Abstract
Abscisic acid (ABA) signaling is critical for seed germination and abiotic stress responses in terrestrial plants. Pre-mRNA splicing is known to regulate ABA signaling. However, the involvement of canonical spliceosomal components in regulating ABA signaling is poorly understood. Here, we show that the spliceosome component Sm core protein SmEb plays an important role in ABA signaling. SmEb expression is up-regulated by ABA treatment, and analysis of Arabidopsis smeb mutant plants suggest that SmEb modulates the alternative splicing of the ABA signaling component HAB1 by enhancing the HAB1.1 splicing variant while repressing HAB1.2. Overexpression of HAB1.1 but not HAB1.2 rescues the ABA-hypersensitive phenotype of smeb mutants. Mutations in the transcription factor ABI3, 4, or 5 also reduce the ABA hypersensitivity of smeb mutants during seed germination. Our results show that the spliceosomal component SmEb plays an important role in ABA regulation of seed germination and early seedling development.
Keywords: ABA signaling, Alternative splicing, Cotyledon greening, HAB1, SmEb
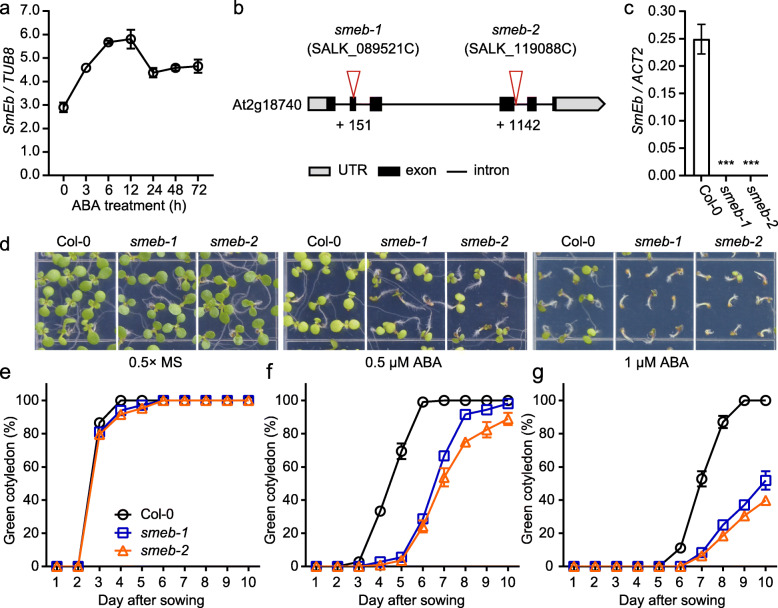
Alternative splicing occurs in many genes involved in diverse processes during plant development and responses to abiotic stresses (Staiger and Brown 2013). mRNA splicing is mainly mediated by the spliceosome consisting of five conserved small nuclear ribonucleoproteins (snRNPs) in eukaryotes. The seven Sm core proteins (B/B′, D1, D2, D3, E, F and G) are common components of the five snRNPs and are essential for their assembly (Reddy 2007; Wahl et al. 2009). Among the Sm core proteins, SmEb has been reported to positively regulate cold stress response by modulating pre-mRNA splicing (Capovilla et al. 2018). In this study, we found that the transcript level of SmEb was induced by ABA treatment (Fig. 1a), which suggests that SmEb might be involved in ABA response. To uncover the molecular mechanism underlying SmEb-modulated ABA response, we first characterized two T-DNA insertion mutant alleles of SmEb, designated smeb-1 and smeb-2. The smeb-1 and smeb-2 mutants have T-DNA insertions in the second exon and fourth intron of the At2g18740 gene, respectively (Fig. 1b). Quantitative reverse transcription-PCR (qRT-PCR) analysis determined that the transcript level of SmEb was nearly undetectable in smeb-1 and smeb-2 mutants (Fig. 1c). Phenotyping of smeb-1 and smeb-2 showed that these two mutants were much more sensitive to ABA treatment than the Col-0 wild type plants in cotyledon greening during seed germination (Fig. 1d). Under normal growth conditions, the rates of cotyledon greening were not different between smeb mutants and wild type plants (Fig. 1e). However, with 0.5 μM ABA supplemented in the medium, the emerged seedlings with green cotyledons in smeb-1 and smeb-2 mutants were only approximately 67% and 54%, in contrast to the near 100% green cotyledons in the wild type at the seventh day of the treatment (Fig. 1f). In addition, compared with the ~ 52% green cotyledon rate in the wild type, only approximately 8.3% of smeb-1 and 6.5% of smeb-2 mutant cotyledons turned green after 1 μM ABA treatment for 7 days (Fig. 1g). Together, these results indicate that SmEb is a positive regulator of ABA responses in Arabidopsis.
