Abstract
Summary
Randomized clinical trials and observational studies on the implementation of clinical governance models, in patients who had experienced a fragility fracture, were examined. Literature was systematically reviewed and summarized by a panel of experts who formulated recommendations for the Italian guideline.
Purpose
After experiencing a fracture, several strategies may be adopted to reduce the risk of recurrent fragility fractures and associated morbidity and mortality. Clinical governance models, such as the fracture liaison service (FLS), have been introduced for the identification, treatment, and monitoring of patients with secondary fragility fractures. A systematic review was conducted to evaluate the association between multidisciplinary care systems and several outcomes in patients with a fragility fracture in the context of the development of the Italian Guidelines.
Methods
PubMed, Embase, and the Cochrane Library were investigated up to December 2020 to update the search of the Scottish Intercollegiate Guidelines Network. Randomized clinical trials (RCTs) and observational studies that analyzed clinical governance models in patients who had experienced a fragility fracture were eligible. Three authors independently extracted data and appraised the risk of bias in the included studies. The quality of evidence was assessed using the Grading of Recommendations Assessment, Development, and Evaluation methodology. Effect sizes were pooled in a meta-analysis using random-effects models. Primary outcomes were bone mineral density values, antiosteoporotic therapy initiation, adherence to antiosteoporotic medications, subsequent fracture, and mortality risk, while secondary outcomes were quality of life and physical performance.
Results
Fifteen RCTs and 62 observational studies, ranging from very low to low quality for bone mineral density values, antiosteoporotic initiation, adherence to antiosteoporotic medications, subsequent fracture, mortality, met our inclusion criteria. The implementation of clinical governance models compared to their pre-implementation or standard care/non-attenders significantly improved BMD testing rate, and increased the number of patients who initiated antiosteoporotic therapy and enhanced their adherence to the medications. Moreover, the treatment by clinical governance model respect to standard care/non-attenders significantly reduced the risk of subsequent fracture and mortality. The integrated structure of care enhanced the quality of life and physical function among patients with fragility fractures.
Conclusions
Based on our findings, clinicians should promote the management of patients experiencing a fragility fracture through structured and integrated models of care. The task force has formulated appropriate recommendations on the implementation of multidisciplinary care systems in patients with, or at risk of, fragility fractures.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11657-023-01316-9.
Keywords: Fracture liaison service, Fragility fracture, Secondary prevention, Systematic review
Introduction
Fragility fractures impose a massive burden on health care systems and the global community [1]. Such fractures are the hallmark of osteoporosis, causing high morbidity, loss of independence, and negatively affecting the quality of life [2]. People who have experienced a fragility fracture (i.e., spontaneous or low-traumatic) have a greater fracture risk immediately after the event [3]. However, antiosteoporotic therapy administered soon after a fragility fracture may mitigate this risk [4]. Unfortunately, temporary or permanent medication discontinuation are frequent (> 50–80%), especially in secondary prevention [5, 6].
The detection of a major fragility fracture (i.e., spontaneous fracture or fracture resulting from a low-impact trauma/fall from standing height or less, occurring at the vertebral bodies, proximal hip, wrist, or humerus) is crucial to identify patients at high risk of subsequent fractures, evaluate bone fragility, and prescribe antiosteoporotic medication [7]. Following an initial fracture, several strategies may be adopted, although secondary preventive measures might not be promptly used. In the last decade, several initiatives (at the various levels—local, regional, national, and international) have been undertaken to improve secondary fracture prevention; these include fracture liaison services (FLS). FLS models were originally introduced in the orthopedic departments of tertiary referral centers as multidisciplinary teams, including coordinators, orthopedic surgeons, bone nurses, bone doctors (internists, endocrinologists, orthogeriatrics, rheumatologists), radiologists, and physiatrists, centered on the fractured patient. At present, these programs also involve primary care in the form of the general practitioner, which is fundamental to promote and support short- and long-term adherence to the antiosteoporotic treatments [8]. These models have proven to be effective in different settings and clinical pathways [9]. Indeed, FLS programs have been demonstrated to reduce fracture-related morbidity and mortality as well as decrease healthcare costs for the secondary prevention of fragility fractures [10].
This systematic review and meta-analysis aims to provide recommendations based on the best available evidence on the efficacy and effectiveness of clinical governance models. The findings may support decision-makers to minimize the cost and social burden associated with fragility fractures.
Methods
We conducted a systematic review to support the Panel of the Italian Fragility Fracture Guidelines (published on the platform of the Italian National Institute of Health) in formulating recommendations. Adopting the GRADE-ADOLOPMENT methodology [11] and the standards defined by the Sistema Nazionale Linee Guida (SNLG [12]), the multidisciplinary panel updated the clinical question of the Scottish guidelines (SIGN, Scottish Intercollegiate Guidelines Network [13]): “Is the use of clinical governance models, such as the so-called fracture liaison services, suitable for the post-fracture patient’s management?”
Inclusion and exclusion criteria
Randomized clinical trials (RCTs) and/or observational studies were selected if they met the following criteria: (1) population: patients who experienced a fragility fracture; (2) intervention: clinical governance models, such as case manager interventions or FLS; (3) comparison: standard care; (4) outcome: (i) primary outcome measures, specifically bone mineral density (BMD) testing rate, antiosteoporotic therapy initiation, adherence to antiosteoporotic medications, subsequent fracture, and mortality risk, and (ii) secondary outcomes were quality of life and physical performance.
Studies were excluded if they (i) were not published in the English language, (ii) did not report original findings (i.e., letters, case report), (iii) did not identify patients affected by a fragility fracture, or (iv) were not before and after studies on the clinical governance model implementation or did not consider standard treatment/non-attenders/another model as a comparator.
Data source and search strategy
We performed a PubMed, Embase, and Cochrane Library search to update the search of the SIGN guidelines, from 2013 up to 17 December 2020, and identified publications on clinical governance models for patients who have sustained a fragility fracture. A systematic review of the available literature was carried out according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) [14] (Supplemental Material, Table S1). The search strategy (Supplemental Material, Table S2) included specific keywords and/or corresponding MeSH terms related to “fragility fracture” AND “integrated models of care.” We checked the reference lists of the studies and the systematic reviews identified during the search process.
Study selection and data extraction
Three independent authors (AB, GP, RR) screened titles and abstracts according to the search strategy and then assessed the full text of the potentially relevant studies. Discrepancies between reviewers were resolved by a consensus meeting. For each included publication, the following information was extracted: (i) first author, year, and country of publication, (ii) study setting, (iii) type of population, (iv) intervention and comparator, and (v) follow-up period.
Quality of studies
The systematic reviews were evaluated using the AMSTAR-2 checklist [15]. The quality of each included publication, derived by our search, was assessed using the Cochrane Risk of Bias (RoB) tool for RCTs [16] and the Newcastle-Ottawa scales [17] for observational studies. The following domains of the Cochrane RoB tool were appraised: selection bias (random sequence generation and allocation concealment), performance bias (blinding of participants and personnel), detection bias (blinding of outcome assessment), attrition bias (incomplete outcome data), reporting bias (selective reporting), and other bias (such as funding bias). Each domain was classified as “high,” “low,” or “unclear” RoB to assess to what extent the publication did not provide sufficient information. In the Newcastle-Ottawa scales, the following domains were evaluated: selection, comparability, and outcome. The threshold for identifying high-quality studies was more than five points.
Quality of evidence
The quality of evidence of each outcome was judged by evaluating five dimensions (risk of bias, consistency of effect, imprecision, indirectness, and publication bias) using the Grading of Recommendations Assessment Development and Evaluation (GRADE) approach [18]. The evidence was downgraded from “high quality” by one level if serious limitations were found for each of the five dimensions, or by two levels if very serious limitations were found.
Statistical analysis
The intervention effect was estimated using the dichotomized measure of risk ratio (RR) to evaluate the effect of clinical governance models. Where possible, we adopted the adjusted RR and pooled adjusted estimates from the original studies. Estimates were summarized if at least three studies reported the association of interest.
Heterogeneity between study-specific estimates was tested using X2 statistics [19] and measured with the I2 index (a measure of the percentage variation across the studies) [20]. Meta-analyses were conducted to combine the outcome data using the DerSimonian random effects model [21], which takes into account both the sampling variance within the studies and the variation in the underlying effect across studies, such as sample characteristics. Furthermore, subgroup analyses according to RCTs were carried out. A publication bias was tested using Egger’s regression and funnel plot visual analysis [22].
All tests were considered statistically significant for p-values less than 0.05. The analyses and the correspondent graphical visualization of forest and funnel plots were respectively performed by using RevMan V.5.4 (Nordic Cochrane Center) and STATA Software Program V.16.1 (STATA).
Results
Study selection
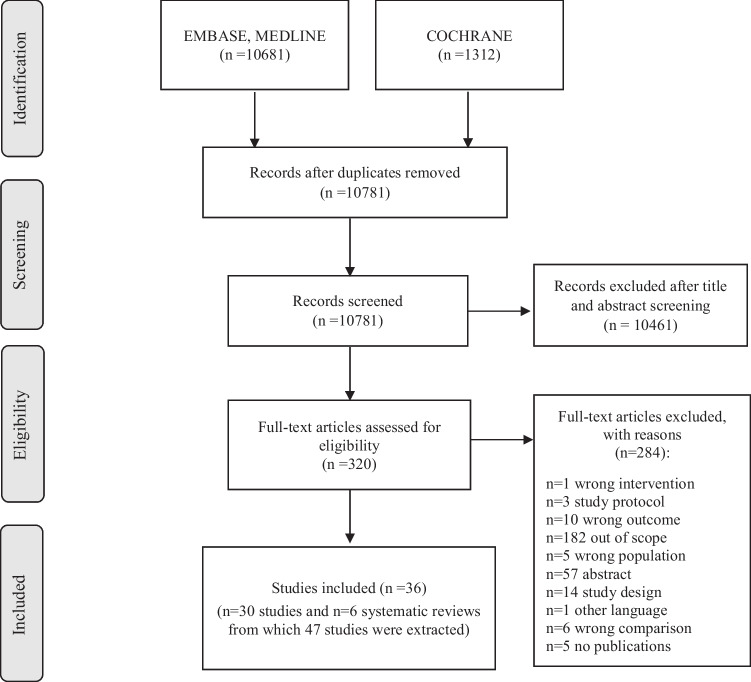
The objective of this study was to evaluate the efficacy of clinical governance implementation. A systematic literature review was carried out using the Embase, Medline, and Cochrane Central databases to update the clinical question elaborated by the SIGN Guideline [13]. As shown in Fig. 1, we identified 10,781 records.
Fig. 1.
Flowchart of study selection
We excluded 10,461 studies because they were unrelated to the issue based on the title and/or abstract. Among the remaining 320 publications assessed for full-text review, we excluded the studies that (i) considered the wrong population (5), intervention (1), comparison (5), or outcome (10); (ii) were study protocol (3) or abstract (57); (iii) had a wrong study design such as letter or case report (14); (iv) were out of scope (182) or not published in the English language (1). Further, the full text of five studies was not available. The remaining 36 publications were considered for the analysis, respectively: 30 primary studies [23–52] and 6 systematic reviews [53–58], from which 47 studies [59–105] were extracted (Table 1; Supplemental Material, Table S3).
Table 1.
