Abstract
Genomic medicine can enhance prevention and treatment. First, we propose that advances in genomics have the potential to enhance assessment of disease risk, improve prognostic predictions, and guide treatment development and application. Clinical implementation of polygenic risk scores (PRSs) has emerged as an area of active research. The pathway from genomic discovery to implementation is an iterative process. Second, we provide examples on how genomic medicine has the potential to solve problems in prevention and treatment using two examples: Lung cancer screening and evidence-based tobacco treatment are both under-utilized and great opportunities for genomic interventions. Third, we discuss the translational process for developing genomic interventions from evidence to implementation by presenting a model to evaluate genomic evidence for clinical implementation, mechanisms of genomic interventions, and patient desire for genomic interventions. Fourth, we present potential challenges in genomic interventions including a great need for evidence in all diverse populations, little evidence on treatment algorithms, challenges in accommodating a dynamic evidence base, and implementation challenges in real world clinical settings. Finally, we conclude that research to identify genomic markers that are associated with smoking cessation success and the efficacy of smoking cessation treatments is needed to empower people of all diverse ancestry. Importantly, genomic data can be used to help identify patients with elevated risk for nicotine addiction, difficulty quitting smoking, favorable response to specific pharmacotherapy, and tobacco-related health problems.
Keywords: Genomics, Polygenic risk, Clinical implementation, Genomic medicine, Precision treatment, Precision prevention, Tobacco, Lung cancer
1. Introduction
1.1. Genomic medicine can enhance prevention and treatment
Advances in genomics have the potential to enhance assessment of disease risk, improve prognostic predictions, and guide treatment development and application. Genomic discoveries may also enable better understanding of both disease and treatment mechanisms and thereby enhance scientific understanding. As such, precision medicine has the potential to meaningfully benefit patient health with personalized prevention and treatment interventions (Fig. 1).
Fig. 1.
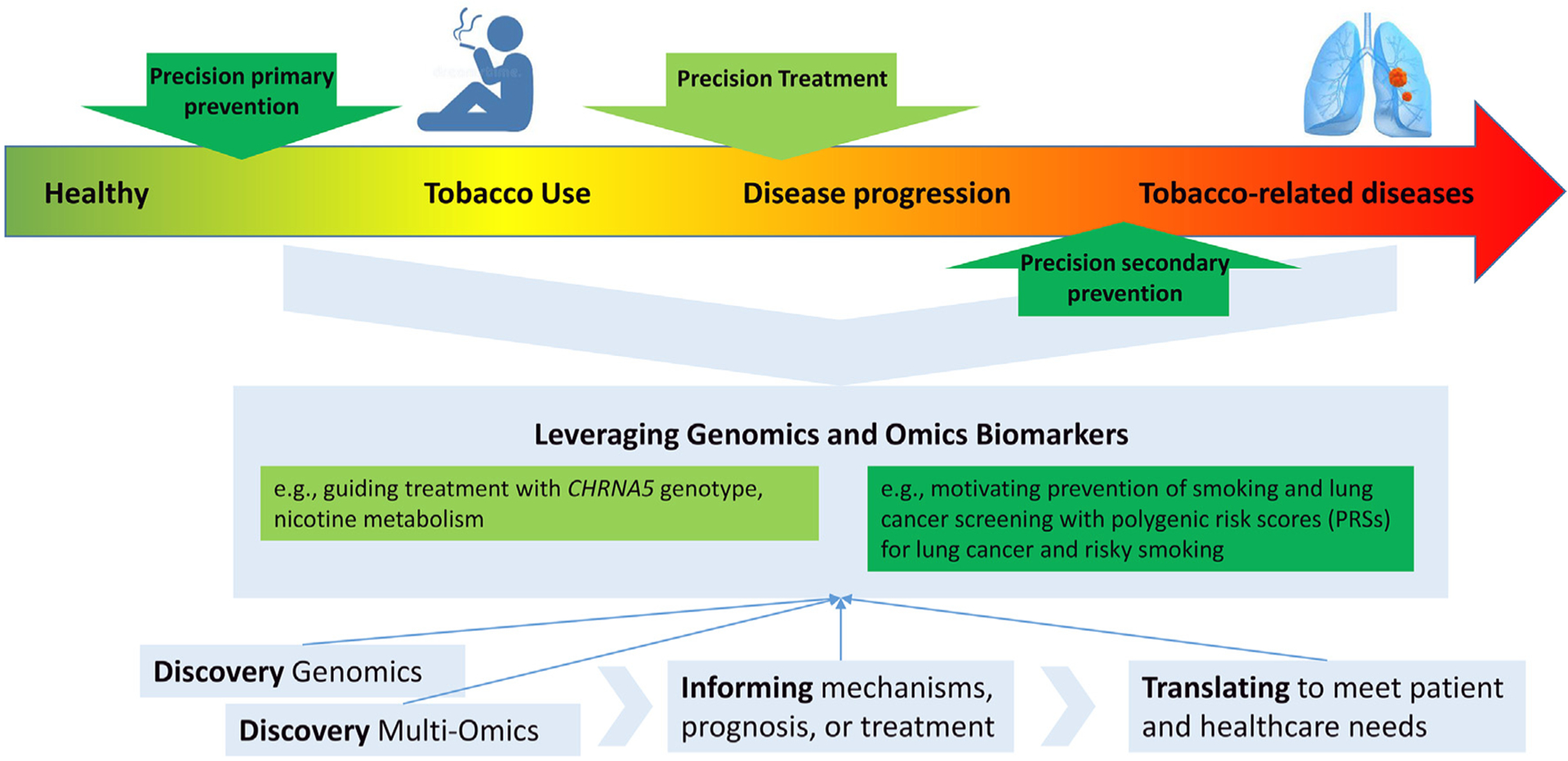
Precision prevention and precision treatment.
Having the potential to capture multiple different biological mechanisms and adapt to a dynamic evidence base, genomics is increasingly recognized as an important source of data that can inform predictions regarding medication efficacy or adverse events [1,2]. Given the growing evidence supporting genetic contributions to drug response, the Clinical Pharmacogenetics Implementation Consortium (CPIC), [3,4] an international consortium, was formed to provide consensus guidelines on interpretation and translation of genotype results into actionable prescribing decisions. Genetic information is now included in US Food and Drug Administration-approved labeling for an increasing number of medications. For example, medication Bupropion (antidepressant and smoking cessation aid) pharmacokinetic properties are affected by CYP2D6 [5]. The likelihood of such benefits is growing with our on-going progress in discovering genetic influences on disease, offering increased insights into disease mechanisms of action and metabolism of medications.
1.2. Clinical implementation of polygenic risk scores (PRSs) is an area of active research
Genome-wide association studies (GWAS) have identified an increasing number of genomic variants significantly associated with common complex human diseases, including nicotine dependence and smoking behaviors [6,7] PRSs provide a single measure of genetic association that aggregates risk alleles across the genome. PRS may confer information on risk magnitude that is equivalent to that conferred by clinical risk factors. Proper clinical implementation of PRS is now an important area of active research across many disease areas [8]. Importantly, we note that PRS could be constructed as a risk or protective factor because it is a weighted sum to capture polygenic effects.
Paving the way for implementing genomic medicine, eMERGE is a national network funded by the National Human Genome Research Institute (NHGRI) that combines DNA biorepositories with electronic medical record (EMR) systems for large scale genetic research [9]. Additionally, the National Institutes of Health (NIH)-funded Implementing GeNomics In praTticE (IGNITE) network was established to support the development and investigation of genomic medicine models to enhance the application and implementation of genomic based practices into routine clinical care [10].
1.3. Advancing science in two key domains — precision prevention and precision treatment
PRS and other biomarkers related to smoking and smoking-related conditions have potential utility for clinical care. Use of PRS has enabled research to evaluate the relative contribution of genetic and non-genetic factors. For example, recent evidence suggest that the adding PRS for smoking cessation significantly improves the prediction of successful smoking cessation beyond use of clinical predictors and that the adding PRS for lung cancer significantly may improve the prediction of lung cancer beyond use of clinical predictors [11,12].
Advances in genomic knowledge is being translated into personalized approaches for both prevention and treatment. One precision prevention opportunity is focused on the primary prevention of smoking among individuals who have not initiated smoking (most notably children, adolescents, and young adults). Another prevention opportunity is the secondary prevention of smoking-related morbidity and mortality among people with a substantial smoking history (most notably those eligible for lung cancer screening). This approach can enhance primary and secondary cancer prevention by providing personalized feedback (e.g., PRS on lung cancer and persistent smoking) to reduce smoking initiation and motivate smoking cessation attempts and cancer screening.
In contrast, precision treatment opportunities leverage the use of pharmacogenomic biomarkers (genetics of smoking, nicotine metabolism) to personalize treatment algorithms, including recommended dosing and selection of medications to maximize effectiveness, minimize side-effects and increase smoking cessation. By working synergistically with common measures, joint efforts to build PRS pipelines and EHR integration, and lessons learned from implementation in real-world settings, the field is positioned to accelerate the advancement of these related yet distinct areas of investigation.
1.4. From genomic discovery to implementation: an iterative process
In this paper, we present a framework for translating genomic discoveries to clinical interventions and provide examples related to prevention of lung cancer and tobacco treatment. Specifically we will discuss both 1) areas that require new research to support personalized tobacco treatment and 2) real world application research that evaluates the clinical needs and readiness for implementing genomic medicine in real world settings. We suggest taking this two-prong parallel approach based on two major reasons: First, there is a great need to reduce the long lag between published science to clinical care [13,14]. Second, a multilevel genomic intervention has the potential as a novel approach that can enhance the implementation and uptake of evidence-based tobacco treatment that are insufficiently used in clinical settings.