Fig. 1.
SmEb is involved in ABA responses during seed germination and cotyledon greening. a The transcription levels of SmEb in 7-day-old wild-type (Col-0) seedlings after exogenous ABA treatment for 0, 3, 6, 12, 24, 48, or 72 h. The transcript levels of TUB 8 were used as an internal control. b Schematic diagram showing the T-DNA insertions in the smeb mutants. c Quantitative RT-PCR analysis of SmEb transcript levels in Col-0, smeb-1, and smeb-2 mutant plants. d 7-day-old seedlings of Col-0, smeb-1, and smeb-2 grown in 0.5× MS medium containing 0, 0.5, and 1 μM ABA. e-g Percentages of green cotyledon of Col-0, smeb-1, and smeb-2 after seed sowing for indicated days in 0.5× MS medium with different concentrations of ABA. Error bars represent the ± SD (n = 3 biological repeats). ***P < 0.001, Student’s t-test
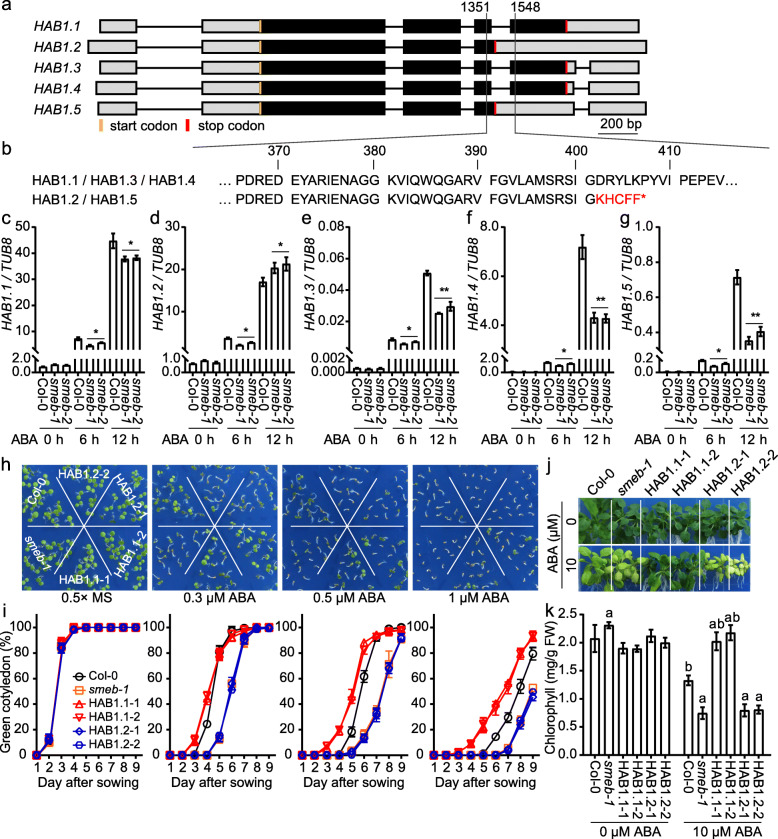
The core ABA signaling pathway is initiated by ABA binding to the PYR/PYL/RCAR receptors, which then interact with and inhibit the type 2C protein phosphatases (PP2Cs) co-receptors such as HYPERSENSITIVE TO ABA1 (HAB1) (Ren et al. 2017; Gong et al. 2020). HAB1 serves as a negative regulator for ABA signaling in seed germination and cotyledon greening, and previous studies have shown that the transcriptional up-regulation of HAB1 is critical for ABA signaling (Melcher et al. 2009; Wang et al. 2018a). As a result, suppression of the accumulation of HAB1 transcripts leads to the activation of ABA signal transduction in plants. Recent reports have shown that splicing factors play critical roles in plant response to ABA through regulating the splicing of HAB1 transcripts (Wang et al. 2015; Zhan et al. 2015; Bi et al. 2021). However, it is unclear whether the canonical spliceosomal component SmEb may contribute to the regulation of HAB1 pre-mRNA splicing and ABA responses. To determine the role of SmEb in the regulation of ABA signaling, we first examined the transcripts of HAB1 by using RT-PCR and Sanger sequencing. As shown in Fig. 2a, alternatively spliced forms of HAB1 transcripts, HAB1.1 to 1.5, were identified under normal and ABA treatment conditions. Only two types of HAB1 polypeptides were encoded by these five spliced forms. The HAB1.1, HAB1.3, and HAB1.4 transcripts are translated into full-length HAB1 protein, named as HAB1.1, while the HAB1.2 and HAB1.5 transcripts produce a truncated HAB1, designated as HAB1.2 (Fig. 2b). Under normal growth conditions, the transcript levels of the five HAB1 spliced forms showed no difference between smeb mutants and Col-0 wild type plants (Fig. 2c - g). However, the transcript levels of all HAB1 variants in smeb mutants were significantly reduced after ABA treatment for 6 h when compared with those of the Col-0 wild type. After 12 h of ABA treatment, the accumulation of HAB1.1, 1.3, 1.4, and 1.5 transcripts were clearly lower in smeb mutants than in Col-0 wild type, whereas the transcript level of HAB1.2 was higher in smeb mutants than in Col-0 wild type plants (Fig. 2c - g). Since the expression level of HAB1.3 is very low compared with the other four variants (Fig. 2e), and the HAB1.1 and HAB1.2 proteins function antagonistically (Wang et al. 2015), it is likely that the reduction of HAB1.1 and HAB1.4 transcripts and thus the decreased level of HAB1.1 protein contributes to the ABA hypersensitive phenotype of smeb mutants.
