Abstract
Purpose
Higher dairy consumption is associated with a lower risk of colorectal cancer (CRC), but no studies thus far have investigated its relation with recurrence in CRC. Few studies have investigated total dairy in relation to mortality in CRC, and yielded inconsistent results.
Methods
In this prospective cohort study, people newly diagnosed with stage I-III CRC filled out a food frequency questionnaire at diagnosis (n = 1812) and six months after diagnosis (n = 1672). We examined associations between pre- and post-diagnostic intake of total dairy, low-fat dairy, high-fat dairy, milk, yoghurt, and cheese with recurrence and all-cause mortality using multivariable Cox proportional hazard models and restricted cubic splines (RCS).
Results
A total of 176 recurrences and 301 deaths occurred during a median follow-up of 3.0 and 5.9 years, respectively. Before diagnosis, a higher low-fat dairy intake was associated with a lower risk of recurrence (HRQ4vsQ1: 0.42, 95% CI 0.26–0.67; PRCS: 0.008) and all-cause mortality (HRQ4vsQ1: 0.58, 95% CI 0.41–0.81; PRCS < 0.001), whereas a higher high-fat dairy consumption tended to be associated with an increased all-cause mortality risk (HRQ4vsQ1: 1.41, 95% CI 0.98–2.01; PRCS: 0.030). After diagnosis, only the associations between low- and high-fat dairy in relation to all-cause mortality remained.
Conclusions
This study demonstrated that higher pre- and post-diagnostic intakes of low-fat dairy were associated with a reduced all-cause mortality risk in people with stage I-III CRC, whereas higher intakes of high-fat dairy were associated with an increased all-cause mortality risk. Also, a higher pre-diagnostic low-fat dairy intake was associated with a reduced risk of recurrence.
Trial registration
Clinical Trials.gov identifier: NCT03191110.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00394-023-03201-0.
Keywords: Low-fat dairy, High-fat dairy, Yoghurt, Colorectal cancer, Recurrence, All-cause mortality
Introduction
Evidence from observational studies suggests that dairy consumption decreases the risk of colorectal cancer (CRC) [1, 2]. Contrasting the large body of evidence and the proposed biological mechanisms [3, 4] available on dairy consumption and CRC risk, a scarcity of studies has been published on the role of dairy intake in CRC prognosis, such as cancer recurrence and all-cause mortality.
For total dairy, two large prospective cohort studies and one pooled analysis of prospective cohort studies observed no association between pre-diagnostic [5, 6] or post-diagnostic [6, 7] dairy intake and CRC-specific or all-cause mortality. However, when stratifying for fat content, one pooled analysis including 1753 individuals with CRC showed that a higher post-diagnostic intake of low-fat dairy products was associated with a reduced risk of all-cause mortality, whereas a higher post-diagnostic intake of high-fat dairy products was associated with an increased risk of all-cause mortality [7]. The authors speculated that potentially beneficial dairy components may be counteracted by fat or fat-related components of dairy [7], which may explain previously observed null associations for total dairy intake with all-cause mortality [5, 6]. We therefore hypothesise that low-fat dairy intake is associated with a reduced risk of all-cause mortality, and that high-fat dairy intake is associated with an increased risk of all-cause mortality.
Inherent to their unique nutritional compositions and in line with existing literature on CRC risk, different dairy products, such as milk, yoghurt, and cheese, are proposed to have a differential influence on CRC prognosis [1, 2, 8–10]. So far, not enough studies have been performed to draw firm conclusions on the association between specific dairy product intake and mortality in CRC. To the best of our knowledge, it is yet unknown whether dairy intake before or after diagnosis is associated with cancer recurrence in people with CRC.
Therefore, we investigated the relation between pre- and post-diagnostic intake of dairy (total, low-fat, high-fat) and specific dairy products (milk, yoghurt, cheese) with cancer recurrence and all-cause mortality in a prospective cohort of newly-diagnosed males and females with stage I-III CRC.
Materials and methods
Study cohort
For these analyses we used data from adults with newly diagnosed CRC who participated in the ‘COlorectal cancer: Longitudinal, Observational study on Nutritional and lifestyle factors that influence colorectal tumour cancer recurrence, survival and quality of life’ (COLON) study (NCT03191110, ClinicalTrials.gov). Participants were recruited from 11 hospitals in the Netherlands between August 2010 and February 2020, and had no hereditary CRC syndrome, a history of CRC or inflammatory bowel disease [11]. Details of this study have been described previously [11]. For the current analyses, we excluded some participants (Fig. 1), among which people diagnosed with stage IV disease (n = 150), as we expect the potential role of lifestyle to be very limited in the poor prognosis of these persons. The final analytical sample sizes were as follows: 1812 (all-cause mortality) and 1341 (recurrence) for pre-diagnostic models, and 1672 (all-cause mortality) and 1236 (recurrence) for post-diagnostic models (Fig. 1). Three participants were lost to follow-up due to moving abroad. The COLON study was approved by a medical ethics committee (region Arnhem-Nijmegen, 2009–349), and all study participants provided written informed consent.
Fig. 1.
Flowchart representing participant selection for the data analyses
Dietary assessment
Upon diagnosis, participants filled out a semi-quantitative food frequency questionnaire (FFQ) of 204 items [12, 13], reflecting dietary intake in the month prior to diagnosis. The exact date of diagnosis was not uniformly available, but in practice participants were included in the study by the health care practitioner only a few days after the diagnosis. The median time between this date of inclusion and completion of the first FFQ was 5 days (IQR: 3–10). At six months after diagnosis participants also completed the FFQ, reflecting dietary intake in the month prior to filling out the FFQ. Dairy product intakes and energy intakes per day were calculated, based on frequency of intake and portion sizes and using the online Dutch Food Composition Table [14], and categorized into total, low-fat and high-fat dairy. Total dairy comprised of all dairy products included in low- and high-fat dairy. Low-fat dairy included low-fat or skimmed versions of milk, yoghurt, custard, and soft curd cheese, and buttermilk. High-fat dairy included whole and condensed milk, full-fat versions of yoghurt, custard and soft curd cheese, all other cheeses, ice cream, whipped cream, and butter. Total milk included all plain and sweetened milk, regardless of fat content. Ready-made breakfast drinks were not included, because not all ready-made breakfast drinks on the Dutch market contain dairy. Total yoghurt included all plain and sweetened yoghurt, yoghurt drinks and soft curd cheese, regardless of fat content. Total cheese included all hard and soft cheese, except for soft curd cheese. Intake of dairy types and products was adjusted for total energy intake using the energy residual method [15]. Further assessment of individual food items, such as skimmed milk, low-fat milk and high-fat milk, was not possible due to low median intakes and a low variation in intake. To improve interpretability, the predicted dairy intake at the median total energy intake was added to individual residuals.
Study outcomes
Recurrence was defined as a local or regional cancer recurrence or a distant metastasis diagnosed after initial tumour resection. Recurrence data were requested from the Netherlands Cancer Registry via the Netherlands Comprehensive Cancer Organisation (IKNL), most recently updated in February 2018. All-cause mortality data were obtained from the municipal registrations (BRP), most recently updated in September 2021.
For recurrence, follow-up time was calculated starting from date of surgery until date of recurrence, until date of death, until the date recurrence status was updated, or until lost to follow-up, whichever came first. Follow-up time for mortality was defined as starting from date of surgery until date of death, until the last date mortality status was updated, or until lost to follow-up, whichever came first. If date of surgery was unavailable (n = 71), date of filling out the questionnaires was used. For post-diagnostic analyses, follow-up time was calculated starting from date of filling out the post-diagnostic questionnaires.
Assessment of covariates
At diagnosis and six months after diagnosis, participants filled out a general questionnaire on demographics, anthropometrics, and lifestyle habits, including questions about age (continuous), sex (male/female), education (low, medium, high), body weight (kg), height (cm), smoking status (current, former, never), and calcium and vitamin D supplement use (including multivitamins, yes/no). Physical activity was assessed using the Short QUestionnaire to ASsess Health-enhancing physical activity (SQUASH) [16]. Moderate-to-vigorous physical activity (hours/week) included all activities with a metabolic equivalent value ≥ 3 [17]. Clinical data, such as disease stage (I-III), tumour location (proximal colon (caecum to the transverse colon), distal colon (splenic flexure to the sigmoid colon), rectum (rectosigmoid junction and rectum)), American Society of Anesthesiologists (ASA) classification (I-IV) and type of treatment (only surgery, surgery and chemotherapy, surgery and radiotherapy, surgery and chemotherapy and radiotherapy, no or unknown surgery, unknown treatment), were collected via the Dutch ColoRectal Audit [18].
To assess possible confounding by other dietary factors previously associated with CRC risk [2], we also calculated total dietary intake of wholegrains, red meat, processed meat, dietary fiber, and alcohol, as defined in [2].
Statistical analysis
Population characteristics are presented as medians [interquartile range (IQR)] or numbers (percentage). Sex-specific quartiles of pre- and post-diagnostic dairy intake were calculated.
