Abstract
Objective: To compare the safety profiles between propofol and sevoflurane in pediatric anesthesia and to investigate risk factors for postoperative adverse reactions. Methods: The data of 194 children who received surgical treatment in Peking Union Medical College Hospital between January 2019 and May 2022 were analyzed retrospectively. According to the different anesthetic drugs the children received, they were divided into a control group (conventional anesthesia with sevoflurane, n=94) and an observation group (anesthesia with both propofol and sevoflurane, n=100). The two groups were compared in terms of anesthetic effect, heart rate, blood oxygen saturation, Ramsay sedation scale (RSS) score during the recovery of anesthesia, and anesthesia safety. Further, the children were grouped based on RSS score to identify the risk factors for agitation during the recovery of anesthesia via logistics regression. Results: The onset time of anesthesia, spontaneous breathing recovery time, extubation time, eye opening time and awake time in the observation group were all significantly shorter than those in the control group (P<0.05). At T1 (during anesthesia induction), T2 (after tracheal intubation) and T3 (after extubation), the observation group showed relatively stable heart rate and blood oxygen saturation than the control group (P<0.05). At the time of awakening, extubation and 30 minutes after extubation, the observation group exhibited significantly lower RSS score than the control group (P<0.05). The observation group also showed a significantly lower incidence of nausea, vomiting and agitation than the control group (P<0.05). Additionally, age ≤6 years old and anesthesia scheme were independent risks for agitation in children during the recovery of anesthesia. The occurrence group had significantly higher risk scores than the non-occurrence group (P<0.05). According to receiver operating characteristic curve-based analysis, the area under the curve of risk score in predicting agitation during the recovery of anesthesia was 0.733. Conclusion: Anesthesia with both propofol and sevoflurane is effective in children undergoing surgical treatment, because the combination can substantially reduce the agitation of children during the recovery of anesthesia and has high anesthesia safety. Propofol combined with sevoflurane is a protective factor against agitation in children during the recovery of anesthesia.
Keywords: Propofol, sevoflurane, pediatric anesthesia, adverse reactions, agitation during the recovery of anesthesia
Introduction
Surgical treatment is significantly effective for many diseases, and anesthetics have become vital in pain alleviation during surgery [1]. However, in surgical anesthesia among children, there are many uncertainties due to the rapid changes of their conditions, compared with adults [2]. Statistics shows that a large number of children worldwide are in need of surgeries and anesthesia each year. For example, in the United States, about 450,000 children undergo surgeries and examinations every year, of which 95% are conducted under general anesthesia [3].
In pediatric anesthesia, parents are increasingly concerned about postoperative complications in children. Frequently adopted general anesthetics in pediatrics include propofol, fentanyl, muscle relaxants, opioids, and inhalation anesthetics, among which propofol and fentanyl are frequently adopted in pediatric general anesthesia [4]. Research has suggested that these drugs, including propofol and fentanyl, might have potential negative impacts on children’s brain development, such as decline in learning and memory abilities and potential permanent neurological damage [5]. Agitation during the recovery of anesthesia is one of the most important adverse effects [6]. Agitation in children during the recovery period occurs in the early period of general anesthesia, and it also manifests as excitement and disorientation in clinical scenarios [7]. In the case of agitation, children may have violent behaviors that hurt themselves and others. In severe cases, agitation can trigger behavioral changes of children, surgical incision dehiscence, catheter removal, etc., prolonging the hospitalization time and incurring additional medical expenses [8]. Sevoflurane is an inhalation anesthetic with advantages of quick onset, strong controllability and stable hemodynamics [9]. Its main mechanism of action is to act on neurotransmitter receptors on neuron cell membranes to produce sedative and anesthetic effects [10]. However, sevoflurane also has its disadvantages. For example, it can easily give rise to agitation and adverse reactions in patients during the recovery of anesthesia [11]. On the contrary, propofol, a new type of fast and short-acting anesthetic for anesthesia induction, anesthesia maintenance and sedation, has the advantages of quick onset, low postoperative adverse reactions, rapid recovery and complete functional recovery [12]. However, the safety profile of propofol combined with sevoflurane in pediatric anesthesia is still unclear.
This study was designed to analyze the safety profile between propofol and sevoflurane in pediatric anesthesia and the risk factors for agitation during postoperative recovery of anesthesia.
