Abstract
Objective
To evaluate epidemiology and outcomes among very preterm infants (<32 weeks’ gestation) with culture-positive and culture-negative late-onset sepsis (LOS).
Design
Cohort study using a nationwide, population-based registry.
Setting
21 neonatal units in Norway.
Participants
All very preterm infants born 1 January 2009–31 December 2018 and admitted to a neonatal unit.
Main outcome measures
Incidences, pathogen distribution, LOS-attributable mortality and associated morbidity at discharge.
Results
Among 5296 very preterm infants, we identified 582 culture-positive LOS episodes in 493 infants (incidence 9.3%) and 282 culture-negative LOS episodes in 282 infants (incidence 5.3%). Extremely preterm infants (<28 weeks’ gestation) had highest incidences of culture-positive (21.6%) and culture-negative (11.1%) LOS. The major causative pathogens were coagulase-negative staphylococci (49%), Staphylococcus aureus (15%), group B streptococci (10%) and Escherichia coli (8%). We observed increased odds of severe bronchopulmonary dysplasia (BPD) associated with both culture-positive (adjusted OR (aOR) 1.7; 95% CI 1.3 to 2.2) and culture-negative (aOR 1.6; 95% CI 1.3 to 2.6) LOS. Only culture-positive LOS was associated with increased odds of cystic periventricular leukomalacia (cPVL) (aOR 2.2; 95% CI 1.4 to 3.4) and severe retinopathy of prematurity (ROP) (aOR 1.8; 95% CI 1.2 to 2.8). Culture-positive LOS-attributable mortality was 6.3%, higher in Gram-negative (15.8%) compared with Gram-positive (4.1%) LOS, p=0.009. Among extremely preterm infants, survival rates increased from 75.2% in 2009–2013 to 81.0% in 2014–2018, p=0.005. In the same period culture-positive LOS rates increased from 17.1% to 25.6%, p<0.001.
Conclusions
LOS contributes to a significant burden of disease in very preterm infants and is associated with increased odds of severe BPD, cPVL and severe ROP.
Keywords: Intensive Care Units, Neonatal; Microbiology; Neonatology; Sepsis
WHAT IS ALREADY KNOWN ON THIS TOPIC
Very preterm infants (VPIs) have higher rates of late-onset sepsis (LOS) and sepsis-associated morbidity and mortality than term infants.
Different pathogens may have different prognosis with regards to acute illness and subsequent association with morbidities of preterm infants at discharge.
WHAT THIS STUDY ADDS
In Norway, among VPIs, the incidence of culture-positive LOS was 9.3% and culture-negative LOS was 5.3%.
Overall culture-positive LOS-attributable mortality was low (6.3%), but higher in Gram-negative (15.8%) than Gram-positive LOS (4.1%).
Culture-positive and culture-negative LOS were associated with a 1.6-fold to 1.7-fold increased adjusted odds for severe BPD.
An improved survival and more proactive care for extremely premature infants in the last 5-year period was concomitantly associated with increasing rates of LOS.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
LOS remains associated with considerable morbidity and mortality, emphasising the need for reducing invasive procedures and a continuous focus on hygiene in preterm infants.
An observed increase in culture-positive LOS among extremely premature infants warrants implementation of quality improvement initiatives to reduce LOS rates.
Background
Very preterm infants (VPIs) have substantially higher rates of sepsis than both term infants, older children and adults.1 A divergent immune-response regulation in case of an invasive infection may partly explain the high vulnerability in this age group.2 The risk of infection is further aggravated by frequent need for invasive therapy and prolonged hospitalisations that increases the risk of colonisation with a nosocomial, antibiotic-resistant commensal flora.3–6 Hospital-acquired late-onset sepsis (LOS) represents a disease that is partially preventable by improved care.7 Despite improvements in hygiene and neonatal care, a substantial proportion of VPI still develop LOS, which has been associated with increased mortality and long-term morbidity.8
Regular surveillance of LOS epidemiology is important to guide prevention and treatment strategies. Epidemiology and distribution of pathogens causing culture-positive LOS changes over time and varies between countries.9 There are few recent population-based studies on LOS in preterm infants.7 Contemporary information regarding LOS rates, risk factors and associated mortality and morbidities are therefore needed. A sepsis diagnosis without growth in blood culture is common in adults.10 In contrast, culture-negative neonatal sepsis is a controversial entity but contributing to substantial antibiotic exposure.11 12 Thus, important data on the proportion of preterm infants treated for a presumed late-onset infection, coined as culture-negative LOS, is often missing.
In this study, we aimed to identify the burden of both culture-positive and culture-negative LOS and its associated mortality and morbidity until discharge in an unselected population of VPIs born in Norway between 2009 and 2018.
Methods
Population and setting
The study includes all preterm infants born below 32 weeks’ gestation admitted to any neonatal intensive care unit (NICU) in Norway during the 10-year period between 1 January 2009 and 31 December 2018. At this time, no national guideline for empirical antibiotic therapy for LOS existed in Norway.
Data source and approvals
Data were obtained from the Norwegian Neonatal Network (NNN), a national population-based registry comprising all 21 NICUs across four health trust regions. The registry includes daily registrations of all investigations, treatments and diagnoses on all infants admitted to one or several Norwegian NICUs until final discharge.13 14 The completeness of data is checked for important diagnostic variables (including if blood cultures are obtained when starting antibiotics) and for central morbidities related to preterm birth. We retrieved background data including date and year of birth, mode of delivery, birth weight (BW), gestational age (GA), clinical risk index for babies 2 (CRIB2) score,15 Apgar score, plurality, sex, antenatal steroids and clinical data including antibiotic use, days of mechanical ventilation (MV) and parenteral nutrition (with/without lipids), blood culture results, the highest C reactive protein (CRP) value during LOS, common morbidities of prematurity, LOS-attributable mortality and overall mortality before discharge. The number of live-born infants was obtained from the Medical Birth Registry of Norway (MBRN). Both the MBRN and NNN collect personal identifiable data without consent according to the MBRN Regulation and the Personal Health Data Filing Systems Act of Norway.
