Abstract
Background
Although a substantial fraction of the US population was infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) during December 2021–February 2022, the subsequent evolution of population immunity reflects the competing influences of waning protection over time and acquisition or restoration of immunity through additional infections and vaccinations.
Methods
Using a Bayesian evidence synthesis model of reported coronavirus disease 2019 (COVID-19) data (diagnoses, hospitalizations), vaccinations, and waning patterns for vaccine- and infection-acquired immunity, we estimate population immunity against infection and severe disease from SARS-CoV-2 Omicron variants in the United States, by location (national, state, county) and week.
Results
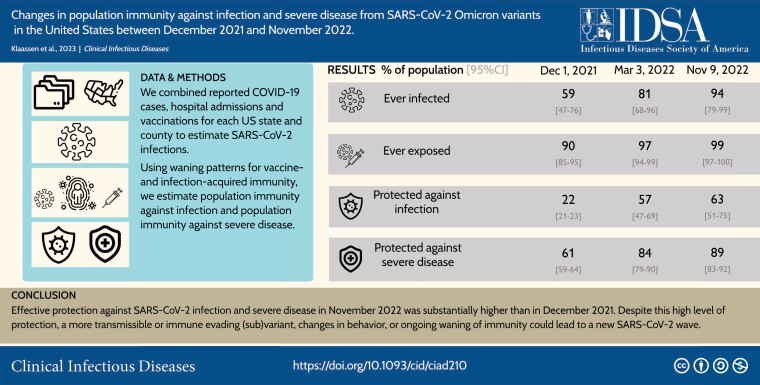
By 9 November 2022, 97% (95%–99%) of the US population were estimated to have prior immunological exposure to SARS-CoV-2. Between 1 December 2021 and 9 November 2022, protection against a new Omicron infection rose from 22% (21%–23%) to 63% (51%–75%) nationally, and protection against an Omicron infection leading to severe disease increased from 61% (59%–64%) to 89% (83%–92%). Increasing first booster uptake to 55% in all states (current US coverage: 34%) and second booster uptake to 22% (current US coverage: 11%) would increase protection against infection by 4.5 percentage points (2.4–7.2) and protection against severe disease by 1.1 percentage points (1.0–1.5).
Conclusions
Effective protection against SARS-CoV-2 infection and severe disease in November 2022 was substantially higher than in December 2021. Despite this high level of protection, a more transmissible or immune evading (sub)variant, changes in behavior, or ongoing waning of immunity could lead to a new SARS-CoV-2 wave.
Keywords: SARS-CoV-2, immunological exposure, effective protection, Omicron variants
Although most of the newly acquired immunity had been accumulated in the December 2021–February 2022 Omicron wave, risk of reinfection and subsequent severe disease remained present at the beginning of the 2022–2023 winter, despite high levels of protection.
Graphical Abstract
Graphical Abstract.
This graphical abstract is also available at Tidbit: https://tidbitapp.io/tidbits/changes-in-population-immunity-against-infection-and-severe-disease-from-sars-cov-2-omicron-variants-in-the-united-states-between-december-2021-and-november-2022-f6e0374e-e984-44b9-814b-2347a8bc7503
In 2022, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Omicron subvariants B.1.1.529 and BA.5 emerged and became dominant in the United States [1]. Several new subvariants have subsequently emerged with potential to outcompete the current dominant variants. During the first Omicron wave (December 2021–February 2022) over 25 million coronavirus disease 2019 (COVID-19) diagnoses were reported in the United States (8% of the population). Many more infections were likely undiagnosed and, due to waning immunity and increased immune evasion, reinfections are occurring more frequently [2], even amongst those vaccinated [3].
In 2022, mask mandates and restrictions on social gatherings were lifted, reducing barriers to the transmission of respiratory pathogens. In the absence of non-pharmaceutical interventions, partial immunity afforded by previous infection and vaccination is the main protection against future infections. By 9 November 2022, 79% of the US population had initiated a vaccination series, 34% had received a first booster and 11% had received a second booster [1]. Levels of vaccination coverage have been heterogeneous geographically and temporally: over three-quarters of the vaccinations in 2022 were administered in January, and the fraction of the population who had received any booster by 9 November 2022 ranged from 20% in North Carolina to 55% in Vermont.
