Abstract
Climatic changes and elevated ambient temperature are significant environmental stressors with a negative impact on birds’ physiological, immunological, and behavioral status, increasing their susceptibility to stressors and immunosuppression and consequently increasing intestinal permeability (leaky gut). Prebiotics have been utilized to stop or diminish the harmful effects of stress in chickens. We aimed to evaluate the role of mannan-oligosaccharides, and beta-d-glucan prebiotics supplements in drinking water against experimentally induced heat stress (HS) on broiler chickens and study their impact on birds’ performance, gut microbiome, and immune response. A total of 120 1-day-old Ross broiler chicks were allocated into four groups (30 birds/group), and each group was subdivided into triplicates (10 birds each). The experimental groups were classified as follows; the 1st (G1) control birds, the 2nd (G2) birds exposed experimentally to HS, the 3rd (G3) birds administered prebiotics in drinking water without exposure to HS, and the 4th (G4) birds exposed to HS and administered prebiotics in drinking water. After each vaccination, blood samples and serum samples were collected to evaluate the birds’ immune status. Fecal samples were also collected for the molecular evaluation of the gut microbiome based on the genetic analyses and sequencing of 16S rRNA gene. The results showed that HS has reduced the birds’ performance and badly affected the birds’ immune response and gut microbiome. However, the addition of prebiotics to drinking water, with or without stress, enhanced the growth rate, maintained a normal gut microbiome, and improved immune parameters. Moreover, the usage of prebiotics improved the chicken gut microbiome and alleviated the negative effect of heat stress. Administering prebiotics significantly (p < 0.05) increased the relative abundance of beneficial bacteria and eradicated pathogenic ones in the birds’ gut microbiome. Prebiotics showed a positive effect on the gut microbiome and the immune status of chickens under HS in addition to their efficacy as a growth promoter.
Subject terms: Microbial communities, Biodiversity, Climate-change ecology
Introduction
The poultry industry in tropical countries is subjected to huge challenges arising from the environmental temperatures that cause heat stress1. Heat stress (HS) has become a dangerous issue; it causes huge losses in broiler production and reduces the performance and productivity of broilers2. Birds that are exposed to HS show behavioral, structural, and immunological changes that negatively affect the poultry business3. The profitability of the broiler production business could be improved by properly managing HS in flocks during the hot season4. The increased quantity of eggs and improved quality of chicken meat during the summer months may aid in covering the general public’s protein needs and resolve food deficiency problems in developing nations5. In this regard, poultry researchers are working regularly and aggressively to create strategies for producers to combat HS problems in poultry farms. The most conventionally successful method to counteract HS effects on birds is using air cooling/ventilation systems, which have many drawbacks including noise from the operating machines, unequally distributed chickens, and being very expensive to cool the entire housing6. Therefore, most approaches applied to mitigate HS have been focused on nutritional approaches7. Antibiotics are one of the feed supplements used in the poultry business for treatment, prevention of diseases, and growth development. However, the presence of antibiotic residues in poultry meat harms the health of consumers by causing resistance to the normal microbiota and the dissemination of pathogenic microorganisms as well as, it resulted in the appearance of microbial resistance against antibiotics8, 9.
Prebiotics are considered the best feed additives as antibiotic substitutes9. Gibson and Roberfroidd10 were the first to define a prebiotic as “a non-digestible non-fermentable food element that benefits the host by selectively encouraging the growing and/or activity of one or a restricted number of bacteria in the colon, hence improving host health”10. This definition was later revised by Gibson et al. who re-defined a prebiotic as “a selectively fermented food that causes specific changes in the development and/or viability of the intestinal microbiota which improves host health and welfare”11. Oligosaccharides have been the most utilized prebiotics in the poultry industry, including mannan oligosaccharides (MOS), inulin, fructo-oligosaccharides (FOS), galacto-oligosaccharides (GOS), xylo-oligosaccharides (XOS), soya-oligosaccharides (SOS), isomalt-oligosaccharides (IMO), pyrodextrins, lactulose, and oligochitosan12. The intestinal microbiota can ferment prebiotics to generate short-chain fatty acids (SCFA), lactic acid, or certain antimicrobial substances like bacteriocin against harmful microbial species13. The inhibition of harmful microbes in the gut may be attributed to the increased intestinal acidity produced by the fermented products of prebiotics12. In turn, prebiotics may help heat-stressed chickens by improving their immune system, gut morphology, oxidative status, and physiological stress responses, which will ultimately improve the growth performance of broiler chickens2. Therefore, we aimed to evaluate the mitigating and prophylactic influences of prebiotics on the birds’ performance, gut microbiome, and immune status of heat-stressed chickens.
Materials and methods
Birds housing and management
This study was ethically approved by Cairo University Institutional Animal Care and Use Committee (CU-IACUC) Veterinary Medical and Agricultural Sciences Sector with code “CU ll F 27 21”. All the animals procedures in this study were conducted according to the guidelines of the Faculty of Veterinary Medicine Cairo University following the “Guide for the Care and Use of Laboratory Animals” published by the Institute of Laboratory Animal Research (Washington, DC, USA). All the animals procedures conducted in this study are following the ARRIVE guidelines 2.0.
A total of 120, 1-day-old commercial male Ross broiler chicks were obtained from a local commercial hatchery. The chickens were raised in the experimental station of the Faculty of Veterinary Medicine, Cairo University during February and March 2022. The pens were disinfected by TH4 (a combination of glutaraldehyde and quaternary ammonium compounds, Sogeval, France) before housing. The poultry house was cleaned, and all manure materials were removed then, water and high-pressure sprayers were used to remove dust materials from windows, floors, ceilings, walls, and ventilation shafts, which were subsequently disinfected with TH4 that had been diluted to 1:200. All removable fittings and equipment (feeders and drinkers) were removed from the building and submerged in 1:200 TH4 dilution baths. Quicklime was sprayed on the ground to destroy parasites and their eggs, such as coccidia. Sixty-watt bulb was hung 2.2 m in the middle of each pen. Light intensity at the level of the birds was approximately 2.66 lx/m2/s14. Dark/light cycle of the broilers during the experiment are demonstrated in Table 1. Chicks were allocated into four groups, each group included 30 chicks. Each group was subdivided into 3 replicates (10 birds each). Chickens’ subgroups were raised in open-side poultry house, and kept on a deep litter system, in separate pens with coarse sawdust bedding material with 10 cm thickness on a concrete floor. The litter was maintained and the wet parts around the drinkers were replaced with dry ones. A good ventilation system was applied to provide continuous airflow and prevent the accumulation of harmful gases such as carbon dioxide and ammonia via air vents, windows, as well as mechanical ventilation by fans and air suction. The birds were administered a balanced commercial diet without any feed additives and administered drinking water (clean and clear tap water, pH 7, room temperature) ad libitum.
Table 1.
Dark/light cycle of the broilers during the experiment.
| Time | Duration of dark (h) | Duration of light (h) |
|---|---|---|
| First 3 days | 0 | 24 |
| 1st week | 1 | 23 |
| 2nd week | 2 | 22 |
| 3rd week | 3 | 21 |
| 4th and 5th week | 4 | 20 |
Temperature and humidity of the poultry house
At the beginning of chicks’ rearing, the temperature was set at 35 °C and steadily decreased at a pace of 2–5 °C/week until it reached 22 °C at the end of the 4th week and then the temperature was kept at 22 °C for the 5th week15. The maximum and minimum thermometer (Taylor Precision 5154 Wall Thermometer Cat# 5154) readings were taken every day to determine the ambient temperature. At weeks 4 and 5, the temperature was kept at 32–35 °C for the two heat-stressed groups using an electrical heater. A wall-mounted thermo-hygrometer (Oumar Hygrothermograph, Cat# TH108) was used to detect indoor relative humidity where the average relative humidity ranged from 60 to 70%. According to the formula described by Farghly et al.1, the average temperature humidity index (THI), was 31.427.
Feeding, and vaccination program for the experimental chickens
All the birds were fed a commercial balanced ration without any feed additives (Cairo Poultry company). Chicks were fed a starter diet for the first 13 days, a grower from 14 to 26 days old, and a finisher diet from 27 to 35 days of age. The percentage of proteins, fats and fibers in the starter, grower and finisher rations are demonstrated in Table 2. The composition of the starter ration was as follow: yellow maize, soybean gain (46%), high fat extruded soybean, yellow corn gluten (60%), DDGS, monocalcium phosphate, limestone powder, common salt, dl-Methionine, and l-lysine hydrochloride, a mixture of vitamin and mineral salt (1/8219) choline chloride. The grower ration composition was as follow: yellow maize, soybean gain (46%), high fat extruded soybean, yellow corn gluten (60%), vegetable oils (cotton seed, sunflower, soy oil), monocalcium phosphate, limestone powder, common salt, dl-Methionine, and l-lysine hydrochloride, a mixture of vitamin and mineral salt (1/8219) choline chloride. The finisher ration composition was as follow: yellow maize, soybean gain (46%), high fat extruded soybean, yellow corn gluten (60%), gluten feed (22%), monocalcium phosphate, limestone powder, common salt, dl-Methionine, and l-lysine hydrochloride, a mixture of vitamin and mineral salt (1/8219) choline chloride. Each ton of this ration contains the nutritional needs of vitamins and minerals in internationally recommended proportions. Vitamins included A, D3, E, B1, B2, B6, B12, niacin, biotin, folic Acid, choline chloride, and pantothenic acid. Mineral elements included manganese, iodine, iron, copper, cobalt, selenium, and zinc. During the first 12 h post-arrival, chicks were supplied with rehydration salts-treated water (glucose anhydrous 2.70 g, trisodium citrate anhydrous 0.58 g, sodium chloride 0.52 g, and potassium chloride 0.30 g for each sachet (4.1 g) of the product. During the experiment, a vaccination program was adopted as follows (Table 3); on day one, all birds were vaccinated against Newcastle disease (ND) and Infectious Bronchitis (IB) using Nobilis Ma5 + Clone 30 live vaccine, via spraying method. On day 7, birds were vaccinated via subcutaneous injection with 0.5 mL at the base of the neck against avian influenza H5N1 using Egymune H5 inactivated reassortant vaccine. The birds were also vaccinated against Gumboro (Infectious Bursal Disease) using Nobilis GUMBORO D78 live vaccine through drinking water on day 12. Finally, the birds were vaccinated again against ND by Nobilis, LaSota strain live vaccine in drinking water on day 17.
Table 2.