Cox proportional hazards regression analyses were used to calculate Hazard Ratios (HR) and 95% Confidence Intervals (95%CI) for the associations between pre- and post-diagnostic dairy intake, and recurrence and all-cause mortality. No violation of the proportionality assumption for the Cox models was observed when visually inspecting log–log curves. For categorical analyses, the lowest quartile was used as the reference category. Ptrend values were computed over quartiles of dairy intake using the medians of the corresponding quartiles. For continuous analyses increments were 100 g/day for total dairy, low-fat dairy, high-fat dairy, milk, and yoghurt, and 30 g/day for cheese. We first created a crude model with the exposure and outcome variables, that was adjusted for age and sex. Then, potential confounders were added to the model one by one, and were included when they changed the HR with > 10%. Based on literature, the following covariates were evaluated: disease stage, primary tumour location, BMI, education level, smoking status, moderate-to-vigorous physical activity, use of calcium supplements, use of vitamin D supplements, and total dietary intake of wholegrains, dietary fiber, red meat, processed meat and alcohol. Additionally, for the post-diagnostic dairy intake analyses, having a stoma, type of treatment, and BMI, smoking status, supplement use and dietary intake at 6 months post-diagnosis were evaluated for confounding. Finally, the adjusted model included age, sex, stage of disease and total energy intake (as part of the energy adjustment method [15]).
We also evaluated restricted cubic splines (RCS) to study linearity of the associations between dairy exposures and CRC prognosis, using the adjusted model. Knots were placed at the 5th, 50th, and 95th percentiles, and the graphs were truncated at the 1st and 99th percentile. The median intake of the first quartile of each exposure was used as the reference.
For linear associations, HRs are displayed continuously as well as in quartiles with p for trend, while for non-linear associations, only quartiles of dairy intake are displayed. All associations are visualized in plots.
Statistical analyses were performed using R Statistical Software (version 4.0.5). P-values below 0.05 and 95% confidence intervals not containing 1 were considered statistically significant.
Results
Population characteristics
Participants were on average 66 years (IQR: 61–72) at CRC diagnosis, and 37% was female (Table 1). Median BMI was 26.2 kg/m2 (IQR: 24.0–28.7) and 10% of participants were current smokers. Furthermore, 66% had a tumour located in the colon, and stage III (43%) was more common than stage I (26%) or stage II (27%) (Table 1).
Table 1.
Population characteristics of people with colorectal cancer stratified by sex-specific quartiles of pre-diagnostic energy-adjusted intake of total dairy, low-fat dairy and high-fat dairy
| Characteristics | Total population (n = 1812) | Quartile of energy-adjusted dairy intake | |||||
|---|---|---|---|---|---|---|---|
| Total dairy | Low-fat dairy | High-fat dairy | |||||
| Q1 (n = 454) | Q4 (n = 454) | Q1 (n = 454) | Q4 (n = 454) | Q1 (n = 454) | Q4 (n = 454) | ||
| Age, years, median [IQR] | 66 [61–72] | 64 [59–70] | 68 [63–74] | 66 [61–71] | 67 [61–74] | 64 [59–69] | 67 [63–73] |
| Female sex, No. (%) | 666 (36.8) | 167 (36.8) | 167 (36.8) | 167 (36.8) | 167 (36.8) | 167 (36.8) | 167 (36.8) |
| BMI, kg/m2, median [IQR] | 26.2 [24.0–28.7] | 26.3 [24.1–28.8] | 26.2 [24.0–28.7] | 25.8 [23.7–28.3] | 26.2 [24.1–28.7] | 25.9 [23.9–28.4] | 25.4 [23.2–28.1] |
| Waist-hip-ratio, median [IQR]a | 0.95 [0.90–1.00] | 0.95 [0.90–1.00] | 0.95 [0.90–1.00] | 0.95 [0.90–1.00] | 0.95 [0.90–1.00] | 0.95 [0.89–1.01] | 0.94 [0.90–1.00] |
| Level of education, No. (%)b | |||||||
| Low | 763 (42.1) | 177 (39.0) | 203 (44.7) | 185 (40.7) | 182 (40.1) | 166 (36.6) | 221 (48.7) |
| Medium | 454 (25.1) | 120 (26.4) | 112 (24.7) | 118 (26.0) | 111 (24.4) | 124 (27.3) | 105 (23.1) |
| High | 591 (32.6) | 156 (34.4) | 136 (30.0) | 150 (33.0) | 159 (35.0) | 163 (35.9) | 125 (27.5) |
| Unknown | 4 (0.2) | 1 (0.2) | 3 (0.7) | 1 (0.2) | 2 (0.4) | 1 (0.2) | 3 (0.7) |
| Smoking status, No. (%) | |||||||
| Current | 184 (10.2) | 65 (14.3) | 40 (8.8) | 69 (15.2) | 33 (7.3) | 41 (9.0) | 57 (12.6) |
| Former | 1057 (58.3) | 263 (57.9) | 265 (58.4) | 253 (55.7) | 261 (57.5) | 269 (59.3) | 254 (55.9) |
| Never | 566 (31.2) | 125 (27.5) | 147 (32.4) | 131 (28.9) | 158 (34.8) | 143 (31.5) | 141 (31.1) |
| Unknown | 5 (0.3) | 1 (0.2) | 2 (0.4) | 1 (0.2) | 2 (0.4) | 1 (0.2) | 2 (0.4) |
| Moderate-to-vigorous physical activity, hours/week, median [IQR] c | 11 [6–20] | 11 [6–20] | 12 [5–21] | 11 [6–20] | 12 [6–19] | 12 [6–20] | 11 [5–19] |
| Calcium supplement use, No. (%)d | 313 (17.3) | 92 (20.3) | 67 (14.8) | 83 (18.3) | 82 (18.1) | 79 (17.4) | 64 (14.1) |
| Vitamin D supplement use, No. (%) | 482 (26.6) | 119 (26.2) | 122 (26.9) | 114 (25.1) | 139 (30.6) | 132 (29.1) | 107 (23.6) |
| Location of the tumor, No. (%)e | |||||||
| Proximal colon | 549 (30.3) | 133 (29.3) | 149 (32.8) | 132 (29.1) | 133 (29.3) | 127 (28.0) | 149 (32.8) |
| Distal colon | 650 (35.9) | 164 (36.1) | 164 (36.1) | 170 (37.4) | 179 (39.4) | 173 (38.1) | 142 (31.3) |
| Rectum | 552 (30.5) | 143 (31.5) | 130 (28.6) | 136 (30.0) | 128 (28.2) | 133 (29.3) | 152 (33.5) |
| Unknown | 61 (3.4) | 14 (3.1) | 11 (2.4) | 16 (3.5) | 14 (3.1) | 21 (4.6) | 11 (2.4) |
| Disease stage, No. (%) | |||||||
| I | 471 (26.0) | 128 (28.2) | 132 (29.1) | 123 (27.1) | 121 (26.7) | 116 (25.6) | 119 (26.2) |
| II | 490 (27.0) | 104 (22.9) | 122 (26.9) | 108 (23.8) | 128 (28.2) | 125 (27.5) | 133 (29.3) |
| III | 770 (42.5) | 200 (44.1) | 183 (40.3) | 203 (44.7) | 185 (40.7) | 187 (41.2) | 190 (41.9) |
| Unknown | 81 (4.5) | 22 (4.8) | 17 (3.7) | 20 (4.4) | 20 (4.4) | 26 (5.7) | 12 (2.6) |
| Type of treatment, No. (%)f | |||||||
| Surgery only | 962 (53.1) | 241 (53.1) | 256 (56.4) | 236 (52.0) | 248 (54.6) | 237 (52.2) | 242 (53.3) |
| Surgery + chemotherapy | 405 (22.4) | 104 (22.9) | 87 (19.2) | 111 (24.4) | 91 (20.0) | 108 (23.8) | 93 (20.5) |
| Surgery + radiotherapy | 189 (10.4) | 40 (8.8) | 55 (12.1) | 38 (8.4) | 50 (11.0) | 38 (8.4) | 59 (13.0) |
| Surgery + chemotherapy + radiotherapy | 182 (10.0) | 52 (11.5) | 42 (9.3) | 50 (11.0) | 47 (10.4) | 49 (10.8) | 47 (10.4) |
| No or unknown surgery | 70 (3.9) | 17 (3.7) | 12 (2.6) | 19 (4.2) | 17 (3.7) | 22 (4.8) | 13 (2.9) |
| Unknown treatment | 4 (0.2) | 0 (0.0) | 2 (0.4) | 0 (0.0) | 1 (0.2) | 0 (0.0) | 0 (0.0) |
| ASA classification, No. (%) | |||||||
| I | 502 (27.7) | 135 (29.7) | 120 (26.4) | 138 (30.4) | 106 (23.3) | 133 (29.3) | 135 (29.7) |
| II | 959 (52.9) | 226 (49.8) | 249 (54.8) | 222 (48.9) | 262 (57.7) | 242 (53.3) | 231 (50.9) |
| III | 204 (11.3) | 48 (10.6) | 58 (12.8) | 52 (11.5) | 51 (11.2) | 38 (8.4) | 58 (12.8) |