Ongoing (re)infections and vaccinations boost population immunity against future infections and subsequent severe disease, which is offset by waning immunity and immune evasion of newer variants In the absence of serological data, estimates of changes in population immunity need to account for the competing mechanisms. Modeling this process is complicated by the emergence of variants with differing transmissibility and immune evasion properties. The protection conferred by different exposure types and the rate at which that protection wanes remains uncertain. With the emergence of the Omicron variant, vaccine protection was strongly reduced as compared to earlier variants [4–8]. Although receiving a booster dose increases the protection against infection [9], with a stronger effect for the bivalent booster [10], low booster uptake has limited this protection in the population. Previous infection offers less protection against reinfection with the Omicron variant compared to earlier variants [11, 12], although Omicron infections offer more protection than pre-Omicron infections [13]. Evidence suggests that protection afforded by a prior infection is higher than that conferred by vaccination alone, and that hybrid immunity offers the most durable protection [14]. The US population at the end of 2022 will reflect a range of prior immunological exposures to SARS-CoV-2, with variable protection against future infections.
In this study we combined estimates of infection and vaccination trends with recent evidence on how immunity wanes after exposure, to estimate the level of population immunity in the United States at national, state, and county levels, for each week between 1 December 2021 and 9 November 2022.
METHODS
Data
We extracted Centers for Disease Control and Prevention (CDC) data for state- and county-level vaccination and booster coverage between 2 December 2021 and 9 November 2022 [1]. We considered 3 vaccination states. First, we used the reported number of individuals that initiated a primary vaccination series as an indicator of being vaccinated. Second, we used the reported number of individuals that received an additional dose after having completed the primary series as an indicator of being boosted. Third, we used the reported number of individuals that received a second booster shot, after having received a primary vaccination series and a first booster dose. Because these data were unavailable at the county level, we assumed the second booster coverage was equal to the encompassing state.
We used a statistical model that renders weekly time-series of SARS-CoV-2 first infections and reinfections, accounting for time-varying under-ascertainment and time lags of reporting and disease progression [15]. Inputs to this model are reported case [16, 17] and hospitalization data [18]. We made 3 updates to this model to better represent disease dynamics under the Omicron variants of SARS-CoV-2. First, infection-induced immunity is modeled to wane and the probability of progressing to more severe disease states is reduced for those reinfected [1, 19]. Second, the infection fatality ratio was updated to reflect decreased mortality risks under the Omicron variants. Third, the model was fitted to hospitalization reports [18], rather than deaths. To facilitate this change, the updated model renders weekly rather than daily estimates (additional eMethods provided in the Supplement). The model used for this analysis was initialized with immunity estimates as of 2 December 2021, as reported by Klaassen et al (2022) [20].
Estimates of Immunological Exposure
For each location and each week, we computed the percentage of the population immunologically exposed, defined having received at least 1 dose of a COVID-19 vaccine, at least 1 prior SARS-CoV-2 infection, or both, using the cumulative reports of first infections and first vaccine doses. Following Klaassen et al (2022) [20], we operationalized the probability of co-occurrence of infection and vaccination as odds ratios for vaccination among individuals with versus without prior infection, based on data collected in the CDC Household Pulse Surveys [21] and validated against independent survey estimates [22].