Methods and data
Ethical statement
This study was approved by the Medical Ethics Committee of Peking Union Medical College Hospital.
Patient information
The data of 194 children who received surgical treatment in Peking Union Medical College Hospital from January 2019 to May 2022 were analyzed retrospectively. Based on different anesthetic drugs the children received, they were divided into a control group (conventional anesthesia with sevoflurane, n=94) and an observation group (anesthesia with propofol combined with sevoflurane, n=100).
Inclusion and exclusion criteria
Inclusion criteria: The age range of the children is from 3 to 12 years old. Patients who met the clinical diagnostic criteria for relevant diseases, underwent preoperative examinations, and met the criteria for general anesthesia; patients who conformed to the indications of relevant surgical treatments, and had good tolerance and stable vital signs; patients who received surgical treatment for the first time; patients without signs of infection and symptoms of poisoning; patients with complete clinical data.
Exclusion criteria: Patients with a history of premature beats or atrial fibrillation; patients with malignant tumor; patients with severe dysfunction of important organs; patients with gastrointestinal bleeding; patients with airway difficulty; patients with abnormal coagulation function; patients with unclear consciousness.
Anesthesia schemes
In order to ensure the safety and success of the surgery, it was essential to provide instructions to the children’s families before anesthesia. This included informing them about the necessary precautions, such as prohibiting water intake for 4 hours and fasting for 6 hours prior to the procedure. At 30 minutes before the anesthesia, each child was injected intramuscularly with 0.01 mg/kg atropine (Dalian Huali Jingang Pharmaceutical Co., Ltd., State Food and Drug Administration (SFDA) approval number: H21021193), and a trocar was placed in the vein at the back of the hand for subsequent intravenous injection. After the children entered surgical room, they were injected intravenously with 0.3 mg/kg dexamethasone (Zhejiang Xianju Pharmaceutical Co., Ltd., SFDA approval number: H33020822) and 4 ml 0.9% sodium chloride solution.
The control group was given inhalation anesthesia with 8% sevoflurane (Jiangsu Heng Rui Pharmaceutical Co., Ltd., SFDA approval number: H20040771), which was given through the mask to induce anesthesia, and then 3%-4% sevoflurane was given to maintain anesthesia. The oxygen flow was maintained at 2 L/min. In the process of surgical treatment, the inhalation dose was adjusted according to the anesthesia depth, and the anesthesia state was strictly controlled.
The observation group was anesthetized with propofol combined with sevoflurane. The 8% sevoflurane, provided by Jiangsu Heng Rui Pharmaceutical Co., Ltd. (SFDA approval number: H20040771), was administered through a mask to initiate anesthesia. Subsequently, a maintenance dose of 3%-4% sevoflurane was used to sustain the anesthetic state. Thereafter, an intravenous infusion of 1-2 mg/kg propofol (H20163404, Sichuan Guorui Pharmaceutical Co., Ltd.) alongside long-chain fat emulsion was administered to further ensure anesthesia. During the surgical procedure, children’s vital signs, including blood pressure and heart rate, were meticulously monitored. This surveillance facilitated the timely identification and resolution of any unexpected situations, thereby ensuring the smooth progression of the operation.
Collection of data
In this study, the clinical data and related indicators of patients were collected from the electronic medical record system and intraoperative records in Peking Union Medical College Hospital. The clinical data included age, sex, body mass index (BMI), family history of hypertension, education time, type of operation, Ramsay sedation scale (RSS) score and postoperative visual analogue scale (VAS) score. Intraoperative records included the onset time of anesthesia, spontaneous breathing recovery time, extubation time, eye opening time, awake time, heart rate and blood oxygen saturation. Adverse reactions were also recorded.
The Ramsay Sedation Scale (RSS) is used to objectively quantify an individual’s sedation levels, which range from 1 (the patient is anxious, restless or both) to 6 (the patient shows no response to light tapping on the forehead or loud auditory stimulation) [13].
The Visual Analog Scale (VAS) is a psychological measurement tool to measure subjective attributes or attitudes that cannot be directly measured. In pain measurement, it consists of a scale line with two endpoints, which define the extremes of the pain experience, with scores ranging from 0-10. Patients were required to mark their pain level on the scale line between the two endpoints representing the severity of pain. The closer the mark is to the endpoint representing the worst pain, the higher the perceived intensity of pain [14].
