Synopsis
While animals swim, crawl, walk, and fly with apparent ease, building robots capable of robust locomotion remains a significant challenge. In this review, we draw attention to mechanosensation—the sensing of mechanical forces generated within and outside the body—as a key sense that enables robust locomotion in animals. We discuss differences between mechanosensation in animals and current robots with respect to (1) the encoding properties and distribution of mechanosensors and (2) the integration and regulation of mechanosensory feedback. We argue that robotics would benefit greatly from a detailed understanding of these aspects in animals. To that end, we highlight promising experimental and engineering approaches to study mechanosensation, emphasizing the mutual benefits for biologists and engineers that emerge from moving forward together.
Introduction
Locomotion in the real world is a significant challenge for current robots because natural environments are highly complex and can change in unpredictable ways (Jayaram and Full 2016). This requires robots to be robust—resistant to perturbations and effective in a wide range of conditions (Jen 2005). While some of the most impressive robots today, like Boston Dynamics’ Atlas (Guizzo 2019) or ANYbotics’ ANYmal (Hutter et al. 2017), are beginning to deliver robust locomotion in natural environments and redefine robot agility, robots still lack the efficiency and effectiveness of animals (Ijspeert 2014; Buschmann et al. 2015). For example, it is still difficult for robots to cope with unexpected perturbations, such as terrain deformation and foot slippage (Ohradzansky et al. 2021; Tranzatto et al. 2022; Chung et al. 2023).
In contrast to robots, animals swim, crawl, walk, and fly robustly with apparent ease. Key to their outstanding performance is a combination of fine-tuned mechanical and sensory feedback (Dickinson et al. 2000). Mechanical feedback is provided by the intrinsic, viscoelastic properties of the musculoskeletal system (Nishikawa et al. 2007). A fast running cockroach, for example, can self-stabilize within milliseconds after being pushed sideways thanks in part due to its viscoelastic legs (Jindrich and Full 2002). From an engineering perspective (Holmes et al. 2006), mechanical feedback is interesting because it helps robots recover from certain perturbations extremely fast at no additional computational cost (Jayaram et al. 2018). As such, it has successfully inspired generations of highly capable multi-legged robots (Altendorfer et al. 2001). Sensory feedback, on the other hand, allows animals to cope with unpredictable environments more broadly. Animals utilize sensory feedback from a variety of different modalities to guide and stabilize locomotion (Dickinson et al. 2000).
In this review, we focus on mechanosensation, the sensing of mechanical forces generated within and outside the body. Mechanosensation is a critical type of sensory feedback for locomotion across animals (Fig. 1; Tuthill and Wilson 2016; Knafo and Wyart 2018; Tuthill and Azim 2018; Boublil et al. 2021; Frigon et al. 2021; Agrawal and Tuthill 2022). Most animals, including humans, rely heavily on mechanosensation for robust locomotion. For example, human patients lacking mechanosensory feedback due to genetic mutations (Chesler et al. 2016) or viral infections (Cole and Katifi 1991; Lajoie et al. 1996) are typically unable to walk. If they do recover, locomotion is anything but robust—each step requires precise planning and visual control. Lack of mechanosensory feedback causes locomotion deficits across species, whether animals walk, fly, crawl, or swim (e.g., Dickinson 1999; Hughes and Thomas 2007; Mendes et al. 2013; Wu et al. 2021; Santuz et al. 2022).
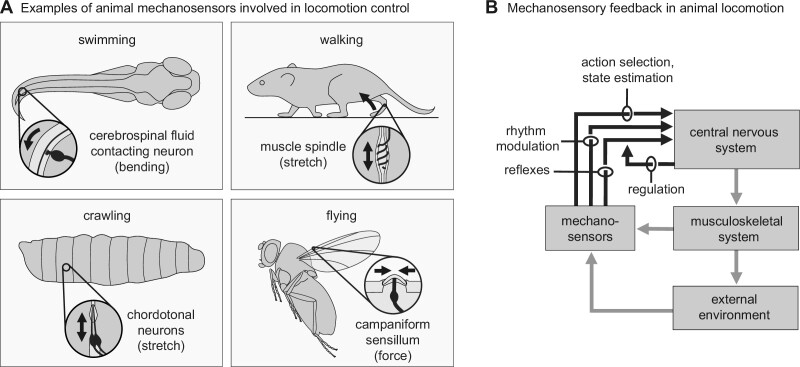
Fig. 1.
Mechanosensory control of locomotion in animals. (A) Animal mechanosensors come in many shapes and forms. Embedded deep within the musculoskeletal system, they detect external and internal forces important for controlling locomotion. Top left: cerebrospinal fluid contacting neuron in the central canal of a larval zebrafish encoding axial bending in the spinal cord through coupling to an internal acellular thread called the Reissner fiber (top view, adapted from Wu et al. 2021). Bottom left: chordotonal neurons in a Drosophila larva encoding muscle stretch (adapted from Agrawal and Tuthill 2022). Top right: muscle spindle in a mouse leg encoding muscle stretch. Bottom right: campaniform sensillum on a Drosophila wing encoding aerodynamic forces. (B) Across species and types of locomotion, mechanosensory feedback informs locomotor circuits in the central nervous system at multiple levels and timescales. Mechanosensory feedback mediates reflexes, shapes internal locomotor rhythms, and contributes to action selection and state estimation. Feedback is flexibly regulated to meet behavioral demands. Control diagram adapted from Dickinson et al. (2000).
In comparison to other modalities like vision, mechanosensation is typically fast, computationally cheap, and equally effective under varying external conditions. These advantages have not been lost on engineers. In fact, mechanosensors—primarily in the form of inertial measurement units (IMUs)—are integral parts of many robotic controller designs. These robotic sensors may even outperform animal mechanosensors in terms of noise and conduction delays (Buschmann et al. 2015). Nevertheless, it has been challenging to take full advantage of mechanosensation in robots to enable animal-like locomotion. Why?
In this review, we draw attention to key differences between mechanosensation in current robots and animals related to (1) the encoding properties and distribution of mechanosensors and (2) the integration and regulation of mechanosensory feedback. We argue that inspiring the next generation of robust robots will be accelerated by a detailed understanding of these aspects in animals. To that end, we highlight promising experimental and engineering approaches to study mechanosensation, emphasizing the mutual benefits for biologists and engineers that emerge from moving forward together.
Encoding properties and distribution of mechanosensors
Encoding properties of mechanosensors
Engineered mechanosensors typically directly encode a physical parameter with fixed sensitivity over the whole parameter range. For example, legged robots are routinely equipped with sensors that linearly encode joint angles or joint forces/torques (Buschmann et al. 2015). At first sight, animal mechanosensors may appear to function similarly. For example, the responses of mammalian muscle spindles and insect chordotonal organs are related to joint angles, while those of mammalian Golgi tendon organs and insect campaniform sensilla are related to forces (Tuthill and Azim 2018). However, unlike engineered sensors, animal mechanosensors often encode physical parameters non-linearly and dependent on stimulus history. Campaniform sensilla, for example, encode the rate of change of force with a power–law relation, their firing rate decreases over time when the force remains constant (adaptation), and they encode the same magnitude of force differently depending on whether it is reached during a force increase or a force decrease (hysteresis; Ridgel et al. 2000). Such complex encoding properties may result from a combination of neural properties—which are in part determined by the neurons’ mechanically gated ion channels (Walker et al. 2000; Cheng et al. 2010; Faucherre et al. 2013; Akitake et al. 2015; Delmas et al. 2022)—and mechanical properties (Sane and McHenry 2009; Barth 2019). Mechanical properties directly affect sensing capabilities because mechanosensory neurons are embedded deep within muscles, tendons, joints, the skin, or the skeleton (Fig. 1A). Campaniform sensilla, for example, are embedded in the insect exoskeleton, including in the legs and in the wings (Dinges et al. 2021). Accordingly, the sensitivity of an individual sensillum strongly depends on how the surrounding cuticle deforms in response to external forces (Dickerson et al. 2021; Dinges et al. 2022).
Distribution of mechanosensors
Engineered sensors differ from animal mechanosensors not only in terms of their encoding properties but also in terms of their distribution across the body. Current robots are equipped with comparatively few mechanosensors. Legged robots, for example, may be equipped with a single sensor per leg joint to encode joint position and/or torque, and a sensor in the foot to encode ground contact (Buschmann et al. 2015). In contrast, animal mechanosensors are orders of magnitudes more numerous. Depending on its size, a single mammalian muscle may contain up to 500 muscle spindles (Prochazka 1996). Even a tiny fruit fly leg contains several hundreds of mechanosensory neurons (Phelps et al. 2021). The higher number of mechanosensors in animals likely contributes to their robust locomotion: Should a subset of mechanosensory neurons be damaged, other neurons can take over with no apparent effect on locomotor performance. In fact, this redundancy makes it difficult to study how specific mechanosensory neurons contribute to locomotion control—a challenge recognized already in early functional studies (Delcomyn 1985) and still present today (see below). But individual mechanosensory neurons of a given type are not necessarily uniform. Among other things, they may differ in their sensitivity. This is seen in many mechanosensory neurons, like the position-sensitive neurons of chordotonal organs in insect legs. With different peak sensitivities, these neurons can divide up the stimulus range, a strategy known as range fractionation (Matheson 1992; Mamiya et al. 2022). As a group, the mechanosensory neurons can then encode a parameter such as joint angle precisely over a broad range.
