Abstract
Retroperitoneal sarcomas (RPSs) are rare findings that can grow into large masses without eliciting severe symptoms. At present, surgical resection is the only radical therapy, whenever it can be performed with the aim to achieve a complete removal of the tumor. The present report describes two consecutive cases of RPSs that resulted in dedifferentiated liposarcomas (DDLPSs) and these patients underwent R0 surgical resection with and without a nephron-sparing procedure. The diagnostic workup, the surgical approach, the impact of late surgical management due to the COVID pandemic and the latest literature on the topic are discussed and analyzed. The patients, who refused to undergo any medical examination during the prior 2 years due to the COVID pandemic, were admitted to Federico II University Hospital (Naples, Italy) complaining about weight loss and general abdominal discomfort. In the first case, a primitive giant abdominal right neoplasm of retroperitoneal origin enveloping and medializing the right kidney was observed. The second patient had a similar primitive retroperitoneal giant left neoplasm, which did not affect the kidney. Given the characteristics of the masses and the absence of distant metastases, after a multidisciplinary discussion, radical surgical removal was carried out for both patients. The lesions appeared well-defined from the surrounding tissues, and markedly compressed all the adjacent organs, without signs of infiltration. In the first patient, the right kidney was surrounded and undetachable from the tumor and it was removed en bloc with the mass. The second patient benefited from a nephron-sparing resection, due to the existence of a clear cleavage plane. The postoperative courses were uneventful. Both the histological examinations were oriented towards a DDLPS and both patients benefited from adjuvant chemotherapy. In conclusion, the treatment of giant RPS is still challenging and requires multidisciplinary treatment as well as, when possible, radical surgical removal. The lack of tissue infiltration and the avoidance of excision or reconstruction of major organs (including the kidney) could lead to an easier postoperative course and an improved prognosis. When possible, surgical management of recurrences or incompletely resected masses must be pursued. Since the COVID pandemic caused limited medicalization of a number of population groups and delayed diagnosis of other oncologic diseases, an increased number of DDLPSs could be expected in the near future.
Keywords: giant retroperitoneal sarcoma, dedifferentiated liposarcoma, nephrectomy, surgical excision, radical resection, COVID, late diagnosis
Introduction
Primary adult retroperitoneal tumors include a wide range of neoplasms, of which mesenchymal tumors are the most common histotype (1,2). Soft-tissue sarcomas are rare malignancies accounting for approximately 1% of all adult tumors (3) with more than 50 different histologic subtypes (2).
Liposarcomas (LPS) are among the most frequent malignant retroperitoneal neoplasms of adipose origin (0.9/100,000 people/year) (4–6). Histologically, they are divided into well-differentiated LPS (WDLPS), dedifferentiated LPS (DDLPS), myxoid LPS, pleomorphic LPS, and myxoid pleomorphic LPS (2).
The most frequent retroperitoneal sarcoma (RPS) is DDLPS (incidence of 0.1/1,000,000 per year) (7), which mainly affects patients in their sixth-seventh decade (8). Due to the retroperitoneum's anatomic characteristics, such lesions can grow considerably and involve multiple contiguous structures before the diagnosis is carried out (5,6). Symptoms are often vague and even large neoplasms are usually diagnosed incidentally (9). As per the majority of solid tumors (10–12), the main treatment of non-metastatic retroperitoneal RPS and DDLPS is radical surgery with the inclusion of involved structures, accompanied or not by radiotherapy in some specialized centers (3,5,13–16), while the advanced or metastatic disease can only benefit from systemic chemotherapy and molecular targeted agents or debulking, when possible (3,13–18).
Among the organs most frequently involved/infiltrated by retroperitoneal giant DDLPS we find the kidneys. Although in these cases nephrectomy is sometimes performed for technical reasons, its validity from an oncological point of view is still debated.
The pandemic had a strong impact on health resources and medical activity, making hospitals and diagnostic services less accessible to the general population. Outpatient prevention and screening services have suffered many limitations due to the relocation of health personnel to COVID departments or services, significantly lengthening the waiting lists for all the other services (19–21). Patients were also reluctant to attend health facilities as they feared contagion. An increase in late-stage diagnoses of malignant diseases, especially breast (22,23), prostate (24), and colon cancer (25) has been recorded worldwide, and Italy was one of the most strongly affected countries. Moreover, many Centers experienced significant delays in the surgical treatment of oncologic diseases, due to operating theatres' schedule rearrangements (26).
Herein, we describe two successful radical surgical removals of giant retroperitoneal DDLPS (a one-time primitive tumor resection and a two-step surgical resection), treated with/without kidney sparing, and describe the available evidence in the literature.
Case report
Case 1
A 56-year-old woman complaining of vague digestive disorders in September 2021 was prescribed an abdominal ultrasound and a gastroscopy, which were not performed due to both the considerable lengthening of the waiting lists and the fear of contracting COVID at the healthcare facilities. Following the worsening of her symptoms, after almost two years and the reduction of the COVID emergency, she finally performed an abdominal ultrasound in May 2022 (General Electrics Ultrasound System, model Logiq S8 XDclear (equipped with 3.5, 5, and 7.5 MHz probes) at our Institution that revealed a giant inhomogeneous abdominal mass. At admission, the patient reported weight loss (7 kg in the previous 2 months) and severe abdominal discomfort. Her medical history was not significant, her blood chemistry tests showed a slight elevation of LDH (250 U/l, normal range 125–243 U/l) and a mild iron deficiency anemia (25 mcg/dl, normal range 50–170 mcg/dl), while all the tumoral and viral markers were negative.
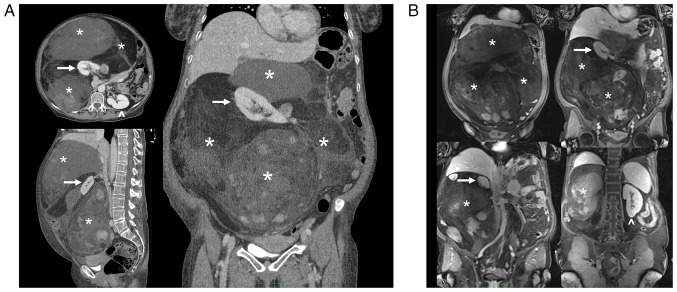
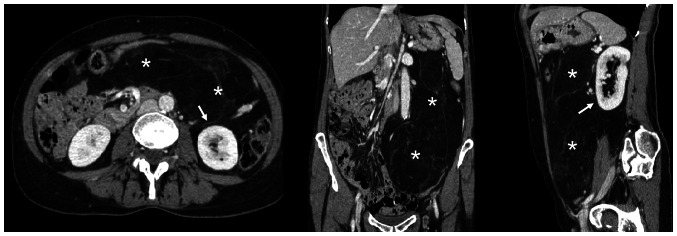
A total body CT scan, MRI, and PET confirmed the presence of the voluminous (max diam. 31 cm) and inhomogeneous mass with intra-lesion cystic components and excluded the presence of distant lesions consistent with an RPS (Figs. 1 and 2).
Figure 1.
Case 1. (A) CT scan and (B) post-contrast MRI (venous phase; T1 fat-saturated) findings showing different components of the giant tumor (asterisk) originating from the right retroperitoneum and enveloping and medializing the ipsilateral kidney. The relationship with the great vessels and other organs is shown. Arrow, right kidney; arrow head, left kidney.
