Key Points
Question
What are the clinical implications of removing race correction for African American patients undergoing surgery for lung cancer?
Findings
Race correction is commonly performed with transition to race-neutral equations associated with an absolute 10% reduction in predicted pulmonary function test (PFT) values for African American patients undergoing surgery. In this quality improvement study, surgeons randomized to the vignette with African American race–corrected PFTs were more likely to recommend lobectomy compared with surgeons randomized to vignettes with other race or multiracial–corrected or race-neutral PFTs.
Meaning
These findings suggest that removing race correction from PFTs may affect surgeon decision-making, and although race correction should be removed from PFTs, this should occur with efforts to improve surgeon education regarding changes to PFTs and investigate alternative diagnostic studies for lung function.
This quality improvement study examines the implications of removing race correction from pulmonary function tests for health care systems, patients, and surgeons.
Abstract
Importance
Removal of race correction in pulmonary function tests (PFTs) is a priority, given that race correction inappropriately conflates race, a social construct, with biological differences and falsely assumes worse lung function in African American than White individuals. However, the impact of decorrecting PFTs for African American patients with lung cancer is unknown.
Objectives
To identify how many hospitals providing lung cancer surgery use race correction, examine the association of race correction with predicted lung function, and test the effect of decorrection on surgeons’ treatment recommendations.
Design, Setting, and Participants
In this quality improvement study, hospitals participating in a statewide quality collaborative were contacted to determine use of race correction in PFTs. For hospitals performing race correction, percent predicted preoperative and postoperative forced expiratory volume in 1 second (FEV1) was calculated for African American patients who underwent lung cancer resection between January 1, 2015, and September 31, 2022, using race-corrected and race-neutral equations. US cardiothoracic surgeons were then randomized to receive 1 clinical vignette that differed by the use of Global Lung Function Initiative equations for (1) African American patients (percent predicted postoperative FEV1, 49%), (2) other race or multiracial patients (percent predicted postoperative FEV1, 45%), and (3) race-neutral patients (percent predicted postoperative FEV1, 42%).
Main Outcomes and Measures
Number of hospitals using race correction in PFTs, change in preoperative and postoperative FEV1 estimates based on race-neutral or race-corrected equations, and surgeon treatment recommendations for clinical vignettes.
Results
A total of 515 African American patients (308 [59.8%] female; mean [SD] age, 66.2 [9.4] years) were included in the study. Fifteen of the 16 hospitals (93.8%) performing lung cancer resection for African American patients during the study period reported using race correction, which corresponds to 473 African American patients (91.8%) having race-corrected PFTs. Among these patients, the percent predicted preoperative FEV1 and postoperative FEV1 would have decreased by 9.2% (95% CI, −9.0% to −9.5%; P < .001) and 7.6% (95% CI, −7.3% to −7.9%; P < .001), respectively, if race-neutral equations had been used. A total of 225 surgeons (194 male [87.8%]; mean [SD] time in practice, 19.4 [11.3] years) were successfully randomized and completed the vignette items regarding risk perception and treatment outcomes (76% completion rate). Surgeons randomized to the vignette with African American race–corrected PFTs were more likely to recommend lobectomy (79.2%; 95% CI, 69.8%-88.5%) compared with surgeons randomized to the other race or multiracial–corrected (61.7%; 95% CI, 51.1%-72.3%; P = .02) or race-neutral PFTs (52.8%; 95% CI, 41.2%-64.3%; P = .001).
Conclusions and Relevance
Given the findings of this quality improvement study, surgeons should be aware of changes in PFT testing because removal of race correction PFTs may change surgeons’ treatment decisions and potentially worsen existing disparities in receipt of lung cancer surgery among African American patients.
Introduction
Removal of race correction in pulmonary function tests (PFTs) is a priority of the US government, health care systems, and practitioners to address inappropriate use of race within clinical algorithms.1,2,3 Many clinical algorithms using race correction inappropriately conflate race, a social construct, with biological differences. Current use of race correction in PFTs is predicated on the false assumption that African American individuals have inherently worse lung function than White individuals.4,5,6 To date, discussion about eliminating race-corrected PFTs has centered on how race-corrected PFT values underdiagnose African American patients with pulmonary conditions, such as asthma or chronic obstructive pulmonary disease.7,8,9,10 However, little is known about the implications for African American patients who need surgery for lung cancer.
Forced expiratory volume in 1 second (FEV1) from preoperative PFTs is commonly used by surgeons to determine a patient’s surgical risk and to provide treatment recommendations. For African American patients, the predicted FEV1 is often race corrected.4,5,6 As a result, the race-corrected percent predicted FEV1 is artificially higher than if a race-neutral (ie, without race correction) predicted FEV1 were used.
On one hand, this artificial inflation in percent predicted FEV1 values may result in African American patients having lung function values that appear too good to receive diagnoses for chronic lung conditions.7,8,9,10 On the other hand, removing race correction (decorrection) may more accurately reflect true lung function but will also lower the percent predicted FEV1, which may lead surgeons to perceive African American individuals as having worse lung function and a higher risk from surgical treatment.11 Therefore, it is important to understand how race-corrected PFTs may affect surgical decision-making among African American patients undergoing surgery for lung cancer.