| IV | 4 (0.2) | 1 (0.2) | 1 (0.2) | 1 (0.2) | 1 (0.2) | 1 (0.2) | 2 (0.4) |
| Unknown | 143 (7.9) | 44 (9.7) | 26 (5.7) | 41 (9.0) | 34 (7.5) | 40 (8.8) | 28 (6.2) |
| Dietary intake, median [IQR] | |||||||
| Total energy intake, kcal/day | 1810 [1506–2172] | 1899 [1520–2237] | 1854 [1546–2261] | 1884 [1517–2209] | 1851 [1568–2201] | 2028 [1703–2356] | 1837 [1517–2189] |
| Energy-adjusted total dairy, g/day | 270 [177–383] | 114 [72–146] | 481 [423–584] | 131 [76–200] | 454 [388–560] | 232 [117–352] | 327 [231–450] |
| Energy-adjusted low-fat dairy, g/dayg | 165 [70–281] | 44 [9–83] | 369 [283–455] | 20 [-5–45] | 372 [315–455] | 212 [96–321] | 110 [33–216] |
| Energy-adjusted high-fat dairy, g/dayh | 77 [46–126] | 59 [28–91] | 96 [55–186] | 96 [56–166] | 64 [38–99] | 24 [5–37] | 184 [150–246] |
| Energy-adjusted milk, g/dayi | 50.4 [14.7–141.8] | 16.0 [-8.0–37.3] | 188.3 [56.1–292.3] | 19.1 [-3.7–41.9] | 167.2 [43.1–281.6] | 34.4 [-2.3–132.6] | 66.6 [20.9–170.6] |
| Energy-adjusted yoghurt, g/dayj | 72.2 [19.5–139.3] | 20.6 [3.1–61.1] | 140.8 [55.0–250.0] | 20.2 [2.7–66.6] | 131.1 [51.2–225.3] | 60.9 [8.5–142.6] | 92.1 [35.6–151.7] |
| Energy-adjusted cheese, g/dayk | 27.7 [15.8–43.0] | 22.3 [11.8–40.3] | 29.6 [17.8–44.4] | 26.4 [14.3–45.7] | 26.2 [14.9–40.0] | 17.8 [8.8–27.5] | 32.3 [18.3–51.7] |
| Dietary fiber, g/day | 19.7 [15.9–24.2] | 19.7 [15.6–23.9] | 19.9 [16.0–24.6] | 19.3 [15.3–23.4] | 20.4 [16.7–24.9] | 22.5 [19.3–26.8] | 18.9 [14.8–24.0] |
| Wholegrains, g/day | 111.1 [75.4–150.8] | 110.3 [74.0–151.3] | 107.9 [75.6–149.5] | 107.4 [68.4–150.9] | 113.9 [78.5–155.6] | 129.4 [85.8–172.1] | 104.6 [71.3–145.2] |
| Red meat, g/day | 35.7 [20.5–49.8] | 36.8 [21.4–53.6] | 34.8 [20.0–48.2] | 35.9 [19.6–51.4] | 35.7 [20.4–48.5] | 37.7 [24.7–52.0] | 34.5 [20.9–48.5] |
| Processed meat, g/day | 28.1 [13.3–44.6] | 33.6 [16.3–51.7] | 25.1 [12.8–41.8] | 30.5 [14.9–45.6] | 26.3 [13.7–45.9] | 35.0 [16.4–53.4] | 25.0 [11.8–40.3] |
| Alcohol, g/day | 8.1 [1.0–20.4] | 13.0 [1.9–26.4] | 4.6 [0.5–14.2] | 9.6 [0.8–23.3] | 5.6 [0.9–16.6] | 12.7 [2.4–26.7] | 4.4 [0.3–14.2] |
| Dietary calcium, mg/day | 855 [650–1087] | 638 [495–833] | 1145 [938–1364] | 689 [520–916] | 1087 [898–1310] | 830 [625–1055] | 951 [770–1207] |
ASA American Society of Anesthesiologists, IQR interquartile range
aData were missing for 4 participants
bLow education was defined as primary school and lower general secondary education; medium as lower vocational training and higher general secondary education; high as higher vocational training and university
cModerate-to-vigorous physical activity included all activities with a metabolic equivalent value ≥ 3 [17]. Data were missing for 8 participants
dData were missing for 1 participant
eProximal colon includes the caecum, appendix, ascending colon, hepatic flexure, and transverse colon. Distal colon includes the splenic flexure, descending colon, and sigmoid colon. Rectum includes the rectosigmoid junction and rectum
fTreatment includes neoadjuvant and adjuvant treatment
gLow-fat dairy included buttermilk and low-fat, reduced-fat or skimmed versions of milk, yoghurt, custard, and soft curd cheese
hHigh-fat dairy included whole and condensed milk, full-fat versions of yoghurt, custard and soft curd cheese, all other cheeses, ice cream, whipped cream, and butter
iTotal milk included all plain and sweetened milk, regardless of fat content
jTotal yoghurt included all plain and sweetened yoghurt, yoghurt drinks and soft curd cheese, regardless of fat content
kTotal cheese included all hard cheese and soft cheese, except for soft curd cheese
Participants in the highest quartile of total pre-diagnostic dairy intake were older, less often current smokers, and more often had a lower level of education, compared to participants in the lowest quartile of total dairy products. Also, they consumed less processed meat and alcohol, consumed more dietary calcium, and less often used calcium supplements. Participants in the highest quartile of low-fat dairy intake were less often current smokers, consumed more dietary calcium and less alcohol, more often used vitamin D supplements, and less often had an ASA score of I, compared to participants in the lowest quartile. Participants in the highest quartile of high-fat dairy products were older and more often had a lower level of education, consumed more dietary calcium, and less processed meat and alcohol and had a lower total energy intake than participants in the lowest quartile (Table 1). Median pre-diagnostic energy-adjusted intakes of total, low-fat and high-fat dairy were 270 (IQR: 177–383), 165 (IQR: 70–281), and 77 (IQR: 46–126) g/day, respectively (Table 1). Median post-diagnostic energy-adjusted intakes of total, low-fat and high-fat dairy were 282 (IQR: 184–392), 161 (IQR: 70–286), and 81 (IQR: 48–136) g/day, respectively (data not shown). Pearson correlation coefficients between pre- and post-diagnostic intakes ranged between 0.41 and 0.61 (data not shown).
For pre-diagnostic analyses, we observed 176 recurrences during a median follow-up time of 3.0 years (IQR: 2.0–4.2), and 301 participants died during a median follow-up of 5.9 years (IQR: 3.9–7.2). For post-diagnostic analyses, we observed 153 recurrences that occurred during a median follow-up time of 2.6 years (IQR: 1.7–3.8), and 245 participants died during a median follow-up of 5.5 years (IQR: 3.8–6.8).
Pre-diagnostic dairy intake in relation to recurrence and all-cause mortality
As shown in Table 2 and Supplementary Fig. 1, when comparing the highest to the lowest quartile, a higher pre-diagnostic intake of total dairy was associated with a lower risk of recurrence (HR: 0.51, 95% CI 0.33–0.80; PRCS: 0.06), and tended to be associated with all-cause mortality (HRQ4 vs Q1: 0.72, 95% CI 0.52–1.01; PRCS: 0.18). A higher intake of low-fat dairy before diagnosis was associated with a lower risk of recurrence (HR: 0.42, 95%CI 0.26–0.67, PRCS: 0.008; Table 2 and Fig. 2A) and all-cause mortality (HRQ4 vs Q1: 0.58, 95% CI 0.41–0.81; PRCS < 0.001). RCS revealed a significant non-linear association between pre-diagnostic low-fat dairy intake and all-cause mortality, with intakes higher than the median of the first quartile (i.e. 21 g/day) associated with a reduced risk of mortality (Fig. 2A). Contrastingly, a higher intake of high-fat dairy before diagnosis tended to be associated with an increased risk of all-cause mortality (HRQ4 vs Q1: 1.41, 95% CI 0.98–2.01; PRCS: 0.030). Pre-diagnostic high-fat dairy intake was not associated with recurrence. A higher yoghurt intake before diagnosis seemed associated with a lower risk of all-cause mortality (HRQ4 vs Q1: 0.70, 95% CI 0.50–0.99; PRCS: 0.09), but not with recurrence. No associations were observed between total milk or cheese intake before diagnosis and recurrence or all-cause mortality.
Table 2.