2. Precision prevention
2.1. Problem – lung cancer screening and tobacco treatment are much under-utilized
Annual lung cancer screening with low-dose computed tomography can reduce lung cancer mortality and is currently recommended in the U.S. for asymptomatic adults aged 50 to 80 years who have a 20 pack-year smoking history and have smoked in the past 15 years [15]. Unfortunately, as few as 2.0% of 7.6 million eligible smokers receive lung cancer screening [16]. Primary care physicians are the predominant referral source for lung cancer screening; however, only a minority ordered lung cancer screening for any screen-eligible patients in the past 12 months [17]. Further, most (>70%) of these lung cancer screening-eligible patients are current smokers, yet smoking cessation pharmacotherapy is prescribed in only 8% of patients who smoke [18], and efficacious treatments are underused when prescribed [19,20].
2.2. Translational opportunity - Precision prevention strategies may boost uptake of lung cancer screening and tobacco treatment
Little progress has been made in incorporating precision medicine into behavioral interventions for tobacco treatment and lung cancer. Two findings offer opportunities to support precision prevention in lung cancer screening and tobacco treatment: 1) clinical and genetic factors inform personalized risk for lung cancer and personalized benefit of smoking cessation, providing the basis for decision support and communication tools for physicians [11,21–25] and 2) patients express a high desire for genetic risk feedback, suggesting its potential to activate behavior change [26–28]. A precision risk intervention leveraging polygenic risk and patient receptivity could help close gaps in ordering and patient uptake of lung cancer screening and tobacco treatment.
2.3. Current evidence on precision prevention: what is known and what is needed?
Growing evidence shows that risk stratification can increase efficiency of lung cancer screening beyond use of existing criteria based on smoking status, pack years, and age. There is growing evidence that genetic variants predict lung cancer risk and likelihood of smoking cessation success (Fig. 2) [6,7,11,21–25].
Fig. 2.
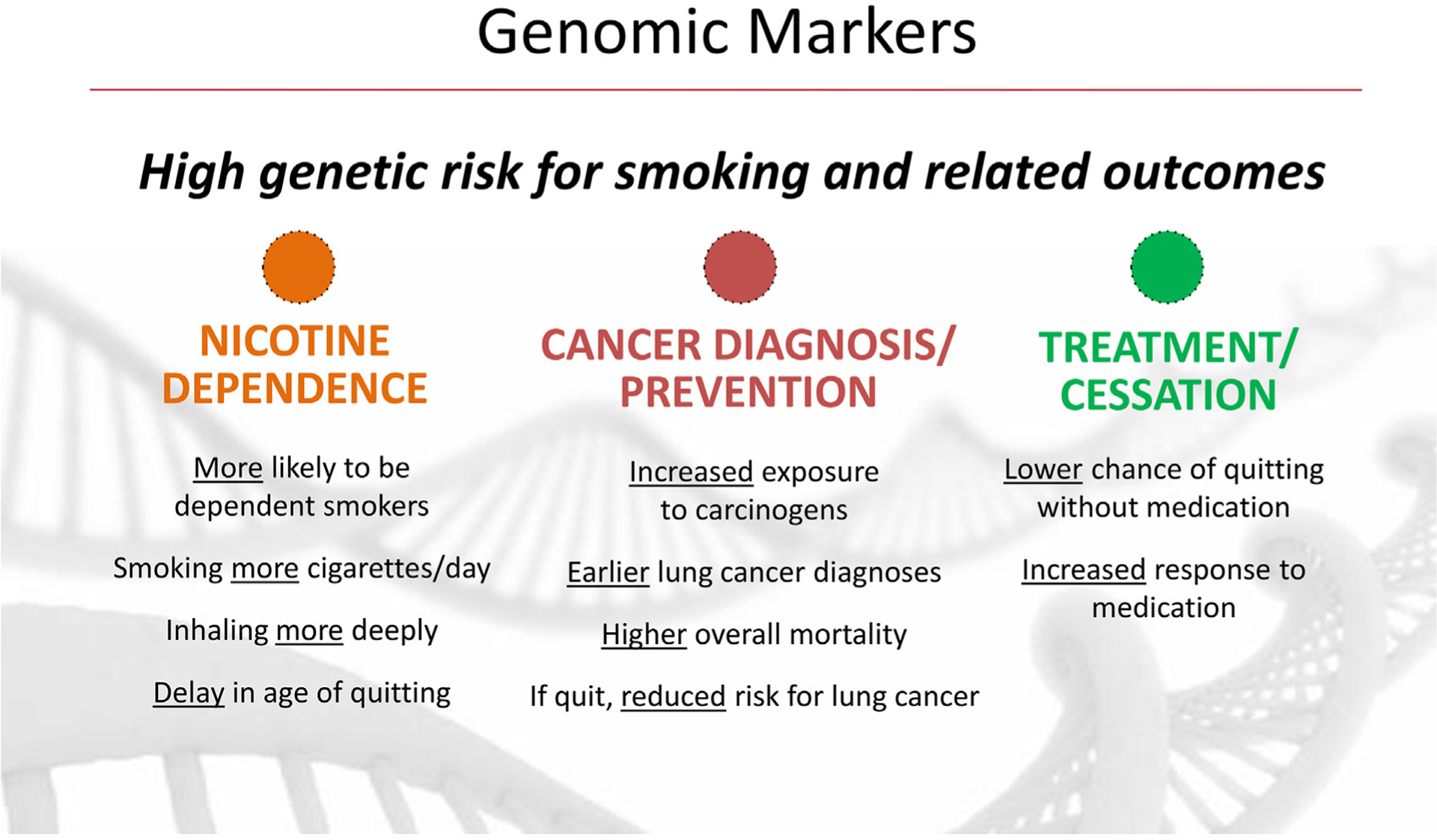
Polygenic score and smoking related outcomes.
2.3.1. What is known about precision risk for lung cancer
There is strong evidence that genetic variants predict lung cancer risk [11,23,24,29–35]. Multiple meta-analyses based on the Integrative Analysis of Lung Cancer Etiology and Risk (INTEGRAL) Consortium, the largest international consortium for lung cancer, show that patients with high genetic risk (CHRNA5 genotypes or polygenic risk) have a 2-fold increase in risk for lung cancer compared to patients with low genetic risk [11,23,24]. Specifically, patients with high genetic risk receive a lung cancer diagnosis about 6 years earlier compared to those at low risk (age 65 vs. 71) [24,36]. These results demonstrate that an individual’s genetic background can inform the precision of lung cancer risk assessment and the urgency for lung cancer screening and smoking cessation.
2.3.2. What is known about precision benefit of smoking cessation in reducing risk for lung cancer
Multiple large genome-wide meta-analyses, by our team with the INTEGRAL Consortium, demonstrate the benefit of smoking cessation based on each patient’s genotypes [23,24,36]. Specifically, patients with high genetic risk (CHRNA5 genotypes or polygenic risk) are less likely to quit successfully when unassisted with medication. Individuals with high genetic risk can benefit from a 50% reduced cancer risk and a 9-year delay in cancer onset (age 71 vs. 62) if they quit. Patients with low genetic risk have a lower risk of cessation failure, but they too can benefit from quitting with a 50% reduction in cancer risk and a 6-year delay in cancer onset (age 74 vs. 68). These results show that an individual’s genetic background not only predicts differential risk of lung cancer and delay in cancer onset but also predicts substantial benefit of cessation even amongst those at low genetic risk.
2.3.3. What is known about precision risk for persistent smoking (or failed smoking cessation)
Risk for nicotine dependence is not only polygenic, but also pleiotropic across multiple related traits such as neuroticism, alcohol use, depression, and schizophrenia. [37] Large genome-wide meta-analyses identified multiple genetic markers for smoking quantity, and smoking cessation, with the most robust signal near the α5 nicotinic cholinergic receptor gene (CHRNA5) [6,7]. Research found that use of this variant and polygenic risk scores has prognostic significance for likelihood of cessation [6,23,24]. Specifically, those with high-risk genetic variants are more likely to 1) smoke heavily [6,7,38], 2) delay age of quitting smoking [24], 3) have lower success with unassisted quit attempts [39], and 4) benefit from cessation medications [39–48], compared to those without the risk variants. Most importantly, use of polygenic risk scores for risk smoking behaviors significantly increases prediction of smoking cessation beyond use of demographic and clinical predictors such as cigarettes per day, carbon monoxide, Fagerström Test for Nicotine Dependence (FTND), age of initiation, anxiety, and depression [12].
2.3.4. What is needed
First, to enhance the evidence base, it is important to note that Genome-wide association studies (GWAS) on lung cancer and smoking behaviors in non-European populations (e.g., African, Hispanic/Latinx, Asian ancestries) are still rare and much smaller in sample size) [6,29,34,49,50]. Larger datasets in African and other underrepresented populations are much needed to identify the ancestry-specific loci and dissect the heterogeneity across different ancestries. Second, there is a missed opportunity to diversity research in countries with varying policy and culture regarding smoking to gain insights in the genomic-environment interactions. Cross-ancestry or international research is still limited and much needed to understand biology [29,38]. Third, to enable precision prevention, we need research to evaluate the clinical utility of a precision intervention and determine if the precision intervention (vs. usual care) generates better clinical outcomes such as increased tobacco treatment and lung cancer screening among eligible patients by effectively motivating health behavior changes.
3. Precision treatment
3.1. Problem - evidence-based tobacco treatment is under-utilized
Innovative interventions are needed to address gaps in tobacco treatment. Smoking remains a leading cause of premature death, causing ~400,000 deaths annually in the U.S. and >50% of all cancer deaths [51–55]. and smoking cessation significantly reduces this risk [56]. However, the public health impact of smoking cessation treatment is limited by a cascade of factors including a low rate of physician prescription of cessation medication (<20%) [55,57–62], a low rate of patient use of cessation medication (~33%) [55,63,64], and limited medication effectiveness (<30%) [65–69]. Thus, the reach and effectiveness of smoking cessation treatment remains limited despite the wide availability of treatment, clear guidance from clinical practice guidelines and considerable efforts to execute health system changes designed to increase smoking treatment implementation and engagement [60,70,71]. The cumulative effects of these multiple constraints yield a very low expected rate of smoking cessation (~2%) among patients in primary care. Existing strategies have had limited success, despite considerable implementation effort with providers and patients. There is a great need for a multilevel intervention that addresses each of these rate limiting factors.