Characteristics of included studies
| Author | Study type | Intervention/N | Control/N | Follow-up period (months) |
|---|---|---|---|---|
| Specialized model vs comparator model | ||||
| Coventry 2017 | A retrospective analysis | Geriatric or comanaged model | Orthopedic model | 12 |
| Majumdar 2011 | RCT | Patients followed by a nurse case-manager, who contacted patients and made clinic appointments for in-person visits and undertook several activities. | Patients followed by a multifaceted intervention, where patients received brief telephone-based counseling, and primary care physicians were faxed patient-specific reminders that notified them that their patient had been treated for a fragility fracture and that this put them at risk of osteoporosis | 6 |
| Vaculík 2017 | Cohort study with survey | Patients with individual recommendations given to the patients and their GPs (detailed recommendation group) | Patients without individual recommendations (general recommendation group) | 6 |
| van Helden 2007 | A prospective observational study | Patients admitted to a reference hospital in which a specialist osteoporosis nurse is employed | Patients admitted to five surrounding hospitals without a nurse | 11-16 weeks |
| Heilmann 2012 | Retrospective, parallel-group, cohort study | At the intervention site, a decentralized clinical-pharmacy-based osteoporosis management service (CPOMS) intervened on postmenopausal women following fracture | One centrally located registered nurse reviewed medical records for all women in the comparison group each month, assessed the appropriateness of either BMD screening or initiation of osteoporosis therapy, and sent recommendations to each patient’s primary care provider via the EMR | 6 |
| Kamel 2000 | A retrospective chart review | Patients were seen by a medical consultant | Patients were not seen by a medical consultant | 24 |
| Rolnick 2001 | RCT |
Group 1 education class on osteoporosis Group 2 education plus BMD |
No intervention | 22 |
| Sietsema 2018 | A retrospective cohort study | Patients who received osteoporosis management service (OP MS) care with a follow-up visit within 90 days of the first fracture | Patients who did not seek OP MS care but had a physician visit within 90 days of the first fracture | 12 |
| Streetan 2006 | A retrospective review of the charts | Patients received a consultation | Patients did not receive a consultation | 48 |
| Brankin 2005 | Observational study | Those who had sustained a fracture or had ≥ 2 osteoporosis risk factors and had not previously been screened for osteoporosis were invited for a dual-energy X-ray absorptiometry scan. | A second group of participants were women within the Coatbridge Local Health Community Cooperative, who had been referred for a DXA scan by their GP on the basis of having risk factors for osteoporosis | 18 |
| Jachna 2003 | A retrospective chart review | Hospitalists consultation at discharge | No consultation | na |
| FLS vs comparator model | ||||
| Murray 2005 | Cohort study | Patients admitted at a centre with a formal fracture liaison service (FLS) responsible for screening fracture patients for osteoporosis | Patients admitted at other centre relied upon individual clinicians to initiate investigation or treatment for osteoporosis in patients following fracture | 6 |
| Ganda 2014 | RCT | 6-monthy follow-up in secondary fracture prevention (SFP) program (FLS). | Patients referral to their primary care physician with a single SFP program visit at 24 months | 24 |
| Wallace 2011 | Practice in two fracture units was audited and compared using the NICE guidelines as an audit standard | Site B is a tertiary referral trauma centre which utilizes a continuous acute orthogeriatric care model for ward patients (one associate specialist 5 days per week, and one staff grade 3.5 days per week). In addition there are three consultant geriatrician-led ward rounds per week | In Site A, care is provided by trauma department doctors and a staff-grade orthogeriatrician (3.5 days per week) who will provide a medical assessment of a significant proportion of the unit’s inpatients. Consultant geriatrician ward-level assessment is available on a referral basis | na |
| Aubry-Rozier 2018 | A nationwide survey | Patients followed by a FLS team | Patients followed by GP | 12 |
| Majumdar 2018 | A pragmatic patient-level parallel-arm comparative effectiveness trial | High intensity FLS with a nurse-led case manager | Low intensity FLS with a low intensity multi-faceted intervention | 6 |
| Before-after specialized model | ||||
| Jones 2005 | A retrospective audit | Patients followed after the introduction of the protocol | Patients followed before the introduction of the protocol | 24 |
| Laslett 2007 | Retrospective study | Patients admitted after the implementation of a clinical pathway | Patients admitted before the implementation of a clinical pathway | 12 |
| Hawker 2003 | Pre-post intervention study | Patients admitted to a simple fracture clinic intervention | Controls patients were selected from among fracture clinic attendees in the 6–9 months preceding the intervention |
Intervention: 3 Control: 3–9 |
| Huntjens 2011 | Before–after impact analysis | Intervention group enrolled in 2004–2006 where a dedicated fracture nurse systematically offered fracture risk evaluation and treatment according to available guidelines | Pre-intervention group enrolled in 1999–2001 | 24 |
| Schuijt 2020 | A retrospective cohort study | Patients admitted to the orthogeriatric trauma unit, implemented on the first of January 2018 | Historical cohort before the implementation of the orthogeriatric trauma unit | 12 |
| Astrand 2012 | Cohort study with survey | Patients admitted from 2002 in a screening program at orthopedics department, where they are assessed by DEXA of the hip and spine, encouraged to see their doctor for decision on treatment regarding osteoporosis, and received written documents containing information, DEXA results, and a letter to their doctor with suggestions regarding blood tests and treatment | A historical control group of patients presented at department 1 year before the screening intervention | 72 |
| Anderson 2017 | Pre–post study design | Patients admitted at a comprehensive geriatric hip fracture program | Patients admitted before the program implementation | na |
| Anighoro 2020 | A retrospective cohort study | Patients admitted after the implementation of a standardized multidisciplinary pathway | Patients admitted before the implementation of a standardized multidisciplinary pathway | 1 |
| Lamb 2017 | A retrospective review of a single institution’s outcomes | Injured patients admitted after the fragility fracture program implementation in 2015. | Patients admitted from 2014 who presented before implementation of the fragility fracture program | na |
| Roy 2011 | Cohort study | Patients admitted after the implementation of an integrated model of care | Patients admitted before the implementation of an integrated model of care | na |
| Fisher 2006 | Prospective observational study with a retrospective (historical) control. | In 1998, a geriatric medicine registrar began overseeing daily medical care with weekly geriatrician consultant review (prospective study) | Between 1995 and 1997, medical problems were managed by a geriatric medicine consultation-only service (retrospective audit) | 48 |
| Sofie 2016 | Retrospective, single-centre study | Patients admitted to the orthopaedic ward after (October through December 2013) implementation of the clinical pathway | Patients admitted to the orthopaedic ward before (October through December 2010) implementation of the clinical pathway | na |
| Soong 2016 | A retrospective pre–post study | The post-intervention period was from January 1, 2012 to December 31, 2013. This group receiving an integrated medical-surgical co-management incorporating continuous improvement methodology | The pre-intervention period was from January 1, 2009 to December 31, 2010 | 24 |
| Sidwell 2004 | An audit | Patients admitted to the orthogetriatric rehabilitation ward after the new protocol implementation | Comparisons were made with a similar group from the same service and same ward but prior to implementation of the protocol | na |
| Hofflich 2014 | Pre-post observational study | Patients followed by a multidisciplinary team (September 1, 2009–June 30, 2010) | Patients followed in the pre-intervention period (July 1, 2008–June 30, 2009) | 12 |
| Johnson 2005 | Prospective study | A simple intervention in a general orthopedic clinic | 6-month pre-intervention group (October 2001 to March 2002) | 6 |
| Brañas 2018 | Observational study | In 2012, a process management systems (PMS) was adopted to improve the quality of care, compliance, and efficiency, implementing it in January 2013 |
Patients admitted during the pre-process period (January 1, 2009, to December 31, 2012) orthogeriatric co-management model |
inhospital deaths |
| Beaton 2017 “Improvements in osteoporosis..” | An interrupted time series analysis | The intervention consisted of assigning a screening coordinator to selected fracture clinics to identify, educate, and follow-up with fragility fracture patients and inform their physicians of the need to evaluate bone health. | At the control hospitals, no specific additions were made to the fracture management program and any care was dependent on the initiative of the usual health care team and their practices. | 12 |
| Ruggiero 2015 | A prospective observational study | Patients treated by a Fracture Prevention Service (FPS), a multidisciplinary integrated model of care | Patients admitted in the pre-intervention phase | 12 |
| Tosi 2008 | A pre-intervention and post-intervention study | Patients admitted to the Own the Bone project | Historical data collection | 10 |
| Jaglal 2009 | Historical control, non-equivalent, pre-post intervention study | Patients admitted at the Integrated post-fracture care model (1 January to 31 December 2005) | Historical controls who received usual care during the year preceding the intervention (1 January to 31 December 2003) |
Mean: Intervention: 17 Control: 8 |
| Abrahamsen 2019 | A prospective observational cohort study with historical control | Patients admitted to a orthogeriatric unit with interprofessional team consisting of orthopaedic surgeons, geriatric specialists, nurses, nursing assistants, physiotherapists, occupational therapists, and dieticians | Historical cohort (September 1, 2013 to January 31, 2014) | 1 |
| Baroni 2019 | A single-center, pre-post intervention observational study | The intervention consisted of implementation of an orthogeriatric comanagement (OGC) and a geriatric consultation service (GCS) that took place from September 1st, 2011, to February 28th, 2012 | The traditional orthopedic control group was obtained from the database of hospital records by looking at patients consecutively admitted to the same ward from March 1st to August 31st, 2011 | 12 |
| Svenoy 2020 | A single-center cohort study with historical controls | The patients in the hip fracture unit (HFU) group were included from May 2014 to May 2015 | The patients in the control group were included from September 2009 to January 2012 These patients were the group randomized to “usual care”, i.e., admission to the orthopedic ward. | 12 |
| Before-after FLS | ||||
