Abstract
Reactivation of herpes simplex virus type 1 (HSV-1) in the trigeminal ganglion (TG) was induced by UV irradiation of the corneas of latently infected mice. Immunocytochemistry was used to monitor the dynamics of cytokine (interleukin-2 [IL-2], IL-4, IL-6, IL-10, gamma interferon [IFN-γ], and tumor necrosis factor alpha [TNF-α]) and viral antigen production in the TG and the adjacent central nervous system on days 1 to 4, 6, 7, and 10 after irradiation. UV irradiation induced increased expression of IL-6 and TNF-α from satellite cells in uninfected TG. In latently infected TG, prior to reactivation, all satellite cells were TNF-α+ and most were also IL-6+. Reactivation, evidenced by HSV-1 antigens and/or infiltrating immune cells, occurred in 28 of 45 (62%) TG samples. Viral antigens were present in the TG in neurons, often disintegrating on days 2 to 6 after irradiation. Infected neurons were usually surrounded by satellite cells and the foci of immune cells producing TNF-α and/or IL-6. IL-4+ cells were detected as early as day 3 and were more numerous by day 10 (a very few IL-2+ and/or IFN-γ+ cells were seen at this time). No IL-10 was detected at any time. Our observations indicate that UV irradiation of the cornea may modulate cytokine production by satellite cells. We confirm that neurons are the site of reactivation and that they probably do not survive this event. The predominance of TNF-α and IL-6 following reactivation parallels primary infection in the TG and suggests a role in viral clearance. The presence of Th2-type cytokines (IL-4 and IL-6) indicates a role for antibody. Thus, several clearance mechanisms may be at work.
The ability of herpes simplex virus (HSV-1) to reactivate from latency in sensory ganglia is central to the pathogenesis of recurrent infection. Several studies have strongly suggested that the neuron is the site of reactivation (16, 21), although the fate of such neurons is still undetermined. After reactivation in vivo in the trigeminal ganglion (TG), only small numbers of virus antigen-positive neurons have been identified, and only small amounts of infectious virus were detected (16, 21). This highly restricted replication may explain the failure to detect virus DNA replication and the transience of expression of productive-cycle transcripts (1). Reactivation will, of course, occur in a host with an immunity already primed against the virus. It is therefore likely that the mounting of a virus-specific secondary immune response will play a major part in the rapid and effective control of infection. This supposition is supported by the observation of focal infiltrates of T cells, both CD4+ and CD8+, in close association with virus antigen-positive neurons as early as 1 day after stimulation to induce reactivation (21). Although these lymphocytes were the predominant infiltrating cell type when virus antigen was being cleared, by day 4 large numbers of B cells were also present, suggesting that local antibody production may also aid the control of reactivated infection. It appears that the efficiency of the immune system in controlling reactivated infection within the sensory ganglion, at least in the mouse, results in a significant proportion of reactivation events being aborted at an early stage, before they can lead to disease or viral shedding at the periphery (21).
After reactivation, the initial presentation of antigen is likely to be mediated by resident major histocompatibility complex (MHC) class II+/F4/80+ immune cells (21). Their presence, together with the rapid appearance of T cells (probably virus-specific memory cells) provide the basis for the secondary immune response. However, a direct cytotoxic role for CD8+ T cells is problematic, since neurons do not normally express MHC class I and are well protected by ensheathing satellite cells. Nevertheless, these T cells may play a role in viral clearance via the production of antiviral cytokines. Evidence for such a function comes from studies on hepatitis B virus infection, where secretion of gamma interferon (IFN-γ) and tumor necrosis factor alpha (TNF-α), by CD8+ T cells can abolish viral gene expression and replication (7).
The production of a range of cytokines in the TG following primary infection with HSV-1 has been investigated by a variety of methods, but there is no consensus on which cytokines are of primary importance during the clearance of virus. For example, using double staining we have demonstrated large numbers of TNF-α+ and/or IL-6+ cells, together with smaller numbers of IFN-γ+ cells, early in the course of infection (day 3), and these were seen in close association with virus antigen (22). mRNA for IFN-γ and TNF-α were detected by reverse-transcriptase (RT)-PCR at a similar time (3). In contrast, in the immunohistochemical study of Liu et al. (13) IFN-γ and IL-4 were the predominant cytokines present early in infection. The role of IL-10 during viral clearance also appears to be equivocal; in our studies no IL-10+ cells were detected, but others have identified small numbers of such cells (13) and Halford et al. (8) found mRNA for this cytokine in 100% of the TG samples taken 5 to 7 days after infection.
In addition to infiltrating immune cells, the resident satellite and Schwann cells of the peripheral nervous system (PNS) and astrocytes in the central nervous system (CNS) at the dorsal root entry zone (DRE) can also be potent sources of TNF-α and IL-6. It has been suggested that, via these cytokines, glial cells may also be involved in viral clearance and in normal homeostatic mechanisms in the nervous system, such as repair and protection of neurons from damage (22).
In contrast to the events in the TG during primary infection, the role of cytokines in reactivation has only recently started to be explored (4, 11, 14). We now report studies on cytokine production (IL-2, IL-4, IL-6, IL-10, IFN-γ, and TNF-α) in response to reactivated virus in the TG with a mouse model in which reactivation and recurrent disease are induced by UV irradiation of the cornea (18). The immunohistochemical method used allows simultaneous detection of virus antigen and cells producing the cytokines.
During primary infection, virus can spread to the CNS via the DRE, resulting in immune cell infiltration and cytokine production at this site. To investigate whether, following reactivation of virus in the TG, similar events occur in the contiguous CNS, our tissue specimens included both the TG and its attached DRE (TG/DRE).