3.2. Translational opportunity - Precision treatment strategies may boost the implementation and effectiveness of tobacco treatment
Precision treatment has the potential to alter clinical practice and patient behavior. First, precision treatment may increase treatment effectiveness by matching patients with the medication that maximizes efficacy and safety. Evidence from meta-analyses, and our work has laid a foundation for precision treatment as described in details below on precision risk, benefit, and treatment choice [36,39,40,43,72–76]. Second, when physicians receive specific recommendations of precision treatment based on biomarkers, they may be more motivated to prescribe medication that is pharmacogenetically matched to their patient [77–80]. Physicians report concerns over medication effectiveness and safety, so a precision medicine approach that addresses these concerns may increase physician outcome expectancies and prescribing [81,82]. Third, when patients receive personalized information on risks and benefits, and personalized treatment recommendations, they may be more motivated to use medication due to increased perception of benefit with reduced side effects [83–85]. Growing evidence suggests that patients who smoke want gene-guided treatment, which may increase their motivation to use it as prescribed [26,86–88].
3.3. Current evidence on precision treatment: what is known and what is needed
In the past decade, evidence has emerged on the role of genetic markers guiding precision treatment based on genetic and metabolic markers for patients who smoke (Fig. 2) [12,23,24,36,39,40,43,73–76].
3.3.1. What is known about precision treatment choice
A recent review by Panagiotou et al.[72] and several landmark trials [36,39,40,43,73–75], demonstrate that patient response to cessation medication is moderated by two major biomarkers: the nicotine metabolite ratio (NMR) and the genotype of rs16969968 in the nicotinic receptor gene CHRNA5.
Metabolic markers
NMR is a biomarker for nicotine metabolism, which can be dichotomized as “normal ” vs. “slow. ” Mounting evidence, including the 2015 landmark Pharmacogenetics of Nicotine Addiction Treatment (PNAT) trial[74] and a recent review [72], showed that NMR can inform choice of cessation medication. These results suggest nicotine replacement therapy (NRT) for slow metabolizers due to similar effectiveness and lower side effect profiles, and varenicline for normal metabolizers due to superior effectiveness. We and others have replicated this finding and meta-analyzed our recent Genetically Informed Smoking Cessation (GISC) trial and the PNAT trial to confirm that NMR moderates cessation medication efficacy [73,89–91]. In addition, our work and existing evidence has shown the value of CYP2A6 genotypes in predicting nicotine metabolism and smoking cessation. [92–95]
Genetic markers
There is mounting evidence that genotypes predict an individual’s response to a specific medication [39,43]., [96–100] The pharmacogenetic evidence is more convergent for African American patients than it is for White patients. The recent review and other evidence show that CHRNA5 may moderate response to NRT in African American patients[72,75]. Patients with rs16969968-GG genotype had higher abstinence at end-of-treatment/12 weeks and 6 months when receiving NRT vs. placebo. However, evidence on treatment for patients with the GA/AA genotypes was lacking.
Our recent GISC trial yielded evidence of differential treatment response for African American patients according to CHRNA5 rs16969968 genotypes: combination NRT [patch and lozenge] was more effective in patients with GG genotypes, whereas varenicline was more effective for patients with GA/AA genotypes [40]. Thus, this finding agrees with evidence and other trials (demonstrating that NRT is more effective in patients with GG genotypes) [72],[75] and suggests that varenicline is more effective for patients with GA/AA genotypes. If these findings are robust, the majority of African American patients will be able to quit smoking more successfully with combination NRT—a less expensive over the counter medication that has fewer side effects than varenicline, but varenicline will be especially effective with the remaining individuals with GA/AA genotypes (~13% of patients).
In addition to CHRNA5 (encoding nicotinic receptor) and CYP2A6 (encoding nicotine metabolism), hundreds of genetic variants have been identified for smoking cessation, in large meta-analyses of genome-wide association studies (GWASs) [6]. We have started to integrate multiple genetic markers as predictors for treatment response [43], and our recent work showed the promise of polygenic risk scores in predicting quit success beyond the prediction using clinical factors alone in European Ancestry patients [12,36].
3.3.2. What is needed
First, to enhance the evidence base, these findings require replication, and research is needed on how the genetic marker can be used with other biomarkers such as NMR to enhance treatment assignment. We need genomic and metabolic data for participants in smoking cessation trials to allow the investigation of multiple markers and treatment response. To optimize a treatment algorithm, a single large trial in which FDA approved medications (combination NRT [patch and lozenge] vs. varenicline) can be evaluated and the contributions of multiple biomarkers can be evaluated regarding their main and interactive effects is needed. In most relevant studies, African American individuals constituted only a small portion of the sample. This means that the size for particular subgroups of participants are small for some key comparisons. Small sample size also occurred in recent reviews [67,72,101]. Indeed, smoking cessation trials focused on pharmacotherapy with African Americans are few in number. We need more and large trials to identify treatment algorithms that optimize multiple biomarkers for African American patients who smoke. Second, to enable precision treatment, we need research to evaluate the clinical utility of a precision intervention and determine if the precision intervention (vs. usual care) generates better clinical outcomes such as increased use of tobacco treatment and overall quit success by effectively motivating health behavior changes and matching treatment.
4. Genomic interventions: from evidence to implementation
4.1. A model to evaluate genomic evidence for clinical implementation
The ACCE framework holds that genetic tests be evaluated based on the four main criteria: Analytic validity, Clinical validity, Clinical utility (ACCE), and associated Ethical Legal and Social Implications (ELSI) and other similar criteria for clinical actionability [102–104]. We propose an evaluation model to guide the clinical actions that can be taken in response to a genomic marker (see Fig. 3). Actionability depends on three factors: (I) discriminative power (e.g., analytic validity, accuracy); (II) performance in comparison to other known risk factors (e.g., clinical validity, impact); and (III) improve patient health outcomes (e.g., clinical utility).
Fig. 3.
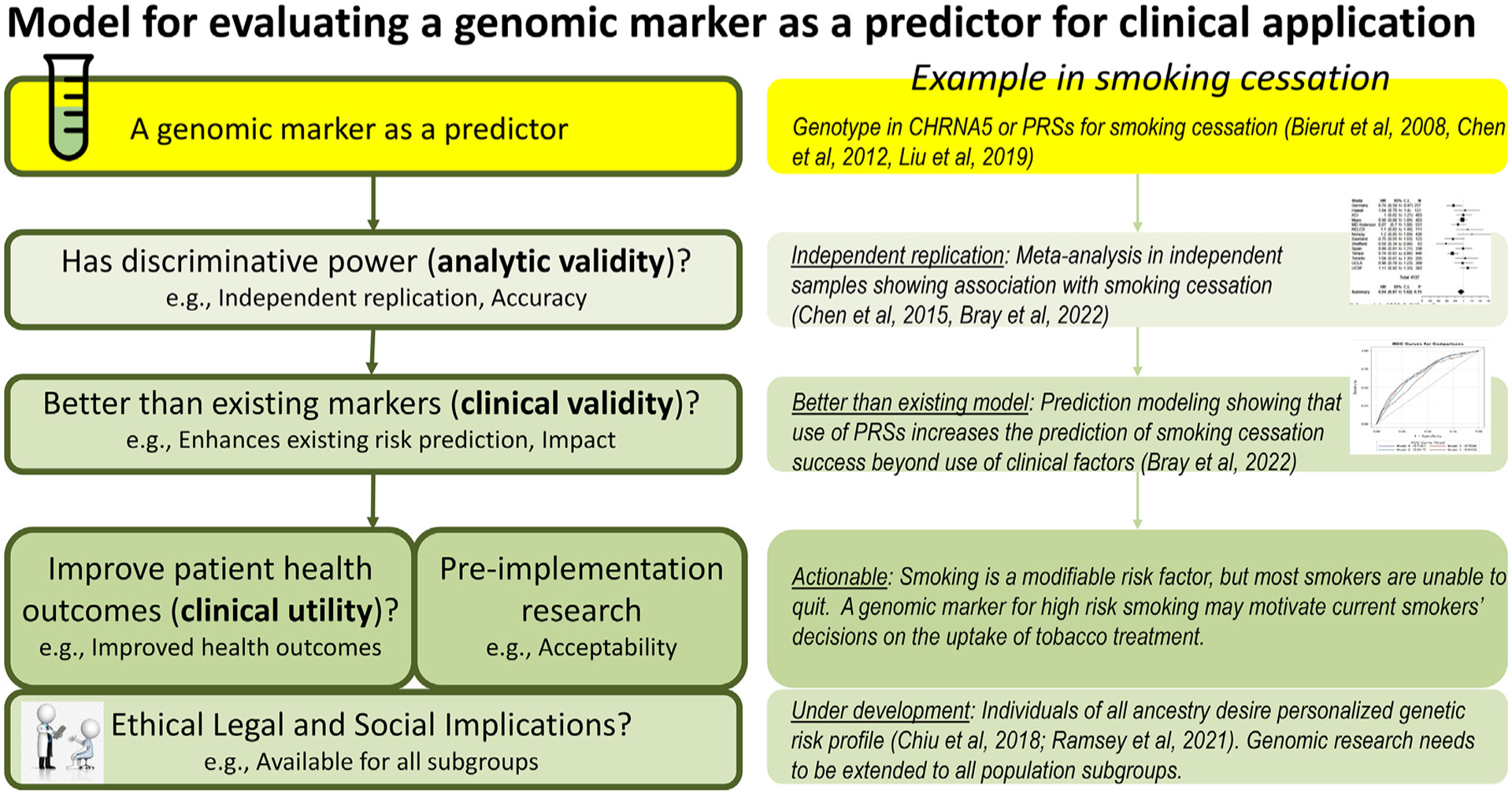
Model for evaluating a genomic marker as a predictor for clinical application.