| Davidson 2017 | Prospective cohort study with an historical control. | Patients admitted during the first 12 months after FLS implementation | Patients admitted during the 4 months before FLS implementation | 36 |
| Singh 2019 | A controlled before-and-after study | At the end of February 2015, the FLS program was implemented, and the intervention group was recruited from the time of FLS implementation to February 2016 (approximately 12 months of recruitment). The intervention group received the FLS program integrated into their orthopaedic clinic visit | Participants were recruited into the study before the FLS program was implemented (approximately 5 months of recruitment; October 2014 to February 2015) and they formed the control group (receiving ‘usual care’) | 6 |
| Bachour 2017 | A retrospective comparative study | Patients admitted after FLS implementation | Patients admitted before FLS implementation | 24 |
| Amphansap 2020 | A prospective cohort study | Patients participated in FLS program from April 1, 2014–March 31, 2019 (5 years implementation). | The data were compared with a previous study, before the commencement of the FLS | 12 |
| Hawley 2016 | Population-based longitudinal study with before–after time-series design | Orthogeriatric and nurse-led FLS models | Patients admitted before models implementation | 24 |
| Cosman 2017 | Cohort study | Patients admitted after the FLS implementation, between February 2010–May 2011 | The pre-FLS cohort included patients admitted for rehabilitation between July 2009 and February 2010 | 6 |
| Wasfie 2019 | A retrospective chart review with a pre-post study design | Patients who presented between January 2015 and December 2017, after fracture liaison service (FLS) | Patients who presented between January 2012 and December 2014, before FLS | 24 |
| Rotman-Pikielny 2018 | A prospective study with historical controls | All patients hospitalized with hip fractures from February to August 2013 when a collaborative Orthopedic-Metabolic team was established, without a coordinator. | The historical controls included hip fracture patients hospitalized from February to August 2012 | 12 |
| Axelsson 2016 | Cohort study | Patients followed during 2013–2014 by FLS | Historic counterparts in 2011–2012 at the same hospital | 24 |
| Amphansap 2016 | A prospective cohort study | Patients admitted after the FLS implementation from April 1, 2014-March 30, 2015. at the Police General Hospital, Bangkok | Patients admitted from a previous study prior to commencement of the FLS project | 12 |
| Chan 2015 | ‘Before and after’ cross-sectional extractions | Patients followed 12 months after 01/04/2009, the date that the primary care fracture liaison nurse started | Patients followed 12 months before 01/04/2009 | 12 |
| Greenspan 2018 | Pre–post study design | The FLS comparison included a prospective study of patients identified with an acute low-trauma fracture followed over six months for the same outcomes assessed above, but with the aid of the FLS model of care and the cloud-based tool. Patients were enrolled between April and December 2014 | The baseline assessment included a retrospective chart review to obtain data on the number of adults who received bone mineral density studies, vitamin D testing, calcium/vitamin D supplementation, and appropriate osteoporosis therapy within six months following a recently diagnosed acute low-trauma fracture | 6 |
| Beaupre 2020 | A population-based time series analysis | The H-FLS consisted of a nurse and physician team working where the nurse identified the patient for inclusion in the H-FLS and discussed the program with the patient and their family/caregiver as appropriate. The H-FLS programs were only offered to patients who resided in the local health zone pre-fracture | Patients admitted prior to H-FLS implementation to represent “usual care” | 12 |
| Specialized model vs standard care | ||||
| Beaton 2017 | Cohort study | Cases came from the BMD fast track program that included full fracture risk assessment and communication of relevant guidelines to the primary care provider (PCP) | Controls were selected from the usual care program | 6 |
| Lih 2011 | Prospective controlled observational study | Patients attending the minimal trauma fracture (MTF) program, a coordinated intervention program | Standard primary care | 48 |
| Cranney 2008 | Cluster RCT | The effect of a multifaceted intervention, directed at both patient and primary care physician, was evaluated. | Usual care | 6 |
| Davis 2007 | RCT | Patient Empowerment and Physician Alerting (PEPA) intervention | Usual care | 6 |
| Leslie 2012 | RCT | Group 1 had mailed notification of the fracture sent to their primary care physicians and Group 2 had notifications sent to both physicians and patients | Usual care | 12 |
| Majumdar 2004 | Non-randomized, controlled trial with blinded ascertainment of outcomes | Faxed physician reminders that contained osteoporosis treatment guidelines endorsed by local opinion leaders and patient education | Control patients received usual care and information about falls and home safety | 6 |
| Majumdar 2008 | RCT | A multifaceted intervention directed at patients in the form of telephone-based education, and their physicians in the form of guidelines endorsed by opinion leaders, supported by reminders | Usual care | 6 |
| Morrish 2009 | RCT | Case manager intervention | Usual care | 12 |
| Roux 2013 | RCT | Group 1: minimal (MIN) intervention; Group2: intensive (INT) intervention. | Standard care | 12 |
| Yuksel 2010 | RCT | Intervention consisted of printed materials, education, and quantitative ultrasound | Usual care | 4 |
| Merle 2017 | RCT | Patients admitted at the post-fracture ‘Prevention of Osteoporosis’ (PREVOST) program, where trained case manager, repeated oral/written education, prompting to visit PCP were implemented | Usual care | 6 |
| Shigemoto 2018 | A retrospective cohort study | Patients treated with a new multidisciplinary approach in 2014-2016 | Patients received conventional treatment in 2012 | 12 |
| Vidan 2005 | RCT | Participants assigned to a daily multidisciplinary geriatric intervention | Usual care | 12 |
| Queally 2013 | RCT | Patient admitted to fracture clinic | Usual care: assessment initiation by the participant’s general practitioner | 3 |
| Miki 2008 | RCT | Inpatient osteoporosis evaluation initiated by orthopaedic surgeons combined with follow-up in a specialized orthopaedic osteoporosis clinic | Usual care where the responsibility of patients evaluation and treatment was placed solely on the primary care physician | 6 |
| Harrington 2005 | Populatioon-based study | Direct referral pilot study (2002) where a nurse managed the direct referral process and contacted the patients to arrange DXA and osteoporosis consultation | Osteoporosis care by primary physicians (2000–2001) | 12 |
| FLS vs standard care | ||||
| Inderjeeth 2018 | A prospective parallel cohort study | Patients admitted to FLS program | Routine care: retrospective group of the same hospital, and prospective group of other hospital | 3-12 |
| Naranjo 2017 | Observational study | Orthogeriatric fracture liaison service (FLS) | Standard care | 6 |
| Huntjens 2014 | Prospective study | FLS group | Non FLS group (standard fracture care) | 24 |
| Henderson 2017 | Cohort study | Orthogeriatric service, a comprehensive geriatric assessment, daily medical involvement of a geriatric team and specialized follow-up assessment of bone and vascular health. | The comparative group received the usual standard of care, which consisted of standard orthopedic care with medical, or geriatric consults received on an as requested basis. | 12 |
| Specialized model vs non-attenders | ||||
| Van der Kallen 2014 | Prospective cohort study | Patients who attended a Fracture Prevention Clinic | Patients who did not attend the clinic | 24 |
| Goltz 2013 | Population-based study | Participants at program of integrated care for osteoporosis in terms of medication supply, fracture incidence and expenses | Controls were also diagnosed with osteoporosis but did not participate in the program | 36 |
| FLS vs non-attenders | ||||
| Nakayama 2016 | Historical cohort study | FLS hospital | No FLS hospital | 36 |
| Sanli 2019 | A prospective cohort-study | Attenders FLS | Non-attenders FLS | 24 |
Study characteristics
The majority of the studies were conducted in Australia (n = 10 [27, 36, 43, 59, 87–90, 94, 95]), Canada (n = 17 [25, 28, 34, 35, 38, 60–70, 102]), USA (n = 18 [23, 29, 31, 32, 37, 46, 77–86, 92, 93]), and European Union (n = 19 [24, 26, 33, 40, 42, 44, 45, 48–50, 76, 91, 96–98, 100, 101, 103, 105]). Five studies were carried out in the Asian continent (Israel, Japan, Thailand, and Lebanon), four publications were from the UK, three studies were performed in Ireland and one in New Zealand.
Fifteen studies were RCTs [28, 60–66, 72, 77, 92, 94, 100, 102, 105], while the remaining papers were observational studies.
Average follow-up was 9 and 17 months respectively for RCT and observational studies, although eight of them did not specify it [23, 24, 26, 32, 75, 83, 93, 104]. Studies were conducted using information from hospital or community-based hospitals or general practitioners [23–26, 28–34, 39–45, 48–50, 52, 59–61, 63, 66–71, 73, 75–79, 81–83, 86, 91, 93–95, 97–101, 103–105], tertiary referral hospital or centers [27, 36, 47, 88–90], or both [87], community pharmacies [64], administrative data [35, 37, 46, 51, 65, 70, 84, 85, 96] or specialized clinics or centers [62, 72, 74, 80, 92, 102]. In general, patients had low trauma fracture, specifically hip [23, 24, 27, 31, 32, 34, 35, 40–42, 44, 45, 47, 49, 51, 52], upper extremity [28], and vertebral [29] fracture.
Only one [46] of the observational studies extracted through the search had an NOS score lower than 6 and was therefore assigned to the category of low-quality study. Generally, “Comparability of cohorts on the basis of the design or analysis” in the comparability section was the domain for which problems were encountered in the most studies (14 [23, 24, 26, 30–32, 34, 35, 38, 39, 45–47, 52]), followed by the domain “Adequacy of follow-up of cohorts” in the outcome section (5 studies [25, 26, 34, 44, 46]) and “Demonstration that outcome of interest was not present at start of study” in the selection section (3 studies [25, 44, 46]).
Two systematic reviews [57, 58] were assessed as low quality, while the remaining were of very low quality (Supplemental Material, Table S4).
Studies considered the following comparisons: (a) after vs before the implementation of a specialized [23, 24, 26, 31, 32, 34, 38, 40, 44, 48, 49, 59, 67, 69, 76, 80, 82, 83, 85, 87, 88, 91, 97, 104] or a FLS [25, 29, 30, 35, 37, 39, 41, 43, 47, 51, 71, 86, 101] model, (b) a specialized model [27, 45, 46, 73, 78, 81, 84, 92, 93, 102, 103] or FLS [28, 33, 74, 75, 94] vs a comparator model, (c) a specialized [52, 60–66, 68, 70, 72, 77, 79, 89, 100, 105] or FLS [36, 42, 98, 99] model vs standard care or a specialized [90, 96] or FLS [50, 95] model vs non-attenders.
Primary outcomes
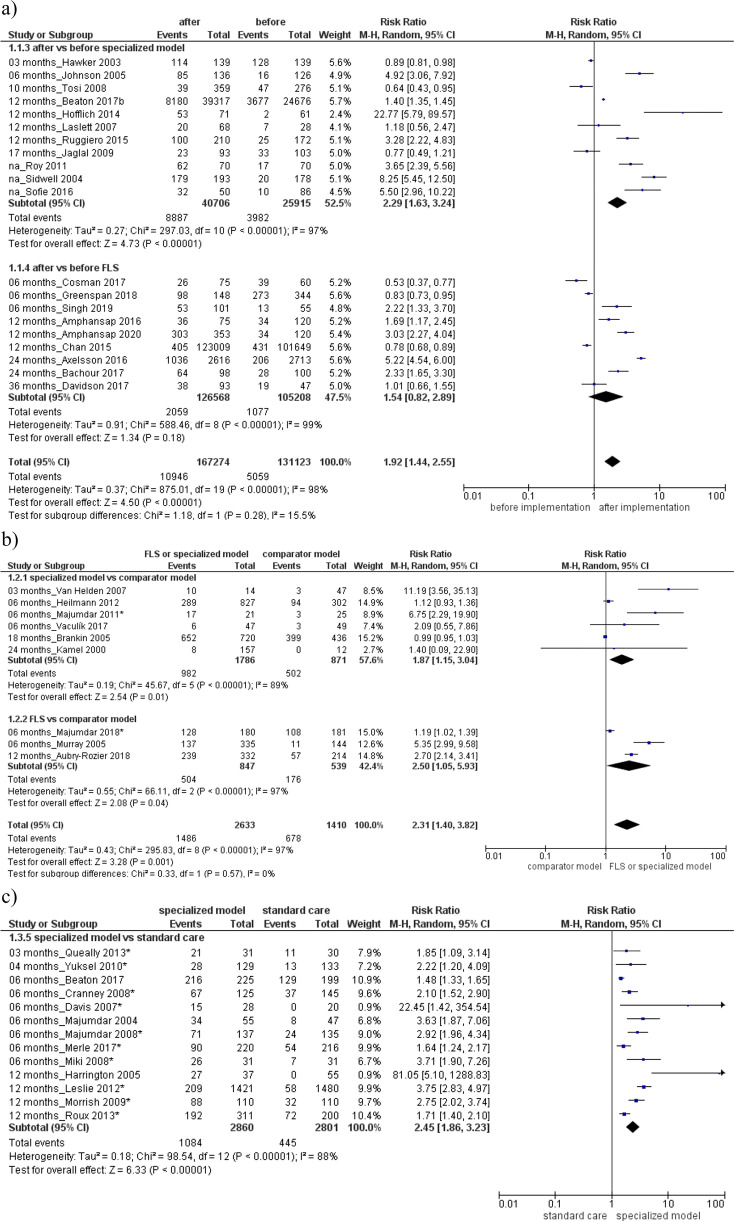
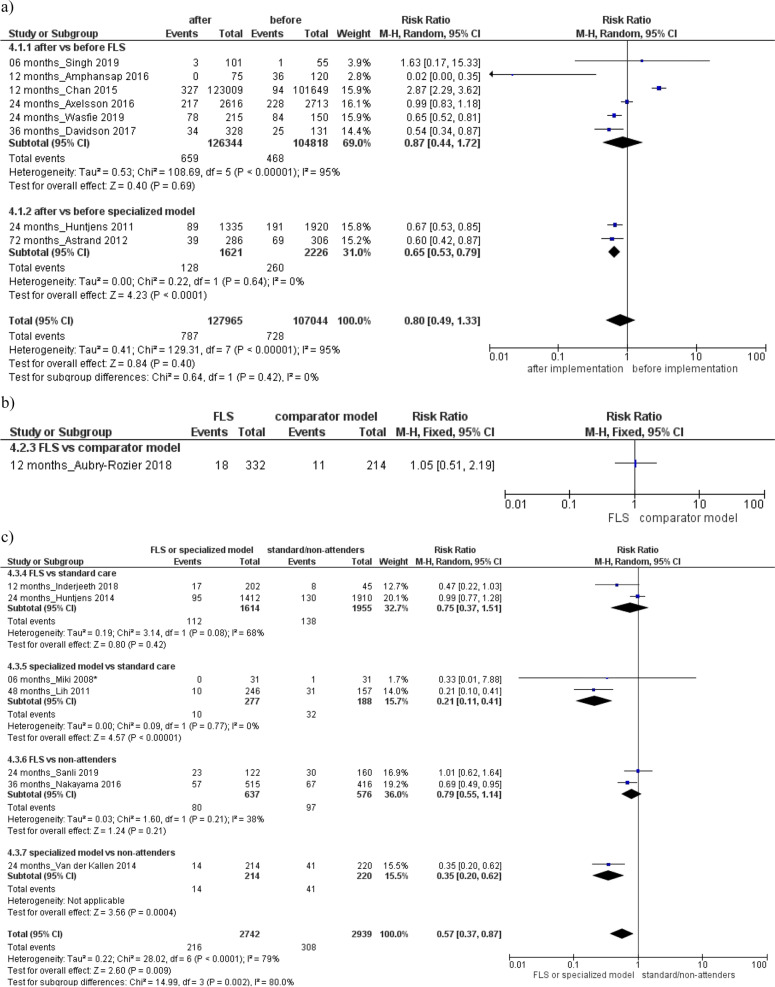
As shown in Fig. 2a, increased BMD testing rate was detected after the implementation of a specialized model or FLS group compared their pre-implementation, respectively 10,946 and 5059. Overall, 20 studies detected a statistically significant RR of 1.92 (95% CI, 1.44 to 2.55) with a high heterogeneity between groups (I2 = 98%) and without publication bias (p = 0.29; Supplemental Material, Figure S1).
