Abstract
Toxoplasmosis is a zoonosis caused by Toxoplasma gondii, an obligate intracellular parasite. Clinical presentation of infection depends on the age and immune status of the patient. In immunocompetent patient, it may present as a non-specific lymphadenopathy and self- limiting illness. In contrast, in immunocompromised patients it can be a life-threatening infection. We present a series of 8 cases of toxoplasma lymphadenitis diagnosed in our institute in last two years. Lymphadenopathy raises a suspicion of malignancy; however, diagnosis of reactive lymphadenitis often marks the end of diagnostic evaluation. Careful morphological evaluation, the classic triad, often can direct at a conclusive diagnosis. We are presenting a case series to draw attention to this entity as majority of the published literature is in the form of case report and serological surveys and very occasional study showing both the histopathology and serology in tandem.
Introduction
Toxoplasmosis is a zoonotic infection caused by Toxoplasma gondii, which is transmitted by contaminated food or water, undercooked meat, or transplacentally [1, 2]. It has various clinical subtypes, including acute, CNS, congenital, ocular, or disseminated toxoplasmosis. The most common presentation is flu-like symptoms with lymphadenopathy, usually observed in the posterior cervical region, axillary, inguinal, or anterior chest wall [3]. Rarely, generalized lymphadenopathy or hepatosplenomegaly can occur. Histologic examination alongwith serologic studies are required for establishing diagnosis. Globally, seroprevalence is estimated to be 30–50%, with higher rates in Latin America and Western Africa. In a study from India an overall seropositivity of 21%, with IgG and IgM seropositivity of 5.7% and 15.3%, respectively [4, 5].
Case Series
Retrospective data analysis was done for patients presenting as lymphadenopathy to our institute during the period of January 2021 to December 2022. These patients had undergone lymph node excision biopsy and histopathological evaluation with haematoxylin and eosin stained slides. Biopsies showing the classically described triad of reactive lymphoid follicles, monocytoid cells along with non-necrotizing epithelioid microgranulomas were included in study and serological testing for toxoplasma was undertaken where possible. None of the patient were seropositive for Human immunodeficiency virus (HIV), Hepatitis B and C virus, which were done as part of prebiopsy workup.
Out of the eight cases histologically suspected to be toxoplasma lymphadenitis, predominantly were males (male: female ratio 7:1). The mean age of the population was 24.25 ± 9.8 years. Cervical lymphadenopathy was the most frequent complaint with three cases showing generalized lymphadenopathy on radiological evaluation. The mean size of lymph node was 2.6 cm (1.3–4.5 cm). All patients had complete blood counts and peripheral smear examination within normal limit. Table 1 shows the relevant clinical features of enrolled patients.
Table 1.
Epidemiological and clinical features of the cases
| Cases | Age | Gender | Lymph node | Presenting complaints | Size |
|---|---|---|---|---|---|
