Abstract
There is controversial literature about the effects of the interleukin-2 (IL-2) cytokine family in COVID-19 pathogenesis and immunity. So we aimed to identify the potential in the role of the IL-2 family in COVID-19. A narrative review search was done through online databases, including PubMed, Scopus, and Web of Science. The search deadline was up to December 2022. We applied no time limits for the searching strategy. After retrieving articles from the databases, the authors summarized the data into two data extraction tables. The first data extraction table described the changes in the IL-2 cytokine family in COVID-19 and the second table described the therapeutic interventions targeting IL-2 family cytokines. The results of the literature on the role of the IL-2 cytokine family do not show a singular rule. IL-2 cytokine family can change during severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Some studies suggest that IL-2 cytokine family rise during the infection and cause severe inflammatory response and cytokine storm. These cytokines are shown to be increased in immunocompromised patients and worsen their prognosis. In individuals without underlying disease, the upregulation of the IL-2 family shows the clinical outcome of the disease and rises with disease severity. However, some other studies show that these cytokines do not significantly change. IL-2 cytokine family is mostly upregulated in healthy individuals who had vaccination, but immunocompromised patients did not show significant changes after a single dose of vaccines, which shows that these patients need booster doses for efficient immunity. IL-2 cytokine family can also be used as immunotherapy agents in COVID-19.
1. Introduction
An unusual case of pneumonia was reported in China toward the end of 2019, and tests confirmed it to be a new coronavirus disease named coronavirus disease 2019 (COVID-19) [1]. COVID-19, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [2], has become a global pandemic burden with devastating health, social, and economic repercussions [2, 3]. Since then, researchers have struggled to determine the possible molecular mechanisms which interfere with disease pathogenicity to find the pharmacologic targets [4–6]. The term “cytokine storm” refers to the excessive release of cytokines caused by the activation of immune system cells in response to a viral invasion [7, 8]. Cytokine storm might have a role in poor outcomes in patients with COVID-19, and immune responses were associated with the emergence and progression of the virus [9]. Interleukins (ILs) were shown to have a crucial role in the development of COVID-19, as demonstrated by the growing body of research on this topic. Serum interleukin levels were significantly higher in severely ill COVID-19 patients than in those with milder illnesses [10].
Moreover, there were variations in cytokine levels among COVID-19 individuals who did and did not survive [11]. IL-2 is a cytokine that is responsible for the regulation of white blood cells, most often lymphocytes. IL-2 binds to IL-2 receptors, expressed on lymphocytes, to exert its actions. Activated CD4+ T cells and CD8+ T cells are the primary producers of IL-2 [12]. IL-2 shares a common structure with other cytokines, including IL-4, 7, 9, 15, and 21. They all possess a four-alpha helix bundle [12]. Previous research on the role of IL-2 in viral respiratory infection shows that the IL-2 levels can be used to determine the prognosis of lung damage in influenza A patients. Considering the evidence on the effects of IL-2 in viral infections, this study aims to review the molecular mechanisms and immunomodulatory effects of IL-2 in COVID-19 and its targeting agents [13].
A narrative review search was done through online databases, including PubMed, Scopus, and Web of Science. The following keywords were used to do the searching strategy: (IL-2 OR IL-2R OR IL-4 OR IL-7 OR IL-9 OR IL-15 OR IL-21) AND (COVID-19 OR SARS-CoV-2). The inclusion criteria were the original studies on the role of the IL-2 cytokine family in COVID-19. The exclusion criteria were non-English studies, studies without full text, letters, and conference papers. We applied no time limits for the searching strategy. The search deadline was up to December 2022.
2. Immunologic Background of COVID-19
The COVID-19 pandemic has created an unprecedented global health crisis, with millions of people infected and hundreds of thousands of deaths worldwide [14]. COVID-19 is caused by a novel coronavirus called SARS-CoV-2 [15], which primarily affects the respiratory system and can cause a wide range of clinical manifestations, from asymptomatic infection to severe respiratory illness and death [4, 16]. The immune system plays a crucial role in the pathogenesis of COVID-19, both in terms of the host response to the virus and the development of disease [3, 17]. The innate immune system is the first line of defense against invading pathogens, including SARS-CoV-2. The innate immune response is initiated by the recognition of pathogen-associated molecular patterns (PAMPs) by pattern recognition receptors (PRRs) expressed on the surface of immune cells [18]. These PRRs can recognize viral RNA [19], lipopolysaccharides [20], and other pathogen-associated molecules and initiate signaling cascades that lead to the production of pro-inflammatory cytokines, chemokines, and other immune mediators. In COVID-19, the innate immune response is activated early after infection and plays a critical role in controlling viral replication and spread [21]. However, the sustained and excessive production of pro-inflammatory cytokines, including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interferon-gamma (IFN-γ), can also lead to severe tissue damage and organ dysfunction, known as a cytokine storm. In COVID-19, the adaptive immune response is critical for the clearance of the virus and the prevention of reinfection [22, 23]. The presence of neutralizing antibodies and virus-specific T cells is associated with a better prognosis and the development of protective immunity. However, the dysregulation of the adaptive immune response can also contribute to the pathogenesis of severe disease [24]. Cytokines are produced by various immune cells, including T cells, B cells, macrophages, and dendritic cells, and they act on a range of target cells, including immune cells, endothelial cells, and epithelial cells [25]. In COVID-19 patients, IL-1 family cytokines are elevated, and they are thought to contribute to the cytokine storm observed in severe cases of COVID-19. IL-1 inhibitors, such as anakinra, have been used in different investigations to reduce the inflammatory response and improve outcomes in COVID-19 patients [26]. IL-6 is another cytokine that is elevated in COVID-19 patients and is thought to play a critical role in the cytokine storm observed in the severe cases of COVID-19. IL-6 is produced by immune cells, including T cells and macrophages, and it promotes inflammation and immune activation. IL-6 inhibitors, such as tocilizumab and sarilumab, have been used in clinical trials to reduce inflammation and improve outcomes in COVID-19 patients [27]. IL-10 is an anti-inflammatory cytokine that can regulate the immune response and prevent tissue damage. In COVID-19 patients, IL-10 levels are reduced, leading to dysregulated immune responses and tissue damage. IL-10 has been proposed as a potential therapeutic target for COVID-19, and IL-10 administration has been shown to reduce the inflammation and improve outcomes in animal models of COVID-19 [28].
Overall, interleukins play a critical role in the immune response to viral infections, including COVID-19. Dysregulation of interleukin production can lead to a cytokine storm and tissue damage, contributing to the severity of COVID-19. Interleukin inhibitors have been used in clinical trials to reduce the inflammation and improve outcomes in the COVID-19 patients, and they represent a promising therapeutic approach for COVID-19. However, further research is needed to fully understand the role of interleukins in COVID-19 pathogenesis and to develop the effective therapeutic strategies (Figure 1).
Figure 1.
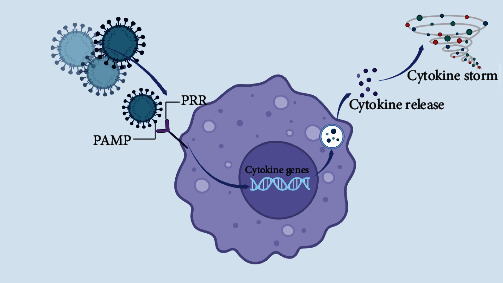
The general mechanism underlying immune system response to SARS-CoV-2 invasion. When the PAMP of the virus attaches to the PRR, the pattern of the virus is detected by immune system. This leads to a signal transduction to the genome of immune cells and as a result, it can activate the cytokine genes. Activation of cytokine genes lead to cytokine release. If the cytokine release happens excessively, the cytokine storm happens.
3. IL-2 Family Overview
IL-2 is a cytokine within the IL-2 cytokine family in which each member contains a four alpha-helix bundle. Other members of this family include IL-4, IL-7, IL-9, IL-15, and IL-21. IL-2 signals via the IL-2 receptor, a complex comprised of three chains named alpha (CD25), beta (CD122), and gamma (CD132). All family members participate in the gamma chain [12]. The IL-2 receptor (IL-2R) subunit binds IL-2 with modest affinity. Due to its short intracellular chain, the interaction between IL-2 and CD25 alone does not result in signal transduction, but it increases IL-2R affinity by hundredfold [29]. T cell signaling depends on the heterodimerization of the subunits of IL-2R. IL-2 may initiate signaling via the intermediate-affinity dimeric CD122/CD132 IL-2R or the high-affinity trimeric CD25/CD122/CD132 IL-2R. Dimeric IL-2R is expressed by memory CD8+ T cells and natural killer (NK) cells, whereas regulatory T cells and activated T cells exhibit large quantities of trimeric IL-2R [30].
