Abstract
Background
The effectiveness of selective COX-2 inhibitors in preventing colorectal cancer recurrence has been demonstrated, however it is unknown how safe and successful they will be over the long term. As a result, we looked at the efficacy, safety, and consequences of adding COX-2 inhibitors to the treatment plan afterward.
Methods
In patients with advanced colorectal cancer, we compared the efficacy of celecoxib at two different doses (200 mg twice day and 400 mg twice daily) with placebo. To evaluate the impacts of post-treatment, several datasets from inception to June 2022 were searched. Response rate, illness control rate, and 3-year survival were the main results. And evaluated several safety outcomes, particularly those that were susceptible to adverse events.
Results
The study comprised a total of 9 randomized controlled trials (3206 participants). Celecoxib and rofecoxib doidn’t significantly improved the 1–3 year remission rate (OR, 1.57 [95% CI: 0.95–2.57]) and disease control rate (OR, 1.08 [95% CI: 0.99–1.17]). Subgroup analysis of different doses showed that 400 mg of celecoxib significantly improved the response rate (OR, 2.82 [95%CI: 1.20–6.61]). 200 mg celecoxib was not significant (OR, 1.28 [95% CI: 0.66–2.49]). Rofecoxib also did not fully improve disease response rates. Celecoxib at any dose improved 3-year survival (OR, 1.21 [95% CI: 1.02–1.45]). It is important to note that COX-2 inhibitors did not significantly enhance the likelihood of adverse events including gastrointestinal or cardiovascular side effects at any dose.
Conclusions
For patients with advanced colorectal cancer, a reasonable chemoprevention regimen can include celecoxib 400 mg twice daily.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12876-023-02918-w.
Keywords: Colorectal cancer, Chemoprevention, COX-2 inhibitor, Celecoxib, meta-analysis
Introduction
In 2018, colorectal cancer (CRC) was the second leading cause of cancer-related death worldwide. In 2018, CRC is anticipated to have been directly responsible for around 244,000 deaths in Europe [1]. Surgery is a highly effective treatment option when a disease is localized to one area of the body. When CRC has progressed to a later stage, it can be difficult to find a treatment plan that has the intended superior effect. Chemotherapy for patients with advanced CRC commonly consists of 5-fluorouracil (5-FU), leucovorin (LV), oxaliplatin, or irinotecan (FOLFOX or FOLFIRI) [2, 3]. Survival rates, quality of life, and the number of patients requiring secondary operations are all factors in the growing importance of optimizing first-line treatment plans.
Prostaglandin production relies heavily on the cyclooxygenase-2 (COX-2) enzyme, which is highly expressed in inflammatory and tumor tissues [4, 5]. COX-1, COX-2, and COX-3 are all COX isomers, however they all have different purposes in the body. It is a universally accepted fact that human cells and tissues always and everywhere express the maintenance enzyme COX-1. However, the splice variant of COX-1 known as COX-3 is expressed in humans but serves no biological purpose [6]. COX-2 is a pro-inflammatory enzyme that is strongly linked to inflammatory diseases. Angiogenesis, tumor tissue invasion, and resistance to apoptosis are all caused by the inducible enzyme COX-2, which has also been linked to inflammatory conditions and carcinogenesis. Consequentially, the COX-2 and prostaglandin cascades play a significant role in the “inflammation of cancer” [7, 8, 9, 10].
90% of lung cancers, 71% of colon cancers, and 56% of breast malignancies have been found to express COX-2 at moderate to high levels [11, 12, 13]. Using multivariate analysis, researchers found that elevated COX-2 levels independently predicted worse outcomes for cancer patients [14]. Multiple lines of evidence suggest that the cyclooxygenase-2 (COX2) enzyme plays a role in the development and progression of colorectal cancer [15]. COX2 has been shown to promote growth, migration, and invasiveness; inhibit apoptosis; and boost angiogenesis. Celecoxib, rofecoxib, etoricoxib, nabumetone, meloxicam, and etodolac, among others, were associated with a reduced risk of death in a population-based retrospective cohort analysis [16]. Consistent aspirin use in conjunction with any dose of COX-2 inhibitors has also been shown to lower mortality and recurrences in colon cancer [17].
Celecoxib is an essential component of tumor therapy because it inhibits the proliferation of tumor cells by promoting apoptosis and shifting the cell cycle. Selective COX-2 inhibitors have been found to increase life expectancy and enhance quality of life when used in conjunction with standard medical care. Therefore, we performed a meta-analysis and a comprehensive literature search to ascertain the impact of drug addition on patient outcomes.
Methods
This study was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) standard.
Search strategy
We found relevant studies after conducting a complete search of PubMed, Medline, and EMBASE up until June 2022. Also, we looked for new research by reading through older systematic reviews. Further information regarding the search strategy is provided in Table 1. The following inclusion criteria were met by randomized controlled trials (RCTs) and long-term follow-up of that: Celecoxib at any dose was the intervention; the placebo or control group was the comparison group; and the number of patients who acquired colorectal neoplasms was the outcome. Individuals with a higher risk of adenomas (≥ 18 years old) participated in the study. Every participant underwent a polypectomy and shown that their colon was polyp-free before they were assigned to a group. All of the participants had a history of adenomas.
Table 1.