The composition of ration used during the experiment.
| Starter | Grower | Finisher | |
|---|---|---|---|
| Crude protein (%) | 23 | 21 | 18 |
| Crude fat (%) | 5.85 | 7.14 | 6.70 |
| Metabolizable energy (kcal/kg) | 3050 | 3150 | 3200 |
| Crude fiber (%) | 3.50 | 3.49 | 3.65 |
Table 3.
Details of the vaccine program adopted during the experiment.
| Vaccine | Disease | Type | Concentration | Method | Time |
|---|---|---|---|---|---|
| Nobilis Ma5 + Clone 30 | Infectious bronchitis (IB) and Newcastle disease (ND) | Live vaccine |
Live I.B. strain Ma5: ≥ 3 log10 EID50 Live ND strain Clone 30: ≥ 6 log10 EID50 |
Spray | Day 1 |
| Egymune H5 | Avian influenza H5N1 | Inactivated reassortant vaccine | 8 log10 EID50/mL | Subcutaneous injection | Day 7 |
| Nobilis GUMBORO D78 | Infectious bursal disease | Live vaccine | ≥ 4 log10 TCID50 | Drinking water | Day 12 |
| Nobilis LaSota strain | Newcastle disease (ND) | Live vaccine | ≥ 6 log10 EID50 | Drinking water | Day 17 |
EID50 Median embryo infectious dose, TCID50 median tissue culture infectious dose.
Prebiotics constitutions and dose
Commercial prebiotic Hydrostar from EGYEURO Company (Cat# 32295000) was applied in drinking water at a dose of 0.5 mL/L (following the manufacturer's instructions) throughout the rearing period from day one till the end of the experiment. The constitutions of the prebiotic Hydrostar are shown in Table 4.
Table 4.
Hydrostar prebiotics (liquid yeast hydrolysate) composition.
| Crude protein, minimum | 6% |
| Crude fat, minimum | 1% |
| Crude fiber, minimum | 2.5% |
| Cerevisiae yeast | 222.222 mg/L |
| Mannan | 8880 mg/L |
| Glucan | 13.330 mg/L |
| Xanthan gum | 11.111 mg/L |
| Acetic acid | 11.111 mg/L |
| Coloring (E-133) | 11 mg/L |
Experimental design
As demonstrated in Table 5, this study was conducted using four groups with two main factors: heat stress (32–35 °C) and the administration of prebiotics. The 1st group (G1) was the control negative group, the 2nd group (G2) birds were exposed to experimentally HS, the 3rd group (G3) was administered prebiotics in drinking water without exposure to HS, and the 4th group (G4) was exposed to HS and administered prebiotics in drinking water. The addition of the prebiotics started on day 1 and until the end of the experiment while HS exposure began at week 4 till the end of the experiment.
Table 5.
Experimental design; prebiotics treatment with or without heat stress of the chicken study groups.
| Factor | Groups | |||
|---|---|---|---|---|
| Control group (G1) | Heat stress group (G2) | Prebiotic group (G3) | Heat stress + prebiotic (G4) | |
| Prebiotics | Absent | Absent | Present | Present |
| Temperature degree | Ambient temperature (22 °C) | High temperature (32–35 °C) | Ambient temperature (22 °C) | High temperature (32–35 °C) |
Body weight (BW), weight gain (WG), and cumulative mortality
Chicks were individually weighed at 1-day old (starting BW) and weekly throughout the experimental period (1–35 d). The live body weight gain (WG) was calculated weekly by subtracting the initial weight from the current weight and expressed as (g/bird/week). The cumulative mortality rate was calculated as the total number of deaths in chickens per group divided by the total population in the same group.
Feed intake (FI) and Feed conversion ratio (FCR)
Each group of chicks received a set amount of feed weekly, and the remaining feed was weighed at the end of each week. The difference between the feed delivered and the rest of the feed was estimated. The weekly consumed feed was divided by the number of birds in each group this week to get the average quantity of feed consumption, so it was expressed as (g/bird/week). Regarding the feed conversion ratio (FCR), it was calculated weekly by dividing the average feed consumption per bird by the average body weight gain per bird.
Sampling
Blood samples
Nine birds were randomly selected weekly from each group and 4.5 mL (0.5 mL per bird) of blood samples were ethically collected from the wing vein into 2 tubes (with and without anticoagulant). The blood samples without anticoagulant were cooled to approximately 4 °C overnight then centrifuged at 3000 rpm for 10 min and sera were transferred to clean tubes using a micropipette (Eppendorf Research Plus micropipette, adjustable volume 20–200 µL). The collected sera were kept at − 20 °C, until the analysis of the hemagglutination inhibition (HI)14. Meanwhile, blood samples taken into tubes with EDTA as an anticoagulant were used for immune cells counting (total and differential leukocytes).
Fecal samples for microbiome analysis
Total genomic DNA was extracted from the fecal sample (200 mg, three samples from each group, a total of 600 mg per group) at three-time intervals (1st, 3rd, and 5th week) using the commercially available QIAamp Fast DNA Stool Mini kit (QIAGEN, Germany), following the manufacturer’s instructions. DNA concentration and quality were assessed using a nanophotometer (NanoPhotometer P360, Implen) and agarose gel electrophoresis. DNA was stored at − 20 °C until further processing. Table 6 illustrates pooled samples from the same group and sample characterization (sampling time, with/without exposure to HS and prebiotics treatment).
Table 6.
Fecal DNA samples for gut microbiome analysis and characterization at exposure to prebiotics with or without heat stress.
| Sample code | Number of pooled samples | Time of sample | Heat stress | Prebiotics |
|---|---|---|---|---|
| S1 | 4 Samples represent all groups before the treatment | Day 1 (Week 1) | No | No |
| S2 | 3 Samples of the control group | Week 3 | No | No |
| S3 | 3 Samples of the prebiotics group | Week 3 | No | Yes |
| S4 | 3 Samples of the control group | Week 5 | No | No |
| S5 | 3 Samples of the prebiotics group | Week 5 | No | Yes |
| S6 | 3 Samples of the heat stress group | Week 5 | Yes | No |
| S7 | 3 Samples of the heat stress + prebiotics group | Week 5 | Yes | Yes |
DNA sequencing and gut microbiome data analysis
Sequencing using Illumina-Miseq™ (2 × 300 bp paired-end protocol) was performed targeting the bacterial V3 and V4 regions of the 16S rRNA gene16. Generated Illumina reads were analyzed using QIIME tool version 1.9.117 as described before18, 19.
Total and differential leukocytic counts
The whole blood samples were used for total and differential leukocytic counts. The total count was carried out microscopically using a hemocytometer (Standard Hemocytometer Weberscientific Cat# 3048-12) after mixing 1 mL (Eppendorf Research Plus micropipette, adjustable volume 100–1000 µL) of Natt and Herrick solution with 10 µL (Eppendorf Research Plus micropipette, adjustable volume 0.5–10 µL) blood and was examined under microscope (Binocular optical microscope Celestron labs) using a hemocytometer. The differential count was done using the commercial Diff Quick stain20.
Antibody (HI) titers to ND and AI vaccines
The hemagglutination inhibition (HI) test was applied to measure the serum level of HI antibodies to the given Newcastle disease (ND) and avian influenza (AI) vaccines. Briefly, two-fold serial dilutions of the sera were mixed with an equal volume of adjusted viral HA antigens. Chicken red blood cells (1% RBCs, v/v) were added, and the end point was indicated by the highest dilution that inhibited the agglutination of the chicken RBCs by observing the button formation rather than agglutination21.
Statistical analysis
Values were expressed as mean ± stranded error (M ± S.E). Statistical comparisons among the means of different experimental groups were constructed with t-test in weeks 1, 2 and 3, and one-way ANOVA in weeks 4 & 5 in all tests. All tests were made by GraphPad Prism version 9.0.2 (GraphPad Software, San Diego, California USA, www.graphpad.com). A probability “P” value of ≤ 0.05 was considered statistically significant.
For the microbiome samples we used PERMANOVA (permutational multivariate analysis of variance) and ANOSIM (analysis of similarities) using Python script embedded in QIIME17, 19 (1.9.1); these tests were used for the analysis of the strength and the statistical significance of sample groupings. We did the differential abundance analysis aiming to find the differences in the relative abundance of each taxa between samples and then we used statistical analysis to assign a significant value to each comparison. We used Kruskal–Wallis and Bonferroni’s multiple comparisons tests using Python scripts embedded in QIIME17, 19 (1.9.1), for each taxa the P value and the false positive adjustment of the P value (FDR P value) corrected by the Benjamini–Hochberg FDR procedure for multiple comparisons were calculated.
Results
Body weight (BW), weight gain (WG), and cumulative mortality
As shown in Table 7, HS reduced the BW of broilers in the 4th and 5th weeks in G2. In the 4th week, birds in G1 showed a higher BW (1831 ± 15.46 g) compared to birds exposed to HS in G2 (1763 ± 18.25 g). The administration of prebiotics to HS-exposed broilers (G3) counteracted the average BW loss in the same week when compared to groups without heat stress. However, in the 5th week, the average BWs of G1 and G2 were 2450 g and 2250 g, respectively while the average BWs of birds in G3 and G4 were 2340 g and 2287 g, respectively.
Table 7.