MATERIALS AND METHODS
Reactivation model.
Specific-pathogen-free, 8-week-old female NIH/OLA inbred mice were obtained from Harlan/Olac; they were maintained as a breeding colony in the Department of Pathology and Microbiology, University of Bristol, Bristol, United Kingdom. Mice were anesthetized with 100 mg of ketamine (Parke-Davies Veterinary, Pontypool, United Kingdom) and 10 mg of xylazine (Bayer plc., Bury St. Edmonds, United Kingdom) per kg of body weight and inoculated by scarification of the left cornea with a 26-gauge needle (24) through a 5-μl drop of medium containing 104 PFU of HSV-1 strain McKrae. Control mice were inoculated in the same way with a preparation of uninfected Vero cells made in the same manner as the virus inoculum (mock inoculum). Twenty-four hours before inoculation, the animals were inoculated intraperitoneally with human serum (Chemicon International, Temecula, Calif.) containing antibodies to HSV-1. The serum was diluted in phosphate-buffered saline (PBS) to give a 50% effective dose of 8,000. Passive immunization was used to protect the eye from the severe damaging effects of HSV-1 disease (19). Only mice that survived primary infection with undamaged eyes were used for reactivation of latent infection. At least 60 days after the inoculation of virus, mice were anesthetized and placed with the left eye proptosed below a Hanovia lamp (emitting a peak of 4.02 mJ/cm2 · s at 320 nm), and the left corneas and lids were irradiated for 90 s (18).
Isolation of virus from eye washings.
Immediately before UV irradiation and on days 1 to 10 after such treatment, mice were anesthetized and eye washings were taken and put onto Vero cells for the isolation of virus (24).
Dissection and processing of tissues.
Anesthetized mice were perfused with periodate-lysine-phosphate buffer (PLP), to which a paraformaldehyde-dextrose solution was added to give a final concentration of 0.5% paraformaldehyde (26). The left TG with its DRE attached was carefully dissected from the skull and placed ventral surface down in curretting cassettes. Other tissues from uninfected mice and unimmunized animals infected on the cornea with HSV-1 were taken as positive control tissues for staining for cytokines and for HSV-1 antigens (22, 27). Tissues were processed as described previously (26). In brief, they were fixed overnight in PLP at 4°C, rapidly dehydrated, and then infiltrated under vacuum with low-temperature paraffin wax. Serial transverse 6-μm sections were cut and transferred to glass microscope slides precoated with poly-l-lysine. Slides were dried overnight at 37°C, wrapped in aluminum foil, and stored desiccated at −20°C.
Immunohistochemical staining for cytokines and HSV-1 antigens.
Sections were first stained for cytokines with monoclonal antibodies and then for virus antigens with a polyclonal antibody. The details of the staining procedure have been described previously (22, 27). The following clones of the rat anti-mouse monoclonal antibodies were used: anti-IL-2 (SB46), anti-IL-4 (BVD4-1D11), anti-IL-6 (MP5-20F3), anti-IL-10 (JES-2A5), and anti-TNF-α (MPG-XT22). A hamster anti-mouse monoclonal antibody clone RP.64 was used to stain for IFN-γ. In addition, the following isotype control monoclonal antibodies were used: immunoglobulin G1 (IgG1; R3-34), IgG2a (R35-95), and IgG2b (R35-38) raised in rats and IgG1 (G235-2356) raised in hamster. The monoclonal antibody to IL-6 was obtained from Cambridge Bioscience; the monoclonal antibodies to IL-2, IL-10, and IFN-γ were from Harlan Sera Lab, and those to IL-4 and TNF-α were from PharMingen. The chromogens were diaminobenzidine for cytokine staining, which gave a brown end product, and Vector VIP (Vector Laboratories, Peterborough, United Kingdom) for virus antigens, which gave a purple end product. Sections were counterstained with methyl green, dehydrated, and mounted in Histomount (National Diagnostics).
Positive and negative control sections were included in each staining run. Positive control sections for cytokine staining were as follows: small intestine with Peyer’s patch stained for IL-2, IL-4, and IL-10; spleen for TNF-α; and eyes 5 days after inoculation of HSV-1 for IL-6 and IFN-γ. Such tissues have been reported to be reliable sources of these cytokines (27). To check the specificity of staining for cytokines, negative control slides were prepared from sections of TG/DRE incubated with diluent instead of the primary antibody or with rat IgG1, IgG2a, or IgG2b or hamster IgG1 isotype controls appropriate to the isotype of, and at the same concentration as, the respective monoclonal antibody against the cytokine. Sections of TG from mice inoculated on the cornea with 104 PFU of HSV-1 strain McKrae 5 days previously were used as positive controls for staining for HSV-1 antigens. Similar sections, in which the primary antibody was replaced by normal rabbit serum, were used as negative controls for staining for HSV-1 antigens.
Counting of cells stained with monoclonal antibodies.
Cytokine-producing cells were identified by the presence of intracellular immunoreactivity by using ×40 or ×100 objective lenses. Extracellular staining in the absence of a coproducer cell was disregarded. In most cases, stained cells were seen in small foci which were not large enough to allow counts beyond one grid area of 0.04 mm2. Three adjacent serial sections were stained for a particular cytokine, the following three for a different cytokine, and so on. Because of technical problems, counts could not be consistently done on all three sections. Therefore, counts for each anti-cytokine antibody were done in the area of maximum staining in one grid area on each of two sections, and this method does not allow any measure of deviation from the mean.