Fig. 2.
Evaluation of BMD testing rate a after vs before the specialized or fracture liaison service (FLS) model implementation, b in the specialized or FLS model vs comparator model, c in the specialized model vs standard care. Squares represent study-specific relative risk estimates (size of the square reflects the study-specific statistical weight, that is, the inverse of the variance); horizontal lines represent 95% CIs; diamonds represent summary relative risk estimates with corresponding 95% CIs; p values are from testing for heterogeneity between study-specific estimates. Asterisk indicates randomized controlled studies. Abbreviations: CI confidence interval, RR relative risk
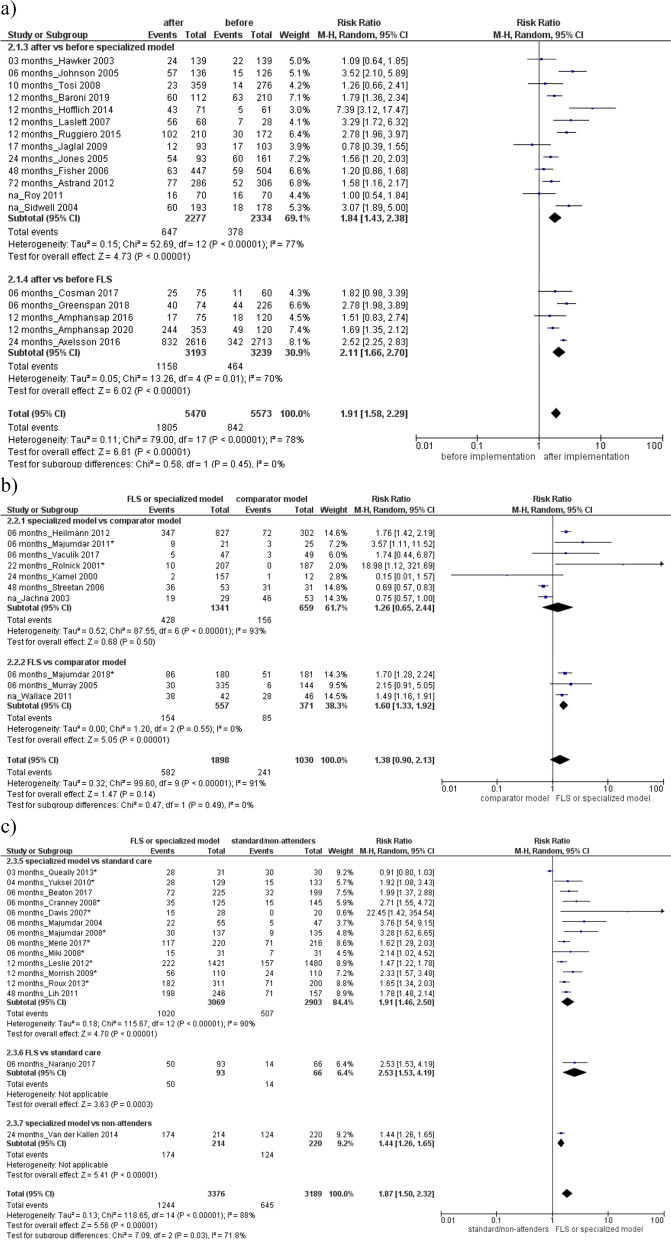
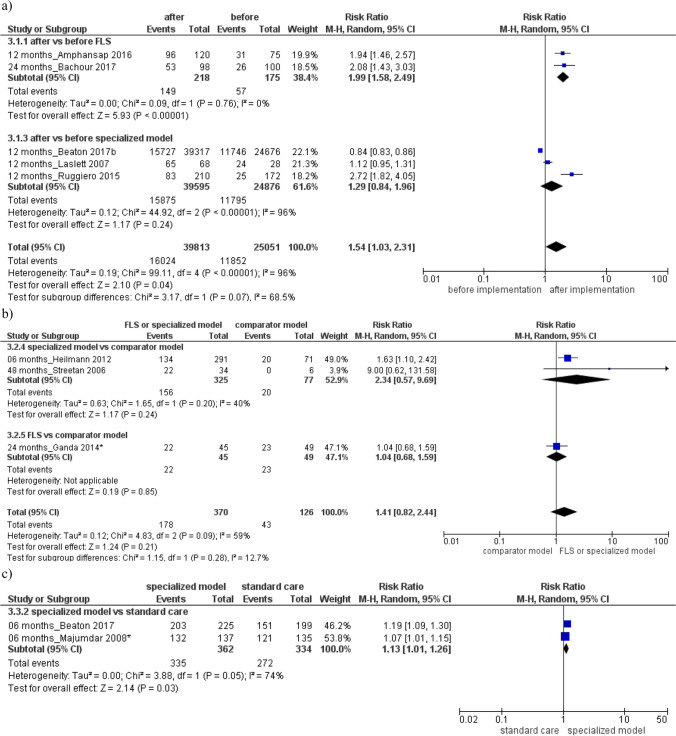
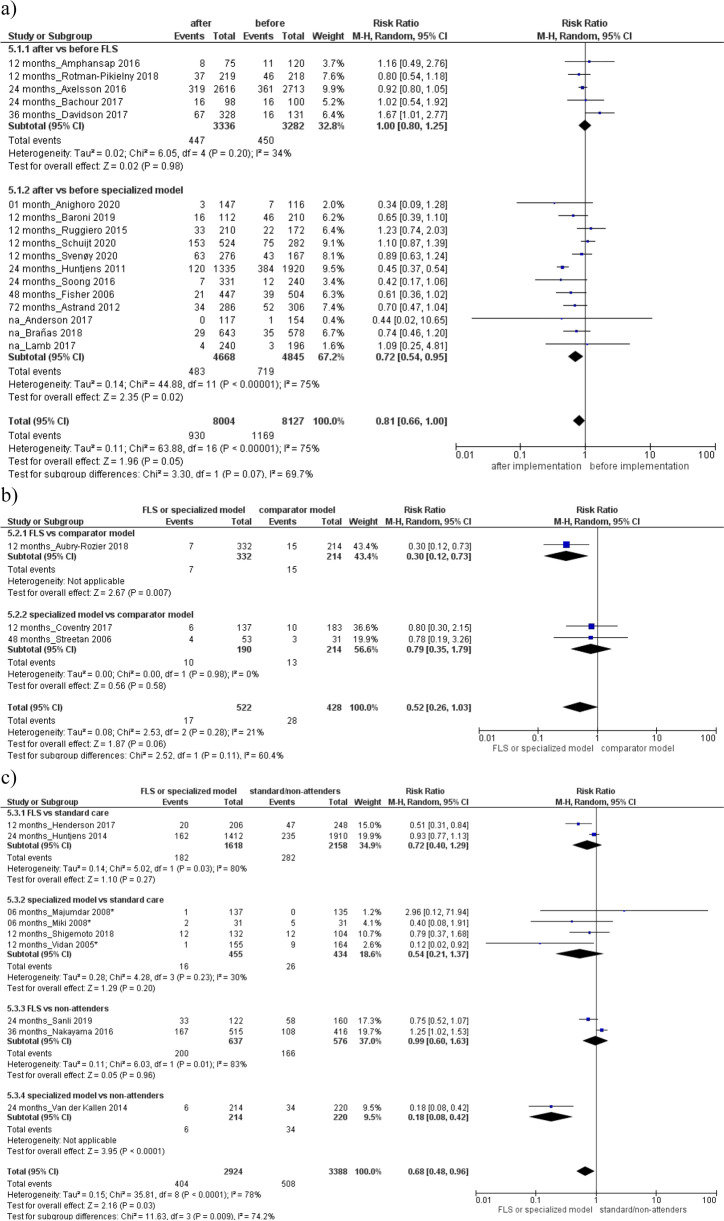
Then, higher BMD testing rate was found in the specialized or FLS model respect to comparator model (Fig. 2b) RR 2.31 (95% CI, 1.40 to 3.82), or standard care (Fig. 2c) RR 2.45 (95% CI, 1.86 to 3.23), with a high heterogeneity among groups (I2 = 97% and 88%). Evaluation of antiosteoporotic therapy showed increased initiation after the specialized or FLS model implementation (RR 1.91, 95% CI 1.58 to 2.29; 18 studies, Fig. 3a) or compared to a standard care/non-attenders (RR 1.87, 95% CI 1.50 to 2.32; 15 studies, Fig. 3c). Furthermore, improved adherence to treatment was detected after the implementation of FLS or specialized model (RR 1.54, 95% CI 1.03–2.31; 5 studies, Fig. 4a) or compared to a standard care (RR 1.31, 95% CI 1.01 to 1.26; 2 studies, Fig. 4c). Both analyses were characterized by high heterogeneity among studies and absence of publication bias (Supplemental Material, Figure S1).
Fig. 3.
Evaluation of antiosteoporotic initiation a after vs before the specialized or fracture liaison service (FLS) model implementation, b in the specialized or FLS model vs comparator model, c in the specialized model or FLS vs standard care/non-attenders. Squares represent study-specific relative risk estimates (size of the square reflects the study-specific statistical weight, that is, the inverse of the variance); horizontal lines represent 95% CIs; diamonds represent summary relative risk estimates with corresponding 95% CIs; p values are from testing for heterogeneity between study-specific estimates. Asterisk indicates randomized controlled studies. Abbreviations: CI confidence interval, RR relative risk
Fig. 4.
Evaluation of antiosteoporotic adherence a after vs before the specialized or fracture liaison service (FLS) model implementation, b in the specialized or FLS model vs comparator model, c in the specialized model vs standard care. Squares represent study-specific relative risk estimates (size of the square reflects the study-specific statistical weight, that is, the inverse of the variance); horizontal lines represent 95% CIs; diamonds represent summary relative risk estimates with corresponding 95% CIs; p values are from testing for heterogeneity between study-specific estimates. Asterisk indicates randomized controlled studies. Abbreviations: CI confidence interval, RR relative risk
Thus, a significant decreased risk of subsequent fracture and a reduction of the mortality rate was found after the specialized or FLS group implementation (subsequent fracture: RR 0.65, 95% CI 0.53 to 0.79; 2 studies; Fig. 5a; mortality: RR: 0.72, 95% CI 0.54 to 0.95; 12 studies, Fig. 6a) or respect to standard care/non-attenders (subsequent fracture: RR 0.57, 95% CI 0.37 to 0.87; 7 studies; Fig. 5c; mortality: RR 0.68, 95% CI 0.48-0.96; 9 studies; Fig. 6c). Both analyses were characterized by high heterogeneity between studies and no existence of publication bias (Supplemental Material, Figure S1).
Fig. 5.