Definitions and outcomes
Culture-positive LOS was defined as growth of bacteria or fungi in a blood culture collected after 3 days of life and antibiotic treatment for at least 5 days or death before 5 days during this LOS episode in an infant with clinical signs of sepsis.14 An infant with growth of coagulase-negative staphylococci (CoNS) in the blood was classified as culture-positive LOS if the infant had clinical symptoms, received minimum 5 days antibiotic therapy and had a CRP >10 mg/L. We also included as culture-positive LOS an episode with polymicrobial growth in the blood culture if there was one significant pathogen along with either a CoNS or another typical contaminant. An episode with a positive blood culture was defined as not LOS based on the following criteria: growth of two or more CoNS species or growth of other typical contaminants (micrococci, propionibacteria, corynebacteria or diphtheroids), antibiotic therapy shorter than 5 days and/or when the clinicians did not label the episode with a sepsis (ICD-10 (International Classification of Diseases 10th Revision), P36.1–8) diagnosis in the national registry. A CLABSI was defined as a case of culture-positive LOS in a patient with an indwelling catheter in place within 2 days prior to the onset of symptoms/infection.16 Multiple positive blood cultures growing the same pathogen within a 7-day period were considered a single LOS episode. Finally, culture-negative LOS (ICD-10, P36.9) was defined according to national criteria that includes: (1) clinical signs of sepsis, (2) maximum CRP >30 mg/L, (3) minimum antibiotic therapy for 5 days and (4) excluded other common causes for the clinical picture.17 Onset of LOS was defined as the day a blood culture was drawn or the day when antibiotic therapy was started for culture-negative LOS. Culture negative LOS-attributable death was defined as death within 7 days of the positive blood culture or start of antibiotics.6
We defined severe BPD as receiving any respiratory support, other than only supplemental oxygen, at 36+0 weeks postmenstrual age (PMA) and severe retinopathy of prematurity (ROP) as treated with laser or antivascular endothelial growth factor in either eye. Intracranial haemorrhage (ICH) grade 3–4 according to Papile and cystic periventricular leukomalacia (cPVL stage ≥2)18 was determined by the most severe cranial ultrasonogram during the hospitalisation. Small for GA (SGA) was defined as birth weight under 10th percentile using Norwegian growth charts.19
Statistical methods
Statistical analyses were performed with SPSS V.26.0 (SPSS, Armork, NY, USA). Results are presented as medians with IQR or proportions (%). Differences between groups are analysed using non-parametric tests (Mann-Whitney or Kruskal-Wallis) for continuous variables and the χ2 test for categorical data, as appropriate. We used logistic regression analysis to evaluate the association between different types of LOS and severe BPD, severe ROP and cPVL. Based on previous reports,20–24 and by drawing directed acyclic graphs, we selected relevant confounders for the different morbidities, including Apgar 5 min score, CRIB2 score, days of MV in the first week of life, days of antibiotic exposure in the first week of life, severe ICH and early-onset sepsis (EOS). During the 10-year study period, in Norway, there was from 2014 a trend towards increasingly proactive management of preterm infants born at 23 weeks’ gestation. Thus, we adjusted our analyses based on the study epochs 2009–2013 and 2014–2018. We present crude and adjusted ORs with 95% CI. Two-tailed p values <0.05 were considered statistically significant.
Results
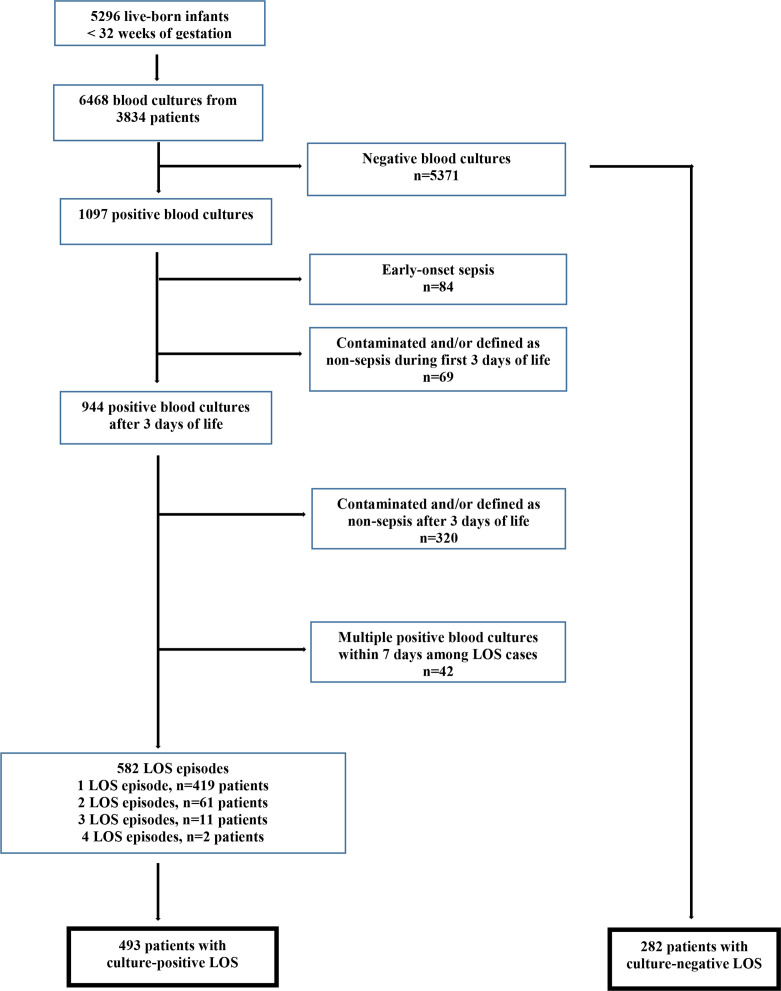
There were 601 668 live-born infants in Norway between 2009 and 2018. Of these, 5296 (0.88%) VPIs (<32 weeks’ gestation) were admitted to a NICU; 3646 (0.61%) infants born at 28–31 weeks’ gestation and 1650 (0.27%) extremely preterm infants (EPIs) born before 28 weeks’ gestation. A total of 6468 blood cultures were obtained from 3834 infants, and 1097 blood cultures showed growth of a potential pathogen (bacterium or fungus). After exclusion of contaminants, EOS episodes and LOS episodes with multiple positive cultures within 7 days or not labelled as sepsis, 582 culture-positive LOS episodes among 493 patients were identified. We also identified 282 patients who were diagnosed with culture-negative LOS and who never had any culture-positive LOS episode (figure 1).