RESULTS
Experimental protocol.
The results are a compilation of two separate experiments. On day 1 after UV irradiation, 14 latently infected mice were killed, and the TG/DRE were removed. On day 2, eight were killed; on days 3, 4, 6, 7, and 10, six mice were killed. TG/DRE samples were removed from two mice given mock inoculum at each timepoint tested. Samples obtained at each timepoint were divided into two equal groups. Serial sections of the entire samples were cut and stained for both HSV-1 antigens and cytokines (group 1 for TNF-α, IL-2, and IFN-γ and group 2 for IL-4, IL-6, and IL-10). Of the approximately 20,000 neurons per ganglion, only small numbers are known to reactivate, and the foci of infiltrating cells are small; therefore, examination of serial sections of the entire TG/DRE samples was essential in order not to miss the areas of interest. Serial sections from three latently infected animals that had not been UV irradiated were stained as a control for resident cytokine-producing cells.
Isolation of virus from eye washings.
Virus was isolated from eye washings of 3 of 32 mice on day 2, from 8 of 24 mice on day 3, from 7 of 18 mice on day 4, and from 1 of 12 mice on days 5 and 6. No virus was isolated on days 7 to 10. The eye washing from one mouse taken before UV irradiation yielded virus; this animal also shed HSV the following day. Such virus was presumably the result of spontaneous reactivation. Amounts of virus isolated from individual mice varied from 1 PFU on a single day to >100 PFU on each of three consecutive days. All animals who shed virus in eye washings had easily recognizable infiltrates of immune cells in the TG. Overall, virus was isolated from 12 (27%) of the 45 latently infected mice subjected to UV irradiation.
Virus antigen.
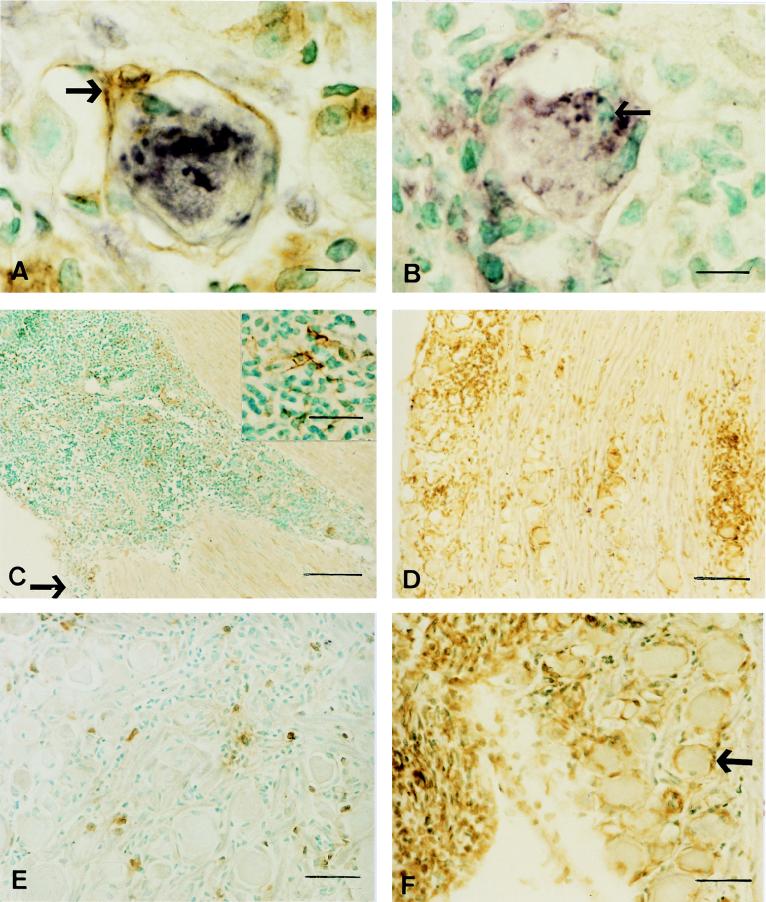
On day 2 after UV irradiation, 4 of 8 ganglia had virus antigen in the TG. This incidence declined to 2 of 6 positive ganglia on days 3 and 4 and 1 of 6 ganglia on day 6 or 7 (Table 1). No virus antigen was detected on day 1 or day 10 after irradiation or in samples from mock-inoculated mice or in the ganglia from latently infected animals not stimulated by irradiation. HSV-1 antigen was not detected in the CNS at the DRE in any sample tested. Antigen was detected in cells which were unequivocally neurons, and such cells were seen in samples obtained on days 2 to 4; their numbers in individual TG ranged from 1 to 6. At the same times, virus antigen was also seen in cells which, from their morphology, appeared to be disintegrating neurons (Fig. 1A and B) and on days 2 to 6 in cells which were probably immune cells. The majority of cells expressing virus antigen were in the mediodorsal part of the TG and unequivocally in the ophthalmic part (TG1), but a smaller number were seen in the area where the TG1 borders the maxillary part (TG2). No virus antigen-positive cells were seen in the mandibular part (TG3). The overall incidence of virus antigen induced by UV irradiation was 9 of 45 samples (20%).
TABLE 1.
Incidence of HSV-1 antigen and immune cell infiltration in the TG/DRE of mice after reactivation of latent infection
| Days after UV irradiation | No. of TG/DREs with:
|
|
|---|---|---|
| HSV antigena | Immune cell infiltration | |
| 1 | 0/13b (0) | 2/13 |
| 2 | 4/8 (0) | 6/8 |
| 3 | 2/6 (3) | 4/6 |
| 4 | 2/6 (3) | 5/6 |
| 6/7 | 1/6 (4) | 6/6 |
| 10 | 0/6 (2) | 5/6 |
The number of mice with virus present in the eye washings, either on the day the animal was killed and/or on previous day(s) after UV irradiation, is given in parentheses.