Effective Protection by Exposure State
We defined mutually exclusive exposure states for different combinations of prior infection and vaccination (eTable 1). Using the estimated immunological exposure and the waning curves, we estimated how individuals transition between these exposure states over time. At each timepoint, we determined the number newly entering the joint vaccinated and infected state through either infection or vaccination, and correspondingly calculated the fraction of new infections and vaccinations occurring in the immune-naïve population. Next, we distributed the reinfections over those previously (re)infected. Finally, the booster and second booster doses were assigned proportionally over the vaccinated and first booster exposure states. We assumed that at each time point only 1 event could take place (eg, it was not possible to be infected and receive a booster in the same week). Infections and reinfections were assigned across the exposures states proportional to susceptibility to (re)infection, whereas vaccinations and boosters were assigned proportional to exposure. We used the population distribution across these exposure states, combined with the waning rates associated with each state, to estimate the level of population immunity against infection and severe disease from an Omicron variant for each week and modelled location, between 2 December 2021 and 9 November 2022. At each time point, we adjusted for all exposure states proportionally for all-cause mortality rate and adjusted the naïve exposure state for the birth rate. We used birth and mortality reports for each US state and each month in 2021 to compute average weekly rates [23]. We assumed the birth and mortality rates for counties were equal to that of their encompassing state.
Initial Levels of Protection and Subsequent Waning
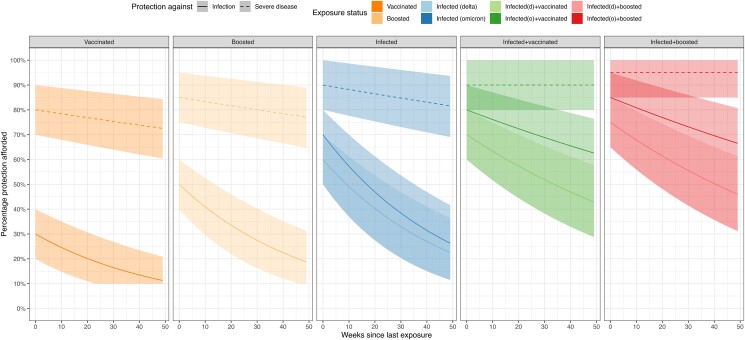
A recent systematic review and meta-analysis synthesized evidence on the waning of protection against infection and severe disease from exposure to Omicron variants [14]. We used this evidence to specify waning curves for different combinations of prior infection and vaccination, supplemented with evidence on the reduced vaccine efficacy against Omicron variants compared to pre-Omicron variants [6, 7]. We assumed that immunity is lost at constant rate, such that each waning curve is parameterized as an initial level of immunity and an exponential rate of decline. The initial level of immunity ranges from 30% for individuals who had only received their primary series [6] to 85% protection for individuals who have received a booster dose and been infected with the Omicron variant [14] (eTable 2 in the Supplement). Based on the evidence reported in the meta-analysis, we assumed that immunity against infection declined at the same rate for all non-hybrid exposures ((re)infection only, vaccination/booster only), and at a lower rate for hybrid exposures (both prior infection and vaccination). For protection against severe disease, we assumed immunity declined at 1% per month for non-hybrid exposures and stayed constant for hybrid exposures. We set a lower bound for the protection against infection at 0.1 and for the protection against severe disease at 0.5, assuming some residual level of protection for individuals with any historical exposure compared to unexposed individuals. The waning curves derived from these assumptions are shown in Figure 1, along with the empirical estimates used to parameterize these curves.
Figure 1.
Assumed initial immunity and waning trends for different combinations of prior infection and vaccination.
We accounted for uncertainty in these assumptions by comparing the base-case results to alternative scenarios with optimistic and pessimistic assumptions for initial immunity and waning rates. In the optimistic scenario, initial immunity estimates were assumed to be 10 percentage points higher than the base-case, and the rate of decline was 50% less. In the pessimistic scenario, initial immunity was assumed to be 10 percentage points lower than the base-case, and the rate of decline was 50% more. In this pessimistic scenario, we also assumed there was no additional benefit of having an Omicron infection in terms of the resulting immunity.
Impact of Higher Booster Uptake
We examined a final scenario estimating the potential impact of greater booster uptake. We operationalized this as an immediate increase in the population fraction that had received a COVID-19 booster and second booster (distributed proportionally across all exposure states eligible for each), from observed levels on 9 November 2022 to 55% coverage of first booster and 20% uptake of the second booster, which represent the highest values for these 2 outcomes across US states (both values taken from Vermont).