Outcome measures
Primary outcome measures
The anesthetic effect (onset time of anesthesia, spontaneous breathing recovery time, extubation time, eye opening time and awake time) of the two groups were compared. The heart rate and blood oxygen saturation of them were also compared at T0 (before anesthesia induction), T1 (during anesthesia induction), T2 (after tracheal intubation) and T3 (after extubation).
Secondary outcome measures
The clinical data, incidence of adverse reactions, changes in RSS score were all compared between the two groups. Based on RSS score, the children were regrouped (a score <3 points indicated agitation), and the risk factors for agitation during the recovery of anesthesia were analyzed through logistics regression, and a prediction model was constructed.
Statistical analyses
This study adopted R language software version 4.1.1 (R Foundation for Statistical Computing, Vienna, Austria) for data cleaning and data analysis, used logistic regression methods to screen influencing factors, and constructed a prediction model. The concordance index (C-index) was calculated using the rms package, and its clinical value was verified through the Receiver Operating Characteristic (ROC) curve. Measurement data were expressed as (X±S) and analyzed by the t-test. Count data were presented as rates and analyzed by the Chi-square test. Data visualization was performed using Graph Pad Prism 8.0. A P-value less than 0.05 was considered statistically significant.
Results
Baseline data
In terms of baseline data, the two groups were not significantly different in age, sex, BMI, family history of hypertension, education time and type of operation (P>0.05, Table 1).
Table 1.
Comparison of baseline data
| Factors | Control group (n=94) | Observation group (n=100) | X2 value | P value |
|---|---|---|---|---|
| Age | 0.058 | 0.808 | ||
| ≥6 years old | 36 | 40 | ||
| <6 years old | 58 | 60 | ||
| Sex | 2.972 | 0.084 | ||
| Male | 41 | 56 | ||
| Female | 53 | 44 | ||
| BMI | 1.039 | 0.308 | ||
| ≥16 kg/m2 | 42 | 52 | ||
| <16 kg/m2 | 52 | 48 | ||
| Family history of hypertension | 0.087 | 0.767 | ||
| Yes | 49 | 50 | ||
| No | 45 | 50 | ||
| Education time | 0.031 | 0.858 | ||
| ≥3 years | 35 | 36 | ||
| <3 years | 59 | 64 | ||
| Type of operation | 1.474 | 0.478 | ||
| Abdominal hernia | 35 | 30 | ||
| Tonsillectomy | 29 | 38 | ||
| Other | 30 | 32 |
Note: BMI: Body mass index.
Comparison of anesthetic effects
The anesthetic effect of the two groups was compared. According to the results, the onset time of anesthesia, spontaneous breathing recovery time, extubation time, eye opening time and awake time in the control group were all significantly longer than those in the observation group (P<0.001, Figure 1).
Figure 1.
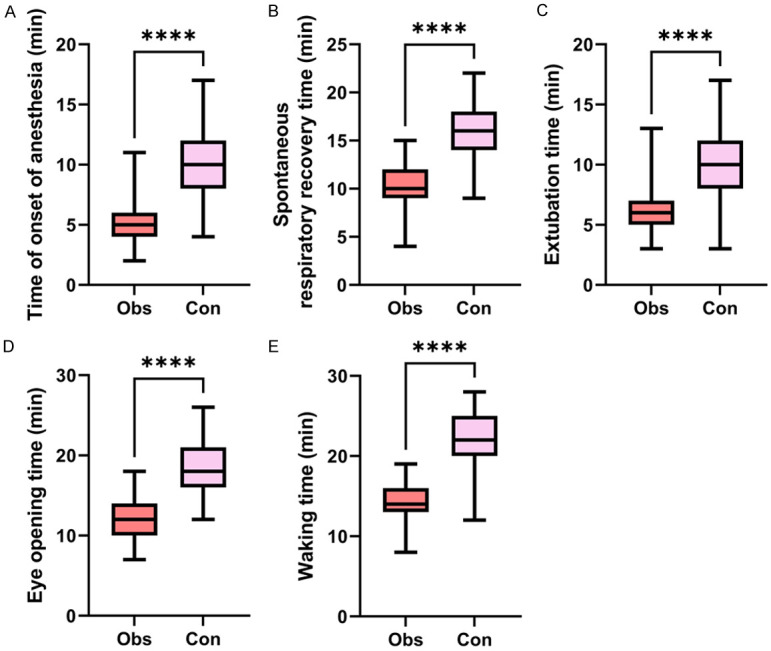
Comparison of anesthetic effect between the observation group and control group. A: Comparison of the onset time of anesthesia; B: Comparison of spontaneous breathing recovery time; C: Comparison of extubation time; D: Comparison of eye opening time; E: Comparison of awake time. Note: ****P<0.0001.
