Abstract
To date very few drugs have favorably influenced the course of transmissible spongiform encephalopathies. In previous studies, the polyene antibiotics amphotericin B (AmB) and MS-8209 prolonged the incubation time in Syrian hamsters of the 263K strain of scrapie, but AmB had no effect against other scrapie strains in Syrian hamsters. In the present experiments using transgenic mice expressing Syrian hamster PrP in neurons only, MS-8209 extended the life spans of animals infected with the 263K strain but not the DY strain. AmB was effective against both 263K and DY and prevented death in 18% of DY-infected animals. The AmB effect against strain 263K was more prominent in mice whose endogenous PrP gene had been inactivated by homologous recombination. It was unclear whether this difference was due to a change in the duration of the disease or to possible interactive effects between the mouse PrP gene and the drugs themselves. The effectiveness of treatment after intracerebral scrapie infection in transgenic mice expressing PrP only in neurons suggested that neurons are important sites of action for these drugs.
Transmissible spongiform encephalopathies (TSE), or prion diseases, are a group of neurodegenerative diseases that includes scrapie in sheep, bovine spongiform encephalopathy in cattle, and Creutzfeldt-Jakob disease in humans (5, 18). These diseases have a long incubation time, with a short clinical phase dominated by neurological symptoms. The pathognomonic feature of TSE diseases is the accumulation in the brain of an abnormal protein, PrP-res, which is derived from a host-encoded protease-sensitive protein, PrP-sen, or prion protein (4, 17). TSE diseases are uniformly fatal, and to date no curative or palliative treatment is available. Nonetheless, some drugs have proven to be partially effective in experimental TSE models (8, 9, 11, 13). For instance, the antifungal polyene antibiotics amphotericin B (AmB) and MS-8209 (a benzylidene derivative of AmB) are able to prolong the incubation time and delay PrP-res accumulation in the brain in certain rodent TSE models (6, 16). This result suggests that polyene antibiotics have potential for use as anti-TSE drugs since they lengthen the incubation time after either intracerebral (i.c.) or intraperitoneal infection, even when administered late in the course of the disease (7). However, in Syrian hamsters (SHa) the AmB effect is dependent on the TSE strain used. Indeed, AmB-induced prolongation of the incubation time occurred in SHa only after infection with the 263K scrapie strain, not after infection with strain 139H or DY (15, 23). In the present experiments, we used transgenic mice (Tg52NSE) in which expression of the hamster PrP (HaPrP) gene is driven by the neuron-specific enolase promoter and occurs only in neurons (19). We chose these mice in order to study whether restriction of HaPrP expression to neurons influences the effectiveness of treatment with polyene antibiotics. The results indicated that the polyene antibiotics were effective against scrapie strain 263K and in some cases against strain DY.
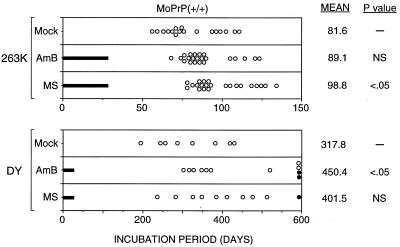
Initially, we studied the effects of AmB and MS-8209 in Tg52NSE mice infected with scrapie strain 263K. Mice were inoculated i.c. with 50 μl of a 1% brain homogenate derived from terminally ill SHa infected by the 263K scrapie strain (∼107 50% infectious doses [ID50]). Animals were monitored regularly for the appearance of clinical signs of scrapie. AmB (Sigma) or MS-8209 (N-methyl glucosamine salt of 1-deoxy-1-amino-4,6-benzylidene-d-fructosyl-AmB; Mayoli-Spindler) was injected intraperitoneally, at 1 mg/kg of body weight for AmB or 5 mg/kg for MS-8209, 6 days per week for 4 weeks starting on the day of infection (Fig. 1). Drugs were dissolved in 5% glucose solution and stored as aliquots at −20°C until use. Control 263K-infected Tg52NSE mice had a mean incubation time of 81.6 days when injected with a 5% glucose solution lacking AmB or MS-8209. The mean incubation period of 263K-infected Tg52NSE mice was prolonged by 7.5 days with AmB treatment and by 17.2 days with MS-8209 treatment. Results with MS-8209 were significantly different from results for mock-treated controls (P < 0.05), but those with AmB were not (Fig. 1).