Moving forward
Although much is known about the encoding properties and distribution of mechanosensors in animals, many details remain unclear, which in turn hampers broader robotic implementation. How exactly do neural and mechanical properties interact to determine the sensing capabilities of mechanosensory neurons? What are the advantages of non-linear, adaptive encoding? How many sensors are needed to control locomotion, and where should they be placed? Moving forward, we believe that computational models and new techniques to fabricate robotic sensors will provide important insights into these questions.
Moving forward with computational models
Computational models are valuable tools to better understand the encoding properties and distribution of animal mechanosensors. Muscle spindles are a good example. Their diverse and complex responses are well documented, but it has been challenging to understand how exactly the responses emerge from body movements in different contexts. Using a computational model of the spindle, Blum et al. (2020) were able to show that the responses can in fact emerge from first principles of muscle contractile mechanics. In another recent example, Mamiya et al. (2022) took advantage of a finite element model based on an X-ray dataset of the fruit fly leg to better understand how leg movement is translated into the very different responses of neighboring neurons of a chordotonal organ. The model revealed that a mechanical structure in the leg effectively pre-processes movement-related signals to give rise to position-sensitivity in some neurons and vibration-sensitivity in others.
To study the potential advantages of biological encoding over standard engineering encoding, Szczecinski et al. (2021) developed a simple computational model that emulates how insect campaniform sensilla adapt to constant forces. The model was used as a real-time filter on a robot leg to encode feedback from strain sensors “campaniform-sensilla-style” (Zyhowski et al. 2023). Thanks to the adaptation, the bio-inspired sensors robustly signaled only relative changes in strain on the leg, making them essentially self-calibrating across contexts. In the future, this may make it easier to determine when a leg is in ground contact and thereby improve the control of inter-leg coordination in walking robots. In another study, Mohren et al. (2018) used a computational model to better understand the encoding and distribution of campaniform sensilla on insect wings. The model revealed that only few sensilla in specific locations are needed to read out information about body movements important for flight control, with biological sensors providing significant advantages over engineered sensors that directly encode strain.
These examples illustrate how computational models can help us understand the encoding properties and distribution of animal mechanosensors and, in turn, provide valuable inspiration for robust sensing in robots. An advantage of computational models is that neural and mechanical properties can be easily changed to explore how they affect sensing capabilities in different contexts (e.g., Weber et al. 2021).
Moving forward with new fabrication and computing techniques
On the engineering side, new fabrication techniques, such as laser micromachined stack laminate manufacturing (Wood et al. 2008; Sreetharan et al. 2012) and 3D printing (Valentine et al. 2017; Skylar-Scott et al. 2019), now enable fast, flexible, and low-cost development of materials that integrate sensors throughout the structure (Gafford et al. 2014; Muth et al. 2014; McDonnell et al. 2022), resulting in smart materials capable of functioning as machines themselves (McEvoy and Correll 2015; McCracken et al. 2020). Embedding sensors directly into a robot’s body may enable the use of generic sensors that—much like the campaniform sensilla discussed above—automatically adapt to behaviorally relevant stimuli via the mechanics of the body (Dickerson et al. 2021), thereby reducing the computational burdens for control. Moreover, these accessible and easy-to-use prototyping techniques are capable of creating bio-inspired mechanosensors that approach sensitivities of biological sensors (Kang et al. 2014; Liu et al. 2022). With such techniques, we expect to see rapid progress in engineered systems utilizing a large number of distributed sensors (Gao et al. 2016; Shih et al. 2020) that have the potential to outperform state-of-the-art systems in terms of adaptability and resilience. Modern bio-inspired sensors are even starting to be capable of simulating the responses of biological mechanosensitive ion channels (Marcotte et al. 2020), which could lead to higher fidelity robophysical models. With computational techniques such as neuromorphic computing (Davies et al. 2021) becoming more accessible, mechanosensory neuron-like encoding can be readily implemented in robotic processors (Dupeyroux et al. 2022). As these robotic implementations mature in design complexity and fidelity, they can increasingly serve as effective robophysical models to elucidate principles of biological mechanosensors, because they are fundamentally grounded in the physics of the real world (Aguilar et al. 2016).
Integration and regulation of mechanosensory feedback
Integration of mechanosensory feedback
In addition to advantageous encoding properties and distributions of mechanosensors, effective mechanosensation in animals is enabled by efficient integration of mechanosensory feedback at multiple levels and timescales to support various locomotor functions (Fig. 1B; Dallmann et al. 2021; Frigon et al. 2021).
At the lowest level, mechanosensory feedback is directly or indirectly integrated in motor neurons to enable fast reflexes that stabilize ongoing locomotion. For example, during the stance phase of walking, mammalian Golgi tendon organs and insect campaniform sensilla provide reflexive, positive feedback to motor neurons of antigravity muscles, thereby reinforcing body weight support (Duysens et al. 2000; Zill et al. 2004). This feedback automatically activates muscles more strongly when the mechanical demands on the body increase, such as when walking uphill or when carrying additional weight. Stabilizing reflexes are critical across species and types of locomotion. In zebrafish, mechanosensory neurons that contact the cerebrospinal fluid in the central canal and detect bending of the spine (Fig. 1A; Djenoune et al. 2014) project to occipital motor neurons in the hindbrain that control head position (Wu et al. 2021). Genetic removal of these neurons demonstrated that they are critical to stabilize posture against rolling during high-speed swimming (Hubbard et al. 2016; Wu et al. 2021). Stabilizing reflexes are even critical for flies, which rapidly flap their wings several hundred times per second during flight. For example, fly halteres—modified hindwings that function as nature’s only biological gyroscope—mediate critical equilibrium reflexes to stabilize the body (Hengstenberg 1988; Nalbach and Hengstenberg 1994; Dickinson 1999; Sherman and Dickinson 2003; Dickerson et al. 2019).
At an intermediate level, mechanosensory feedback is integrated in control circuits in the spinal cord (vertebrates) or ventral nerve cord (invertebrates), where it tunes locomotor speed and body coordination. Parts of these control circuits are intrinsically rhythmic and referred to as locomotor central pattern generators (CPGs; Guertin 2009; Bidaye et al. 2018; Grillner and Kozlov 2021).
The effects of mechanosensory feedback on speed control can be seen in fictive preparations, where CPG circuits are isolated and disconnected from mechanosensory feedback altogether. Without mechanosensory feedback, rhythmic locomotor-like activity in circuits normally controlling walking, flight, swimming, or crawling is significantly slower (Wilson 1961; Grillner and Zangger 1979; Wallén and Williams 1984; Ryckebusch and Laurent 1993; Büschges et al. 1995; Pulver et al. 2015), suggesting that mechanosensory feedback enhances locomotor speed. This effect was confirmed in genetic model organisms by manipulating specific mechanosensory neurons during swimming and crawling (Hughes and Thomas 2007; Böhm et al. 2016; Knafo et al. 2017). For example, activating mechanosensory Rohon beard neurons in larval zebrafish that detect pressure on the skin from the external fluid activates premotor excitatory V2a neurons (Knafo et al. 2017), which are involved in setting locomotor speed (McLean et al. 2008; Menelaou and McLean 2019). Similarly, the cerebrospinal fluid contacting neurons mentioned above and other mechanosensory neurons detecting bending of the spine contribute to increasing movement speed (Böhm et al. 2016; Picton et al. 2021; Wu et al. 2021). Likewise, in crawling Drosophila larvae, different types of mechanosensory neurons that monitor body wall deformation (Vaadia et al. 2019) provide feedback to speed up locomotion (Hughes and Thomas 2007).
The effects of mechanosensory feedback on body coordination are also seen across species and types of locomotion. During walking, both vertebrates and invertebrates integrate feedback from various types of mechanosensory neurons to tune the duration of stance and swing phases, spatial foot placement, and the temporal coordination of legs on a step-by-step basis (Prochazka 1996; Pearson 2004; Bidaye et al. 2018). For example, both movement- and force-related feedback interact with CPG circuits to time each leg’s stance-to-swing transition (Duysens and Pearson 1980; Cruse 1985; Hiebert et al. 1996). In addition, coordination across multiple legs is aided by long-range, inter-segmental mechanosensory pathways (Grabowska et al. 2022). Similarly, mechanosensory feedback is required to coordinate different body segments during crawling (Hughes and Thomas 2007; Song et al. 2007). In insect flight, mechanosensory feedback tunes the flight rhythm on a stroke-by-stroke basis, with mechanosensory feedback from the hindwing playing a major role in structuring the motion of its anterior serial homologue (Gettrup 1965; Frye 2001; Dickerson 2020). This is the case for both locusts, in which campaniform sensilla encode hindwing motion that in turn controls forewing twist (Gettrup 1965), and moths, in which the hindwing stretch receptor helps control lift generation by the forewing (Frye 2001). Although flies do not possess a true hindwing, recent work suggests that the aforementioned haltere plays a similar role (Dickerson et al. 2019).