Fig. 2.
SmEb modulates the alternative splicing of HAB1 in response to ABA. a Different alternative splicing variants of HAB1 as determined by sequence analysis of cloned PCR products. The lines between boxes indicate introns. The grey and black boxes represent the UTR and exon, respectively. b Amino acid sequence resulting from alternative splicing and the formation of a full-length and truncated HAB1 proteins in the close-up view. HAB1.1, HAB1.3 and HAB1.5 encode the full-length HAB1 protein, while the HAB1.2 and HAB1.4 encode the truncated HAB1 protein. c-g Relative transcript levels of different alternative splicing variants of HAB1 in Col-0, smeb-1 and smeb-2 plants subjected to 50 μM ABA for 0, 6, or 12 h, as determined by qRT-PCR analysis. Values are mean ± SD (n = 3), * P < 0.05, ** P < 0.01, Student’s t-test. h Cotyledon greening assay of Col-0, smeb-1, HAB1.1-1, HAB1.1-2, HAB1.2-1, and HAB1.2-2 after seed sowing for 6 days in 0.5× MS medium containing 0, 0.3, 0.5, or 1 μM ABA. HAB1.1-1 and HAB1.1-2, two independent lines of smeb-1 expressing 35S:HAB1.1-3 × FLAG transgene. HAB1.2-1 and HAB1.2-2, two independent lines of smeb-1 expressing 35S:HAB1.2-3 × FLAG transgene. i Percentages of green cotyledon of Col-0, smeb-1, HAB1.1-1, HAB1.1-2, HAB1.2-1, and HAB1.2-2 after seed sowing for indicated days in 0.5× MS medium supplementing 0, 0.3, 0.5, or 1 μM ABA. Error bars represent ± SD (n = 6). j Seven-day-old Col-0, smeb-1, HAB1.1-1, HAB1.1-2, HAB1.2-1, and HAB1.2-2 seedlings grown in 0.5× MS medium were transferred to 0.5× MS medium with or without 10 μM ABA and grown for an additional 3 weeks. k The chlorophyll content of rosette leaves of Col-0, smeb-1, HAB1.1-1, HAB1.1-2, HAB1.2-1, and HAB1.2-2 plants shown in (j). Values are mean ± SD (n = 6). The letters a and b above the columns indicate a significant difference relative to Col-0 and smeb-1 mutant, respectively (P < 0.05, Student’s t-test)
To test the hypothesis that the reduction of HAB1.1 protein is responsible for the ABA hypersensitive response phenotype of smeb mutants, we expressed the full-length cDNAs of HAB1.1 and HAB1.2 driven by the CaMV 35S promoter in smeb-1 mutant plants. Two independent homozygous lines of 35S:HAB1.1-3 × FLAG/smeb-1 (designated as HAB1.1-1 and HAB1.1-2) and 35S:HAB1.2-3 × FLAG/smeb-1 (named as HAB1.2-1 and HAB1.2-2) were identified, and qRT-PCR analysis showed that the expression levels of HAB1.1 and HAB1.2 in the overexpression lines are approximately six- to nine-fold higher than the wild type controls. Cotyledon greening assays showed that overexpression of HAB1.1 but not HAB1.2 could fully rescue the hypersensitive phenotype of semb-1 in response to ABA treatment (Fig. 2h and i). These results further support that HAB1 is a downstream target of SmEb and decreased accumulation of HAB1.1 protein is responsible for the smeb mutant phenotype. The transgenic seedlings were further tested on media supplemented with 10 μM ABA. The results showed that the ectopic expression of HAB1.1 but not HAB1.2 also suppressed the chlorosis phenotype of smeb-1 in response to ABA treatment (Fig. 2j and k). These results indicate that the HAB1.1 but not the HAB1.2 spliced form is the functional variant in SmEb-modulated ABA response, and it is very likely that the reduction of HAB1.1 protein contributes to the ABA-hypersensitive phenotype of smeb-1 mutant plants.