FIG. 1.
Incubation times of Tg52NSE mice infected with hamster scrapie strain 263K or DY after treatment with 1 mg of AmB per kg or 5 mg of MS-8209 (MS) per kg. Treatment was done 6 days per week for 4 weeks following infection (treatment times are indicated by solid bars). Each point represents one animal. Solid dots represent mice healthy at 600 days. The mean incubation times, in days, and the P values for comparison of groups, as analyzed by a one-tailed Mann-Whitney U test (Instat 2.0, Graphpad 1993), are indicated on the right (NS, not significant). Mean incubation time was calculated by using a value of 600 days for survivors at that time.
To determine whether treatment with these drugs might be effective against a different TSE strain, Tg52NSE mice were inoculated i.c. with the DY strain and treated with AmB or MS-8209 by the protocol described above. DY is a slow-replicating TSE strain which differs from 263K in disease tempo, clinical signs, and regions of brain infected (2). PrP-res from brains of animals infected with DY is 1 to 2 kDa lower in molecular mass than that observed with the 263K strain (3). In the present study, Tg52NSE mice were infected with 50 μl of a 1% brain homogenate from terminally ill SHa infected by the DY strain (∼105 ID50) and subsequently treated with AmB or MS-8209. All mock-treated DY-infected Tg52NSE mice developed disease with an average incubation time of 318 days (Fig. 1). At the end of the study (600 days), 2 of 11 (18%) of the AmB-treated mice and 1 of 10 (10%) of the MS-8209-treated mice were clinically normal (Fig. 1). This is the first study showing AmB to be effective against DY infection, in contrast with the negative results previously reported against the DY strain in SHa (15).
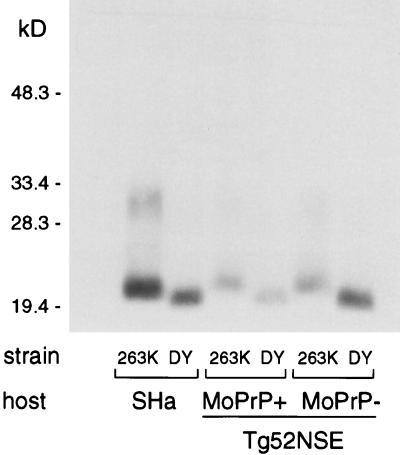
Because of this discrepancy, we wished to confirm that the DY strain was not modified by passage in Tg52NSE mice. Therefore, we compared the molecular masses of PrP-res from DY-infected and 263K-infected mice. Brain homogenates (∼10% [wt/vol] were digested by proteinase K (Boehringer Mannheim) (20 μg/ml, 37°C, 1 h), methanol precipitated, and then digested by PNGase F (New England Biolabs) (37°C, 6 h) as previously reported (12). The apparent molecular mass of PrP-res from DY-infected animals, as detected by immunoblotting with HaPrP-reactive 3F4 antibody (10), was, as expected, 1 to 2 kDa lower than that of 263K-infected mice (Fig. 2). Thus, this property of the DY strain was not altered by infection of Tg52NSE mice.
FIG. 2.
Immunodetection of PrP-res in Tg mice infected with strain 263K or DY. Samples were digested with PK and PNGase F, as described previously (12), and were run on 16% polyacrylamide Novex gels. Blots were developed with 3F4 monoclonal antibody (10) by using the ECL system (Amersham).