The mouse which was shedding virus before UV irradiation (spontaneous shedder) was excluded.
FIG. 1.
Immunohistochemical detection of HSV-1 antigen (purple) and cytokines (brown) in the TG of latently infected mice after reactivation of HSV-1 by UV irradiation of the corneas. (A) HSV-1 antigen in a disintegrating neuron on day 4 after irradiation. Note the TNF-α+ cells, including the satellite cell (arrow). (B) HSV-1 antigen in a disintegrating neuron on day 2, also stained for IL-2. Note that the infiltrating cells are negative for this cytokine. Some immune cells are adhering to the neuronal cell membrane (arrow). (C) Massive infiltrate in the axon-rich peripheral part of the TG and under the arachnoid membrane (arrow) on day 1. The higher-magnification inset shows that some of the infiltrating cells are TNF-α+ and have a dendritic morphology. (D) Two foci of IL-6+ cells on day 4. (E) Scattered infiltrating cells on day 10, some of which are IL-4+. (F) Large numbers of IL-6+ cells under the arachnoid membrane on day 4. Note also IL-6+ satellite cells (arrow). Bars, 100 μm (C and D), 50 μm (E and F), 25 μm (C inset), and 10 μm (A and B).
Cytokines in the TG/DRE from mock-inoculated UV-irradiated animals and unirradiated latently infected mice.
At all timepoints tested after UV irradiation, nearly all satellite cells in the TG of mock-inoculated mice stained for TNF-α. On days 1 and 2 after UV irradiation, nearly all of these cells also stained for IL-6. On subsequent days, the number of satellite cells expressing IL-6 declined to approximately 10% of the total number. At the DRE, positive staining for TNF-α and/or IL-6 was seen in cells with a dendritic morphology that lay in a line in the CNS. No other cytokines were detected in the TG/DRE.
In unirradiated latently infected mice, TNF-α expression was detected in nearly all of the satellite cells and most satellite cells also showed weak staining for IL-6. A small number of other TNF-α+ cells (<1 cell/grid area) were scattered throughout the TG. At the DRE, in addition to the stained cells seen in samples from mock-inoculated mice, there were a few TNF-α+ cells, which by their morphology appeared to be astrocytes. No other cytokines were detected.
Cytokine production in latently infected ganglia after UV irradiation to stimulate reactivation.
Immune cell infiltration of the TG/DRE was very rapid (Fig. 1B and C). On day 1 after UV irradiation, 2 of 13 samples had very small foci of infiltrating cells. By day 2, the foci were larger, and their incidence had increased to 6 of 8 (75%). These foci were often in close association with HSV-1 antigen-positive neurons, and some of the immune cells even adhered to the neuronal cell membrane (Fig. 1B). The foci contained over 200 cells per grid area. At later time points the distribution of immune cells was less dense and more widespread over the TG1, and in some samples the cells had also infiltrated the TG2. Infiltrating cells were seen in the PNS and CNS in the ophthalmic part of the DRE in one TG/DRE sample taken on day 3 and in two samples taken on day 4. The overall incidence of infiltrating immune cells in the TG/DRE induced by UV irradiation was 28 of 45 samples (62%).
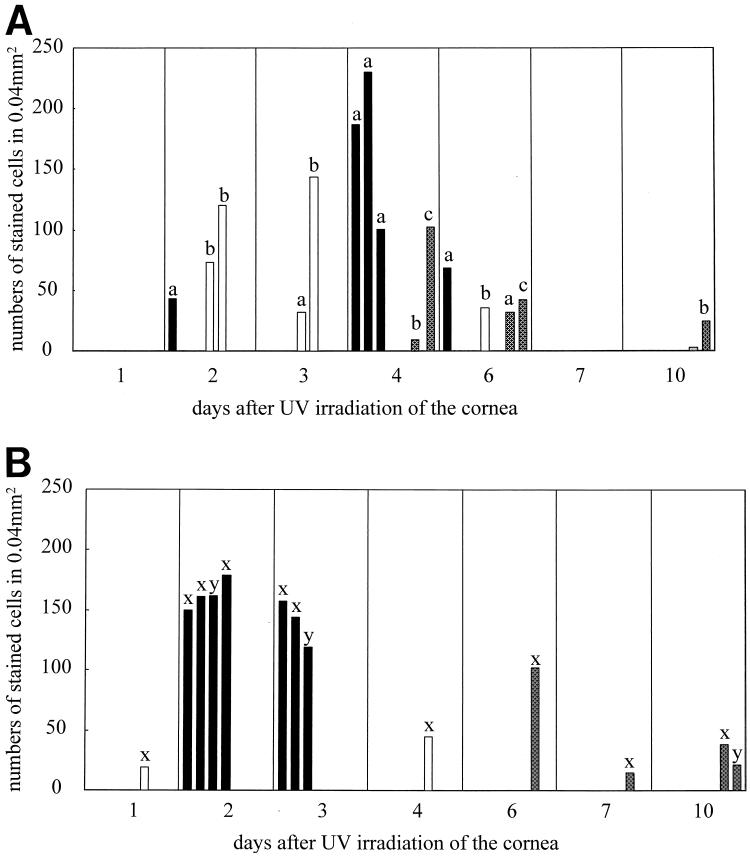
TNF-α and IL-6 were the only cytokines detected within the immune cells that formed the focal infiltrates (Fig. 1A and D). The numbers of cells expressing these cytokines were large; for example, in many foci more than 100 cells stained for IL-6 and more than 140 stained for TNF-α (Fig. 2). In addition, the intensity of the staining suggested that large amounts of these cytokines were being produced. Many of these cytokine-producing cells were nondendritic and mononuclear, suggesting that they were lymphocytes. In most cases, the foci were not large enough to allow staining of serial sections for all cytokines. Hence, in some cases the apparent absence of TNF-α or IL-6 may result from the lack of sufficient sections. At later time points, when the infiltrate had become more diffuse, the number of cells producing IL-6 and/or TNF-α (Fig. 2A and B) and the intensity of the staining declined.