Associations of pre-diagnostic energy-adjusted intake of total dairy, low-fat dairy, high-fat dairy, and dairy products with recurrence (n = 1315) and all-cause mortality (n = 1731) in sex-specific quartiles of people with stage I-III colorectal cancer
| Dietary variable | Recurrencea | All-cause mortality | ||||||
|---|---|---|---|---|---|---|---|---|
| Median energy-adjusted intake [IQR]b | n | No. of recurrences/person-years | Multivariable HR (95% CI)c | Median energy-adjusted intake [IQR]b | n | No. of deaths/person-years | Multivariable HR (95% CI)c | |
| Total dairy | ||||||||
| Q1 | 109.8 [69.7, 142.4] | 331 | 55/1033 | 1.00 | 114.3 [73.0, 147.3] | 432 | 74/2534 | 1.00 |
| Q2 | 220.3 [184.3, 253.9] | 330 | 46/1063 | 0.80 (0.54–1.19) | 224.5 [190.0, 247.3] | 432 | 78/2504 | 0.98 (0.71–1.35) |
| Q3 | 323.2 [278.1, 351.3] | 326 | 42/1049 | 0.72 (0.48–1.08) | 322.5 [279.1, 348.9] | 430 | 64/2485 | 0.77 (0.55–1.09) |
| Q4 | 489.6 [426.1, 591.02] | 328 | 30/1106 | 0.51 (0.33–0.80) | 481.0 [423.7, 581.2] | 437 | 66/2582 | 0.72 (0.52–1.01) |
| Ptrend | NA | NA | NA | 0.003 | NA | NA | NA | 0.029 |
| Continuous (per 100 g/day) | 267.9 [172.1, 385.4] | 1315 | 173/4251 | 0.89 (0.81–0.99) | 269.6 [177.6, 384.6] | 1731 | 282/10105 | 0.96 (0.89–1.03) |
| Low-fat dairyd | ||||||||
| Q1 | 20.5 [− 4.8, 42.3] | 333 | 56/1076 | 1.00 | 20.5 [− 3.5, 44.6] | 434 | 87/2511 | 1.00 |
| Q2 | 120.1 [94.8, 142.0] | 329 | 46/1034 | 0.83 (0.56–1.23) | 119.7 [94.9, 143.7] | 438 | 74/2503 | 0.84 (0.62–1.15) |
| Q3 | 223.0 [184.0, 254.9] | 325 | 47/1035 | 0.84 (0.57–1.24) | 219.6 [185.9, 253.4] | 425 | 63/2474 | 0.69 (0.50–0.96) |
| Q4 | 374.3 [317.8, 465.4] | 328 | 24/1108 | 0.42 (0.26–0.67) | 371.9 [315.4, 452.4] | 434 | 58/2618 | 0.58 (0.41–0.81) |
| Ptrend | NA | NA | NA | < .001 | NA | NA | NA | < 0.001 |
| Continuous (per 100 g/day) | 163.3 [68.2, 281.4] | 1315 | 173/4251 | 0.86 (0.77–0.96) | 164.8 [69.7, 281.2] | 1731 | 282/10105 | * |
| High-fat dairye | ||||||||
| Q1 | 25.4 [6.3, 36.9] | 328 | 45/1044 | 1.00 | 24.5 [5.5, 36.9] | 428 | 49/2553 | 1.00 |
| Q2 | 60.7 [50.9, 68.7] | 329 | 38/1065 | 0.80 (0.51–1.24) | 61.3 [51.5, 69.0] | 430 | 67/2525 | 1.13 (0.78–1.66) |
| Q3 | 96.7 [85.9, 109.2] | 325 | 43/1068 | 0.93 (0.60–1.44) | 96.8 [86.2, 109.2] | 431 | 80/2483 | 1.33 (0.92–1.93) |
| Q4 | 180.3 [149.6, 229.8] | 333 | 47/1075 | 0.97 (0.64–1.47) | 182.9 [149.8, 244.3] | 442 | 86/2545 | 1.41 (0.98–2.01) |
| Ptrend | NA | NA | NA | 0.83 | NA | NA | NA | 0.06 |
| Continuous (per 100 g/day) | 76.0 [45.6, 128.5] | 1315 | 173/4251 | 1.07 (0.91–1.26) | 77.7 [46.5, 127.9] | 1731 | 282/10105 | 1.15 (1.03–1.28) |
| Total milk | ||||||||
| Q1 | − 4.2 [-19.1, 7.1] | 331 | 43/1055 | 1.00 | − 4.9 [-19.4, 6.7] | 424 | 60/2508 | 1.00 |
| Q2 | 31.8 [21.2, 41.4] | 328 | 51/984 | 1.06 (0.69–1.64) | 30.9 [21.1, 40.5] | 432 | 75/2399 | 1.07 (0.75–1.55) |
| Q3 | 93.8 [69.6, 120.4] | 330 | 40/1100 | 0.89 (0.57–1.40) | 89.6 [67.4, 116.5] | 439 | 74/2558 | 1.09 (0.76–1.55) |
| Q4 | 231.5 [175.1, 304.3] | 326 | 39/1112 | 0.81 (0.52–1.27) | 227.0 [171.1, 297.5] | 436 | 73/2641 | 0.91 (0.64–1.29) |
| Ptrend | NA | NA | NA | 0.23 | NA | NA | NA | 0.39 |
| Continuous (per 100 g/day) | 52.2 [15.3, 145.8] | 1315 | 173/4251 | 0.90 (0.78–1.04) | 51.2 [15.5, 142.5] | 1731 | 282/10105 | 1.02 (0.92–1.12) |
| Total yoghurt | ||||||||
| Q1 | 2.4 [− 7.1, 9.9] | 325 | 43/1088 | 1.00 | 2.4 [− 6.1, 11.3] | 427 | 78/2531 | 1.00 |
| Q2 | 45.8 [22.2, 62.0] | 328 | 48/1040 | 1.14 (0.75–1.72) | 45.3 [22.5, 68.0] | 429 | 80/2449 | 0.91 (0.66–1.25) |
| Q3 | 102.3 [76.1, 122.0] | 333 | 49/1074 | 1.09 (0.72–1.64) | 102.3 [76.4, 124.7] | 438 | 68/2588 | 0.74 (0.53–1.02) |
| Q4 | 205.0 [147.1, 277.2] | 329 | 33/1049 | 0.78 (0.50–1.24) | 204.6 [147.5, 273.6] | 437 | 56/2537 | 0.70 (0.50–0.99) |
| Ptrend | NA | NA | NA | 0.21 | NA | NA | NA | 0.029 |
| Continuous (per 100 g/day) | 70.8 [18.5, 138.4] | 1315 | 173/4251 | 0.96 (0.83–1.13) | 72.8 [19.9, 140.1] | 1731 | 282/10105 | 0.86 (0.75–0.99) |
| Total cheese | ||||||||
| Q1 | 9.0 [2.5, 12.6] | 329 | 47/1065 | 1.00 | 8.6 [2.4, 12.4] | 429 | 63/2518 | 1.00 |
| Q2 | 21.6 [19.4, 25.1] | 325 | 35/1048 | 0.76 (0.49–1.18) | 21.5 [19.0, 24.8] | 429 | 64/2523 | 0.92 (0.65–1.32) |
| Q3 | 34.8 [31.1, 38.1] | 331 | 47/1007 | 0.97 (0.64–1.46) | 34.5 [31.1, 37.4] | 436 | 78/2449 | 1.08 (0.77–1.51) |
| Q4 | 57.2 [49.1, 73.3] | 330 | 44/1131 | 0.91 (0.60–1.38) | 57.6 [49.1, 74.0] | 437 | 77/2615 | 1.04 (0.75–1.46) |
| Ptrend | NA | NA | NA | 0.95 | NA | NA | NA | 0.61 |
| Continuous (per 30 g/day) | 28.0 [16.1, 43.7] | 1315 | 173/4251 | 1.03 (0.85–1.24) | 27.8 [15.9, 43.2] | 1731 | 282/10105 | 1.03 (0.89–1.19) |
CI confidence interval, HR hazard ratio, IQR inter-quartile range. Cut-off points for female quartiles were as follows: 220.8, 308.1 and 441.3 g/day for total dairy; 101.4, 201.4 and 313.5 g/day for low-fat dairy; 58.0, 84.0 and 129.1 g/day for high-fat dairy; 23.6, 60.0 and 149.6 g/day for milk; 40.7, 104.6 and 164.6 g/day for yoghurt; 17.9, 29.8 and 44.8 g/day for cheese. Cut-off points for male quartiles were as follows: 152.1, 244.1 and 358.0 g/day for total dairy; 56.6, 145.7 and 264.0 g/day for low-fat dairy; 38.0, 72.5 and 123.1 g/day for high-fat dairy; 9.4, 46.0 and 137.2 g/day for milk; 9.7, 54.7 and 120.5 g/day for yoghurt; 14.3, 26.2 and 41.9 g/day for cheese
*This association was observed to be non-linear in restricted cubic splines. See Fig. 2 for the continuous analysis
aRecurrence is defined as a locoregional recurrence or distant metastasis. Participants were excluded from data analysis when they had missing data on recurrence status (n = 463) or when they had no surgery (n = 8)
bDietary intake was adjusted for energy using the energy residual method. To improve interpretability, the predicted nutrient intake at the median total energy intake was added to individual residuals [15]
cThe adjusted model was adjusted for age, sex, stage of disease and pre-diagnostic energy intake
dLow-fat dairy included buttermilk and low-fat, reduced-fat or skimmed versions of milk, yoghurt, custard, and soft curd cheese
eHigh-fat dairy included whole and condensed milk, full-fat versions of yoghurt, custard and soft curd cheese, all other cheeses, ice cream, whipped cream, and butter
Fig. 2.