T helper 2 (Th2) cells are derived from naïve helper T cells (Th0 cells), and IL-4 is a cytokine that induces this development [31, 32]. Th2 cells undergo a positive feedback loop wherein they create more IL-4 after being activated by the cytokine. Th2 cells, mast cells, basophils, and eosinophils are the primary cells responsible for IL-4 production [33]. One-half of the structure of IL-4 is dominated by a four alpha-helix bundle with a left-handed twist; the helices are antiparallel with two overhand connections, and they fall into a two-stranded antiparallel beta-sheet. IL-4 has a tight, globular fold, stabilized by three disulfide bonds [34].
IL-7 is a hematopoietic growth factor released by bone marrow and thymus stromal cells. It is also generated by keratinocytes, dendritic cells, hepatocytes, neurons, and epithelial cells, but not by normal lymphocytes [35]. The 3D structure of IL-7 was determined by X-ray. The IL-7 receptor is a heterodimer composed of the alpha and gamma subunits of the IL-7 receptor [36]. IL-7 is a cytokine that promotes the growth of B and T cells. IL-7 and the hepatocyte growth factor (HGF) create a heterodimer that acts as a growth factor for pre–pro-B cells. During early T cell development, this cytokine is discovered to be a cofactor for V(D)J rearrangement of the T cell receptor beta (TCR-ß) [37].
IL-9 is generated at varying levels, including NKT cells, mast cells, Th2, Treg, Th17, ILC2, and Th9 cells. Th9 cells are the most critical CD4+ T cells that generate IL-9 [38]. In order to transmit signals, the gamma chain of the IL-2 receptor interacts with the IL-9 receptor, which is a member of the hemopoietin receptor superfamily. Although the Jak-STAT pathway has been the focus of much signal transduction research due to its centrality in several IL-9 bioactivities, the 4PS/IRS2 adaptor protein may also play a crucial role in IL-9 signaling [39]. IL-15 is another cytokine from the IL-2 cytokine family. A combination comprising the IL-2/IL-15 receptor beta chain (CD122) and the typical gamma chain mediates the binding and signaling of IL-15, as it does for IL-2 (gamma-C, CD132) [40]. When confronted with a viral infection, mononuclear phagocytes (and other cells) release IL-15 (es). Naturally occurring killer cells (NKCs), whose primary function is to eliminate virus-infected cells, are stimulated to multiply in response to this cytokine [41].
IL-21 is a cytokine that stimulates the cell division and proliferation in its target cells. IL-21 has significant regulatory effects on immune cells, such as NK and cytotoxic T cells, which may eliminate virally infected or malignant cells [42, 43]. T cells, B cells, and NK cells all have IL-21 receptors (IL-21R) on their surfaces. Similar to cytokine receptors like IL-2R [44], the IL-21 receptor (IL-21R) dimerizes with the common gamma chain (c) to bind IL-21 [45, 46].
Inflammatory signaling pathways can affect the expression of IL-2 cytokines by activating transcription factors that bind to the IL-2 gene promoter and enhance transcription. One such pathway is the NF-κB pathway, which is activated by various stimuli, including pro-inflammatory cytokines, such as IL-1, TNF-α, and IL-6 [47]. Activation of the NF-κB pathway leads to the phosphorylation and degradation of its inhibitor, IκB, and the translocation of NF-κB to the nucleus, where it binds to the promoter region of the IL-2 gene and enhances its transcription [48, 49]. Additionally, other signaling pathways, such as MAPK and PI3K, can also affect the expression of IL-2 cytokines. Activation of these pathways leads to the activation of transcription factors, such as AP-1 and NFAT, which can also bind to the promoter region of the IL-2 gene and enhance its transcription [49].
4. A Brief Description of the Role of the IL-2 Family in Viral Infection
Cytokines play a significant role in viral infection by regulating cell signaling pathways. It has been shown that cytokines can be involved directly in disease pathogenesis [50]. Viruses led to the induction of cytokine genes, which can help viral genome replication. Some cytokines are known as viral inhibitors, especially IL-12 and interferons, which downregulate viral replication. However, it has been demonstrated that cytokines from the IL-2 family, such as IL-4, can increase the pathogenicity of the virus [51]. Research has indicated the remarkable role of IL-2 on HIV-infected T cells. This cytokine was demonstrated to increase the viral replication and its receptors, including CCR5 and CXCR4. Additionally, patients with HIV were shown to have IL-2 and decreased expression of transcription factors which repress viral replication inhibitors [52]. It has been demonstrated that IL-7 could exhibit antiviral properties by targeting CD8+ cells and affecting their function. IL-7 was used as an antiviral treatment in mice infected with viruses. The results showed that IL-7 can lead to a significant proliferation of B and T cells and played a role in viral clearance in mice after the cytokine therapy. It was also shown that IL-7 could upregulate T-cell populations and increase their transportation from the thymus [53]. The role of IL-9 in viral infections was not clearly defined by the previous literature. However, it was shown that IL-9 could modulate CD4+ responses in some viral infections, including respiratory syncytial virus infection [54] and coxsackievirus B3-induced myocarditis [55]. IL-15 has been known for its regulatory role in antiviral responses of NK cells and CD8+ cells in activated immune reactions. The expression of IL-15 is affected by infectious agents.
Then IL-15 can play its regulatory role through modulation of these cells in the infection site, where NK cells and T-cells migrate and expand. IL-15 upregulation also increases the phagocytosis of viral pathogens by inducing proinflammatory factor release and chemotaxis of monocytes and macrophages [41]. IL-21 is a crucial element in the management of chronic viral infections. Mice lacking IL-21 (or IL-21R) that had been chronically infected with LCMV (lymphocytic choriomeningitis virus) were less successful in fighting off the illness.
Furthermore, LCMV-specific CD8+ T cells were depleted to a greater extent in mice with impaired IL-21 signaling, indicating that IL-21 produced by CD4+ T cells is necessary for sustained CD8+ T-cell effector activity and, consequently, for maintaining immunity to resolve persistent viral infection [56]. It has been shown that HIV-specific CD4+ T cells from “HIV controllers” (rare individuals who do not progress to AIDS by controlling the virus replication without treatment) are able to produce significantly more IL-21 than those of progressors. Furthermore, IL-21-generating virus-specific CD8 T cells were also seen in HIV-infected individuals [57].
5. The Role of the IL-2 Family in COVID-19
A deep review of literature in the online databases showed that the exact mechanism of action of the IL-2 cytokine family in the pathogenesis of COVID-19 was not clearly defined. However, there are some evidences which could demonstrate the changes in cytokines with or without an intervention in COVID-19 patients.
Some studies investigated cytokine changes in patients with COVID-19 who suffered from underlying diseases. Maranatha et al. [58] designed a study to identify how TNF-α, TGF- β1, amphiregulin, IL-2, and estimated glomerular filtration rate changed in COVID-19 patients with pulmonary fibrosis. They found that none of the cytokines except TNF-α was associated with COVID-19 severity and pulmonary fibrosis. This study demonstrated that IL-2 did not correlate with COVID-19 severity and pulmonary fibrosis. A similar study by Ghazavi et al. [59] also showed that there was no change in IL-9, a cytokine from IL-2 family, in COVID-19 patients. They showed that only TGF-β can act as a prognostic factor for COVID-19. However other studies showed that TNF-α changes during inflammation can destroy the epithelial tissues [60].
On the other hand, Tappe et al. [61] showed that T-cell immunity (IFN-γ, IL-2, CXCL9, and CXCL10 cytokines) and innate immunity were diminished in patients with COVID-19. As a result, this downregulation could increase the risk of superinfection in these patients by weakening the antifungal immune mechanisms [61]. While Tappe et al's. [61] study shows a downregulation in IL-2 cytokine, other studies represent a different result in other patients with different underlying diseases. In a study by Rodríguez-Morales et al. [62], in two patients with a brain tumor who had COVID-19, the cytokine profile was upregulated. Increased amounts of IL-13, IFN-γ, and IL-2 in these patients disrupted the blood-brain barrier by activating the microglia and an exacerbated immune response.