Comparison of heart rate and oxygen saturation at different time points
The heart rate and blood oxygen saturation were compared between the two groups at different time points. According to the results, at T0 and T3, the heart rate and blood oxygen saturation of the two groups were not significantly different (P>0.05, Figure 2), while at T1 and T2, the observation group showed significantly lower heart rate and significantly higher blood oxygen saturation than the control group (P<0.05, Figure 2).
Figure 2.
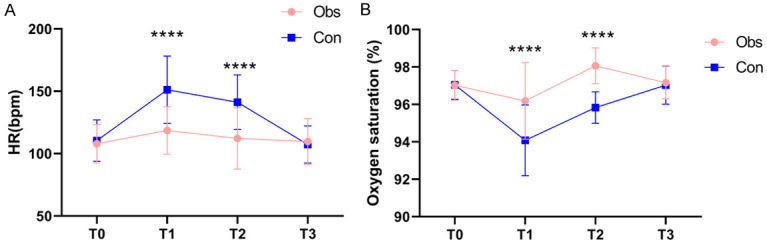
Comparison of heart rate and blood oxygen saturation at different time points. A: Comparison of heart rate; B: Comparison of oxygen saturation. Note: HR: heart rate. ****P<0.0001.
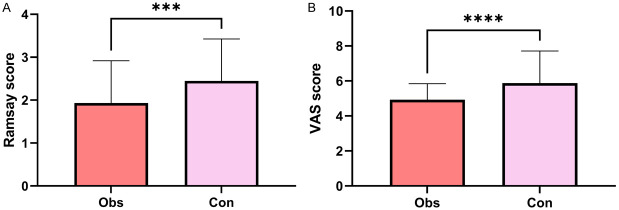
Comparison of agitation score and postoperative pain score during the recovery of anesthesia
The RSS score during the recovery of anesthesia was compared between the two groups. Significantly lower RSS score during the recovery of anesthesia was found in the observation group than that in the control group (P<0.05, Figure 3A). In addition, the observation group demonstrated significantly lower VAS score than the control group (P<0.05, Figure 3B).
Figure 3.
Comparison of RRS and VAS scores between the observation group and control group. A: Comparison of RRS score; B: Comparison of VAS score. Note: ***P<0.001, ****P<0.0001. RSS: Ramsay sedation scale; VAS: Visual analogue scale.
Adverse reactions
In terms of adverse reactions, a lower incidence of nausea, vomiting and agitation was found in the observation group than that in the control group (P<0.05, Table 2).
Table 2.
Adverse reactions
| Group | Nausea and vomiting | Respiratory depression | Hypotension | Agitation during the recovery of anesthesia |
|---|---|---|---|---|
| Control group (n=94) | 10 | 4 | 6 | 44 |
| Observation group (n=100) | 3 | 1 | 2 | 27 |
| X2 value | 4.522 | 2.045 | 2.354 | 8.194 |
| P value | 0.033 | 0.152 | 0.124 | 0.004 |
Analysis of risk factors for agitation during the recovery of anesthesia and construction of a prediction model
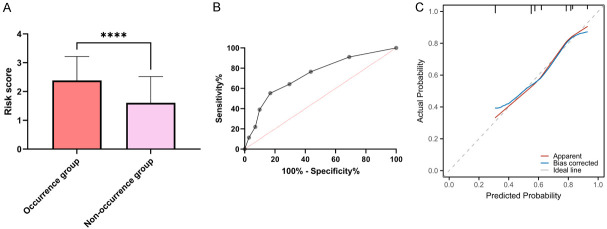
Based on RSS score, the children were grouped into an occurrence group and a non-occurrence group. According to univariate analysis, age, postoperative VAS score, and anesthesia scheme were the risk factors for agitation in the children during the recovery of anesthesia (P<0.05, Table 3). Then, through backward LR multivariate logistics regression analysis, age, postoperative VAS score and anesthesia scheme were found to be the independent risk factors (P<0.05, Table 4). A risk prediction model was constructed based on β coefficient of logistics regression. The risk formula is: Anesthesia mode * 1.005 + Age * 1.261 + VAS score * 1.093. By calculating the risk score of each child, it was found that the occurrence group exhibited significantly higher risk scores than the non-occurrence group (P<0.05, Figure 4A). In addition, according to ROC curve-based analysis, the area under the curve of the risk score for predicting the agitation in the recovery period was 0.733 (Figure 4B). Subsequently, the Bootstrap method (after the original data was repeatedly sampled for 1 000 times) was adopted for internal validation of the model. The results showed that the C-index in internal validation was 0.734 (95 CI%: 0.661-0.806), and the calibration curves were well fitted with the ideal curves (Figure 4C).