Figure 2.
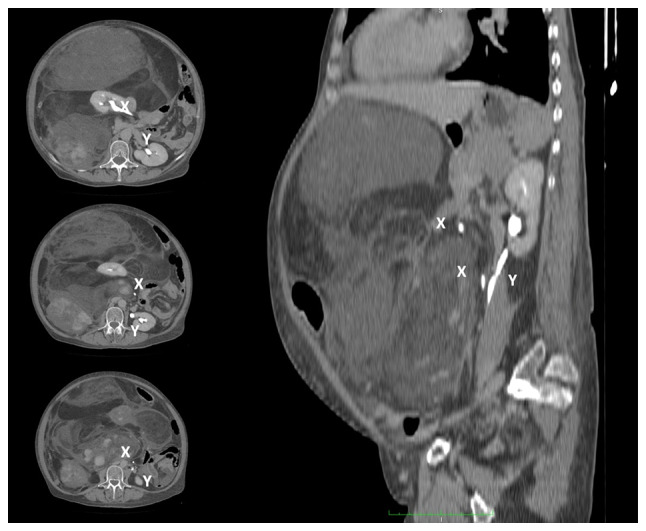
Case 1. CT-scan showing the medialization of the right kidney by the giant retroperitoneal tumor. The ureter is shifted contralaterally and runs parallel to the left ureter. X, right ureter; Y, left ureter.
For the CT scan we used a Toshiba Aquilion 64 four-phase CT scan (Toshiba Aquilion 64, Japan) with a protocol including basal and post contrast medium IV injection (30–35 sec after injection for the arterial phase; 75–80 sec after contrast injection to obtain the venous phase; 3–5 min after injection for the delayed phase).
For the MRI we used Siemens Magnetom Trio High-field MRI (Magnetom Trio 3T, Siemens Medical Imaging, Erlangen, Germany) with a protocol including a T1 and T2 weighted sequences, also with TE opposed-phase and fat suppression, completed with DWI study and acquisition before, during and after e.v. injection of paramagnetic contrast media.
For the CT-PET we used a Philips Ingenuity 64 and the exam was performed after intravenous injection of (18F) FDG. Images of the isotope distribution were acquired in 15 min. PET images were reconstructed with 4 mm thick tomographic slices. The CT exam was performed with tomographic sections of 2 mm (120 kV) and dose modulation in relation to the patient's build.
An oncologic consultation suggested a CT-PET-guided biopsy, which was oriented toward a mesenchymal tumor. Finally, the patient underwent surgical removal of the mass.
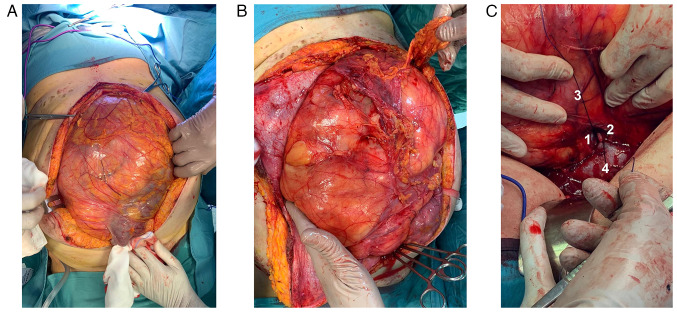
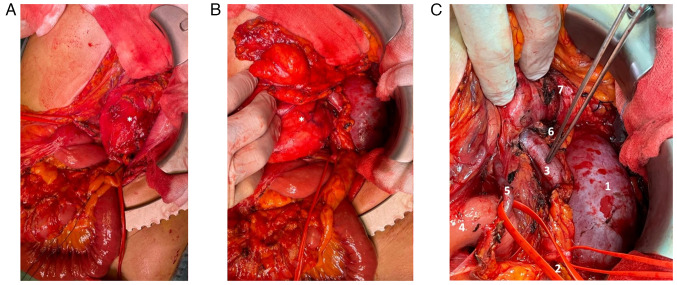
Ureteral stents were placed bilaterally, and a xipho-pubic incision with supra-umbilical right lateral split was performed. The lesion significantly protruded out of the abdomen, compressing all the viscera, and determining important abdominal hypertension. However, it appeared non-infiltrating and well-defined from the surrounding tissues (Fig. 3A and B). The vena cava was used as a landmark to isolate the renal veins: the ureters were running in parallel, and the right kidney was completely embedded and undetachable from the mass: therefore, a right nephrectomy was mandatory (Fig. 3C). After the exposure of the retroperitoneal plane and the diaphragm, the mass was removed. An inter-aorto-caval lymph-node picking, right adrenalectomy, and cholecystectomy were performed.
Figure 3.
Case 1. Intraoperative findings. After (A) median incision and (B) right lateral split, the giant retroperitoneal tumor spontaneously bulges out of the abdominal cavity. (C) Detail of the right kidney's vein and artery during vascular control, prior to the organ's removal (en bloc with the tumor): The organ is completely enveloped by the tumor. 1, renal vein (ligated); 2, renal artery (ligated); 3, tumor; and 4, vena cava.
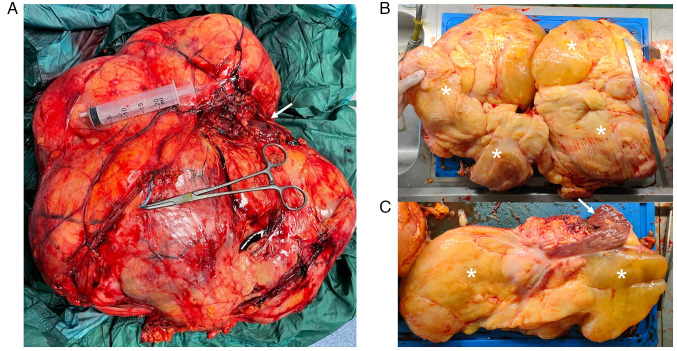
Following surgery two PRBCs were infused due to mild anemia. The further postoperative course was uneventful. The lesion's diameters were 41×36×16 cm and it weighed 13.56 Kg (Fig. 4), with yellow, lobulated, and partly gelatinous sections, and a focal bone metaplastic area of 3.5 cm. No liquid and/or cystic areas were observed. The right renal parenchyma appeared twisted and deformed, but not infiltrated. Surgical margins were negative (R0) and none of the isolated lymph nodes and intraoperative specimens showed neoplastic features (N0, M0).
Figure 4.
Case 1. (A) Whole and (B) sectioned specimen. (C) Relationship between the tumor and the kidney. Asterisks, tumor; arrows, kidney.