Impacts of prebiotics with or without heat stress on the body weight, weight gain, feed intake, and feed conversion ratio (FCR) of broilers.
| Time | Groups | Body weight (g) | Weight gain (g) | Feed intake (g) | FCR |
|---|---|---|---|---|---|
| Week 1 | Control | 185 ± 2.88a | 146 ± 2.88a | 163.3 ± 12.81 | 1.117 ± 0.06 |
| Prebiotics | 187 ± 2.79b | 148 ± 2.79b | 159.2 ± 9.33 | 1.074 ± 0.04 | |
| Week 2 | Control | 523 ± 9.66a | 338 ± 6.77a | 420.5 ± 18.58 | 1.243 ± 0.03a |
| Prebiotics | 534 ± 8.59b | 347 ± 5.79b | 426.4 ± 16.81 | 1.228 ± 0.02b | |
| Week 3 | Control | 1089 ± 12.88a | 566 ± 3.21a | 734.3 ± 34.42a | 1.297 ± 0.05a |
| Prebiotics | 1064 ± 12.03b | 530 ± 3.43b | 682.4 ± 32.3b | 1.287 ± 0.05b | |
| Week 4 | Control | 1831 ± 15.46 | 742 ± 2.57a | 1047 ± 51.39 | 1.41 ± 0.06 |
| Prebiotics | 1785 ± 15.03 | 721 ± 3.00b | 1049 ± 58.6 | 1.455 ± 0.07 | |
| Heat stress | 1763 ± 18.25 | 674 ± 5.36c | 949 ± 65.53 | 1.407 ± 0.08 | |
| Heat stress prebiotics | 1759 ± 10.74 | 695 ± 1.28d | 1008 ± 51.67 | 1.45 ± 0.07 | |
| Week 5 | Control | 2450 ± 19.05a | 619 ± 4.04a | 1278 ± 61.2 | 2.063 ± 0.08 |
| Prebiotics | 2340 ± 12.7b | 555 ± 2.3b | 1126 ± 31.47 | 2.029 ± 0.06 | |
| Heat stress | 2250 ± 23.62c | 487 ± 5.36c | 1121 ± 64.63 | 2.300 ± 0.1 | |
| Heat stress prebiotics | 2287 ± 17.18b,c | 527.8 ± 6.64d | 1064 ± 52.65 | 2.013 ± 0.07 | |
| Final (cumulative day 1–35) | Control | – | 2411.00 ± 19.06a | 3642 ± 178.1 | 1.51 ± 0.06 |
| Prebiotics | – | 2301.00 ± 21.36b | 3443 ± 148.4 | 1.496 ± 0.05 | |
| Heat stress | – | 2211.00 ± 23.67c | 3389 ± 196 | 1.531 ± 0.07 | |
| Heat stress prebiotics | – | 2260.00 ± 21.07b,c | 3340 ± 163.1 | 1.485 ± 0.06 |
Each value represents mean ± S.E. Significant difference between groups in body weight, weight gain, feed intake, and FCR was analyzed by t-test in weeks 1, 2 and 3, and one-way ANOVA in weeks 4 and 5 at p ≤ 0.05.
Parameters in the same column followed by different superscript letters are significantly different (p ≤ 0.05).
During the 4th week, the WG in the birds in G1 and G2 were 742 g and 674 g, respectively while in G3 and G4 were 721 g and 695 g, respectively. During the 5th week, the WG in the birds in G1 and G2 were 619 g and 487 g, respectively while, in G3 and G4 they were 555 g and 527 g, respectively. Final WG in the birds in G1 and G2 were 2411 g and 2211 g, respectively while, in G3 and 4, they were 2301 g and 2260 g, respectively.
There was a significant increase in BW of G3 than G1 in the first 2 weeks (p = 0.0023), (p = 0.0094) respectively, and a significant decrease in BW in the same group (p = 0.0012) in week 3. Meanwhile, in the 4th week, there was a significant increase between G1 and G4 (p = 0.0396). In the 5th week, there was a significant increase between G1 and G4, G1 and G3, G1 and G2, and G3 and G2 (p = 0.0012), (p = 0.0129), (p = 0.0003), (p = 0.0364) respectively.
Concerning the WG, there was a significant increase between G3 and G1 in the first 2 weeks (p = 0.0023) and (p = 0.0116) respectively, in contrast, a significant decrease in WG in the same groups (p ≤ 0.0001) in week 3. While in week 4, there was a significant increase among all groups G1 & G4, G1 & G2, G3 & G2 (p ≤ 0.0001), and a significant increase in G1 & G3, G3 & G4 (p = 0.0102) and (p = 0.0028) respectively. Finally, there was a significant decrease in G2 & G4 (p = 0.0102).
In week 5, there were a significant increase between G1 & G4, G1 & G3, G1 & G2, G3 & G2 (p < 0.0001), while a significant increase between G3 & G4 (p = 0.0176), and a significant decrease between G2 & G4 (p = 0.0016).
Finally, WG (1–35) shows a significant increase between G1 & G3, G1 & G2, and G1 & G4 (p = 0.0271), (p = 0.0008) and (p = 0.0046), respectively. The cumulative mortalities have been recorded to be at the highest level of 10% (3/30 birds) in G2 (HS group) followed by G1, and G4 (control group and heat stress prebiotics group) with a rate of 3% (1/30 birds) for each group while no mortalities were recorded in G3 (prebiotics group) all over the experimental period.
Feed intake (FI) and Feed conversion ratio (FCR)
Birds in G2 and G4 showed a mathematically reduced FI than those in G1 and G3. The FI masses of chickens in the 4th week in G1 and G3 were 1047 g and 1049 g, respectively, while in G2 and G4 the FI values were 949 g and 1008 g, respectively. In the 5th week, the FI of chickens in G1 and G3 were 1278 g and 1126 g, respectively, while in G2 and G4 they were 1121 g and 1064 g, respectively. Unexpectedly, the administration of prebiotics to chickens of the control group led to a drastic decline in the FI while the decline was not that much in heat-stressed chickens. The addition of prebiotics to heat-treated birds did not cause a severe decline in FI when compared to the control group of the same week which caused a great decrease in FI.
In week 4, G1 & G2 were 1047 and 949, while G3 & G4 were1049 and 1008, respectively. At week 5, G1 & G2 were 1278 and 1121, while G3 & G4 were 1126 and 1064, respectively.
The overall FI masses in G1 & G2 were 3642 and 3389, while in G3& G4 they were 3443 g and 3340, respectively, and there was a significant increase in FI only in week 3 (p = 0.0017) when G1 compared with G3. In the 4th week, the FCR in the chickens of G2 and G4 were 1.40 and 1.45, respectively while G1 and G3 showed FCR values of 1.41 and 1.45, respectively. In the 5th week, the FCRs in G2, G4, G1, and G3 were 2.30, 2.01, 2.06, and 2.02, respectively. The overall FCRs were 1.53, 1.48, 1.51, and 1.49, in the same group order respectively. The overall FCRs in prebiotics-treated groups were better than those in other groups. There was a significant increase between G1, and G3, in week 2 & 3 (p = 0.0229) and (p = 0.0131), respectively. Table 4 depicts the BW, WG, and FCR of broilers as related to prebiotic administration and heat stress.
Total and differential leukocytic counts
Data concerning the total white blood cells count (TWBC) X 103/mm3 were presented by means ± standard errors (Mean ± S.E.) as summarized in Table 8. A significant difference was noticed between the TWBC of chickens of different groups in week 4. The corresponding p values with significant increase were (p ≤ 0.0001) for G1 vs G3, G1 vs G2, G1 vs G4, G3 vs G2, significant increase (p = 0.0011) for G3 vs G4, and significant decrease (p = 0.0212) for G2 vs G4. At week 5, there was a significant increase between G1 when compared with G3, and G1 with G4 (p ≤ 0.0001), G1 with G2 (p = 0.0024), G3 with G4 (p = 0.0191), and G2 with G4 (p = 0.0007).
Table 8.
Effects of prebiotics with or without heat stress, and their interaction on total and differential leukocyte count.
| Time | Groups | WBC × 103 cells/mm3 | Lymphocyte% | Monocyte% | Heterophil% | Eosinophil% |
|---|---|---|---|---|---|---|
| Week 2 | Control | 72.78 ± 4.573 | 48.444 ± 0.930a | 12.111 ± 1.467a | 38.444 ± 1.684 | 1.000 ± 0.577 |
| Prebiotics | 81.11 ± 4.47 | 37.333 ± 1.658b | 27.333 ± 2.048b | 35.333 ± 0.408 | 0.000 ± 0.000 | |
| Week 3 | Control | 67.22 ± 3.84 | 58.000 ± 1.247 | 17.778 ± 0.909a | 22.667 ± 1.054 | 1.556 ± 0.294 |
| Prebiotics | 65.78 ± 1.47 | 55.778 ± 1.024 | 23.556 ± 1.144b | 20.667 ± 1.915 | 0.000 ± 000 | |
| Week 4 | Control | 83.33 ± 1.78a | 58.667 ± 0.667a | 21.111 ± 0.351a | 19.111 ± 0.588a | 1.111 ± 0.351 |
| Prebiotics | 70.33 ± 0.781b | 61.111 ± 0.351b | 21.333 ± 1.106a | 17.556 ± 1.281a | 0.000 ± 0.000 | |
| Heat stress | 56.57 ± 0.781c | 62.444 ± 1.281c | 29.111 ± 0.676b | 8.444 ± 0.930b | 0.000 ± 0.000 | |
| Heat stress prebiotics | 62.44 ± 1.634d | 62.667 ± 0.745b,c | 26.889 ± 0.351b,c | 10.000 ± 0.471b | 0.444 ± 0.294 | |
| Week 5 | Control | 91.56 ± 3.245a | 44.667 ± 1.054 | 16.667 ± 1.106a | 35.333 ± 1.054a | 3.333 ± 0.333 |
| Prebiotics | 74.22 ± 3.022b | 45.333 ± 0.667 | 23.444 ± 1.119b | 29.222 ± 1.451b | 2 ± 0.333 | |
| Heat stress | 78.44 ± 0.987c | 42 ± 0.969a | 40.889 ± 1.338c | 16.444 ± 1.281c | 0.444 ± 0.294 | |
| Heat stress prebiotics | 63.78 ± 1.341d | 46.444 ± 0.801b | 34.222 ± 1.47d | 18.444 ± 1.281c | 0.889 ± 0.351 |
Each value represents mean ± S.E. Significant difference between groups in total WBCS count and differential leukocytic counts was analyzed by t-test in weeks 2 and 3, and one-way ANOVA in weeks 4 and 5 at p ≤ 0.05.
Parameters in the same column followed by different superscript letters are significantly different (p ≤ 0.05).
Data concerning differential leukocytic counts are summarized also in Table 8 which shows that lymphocyte % decreased significantly, and monocyte % increased significantly (p ≤ 0.0001) in week 2 when the prebiotics-treated birds are compared to control groups. In week 3, the monocyte% showed a significant increase (p = 0.0004). In week 4 lymphocyte% shows a significant decrease (p = 0.0002), (p = 0.0407) and (p ≤ 0. 0001) in G1 vs G2, G1 vs G3, and G1 vs G4 respectively, while in monocyte% show a significant decrease in G1 when compared with G2, G1 with G4, G3 with G2, and G3 with G4 (p ≤ 0.0001), also heterophil showed a significant increase between G1 & G2, G1 & G4, G3 & G2 and G3& G4 (p ≤ 0.0001). In week 5, lymphocyte % show a significant decrease (p = 0.0048) in G2 with G4, while monocyte % show a significant decrease in G1 when compared with G3, G1 with G2, G1 with G4, G3 with G2, G3 with G4, and significantly increase when G2 compared with G4 at p value (p ≤ 0. 0001), but heterophil % shows a significant increase in G1 when compared with G3, G1 with G2, G1 with G4, G3 with G2, and G3 with G4 (p ≤ 0.0001).