Characteristics of included studies
| Study | Location | Phase | Study period | Mean age (Year) |
ECOG or WHO PS | Sample size (Number of case/control) |
Co-treatment regimen | Celecoxib treatment program (Drugs/dosage (mg/m2)/d/frequency of cycles) |
|---|---|---|---|---|---|---|---|---|
| Jin 2011 | China | II | 2005.6-2008.1 | - | 0–2 | 88(58/30) | folinic acid + fluorouracil + oxaliplatin | 200 mg twice daily no fewer than 8 weeks |
| Köhne 2007 | USA | II | 2002.6-2005.11 | about 70 | 0–2 | 44(23/21) | irinotecan + FA + 5-FU | 200 mg twice daily,800 mg |
| USA | II | 2002.6-2005.11 | about 70 | 1–2 | 41(19/22) | irinotecan + capecitabine | 200 mg twice daily,800 mg | |
| Maiello 2006 | Italy | II | 2003.1-2004.12 | 64 | 0–2 | 81(41/40) | irinotecan + FA + 5-FU | 400 mg twice daily,repeated every 2weeks |
| Haldar 2020 | USA | II | - | 58 | 0–2 | 34(16/18) | etodolac + propranolol | 400 mg twice daily |
| Fenwick 2003 | UK | II | 2000.12-2002.2 | about 65 | 0–2 | 44(23/21) | placebo | 25 mg twice daily,rofecoxib |
| Mostafa 2022 | Egypt | II | 2018.10-2020.7 | about 44 | 0–2 | 54(26/28) | irinotecan + FA + 5-FU | 200 mg twice daily last 3 months |
| Niu 2010 | China | II | 2006.1-2008.12 | 56 | 0–1 | 60(30/30) | irinotecan | 200 mg twice daily |
| Li 2018 | China | II | 2015.2-2016.11 | 63 | 0–2 | 122(61/61) | oxycodone hydrochloride | 200 mg twice daily last 1 months |
| Meyerhardt 2021 | USA | III | 2010.6-2015.11 | 61 | 0–2 | 2524(1263/1261) | folinic acid + fluorouracil + oxaliplatin | 400 mg twice daily |
| Debucquoy 2009 | USA | II | - | - | 0–2 | 80(35/45) | CRT + 5-FU | 400 mg twice daily |
| Hu 2022 | China | II | 2019.5-2021.4 | 18–75 | 0–2 | 34(17/17) | toripalimab | 200 mg twice daily last 2 weeks |
Note:FA = folinic acid, 5-FU = 5-fluorouracil, CRT = Chemotherapy treatment
Outcomes of interest
One of the most notable effectiveness results that stood out to people was the number of colorectal adenomas that returned more than once (advanced adenomas and any adenomas). Each of the following was true of advanced adenomas: a diameter of at least one centimeter; villous or tubulovillous histology; high-grade dysplasia; intramucosal carcinoma; invasive malignancy. On the spectrum of adenoma development, from benign to malignant, we find invasive adenomas (classified as one or two small [1 cm] tubular adenomas or serrated polyps without cytologic dysplasia). Colorectal cancer rates, all-cause mortality rates, serious adverse events, cardiovascular disease rates, kidney disease rates, blood pressure levels, and any reports of post-randomization follow-up were also considered. The study authors determined that adverse events were considered serious if they led to death, hospitalization, severe gastrointestinal bleeding, cardiovascular problems, or the cessation of an intervention. Cardiovascular death, myocardial infarction, stroke, heart failure, and thromboembolic event were all categorized as “serious cardiovascular events” by the study’s authors. Renal and hypertensive effects include elevated blood creatinine levels, fluid retention and edema, hypertension, proteinuria, and renal failure. On top of that, hypertension has been linked to at least a few deaths in recent history. We analyzed the effects of celecoxib on the recurrence of colorectal neoplasia in patients who had been on the drug for at least two years after treatment had ended.
Data extraction and quality assessment
Two reviewers reviewed the primary papers and then used a standardized form to record data on the study, the participants, and the treatment. There was no more confusion after the group discussion than there had been before it. The efficacy results were gathered using an updated version of the intention-to-treat analysis (i.e., subjects who received at least one dose of celecoxib at any dose and had at least one colonoscopy after randomization). The data on safety outcomes were compiled and analyzed using the intention-to-treat principle and the original trial participants who were randomly allocated to each study arm. Those who were unable to be reached for further evaluation were found to have experienced no negative outcomes. The Rob2.0 tool for evaluating the quality of randomized parallel and crossover trials was used for this study. This tool includes the following content: randomization/allocation process, deviations from expected interventions, missing outcome data, outcome measurement, and selective outcome reporting [18, 19]. By reviewing the papers until a consensus was reached, the reviewers were able to address their concerns about the inclusion of certain studies, the collection and processing of data, and the appraisal of the potential for bias.
Data synthesis and statistical analysis
Review Manager’s meta-analysis made use of the random-effects model developed by DerSimonian and Laird. Therefore, we were able to estimate pooled risk ratios and 95% confidence intervals that took into account heterogeneity both within and between trials. We used I2 statistics to assess whether or not primary outcomes varied significantly across trials, with values above 50% indicating significant heterogeneity. Due to the small sample size, it was not able to determine whether or not publication bias had occurred. As a result, it became more difficult to determine whether or not an observed disparity was the product of a genuine cause or a mere coincidence. Subgroup analysis was performed with respect to the different celecoxib dosages, including 200 mg twice day (400 mg/day) and 400 mg twice daily (800 mg/day). Sensitivity analysis was also carried out to further ensure the reliability of the findings.