Evaluation of the risk of subsequent fracture a after vs before the specialized or fracture liaison service (FLS) model implementation, b in the FLS model vs comparator model, c in the specialized or FLS model vs standard care/non-attenders. Squares represent study-specific relative risk estimates (size of the square reflects the study-specific statistical weight, that is, the inverse of the variance); horizontal lines represent 95% CIs; diamonds represent summary relative risk estimates with corresponding 95% CIs; p values are from testing for heterogeneity between study-specific estimates. Asterisk indicates randomized controlled studies. Abbreviations: CI confidence interval, RR relative risk
Fig. 6.
Evaluation of the risk of mortality a after vs before the specialized or fracture liaison service (FLS) model implementation, b in the specialized or FLS model vs comparator model, c in the specialized or FLS model vs standard care/non-attenders. Squares represent study-specific relative risk estimates (size of the square reflects the study-specific statistical weight, that is, the inverse of the variance); horizontal lines represent 95% CIs; diamonds represent summary relative risk estimates with corresponding 95% CIs; p values are from testing for heterogeneity between study-specific estimates. Asterisk indicates randomized controlled studies. Abbreviations: CI confidence interval, RR relative risk
For all of the aforementioned outcomes, the certainty of the evidence was downgraded from low to very low due to serious inconsistency and study design (Supplemental Material, Table S5).
All the above mentioned results are summarized in Supplemental Material, Table S6.
Subgroup analyses
Previous findings regarding the BMD testing rate and antiosteoporotic initiation were confirmed based only on RCTs, specifically for the specialized or FLS model implementation compared to standard care/non-attenders (Supplemental Material, Figures S2-3). Moreover, an increased antiosteoporotic initiation was found for the specialized or FLS model implementation respect to a comparator model (Supplemental Material, Figure S3). Conversely, the summary estimate of the RCTs showed a non-significant reduction in the adherence to antiosteoporotic treatment, risk of subsequent fracture or mortality (Supplemental Material, Figure S2-6).
Secondary outcomes
A systematic review [58] evaluated the effect of clinical care pathways that enrolled patients of over 50 years of age who had sustained a hip fracture. Twenty-two studies evaluated these secondary preventive measures compared to usual care. Twelve studies measured the health-related quality of life (HRQoL) between 3 and 12 months, which improved compared with usual care patients following hip fracture. Moreover, 19 studies estimated the physical function between 3 and 12 months that increased with respect to standard treatment. When the meta-analyses were stratified by length of follow-up, a greater HRQoL measure and physical function were found compared to usual care between 3 and 12 months.
Discussion
This systematic review evaluated a clinical question of the Italian Guideline [106] and a panel of experts formulated recommendations through a structured and transparent process. Specifically, we conducted a systematic review and meta-analysis on the efficacy of clinical governance models (i.e., FLS, structured service delivery models, nurse-led clinics) versus the pre-implementation, a comparator model or standard care/non-attenders in low-income and developed countries. These results highlighted that implementation of clinical governance significantly improved BMD testing rate, antiosteoporotic therapy initiation, adherence as well as reduced the risk of subsequent fracture or mortality compared to the standard care/non-attenders. Moreover, a higher BMD testing rate, number of patients who initiated antiosteoporotic therapy and adherence to the medications was found after the FLS or specialized model implementation respect to their pre-implementation.
The benefits of the abovementioned results were more evident considering the RCT that underlined the effectiveness of the integrated structure of care versus standard treatment, specifically for the BMD testing rate and the antiosteoporotic initiation, or versus a comparator model, specifically for the antiosteoporotic initiation. These findings are consistent with studies that evaluated the implementation of an FLS, which similarly to our study underlined the effectiveness in reducing the bone fragility evaluation and treatment gaps, and subsequent fractures and mortality rates [57, 58, 107]. The results of this meta-analysis enabled us to recommend the management of patients with fragility fractures through multidisciplinary care systems (e.g., FLS) which ensures patients' transition to out-hospital services.
The primary objective of an FLS is the prevention of subsequent fragility fractures, associated with indirect and direct costs attributable to the antiosteoporotic treatment, which should be administered for prolonged periods to maintain therapy in subjects at high risk of fracture [5, 6]. Recently, the scientific community has focused on the impact of fragility fractures and their clinical consequences. Structures such as the multidisciplinary FLS are becoming increasingly popular in medical communities around the world. In the last decade, these programs have been promoted and supported by international scientific organizations, such as the International Osteoporosis Foundation (IOF), the American Society for Bone and Mineral Research (ASBMR) and the European League Against Rheumatism (EULAR) together with the European Federation of National Associations of Orthopaedics and Traumatology (EFORT) [9, 108–111]. International scientific societies have largely endorsed and promoted the establishment of coordinated, multidisciplinary clinical care governance for the management of patients with recent major fragility fractures in various parts of the world [112–116].
Regarding secondary outcomes, the establishment of clinical care pathways compared to usual care was demonstrated to improve HRQoL and physical performance in a meta-analysis that included patients over 50 years. This acquires particular importance in older patients with comorbidities and potentially improves the cost-effectiveness of these systems in clinical practice, given the various comorbidities displayed by these subjects.
Limitations and strengths
Some limitations must be acknowledged. First, we considered different models of clinical governance, which may reduce the reliability of our findings. Moreover, the majority of studies were conducted in Europe or America, which may limit the generalizability of the results. Second, we have some concerns regarding heterogeneous multidisciplinary programs, characteristics of patients, fracture site at baseline, and length of follow-up. Third, the certainty of the evidence for the assessed outcomes was judged as “very low” or “low” due to the inconsistency of the estimates and the inclusion of observational studies with a modest sample size. Fourth, the majority of the included studies did not account for competing risks of death, which could have affected the results. Fifth, although falls may influence and increase the risk of subsequent fracture, this determinant was not an outcome of interest of the present meta-analysis. However, the role of falls will be investigated in a clinical question of the Italian Guideline and will be converted into a scientific article.
Despite the above limitations, this study presents points of strength. The exhaustive search strategy identified an overview of studies on the implementation of clinical governance programs. Then, the internal validity of the included studies was assessed using the Newcastle-Ottawa Scale for observational studies and the RoB tool for RCTs. Finally, preliminary performance indicators of FLS efficacy might be represented by BMD testing rate and initiation of treatment [109].
Perspectives
Rigorous RCT testing the efficacy and effectiveness of models of clinical governance in secondary fracture prevention (i.e., FLS) against “standard care” will not likely be furtherly pursued in the future, mainly for ethical reasons. Therefore, longitudinal, large “real-world” studies, preferably designed and homogenized for including specific Key Performance Indicators of the efficacy of FLS, as advised by the international initiative IOF Capture The Fracture initiative-Best Practice Framework [117], are expected to be included in future systematic analyses in this field to reinforce the results. With this respect, also results coming from the surveys carried out within National Registries, which are now at an advanced stage of development worldwide [118–123], will be likely incorporated in these future assessments.
Conclusion
This systematic review and meta-analysis indicate that the implementation of structured and integrated models of care increased the BMD testing rate, antiosteoporotic initiation and adherence to medication as well as reduced the risk of subsequent fracture and mortality and improved HRQoL and the physical function of patients experiencing a fragility fracture. The task force formulated recommendations on the introduction of these programs, although our systematic review judged outcomes affected by “very low” to “low” quality evidence.
Supplementary information
Supplemental Table S1. Prisma Checklist. (PDF 888 kb)
Supplemental Table S2. Search Strategy. Supplemental Table S3. Characteristics of included studies. Supplemental Table S4. Quality evaluation. Supplemental Table S5. Summary of findings, GRADE approach. Supplemental Table S6. Summary results. Supplemental Figure S1. Funnel plot and Egger’s test. Supplemental Figure S2. BMD testing rate in FLS, RCT studies. Supplemental Figure S3. Antiosteoporotic initiation in FLS, RCT studies. Supplemental Figure S4. Antiosteoporotic adherence in FLS, RCT studies. Supplemental Figure S5. Subsequent fracture risk in FLS, RCT studies. Supplemental Figure S6. Mortality risk in FLS, RCT studies. Complete list of experts involved. (DOCX 353 kb)
Acknowledgements
We thank the Charlesworth Author Services for the English Academic Editing.
Patient and public involvement statement
This research was done without patient involvement. Patients were not invited to comment on the study design and were not consulted to develop patient-relevant outcomes or interpret the results. Patients were not invited to contribute to the writing or editing of this document for readability or accuracy.
Data sharing
No additional data is available.
Transparency declaration
The lead author (the manuscript’s guarantor) affirms that the manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
Funding
Open access funding provided by Università degli Studi di Milano - Bicocca within the CRUI-CARE Agreement. The Italian guideline was funded by ALTIS Omnia Pharma Service, which did not affect the content of the document.