In this context, we used a multimethods approach to examine the implications of removing race correction in PFTs for health care systems, patients, and surgeons. First, we surveyed hospitals in a statewide quality collaborative to determine the scale of race correction in PFTs among surgical patients. Second, for hospitals reporting race correction, we evaluated the change in percent predicted FEV1 when decorrecting PFTs from race-corrected to race-neutral PFTs among African American patients who underwent resection for lung cancer. Third, we conducted a randomized clinical vignette study of board-certified cardiothoracic surgeons to test the association of decorrection in PFTs with treatment recommendations and risk perception for a hypothetical African American patient with lung cancer.
Methods
This quality improvement study was exempted from regulation by the University of Michigan Institutional Review Board because it is a quality improvement study with secondary analysis of deidentified data and the survey was deemed as research involving benign behavioral interventions in conjunction with the collection of information from an adult through written or oral consent. Participants in the clinical vignette survey provided written consent before randomization. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline.
Statewide Collaborative Assessment
Data Source
We identified all African American patients who received lung cancer surgery between January 1, 2015, and September 31, 2022, in the Michigan Society of Thoracic and Cardiovascular Surgeons Quality Collaborative (MSTCVS-QC) General Thoracic Surgery database. This quality collaborative includes hospitals in Michigan who perform lung cancer resection and report data to the Society of Thoracic Surgeons General Thoracic Database. Approximately 65% of all lung cancer operations in Michigan are performed at hospitals participating in the quality collaborative. Sixteen of 21 hospitals in the quality collaborative performed operations on at least 1 African American patient during the study period. First, we contacted each of these 16 hospitals by telephone, and pulmonary function laboratory staff at each hospital were asked whether race correction was used in testing. Second, we identified all African American patients in the MSTCVS-QC database who received lung cancer surgery and extracted their patient data, including sex, age, height, percent predicted FEV1, self-reported race, tumor location, and operation performed. Lung cancer diagnosis was identified with International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) and International Statistical Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM). Operations were coded based on Current Procedural Terminology codes.
The racial group African American is used in this manuscript to reflect the current racial and ethnic group that has a Global Lung Function Initiative (GLI) reference equation. Notably, the African American GLI reference equation is supposed to be applied only to African American individuals as opposed to being applied to the diversity of ethnicity, nationality, and ancestry reflected among individuals who may self-identify as racially Black.
Statistical Analysis
All African American patients who underwent an operation for lung cancer at a hospital that reported using race correction in PFTs were included in the analysis. First, patient age, sex, and height were used with the GLI reference equations to calculate predicted African American or race-neutral FEV1 in liters.12 On the basis of each patient’s documented preoperative percent predicted FEV1 from MSTCVS data, an actual preoperative FEV1 value in liters was calculated by multiplying the preoperative percent predicted FEV1 with the African American race–corrected predicted FEV1. This actual FEV1 value in liters was then divided by each patient’s predicted race-neutral FEV1 in liters to calculate the preoperative percent predicted FEV1 using the race-neutral equation. For patients with documented tumor location and laterality, percent predicted postoperative FEV1 values were also calculated using African American race–corrected and race-neutral equations. Paired t tests were conducted to evaluate the difference in percent predicted FEV1 based on African American or race-neutral equations.
Clinical Vignette Survey
Participant Recruitment
We recruited volunteers from a proprietary email list of board-certified thoracic surgeons in August 2022. Surgeons were thoracic surgeons, but subspecialization in general thoracic surgery, adult cardiac surgery, or congenital heart surgery could not be delineated. The email framed the study on surgical decision-making for patients with early-stage lung cancer without referring to topics of race, ethnicity, or race correction in PFTs to avoid biasing participant enrollment. Participant recruitment was conducted similar to a randomized clinical trial. After agreeing to take part in the study, participants were randomized 1:1:1 to 1 clinical vignette that differed only by percent predicted postoperative FEV1.13 By randomizing participants after enrollment, response bias may be reduced if successful randomization is achieved.
Vignette Instrument
We created a single vignette of a hypothetical 170-cm tall, 71-year-old African American woman with a 3.6-cm mass confirmed to be adenocarcinoma without nodal involvement (stage 1B) being evaluated for a right upper lobectomy. The vignette included age, history of present illness, medical history, smoking history, vital signs, physical examination findings, percent predicted postoperative FEV1, and diffusion capacity for carbon monoxide (Dlco). We also included data on functional status, chest computed tomography findings, positron emission tomography/computed tomography findings, computed tomography–guided biopsy results, and invasive mediastinal staging. The vignette included a photograph of a hypothetical patient that had been previously validated for race and age.14 The full vignette can be found in the eAppendix in Supplement 1.
PFT Values in the Vignette
Within this single vignette, participants were randomized to receive 1 of 3 percent predicted postoperative FEV1 values. We randomized percent predicted postoperative FEV1 values instead of including all PFT values, because percent predicted postoperative FEV1 is a major determinant of surgical risk and used in clinical guidelines for treatment recommendations.15 Only spirometry values are race corrected; therefore, although Dlco is also important to surgical decision-making, Dlco values are not race corrected and so did not vary in the vignette.
Lobectomy, representing formal anatomical resection, was the primary treatment focus given current National Comprehensive Cancer Network recommendations for treatment for stage 1B disease.15 Prior recommendations have identified percent predicted postoperative FEV1 values of less than 30% as a contraindication for lobectomy, whereas patients with values above 60% are considered candidates for lobectomy.16 For patients with values between 30% and 60%, surgeon approaches to further testing and treatment vary.11,16 For this reason, we chose to use values within this range for the hypothetical patient in the vignette to highlight implications for patients for whom there are no clear recommendations.