At the highest level, mechanosensory feedback is integrated in control circuits in the brain, where it helps to elicit appropriate changes in motor behavior and estimate the state of the body. For example, activating chordotonal neurons in the legs of Drosophila can elicit forward walking (Medeiros et al. 2022). When walking flies encounter a dead-end, touch-sensitive neurons on the body and legs can elicit backward walking (Sen et al. 2019). These changes in motor behavior are mediated by circuits in the brain (Sen et al. 2019; Medeiros et al. 2022). While some fly mechanosensors may directly project to the brain (Tsubouchi et al. 2017), most mechanosensory feedback is carried by ascending neurons (Chen et al. 2023). In the brain, mechanosensory feedback can directly modulate descending neurons that control locomotion (Sen et al. 2019) or be integrated with other types of sensory feedback in deeper layers. For example, the central complex—the navigational center of the insect brain—integrates mechanosensory feedback from the legs (Lu et al. 2022; Lyu et al. 2022) and halteres (Kathman and Fox 2019) with other sensory feedback to keep track of the fly’s heading during walking and flight. Likewise, in mammals, mechanosensory feedback ascends to multiple brain regions, including the brainstem, the cerebellum, the thalamus, and the cerebral cortex, where it provides critical information about the state of the locomotor system (Frigon et al. 2021).
Regulation of mechanosensory feedback
A fundamental feature of mechanosensory feedback in animals is that its effects are not fixed but can be flexibly attenuated when disruptive or self-generated and enhanced when advantageous (Azim and Seki 2019).
Mechanosensory feedback can be adjusted already in the periphery. A well-documented example is the muscle spindle, whose sensitivity is tuned by the activity of gamma motor neurons that regulate the level of tension in the muscle fibers of the spindle (Macefield and Knellwolf 2018). Another example are fly halteres. Like the wings, halteres possess a set of steering muscles that receive visual feedback to regulate muscle activity and, with that, haltere motion. This way, visual feedback directly affects the mechanosensory feedback provided by the halteres and, in turn, the halteres' effects on flight control (Dickerson et al. 2019). A conserved mechanism for regulating mechanosensory feedback in the periphery across species is presynaptic inhibition, where specialized interneurons inhibit the mechanosensory axons as they enter the spinal cord or ventral nerve cord in a task- and context-dependent manner (Watson 1992; Rudomin and Schmidt 1999; Rossignol et al. 2006). For example, mechanosensory axons of locusts and mice receive presynaptic inhibition during walking (Wolf and Burrows 1995; Koch et al. 2017). Removal of this inhibition in mice results in excessive flexion and perturbed gait, suggesting that presynaptic inhibition is critical to prevent reflexive responses that would otherwise disrupt rhythmic stepping (Koch et al. 2017).
Regulating the effects of mechanosensory feedback is not limited to the periphery. Feedback can be flexibly adjusted at multiple levels, from circuits in the spinal cord or ventral nerve cord to circuits in the brain (Azim and Seki 2019). In stick insects, for example, a balance of excitation and inhibition within a population of specific interneurons in the ventral nerve cord is thought to reverse the sign of postural reflexes during locomotion (Driesang and Büschges 1996).
Moving forward
The above examples illustrate that mechanosensory feedback is integrated in control circuits at multiple levels to support a variety of locomotor functions. Mechanosensory feedback mediates fast reflexes, shapes locomotor rhythms, and contributes to action selection and state estimation. Task- and context-dependent enhancement or attenuation of feedback guarantees smooth, robust movements. Despite our broad knowledge about the integration and regulation of mechanosensory feedback in animals, many critical details remain unclear, which hampers broader robotic implementation. How exactly are feedback pathways organized in control circuits? How do specific feedback pathways contribute to robust locomotion? Moving forward, we believe that connectomics, genetic tools, and computational and physical models will provide important insights into these questions.
Moving forward with connectomics
One obstacle to understanding mechanosensory feedback concerns the organization of feedback pathways in the central nervous system. Although much is known about the organization already (Abraira and Ginty 2013; Tuthill and Wilson 2016; Wu et al. 2021), we lack comprehensive wiring diagrams. Such wiring diagrams would be powerful tools to generate and test hypotheses about the integration of mechanosensory feedback in control circuits and constrain computational models (Zarin et al. 2019; Lappalainen et al. 2023). Thanks to recent advances in electron-microscopy-based connectomics, mechanosensory wiring diagrams are now in reach. Connectomics has developed extremely rapidly in the last five years, particularly for Drosophila (Eschbach and Zlatic 2020; Phelps et al. 2021; Galili et al. 2022; Winding et al. 2023; Takemura et al. 2023). There now exist multiple connectomes of the Drosophila brain and ventral nerve cord in various stages of reconstruction. Reconstruction and analysis efforts are massively accelerated by collaborations in online communities and new machine learning tools that can reconstruct neurons, identify synapses, and predict neurotransmitter identity with high accuracy (Azevedo et al. 2022; Dorkenwald et al. 2022). Studies have already begun to reveal the anatomical diversity and distinct projection patterns of mechanosensory neurons from different body parts and how they interface with central circuits to influence behavior (Ohyama et al. 2015; Hampel et al. 2020; Phelps et al. 2021; Dorkenwald et al. 2022; Eichler et al. 2023). One of the earliest studies identified how mechanosensory feedback from chordotonal organs is integrated in a multilevel circuit in the Drosophila larva to trigger rolling (Ohyama et al. 2015). Similar efforts are underway to identify mechanosensory feedback circuits for walking and flight in the adult. A challenge for reconstructing mechanosensory pathways is that connectomics datasets do not include the mechanosensory cell bodies in the periphery. Therefore, mechanosensory axons must be identified with the help of other datasets, such as light microscopy images (Eichler et al. 2023). Whole-nervous system connectomics is not yet within reach for mammalian-sized nervous systems, but insect species other than Drosophila are potential targets. Comparative connectomics will be useful to identify which feedback pathways are conserved and which represent species-specific adaptations (Galili et al. 2022).
Moving forward with genetic tools
Where connectomics can provide a static roadmap of mechanosensory pathways, genetic tools allow recording and manipulating these pathways in behaving animals with ever-increasing specificity. In Drosophila, zebrafish, and mice, neural activity can now be imaged in the spinal cord or ventral nerve cord during tethered locomotion (Nelson et al. 2019; Vaadia et al. 2019; Böhm et al. 2022; Hermans et al. 2022). Experimental access to the activity of mechanosensors and their downstream targets is a huge advantage. As outlined above, activity patterns cannot easily be predicted from movement, because the nervous system can adjust feedback in a task- and context-dependent manner (Azim and Seki 2019). Imaging the activity of locomotor circuits at multiple levels during behavior can reveal when and where in the processing hierarchy are specific types of feedback available for control. In addition, chronic knock-outs and acute optogenetic tools allow manipulating these feedback pathways in behaving animals (Böhm et al. 2016; DeAngelis et al. 2020; Wu et al. 2021; Chockley et al. 2022). Importantly, these tools allow targeting pathways that cannot be targeted with traditional techniques. A good example are the internal, cerebrospinal fluid contacting neurons mentioned above (Fig. 1A). Targeting these cells with genetic knock-outs, optogenetics, and calcium imaging in behaving zebrafish revealed that their feedback contributes to optimizing locomotion speed (Böhm et al. 2016; Wu et al. 2021). Likewise, insect chordotonal neurons and mammalian muscle spindles are notoriously difficult to manipulate in locomoting animals with traditional techniques, because they are deeply embedded in the legs (Fig. 1A). Studies in Drosophila and mice have started to use genetic manipulations to study the contribution of these mechanosensors to the control of locomotion (Akay et al. 2014; Chockley et al. 2022; Santuz et al. 2022). Albeit powerful, genetic manipulations come with their own set of challenges (Wolff and Ölveczky 2018). Due to the redundancy of mechanosensory feedback pathways and their interactions with locomotor circuits at multiple levels, manipulations can have little effect on locomotion or unintended effects that are difficult to interpret (Dallmann et al. 2021). In particular, the effects of silencing specific mechanosensors might be compensated for during development considering the importance of locomotion for animals. A promising strategy is to complement manipulation experiments with computational and physical models.
Moving forward with computational models
In the context of goal-directed locomotion, a useful framework to design and analyze manipulation experiments is control theory (Madhav and Cowan 2020). Based on system identification, this engineering framework allows generating testable hypotheses of how different types of feedback contribute to locomotor performance. Roth et al. (2016) used this framework to study the relative contribution of mechanosensory and visual feedback to the flower tracking behavior of feeding hawkmoths during flight. Using an artificial flower, the authors provided independent and conflicting visual and mechanical flower stimuli. The analysis revealed that feedback from mechanosensory neurons in the proboscis is sufficient to perform tracking behavior, and is weighted linearly with parallel feedback from visual neurons. In another study, Cellini and Mongeau (2022) took advantage of control theory to tease apart the contribution of mechanosensory and visual feedback to gaze stabilization in flying flies. The analysis revealed that mechanosensory feedback from halteres actively damps head movements when body motion is self-generated. Similar studies have helped to understand how mechanosensory feedback contributes to the task-level control of rapid running (Cowan et al. 2006; Mongeau et al. 2015).
When locomotion is not related to a specific task like flower tracking or gaze stabilization, determining and measuring the inputs and outputs of the locomotor system required for a top-down, control-theoretic approach is challenging. An alternative approach is bottom-up modeling, where the locomotor system is synthesized from low-level principles, including physiologically plausible feedback pathways. Markin et al. (2016) took this approach and developed a neuromechanical CPG model of the cat hindlimb with physiologically plausible feedback from cutaneous mechanosensors in the paw and muscle spindles and Golgi tendon organs in the leg. The model allowed the authors to selectively remove mechanosensory feedback pathways and test the resulting effects on walking. For example, without muscle spindle feedback from the calf muscle, the simulated cat hindlimb was still able to walk, but without Golgi tendon organ feedback from the same muscle, it was unable to produce robust walking and collapsed after a few strides. Similar kinds of models are being used to study the relative contribution of mechanosensory feedback pathways in other walking, crawling, and swimming animals (Proctor and Holmes 2018; Schilling and Cruse 2020; Sun et al. 2022; Hamlet et al. 2023; Russo et al. 2023). The ability to easily change the gain of feedback pathways in these models helps reveal the effectiveness of feedback across the parameter space and generate specific, testable hypotheses for experiments. Moving forward, progress will be accelerated by using realistic computational models that are openly shared for use across labs (Lobato-Rios et al. 2022).