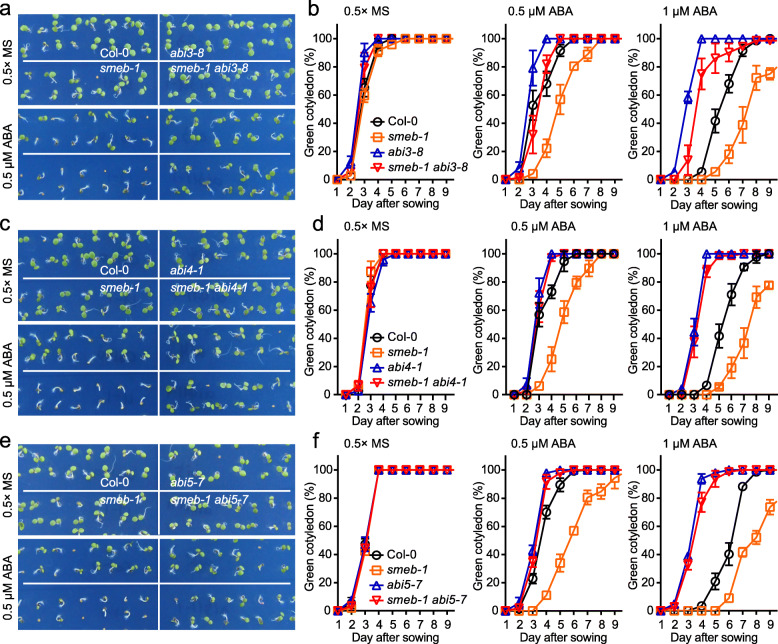
Inhibition of the full length HAB1 protein results in the activation of SnRK2 protein kinases, leading to the activation of downstream transcription factors such as ABA INSENSITIVE (ABI) 3, ABI4, and ABI5 in the ABA signaling pathway (Feng et al. 2014; Zhu 2016). Although ABI3, ABI4, and ABI5 belong to different transcription factor families, they are all positive regulators in plant ABA responses (Skubacz et al. 2016). The genetic interaction between SmEb and ABI3, ABI4, and ABI5 in modulating seed germination and cotyledon greening in response to ABA treatment was studied by generating smeb-1 abi3-8, smeb-1 abi4-1, and smeb-1 abi5-7 double mutant lines. Cotyledon greening assays revealed that the abi3, abi4, or abi5 mutation could rescue the ABA-hypersensitive phenotype of smeb-1 mutant plants (Fig. 3). However, among the three abi mutations, abi3 showed weaker suppression of the smeb-1 mutant phenotype (Fig. 3), suggesting that the effect of smeb mutation on ABA response is partially independent of ABI3. Taken together, it is likely that the smeb-1 mutation causes reduced production of HAB1.1 and/or other PP2Cs protein, which leads to enhanced ABA signaling and activation of ABA responses that are dependent on the downstream transcription factors ABI3, 4, and 5, resulting in ABA-hypersensitivity of the smeb-1 mutant.
Fig. 3.