Result
Characteristics of included studies
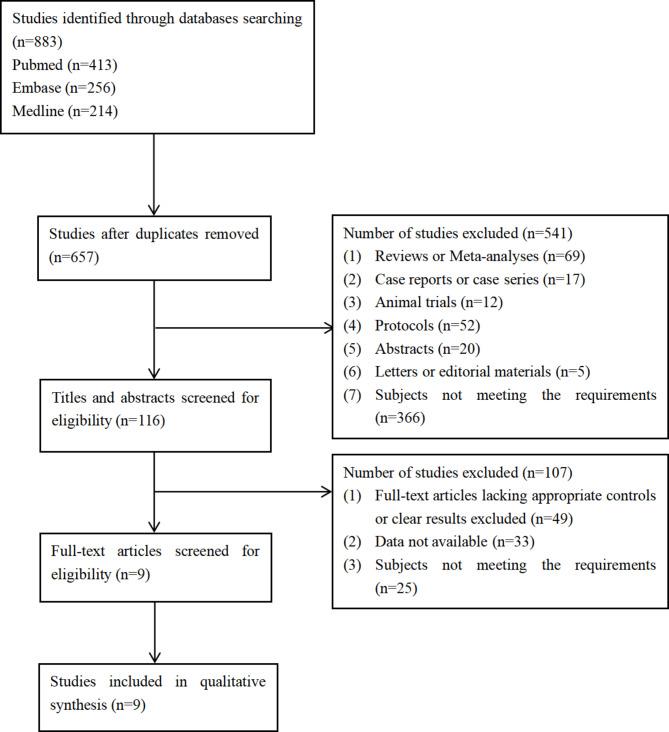
Figure 1 shows the screening process of this study. A total of 9 articles [20, 21, 22, 23, 24, 25, 26, 27, 28] were included for data extraction and analysis. Table 1 describes the characteristics of 9 RCTs. A total of 3206 patients were recruited in nine studies. The gender distribution included in the assessment was equal. Of these, 1594 patients received standard of care or placebo regimens, main drugs are folinic acid + fluorouracil + oxaliplatin, irinotecan + FA + 5-FU, oxycodone hydrochloride or others. And 1612 received supplemental COX-2 inhibitor therapy (Celecoxib), the frequency of cycles is 200 mg (7 articles) or 400 mg (4 articles) twice daily for more than 2 weeks. In another study, the drug regimen was 25 mg rofecoxib twice daily. 9 studies were of good quality according to the quality assessment of the modified Jadad scale. In studies with lower scores, random number generation was not reported, nor did they report on how randomization was concealed.
Fig. 1.
Screening flow chart of included literature
Quality assessment
Supplement S1 indicates the risk bias of the included studies. All studies did not provide a specific allocation plan, but were considered to be of high quality.
Effects on the primary efficacy outcomes
Benefit of response rate(RR)
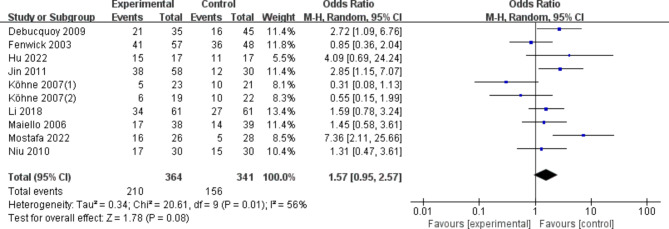
According to the results of the meta-analysis, the use of any dose (200 mg or 400 mg of celecoxib) or rofecoxib did not statistically significantly improve response in patients with advanced CRC compared with placebo or control regimens (OR, 1.57 [95% CI: 0.95–2.57], Fig. 2). In the sensitivity analysis, the results were consistent with the primary analysis. Excluding studies using rofecoxib, the results were still robust (OR, 1.70 [95%CI: 0.99–2.90]). Using dose as a grouping criterion, the use of 200 mg of celecoxib resulted in the same results (OR, 1.28 [95% CI: 0.66–2.49]), and the use of 400 mg of celecoxib significantly improved the response of patients with advanced CRC (OR, 2.82 [95%CI: 1.20–6.61]).
Fig. 2.
OR of COX-2 inhibitors compared to placebo in CRC treatment
Benefit of disease control rate(DCR)
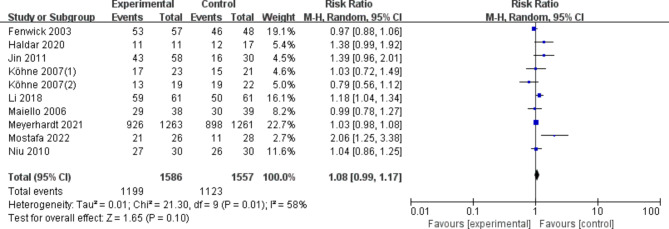
According to the results of the meta-analysis, the use of any dose (200 mg or 400 mg of celecoxib) or rofecoxib did not statistically significantly improve disease control rates in patients with advanced CRC compared with placebo or control regimens (OR, 1.08 [95%CI: 0.99–1.17], Fig. 3). In the sensitivity analysis, the results were consistent with the primary analysis. Excluding studies using rofecoxib, the results were still robust (OR, 1.11 [95%CI: 0.99–1.23]). Using dose as a grouping criterion, the same results were obtained with celecoxib 200 mg (OR, 1.19 [95% CI: 0.95–1.49]) and celecoxib 400 mg (OR, 1.09 [95% CI: 0.95–1.25]).