Model Implementation
We executed the analysis in R [24] and the rstan package [25] (https://github.com/COVIDestim/COVIDestim/tree/severe-protection). For state-level results, we report uncertainty using equal-tailed 95% credible intervals (95% CrI). We calculated national estimates and conservative uncertainty intervals by summing state-level estimates and upper and lower bounds of state-level intervals. County-level estimates were produced using an optimization routine [6] that produces point estimates without uncertainty intervals. For 91 counties we could not compute estimates of immunity due to missing data.
RESULTS
Immunological Exposure
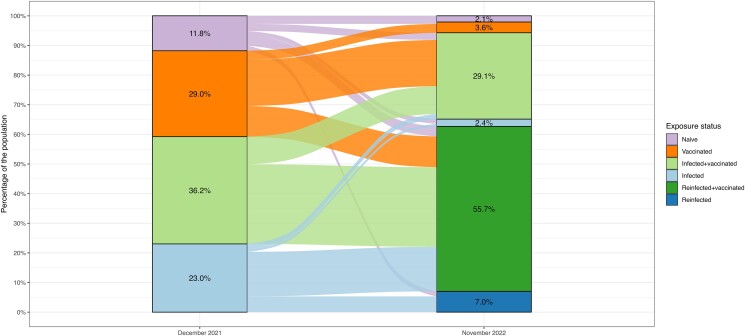
By 9 November 2022, 94% (95% CrI: 79%–99%) of the US population were estimated to have been infected by SARS-CoV-2 at least once, and 65% (35%–88%) were estimated to have experienced multiple infections over the course of the pandemic. Between 2 December 2021 and 9 November 2022, 116 (67–135) million first infections were estimated to have occurred, and 209 (118–292) million reinfections (corresponding to approximately 32% and 65% of the US population, respectively; on average, every person got infected 0.97 times). Combined with vaccination, 97% (95%–99%) were estimated to have some prior immunological exposure to SARS-CoV-2 by 9 November 2022. Figure 2 shows how the US population was assumed to transition through exposure states between 2 December 2021 and 9 November 2022. eFigures 1 and 2 show the immunological exposure states over time between 2 December 2021 and 9 November 2022 at national and state level, respectively.
Figure 2.
Flow of the US population between COVID-19 starting exposure states on 1 December 2021 to ending exposure state on 9 November 2022. F Individuals may make multiple transitions over this time period. Abbreviation: COVID-19, coronavirus disease 2019.
Population Immunity Against Infection
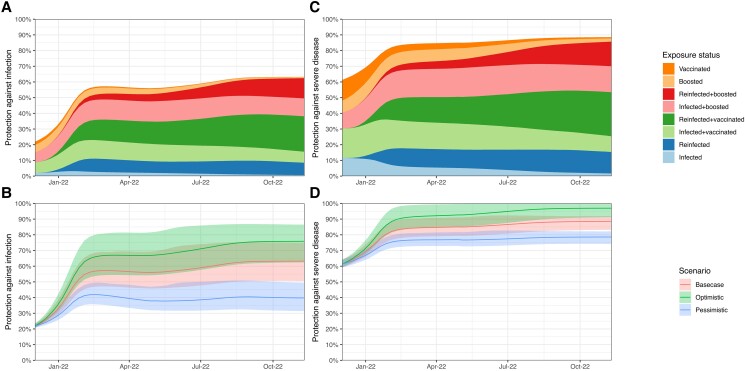
Between 2 December 2021 and 9 November 2022, population protection against an Omicron infection rose from 22% (21%–23%) to 63% (51%–75%) nationally (Figure 3A and 3B , eFigures 3 and 5). In contrast, population immunity against infection by the then circulating pre-Omicron variants in the fall of 2021 (9 November) was estimated to be 51% (47%–56%) [20]. Most of the additional population immunity accrued over the study period was acquired during the initial Omicron surge, with protection against infection estimated as 57% (47%–69%) on 3 March 2022.