Table 3.
Univariate analysis of agitation during the recovery of anesthesia
| Factors | Occurrence group (n=71) | No-occurrence group (n=123) | X2 value | P value |
|---|---|---|---|---|
| Age | 13.013 | <0.001 | ||
| ≥6 years old | 16 | 60 | ||
| <6 years old | 55 | 63 | ||
| Sex | 1.799 | 0.179 | ||
| Male | 31 | 66 | ||
| Female | 40 | 57 | ||
| BMI | 0.513 | 0.473 | ||
| ≥16 kg/m2 | 32 | 62 | ||
| <16 kg/m2 | 39 | 61 | ||
| Family history of hypertension | 0.681 | 0.409 | ||
| Yes | 39 | 60 | ||
| No | 32 | 63 | ||
| Education time | 0.388 | 0.532 | ||
| ≥3 years | 28 | 43 | ||
| <3 years | 43 | 80 | ||
| Type of operation | 1.595 | 0.450 | ||
| Ophthalmic surgery | 24 | 50 | ||
| Head/neck surgery | 25 | 43 | ||
| Other | 23 | 30 | ||
| Onset time of anesthesia (min) | 8.20±3.78 | 7.14±3.20 | 0.488 | 0.625 |
| Spontaneous breathing recovery time (min) | 14.11±3.79 | 12.49±3.54 | 0.567 | 0.571 |
| Extubation time (min) | 8.86±3.08 | 7.87±2.95 | 0.761 | 0.447 |
| Eye opening time (min) | 15.7±4.10 | 14.7±4.17 | 1.626 | 0.105 |
| Awake time (min) | 19.06±4.95 | 17.35±4.86 | 0.186 | 0.852 |
| Postoperative VAS score | 6.38±1.47 | 4.82±1.21 | 7.994 | <0.001 |
| Heart rate at T0 (bpm) | 108.2±16.17 | 109.65±16.06 | 0.605 | 0.545 |
| Oxygen saturation at T0 (%) | 97.14±0.82 | 96.97±0.77 | 1.448 | 0.149 |
Notes: BMI: Body mass index; VAS: Visual analogue scale.
Table 4.
Multivariate analysis of agitation during the recovery of anesthesia
| Factors | β value | Standard error | X2 value | P value | OR value | 95% CI | |
|---|---|---|---|---|---|---|---|
|
| |||||||
| Lower limit | Upper limit | ||||||
| Anesthesia mode | 1.005 | 0.330 | 9.280 | 0.002 | 2.733 | 1.431 | 5.218 |
| Age | 1.261 | 0.356 | 12.539 | <0.001 | 3.529 | 1.756 | 7.093 |
| VAS score | 1.093 | 0.343 | 10.167 | 0.001 | 2.983 | 1.524 | 5.839 |
Note: VAS: Visual analogue scale.
Figure 4.
The value of risk model in predicting agitation during the recovery of anesthesia. A: The risk score in children with agitation during the recovery of anesthesia; B: ROC curve of the risk score; C: Risk model correction curve. Notes: ROC: receiver operating characteristic; ****P<0.0001.