Other studies investigated the changes in cytokines in patients with COVID-19 without any underlying diseases. In these studies, critical COVID-19 outcomes [63], extended hospitalization and lymphopenia in COVID-19 patients [64], and poor prognosis and disease severity [65] were associated with a significant increase in IL-2 cytokine family profile, including IL-2, IL-4, and IL-15. Perpiñan et al. [66] showed that resistin and IL-15 are predictors of invasive mechanical ventilation in COVID-19 patients. They also mentioned that cytokine storm is a predictor of severity in viral pneumonia in patients with metabolic syndrome. However, there was no association between increased levels of IL-15 and metabolic syndrome. In a similar study, Vaz de Paula et al. [67] showed that cytokine storms caused by SARS-CoV-2 could enhance the alveolar damage and increase the cell proliferation of fibrosis tissue in the lungs. IL-4 was significantly increased in lung autopsies. IL-4 can increase lung damage by activating Th1, Th2, and Th17 responses, and it is the key cytokine in inflammatory response [68].
Previously, the effect of IL-2 on cellular immunity was investigated. It was noted that IL-2/2R could activate JAK1–STAT5 signaling and therefore activate the CD8+ cells [69]. In a study by Shi et al. [70], it was shown that IL-2 was significantly upregulated in the plasma of COVID-19 patients. However, the expression of IL-2R and JAK1–STAT5 pathway was decreased in COVID-19 patients with severe clinical manifestations. This group demonstrated that the inhibition of IL-2R and an increase in IL-2 levels could lead to a downregulation of CD8+ cells. As a result, it can be noted that targeting IL-2 could effectively prevent fatal outcomes of the disease and increase the quality of life of patients with COVID-19 (Table 1, Figure 2).
Table 1.
The role of the IL-2 cytokine family in COVID-19.
| Sample | Cytokine | Changes in COVID-19 patients | Outcome in disease | Reference |
|---|---|---|---|---|
| Seventy-four COVID-19 patients with pulmonary fibrosis | TNF-α, TGF- β1, amphiregulin, IL-2, and EGFR | ↑TNF-α other cytokines no changes | Association with severity of COVID-19 and pulmonary fibrosis | [58] |
|
| ||||
| Twelve COVID-19 patients with Aspergillus fumigatus and Rhizopus arrhizus antigens | IFN-γ, IL-2, CXCL9, CXCL10 | ↓IFN-γ, IL-2, CXCL9, CXCL10 | ↓ T-cell and innate immunity function, weakened anti-mold immune responses | [61] |
|
| ||||
| Two patients with a brain tumor and COVID-19 | IL-13, IFN-γ, and IL-2 | ↑IL-13, IFN-γ, and IL-2 | Activation of the microglia and an exacerbated immune response; disruption of BBB | [62] |
|
| ||||
| One hundred three patients with COVID-19 | IP-10, IL-4, IL-2 | ↑IP-10, IL-4, IL-2 | Critical COVID-19 outcomes | [63] |
|
| ||||
| One hundred thirty COVID-19 patients and 16 control group | IL-6, IL-15 | ↑IL-6, IL-15 | Extended hospitalization and lymphopenia in COVID-19 patients | [64] |
|
| ||||
| One hundred forty-six COVID-19 patients | Resistin, IL-6, IL-8, IL-15, MCP-1 and TNF-α | ↑Resistin, IL-6, IL-8, IL-15, MCP-1 and TNF-α | Prediction of invasive mechanical ventilation | [66] |
|
| ||||
| Sixty-three COVID-19 patients and 33 control group | IFN-γ, IL-5, IL-8, IL-9, IL-17, TGF-β | ↑IFN-γ, TGF-β, IL-17, IL-8 | TGF-β can be a predictive prognosis factor | [59] |
|
| ||||
| Thirty COVID-19 patients and 10 control group | IFN, TNF, IL-21 | ↑ IFN, TNF, IL-21 | Poor prognosis and disease severity | [65] |
|
| ||||
| Fifty-four patients with COVID-19 | IL-2, IL-2R | ↓IL-2R, ↑IL-2 in severe patients and ↓in critical patients | CD8+ T cell and lymphocyte decrease through JAK1–STAT5 in critical patients | [70] |
|
| ||||
| Six COVID-19 patients and 10 control group | IL-4, IL-13, TGF-β | ↑IL-4 | Lung remodeling | [67] |
∗BBB, blood–brain barrier; EGFR, estimated glomerular filtration rate.
Figure 2.
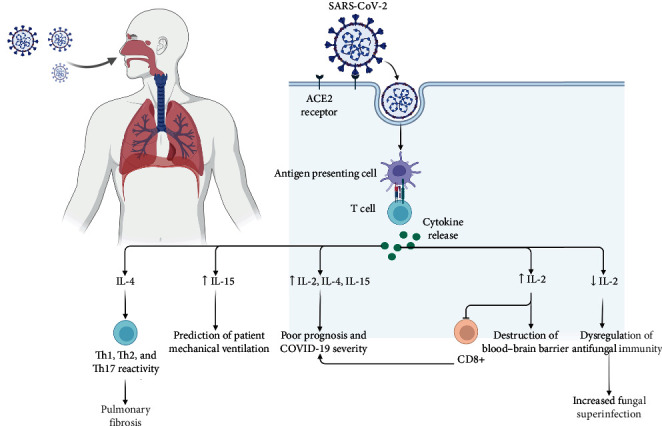
IL-2 cytokine family changes after SARS-CoV-2 infection.
6. IL-2 Family in COVID-19 Infection
There is limited research on the role of IL-2 in different COVID-19 variants. However, some studies have suggested that IL-2 levels may differ in different COVID-19 variants. In Sadhu et al. [71] study, it was showed that IL-9 aggravates SARS-CoV-2 infection. They found that IL-9 level is associated with moderate disease severity [72].
Several preventive and therapeutic strategies have been found for COVID-19 [3]. Targeting interleukins involved in COVID-19 has been a therapeutic option for COVID-19 since the start of the outbreak. Inhibition of these molecules could, most of the time, lead to the prevention of fatal disease [73, 74].
6.1. IL-2 Family in COVID-19 Infection: The Role of Nutritional Supplements
Nutrition supplements have always been a choice for the prevention of diseases. In COVID-19 era, the use of these supplements have increased and the need to investigate the effectiveness of these supplements became vital [75]. Nutrition Bio-shield Superfood (NBS) is a wheat germ-based product that contains alpha-linolenic acid, glutathione, fibers, vitamins, minerals, and phytochemicals. It was previously shown that this dietary supplement could help to increase white blood cells and neutrophil count, which can protect the body against pathogenic agents [76]. Jalilian et al. [77] demonstrated that NBS (2 g/day for 4 week) could effectively decrease the cytokines profile of the patients, including IL-2, IL-6, and TNF-α was decreased significantly. So the inflammatory process of the disease was controlled to some extent. Although this supplement was an effective anti-inflammatory agent, it could not change the overall disease severity. This indicates the adjuvant role of supplements, but not their therapeutic activity alone. Palmitoylethanolamide (PEA) is a novel supplementary, endocannabinoid-like lipid mediator with a background of neuroprotective, immunoprotective, and anti-inflammatory activity [78]. Levagen+ is a supplementary drug based on PEA. In a randomized trial performed by Fessler et al. [79], the effect of Levagen (600 mg twice daily for 4 weeks) was investigated on COVID-19 patients and compared to the control group. The participants' inflammatory cytokine expression (sP-selectin, IL-1β, and IL-2) was evaluated. The results showed that this supplement could significantly prevent the inflammatory process in nonhospitalized patients.
6.2. Targeting IL-2 Family in COVID-19 Infection: The Role of Vaccination
Vaccination is now available for the prevention of COVID-19 around the world. mRNA, viral vector, and inactivated vaccines are examples of the technologies used to develop an efficient vaccine against COVID-19. However, the immunity caused by these vaccines is still questionable [3]. The effects of COVID-19 on the expression of IL-2 family cytokines were evaluated in several studies after different vaccine doses.
While the elevated levels of IL-2 family in COVID-19 is pathogenic and might cause severe inflammation in patients, the elevated IL-2 in individuals who are not currently infected with the virus can boost their cellular immunity by the activation of T cells by IL-2 family. The aim of the clinical researchers on the effects of COVID-19 vaccination is to increase cellular immunity to win the battle against the virus. Third/fourth vaccination with Comirnaty® and Spikevax [80], two doses of COVID-19 mRNA-based vaccines (BNT162b2 (Pfizer-BioNTech) or mRNA-1273 (Moderna)) [81], single dose COVID-19 vaccination with inactivated vaccines (CoronaVac, BBIBP-CorV, or WIBP-CorV) [82], and first and second dose of BNT162b2 mRNA [83] showed a significant in IL-2 family expression, including IL-2 and IL-15 which lead to partial viral immunity in vaccinated individuals. However, in some studies, no changes were seen in the IL-2 family profile, and as a result, boosted immunity after single dose COVID-19 vaccination with BNT162b2 in patients with a rheumatic disease [84]. So the efficacy of a single dose of the COVID-19 vaccine might not be satisfying, and the administration of a booster might help these patients have better immunity against SARS-CoV-2 [85].