FIG. 2.
Cytokine-producing cells infiltrating the TG of latently infected mice after reactivation of HSV-1 by UV irradiation of the cornea. IL-6+ (A) and TNF-α+ (B) cells were identified by immunohistochemical staining in paraffin-embedded tissue. Columns: ■, stained cells in foci with virus antigen; □, stained cells in foci without virus antigen;  , scattered stained cells in samples without virus antigen. Except for samples with scattered stained cells, each bar represents a single focus. Since some TG samples contained several foci, each of which were counted, individual animals taken at each time point are identified by the following letters: a, b, and c for samples stained for IL-6, and x and y for samples stained for TNF-α. Before irradiation, the only immune cells expressing cytokines in latently infected TG were very small numbers of TNF-α+ cells (>1 cell per grid area).
, scattered stained cells in samples without virus antigen. Except for samples with scattered stained cells, each bar represents a single focus. Since some TG samples contained several foci, each of which were counted, individual animals taken at each time point are identified by the following letters: a, b, and c for samples stained for IL-6, and x and y for samples stained for TNF-α. Before irradiation, the only immune cells expressing cytokines in latently infected TG were very small numbers of TNF-α+ cells (>1 cell per grid area).
Very small numbers of IL-4+ cells were seen in some samples taken on days 3 and 4 (one to four cells per grid area). By day 10, all of the samples had IL-4+ cells, although there was considerable variation among the samples in the numbers (ranging from 3 to 24 cells per grid area). In all cases, the IL-4+ cells were evenly scattered over TG1 (Fig. 1E). Only occasional cells expressing IL-2 or IFN-γ were detected, and these were usually in the large masses of cells under the arachnoid membrane or in the axon-rich peripheral part of the TG. No IL-10+ cells were seen.
In some TG samples, particularly those which had evidence of multiple sites of reactivation, infiltrating cells were seen on both the PNS and CNS sides of the DRE. Such cells stained for TNF-α and/or IL-6 at times when there were focal infiltrates of immune cells staining for these cytokines in the TG. In addition, there appeared to be an upregulation of expression of TNF-α and/or IL-6 in the cells with a dendritic morphology arranged linearly at the DRE in the CNS. At later time points, small numbers of IL-4+ cells were also detected in a focal area on both sides of the TG1 part of the DRE. These areas appeared to contain the axons of neuron cell bodies that lay within reactivation foci.
At all of the time points tested, as in the mock-inoculated control animals, nearly all satellite cells in TG from latently infected mice treated with UV irradiation stained for TNF-α. Their production of IL-6 was more variable.
In the majority of samples and, unlike the unirradiated controls, early after UV irradiation the satellite cells did not stain for IL-6. From day 3, in the majority of the samples, a small percentage of these cells (approximately 10%) were IL-6+; in some samples all such cells were IL-6+.
Large numbers (>200 per grid area) of infiltrating cells were also detected in the axon-rich peripheral portion of the dorsal part of the TG1 (Fig. 1C) and under the arachnoid membrane, particularly that which sheathed the lateral and dorsal surfaces of the TG1 (Fig. 1C and F). Cells at these locations were present in 4 of the 13 TG samples taken on day 1; 2 of these 4 also had small foci of infiltrating cells within the ganglion. On subsequent days, all of the TG which showed evidence of reactivation (HSV-1 antigen and/or infiltrating immune cells) had large numbers of cells in the axon-rich peripheral portion of the dorsal part of TG1 and under the arachnoid membrane. At early time points, TNF-α and IL-6 were the only cytokines detected at these sites (Fig. 1C and F), and some TG samples had large numbers of cells, with dendritic processes, that stained for TNF-α (Fig. 1C, inset). By day 7, very small numbers of IL-4+ and/or IL-2+ cells (<5 per grid area) were also detected. No infiltrating cells in the axon-rich peripheral portion of the dorsal surface of the TG1 or under the arachnoid membrane were seen in samples from mock-inoculated mice or in the ganglia from latently infected animals not stimulated by UV irradiation.
DISCUSSION
The present study extends our previous observations with this mouse model (18, 20, 21) and supports the following picture of reactivation. (i) Reactivation occurs rapidly, in some cases probably earlier than 24 h after UV irradiation. In the mouse hyperthermia model of reactivation, others have reported virus isolation from ganglia as early as 14 h (16), and we and others have detected antigen-positive neurons at 24 h after stimulation (16, 21). (ii) Neurons are the sites of reactivation and these undergo severe degenerative changes which makes their survival unlikely. (iii) Reactivation is not synchronous between TG samples or within a single TG. (iv) The majority of reactivating neurons are in the mediodorsal part of the TG1. This corresponds to the location of the neuronal cell bodies which supply sensory fibers to the cornea. (vi) UV irradiation of the corneas of mock-inoculated control mice induced no inflammatory infiltration in the TG (21). Thus, as in the previous study (21), we have interpreted both the foci of infiltrating immune cells which appear in the TG of latently infected mice and the appearance of virus antigen in the TG as evidence of reactivation after UV irradiation. Using these criteria, the incidence of reactivation in this study is high (28 of 45 animals, or 62%). (vii) The immune response in the TG is very rapid. (viii) A significant proportion, possibly 50%, of the reactivation events in the TG do not result in evidence of infection in the peripheral tissue. (vii) Spontaneous shedding of HSV-1 in the tear film is rare (in this study, virus was isolated from 1 of 46 eye washings taken prior to UV irradiation).