Discussion
With advantages of rapid onset, low irritation, strong controllability and hemodynamic stability, sevoflurane is extensively adopted as an inhalation anesthetic [10]. However, under lax control of the dosage of sevoflurane, children are prone to violent agitation during the recovery of anesthesia, which greatly endangers their safety and prognosis [15,16]. Therefore, application of sevoflurane should be carried out with other safer and more effective drugs together. In contrast, propofol is an alkyl acid short-acting intravenous anesthetic extensively adopted in clinical scenarios. It has the advantages of rapid recovery, rapid anesthesia induction, low adverse reactions and complete functional recovery [17]. In addition, propofol can strongly protect the cardiovascular system and nervous system, and guarantee the stability of children during surgery, greatly reducing the incidence of agitation after anesthesia [18]. However, the effect of propofol alone is not remarkable. In this study, the onset time of anesthesia, spontaneous breathing recovery time, extubation time, eye opening time and awake time in the observation group were all notably shorter than those in the control group, and the observation group showed notably lower postoperative pain than the control group. Additionally, at T1, T2, and T3, the heart rate of the observation group was lower than that of the control group, and the blood oxygen saturation was significantly more stable than that of the control group. The control group showed a significantly higher incidence of adverse reactions than the observation group. These results indicate that the adoption of propofol combined with sevoflurane for pediatric anesthesia has the advantages of rapid recovery, rapid anesthesia induction, stable heart rate and blood oxygen, and less adverse reactions. Song et al. [19] revealed that compared with the control, the sevoflurane combined with propofol resulted notably shorter time of unconsciousness and recovery. Another study reported that children received propofol and sevoflurane showed a notably lower incidence of adverse reactions (intraoperative cough, breath holding, body movement, bronchospasm and laryngospasm) than those received propofol and remifentanil [20]. We believe that this is because propofol mainly acts on GABA receptors and the central nervous system, reducing the release of excitatory transmitters to play a sedative role. Moreover, propofol can substantially reduce the occurrence of nausea and vomiting, as well as the stress reaction during extubation, and thus ensure the stability of hemodynamics after operation [21].
Agitation refers to the separation of behaviors, mental state and consciousness of patients during the recovery of general anesthesia. Patients may have symptoms such as irritability, excitement and delusion, with unconscious movements as the main sign [22]. Compared with adults, children have a relatively higher incidence of agitation after surgical anesthesia due to their relatively poorer brain regulation capabilities [23]. This study compared the RSS scores of the two groups during recovery. According to the results, the control group exhibited notably higher RSS scores than the observation group. This shows that propofol combined with sevoflurane can substantially lower the incidence of agitation in children after anesthesia. Prior research revealed no significant difference in agitation during recovery and other adverse events between children receiving sevoflurane or propofol alone and those receiving additional propofol [24]. However, another study revealed a lower incidence of agitation during recovery in children receiving propofol anesthesia alone than those receiving sevoflurane [25]. In order to understand the risk factors for agitation during the recovery period, logistics regression analysis was conducted. As a result, age ≥6 years old, postoperative VAS score ≥6 points and anesthesia with sevoflurane were found to be the risk factors for agitation. Age is closely related to the incidence of agitation. Patients at a younger age face a higher rate of agitation. With incomplete physical and psychological development, younger children are prone to agitation because of fear in unfamiliar environment [26]. Postoperative pain is a common symptom that needs to be treated with analgesia. Insufficient analgesia can easily give rise to agitation. For children anesthetized with sevoflurane inhalation, the risk of agitation during recovery is high [7,27]. This is because anesthesia induction with sevoflurane will cause changes in EEG, similar to seizures. Sevoflurane inhibits different parts of the central nervous system, so the subcortical center can recover normally during awakening, but a few cerebral cortices may still be inhibited, disrupting the sensory response and processing ability of children, and triggering over-excitation and agitation symptoms.
Finally, this study constructed a risk model to predict the agitation in children during awakening. According to comparison, the occurrence group had significantly higher risk scores than the non-occurrence group. In addition, ROC curve analysis revealed that the area under the curve of risk score in predicting the agitation during awakening was 0.734. The internal validation suggested a certain universality of the risk model. However, this study still has some limitations. This is a retrospective study, with all the samples collected from a single center, so whether this model is universal in other settings needs further confirmation. We hope to carry out multi-center experiments in future studies to verify the efficacy of the constructed model.
In conclusion, anesthesia with both propofol and sevoflurane is effective in children undergoing surgical treatment, because this combination can effectively reduce the agitation of children during the recovery of anesthesia, and it shows a high safety profile. Propofol combined with sevoflurane is a protective factor against agitation during the recovery of anesthesia in children.