6.3. Targeting IL-2 Family in COVID-19 Infection: The Role of Immunotherapy
Targeting severe inflammatory responses to a viral infection is an option to prevent the destructive effects of cytokine storm, disease morbidity, and mortality in patients with COVID-19 [86]. The innate immunity cells, including NK cells, are not specified to identify SARS-CoV-2. Neutralizing antibodies can bind to the subunits of the entering receptor of SARS-CoV-2, named angiotensin-converting enzyme 2 (ACE2). Although the NK cells cannot express ACE2 specified receptors on their cellular surface, finding a way to engineer these cells to express ACE2 might be an excellent option to prevent infection [87]. In a study by Lu et al. [88], NK cells were isolated and chimeric antigen receptor (CAR) NK cells were engineered, and a soluble human IL-15 was added to this complex. Administration of this complex to the A549 cell line and Humanized K18-hACE2 mouse model resulted in enhanced NK cell viral protection and increased expression of inflammatory cytokines including TNF-α and IFN-γ. The mentioned experiment can be used in further clinical investigations in patients with COVID-19 who face severe and drug-resistant disease. Another immunotherapy for COVID-19 with a member of the IL-2 cytokine family, IL-7, was a trial performed by Laterre et al. [89]. IL-7 is a cytokine which plays a role in increasing lymphocytes. It is currently available immunotherapy previously used in oncology. IL-7 therapy can also lead to decreased viral load and prognosis in fatal viral infections [90]. So, in immunosuppressed COVID-19 patients, immunotherapy with IL-7 can restore immunity and result in better clinical outcomes [89] (Table 2).
Table 2.
The role of different interventions in targeting the IL-2 cytokine family in COVID-19.
| Intervention | Samples | Targeted cytokine | Cytokine changes | Clinical/experimental outcome | Reference |
|---|---|---|---|---|---|
| Nutrition Bio-shield Superfood 2 g/day for 4 weeks | Forty-seven patients with COVID-19 (24 intervention; 23 = control) | IL-2, IL-6, IL-17, IFNγ, and TNFα | ↓IL-2, IL-6, and TNF-α | Beneficial anti-inflammatory effect in patients | [77] |
|
| |||||
| 600 mg Levagen + twice daily for 4 weeks | Sixty patients with COVID-19 (30 intervention; 30 control) | sP-selectin, IL-1β, IL-2 | ↓sP-selectin, IL-1β, IL-2 | Anti-inflammatory effects in nonhospitalized patients | [79] |
|
| |||||
| Third/fourth vaccination with Comirnaty® and Spikevax | Thirty-two kidney transplant patients after the booster injection | IFN-γ, IL-2 | ↓IFN-γ, ↑IL-2 | Partly protection of patients with antibody release and IL-2 production | [80] |
|
| |||||
| Two doses of COVID-19 mRNA-based vaccine (BNT162b2 (Pfizer-BioNTech) or mRNA-1273 (Moderna)) | Forty-three MS patients and 34 controls | IFNγ, IL-2 | ↑IFN-γ, IL-2 | Partial adaptive immune response to COVID-19 vaccination | [81] |
|
| |||||
| Single dose COVID-19 vaccination with inactivated vaccines (CoronaVac, BBIBP-CorV, or WIBP-CorV) | Fifty-one COVID-19 recovered subjects and 63 healthy individuals | IFNγ, IL-2, TNF-α | ↑IFN-γ, IL-2, TNF-α | Robust humoral and cellular immune response | [82] |
|
| |||||
| Single dose COVID-19 vaccination with BNT162b2 | Forty patients with rheumatic diseases and 24 healthy patients | IFNγ, IL-2, TNF-α | No difference between two groups in IFNγ, IL-2, TNF-α | No changes in immune response in patients under immunosuppressive treatment | [84] |
|
| |||||
| mACE2-CAR_sIL15 NK cell therapy | A549 cell line and Humanized K18-hACE2 mouse model | IL-15, IFN-γ, TNFα | ↑IL-15, IFN-γ, TNFα | Decreased viral load and prolonged survival | [88] |
|
| |||||
| First and second doses of BNT162b2 mRNA | Sixty-three COVID-19 patients | IFN-γ, IL-15, IP-10/CXCL10 | ↑IFN-γ, IL-15, IP-10/CXCL10 | Effective immune response to the vaccine | [83] |
|
| |||||
| 3–10 µg/kg IL-7 | Twelve COVID-19 patients | TNF-α, IL-6, IL-1β | No changes | Restored lymphocyte count and disease severity | [89] |
∗MS: multiple sclerosis.
7. Deep Insight in the Molecular Mechanisms of IL-2 Family
The Janus Kinase (JAK)/Signal Transducers and Activators of Transcription (STAT) is a signaling pathway enrolled in the SARS-CoV-2 cytokine storm and inflammatory reactions. JAK/STAT pathway is regulated through a cytokine receptor signaling. In cells with no inflammatory activity, STAT proteins with SH2 domain are not active. After the initiation of inflammatory response to a stimulant, STAT proteins bind to phosphortyrosine of the receptor through JAK signaling. The phosphorylated STAT proteins leads to dimerization of STAT. This dimerization helps the STAT proteins to translocate into the nucleus and, as a result, immune-related changes, including apoptosis and cell differentiation occur [2, 91]. When SARS-CoV-2 enters the lung through inspiration of viral particles, the antigen presenting cells (APCs) present the antigen to t helper and cytotoxic cells. Then these cells produce the IL-2 cytokine family. When IL-2 cytokine family, including IL-2, IL-4, IL7, IL-9, IL-15, and IL-21 attach to the IL-2R, the JAK/STAT will be activated. The activation of JAK/STAT pathway leads to an inflammatory response including fibrin formation and lung fibrosis, activation of a cytokine cascade, and fluid leak from the vessels into the alveoli, which in turn causes acute respiratory distress (Figure 3) [92].
Figure 3.
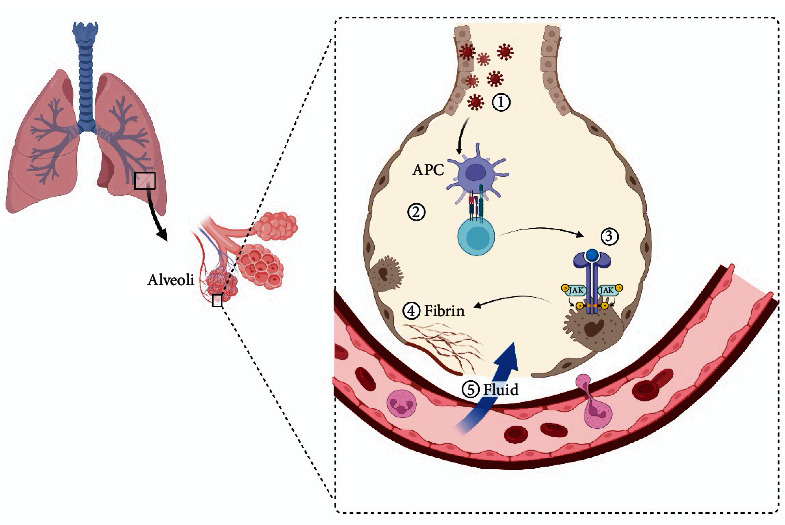
1: Coronavirus infects lung cells; 2: antigen presenting cells (APCs) attach to T helper and T cytotoxic cells, which in turn produce IL-2 cytokine family cytokines, creating a cycle of inflammation that damages the lung cells; 3–5: lung fibrosis can occur through the formation of fibrin. Blood vessels are filled with fluid leading to respiratory failure and patient poor prognosis.