HI antibody titers against Newcastle disease and Avian Influenza vaccines
As titrated by the hemagglutination inhibition (HI) test, levels of HI antibodies (log 2) against ND & AI vaccines are presented by mean ± S.E. in Table 9. In week 3, there was a significant decrease in antibody titers when G1 compared with G3 against Al (p = 0.0007). In week 4, there was a significant decrease against ND when G1 compared with G3 (p = 0.0017), G1 compared with G4 (p = 0.0332), and G2 compared with G4 (p = 0.0017). Finally, there was a significant increase when G3 compared with G2 (p ≤ 0.0001).
Table 9.
Impact of prebiotics with or without heat stress on hemagglutination inhibition (HI) antibody titers against Newcastle disease (ND) and avian influenza (AI) vaccines.
| Time | Groups | |||
|---|---|---|---|---|
| Control HI titers (Log2) |
Prebiotics HI titers (Log2) |
Heat stress HI titers (Log2) |
Heat stress prebiotics HI titers (Log2) |
|
| Week 2 (ND) | 2.22 ± 0.147 | 2.667 ± 0.166 | – | – |
| Week 3 (Al) | 3.778 ± 0.277a | 5.333 ± 0.235b | – | – |
| Week 4 (ND) | 1 ± 0a | 1.778 ± 0.147b | 0.777 ± 0.147a,c | 1.556 ± 0.175b,d |
| Week 5 (ND) | 1.222 ± 0.147 | 1.444 ± 0.175 | 0.888 ± 0.2 | 1.444 ± 0.175 |
Each value represents mean ± S.E. A significant difference between groups was analyzed by t-test in weeks 2 and 3, and one-way ANOVA in weeks 4 and 5 at p ≤ 0.05. “–” means that the test was not performed as the heat stress started from week 4.
Parameters in the same row followed by different superscript letters are significantly different (p ≤ 0.05).
Chicken gut microbiome analysis
The high-throughput Illumina-MiSeq sequencing was used to investigate the dynamics of gut microbiota in the investigated chicken groups. A total of 22 samples were pooled into 7 samples (Table 6) that were sequenced and the sequencing output was analyzed. The sequenced samples represented different time intervals of the experiment and the four chicken groups investigated. Sequencing of the 7 fecal samples yielded a total of 888,081 16S rRNA gene sequence reads with an average length of 460 bp after the sequence quality-filtering step. The number of sequences per sample ranged from 189,884 to 97,647 with an average of 126,868.714 ± 27,880.175. The number of observed OTUs per sample ranged from 5206 to 2280 with an average of 3521 ± 934.626. The Good’s coverage of the sequenced samples ranged from 98.08 to 99.29%.
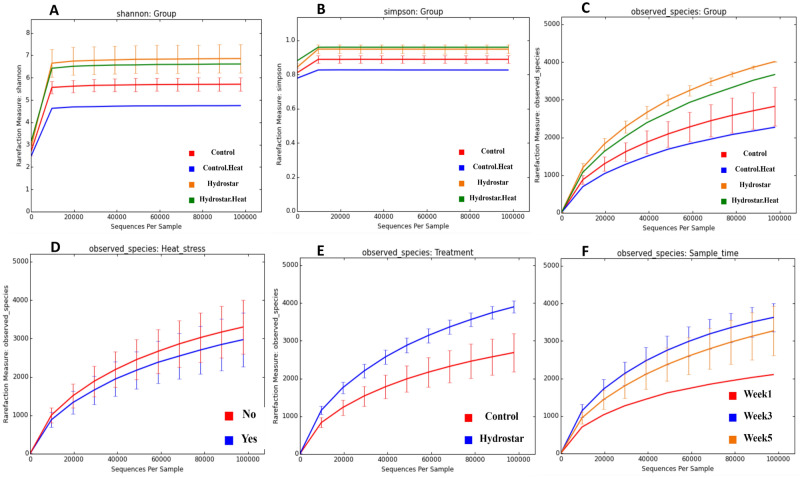
The alpha diversity in the four tested groups was calculated and evaluated. The species diversity was investigated using Simpson’s and Shannon’s metrics (Fig. 1A,B). There was a significant difference recorded between the prebiotics-treated groups and the other groups in terms of Simpson’s and Shannon’s metrics (Monte Carlo permutations (999), (p = 0.035 and 0.056), respectively. The number of species observed was the highest in G3 followed by G4, G1 then G2 (Fig. 1C). There was no significant effect of the HS on the number of observed species within samples that were treated with prebiotics when compared to the control samples (Monte Carlo permutations (999), (p = 0.689) (Fig. 1D). However, treatment with prebiotics significantly increased the number of observed species within samples (Monte Carlo permutations (999), (p = 0.054) (Fig. 1E). Regarding the sampling time, the least number of observed species was during week 1 followed by week 5 and then week 3 with no recorded significant difference (Monte Carlo permutations (999), (p = 0.963) (Fig. 1F).
Figure 1.
Rarefaction curves of alpha diversity metrics assessed for the sequenced fecal samples. (A) Shannon index representing the diversity within samples. (B) Simpson’s index (similarity index) revealing species’ evenness. (C–F) The observed species representing the true species richness within different sampling groups (Heat stress, Prebiotics treatment (Hydrostar) and Sample time, respectively).
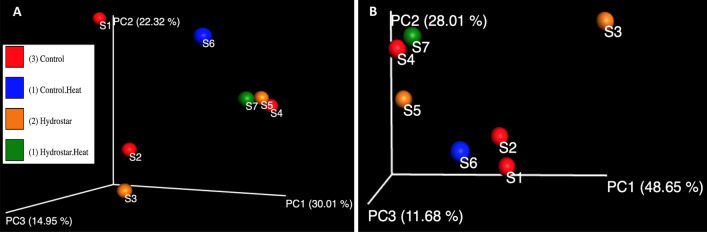
The unweighted and weighted beta diversity between G1, G2, G3, and G4 samples was investigated using the Unifrac distance metrics (Fig. 2A,B). Based on the principal coordinates analysis plot (PCoA), all the prebiotics samples in week 5 (with and without HS) clustered together away from the HS sample (S6) in the same week (Fig. 2A,B). The impact of the prebiotics administration and HS application on the overall microbial community diversity between samples was investigated. There was a significant effect of prebiotics administration on the overall microbial community composition between individual samples (PERMANOVA, p = 0.058). The application of HS was accompanied by a significant effect on the overall microbial community composition between individual samples (PERMANOVA, p = 0.017).
Figure 2.
Principal coordinates analysis (PCoA) demonstrating the beta diversity evaluated by the unweighted UNIFRAC (A) and weighted UNIFRAC (B) method. Individual datasets are designated as spheres which are colored in relation to their sample treatment as follows; red: control (no heat stress or prebiotics), blue: control under heat stress, orange: prebiotics (hydrostar®), and Green (dark): prebiotic under heat stress.
The effect of the sampling time on the microbial community composition was analyzed. The microbial populations of week 1, week 3, and week 5 were compared to test the significance of sampling time on the total microbial diversity. There was a significant effect of sampling time on the overall community composition between individual samples (PERMANOVA, p = 0.008 and ANOSIM, p = 0.004).
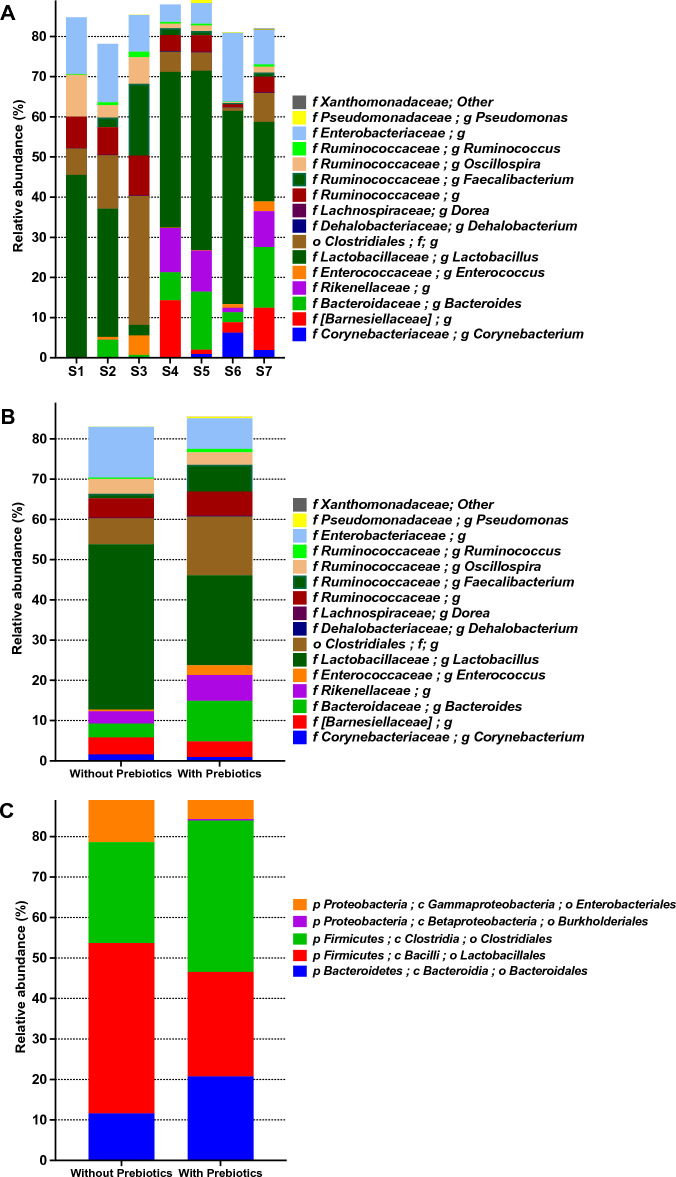
The composition of the microbiota was further examined (Figs. 3, 4). In the prebiotics-treated samples, the major components at the genus level belonged to Lactobacillus (22.3%) (Fig. 3A,B). In addition, undefined genera of the order Clostridiales (14.6%), family Bacteroidaceae (10.0%), and family Enterobacteriaceae (7.6%) were detected. On the contrary, in samples whose chickens were not treated by prebiotics the relative abundance of those genera was (41.0%), (6.4%), (3.4%) and (12.5%), respectively, with no significant differences (Kruskal–Wallis, Bonferroni’s multiple comparisons tests, (p > 0.1) (Fig. 3B, Table S1). However, there was a significant difference in other bacterial categories as Pseudomonadaceae, Dorea, an undefined genus of order Lactobacillales, Dehalobacterium, and an undefined genus of family Xanthomonadaceae (p = 0.077) with relative abundance in prebiotics-treated and non-prebiotics-treated samples of (0.34% and 0.02%), (0.133% and 0.067%), (0.06% and 0.011%), (0.0147% and 0.0024%) and (0.06% and 0%) respectively (Fig. 3B, Table S1). Figure 3C describes the relative abundance of the most dominant orders in prebiotics-treated and non-prebiotics-treated samples as follows: Enterobacteriales (7.73% and 12.81%), Burkholderiales (0.36% and 0.02%), Clostridiales (37.39% and 24.91%), Lactobacillales (25.80% and 42.09%) and Bacteroidales (20.76% and 11.61%), respectively.