Figure 3.
Population protection against infection (A and B) and severe disease (C and D) from the Omicron variant in the United States population between 1 December 2021 and 9 November 2022, by exposure state (A and C) and waning scenario (B and D).
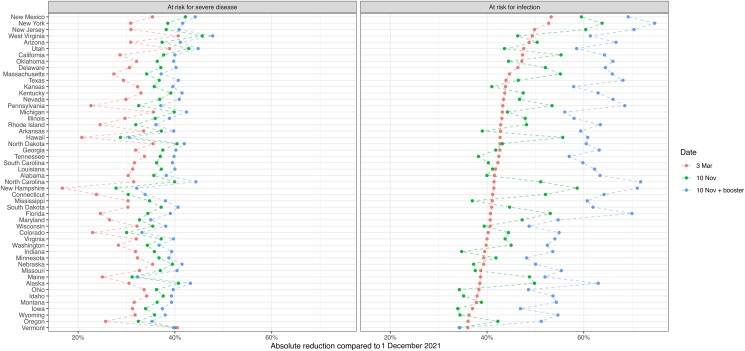
At the state level, protection against infection ranged from 55% (Mississippi, 43%–72%) to 69% (Maine, 54%–83%) as of 9 November 2022. By this date, we estimated that half of the states had time trends in protection against infection that had started to decline, that is, the existing immunity waned more quickly than new immunity was being accumulated (eFigure 3 in the Supplement).
Under the pessimistic waning scenario, national protection against infection on 9 November 2022 was 40% (31%–50%), whereas under the optimistic waning scenario, it was 76% (62%–87%) (Figure 3C and 3D ). Figure 4 shows state-level results for how population protection has increased from 2 December 2021, to 3 March 2022, and then to 9 November 2022. In all but 8 states, population immunity against a new Omicron infection was estimated to have increased or stayed constant between the end of the December–February surge and the end of the study period. In more than one fourth of the counties the population immunity against a new Omicron infection decreased between 3 March 2022 and 9 November 2022 (eFigure 6).
Figure 4.
State-level changes in population protection against infection and severe disease from the Omicron variant between 1 December 2021 and 9 November 2022.
Population Immunity Against Severe Disease
Population protection against severe disease was estimated to have increased over the study period, both nationally (Figure 3C and 3D ) and within every state (Figure 4). Nationally, protection against severe disease resulting from a new Omicron infection rose from 61% (59%–64%) on 2 December 2021, to 84% (79%–90%) on 3 March 2022, to 89% (83%–91%) by 9 November 2022.
Booster Uptake Scenarios
On 9 November, 79% of the US population has initiated a vaccination series (range: 60% Wyoming to 95% Vermont and Maine), 34% has received a first booster (range: 20% North Carolina to 55% Vermont), and 11% has received a second booster (range: 5% North Carolina to 22% Vermont). We evaluated the impact on immunity if all states were able to increase coverage of first and second boosters to the levels seen in the best performing state (55% and 22%, respectively, Vermont). We assumed initial vaccine coverage was saturated. In this hypothetical scenario, national protection against infection would increase 4 percentage points (state range, excluding Vermont: 1.6 [Maine] to 12 [Mississippi]), compared to an increase of 6 percentage points between 3 March and 9 November. Protection against severe disease would go up by 1.1 percentage point (state range, excluding Vermont: 0.5 [Maine] to 2.4 [Florida]), compared to an increase of 5 percentage points between 3 March and 9 November.
DISCUSSION
In this study, we estimated time trends in effective protection against infection and severe disease caused by SARS-CoV-2 Omicron variants in the United States between December 2021 and November 2022. The results of this analysis show that effective protection against SARS-CoV-2 infection and severe disease on 9 November 2022 was substantially higher than on 2 December 2021, before the initial Omicron surge. We also found that population immunity in November 2022 was higher than in the immediate aftermath of the initial surge, with the impact of ongoing transmission and vaccination over the middle of 2022 outweighing the losses in protection due to waning of immunity, both nationally and for the large majority of US states.