Figure 1.
Study flow: participants and blood cultures.
Table 1 shows background characteristics, LOS incidence according to different pathogens, selected therapies, CRP response and major outcomes in all VPIs without and with LOS and presented by LOS-subtypes. During the 10-year study period, the overall culture-positive LOS incidence in all VPIs was 9.3% (493/5296); for 28–31 weeks infants 3.8% (139/3646) and for EPIs 21.6% (354/1650). The overall incidence of culture-negative LOS in all VPIs was 5.3% (282/5296); for 28–31 weeks infants 2.7% (99/3646) and for EPIs 11.1% (183/1650). Infants with all subtypes of LOS had significantly lower BW and were more immature compared with infants without LOS. The median (IQR) age at onset of the first LOS episode was 15 (9–28) days; 16 (9–30) days for 28–31 weeks infants and 14 (9–24) days for EPIs.
Table 1.
Background characteristics and outcomes in 5232 very preterm infants (<32 weeks’ gestation) without and with late-onset sepsis (LOS)
| Infants without LOS n=4457* |
Culture-negative LOS n=282 |
Culture-positive LOS n=493 |
Gram-positive, excl. CoNS-LOS n=163 |
CoNS LOS n=224 |
Gram-negative LOS n=95 |
Candida LOS n=11 |
|
| Perinatal and neonatal data | |||||||
| Antenatal steroids (%) | 84.3 | 90.6 | 92.9 | 90.2 | 93.8 | 98.9 | 90.9 |
| Birth weight (g), median (IQR) | 1277 (980,1555) | 838 (652,1112) | 830 (676,1034) | 853 (697,1030) | 806 (658,1039) | 839 (700,1034) | 725 (635,856) |
| Small for gestational age (%) | 25.9 | 33.0 | 29.4 | 33.8 | 31.7 | 17.9 | 9.1 |
| Gestational age (weeks), median (IQR) | 29+6 (28+0, 31+1) | 26+6 (25+1, 28+6) | 26+4 (25+0, 28+2) | 26+4 (25+4, 28+6) | 26+4 (24+6, 28+1) | 26+5 (25+0, 28+1) | 24+5 (24+2, 26+1) |
| Caesarean delivery (%) | 51.1 | 56.0 | 44 | 50.3 | 43.8 | 36.8 | 27.3 |
| Female (%) | 45.6 | 40.8 | 41.6 | 41.2 | 41.5 | 44.2 | 27.3 |
| Apgar 5 min, median (IQR) | 8 (7, 9) | 7 (5, 8) | 7 (6, 8) | 7 (6, 8) | 7 (6, 8) | 7 (6, 8) | 6 (5, 8) |
| Mechanical ventilation days, median (IQR) | 0 (0,2) | 11 (2,33) | 11 (3,30.5) | 10 (2,31) | 12.5 (4,31) | 9 (3,23) | 36 (29,48) |
| Central catheters days, median (IQR) | 1.0 (0,1) | 15.0 (8,27) | 17.0 (10,26) | 15.0 (7,25) | 17.5 (12,28) | 16.0 (9,27) | 27.0 (18,53) |
| Parenteral nutrition days, median (IQR) | 3.0 (0,8) | 13.0 (7,21) | 13.0 (9,24) | 11 (7,19) | 15 (11,27) | 12 (8,20) | 20 (11,45) |
| Incidence LOS (%) | NA | 5.3 | 9.3 | 3.1 | 4.2 | 1.8 | 0.2 |
| Proportion CLABSI among LOS (%) | NA | NA | 41.6 | 25.8 | 51.8 | 42.1 | 72.7 |
| Outcomes | |||||||
| Day of life onset LOS, median (IQR) | NA | 27 (15,45) | 15 (9,28) | 25 (14,40) | 12 (8,21) | 13 (8,20) | 17 (10,32) |
| LOS-attributable mortality (%) | NA | 2.1 | 6.3 | 4.3 | 4.0 | 15.8 | 0 |
| Days to death from LOS onset, median (IQR) | NA | 4 (3,5) | 1 (1,2) | 1 (1,3) | 2 (1,2) | 2 (0,3) | NA |
| C reactive protein † (mg/L), median (IQR) | NA | 75 (48,138) | 53 (28,53) | 62 (35,110) | 46 (27,75) | 70 (26,131) | 47 (30,72) |
| Severe BPD (%) | 9 | 41.1 | 37.9 | 39.3 | 40.6 | 30.5 | 45.5 |
| Severe ROP (%) | 1.8 | 8.2 | 9.3 | 9.2 | 9.4 | 10.5 | 0 |
| Cystic PVL (%) | 2.3 | 3.2 | 7.3 | 8.6 | 5.4 | 10.5 | 9.1 |
| Severe ICH (%) | 3.4 | 8.2 | 9.7 | 7.4 | 8.4 | 14.7 | 16.7 |
| Surgically treated NEC (%) | 0.7 | 6.8 | 8.1 | 3.7 | 9.3 | 12.6 | 8.3 |
| Postnatal steroids (%) | 7.2 | 37.2 | 39.1 | 37.4 | 43.6 | 28.4 | 72.7 |
| Days of hospitalisation, median (IQR) | 46 (33, 66) | 89 (62,115) | 88 (61, 115) | 91 (61, 114) | 89 (67, 118) | 78 (48, 108) | 113 (92, 126) |
| PMA at discharge (weeks), median (IQR) | 36+4 (35+3, 38+1) | 39+4 (37+2, 42+2) | 39+2 (37+6, 41+6) | 39+2 (37+5, 39+2) | 39+3 (37+0, 42+2) | 38+2 (35+5, 41+3) | 40+3 (38+5, 43+0) |
LOS attributable mortality; defined as death within 7 days of positive blood culture or for culture neg. when blood culture was obtained divided by total affected infants. Severe bronchopulmonary dysplasia (BPD); need for any respiratory support, other than only O2, at 36+0 weeks postmenstrual age (PMA). Severe retinopathy of prematurity (ROP); receiving therapy with laser or antivascular endothelial growth factor. Cystic periventricular leukomalacia. Intracranial haemorrhage (ICH): grade 3–4 according to Papile. Central catheter days include any of the following: umbilical artery and/or vein, peripherally inserted central catheter and central venous catheter. Parenteral nutrition days; total days of parenteral nutrition, with or without lipids.