Declarations
Conflicts of interest
GA declares personal fees from Theramex, Amgen, BMS, Lilly, Fresenius Kabi and Galapagos. LC declares personal fees from UCB Pharma, Abiogen Pharma, Bruno Farmaceutici, Sandoz, Metagenics. DG has received honoraria as consultant for Eli-Lilly, Organon, MSD Italia. SG has received honoraria as consultant for UCB Pharma. SM has received honoraria as consultant for UCB, Eli-Lilly, Amgen. MLB has received (i) honoraria from Amgen, Bruno Farmaceutici, Calcilytix, Kyowa Kirin, UCB; (ii) grants and/or speaker: Abiogen, Alexion, Amgen, Bruno Farmaceutici, Echolight, Eli Lilly, Kyowa Kirin, SPA, Theramex, UCB Pharma; (iii) consultant: Alexion, Amolyt, Bruno Farmaceutici, Calcilytix, Kyowa Kirin, UCB Pharma. GC received research support from the European Community (EC), the Italian Agency of Drug (AIFA), and the Italian Ministry for University and Research (MIUR). He took part to a variety of projects that were funded by pharmaceutical companies (i.e., Novartis, GSK, Roche, AMGEN, and BMS). He also received honoraria as member of Advisory Board from Roche. No other potential conflicts of interest relevant to this article were disclosed. MR declares personal fees from Amgen, ABBvie, BMS, Eli Lilly, Galapagos, Menarini, Novartis, Pfizer, Sandoz, Theramex and UCB outside the submitted work. RM took part to a project funded by Abiogen Pharma. GI received honoraria as speaker by Eli-Lilly, Menarini, UCB Pharma. The other authors declare that they have no conflict of interest.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
G. Porcu, Email: gloria.porcu@unimib.it
R. Ronco, Email: raffaella.ronco@unimib.it
A. Biffi, Email: annalisa.biffi@unimib.it
References
- 1.Borgström F, Karlsson L, Ortsäter G, Norton N, Halbout P, Cooper C, et al. Fragility fractures in Europe: burden, management and opportunities. Arch Osteoporos. 2020;15:59. doi: 10.1007/s11657-020-0706-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Compston JE, McClung MR, Leslie WD. Osteoporosis. Lancet. 2019;393:364–376. doi: 10.1016/S0140-6736(18)32112-3. [DOI] [PubMed] [Google Scholar]
- 3.Wong RMY, Wong PY, Liu C, Wong HW, Chung YL, Chow SKH, et al. The imminent risk of a fracture-existing worldwide data: a systematic review and meta-analysis. Osteoporos Int. 2022;33(12):2453–2466. doi: 10.1007/s00198-022-06473-0. [DOI] [PubMed] [Google Scholar]
- 4.Kanis JA, Cooper C, Rizzoli R, Reginster JY. Scientific Advisory Board of the European Society for Clinical and Economic Aspects of Osteoporosis (ESCEO) and the Committees of Scientific Advisors and National Societies of the International Osteoporosis Foundation (IOF). European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int. 2019;30:3–44. doi: 10.1007/s00198-018-4704-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Åkesson KE, McGuigan FEA. Closing the Osteoporosis Care Gap. Curr Osteoporos Rep. 2021;19:58–65. doi: 10.1007/s11914-020-00644-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Curtis EM, Dennison EM, Cooper C, Harvey NC. Osteoporosis in 2022: care gaps to screening and personalised medicine. Best Pract Res Clin Rheumatol 2022;101754. [DOI] [PMC free article] [PubMed]
- 7.Mitchell PJ, Cooper C, Fujita M, Halbout P, Åkesson K, Costa M, et al. Quality improvement initiatives in fragility fracture care and prevention. Curr Osteoporos Rep. 2019;17:510–520. doi: 10.1007/s11914-019-00544-8. [DOI] [PubMed] [Google Scholar]
- 8.Geusens P, Bours SPG, Wyers CE, van den Bergh JP. Fracture liaison programs. Best Pract Res Clin Rheumatol. 2019;33:278–289. doi: 10.1016/j.berh.2019.03.016. [DOI] [PubMed] [Google Scholar]
- 9.Javaid MK. Efficacy and efficiency of fracture liaison services to reduce the risk of recurrent osteoporotic fractures. Aging Clin Exp Res. 2021;33:2061–2067. doi: 10.1007/s40520-021-01844-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lewiecki EM, Ortendahl JD, Vanderpuye-Orgle J, Grauer A, Arellano J, Lemay J, et al. Healthcare policy changes in osteoporosis can improve outcomes and reduce costs in the United States. JBMR Plus. 2019;3:e10192. doi: 10.1002/jbm4.10192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schünemann HJ, Wiercioch W, Brozek J, Etxeandia-Ikobaltzeta I, Mustafa RA, Manja V, et al. GRADE Evidence to Decision (EtD) frameworks for adoption, adaptation, and de novo development of trustworthy recommendations: GRADE-ADOLOPMENT. J Clin Epidemiol. 2017;81:101–110. doi: 10.1016/j.jclinepi.2016.09.009. [DOI] [PubMed] [Google Scholar]
- 12.Centro Nazionale per l’Eccellenza Clinica, la Qualità e la Sicurezza delle Cure (2019) Manuale metodologico per la produzione di linee guida di pratica clinica. Istituto Superiore di Sanità. https://www.iss.it/-/snlg-manualemetodologico. Accessed 20 Dec 2022
- 13.Scottish Intercollegiate Guidelines Network . Management of osteoporosis and the prevention of fragility fractures: a national clinical guideline. Scottish Intercollegiate Guidelines Network; 2015. [Google Scholar]
- 14.Page MJ, Moher D. Evaluations of the uptake and impact of the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) Statement and extensions: a scoping review. Syst Rev. 2017;6:263. doi: 10.1186/s13643-017-0663-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shea BJ, Reeves BC, Wells G, Thuku M, Hamel C, Moran J, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. doi: 10.1136/bmj.j4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Higgins JPT, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603–605. doi: 10.1007/s10654-010-9491-z. [DOI] [PubMed] [Google Scholar]
- 18.Balshem H, Helfand M, Schünemann HJ, Oxman AD, Kunz R, Brozek J, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol. 2011;64:401–406. doi: 10.1016/j.jclinepi.2010.07.015. [DOI] [PubMed] [Google Scholar]
- 19.Cochran WG. The combination of estimates from different experiments. Biometrics. 1954;10:101–129. doi: 10.2307/3001666. [DOI] [Google Scholar]
- 20.Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 22.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lamb LC, Montgomery SC, Wong Won B, Harder S, Meter J, Feeney JM. A multidisciplinary approach to improve the quality of care for patients with fragility fractures. J Orthop. 2017;14:247–251. doi: 10.1016/j.jor.2017.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Brañas F, Ruiz-Pinto A, Fernández E, Del Cerro A, de Dios R, Fuentetaja L, et al. Beyond orthogeriatric co-management model: benefits of implementing a process management system for hip fracture. Arch Osteoporos. 2018;13:81. doi: 10.1007/s11657-018-0497-6. [DOI] [PubMed] [Google Scholar]
- 25.Singh S, Whitehurst DG, Funnell L, Scott V, MacDonald V, Leung PM, et al. Breaking the cycle of recurrent fracture: implementing the first fracture liaison service (FLS) in British Columbia, Canada. Arch Osteoporos. 2019;14:116. doi: 10.1007/s11657-019-0662-6. [DOI] [PubMed] [Google Scholar]
- 26.Sofie S, Yves P, Barbara V, Margareta L, Raf VH, Bruno V, et al. Building for better bones: evaluation of a clinical pathway in the secondary prevention of osteoporotic fractures. Eur J Hosp Pharm. 2018;25:210–213. doi: 10.1136/ejhpharm-2016-000906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Coventry LS, Nguyen A, Karahalios A, Roshan-Zamir S, Tran P. Comparison of 3 different perioperative care models for patients with hip fractures within 1 health service. Geriatr Orthop Surg Rehabil. 2017;8:87–93. doi: 10.1177/2151458517692651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Majumdar SR, McAlister FA, Johnson JA, Rowe BH, Bellerose D, Hassan I, et al. Comparing strategies targeting osteoporosis to prevent fractures after an upper extremity fracture (C-STOP Trial): a randomized controlled trial. J Bone Miner Res. 2018;33:2114–2121. doi: 10.1002/jbmr.3557. [DOI] [PubMed] [Google Scholar]
- 29.Wasfie T, Jackson A, Brock C, Galovska S, McCullough JR, Burgess JA. Does a fracture liaison service program minimize recurrent fragility fractures in the elderly with osteoporotic vertebral compression fractures? Am J Surg. 2019;217:557–560. doi: 10.1016/j.amjsurg.2018.09.027. [DOI] [PubMed] [Google Scholar]
- 30.Bachour F, Rizkallah M, Sebaaly A, Barakat A, Razzouk H, El Hage R, et al. Fracture liaison service: report on the first successful experience from the Middle East. Arch Osteoporos. 2017;12:79. doi: 10.1007/s11657-017-0372-x. [DOI] [PubMed] [Google Scholar]
- 31.Anighoro K, Bridges C, Graf A, Nielsen A, Court T, McKeon J, et al. From ER to OR: results after implementation of multidisciplinary pathway for fragility hip fractures at a level I trauma center. Geriatr Orthop Surg Rehabil. 2020;11:2151459320927383. doi: 10.1177/2151459320927383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Anderson ME, Mcdevitt K, Cumbler E, Bennett H, Robison Z, Gomez B, et al. Geriatric hip fracture care: fixing a fragmented system. Perm J. 2017;21:16–104. doi: 10.7812/TPP/16-104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Aubry-Rozier B, Stoll D, Gonzalez Rodriguez E, Hans D, Prudent V, Seuret A, et al. Impact of a fracture liaison service on patient management after an osteoporotic fracture: the CHUV FLS. Swiss Med Wkly. 2018;148:w14579. doi: 10.4414/smw.2018.14579. [DOI] [PubMed] [Google Scholar]
- 34.Soong C, Cram P, Chezar K, Tajammal F, Exconde K, Matelski J, et al. Impact of an integrated hip fracture inpatient program on length of stay and costs. J Orthop Trauma. 2016;30:647–652. doi: 10.1097/BOT.0000000000000691. [DOI] [PubMed] [Google Scholar]
- 35.Beaupre LA, Moradi F, Khong H, Smith C, Evens L, Hanson HM, et al. Implementation of an in-patient hip fracture liaison services to improve initiation of osteoporosis medication use within 1-year of hip fracture: a population-based time series analysis using the RE-AIM framework. Arch Osteoporos. 2020;15:83. doi: 10.1007/s11657-020-00751-2. [DOI] [PubMed] [Google Scholar]
- 36.Inderjeeth CA, Raymond WD, Briggs AM, Geelhoed E, Oldham D, Mountain D. Implementation of the Western Australian Osteoporosis Model of Care: a fracture liaison service utilising emergency department information systems to identify patients with fragility fracture to improve current practice and reduce re-fracture rates: a 12-month analysis. Osteoporos Int. 2018;29:1759–1770. doi: 10.1007/s00198-018-4526-5. [DOI] [PubMed] [Google Scholar]
- 37.Greenspan SL, Singer A, Vujevich K, Marchand B, Thompson DA, Hsu YJ, et al. Implementing a fracture liaison service open model of care utilizing a cloud-based tool. Osteoporos Int. 2018;29:953–960. doi: 10.1007/s00198-017-4371-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Beaton DE, Mamdani M, Zheng H, Jaglal S, Cadarette SM, Bogoch ER, et al. Improvements in osteoporosis testing and care are found following the wide scale implementation of the Ontario Fracture Clinic Screening Program: an interrupted time series analysis. Medicine (Baltimore) 2017;96:e9012. doi: 10.1097/MD.0000000000009012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chan T, de Lusignan S, Cooper A, Elliott M. Improving osteoporosis management in primary care: an audit of the impact of a community based fracture liaison nurse. PLoS One. 2015;10:e0132146. doi: 10.1371/journal.pone.0132146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Schuijt HJ, Kusen J, van Hernen JJ, van der Vet P, Geraghty O, Smeeing DPJ, et al. Orthogeriatric trauma unit improves patient outcomes in geriatric hip fracture patients. Geriatr Orthop Surg Rehabil. 2020;11:2151459320949476. doi: 10.1177/2151459320949476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rotman-Pikielny P, Frankel M, Lebanon OT, Yaacobi E, Tamar M, Netzer D, et al. Orthopedic-metabolic collaborative management for osteoporotic hip fracture. Endocr Pract. 2018;24:718–725. doi: 10.4158/EP-2018-0082. [DOI] [PubMed] [Google Scholar]