| Case 1 | 19 | M | Submental lymph nodes | submental neck swelling | 4.5 × 3 × 2 cm |
| Case 2 | 22 | M | Right cervical level IV lymph nodes | generalised lymphadenopathy 1 month | 2 × 1.5 × 1.2 cm |
| Case 3 | 17 | M | Right cervical nodes | lymphadenopathy 2 months duration | NA |
| Case 4 | 39 | M | Right axillary lymph nodes | lymphadenopathy | 4.5 × 2 × 1.5 cm |
| Case 5 | 43 | F | Right level II lymph nodes | generalised lymphadenopathy | 2.5 × 1.5 × 1.3 cm |
| Case 6 | 17 | M | Right level 5 lymph nodes | generalized lymphadenopathy | 1.4 × 1.1 × 0.3 cm |
| Case 7 | 20 | M | Submental neck swelling | lymphadenopathy | 1.3 × 1.2 × 0.7 cm |
| Case 8 | 17 | M | Multiple neck nodes | multiple neck nodes | 2 × 1.8 × 0.2 cm |
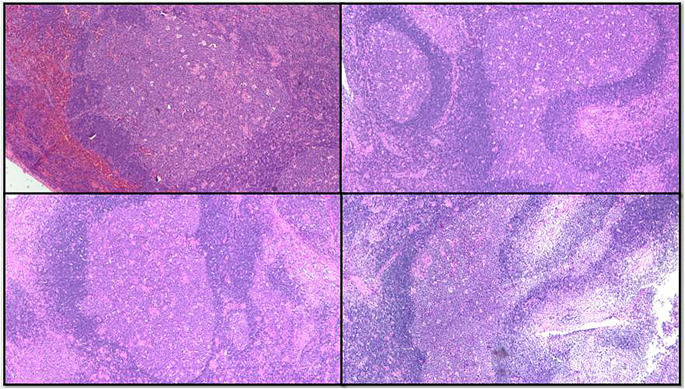
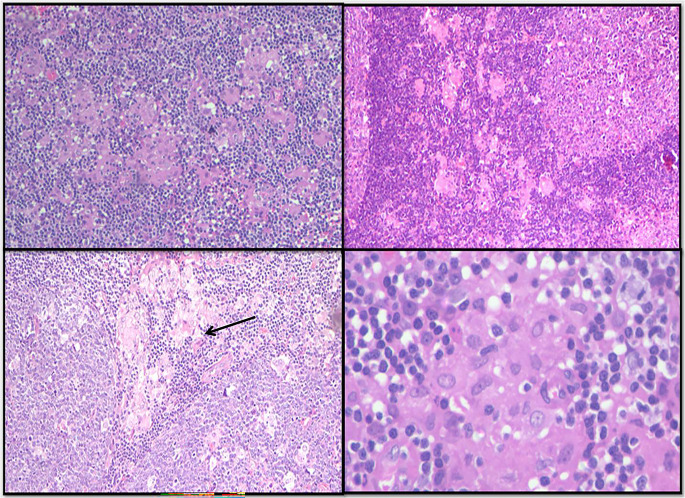
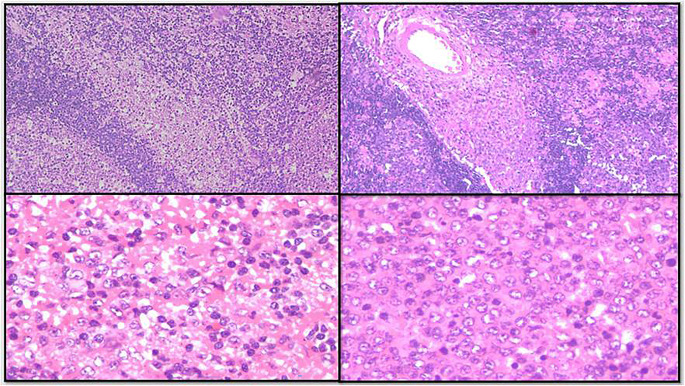
Histomorphology features in the lymph nodes showed preserved architecture with minimal capsular involvement. The classical triad of florid follicular hyperplasia with prominent germinal centres showing indistinct margins and numerous tingible body macrophages (Fig. 1), numerous microgranulomas (collections of epithelioid cells with less than 25 nuclei) (Fig. 2) diffusely scattered in interfollicular and paracortical regions with few seen abutting the mantle zones and monocytoid B cells characterized by an abundant pale eosinophilic cytoplasm, indented nuclei with inconspicuous nucleoli and open chromatin were seen. These cells are commonly located in the perivascular region or adjacent to sinuses (Fig. 3). None of the cases showed necrosis, macrogranulomas or giant cells.
Fig. 1.
Photomicrograph show the enlarged and expanded follicles with reactive germinal centre
Fig. 2.
Numerous clusters of epithelioid histiocytes are seen in interfollicular areas of the lymph node. A cluster of epithelioid histiocytes is seen encroaching a germinal centre (arrow)
Fig. 3.
- Monocytoid B cells expansion in sinuses and perivascular location
Six out of eight patients underwent serological test for Toxoplasma IgM and IgG antibodies by chemiluminescent microparticle immunoassay. The cut off points for positive results for IgM and IgG were > 0.60 IU/mL and > 3 IU/mL, respectively. All six patients showed positivity for IgG with mean titres of 1068 IU/mL, however, all but one showed positivity for IgM antibodies with mean titres of 24.5 IU/mL.
Discussion
In Indian subcontinent, persistent lymphadenopathy is common, due to infections like tuberculosis or haematolymphoid malignancies [6]. In this study, all patients had a history of lymph node enlargement lasting more than one month, leading to concerns and subsequent evaluation through lymph node biopsy. Although none of the patients had fever or weight loss at presentation, few reported a history of fever during detailed evaluation. The above presentation is typical in acute toxoplasmosis infection, where patients may be asymptomatic or experience an indolent flu-like symptoms not necessitating hospital visit. Lymph node enlargement may occur, generally clinically insignificant, or unnoticeable. These enlarged lymph nodes may persist for up to three months before subsiding.