The hypothetical patient had a 3.6-cm mass in the right upper lobe, and percent predicted postoperative FEV1 values were calculated for a right upper lobectomy using African American race–corrected, other race or multiracial–corrected, and without a race correction factor (ie, race-neutral) GLI equations.12 Using the same base vignette of the hypothetical patient, surgeons were 1:1:1 randomized to receive 1 of 3 percent predicted postoperative FEV1 values based on the GLI reference equations for (1) African American (percent predicted postoperative FEV1, 49%), (2) other race or multiracial (percent predicted postoperative FEV1, 45%), and (3) race-neutral (percent predicted postoperative FEV1, 42%) patients. All other information in the clinical vignette was held constant. All equations used are included the eAppendix in Supplement 1.
Outcomes
The primary outcome was choice of surgical treatment (lobectomy, wedge resection, or stereotactic body radiation therapy). Secondary outcomes included surgical risk perception for postoperative complications (measured on a scale of 0%-100%) and predicted postoperative mortality (measured on a scale of 0%-100%).
Vignette Administration
All vignettes were pilot tested within the University of Michigan Section of Thoracic Surgery and the University of Michigan Center for Bioethics and Social Sciences in Medicine Working group, a group of multidisciplinary survey experts. An email containing a link to the clinical vignette was sent from the research study team to board-certified thoracic surgeons. The vignettes were administered online using Qualtrics (Qualtrics International Inc), a survey development platform. Participation in the study was voluntary, and participants were offered the opportunity to enroll in a lottery for a $100 gift card after study completion. Four reminder emails were sent to all surgeons.
Statistical Analysis
Similar to a clinical trial, we report a recruitment rate defined as the proportion of all eligible participants who enrolled in the study and a completion rate defined as the proportion of all participants completing the study similar to a dropout rate. Respondent demographic information was collected at the end of the survey, including gender, race, ethnicity, place of birth, geographic region of clinical practice, practice location, practice setting, years in practice, and percentage of practice that is general thoracic surgery. Assessment of randomization was evaluated based on any differences in surgeon characteristics across the 3 randomized groups. Multinomial logistic and linear regressions were performed to assess the outcomes of treatment recommendation and risk perception with pairwise comparison among the 3 groups. Analyses were performed using Stata software, version 17 (StataCorp).
Results
Statewide Collaborative Assessment
A total of 515 African American patients (308 [59.8%] female and 207 [40.2%] male; mean [SD] age, 66.2 [9.4] years) were included in the study (Table 1). Fifteen of the 16 hospitals (93.8%) performing lung cancer resection for African American patients during the study period reported using race correction, which corresponds to 473 African American patients (91.8%) having race-corrected PFTs.
Table 1. African American Patient Demographic Characteristics According to MSTCVS-QC Data.
| Characteristic | Value (N = 515)a |
|---|---|
| Sex | |
| Female | 308 (59.8) |
| Male | 207 (40.2) |
| Age, mean (SD), y | 66.2 (9.4) |
| Height, mean (SD), cm | 168.7 (10.2) |
| Weight, mean (SD), kg | 80.6 (19.5) |
| Predicted FEV1, median (IQR), % | 86 (71-100) |
| Race correction used | 473 (91.8) |
| Tumor locationb | |
| Right upper lobe | 103 (20.0) |
| Right middle lobe | 39 (7.6) |
| Right lower lobe | 20 (3.9) |
| Left upper lobe | 61 (11.8) |
| Left lower lobe | 29 (5.6) |
| Undocumented | 263 (51.1) |
| Operation performed | |
| Bilobectomy | 7 (1.3) |
| Lobectomy | 341 (67.1) |
| Segmentectomy | 29 (5.7) |
| Wedge resection | 79 (15.6) |
| Pneumonectomy | 5 (1.0) |
| Other | 47 (9.3) |
Abbreviations: FEV1, forced expiratory volume in 1 second; MSTCVS-QC, Michigan Society of Thoracic and Cardiovascular Surgeons Quality Collaborative.
Data are presented as number (percentage) of patients unless otherwise indicated.
A total of 252 patients had full documentation of both the lobe involved and laterality.