Our estimate for national protection against a new Omicron infection (mean estimate 63% on 9 November 2022) is comparable to estimates produced by other studies (IHME [26]: 58% against infection with BA.5 variant, 62% against infection with B.1.1 variant; and McKinsey [27]: 75%). To account for current uncertainty in how immunity wanes we present multiple scenarios, rendering a wide range of potential levels of immunity. In particular, the pessimistic scenario presented shows that population protection against infection would be only around 40% nationally if prior Omicron infections do not offer additional protection against a new (sub)variant. This value is closer to the 22% population immunity estimated for the (at that time) new Omicron variants in December 2021 [20], as compared to population immunity values in November 2022.
By the end of our study period, over 94% of the US population was estimated to have been infected at least once with SARS-CoV-2, and a majority has been infected more than 1 time. Despite these high exposure numbers, there was still substantial population susceptibility to infection with an Omicron variant. In a hypothetical scenario of improved booster coverage, we found that increases in first and second booster uptake to the levels seen in the best-performing state would produce an appreciable improvement in population immunity, with greater relative impact for protection against infection versus severe disease. This additional protection results from the recovery of immunity lost due to waning. A high level of population immunity will reduce the relative benefit of booster vaccines. However, this reduction in relative benefit should not be interpreted as indicating that the absolute benefit is small, particularly for individuals with no recent infection or vaccination, or with elevated susceptibility to severe disease, for whom these population-average results will not apply.
This study has several limitations. First, vaccination data as reported by the CDC are known to be biased for some locations [28], and there is no known alternative data source available that covers the full set of modelled locations accurately. Although the underreporting of vaccination coverage for some locations could lead to underestimation of population immunity, this bias is likely small because, due to high infection rates, most of the population already has some level of immunity. Second, there is substantial uncertainty around the estimates of prior infectious exposure. Our analysis quantified many of the sources of uncertainty in the estimation approach, reflected in broad intervals for the estimated time course of infectious exposure in the analysis. However, this analysis was based on an analytic model that makes structural assumptions about the natural history processes of COVID-19 and how these change across states and counties (described in detail in Chitwood, Russi et al 2021 [15]), not all of which can be represented in the uncertainty analysis. Third, we did not distinguish the levels of immune evasion achieved by successive Omicron-subvariants. Similarly, we did not distinguish the level of immunity conferred by different pre-Omicron variants, or different vaccine products. We did not account for differences in effectiveness between different booster products in the main results, nor in the hypothetical scenario. In the assignment of exposure states, we only explicitly make assumptions about the joint probability of vaccination and first infection and do not account for behavior that changes the probability of changing exposure state (eg, not getting boosted right after an infection).
A substantial fraction of the US population was infected with SARS-CoV-2 infection during December 2021–February 2022, increasing population immunity against Omicron variants in the US population. This level of immunity continued to increase slowly over the course of 2022, as newly acquired immunity from ongoing transmission and vaccination outweighed the effects of immune waning for the majority of modeled locations. Despite the high level of protection at the beginning of the 2022–2023 winter, risk of reinfection and subsequent severe disease remained. A more transmissible or immune evading (sub)variant, changes in behavior, or ongoing waning of immunity could lead to a new SARS-CoV-2 wave, as was observed at the end of 2021.
Supplementary Data
Supplementary materials are available at Clinical Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Notes
Author Contributions. T. C., N. A. M., and J. A. S. conceived and supervised the project. T. C., N. A. M., and J. A. S. acquired funding. F. K. wrote the model code, drafted the original manuscript, and visualized the results. F. K. and M. R. curated the data and executed the analysis. All authors contributed to the development of the methodology, and reviewed and edited the original manuscript.
Disclaimer. The findings, conclusions, and views expressed are those of the author(s) and do not necessarily represent the official position of the Centers for Disease Control and Prevention (CDC), Council of State and Territorial Epidemiologists (CSTE), or National Institutes of Health (NIH).