Moving forward with physical models
Another approach to evaluate a specific mechanosensory feedback mechanism is to implement it on a robot (Ijspeert 2014). Force feedback during walking is a good example. A series of experimental studies suggest that force feedback from mammalian Golgi tendon organs and insect campaniform sensilla provides reflexive, positive feedback that can contribute to inter-leg coordination (Duysens and Pearson 1980; Duysens et al. 2000; Zill et al. 2009; Dallmann et al. 2017). However, due to redundancy of feedback pathways, it has been challenging to provide causal evidence for this mechanism in walking animals. For example, position and movement feedback is thought to contribute to inter-leg coordination as well (Cruse 1985; Hiebert et al. 1996). But using a minimalistic legged robot, Owaki and Ishiguro (2017) confirmed the sufficiency of the load-based mechanism for establishing inter-leg coordination. In their experiments, each robotic leg was driven independently of the others by its own CPG. Incorporating force feedback from the legs was sufficient to coordinate the CPGs and generate different stepping patterns. Robots have been similarly useful to test the effectiveness of mechanosensory feedback pathways in other animals and types of locomotion (Ijspeert 2001; Ijspeert et al. 2007; Righetti and Ijspeert 2008; Hagen et al. 2021; Sivitilli et al. 2022). The advantage of robots over simulations in such studies is that the effects of mechanosensory feedback on locomotion can be tested in environments similar to those encountered by animals. The physical interactions with the natural environment provide a realistic source of sensory noise and other complex conditions that may be simplified or nonexistent in simulations. Moving forward, this “robotics-inspired biology” approach will be accelerated by decreasing cost and increasing availability of electronic components, actuators, sensors, fabrication equipment, and access to open-source software (Gravish and Lauder 2018). In addition, size-agnostic design and fabrication workflows will facilitate building robots at animal scales, including that of small insects (Jayaram et al. 2020).
While incorporating simple proprioceptive feedback has proven successful for controlling robots across sizes (Wensing et al. 2017; Jayaram, Jafferis, et al. 2018; Doshi et al. 2019; Gong et al. 2019), incorporating more advanced forms of mechanosensory feedback has been limited due to several implementation challenges. For example, physiologically plausible controllers can become highly complex and laborious to tune. This has led to the emergence of model-free reinforcement learning as an alternative approach for developing locomotion controllers (Lee et al. 2020). Rather than emulating specific neural pathways, these controllers learn how to process the stream of mechanosensory information optimally for the locomotor tasks at hand. Intriguingly, current robots require extremely high-bandwidth closed-loop controllers (∼1 kHz) for effective locomotion (Chung et al. 2023), whereas animals operate at much lower bandwidth (∼10 Hz; Nishikawa et al. 2007). One possible explanation is that robotic controllers do not yet implement the parallelization, distribution, and hierarchical organization of mechanosensory pathways that make processing in nervous systems so effective (Merel et al. 2019). A promising approach for achieving this is to move away from conventional computing to neuromorphic computing—efficient, parallel, low-power computing inspired by the nervous system (Calimera et al. 2013; Thakur et al. 2018; de Croon et al. 2022).
Ultimately, when looking for inspiration for engineered systems, it is important to consider that biological solutions are not necessarily optimized to perform one particular function (Haberland and Kim 2015). For example, some fly mechanosensory neurons involved in walking are also involved in grooming (Ravbar et al. 2021) and courtship (McKelvey et al. 2021). In addition, non-locomotor factors like development and physiology can constrain the distribution of mechanosensory neurons (Aiello et al. 2021). Therefore, it will be crucial to ask which aspects of animal mechanosensation are truly necessary to improve robotic performance, and which are bugs. An example of the latter are sensory delays, which can constrain the effectiveness of mechanosensation at high-speed locomotion in animals (More and Donelan 2018). Robots can easily outperform animals in terms of conduction delays (conduction speed through an electrical wire approaches the speed of light, resulting in insignificant delays) and other component specifications (e.g., onboard processing power).
Conclusions
Mechanosensation is a key sense that enables animals to move robustly in complex, natural environments, and it holds great promise for the design of the next generation of locomoting robots. We argue that bio-inspired engineering would benefit greatly from (1) a detailed understanding of the encoding properties and distribution of animal mechanosensors and (2) the integration and regulation of mechanosensory feedback in the nervous system. Moving forward will require complementary experimental and engineering approaches, including connectomics to map mechanosensory feedback pathways in the nervous system, genetic tools to record from and manipulate these pathways, computational and physical models to evaluate their effectiveness, and new fabrication and computing techniques to emulate them in robotics. We envision that these approaches will help us study the mechanosensory control of locomotion under increasingly natural conditions, which may reveal additional functions that are not apparent in simplified laboratory settings. As the space of possible experiments and analyses grows, there is an increased need for interdisciplinary collaborations of engineers and biologists taking a comparative approach across species and types of locomotion to identify general principles worthy of imitation. Interdisciplinary meetings like the 2023 SICB symposium “The role of mechanosensation in robust locomotion” are invaluable for moving forward together.
Acknowledgement
We thank the organizers Hilary Katz and Katie Stanchak and the participants of the 2023 SICB symposium “The role of mechanosensation in robust locomotion” for an inspiring, interdisciplinary meeting. We also thank two anonymous reviewers for helpful comments on the manuscript. This review benefited greatly from presentations and discussion in the Kavli Institute of Theoretical Physics (KITP), University of California in Santa Barbara, during the Neurophysics of Locomotion course 2022 that was supported in part by the National Science Foundation under Grant No. NSF PHY-1748958 and the Gordon and Betty Moore Foundation Grant No. 2919.02.
Notes
From the symposium "The role of mechanosensation in robust locomotion" presented at the annual meeting of the Society for Integrative and Comparative Biology, January 3–7, 2023, and SICB+ Virtual, January 16–March 31, 2023.
Contributor Information
Chris J Dallmann, Department of Physiology and Biophysics, University of Washington, Seattle, WA 98195, USA.
Bradley H Dickerson, Princeton Neuroscience Institute, Princeton University, Princeton, NJ 08544, USA.
Julie H Simpson, Department of Molecular, Cellular, and Developmental Biology and Neuroscience Research Institute, University of California Santa Barbara, Santa Barbara, CA 93106, USA.
Claire Wyart, Institut du Cerveau et de la Moelle épinière (ICM), Sorbonne Université, Paris 75005, France.
Kaushik Jayaram, Paul M Rady Department of Mechanical Engineering, University of Colorado Boulder, Boulder, CO 80309, USA.
Funding
The work in the Dickerson Lab was supported by National Science Foundation award 2221458; a Searle Scholar award; and a McKnight Scholar award. Work in the Simpson Lab was supported by the National Institutes of HealthR01-NS110866 “Sensory control of a motor sequence.” Work in the Wyart Lab was supported by the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska–Curie grant agreement [grant number #813457]; New York Stem Cell Foundation (NYSCF) Robertson Award 2016 [grant number 332; NYSCF-R-NI39]; the National Institutes of Health [grant number 1U19NS104653-01]; 2020 European Research Council Consolidator [grant number #101002870; 2021–2026] “Circuit mechanisms underlying sensory-evoked navigation”; “Fondation pour la Recherche Médicale” [FRM- EQU202003010612]; and Fondation Bettencourt-Schueller [FBS-don-0031]. Work in the Jayaram Lab was supported by the US Army Research Office (ARO) [grant number #W911NF-23-1-0039] and the Meta Foundation.
Conflict of interest
The authors have no conflicts of interest to declare.
Data availability statement
Data sharing is not applicable as no new data were generated or analyzed in this study.