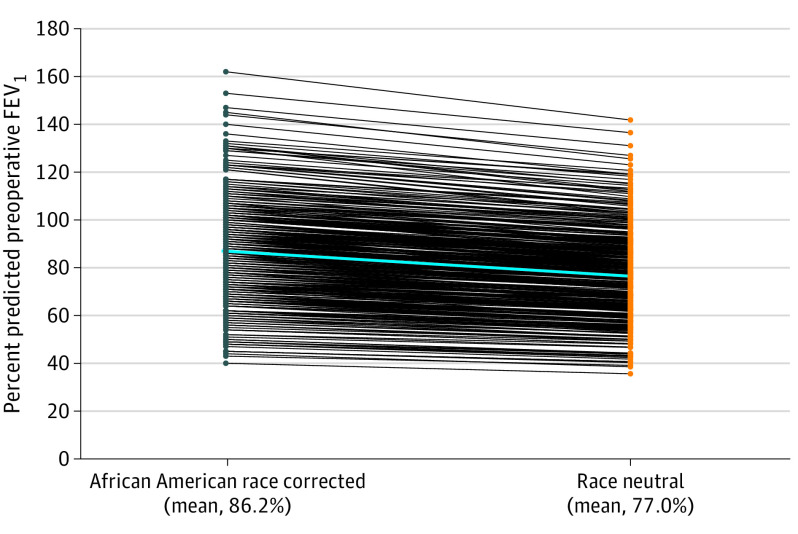
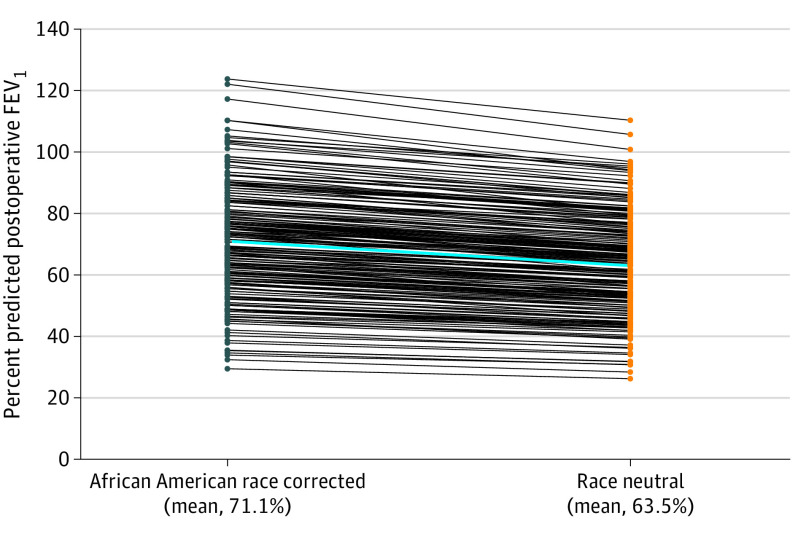
Among all patients, the mean percent predicted FEV1 with race correction was 86.2% (95% CI, 84.%-88.2%) compared with 77.0% (95% CI, 75.2%-78.7%) with race-neutral equations. For these patients, decorrection from African American race–corrected to race-neutral PFTs resulted in a significant reduction in preoperative percent predicted FEV1 of 9.2% (95% CI, −9.0% to −9.5%; P < .001) (Figure 1). Of the 224 patients with recorded laterality available to calculate postoperative percent predicted FEV1 for lobectomy, a change from race-corrected to race-neutral PFTs resulted in a significant reduction in postoperative percent predicted FEV1 of 7.6% (95% CI, −7.3% to −7.9%; P < .001) (Figure 2). Among all patients, the mean postoperative percent predicted FEV1 with race correction was significantly higher than the mean postoperative percent predicted FEV1 with the race-neutral equation (71.1% vs 63.5%; P < .001).
Figure 1. Percent Predicted Preoperative Forced Expiratory Volume in 1 Second (FEV1) Values Based on African American Race–Corrected and Race-Neutral Global Lung Initiative Equations.
The blue line represents the mean population change in percent predicted preoperative FEV1 when transitioning from African American race–corrected to race-neutral equations. Mean change was 9.2% (95% CI, −9.0% to −9.5%; P < .001).
Figure 2. Percent Predicted Postoperative Forced Expiratory Volume in 1 Second (FEV1) Values Based on African American Race–Corrected and Race-Neutral Global Lung Initiative Equations.
The blue line represents the mean population change in percent predicted postoperative FEV1 when transitioning from African American corrected to race neutral equations. Mean change was 7.6% (95% CI, −7.3% to −7.9%; P < .001).
Clinical Vignette
Surgeon Characteristics
Of the 4521 US cardiothoracic surgeons who were emailed, 297 surgeons volunteered to participate (6.6% recruitment rate). Of these surgeons, 225 (194 [87.8%] men and 27 [12.2%] women; 27 [12.4%] Asian, 7 [3.2%] Black or African American, 1 [0.5%] Native Hawaiian or Pacific Islander, 173 [79.7%] White, and 9 [4.2%] of other race; and 13 [5.9%] Hispanic or Latino) fully completed the vignette items regarding risk perception and treatment outcomes (75.8% completion rate). Participant characteristics were well balanced across the 3 randomized groups (Table 2). The surgeons had a mean (SD) of 19.4 (11.3) years of practice with general thoracic surgery, representing a mean (SD) of 68.5% (36.1%) of their clinical practice. Surgeons represented all geographic areas and clinical practices, including academic (88 [39.8%]), community practice (73 [33.0%]), private practice (57 [26.0%]), and Veterans Affairs (3 [1.4%]).
Table 2. Surgeon Demographic Characteristics by Race-Corrected Pulmonary Function Test Groupa.