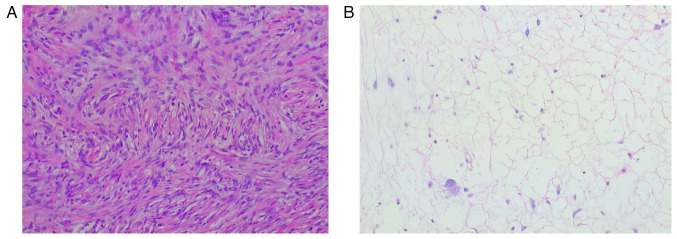
The histological examination was performed through a hematoxylin and eosin staining, carried out on 4-µm thick sections from formalin fixed, paraffin embedded tissues. The slides underwent an automated processing step (Fig. 5). Immunohistochemical staining was not performed due to the peculiar characteristics of the lesion, that suggested a DDLPS diagnosis: the sections showed morphological features typical of dedifferentiated liposarcoma, such as the presence of areas of atypical lipomatous tumor (ALT), with mature adipocytes in which substantial variation in cell size is appreciated, and areas of non-lipogenic sarcoma with low grade dedifferentiation showing fibroblastic spindle cells with moderate nuclear atypia, organized in a fascicular architectural pattern, and exhibiting medium to high cellularity. Transition between the two areas was abrupt. Hence, immunohistochemical staining was considered not necessary for this diagnosis. The histological features, together with the outcome of the imaging exams, the clinical trend and the intraoperative findings were all compatible with the diagnosis of DDLPS, Stage IIIb (larger than 10 cm, not spread to nearby lymph nodes or to distant sites) (27). The patient started adjuvant chemotherapy: the schedule consisted of doxorubicin (70 mg/m2) intravenously for 20 min and ifosfamide (4 g/m2) with mesna intravenously for 24 h at day 1, and pegfilgrastim subcutaneously, starting 24 h after completing ifosfamide. Four cycles were given at 3 week intervals (28,29). The control CT scan performed on POD8 and a control MRI at eight months showed no signs of recurrence; at discharge, LDH was 290 U/l and iron levels were at 30 mcg/dl, thus iron supplementation was prescribed. Even if the patient was referred to nephrological follow-up, apparently the nephrectomy and the subsequent chemotherapy had no reflections on the renal function, and the left kidney fully compensated for the loss of the contralateral organ: the creatinine value at the last follow-up was 1.1 mg/dl (normal range: 0.6–1.1 mg/dl). The next check-up at our outpatient clinic is set at 12 months after surgery.
Figure 5.
Case 1. Microscopic findings (haematoxylin and eosin; original magnification, ×20). (A) Dedifferentiated component, similar to a fibro-istocitoma, with a storiform architectural pattern. (B) Well-differentiated liposarcoma with components similar to a lipoma with atypical cells scattered among the mature adipocytes.
Case 2
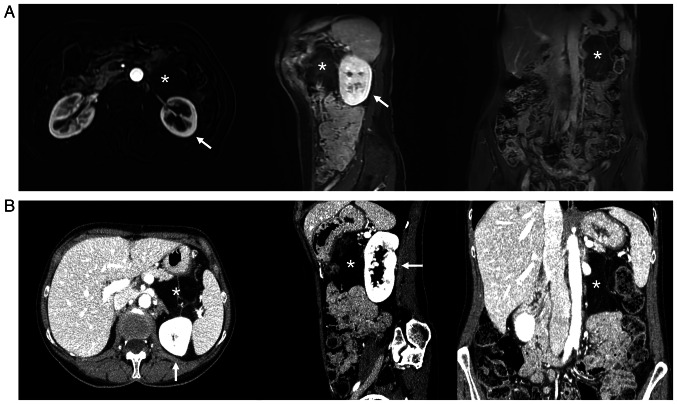
With the same timing and due to the same COVID-related delays of the first patient, a 61-year-old woman complaining about worsening general abdominal discomfort for two years finally underwent an abdominal US in February 2022 at another hospital (although the first indication to perform this diagnostic test was given in January 2021), that revealed a giant abdominal inhomogeneous mass. A preoperative CT scan showed a giant inhomogeneous non-infiltrating retroperitoneal lesion (23×17×6.5 cm) (Fig. 6).
Figure 6.
Case 2. CT-scan findings showing a giant tumor (asterisk) originating from the left retroperitoneum, and its relationship to other organs and the ipsilateral kidney (arrow).
Blood chemistry tests were the norm, as well as viral and tumoral markers. Given the worsening of her symptoms, the patient underwent laparoscopic excision of the mass in April 2022, in another Institute. When approaching the anterior aspect of the pancreas and the III–IV duodenal portion the resection resulted difficult, so excision of this level was interrupted, resulting in an incomplete resection (R2). The postoperative course was complicated by acute pancreatitis. Histologically, the lesion appeared to be a DDLPS with a maximum diameter of 19 cm.
As soon as postoperative pancreatitis subsided in May 2022, a control US, MRI, and PET (performed at our Institution, with the same equipment and protocols described above) (Fig. 7) revealed the presence of a recurrence/residue of disease (9×6.5×4.6 cm), close to the Treitz ligament and anteriorly to the left kidney, but the absence of distant lesions. The presence of disappearing fat in the fat-suppressor sequences, the constant isointensity of the solid component with that of neighboring muscle structures, the inhomogeneous uptake/absence of uptake of the contrast medium/beta-emitting tracer in some areas of the mass, and the presence of necrotic areas all made us suspect a DDLPS (Fig. 7).
Figure 7.
Case 2. Liposarcoma relapse (asterisk), ventrally to the left kidney (arrow). (A) MRI. Axial (T1; arterial phase; fat-saturated), coronal and sagittal (T1 venous phase; fat-saturated). (B) CT scan.
The patient was admitted to our Institution reporting weight loss (5 Kg in 3 months) and was enlisted for radical resection, scheduled in June 2022. After a median incision and a wide adhesions ablation, the mass appeared non-infiltrative and detachable from the left kidney, pancreatic tail, left colic flexure, and splenic hilum. After the individuation of a resection plane, the mass was removed en bloc without resecting other structures (Fig. 8).
Figure 8.
Case 2. Intraoperative findings. (A) Tumor recurrence (asterisk). (B) The latero-medial isolation of the tumor allows its mobilization from the left kidney and its hilar elements to perform a nephron sparing resection. (C) Operating field after removal of the tumor recurrence. 1, left kidney; 2, left ureter; 3, left renal vein; 4, Treitz ligament; 5, inferior mesenteric vein; 6, left adrenal gland; and 7, pancreas tail.