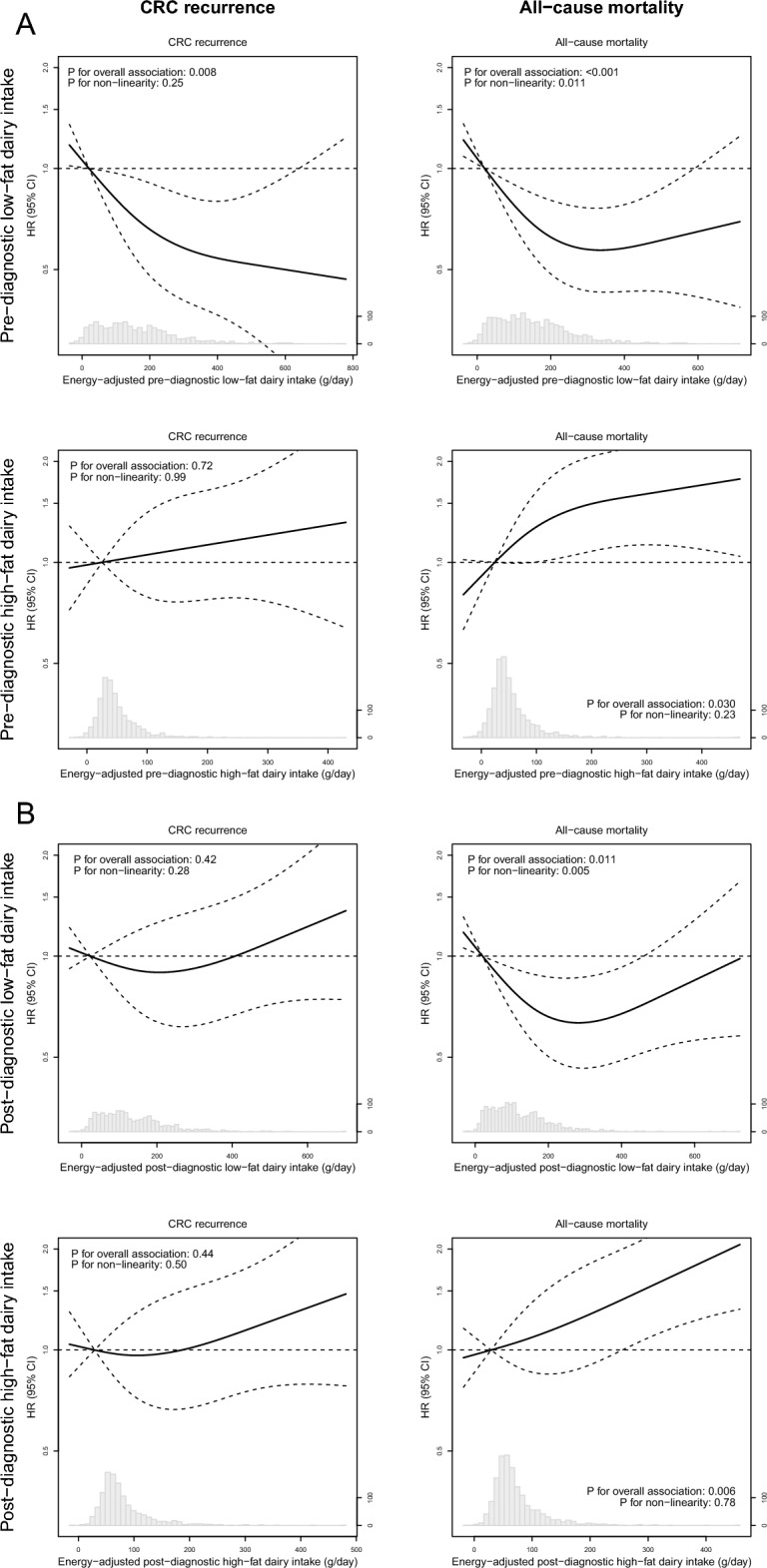
Associations between pre-diagnostic (A) and post-diagnostic (B) intake of low- and high-fat dairy and colorectal cancer prognosis. Solid lines are restricted cubic splines, and dashed lines are 95% confidence intervals. The reference values were set at the median of the first quartile of dairy intake. Knots were located at the 5th, 50th and 95th percentile, and the graphs were truncated at the 1st and 99th percentile. Analyses are adjusted for age, sex, disease stage and energy intake
Post-diagnostic dairy intake in relation to recurrence and all-cause mortality
As shown in Table 3 and Supplementary Fig. 1, post-diagnostic total dairy intake was not associated with recurrence or all-cause mortality. A higher intake of low-fat dairy after diagnosis tended to be associated with a lower risk of all-cause mortality (HRQ4 vs Q1: 0.76, 95% CI 0.53–1.08; PRCS: 0.011; Table 3 and Fig. 2B). RCS revealed a significant non-linear association between post-diagnostic low-fat dairy intake and all-cause mortality, with intakes higher than the median of the first quartile (i.e. 20 g/day) associated with a reduced risk of mortality (Fig. 2B). Post-diagnostic low- and high-fat dairy intakes were not associated with recurrence. A higher intake of high-fat dairy after diagnosis was associated with an increased risk of all-cause mortality (HRQ4 vs Q1: 1.60, 95% CI 1.10–2.33; PRCS: 0.006). Post-diagnostic milk, yoghurt, and cheese intake were not associated with recurrence or all-cause mortality.
Table 3.
Associations of post-diagnostic energy-adjusted intake of total dairy, low-fat dairy, high-fat dairy, and dairy products with recurrence (n = 1215) and all-cause mortality (n = 1605) in sex-specific quartiles of people with stage I-III colorectal cancer
| Dietary variable | Recurrencea | All-cause mortality | ||||||
|---|---|---|---|---|---|---|---|---|
| Median energy-adjusted intake [IQR]b | n | No. of recurrences/person-years | Adjusted model, HR (95% CI)c | Median energy-adjusted intake [IQR]b | n | No. of deaths/ person-years |
Adjusted model, HR (95% CI)c | |
| Total dairy | ||||||||
| Q1 | 125.3 [92.7, 157.9] | 305 | 31/889 | 1.00 | 125.7 [90.8, 157.9] | 396 | 51/2211 | 1.00 |
| Q2 | 227.7 [206.4, 257.8] | 304 | 41/899 | 1.38 (0.86–2.21) | 228.1 [206.6, 254.1] | 403 | 59/2186 | 1.06 (0.73–1.55) |
| Q3 | 341.6 [318.2, 365.7] | 303 | 39/842 | 1.31 (0.81–2.10) | 335.1 [303.5, 362.6] | 400 | 60/2161 | 1.07 (0.73–1.55) |
| Q4 | 485.9 [440.2, 606.7] | 303 | 39/853 | 1.27 (0.79–2.04) | 481.4 [434.8, 593.7] | 406 | 65/2272 | 1.00 (0.69–1.45) |
| Ptrend | NA | NA | NA | 0.47 | NA | NA | NA | 0.94 |
| Continuous (per 100 g/day) | 285.1 [184.7, 396.6] | 1215 | 150/3484 | 1.06 (0.97–1.16) | 282.7 [185.9, 393.7] | 1605 | 235/8829 | 1.03 (0.95–1.10) |
| Low-fat dairyd | ||||||||
| Q1 | 23.0 [0.3, 46.7] | 302 | 37/871 | 1.00 | 20.1 [− 1.7, 46.2] | 396 | 66/2151 | 1.00 |
| Q2 | 120.1 [94.4, 140.1] | 305 | 36/897 | 0.95 (0.60–1.50) | 119.3 [94.9, 139.8] | 402 | 58/2220 | 0.83 (0.58–1.18) |
| Q3 | 221.7 [187.0, 259.4] | 304 | 36/852 | 1.03 (0.65–1.63) | 216.9 [180.6, 255.5] | 402 | 52/2211 | 0.70 (0.49–1.01) |
| Q4 | 382.6 [322.3, 470.0] | 304 | 41/864 | 1.14 (0.73–1.78) | 380.3 [322.6, 469.4] | 405 | 59/2247 | 0.76 (0.53–1.08) |
| Ptrend | NA | NA | NA | 0.48 | NA | NA | NA | 0.12 |
| Continuous (per 100 g/day) | 161.0 [71.3, 288.0] | 1215 | 150/3484 | 1.03 (0.94–1.13) | 161.2 [72.0, 286.6] | 1605 | 235/8829 | * |
| High-fat dairye | ||||||||
| Q1 | 29.2 [13.2, 40.9] | 307 | 40/853 | 1.00 | 29.2 [13.5, 40.0] | 400 | 43/2213 | 1.00 |
| Q2 | 64.7 [54.8, 73.0] | 304 | 33/902 | 0.80 (0.50–1.30) | 64.9 [54.0, 73.0] | 403 | 61/2232 | 1.27 (0.85–1.89) |
| Q3 | 102.9 [90.7, 115.9] | 305 | 32/895 | 0.77 (0.48–1.24) | 102.2 [90.4, 116.2] | 405 | 49/2249 | 0.94 (0.62–1.44) |
| Q4 | 209.7 [168.5, 291.9] | 299 | 45/834 | 1.02 (0.66–1.57) | 202.7 [166.1, 273.6] | 397 | 82/2135 | 1.60 (1.10–2.33) |
| Ptrend | NA | NA | NA | 0.62 | NA | NA | NA | 0.010 |
| Continuous (per 100 g/day) | 81.2 [48.4, 136.6] | 1215 | 150/3484 | 1.08 (0.93–1.24) | 81.2 [48.5, 135.9] | 1605 | 235/8829 | 1.18 (1.06–1.32) |
| Total milk | ||||||||
| Q1 | − 0.4 [− 14.4, 8.4] | 307 | 32/863 | 1.00 | − 1.1 [− 15.3, 7.0] | 394 | 57/2106 | 1.00 |
| Q2 | 32.9 [25.6, 42.8] | 301 | 42/873 | 1.31 (0.82–2.10) | 31.9 [24.1, 40.5] | 401 | 54/2252 | 0.79 (0.53–1.17) |
| Q3 | 104.6 [78.9, 133.8] | 305 | 30/897 | 1.01 (0.61–1.67) | 101.2 [75.7, 128.6] | 403 | 50/2209 | 0.78 (0.53–1.15) |
| Q4 | 256.2 [191.9, 306.5] | 302 | 46/851 | 1.39 (0.88–2.19) | 237.1 [183.6, 299.5] | 407 | 74/2261 | 0.99 (0.70–1.42) |
| Ptrend | NA | NA | NA | 0.28 | NA | NA | NA | 0.54 |
| Continuous (per 100 g/day) | 58.8 [16.9, 159.8] | 1215 | 150/3484 | 1.05 (0.93–1.18) | 56.9 [16.4, 155.6] | 1605 | 235/8829 | 1.02 (0.92–1.12) |
| Total yoghurt | ||||||||
| Q1 | 4.5 [− 4.3, 11.7] | 301 | 30/888 | 1.00 | 4.2 [-4.4, 12.1] | 397 | 62/2229 | 1.00 |
| Q2 | 45.7 [29.4, 58.9] | 303 | 44/862 | 1.38 (0.86–2.20) | 47.4 [29.1, 61.2] | 402 | 62/2171 | 0.99 (0.70–1.41) |
| Q3 | 104.1 [84.9, 121.7] | 305 | 33/897 | 1.03 (0.63–1.69) | 103.8 [82.9, 121.1] | 402 | 56/2215 | 0.84 (0.58–1.20) |
| Q4 | 187.3 [144.2, 266.7] | 306 | 43/836 | 1.51 (0.96–2.39) | 187.8 [143.8, 269.6] | 404 | 55/2214 | 0.89 (0.62–1.28) |
| Ptrend | NA | NA | NA | 0.39 | NA | NA | NA | 0.17 |
| Continuous (per 100 g/day) | 76.3 [20.5, 136.8] | 1215 | 150/3484 | 1.07 (0.91–1.27) | 75.5 [21.7, 137.2] | 1605 | 235/8829 | 0.96 (0.83–1.11) |
| Total cheese | ||||||||
| Q1 | 8.9 [3.3, 12.9] | 303 | 37/848 | 1.00 | 8.1 [2.1, 12.6] | 397 | 55/2133 | 1.00 |
| Q2 | 21.2 [18.9, 23.8] | 301 | 47/825 | 1.37 (0.88–2.12) | 21.1 [18.6, 23.4] | 403 | 66/2193 | 1.02 (0.71–1.48) |
| Q3 | 32.9 [29.6, 36.9] | 305 | 34/889 | 0.92 (0.57–1.46) | 32.7 [29.3, 36.5] | 403 | 55/2247 | 0.86 (0.59–1.25) |
| Q4 | 53.1 [46.8, 67.5] | 306 | 32/921 | 0.83 (0.51–1.33) | 52.4 [46.5, 66.4] | 402 | 59/2257 | 0.89 (0.61–1.29) |
| Ptrend | NA | NA | NA | 0.18 | NA | NA | NA | 0.40 |
| Continuous (per 30 g/day) | 26.3 [16.3, 40.9] | 1215 | 150/3484 | 0.85 (0.68–1.07) | 26.1 [15.9, 40.4] | 1605 | 235/8829 | 0.93 (0.78–1.11) |
CI confidence interval, HR hazard ratio; IQR inter-quartile range. Cut-off points for female quartiles were as follows: 208.8, 317.5 and 418.1 g/day for total dairy; 91.2, 181.8 and 303.1 g/day for low-fat dairy; 61.0, 89.8 and 145.5 g/day for high-fat dairy; 21.7, 52.0 and 153.5 g/day for milk; 45.1, 102.9 and 152.5 g/day for yoghurt; 18.5, 28.2 and 42.4 g/day for cheese. Cut-off points for male quartiles were as follows: 166.9, 263.2 and 375.3 g/day for total dairy; 61.6, 147.3 and 270.6 g/day for low-fat dairy; 42.5, 74.0 and 129.1 g/day for high-fat dairy; 12.7, 59.4 and 155.5 g/day for milk; 14.0, 58.7 and 121.9 g/day for yoghurt; 14.1, 24.4 and 39.4 g/day for cheese
*This association was observed to be non-linear in restricted cubic splines. See Fig. 2 for the continuous analysis