Immune response against Toxoplasma is triggered when the parasite enters the draining lymph nodes and are captured by subcapsular sinus macrophages. In the early stages of infection, neutrophils clusters maybe seen within lymph nodes with high infection rates. Later stages, cytotoxic T cells move to these sites of infection and form antigen-specific clusters alongwith infected macrophages at the subcapsular sinus containing the parasites [7–9]. As a result, when these nodes are morphologically evaluated, the parasite is not detected. Instead, the various immunological reactions to the parasite can be observed in the form of the histopathological triad. In immunocompromised hosts, parasites evade elimination causing life-threatening infection, mostly affecting the central nervous system. They form cysts containing bradyzoites. The presence of tachyzoites within the cyst wall is the most common histopathological feature in this scenario. A granulomatous reaction to it is also commonly observed.
In a large retrospective study conducted at a tertiary referral hospital in India, lymph node excision biopsy was performed on 1724 patients, with tuberculous adenitis being observed in 31% of cases, malignancy in 26%, and non-specific lymphadenitis in 31% [6]. The remaining patients could not be attributed a specific etiology. Another study anticipated that 15% of unexplained lymphadenopathy is due to toxoplasmosis, typically affecting the cervical lymph nodes [10]. In our series, all patients presented with cervical lymphadenopathy, and three had generalized lymphadenopathy. The definitive diagnosis is usually established through lymph node biopsy of the most representative enlarged node. Excision biopsy is the preferred method as it allows for an evaluation of the entire nodal architecture, which is essential for arriving at a diagnosis. The diagnosis of reactive lymphadenitis often marks the end of the diagnostic evaluation.
In the present study, the histopathological triad was observed in 100% of cases. However, a study by Eapen et al. showed triad was highly specific (96.6%), with sensitivity of only 44.4% [11]. Out of the nine cases of toxoplasmosis in the above study, the limiting factor was the presence of monocytoid B cells, which were only seen in four. Microgranulomas are the most definitive feature of toxoplasmosis, as follicular hyperplasia and monocytoid cells can be seen in other conditions like infectious mononucleosis and rarely Hodgkin lymphoma. In the present study, cases received for an opinion were mainly diagnosed as Hodgkin lymphoma elsewhere. Firstly, the immunoblastic proliferation can mimic Reed-Sternberg (RS) cells, secondly, granulomas are also a feature described in Hodgkin lymphoma. Immunohistochemistry can help to exclude the lymphomatous process, as the immunoblasts are positive for CD20 and CD30, while RS cells are predominantly CD20 negative.
Serology testing for IgG and IgM antibodies is required for diagnosis of toxoplasmosis. IgM titers typically increase after one-week upto three months, gradually declining, reaching low levels after a year. IgG antibodies, on the other hand, appear two weeks after infection, peak at three months and may persist for the patient’s lifetime [12]. When IgM titers are low, an IgG avidity test is performed to confirm the infection timeline, with low levels indicating acute infection [13].
Follow-up of patients showed regression of lymphadenopathy, indicating a self-limiting infection. Other studies have also emphasized that symptoms are self-limiting and resolve within one to two months in 60% of immunocompetent patients, without antiparasitic treatment. However, 25% of patients may take two to four months to return to normal, and it may take four to six months in 8% of cases. Reactivation of the infection may occur if the patient becomes immunosuppressed [14].
Infected women, particularly during pregnancy, can lead to fatal congenital toxoplasmosis infections, pregnancy loss, stillbirth, and intrauterine malformations in the foetus. These infections are also part of the TORCH spectrum [15].
In conclusion, a good clinical examination, histomorphology, substantiated with serology, can aid the diagnosis of toxoplasma lymphadenitis. The presence of the triad comprising follicular hyperplasia, microgranulomas, and monocytoid B cells should raise suspicion for this condition. The accurate and timely identification of this condition can end the diagnostic evaluation chain.
Author contribution
Manasi Chetan Mundada, Faiq Ahmed and Snehil Kumar formulated the case series. Snehil Kumar drafted the initial manuscript. Manasi Chetan Mundada, Faiq Ahmed, K Suseela served as senior consultant from Department of Laboratory Medicine while L.M. Chandrasekhara Rao.S supervised and served as senior consultants from Department of Head and Neck Oncology. All senior consultants reviewed and revised the manuscript, and approved the final manuscript as submitted.
Declarations
Financial Support and Sponsorship
Nil.
Disclosure of Potential Conflicts of Interest
There are no conflicts of interest by any authors.
Research involving human participants and/or animals: No new patients or samples from patients were recruited in the study. No animal experiments were performed during the study.
Informed Consent
Informed consent is obtained from all patients evaluated in our institute and duly archived under Department of Medical records.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Manasi Chetan Mundada, Email: manasicmundada@gmail.com.