The abi3, abi4 and abi5 mutations suppress the ABA-hypersensitivity of smeb mutant. a Cotyledon greening assay of Col-0, smeb-1, abi3-8, and smeb-1 abi3-8 after seed sowing for 5 days in 0.5× MS medium with or without 0.5 μM ABA. b The percentage of green cotyledon of Col-0, smeb-1, abi3-8, and smeb-1 abi3-8 in response to ABA. Green cotyledons were recorded after seed sowing on medium supplemented with 0, 0.5, or 1 μM ABA. Error bars represent ± SD (n = 6). c Cotyledon greening assay of Col-0, smeb-1, abi4-1, and smeb-1 abi4-1 after seed sowing for 5 days in 0.5× MS medium with or without 0.5 μM ABA. d The percentage of green cotyledon of Col-0, smeb-1, abi4-1, and smeb-1 abi4-1 in response to ABA. Green cotyledons were recorded after seed sowing on medium containing 0, 0.5, or 1 μM ABA. Error bars represent ± SD (n = 6). e Cotyledon greening of Col-0, smeb-1, abi5-7, and smeb-1 abi5-7 after seed sowing for 5 days in 0.5× MS medium with or without 0.5 μM ABA. f The percentage of green cotyledon of Col-0, smeb-1, abi5-7, and smeb-1 abi5-7 after seed sowing for indicated days in 0.5× MS medium supplementing 0, 0.5, or 1 μM ABA. Error bars represent ± SD (n = 6)
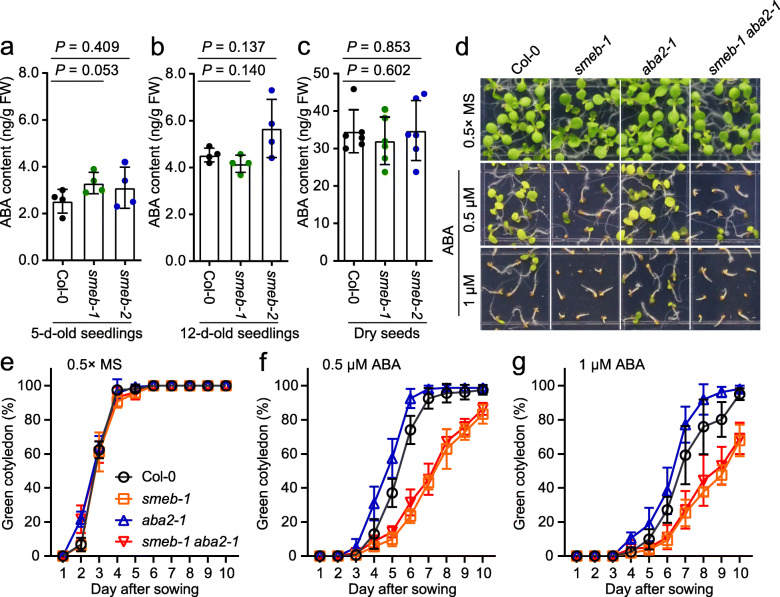
In addition to ABA signaling, ABA biosynthesis plays a role in controlling plant ABA responses (Nakashima and Yamaguchi-Shinozaki 2013; Wang et al. 2018a). The de novo ABA biosynthesis pathway is well known in Arabidopsis, and the xanthoxin dehydrogenase encoded by ABA DEFICIENT 2 (ABA2) is a critical enzyme for ABA biosynthesis (Seo and Koshiba 2002). To determine whether the ABA-hypersensitive phenotype of smeb mutants may be due to enhanced ABA accumulation, we measured ABA contents, and found that the two smeb mutants and Col-0 wild type had comparable cellular ABA levels in seedlings and dry seeds (Fig. 4a - c). This result indicates that the smeb mutations do not affect ABA biosynthesis. We further tested the genetic interaction between aba2 and smeb-1 by analyzing the double mutant smeb-1 aba2-1. The aba2 mutant is hyposensitive to ABA in seed germination (Lin et al. 2007). The results indicated that the aba2 mutation could not rescue the ABA-hypersensitive phenotype of smeb-1 mutant in cotyledon greening during seed germination (Fig. 4d - g). These results further support that SmEb modulates ABA responses by regulating ABA signaling rather than ABA accumulation.
Fig. 4.
Effect of ABA biosynthesis on ABA sensitivity of smeb-1 mutant. a, b The ABA contents were measured in seedlings of Col-0, smeb-1, and smeb-2 seedlings under normal conditions after seed sowing for 5 (a) and 12 (b) days, respectively. Values are mean ± SD (n = 4). c The ABA contents in dry seeds of Col-0, smeb-1, and smeb-2. Error bars represent ± SD (n = 6), P values are determined by Student’s t-test. d Cotyledon greening assay of Col-0, smeb-1, aba2-1 and smeb-1 aba2-1 after seed sowing for 7 days in 0.5× MS medium containing 0, 0.5, or 1 μM ABA. e-g The percentage of green cotyledon of Col-0, smeb-1, aba2-1 and smeb-1 aba2-1 after seed sowing for indicated days in 0.5× MS medium supplementing 0, 0.5, or 1 μM ABA. Error bars represent ± SD (n = 6)
In conclusion, we have identified that SmEb, a core spliceosome component involved in pre-mRNA splicing, modulates the alternative splicing of HAB1 that encodes an ABA co-receptor critical for ABA responses in Arabidopsis. Our results suggest that this regulatory mechanism helps to fine-tune ABA signaling during seed maturation, germination and cotyledon greening by maintaining proper levels of HAB1 splicing variants in response to changes in endogenous ABA contents. When ABA concentration is low, SmEb is not critical for HAB1 pre-mRNA splicing. When ABA concentration increases, SmEb expression is up-regulated, and SmEb becomes critical for maintaining a proper ratio of the two splicing variants that encode antagonistic HAB1 proteins, HAB1.1 and HAB1.2. Our findings highlight the crucial roles of mRNA splicing control of key genes in plant hormonal signaling and stress responses. Future work will determine how increased SmEb may affect the splicing of HAB1 pre-mRNAs in response to ABA treatment.