8. The Role of IL-2 in the Diagnosis and Prognosis of COVID-19
Biomarkers are the molecules which are used in diagnosis and prognosis of a disease. There are several factors for diagnosis and prognosis of the COVID-19 [17, 93]. The immunologic response of the patients with COVID-19 was investigated in several literature to show the impact of IL-2 family for predicting the prognosis of the disease. To achieve this goal, the serum levels of these cytokines were compared in patients with severe COVID-19 and nonsevere COVID-19. The results of the studies showed a controversial concept which requires more attention for further research. In a study on 69 patients with prolonged COVID-19 showed that, high IL-2R in serum is correlated with a prolonged disease in COVID-19 patients. The results of this study indicate that neutrophils and T cells are enrolled in the evolution of COVID-19 [94]. On the counter, in a study on 115 patients, the blood sample tests demonstrated that serum IL-2 was lower in patients with severe or moderate COVID-19 than individuals with asymptomatic and mild COVID-19 [95]. A study by Hou et al. [96] also evaluated biomarkers for the early detection of severe COVID-19. On the counter, this study showed that IL-2R is a significant cytokine which needs critical attention for the detection of severe COVID-19 cases and could predict the clinical progress of the patients. An analysis of cytokines Iraqi patients with COVID-19 demonstrated that the serum levels of IL-4 were significantly higher in patients with severe form of COVID-19 [97]. Serum IL-7, a member of IL-2 family was also compared in patients with both severe and nonsevere COVID-19. The results of the study showed that IL-7 high levels is associated with a more severe disease [98].
A systematic review and meta-analysis showed that serum level of IL-2 is indicative of COVID-19 infection compared to the healthy individuals. However, the results of that study showed that IL-2 and IL-4 are not significantly altered in severe and nonsevere COVID-19 patients [99]. Another study evaluated cytokine profile of patients with the severe and nonsevere COVID-19. The results also showed that the level of IL-9 did not differ among controls and COVID-19 patients [59].
COVID-19 has been associated with an increased risk of venous thromboembolism (VTE), which is thought to be related to a complex interplay between inflammatory cytokines, coagulation factors, and endothelial dysfunction. IL-2 may contribute to this process by promoting the activation of both immune cells and platelets, thereby promoting inflammation and thrombosis [100]. It has been demonstrated that some pro-inflammatory cytokines could potentially induce coagulation and lead to thrombosis. IL-2 can activate cytokines which are enrolled in the impairment of anticoagulation system in the intima of the vessels which leads to the induction of coagulation system [101].
9. Conclusions and Perspectives
The results of the literature on the role of the IL-2 cytokine family do not show a singular rule. IL-2 cytokine family can change during SARS-CoV-2 infection under different circumstances. So investigating the association between the expression of IL-2 in different clinical situations is vital.
Some studies suggest that the IL-2 cytokine family rises during the infection and cause severe inflammatory response and cytokine storm. Cytokine storm is a common cause of death in patients with COVID-19 [102]. Many studies suggest that the prevention of cytokine storm can be performed through inhibition of many different cytokines, including IL-6 [103], TNF-α [104], and IL-1 family [11]. These cytokines are shown to be increased in immunocompromised patients and worsen their prognosis. IL-2 family is responsible for severe inflammatory responses in COVID-19, which can similarly affect the prognosis of patients with underlying diseases, including patients with fungal infections, cancer, and metabolic disorders.
In individuals without underlying disease, the upregulation of the IL-2 family shows the clinical outcome of the disease and rises with disease severity. However, some other studies show that these cytokines do not significantly change. IL-2 cytokine family is predominantly upregulated in healthy individuals who had the vaccination, but immunocompromised patients did not show significant changes after a single dose of vaccines, which shows that these patients need booster doses for efficient immunity. IL-2 cytokine family can also be used as immunotherapy agents to prevent the severe outcomes of COVID-19.
The future perspectives on the role of the IL-2 cytokine family in COVID-19 are promising but require further investigation. First, there is a need for more extensive research to investigate the association between the expression of IL-2 in different clinical situations, including patients with underlying diseases, those with severe or mild symptoms, and vaccinated individuals. This will help researchers to identify the specific patient populations that are at-risk of developing severe inflammatory responses and cytokine storms, and develop targeted therapies to prevent such outcomes.
Second, cytokine storm is a common cause of death in COVID-19 patients, and many studies suggest that the prevention of cytokine storm can be performed through inhibition of various cytokines, including the IL-2 family. Therefore, future research should focus on identifying specific inhibitors, including monoclonal antibodies and corticosteroids that can effectively control the upregulation of the IL-2 cytokine family and prevent severe inflammatory responses in COVID-19 patients [105, 106].
Third, the upregulation of the IL-2 cytokine family has been shown to be a clinical outcome of the disease and is associated with disease severity. Therefore, IL-2 cytokine family can be used as a potential biomarker to monitor the disease progression and treatment efficacy.
Finally, IL-2 cytokine family can be used as mmunotherapy agents to prevent the severe outcomes of COVID-19. Future research should focus on developing the IL-2-based immunotherapies that can effectively modulate the immune response and prevent severe inflammatory responses in COVID-19 patients. Overall, the role of the IL-2 cytokine family in COVID-19 is complex and requires further investigation to develop targeted therapies and improve patient outcomes.
Data Availability
All data generated or analyzed during this study are included in this published article.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Authors' Contributions
D N, P K, and F A A contributed in the conceptualization; L G N contributed in the validation; F K contributed in the data curation; L G N, D N, and F A A contributed in writing the original draft preparation; all authors contributed in writing—review and editing. All authors have read and agreed to the published version of the manuscript.
References
- 1.Samieefar N., Boroujeni R. Y., Jamee M., et al. Country quarantine during COVID-19: critical or not? Disaster Medicine and Public Health Preparedness . 2021;15(4):e24–e25. doi: 10.1017/dmp.2020.384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Abdi J., Jian H., Chang H. Role of micro-RNAs in drug resistance of multiple myeloma. Oncotarget . 2016;7(37):60723–60735. doi: 10.18632/oncotarget.11032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tabari M. A. K., Najary S., Khadivi G., Yousefi M. J., Samieefar N., Abdollahimajd F. Oral lesions after COVID-19 vaccination: immune mechanisms and clinical approach. Infectious Medicine . 2022;1(3):171–179. doi: 10.1016/j.imj.2022.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tabari M. A. K., Khoshhal H., Tafazoli A., Khandan M., Bagheri A. Applying computer simulations in battling with COVID-19, using pre-analyzed molecular and chemical data to face the pandemic. Informatics in Medicine Unlocked . 2020;21 doi: 10.1016/j.imu.2020.100458.100458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sánchez-García S., Ruiz-Hornillos J., Bernaola M., et al. Effect of the SARS-CoV-2 pandemic on the control and severity of pediatric asthma. Allergologia et Immunopathologia . 2022;50(1):99–103. doi: 10.15586/aei.v50i1.492. [DOI] [PubMed] [Google Scholar]