*64 infants with EOS, but without LOS, were excluded from this presentation/analysis.
†C reactive protein (CRP) maximum value; 211/775 (27%) of CRP values were missing. However, for all CoNS-LOS cases, the CRP value was traced and reported.
CLABSI, central line associated bloodstream infection; CoNS, coagulase-negative staphylococci; PVL, periventricular leukomalacia.
Table 2 shows that CoNS was involved in 49% of culture-positive LOS followed by Staphylococcus aureus (15%), group B streptococci (10%) and Escherichia coli (8%). The overall culture-positive LOS-attributable mortality was 6.3%, but significantly higher in Gram-negative (15.8%) versus Gram-positive (4.0%) LOS, p=0.009. The culture-negative LOS-attributable mortality rate was 2.1%.
Table 2.
Pathogen distribution in 582 late-onset sepsis (LOS) episodes and LOS-attributable mortality in 493 very preterm infants
| Culture-positive LOS episodes n=582 (%*) |
LOS attributable death in 493 patients with a positive blood culture (%†) |
|
| Gram positive bacteria, excl. CoNS | 183 (31.4) | 7/163 (4.3) |
| Staphylococcus aureus‡ | 89 (15.1) | 2 (2.2) |
| Group B streptococcus | 55 (9.5) | 2 (3.6) |
| Enteroccocus spp. | 24 (4.1) | 2 (8.3) |
| Bacillus spp. | 4 (0.7) | 1 (2.5) |
| Other | 11 (1.9) | 0 (0) |
| CoNS | 282 (48.5) | 9/224 (4.0) |
| Gram-negative bacteria | 105 (18.0) | 15/95 (15.8) |
| Escherichia coli | 49 (8.4) | 7 (14) |
| Klebsiella spp. | 26 (4.5) | 5 (19.2) |
| Enterobacter spp. | 18 (3.1) | 1 (5.6) |
| Serratia spp. | 4 (0.7) | 0 (0) |
| Other | 4 (0.7) | 0 (0) |
| Salmonella spp. | 2 (0.3) | 1 (50) |
| Pseudomonas spp. | 1 (0.2) | 1 (100) |
| Acinetobacter spp. | 1 (0.2) | 0 (0) |
| Candida spp. | 11 (1.9) | 0/11 (0) |
| Other | 1 (0.2) | 0 (0) |
| Total | 582 | 31/493 (6.3) |
LOS attributable death was defined as death within 7 days of positive blood culture.
* Percentage of all LOS cases.
†Percentage infants in each pathogen category.
‡Only one out of 89 S. aureus cases was methicillin resistant.
CoNS, coagulase-negative staphylococci.
Overall survival to discharge was similar in the two 5-year periods 2009–2013 (90.9%) and 2014–2018 (92.0%). However, among EPIs, survival increased from 75.2% in 2009–2013 to 81.0% in 2014–2018, p=0.005 (online supplemental table 1). In the second 5-year period, these EPIs also had an increase in duration of parenteral nutrition from median 10 to 12 days, p<0.001 (online supplemental table 1). The improved survival was associated with a concomitant increase in the overall rates of LOS in extremely preterm infants, increasing from 17.1% in 2009–2013 to 25.6% in 2014–2018, p<0.001.
fetalneonatal-2022-324977supp001.pdf (33.2KB, pdf)
Table 3 shows associations between LOS and severe BPD, cPVL and severe ROP. After adjusting for confounders, we found that both culture-positive and culture-negative LOS were associated with increased odds of severe BPD. Only culture-positive LOS was associated with increased odds of cPVL and severe ROP.
Table 3.