Materials and methods
Plant materials and growth conditions
All Arabidopsis (Arabidopsis thaliana) plants used in this study are in the Columbia-0 background. The T-DNA insertion mutants, including smeb-1 (SALK_089521C) and smeb-2 (SALK_119088C), were obtained from the Arabidopsis Biological Resource Center (ABRC; The Ohio State University, Columbus, OH, USA). The abi3-8, abi4-1, abi5-7, and aba2-1 mutants were collected from previous studies. The double mutants, including smeb-1 abi3-8, smeb-1 abi4-1, smeb-1 abi5-7, and smeb-1 aba2-1, were generated by genetic crossing and subsequent PCR-based genotyping in the F2 population. Seeds were surface-sterilized and stratified at 4 °C for 2 days, and then sown in 0.5× Murashige and Skoog (MS) medium plates and grown at 22 °C under 16 h light / 8 h dark photoperiod.
To construct the HAB1.1 and HAB1.2 transgenic plants, the coding sequence (CDS) of two HAB1 splicing isoforms, HAB1.1 and HAB1.2, were amplified and cloned into pCambia1305 binary vector (Hong et al. 2020) to obtain the 35S:HAB1.1-3 × FLAG and 35S:HAB1.2-3 × FLAG constructs, respectively. These two constructs were then respectively introduced into smeb-1 mutant plants by using Agrobacterium-mediated floral dip method (Clough and Bent 1998). The seeds of T3 homozygous transgenic plants were identified for further analysis.
Physiological phenotype assays
For seed germination assay, Arabidopsis seeds were surface-sterilized and stratified at 4 °C for 2 days before sowing on 0.5× MS medium plates containing indicated concentrations of ABA. The plates were kept in a growth chamber at 22 °C under 16 h light / 8 h dark conditions. The percentages of green cotyledon were calculated with three to six replicates for indicated days after sowing. For analysis of ABA sensitivity at post-germination stage, seven-day-old seedlings of Col-0, smeb-1 and transgenic plants grown in 0.5× MS plates were transferred to 0.5× MS medium with or without 10 μM ABA for additional 3 weeks, and the chlorophyll content were then determined in rosette leaves. The average content of total chlorophyll was calculated from six independent experiments (n = 3 plants).
Quantification of HAB1 splice variants
The transcripts of HAB1 gene were amplified by RT-PCR from the cDNA of 7-day-old Col-0 and smeb mutant seedlings after 50 μM ABA treatment for 12 h. The PCR products with three bands were cloned into the vector using pMD™18-T Vector Cloning Kit (TaKaRa), followed by Sanger sequencing. The HAB1 alternative splicing variants were distinguished in qRT-PCR by using primer pairs that only amplify one of the five variants, which were designed according to the last three exons and 3’UTR of At1g72770. The primer pairs used to determine the transcript levels of the five splicing variants of HAB1 by qRT-PCR are as follows. 5′-TCTAGGTCCATCGGTGACAGATATCT-3′ as forward primer of HAB1.1, 1.3, and 1.4; 5′-GCACATCACACAAACGTGGCTTG-3′ as forward primer of HAB1.2 and 1.5; 5′-TACGAAAACTCGAAACTTACCCCCA-3′ as reverse primer of HAB1.1; 5′-ACCTGTCGAAATTAGATCCTTAACGATCT-3′ as reverse primer of HAB1.2; 5′-TTTTCTTTATTTTTTTTACCCCCAGT-3′ as reverse primer of HAB1.3; and 5′-TTCTTTATTTTTTCTCCCTTCCCCAGTA-3′ as reverse primer of HAB1.4 and 1.5.
Determination of chlorophyll and ABA content
The chlorophyll and ABA contents were determined as described in previous study (Wang et al. 2018,b). Briefly, to quantify chlorophyll contents, the chlorophyll was extracted from 100 mg rosette leaves with 0.6 mL 80% (v/v) acetone buffer. The extract was then centrifuged for 5 min at 4 °C, and the supernatant was collected and the absorbance of chlorophyll at wavelengths 645 and 663 nm was measured using NanoDrop spectrophotometer 2000C (Thermo Scientific). To determinate ABA contents, endogenous ABA was extracted from 50 mg fresh leaf tissue of each sample using 0.5 mL homogeneous buffer (70% methanol, 0.1% formic acid). 2 ng ABA-d6 (Olchemim, Olomouc, Czech) was added to the extracts as an internal standard. The mixture was diluted 2 times with deionized water, and the ABA content of 50 μL dilution of each sample was determined by the UPLC-Triple TOF 5600+ system (Sciex, Concord, Canada).