Fig. 3.
DCR of COX-2 inhibitors compared to placebo in CRC treatment
Benefit of survival and quality of life improvement
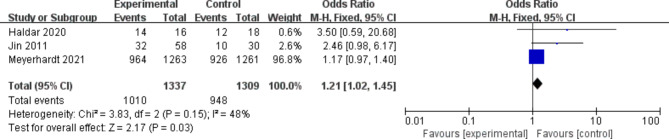
According to the results of the meta-analysis, the use of any dose (200 mg or 400 mg of celecoxib) statistically significantly improved 3-year survival in patients with advanced CRC compared with placebo or control regimens (OR, 1.21 [95% CI: 1.02–1.45], Fig. 4). In the sensitivity analysis, the results were consistent with the primary analysis. Using dose as a grouping criterion, the same results were obtained with celecoxib 200 mg (OR, 2.46 [95% CI: 0.98–6.17]) and celecoxib 400 mg (OR, 1.18 [95% CI: 0.99–1.41]) The results did not improve the patient’s condition. What is more significant is that the improvement rate of the quality of life of the patients in the control group is 40%, while the improvement rate of the patients in the experimental group is as high as 66%, and various physiological functions have recovered to a certain extent.
Fig. 4.
3-year survival of COX-2 inhibitors compared to placebo in CRC treatment
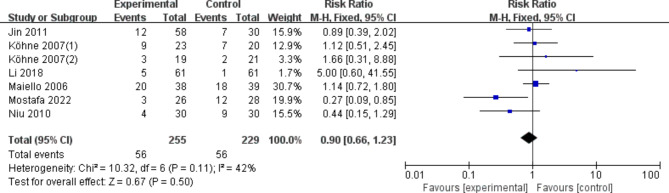
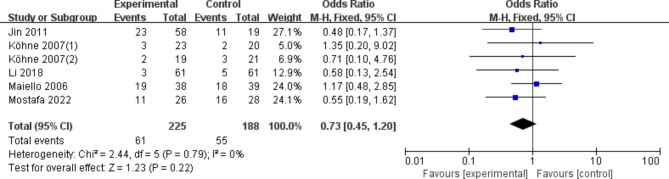
Effects on the primary safety outcomes
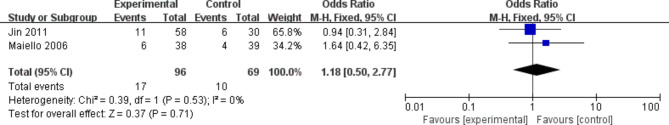
According to the results of the meta-analysis, the use of either dose (200 mg or 400 mg of celecoxib) did not statistically significantly improve the nausea/vomiting response rate (OR, 0.73 [95% CI: 0.45–1.20], Fig. 5) compared with placebo or control regimens. In the sensitivity analysis, the results were consistent with the primary analysis. Using dose as a grouping criterion, the same results were obtained with celecoxib 200 mg (OR, 0.62 [95%CI: 0.30–1.26]) and celecoxib 400 mg (OR, 0.86 [95% CI: 0.43–1.70]). There was no statistically significant increase in the incidence of diarrhea with either dose (200 mg or 400 mg of celecoxib) compared with placebo or control regimens (OR, 0.90 [95% CI: 0.66–1.23], Fig. 6). In In the sensitivity analysis, the results were consistent with the preliminary analysis. Using dose as a grouping criterion, the same results were obtained with celecoxib 200 mg (OR, 1.00 [95% CI: 0.64–1.58]) and celecoxib 400 mg (OR, 0.80 [95% CI: 0.52–1.21]). The use of either dose (200 mg or 400 mg of celecoxib) did not statistically significantly increase the incidence of Oral mucositis (OR, 1.18 [95% CI: 0.50–2.77], Fig. 7) compared with placebo or control regimens. Using dose as a grouping criterion, the same results were obtained with celecoxib 200 mg (OR, 0.94 [95% CI: 0.31–2.84]) and celecoxib 400 mg (OR, 1.64 [95% CI: 0.42–6.35]). In addition, neurotoxicity, Myelosuppression, etc. showed no significant difference.
Fig. 5.
Nausea/vomiting of COX-2 inhibitors compared to placebo in CRC treatment
Fig. 6.
Diarrhoea of COX-2 inhibitors compared to placebo in CRC treatment
Fig. 7.
Oral mucositis of COX-2 inhibitors compared to placebo in CRC treatment
Discussion
COX-2 drugs are considerably protective against CRC recurrence, according to prior meta-analyses [29, 30, 31, 32, 33]. The nature of each COX-2 inhibitor and the intricate interactions between dose and baseline have prevented broader attention, even if the data from numerous studies are encouraging, particularly the rise in adverse effects. One of the few selective COX-2 inhibitors studied in numerous CRC preventive studies is celecoxib. Further research on his advantages is also made possible by the extensive application.
The preferred technique for preventing sporadic CRC is screening colonoscopy with removal of adenomatous polyps because it is associated with lower mortality [34, 35]. Additionally, despite routine screening, a small percentage of persons still develop CRC before the advised interval for surveillance, presumably as a result of missed or insufficient polyp removal or quickly growing tumors [36, 37]. COX-2 inhibitors are significant possibilities for easing the burden of CRC, according to numerous research.