- 6.Al-kuraishy H. M., Al-Gareeb A. I., El-Saber Batiha G. The possible role of ursolic acid in Covid-19: a real game changer. Clinical Nutrition ESPEN . 2022;47:414–417. doi: 10.1016/j.clnesp.2021.12.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mehta P., McAuley D. F., Brown M., Sanchez E., Tattersall R. S., Manson J. J. COVID-19: consider cytokine storm syndromes and immunosuppression. The Lancet . 2020;395(10229):1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Al-Kuraishy H. M., Al-Gareeb A. I., Al-Niemi M. S., Al-Buhadily A. K., Al-Harchan N. A., Lugnier C. COVID-19 and phosphodiesterase enzyme type 5 inhibitors. Journal of Microscopy and Ultrastructure . 2020;8(4):141–145. doi: 10.4103/JMAU.JMAU_63_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hojyo S., Uchida M., Tanaka K., et al. How COVID-19 induces cytokine storm with high mortality. Inflammation and Regeneration . 2020;40 doi: 10.1186/s41232-020-00146-3.37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chen R., Sang L., Jiang M., et al. Longitudinal hematologic and immunologic variations associated with the progression of COVID-19 patients in China. The Journal of Allergy and Clinical Immunology . 2020;146(1):89–100. doi: 10.1016/j.jaci.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Makaremi S., Asgarzadeh A., Kianfar H., Mohammadnia A., Asghariazar V., Safarzadeh E. The role of IL-1 family of cytokines and receptors in pathogenesis of COVID-19. Inflammation Research . 2022;71:923–947. doi: 10.1007/s00011-022-01596-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liao W., Lin J.-X., Leonard W. J. IL-2 family cytokines: new insights into the complex roles of IL-2 as a broad regulator of T helper cell differentiation. Current Opinion in Immunology . 2011;23(5):598–604. doi: 10.1016/j.coi.2011.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kai McKinstry K., Alam F., Flores-Malavet V., et al. Memory CD4 T cell-derived IL-2 synergizes with viral infection to exacerbate lung inflammation. PLOS Pathogens . 2019;15(8) doi: 10.1371/journal.ppat.1007989.e1007989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Momtazmanesh S., Samieefar N., Uddin L. Q., et al. Coronavirus Disease-COVID-19 . Springer; 2021. Socialization during the COVID-19 pandemic: the role of social and scientific networks during social distancing; pp. 911–921. [DOI] [PubMed] [Google Scholar]
- 15.Al-Kuraishy H. M., Al-Gareeb A. I. From SARS-CoV to nCoV-2019: ruction and argument. Archives of Clinical Infectious Diseases . 2020;15 doi: 10.5812/archcid.102624.e102624 [DOI] [Google Scholar]
- 16.Tabari M. A. K., Iranpanah A., Bahramsoltani R., Rahimi R. Flavonoids as promising antiviral agents against SARS-CoV-2 infection: a mechanistic review. Molecules . 2021;26(13) doi: 10.3390/molecules26133900.3900 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Deepak Bethineedi L., Baghsheikhi H., Soltani A., et al. Human T2R38 bitter taste receptor expression and COVID-19: from immunity to prognosis. Avicenna Journal of Medical Biotechnology . 2023;15(2):118–123. doi: 10.18502/ajmb.v15i2.12022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kindler E., Thiel V. To sense or not to sense viral RNA—essentials of coronavirus innate immune evasion. Current Opinion in Microbiology . 2014;20:69–75. doi: 10.1016/j.mib.2014.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Torres-Pedraza S., Betancur J. G., Urcuqui-Inchima S. Viral recognition by the innate immune system: the role of pattern recognition receptors. Colombia Médica . 2010;41(4):377–387. doi: 10.25100/cm.v41i4.731. [DOI] [Google Scholar]
- 20.Moriyama K., Nishida O. Targeting cytokines, pathogen-associated molecular patterns, and damage-associated molecular patterns in sepsis via blood purification. International Journal of Molecular Sciences . 2021;22(16) doi: 10.3390/ijms22168882.8882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Paludan S. R., Mogensen T. H. Innate immunological pathways in COVID-19 pathogenesis. Science Immunology . 2022;7(67) doi: 10.1126/sciimmunol.abm5505.eabm5505 [DOI] [PubMed] [Google Scholar]
- 22.Paces J., Strizova Z., Smrz D., Cerny J. COVID-19 and the immune system. Physiological Research . 2020;69:379–388. doi: 10.33549/physiolres.934492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Al-Kuraishy H. M., Al-Gareeb A. I., Mostafa-Hedeab G., et al. Effects of β-blockers on the sympathetic and cytokines storms in COVID-19. Frontiers in Immunology . 2021;12 doi: 10.3389/fimmu.2021.749291.749291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cox R. J., Brokstad K. A. Not just antibodies: B cells and T cells mediate immunity to COVID-19. Nature Reviews Immunology . 2020;20:581–582. doi: 10.1038/s41577-020-00436-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Al-Kuraishy H. M., Al-Gareeb A. I., Alkazmi L., Habotta O. A., Batiha G. E.-S. High-mobility group box 1 (HMGB1) in COVID-19: extrapolation of dangerous liaisons. Inflammopharmacology . 2022;30:811–820. doi: 10.1007/s10787-022-00988-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Huet T., Beaussier H., Voisin O., et al. Anakinra for severe forms of COVID-19: a cohort study. The Lancet Rheumatology . 2020;2(7):e393–e400. doi: 10.1016/S2665-9913(20)30164-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kaye A. G., Siegel R. The efficacy of IL-6 inhibitor tocilizumab in reducing severe COVID-19 mortality: a systematic review. PeerJ . 2020;8 doi: 10.7717/peerj.10322.e10322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lu L., Zhang H., Dauphars D. J., He Y.-W. A potential role of interleukin 10 in COVID-19 pathogenesis. Trends in Immunology . 2021;42(1):3–5. doi: 10.1016/j.it.2020.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang X., Rickert M., Christopher Garcia K. Structure of the quaternary complex of interleukin-2 with its α, ß, and γc receptors. Science . 2005;310(5751):1159–1163. doi: 10.1126/science.1117893. [DOI] [PubMed] [Google Scholar]
- 30.Arenas-Ramirez N., Woytschak J., Boyman O. Interleukin-2: biology, design and application. Trends in immunology . 2015;36(12):763–777. doi: 10.1016/j.it.2015.10.003. [DOI] [PubMed] [Google Scholar]
- 31.Pritam P., Manna S., Sahu A., et al. Eosinophil: a central player in modulating pathological complexity in asthma. Allergologia et Immunopathologia . 2021;49(2):191–207. doi: 10.15586/aei.v49i2.50. [DOI] [PubMed] [Google Scholar]
- 32.Wu J., Chen S., Li X. Correlation between B-cell lymphoma 6 with the balance of T helper-1/2 and severity of allergic rhinitis. Allergologia et Immunopathologia . 2023;51(1):1–8. doi: 10.15586/aei.v51i1.673. [DOI] [PubMed] [Google Scholar]
- 33.Gadani S. P., Cronk J. C., Norris G. T., Kipnis J. IL-4 in the brain: a cytokine to remember. The Journal of Immunology . 2012;189(9):4213–4219. doi: 10.4049/jimmunol.1202246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Walter M. R., Cook W. J., Zhao B. G., et al. Crystal structure of recombinant human interleukin-4. Journal of Biological Chemistry . 1992;267(28):20371–20376. doi: 10.1016/S0021-9258(19)88711-2. [DOI] [PubMed] [Google Scholar]
- 35.Buffière A., Uzan B., Aucagne R., et al. T-cell acute lymphoblastic leukemia displays autocrine production of interleukin-7. Oncogene . 2019;38:7357–7365. doi: 10.1038/s41388-019-0921-4. [DOI] [PubMed] [Google Scholar]
- 36.Noguchi M., Nakamura Y., Russell S. M., et al. Interleukin-2 receptor γ chain: a functional component of the interleukin-7 receptor. Science . 1993;262(5141):1877–1880. doi: 10.1126/science.8266077. [DOI] [PubMed] [Google Scholar]
- 37.Muegge K., Vila M. P., Durum S. K. Interleukin-7: a cofactor for V(D)J rearrangement of the T cell receptor β gene. Science . 1993;261(5117):93–95. doi: 10.1126/science.7686307. [DOI] [PubMed] [Google Scholar]
- 38.Rojas-Zuleta W. G., Sanchez E. IL-9: function, sources, and detection. In: Goswami R., editor. Th9 Cells . Springer; 2017. pp. 21–35. [DOI] [PubMed] [Google Scholar]
- 39.Demoulin J.-B., Renauld J.-C. Interleukin 9 and its receptor: an overview of structure and function. International Reviews of Immunology . 1998;16(3-4):345–364. doi: 10.3109/08830189809043001. [DOI] [PubMed] [Google Scholar]
- 40.Ma A., Koka R., Burkett P. Diverse functions of IL-2, IL-15, and IL-7 in lymphoid homeostasis. Annual Review of Immunology . 2006;24:657–679. doi: 10.1146/annurev.immunol.24.021605.090727. [DOI] [PubMed] [Google Scholar]
- 41.Verbist K. C., Klonowski K. D. Functions of IL-15 in anti-viral immunity: multiplicity and variety. Cytokine . 2012;59(3):467–478. doi: 10.1016/j.cyto.2012.05.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Parrish-Novak J., Dillon S. R., Nelson A., et al. Interleukin 21 and its receptor are involved in NK cell expansion and regulation of lymphocyte function. Nature . 2000;408:57–63. doi: 10.1038/35040504. [DOI] [PubMed] [Google Scholar]