| Characteristic | Total (N = 221)b | Race neutral (n = 69) | Other race or multiracial corrected (n = 80) | African American race corrected (n = 72) | P value |
|---|---|---|---|---|---|
| Gender | |||||
| Man | 194 (87.8) | 60 (87.0) | 69 (86.3) | 65 (90.3) | .73 |
| Woman | 27 (12.2) | 9 (13.0) | 11 (13.8) | 7 (9.7) | |
| Race | |||||
| Asian | 27 (12.4) | 10 (14.5) | 6 (7.9) | 11 (15.3) | .46 |
| Black or African American | 7 (3.2) | 2 (2.9) | 4 (5.3) | 1 (1.4) | |
| Native Hawaiian or Pacific Islander | 1 (0.5) | 0 | 1 (1.3) | 0 | |
| White | 173 (79.7) | 53 (76.8) | 61 (80.3) | 59 (81.9) | |
| Other | 9 (4.2) | 4 (5.8) | 4 (5.3) | 1 (1.4) | |
| Ethnicity | |||||
| Hispanic or Latino | 13 (5.9) | 2 (2.9) | 7 (8.8) | 4 (5.6) | .32 |
| Not Hispanic or Latino | 208 (94.1) | 67 (97.1) | 73 (91.3) | 68 (94.4) | |
| Place of birth | |||||
| US | 178 (80.5) | 56 (81.2) | 62 (77.5) | 60 (83.3) | .66 |
| Outside US | 43 (19.5) | 13 (18.8) | 18 (22.5) | 12 (16.7) | |
| Geographic region | |||||
| West | 39 (17.7) | 18 (26.1) | 10 (12.5) | 11 (15.3) | .17 |
| Midwest | 66 (29.9) | 16 (23.2) | 28 (35.0) | 22 (30.6) | |
| South | 67 (30.3) | 24 (34.8) | 24 (30.0) | 19 (26.4) | |
| Northeast | 49 (22.2) | 11 (15.9) | 18 (22.5) | 20 (27.8) | |
| Practice location | |||||
| Rural | 17 (7.7) | 8 (11.6) | 5 (6.3) | 4 (5.6) | .64 |
| Suburban | 62 (28.1) | 20 (29.0) | 23 (28.8) | 19 (26.4) | |
| Urban | 142 (64.3) | 41 (59.4) | 52 (65.0) | 49 (68.1) | |
| Practice setting | |||||
| Academic | 88 (39.8) | 22 (31.9) | 36 (45.0) | 30 (41.7) | .51 |
| Community practice | 73 (33.0) | 23 (33.3) | 23 (28.8) | 27 (37.5) | |
| Private practice | 57 (26.0) | 23 (33.3) | 20 (25.0) | 14 (19.4) | |
| Veterans Affairs | 3 (1.4) | 1 (1.5) | 1 (1.25) | 1 (1.4) | |
| Time in practice, mean (SD), y | 19.4 (11.3) | 21.2 (11.7) | 18.9 (11.3) | 18.3 (10.9) | .27 |
| Part of practice as general thoracic surgery, mean (SD), % | 68.5 (36.1) | 57.3 (37.9) | 71.5 (35.0) | 75.8 (33.3) | .57 |
Data are presented as number (percentage) of surgeons unless otherwise indicated.
Four surgeons did not provide complete demographic information.
Treatment Recommendation
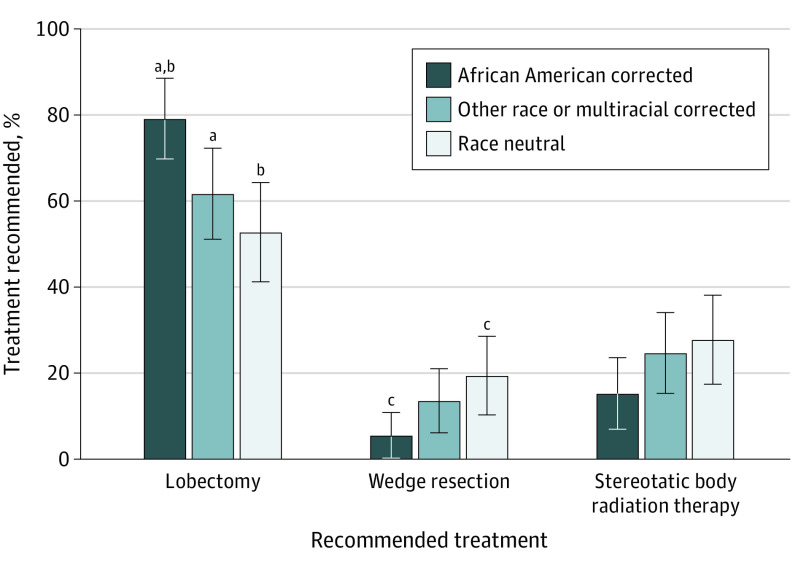
Surgeons randomized to receive postoperative percent predicted FEV1 values that used the African American race–corrected GLI equation were significantly more likely to recommend lobectomy (79.2%; 95% CI, 69.8%-88.5%) compared with surgeons randomized to the other race or multiracial–corrected equation (61.7%; 95% CI, 51.1%-72.3%; P = .02) or race-neutral equation (52.8%; 95% CI, 41.2%-64.3%; P = .001) (Figure 3). Surgeons randomized to the race-neutral equation were significantly more likely to recommend wedge resection (19.4%; 95% CI, 10.3%-28.6%) than surgeons randomized to the African American race–corrected equation (5.6%; 95% CI, 0.26%-10.9%; P = .01) (Figure 3). Although surgeons randomized to the African American race–corrected equation recommended stereotactic body radiation therapy less frequently (15.3%; 95% CI, 7.0%-23.6%) than those randomized to the other race or multiracial group (24.7%; 95% CI, 15.30%-34.08%) and race-neutral group (27.8%; 95% CI, 17.4%-38.1%), there was no significant difference among the groups.
Figure 3. Surgeon Treatment Recommendation Depending on Whether Race Correction Is Used in Pulmonary Function Tests .