The results of the initial randomized controlled trial and other meta-analyses are expanded upon in our study. The probability of advanced metachronous cancers was dramatically decreased by COX-2 inhibitors, particularly celecoxib. We discovered that in individuals with high-risk tumors at baseline, the advantages of COX-2 inhibitors may outweigh the risks of significant side effects. The long-term cardiovascular safety of COX-2 inhibitors is of concern, in addition to the short-term risk of major side events that we saw in our meta-analysis [38]. It’s important to note that such incidents are mostly observed in populations with a history of cardiovascular risk factors or disease. Conventional NSAIDs may not be linked to an elevated cardiovascular risk, according to recent literature [39]. Therefore, in populations with a low baseline risk of cardiovascular disease and an intermediate-high baseline risk of CRC, COX-2 inhibitors may be thought of as chemopreventive medicines.
The most popular COX-2 inhibitors in the FOLFIRI regimen, celecoxib and rofecoxib, were observed to significantly improve ORR and DCR in this study. The response rate shown a rise that was statistically significant, particularly in the short-term efficacy. These results are in line with a prior study that found patients with locally advanced rectal cancer who received celecoxib in combination to preoperative chemoradiation experienced an increase in excellent response [40, 41, 42]. The fact that celecoxib was well tolerated and there was no discernible difference in the frequency of adverse effects between the two groups is more concerning. Celecoxib was well tolerated at both higher dosages (800 mg/day) and lower doses (400 mg/day), according to earlier clinical investigations in CRC patients. Additionally, the current data are consistent with earlier studies that suggested celecoxib with chemotherapy did not increase toxicity in comparison to placebo [43, 44].
The serum VEGF levels were markedly lowered in the celecoxib/FOLFIRI group [45]. Celecoxib inhibits angiogenesis by lowering VEGF levels with the use of FOLFIRI and celecoxib. According to reports, celecoxib inhibits NF-kB by boosting the IkB inhibitor protein, lowering blood levels of CXCL5, and blocking the AKT/NF-kB pathway that is involved in cancer and angiogenesis [46]. Our positive findings regarding celecoxib’s impact on CXCL5 are in line with earlier research showing that increased CXCL5 levels are linked to colorectal metastases and a bad prognosis. Celecoxib and other selective COX-2 inhibitors have both shown antiangiogenic effects in a variety of in vitro and in vivo models [47, 48]. Selective COX-2 drugs have antiangiogenic action, however the exact mechanism is unknown. Endothelial cells may directly respond to selective COX-2 inhibitors [49, 50]. Due to CRC metastases, rofecoxib medication was linked to a 60% decrease in in vitro PGE2 synthesis. In fact, early results from a phase II trial involving celecoxib in combination with a variety of chemotherapy treatments for patients with metastatic colorectal cancer suggest that this strategy merits a controlled clinical examination.
However, this study has some limitations. Firstly, due to limited literature and research methods, it is not possible to conduct all subgroup analyses based on different drugs and doses. Secondly, it is difficult to unify dose equivalents across different trials and treatment plans. Although we attempted to standardize this definition during the data extraction process, it may not be fully effective. Third, the research on adding COX-2 in the treatment scheme is really lacking, and more high-quality randomized Scientific control are still needed.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplement S1 Risk of bias graph and risk of bias summary.
Acknowledgements
Not applicable.
Authors’ contributions
Ting Hu and Cheng-Jiang Liu designed the study and supervised the overall project; Xiaoming Yin and WenJuan Tang participated in collecting data; LanFang Yin, Hui Bai and FangFang Liu participated in data collecting and analysis; Dan Wang and YiLei Li provided the statistical analysis and wrote the manuscript.
Funding
No Funding.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This is a systematic review and meta-analysis, ethics approval and consent to participate are not applicable.
Consent for publication
Not applicable. This study does not involve human participants.