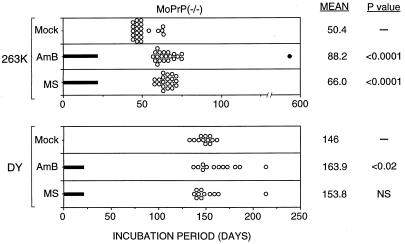
The marginal effect of AmB on the incubation period of 263K-infected Tg52NSE mice was in marked contrast to the higher effectiveness of AmB in the SHa model (1, 23). One major difference between the two hosts is the presence of the mouse PrP (MoPrP) protein in the brain of Tg52NSE mice. Coexpression of MoPrP and HaPrP is known to delay the onset of disease (19, 22) and could possibly interfere with treatment. Therefore, we carried out the same treatments in Tg52NSE mice which lacked a functional MoPrP gene. These mice were generated by breeding Tg52NSE mice with MoPrP(−/−) mice whose PrP gene was inactivated by homologous recombination (14). The mice were infected i.c. with the same 263K and DY inocula as before, but the drugs were administered for 6 days per week for 3 weeks instead of 4 weeks. 263K-infected mock-treated mice had a mean incubation time of 50.4 days (Fig. 3). After a 3-week course of treatment, results AmB- and MS-8209-treated groups were both significantly different from controls (P < 0.0001). Therefore, in contrast to results with Tg52NSE mice, both drugs had a significant effect in 263K-infected Tg52NSE/MoPrP(−/−) mice.
FIG. 3.
Incubation times of Tg52NSE/MoPrP(−/−) mice infected with hamster scrapie strain 263K or DY after treatment with 1 mg of AmB per kg or 5 mg of MS-8209 (MS) per kg. Treatment was done 6 days per week for 3 weeks following infection (treatment times are indicated by solid bars). Each dot represents one animal; the solid dot represents one mouse healthy at 600 days. The mean incubation times, in days, and the P values, as analyzed by a one-tailed Mann-Whitney U test (Instat 2.0, Graphpad 1993), are indicated on the right (NS, not significant). Mean incubation time was calculated by using a value of 600 days for survivors at that time.
Because of the increased effect seen with AmB in 263K-infected Tg52NSE mice lacking a MoPrP gene, these mice were infected i.c. with the DY strain and drug treatment was studied. In the mock-treated group, the mean incubation time was 146 days; the incubation time was prolonged after administration of either AmB or MS-8209 (Fig. 3). The AmB-treated group was significantly different from the control group (P < 0.02), while the MS-8209-treated group was not. In conclusion, in both Tg52NSE mice and Tg52NSE/MoPrP(−/−) mice, only AmB was able to induce a significant increase in the incubation time in DY-infected mice.
In the present study, AmB and MS-8209 were efficient in delaying the appearance of clinical signs in 263K-infected Tg52NSE/MoPrP(−/−) mice. This result is consistent with earlier results reported with SHa (1). In our system, HaPrP expression was restricted to neurons; therefore, it is possible that polyene antibiotics have an effect directly on neurons during scrapie infection. However, our results do not exclude the possibility that in hamsters or nontransgenic mice these drugs might work on nonneuronal PrP-positive cells such as astrocytes or microglia. To determine whether there is any restriction on cell specificity for PrP expression regarding the inhibitory effect of polyene antibiotics, it would be of interest to study these drugs in GFAP (glial fibrillary acidic protein)-HaPrP transgenic mice, which express HaPrP in astrocytes only (20).
This is the first time that treatment with AmB has led to alteration of clinical disease in DY-infected animals. In contrast, previous data indicated that long-term treatment from the day of inoculation with 1 mg of AmB per kg in DY-infected SHa had no effect (15). The difference in the duration of treatment cannot account for this discrepancy, because we treated our mice for a shorter time than was used previously for SHa. Because the transgenic mouse model we used is different from SHa, both drug distribution and scrapie strain properties might be different. However, we showed here that the PrP-res of the DY strain displayed its unique molecular mass in both Tg52NSE and Tg52NSE/MoPrP(−/−) mice (Fig. 2). Therefore, the most likely explanation for the efficiency of AmB in DY-infected transgenic mice may be differences in drug distribution in the brain.
The AmB derivative MS-8209 had a significant effect on 263K-infected Tg mice but no effect on DY-infected Tg mice. Thus, the mechanisms of inhibition of AmB and MS-8209 on TSE diseases may not be identical. This phenomenon could be due to the addition of the benzylidene group to AmB in the synthesis of MS-8209 (21), leading to slight differences in mechanisms of inhibition of TSE diseases. These differences suggest that it might be possible to design improved AmB derivatives which could be effective against “AmB-resistant” TSE strains.