References
- Abraira VE, Ginty DD.. 2013. The sensory neurons of touch. Neuron. 79:618–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agrawal S, Tuthill JC.. 2022. The two-body problem: proprioception and motor control across the metamorphic divide. Curr Opin Neurobiol. 74:102546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aguilar J, Zhang T, Qian F, Kingsbury M, McInroe B, Mazouchova N, Li C, Maladen R, Gong C, Travers Met al. 2016. A review on locomotion robophysics: the study of movement at the intersection of robotics, soft matter and dynamical systems. Rep Prog Phys. 79:110001. [DOI] [PubMed] [Google Scholar]
- Aiello BR, Stanchak KE, Weber AI, Deora T, Sponberg S, Brunton BW.. 2021. Spatial distribution of campaniform sensilla mechanosensors on wings: form, function, and phylogeny. Curr Opin Insect Sci. 48:8–17. [DOI] [PubMed] [Google Scholar]
- Akay T, Tourtellotte WG, Arber S, Jessell TM.. 2014. Degradation of mouse locomotor pattern in the absence of proprioceptive sensory feedback. Proc Natl Acad Sci USA. 111:16877–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akitake B, Ren Q, Boiko N, Ni J, Sokabe T, Stockand JD, Eaton BA, Montell C.. 2015. Coordination and fine motor control depend on Drosophila TRPγ. Nat Commun. 6:7288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altendorfer R, Moore N, Komsuolu H, Buehler M, Brown HB, McMordie D, Saranli U, Full R, Koditschek DE.. 2001. RHex: a biologically inspired hexapod runner. Auton Robots. 11:207–13. [Google Scholar]
- Azevedo A, Lesser E, Mark B, Phelps J, Elabbady L, Kuroda S, Sustar A, Moussa A, Kandelwal A, Dallmann CJet al. 2022. Tools for comprehensive reconstruction and analysis of Drosophila motor circuits. bioRxiv 2022.12.15.520299. 10.1101/2022.12.15.520299 [DOI] [Google Scholar]
- Azim E, Seki K.. 2019. Gain control in the sensorimotor system. Curr Opin Physiol. 8:177–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barth FG. 2019. Mechanics to pre-process information for the fine tuning of mechanoreceptors. J Comp Physiol A. 205:661–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bidaye SS, Bockemühl T, Büschges A.. 2018. Six-legged walking in insects: how CPGs, peripheral feedback, and descending signals generate coordinated and adaptive motor rhythms. J Neurophysiol. 119:459–75. [DOI] [PubMed] [Google Scholar]
- Blum KP, Campbell KS, Horslen BC, Nardelli P, Housley SN, Cope TC, Ting LH.. 2020. Diverse and complex muscle spindle afferent firing properties emerge from multiscale muscle mechanics. eLife. 9:e55177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böhm UL, Kimura Y, Kawashima T, Ahrens MB, Higashijima S, Engert F, Cohen AE.. 2022. Voltage imaging identifies spinal circuits that modulate locomotor adaptation in zebrafish. Neuron. 110:1211–1222.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böhm UL, Prendergast A, Djenoune L, Nunes Figueiredo S, Gomez J, Stokes C, Kaiser S, Suster M, Kawakami K, Charpentier Met al. 2016. CSF-contacting neurons regulate locomotion by relaying mechanical stimuli to spinal circuits. Nat Commun. 7:10866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boublil BL, Diebold CA, Moss CF.. 2021. Mechanosensory hairs and hair-like structures in the animal kingdom: specializations and shared functions serve to inspire technology applications. Sensors. 21:6375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Büschges A, Schmitz J, Bässler1 U.. 1995. Rhythmic patterns in the thoracic nerve cord of the stick insect induced by pilocarpine. J Exp Biol. 198:435–56. [DOI] [PubMed] [Google Scholar]
- Buschmann T, Ewald A, von Twickel A, Büschges A. 2015. Controlling legs for locomotion—insights from robotics and neurobiology. Bioinspir Biomim. 10:041001. [DOI] [PubMed] [Google Scholar]
- Calimera A, Macii E, Poncino M.. 2013. The human brain project and neuromorphic computing. Funct Neurol. 28:191. [PMC free article] [PubMed] [Google Scholar]
- Cellini B, Mongeau J-M.. 2022. Nested mechanosensory feedback actively damps visually guided head movements in Drosophila. eLife. 11:e80880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C-L, Aymanns F, Minegishi R, Matsuda VDV, Talabot N, Günel S, Dickson BJ, Ramdya P.. 2023. Ascending neurons convey behavioral state to integrative sensory and action selection brain regions. Nat Neurosci. 26: 682–695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng LE, Song W, Looger LL, LY Jan, Jan YN.. 2010. The role of the TRP channel NompC in Drosophila larval and adult locomotion. Neuron. 67:373–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chesler AT, Szczot M, Bharucha-Goebel D, Čeko M, Donkervoort S, Laubacher C, Hayes LH, Alter K, Zampieri C, Stanley Cet al. 2016. The role of PIEZO2 in human mechanosensation. N Engl J Med. 375:1355–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chockley AS, Dinges GF, Di Cristina G, Ratican S, Bockemühl T, Büschges A.. 2022. Subsets of leg proprioceptors influence leg kinematics but not interleg coordination in Drosophila melanogaster walking. J Exp Biol. 225:jeb244245. [DOI] [PubMed] [Google Scholar]
- Chung TH, Orekhov V, Maio A.. 2023. Into the Robotic Depths: analysis and Insights from the DARPA Subterranean Challenge. Annu Rev Control Robot Auton Syst. 6:477. [Google Scholar]
- Cole JD, Katifi HA.. 1991. Evoked potentials in a man with a complete large myelinated fibre sensory neuropathy below the neck. Electroencephalogr Clin Neurophysiol Evoked Potentials Section. 80:103–7. [DOI] [PubMed] [Google Scholar]
- Cowan NJ, Lee J, Full RJ.. 2006. Task-level control of rapid wall following in the American cockroach. J Exp Biol. 209:1617–29. [DOI] [PubMed] [Google Scholar]
- Cruse H. 1985. Which parameters control the leg movement of a walking insect?: II. The start of the swing phase. J Exp Biol. 116:357–62. [Google Scholar]
- Dallmann CJ, Hoinville T, Dürr V, Schmitz J.. 2017. A load-based mechanism for inter-leg coordination in insects. Proc R Soc B. 284:20171755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallmann CJ, Karashchuk P, Brunton BW, Tuthill JC.. 2021. A leg to stand on: computational models of proprioception. Curr Opin Physiol. 22:100426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies M, Wild A, Orchard G, Sandamirskaya Y, Guerra GAF, Joshi P, Plank P, Risbud SR. 2021. Advancing neuromorphic computing with loihi: a survey of results and outlook. Proc IEEE. 109:911–34. [Google Scholar]
- de Croon GCHE, Dupeyroux JJG, Fuller SB, Marshall JAR.. 2022. Insect-inspired AI for autonomous robots. Sci Robot. 7:eabl6334. [DOI] [PubMed] [Google Scholar]
- DeAngelis BD, Zavatone-Veth JA, Gonzalez-Suarez AD, Clark DA.. 2020. Spatiotemporally precise optogenetic activation of sensory neurons in freely walking Drosophila. eLife. 9:e54183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delcomyn F. 1985. Factors regulating insect walking. Annu Rev Entomol. 30:239–56. [Google Scholar]
- Delmas P, Parpaite T, Coste B.. 2022. PIEZO channels and newcomers in the mammalian mechanosensitive ion channel family. Neuron. 110:2713–27. [DOI] [PubMed] [Google Scholar]
- Dickerson BH, de Souza AM, Huda A, Dickinson MH.. 2019. Flies regulate wing motion via active control of a dual-function gyroscope. Curr Biol. 29:3517–3524.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickerson BH, Fox JL, Sponberg S.. 2021. Functional diversity from generic encoding in insect campaniform sensilla. Curr Opin Physiol. 19:194–203. [Google Scholar]
- Dickerson BH. 2020. Timing precision in fly flight control: integrating mechanosensory input with muscle physiology. Proc R Soc B. 287:20201774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickinson MH, Farley CT, Full RJ, Koehl MAR, Kram R, Lehman S.. 2000. How animals move: an integrative view. Science. 288:100–6. [DOI] [PubMed] [Google Scholar]
- Dickinson MH. 1999. Haltere–mediated equilibrium reflexes of the fruit fly, Drosophila melanogaster. Phil Trans R Soc Lond B. 354:903–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinges GF, Bockemühl T, Iacoviello F, Shearing PR, Büschges A, Blanke A.. 2022. Ultra high-resolution biomechanics suggest that substructures within insect mechanosensors decisively affect their sensitivity. J R Soc Interface. 19:20220102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinges GF, Chockley AS, Bockemühl T, Ito K, Blanke A, Büschges A.. 2021. Location and arrangement of campaniform sensilla in Drosophila melanogaster. J Comp Neurol. 529:905–25. [DOI] [PubMed] [Google Scholar]
- Djenoune L, Khabou H, Joubert F, Quan FB, Nunes Figueiredo S, Bodineau L, Del Bene F, Burcklé C, Tostivint H, Wyart C. 2014. Investigation of spinal cerebrospinal fluid-contacting neurons expressing PKD2L1: evidence for a conserved system from fish to primates. Front Neuroanat. 8:26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorkenwald S, McKellar CE, Macrina T, Kemnitz N, Lee K, Lu R, Wu J, Popovych S, Mitchell E, Nehoran Bet al. 2022. FlyWire: online community for whole-brain connectomics. Nat Methods. 19:119–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doshi N, Jayaram K, Castellanos S, Kuindersma S, Wood RJ.. 2019. Effective locomotion at multiple stride frequencies using proprioceptive feedback on a legged microrobot. Bioinspir Biomim. 14:056001. [DOI] [PubMed] [Google Scholar]