Competing interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ting Hu and Cheng-Jiang Liu contributed equally to this work.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Douillard JY, Cunningham D, Roth AD, Navarro M, James RD, Karasek P, Jandik P, Iveson T, Carmichael J, Alakl M, Gruia G, Awad L, Rougier P. Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet. 2000;355(9209):1041-7. doi: 10.1016/s0140-6736(00)02034-1. Erratum in: Lancet 2000;355(9212):1372. [DOI] [PubMed]
- 3.de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer G, Papamichael D, Le Bail N, Louvet C, Hendler D, de Braud F, Wilson C, Morvan F, Bonetti A. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18(16):2938–47. doi: 10.1200/JCO.2000.18.16.2938. [DOI] [PubMed] [Google Scholar]
- 4.Cao Y, Prescott SM. Many actions of cyclooxygenase-2 in cellular dynamics and in cancer. J Cell Physiol. 2002;190(3):279–86. doi: 10.1002/jcp.10068. [DOI] [PubMed] [Google Scholar]
- 5.Williams CS, Mann M, DuBois RN. The role of cyclooxygenases in inflammation, cancer, and development. Oncogene. 1999;18(55):7908–16. doi: 10.1038/sj.onc.1203286. [DOI] [PubMed] [Google Scholar]
- 6.Liu B, Qu L, Yan S. Cyclooxygenase-2 promotes tumor growth and suppresses tumor immunity. Cancer Cell Int. 2015;15:106. doi: 10.1186/s12935-015-0260-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sharma S, Stolina M, Yang SC, Baratelli F, Lin JF, Atianzar K, Luo J, Zhu L, Lin Y, Huang M, Dohadwala M, Batra RK, Dubinett SM. Tumor cyclooxygenase 2-dependent suppression of dendritic cell function. Clin Cancer Res. 2003;9(3):961–8. [PubMed] [Google Scholar]
- 8.Tsujii M, Kawano S, Tsuji S, Sawaoka H, Hori M, DuBois RN. Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell. 1998;93(5):705–16. doi: 10.1016/s0092-8674(00)81433-6. [DOI] [PubMed] [Google Scholar]
- 9.Tsujii M, DuBois RN. Alterations in cellular adhesion and apoptosis in epithelial cells overexpressing prostaglandin endoperoxide synthase 2. Cell. 1995;83(3):493–501. doi: 10.1016/0092-8674(95)90127-2. [DOI] [PubMed] [Google Scholar]
- 10.Nzeako UC, Guicciardi ME, Yoon JH, Bronk SF, Gores GJ. COX-2 inhibits Fas-mediated apoptosis in cholangiocarcinoma cells. Hepatology. 2002;35(3):552–9. doi: 10.1053/jhep.2002.31774. [DOI] [PubMed] [Google Scholar]
- 11.Soslow RA, Dannenberg AJ, Rush D, Woerner BM, Khan KN, Masferrer J, Koki AT. COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer. 2000;89(12):2637–45. doi: 10.1002/1097-0142(20001215)89:12<2637::aid-cncr17>3.0.co;2-b. [DOI] [PubMed] [Google Scholar]
- 12.Brabender J, Park J, Metzger R, Schneider PM, Lord RV, Hölscher AH, Danenberg KD, Danenberg PV. Prognostic significance of cyclooxygenase 2 mRNA expression in non-small cell lung cancer. Ann Surg. 2002;235(3):440-3. doi: 10.1097/00000658-200203000-00017. Erratum in: Ann Surg 2002;235(6):896-7. [DOI] [PMC free article] [PubMed]
- 13.Lim SJ, Kim HJ, Kim JY, Park K, Lee CM. Expression of HuR is associated with increased cyclooxygenase-2 expression in uterine cervical carcinoma. Int J Gynecol Pathol. 2007;26(3):229–34. doi: 10.1097/01.pgp.0000236946.82334.07. [DOI] [PubMed] [Google Scholar]
- 14.Urakawa H, Nishida Y, Naruse T, Nakashima H, Ishiguro N. Cyclooxygenase-2 overexpression predicts poor survival in patients with high-grade extremity osteosarcoma: a pilot study. Clin Orthop Relat Res. 2009;467(11):2932–8. doi: 10.1007/s11999-009-0814-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dannenberg AJ, Altorki NK, Boyle JO, Dang C, Howe LR, Weksler BB, Subbaramaiah K. Cyclo-oxygenase 2: a pharmacological target for the prevention of cancer. Lancet Oncol. 2001;2(9):544–51. doi: 10.1016/S1470-2045(01)00488-0. [DOI] [PubMed] [Google Scholar]
- 16.Huang WW, Hsieh KP, Huang RY, Yang YH. Role of cyclooxygenase-2 inhibitors in the survival outcome of colorectal cancer patients: a population-based cohort study. Kaohsiung J Med Sci. 2017;33(6):308–14. doi: 10.1016/j.kjms.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 17.Ng K, Meyerhardt JA, Chan AT, Sato K, Chan JA, Niedzwiecki D, Saltz LB, Mayer RJ, Benson AB, 3rd, Schaefer PL, Whittom R, Hantel A, Goldberg RM, Venook AP, Ogino S, Giovannucci EL, Fuchs CS. Aspirin and COX-2 inhibitor use in patients with stage III colon cancer. J Natl Cancer Inst. 2014;107(1):345. doi: 10.1093/jnci/dju345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 19.Higgins JPT, Li T, Deeks JJ,Chapter 6: Choosing e!ect measures and computing estimates of e!ect. In:, Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editors. Cochrane handbook for systematic reviews of interventions version 6.0 (updated July 2019). Cochrane; 2019. Available from www.training.cochrane.org/handbook (http://www.training.cochrane.org/handbook).