Quantification of gene expression
Total RNAs were extracted from rosette leaves of 7-day-old seedlings grown in 0.5× MS liquid medium with or without 50 μM ABA treatment using Quick RNA Isolation Kit (Huayueyang). Reverse transcription was carried out using iScript cDNA synthesis kit (Bio-RAD), followed by quantitative PCR on a CFX96™ Real-Time system (BIO-RAD) with AceQ qPCR SYBR Green Master Mix (Vazyme Biotech co., ltd). The data of expression profiles were analyzed and illustrated by temporal changes using the Prism 7 (GraphPad Software). TUB8 was used as an internal control.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant 32000206 to Z.W.); the Youth Innovation Promotion Association (2020273 to Z.W.) of the Chinese Academy of Sciences.
Accession numbers
Sequence data from this article could be found on the website of Arabidopsis Information Resource (www.arabidopsis.org) under the following accession numbers: SmEb, At2g18740; HAB1, At1g72770; ABI3, At3g24650; ABI4, At2g40220; ABI5, At2g36270; TUB8, At5g23860.
Abbreviations
- ABA
Abscisic acid
- T-DNA
Transfer DNA
- qRT-PCR
Quantitative reverse transcription-PCR
- PP2C
Type 2C protein phosphatases
- HAB1
HYPERSENSITIVE TO ABA1
- ABI3
ABA INSENSITIVE 3
- ABI4
ABA INSENSITIVE 4
- ABI5
ABA INSENSITIVE 5
- ABA2
ABA DEFICIENT 2
- MS
Murashige and Skoog
Authors’ contributions
H.S. and Z.W. designed the experiments; Y.H., H.S., and Z.W. analyzed the data; Y.H., Y.C., J.Y., and Z.W. performed experiments; H.S. and Z.W. drafted the article; H.S., J.-K.Z., and Z.W. revised and finalized the manuscript. The author(s) read and approved the final manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (grant 32000206 to Z.W.); the Youth Innovation Promotion Association (2020273 to Z.W.) of the Chinese Academy of Sciences.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration
Competing interests
Author H.Z. and J.-K.Z. are members of the Editorial Board, and were not involved in the journal's review of, or decisions related to, this manuscript.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yechun Hong and Juanjuan Yao contributed equally to this work.
References
- Bi Y, Deng ZP, Ni WM, Shrestha R, Savage D, Hartwig T, Patil S, Hong SH, Zhang ZZ, Oses-Prieto JA, Li KH, Quail PH, Burlingame AL, Xu SL, Wang ZY. Arabidopsis ACINUS is O-glycosylated and regulates transcription and alternative splicing of regulators of reproductive transitions. Nat Commun. 2021;12(1):945. doi: 10.1038/s41467-021-20929-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capovilla G, Delhomme N, Collani S, Shutava I, Bezrukov I, Symeonidi E, Amorim MD, Laubinger S, Schmid M. PORCUPINE regulates development in response to temperature through alternative splicing. Nat Plants. 2018;4(8):534–539. doi: 10.1038/s41477-018-0176-z. [DOI] [PubMed] [Google Scholar]
- Clough SJ, Bent AF (1998) Floral dip: a simplified method for agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16(6):735–743. 10.1046/j.1365-313x.1998.00343.x [DOI] [PubMed]
- Feng CZ, Chen Y, Wang C, Kong YH, Wu WH, Chen YF. Arabidopsis RAV1 transcription factor, phosphorylated by SnRK2 kinases, regulates the expression of ABI3, ABI4, and ABI5 during seed germination and early seedling development. Plant J. 2014;80(4):654–668. doi: 10.1111/tpj.12670. [DOI] [PubMed] [Google Scholar]
- Gong ZZ, Xiong LM, Shi HZ, Yang SH, Herrera-Estrella LR, Xu GH, Chao DY, Li JR, Wang PY, Qin F, Li JJ, Ding YL, Shi YT, Wang Y, Yang YQ, Guo Y, Zhu JK. Plant abiotic stress response and nutrient use efficiency. Sci China-Life Sci. 2020;63(5):635–674. doi: 10.1007/s11427-020-1683-x. [DOI] [PubMed] [Google Scholar]