Faiq Ahmed, Email: dr.faiq@gmail.com.
Snehil Kumar, Email: snehilsimulation25@gmail.com.
Suseela Kodandapani, Email: suseela.kpani@gmail.com.
L.M. Chandrasekhara Rao, Email: drcsrao@induscancer.com.
References
- 1.Tenter AM, Heckeroth AR, Weiss LM (2000 Nov) Toxoplasma gondii: from animals to humans. Int J Parasitol 30(12–13):1217–1258 [DOI] [PMC free article] [PubMed]
- 2.Tenter AM (2009 Mar) Toxoplasma gondii in animals used for human consumption. Mem Inst Oswaldo Cruz 104:364–369 [DOI] [PubMed]
- 3.Pol JN, Patil SJ, Nisar Z, Jagdale RV (2021) Cyto-serological correlation in toxoplasma lymphadenitis: is bypassing biopsy plausible? IP Arch Cytol Histopathol Res. 15(2):103–107
- 4.Flegr J, Prandota J, Sovičková M, Israili ZH. Toxoplasmosis–a global threat. Correlation of latent toxoplasmosis with specific disease burden in a set of 88 countries. PLoS ONE. 2014;9(3):e90203. doi: 10.1371/journal.pone.0090203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mewara A, Singh S, Khurana S, Gupta P, Sehgal R. Seroprevalence of Toxoplasmosis at a Tertiary Care Centre in North India from 2004 to 2014. Indian J Med Microbiol. 2019;37(3):351–357. doi: 10.4103/ijmm.IJMM_19_327. [DOI] [PubMed] [Google Scholar]
- 6.Mohan A, Reddy MK, Phaneendra BV, Chandra A. Aetiology of peripheral lymphadenopathy in adults: analysis of 1724 cases seen at a tertiary care teaching hospital in southern India. Natl Med J India. 2007;20(2):78–80. [PubMed] [Google Scholar]
- 7.Yap GS, Ling Y, Zhao Y. Autophagic elimination of intracellular parasites: convergent induction by IFN-gamma and CD40 ligation? Autophagy. 2007;3(2):163–165. doi: 10.4161/auto.3655. [DOI] [PubMed] [Google Scholar]
- 8.Stafford JL, Neumann NF, Belosevic M. Macrophage-mediated innate host defense against protozoan parasites. Crit Rev Microbiol. 2002;28(3):187–248. doi: 10.1080/1040-840291046731. [DOI] [PubMed] [Google Scholar]
- 9.Taylor GA, Feng CG, Sher A. Control of IFN-gamma-mediated host resistance to intracellular pathogens by immunity-related GTPases (p47 GTPases) Microbes Infect. 2007;9(14–15):1644–1651. doi: 10.1016/j.micinf.2007.09.004. [DOI] [PubMed] [Google Scholar]
- 10.: WHO Meeting of Investigators on Toxoplasmosis, Geneva S, Organization WH (1968) Toxoplasmosis: report of a WHO meeting of investigators [meeting held in Geneva from 25 to 29 November 1968] [Internet]. World Health Organization; 1969 [cited 2023 Apr 11]. Available from: https://apps.who.int/iris/handle/10665/40753
- 11.Eapen M, Mathew CF, Aravindan KP (2005 Nov) Evidence based criteria for the histopathological diagnosis of toxoplasmic lymphadenopathy. J Clin Pathol 58(11):1143–1146 [DOI] [PMC free article] [PubMed]
- 12.Montoya JG Laboratory diagnosis of Toxoplasma gondii infection and toxoplasmosis.J Infect Dis. 2002 Feb15;185(Supplement_1):S73–82 [DOI] [PubMed]
- 13.Villard O, Cimon B, L’Ollivier C, Fricker-Hidalgo H, Godineau N, Houze S et al (2016 Jan) Serological diagnosis of Toxoplasma gondii infection: recommendations from the French National Reference Center for Toxoplasmosis. Diagn Microbiol Infect Dis 84(1):22–33 [DOI] [PubMed]
- 14.Rubinstein E, Keynan Y Lymphadenopathy - ClinicalKey [Internet]. 4th ed. Elsevier; [cited 2023 Apr 11]. 136–145 p. Available from:https://www.clinicalkey.com/#!/content/3-s2.0-B9780702062858000150?scrollTo=%23c00015
- 15.Deganich M, Boudreaux C, Benmerzouga I (2023 Jan) Toxoplasmosis infection during pregnancy. Trop Med Infect Dis 8(1):3 [DOI] [PMC free article] [PubMed]