We reported previously that in the normal mouse TG the only cytokine detected was TNF-α and was limited, almost exclusively, to satellite cells, of which approximately 30% were TNF-α+ (22). This pattern was changed significantly after UV irradiation of the cornea of mock-inoculated mice (taken to be equivalent to the normal animals used previously), since there was transient expression of IL-6 by satellite cells and more long-term expression of TNF-α in nearly all such cells. Both of these cytokines have been demonstrated in the CNS after injury (2, 10), and upregulated immunoreactivity to IL-6 in Schwann cells of the PNS has been demonstrated after nerve damage (9). Therefore, both IL-6 and TNF-α may be involved in neuronal repair and protection, and it is likely that they have similar functions in the TG. The upregulation of IL-6 in satellite cells is also of particular interest in view of the suggested involvement of this cytokine in the induction of reactivation itself (see below).
In the latently infected animals before irradiation, the expression of cytokines by the satellite cells was slightly different since, in contrast to the 30% that were TNF-α+ in uninfected animals, nearly all such cells were TNF-α+ and most showed weak staining for IL-6. This suggests that either the presence of latent virus and/or the previous infection events in the ganglion may produce a persisting change in the pattern of cytokine expression. Moreover, in the latently infected animals, this pattern of expression did not change after UV irradiation, although there was a suggestion that, in some animals, the extent of IL-6 expression in satellite cells was decreased.
It has been suggested from studies of latently infected TG from nonimmunized mice that the persistence of lymphocytes and the cytokines they produce may be involved in the maintenance of latency (22, 8). This hypothesis appears unlikely, since we detected only low numbers of persisting immune cells (21) in the ganglia from latently infected, passively immunized mice and, of these, only very small numbers were TNF-α+. However, it is now clear that in the TG, satellite cells are a further source of cytokines, TNF-α and IL-6 in particular. Indeed, as mentioned above, in the latently infected TG, the extent of expression of these cytokines seems to be greater than normal. The possible role of cytokines from this source in mediating virus-cell interactions such as latency remains to be established.
The major routes by which immune cells infiltrate the TG in response to infection appear to be via the arachnoid membrane, which sheaths the ganglion and through channels in the axon-rich peripheral portion of the TG. Heavy immune cell infiltration of the arachnoid membrane has been seen during primary HSV-1 infection of the TG (5). In this present study, two TG samples obtained 1 day after UV irradiation that had no evidence of reactivation, as defined above, had large masses of infiltrating cells (some of which were TNF-α+) in the peripheral portion of the TG. These cells were likely to be infiltrating in response to infected neurons at very early stages of reactivation.
TNF-α and IL-6 produced by immune cells were the predominant cytokines detected in the TG after reactivation. These cytokines were seen in cells close to virus antigen-positive neurons, suggesting that they may, as proposed for primary infections (22), play a role in viral clearance. Indeed, some TNF-α+ cells had breached the satellite cell “barrier” and adhered to the membrane of the neuronal cell body. Similarly adhering cells, identified as γδ T cells, were observed early in primary infection with HSV-1 in the mouse TG (13), and there is evidence that these cells are important in restricting virus replication in the nervous system (17). In some cells, TNF-α can induce apoptosis and, if this occurs in neurons, such cell death could halt virus replication. However, apoptosis was not observed in dorsal root ganglion neurons during primary infection with HSV-2 in the mouse (15). There is no consensus on the role of TNF-α in viral infections. For example, TNF-α may have a direct antiviral effect on HSV-1 infection (6) and in the case of another virus, hepatitis B virus, gene expression is decreased (7). In contrast, in vitro studies have provided evidence that this cytokine may promote HSV-1 reactivation (25).
Macrophages and F4/80+ dendritic cells were the most likely immune cell sources of TNF-α and IL-6 when virus was being cleared during primary infection, since these were the predominant infiltrating cells at this time (22). In contrast, the foci of infiltrating cells (each with approximately 200 cells) formed after reactivation contained almost equal numbers of macrophages or F4/80+ dendritic cells and T cells (both CD4+ and CD8+) (21). Within these foci, approximately 75% of the cells stained for TNF-α and about a 50% stained for IL-6; many of these stained cells were mononuclear and lacked dendritic processes. This suggests that after reactivation, T cells are likely to be a major source of these cytokines and that some cells within these foci may be producing both cytokines. Lymphocytes at inflammatory sites may produce large amounts of IL-6, and in mice the producer cells are mainly Th2 in type. Moreover, T cells are a well-recognized source of TNF-α, and it has been suggested that CD8+ T cells may produce this cytokine during primary infection of the TG with HSV-1 (13).
Virus produced in neurons after reactivation may be transported along axons towards the CNS, as well as to the periphery. Although no HSV-1 antigen was detected in the CNS at the DRE, small foci of infiltrating immune cells were seen in some samples on both sides of the DRE. These cells were between axons derived from areas containing foci of reactivation and were only in the TG where such foci were multiple, i.e., where a relatively large amount of virus was likely to be produced. As in the TG during reactivation and in the DRE during primary infection (22), IL-6 and TNF-α were the predominant cytokines at the DRE, emphasizing again their importance in the nervous system in the immune response to HSV-1 infection.