- 20.Jin CH, Wang AH, Chen JM, Li RX, Liu XM, Wang GP, Xing LQ. Observation of curative efficacy and prognosis following combination chemotherapy with celecoxib in the treatment of advanced colorectal cancer. J Int Med Res. 2011;39(6):2129–40. doi: 10.1177/147323001103900609. [DOI] [PubMed] [Google Scholar]
- 21.Köhne CH, De Greve J, Hartmann JT, Lang I, Vergauwe P, Becker K, Braumann D, Joosens E, Müller L, Janssens J, Bokemeyer C, Reimer P, Link H, Späth-Schwalbe E, Wilke HJ, Bleiberg H, Van Den Brande J, Debois M, Bethe U, Van Cutsem E. Irinotecan combined with infusional 5-fluorouracil/folinic acid or capecitabine plus celecoxib or placebo in the first-line treatment of patients with metastatic colorectal cancer. EORTC study 40015. Ann Oncol. 2008;19(5):920–6. doi: 10.1093/annonc/mdm544. [DOI] [PubMed] [Google Scholar]
- 22.Maiello E, Giuliani F, Gebbia V, Di Renzo N, Pezzella G, Romito S, Mallamaci R, Lopez M, Colucci G. Gruppo Oncologico dell’Italia Meridionale. FOLFIRI with or without celecoxib in advanced colorectal cancer: a randomized phase II study of the Gruppo Oncologico dell’Italia Meridionale (GOIM) Ann Oncol. 2006;17(Suppl 7):vii55–9. doi: 10.1093/annonc/mdl952. [DOI] [PubMed] [Google Scholar]
- 23.Haldar R, Ricon-Becker I, Radin A, Gutman M, Cole SW, Zmora O, Ben-Eliyahu S. Perioperative COX2 and β-adrenergic blockade improves biomarkers of tumor metastasis, immunity, and inflammation in colorectal cancer: a randomized controlled trial. Cancer. 2020;126(17):3991–4001. doi: 10.1002/cncr.32950. [DOI] [PubMed] [Google Scholar]
- 24.Fenwick SW, Toogood GJ, Lodge JP, Hull MA. The effect of the selective cyclooxygenase-2 inhibitor rofecoxib on human colorectal cancer liver metastases. Gastroenterology. 2003;125(3):716–29. doi: 10.1016/s0016-5085(03)01061-8. [DOI] [PubMed] [Google Scholar]
- 25.Mostafa TM, Alm El-Din MA, Rashdan AR. Celecoxib as an adjuvant to chemotherapy for patients with metastatic colorectal cancer: a randomized controlled clinical study. Saudi Med J. 2022;43(1):37–44. doi: 10.15537/smj.2022.43.1.20210574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meyerhardt JA, Shi Q, Fuchs CS, Meyer J, Niedzwiecki D, Zemla T, Kumthekar P, Guthrie KA, Couture F, Kuebler P, Bendell JC, Kumar P, Lewis D, Tan B, Bertagnolli M, Grothey A, Hochster HS, Goldberg RM, Venook A, Blanke C, O’Reilly EM, Shields AF. Effect of Celecoxib vs Placebo added to Standard Adjuvant Therapy on Disease-Free Survival among patients with stage III Colon cancer: the CALGB/SWOG 80702 (Alliance) Randomized Clinical Trial. JAMA. 2021;325(13):1277–86. doi: 10.1001/jama.2021.2454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Debucquoy A, Roels S, Goethals L, Libbrecht L, Van Cutsem E, Geboes K, Penninckx F, D’Hoore A, McBride WH, Haustermans K. Double blind randomized phase II study with radiation + 5-fluorouracil+/-celecoxib for resectable rectal cancer. Radiother Oncol. 2009;93(2):273–8. doi: 10.1016/j.radonc.2009.08.006. [DOI] [PubMed] [Google Scholar]
- 28.Hu H, Kang L, Zhang J, Wu Z, Wang H, Huang M, Lan P, Wu X, Wang C, Cao W, Hu J, Huang Y, Huang L, Wang H, Shi L, Cai Y, Shen C, Ling J, Xie X, Cai Y, He X, Dou R, Zhou J, Ma T, Zhang X, Luo S, Deng W, Ling L, Liu H, Deng Y. Neoadjuvant PD-1 blockade with toripalimab, with or without celecoxib, in mismatch repair-deficient or microsatellite instability-high, locally advanced, colorectal cancer (PICC): a single-centre, parallel-group, non-comparative, randomised, phase 2 trial. Lancet Gastroenterol Hepatol. 2022;7(1):38–48. doi: 10.1016/S2468-1253(21)00348-4. [DOI] [PubMed] [Google Scholar]
- 29.Dulai PS, Singh S, Marquez E, Khera R, Prokop LJ, Limburg PJ, Gupta S, Murad MH. Chemoprevention of colorectal cancer in individuals with previous colorectal neoplasia: systematic review and network meta-analysis. BMJ. 2016;355:i6188. doi: 10.1136/bmj.i6188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Veettil SK, Saokaew S, Lim KG, Ching SM, Phisalprapa P, Chaiyakunapruk N. Comparative effectiveness of chemopreventive interventions for colorectal cancer: protocol for a systematic review and network meta-analysis of randomised controlled trials. J Gastrointest Oncol. 2016;7(4):595–602. doi: 10.21037/jgo.2016.04.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rostom A, Dubé C, Lewin G, Tsertsvadze A, Barrowman N, Code C, Sampson M, Moher D, U.S. Preventive Services Task Force Nonsteroidal anti-inflammatory drugs and cyclooxygenase-2 inhibitors for primary prevention of colorectal cancer: a systematic review prepared for the U.S. Preventive Services Task Force. Ann Intern Med. 2007;146(5):376–89. doi: 10.7326/0003-4819-146-5-200703060-00010. [DOI] [PubMed] [Google Scholar]
- 32.Dubé C, Rostom A, Lewin G, Tsertsvadze A, Barrowman N, Code C, Sampson M, Moher D, U.S. Preventive Services Task Force The use of aspirin for primary prevention of colorectal cancer: a systematic review prepared for the U.S. Preventive Services Task Force. Ann Intern Med. 2007;146(5):365–75. doi: 10.7326/0003-4819-146-5-200703060-00009. [DOI] [PubMed] [Google Scholar]