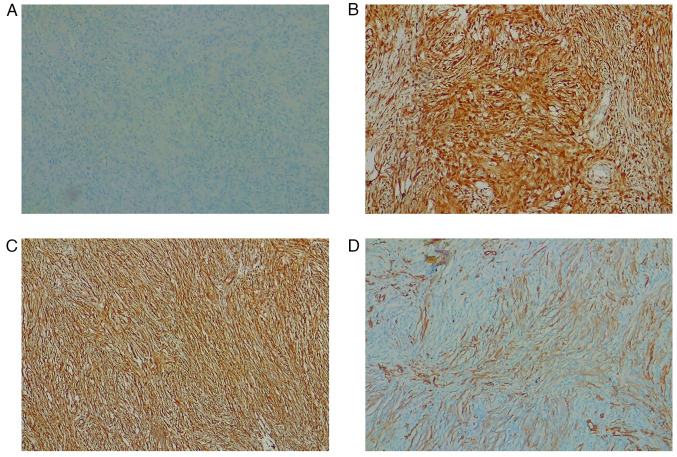
The postoperative course was uneventful, and also this patient underwent adjuvant chemotherapy with the same protocol as in case 1. Histological and immunohistochemical examination confirmed the previously stated diagnosis of DDLPS. Hematoxylin and eosin staining was performed as described above, but the results were not as decisive as in the first case so to better define the diagnosis we performed other tests. Thus, immunohistochemical analysis was performed, using 4-µm-thick formalin-fixed, paraffin-embedded tissue sections. Sections were stained for immunohistochemical analysis by using standard techniques and the antibodies' dilutions were performed according to the instructions predefined by the manufacturer. Briefly, consecutive parallel sections were stained with the following antibodies: anti-s100 (rabbit polyclonal antibody anti-s100 protein, CONFIRM, catalog Number: 760-2523), anti-p16 protein (mouse monoclonal anti-p16 antibody, clone E6H4, CINtec, catalog Number: 705-4793), anti-Vimentin (mouse monoclonal antibody, clone V9, VENTANA, catalog Number: 790-2917), anti-SMA (mouse monoclonal Primary Antibody, clone 1A4, VENTANA, catalog number: 760-2833). Our sample was S-100-negative, p16-positive, Vimentin positive, and SMA-positive (Fig. 9); CDK4, MDM2 and STAT6 immunohistochemical analysis were not performed due to the lack of antibodies. Overall, the clinical and instrumental data were all compatible with the diagnosis of DDLPS, Stage IIIa (larger than 5 cm but not more than 10 cm, not spread to nearby lymph nodes or to distant sites) (27). Two postoperative CT scans at one week and 7 months revealed no signs of recurrence, and a good creatinine value (0.8 mg/dl) was found at the last follow-up. The next check-up at our outpatient clinic is set at 12 months after surgery.
Figure 9.
Case 2. Immunohistochemical analysis of giant liposarcoma tissues (original magnification, ×20). (A) S-100-negative. (B) p16-positive. (C) Vimentin-positive. (D) Smooth muscle actin-positive. CDK4, MDM2 and STAT6 immunohistochemical analysis were not performed due to the lack of antibodies.
Discussion
Surgical resection is the gold standard treatment of RPS, as several guidelines (3,13–15) and case series (30–32) confirm. Some papers including one of the largest series to date (11,33), comprising 8653 patients undergoing surgery for primary RPS, advise against the routine use of chemotherapy since the currently available regimens do not confer a significant survival advantage. However, the superiority of a radical excision comprising surrounding organs vs. the resection of the tumor alone is still under debate (15,31,34–38).
Given their rarity and heterogeneity, the available literature often groups all RPS in cohorts spanning a considerable number of years. In a 28-year study, a single-centered series of 675 patients (31), determined that R1 resection (along with histologic type, age, and tumor size) was independently associated with local recurrence. Moreover, some histological types, such as liposarcomas and leiomyosarcomas, were associated with recurrence also until 15 years from diagnosis, and with lower disease-specific overall survival (OS) (31,32).
RPS is associated with a considerable recurrence rate, accounting for 70.5% of a recent multicentric series of 567 RPS patients undergoing surgery (39). The 5-years OS depends on the recurrence site (local, distal, or both), the possibility of undergoing a second resection, and its timing. MacNeill et al (30) reported the post-relapse outcomes for 1007 RPS surgical patients: recurrence is predictive of high mortality and a new surgical treatment should be considered when radicality is possible, as we did for our second case.
Surgical series of DDLPSs are scarce, given the rarity of these tumors and the tendency of grouping them with other RPSs. A recent cohort of 32 patients undergoing resection (18 DDLPSs vs. 14 WDLPS) showed a significant difference in recurrence-free survival times at 2 years (26% vs. 59%, respectively; P=0.005), but could not confirm a difference in OS (40). Local recurrence of DDLPS develops four times more frequently compared with WDLPS (38), especially if the dedifferentiated component is found at the resection margin (15,35–37,41). All these considerations gain even more validity for bigger lesions and giant tumors (5,42–45).
The definition of giant tumors is vague (5,42–44), but guidelines and literature reports support surgery as a valid treatment for both small and giant DDLPSs (1,3,5,6,13,15,34,36). However, despite being the optimal therapy, the surgical removal of giant DDLPSs presents a great degree of difficulty and a low R0 rate (5,44).
DDLPSs are the most frequent histotype of RPS. They appear as de novo lesions in 90% of cases, while the remaining DDLPSs progress after a previous well-differentiated RPS develops recurrence, most likely in a time-dependent dedifferentiation process (2). For this reason, giant DDLPSs often present de-differentiated areas (4,13,31,38). DDLPSs appear as predominantly non-lipomatous neoplasms that are adjacent, inside or encompass a fatty mass (46).
Imaging is crucial in the diagnosis and preoperative management of DDLPSs (9,14,46). All the investigations carried out preoperatively were fundamental for the selection of the correct treatment: the absence of infiltration of large retroperitoneal vessels and the lack of distant metastases led to choosing an aggressive treatment instead of palliation (3,15,17,18,37). At the same time, the finding of morphological and functional characteristics at the operating table (absence of cystic areas and larger-than-expected dimensions) did not negatively affect surgical removal, as we observed in our first case.
The biopsy's goal is to evaluate both surgical and medical management and eventually perform neoadjuvant therapy, as recommended by international guidelines (3,15,47). In our first case, given the primitive nature of the lesion, a preoperative biopsy was performed percutaneously, as surgical biopsy could favor abdominal wall seeding due to accidental tumor rupture (1,15,47). When deemed necessary, given their common inhomogeneity, we suggest a TC-PET-guided core needle biopsy of the lesions to obtain a more substantial and contributive tissue sample. However, neoadjuvant chemotherapy is not routinely recommended (16) and, in the case of symptomatic giant masses, there is no possibility of waiting for the chemotherapy's effects. Moreover, the presence of distant lesions had already been excluded by preoperative imaging. Therefore, a biopsy could be avoided in symptomatic upfront resectable giant RPS.
Our patients were not candidates for neoadjuvant radiotherapy, due to the extension of the lesions, the onset of symptoms, and their proximity to radiosensitive organs (3,14,15). For this reason, a more invasive but radical surgical approach was selected.
Radiologic embolization can be considered a preoperative treatment whenever a giant mass has a clear vascular axis, both to shrink the mass and to reduce intraoperative blood loss (3). This procedure was taken into consideration for both patients but was finally excluded due to the absence of a single clear tributary vessel. Moreover, a preoperative kidney embolization could be considered when an organ-preserving resection is not possible (45) and the removal of the kidney is certain. In our first patient, although the kidney was enveloped by the tumor, renal vessels' embolization was not contemplated because of the preoperative uncertainty of being able to take away all the mass containing the kidney, while the second patient did not show a clear renal involvement. Moreover, preoperative embolization of RPS is scarcely reported in the literature and can lead to severe complications (i.e.: post-embolization syndrome).
Giant lesions' invasiveness and boundaries are assessed mostly intraoperatively, and, even after an extensive preoperative discussion, it is often difficult to anticipate the surgical management in detail (5,9,36,42). This is particularly true for unexpected vascular invasion, which has a worse prognosis by definition and could hamper the margins' radicality. Vascular resections, especially when retroperitoneal vessels are involved, are difficult to couple with an en bloc resection, as they often require an incomplete R1 debulking before reaching optimal exposure, leading to a higher rate of peritoneal seeding (37,42). Our first case benefited from an en-bloc resection without vascular reconstruction, allowing a no-touch technique and avoiding seeding. Although visceral or vascular invasion after recurrence or incomplete resection is common (30,36,39) our second patient benefited from an early and prompt radical second look without the need for organ removals or vascular reconstruction. This reinforces our belief that patients with recurrence or previous R1 resections should be thoroughly studied and evaluated for surgery, when possible. Both cases showed the lesions' confinement to the surgical field and surgery was performed without the exposure of tumoral tissue; however, after surgical manipulation, adjuvant chemotherapy and a strict follow-up are necessary (32,33).