Model adapted from ACCE framework by Teutsch SM et al. 2009, and other similar criteria proposed by Klein RJ et al. 2022, Wand H, 2021.
For the use of genomic markers (CHRNA5 genotypes or PRSs for high-risk smoking) for predicting risk of lung cancer occurrence and smoking cessation, current research includes evidentiary support from meta-analyses for analytic validity[6,39,105] and meta-analytic evidence of clinical validity (e.g., use of PRS for high-risk smoking significantly increases the predictive accuracy of smoking cessation success beyond use of demographic and clinical factors) [12,23,24]. However, we need more evidence with regard to clinical utility and we need further investigation of issues around ELSI.
Based on our evaluative model (Fig. 3) for assessing the evidentiary basis for the clinical utility of a genomic marker in predicting treatment choices, we note that there is strong evidence of analytic validity, moderate evidence from meta-analyses and a recent review for clinical validity, but that there is little research regarding the criteria, clinical utility, and ELSI.
4.2. Mechanisms of genomic interventions
Knowledge of personalized genomic risk may motivate behavior change. Studies have examined behavior change following the return of genetic information. Among underserved populations [30], consumers of genomic testing [31], and college students[102] who smoke, returning genetic susceptibility results, including results on nicotine dependence [102], has not been shown to effect psychological or behavioral harms, even among populations at high-risk for adverse reactions. A lack of iatrogenic effects has been supported in multiple meta-analyses [103,104], alleviating concerns about negative behavioral or psychological effects (e.g., sense of fatalism, risk compensation, depression or anxiety) of feedback of genetic risk. Although these meta-analyses confirm a low risk of adverse effects, they report mixed findings on the potential of genetic susceptibility results to motivate positive behavior change. One meta-analysis revealed little to no behavioral change [103], whereas a newer meta-analysis found moderate increases in healthy behaviors [104].
A novel care paradigm may transform precision tobacco treatment. Genetic information may boost physician prescribing, motivate patients’ use of medication, and enhance medication effectiveness. However, additional research is needed to inform the implementation of a new precision care paradigm that is theory-driven, addresses clinician and patient barriers, and integrated in healthcare settings and practices, factors that are critical to behavior change efforts [106,107].
Drawing from the teachable moment hypothesis in the healthcare context, precision prevention for smoking enables multiple pathways to increase engagement and motivation for behavior change, including 1) communicating personalized risk of lung cancer and the potential benefits of lung cancer screening and tobacco treatment, 2) increasing urgency for action alongside positively-framed, action-oriented messages, and 3) reducing self-stigmatizing views. Translating these genomic findings into effective motivational or behavior change strategies is a promising frontier for engaging both patients and physicians in lung cancer screening and tobacco treatment actions [47,93–96].
While there is emerging evidence to support translation of precision prevention into lung cancer screening and tobacco treatment clinical practice, extant evidence now permits rigorous tests of the utility of incorporating genetic information into motivational interventions for screening and treatment in real-world settings. Such efforts could address the need to drive the uptake of lung cancer screening and tobacco treatment at point-of-care [97,98]. Enriching behavioral interventions with genetic prognostic information may prompt and guide physician ordering and motivate and facilitate patient uptake of annual lung cancer screening and tobacco treatment [93,94].
4.3. Patient desire and the promise of genomic intervention
Research suggest that patients have high demand for personalized genetics [29–32]. We found that patients who smoke strongly endorsed the importance of receiving genetic risk results, in particular to learn the following: risk for developing lung cancer (96%) and guidance regarding smoking cessation (95%) [29].
Patients report increased motivation to use personalized tobacco treatment [28]: In our recent work, 85% of current smokers reported high interest in receiving genetic testing results, with a significantly higher proportion desiring to take medication when they were informed that the likelihood of medication benefit was above average, compared to below average [28]. These data suggest that personalized genetic information on risk and risk modification might motivate smokers for positive behavior change such as use of tobacco treatment [28].
Existing research including ours suggests that smokers not only desire knowledge of personalized risk, but also report that such knowledge will motivate cancer prevention behaviors such as seeking tobacco treatment [28–32]. The robust desire and perceived importance of personalized genetic data suggested strong demand among smokers, motivating the development and evaluation of returning personalized genetic risk as an intervention to promote health behaviors such as smoking cessation. In our recent work to motivate patients who smoke for positive behavior change, we designed RiskProfile as an intervention to communicate personalized genetic risk for developing lung cancer and difficulty in quitting smoking [30–32]. RiskProfile demonstrates high acceptability and utility as a behavior change tool to reduce smoking among individuals who smoke [27]. In a proof-of-concept trial, we found that 69% of participants reported increased readiness to quit after receiving RiskProfile, and 65% reported reducing their cigarette smoking. Overall, 77% reported making at least one smoking-related behavior change (quit attempt, fewer cigarettes, tried cessation medication) [86]. Importantly, we found that patients of high genetic risk substantially reduced their smoking (defeating the bias of “genetic determinism”); likewise, patients of low genetic risk also reduced their smoking (defeating the bias of a “health certificate effect”) [86]. These results showed the potential of a genetically informed intervention for behavior change.
5. Potential challenges in genomic interventions
Genomic medicine is encouraged by advances in genomic discoveries, but hampered by multiple problems: 1) It is critical to enhance evidence in all diverse populations with studies with larger sample sizes, linkage between genomic and phenotypic data, and robust measures. 2) We need to develop and evaluate treatment algorithms that include both genomic and non-genomic factors. 3) Genomic interventions need to be based on most current evidence with a mechanism to incorporate ongoing advances in the knowledge base. 4) Implementation challenges of genomic interventions depends on cost-effectiveness, level of integration with health informatics, and co-design with stakeholders in the healthcare systems.
5.1. Need more evidence in all diverse populations
We need research with large sample sizes, genomic data linked with highly granular phenotypic data, and robust measures such as specific pharmacotherapy, adherence, and bioverified abstinence outcomes. For example, current evidence in African American patients is based on only a few small trials, and research has not explored how biomarkers can be used together and with non-biologic variables to enhance cessation outcomes. Unfortunately, little research has focused on African American patients even though they have disproportionally high rates of tobacco-related cancer and cancer mortality [108], and despite evidence that smoking cessation treatments are often less effective in African American patients than in other groups [109–112]. A well-powered trial with African American patients who smoke is needed to develop biomarker-informed treatment, so that it can then be experimentally evaluated as a precision treatment algorithm in this under-studied population to reduce smoking-related disparities.
5.2. Little evidence on treatment algorithms
Precision treatment algorithms will likely include both metabolic and genetic markers. Work has started to explore the use of NMR in guiding treatment. However, rapidly emerging genomic evidence suggests the potential value of including genetic markers in precision treatment. First, genetic data are increasingly available via EHR with multiple national initiatives on genomic medicine and consumer genomics; thus, gene-based interventions are likely to be highly scalable [1,2]. Second, genetic data have the potential to capture multiple distinct biological pathways (both nicotinic receptor gene function and nicotine metabolism) and adapt to a dynamic evidence base. Third, genetic data may be uniquely informative in personalizing an individual’s risk, benefit, and recommended treatment, capturing multiple mechanisms to optimize treatment success. Given the evidence, evaluating the use of both metabolic and genetic markers in a multilevel intervention using behavior change theories may advance the field of tobacco treatment.
Research is needed to develop and evaluate different treatment algorithms to optimize smoking cessation success: 1) a non-biologic algorithm (using non-biologic predictors such as demographics, smoking quantity, and past treatment history), 2) a biologic algorithm (using either or both NMR and genetic markers), and 3) a hybrid algorithm comprising both non-biologic predictors and biomarkers. For example, our recent work showed the promise of polygenic risk scores in predicting quit success beyond the prediction using clinical factors alone in European Ancestry patients [12],[36] Statistical approaches should be used to identify treatment algorithms and evaluate the utility of these algorithms with regard to optimal prediction of abstinence. The outcomes of algorithms will be appraised with regard to prediction of successful smoking cessation and associated costs (e.g., biomarker assays, medication, time). Development and validation of treatment algorithms to incorporate new evidence will lay the foundation for up to date genomic medicine [113].
5.3. Challenge in accommodating a dynamic evidence base
A mechanism to incorporate a rapidly evolving evidence base is needed. One major challenge of precision treatment is the rapidly evolving evidence that identifies novel biomarkers in treatment. For example, our recent publication suggests the potential of polygenic risk scores in guiding treatment in the future [12]. Despite the presence of actionable precision treatment findings, the dynamic evidence base and perception that superior data are on the horizon has had a stifling effect on implementation [114,115]. We propose research to implement genomic medicine using state-of-the-art, biology-based metabolic and genetic markers that provide robust evidence that will be updated over time to inform both prevention and treatment. This effort will reduce the time lag from evidence to implementation, especially when precision medicine applies an active dynamic evidence base.
5.4. Implementation challenges/clinical utility
Precision tobacco treatment using genomic or biomarkers in healthcare settings may face multiple barriers. First, patients may not be willing to delay treatment due to genomic or biomarker testing. Second, current primary care clinic settings may not permit easy access to genomic or biomarker testing without significant changes in workflows. Some early work has started to explore assessment of biomarkers as part of smoking cessation treatments in research and clinical settings [39,40,74,116]. Early data on metabolism-informed treatment also suggests high feasibility and acceptability of biomarker testing in clinical settings [116]. In our work of returning smoking-related genetic results, conducted in both in-person and virtual visits, we found a high patient completion rate of biomarker assays (>98%) that was robust across categories of age and race [86,117]. Yet, turnaround time for genomic and biomarker assays remain a major challenge. To overcome the multilevel implementation challenges for genomic medicine and empower patients and physicians in making precision decisions, implementation strategies leveraging health informatics and learning health systems may be helpful [118–121].