- 42.Naranjo A, Fernández-Conde S, Ojeda S, Torres-Hernández L, Hernández-Carballo C, Bernardos I, et al. Preventing future fractures: effectiveness of an orthogeriatric fracture liaison service compared to an outpatient fracture liaison service and the standard management in patients with hip fracture. Arch Osteoporos. 2017;12:112. doi: 10.1007/s11657-017-0373-9. [DOI] [PubMed] [Google Scholar]
- 43.Davidson E, Seal A, Doyle Z, Fielding K, McGirr J. Prevention of osteoporotic refractures in regional Australia. Aust J Rural Health. 2017;25:362–368. doi: 10.1111/ajr.12355. [DOI] [PubMed] [Google Scholar]
- 44.Svenøy S, Watne LO, Hestnes I, Westberg M, Madsen JE, Frihagen F. Results after introduction of a hip fracture care pathway: comparison with usual care. Acta Orthop. 2020;91:139–145. doi: 10.1080/17453674.2019.1710804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Vaculík J, Stepan JJ, Dungl P, Majerníček M, Čelko A, Džupa V. Secondary fracture prevention in hip fracture patients requires cooperation from general practitioners. Arch Osteoporos. 2017;12:49. doi: 10.1007/s11657-017-0346-z. [DOI] [PubMed] [Google Scholar]
- 46.Sietsema DL, Araujo AB, Wang L, Boytsov NN, Pandya SA, Haynes VS, et al. The effectiveness of a private orthopaedic practice-based osteoporosis management service to reduce the risk of subsequent fractures. J Bone Joint Surg Am. 2018;100:1819–1828. doi: 10.2106/JBJS.17.01388. [DOI] [PubMed] [Google Scholar]
- 47.Amphansap T, Stitkitti N, Arirachakaran A. The effectiveness of Police General Hospital’s fracture liaison service (PGH’s FLS) implementation after 5 years: a prospective cohort study. Osteoporos Sarcopenia. 2020;6:199–204. doi: 10.1016/j.afos.2020.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Abrahamsen C, Nørgaard B, Draborg E, Nielsen MF. The impact of an orthogeriatric intervention in patients with fragility fractures: a cohort study. BMC Geriatr. 2019;19:268. doi: 10.1186/s12877-019-1299-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Baroni M, Serra R, Boccardi V, Ercolani S, Zengarini E, Casucci P, et al. The orthogeriatric comanagement improves clinical outcomes of hip fracture in older adults. Osteoporos Int. 2019;30:907–916. doi: 10.1007/s00198-019-04858-2. [DOI] [PubMed] [Google Scholar]
- 50.Sanli I, van Helden SH, Ten Broeke RHM, Geusens P, Van den Bergh JPW, Brink PRG, et al. The role of the Fracture Liaison Service (FLS) in subsequent fracture prevention in the extreme elderly. Aging Clin Exp Res. 2019;31:1105–1111. doi: 10.1007/s40520-018-1054-2. [DOI] [PubMed] [Google Scholar]
- 51.Hawley S, Javaid MK, Prieto-Alhambra D, Lippett J, Sheard S, Arden NK, et al. Clinical effectiveness of orthogeriatric and fracture liaison service models of care for hip fracture patients: population-based longitudinal study. Age Ageing. 2016;45:236–242. doi: 10.1093/ageing/afv204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Shigemoto K, Sawaguchi T, Goshima K, Iwai S, Nakanishi A, Ueoka K. The effect of a multidisciplinary approach on geriatric hip fractures in Japan. J Orthop Sci. 2019;24:280–285. doi: 10.1016/j.jos.2018.09.012. [DOI] [PubMed] [Google Scholar]
- 53.Ganda K, Puech M, Chen JS, Speerin R, Bleasel J, Center JR, et al. Models of care for the secondary prevention of osteoporotic fractures: a systematic review and meta-analysis. Osteoporos Int. 2013;24:393–406. doi: 10.1007/s00198-012-2090-y. [DOI] [PubMed] [Google Scholar]
- 54.Bell K, Strand H, Inder WJ. Effect of a dedicated osteoporosis health professional on screening and treatment in outpatients presenting with acute low trauma non-hip fracture: a systematic review. Arch Osteoporos. 2014;9:167. doi: 10.1007/s11657-013-0167-7. [DOI] [PubMed] [Google Scholar]
- 55.Chang YF, Huang CF, Hwang JS, Kuo JF, Lin KM, Huang HC, et al. Fracture liaison services for osteoporosis in the Asia-Pacific region: current unmet needs and systematic literature review. Osteoporos Int. 2018;29:779–792. doi: 10.1007/s00198-017-4347-y. [DOI] [PubMed] [Google Scholar]
- 56.Wu CH, Chen CH, Chen PH, Yang JJ, Chang PC, Huang TC, et al. Identifying characteristics of an effective fracture liaison service: systematic literature review. Osteoporos Int. 2018;29:1023–1047. doi: 10.1007/s00198-017-4370-z. [DOI] [PubMed] [Google Scholar]
- 57.Wu CH, Tu ST, Chang YF, Chan DC, Chien JT, Lin CH, et al. Fracture liaison services improve outcomes of patients with osteoporosis-related fractures: a systematic literature review and meta-analysis. Bone. 2018;111:92–100. doi: 10.1016/j.bone.2018.03.018. [DOI] [PubMed] [Google Scholar]
- 58.Talevski J, Sanders KM, Duque G, Connaughton C, Beauchamp A, Green D, et al. Effect of clinical care pathways on quality of life and physical function after fragility fracture: a meta-analysis. J Am Med Dir Assoc. 2019;20:926.e1–926.e11. doi: 10.1016/j.jamda.2019.02.022. [DOI] [PubMed] [Google Scholar]
- 59.Laslett LL, Whitham JN, Gibb C, Gill TK, Pink JA, McNeil JD. Improving diagnosis and treatment of osteoporosis: evaluation of a clinical pathway for low trauma fractures. Arch Osteoporos. 2007;2:1–6. [Google Scholar]
- 60.Davis JC, Guy P, Ashe MC, Liu-Ambrose T, Khan K. HipWatch: osteoporosis investigation and treatment after a hip fracture: a 6-month randomized controlled trial. J Gerontol A Biol Sci Med Sci. 2007;62:888–891. doi: 10.1093/gerona/62.8.888. [DOI] [PubMed] [Google Scholar]
- 61.Cranney A, Lam M, Ruhland L, Brison R, Godwin M, Harrison MM, et al. A multifaceted intervention to improve treatment of osteoporosis in postmenopausal women with wrist fractures: a cluster randomized trial. Osteoporos Int. 2008;19:1733–1740. doi: 10.1007/s00198-008-0669-0. [DOI] [PubMed] [Google Scholar]
- 62.Majumdar SR, Johnson JA, McAlister FA, Bellerose D, Russell AS, Hanley DA, et al. Multifaceted intervention to improve diagnosis and treatment of osteoporosis in patients with recent wrist fracture: a randomized controlled trial. CMAJ. 2008;178:569–575. doi: 10.1503/cmaj.070981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Morrish DW, Beaupre LA, Bell NR, Cinats JG, Hanley DA, Harley CH, et al. Facilitated bone mineral density testing versus hospital-based case management to improve osteoporosis treatment for hip fracture patients: additional results from a randomized trial. Arthritis Rheum. 2009;61:209–215. doi: 10.1002/art.24097. [DOI] [PubMed] [Google Scholar]
- 64.Yuksel N, Majumdar SR, Biggs C, Tsuyuki RT. Community pharmacist-initiated screening program for osteoporosis: randomized controlled trial. Osteoporos Int. 2010;21:391–398. doi: 10.1007/s00198-009-0977-z. [DOI] [PubMed] [Google Scholar]
- 65.Leslie WD, LaBine L, Klassen P, Dreilich D, Caetano PA. Closing the gap in postfracture care at the population level: a randomized controlled trial. CMAJ. 2012;184:290–296. doi: 10.1503/cmaj.111158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Roux S, Beaulieu M, Beaulieu MC, Cabana F, Boire G. Priming primary care physicians to treat osteoporosis after a fragility fracture: an integrated multidisciplinary approach. J Rheumatol. 2013;40:703–711. doi: 10.3899/jrheum.120908. [DOI] [PubMed] [Google Scholar]
- 67.Hawker G, Ridout R, Ricupero M, Jaglal S, Bogoch E. The impact of a simple fracture clinic intervention in improving the diagnosis and treatment of osteoporosis in fragility fracture patients. Osteoporos Int. 2003;14:171–178. doi: 10.1007/s00198-003-1377-4. [DOI] [PubMed] [Google Scholar]
- 68.Majumdar SR, Rowe BH, Folk D, Johnson JA, Holroyd BH, Morrish DW, et al. A controlled trial to increase detection and treatment of osteoporosis in older patients with a wrist fracture. Ann Intern Med. 2004;141:366–373. doi: 10.7326/0003-4819-141-5-200409070-00011. [DOI] [PubMed] [Google Scholar]
- 69.Jaglal SB, Hawker G, Bansod V, Salbach NM, Zwarenstein M, Carroll J, et al. A demonstration project of a multi-component educational intervention to improve integrated post-fracture osteoporosis care in five rural communities in Ontario. Canada. Osteoporos Int. 2009;20:265–274. doi: 10.1007/s00198-008-0654-7. [DOI] [PubMed] [Google Scholar]
- 70.Beaton DE, Vidmar M, Pitzul KB, Sujic R, Rotondi NK, Bogoch ER, et al. Addition of a fracture risk assessment to a coordinator’s role improved treatment rates within 6 months of screening in a fragility fracture screening program. Osteoporos Int. 2017;28:863–869. doi: 10.1007/s00198-016-3794-1. [DOI] [PubMed] [Google Scholar]
- 71.Amphansap T, Stitkitti N, Dumrongwanich P. Evaluation of Police General Hospital’s Fracture Liaison Service (PGH’s FLS): the first study of a Fracture Liaison Service in Thailand. Osteoporos Sarcopenia. 2016;2:238–243. doi: 10.1016/j.afos.2016.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Queally JM, Kiernan C, Shaikh M, Rowan F, Bennett D. Initiation of osteoporosis assessment in the fracture clinic results in improved osteoporosis management: a randomised controlled trial. Osteoporos Int. 2013;24:1089–1094. doi: 10.1007/s00198-012-2238-9. [DOI] [PubMed] [Google Scholar]
- 73.Brankin E, Mitchell C, Munro R, Lanarkshire Osteoporosis Service Closing the osteoporosis management gap in primary care: a secondary prevention of fracture programme. Curr Med Res Opin. 2005;21:475–482. doi: 10.1185/030079905X38150. [DOI] [PubMed] [Google Scholar]
- 74.Murray AW, McQuillan C, Kennon B, Gallacher SJ. Osteoporosis risk assessment and treatment intervention after hip or shoulder fracture. A comparison of two centres in the United Kingdom. Injury. 2005;36:1080–1084. doi: 10.1016/j.injury.2005.03.012. [DOI] [PubMed] [Google Scholar]
- 75.Wallace I, Callachand F, Elliott J, Gardiner P. An evaluation of an enhanced fracture liaison service as the optimal model for secondary prevention of osteoporosis. JRSM Short Rep. 2011;2:8. doi: 10.1258/shorts.2010.010063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ruggiero C, Zampi E, Rinonapoli G, Baroni M, Serra R, Zengarini E, et al. Fracture prevention service to bridge the osteoporosis care gap. Clin Interv Aging. 2015;10:1035–1042. doi: 10.2147/CIA.S76695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Miki RA, Oetgen ME, Kirk J, Insogna KL, Lindskog DM. Orthopaedic management improves the rate of early osteoporosis treatment after hip fracture. A randomized clinical trial. J Bone Joint Surg Am. 2008;90:2346–2353. doi: 10.2106/JBJS.G.01246. [DOI] [PubMed] [Google Scholar]
- 78.Kamel HK, Hussain MS, Tariq S, Perry HM, Morley JE. Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture. Am J Med. 2000;109:326–328. doi: 10.1016/s0002-9343(00)00457-5. [DOI] [PubMed] [Google Scholar]
- 79.Harrington JT, Barash HL, Day S, Lease J. Redesigning the care of fragility fracture patients to improve osteoporosis management: a health care improvement project. Arthritis Rheum. 2005;53:198–204. doi: 10.1002/art.21072. [DOI] [PubMed] [Google Scholar]
- 80.Johnson SL, Petkov VI, Williams MI, Via PS, Adler RA. Improving osteoporosis management in patients with fractures. Osteoporos Int. 2005;16:1079–1085. doi: 10.1007/s00198-004-1814-z. [DOI] [PubMed] [Google Scholar]
- 81.Streeten EA, Mohamed A, Gandhi A, Orwig D, Sack P, Sterling R, et al. The inpatient consultation approach to osteoporosis treatment in patients with a fracture. Is automatic consultation needed? J Bone Joint Surg Am. 2006;88:1968–1974. doi: 10.2106/JBJS.E.01072. [DOI] [PubMed] [Google Scholar]
- 82.Tosi LL, Gliklich R, Kannan K, Koval KJ. The American Orthopaedic Association’s “own the bone” initiative to prevent secondary fractures. J Bone Joint Surg Am. 2008;90:163–173. doi: 10.2106/JBJS.G.00682. [DOI] [PubMed] [Google Scholar]