While our observations clearly suggest that IL-6 may have an important function in the immune response to primary and reactivated infection, as previously mentioned, recent evidence has suggested that IL-6 may also have a role in the induction of reactivation itself. A very early (2 h) and transient rise in IL-6 mRNA was observed in latently infected TG in vivo after a period of brief hyperthermia (14), a stimulus known to reactivate latent virus. Moreover, there was a reduction in the incidence of induced viral shedding in the tears of latently infected mice after treatment with antibodies to IL-6 (11) and IL-6 response elements were located in the LAT region of the viral genome (12). In our study, however, all of the satellite cells in latently infected ganglia were already expressing IL-6 before irradiation. Therefore, detection of any upregulation of this expression would require methods more sensitive than immunohistochemistry. As previously mentioned, however, there was a general upregulation of IL-6 in satellite cells throughout the ganglion after UV irradiation of mock-inoculated animals.
Despite the large numbers of infiltrating T cells, IL-2+ and IFN-γ+ cells were very sparse, suggesting that a local Th1 response was unlikely. Local T-cell proliferation also appears unlikely, a strategy that may protect the nervous system from damage. Although we did not detect any IL-10, the presence of large numbers of IL-6+ cells, together with IL-4+ cells, was more characteristic of a Th2 type response. These IL-4+ cells may aid in the induction of cytotoxic T-cell differentiation and promote growth of B cells (such cells also infiltrate the TG after reactivation [21]). The continued presence of IL-6 would allow the terminal differentiation of B cells to plasma cells. Thus, in addition to the cell-mediated response to HSV-1, local antibody production may also play a role in the viral clearance after reactivation. Indeed, such an activity may explain the early observations of Stevens and Cook (23) that antiviral immunoglobulins were capable of suppressing the reactivation of HSV in ganglion explants in vitro.
In conclusion, these studies have further characterized reactivation events in the TG and have confirmed that, as in primary infections, TNF-α and IL-6 produced by both infiltrating immune cells and the resident glial cells may have a role both in viral clearance and in the protection and repair of the nervous system after the reactivation of HSV-1. The use of IL-6 and/or TNF-α-deficient mice or specific inhibitors of these cytokines will permit further investigation of the roles of these cytokines in HSV-1 infection of the nervous system.
ACKNOWLEDGMENTS
This work was supported by the Wellcome Trust and the Henry Smith’s Charity.
We thank Tanya Searle for technical assistance.
REFERENCES
- 1.Bloom D C, Dei-Rao G B, Hill J M, Stevens J G, Wagner E K. Molecular analysis of herpes simplex virus type 1 during epinephrine-induced reactivation of latently infected rabbits in vivo. J Virol. 1994;68:1283–1292. doi: 10.1128/jvi.68.3.1283-1292.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bruce A J, Boling W, Kindy M S, Peschon J, Kraemer P J, Carpenter M K, Holtsberg F W, Mattson M P. Altered neuronal and microglial responses to excitotoxic and ischemic brain injury in mice lacking TNF receptors. Nat Med. 1996;2:788–794. doi: 10.1038/nm0796-788. [DOI] [PubMed] [Google Scholar]
- 3.Cantin E M, Hinton D R, Chen J, Openshaw H. Gamma interferon expression during acute and latent nervous system infection by herpes simplex virus type 1. J Virol. 1995;69:4898–4905. doi: 10.1128/jvi.69.8.4898-4905.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carr D J J, Noisakran S, Halford W P, Lukacs N, Asensio V, Campbell I L. Cytokine and chemokine production in HSV-1 latently infected trigeminal ganglion cell cultures: effects of hyperthermic stress. J Neuroimmunol. 1998;85:111–121. doi: 10.1016/s0165-5728(97)00206-3. [DOI] [PubMed] [Google Scholar]
- 5.Cunningham E T, Stalder A K, Sanna P P, Liu S S, Bloom F E, Howes E L, Campbell I L, Margolis T P. Distribution of tumor necrosis factor receptor messenger RNA in normal and herpes simplex virus-infected trigeminal ganglia in the mouse. Brain Res. 1997;758:99–106. doi: 10.1016/s0006-8993(97)00169-8. [DOI] [PubMed] [Google Scholar]
- 6.Feduchi E, Alonso M A, Carrasco L. Human gamma interferon and tumor necrosis factor exert a synergistic blockade on the replication of herpes simplex virus. J Virol. 1989;63:1354–1359. doi: 10.1128/jvi.63.3.1354-1359.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guidotti L G, Chisari F V. To kill or to cure: options in host defense against viral infection. Curr Opin Immunol. 1996;8:478–483. doi: 10.1016/s0952-7915(96)80034-3. [DOI] [PubMed] [Google Scholar]
- 8.Halford W P, Gebhardt B M, Carr D J J. Persistent cytokine expression in trigeminal ganglion latently infected with herpes simplex virus type 1. J Immunol. 1996;157:3542–3549. [PubMed] [Google Scholar]