6. Conclusion and recommendation
The use of genomic markers in medical treatment is increasing for conditions such as cancer care and nicotine dependence. Research is identifying increasing numbers of genomic markers that are associated with smoking cessation success and the efficacy of smoking cessation treatments. Further, adding genomic data to typical clinical and epidemiological study designs has proven to be informative and important. The potential of genetically informed treatment makes it increasingly important that investigators collect biological samples within clinical trials and integrate their analysis and interpretation with the goals of the trial. For example, genomic data can be used to understand smoking cessation, response to tobacco treatment, tobacco-related health problems, and personalized benefit of annual lung cancer screening. In addition, genomic data can be used to help identify patients with elevated risk for nicotine addiction, difficulty quitting smoking, favorable response to specific pharmacotherapy, and tobacco-related health problems.
These results encourage smoking cessation researchers to engage in research that evaluates such topics as: 1) Enhancing the evidence on the extent to which the benefits of genomic interventions generalize across healthcare sites and patient populations. 2) Building the treatment algorithm that incorporates both biological and non-biological predictors. 3) Starting an iterative process to translate science to interventions. 4) Evaluating clinical utility of genomic interventions and the extent to which such interventions can be implemented consistently and effectively over time in real world healthcare outside of formal research studies.
A major challenge in identifying genomic biomarkers will be to obtain adequate sample sizes that cover diverse ancestry groups. Consortium-based approaches will likely be necessary to yield real successes. For pharmacogenomic studies, meta-analysis of data from individual smoking cessation trials will be crucial and will require comparable trial designs and outcomes [122–124]. Importantly, identifying an optimal pharmacogenomic strategy is highly complex, as treatment trials vary in study designs, the type and intensity of the counseling treatment provided to all groups including the placebo arm, subject inclusion/exclusion criteria, and other experimental methods.
To foster collection of high-quality genomic data in clinical studies, we recommend: (1) genetically informed study designs, (2) biological samples (collection requirements, storage, and analysis with a focus on genomic data) collected as a routine part of healthcare with patients who smoke, (3) participant consent and genomic data sharing requirements for Institutional Review Board (IRB) approvals, and (4) collaborative consortia with harmonization on phenotype characterization for meta-analyses with details described in recent publications by the SRNT Genomics and Omics workgroup [123,124].
We recommend to start an iterative process for translating genomic discovery to implementation. A mechanism to incorporate a rapidly evolving evidence base is needed. One major challenge of precision treatment is the evolving evidence that identifies novel biomarkers in treatment. The dynamic evidence base and perception that superior data are on the horizon has had a stifling effect on implementation [114,115]. This effort will reduce the time lag from evidence to implementation, especially when precision medicine applies an active dynamic evidence base.
To facilitate implementation of genomic interventions, we need research to evaluate clinical utility. Most importantly, we need research to 1) evaluate the needs of patients and physicians, 2) develop/test interventions based on genomic knowledge and user input, and 3) evaluate physician and patient-level effects of genomic interventions in healthcare settings.
In summary, we recommend research to enable the translational roadmap where people of diverse ancestry can make well-informed decisions about their genomic data and health decisions including prevention and treatment [125]. Our goal is to develop not only knowledge bases for genomic medicine, but also strategies for implementing genomic medicine in clinical care in a learning health system to realize precision medicine in reducing tobacco and related disorders.
Source of funding
This work was supported by the National Institutes of Health (P30 CA091842-19S5 (LSC), P50 CA244431 (LSC, AR), R01DA038076 (LSC), R01CA268030 (LSC, AR), R01DA056050 (LSC,AR), R34 DA052928 (AR), U19 CA203654 (LSC, CIA, LJB), P01 CA180945 (TBB); and Alvin J. Siteman Cancer Center Investment Program 5129 - Barnard Trust and The Foundation of Barnes Jewish Hospital Cancer Frontier Fund.
Footnotes
Declaration of Competing Interest
Dr. Bierut is listed as inventor on issued U.S. patent 8080,371, “Markers for Addiction” covering the use of certain SNPs in determining the diagnosis, prognosis, and treatment of addiction. All other authors declare no potential conflict of interest. Dr. Baker has a Glaxo-Wellcome Chair in the Department of Medicine.
Submission to: NIDA special issue on Biomarkers for Smoking Cessation and Treatment to Addiction Neuroscience.
Data availability
No data was used for the research described in the article.
References
- [1].Ayatollahi H, Hosseini SF, Hemmat M, Integrating genetic data into electronic health records: medical geneticists’ perspectives, Healthc Inform Res 25 (4) (Oct 2019) 289–296, doi: 10.4258/hir.2019.25.4.289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Gottesman O, Kuivaniemi H, Tromp G, et al. , The electronic medical records and genomics (eMERGE) network: past, present, and future, Genet. Med 15 (10) (Oct 2013) 761–771, doi: 10.1038/gim.2013.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Pritchard D, Patel JN, Stephens LE, McLeod HL, Comparison of FDA table of pharmacogenetic associations and clinical pharmacogenetics implementation consortium guidelines, Am. J. Health Syst. Pharm 79 (12) (Jun 7 2022) 993–1005, doi: 10.1093/ajhp/zxac064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Caudle KE, Klein TE, Hoffman JM, et al. , Incorporation of pharmacogenomics into routine clinical practice: the Clinical Pharmacogenetics Implementation Consortium (CPIC) guideline development process, Curr. Drug Metab 15 (2) (Feb 2014) 209–217, doi: 10.2174/1389200215666140130124910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].FDA. Food and Drug Administration. Table of Pharmacogenomic Biomarkers in Drug Labeling.
- [6].Liu M, Jiang Y, Wedow R, et al. , Association studies of up to 1.2 million individuals yield new insights into the genetic etiology of tobacco and alcohol use, Nat. Genet 51 (2) (Feb 2019) 237–244, doi: 10.1038/s41588-018-0307-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Xu K, Li B, McGinnis KA, et al. , Genome-wide association study of smoking trajectory and meta-analysis of smoking status in 842,000 individuals, Nat. Commun 11 (1) (Oct 20 2020) 5302, doi: 10.1038/s41467-020-18489-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Hao L, Kraft P, Berriz GF, et al. , Development of a clinical polygenic risk score assay and reporting workflow, Nat. Med 28 (5) (May 2022) 1006–1013, doi: 10.1038/s41591-022-01767-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].eMerge Consortium, Harmonizing Clinical Sequencing and Interpretation for the eMERGE III Network, Am. J. Hum. Genet 105 (3) (Sep 5 2019) 588–605, doi: 10.1016/j.ajhg.2019.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Ginsburg GS, Cavallari LH, Chakraborty H, et al. , Establishing the value of genomics in medicine: the IGNITE Pragmatic Trials Network, Genet. Med 23 (7) (Jul 2021) 1185–1191, doi: 10.1038/s41436-021-01118-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Hung RJ, Warkentin MT, Brhane Y, et al. , Assessing Lung Cancer Absolute Risk Trajectory Based on a Polygenic Risk Model, Cancer Res. 81 (6) (Mar 15 2021) 1607–1615, doi: 10.1158/0008-5472.CAN-20-1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Bray M, Chang Y, Baker TB, et al. , The promise of polygenic risk prediction in smoking cessation: evidence from two treatment trials, Nicotine Tob. Res (Feb 16 2022), doi: 10.1093/ntr/ntac043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Morris ZS, Wooding S, Grant J, The answer is 17 years, what is the question: understanding time lags in translational research, J. R. Soc. Med 104 (12) (Dec 2011) 510–520, doi: 10.1258/jrsm.2011.110180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Munro CL, Savel RH, Narrowing the 17-Year Research to Practice Gap, Am. J. Crit. Care 25 (3) (May 2016) 194–196, doi: 10.4037/ajcc2016449. [DOI] [PubMed] [Google Scholar]
- [15].USPSTF. Lung Cancer: Screening. https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/lung-cancer-screening
- [16].Pham D, Bhandari S, Pinkston C, Oechsli M, Kloecker G, Lung Cancer Screening Registry Reveals Low-dose CT Screening Remains Heavily Underutilized, Clin. Lung Cancer 21 (3) (May 2020) e206–e211, doi: 10.1016/j.cllc.2019.09.002. [DOI] [PubMed] [Google Scholar]
- [17].Lewis JA, Petty WJ, Tooze JA, et al. , Low-Dose CT Lung Cancer Screening Practices and Attitudes among Primary Care Providers at an Academic Medical Center, Cancer Epidemiol. Biomarkers Prev 24 (4) (Apr 2015) 664–670, doi: 10.1158/1055-9965.EPI-14-1241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Jamal A, Dube SR, Malarcher AM, et al. , Tobacco use screening and counseling during physician office visits among adults–National Ambulatory Medical Care Survey and National Health Interview Survey, United States, 2005–2009, MMWR Morb. Mortal. Wkly. Rep 61 (Suppl) (Jun 15 2012) 38–45. [PubMed] [Google Scholar]