aRecurrence is defined as a locoregional recurrence or distant metastasis. Participants were excluded from data analysis when they had missing data on recurrence status (n = 419), when they had no surgery (n = 8) or when they had a recurrence before filling out the post-diagnostic FFQ (n = 9)
bDietary intake was adjusted for energy using the energy residual method. To improve interpretability, the predicted nutrient intake at the median total energy intake was added to individual residuals [15]
cThe adjusted model was adjusted for age, sex, stage of disease and post-diagnostic energy intake
dLow-fat dairy included buttermilk and low-fat, reduced-fat or skimmed versions of milk, yoghurt, custard, and soft curd cheese
eHigh-fat dairy included whole and condensed milk, full-fat versions of yoghurt, custard and soft curd cheese, all other cheeses, ice cream, whipped cream, and butter
Except for pre- and post-diagnostic low-fat dairy in relation to all-cause mortality, we observed no evidence of non-linearity in our RCS analyses, and findings of these analyses were in line with the Cox proportional hazard regression analyses (Fig. 2 and Supplementary Figure 1).
Discussion
This study demonstrated that a higher low-fat dairy intake was associated with a reduced risk of all-cause mortality in people with stage I-III CRC, whereas a higher intake of high-fat dairy was associated with an increased risk of all-cause mortality, both before and after diagnosis. Furthermore, a higher pre-diagnostic intake of yoghurt was associated with a reduced risk of all-cause mortality. Also, to our knowledge, this is the first study to demonstrate that higher pre-diagnostic intakes of total and low-fat dairy were associated with a reduced risk of recurrence.
Our findings on post-diagnostic low- and high-fat dairy intake in relation to all-cause mortality are in line with the results of a recent pooled analysis of the Nurses’ Health Study and Health Professionals Follow-up Study in people with stage I-IV CRC, that also demonstrated a reduced risk of all-cause mortality with higher post-diagnostic low-fat dairy intakes, and an increased risk with higher post-diagnostic high-fat dairy intakes [7]. Previous large cohort studies observed no statistically significant associations between pre- [5, 6] or post-diagnostic [6, 7] total dairy intake and all-cause mortality. Therefore, we stress the importance of considering low- and high-fat dairy separately when studying health effects of dairy.
The mechanisms linking dairy consumption to prognostic outcomes in CRC are not clear and current hypotheses are largely derived from research focussing on CRC aetiology [3, 4]. Calcium has been suggested to reduce CRC risk via binding secondary bile acids and free fatty acids in the colonic lumen, thereby reducing oxidative stress and reactive proliferation of the epithelium [19, 20]. Also, clinical trials have demonstrated that calcium can alter molecular pathways that lead to direct inhibition of cell proliferation, promotion of cell differentiation, and induction of apoptosis in tumour cells [20–25]. Other dairy components, such as conjugated linoleic acid, lactic acid-producing bacteria in fermented dairy, lactoferrin, and vitamin D in fortified dairy foods, may also reduce CRC risk [2, 3, 21, 26, 27]. Previous work from our group showed that, although no clear associations were observed, the HRs for recurrence and all-cause mortality tended to be lower for those with a higher dietary calcium intake [28]. The present study includes a larger study population and more events for recurrence and all-cause mortality. To provide insight into whether observed associations in the current study are independent of dietary calcium intake, we carried out additional exploratory analyses where we adjusted our models for dietary calcium intake (data not shown). Except for the association between cheese intake and risk of recurrence, which changed to a more positive, but still non-significant association, the observed associations did not substantially change. Furthermore, considering the current uncertainty about the biological mechanism linking dairy to CRC outcomes, this study focuses on food groups rather than micronutrients, which is closer to people’s perception of the diet and easier to translate into dietary guidelines.
Our study suggests that pre-diagnostic, but not post-diagnostic, intake of low-fat dairy is associated with a reduced risk of recurrence. A potential explanation for this time-dependent finding could be that pre-diagnostic consumption may well reflect habitual dietary consumption in the decades before cancer diagnosis, implying low-fat dairy may directly or indirectly influence carcinogenesis early in the CRC continuum [9]. Also, methodological issues, such as differences in number of events and duration of follow-up time, or differences in dairy intake could explain this time-dependent finding. For post-diagnostic intake, our study showed only associations with all-cause mortality, not with recurrence, which may suggest that post-diagnostic dairy consumption influences CRC prognosis via other ways than recurrence, such as risk of cardiovascular disease [29].
In our study, a higher pre-diagnostic intake of yoghurt intake was associated with a reduced risk of all-cause mortality. Previous large prospective cohort studies showed no statistically significant association between pre-diagnostic yoghurt consumption and CRC-specific mortality [5, 9]. Possibly, yoghurt consumption influences cardiovascular risk factors rather than CRC [30], explaining why yoghurt may be associated with all-cause mortality in the current study, but not with CRC-specific mortality in other studies. Therefore, future studies should include cause-specific death information when studying associations between yoghurt consumption and CRC prognosis.
We observed no associations between pre- or post-diagnostic total milk or cheese intake and CRC prognosis. Previous studies on milk intake demonstrated a marginal positive [5] or no association [6, 7] between pre-diagnostic total milk intake and mortality. For post-diagnostic intake, the two US cohorts observed a reduced risk of all-cause mortality with higher intakes of low-fat milk [7] and total milk [6]. In contrast to the milk studied in the US cohorts [6, 7], Dutch milk is not fortified with vitamin D. As higher concentrations of 25-hydroxyvitamin D in the blood have previously been associated with improved survival in CRC [31], this might explain the discrepancy between findings. Also, compared to an estimated median milk intake of ~ 100–150 g/day in previous studies, our study population had a relatively low milk intake with a median intake of around 50 g/day. However, our study population contained many non-consumers of milk, and mean intakes were in line with the most recent national food consumption survey [32]. Only two previous cohorts studied cheese separately, and also demonstrated no association between pre- [5] or post-diagnostic [7] cheese intake and mortality in CRC. Future studies should investigate unfortified low-fat milk intake in relation to mortality in CRC.