Adjusted and crude OR for severe morbidities in relation to different types of LOS
| Comparison | Severe BPD* | Severe ROP† | Cystic PVL‡ | |||
| 22–31 weeks’ GA | 22–27 weeks | 22–31 weeks | 22–27 weeks | 22–31 weeks | 22–27 weeks | |
| Culture-positive LOS versus no sepsis§ | ||||||
| Adjusted OR | 1.7 (1.3–2.2) | 1.5 (1.1–2.1) | 1.8 (1.2–2.8) | 1.8 (1.2–2.9) | 2.2 (1.4–3.4) | 2.1 (1.2–3.4) |
| Crude OR | 6.3 (5.1–7.8) | 3.2 (2.5–4.1) | 5.7 (3.9–8.3) | 2.5 (1.6–3.7) | 3.4 (2.3–5.0) | 2.0 (1.3–3.3) |
| Gram negative LOS versus no sepsis | 2.5 | |||||
| Adjusted OR | 1.3 (0.7–2.2) | 1.2 (0.7–2.4) | 1.6 (0.7–3.7) | 1.7 (0.8–4.0) | 3.2 (1.5–7.0) | 2.8 (1.2–6.7) |
| Crude OR | 4.5 (2.9–7.1) | 2.5 (1.5–4.1) | 6.5 (3.3–13.0) | 2.6 (1.2–5.5) | 5.1 (2.6–10.1) | 2.5 (1.1–5.7) |
| Gram-positive LOS versus no sepsis | ||||||
| Adjusted OR | 2.1 (1.3–3.2) | 1.8 (1.1–2.9) | 2.2 (1.2–4.1) | 2.2 (1.2–4.1) | 3.3 (1.8–6.2) | 3.0 (1.5–6.0 |
| Crude OR | 6.7 (4.8–9.3) | 3.5 (2.3–5.2) | 5.6 (3.2–10.0) | 2.7 (1.5–4.9) | 4.1 (2.3–7.3) | 2.6 (1.3–5.1) |
| CoNS LOS versus no sepsis | ||||||
| Adjusted OR | 1.5 (1.1–2.3) | 1.6 (1.1–2.4) | 1.6 (0.9–3.0) | 1.6 (0.9–3.0) | 1.3 (0.6–2.6) | 1.4 (0.7–3.0) |
| Crude OR | 7.0 (5.3–9.3) | 3.5 (2.5–4.9) | 5.4 (3.5–9.4) | 2.4 (1.4–4.2) | 2.4 (1.3–4.5) | 1.6 (0.8–3.2) |
| Culture-negative LOS versus no sepsis | ||||||
| Adjusted OR | 1.6 (1.3–2.6) | 2.2 (1.4–3.2) | 1.8 (1.0–3.0) | 1.6 (0.9–2.8) | 1.0 (0.5–2.0) | 1.0 (0.4–2.3) |
| Crude OR | 7.3 (5.6–9.4) | 4.6 (3.3–6.4) | 4.9 (3.1–8.0) | 2.1 (1.3–3.7) | 1.4 (0.7–2.9) | 0.9 (0.4–2.0) |
Severe bronchopulmonary dysplasia (BPD); 1 value was missing. Severe retinopathy of prematurity (ROP), 9 values were missing.
*Adjusted for CRIB2, mechanical ventilation, antibiotic exposure in the first week of life and year of birth (2009–2013 vs 2014–2018).
†Adjusted for CRIB2, mechanical ventilation in the first week of life and year of birth (2009–2013 vs 2014–2018).
‡Adjusted for CRIB2, Apgar 5 min, EOS, intracranial haemorrhage grade 3–4 and year of birth (2009–2013 vs 2014–2018).
§No sepsis; neither culture-positive EOS nor LOS, n=4457 infants.
CRIB2, clinical risk index for babies 2; EOS, early-onset sepsis; LOS, late-onset sepsis; PVL, periventricular leukomalacia.
Discussion
In this 10-year population-based study, we present the burden of LOS in an unselected, contemporary cohort of more than 5000 infants born below 32 weeks’ gestation. LOS was uncommon in preterm infants between 28 and 31 weeks’ gestation. In contrast, more than 20% of EPIs developed a culture-positive LOS, and an additional 11% had an episode coined as culture-negative LOS. In EPIs, an improved survival in the last 5-year period was associated with a modest increase in duration of parenteral nutrition, but also a significant increase in culture-positive LOS incidence. The overall LOS-attributable mortality was low, but infants diagnosed with LOS had subsequent increased risk of inflammation-associated conditions like BPD, cPVL and ROP.2
There are divergent reports on the epidemiology of culture-positive LOS among VPIs.4 7–9 25–27 This is partly due to variations in definition of timing of LOS (defined as onset within 48 hours or up to 7 days after birth),5 28 but even more important due to different definitions of CoNS infections.25 28–30 Our definition of CoNS-LOS aligns with definitions suggested by the Vermont Oxford Network and a recent Dutch multicentre study, both requiring a single positive blood culture along with clinical symptoms and minimum 5-day antibiotic therapy.31 32 In the Dutch study, an increased inflammatory response (CRP >10 mg/L) was also added.31 In contrast, the revised CDC guidelines require two independently drawn positive blood cultures,29 despite common practice in many NICUs is to obtain only one culture before therapy. These discrepancies have great impact on incidence data comparison. The overall culture-positive LOS incidence of 9.3% in all VPIs and 21.6% in EPIs in our study is in the lower range of LOS incidences reported, ranging from 12% to 30% in VPIs and 20% to 40% in VPIs.3 4 31 33–35 However, a recent Swiss population-based study reported much lower LOS-incidences both among VPIs (6%) and EPIs (11%), respectively, but excluded CoNS infections in the absence of a catheter at the time of blood culture collection.7
The LOS pathogen distribution among all infants in our study is also in line with other recent international reports.4 7 25 28 36 CoNS was by far the most prevalent pathogen and induced a marked inflammatory response in most preterm infants, reflected by elevated CRP values. More than half of all CoNS-LOS episodes were classified as CLABSIs, and nearly all patients with CoNS-LOS had received parenteral nutrition. Increasing rates of CoNS infections are multifactorial, often attributed to invasive procedures, fragile skin, use of central lines and parental nutrition.30 Care for the VPIs implies weighing the potential risks and benefits of a given treatment, and the use of central lines is particularly essential for providing adequate parenteral nutrition. Faster advancement of enteral feeds and earlier removal of central lines is feasible and has been associated with lower rates of LOS in some, but not all studies.31 37 38 During the study period, there were no uniform Norwegian guideline on parenteral nutrition for VPIs.