Our first patient is one of the few giant retroperitoneal DDLPS reported in the literature that could be resected with negative margins and no vascular resections, despite being >40 cm of maximum diameter in a non-obese patient (43,44,48). Incidental primitive lesions in otherwise healthy patients are even rarer (43,48).
In both cases, surgery was performed respecting the lesion's plane without needing bowel resections or reconstruction of major structures. Mortality from RPLS usually results from local recurrence, underlining the importance of an R0 resection, sometimes hard to reach for the difficulty in differentiating the liposarcoma from adjacent normal fat, and for the absence of an evident anatomical vascular peduncle (49). Although this, the prognosis from RPS following R0 resection also depends on its histologic type, indeed the 5-year survival rate for well-differentiated subtypes is 90, 75% for dedifferentiated, 60–90% for myxoid/round cell, and 30–50% for pleomorphic subtypes (50). It has also been demonstrated that tumor burden and nephrectomy are not associated with disease-specific survival (51).
Due to the tumor's location, en bloc resection of close organs together with RLPS is not always avoidable. The necessity of multi-visceral resection (≥3 organs) has been correlated to a higher local recurrence rate and worse survival rate (15,31,36,37). However, a recent systematic review seems to dismiss this suggestion (35). In case of kidney invasion, it is necessary to balance the benefit of free margin resection against medical complications and quality of life loss. In the first patient, a nephrectomy was unavoidable due to the complete embedding of the right kidney into the lesion.
The second patient benefited from a nephron-sparing resection thanks to an early diagnosis that possibly favored the presence of an adequate cleavage plane and the absence of infiltration.
These two cases show the duality of kidney resection in the radical surgical treatment of giant DDLPSs: the first case was a first presentation of DDLPs and unfortunately the kidney was removed for technical reasons as it was surrounded by the tumor even if this organ was not infiltrated. At the same time, in the second case we treated a large DDLPS recurrence, that often requires contextual extensive organs removals, through a kidney-sparing procedure, achieving nonetheless a radical resection. Our cases showed how nephrectomy should be evaluated case by case and, in selected cases it is still possible achieve radical surgical removal of a DDLPs through nephron-sparing surgery, even in case of a recurrence.
So in conclusion the surgical aggression of a recurrence does not necessarily have to foresee the removal of the kidney as well as the presence of a giant liposarcoma involving but not infiltrating the kidney can basically foresee the removal of the mass saving the organ.
In a case series of 228 patients undergoing surgery for RLPS, the group of those not requiring multi-organ resection had a higher 10-year survival rate as opposed to those requiring multi-organ resection (35% vs. 26%) (52). This suggests that, although invasion to renal parenchyma and the possibility of future recurrence from that site, the choice of performing a nephron-sparing resection must be balanced on the tumor's histologic type, patient's age, and increased morbidity from resection (53).
Despite the existence of reports describing excision through a minimally-invasive approach (54,55), our experience and literature data suggest performing an open approach when dealing with a giant retroperitoneal mass since the retroperitoneal plane is often narrow and movements could be restricted by the mass, abdominal hypertension and visceral dislocation (5,43,56). The proximity with important vessels and anatomic structures must be assessed carefully, and severe intraoperative complications are possible when an adequate surgical field is difficult to attain (57,58). However, to reach definitive statements on the matter it is necessary to wait for case series with a substantial number of cases and a long-term follow-up.
As well as because of mild symptoms, the late diagnosis could also be attributed to the COVID pandemic, due to the patient's resistance to going through a diagnostic process at the first warning signs. Moreover, the accessibility of diagnostic services was limited due to the overload of hospitalized COVID patients, which caused important delays and undertreatment of many non-respiratory disease patients and bore a dramatic impact on oncologic disease patients (21,59).
The COVID-19 pandemic has resulted in a massive backlog of the elective case as pointed out in a retrospective case-control study; anonymized case-related routine data of a Germany-wide voluntary hospital association (CLINOTEL association) (60).
Delays in the provision of ‘elective’ surgical care have dire consequences for the patient: progression of disease and comorbidities, higher complication rates, and lower overall survival. Delays in elective surgery also have significant consequences for the healthcare system: added emotional strain on healthcare workers, reduction in training opportunities, rising costs, and increased inequality in health-service provision (61). The COVID-19 pandemic led to disruptions in the delivery of cancer treatments and the most relevant factors associated with this delay are subjective patient issues and local healthcare organizations (62).
This was particularly important in Italy, which suffered among the strongest restrictions in Europe at that time (19,22,24). Healthcare workflow was heavily affected as well since oncologic multidisciplinary discussions could only be performed via online meetings and outpatient visits were reduced to the minimum, following necessary anti-COVID tests (20,25,26). To the best of our knowledge, our cases represent the first report of surgically treated giant DDLPS during the COVID era. Moreover, these two cases were discovered and treated in a short time span (two months). Although there are no studies comparing the stage of disease at diagnosis in pre- and post-COVID era, given the growth pattern of DDLPS (2) and the recent delay of medical attention to non-respiratory diseases, an increased number of RPS and DDLPS cases could be expected shortly.
In conclusion, the treatment of giant DDLPS is still challenging and requires multidisciplinary treatment as well as, when possible, radical surgical removal. The lack of tissue infiltration and the avoidance of major organs' excision or reconstruction could lead to an easier postoperative course and a better prognosis. In the current state of knowledge for the treatment of giant DDLPS, an open approach is preferable over minimally invasive techniques. In the same way, considering the technical and oncological point of view, the excision of the kidneys together with the neoplastic mass should be avoided and the surgical management of recurrences or incompletely resected masses must be pursued. As the COVID pandemic caused a limited medicalization of many population groups and a late diagnosis of other oncologic diseases, an increased number of DDLPSs could be expected in the next future.
Acknowledgements
Not applicable.
Funding Statement
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
FP, MLS, NC and VD participated in the multidisciplinary meetings prior to surgeries, operated the patients, conceived and designed the manuscript, acquired and confirmed the authenticity of all the raw data, drafted the final version of the manuscript, and provided the surgical details described in the manuscript. DS, LC, MI and CGM performed the radiologic imaging of all patients in the present study, and provided radiologic advice and the machinery details described in the present study. LI and RA performed histologic analysis of the specimens and provided all pathologic details described in the present study. MG provided oncologic advice and treated both patients, providing all details of the adjuvant treatment described in the present study. AS, LP, NC and AJ performed a critical literature review, contributed to the acquisition, analysis and interpretation of data, contributed to the drafting of the Introduction and Discussion, and provided critical surgical advice. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Given the retrospective nature of the present study, ethics committee approval was not necessary.