- 83.Roy A, Heckman MG, O’Connor MI. Optimizing screening for osteoporosis in patients with fragility hip fracture. Clin Orthop Relat Res. 2011;469:1925–1930. doi: 10.1007/s11999-011-1839-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Heilmann RMF, Friesleben CR, Billups SJ. Impact of a pharmacist-directed intervention in postmenopausal women after fracture. Am J Health Syst Pharm. 2012;69:504–509. doi: 10.2146/ajhp110309. [DOI] [PubMed] [Google Scholar]
- 85.Hofflich HL, Oh DK, Choe CH, Clay B, Tibble C, Kulasa KM, et al. Using a triggered endocrinology service consultation to improve the evaluation, management, and follow-up of osteoporosis in hip-fracture patients. Jt Comm J Qual Patient Saf. 2014;40:228–234. doi: 10.1016/s1553-7250(14)40030-8. [DOI] [PubMed] [Google Scholar]
- 86.Cosman F, Nicpon K, Nieves JW. Results of a fracture liaison service on hip fracture patients in an open healthcare system. Aging Clin Exp Res. 2017;29:331–334. doi: 10.1007/s40520-016-0545-2. [DOI] [PubMed] [Google Scholar]
- 87.Jones G, Warr S, Francis E, Greenaway T. The effect of a fracture protocol on hospital prescriptions after minimal trauma fractured neck of the femur: a retrospective audit. Osteoporos Int. 2005;16:1277–1280. doi: 10.1007/s00198-005-1960-y. [DOI] [PubMed] [Google Scholar]
- 88.Fisher AA, Davis MW, Rubenach SE, Sivakumaran S, Smith PN, Budge MM. Outcomes for older patients with hip fractures: the impact of orthopedic and geriatric medicine cocare. J Orthop Trauma. 2006;20:172–8; discussion 179-180. doi: 10.1097/01.bot.0000202220.88855.16. [DOI] [PubMed] [Google Scholar]
- 89.Lih A, Nandapalan H, Kim M, Yap C, Lee P, Ganda K, et al. Targeted intervention reduces refracture rates in patients with incident non-vertebral osteoporotic fractures: a 4-year prospective controlled study. Osteoporos Int. 2011;22:849–858. doi: 10.1007/s00198-010-1477-x. [DOI] [PubMed] [Google Scholar]
- 90.Van der Kallen J, Giles M, Cooper K, Gill K, Parker V, Tembo A, et al. A fracture prevention service reduces further fractures two years after incident minimal trauma fracture. Int J Rheum Dis. 2014;17:195–203. doi: 10.1111/1756-185X.12101. [DOI] [PubMed] [Google Scholar]
- 91.Astrand J, Nilsson J, Thorngren KG. Screening for osteoporosis reduced new fracture incidence by almost half: a 6-year follow-up of 592 fracture patients from an osteoporosis screening program. Acta Orthop. 2012;83:661–665. doi: 10.3109/17453674.2012.747922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rolnick SJ, Kopher R, Jackson J, Fischer LR, Compo R. What is the impact of osteoporosis education and bone mineral density testing for postmenopausal women in a managed care setting? Menopause. 2001;8:141–148. doi: 10.1097/00042192-200103000-00010. [DOI] [PubMed] [Google Scholar]
- 93.Jachna CM, Whittle J, Lukert B, Graves L, Bhargava T. Effect of hospitalist consultation on treatment of osteoporosis in hip fracture patients. Osteoporos Int. 2003;14:665–671. doi: 10.1007/s00198-003-1413-4. [DOI] [PubMed] [Google Scholar]
- 94.Ganda K, Schaffer A, Pearson S, Seibel MJ. Compliance and persistence to oral bisphosphonate therapy following initiation within a secondary fracture prevention program: a randomised controlled trial of specialist vs. non-specialist management. Osteoporos Int. 2014;25:1345–1355. doi: 10.1007/s00198-013-2610-4. [DOI] [PubMed] [Google Scholar]
- 95.Nakayama A, Major G, Holliday E, Attia J, Bogduk N. Evidence of effectiveness of a fracture liaison service to reduce the re-fracture rate. Osteoporos Int. 2016;27:873–879. doi: 10.1007/s00198-015-3443-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Goltz L, Degenhardt G, Maywald U, Kirch W, Schindler C. Evaluation of a program of integrated care to reduce recurrent osteoporotic fractures. Pharmacoepidemiol Drug Saf. 2013;22:263–270. doi: 10.1002/pds.3399. [DOI] [PubMed] [Google Scholar]
- 97.Huntjens KMB, van Geel TCM, Geusens PP, Winkens B, Willems P, van den Bergh J, et al. Impact of guideline implementation by a fracture nurse on subsequent fractures and mortality in patients presenting with non-vertebral fractures. Injury. 2011;42(Suppl 4):S39–S43. doi: 10.1016/S0020-1383(11)70011-0. [DOI] [PubMed] [Google Scholar]
- 98.Huntjens KMB, van Geel TACM, van den Bergh JPW, van Helden S, Willems P, Winkens B, et al. Fracture liaison service: impact on subsequent nonvertebral fracture incidence and mortality. J Bone Joint Surg Am. 2014;96:e29. doi: 10.2106/JBJS.L.00223. [DOI] [PubMed] [Google Scholar]
- 99.Henderson CY, Shanahan E, Butler A, Lenehan B, O’Connor M, Lyons D, et al. Dedicated orthogeriatric service reduces hip fracture mortality. Ir J Med Sci. 2017;186:179–184. doi: 10.1007/s11845-016-1453-3. [DOI] [PubMed] [Google Scholar]
- 100.Vidán M, Serra JA, Moreno C, Riquelme G, Ortiz J. Efficacy of a comprehensive geriatric intervention in older patients hospitalized for hip fracture: a randomized, controlled trial. J Am Geriatr Soc. 2005;53:1476–1482. doi: 10.1111/j.1532-5415.2005.53466.x. [DOI] [PubMed] [Google Scholar]
- 101.Axelsson KF, Jacobsson R, Lund D, Lorentzon M. Effectiveness of a minimal resource fracture liaison service. Osteoporos Int. 2016;27:3165–3175. doi: 10.1007/s00198-016-3643-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Majumdar SR, Johnson JA, Bellerose D, McAlister FA, Russell AS, Hanley DA, et al. Nurse case-manager vs multifaceted intervention to improve quality of osteoporosis care after wrist fracture: randomized controlled pilot study. Osteoporos Int. 2011;22:223–230. doi: 10.1007/s00198-010-1212-7. [DOI] [PubMed] [Google Scholar]
- 103.van Helden S, Cauberg E, Geusens P, Winkes B, van der Weijden T, Brink P. The fracture and osteoporosis outpatient clinic: an effective strategy for improving implementation of an osteoporosis guideline. J Eval Clin Pract. 2007;13:801–805. doi: 10.1111/j.1365-2753.2007.00784.x. [DOI] [PubMed] [Google Scholar]
- 104.Sidwell AI, Wilkinson TJ, Hanger HC. Secondary prevention of fractures in older people: evaluation of a protocol for the investigation and treatment of osteoporosis. Intern Med J. 2004;34:129–132. doi: 10.1111/j.1444-0903.2004.00554.x. [DOI] [PubMed] [Google Scholar]
- 105.Merle B, Chapurlat R, Vignot E, Thomas T, Haesebaert J, Schott AM. Post-fracture care: do we need to educate patients rather than doctors? The PREVOST randomized controlled trial. Osteoporos Int. 2017;28:1549–1558. doi: 10.1007/s00198-017-3953-z. [DOI] [PubMed] [Google Scholar]
- 106.Sistema Nazionale Linee Guida (2021). Diagnosi, Stratificazione del rischio e continuità assistenziale delle Fratture da Fragilità. https://snlg.iss.it/. Accessed 20 Dec 2022
- 107.Li N, Hiligsmann M, Boonen A, van Oostwaard MM, de Bot RTAL, Wyers CE, et al. The impact of fracture liaison services on subsequent fractures and mortality: a systematic literature review and meta-analysis. Osteoporos Int. 2021;32:1517–1530. doi: 10.1007/s00198-021-05911-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Åkesson KE, Ganda K, Deignan C, Oates MK, Volpert A, Brooks K, et al. Post-fracture care programs for prevention of subsequent fragility fractures: a literature assessment of current trends. Osteoporos Int. 2022;33(8):1659–1676. doi: 10.1007/s00198-022-06358-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Javaid MK, Sami A, Lems W, Mitchell P, Thomas T, Singer A, et al. A patient-level key performance indicator set to measure the effectiveness of fracture liaison services and guide quality improvement: a position paper of the IOF Capture the Fracture Working Group, National Osteoporosis Foundation and Fragility Fracture Network. Osteoporos Int. 2020;31:1193–1204. doi: 10.1007/s00198-020-05377-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Eisman JA, Bogoch ER, Dell R, Harrington JT, McKinney RE, McLellan A, et al. Making the first fracture the last fracture: ASBMR task force report on secondary fracture prevention. J Bone Miner Res. 2012;27:2039–2046. doi: 10.1002/jbmr.1698. [DOI] [PubMed] [Google Scholar]
- 111.Lems WF, Dreinhöfer KE, Bischoff-Ferrari H, Blauth M, Czerwinski E, da Silva J, et al. EULAR/EFORT recommendations for management of patients older than 50 years with a fragility fracture and prevention of subsequent fractures. Ann Rheum Dis. 2017;76:802–810. doi: 10.1136/annrheumdis-2016-210289. [DOI] [PubMed] [Google Scholar]
- 112.Kanis JA, Cooper C, Rizzoli R, Reginster JY. Scientific Advisory Board of the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) and the Committees of Scientific Advisors and National Societies of the International Osteoporosis Foundation (IOF). Executive summary of European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Aging Clin Exp Res. 2019;31:15–17. doi: 10.1007/s40520-018-1109-4. [DOI] [PubMed] [Google Scholar]
- 113.Kanis JA, Cooper C, Rizzoli R, Abrahamsen B, Al-Daghri NM, Brandi ML, et al. Identification and management of patients at increased risk of osteoporotic fracture: outcomes of an ESCEO expert consensus meeting. Osteoporos Int. 2017;28:2023–2034. doi: 10.1007/s00198-017-4009-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Anderson PA, Freedman BA, Brox WT, Shaffer WO. Osteoporosis: recent recommendations and positions of the American Society for Bone and Mineral Research and the International Society for Clinical Densitometry. J Bone Joint Surg Am. 2021;103:741–747. doi: 10.2106/JBJS.20.01248. [DOI] [PubMed] [Google Scholar]
- 115.Shoback D, Rosen CJ, Black DM, Cheung AM, Murad MH, Eastell R. Pharmacological Management of Osteoporosis in Postmenopausal Women: an Endocrine Society Guideline update. J Clin Endocrinol Metab. 2020;105:dgaa048. doi: 10.1210/clinem/dgaa048. [DOI] [PubMed] [Google Scholar]
- 116.Arceo-Mendoza RM, Camacho PM. Postmenopausal osteoporosis: latest guidelines. Endocrinol Metab Clin North Am. 2021;50:167–178. doi: 10.1016/j.ecl.2021.03.009. [DOI] [PubMed] [Google Scholar]
- 117.Akesson K, Marsh D, Mitchell PJ, McLellan AR, Stenmark J, Pierroz DD, et al. Capture the Fracture: a Best Practice Framework and global campaign to break the fragility fracture cycle. Osteoporos Int. 2013;24:2135–2152. doi: 10.1007/s00198-013-2348-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Asia Pacific Fragility Fracture Alliance (APFFA). The Hip Fracture Registry (HFR) Toolbox. Available from:https://apfracturealliance.org/hfr-toolbox/. [cited 22 May 2023].
- 119.Australian and New Zealand Fragility Fracture Registry (ANZFFR). Fragility Fracture Registry NZ. Available from: https://fragilityfracture.co.nz/. [cited 22 May 2023].
- 120.Australian Fragility Fracture Foundation. Australian & New Zealand Fragility Fracture Registry. Available from: https://fragilityfracture.com.au/. [cited 22 May 2023].
- 121.The Irish Institute of Trauma and Orthopaedic Surgery. Fracture Liaison Service Database. Available from: https://iitos.com/fracture-liaison-service-database-2/. [cited 22 May 2023].
- 122.Royal College of Physicians. Fracture Liaison Service Database (FLS-DB). Available from: https://www.rcplondon.ac.uk/projects/fracture-liaison-service-database-fls-db. [cited 22 May 2023].
- 123.American Orthopaedic Association. Own the Bone. Available from: https://www.ownthebone.org/. [cited 22 May 2023].
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Table S1. Prisma Checklist. (PDF 888 kb)
Supplemental Table S2. Search Strategy. Supplemental Table S3. Characteristics of included studies. Supplemental Table S4. Quality evaluation. Supplemental Table S5. Summary of findings, GRADE approach. Supplemental Table S6. Summary results. Supplemental Figure S1. Funnel plot and Egger’s test. Supplemental Figure S2. BMD testing rate in FLS, RCT studies. Supplemental Figure S3. Antiosteoporotic initiation in FLS, RCT studies. Supplemental Figure S4. Antiosteoporotic adherence in FLS, RCT studies. Supplemental Figure S5. Subsequent fracture risk in FLS, RCT studies. Supplemental Figure S6. Mortality risk in FLS, RCT studies. Complete list of experts involved. (DOCX 353 kb)