Fungal LOS was rare in our study population. This stands in sharp contrast to a population-based Norwegian study from 1999 to 2000 in which Candida species emerged as the second most frequent pathogen with 12% of all LOS isolates.38 Increasing use of fungal prophylaxis in EPIs,39 although not universally in Norwegian tertiary NICUs, may partly explain this striking reduction in fungal infections.
LOS-attributable mortality in preterm infants remains a great concern but seems to be decreasing.40 In our study, Gram-negative LOS had the highest mortality rate of 15.8%, but it was still lower than in some previous studies reporting mortality rates up to 30%–40%.4–6 Culture-positive LOS-attributable mortality in our study (6.3%) was considerably lower than reported in most other studies,6 29 36 but slightly higher than in a recent report from the German Neonatal Network with 4.2%.34 Multiple reasons may explain the relatively low mortality rate, including a general improvement in neonatal care over the last decade. Prevalence of antimicrobial resistance in children is also low in Norway,41 leading to lower risk of antibiotic therapy failure.
We found higher adjusted odds of subsequent severe BPD, cPVL and severe ROP in patients who had experienced one or more culture-positive LOS episodes when compared with non-infected infants. These results are consistent with the existing literature indicating the detrimental effect of LOS and inflammation on both the lungs, eyes and the brain.2 4 7 21 25 34 36
There is a paucity of epidemiological data on culture-negative LOS in preterm infants, and whether culture-negative-LOS is a clinically significant entity remains controversial.11 12 Blood cultures may be falsely negative because of inappropriate sampling volume or if cultures are collected after commencement of antibiotic therapy. It has also been suggested that culture-negative LOS may reflect a milder form of LOS compared with culture-positive LOS.42 In our study, infants with culture-negative LOS had many features in common with those having a culture-positive LOS, apart from a lower LOS-attributable mortality, which was also observed in large study from China.42
The primary strength of this study is prospective collection of data on a daily basis with a standardised electronic registration. The population-based design avoids selection bias. The study period over 10 years allows evaluation of trends in incidence, as consistent diagnostic criteria were applied throughout the whole study period. Inaccuracies in data entry are inevitable in register studies but unlikely to change over time or geographic location. The main limitation is the reliance on a large number of physicians to register data. Given a long observation period, clinical practice changes may influence some of the outcomes. However, except for sporadic use of probiotics to EPIs during 2014–2017, there were no other supportive therapies for sepsis implemented in Norway during the study period. We also did not have information on blood culture volume or other inflammatory biomarkers. Finally, in the logistic regression analyses, we attempted to correct for relevant confounders, but we cannot exclude residual confounding.
A fundamental question is whether it is possible to reduce LOS incidence in the most immature infants and, if so, how? In the Canadian neonatal network, a continuous quality improvement project reduced LOS rates in 26–28 week-old infants, whereas LOS rates remained high at 31%–36% in infants below 26 weeks.35 We observed an unfortunate increase in LOS incidence in EPIs concomitantly with more proactive care, higher survival and intensification of parenteral nutrition. However, these associations do not have to represent any causal relationship.
In summary, this study describes the burden of LOS, both culture-positive but also the controversial entity culture-negative LOS, in an unselected population of VPIs. LOS remains associated with a considerable morbidity, and mortality was particularly high among Gram-negative LOS cases. Potentially preventable hospital-acquired infections are often caused by CoNS, and this pathogen caused around half of all LOS episodes. The increase in LOS incidence in the most immature infants is of great concern. It is paramount to explore preventative factors for LOS and to implement evidence-based care bundles rates,26 33 aiming to reduce LOS incidence also in the youngest and most vulnerable preterm infants.
Footnotes
Twitter: @ClausKlingenbe1
Collaborators: We would like to thank the neonatal units from the following hospital in Norway for contributing with data to the Norwegian Neonatal Network and thus making this study possible: Oslo University Hospital-Rikshospitalet, Oslo; Oslo University Hospital-Ullevål, Oslo; Akershus University Hospital, Lørenskog; Drammen Hospital, Drammen; Østfold Hospital Trust, Fredrikstad; Innlandet Hospital Trust, Lillehammer; Innlandet Hospital Trust, Elverum; Telemark Health Trust, Skien; Hospital of Southern Norway, Kristiansand; Hospital of Southern Norway, Arendal; Stavanger University Hospital, Stavanger; Haukeland University Hospital, Bergen; Fonna Health Trust, Haugesund; Health Sunnmøre Trust, Ålesund; Førde Health Trust, Førde; St. Olav University Hospital, Trondheim; Nord-Trøndelag Health Trust, Levanger; Nordland Central Hospital, Bodø; University Hospital of North Norway, Tromsø and Finnmark Health Trust, Hammerfest.
Contributors: ZH carried out the analyses, drafted the initial manuscript and revised the manuscript. HJS was responsible for data retrieval and processing, contributed to data analyses and reviewed the manuscript. AV contributed to study design, data collection and reviewed the manuscript. RS, BS and AKB contributed to data collection, reviewed and contributed to the process of finalising the manuscript. KAAØ contributed to statistical analyses, reviewed the manuscript and contributed to the process of finalizing the manuscript. AR conceptualised the study and ensured complete data collection and reviewed the manuscript. CK conceptualised and designed the study, contributed during data analyses, and drafted and revised the manuscript. ZH, HJS and CK had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. CK is guarantor.