Patient consent for publication
The patients signed a specific informed consent that included the acquirement of clinical data and pictures in an anonymous form for publication purposes.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Improta L, Tzanis D, Bouhadiba T, Abdelhafidh K, Bonvalot S. Overview of primary adult retroperitoneal tumours. Eur J Surg Oncol. 2020;46:1573–1579. doi: 10.1016/j.ejso.2020.04.054. [DOI] [PubMed] [Google Scholar]
- 2.Sbaraglia M, Bellan E, Dei Tos AP. The 2020 WHO Classification of Soft Tissue Tumours: News and perspectives. Pathologica. 2021;113:70–84. doi: 10.32074/1591-951X-213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.von Mehren M, Randall RL, Benjamin RS, Boles S, Bui MM, Ganjoo KN, George S, Gonzalez RJ, Heslin MJ, Kane JM, et al. Soft tissue sarcoma, version 2.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018;16:536–563. doi: 10.6004/jnccn.2018.0025. [DOI] [PubMed] [Google Scholar]
- 4.Ducimetière F, Lurkin A, Ranchère-Vince D, Decouvelaere AV, Péoc'h M, Istier L, Chalabreysse P, Muller C, Alberti L, Bringuier PP, et al. Incidence of sarcoma histotypes and molecular subtypes in a prospective epidemiological study with central pathology review and molecular testing. PLoS One. 2011;6:e20294. doi: 10.1371/journal.pone.0020294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bachmann R, Eckert F, Gelfert D, Strohäker J, Beltzer C, Ladurner R. Perioperative strategy and outcome in giant retroperitoneal dedifferentiated liposarcoma-results of a retrospective cohort study. World J Surg Oncol. 2020;18:296. doi: 10.1186/s12957-020-02069-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hassan I, Park SZ, Donohue JH, Nagorney DM, Kay PA, Nasciemento AG, Schleck CD, Ilstrup DM. Operative management of primary retroperitoneal sarcomas: A reappraisal of an institutional experience. Ann Surg. 2004;239:244–250. doi: 10.1097/01.sla.0000108670.31446.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hirata M, Asano N, Katayama K, Yoshida A, Tsuda Y, Sekimizu M, Mitani S, Kobayashi E, Komiyama M, Fujimoto H, et al. Integrated exome and RNA sequencing of dedifferentiated liposarcoma. Nat Commun. 2019;10:5683. doi: 10.1038/s41467-019-13286-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nishio J, Nakayama S, Nabeshima K, Yamamoto T. Biology and management of dedifferentiated liposarcoma: State of the art and perspectives. J Clin Med. 2021;10:3230. doi: 10.3390/jcm10153230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Messiou C, Moskovic E, Vanel D, Morosi C, Benchimol R, Strauss D, Miah A, Douis H, van Houdt W, Bonvalot S. Primary retroperitoneal soft tissue sarcoma: Imaging appearances, pitfalls and diagnostic algorithm. Eur J Surg Oncol. 2017;43:1191–1198. doi: 10.1016/j.ejso.2016.10.032. [DOI] [PubMed] [Google Scholar]
- 10.Capece M, Creta M, Calogero A, La Rocca R, Napolitano L, Barone B, Sica A, Fusco F, Santangelo M, Dodaro C, et al. Does physical activity regulate prostate carcinogenesis and prostate cancer outcomes? A narrative review. Int J Environ Res Public Health. 2020;17:1441. doi: 10.3390/ijerph17041441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Strocchi E, Iaffaioli RV, Facchini G, Mantovani G, Ricci S, Cavallo G, Tortoriello A, D'Angelo R, Formato R, Rosato G, et al. Stop-flow technique for loco-regional delivery of high dose chemotherapy in the treatment of advanced pelvic cancers. Eur J Surg Oncol. 2004;30:663–670. doi: 10.1016/j.ejso.2004.04.005. [DOI] [PubMed] [Google Scholar]
- 12.Santangelo M, Esposito A, Tammaro V, Calogero A, Criscitiello C, Roberti G, Candida M, Rupealta N, Pisani A, Carlomagno N. What indication, morbidity and mortality for central pancreatectomy in oncological surgery? A systematic review. Int J Surg. 2016;28((Suppl 1)):S172–S176. doi: 10.1016/j.ijsu.2015.12.046. [DOI] [PubMed] [Google Scholar]
- 13.Kilpatrick SE. Dedifferentiated liposarcoma: A comprehensive historical review with proposed evidence-based guidelines regarding a diagnosis in need of further clarification. Adv Anatomic Pathol. 2021;28:426–438. doi: 10.1097/PAP.0000000000000314. [DOI] [PubMed] [Google Scholar]
- 14.Baldini EH, Wang D, Haas RL, Catton CN, Indelicato DJ, Kirsch DG, Roberge D, Salerno K, Deville C, Guadagnolo BA, et al. Treatment guidelines for preoperative radiation therapy for retroperitoneal sarcoma: Preliminary consensus of an international expert panel. Int J Radiat Oncol Biol Phys. 2015;92:602–612. doi: 10.1016/j.ijrobp.2015.02.013. [DOI] [PubMed] [Google Scholar]
- 15.Casali P, Abecassis N, Aro HT, Bauer S, Biagini R, Bielack S, Bonvalot S, Boukovinas I, Bovee JVMG, Brodowicz T, et al. Soft tissue and visceral sarcomas: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29((Suppl 4)):iv51–iv67. doi: 10.1093/annonc/mdy310. [DOI] [PubMed] [Google Scholar]
- 16.Callegaro D, Raut CP, Keung EZ, Kim T, Le Pechoux C, Martin-Broto J, Gronchi A, Swallow C, Gladdy R. Strategies for care of patients with gastrointestinal stromal tumor or soft tissue sarcoma during COVID-19 pandemic: A guide for surgical oncologists. J Surg Oncol. 2021;123:12–23. doi: 10.1002/jso.26246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Keung EZ, Lazar AJ, Torres KE, Wang WL, Cormier JN, Ashleigh Guadagnolo B, Bishop AJ, Lin H, Hunt KK, Bird J, et al. Phase II study of neoadjuvant checkpoint blockade in patients with surgically resectable undifferentiated pleomorphic sarcoma and dedifferentiated liposarcoma. BMC Cancer. 2018;18:913. doi: 10.1186/s12885-018-4829-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roland CL, Keung EZ-Y, Lazar AJ, Torres KE, Wang WL, Guadagnolo A, Bishop AJ, Lin HY, Hunt Y, Feig BW, et al. Preliminary results of a phase II study of neoadjuvant checkpoint blockade for surgically resectable undifferentiated pleomorphic sarcoma (UPS) and dedifferentiated liposarcoma (DDLPS) Am Soc Clin Oncol. 2020;38:11505. doi: 10.1200/JCO.2020.38.15_suppl.11505. [DOI] [Google Scholar]