Financial support. This project has been funded (in part) by contract number 200-2016-91779 with the Centers for Disease Control and Prevention (CDC). V. E. P. reports grants from National Institute of Allergy and Infectious Diseases (NIAID) (grant number R01 AI137093). T. C. reports grants from NIAID (grant number R01 AI112438). N. A. M. reports grants from NIAID R01 (grant number AI146555-01A1), the CDC through the Council of State and Territorial Epidemiologists (grant number NU38OT000297-03), and the CDC (grant number 75D30121F0003). J. A. S. reports funding from the CDC though the Council of State and Territorial Epidemiologists (grant number NU38OT000297-02) and the National Institute on Drug Abuse (grant number 3R37DA01561217S1). N. A. S. reports support from Council of State and Territorial Epidemiologists. F. K. reports support from CDC, NIAID, and Council of State and Territorial Epidemiologists (paid to institution). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplementary Material
Contributor Information
Fayette Klaassen, Department of Global Health and Population, Harvard T.H. Chan School of Public Health, Boston, Massachusetts, USA.
Melanie H Chitwood, Department of Epidemiology of Microbial Diseases and Public Health Modeling Unit, Yale School of Public Health, New Haven, Connecticut, USA.
Ted Cohen, Department of Epidemiology of Microbial Diseases and Public Health Modeling Unit, Yale School of Public Health, New Haven, Connecticut, USA.
Virginia E Pitzer, Department of Epidemiology of Microbial Diseases and Public Health Modeling Unit, Yale School of Public Health, New Haven, Connecticut, USA.
Marcus Russi, Department of Epidemiology of Microbial Diseases and Public Health Modeling Unit, Yale School of Public Health, New Haven, Connecticut, USA.
Nicole A Swartwood, Department of Global Health and Population, Harvard T.H. Chan School of Public Health, Boston, Massachusetts, USA.
Joshua A Salomon, Department of Health Policy, Stanford University School of Medicine, Stanford, California, USA.
Nicolas A Menzies, Department of Global Health and Population, Harvard T.H. Chan School of Public Health, Boston, Massachusetts, USA.
References
- 1. Centers for Disease Control and Prevention . COVID data tracker. Available at: https://covid.cdc.gov/covid-data-tracker. Published 2020. Accessed 22 October 2021.
- 2. Ribeiro Xavier C, Sachetto Oliveira R, da Fonseca Vieira V, Lobosco M, Weber Dos Santos R. Characterisation of Omicron variant during COVID-19 pandemic and the impact of vaccination, transmission rate, mortality, and reinfection in South Africa, Germany, and Brazil. BioTech (Basel) 2022; 11:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Carazo S, Skowronski DM, Brisson M, et al. . Estimated protection of prior SARS-CoV-2 infection against reinfection with the Omicron variant among messenger RNA–vaccinated and nonvaccinated individuals in Quebec, Canada. JAMA Network Open 2022; 5:e2236670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Suzuki R, Yamasoba D, Kimura I, et al. . Attenuated fusogenicity and pathogenicity of SARS-CoV-2 Omicron variant. Nature 2022; 603:700–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Willett BJ, Grove J, MacLean OA, et al. . SARS-CoV-2 Omicron is an immune escape variant with an altered cell entry pathway. Nat Microbiol 2022; 7:1161–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chin ET, Leidner D, Lamson L, et al. . Protection against Omicron from vaccination and previous infection in a prison system. N Engl J Med 2022; 387:1770–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Powell AA, Kirsebom F, Stowe J, et al. . Protection against symptomatic infection with Delta (B.1.617.2) and Omicron (B.1.1.529) BA.1 and BA.2 SARS-CoV-2 variants after previous infection and vaccination in adolescents in England, August, 2021–March, 2022: a national, observational, test-negative, case-control study. Lancet Infect Dis 2023; 23: 435–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Ferdinands JM, Rao S, Dixon BE, et al. . Waning of vaccine effectiveness against moderate and severe