Disclosure of conflict of interest
None.
References
- 1.Kurth CD, Hyman D. Worldwide journey in pediatric anesthesia quality and safety. Paediatr Anaesth. 2022;32:1181–1184. doi: 10.1111/pan.14534. [DOI] [PubMed] [Google Scholar]
- 2.Chua GCC, Cyna AM. Satisfaction measures in pediatric anesthesia and perioperative care. Paediatr Anaesth. 2021;31:746–754. doi: 10.1111/pan.14197. [DOI] [PubMed] [Google Scholar]
- 3.Suresh S, Ecoffey C, Bosenberg A, Lonnqvist PA, de Oliveira GS Jr, de Leon Casasola O, de Andres J, Ivani G. The European Society of Regional Anaesthesia and Pain Therapy/American society of regional anesthesia and pain medicine recommendations on local anesthetics and adjuvants dosage in pediatric regional anesthesia. Reg Anesth Pain Med. 2018;43:211–216. doi: 10.1097/AAP.0000000000000702. [DOI] [PubMed] [Google Scholar]
- 4.Stepanovic B, Sommerfield D, Lucas M, von Ungern-Sternberg BS. An update on allergy and anaphylaxis in pediatric anesthesia. Paediatr Anaesth. 2019;29:892–900. doi: 10.1111/pan.13703. [DOI] [PubMed] [Google Scholar]
- 5.Cote CJ. Risk, error, outcome, and prevention in pediatric anesthesia: so many issues, lots of good solutions, but where do we find the resources? Paediatr Anaesth. 2011;21:713–715. doi: 10.1111/j.1460-9592.2011.03620.x. [DOI] [PubMed] [Google Scholar]
- 6.Shen QH, Xu-Shen, Lai L, Chen YJ, Liu K, Sun LJ. The effect of magnesium sulfate on emergence agitation in children undergoing general anesthesia: a systematic review and meta-analysis. J Clin Anesth. 2022;78:110669. doi: 10.1016/j.jclinane.2022.110669. [DOI] [PubMed] [Google Scholar]
- 7.Kanaya A. Emergence agitation in children: risk factors, prevention, and treatment. J Anesth. 2016;30:261–267. doi: 10.1007/s00540-015-2098-5. [DOI] [PubMed] [Google Scholar]
- 8.Zou Y, Liu SH, Xue FS. Emergence agitation or delirium in children. J Anesth. 2022;36:156. doi: 10.1007/s00540-020-02785-9. [DOI] [PubMed] [Google Scholar]
- 9.Franzen S, Semenas E, Taavo M, Martensson J, Larsson A, Frithiof R. Renal function during sevoflurane or total intravenous propofol anaesthesia: a single-centre parallel randomised controlled study. Br J Anaesth. 2022;128:838–848. doi: 10.1016/j.bja.2022.02.030. [DOI] [PubMed] [Google Scholar]
- 10.Sun M, Xie Z, Zhang J, Leng Y. Mechanistic insight into sevoflurane-associated developmental neurotoxicity. Cell Biol Toxicol. 2022;38:927–943. doi: 10.1007/s10565-021-09677-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xiao Y, Jin X, Zhang Y, Huang T, Zhou L, Gao J. Efficacy of propofol for the prevention of emergence agitation after sevoflurane anaesthesia in children: a meta-analysis. Front Surg. 2022;9:1031010. doi: 10.3389/fsurg.2022.1031010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Key KL, Rich C, DeCristofaro C, Collins S. Use of propofol and emergence agitation in children: a literature review. AANA J. 2010;78:468–473. [PubMed] [Google Scholar]
- 13.Lu Q, Wu C, Wu Z, Wang X, Cheng H. Effects of different doses of dex anesthesia on inflammatory factors and hemodynamics in patients undergoing neurosurgery and its relationship with RSS score. Biomed Res Int. 2022;2022:6447407. doi: 10.1155/2022/6447407. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 14.Hwang WY, Kim K, Cho HY, Yang EJ, Suh DH, No JH, Lee JR, Hwang JW, Do SH, Kim YB. The voiding VAS score is a simple and useful method for predicting POUR after laparoscopy for benign gynaecologic diseases: a pilot study. J Obstet Gynaecol. 2022;42:2469–2473. doi: 10.1080/01443615.2022.2071149. [DOI] [PubMed] [Google Scholar]