- Driesang RB, Büschges A.. 1996. Physiological changes in central neuronal pathways contributing to the generation of a reflex reversal. J Comp Physiol A. 179:45–57. [Google Scholar]
- Dupeyroux J, Stroobants S, De Croon GCHE.. 2022. A toolbox for neuromorphic perception in robotics. Presented at the 2022 8th International Conference on Event-Based Control, Communication, and Signal Processing (EBCCSP). p. 1–7.. arXiv:2103.02751. [Google Scholar]
- Duysens J, Clarac F, Cruse H.. 2000. Load-regulating mechanisms in gait and posture: comparative aspects. Physiol Rev. 80:83–133. [DOI] [PubMed] [Google Scholar]
- Duysens J, Pearson KG.. 1980. Inhibition of flexor burst generation by loading ankle extensor muscles in walking cats. Brain Res. 187:321–32. [DOI] [PubMed] [Google Scholar]
- Eichler K, Hampel S, Alejandro-García A, Calle-Schuler SA, Santana-Cruz A, Kmecova L, Blagburn JM, Hoopfer ED, Seeds AM. 2023. Somatotopic organization among parallel sensory pathways that promote a grooming sequence in Drosophila. Neuroscience. bioRxiv 2023.02.11.528119. 10.1101/2023.02.11.528119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eschbach C, Zlatic M.. 2020. Useful road maps: studying Drosophila larva’s central nervous system with the help of connectomics. Curr Opin Neurobiol. 65:129–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faucherre A, Nargeot J, Mangoni ME, Jopling C. 2013. piezo2b regulates vertebrate light touch response. J Neurosci. 33:17089–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frigon A, Akay T, Prilutsky BI.. 2021. Control of mammalian locomotion by somatosensory feedback. In: Terjung R, editor. Comprehensive physiology. 1st ed. Wiley. p. 2877–947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frye MA. 2001. Effects of stretch receptor ablation on the optomotor control of lift in the hawkmoth Manduca sexta. J Exp Biol. 204:3683–91. [DOI] [PubMed] [Google Scholar]
- Gafford JB, Kesner SB, Degirmenci A, Wood RJ, Howe RD, Walsh CJ.. 2014. A monolithic approach to fabricating low-cost, millimeter-scale multi-axis force sensors for minimally-invasive surgery. 2014 IEEE International Conference on Robotics and Automation (ICRA), Hong Kong, China. p. 1419–25. [Google Scholar]
- Galili DS, Jefferis GS, Costa M.. 2022. Connectomics and the neural basis of behaviour. Curr Opin Insect Sci. 54:100968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao W, Emaminejad S, Nyein HYY, Challa S, Chen K, Peck A, Fahad HM, Ota H, Shiraki H, Kiriya Det al. 2016. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature. 529:509–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gettrup E. 1965. Sensory mechanisms in locomotion: the campaniform sensilla of the insect wing and their function during flight. Cold Spring Harbor Symp Quant Biol. 30:615–22. [DOI] [PubMed] [Google Scholar]
- Gong Y, Hartley R, Da X, Hereid A, Harib O, Huang J-K, Grizzle J.. 2019. Feedback control of a cassie bipedal robot: walking, standing, and riding a segway. 2019 American Control Conference (ACC), Philadelphia, PA, USA. p. 4559–66. [Google Scholar]
- Grabowska M, Toth TI, Büschges A, Daun S.. 2022. Existence of a long-range caudo-rostral sensory influence in terrestrial locomotion. J Neurosci. 42:4841–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gravish N, Lauder GV.. 2018. Robotics-inspired biology. J Exp Biol. 221:jeb138438. [DOI] [PubMed] [Google Scholar]
- Grillner S, Kozlov A.. 2021. The CPGs for limbed locomotion—facts and fiction. IJMS. 22:5882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grillner S, Zangger P.. 1979. On the central generation of locomotion in the low spinal cat. Exp Brain Res. 34:241–61. [DOI] [PubMed] [Google Scholar]
- Guertin PA. 2009. The mammalian central pattern generator for locomotion. Brain Res Rev. 62:45–56. [DOI] [PubMed] [Google Scholar]
- Guizzo E. 2019. The next generation of Boston dynamics’ ATLAS robot is quiet, robust, and tether free. IEEE Spectrum. (https://spectrum.ieee.org/next-generation-of-boston-dynamics-atlas-robot). [Google Scholar]
- Haberland M, Kim S.. 2015. On extracting design principles from biology: I. Method—general answers to high-level design questions for bioinspired robots. Bioinspir Biomim. 10:016010. [DOI] [PubMed] [Google Scholar]
- Hagen DA, Marjaninejad A, Loeb GE, Valero-Cuevas FJ.. 2021. insideOut: a bio-inspired machine learning approach to estimating posture in robots driven by compliant tendons. Front Neurorobot. 15:679122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamlet C, Fauci L, Morgan JR, Tytell ED.. 2023. Proprioceptive feedback amplification restores effective locomotion in a neuromechanical model of lampreys with spinal injuries. Proc Natl Acad Sci USA. 120:e2213302120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hampel S, Eichler K, Yamada D, Bock DD, Kamikouchi A, Seeds AM.. 2020. Distinct subpopulations of mechanosensory chordotonal organ neurons elicit grooming of the fruit fly antennae. eLife. 9:e59976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hengstenberg R. 1988. Mechanosensory control of compensatory head roll during flight in the blowflyCalliphora erythrocephala Meig. J Comp Physiol. 163:151–65. [Google Scholar]
- Hermans L, Kaynak M, Braun J, Ríos VL, Chen C-L, Friedberg A, Günel S, Aymanns F, Sakar MS, Ramdya P.. 2022. Microengineered devices enable long-term imaging of the ventral nerve cord in behaving adult Drosophila. Nat Commun. 13:5006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiebert GW, Whelan PJ, Prochazka A, Pearson KG.. 1996. Contribution of hind limb flexor muscle afferents to the timing of phase transitions in the cat step cycle. J Neurophysiol. 75:1126–37. [DOI] [PubMed] [Google Scholar]
- Holmes P, Full RJ, Koditschek DE, Guckenheimer J.. 2006. The dynamics of legged locomotion: models, analyses, and challenges. SIAM Rev. 48:207–304. [Google Scholar]
- Hubbard JM, Böhm UL, Prendergast A, Tseng P-EB, Newman M, Stokes C, Wyart C.. 2016. Intraspinal sensory neurons provide powerful inhibition to motor circuits ensuring postural control during locomotion. Curr Biol. 26:2841–53. [DOI] [PubMed] [Google Scholar]
- Hughes CL, Thomas JB.. 2007. A sensory feedback circuit coordinates muscle activity in Drosophila. Mol Cell Neurosci. 35:383–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutter M, Gehring C, Lauber A, Gunther F, Bellicoso CD, Tsounis V, Fankhauser P, Diethelm R, Bachmann S, Bloesch Met al. 2017. ANYmal—toward legged robots for harsh environments. Adv Robot. 31:918–31. [Google Scholar]
- Ijspeert AJ, Crespi A, Ryczko D, Cabelguen J-M.. 2007. From swimming to walking with a salamander robot driven by a spinal cord model. Science. 315:1416–20. [DOI] [PubMed] [Google Scholar]
- Ijspeert AJ. 2001. A connectionist central pattern generator for the aquatic and terrestrial gaits of a simulated salamander. Biol Cybern. 84:331–48. [DOI] [PubMed] [Google Scholar]
- Ijspeert AJ. 2014. Biorobotics: using robots to emulate and investigate agile locomotion. Science. 346:196–203. [DOI] [PubMed] [Google Scholar]
- Jayaram K, Full RJ.. 2016. Cockroaches traverse crevices, crawl rapidly in confined spaces, and inspire a soft, legged robot. Proc Natl Acad Sci USA. 113:E950–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayaram K, Jafferis NT, Doshi N, Goldberg B, Wood RJ.. 2018. Concomitant sensing and actuation for piezoelectric microrobots. Smart Mater Struct. 27:065028. [Google Scholar]
- Jayaram K, Mongeau J-M, Mohapatra A, Birkmeyer P, Fearing RS, Full RJ.. 2018. Transition by head-on collision: mechanically mediated manoeuvres in cockroaches and small robots. J R Soc Interface. 15:20170664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayaram K, Shum J, Castellanos S, Helbling EF, Wood RJ.. 2020. Scaling down an insect-size microrobot, HAMR-VI into HAMR-Jr. 2020 IEEE International Conference on Robotics and Automation (ICRA), Paris, France. p. 10305–11. [Google Scholar]
- Jen E. 2005. Robust design: a repertoire of biological, ecological, and engineering case studies. New York, New York 10016: Oxford University Press. [Google Scholar]
- Jindrich DL, Full RJ.. 2002. Dynamic stabilization of rapid hexapedal locomotion. J Exp Biol. 205:2803–23. [DOI] [PubMed] [Google Scholar]
- Kang D, Pikhitsa PV, Choi YW, Lee C, Shin SS, Piao L, Park B, Suh K-Y, Kim T, Choi M.. 2014. Ultrasensitive mechanical crack-based sensor inspired by the spider sensory system. Nature. 516:222–6. [DOI] [PubMed] [Google Scholar]