- 19.Ferrara G, De Vincentiis L, Ambrosini-Spaltro A, Barbareschi M, Bertolini V, Contato E, Crivelli F, Feyles E, Mariani MP, Morelli L, et al. Cancer diagnostic delay in northern and central Italy during the 2020 lockdown due to the coronavirus disease 2019 pandemic. Am J Clin Pathol. 2021;155:64–68. doi: 10.1093/ajcp/aqaa177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Battisti F, Falini P, Gorini G, Sassoli de Bianchi P, Armaroli P, Giubilato P, Giorgi Rossi P, Zorzi M, Battagello J, Senore C, et al. Cancer screening programmes in Italy during the COVID-19 pandemic: An update of a nationwide survey on activity volumes and delayed diagnoses. Ann Ist Super Sanita. 2022;58:16–24. doi: 10.4415/ANN_22_01_03. [DOI] [PubMed] [Google Scholar]
- 21.Bracale U, Podda M, Castiglioni S, Peltrini R, Sartori A, Arezzo A, Corcione F, Agresta F, CLOUD-19 Collaborative Group Changes in surgicaL behaviOrs dUring the CoviD-19 pandemic. The SICE CLOUD19 Study. Updates Surg. 2021;73:731–744. doi: 10.1007/s13304-021-01010-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mentrasti G, Cantini L, Vici P, D'Ostilio N, La Verde N, Chiari R, Paolucci V, Crocetti S, De Filippis C, Pecci F, et al. Rising incidence of late stage breast cancer after COVID-19 outbreak. Real-world data from the Italian COVID-DELAY study. Breast. 2022;65:164–171. doi: 10.1016/j.breast.2022.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Piscitelli P, Santoriello A, Buonaguro FM, Di Maio M, Iolascon G, Gimigliano F, Marinelli A, Distante A, Serravezza G, Sordi E, et al. Incidence of breast cancer in Italy: Mastectomies and quadrantectomies performed between 2000 and 2005. J Exp Clin Cancer Res. 2009;28:86. doi: 10.1186/1756-9966-28-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pepe P, Pepe L, Pennisi M, Fraggetta F. Prostate cancer diagnosis and management during one year of the COVID-19 pandemic. Anticancer Res. 2021;41:3127–3130. doi: 10.21873/anticanres.15097. [DOI] [PubMed] [Google Scholar]
- 25.Ricciardiello L, Ferrari C, Cameletti M, Gaianill F, Buttitta F, Bazzoli F, Luigi de'Angelis G, Malesci A, Laghi L. Impact of SARS-CoV-2 pandemic on colorectal cancer screening delay: Effect on stage shift and increased mortality. Clin Gastroenterol Hepatol. 2021;19:1410–1417.e9. doi: 10.1016/j.cgh.2020.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lazzerini M, Barbi E, Apicella A, Marchetti F, Cardinale F, Trobia G. Delayed access or provision of care in Italy resulting from fear of COVID-19. Lancet Child Adolesc Health. 2020;4:e10–e11. doi: 10.1016/S2352-4642(20)30108-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR, Winchester DP. The Eighth edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more ‘personalized’ approach to cancer staging. CA Cancer J Clin. 2017;67:93–99. doi: 10.3322/caac.21388. [DOI] [PubMed] [Google Scholar]
- 28.Woll PJ, Reichardt P, Le Cesne A, Bonvalot S, Azzarelli A, Hoekstra HJ, Leahy M, Van Coevorden F, Verweij J, Hogendoorn PC, et al. Adjuvant chemotherapy with doxorubicin, ifosfamide, and lenograstim for resected soft-tissue sarcoma (EORTC 62931): A multicentre randomised controlled trial. Lancet Oncol. 2012;13:1045–1054. doi: 10.1016/S1470-2045(12)70346-7. [DOI] [PubMed] [Google Scholar]
- 29.Gronchi A, Miah AB, Dei Tos AP, Abecassis N, Bajpai J, Bauer S, Biagini R, Bielack S, Blay JY, Bolle S, et al. Soft tissue and visceral sarcomas: ESMO-EURACAN-GENTURIS Clinical Practice Guidelines for diagnosis, treatment and follow-up(★) Ann Oncol. 2021;32:1348–1365. doi: 10.1016/j.annonc.2021.07.006. [DOI] [PubMed] [Google Scholar]
- 30.MacNeill AJ, Miceli R, Strauss DC, Bonvalot S, Hohenberger P, Van Coevorden F, Rutkowski P, Callegaro D, Hayes AJ, Honoré C, et al. Post-relapse outcomes after primary extended resection of retroperitoneal sarcoma: A report from the Trans-Atlantic RPS Working Group. Cancer. 2017;123:1971–1978. doi: 10.1002/cncr.30572. [DOI] [PubMed] [Google Scholar]
- 31.Tan MC, Brennan MF, Kuk D, Agaram NP, Antonescu CR, Qin LX, Moraco N, Crago AM, Singer S. Histology-based classification predicts pattern of recurrence and improves risk stratification in primary retroperitoneal sarcoma. Ann Surg. 2016;263:593–600. doi: 10.1097/SLA.0000000000001149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhao X, Li P, Huang X, Chen L, Liu N, She Y. Prognostic factors predicting the postoperative survival period following treatment for primary retroperitoneal liposarcoma. Chin Med J. 2015;128:85–90. doi: 10.4103/0366-6999.147822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Miura JT, Charlson J, Gamblin TC, Eastwood D, Banerjee A, Johnston FM, Turaga KK. Impact of chemotherapy on survival in surgically resected retroperitoneal sarcoma. Eur J Surg Oncol. 2015;41:1386–1392. doi: 10.1016/j.ejso.2015.07.014. [DOI] [PubMed] [Google Scholar]
- 34.Villano AM, Zeymo A, Nigam A, Chan KS, Shara N, Unger KR, Al-Refaie WB. Radical excision for retroperitoneal soft tissue sarcoma: A national propensity-matched outcomes analysis. Surgery. 2020;168:831–837. doi: 10.1016/j.surg.2020.05.031. [DOI] [PubMed] [Google Scholar]
- 35.Guo Q, Zhao J, Du X, Huang B. Survival outcomes of surgery for retroperitoneal sarcomas: A systematic review and meta-analysis. PLoS One. 2022;17:e0272044. doi: 10.1371/journal.pone.0272044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bonvalot S, Miceli R, Berselli M, Causeret S, Colombo C, Mariani L, Bouzaiene H, Le Péchoux C, Casali PG, Le Cesne A, et al. Aggressive surgery in retroperitoneal soft tissue sarcoma carried out at high-volume centers is safe and is associated with improved local control. Ann Surg Oncol. 2010;17:1507–1514. doi: 10.1245/s10434-010-1057-5. [DOI] [PubMed] [Google Scholar]
- 37.Bonvalot S, Rivoire M, Castaing M, Stoeckle E, Le Cesne A, Blay JY, Laplanche A. Primary retroperitoneal sarcomas: A multivariate analysis of surgical factors associated with local control. J Clin Oncol. 2009;27:31–37. doi: 10.1200/JCO.2008.18.0802. [DOI] [PubMed] [Google Scholar]
- 38.Singer S, Antonescu CR, Riedel E, Brennan MF. Histologic subtype and margin of resection predict pattern of recurrence and survival for retroperitoneal liposarcoma. Ann Surg. 2003;238:358–371. doi: 10.1097/01.sla.0000086542.11899.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.van Houdt WJ, Fiore M, Barretta F, Rutkowski P, Blay JY, Lahat G, Strauss D, Gonzalez RJ, Ahuja N, Grignani G, et al. Patterns of recurrence and survival probability after second recurrence of retroperitoneal sarcoma: A study from TARPSWG. Cancer. 2020;126:4917–4925. doi: 10.1002/cncr.33139. [DOI] [PubMed] [Google Scholar]
