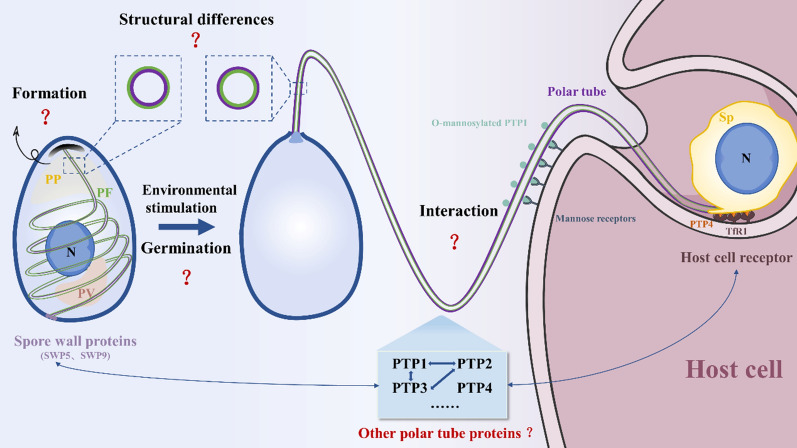
Abstract
Microsporidia are a class of obligate intracellular parasitic unicellular eukaryotes that infect a variety of hosts, even including humans. Although different species of microsporidia differ in host range and specificity, they all share a similar infection organelle, the polar tube, which is also defined as the polar filament in mature spores. In response to the appropriate environmental stimulation, the spore germinates with the polar filament everted, forming a hollow polar tube, and then the infectious cargo is transported into host cells via the polar tube. Hence, the polar tube plays a key role in microsporidian infection. Here, we review the origin, structure, composition, function, and application of the microsporidian polar tube, focusing on the origin of the polar filament, the structural differences between the polar filament and polar tube, and the characteristics of polar tube proteins. Comparing the three-dimensional structure of PTP6 homologous proteins provides new insight for the screening of additional novel polar tube proteins with low sequence similarity in microsporidia. In addition, the interaction of the polar tube with the spore wall and the host are summarized to better understand the infection mechanism of microsporidia. Due to the specificity of polar tube proteins, they are also used as the target in the diagnosis and prevention of microsporidiosis. With the present findings, we propose a future study on the polar tube of microsporidia.
Graphical Abstract
Keywords: Microsporidia, Polar tube, Polar filament, Structure, Polar tube proteins, Interaction, Diagnosis
Background
Microsporidia are a class of obligate intracellular parasitic unicellular eukaryotes. In the mid-nineteenth century, Nosema bombycis was first identified as the pathogen causing severe Pébrine disease in silkworms [1]. Initially, microsporidia were classified as protists [2]. With the phylogenetic analysis of the conserved gene (α-tubulin, β-tubulin, RNA polymerase II, heat shock protein 70), data have confirmed that microsporidia are a clade or sister group to fungi [3–13].
Microsporidia have a wide range of hosts, including invertebrates, vertebrates, and even humans [14]. Approximately half of all animal phyla have been reported to be infected with microsporidia [15]. The wide host range enables microsporidia to obtain more survival resources and opportunities [16–18]. More than 200 genera and 1700 species of microsporidia have been identified, 17 of which are known to infect humans, including Anncaliia connori, Anncaliia algerae, Anncaliia vesicularum, Enterocytozoon bieneusi, Encephalitozoon cuniculi, Encephalitozoon hellem, Encephalitozoon intestinalis, Pleistophora sp., Pleistophora ronneafiei, Trachipleistophora anthropophthera, Trachipleistophora hominis, Tubulinosema acridophagus, Vittaforma corneae, Nosema ocularum, Endoreticulatus sp., Microsporidium africanum, and Microsporidium ceylonensis [19–25]. Although human-infecting microsporidia were generally thought to infect immunocompromised patients, they have now been reported in immunocompetent individuals as well [23, 26–28].
Microsporidia differ in host range and host specificity; however, they all have a unique infection organelle, the polar tube, which is also defined as the polar filament in mature spores. In response to the appropriate environmental stimulation, the spore germinates with the polar filament everted, like a reversed finger of a glove, forming a hollow polar tube [29, 30]. The ejection process of the polar filament is so rapid that Encephalitozoon species take less than 500 ms for polar filament firing and passing of the infectious cargo through the polar tube, while the velocity of A. algerae emerging in the polar tube is up to 300 µm/s, with only 1.6 s required for the entire process [31]. Finally, the infectious sporoplasm is transported into the host cell to complete the microsporidian invasion [32]. However, the process by which the polar tube mediates the sporoplasm entry into host cells is still unclear. Currently, two phenomena have been observed: one in which the polar tube penetrates the host cell membrane to directly deliver the infectious sporoplasm into the host [33, 34], and the other wherein the polar tube attaches to the host cell membrane, forming invasive synapses to create a protected microenvironment for the sporoplasm, after which the sporoplasm is transported into the host cells by endocytosis [35]. Therefore, the polar tube plays an important role in microsporidian infection.
The polar tube is mainly composed of polar tube proteins (PTPs). With the development of proteomics technology, an increasing number of potential PTPs have been successfully identified [36], which lays a foundation for further research on the infection mechanism of microsporidia. In this paper, we review the origin, structure, composition, function, and application of the microsporidian polar tube in recent years, and provide new insights into the study of this unique infection organelle.