Funding: This work was supported by the Western and Northern Norway Regional Health Trusts with the respective grant numbers F-11639 and HNF1512-20.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available on reasonable request. The raw data supporting the conclusion of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
This study involves human participants and was approved by REK Helse Sør Øst 2012/544-1.
References
- 1. Agyeman PKA, Schlapbach LJ, Giannoni E, et al. Epidemiology of blood culture-proven bacterial sepsis in children in switzerland: a population-based cohort study. Lancet Child Adolesc Health 2017;1:124–33. 10.1016/S2352-4642(17)30010-X [DOI] [PubMed] [Google Scholar]
- 2. Humberg A, Fortmann I, Siller B, et al. Preterm birth and sustained inflammation: consequences for the neonate. Semin Immunopathol 2020;42:451–68. 10.1007/s00281-020-00803-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Tröger B, Göpel W, Faust K, et al. Risk for late-onset blood-culture proven sepsis in very-low-birth weight infants born small for gestational age: a large multicenter study from the German neonatal network. Pediatr Infect Dis J 2014;33:238–43. 10.1097/INF.0000000000000031 [DOI] [PubMed] [Google Scholar]
- 4. Stoll BJ, Hansen N, Fanaroff AA, et al. Late-onset sepsis in very low birth weight neonates: the experience of the NICHD neonatal research network. Pediatrics 2002;110:285–91. 10.1542/peds.110.2.285 [DOI] [PubMed] [Google Scholar]
- 5. Tsai M-H, Hsu J-F, Chu S-M, et al. Incidence, clinical characteristics and risk factors for adverse outcome in neonates with late-onset sepsis. Pediatr Infect Dis J 2014;33:e7–13. 10.1097/INF.0b013e3182a72ee0 [DOI] [PubMed] [Google Scholar]
- 6. Levit O, Bhandari V, Li F-Y, et al. Clinical and laboratory factors that predict death in very low birth weight infants presenting with late-onset sepsis. Pediatr Infect Dis J 2014;33:143–6. 10.1097/INF.0000000000000024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Giannoni E, Agyeman PKA, Stocker M, et al. Neonatal sepsis of early onset, and hospital-acquired and community-acquired late onset: a prospective population-based cohort study. J Pediatr 2018;201:106–14. 10.1016/j.jpeds.2018.05.048 [DOI] [PubMed] [Google Scholar]
- 8. Greenberg RG, Kandefer S, Do BT, et al. Late-onset sepsis in extremely premature infants: 2000-2011. Pediatr Infect Dis J 2017;36:774–9. 10.1097/INF.0000000000001570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Cailes B, Kortsalioudaki C, Buttery J, et al. Epidemiology of UK neonatal infections: the neonin infection surveillance network. Arch Dis Child Fetal Neonatal Ed 2018;103:F547–53. 10.1136/archdischild-2017-313203 [DOI] [PubMed] [Google Scholar]
- 10. Singer M, Deutschman CS, Seymour CW, et al. The third International consensus definitions for sepsis and septic shock (sepsis-3). JAMA 2016;315:801–10. 10.1001/jama.2016.0287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Klingenberg C, Kornelisse RF, Buonocore G, et al. Culture-negative early-onset neonatal sepsis - at the crossroad between efficient sepsis care and antimicrobial stewardship. Front Pediatr 2018;6:285. 10.3389/fped.2018.00285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Mukhopadhyay S, Puopolo KM, Hansen NI, et al. Neurodevelopmental outcomes following neonatal late-onset sepsis and blood culture-negative conditions. Arch Dis Child Fetal Neonatal Ed 2021;106:467–73. 10.1136/archdischild-2020-320664 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Norwegian neonatal network N. Available: https://www.kvalitetsregistre.no/register/skade-og-intensiv-barn/norsk-nyfodtmedisinsk-kvalitetsregister [Accessed 31 Dec 2022].
- 14. Stensvold HJ, Klingenberg C, Stoen R, et al. Neonatal morbidity and 1-year survival of extremely preterm infants. Pediatrics 2017;139:e20161821. 10.1542/peds.2016-1821 [DOI] [PubMed] [Google Scholar]
- 15. Parry G, Tucker J, Tarnow-Mordi W, et al. Crib II: an update of the clinical risk index for babies score. Lancet 2003;361:1789–91. 10.1016/S0140-6736(03)13397-1 [DOI] [PubMed] [Google Scholar]
- 16. Sinha AK, Murthy V, Nath P, et al. Prevention of late onset sepsis and central-line associated blood stream infection in preterm infants. Pediatr Infect Dis J 2016;35:401–6. 10.1097/INF.0000000000001019 [DOI] [PubMed] [Google Scholar]
- 17. Fjalstad JW, Stensvold HJ, Bergseng H, et al. Early-onset sepsis and antibiotic exposure in term infants: a nationwide population-based study in norway. Pediatr Infect Dis J 2016;35:1–6. 10.1097/INF.0000000000000906 [DOI] [PubMed] [Google Scholar]
- 18. de Vries LS, Eken P, Dubowitz LM. The spectrum of leukomalacia using cranial ultrasound. Behav Brain Res 1992;49:1–6. 10.1016/s0166-4328(05)80189-5 [DOI] [PubMed] [Google Scholar]
- 19. Skjaerven R, Gjessing HK, Bakketeig LS. Birthweight by gestational age in Norway. Acta Obstet Gynecol Scand 2000;79:440–9. 10.1034/j.1600-0412.2000.079006440.x [DOI] [PubMed] [Google Scholar]
- 20. Baker EK, Davis PG. Bronchopulmonary dysplasia outcome estimator in current neonatal practice. Acta Paediatr 2021;110:166–7. 10.1111/apa.15427 [DOI] [PubMed] [Google Scholar]