- 33.Veettil SK, Lim KG, Ching SM, Saokaew S, Phisalprapa P, Chaiyakunapruk N. Effects of aspirin and non-aspirin nonsteroidal anti-inflammatory drugs on the incidence of recurrent colorectal adenomas: a systematic review with meta-analysis and trial sequential analysis of randomized clinical trials. BMC Cancer. 2017;17(1):763. doi: 10.1186/s12885-017-3757-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nishihara R, Wu K, Lochhead P, Morikawa T, Liao X, Qian ZR, Inamura K, Kim SA, Kuchiba A, Yamauchi M, Imamura Y, Willett WC, Rosner BA, Fuchs CS, Giovannucci E, Ogino S, Chan AT. Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med. 2013;369(12):1095–105. doi: 10.1056/NEJMoa1301969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Brenner H, Stock C, Hoffmeister M. Effect of screening sigmoidoscopy and screening colonoscopy on colorectal cancer incidence and mortality: systematic review and meta-analysis of randomised controlled trials and observational studies. BMJ. 2014;348:g2467. doi: 10.1136/bmj.g2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Singh S, Singh PP, Murad MH, Singh H, Samadder NJ. Prevalence, risk factors, and outcomes of interval colorectal cancers: a systematic review and meta-analysis. Am J Gastroenterol. 2014;109(9):1375–89. doi: 10.1038/ajg.2014.171. [DOI] [PubMed] [Google Scholar]
- 37.Adler J, Robertson DJ. Interval Colorectal Cancer after Colonoscopy: exploring explanations and solutions. Am J Gastroenterol. 2015;110(12):1657–64. doi: 10.1038/ajg.2015.365. [DOI] [PubMed] [Google Scholar]
- 38.Rostom A, Dube C, Lewin GUS. Preventive services Task Force evidence Syntheses, formerly systematic evidence reviews. Use of aspirin and NSAIDs to prevent Colorectal Cancer. Rockville (MD).Agency for Healthcare Research and Quality; 2007. [PubMed]
- 39.McGettigan P, Henry D. Cardiovascular risk and inhibition of cyclooxygenase: a systematic review of the observational studies of selective and nonselective inhibitors of cyclooxygenase 2. JAMA. 2006;296(13):1633-44. doi: 10.1001/jama.296.13.jrv60011. Epub 2006 Sep 12. [DOI] [PubMed]
- 40.Yi L, Zhang W, Zhang H, Shen J, Zou J, Luo P, Zhang J. Systematic review and meta-analysis of the benefit of celecoxib in treating advanced non-small-cell lung cancer. Drug Des Devel Ther. 2018;12:2455–66. doi: 10.2147/DDDT.S169627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Guo Q, Li Q, Wang J, Liu M, Wang Y, Chen Z, Ye Y, Guan Q, Zhou Y. A comprehensive evaluation of clinical efficacy and safety of celecoxib in combination with chemotherapy in metastatic or postoperative recurrent gastric cancer patients: a preliminary, three-center, clinical trial study. Med (Baltim) 2019;98(27):e16234. doi: 10.1097/MD.0000000000016234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhou X, Wang X, Zhao Y, Yi C. The role of celecoxib for colorectal cancer treatment: a systematic review. Transl Cancer Res. 2018;7:1527–36. doi: 10.21037/tcr.2018.11.22. [DOI] [Google Scholar]
- 43.Chen EY, Blanke CD, Haller DG, Benson AB, Dragovich T, Lenz HJ, Robles C, Li H, Mori M, Mattek N, Sanborn RE, Lopez CD. A phase II study of Celecoxib with Irinotecan, 5-Fluorouracil, and leucovorin in patients with previously untreated Advanced or metastatic colorectal Cancer. Am J Clin Oncol. 2018;41(12):1193–8. doi: 10.1097/COC.0000000000000465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hammond WA, Swaika A, Mody K. Pharmacologic resistance in colorectal cancer: a review. Ther Adv Med Oncol. 2016;8(1):57–84. doi: 10.1177/1758834015614530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xie C, Xu X, Wang X, Wei S, Shao L, Chen J, Cai J, Jia L. Cyclooxygenase-2 induces angiogenesis in pancreatic cancer mediated by prostaglandin E2. Oncol Lett. 2018;16(1):940–8. doi: 10.3892/ol.2018.8786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zuo C, Hong Y, Qiu X, Yang D, Liu N, Sheng X, Zhou K, Tang B, Xiong S, Ma M, Liu Z. Celecoxib suppresses proliferation and metastasis of pancreatic cancer cells by down-regulating STAT3 / NF-kB and L1CAM activities. Pancreatology. 2018;18(3):328–33. doi: 10.1016/j.pan.2018.02.006. [DOI] [PubMed] [Google Scholar]
- 47.Masferrer JL, Leahy KM, Koki AT, Zweifel BS, Settle SL, Woerner BM, Edwards DA, Flickinger AG, Moore RJ, Seibert K. Antiangiogenic and antitumor activities of cyclooxygenase-2 inhibitors. Cancer Res. 2000;60(5):1306–11. [PubMed] [Google Scholar]
- 48.Sawaoka H, Tsuji S, Tsujii M, Gunawan ES, Sasaki Y, Kawano S, Hori M. Cyclooxygenase inhibitors suppress angiogenesis and reduce tumor growth in vivo. Lab Invest. 1999;79(12):1469–77. [PubMed] [Google Scholar]
- 49.Leahy KM, Ornberg RL, Wang Y, Zweifel BS, Koki AT, Masferrer JL. Cyclooxygenase-2 inhibition by celecoxib reduces proliferation and induces apoptosis in angiogenic endothelial cells in vivo. Cancer Res. 2002;62(3):625–31. [PubMed] [Google Scholar]
- 50.Dicker AP, Williams TL, Grant DS. Targeting angiogenic processes by combination rofecoxib and ionizing radiation. Am J Clin Oncol. 2001;24(5):438–42. doi: 10.1097/00000421-200110000-00005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplement S1 Risk of bias graph and risk of bias summary.
Data Availability Statement
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.