- Kathman ND, Fox JL.. 2019. Representation of haltere oscillations and integration with visual inputs in the fly central complex. J Neurosci. 39:4100–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knafo S, Fidelin K, Prendergast A, Tseng P-EB, Parrin A, Dickey C, Böhm UL, Figueiredo SN, Thouvenin O, Pascal-Moussellard Het al. 2017. Mechanosensory neurons control the timing of spinal microcircuit selection during locomotion. eLife. 6:e25260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knafo S, Wyart C.. 2018. Active mechanosensory feedback during locomotion in the zebrafish spinal cord. Curr Opin Neurobiol. 52:48–53. [DOI] [PubMed] [Google Scholar]
- Koch SC, Del Barrio MG, Dalet A, Gatto G, Günther T, Zhang J, Seidler B, Saur D, Schüle R, Goulding M. 2017. RORβ spinal interneurons gate sensory transmission during locomotion to secure a fluid walking gait. Neuron. 96:1419–1431.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lajoie Y, Teasdale N, Cole JD, Burnett M, Bard C, Fleury M, Forget R, Paillard J, Lamarre Y.. 1996. Gait of a deafferented subject without large myelinated sensory fibers below the neck. Neurology. 47:109–15. [DOI] [PubMed] [Google Scholar]
- Lappalainen JK, Tschopp FD, Prakhya S, McGill M, Nern A, Shinomiya K, Takemura S, Gruntman E, Macke JH, Turaga SC.. 2023. Connectome-constrained deep mechanistic networks predict neural responses across the fly visual system at single-neuron resolution. Neurosciencepublished online (doi: 10.1101/2023.03.11.532232). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J, Hwangbo J, Wellhausen L, Koltun V, Hutter M.. 2020. Learning quadrupedal locomotion over challenging terrain. Sci Robot. 5:eabc5986. [DOI] [PubMed] [Google Scholar]
- Liu L, Niu S, Zhang J, Mu Z, Li J, Li B, Meng X, Zhang C, Wang Y, Hou Tet al. 2022. Bioinspired, omnidirectional, and hypersensitive flexible strain sensors. Adv Mater. 34:2200823. [DOI] [PubMed] [Google Scholar]
- Lobato-Rios V, Ramalingasetty ST, Özdil PG, Arreguit J, Ijspeert AJ, Ramdya P.. 2022. NeuroMechFly, a neuromechanical model of adult Drosophila melanogaster. Nat Methods. 19:620–7. [DOI] [PubMed] [Google Scholar]
- Lu J, Behbahani AH, Hamburg L, Westeinde EA, Dawson PM, Lyu C, Maimon G, Dickinson MH, Druckmann S, Wilson RI.. 2022. Transforming representations of movement from body- to world-centric space. Nature. 601:98–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyu C, Abbott LF, Maimon G.. 2022. Building an allocentric travelling direction signal via vector computation. Nature. 601:92–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macefield VG, Knellwolf TP.. 2018. Functional properties of human muscle spindles. J Neurophysiol. 120:452–67. [DOI] [PubMed] [Google Scholar]
- Madhav MS, Cowan NJ.. 2020. The synergy between neuroscience and control theory: the nervous system as inspiration for hard control challenges. Annu Rev Control Robot Auton Syst. 3:243–67. [Google Scholar]
- Mamiya A, Gurung P, Siwanowicz I, Sustar A, Chen C, Phelps JS, Kuan AT, Pacureanu A, Lee W-CA, Mhatre Net al. 2022. Biomechanical origins of proprioceptive maps in the Drosophila leg. Neurosciencepublished online (doi: 10.1101/2022.08.08.503192). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcotte A, Mouterde T, Niguès A, Siria A, Bocquet L.. 2020. Mechanically activated ionic transport across single-digit carbon nanotubes. Nat Mater. 19:1057–61. [DOI] [PubMed] [Google Scholar]
- Markin SN, Klishko AN, Shevtsova NA, Lemay MA, Prilutsky BI, Rybak IA.. 2016. A neuromechanical model of spinal control of locomotion. In: Prilutsky BI, Edwards DH, editors. Neuromechanical modeling of posture and locomotion. Springer series in Computational neuroscience. New York, NY: Springer. p. 21–65. [Google Scholar]
- Matheson T. 1992. Range fractionation in the locust metathoracic femoral chordotonal organ. Journal of Comparative Physiology A. 170:509–520. [Google Scholar]
- McCracken JM, Donovan BR, White TJ.. 2020. Materials as machines. Adv Mater. 32:1906564. [DOI] [PubMed] [Google Scholar]
- McDonnell W, Jayaram K, 2022. Enabling Power and Control Autonomy for Insect Scale Robo-Physical Models. In APS March Meeting Abstracts.2022: W03–010. [Google Scholar]
- McEvoy MA, Correll N.. 2015. Materials that couple sensing, actuation, computation, and communication. Science. 347:1261689. [DOI] [PubMed] [Google Scholar]
- McKelvey EGZ, Gyles JP, Michie K, Barquín Pancorbo V, Sober L, Kruszewski LE, Chan A, Fabre CCG. 2021. Drosophila females receive male substrate-borne signals through specific leg neurons during courtship. Curr Biol. 31:3894–3904.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLean DL, Masino MA, Koh IYY, Lindquist WB, Fetcho JR.. 2008. Continuous shifts in the active set of spinal interneurons during changes in locomotor speed. Nat Neurosci. 11:1419–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medeiros AM, Hobbiss AF, Borges G, Moita M, Mendes CS.. 2022. Mechanosensory stimulation triggers sustained local motor activity in Drosophila melanogaster. Neurosciencepublished online (doi: 10.1101/2022.07.19.500315). [DOI] [PubMed] [Google Scholar]
- Mendes CS, Bartos I, Akay T, Márka S, Mann RS.. 2013. Quantification of gait parameters in freely walking wild type and sensory deprived Drosophila melanogaster. eLife. 2:e00231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menelaou E, McLean DL.. 2019. Hierarchical control of locomotion by distinct types of spinal V2a interneurons in zebrafish. Nat Commun. 10:4197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merel J, Botvinick M, Wayne G.. 2019. Hierarchical motor control in mammals and machines. Nat Commun. 10:5489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohren TL, Daniel TL, Brunton SL, Brunton BW.. 2018. Neural-inspired sensors enable sparse, efficient classification of spatiotemporal data. Proc Natl Acad Sci USA. 115:10564–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mongeau J-M, Sponberg SN, Miller JP, Full RJ.. 2015. Sensory processing within antenna enables rapid implementation of feedback control for high-speed running maneuvers. J Exp Biol. 218:2344–54. [DOI] [PubMed] [Google Scholar]
- More HL, Donelan JM.. 2018. Scaling of sensorimotor delays in terrestrial mammals. Proc R Soc B. 285:20180613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muth JT, Vogt DM, Truby RL, Mengüç Y, Kolesky DB, Wood RJ, Lewis JA.. 2014. Embedded 3D printing of strain sensors within highly stretchable elastomers. Adv Mater. 26:6307–12. [DOI] [PubMed] [Google Scholar]
- Nalbach G, Hengstenberg R.. 1994. The halteres of the blowfly Calliphora: II. Three-dimensional organization of compensatory reactions to real and simulated rotations. J Comp Physiol A. 175:695–708. [Google Scholar]
- Nelson NA, Wang X, Cook D, Carey EM, Nimmerjahn A.. 2019. Imaging spinal cord activity in behaving animals. Exp Neurol. 320:112974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishikawa K, Biewener AA, Aerts P, Ahn AN, Chiel HJ, Daley MA, Daniel TL, Full RJ, Hale ME, Hedrick TLet al. 2007. Neuromechanics: an integrative approach for understanding motor control. Integr Comp Biol. 47:16–54. [DOI] [PubMed] [Google Scholar]
- Ohradzansky MT, Rush ER, Riley DG, Mills AB, Ahmad S, McGuire S, Biggie H, Harlow K, Miles MJ, Frew EWet al. 2021. Multi-agent autonomy: advancements and challenges in subterranean exploration. arXiv preprint arXiv:2110.04390. [Google Scholar]
- Ohyama T, Schneider-Mizell CM, Fetter RD, Aleman JV, Franconville R, Rivera-Alba M, Mensh BD, Branson KM, Simpson JH, Truman JWet al. 2015. A multilevel multimodal circuit enhances action selection in Drosophila. Nature. 520:633–9. [DOI] [PubMed] [Google Scholar]
- Owaki D, Ishiguro A.. 2017. A quadruped robot exhibiting spontaneous gait transitions from walking to trotting to galloping. Sci Rep. 7:277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson KG. 2004. Generating the walking gait: role of sensory feedback. Prog Brain Res. 143:123–9. [DOI] [PubMed] [Google Scholar]
- Phelps JS, Hildebrand DGC, Graham BJ, Kuan AT, Thomas LA, Nguyen TM, Buhmann J, Azevedo AW, Sustar A, Agrawal Set al. 2021. Reconstruction of motor control circuits in adult Drosophila using automated transmission electron microscopy. Cell. 184:759–774.e18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picton LD, Bertuzzi M, Pallucchi I, Fontanel P, Dahlberg E, Björnfors ER, Iacoviello F, Shearing PR, El Manira A.. 2021. A spinal organ of proprioception for integrated motor action feedback. Neuron. 109:1188–1201.e7. [DOI] [PubMed] [Google Scholar]
- Prochazka A. 1996. Proprioceptive feedback and movement regulation. In: In: Handbook of Physiology. Section 12. Exercise: Regulation and Integration of Multiple Systems. Rowell L., Sheperd J.T., editor. 1st ed.Wiley: New York. 89–127. [Google Scholar]
- Proctor JL, Holmes P.. 2018. The effects of feedback on stability and maneuverability of a phase-reduced model for cockroach locomotion. Biol Cybern. 112:387–401. [DOI] [PubMed] [Google Scholar]