Figure 3.
The relative abundance of the most dominant categories of bacteria in prebiotics-treated samples. (A) The relative abundance of the most dominant bacteria genera in the different samples. S1 represents all groups before any treatment on day zero. S2 and S4 represent the control group (without prebiotics treatment and without heat stress) in Week 3 and week 5, respectively. S3 and S5 represent the prebiotics group in Week 3 and week 5, respectively. S6 represents the heat stress group in week 5. S7 represents the heat stress + prebiotics group in week 5. (B) The relative abundance of the most dominant genera in prebiotics-treated and non-prebiotics-treated samples. (C) The relative abundance of the most dominant order in prebiotics-treated and non-prebiotics-treated samples.
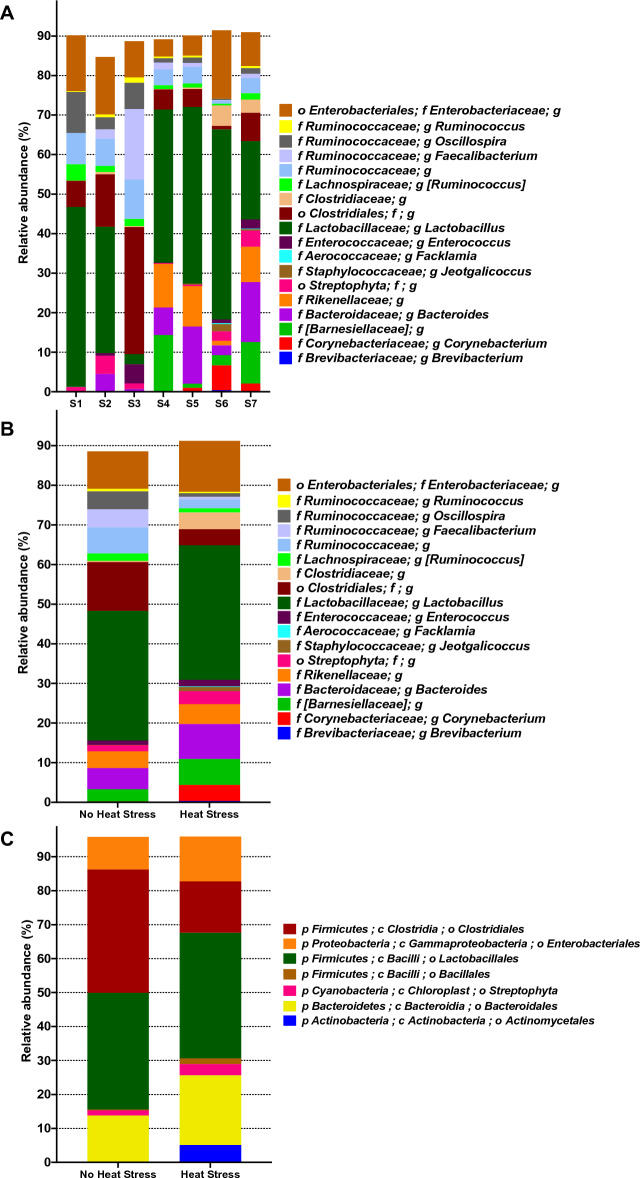
Figure 4.
The relative abundance of the most dominant bacteria in heat-stressed samples. (A) The relative abundance of the most dominant bacterial genera in the different samples. S1 represents all groups before any treatment on day zero. S2 and S4 represent the control group (without prebiotics treatment and without heat stress) in week 3 and week 5, respectively. S3 and S5 represent the prebiotics group in week 3 and week 5, respectively. S6 represents the heat stress group in week 5. S7 represents the heat stress + prebiotics group in week 5. (B) The relative abundance of the most dominant genera in heat-stressed and non-heat-stressed samples. (C) The relative abundance of the most dominant orders in heat-stressed and non-heat-stressed samples.
The major bacterial taxa and their relative abundance in the fecal samples in both heat-stressed and non-stressed groups were examined (Fig. 4A,B). In the heat-stressed groups, the major components at the genus level were belonging to Lactobacillus (33.9%), undefined genus of family Enterobacteriaceae (12.9%), Bacteroides (8.8%), undefined genus of family Barnesiellaceae (6.5%), undefined genus of family Rikenellaceae (5.0%), an undefined genus of family Clostridiaceae (4.0%), an unknown genus of order Streptophyta (3.2%), Enterococcus (1.6%), and Ruminococcus (1.0%) (Fig. 4B). Concerning the non-stressed bird samples, the relative abundances of those genera were (32.7%), (9.4%), (5.3%), (3.0%), (4.2%), (12.3%), (1.5%), (1.1%) and (1.9%), respectively, with no significant differences (Kruskal–Wallis, Bonferroni’s multiple comparisons tests, p > 0.1) (Fig. 4B, Table S2). However, there was a significant difference in other bacterial genera as an undefined genus of family Clostridiaceae, Corynebacterium, Jeotgalicoccus, and Brevibacterium (p = 0.05) with relative abundance in heat-stressed and non-stressed samples (4.25% and 0.25%), (4.09% and 0.19%), (1.08% and 0.04%), and (0.2718% and 0.0076%) respectively (Table S2). In addition to genus Facklamia (p = 0.04) with relative abundance in heat-stressed and non-stressed samples (0.1460% and 0.0029%) (Table S2). Figure 4C illustrates the relative abundance of the most dominant orders in heat-stressed and non-heat-stressed samples as follows: Clostridiales (15.13% and 36.31%), Enterobacteriales (13.19% and 9.62%), Lactobacillales (37.02%, 37.35%), Bacillales (1.96% and 0.240%), Streptophyta (3.26% and 1.53%), Bacteroidales (20.51% and 13.55%) and Actinomycetales (5.15% and 0.25%), respectively.
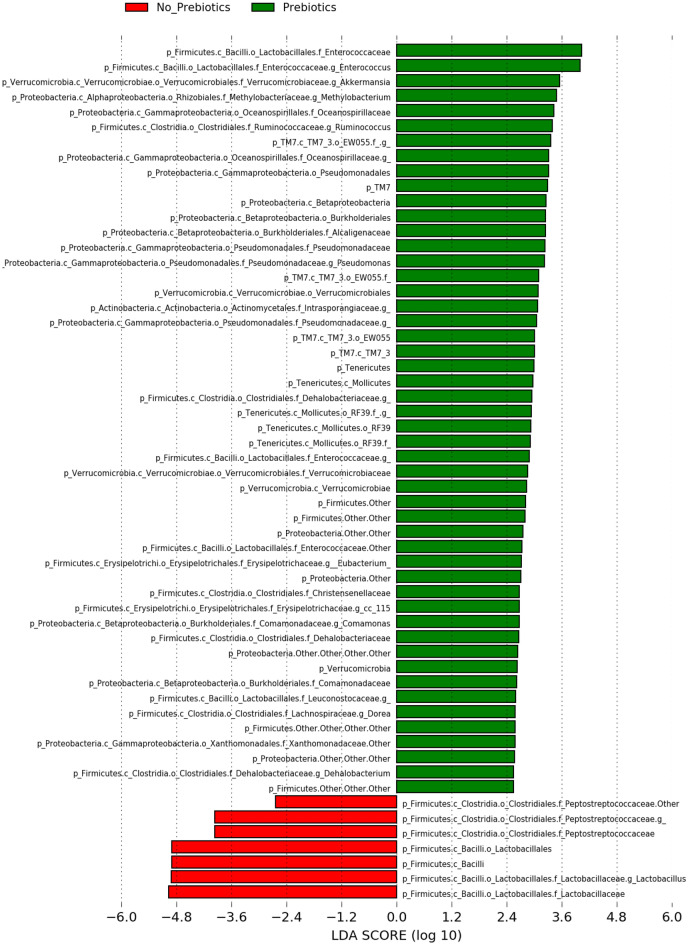
The LEfSe classification tool was used to characterize the differences between the core microbiome in the prebiotics sample and samples without prebiotics treatment (Fig. 5), in addition to the differences between the core microbiome in the heat-stressed and non-stressed samples at different levels (Fig. 6). The recorded results showed that the core microbiome in the prebiotics samples differed significantly from those samples without prebiotics treatment. The core microbiome in the prebiotics samples belonged to phyla Proteobacteria and Firmicutes including families Erysipelotrichaceae, Xanthomonadaceae, Intrasporangiaceae and Oceanospirillaceae, and genera Eubacterium, Dorea, Methylobacterium, Pseudomonas, Dehalobacterium and Comamonas (Fig. 5).
Figure 5.
LEfSe core microbiome analysis. Data is presented as a histogram of the linear discriminative analysis (LDA) scores processed to feature the differentially rich taxa (core microbiome) between prebiotics and non-prebiotics samples.