- [19].Cadham CJ, Jayasekera JC, Advani SM, et al. , Smoking cessation interventions for potential use in the lung cancer screening setting: a systematic review and meta-analysis, Lung Cancer 135 (Sep 2019) 205–216, doi: 10.1016/j.lungcan.2019.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Kumar P, Gareen IF, Lathan C, et al. , Racial Differences in Tobacco Cessation and Treatment Usage After Lung Screening: an Examination of the National Lung Screening Trial, Oncologist 21 (1) (Jan 2016) 40–49, doi: 10.1634/theoncologist.2015-0325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Tammemagi MC, Berg CD, Riley TL, Cunningham CR, Taylor KL, Impact of lung cancer screening results on smoking cessation, J. Natl. Cancer Inst 106 (6) (Jun 2014) dju084 doi:dju084 [pii], doi: 10.1093/jnci/dju084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Tammemagi MC, Katki HA, Hocking WG, et al. , Selection criteria for lungcancer screening, N. Engl. J. Med 368 (8) (Feb 21 2013) 728–736, doi: 10.1056/NEJMoa1211776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Chen LS, Baker T, Hung RJ, et al. , Genetic Risk Can Be Decreased: quitting Smoking Decreases and Delays Lung Cancer for Smokers With High and Low CHRNA5 Risk Genotypes - A Meta-analysis, EBioMedicine (Aug 10 2016), doi: 10.1016/j.ebiom.2016.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Chen LS, Hung RJ, Baker T, et al. , CHRNA5 risk variant predicts delayed smoking cessation and earlier lung cancer diagnosis–a meta-analysis, J. Natl. Cancer Inst 107 (5) (May 2015), doi: 10.1093/jnci/djv100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Ten Haaf K, Bastani M, Cao P, et al. , A Comparative Modeling Analysis of Risk-Based Lung Cancer Screening Strategies, J. Natl. Cancer Inst 112 (5) (May 1 2020,) 466–479, doi: 10.1093/jnci/djz164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Chiu A, Hartz S, Smock N, et al. , Most Current Smokers Desire Genetic Susceptibility Testing and Genetically-Efficacious Medication, J Neuroimmune Pharmacol 13 (4) (Dec 2018) 430–437, doi: 10.1007/s11481-018-9818-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Ramsey AT, Bray M, Acayo Laker P, et al. , Participatory Design of a Personalized Genetic Risk Tool to Promote Behavioral Health, Cancer Prev. Res. (Phila.) 13 (7) (Jul 2020) 583–592, doi: 10.1158/1940-6207.CAPR-20-0029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Hartz SM, Olfson E, Culverhouse R, et al. , Return of individual genetic results in a high-risk sample: enthusiasm and positive behavioral change, Genet. Med 17 (5) (May 2015) 374–379 doi:gim2014110 [pii], doi: 10.1038/gim.2014.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Dai J, Lv J, Zhu M, et al. , Identification of risk loci and a polygenic risk score for lung cancer: a large-scale prospective cohort study in Chinese populations, Lancet Respir Med 7 (10) (Oct 2019) 881–891, doi: 10.1016/S2213-2600(19)30144-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Zhang YD, Hurson AN, Zhang H, et al. , Assessment of polygenic architecture and risk prediction based on common variants across fourteen cancers, Nat. Commun 11 (1) (Jul 3 2020) 3353, doi: 10.1038/s41467-020-16483-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Amos CI, Wu X, Broderick P, et al. , Genome-wide association scan of tag SNPs identifies a susceptibility locus for lung cancer at 15q25.1, Nat. Genet 40 (5) (2008) 616–622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Saccone NL, Culverhouse RC, Schwantes-An TH, et al. , Multiple independent loci at chromosome 15q25.1 affect smoking quantity: a meta-analysis and comparison with lung cancer and COPD, PLos Genet. 6 (8) (Aug 2010) 1–16, doi: 10.1371/journal.pgen.1001053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Thorgeirsson TE, Geller F, Sulem P, et al. , A variant associated with nicotine dependence, lung cancer and peripheral arterial disease, Nature 452 (3) (2008) 638–642 April 3, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].McKay JD, Hung RJ, Han Y, et al. , Large-scale association analysis identifies new lung cancer susceptibility loci and heterogeneity in genetic susceptibility across histological subtypes, Nat. Genet 49 (7) (Jul 2017) 1126–1132, doi: 10.1038/ng.3892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Hung RJ, McKay JD, Gaborieau V, et al. , A susceptibility locus for lung cancer maps to nicotinic acetylcholine receptor subunit genes on 15q25, Nature 452 (7187) (2008) 633–637. [DOI] [PubMed] [Google Scholar]
- [36].Chen LS, Baker TB, Bray M, Pham G, Bierut LJ Predicting lung cancer risk and smoking cessation treatment outcomes with genetic risk scores presented at: world Congress of Psychiatric Genetics; 2021; Virtual due to COVID. [Google Scholar]
- [37].Risner VA, Benca-Bachman CE, Bertin L, et al. , Multi-Polygenic Analysis of Nicotine Dependence in Individuals of European Ancestry, Nicotine Tob. Res 23 (12) (Nov 5 2021) 2102–2109, doi: 10.1093/ntr/ntab105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Chen LS, Saccone NL, Culverhouse RC, et al. , Smoking and genetic risk variation across populations of European, Asian, and African American ancestry–a metaanalysis of chromosome 15q25, Genet. Epidemiol 36 (4) (May 2012) 340–351, doi: 10.1002/gepi.21627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Chen LS, Baker TB, Piper ME, et al. , Interplay of genetic risk factors (CHRNA5-CHRNA3-CHRNB4) and cessation treatments in smoking cessation success, Am. J. Psychiatry 169 (7) (Jul 1 2012) 735–742 doi:1169679 [pii], doi: 10.1176/appi.ajp.2012.11101545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Chen LS, Baker TB, Miller JP, et al. , Genetic Variant in CHRNA5 and Response to Varenicline and Combination Nicotine Replacement in a Randomized PlaceboControlled Trial, Clin. Pharmacol. Ther (Jun 29 2020), doi: 10.1002/cpt.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Chen LS, Baker TB, Piper ME, et al. , Interplay of genetic risk (CHRNA5) and environmental risk (partner smoking) on cigarette smoking reduction, Drug Alcohol Depend. 143 (Oct 1 2014) 36–43 doi:S0376–8716(14)00953–3 [pii], doi: 10.1016/j.drugalcdep.2014.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Chen LS, Bierut LJ, Genomics and personalized medicine: and smoking cessation treatment, J Food Drug Anal 21 (4) (Dec 2013) S87–S90, doi: 10.1016/j.jfda.2013.09.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Chen LS, Bloom AJ, Baker TB, et al. , Pharmacotherapy effects on smoking cessation vary with nicotine metabolism gene (CYP2A6), Addiction 109 (1) (Jan 2014) 128–137, doi: 10.1111/add.12353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Chen LS, Horton A, Bierut L, Pathways to precision medicine in smoking cessation treatments, Neurosci. Lett 669 (Mar 16 2018) 83–92, doi: 10.1016/j.neulet.2016.05.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Bergen AW, Javitz HS, Krasnow R, et al. , Nicotinic acetylcholine receptor variation and response to smoking cessation therapies, Pharmacogenet Genomics 23 (2) (Feb 2013) 94–103, doi: 10.1097/FPC.0b013e32835cdabd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Munafo MR, Johnstone EC, Walther D, Uhl GR, Murphy MF, Aveyard P, CHRNA3 rs1051730 Genotype and Short-Term Smoking Cessation, Nicotine Tob. Res 13 (10) (Oct 2011) 982–988 doi:ntr106 [pii], doi: 10.1093/ntr/ntr106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Sarginson JE, Killen JD, Lazzeroni LC, et al. , Response to Transdermal Selegiline Smoking Cessation Therapy and Markers in the 15q24 Chromosomal Region, Nicotine Tob. Res (Jan 8 2015) doi:ntu273 [pii], doi: 10.1093/ntr/ntu273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Sarginson JE, Killen JD, Lazzeroni LC, et al. , Markers in the 15q24 nicotinic receptor subunit gene cluster (CHRNA5-A3-B4) predict severity of nicotine addiction and response to smoking cessation therapy, American Journal of Medical Genetics Part B: Neuropsychiatric Genetics 156 (3) (2011) 275–284. [DOI] [PubMed] [Google Scholar]
- [49].Zanetti KA, Wang Z, Aldrich M, et al. , Genome-wide association study confirms lung cancer susceptibility loci on chromosomes 5p15 and 15q25 in an African-American population, Lung Cancer 98 (Aug 2016) 33–42, doi: 10.1016/j.lungcan.2016.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].David SP, Hamidovic A, Chen GK, et al. , Genome-wide metaanalyses of smoking behaviors in African Americans. Original Article, Transl Psychiatry 2 (2012) e119 05/22/online https://www.nature.com/articles/tp201241#supplementary-information, doi: 10.1038/tp.2012.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Schroeder SA, New evidence that cigarette smoking remains the most important health hazard, N. Engl. J. Med 368 (4) (Jan 24 2013) 389–390, doi: 10.1056/NEJMe1213751. [DOI] [PubMed] [Google Scholar]
- [52].Thun MJ, Carter BD, Feskanich D, et al. , 50-year trends in smoking-related mortality in the United States, N. Engl. J. Med 368 (4) (Jan 24 2013) 351–364, doi: 10.1056/NEJMsa1211127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Whiteford HA, Baxter AJ, The Global Burden of Disease 2010 Study: what does it tell us about mental disorders in Latin America? Rev Bras Psiquiatr 35 (2) (Apr 2013) 111–112 doi:S1516–44462013000200111 [pii]. [DOI] [PubMed] [Google Scholar]
- [54].Danaei G, Ding EL, Mozaffarian D, et al. , The preventable causes of death in the United States: comparative risk assessment of dietary, lifestyle, and metabolic risk factors, PLoS Med. 6 (4) (Apr 28 2009) e1000058, doi: 10.1371/journal.pmed.1000058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Babb S, Malarcher A, Schauer G, Asman K, Jamal A, Quitting Smoking Among Adults - United States, 2000–2015, MMWR Morb. Mortal. Wkly. Rep 65 (52) (Jan 6 2017) 1457–1464, doi: 10.15585/mmwr.mm6552a1. [DOI] [PubMed] [Google Scholar]