This study has several strengths, including the prospective study design and the availability of detailed information on cancer treatment, recurrence and both pre- and post-diagnostic dietary intake and supplement use data. Furthermore, considering the current global transition towards more plant-based diets [33–35], it is highly relevant to study potential health effects of dairy in cancer survivorship. A limitation of the current study is the lack of information on the specific causes of death. However, we believe all-cause mortality is a highly relevant study outcome with strong clinical relevance besides the risk of recurrence, as people with CRC are also more prone to develop other chronic diseases compared to the general population [29]. A second potential limitation is the possibility of underestimation of self-reported dietary intake data inherent to the chosen dietary assessment method. However, FFQs are commonly used in cohort studies for feasibility reasons, and literature has previously shown to capture intake of dairy reasonably well by FFQs in general [36–38]. Also, even though we tested for confounding of many different lifestyle factors, we cannot exclude the possibility of residual confounding. Lastly, our participants were primarily Caucasian, limiting the generalizability of our study. Future studies should make an effort to recruit a diverse range of ethnicities.
Conclusion
This study found that higher pre- and post-diagnostic intakes of low-fat dairy were associated with a reduced risk of all-cause mortality in people with stage I-III CRC, whereas higher intakes of high-fat dairy were associated with an increased risk of all-cause mortality. We also demonstrated that a higher pre-diagnostic intake of low-fat dairy was associated with a reduced risk of recurrence in persons diagnosed with stage I-III CRC. Future research should differentiate between low-and high-fat dairy when investigating associations with recurrence and mortality. When replicated in other large prospective cohorts and ultimately intervention studies, our results can contribute to specific dietary guidelines for CRC survivors who aim to improve their prognosis via lifestyle change.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors would like to thank all participants, the involved co-workers in the participating hospitals, and the COLON investigators at Wageningen University & Research. Also, the authors would like to thank the registration team of the Netherlands Comprehensive Cancer Organisation (IKNL) for the collection of data for the Netherlands Cancer Registry.
Author contributions
Conceptualization: ASL, DEK, EK, FJBD. Formal analysis: ASL. Funding acquisition: DEK, RMW, EK, FJBD. Investigation: ASL, DEK, RMW, EW, FJBD. Methodology: ASL, DEK, EK, FJBD. Project administration: ASL, DEK, EK, FJBD. Resources: HKH, JHWW. Supervision: DEK, EK, FJBD. Visualization: ASL. Writing—original draft: ASL. Writing: reviewing and editing: all co-authors.
Funding
This work was supported by Wereld Kanker Onderzoek Fonds (WKOF) & World Cancer Research Fund International (WCRF International) as well as by funding (2014/1179, IIG_FULL_2021_022 and IIG_FULL_2021_023) obtained from the Wereld Kanker Onderzoek Fonds (WKOF) as part of the World Cancer Research Fund International grant programme; Alpe d’Huzes/Dutch Cancer Society (UM 2012–5653, UW 2013–5927, UW 2015–7946); and ERA-NET on Translational Cancer Research (TRANSCAN: Dutch Cancer Society (UW2013-6397, UW2014-6877) and the Netherlands Organization for Health Research and Development (ZonMw), the Netherlands) and the Regio Deal Foodvalley (162135). The funders had no role in the design of the study; the collection, analysis and interpretation of the data; the writing of the manuscript; and the decision to submit the manuscript for publication.
Availability of data and materials
Because the data consist of identifying cohort information, some access restrictions apply, and therefore they cannot be made publicly available. Data will be shared with permission from the acting committee of the COLON study. Requests for data can be sent to dr. Fränzel van Duijnhoven, Division of Human Nutrition and Health, Wageningen University & Research, Netherlands (e-mail: franzel.vanduijnhoven@wur.nl).
Declarations
Conflicts of interest
The authors declare no conflicts of interest.
References
- 1.Jin S, Kim Y, Je Y. Dairy consumption and risks of colorectal cancer incidence and mortality: a meta-analysis of prospective cohort studies. Cancer Epidemiol Biomarkers Prev. 2020;29(11):2309–2322. doi: 10.1158/1055-9965.Epi-20-0127. [DOI] [PubMed] [Google Scholar]
- 2.World Cancer Research Fund/American Institute for Cancer Research (2018) Diet, nutrition, physical activity and colorectal cancer
- 3.Norat T, Riboli E. Dairy products and colorectal cancer. A review of possible mechanisms and epidemiological evidence. Eur J Clin Nutr. 2003;57(1):1–17. doi: 10.1038/sj.ejcn.1601522. [DOI] [PubMed] [Google Scholar]
- 4.World Cancer Research Fund/American Institute for Cancer Research (2018) Diet, nutrition, physical activity and cancer: a global perspective
- 5.Dik VK, Murphy N, Siersema PD, Fedirko V, Jenab M, Kong SY, Hansen CP, Overvad K, Tjønneland A, Olsen A, Dossus L, Racine A, Bastide N, Li K, Kühn T, Boeing H, Aleksandrova K, Trichopoulou A, Trichopoulos D, Barbitsioti A, Palli D, Contiero P, Vineis P, Tumino R, Panico S, Peeters PH, Weiderpass E, Skeie G, Hjartåker A, Amiano P, Sánchez MJ, Fonseca-Nunes A, Barricarte A, Chirlaque MD, Redondo ML, Jirström K, Manjer J, Nilsson LM, Wennberg M, Bradbury KE, Khaw KT, Wareham N, Cross AJ, Riboli E, Bueno-de-Mesquita HB. Prediagnostic intake of dairy products and dietary calcium and colorectal cancer survival–results from the EPIC cohort study. Cancer Epidemiol Biomarkers Prev. 2014;23(9):1813–1823. doi: 10.1158/1055-9965.Epi-14-0172. [DOI] [PubMed] [Google Scholar]
- 6.Yang B, McCullough ML, Gapstur SM, Jacobs EJ, Bostick RM, Fedirko V, Flanders WD, Campbell PT. Calcium, vitamin D, dairy products, and mortality among colorectal cancer survivors: the cancer prevention study-II nutrition cohort. J Clin Oncol. 2014;32(22):2335–2343. doi: 10.1200/jco.2014.55.3024. [DOI] [PubMed] [Google Scholar]
- 7.Liu X, Yang W, Wu K, Ogino S, Wang W, He N, Chan AT, Zhang Z-F, Meyerhardt JA, Giovannucci E, Zhang X. Postdiagnostic dairy products intake and colorectal cancer survival in US males and females. Am J Clin Nutr. 2021 doi: 10.1093/ajcn/nqab059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Barrubés L, Babio N, Becerra-Tomás N, Rosique-Esteban N, Salas-Salvadó J. Association between dairy product consumption and colorectal cancer risk in adults: A systematic review and meta-analysis of epidemiologic studies. Adv Nutr. 2019;10(suppl 2):S190–s211. doi: 10.1093/advances/nmy114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Michels KB, Willett WC, Vaidya R, Zhang X, Giovannucci E. Yogurt consumption and colorectal cancer incidence and mortality in the Nurses' Health Study and the health professionals follow-up study. Am J Clin Nutr. 2020;112(6):1566–1575. doi: 10.1093/ajcn/nqaa244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cho E, Smith-Warner SA, Spiegelman D, Beeson WL, van den Brandt PA, Colditz GA, Folsom AR, Fraser GE, Freudenheim JL, Giovannucci E, Goldbohm RA, Graham S, Miller AB, Pietinen P, Potter JD, Rohan TE, Terry P, Toniolo P, Virtanen MJ, Willett WC, Wolk A, Wu K, Yaun SS, Zeleniuch-Jacquotte A, Hunter DJ. Dairy foods, calcium, and colorectal cancer: a pooled analysis of 10 cohort studies. J Natl Cancer Inst. 2004;96(13):1015–1022. doi: 10.1093/jnci/djh185. [DOI] [PubMed] [Google Scholar]
- 11.Winkels RM, Heine-Bröring RC, van Zutphen M, van Harten-Gerritsen S, Kok DE, van Duijnhoven FJ, Kampman E. The COLON study: colorectal cancer: longitudinal, observational study on nutritional and lifestyle factors that may influence colorectal tumour recurrence, survival and quality of life. BMC Cancer. 2014;14:374. doi: 10.1186/1471-2407-14-374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Verkleij-Hagoort AC, de Vries JH, Stegers MP, Lindemans J, Ursem NT, Steegers-Theunissen RP. Validation of the assessment of folate and vitamin B12 intake in women of reproductive age: the method of triads. Eur J Clin Nutr. 2007;61(5):610–615. doi: 10.1038/sj.ejcn.1602581. [DOI] [PubMed] [Google Scholar]