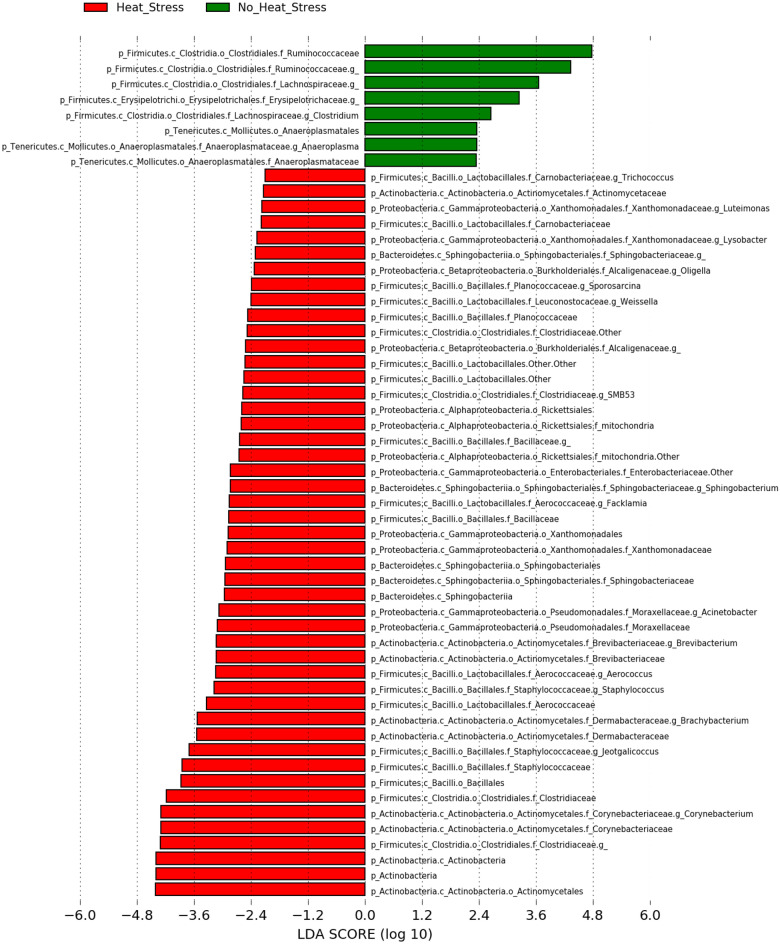
Figure 6.
LEfSe core microbiome analysis. Data is presented as a histogram of the linear discriminative analysis (LDA) scores processed to feature differentially rich taxa (core microbiome) between heat-stressed and non-heat-stressed samples.
On the other hand, the LEfSe analysis showed that the core microbiome in the heat-stressed birds differed significantly from non-stressed ones (Fig. 6). The core microbiome in the heat-stressed bird included order Lactobacillales, families Clostridiaceae, Corynebacteriaceae, Staphylococcaceae, Dermabacteriaceae, Brevibacteriaceae, Enterobacteriaceae, Bacillaceae, Aerococcaceae, Leuconostocaceae, Aerococcaceae, Xanthomonadaceae, Planococcaceae, Alcaligenaceae, Aerococcaceae, Carnobacteriaceae, genus Trichococcus, Aerococcus, Staphylococcus, Sporosarcina, Oligella, Luteimonas, Facklamia, Lysobacter, Brevibacterium, Corynebacterium, Jeotgalicoccus, Brachybacterium, and Weissella (Fig. 6). The core microbiome of the non-heat stressed samples was affiliated to the families Ruminococcaceae and Erysipelotrichaceae (Fig. 6).
Discussion
Concerns about how climate change would affect animal output are mounting. The average temperature is predicted to rise by 2 °C to 6 °C by the year 2100, posing a climatic challenge and increasing the likelihood of heat stress incidents in livestock species22. Our research demonstrated that chicken performance was negatively impacted by high ambient temperatures however, we proved that supplementing the birds with prebiotics reduced the effects of heat stress.
One of the major issues confronting the poultry business, particularly in tropical countries, is broiler breeding under high temperature. However, there is insufficient data on the negative effects of increased temperatures on the productivity output, body weight, gut microbiome, and immunological response in broilers as measured by antibody titer to different vaccines as well as total and differential leukocytic counts4.
In this study, exposure to HS revealed negative consequences on body weight, gut microbiome, and immunological response in broiler chickens. Results of the current study showed that HS (32–35 °C) had a negative impact on broiler BW, reduced appetite, lowered feed intake, and lowered final body weight. This declined growth of chickens bred under HS could be attributed to the fact that the elevated external temperature excited peripheral thermal sensors, which sent inhibitory nerve impulses to the hunger center in the hypothalamus, resulting in a reduction in the feed intake and increased water uptake to compensate for increased evaporative heat loss needs14, 23.
Our experiment showed that the (BW) in the first 2 weeks in the prebiotics groups was greater than those of the non-prebiotics groups. The overall FI masses in G1 and G2 were 3642 and 3389, while in G3 and G4 they were 3443 g and 3340, respectively. So, we concluded that prebiotic treatment maintains the feed capacity of birds while control groups show a severe decline in FI. In the 5th week (last week of our experiment), the bird performance in the groups administered prebiotics either under normal conditions (G3) or under HS (G4) showed improvement in BW and feed conversion ratio (FCR) in comparison with the other groups that didn’t receive any prebiotics either under normal conditions (G1) or under HS (G2). These results agree with those obtained by Sohail et al.24 who recorded that the addition of mannan-oligosaccharides and probiotics improved the performance of broiler chickens exposed to chronic HS. In addition, He et al.25 demonstrated that HS could cause weight loss in ducks and Luo et al.26 confirmed the same result in broiler chickens. These findings could be explained as the addition of prebiotics improves appetite, increases feed consumption, improves the gut microbiome, increases intestinal villi length, enhances nutrients absorption, and increases pathogen resistance that has a positive impact on the BW and lowers FCR27.
Interestingly, our prebiotics groups recorded decreased mortality rate even with heat stress in comparison to control groups, and this agrees with the study of Sugiharto who reported that prebiotics improved host defense and decreased avian mortality brought on by gut pathogen attack12.
Our results showed that the control group has a higher significant TWBC than the prebiotics group in weeks 4 and 5, and this result agrees with Amer et al.28 who reported that WBC levels of the control group were significantly higher than all immune treated in week 4 and 5. In addition, Al-Mansour et al.29 recorded that chicks fed yeast culture at a rate of 1.5 g/kg had significantly (p < 0.05) reduced leucocytic counts than controls. Also, we found that the groups exposed to HS recorded lower total white blood cells count (TWBC) which agrees with the findings of other studies30. These findings may be explained by the atrophy of all lymphoid tissues (bursa, thymus, spleen, or liver) since heat stress drastically decreased their weights. This may have happened as a result of the birds eating less feed, which would have provided them with fewer nutrients for their organs’ healthy growth31. According to Abo-Al-Ela et al.32, birds exposed to high ambient temperatures had a rise in plasma corticosterone, which in turn reduced the activity of their lymphoid tissues and their overall leucocyte count. However, G4 showed higher TWBC than G2 in week 4, and this agrees with Attia et al.33 who observed that administration of mannanoligosaccharides was accompanied by higher white blood cells (WBCs) than the other groups.
Regarding differential leukocytic count, monocyte showed a significant increase in prebiotics groups than the control group in weeks 2, 3, and 5. Our study agrees with Amer et al.28 who reported that beta glucan-treated groups show higher values of monocyte than other groups. Also, lymphocytes showed a significant increase in prebiotics groups than control in week 4, and this result matched with the other studies28, 34, 35. In addition to that the lymphocytes ratios were elevated in all groups in our study, and this is in accordance with Amer et al.28 who observed that lymphocytes represented the majority of total white blood cells in birds. This can be attributed to the vaccines given for all groups as these vaccines are viral types and the major reaction is cell-mediated immunity based. Therefore, it is logical to find a higher proportion of lymphocytes.
Monocytes showed a significant increase in the heat-stressed group when compared to the non-stressed groups and this finding agrees with that of Rocchi et al.36. It has been speculated that heat stress causes severe inflammation, especially in the gut resulting in increased intestinal permeability (leaky gut), damage of intestinal integrity due to the minimized expression of tight junction protein, and enhanced bacterial expansion36.
Average serum HI antibody titers against avian influenza (AI) and Newcastle disease (ND) vaccines indicated a significant increase in the prebiotics group than the control group in weeks 3 and 4, and this matched with previous studies28, 37, 38 which indicated that the β-1,3-d glucans molecules are powerful reticuloendothelial-modulators, whose immunobiological action is mediated via inducing the production of proinflammatory cytokines. Also, average serum HI antibody titers were significantly influenced by HS. It has been mentioned that HS causes oxidative stress, which lowers lymphoid organs’ weight and may be the primary factor in lower antibody titers for infectious illnesses like Al and ND. In addition, HS increases adrenal gland activity, which increases serum corticosteroid synthesis and inhibits interleukin-2 resulting in reduced titer of antibodies39. Meanwhile, birds administrated prebiotics in the drinking water (G3) showed a good immune response against the ND vaccine in comparison with the control group (G1) and the HS group (G2). Once again, this could be correlated to the overproduction of reactive oxygen species (ROS) in chickens exposed to environmental stressors like HS, which increases the production of free radicals that damage proteins and DNA through lipid peroxidation40. Prebiotics have been shown to boost the immune system, and the supplement’s ingredients, beta-d-glucan, and mannan-oligosaccharides, bind to the receptor mannose-specific type-1 fimbriae and stimulate gut immune cells to block pathogen colonization while favoring beneficial microbes like Faecalibacterium, which are frequently linked to gut health12.
Chicken droppings are considered the best choice of sampling to monitor the cecal microbiota because of their diversity, richness, and abundance. This enables researchers to collect samples without troubling the microbiota and to diminish stress on the chickens41. A greater understanding of the role of feed additives such as prebiotics on the microbial population and their diversity in response to HS could aid in the development of focused approaches to HS relief2, 42. High throughput sequencing was used to estimate the gut microbiota and their microecological composition in poultry16, 43.
Regarding the number of observed bacterial microbiota species within samples in our study, birds showed higher diversity in week 3 than week 5. This may be as a result of the chickens being under stress in the last week of rearing due to the sudden increase in their body weights. In week 1 the species’ richness begins to grow and propagate. High ambient temperature is an important environmental indicator that regulates bacterial expansion and progression44. Continuously elevated temperatures can lead to functional problems, such as intestinal dysbiosis44, 45.
In this study, there was a significant difference recorded between control groups and prebiotics-treated groups in terms of observed species richness. Prebiotics-treated groups showed higher diversity than the control groups as prebiotics increased diversity. HS resulted in a significant decrease in population, but the prebiotics could compensate for the effect of HS and maintain its bacterial population.
The rarefaction curves and alpha diversity parameters were evaluated, and there was a significant difference in the alpha diversity indices, showing that HS had a major effect on gut microbiota diversity. The data revealed that the chicken gut microbiome was mainly comprised of five phyla: Firmicutes, Proteobacteria, Bacteroidetes, Actinobacteria, and Cyanobacteria. Firmicutes was the major microbial group, accounting for more than 45% of the microbiome. In addition, we found Tenericutes, Verrucomicrobia and Acidobacteria in lower concentrations (less than 0.2%) in the broilers’ gut. Our study agrees with the study of Wen et al.46 who found that Firmicutes accounted for more than 60% of the chicken gut microbiota.