- Pulver SR, Bayley TG, Taylor AL, Berni J, Bate M, Hedwig B.. 2015. Imaging fictive locomotor patterns in larval Drosophila. J Neurophysiol. 114:2564–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ravbar P, Zhang N, Simpson JH.. 2021. Behavioral evidence for nested central pattern generator control of Drosophila grooming. eLife. 10:e71508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridgel AL, Frazier SF, DiCaprio RA, Zill SN.. 2000. Encoding of forces by cockroach tibial campaniform sensilla: implications in dynamic control of posture and locomotion. J Comp Physiol A Sens Neural Behav Physiol. 186:359–74. [DOI] [PubMed] [Google Scholar]
- Righetti L, Ijspeert AJ.. 2008. Pattern generators with sensory feedback for the control of quadruped locomotion. 2008 IEEE International Conference on Robotics and Automation, Pasadena, CA, USA. p. 819–24. [Google Scholar]
- Rossignol S, Dubuc R, Gossard J-P.. 2006. Dynamic sensorimotor interactions in locomotion. Physiol Rev. 86:89–154. [DOI] [PubMed] [Google Scholar]
- Roth E, Hall RW, Daniel TL, Sponberg S.. 2016. Integration of parallel mechanosensory and visual pathways resolved through sensory conflict. Proc Natl Acad Sci USA. 113:12832–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudomin P, Schmidt RF.. 1999. Presynaptic inhibition in the vertebrate spinal cord revisited. Exp Brain Res. 129:1–37. [DOI] [PubMed] [Google Scholar]
- Russo AD, Stanev D, Sabnis A, Danner SM, Ausborn J, Armand S, Ijspeert A. 2023. Investigating the roles of reflexes and central pattern generators in the control and modulation of human locomotion using a physiologically plausible neuromechanical model. Neurosciencepublished online (doi: 10.1101/2023.01.25.525432). [DOI] [PubMed] [Google Scholar]
- Ryckebusch S, Laurent G.. 1993. Rhythmic patterns evoked in locust leg motor neurons by the muscarinic agonist pilocarpine. J Neurophysiol. 69:1583–95. [DOI] [PubMed] [Google Scholar]
- Sane SP, McHenry MJ.. 2009. The biomechanics of sensory organs. Integr Comp Biol. 49:i8–i23. [Google Scholar]
- Santuz A, Laflamme OD, Akay T.. 2022. The brain integrates proprioceptive information to ensure robust locomotion. J Physiol. 600:5267–94. [DOI] [PubMed] [Google Scholar]
- Schilling M, Cruse H.. 2020. Decentralized control of insect walking: a simple neural network explains a wide range of behavioral and neurophysiological results. PLoS Comput Biol. 16:e1007804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sen R, Wang K, Dickson BJ.. 2019. TwoLumps ascending neurons mediate touch-evoked reversal of walking direction in Drosophila. Curr Biol. 29:4337–4344.e5. [DOI] [PubMed] [Google Scholar]
- Sherman A, Dickinson MH.. 2003. A comparison of visual and haltere-mediated equilibrium reflexes in the fruit fly Drosophila melanogaster. J Exp Biol. 206:295–302. [DOI] [PubMed] [Google Scholar]
- Shih B, Shah D, Li J, Thuruthel TG, Park Y-L, Iida F, Bao Z, Kramer-Bottiglio R, Tolley MT.. 2020. Electronic skins and machine learning for intelligent soft robots. Sci Robot. 5:eaaz9239. [DOI] [PubMed] [Google Scholar]
- Sivitilli DM, Smith JR, Gire DH.. 2022. Lessons for robotics from the control architecture of the octopus. Front Robot AI. 9: 862391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skylar-Scott MA, Mueller J, Visser CW, Lewis JA. 2019. Voxelated soft matter via multimaterial multinozzle 3D printing. Nature. 575:330–5. [DOI] [PubMed] [Google Scholar]
- Song W, Onishi M, LY Jan, Jan YN.. 2007. Peripheral multidendritic sensory neurons are necessary for rhythmic locomotion behavior in Drosophila larvae. Proc Natl Acad Sci USA. 104:5199–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sreetharan PS, Whitney JP, Strauss MD, Wood RJ.. 2012. Monolithic fabrication of millimeter-scale machines. J Micromech Microeng. 22:055027. [Google Scholar]
- Sun X, Liu Y, Liu C, Mayumi K, Ito K, Nose A, Kohsaka H.. 2022. A neuromechanical model for Drosophila larval crawling based on physical measurements. BMC Biol. 20:130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szczecinski NS, Dallmann CJ, Quinn RD, Zill SN.. 2021. A computational model of insect campaniform sensilla predicts encoding of forces during walking. Bioinspir Biomim. 16:065001. [DOI] [PubMed] [Google Scholar]
- Takemura S, Hayworth KJ, Huang GB, Januszewski M, Lu Z, Marin EC, Preibisch S, Xu CS, Bogovich J, Champion ASet al. 2023. A connectome of the male Drosophila ventral nerve cord. bioRxiv 2023.06.05.543757. [Google Scholar]
- Thakur CS, Molin JL, Cauwenberghs G, Indiveri G, Kumar K, Qiao N, Schemmel J, Wang R, Chicca E, Olson Hasler Jet al. 2018. Large-scale neuromorphic spiking array processors: a quest to mimic the brain. Front Neurosci. 12:891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tranzatto M, Miki T, Dharmadhikari M, Bernreiter L, Kulkarni M, Mascarich F, Andersson O, Khattak S, Hutter M, Siegwart Ret al. 2022. CERBERUS in the DARPA Subterranean Challenge. Sci Robot. 7:eabp9742. [DOI] [PubMed] [Google Scholar]
- Tsubouchi A, Yano T, Yokoyama TK, Murtin C, Otsuna H, Ito K.. 2017. Topological and modality-specific representation of somatosensory information in the fly brain. Science. 358:615–23. [DOI] [PubMed] [Google Scholar]
- Tuthill JC, Azim E.. 2018. Proprioception. Curr Biol. 28:R194–203. [DOI] [PubMed] [Google Scholar]
- Tuthill JC, Wilson RI.. 2016. Mechanosensation and adaptive motor control in insects. Curr Biol. 26:R1022–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaadia RD, Li W, Voleti V, Singhania A, Hillman EMC, Grueber WB.. 2019. Characterization of proprioceptive system dynamics in behaving Drosophila larvae using high-speed volumetric microscopy. Curr Biol. 29:935–944.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valentine AD, Busbee TA, Boley JW, Raney JR, Chortos A, Kotikian A, Berrigan JD, Durstock MF, Lewis JA.. 2017. Hybrid 3D printing of soft electronics. Adv Mater. 29:1703817. [DOI] [PubMed] [Google Scholar]
- Walker RG, Willingham AT, Zuker CS.. 2000. A Drosophila mechanosensory transduction channel. Science. 287:2229–34. [DOI] [PubMed] [Google Scholar]
- Wallén P, Williams TL.. 1984. Fictive locomotion in the lamprey spinal cord in vitro compared with swimming in the intact and spinal animal. J Physiol. 347:225–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson AHD. 1992. Presynaptic modulation of sensory afferents in the invertebrate and vertebrate nervous system. Comp Biochem Physiol A Physiol. 103:227–39. [DOI] [PubMed] [Google Scholar]
- Weber AI, Daniel TL, Brunton BW.. 2021. Wing structure and neural encoding jointly determine sensing strategies in insect flight. PLoS Comput Biol. 17:e1009195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wensing PM, Wang A, Seok S, Otten D, Lang J, Kim S.. 2017. Proprioceptive actuator design in the MIT Cheetah: impact mitigation and high-bandwidth physical interaction for dynamic legged robots. IEEE Trans Robot. 33:509–22. [Google Scholar]
- Wilson DM. 1961. The central nervous control of flight in a locust. J Exp Biol. 38:471–90. [Google Scholar]
- Winding M, Pedigo BD, Barnes CL, Patsolic HG, Park Y, Kazimiers T, Fushiki A, Andrade IV, Khandelwal A, Valdes-Aleman Jet al. 2023. The connectome of an insect brain. Science. 379:eadd9330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf H, Burrows M.. 1995. Proprioceptive sensory neurons of a locust leg receive rhythmic presynpatic inhibition during walking. J Neurosci. 15:5623–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolff SB, Ölveczky BP.. 2018. The promise and perils of causal circuit manipulations. Curr Opin Neurobiol. 49:84–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wood RJ, Avadhanula S, Sahai R, Steltz E, Fearing RS.. 2008. Microrobot design using fiber reinforced composites. J Mech Des. 130:052304. [Google Scholar]
- Wu M-Y, Carbo-Tano M, Mirat O, Lejeune F-X, Roussel J, Quan FB, Fidelin K, Wyart C.. 2021. Spinal sensory neurons project onto the hindbrain to stabilize posture and enhance locomotor speed. Curr Biol. 31:3315–3329.e5. [DOI] [PubMed] [Google Scholar]
- Zarin AA, Mark B, Cardona A, Litwin-Kumar A, Doe CQ.. 2019. A multilayer circuit architecture for the generation of distinct locomotor behaviors in Drosophila. eLife. 8:e51781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zill SN, Keller BR, Duke ER.. 2009. Sensory signals of unloading in one leg follow stance onset in another leg: transfer of load and emergent coordination in cockroach walking. J Neurophysiol. 101:2297–304. [DOI] [PubMed] [Google Scholar]
- Zill SN, Schmitz J, Büschges A.. 2004. Load sensing and control of posture and locomotion. Arthropod Struct Dev. 33:273–86. [DOI] [PubMed] [Google Scholar]
- Zyhowski WP, Zill SN, Szczecinski NS.. 2023. Adaptive load feedback robustly signals force dynamics in robotic model of Carausius morosus stepping. Front Neurorobot. 17:1125171. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing is not applicable as no new data were generated or analyzed in this study.