- Hong YC, Wang Z, Shi HZ, Yao JJ, Liu X, Wang FX, Zeng L, Xie Z, Zhu JK. Reciprocal regulation between nicotinamide adenine dinucleotide metabolism and abscisic acid and stress response pathways in Arabidopsis. PLoS Genet. 2020;16(6):e1008892. doi: 10.1371/journal.pgen.1008892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin PC, Hwang SG, Endo A, Okamoto M, Koshiba T, Cheng WH. Ectopic expression of ABSCISIC ACID 2/GLUCOSE INSENSITIVE 1 in Arabidopsis promotes seed dormancy and stress tolerance. Plant Physiol. 2007;143(2):745–758. doi: 10.1104/pp.106.084103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melcher K, Ng LM, Zhou XE, Soon FF, Xu Y, Suino-Powell KM, Park SY, Weiner JJ, Fujii H, Chinnusamy V, Kovach A, Li J, Wang YH, Li JY, Peterson FC, Jensen DR, Yong EL, Volkman BF, Cutler SR, Zhu JK, Xu HE. A gate-latch-lock mechanism for hormone signalling by abscisic acid receptors. Nature. 2009;462(7273):602–U672. doi: 10.1038/nature08613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakashima K, Yamaguchi-Shinozaki K. ABA signaling in stress-response and seed development. Plant Cell Rep. 2013;32(7):959–970. doi: 10.1007/s00299-013-1418-1. [DOI] [PubMed] [Google Scholar]
- Reddy ASN. Alternative splicing of pre-messenger RNAs in plants in the genomic era. Annu Rev Plant Biol. 2007;58(1):267–294. doi: 10.1146/annurev.arplant.58.032806.103754. [DOI] [PubMed] [Google Scholar]
- Ren ZZ, Wang Z, Zhou XE, Shi HZ, Hong YC, Cao MJ, Chan ZL, Liu X, Xu HE, Zhu JK. Structure determination and activity manipulation of the turfgrass ABA receptor FePYR1. Sci Rep. 2017;7(1):14022. doi: 10.1038/s41598-017-14101-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seo M, Koshiba T. Complex regulation of ABA biosynthesis in plants. Trends Plant Sci. 2002;7(1):41–48. doi: 10.1016/S1360-1385(01)02187-2. [DOI] [PubMed] [Google Scholar]
- Skubacz A, Daszkowska-Golec A, Szarejko I. The role and regulation of ABI5 (ABA-Insensitive 5) in plant development, abiotic stress responses and phytohormone crosstalk. Front Plant Sci. 2016;7:1884. doi: 10.3389/fpls.2016.01884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staiger D, Brown JWS. Alternative splicing at the intersection of biological timing, development, and stress responses. Plant Cell. 2013;25(10):3640–3656. doi: 10.1105/tpc.113.113803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wahl MC, Will CL, Luhrmann R. The spliceosome: design principles of a dynamic RNP machine. Cell. 2009;136(4):701–718. doi: 10.1016/j.cell.2009.02.009. [DOI] [PubMed] [Google Scholar]
- Wang K, He JN, Zhao Y, Wu T, Zhou XF, Ding YL, Kong LY, Wang XJ, Wang Y, Li JG, Song CP, Wang BS, Yang SH, Zhu JK, Gong ZZ. EAR1 negatively regulates ABA signaling by enhancing 2C protein phosphatase activity. Plant Cell. 2018;30(4):815–834. doi: 10.1105/tpc.17.00875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z, Wang FX, Hong YC, Yao JJ, Ren ZZ, Shi HZ, Zhu JK. The flowering repressor SVP confers drought resistance in Arabidopsis by regulating abscisic acid catabolism. Mol Plant. 2018;11(9):1184–1197. doi: 10.1016/j.molp.2018.06.009. [DOI] [Google Scholar]
- Wang ZJ, Ji HT, Yuan BJ, Wang SF, Su C, Yao B, Zhao HT, Li X. ABA signalling is fine-tuned by antagonistic HAB1 variants. Nat Commun. 2015;6(1):8138. doi: 10.1038/ncomms9138. [DOI] [PubMed] [Google Scholar]
- Zhan XQ, Qian BL, Cao FQ, Wu WW, Yang L, Guan QM, Gu XB, Wang PC, Okusolubo TA, Dunn SL, Zhu JK, Zhu JH. An Arabidopsis PWI and RRM motif-containing protein is critical for pre-mRNA splicing and ABA responses. Nat Commun. 2015;6(1):8139. doi: 10.1038/ncomms9139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu JK. Abiotic stress signaling and responses in plants. Cell. 2016;167(2):313–324. doi: 10.1016/j.cell.2016.08.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.