- 43.Parrish-Novak J., Foster D. C., Holly R. D., Clegg C. H. Interleukin-21 and the IL-21 receptor: novel effectors of NK and T cell responses. Journal of Leukocyte Biology . 2002;72(5):856–863. doi: 10.1189/jlb.72.5.856. [DOI] [PubMed] [Google Scholar]
- 44.Ozaki K., Kikly K., Michalovich D., Young P. R., Leonard W. J. Cloning of a type I cytokine receptor most related to the IL-2 receptor β chain. Proceedings of the National Academy of Sciences . 2000;97(21):11439–11444. doi: 10.1073/pnas.200360997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Han Y., Kozel T. R., Zhang M. X., MacGill R. S., Carroll M. C., Cutler J. E. Complement is essential for protection by an IgM and an IgG3 monoclonal antibody against experimental, hematogenously disseminated candidiasis. The Journal of Immunology . 2001;167(3):1550–1557. doi: 10.4049/jimmunol.167.3.1550. [DOI] [PubMed] [Google Scholar]
- 46.Habib T., Senadheera S., Weinberg K., Kaushansky K. The common γ chain (γc) is a required signaling component of the IL-21 receptor and supports IL-21-induced cell proliferation via JAK3. Biochemistry . 2002;41(27):8725–8731. doi: 10.1021/bi0202023. [DOI] [PubMed] [Google Scholar]
- 47.Liu T., Zhang L., Joo D., Sun S.-C. NF-κB signaling in inflammation. Signal Transduction and Targeted Therapy . 2017;2 doi: 10.1038/sigtrans.2017.23.17023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Arima N., Kuziel W. A., Grdina T. A., Greene W. C. IL-2-induced signal transduction involves the activation of nuclear NF-kappa B expression. The Journal of Immunology . 1992;149(1):83–91. doi: 10.4049/jimmunol.149.1.83. [DOI] [PubMed] [Google Scholar]
- 49.Pahl H. L. Activators and target genes of Rel/NF-κB transcription factors. Oncogene . 1999;18:6853–6866. doi: 10.1038/sj.onc.1203239. [DOI] [PubMed] [Google Scholar]
- 50.Mao K., Li D., Liu J., Sun J., Zhang R. Investigation of serum IL-12, IL-16, and IL-17A as diagnostic biomarkers in children with cow’s milk protein allergy. Allergologia et Immunopathologia . 2022;50(5):162–168. doi: 10.15586/aei.v50i5.592. [DOI] [PubMed] [Google Scholar]
- 51.Sanapala P., Pola S. Role of cytokines in infectious viral disease. In: Bramhachari P., editor. Dynamics of Immune Activation in Viral Diseases . Singapore: Springer; 2020. pp. 81–98. [DOI] [Google Scholar]
- 52.Bahr G. M., Darcissac E. C., Mouton Y. Discordant effects of interleukin-2 on viral and immune parameters in human immunodeficiency virus-1-infected monocyte-derived mature dendritic cells. Clinical and Experimental Immunology . 2003;132(2):289–296. doi: 10.1046/j.1365-2249.2003.02143.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bordon Y. IL-7 goes antiviral. Nature Reviews Immunology . 2011;11:159–159. doi: 10.1038/nri2949. [DOI] [PubMed] [Google Scholar]
- 54.Dodd J. S., Lum E., Goulding J., Muir R., Van Snick J., Openshaw P. J. M. IL-9 regulates pathology during primary and memory responses to respiratory syncytial virus infection. The Journal of Immunology . 2009;183(11):7006–7013. doi: 10.4049/jimmunol.0900085. [DOI] [PubMed] [Google Scholar]
- 55.Yu M., Long Q., Li H.-H., et al. IL-9 inhibits viral replication in coxsackievirus b3-induced myocarditis. Frontiers in Immunology . 2016;7 doi: 10.3389/fimmu.2016.00409.409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Johnson L. D. S., Jameson S. C. A chronic need for IL-21. Science . 2009;324(5934):1525–1526. doi: 10.1126/science.1176487. [DOI] [PubMed] [Google Scholar]
- 57.Chevalier M. F., Jülg B., Pyo A., et al. HIV-1-specific interleukin-21+ CD4+ T cell responses contribute to durable viral control through the modulation of HIV-specific CD8+ T cell function. Journal of Virology . 2011;85(2):733–741. doi: 10.1128/jvi.02030-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Maranatha D., Hasan H., Bakhtiar A., Widyoningroem A., Aryati Association of TNF-α, TGF-β1, amphiregulin, IL-2, and EGFR with pulmonary fibrosis in COVID-19. Journal of Infection and Public Health . 2022;15(10):1072–1075. doi: 10.1016/j.jiph.2022.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ghazavi A., Ganji A., Keshavarzian N., Rabiemajd S., Mosayebi G. Cytokine profile and disease severity in patients with COVID-19. Cytokine . 2021;137 doi: 10.1016/j.cyto.2020.155323.155323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fan K., Jin L., Yu S. Roles of regulatory B cells in the pathogenesis of allergic rhinitis. Allergologia et Immunopathologia . 2022;50(5):7–15. doi: 10.15586/aei.v50i5.615. [DOI] [PubMed] [Google Scholar]
- 61.Tappe B., Lauruschkat C. D., Strobel L., et al. COVID-19 patients share common, corticosteroid-independent features of impaired host immunity to pathogenic molds. Frontiers in Immunology . 2022;13 doi: 10.3389/fimmu.2022.954985.954985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Rodríguez-Morales J., Guartazaca-Guerrero S., Rizo-Téllez S. A., et al. Blood–brain barrier damage is pivotal for SARS-CoV-2 infection to the central nervous system. Experimental Neurobiology . 2022;31(4):270–276. doi: 10.5607/en21049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Torres-Ruiz J., Lomelín-Gascón J., Vargas-Castro A. S., et al. Clinical and immunological features associated to the development of a sustained immune humoral response in COVID-19 patients: results from a cohort study. Frontiers in Immunology . 2022;13 doi: 10.3389/fimmu.2022.943563.943563 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Singh A. K., Kasarpalkar N., Bhowmick S., et al. Opposing roles for sMAdCAM and IL-15 in COVID-19 associated cellular immune pathology. Journal of Leukocyte Biology . 2022;111(6):1287–1295. doi: 10.1002/JLB.3COVBCR0621-300R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Khan M., Mathew B. J., Gupta P., et al. Gut dysbiosis and IL-21 response in patients with severe COVID-19. Microorganisms . 2021;9(6) doi: 10.3390/microorganisms9061292.1292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Perpiñan C., Bertran L., Terra X., et al. Resistin and IL-15 as predictors of invasive mechanical ventilation in COVID-19 pneumonia irrespective of the presence of obesity and metabolic syndrome. Journal of Personalized Medicine . 2022;12(3) doi: 10.3390/jpm12030391.391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.de Paula C. B. V., de Azevedo M. L. V., Nagashima S., et al. IL-4/IL-13 remodeling pathway of COVID-19 lung injury. Scientific Reports . 2020;10 doi: 10.1038/s41598-020-75659-5.18689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nepolo E. P., Nkambule B. B., Dludla P. V., Ndevahoma F., Nyambuya T. M. Association between the type of allergen and T-helper 2 mediated inflammation in allergic reactions: a systematic review and a meta-analysis. Allergologia et Immunopathologia . 2022;50(1):37–50. doi: 10.15586/aei.v50i1.470. [DOI] [PubMed] [Google Scholar]
- 69.Fu X., Xu M., Song Y., et al. Enhanced interaction between SEC2 mutant and TCR Vβ induces MHC II–independent activation of T cells via PKCθ/NF-κB and IL-2R/STAT5 signaling pathways. Journal of Biological Chemistry . 2018;293(51):19771–19784. doi: 10.1074/jbc.RA118.003668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Shi H., Wang W., Yin J., et al. The inhibition of IL-2/IL-2R gives rise to CD8+ T cell and lymphocyte decrease through JAK1-STAT5 in critical patients with COVID-19 pneumonia. Cell Death & Disease . 2020;11 doi: 10.1038/s41419-020-2636-4.429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sadhu S., Dalal R., Dandotiya J., et al. IL-9 aggravates SARS-CoV-2 infection and exacerbates associated airway inflammation. Nature Communications . 2023;14 doi: 10.1038/s41467-023-39815-5.4060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Korobova Z. R., Arsentieva N. A., Liubimova N. E., et al. Cytokine profiling in different SARS-CoV-2 genetic variants. International Journal of Molecular Sciences . 2022;23(22) doi: 10.3390/ijms232214146.14146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Dzobo K., Chiririwa H., Dandara C., Dzobo W. Coronavirus disease-2019 treatment strategies targeting interleukin-6 signaling and herbal medicine. OMICS: A Journal of Integrative Biology . 2021;25(1):13–22. doi: 10.1089/omi.2020.0122. [DOI] [PubMed] [Google Scholar]
- 74.Al-kuraishy H. M., Al-Gareeb A. I., Qusty N., Alexiou A., Batiha G. E.-S. Impact of sitagliptin on non-diabetic COVID-19 patients. Current Molecular Pharmacology . 2022;15(4):683–692. doi: 10.2174/1874467214666210902115650. [DOI] [PubMed] [Google Scholar]