With the 263K strain we obtained a different response to drug treatment in the presence or absence of expression of MoPrP. One possible explanation could be that the duration of drug administration was shorter relative to the incubation period in Tg52NSE mice than in Tg52NSE/MoPrP(−/−) mice. In contrast, in DY-infected animals, expression of MoPrP did not decrease the effectiveness of AmB treatment. This discrepancy perhaps reflects differences in the roles of PrP in the replication process or the types of PrP-res generated by 263K and DY strains.
With a mean incubation period of 50 days, Tg52NSE/MoPrP(−/−) mice are an extremely rapid model for the study of experimental TSE disease (Fig. 3). In comparison to SHa, in which the incubation period is around 70 days, these transgenic mice are much more economical and efficient to maintain and therefore should prove to be extremely useful in future evaluations of other TSE therapeutic agents. In addition, they should provide useful information regarding the role of neuronal PrP expression in the therapeutic effectiveness of candidate drugs.
Acknowledgments
We thank R. Bessen for the DY inoculum and M. Seman for MS-8209 samples. We thank S. Priola, U. Dittmer, and D. Lodmell for careful reading of the manuscript.
REFERENCES
- 1.Adjou K T, Demaimay R, Lasmezas C, Deslys J-P, Seman M, Dormont D. MS-8209, a new amphotericin B derivative, provides enhanced efficacy in delaying hamster scrapie. Antimicrob Agents Chemother. 1995;39:2810–2812. doi: 10.1128/aac.39.12.2810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bessen R A, Marsh R F. Identification of two biologically distinct strains of transmissible mink encephalopathy in hamsters. J Gen Virol. 1992;73:329–334. doi: 10.1099/0022-1317-73-2-329. [DOI] [PubMed] [Google Scholar]
- 3.Bessen R A, Marsh R F. Distinct PrP properties suggest the molecular basis of strain variation in transmissible mink encephalopathy. J Virol. 1994;68:7859–7868. doi: 10.1128/jvi.68.12.7859-7868.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bolton D C, McKinley M P, Prusiner S B. Identification of a protein that purifies with the scrapie prion. Science. 1982;218:1309–1311. doi: 10.1126/science.6815801. [DOI] [PubMed] [Google Scholar]
- 5.Caughey B, Chesebro B. Prion protein and the transmissible spongiform encephalopathies. Trends Cell Biol. 1997;7:56–62. doi: 10.1016/S0962-8924(96)10054-4. [DOI] [PubMed] [Google Scholar]
- 6.Demaimay R, Adjou K, Lasmezas C, Lazarini F, Cherifi K, Seman M, Deslys J P, Dormont D. Pharmacological studies of a new derivative of amphotericin B, MS-8209, in mouse and hamster scrapie. J Gen Virol. 1994;75:2499–2503. doi: 10.1099/0022-1317-75-9-2499. [DOI] [PubMed] [Google Scholar]
- 7.Demaimay R, Adjou K T, Beringue V, Demart S, Lasmézas C I, Deslys J-P, Seman M, Dormont D. Late treatment with polyene antibiotics can prolong the survival time of scrapie-infected animals. J Virol. 1997;71:9685–9689. doi: 10.1128/jvi.71.12.9685-9689.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ehlers B, Diringer H. Dextran sulphate 500 delays and prevents mouse scrapie by impairment of agent replication in spleen. J Gen Virol. 1984;65:1325–1330. doi: 10.1099/0022-1317-65-8-1325. [DOI] [PubMed] [Google Scholar]
- 9.Farquhar C F, Dickinson A G. Prolongation of scrapie incubation period by an injection of dextran sulphate 500 within the month before or after infection. J Gen Virol. 1986;67:463–473. doi: 10.1099/0022-1317-67-3-463. [DOI] [PubMed] [Google Scholar]