- [56].Jha P, The 21st century benefits of smoking cessation in Europe, Eur. J. Epidemiol 28 (8) (Aug 2013) 617–619, doi: 10.1007/s10654-013-9835-6. [DOI] [PubMed] [Google Scholar]
- [57].Kalkhoran S, Inman EM, Kelley JHK, Ashburner JM, Rigotti NA, Proactive Population Health Strategy to Offer Tobacco Dependence Treatment to Smokers in a Primary Care Practice Network, J. Gen. Intern. Med 34 (8) (Aug 2019) 1571–1577, doi: 10.1007/s11606-019-05079-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [58].Kruger J, O’Halloran A, Rosenthal AC, Babb SD, Fiore MC, Receipt of evidence-based brief cessation interventions by health professionals and use of cessation assisted treatments among current adult cigarette-only smokers: national Adult Tobacco Survey, 2009–2010, BMC Public Health 16 (Feb 11 2016) 141, doi: 10.1186/s12889-016-2798-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Ramsey AT, Baker TB, Pham G, et al. , Low Burden Strategies Are Needed to Reduce Smoking in Rural Healthcare Settings: a Lesson from Cancer Clinics, Int. J. Environ. Res. Public Health 17 (5) (Mar 6 2020), doi: 10.3390/ijerph17051728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Ramsey AT, Chiu A, Baker T, et al. , Care-paradigm shift promoting smoking cessation treatment among cancer center patients via a low-burden strategy, Electronic Health Record-Enabled Evidence-Based Smoking Cessation Treatment, Transl Behav Med (Jul 17 2019), doi: 10.1093/tbm/ibz107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Thorndike AN, Regan S, Rigotti NA, The treatment of smoking by US physicians during ambulatory visits: 1994 2003, Am. J. Public Health 97 (10) (Oct 2007) 1878–1883, doi: 10.2105/AJPH.2006.092577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Dietz PM, Homa D, England LJ, et al. , Estimates of nondisclosure of cigarette smoking among pregnant and nonpregnant women of reproductive age in the United States, Am. J. Epidemiol 173 (3) (Feb 1 2011) 355–359 doi:kwq381 [pii], doi: 10.1093/aje/kwq381. [DOI] [PubMed] [Google Scholar]
- [63].Kalkhoran S, Kruse GR, Chang Y, Rigotti NA, Smoking-Cessation Efforts by US Adult Smokers with Medical Comorbidities, Am. J. Med 131 (3) (Mar 2018) 318 e1–318 e8, doi: 10.1016/j.amjmed.2017.09.025. [DOI] [PubMed] [Google Scholar]
- [64].Yang M, Mehta HB, Bhowmik D, Essien EJ, Abughosh SM, Predictors of smoking cessation medication use among nonobese and obese smokers, Subst. Use Misuse 49 (6) (May 2014) 752–761, doi: 10.3109/10826084.2014.880121. [DOI] [PubMed] [Google Scholar]
- [65].Gomez-Coronado N, Walker AJ, Berk M, Dodd S, Current and emerging pharmacotherapies for cessation of tobacco smoking, Pharmacotherapy 38 (2) (Feb 2018) 235–258, doi: 10.1002/phar.2073. [DOI] [PubMed] [Google Scholar]
- [66].Hartmann-Boyce J, Chepkin SC, Ye W, Bullen C, Lancaster T, Nicotine replacement therapy versus control for smoking cessation, Cochrane Database Syst. Rev 5 (May 31 2018,) CD000146, doi: 10.1002/14651858.CD000146.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [67].Piper ME, Smith SS, Schlam TR, et al. , A randomized placebo-controlled clinical trial of 5 smoking cessation pharmacotherapies, Arch. Gen. Psychiatry 66 (11) (Nov 2009) 1253–1262 doi:66/11/1253 [pii], doi: 10.1001/archgenpsychiatry.2009.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Gonzales D, Rennard SI, Nides M, et al. , Varenicline, an alpha4beta2 nicotinic acetylcholine receptor partial agonist, vs sustained-release bupropion and placebo for smoking cessation: a randomized controlled trial, JAMA 296 (1) (Jul 5 2006) 47–55 doi:296/1/47 [pii], doi: 10.1001/jama.296.1.47. [DOI] [PubMed] [Google Scholar]
- [69].Jorenby DE, Hays JT, Rigotti NA, et al. , Efficacy of varenicline, an alpha4beta2 nicotinic acetylcholine receptor partial agonist, vs placebo or sustained-release bupropion for smoking cessation: a randomized controlled trial, JAMA 296 (1) (Jul 5 2006) 56–63 doi:296/1/56 [pii], doi: 10.1001/jama.296.1.56. [DOI] [PubMed] [Google Scholar]
- [70].Fiore MC, Jaen CR, TB B, al. e. Treating tobacco use and dependence: 2008 update. 2008. http://www.ahrq.gov/path/tobacco.htm#Clinic
- [71].Fiore M, Adsit R, Zehner M, et al. , An electronic health record-based interoperable eReferral system to enhance smoking Quitline treatment in primary care, J. Am. Med. Inform. Assoc 26 (8–9) (Aug 1 2019) 778–786, doi: 10.1093/jamia/ocz044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Panagiotou OA, Schuit E, Munafo MR, Bennett DA, Bergen AW, David SP, Smoking Cessation pharmacotherapy based on genetically-informed biomarkers: what is the evidence? Nicotine Tob. Res 21 (9) (Aug 19 2019) 1289–1293, doi: 10.1093/ntr/ntz009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Chen LS, Baker TB, Bray M, Pham G, Bierut LJ, Predicting Smoking Cessation Treamtent Outcomes With Genetic Risk Scores And Biomarkers, presented at: Society of Research for Nicotine and Tobacco, 2021 Virtual due to COVID. [Google Scholar]
- [74].Lerman C, Schnoll RA, Hawk LW Jr., et al. , Use of the nicotine metabolite ratio as a genetically informed biomarker of response to nicotine patch or varenicline for smoking cessation: a randomised, double-blind placebo-controlled trial, Lancet Respir. Med 3 (2) (Feb 2015) 131–138, doi: 10.1016/S2213-2600(14)70294-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [75].Zhu AZ, Zhou Q, Cox LS, et al. , Association of CHRNA5-A3-B4 SNP rs2036527 with smoking cessation therapy response in African-American smokers, Clin. Pharmacol. Ther (Apr 14 2014) doi:clpt201488 [pii], doi: 10.1038/clpt.2014.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Salloum NC, Buchalter ELF, Chanani S, et al. , From genes to treatments: a systematic review of the pharmacogenetics in smoking cessation, Pharmacogenomics 19 (10) (Jul 1 2018) 861–871, doi: 10.2217/pgs-2018-0023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Levy DE, Youatt EJ, Shields AE, Primary care physicians’ concerns about offering a genetic test to tailor smoking cessation treatment, Genet. Med 9 (12) (Dec 2007) 842–849, doi: 10.1097/gim.0b013e31815bf953. [DOI] [PubMed] [Google Scholar]
- [78].Schnoll RA, Shields AE, Physician barriers to incorporating pharmacogenetic treatment strategies for nicotine dependence into clinical practice, Clin. Pharmacol. Ther 89 (3) (Mar 2011) 345–347, doi: 10.1038/clpt.2010.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [79].Shields AE, Blumenthal D, Weiss KB, Comstock CB, Currivan D, Lerman C, Barriers to translating emerging genetic research on smoking into clinical practice. Perspectives of primary care physicians, J. Gen. Intern. Med 20 (2) (Feb 2005) 131–138, doi: 10.1111/j.1525-1497.2005.30429.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [80].Shields AE, Levy DE, Blumenthal D, et al. , Primary care physicians’ willingness to offer a new genetic test to tailor smoking treatment, according to test characteristics, Nicotine Tob. Res 10 (6) (Jun 2008) 1037–1045, doi: 10.1080/14622200802087580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [81].Marti J, Assessing preferences for improved smoking cessation medications: a discrete choice experiment, Eur. J. Health Econ 13 (5) (Oct 2012) 533–548, doi: 10.1007/s10198-011-0333-z. [DOI] [PubMed] [Google Scholar]
- [82].Vogt F, Hall S, Marteau TM, General practitioners’ beliefs about effectiveness and intentions to prescribe smoking cessation medications: qualitative and quantitative studies, BMC Public Health 6 (Nov 8 2006) 277, doi: 10.1186/1471-2458-6-277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [83].Cox LS, Bronars CA, Thomas JL, et al. , Achieving high rates of consent for genetic testing among African American smokers, Nicotine Tob. Res 9 (6) (Jun 2007) 711–716, doi: 10.1080/14622200701365228. [DOI] [PubMed] [Google Scholar]
- [84].Park ER, Kleimann S, Youatt EJ, et al. , Black and White adults’ perspectives on the genetics of nicotine addiction susceptibility, Addict. Behav 36 (7) (Jul 2011) 769–772, doi: 10.1016/j.addbeh.2011.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [85].Quaak M, Smerecnik C, van Schooten FJ, de Vries H, van Schayck CP, Knowledge, attitudes and preferences regarding genetic testing for smoking cessation. A cross-sectional survey among Dutch smokers, BMJ Open 2 (2012) e000321, doi: 10.1136/bmjopen-2011-000321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [86].Ramsey AT, Bourdon JL, Bray M, et al. , Proof of concept of a personalized genetic risk tool to promote smoking cessation: high acceptability and reduced cigarette smoking, Cancer Prev. Res. (Phila.) 14 (2) (Feb 2021) 253–262, doi: 10.1158/1940-6207.CAPR-20-0328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [87].Senft N, Sanderson M, Selove R, et al. , Attitudes toward precision treatment of smoking in the southern community cohort study, Cancer Epidemiol. Biomarkers Prev 28 (8) (Aug 2019) 1345–1352, doi: 10.1158/1055-9965.EPI-19-0179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [88].Vogt F, Hall S, Marteau TM, Understanding why smokers do not want to use nicotine dependence medications to stop smoking: qualitative and quantitative studies, Nicotine Tob. Res 10 (8) (Aug 2008) 1405–1413 doi:901415516 [pii], doi: 10.1080/14622200802239280. [DOI] [PubMed] [Google Scholar]