In the current study, in week 5 under heat stress, the Firmicutes level was higher in the control group than in the prebiotics groups while Bacteroidetes were higher in prebiotics groups than in the control groups under heat stress applications. Adding prebiotics to the drinking water of birds increases the count of beneficial microbiome especially Lactobacillus which improve performance and plays an important function in food fermentation47. Lactobacillus dominance in the intestine has been linked to resistance to pathogens and illnesses48. Our data revealed that L. murinus, L. ruminus, and L. casei were the most abundant lactobacilli in our prebiotics-treated samples. Lactobacillus has been found to be extremely abundant in the distal gut and its deficiency is usually linked to the disease conditions49, 50.
Bacteroidetes are crucial complex carbohydrate degraders and short-chain fatty acid (SCFA) makers that are well-suited to the distal gut and serve as significant commensals in the chicken gut51, 52. Firmicutes primarily break down oligosaccharides or convert lactate or acetate, two metabolic waste products, into butyrate53. The Firmicutes-to-Bacteroidetes ratio was considerably higher when more Firmicutes and fewer Bacteroidetes were found in the gut microbiota of stressed broilers54. In our study Firmicutes-to-Bacteroidetes in control and prebiotics groups was 4.08 and 3.07, respectively.
Proteobacteria have been found to grow as a result of antibiotic use or inflammation in both people and animals, and this phenomenon is thought to be a microbial indicator of gut disturbance and epithelial malfunction55, 56. In our investigation, the total percentage of Proteobacteria in the control groups (38.3%) was higher than that in the prebiotics groups (26.7%), this strongly proves the added value of using prebiotics to improve the avian gut microbiome.
Animal-developed Enterobacteriaceae bacteria such as Klebsiella pneumoniae, Escherichia coli, Proteus mirabilis, and Salmonella spp. especially serotypes Salmonella Pullorum and Salmonella Gallinarum are significant food-borne zoonotic bacteria that are prevalent in the broiler breeding sector57, 58. In our results, the overall prevalence of Enterobacteriaceae was 36.75% in control groups and 23.19% in prebiotics groups. Moreover, during the last week under heat stress exposure, the prevalence of Enterobacteriaceae in control is higher than in prebiotics groups. This again explored the added value of using prebiotics to improve the avian gut microbiome and alleviate the effect of heat stress.
In our experiment genus Brevibacterium, and Facklamia showed a significant increase in the heat-stressed group than the non-heat-stressed group. Other research indicated that few Brevibacterium-like microbes isolated from infected chicken were obtained from aspirates taken during necropsy causing bumblefoot (granuloma) lesions, Moreover, some species-level identification was not established59. Facklamia has been reported to produce septicemia and meningitis, both of which can be fatal60. This reflects the negative impact of heat stress on the avian gut microbiome and valorizes the positive impact of using prebiotics to alleviate the effect of heat stress as the abundance of these pathogens was either eradicated or significantly decreased in the prebiotics-treated groups.
Conclusion
The study of microbial structure and its interaction is useful for discovering the effect of high temperature on the intestinal barrier and can provide a hypothetical foundation and experimental suggestions for restoring body destruction affected by heat stress. In our experiments, the birds’ exposure to heat stress reflected badly on the bird performance and reduced bird immunity. Meanwhile, the addition of prebiotics in the drinking water during the rearing period of broiler chickens alleviated the negative impact of heat stress. Prebiotics were able to improve bird appetite, increase feed intake, lower FCR, improve the immunological parameters of the bird, boost the bird immune response against infections, and increase the effectiveness of bird immune response to vaccines. Moreover, using prebiotics improved the birds’ gut microbiome and alleviated the negative effect of heat stress. Administering prebiotics significantly increased the relative abundance of beneficial bacteria and eradicated pathogenic ones in the chicken gut microbiome.
Supplementary Information
Author contributions
Y.S.: conceptualization, methodology, validation, software, visualization, and writing original draft. M.H.: conceptualization, supervision, methodology, validation, software, visualization, review and editing manuscript. H.M.S.: conceptualization, supervising, review and editing manuscript. K.A.: conceptualization, supervising, review. G.E.E.: conceptualization, supervising, review and editing manuscript.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Data availability
16S sequence data was submitted to the Sequence Read Archive (SRA) under Bioproject (PRJNA977599) and SRA study (SRR24784609-15). The data was assigned Accession Numbers SAMN35526087, SAMN35526088, SAMN35526089, SAMN35526090, SAMN35526091, SAMN35526092 and SAMN35526093. The data has been released and it is available via the following link: https://submit.ncbi.nlm.nih.gov/subs/bioproject/SUB13482173/overview.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yara Sayed, Email: Yara.sayed@cu.edu.eg.
Mariam Hassan, Email: mariam.hassan@pharma.cu.edu.eg.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-023-40997-7.
References
- 1.Farghly MFA, et al. Implementation of different feed withdrawal times and water temperatures in managing turkeys during heat stress. Poult. Sci. 2018;97:3076–3084. doi: 10.3382/ps/pey173. [DOI] [PubMed] [Google Scholar]
- 2.Awad EA, Zulkifli I, Ramiah SK, Khalil ES, Abdallh ME. Prebiotics supplementation: An effective approach to mitigate the detrimental effects of heat stress in broiler chickens. Worlds Poult. Sci. J. 2021;77:135–151. doi: 10.1080/00439339.2020.1759222. [DOI] [Google Scholar]
- 3.Lara LJ, Rostagno MH. Impact of heat stress on poultry production. Animals. 2013;3:356–369. doi: 10.3390/ani3020356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Salem HM, et al. Heat stress in poultry with particular reference to the role of probiotics in its amelioration: An updated review. J. Therm. Biol. 2022;108:103302. doi: 10.1016/j.jtherbio.2022.103302. [DOI] [PubMed] [Google Scholar]
- 5.Saeed M, et al. Heat stress management in poultry farms: A comprehensive overview. J. Therm. Biol. 2019;84:414–425. doi: 10.1016/j.jtherbio.2019.07.025. [DOI] [PubMed] [Google Scholar]
- 6.Jiang S, Yan F-F, Hu J-Y, Mohammed A, Cheng H-W. Bacillus subtilis-based probiotic improves skeletal health and immunity in broiler chickens exposed to heat stress. Animals. 2021;11:1494. doi: 10.3390/ani11061494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Horváth M, Babinszky L. Impact of selected antioxidant vitamins (Vitamin A, E and C) and micro minerals (Zn, Se) on the antioxidant status and performance under high environmental temperature in poultry. A review. Acta Agric. Scand. Sect. A Anim. Sci. 2018;68:152–160. doi: 10.1080/09064702.2019.1611913. [DOI] [Google Scholar]
- 8.Abd El-Hack ME, et al. Alternatives to antibiotics for organic poultry production: Types, modes of action and impacts on bird's health and production. Poult. Sci. 2022;101:101696. doi: 10.1016/j.psj.2022.101696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McEwen SA, Angulo FJ, Collignon PJ, Conly JM. Unintended consequences associated with national-level restrictions on antimicrobial use in food-producing animals. Lancet Planet. Health. 2018;2:e279–e282. doi: 10.1016/S2542-5196(18)30138-4. [DOI] [PubMed] [Google Scholar]
- 10.Gibson GR, Roberfroid MB. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995;125:1401–1412. doi: 10.1093/jn/125.6.1401. [DOI] [PubMed] [Google Scholar]
- 11.Gibson GR, Probert HM, Loo JV, Rastall RA, Roberfroid MB. Dietary modulation of the human colonic microbiota: Updating the concept of prebiotics. Nutr. Res. Rev. 2004;17:259–275. doi: 10.1079/NRR200479. [DOI] [PubMed] [Google Scholar]
- 12.Sugiharto S. Role of nutraceuticals in gut health and growth performance of poultry. J. Saudi Soc. Agric. Sci. 2016;15:99–111. doi: 10.1016/j.jssas.2014.06.001. [DOI] [Google Scholar]
- 13.Teng PY, Kim WK. Review: Roles of prebiotics in intestinal ecosystem of broilers. Front. Vet. Sci. 2018;5:245. doi: 10.3389/fvets.2018.00245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ahmed E, Abdelrahman M, Gahreeb K. Effect of probiotic on growth performance, carcass traits, and clinical health parameters of broilers reared under heat stress in upper Egypt. SVU Int. J. Vet. Sci. 2019;2:27–44. doi: 10.21608/svu.2019.11221.1012. [DOI] [Google Scholar]
- 15.Ghareeb K, Awad WA, Sid-Ahmed OE, Böhm J. Insights on the host stress, fear and growth responses to the deoxynivalenol feed contaminant in broiler chickens. PLoS ONE. 2014;9:e87727. doi: 10.1371/journal.pone.0087727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Darwish N, Shao J, Schreier LL, Proszkowiec-Weglarz M. Choice of 16S ribosomal RNA primers affects the microbiome analysis in chicken ceca. Sci. Rep. 2021;11:11848. doi: 10.1038/s41598-021-91387-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Caporaso JG, et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods. 2010;7:335–336. doi: 10.1038/nmeth.f.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hassan M, Essam T, Mira A, Megahed S. Biomonitoring detoxification efficiency of an algal-bacterial microcosm system for treatment of coking wastewater: Harmonization between Chlorella vulgaris microalgae and wastewater microbiome. Sci. Total Environ. 2019;677:120–130. doi: 10.1016/j.scitotenv.2019.04.304. [DOI] [PubMed] [Google Scholar]