- 21. Bonafiglia E, Gusson E, Longo R, et al. Early and late onset sepsis and retinopathy of prematurity in a cohort of preterm infants. Sci Rep 2022;12:11675. 10.1038/s41598-022-15804-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Cantey JB, Huffman LW, Subramanian A, et al. Antibiotic exposure and risk for death or bronchopulmonary dysplasia in very low birth weight infants. J Pediatr 2017;181:289–93. 10.1016/j.jpeds.2016.11.002 [DOI] [PubMed] [Google Scholar]
- 23. Hatzidaki E, Giahnakis E, Maraka S, et al. Risk factors for periventricular leukomalacia. Acta Obstet Gynecol Scand 2009;88:110–5. 10.1080/00016340802503039 [DOI] [PubMed] [Google Scholar]
- 24. Resch B, Neubauer K, Hofer N, et al. Episodes of hypocarbia and early-onset sepsis are risk factors for cystic periventricular leukomalacia in the preterm infant. Early Hum Dev 2012;88:27–31. 10.1016/j.earlhumdev.2011.06.011 [DOI] [PubMed] [Google Scholar]
- 25. Gowda H, Norton R, White A, et al. Late-onset neonatal sepsis-A 10-year review from north queensland, australia. Pediatr Infect Dis J 2017;36:883–8. 10.1097/INF.0000000000001568 [DOI] [PubMed] [Google Scholar]
- 26. Bizzarro MJ, Shabanova V, Baltimore RS, et al. Neonatal sepsis 2004-2013: the rise and fall of coagulase-negative staphylococci. J Pediatr 2015;166:1193–9. 10.1016/j.jpeds.2015.02.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Härtel C, Faust K, Avenarius S, et al. Epidemic microclusters of blood-culture proven sepsis in very-low-birth weight infants: experience of the German neonatal network. PLoS One 2012;7:e38304. 10.1371/journal.pone.0038304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Berardi A, Sforza F, Baroni L, et al. Epidemiology and complications of late-onset sepsis: an italian area-based study. PLoS One 2019;14:e0225407. 10.1371/journal.pone.0225407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Hornik CP, Fort P, Clark RH, et al. Early and late onset sepsis in very-low-birth-weight infants from a large group of neonatal intensive care units. Early Hum Dev 2012;88 Suppl 2:S69–74. 10.1016/S0378-3782(12)70019-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Isaacs D, Australasian Study Group For Neonatal Infections . A ten year, multicentre study of coagulase negative staphylococcal infections in australasian neonatal units. Arch Dis Child Fetal Neonatal Ed 2003;88:F89–93. 10.1136/fn.88.2.f89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. El Manouni El Hassani S, Berkhout DJC, Niemarkt HJ, et al. Risk factors for late-onset sepsis in preterm infants: a multicenter case-control study. Neonatology 2019;116:42–51. 10.1159/000497781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Vermont NO. Manual of operations: part2 data definitions and infant data forms 2019; 2022.
- 33. Alcock G, Liley HG, Cooke L, et al. Prevention of neonatal late-onset sepsis: a randomised controlled trial. BMC Pediatr 2017;17:98. 10.1186/s12887-017-0855-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Köstlin-Gille N, Härtel C, Haug C, et al. Epidemiology of early and late onset neonatal sepsis in very low birthweight infants: data from the German neonatal network. Pediatr Infect Dis J 2021;40:255–9. 10.1097/INF.0000000000002976 [DOI] [PubMed] [Google Scholar]
- 35. Shah PS, Dunn M, Aziz K, et al. Sustained quality improvement in outcomes of preterm neonates with a gestational age less than 29 weeks: results from the evidence-based practice for improving quality phase 3 1 Can J Physiol Pharmacol 2019;97:213–21. 10.1139/cjpp-2018-0439 [DOI] [PubMed] [Google Scholar]
- 36. Shah J, Jefferies AL, Yoon EW, et al. Risk factors and outcomes of late-onset bacterial sepsis in preterm neonates born at < 32 weeks’ gestation. Am J Perinatol 2015;32:675–82. 10.1055/s-0034-1393936 [DOI] [PubMed] [Google Scholar]
- 37. Dorling J, Abbott J, Berrington J, et al. Controlled trial of two incremental milk-feeding rates in preterm infants. N Engl J Med 2019;381:1434–43. 10.1056/NEJMoa1816654 [DOI] [PubMed] [Google Scholar]
- 38. Rønnestad A, Abrahamsen TG, Medbø S, et al. Late-onset septicemia in a norwegian national cohort of extremely premature infants receiving very early full human milk feeding. Pediatrics 2005;115:e269–76. 10.1542/peds.2004-1833 [DOI] [PubMed] [Google Scholar]
- 39. Cleminson J, Austin N, McGuire W. Prophylactic systemic antifungal agents to prevent mortality and morbidity in very low birth weight infants. Cochrane Database Syst Rev 2015;2015:CD003850. 10.1002/14651858.CD003850.pub5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Patel RM, Kandefer S, Walsh MC, et al. Causes and timing of death in extremely premature infants from 2000 through 2011. N Engl J Med 2015;372:331–40. 10.1056/NEJMoa1403489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Thaulow CM, Lindemann PC, Klingenberg C, et al. Epidemiology and antimicrobial susceptibility of invasive bacterial infections in children-A population-based study from norway. Pediatr Infect Dis J 2021;40:403–10. 10.1097/INF.0000000000003013 [DOI] [PubMed] [Google Scholar]
- 42. Jiang S, Yang Z, Shan R, et al. Neonatal outcomes following culture-negative late-onset sepsis among preterm infants. Pediatr Infect Dis J 2020;39:232–8. 10.1097/INF.0000000000002558 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
fetalneonatal-2022-324977supp001.pdf (33.2KB, pdf)
Data Availability Statement
Data are available on reasonable request. The raw data supporting the conclusion of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.