- [89].Ho MK, Mwenifumbo JC, Al Koudsi N, et al. , Association of nicotine metabolite ratio and CYP2A6 genotype with smoking cessation treatment in African-American light smokers, Clin. Pharmacol. Ther 85 (6) (Jun 2009) 635–643 doi:clpt200919 [pii], doi: 10.1038/clpt.2009.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [90].Lerman C, Jepson C, Wileyto EP, et al. , Genetic variation in nicotine metabolism predicts the efficacy of extended-duration transdermal nicotine therapy, Clin. Pharmacol. Ther 87 (5) (May 2010) 553–557 doi:clpt20103 [pii], doi: 10.1038/clpt.2010.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [91].Lerman C, Tyndale R, Patterson F, et al. , Nicotine metabolite ratio predicts efficacy of transdermal nicotine for smoking cessation, Clin. Pharmacol. Ther 79 (6) (Jun 2006) 600–608 doi:S0009–9236(06)00071–3 [pii], doi: 10.1016/j.clpt.2006.02.006. [DOI] [PubMed] [Google Scholar]
- [92].Chen LS, Hartz SM, Baker TB, Ma Y, LS N, Bierut LJ, Use of polygenic risk scores of nicotine metabolism in predicting smoking behaviors, Pharmacogenomics 19 (18) (Dec 2018) 1383–1394, doi: 10.2217/pgs-2018-0081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [93].El-Boraie A, Taghavi T, Chenoweth MJ, et al. , Evaluation of a weighted genetic risk score for the prediction of biomarkers of CYP2A6 activity, Addict. Biol 25 (1) (Jan 2020) e12741, doi: 10.1111/adb.12741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [94].Chenoweth MJ, Ware JJ, Zhu AZX, et al. , Genome-wide association study of a nicotine metabolism biomarker in African American smokers: impact of chromosome 19 genetic influences, Addiction 113 (3) (Mar 2018) 509–523, doi: 10.1111/add.14032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [95].Bergen AW, McMahan CS, McGee S, et al. , Multiethnic Prediction of Nicotine Biomarkers and Association With Nicotine Dependence, Nicotine Tob. Res 23 (12) (Nov 5 2021) 2162–2169, doi: 10.1093/ntr/ntab124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [96].Gold AB, Lerman C, Pharmacogenetics of smoking cessation: role of nicotine target and metabolism genes, Hum. Genet (Jan 31 2012), doi: 10.1007/s00439-012-1143-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [97].Rose JE, Behm FM, Drgon T, Johnson C, Uhl GR, Personalized smoking cessation: interactions between nicotine dose, dependence and quit-success genotype score, Mol. Med 16 (7–8) (Jul-Aug 2010) 247–253 doi:molmed.2009.00159 [pii], doi: 10.2119/molmed.2009.00159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [98].Uhl GR, Drgon T, Johnson C, Ramoni MF, Behm FM, Rose JE, Genome-wide association for smoking cessation success in a trial of precessation nicotine replacement, Mol. Med 16 (11–12) (Nov-Dec 2010) 513–526 doi:molmed.2010.00052 [pii], doi: 10.2119/molmed.2010.00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [99].McGeary JE, Knopik VS, Hayes JE, Palmer RH, Monti PM, Kalman D, Predictors of relapse in a bupropion trial for smoking cessation in recently-abstinent alcoholics: preliminary results using an aggregate genetic risk score, Subst. Abuse 6 (2012) 107–114 [pii], doi: 10.4137/SART.S8866sart-6-2012-107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [100].David SP, Johnstone EC, Churchman M, Aveyard P, Murphy MF, Munafo MR, Pharmacogenetics of smoking cessation in general practice: results from the patch II and patch in practice trials, Nicotine Tob. Res 13 (3) (Mar 2011) 157–167, doi: 10.1093/ntr/ntq246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [101].Ahluwalia JS, Okuyemi K, Nollen N, et al. , The effects of nicotine gum and counseling among African American light smokers: a 2 × 2 factorial design, Addiction 101 (6) (Jun 2006) 883–891, doi: 10.1111/j.1360-0443.2006.01461.x. [DOI] [PubMed] [Google Scholar]
- [102].Teutsch SM, Bradley LA, Palomaki GE, et al. , The Evaluation of Genomic Applications in Practice and Prevention (EGAPP) Initiative: methods of the EGAPP Working Group, Genet. Med 11 (1) (Jan 2009) 3–14, doi: 10.1097/GIM.0b013e318184137c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [103].Klein RJ, Gumus ZH, Are polygenic risk scores ready for the cancer clinic?-a perspective, Transl. Lung Cancer Res 11 (5) (May 2022) 910–919, doi: 10.21037/tlcr-21-698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [104].Wand H, Lambert SA, Tamburro C, et al. , Improving reporting standards for polygenic scores in risk prediction studies, Nature 591 (7849) (Mar 2021) 211–219, doi: 10.1038/s41586-021-03243-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [105].Bierut LJ, Stitzel JA, Wang JC, et al. , Nicotine dependence and the a5-a3-b4 nicotinic receptor gene cluster: variants in the nicotinic receptors alter the risk for nicotine dependence, Am. J. Psychiatry 9 (165) (2008) 1163–1171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [106].Bloss CS, Madlensky L, Schork NJ, Topol EJ, Genomic information as a behavioral health intervention: can it work? Per. Med 8 (6) (Nov 2011) 659–667, doi: 10.2217/pme.11.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [107].Klein WMP, McBride CM, Allen CG, et al. , Optimal integration of behavioral medicine into clinical genetics and genomics, Am. J. Hum. Genet 104 (2) (Feb 7 2019) 193–196, doi: 10.1016/j.ajhg.2019.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [108].Cancer Facts & Figures For African Americans 2019–2021, Society AC, 2019. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/cancer-facts-and-figures-for-african-americans/cancer-facts-and-figures-for-african-americans-2019-2021.pdf.
- [109].Nollen NL, Mayo MS, Sanderson Cox L, et al. , Factors that explain differences in abstinence between black and white smokers: a prospective intervention study, J. Natl. Cancer Inst 111 (10) (Oct 1 2019) 1078–1087, doi: 10.1093/jnci/djz001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [110].Shiffman S, Brockwell SE, Pillitteri JL, Gitchell JG, Individual differences in adoption of treatment for smoking cessation: demographic and smoking history characteristics, Drug Alcohol Depend. 93 (1–2) (Jan 11 2008) 121–131, doi: 10.1016/j.drugalcdep.2007.09.005. [DOI] [PubMed] [Google Scholar]
- [111].Burgess DJ, van Ryn M, Noorbaloochi S, et al. , Smoking cessation among African American and white smokers in the Veterans Affairs health care system, Am. J. Public Health 104 (Suppl 4) (Sep 2014) S580–S587, doi: 10.2105/AJPH.2014.302023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [112].Cox LS, Nollen NL, Mayo MS, et al. , Bupropion for smoking cessation in African American light smokers: a randomized controlled trial, J. Natl. Cancer Inst 104 (4) (Feb 22 2012) 290–298, doi: 10.1093/jnci/djr513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [113].Weiss J, Kuusisto F, Boyd K, Liu J, Page D, Machine Learning for Treatment Assignment: improving Individualized Risk Attribution, AMIA Annu. Symp. Proc 2015 (2015) 1306–1315. [PMC free article] [PubMed] [Google Scholar]
- [114].Ramsey AT, Chen LS, Hartz SM, et al. , Toward the implementation of genomic applications for smoking cessation and smoking-related diseases, Transl. Behav. Med 8 (1) (Jan 29 2018) 7–17, doi: 10.1093/tbm/ibx060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [115].Ramsey AT, Proctor EK, Chambers DA, et al. , Designing for accelerated translation (DART) of Emerging Innovations in Health, J. Clin. Transl. Sci 3 (2–3) (Jun 2019) 53–58, doi: 10.1017/cts.2019.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [116].Scoville EA, Tindle HA, Wells QS, et al. , Precision nicotine metabolism-informed care for smoking cessation in Crohn’s disease: a pilot study, PLoS ONE 15 (3) (2020) e0230656, doi: 10.1371/journal.pone.0230656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [117].Ramsey AT, Rammaha T, Salyer P, Chang Y, Bourdon J, Chen L-S, Bierut L, Emerging Evidence of Feasibility and Behavior Change Associated With Fully-Remote Delivery of a Genetically-Informed Smoking Cessation Intervention, presented at: The College on Problems of Drug Dependence (CPDD), 2022. [Google Scholar]
- [118].D’Angelo H, Ramsey AT, Rolland B, et al. , Pragmatic application of the RE-AIM framework to evaluate the implementation of tobacco cessation programs within NCI-designated cancer centers, Front. Public Health 8 (2020) 221, doi: 10.3389/fpubh.2020.00221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [119].D’Angelo H, Rolland B, Adsit R, et al. , Tobacco treatment program implementation at NCI Cancer Centers: progress of the NCI cancer moonshot-funded cancer center cessation initiative, Cancer Prev. Res. (Phila.) 12 (11) (Nov 2019) 735–740, doi: 10.1158/1940-6207.CAPR-19-0182. [DOI] [PubMed] [Google Scholar]
- [120].Salloum RG, D’Angelo H, Theis RP, et al. , Mixed-methods economic evaluation of the implementation of tobacco treatment programs in National Cancer Institute-designated cancer centers, Implement. Sci. Commun 2 (1) (Apr 9 2021) 41, doi: 10.1186/s43058-021-00144-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [121].Fiore MC, D’Angelo H, Baker T, Effective cessation treatment for patients with cancer who smoke-the fourth pillar of cancer care, JAMA Netw. Open 2 (9) (Sep 4 2019) e1912264, doi: 10.1001/jamanetworkopen.2019.12264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [122].Chen LS, Baker TB, Bierut LJ, The value of control conditions for evaluating pharmacogenetic effects, Pharmacogenomics 16 (18) (2015) 2005–2006, doi: 10.2217/pgs.15.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [123].Chen LS, Zawertailo L, Piasecki TM, et al. , Leveraging genomic data in smoking cessation trials in the era of precision medicine: why and how, Nicotine Tob. Res 20 (4) (Mar 6 2018) 414–424, doi: 10.1093/ntr/ntx097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [124].Saccone NL, Baurley JW, Bergen AW, et al. , The value of biosamples in smoking cessation trials: a review of genetic, metabolomic, and epigenetic findings, Nicotine Tob. Res 20 (4) (Mar 6 2018) 403–413, doi: 10.1093/ntr/ntx096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [125].The All of Us Research Program. https://allofus.nih.gov/
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data was used for the research described in the article.