- 40.Osuna-Soto J, Caro Cuenca T, Sanz-Zorrilla A, Torrecilla-Martínez A, Ortega Salas R, Leiva-Cepas F. Prognosis and survival of patients diagnosed with well-differentiated and dedifferentiated retroperitoneal liposarcoma. Cir Esp (Engl Ed) 2022;100:622–628. doi: 10.1016/j.ciresp.2021.06.010. [DOI] [PubMed] [Google Scholar]
- 41.Dehner CA, Hagemann IS, Chrisinger JSA. Retroperitoneal dedifferentiated liposarcoma. Am J Clin Pathol. 2021;156:920–925. doi: 10.1093/ajcp/aqab051. [DOI] [PubMed] [Google Scholar]
- 42.Fodor M, Maglione M, Kogler P, Kafka-Ritsch R, Ofner D, Perathoner A. Challenges in the treatment of a giant retroperitoneal liposarcoma. Ann Ital Chir. 2020;9 S2239253X20033162. [PubMed] [Google Scholar]
- 43.Inoue K, Higaki Y, Yoshida H. Giant retroperitoneal liposarcoma. Int J Urol. 2005;12:220–222. doi: 10.1111/j.1442-2042.2005.01019.x. [DOI] [PubMed] [Google Scholar]
- 44.Kanthala L, Ray S, Aurobindo Prasad Das S, Nundy S, Mehta N. Recurrent giant retroperitoneal liposarcoma: Review of literature and a rare case report. Ann Med Surg. 2021;65:102329. doi: 10.1016/j.amsu.2021.102329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Galatola R, Stanzione A, Sirignano C, Mainolfi C, Guadagno E, Carlomagno N, Insabato L, Santangelo M, Maurea S. Giant Epithelioid Angiomyolipoma: An imaging-related differential diagnosis among Fat-containing renal masses. Clin Genitourin Cancer. 2020;18:e5–e9. doi: 10.1016/j.clgc.2019.10.028. [DOI] [PubMed] [Google Scholar]
- 46.Czeyda-Pommersheim F, Menias C, Boustani A, Revzin M. Diagnostic approach to primary retroperitoneal pathologies: What the radiologist needs to know. Abdom Radiol (NY) 2021;46:1062–1081. doi: 10.1007/s00261-020-02752-8. [DOI] [PubMed] [Google Scholar]
- 47.Trans-Atlantic RPS Working Group Management of recurrent retroperitoneal sarcoma (RPS) in the adult: A consensus approach from the Trans-Atlantic RPS Working Group. Ann Surg Oncol. 2016;23:3531–3540. doi: 10.1245/s10434-016-5336-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dubois-Silva A, Barbagelata-Lopez C. Retroperitoneal dedifferentiated liposarcoma. Intern Emerg Med. 2019;14:619–620. doi: 10.1007/s11739-018-2004-x. [DOI] [PubMed] [Google Scholar]
- 49.Nijhuis PH, Sars PR, Plaat BE, Molenaar WM, Sluiter WJ, Hoekstra HJ. Clinico-pathological data and prognostic factors in completely resected AJCC stage I–III liposarcomas. Ann Surg Oncol. 2000;7:535–543. doi: 10.1007/s10434-000-0535-6. [DOI] [PubMed] [Google Scholar]
- 50.Karadayi K, Yildiz C, Karakus S, Kurt A, Bozkurt B, Soylu S, Cicekli AA, Egilmez R, Cetin A. Well-differentiated abdominal liposarcoma: Experience of a tertiary care center. World J Surg Oncol. 2015;13:166. doi: 10.1186/s12957-015-0580-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tseng WW, Madewell JE, Wei W, Somaiah N, Lazar AJ, Ghadimi MP, Hoffman A, Pisters PW, Lev DC, Pollock RE. Locoregional disease patterns in well-differentiated and dedifferentiated retroperitoneal liposarcoma: Implications for the extent of resection? Ann Surg Oncol. 2014;21:2136–2143. doi: 10.1245/s10434-014-3643-4. [DOI] [PubMed] [Google Scholar]
- 52.Stilidi IS, Nikulin MP, Nered SN, Davydov MM, Bolotskiĭ VI, Gubina GI. Combined operations by retroperitoneal liposarcomas. Khirurgiia (Mosk) 2013:20–25. (In Russian) [PubMed] [Google Scholar]
- 53.Argadjendra M, Napitupulu R, Yudadi R, Hoetama S, Wibowo HS. Kidney sparing giant retroperitoneal liposarcoma: Case report and literature review. Int J Surg Case Rep. 2019;56:70–73. doi: 10.1016/j.ijscr.2019.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Agrusa A, Di Buono G, Buscemi S, Randisi B, Gulotta L, Sorce V, Badalamenti G, Albano D, Galia M, Romano G, Gulotta G. Dedifferentiated retroperitoneal large liposarcoma and laparoscopic treatment: Is it possible and safe? The first literature case report. Int J Surg Case Rep. 2019;57:113–117. doi: 10.1016/j.ijscr.2019.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Xiao J, Liu J, Chen M, Liu W, He X. Diagnosis and prognosis of retroperitoneal liposarcoma: A single Asian center cohort of 57 Cases. J Oncol. 2021;2021:7594027. doi: 10.1155/2021/7594027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ferrarese A, Pozzi G, Borghi F, Marano A, Delbon P, Amato B, Santangelo M, Buccelli C, Niola M, Martino V, Capasso E. Malfunctions of robotic system in surgery: Role and responsibility of surgeon in legal point of view. Open Med (Wars) 2016;11:286–291. doi: 10.1515/med-2016-0055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Liu H, Hu T, Li Y, Yue Z, Zhang F, Fu J. Successful intraoperative management in patients with abdominal compartment syndrome induced by giant liposarcomas: Two case reports. Medicine. 2020;99:e22575. doi: 10.1097/MD.0000000000022575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Santangelo M, De Rosa P, Spiezia S, Spinosa G, Grassia S, Zuccaro M, Renda A. Healing of surgical incision in kidney transplantation: A single transplant center's experience. Transplant Proc. 2006;38:1044–1046. doi: 10.1016/j.transproceed.2006.03.005. [DOI] [PubMed] [Google Scholar]
- 59.Effect of COVID-19 pandemic lockdowns on planned cancer surgery for 15 tumour types in 61 countries: An international, prospective, cohort study. Lancet Oncol. 2021;22:1507–1517. doi: 10.1016/S1470-2045(21)00493-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hunger R, König V, Stillger R, Mantke R. Impact of the COVID-19 pandemic on delays in surgical procedures in Germany: A multi-center analysis of an administrative registry of 176,783 patients. Patient Saf Surg. 2022;16:22. doi: 10.1186/s13037-022-00331-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Nel D. Strategies for recovery of a surgical service in the COVID-19 era. S Afr J Surg. 2022;60:154–159. doi: 10.17159/2078-5151/sajs3783. [DOI] [PubMed] [Google Scholar]
- 62.Mullangi S, Aviki EM, Chen Y, Robson M, Hershman DL. Factors associated with cancer treatment delay among patients diagnosed with COVID-19. JAMA Netw Open. 2022;5:e2224296. doi: 10.1001/jamanetworkopen.2022.24296. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.