- 9.Hirota H, Kiyama H, Kishimoto T, Taga T. Accelerated nerve regeneration in mice by upregulated expression of interleukin (IL)-6 and IL-6 receptor after trauma. J Exp Med. 1996;183:2627–2634. doi: 10.1084/jem.183.6.2627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Klein M A, Möller J C, Jones L L, Bluethmann H, Kreutzberg G W, Raivich G. Impaired neuroglial activation in interleukin-6 deficient mice. Glia. 1997;19:227–233. doi: 10.1002/(sici)1098-1136(199703)19:3<227::aid-glia5>3.0.co;2-w. [DOI] [PubMed] [Google Scholar]
- 11.Kriesel J D, Gebhardt B M, Hill J M, Maulden S A, Hwang I P, Clinch T E, Cao X, Spruance S L, Araneo B A. Anti-interleukin-6 antibodies inhibit herpes simplex virus reactivation. J Infect Dis. 1997;175:821–827. doi: 10.1086/513977. [DOI] [PubMed] [Google Scholar]
- 12.Kriesel J D, Ricigliano J, Spruance S L, Garza H H, Hill J M. Neuronal reactivation of herpes simplex virus may involve interleukin 6. J Neurovirol. 1997;3:441–448. doi: 10.3109/13550289709031190. [DOI] [PubMed] [Google Scholar]
- 13.Liu T, Tang Q, Hendricks R L. Inflammatory infiltration of the trigeminal ganglion after herpes simplex virus type 1 corneal infection. J Virol. 1996;70:264–271. doi: 10.1128/jvi.70.1.264-271.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Noisakran S, Halford W P, Veress L, Carr D J J. Role of the hypothalamic pituitary adrenal axis and IL-6 in stress-induced reactivation of latent herpes simplex virus type 1. J Immunol. 1998;160:5441–5447. [PubMed] [Google Scholar]
- 15.Ozaki N, Sugiura Y, Yamamoto M, Yokoya S, Wanaka A, Nishiyama Y. Apoptosis induced in the spinal cord and dorsal root ganglion by infection of herpes simplex virus type 2 in the mouse. Neurosci Lett. 1997;228:99–102. doi: 10.1016/s0304-3940(97)00364-9. [DOI] [PubMed] [Google Scholar]
- 16.Sawtell N M, Thompson R L. Rapid in vivo reactivation of herpes simplex virus in latently infected murine ganglionic neurons after transient hyperthermia. J Virol. 1992;66:2150–2156. doi: 10.1128/jvi.66.4.2150-2156.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sciammas R, Kodukula P, Tang Q, Hendricks R L, Bluestone J A. T cell receptor-γ/δ cells protect mice from herpes simplex virus type 1-induced lethal encephalitis. J Exp Med. 1997;185:1969–1975. doi: 10.1084/jem.185.11.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shimeld C, Hill T, Blyth B, Easty D. An improved model of recurrent eye disease in mice. Curr Eye Res. 1989;8:1193–1205. doi: 10.3109/02713688909000044. [DOI] [PubMed] [Google Scholar]
- 19.Shimeld C, Hill T J, Blyth W A, Easty D L. Passive immunization protects the mouse eye from damage after herpes simplex virus infection by limiting spread of virus in the nervous system. J Gen Virol. 1990;71:681–687. doi: 10.1099/0022-1317-71-3-681. [DOI] [PubMed] [Google Scholar]
- 20.Shimeld C, Whiteland J L, Nicholls S M, Easty D L, Hill T J. Immune cell infiltration in corneas of mice with recurrent herpes simplex virus disease. J Gen Virol. 1996;77:977–985. doi: 10.1099/0022-1317-77-5-977. [DOI] [PubMed] [Google Scholar]
- 21.Shimeld C, Whiteland J L, Williams N A, Easty D L, Hill T J. Reactivation of herpes simplex virus type 1 in the mouse: an in vivo study of virus antigen and immune cell infiltration. J Gen Virol. 1996;77:2583–2590. doi: 10.1099/0022-1317-77-10-2583. [DOI] [PubMed] [Google Scholar]
- 22.Shimeld C, Whiteland J L, Williams N A, Easty D L, Hill T J. Cytokine production in the nervous system of mice during acute and latent infection with herpes simplex virus type 1. J Gen Virol. 1997;78:3317–3325. doi: 10.1099/0022-1317-78-12-3317. [DOI] [PubMed] [Google Scholar]
- 23.Stevens J G, Cook M L. Maintenance of latent herpetic infection: an apparent role for anti-viral IgG. J Immunol. 1974;113:1685–1693. [PubMed] [Google Scholar]
- 24.Tullo A B, Shimeld C, Blyth W A, Hill T J, Easty D L. Ocular infection with herpes simplex virus in nonimmune and immune mice. Arch Ophthalmol. 1983;101:961–964. doi: 10.1001/archopht.1983.01040010961023. [DOI] [PubMed] [Google Scholar]
- 25.Walev I, Podlech J, Falke D. Enhancement by TNFα of reactivation and replication of latent herpes simplex virus from trigeminal ganglia of mice. Arch Virol. 1995;140:987–992. doi: 10.1007/BF01315409. [DOI] [PubMed] [Google Scholar]
- 26.Whiteland J L, Nicholls S M, Shimeld C, Easty D L, Williams N A, Hill T J. Immunohistochemical detection of T-cell subsets and other leukocytes in paraffin-embedded rat and mouse tissues with monoclonal antibodies. J Histochem Cytochem. 1995;43:313–320. doi: 10.1177/43.3.7868861. [DOI] [PubMed] [Google Scholar]
- 27.Whiteland J L, Shimeld C, Nicholls S M, Easty D L, Williams N A, Hill T J. Immunohistochemical detection of cytokines in paraffin-embedded mouse tissues. J Immunol Methods. 1997;210:103–108. doi: 10.1016/s0022-1759(97)00167-1. [DOI] [PubMed] [Google Scholar]