- 75.Costagliola G., Spada E., Comberiati P., Peroni D. G. Could nutritional supplements act as therapeutic adjuvants in COVID-19? Italian Journal of Pediatrics . 2021;47 doi: 10.1186/s13052-021-00990-0.32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Bayat A., Khalkhali A., Mahjoub A. R. Nutrition bio-shield superfood: healthy and live herbal supplement for immune system enhancement. International Journal of Nutrition and Food Engineering . 2014;15(1):6–9. [Google Scholar]
- 77.Jalilian F. A., Keshavarz G., Khazaei S., et al. The effects of nutrition bio-shield superfood powder on immune system function: a clinical trial study among patients with COVID-19. Frontiers in Immunology . 2022;13 doi: 10.3389/fimmu.2022.919402.919402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Clayton P., Hill M., Bogoda N., Subah S., Venkatesh R. Palmitoylethanolamide: a natural compound for health management. International Journal of Molecular Sciences . 2021;22(10) doi: 10.3390/ijms22105305.5305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Fessler S. N., Liu L., Chang Y., Yip T., Johnston C. S. Palmitoylethanolamide reduces proinflammatory markers in unvaccinated adults recently diagnosed with COVID-19: a randomized controlled trial. The Journal of Nutrition . 2022;152(10):2218–2226. doi: 10.1093/jn/nxac154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Thümmler L., Gäckler A., Bormann M., et al. Cellular and humoral immunity against different SARS-CoV-2 variants is detectable but reduced in vaccinated kidney transplant patients. Vaccines . 2022;10(8) doi: 10.3390/vaccines10081348.1348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Alfonso-Dunn R., Lin J., Kirschner V., et al. Strong T-cell activation in response to COVID-19 vaccination in multiple sclerosis patients receiving B-cell depleting therapies. Frontiers in Immunology . 2022;13 doi: 10.3389/fimmu.2022.926318.926318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Liang B., Xiang T., Wang H., et al. Robust humoral and cellular immune responses in long-term convalescent COVID-19 individuals following one-dose SARS-CoV-2 inactivated vaccination. Frontiers in Immunology . 2022;13 doi: 10.3389/fimmu.2022.966098.966098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bergamaschi C., Terpos E., Rosati M., et al. Systemic IL-15, IFN-γ, and IP-10/CXCL10 signature associated with effective immune response to SARS-CoV-2 in BNT162b2 mRNA vaccine recipients. Cell Reports . 2021;36(6) doi: 10.1016/j.celrep.2021.109504.109504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Udaondo C., Cámara C., Berenguel L. M., et al. Humoral and cellular immune response to mRNA SARS-CoV-2 BNT162b2 vaccine in adolescents with rheumatic diseases. Pediatric Rheumatology . 2022;20 doi: 10.1186/s12969-022-00724-4.64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lee A. R. Y. B., Wong S. Y., Chai L. Y. A., et al. Efficacy of COVID-19 vaccines in immunocompromised patients: systematic review and meta-analysis. BMJ . 2022;376 doi: 10.1136/bmj-2021-068632.e068632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.van de Veerdonk F. L., Giamarellos-Bourboulis E., Pickkers P., et al. A guide to immunotherapy for COVID-19. Nature Medicine . 2022;28:39–50. doi: 10.1038/s41591-021-01643-9. [DOI] [PubMed] [Google Scholar]
- 87.Brouwer P. J. M., Caniels T. G., van der Straten K., et al. Potent neutralizing antibodies from COVID-19 patients define multiple targets of vulnerability. Science . 2020;369(6504):643–650. doi: 10.1126/science.abc5902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lu T., Ma R., Dong W., et al. Off-the-shelf CAR natural killer cells secreting IL-15 target spike in treating COVID-19. Nature Communications . 2022;13 doi: 10.1038/s41467-022-30216-8.2576 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Laterre P. F., François B., Collienne C., et al. Association of Interleukin 7 immunotherapy with lymphocyte counts among patients with severe coronavirus disease 2019 (COVID-19) JAMA Network Open . 2020;3(7) doi: 10.1001/jamanetworkopen.2020.16485.e2016485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Barata J. T., Durum S. K., Seddon B. Flip the coin: IL-7 and IL-7R in health and disease. Nature Immunology . 2019;20:1584–1593. doi: 10.1038/s41590-019-0479-x. [DOI] [PubMed] [Google Scholar]
- 91.Satarker S., Tom A. A., Shaji R. A., Alosious A., Luvis M., Nampoothiri M. JAK-STAT pathway inhibition and their implications in COVID-19 therapy. Postgraduate Medicine . 2021;133(5):489–507. doi: 10.1080/00325481.2020.1855921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Bhaskar S., Sinha A., Banach M., et al. Cytokine storm in COVID-19—immunopathological mechanisms, clinical considerations, and therapeutic approaches: the REPROGRAM consortium position paper. Frontiers in Immunology . 2020;11 doi: 10.3389/fimmu.2020.01648.1648 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Babalghith A. O., Al-Kuraishy H. M., Al-Gareeb A. I., et al. The potential role of growth differentiation factor 15 in COVID-19: a corollary subjective effect or not? Diagnostics . 2022;12(9) doi: 10.3390/diagnostics12092051.2051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Ma A., Zhang L., Ye X., et al. High levels of circulating IL-8 and soluble IL-2R are associated with prolonged illness in patients with severe COVID-19. Frontiers in Immunology . 2021;12 doi: 10.3389/fimmu.2021.626235.626235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tjan L. H., Furukawa K., Nagano T., et al. Early differences in cytokine production by severity of coronavirus disease 2019. The Journal of Infectious Diseases . 2021;223(7):1145–1149. doi: 10.1093/infdis/jiab005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Hou H., Zhang B., Huang H., et al. Using IL-2R/lymphocytes for predicting the clinical progression of patients with COVID-19. Clinical and Experimental Immunology . 2020;201(1):76–84. doi: 10.1111/cei.13450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Merza M. Y., Hwaiz R. A., Hamad B. K., Mohammad K. A., Hama H. A., Karim A. Y. Analysis of cytokines in SARS-CoV-2 or COVID-19 patients in Erbil city, Kurdistan Region of Iraq. PLOS ONE . 2021;16(4) doi: 10.1371/journal.pone.0250330.e0250330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Chi Y., Ge Y., Wu B., et al. Serum cytokine and chemokine profile in relation to the severity of coronavirus disease 2019 in China. The Journal of Infectious Diseases . 2020;222(5):746–754. doi: 10.1093/infdis/jiaa363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Chang Y., Bai M., You Q. Associations between serum interleukins (IL-1β, IL-2, IL-4, IL-6, IL-8, and IL-10) and disease severity of COVID-19: a systematic review and meta-analysis. BioMed Research International . 2022;2022:15. doi: 10.1155/2022/2755246.2755246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Sarfraz A., Sarfraz Z., Razzack A. A., Patel G., Sarfraz M. Venous thromboembolism, corticosteroids and COVID-19: a systematic review and meta-analysis. Clinical and Applied Thrombosis/Hemostasis . 2021;27 doi: 10.1177/1076029621993573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zhou X., Cheng Z., Hu Y. COVID-19 and venous thromboembolism: from pathological mechanisms to clinical management. Journal of Personalized Medicine . 2021;11(12) doi: 10.3390/jpm11121328.1328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hu B., Huang S., Yin L. The cytokine storm and COVID-19. Journal of Medical Virology . 2021;93(1):250–256. doi: 10.1002/jmv.26232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Du P., Geng J., Wang F., Chen X., Huang Z., Wang Y. Role of IL-6 inhibitor in treatment of COVID-19-related cytokine release syndrome. International Journal of Medical Sciences . 2021;18(6):1356–1362. doi: 10.7150/ijms.53564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Moradian N., Gouravani M., Salehi M. A., et al. Cytokine release syndrome: inhibition of pro-inflammatory cytokines as a solution for reducing COVID-19 mortality. European Cytokine Network . 2020;31(3):81–93. doi: 10.1684/ecn.2020.0451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.El-Saber Batiha G., Al-Gareeb A. I., Saad H. M., Al-Kuraishy H. M. COVID-19 and corticosteroids: a narrative review. Inflammopharmacology . 2022;30:1189–1205. doi: 10.1007/s10787-022-00987-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Tabari M. A. K., Khandan M., Mirjalili R., et al. Corticosteroids therapy for COVID-19-induced hyposmia and anosmia. B-ENT . 2022;18(4):284–290. doi: 10.5152/B-ENT.2022.21792. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.