- 19.Hassan M, Essam T, Megahed S. Illumina sequencing and assessment of new cost-efficient protocol for metagenomic-DNA extraction from environmental water samples. Braz. J. Microbiol. 2018;49(Suppl 1):1–8. doi: 10.1016/j.bjm.2018.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hernández-Granados MJ, Ortiz-Basurto RI, Jiménez-Fernández M, García-Munguía CA, Franco-Robles E. Dietary encapsulated Bifidobacterium animalis and Agave fructans improve growth performance, health parameters, and immune response in broiler chickens. Anim. Biosci. 2021;35:587–595. doi: 10.5713/ab.21.0213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oberländer B, et al. Evaluation of newcastle disease antibody titers in backyard poultry in Germany with a vaccination interval of twelve weeks. PLoS ONE. 2020;15:e0238068. doi: 10.1371/journal.pone.0238068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kpomasse CC, Oke OE, Houndonougbo FM, Tona K. Broiler production challenges in the tropics: A review. Vet. Med. Sci. 2021;7:831–842. doi: 10.1002/vms3.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sritharet N, Hara H, Yoshida Y, Hanzawa K, Watanabe S. Effects of heat stress on histological features in pituicytes and hepatocytes, and enzyme activities of liver and blood plasma in Japanese quail (Coturnix japonica) J. Poult. Sci. 2002;39:167–178. doi: 10.2141/jpsa.39.167. [DOI] [Google Scholar]
- 24.Sohail MU, et al. Alleviation of cyclic heat stress in broilers by dietary supplementation of mannan-oligosaccharide and Lactobacillus-based probiotic: Dynamics of cortisol, thyroid hormones, cholesterol, C-reactive protein, and humoral immunity. Poult. Sci. 2010;89:1934–1938. doi: 10.3382/ps.2010-00751. [DOI] [PubMed] [Google Scholar]
- 25.He J, et al. Associations of gut microbiota with heat stress-induced changes of growth, fat deposition, intestinal morphology, and antioxidant capacity in ducks. Front. Microbiol. 2019;10:903. doi: 10.3389/fmicb.2019.00903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Luo J, et al. Effect of epigallocatechin gallate on growth performance and serum biochemical metabolites in heat-stressed broilers. Poult. Sci. 2018;97:599–606. doi: 10.3382/ps/pex353. [DOI] [PubMed] [Google Scholar]
- 27.Sohail MU, et al. Effect of supplementation of prebiotic mannan-oligosaccharides and probiotic mixture on growth performance of broilers subjected to chronic heat stress. Poult. Sci. 2012;91:2235–2240. doi: 10.3382/ps.2012-02182. [DOI] [PubMed] [Google Scholar]
- 28.Amer MM, Tammam SM, Dahshan AHM, AlAmir A. Studies on the effect of different immunostimulants on chick's immune response to inactivated avian influenza and Newcastle vaccines. J. Vet. Med. Res. 2017;24:176–185. doi: 10.21608/jvmr.2017.43281. [DOI] [Google Scholar]
- 29.Mansour SA, Khalf A, Homidan IA, Fathi MH. Feed efficiency and blood hematology of broiler chicks given a diet supplemented with yeast culture. Int. J. Poult. Sci. 2011;10:603–607. doi: 10.3923/ijps.2011.603.607. [DOI] [Google Scholar]
- 30.Silva JE. The thermogenic effect of thyroid hormone and its clinical implications. Ann. Intern. Med. 2003;139:205–213. doi: 10.7326/0003-4819-139-3-200308050-00010. [DOI] [PubMed] [Google Scholar]
- 31.Bartlett JR, Smith MO. Effects of different levels of zinc on the performance and immunocompetence of broilers under heat stress. Poult. Sci. 2003;82:1580–1588. doi: 10.1093/ps/82.10.1580. [DOI] [PubMed] [Google Scholar]
- 32.Abo-Al-Ela HG, et al. Stress and immunity in poultry: Light management and nanotechnology as effective immune enhancers to fight stress. Cell Stress Chaperones. 2021;26:457–472. doi: 10.1007/s12192-021-01204-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Attia YA, Bakhashwain AA, Bertu NK. Thyme oil (Thyme vulgaris L.) as a natural growth promoter for broiler chickens reared under hot climate. Ital. J. Anim. Sci. 2017;16:275–282. doi: 10.1080/1828051X.2016.1245594. [DOI] [Google Scholar]
- 34.Kong X, Hu Y, Rui R, Wang D, Li X. Effects of Chinese herbal medicinal ingredients on peripheral lymphocyte proliferation and serum antibody titer after vaccination in chicken. Int. Immunopharmacol. 2004;4:975–982. doi: 10.1016/j.intimp.2004.03.008. [DOI] [PubMed] [Google Scholar]
- 35.Zhao X, Hu Y, Wang D, Liu J, Guo L. The comparison of immune-enhancing activity of sulfated polysaccharidses from Tremella and Condonpsis pilosula. Carbohydr. Polym. 2013;98:438–443. doi: 10.1016/j.carbpol.2013.06.043. [DOI] [PubMed] [Google Scholar]
- 36.Rocchi A, et al. Experimental cyclic heat stress on intestinal permeability, bone mineralization, leukocyte proportions and meat quality in broiler chickens. Animals. 2022;12:73. doi: 10.3390/ani12101273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li B, et al. Yeast glucan particles activate murine resident macrophages to secrete proinflammatory cytokines via MyD88- and Syk kinase-dependent pathways. Clin. Immunol. 2007;124:170–181. doi: 10.1016/j.clim.2007.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ichinohe T, et al. Induction of cross-protective immunity against influenza A virus H5N1 by an intranasal vaccine with extracts of mushroom mycelia. J. Med. Virol. 2010;82:128–137. doi: 10.1002/jmv.21670. [DOI] [PubMed] [Google Scholar]
- 39.Khan RU, et al. Physiological dynamics in broiler chickens under heat stress and possible mitigation strategies. Anim. Biotechnol. 2023;34:438–447. doi: 10.1080/10495398.2021.1972005. [DOI] [PubMed] [Google Scholar]
- 40.Mohammed AA, Jiang S, Jacobs JA, Cheng HW. Effect of a synbiotic supplement on cecal microbial ecology, antioxidant status, and immune response of broiler chickens reared under heat stress. Poult. Sci. 2019;98:4408–4415. doi: 10.3382/ps/pez246. [DOI] [PubMed] [Google Scholar]
- 41.Takeshita N, Watanabe T, Ishida-Kuroki K, Sekizaki T. Transition of microbiota in chicken cecal droppings from commercial broiler farms. BMC Vet. Res. 2021;17:10. doi: 10.1186/s12917-020-02688-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhou J, et al. Yeast cell-wall polysaccharides improve immunity and attenuate inflammatory response via modulating gut microbiota in LPS-challenged laying hens. Int. J. Biol. Macromol. 2023;224:407–421. doi: 10.1016/j.ijbiomac.2022.10.133. [DOI] [PubMed] [Google Scholar]
- 43.Wang Y, et al. Effect of probiotics on the meat flavour and gut microbiota of chicken. Sci. Rep. 2017;7:6400. doi: 10.1038/s41598-017-06677-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kovats RS, Hajat S. Heat stress and public health: A critical review. Annu. Rev. Public Health. 2008;29:41–55. doi: 10.1146/annurev.publhealth.29.020907.090843. [DOI] [PubMed] [Google Scholar]
- 45.He J, et al. Heat stress affects fecal microbial and metabolic alterations of primiparous sows during late gestation. J. Anim. Sci. Biotechnol. 2019;10:84. doi: 10.1186/s40104-019-0391-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wen C, et al. Heat stress alters the intestinal microbiota and metabolomic profiles in mice. Front. Microbiol. 2021;12:706772. doi: 10.3389/fmicb.2021.706772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Heeney DD, Gareau MG, Marco ML. Intestinal Lactobacillus in health and disease, a driver or just along for the ride? Curr. Opin. Biotechnol. 2018;49:140–147. doi: 10.1016/j.copbio.2017.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Reid G, Burton J. Use of Lactobacillus to prevent infection by pathogenic bacteria. Microbes Infect. 2002;4:319–324. doi: 10.1016/s1286-4579(02)01544-7. [DOI] [PubMed] [Google Scholar]
- 49.Rossi M, et al. Mining metagenomic whole genome sequences revealed subdominant but constant Lactobacillus population in the human gut microbiota. Environ. Microbiol. Rep. 2016;8:399–406. doi: 10.1111/1758-2229.12405. [DOI] [PubMed] [Google Scholar]
- 50.Tabashsum Z, et al. Competitive reduction of poultry-borne enteric bacterial pathogens in chicken gut with bioactive Lactobacillus casei. Sci. Rep. 2020;10:16259. doi: 10.1038/s41598-020-73316-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fan Y, Ju T, Bhardwaj T, Korver DR, Willing BP. Week-old chicks with high bacteroides abundance have increased short-chain fatty acids and reduced markers of gut inflammation. Microbiol. Spectr. 2023;11:e0361622. doi: 10.1128/spectrum.03616-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Śliżewska K, Markowiak-Kopeć P, Żbikowski A, Szeleszczuk P. The effect of symbiotic preparations on the intestinal microbiota and her metabolism in broiler chickens. Sci. Rep. 2020;10:4281. doi: 10.1038/s41598-020-61256-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kollarcikova M, et al. Different bacteroides species colonise human and chicken intestinal tract. Microorganisms. 2020;8:1483. doi: 10.3390/microorganisms8101483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.He Y, Maltecca C, Tiezz F. Potential use of gut microbiota composition as a biomarker of heat stress in monogastric species: A review. Animals. 2021;11:1833. doi: 10.3390/ani11061833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Litvak Y, Byndloss MX, Tsolis RM, Bäumler AJ. Dysbiotic proteobacteria expansion: A microbial signature of epithelial dysfunction. Curr. Opin. Microbiol. 2017;39:1–6. doi: 10.1016/j.mib.2017.07.003. [DOI] [PubMed] [Google Scholar]
- 56.Dehau T, Cherlet M, Croubels S, van Immerseel F, Goossens E. A high dose of dietary berberine improves gut wall morphology, despite an expansion of Enterobacteriaceae and a reduction in beneficial microbiota in broiler chickens. mSystems. 2023;8:e0123922. doi: 10.1128/msystems.01239-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vaid RK, Thakur Z, Anand T, Kumar S, Tripathi BN. Comparative genome analysis of Salmonella enterica serovar Gallinarum biovars Pullorum and Gallinarum decodes strain specific genes. PLoS ONE. 2021;16:e0255612. doi: 10.1371/journal.pone.0255612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Su Y, et al. Drug resistance analysis of three types of avian-origin carbapenem-resistant Enterobacteriaceae in Shandong Province, China. Poult. Sci. 2023;102:102483. doi: 10.1016/j.psj.2023.102483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Pascual C, Collins MD. Brevibacterium avium sp. nov., isolated from poultry. Int. J. Syst. Bacteriol. 1999;49:1527–1530. doi: 10.1099/00207713-49-4-1527. [DOI] [PubMed] [Google Scholar]
- 60.Li A, et al. Long-term hexavalent chromium exposure disturbs the gut microbial homeostasis of chickens. Ecotoxicol. Environ. Saf. 2022;237:113532. doi: 10.1016/j.ecoenv.2022.113532. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
16S sequence data was submitted to the Sequence Read Archive (SRA) under Bioproject (PRJNA977599) and SRA study (SRR24784609-15). The data was assigned Accession Numbers SAMN35526087, SAMN35526088, SAMN35526089, SAMN35526090, SAMN35526091, SAMN35526092 and SAMN35526093. The data has been released and it is available via the following link: https://submit.ncbi.nlm.nih.gov/subs/bioproject/SUB13482173/overview.