Data reflect surgeon recommendation for treatment (lobectomy, wedge resection, or stereotactic body radiation therapy) based on randomization to the African American race–corrected (forced expiratory volume in 1 second [FEV1], 49%), other race or multiracial–corrected (FEV1, 45%), or race-neutral (FEV1, 42%) groups. All surgeons received a vignette about a 71-year-old African American woman with a 20–pack-year smoking history and a 3.6-cm right upper lobe mass biopsy specimen proven to be invasive adenocarcinoma without nodal disease on positron emission tomography/computed tomography and invasive mediastinal staging. Error bars indicate 95% CIs of treatment recommended by surgeons.
aAfrican American corrected vs other race or multiracial–corrected for lobectomy (P = .02).
bAfrican American race corrected vs race neutral for lobectomy (P = .001).
cAfrican American race corrected vs race neutral for wedge resection (P = .01).
Risk Estimation
There was no difference in surgeon estimation of the probability of complications following right lobectomy among the 3 groups (African American race–corrected: 20.1%; 95% CI, 16.7%-23.4%; other race or multiracial-corrected: 22.2%; 95% CI, 19.0%-25.3%; and race neutral, 21.0%; 95% CI, 17.7%-24.4%) (eFigure 1 in Supplement 1). Surgeons estimated a higher probability of mortality in the other race or multiracial–corrected group (5.7%; 95% CI, 4.5%-7.0%) compared with the African American race–corrected group (3.8%; 95% CI, 2.4%-5.1%; P = .03) (eFigure 2 in Supplement 1).
Discussion
We identified several important findings that highlight major implications of removal of race correction from PFTs for lung cancer surgery. First, we found that 91.8% of African American patients undergoing surgery for lung cancer had race-corrected PFT values. Second, if these patients’ PFT values were switched from race corrected to race neutral, the percent predicted FEV1 values would decrease by approximately 10 percentage points. Third, we found that the use of race-corrected or race-neutral PFT values directly influences surgeon decision-making for patients with lung cancer through the use of randomized clinical vignettes. Surgeons were more than 25 percentage points less likely to recommend lobectomy when race-neutral values were used compared with African American race–corrected values. Taken together, these results demonstrate that transitioning from race-corrected to race-neutral equations could have broad impact, could result in unintended harm to African American patients with lung cancer, and may widen existing disparities in lung cancer treatment.17,18
Prior research4,7,8,9,10 focused on race correction in PFTs has focused on medical conditions without considering the effect on surgical decision-making. For instance, a prior assessment10 of the use of race correction in PFTs among biracial pediatric patients demonstrated a 15% difference in predicted FEV1 values based on whether the patient’s racial category was selected as African American or White. Furthermore, race correction has been associated with underestimating the severity of chronic obstructive pulmonary disease.19 Race correction has also been shown to have limited clinical benefit in predicting outcomes among patients with chronic respiratory disease.8,9 Our findings expand on this prior work by demonstrating that race correction in PFTs is incredibly common for surgical patients, is associated with a major change in diagnostic testing values, and directly influences surgeons’ treatment recommendations.
To the best of our knowledge, there have been no empirical evaluations of the role race correction in PFTs plays in surgical decision-making. Our findings highlight the potential problems that may arise if race correction is removed from PFTs. Among African American patients in our study, applying race-neutral testing was associated with a nearly 10-percentage-point absolute reduction in percent predicted FEV1 and postoperative percent predicted FEV1. This reduction occurs even though there is no actual change in a patient’s FEV1 but is solely due to the switch from using race-corrected to race-neutral prediction equations.
This reduction in estimated lung function has important implications for treatment recommendations, operative risk, and postoperative patient-reported outcomes, such as physical function or dyspnea.14,20 As a comparison, the use of race correction in estimated glomerular filtration rate (eGFR) has historically been associated with a 16% to 21% difference in GFR values compared with race-neutral values and has been associated with racial disparities in medical and surgical care for patients with chronic kidney disease.21 This finding has led to large-scale movement across health care systems, transplant centers, specialties, and professional societies to eliminate the use of GFR and investigate the use of other diagnostic tests of kidney function.21,22,23,24,25 In comparison, similar broad investigation regarding the implications of race correction in PFTs across medical and surgical patients and the need for different assessments of lung function has been more limited.26
Our findings raise concern that if PFTs become race neutral African American patients may be offered potentially curative lung cancer surgery less frequently if surgeons do not realize that lower predicted PFT values are a function of a change in equations and not a change in actual lung function. Surgical decision-making reflects a complex process for which PFTs help guide overall risk perception. We found that the use of race-neutral PFTs resulted in surgeons being less likely to offer lobectomy and more likely to perform wedge resection compared with when African American race–corrected PFT values were used, which occurred despite no actual change in actual risk or lung function. This finding raises concern that for African American patients who are borderline candidates for lung cancer resection, the removal of race correction, without additional diagnostic testing or surgeon education, may inadvertently exacerbate well-documented racial inequities in the receipt of lobectomy and worsen long-term oncologic outcomes.18,27
Our work has several important implications, given the US Department of Health and Human Services’ proposed recent rule change that would prohibit the use of discriminatory clinical algorithms and the American Thoracic Society’s recent guidelines to remove race correction in PFTs.1,2,3,28 First, these results should not be used as a justification for the continuance of race correction in PFTs, given the false conflation of race, a social construct based on physical appearance, with biology. The presence of race correction in clinical algorithms, including PFTs, is rooted in racist beliefs regarding the biological inferiority of African Americans.4,5,6,17 However, our findings highlight that removing race correction cannot happen in isolation and must occur alongside large-scale efforts to develop interventions to educate clinicians, improve shared decision-making with patients, and create new guidelines and preoperative diagnostic studies of lung function for lung cancer surgery.
