastrodin participating in improving the cerebral ischemia-reperfusion injury through Notch 1 and NF-κB signaling pathways. J Biomater Tissue Eng. 2021;11:271–275. [Google Scholar]
- 4.Hou J, Li H, Xue C, Ma J. Lidocaine relieves spinal cord ischemia-reperfusion injury via long non-coding RNA MIAT-mediated Notch1 downregulation. J Biochem. 2022;171:411–420. doi: 10.1093/jb/mvab150. [DOI] [PubMed] [Google Scholar]
- 5.Tang D, Liu X, Chen J. Mitoquinone intravitreal injection ameliorates retinal ischemia-reperfusion injury in rats involving SIRT1/Notch1/NADPH axis. Drug Dev Res. 2022;83:800–810. doi: 10.1002/ddr.21911. [DOI] [PubMed] [Google Scholar]
- 6.Ren YF, Jiang YJ, Zhang XL, Jiang S, Wang YH. Effect of Notch signaling pathway on oxidative stress in human trophoblast cells. Shandong Med J. 2019;59:44–46. [Google Scholar]
- 7.Zhao H, Chen Z, Xie LJ, Liu GF. Suppression of TLR4/NF-κB signaling pathway improves cerebral ischemia-reperfusion injury in rats. Mol Neurobiol. 2018;55:4311–4319. doi: 10.1007/s12035-017-0552-0. [DOI] [PubMed] [Google Scholar]
- 8.Yu L, Li Z, Dong X, Xue X, Liu Y, Xu S, Zhang J, Han J, Yang Y, Wang H. Polydatin protects diabetic heart against ischemia-reperfusion injury via Notch1/Hes1-mediated activation of Pten/Akt signaling. Oxid Med Cell Longev. 2018;2018:2750695. doi: 10.1155/2018/2750695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wei X, Zhu QF. Protective effect of Zhenlong Waking Brain Capsule on focal cerebral ischemia-reperfusion injury in rats. Chin Herb Med. 2015;46:6. [Google Scholar]
- 10.Yu XF, Zhang YH, Ma D. Clinical study on Zhenlong Xingnao Capsules combined with butylphthalide in treatment of acute cerebral infarction. Drugs Clinic. 2022;37:988–992. [Google Scholar]
- 11.Longa EZ, Weinstein PR, Carlson S, Cummins R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke. 1989;20:84–91. doi: 10.1161/01.str.20.1.84. [DOI] [PubMed] [Google Scholar]
- 12.Wang T, Chen H, Xia S, Chen X, Sun H, Xu Z. Ameliorative effect of parishin C against cerebral ischemia-induced brain tissue injury by reducing oxidative stress and inflammatory responses in rat model. Neuropsychiatr Dis Treat. 2021;17:1811–1823. doi: 10.2147/NDT.S309065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gao XJ, Xie GN, Liu L, Fu ZJ, Zhang ZW, Teng LZ. Sesamol attenuates oxidative stress, apoptosis and inflammation in focal cerebral ischemia/reperfusion injury. Exp Ther Med. 2017;14:841–847. doi: 10.3892/etm.2017.4550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ma M, Uekawa K, Hasegawa Y, Nakagawa T, Katayama T, Sueta D, Toyama K, Kataoka K, Koibuchi N, Kuratsu J, Kim-Mitsuyama S. Pretreatment with rosuvastatin protects against focal cerebral ischemia/reperfusion injury in rats through attenuation of oxidative stress and inflammation. Brain Res. 2013;1519:87–94. doi: 10.1016/j.brainres.2013.04.040. [DOI] [PubMed] [Google Scholar]
- 15.Tian T, Cao L, He C, Ye Q, Liang R, You W, Zhang H, Wu J, Ye J, Tannous BA, Gao J. Targeted delivery of neural progenitor cell-derived extracellular vesicles for anti-inflammation after cerebral ischemia. Theranostics. 2021;11:6507–6521. doi: 10.7150/thno.56367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gao W, Ning Y, Peng Y, Tang X, Zhong S, Zeng H. LncRNA NKILA relieves astrocyte inflammation and neuronal oxidative stress after cerebral ischemia/reperfusion by inhibiting the NF-κB pathway. Mol Immunol. 2021;139:32–41. doi: 10.1016/j.molimm.2021.08.002. [DOI] [PubMed] [Google Scholar]
- 17.Liu JD, Chen SH, Wang YCH. Ameliorative effect of β-stigmasterol on Notch1/NF-κB signaling axis in rats with cerebral ischemia-reperfusion injury. J Third Military Med Univ. 2021;43:8. [Google Scholar]
- 18.Perez-Fidalgo JA, Ortega B, Simon S, Samartzis EP, Boussios S. NOTCH signalling in ovarian cancer angiogenesis. Ann Transl Med. 2020;8:1705. doi: 10.21037/atm-20-4497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ho DM, Artavanis-Tsakonas S, Louvi A. The Notch pathway in CNS homeostasis and neurodegeneration. Wiley Interdiscip Rev Dev Biol. 2020;9:e358. doi: 10.1002/wdev.358. [DOI] [PubMed] [Google Scholar]
- 20.Liang W, Lin C, Yuan L, Chen L, Guo P, Li P, Wang W, Zhang X. Preactivation of Notch1 in remote ischemic preconditioning reduces cerebral ischemia-reperfusion injury through crosstalk with the NF-κB pathway. J Neuroinflammation. 2019;16:181. doi: 10.1186/s12974-019-1570-9. [DOI] [PMC free article] [PubMed] [Google Scholar]