- 15.Liang TY, Peng SY, Ma M, Li HY, Wang Z, Chen G. Protective effects of sevoflurane in cerebral ischemia reperfusion injury: a narrative review. Med Gas Res. 2021;11:152–154. doi: 10.4103/2045-9912.318860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Apai C, Shah R, Tran K, Pandya Shah S. Anesthesia and the developing brain: a review of sevoflurane-induced neurotoxicity in pediatric populations. Clin Ther. 2021;43:762–778. doi: 10.1016/j.clinthera.2021.01.024. [DOI] [PubMed] [Google Scholar]
- 17.Walsh CT. Propofol: milk of amnesia. Cell. 2018;175:10–13. doi: 10.1016/j.cell.2018.08.031. [DOI] [PubMed] [Google Scholar]
- 18.Stogiannou D, Protopapas A, Protopapas A, Tziomalos K. Is propofol the optimal sedative in gastrointestinal endoscopy? Acta Gastroenterol Belg. 2018;81:520–524. [PubMed] [Google Scholar]
- 19.Song YS, Yang ZZ, Hao YB, Liu WW, Zhang SS, Wang P, Fan TL, Zhang Y. Anesthetic effect and safety of sevoflurane combined with propofol in removing tracheobronchial foreign bodies in children. Eur Rev Med Pharmacol Sci. 2020;24:11315–11322. doi: 10.26355/eurrev_202011_23622. [DOI] [PubMed] [Google Scholar]
- 20.Kanaya A, Kuratani N, Satoh D, Kurosawa S. Lower incidence of emergence agitation in children after propofol anesthesia compared with sevoflurane: a meta-analysis of randomized controlled trials. J Anesth. 2014;28:4–11. doi: 10.1007/s00540-013-1656-y. [DOI] [PubMed] [Google Scholar]
- 21.Hausburg MA, Banton KL, Roman PE, Salgado F, Baek P, Waxman MJ, Tanner A 2nd, Yoder J, Bar-Or D. Effects of propofol on ischemia-reperfusion and traumatic brain injury. J Crit Care. 2020;56:281–287. doi: 10.1016/j.jcrc.2019.12.021. [DOI] [PubMed] [Google Scholar]
- 22.Zhang C, Li J, Zhao D, Wang Y. Prophylactic midazolam and clonidine for emergence from agitation in children after emergence from sevoflurane anesthesia: a meta-analysis. Clin Ther. 2013;35:1622–1631. doi: 10.1016/j.clinthera.2013.08.016. [DOI] [PubMed] [Google Scholar]
- 23.Urits I, Peck J, Giacomazzi S, Patel R, Wolf J, Mathew D, Schwartz R, Kassem H, Urman RD, Kaye AD, Viswanath O. Emergence delirium in perioperative pediatric care: a review of current evidence and new directions. Adv Ther. 2020;37:1897–1909. doi: 10.1007/s12325-020-01317-x. [DOI] [PubMed] [Google Scholar]
- 24.Wu G, Xu X, Fu G, Zhang P. General anesthesia maintained with sevoflurane versus propofol in pediatric surgery shorter than 1 hour: a randomized single-blind study. Med Sci Monit. 2020;26:e923681. doi: 10.12659/MSM.923681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chai J, Wu XY, Han N, Wang LY, Chen WM. A retrospective study of anesthesia during rigid bronchoscopy for airway foreign body removal in children: propofol and sevoflurane with spontaneous ventilation. Paediatr Anaesth. 2014;24:1031–1036. doi: 10.1111/pan.12509. [DOI] [PubMed] [Google Scholar]
- 26.Reynolds T, Sankaran S, Chimbira WT, Phan T, Nafiu OO. Severe obesity and sleep-disordered breathing as risk factors for emergence agitation in pediatric ambulatory surgery. J Perianesth Nurs. 2018;33:304–311. doi: 10.1016/j.jopan.2016.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Barreto ACTP, Rangel da Rocha Paschoal AC, Barbosa Farias C, Gomes Nogueira Borges PS, Gonelli Albanez da Cunha Andrade R, de Orange FA. Risk factors associated with anesthesia emergence delirium in children undergoing outpatient surgery. Braz J Anesthesiol. 2018;68:162–167. doi: 10.1016/j.bjane.2017.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]