Limitations
This study should be interpreted in light of several limitations. First, the overall recruitment rate for participation in this study was low. However, the internal validity of our study was maintained given successful randomization of surgeons into the 3 vignette study groups. Participants in this study also broadly match the demographic characteristics of cardiothoracic surgeons nationally, which supports external validity.29,30 Second, our vignettes did not allow for surgeons to ask for additional preoperative testing, such as cardiopulmonary exercising testing, which some surgeons may use as supplementary tests to guide decision-making. Third, we did not include segmentectomy in this analysis as a surgical option because of its low overall use compared with other surgical methods and in order to maximize statistical power for analyses. In addition, we did not include actual FEV1 values in liters in our vignette; however, postoperative percent predicted FEV1 is the recommended value that is used to determine risk.14
Conclusions
In this quality improvement study, the use of race correction was common, and the switch from race-corrected to race-neutral PFTs resulted in a nearly 10-percentage-point reduction in PFT values. Surgeons exposed to race-neutral PFT values were less likely to offer surgery than surgeons exposed to race-corrected PFTs. These results highlight the need to carefully consider the potential unintended consequences of removing race correction from PFTs to avoid exacerbating existing racial disparities in lung cancer surgery.
eAppendix. Vignette Instrument
eFigure 1. Surgeon Estimation of the Probability of Post-Operative Complications for Right Upper Lobectomy Depending on Whether Race-Correction Is Used in Pulmonary Function Tests (PFTs)
eFigure 2. Surgeon Estimation of the Probability of Post-Operative Mortality for Right Upper Lobectomy Depending on Whether Race-Correction Is Used in Pulmonary Function Tests (PFTs)
Data Sharing Statement
References
- 1.Khazanchi R, Tsai J, Eneanya ND, Han J, Maybank A. Leveraging Affordable Care Act section 1557 to address racism in clinical algorithms. Health Affairs Forefront. September 30, 2022. Accessed June 28, 2023. https://www.healthaffairs.org/content/forefront/leveraging-affordable-care-act-section-1557-address-racism-clinical-algorithms
- 2.Shachar C, Gerke S. Prevention of bias and discrimination in clinical practice algorithms. JAMA. 2023;329(4):283-284. doi: 10.1001/jama.2022.23867 [DOI] [PubMed] [Google Scholar]
- 3.Goodman KE, Morgan DJ, Hoffmann DE. Clinical algorithms, antidiscrimination laws, and medical device regulation. JAMA. 2023;329(4):285-286. doi: 10.1001/jama.2022.23870 [DOI] [PubMed] [Google Scholar]
- 4.Vyas DA, Eisenstein LG, Jones DS. Hidden in plain sight - reconsidering the use of race correction in clinical algorithms. N Engl J Med. 2020;383(9):874-882. doi: 10.1056/NEJMms2004740 [DOI] [PubMed] [Google Scholar]
- 5.Braun L. Race correction and spirometry: why history matters. Chest. 2021;159(4):1670-1675. doi: 10.1016/j.chest.2020.10.046 [DOI] [PubMed] [Google Scholar]
- 6.Braun L. Race, ethnicity and lung function: a brief history. Can J Respir Ther. 2015;51(4):99-101. [PMC free article] [PubMed] [Google Scholar]
- 7.Moffett AT, Eneanya ND, Halpern SD, Weissman GE. The impact of race correction on the interpretation of pulmonary function testing among Black patients. Am J Respir Crit Care Med. 2021;203:A1030. doi: 10.1164/ajrccm-conference.2021.203.1_MeetingAbstracts.A1030 [DOI] [Google Scholar]
- 8.Baugh AD, Shiboski S, Hansel NN, et al. Reconsidering the utility of race-specific lung function prediction equations [published correction appears in Am J Respir Crit Care Med. 2022;206(2):230]. Am J Respir Crit Care Med. 2022;205(7):819-829. doi: 10.1164/rccm.202105-1246OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Elmaleh-Sachs A, Balte P, Oelsner EC, et al. Race/ethnicity, spirometry reference equations, and prediction of incident clinical events: the Multi-Ethnic Study of Atherosclerosis (MESA) Lung Study. Am J Respir Crit Care Med. 2022;205(6):700-710. doi: 10.1164/rccm.202107-1612OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pittman JE, Van Rie A, Davis SD. Spirometry in biracial children: how adequate are race-based reference equations? Arch Pediatr Adolesc Med. 2011;165(6):573-574. doi: 10.1001/archpediatrics.2011.76 [DOI] [PubMed] [Google Scholar]
- 11.Pennathur A, Brunelli A, Criner GJ, et al. ; AATS Clinical Practice Standards Committee: Thoracic Surgery . Definition and assessment of high risk in patients considered for lobectomy for stage I non-small cell lung cancer: the American Association for Thoracic Surgery expert panel consensus document. J Thorac Cardiovasc Surg. 2021;162(6):1605-1618.e6. doi: 10.1016/j.jtcvs.2021.07.030 [DOI] [PubMed] [Google Scholar]
- 12.Quanjer PH, Stanojevic S, Cole TJ, Stocks J. Quanjer GLI-2012 Regression Equation and Lookup Tables. Global Lung Function Initiative. Accessed June 28, 2023. https://www.ers-education.org/lr/show-details/?idP=138978
- 13.Evans SC, Roberts MC, Keeley JW, et al. Vignette methodologies for studying clinicians’ decision-making: Validity, utility, and application in ICD-11 field studies. Int J Clin Health Psychol. 2015;15(2):160-170. doi: 10.1016/j.ijchp.2014.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Minear M, Park DC. A lifespan database of adult facial stimuli. Behav Res Methods Instrum Comput. 2004;36(4):630-633. doi: 10.3758/BF03206543 [DOI] [PubMed] [Google Scholar]
- 15.National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Non-Small Cell Lung Cancer. Vol 2. National Comprehensive Cancer Network; 2023.