- 10.Kascsak R J, Rubenstein R, Merz P A, Tonna-DeMasi M, Fersko R, Carp R I, Wisniewski H M, Diringer H. Mouse polyclonal and monoclonal antibody to scrapie-associated fibril proteins. J Virol. 1987;61:3688–3693. doi: 10.1128/jvi.61.12.3688-3693.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kimberlin R H, Walker C A. Suppression of scrapie infection in mice by heteropolyanion 23, dextran sulfate, and some other polyanions. Antimicrob Agents Chemother. 1986;30:409–413. doi: 10.1128/aac.30.3.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kocisko D A, Lansbury P T, Jr, Caughey B. Partial unfolding and refolding of scrapie-associated prion protein: evidence for a critical 16-kDa C-terminal domain. Biochemistry. 1996;35:13434–13442. doi: 10.1021/bi9610562. [DOI] [PubMed] [Google Scholar]
- 13.Ladogana A, Casaccia P, Ingrosso L, Cibati M, Salvatore M, Xi Y G, Masullo C, Pocchiari M. Sulphate polyanions prolong the incubation period of scrapie-infected hamsters. J Gen Virol. 1992;73:661–665. doi: 10.1099/0022-1317-73-3-661. [DOI] [PubMed] [Google Scholar]
- 14.Manson J C, Clarke A R, McBride P A, McConnell I, Hope J. PrP gene dosage determines the timing but not the final intensity or distribution of lesions in scrapie pathology. Neurodegeneration. 1994;3:331–340. [PubMed] [Google Scholar]
- 15.McKenzie D, Kaczkowski J, Marsh R, Aiken J. Amphotericin B delays both scrapie agent replication and PrP-res accumulation early in infection. J Virol. 1994;68:7534–7536. doi: 10.1128/jvi.68.11.7534-7536.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pocchiari M, Schmittinger S, Masullo C. Amphotericin B delays the incubation period of scrapie in intracerebrally inoculated hamsters. J Gen Virol. 1987;68:219–223. doi: 10.1099/0022-1317-68-1-219. [DOI] [PubMed] [Google Scholar]
- 17.Prusiner S B. Novel proteinaceous infectious particles cause scrapie. Science. 1982;216:136–144. doi: 10.1126/science.6801762. [DOI] [PubMed] [Google Scholar]
- 18.Prusiner S B. Molecular biology of prion diseases. Science. 1991;252:1515–1522. doi: 10.1126/science.1675487. [DOI] [PubMed] [Google Scholar]
- 19.Race R E, Priola S A, Bessen R A, Ernst D R, Dockter J, Rall G F, Mucke L, Chesebro B, Oldstone M B A. Neuron-specific expression of a hamster prion protein minigene in transgenic mice induces susceptibility to hamster scrapie agent. Neuron. 1995;15:1183–1191. doi: 10.1016/0896-6273(95)90105-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Raeber A J, Race R E, Brandner S, Priola S A, Sailer A, Bessen R A, Mucke L, Manson J, Aguzzi A, Oldstone M B A, Weissman C, Chesebro B. Astrocyte-specific expression of hamster prion protein (PrP) renders PrP knockout mice susceptible to hamster scrapie. EMBO J. 1997;16:6057–6065. doi: 10.1093/emboj/16.20.6057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saint-Julien L, Joly V, Seman M, Carbon C, Yeni P. Activity of MS-8209, a nonester amphotericin B derivative, in treatment of experimental systemic mycoses. Antimicrob Agents Chemother. 1992;36:2722–2728. doi: 10.1128/aac.36.12.2722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Scott M, Foster D, Mirenda C, Serban D, Coufal F, Walchli M, Torchia M, Groth D, Carlson G, DeArmond S J, Westaway D, Prusiner S B. Transgenic mice expressing hamster prion protein produce species-specific scrapie infectivity and amyloid plaques. Cell. 1989;59:847–857. doi: 10.1016/0092-8674(89)90608-9. [DOI] [PubMed] [Google Scholar]
- 23.Xi Y G, Ingrosso L, Ladogana A, Masullo C, Pocchiari M. Amphotericin B treatment dissociates in vivo replication of the scrapie agent from PrP accumulation. Nature. 1992;356:598–601. doi: 10.1038/356598a0. [DOI] [PubMed] [Google Scholar]