COVID-19 among adults in the US from the VISION network: test negative, case-control study. BMJ 2022; 379:e072141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Regev-Yochay G, Gonen T, Gilboa M, et al. . Efficacy of a fourth dose of Covid-19 mRNA vaccine against Omicron. N Engl J Med 2022; 386:1377–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Chalkias S, Eder F, Essink B, et al. . Safety, immunogenicity and antibody persistence of a bivalent Beta-containing booster vaccine against COVID-19: a phase 2/3 trial. Nat Med 2022; 28:2388–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Altarawneh HN, Chemaitelly H, Hasan MR, et al. . Protection against the Omicron variant from previous SARS-CoV-2 infection. N Engl J Med 2022; 386:1288–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Stein C, Nassereldine H, Sorensen RJD, et al. . Past SARS-CoV-2 infection protection against re-infection: a systematic review and meta-analysis. Lancet 2023; 401:833–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Altarawneh HN, Chemaitelly H, Ayoub HH, et al. . Protective effect of previous SARS-CoV-2 infection against Omicron BA.4 and BA.5 subvariants. N Engl J Med 2022; 387:1620–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Bobrovitz N, Ware H, Ma X, et al. . Protective effectiveness of prior SARS-CoV-2 infection and hybrid immunity against Omicron infection and severe disease: a systematic review and meta-regression. Lancet Infect Dis 2023; 23:556–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Chitwood MH, Russi M, Gunasekera K, et al. . Reconstructing the course of the COVID-19 epidemic over 2020 for US states and counties: results of a Bayesian evidence synthesis model. PLoS Comput Biol 2022; 18:e1010465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Dong E, Du H, Gardner L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect Dis 2020; 20:533–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. The New York Times . Coronavirus (Covid-19) data in the United States. Available at: https://github.com/nytimes/covid-19-data. Published 2021. Accessed 10 November 2022.
- 18. Healthdata.gov . Healthdata.gov hospitalizations by HSA. Available at: https://healthdata.gov/Hospital/COVID-19-Reported-Patient-Impact-and-Hospital-Capa/anag-cw7u. Published 2022. Accessed 15 November 2022.
- 19. The COVID tracking project . Available at: https://covidtracking.com/. Accessed 29 October 2021.
- 20. Klaassen F, Chitwood MH, Cohen T, et al. . Population immunity to pre-Omicron and Omicron severe acute respiratory syndrome coronavirus 2 variants in US states and counties through 1 December 2021. Clin Infect Dis 2023; 76:e350–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Household Pulse Survey . US Census Bureau. Available at: https://www.census.gov/data/experimental-data-products/household-pulse-survey.html. Accessed 29 October 2021.
- 22. Ipsos . Axios-Ipsos coronavirus index, waves 34–48. Ann Arbor: Inter-University Consortium for Political and Social Research; [distributor], 2021. Accessed 1 December 2021. [Google Scholar]
- 23. Statistics NCfH . State and national provisional counts. Available at: https://www.cdc.gov/nchs/nvss/vsrr/provisional-tables.htm. Published 2022. Accessed on 14 November 2022.
- 24. R Core Team . R: A language and environment for statistical computing [computer program]. Vienna: R Foundation for Statistical Computing; 2021. [Google Scholar]
- 25. Stan Development . RStan: the R interface to Stan [computer program]. Version R package version 2.21.22020.
- 26. Institute for Health Metrics and Evaluation (IHME) . COVID-19 results briefing. Seattle: IHME, University of Washington, 2022. [Google Scholar]
- 27. Charumilind S, Craven M, Lamb J, Sabow A, Singhal S, Wilson M. When will the COVID-19 pandemic end? McKinsey & Company. Published 2022. Updated 28 July 2022. Accessed 1 November 2022.
- 28. Tiu A, Susswein Z, Merritt A, Bansal S. Characterizing the spatiotemporal heterogeneity of the COVID-19 vaccination landscape. Am J Epidemiol 2022; 191:1792–1802. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.