- 16.Detterbeck FC, Lewis SZ, Diekemper R, Addrizzo-Harris D, Alberts WM. Executive Summary: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5)(suppl):7S-37S. doi: 10.1378/chest.12-2377 [DOI] [PubMed] [Google Scholar]
- 17.Bonner SN, Wakeam E. The end of race correction in spirometry for pulmonary function testing and surgical implications. Ann Surg. 2022;276(1):e3-e5. doi: 10.1097/SLA.0000000000005431 [DOI] [PubMed] [Google Scholar]
- 18.Wolf A, Alpert N, Tran BV, Liu B, Flores R, Taioli E. Persistence of racial disparities in early-stage lung cancer treatment. J Thorac Cardiovasc Surg. 2019;157(4):1670-1679.e4. doi: 10.1016/j.jtcvs.2018.11.108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Beaverson S, Ngo VM, Pahuja M, Dow A, Nana-Sinkam P, Schefft M. Things We Do for No Reason™: race adjustments in calculating lung function from spirometry measurements. J Hosp Med. Published online October 7, 2022. doi: 10.1002/jhm.12974 [DOI] [PubMed] [Google Scholar]
- 20.Shin S, Kong S, Kang D, et al. Longitudinal changes in pulmonary function and patient-reported outcomes after lung cancer surgery. Respir Res. 2022;23(1):224. doi: 10.1186/s12931-022-02149-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Boulware LE, Mohottige D, Maciejewski ML. Race-free estimation of kidney function: clearing the path toward kidney health equity. JAMA. 2022;327(23):2289-2291. doi: 10.1001/jama.2022.7310 [DOI] [PubMed] [Google Scholar]
- 22.Mohottige D, Purnell TS, Boulware LE. Redressing the harms of race-based kidney function estimation. JAMA. 2023;329(11):881-882. doi: 10.1001/jama.2023.2154 [DOI] [PubMed] [Google Scholar]
- 23.Inker LA, Eneanya ND, Coresh J, et al. ; Chronic Kidney Disease Epidemiology Collaboration . New creatinine- and cystatin C-based equations to estimate GFR without race. N Engl J Med. 2021;385(19):1737-1749. doi: 10.1056/NEJMoa2102953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Delgado C, Baweja M, Crews DC, et al. A unifying approach for gfr estimation: recommendations of the NKF-ASN Task Force on reassessing the inclusion of race in diagnosing kidney disease. J Am Soc Nephrol. 2021;32(12):2994-3015. doi: 10.1681/ASN.2021070988 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gutiérrez OM, Sang Y, Grams ME, et al. ; Chronic Kidney Disease Prognosis Consortium . Association of estimated GFR calculated using race-free equations with kidney failure and mortality by Black vs non-Black race. JAMA. 2022;327(23):2306-2316. doi: 10.1001/jama.2022.8801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bhakta NR, Kaminsky DA, Bime C, et al. Addressing race in pulmonary function testing by aligning intent and evidence with practice and perception. Chest. 2022;161(1):288-297. doi: 10.1016/j.chest.2021.08.053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Blom EF, Ten Haaf K, Arenberg DA, de Koning HJ. Disparities in receiving guideline-concordant treatment for lung cancer in the United States. Ann Am Thorac Soc. 2020;17(2):186-194. doi: 10.1513/AnnalsATS.201901-094OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bhakta NR, Bime C, Kaminsky DA, et al. Race and ethnicity in pulmonary function test interpretation: an official American Thoracic Society Statement. Am J Respir Crit Care Med. 2023;207(8):978-995. doi: 10.1164/rccm.202302-0310ST [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ortmeyer KA, Raman V, Tiko-Okoye C, Espinosa J, Cooke DT, Erkmen CP. Women and minorities underrepresented in academic cardiothoracic surgery: it’s time for next steps. Ann Thorac Surg. 2021;112(4):1349-1355. doi: 10.1016/j.athoracsur.2020.08.057 [DOI] [PubMed] [Google Scholar]
- 30.Olive JK, Mansoor S, Simpson K, et al. Demographic landscape of cardiothoracic surgeons and residents at United States training programs. Ann Thorac Surg. 2022;114(1):108-114. doi: 10.1016/j.athoracsur.2021.07.076 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eAppendix. Vignette Instrument
eFigure 1. Surgeon Estimation of the Probability of Post-Operative Complications for Right Upper Lobectomy Depending on Whether Race-Correction Is Used in Pulmonary Function Tests (PFTs)
eFigure 2. Surgeon Estimation of the Probability of Post-Operative Mortality for Right Upper Lobectomy Depending on Whether Race-Correction Is Used in Pulmonary Function Tests (PFTs)
Data Sharing Statement