- 13.Feunekes IJ, Van Staveren WA, Graveland F, De Vos J, Burema J. Reproducibility of a semiquantitative food frequency questionnaire to assess the intake of fats and cholesterol in The Netherlands. Int J Food Sci Nutr. 1995;46(2):117–123. doi: 10.3109/09637489509012539. [DOI] [PubMed] [Google Scholar]
- 14.National Institute for Public Health and the Environment (2019) NEVO online version 2011/3.0
- 15.Willett WC, Howe GR, Kushi LH. Adjustment for total energy intake in epidemiologic studies. Am J Clin Nutr. 1997;65(4 suppl):1220–1228. doi: 10.1093/ajcn/65.4.1220S. [DOI] [PubMed] [Google Scholar]
- 16.Wendel-Vos GC, Schuit AJ, Saris WH, Kromhout D. Reproducibility and relative validity of the short questionnaire to assess health-enhancing physical activity. J Clin Epidemiol. 2003;56(12):1163–1169. doi: 10.1016/s0895-4356(03)00220-8. [DOI] [PubMed] [Google Scholar]
- 17.Ainsworth BE, Haskell WL, Herrmann SD, Meckes N, Bassett DR, Jr, Tudor-Locke C, Greer JL, Vezina J, Whitt-Glover MC, Leon AS. 2011 compendium of physical activities: a second update of codes and MET values. Med Sci Sports Exerc. 2011;43(8):1575–1581. doi: 10.1249/MSS.0b013e31821ece12. [DOI] [PubMed] [Google Scholar]
- 18.Van Leersum NJ, Snijders HS, Henneman D, Kolfschoten NE, Gooiker GA, ten Berge MG, Eddes EH, Wouters MW, Tollenaar RA, Bemelman WA, van Dam RM, Elferink MA, Karsten TM, van Krieken JH, Lemmens VE, Rutten HJ, Manusama ER, van de Velde CJ, Meijerink WJ, Wiggers T, van der Harst E, Dekker JW, Boerma D. The Dutch surgical colorectal audit. Eur J Surg Oncol. 2013;39(10):1063–1070. doi: 10.1016/j.ejso.2013.05.008. [DOI] [PubMed] [Google Scholar]
- 19.Newmark HL, Wargovich MJ, Bruce WR. Colon cancer and dietary fat, phosphate, and calcium: a hypothesis. J Natl Cancer Inst. 1984;72(6):1323–1325. [PubMed] [Google Scholar]
- 20.Lamprecht SA, Lipkin M. Chemoprevention of colon cancer by calcium, vitamin D and folate: molecular mechanisms. Nat Rev Cancer. 2003;3(8):601–614. doi: 10.1038/nrc1144. [DOI] [PubMed] [Google Scholar]
- 21.Holt PR, Atillasoy EO, Gilman J, Guss J, Moss SF, Newmark H, Fan K, Yang K, Lipkin M. Modulation of abnormal colonic epithelial cell proliferation and differentiation by low-fat dairy foods: a randomized controlled trial. JAMA. 1998;280(12):1074–1079. doi: 10.1001/jama.280.12.1074. [DOI] [PubMed] [Google Scholar]
- 22.Fedirko V, Bostick RM, Flanders WD, Long Q, Sidelnikov E, Shaukat A, Daniel CR, Rutherford RE, Woodard JJ. Effects of vitamin D and calcium on proliferation and differentiation in normal colon mucosa: a randomized clinical trial. Cancer Epidemiol Biomark Prev. 2009;18(11):2933–2941. doi: 10.1158/1055-9965.Epi-09-0239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Holt PR, Wolper C, Moss SF, Yang K, Lipkin M. Comparison of calcium supplementation or low-fat dairy foods on epithelial cell proliferation and differentiation. Nutr Cancer. 2001;41(1–2):150–155. doi: 10.1080/01635581.2001.9680626. [DOI] [PubMed] [Google Scholar]
- 24.Ahearn TU, Shaukat A, Flanders WD, Rutherford RE, Bostick RM. A randomized clinical trial of the effects of supplemental calcium and vitamin D3 on the APC/β-catenin pathway in the normal mucosa of colorectal adenoma patients. Cancer Prev Res (Phila) 2012;5(10):1247–1256. doi: 10.1158/1940-6207.Capr-12-0292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rozen P, Lubin F, Papo N, Knaani J, Farbstein H, Farbstein M, Zajicek G. Calcium supplements interact significantly with long-term diet while suppressing rectal epithelial proliferation of adenoma patients. Cancer. 2001;91(4):833–840. doi: 10.1002/1097-0142(20010215)91:4<833::AID-CNCR1071>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- 26.Pufulete M. Intake of dairy products and risk of colorectal neoplasia. Nutr Res Rev. 2008;21(1):56–67. doi: 10.1017/s0954422408035920. [DOI] [PubMed] [Google Scholar]
- 27.Castro-Gómez P, Rodríguez-Alcalá LM, Monteiro KM, Ruiz AL, Carvalho JE, Fontecha J. Antiproliferative activity of buttermilk lipid fractions isolated using food grade and non-food grade solvents on human cancer cell lines. Food Chem. 2016;212:695–702. doi: 10.1016/j.foodchem.2016.06.030. [DOI] [PubMed] [Google Scholar]
- 28.Wesselink E, Kok DE, Bours MJL, de Wilt JHW, van Baar H, van Zutphen M, Geijsen A, Keulen ETP, Hansson BME, van den Ouweland J, Witkamp RF, Weijenberg MP, Kampman E, van Duijnhoven FJB. Vitamin D, magnesium, calcium, and their interaction in relation to colorectal cancer recurrence and all-cause mortality. Am J Clin Nutr. 2020;111(5):1007–1017. doi: 10.1093/ajcn/nqaa049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Baraghoshi D, Hawkins ML, Abdelaziz S, Park J, Wan Y, Fraser AM, Smith KR, Deshmukh V, Newman M, Rowe KG, Snyder J, Lloyd S, Samadder NJ, Hashibe M. Long-term risk of cardiovascular disease among colorectal cancer survivors in a population-based cohort study. J Clin Oncol. 2018;36(7):113–113. doi: 10.1200/JCO.2018.36.7_suppl.113. [DOI] [Google Scholar]
- 30.Cruijsen E, Jacobo Cejudo MG, Küpers LK, Busstra MC, Geleijnse JM. Dairy consumption and mortality after myocardial infarction: a prospective analysis in the Alpha Omega Cohort. Am J Clin Nutr. 2021;114(1):59–69. doi: 10.1093/ajcn/nqab026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wu G, Xue M, Zhao Y, Han Y, Zhang S, Zhang J, Li C, Xu J. 2020. Low circulating 25-hydroxyvitamin D level is associated with increased colorectal cancer mortality: a systematic review and dose-response meta-analysis. Biosci Rep. [DOI] [PMC free article] [PubMed]
- 32.National Institute for Public Health and the Environment (2020) The diet of the Dutch: results of the Dutch National Food Consumption Survey 2012–2016
- 33.Willett W, Rockström J, Loken B, Springmann M, Lang T, Vermeulen S, Garnett T, Tilman D, DeClerck F, Wood A, Jonell M, Clark M, Gordon LJ, Fanzo J, Hawkes C, Zurayk R, Rivera JA, De Vries W, Majele Sibanda L, Afshin A, Chaudhary A, Herrero M, Agustina R, Branca F, Lartey A, Fan S, Crona B, Fox E, Bignet V, Troell M, Lindahl T, Singh S, Cornell SE, Srinath Reddy K, Narain S, Nishtar S, Murray CJL. Food in the Anthropocene: the EAT-Lancet Commission on healthy diets from sustainable food systems. The Lancet. 2019;393(10170):447–492. doi: 10.1016/S0140-6736(18)31788-4. [DOI] [PubMed] [Google Scholar]
- 34.Schmidt P (2018) Promoting healthy and sustainable diets in the EU. European Economic and Social Committee: Section for Agriculture, Rural Development and the Environment
- 35.Health Council of the Netherlands (2011) Guidelines for a healthy diet: the ecological perspective
- 36.Shu XO, Yang G, Jin F, Liu D, Kushi L, Wen W, Gao YT, Zheng W. Validity and reproducibility of the food frequency questionnaire used in the Shanghai Women's Health Study. Eur J Clin Nutr. 2004;58(1):17–23. doi: 10.1038/sj.ejcn.1601738. [DOI] [PubMed] [Google Scholar]
- 37.Steinemann N, Grize L, Ziesemer K, Kauf P, Probst-Hensch N, Brombach C. Relative validation of a food frequency questionnaire to estimate food intake in an adult population. Food Nutr Res. 2017;61(1):1305193–1305193. doi: 10.1080/16546628.2017.1305193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Haftenberger M, Heuer T, Heidemann C, Kube F, Krems C, Mensink GBM. Relative validation of a food frequency questionnaire for national health and nutrition monitoring. Nutr J. 2010;9(1):36. doi: 10.1186/1475-2891-9-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Wu G, Xue M, Zhao Y, Han Y, Zhang S, Zhang J, Li C, Xu J. 2020. Low circulating 25-hydroxyvitamin D level is associated with increased colorectal cancer mortality: a systematic review and dose-response meta-analysis. Biosci Rep. [DOI] [PMC free article] [PubMed]
Supplementary Materials
Data Availability Statement
Because the data consist of identifying cohort information, some access restrictions apply, and therefore they cannot be made publicly available. Data will be shared with permission from the acting committee of the COLON study. Requests for data can be sent to dr. Fränzel van Duijnhoven, Division of Human Nutrition and Health, Wageningen University & Research, Netherlands (e-mail: franzel.vanduijnhoven@wur.nl).