Origin
In general, the life cycle of microsporidia is divided into three stages: infective phase, proliferative phase, and sporogonic phase [37–39]. Microsporidian development starts with mature spores and ends with mature spores. Mature spore germination initiates the infective phase. One of the most widely accepted hypotheses regarding microsporidian germination is that under a series of stimuli, the water from the environment flows into mature spores to increase the intracellular osmotic pressure, which results in the disordering of the polaroplasts and swelling of the posterior vacuole, pushing the discharged polar filament. There are many factors (such as temperature, ultraviolet radiation, pH, metal ions, digestive enzymes, and so on) that trigger this germination process [40–49]. Next, the infectious sporoplasm transported into host cells begins the proliferative phase, which is the first stage of microsporidian intracellular parasitic life. The initial merogony division produces multiple merozoites, which transform into the sporont by binary fission or multiple fission. Then the spore wall begins to gradually thicken outside the plasma membrane, indicating the entry of the sporogonic stage. In this phase, the sporont undergoes binary fission to form a sporoblast, which finally develops as mature spores. The formation of the extrusion apparatus, involving the polar filament, polaroplast, and posterior vacuole, usually begins during this period. Finally, mature spores are released from the infected host to begin the next new life cycle [37].
Microsporidia do not have the typical Golgi complex (GC), but rather a GC-like structure, which is considered as clusters of vesicles derived from a nuclear envelope and endoplasmic reticulum (ER) in the early proliferate stage [50, 51]. And it was thought to be 300-nm networks of thin branching or varicose tubules in the late sporoblasts and young mature spores of Paranosema grylli and Paranosema locustae [51, 52]. The polar filament considered to originate from the GC-like structure was first observed in the early sporoblast [30, 52, 53]. Thiamine pyrophosphatase (TPPase), a histochemical marker of the GC, was found on the membranes and the high-electron-dense region of the polar filament in the fish microsporidium Glugea stephani, suggesting that the GC-like structure was indeed associated with the polar filament formation [53]. In addition, the ER is thought to participate in polar filament formation. It was found the signal of nucleoside diphosphatase (NDPase), a histochemical marker of ER, was in the core and outer sheath of the polar filament in G. stephani [54]. Weidner [55] suggested that the central core of the polar filament was from the GC-like structure vesicles, whereas the outer envelope was derived from the ER. Hence, the end of the tubular vesicle of the ER containing high-electron-dense material may be transported to the GC-like structure to gradually develop the extrusion apparatus [37].
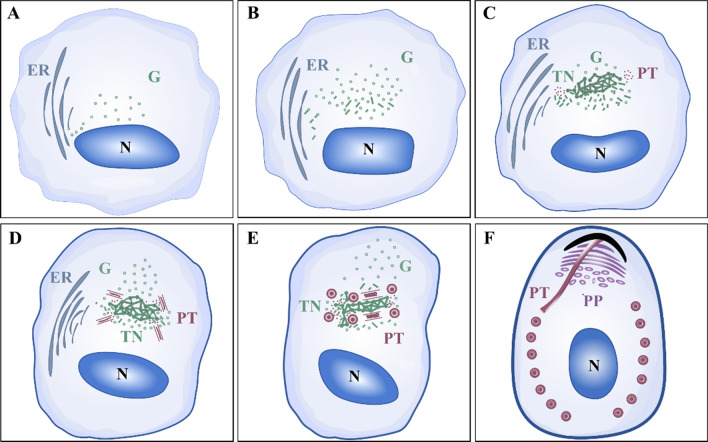
The formation of the polar filament is a relatively complex process, and a hypothesis has been proposed for microsporidian polar filament formation (Fig. 1A–F). Firstly, electron-dense vesicles appear close to the nucleus (Fig. 1A), like the matrix-arranged clusters of small vesicles (CsVm) in Saccharomyces cerevisiae as the “ER-to-Golgi intermediates” [51, 56, 57]. Then additional vesicles transform into tightly packed short tubules performing as cis-GC in sporonts (Fig. 1B), similar to tubular clusters of small vesicles (CsVt) in S. cerevisiae and vesicular tubular clusters (VTCs) in mammalian cells [51, 56, 57]. With the development of microsporidia, the tubular network (TN) becomes more prominent (Fig. 1C). In addition, the PTPs are synthesized and then are concentrated and modified in the TN [52, 58]. The PTPs gradually accumulate at the edge of the TN to connect with it, forming the core and envelope of the polar filament (Fig. 1D) [52]. Finally, the polar filament is gradually assembled into layers (Fig. 1E, F) [37], depending on its maturity [30, 59].
Fig. 1.
The hypothesis of the formation process of the microsporidian polar filament. A In the early proliferate stages, the GC-like structure (G) is considered as clusters of vesicles derived from the nucleus (N) and endoplasmic reticulum (ER). B The GC-like structure gradually appears as tubules. C With the tubular network (TN) formed, the PTPs are concentrated and modified in the TN and gradually accumulate at its edge. D With the assembly of PTPs, the core and envelope of the polar filaments are first formed. E, F They further develop into layers to develop mature polar filaments
Structure
The study of the polar filament structure began in 1960 [49]. The polar filament is composed of two parts: the anterior end is a vertical linear region (the manubrium portion), attached through an anchoring disc and surrounded by the polaroplast, and the other part is the helical region to protect the nucleus at the mid-posterior part of the mature spore [50]. The spiral number of polar filaments usually varies from four to 30 coils, depending on the microsporidian species [30, 31, 50, 60]. The polar filament is a right-handed helix packed at a special angle relative to the anterior–posterior (A–P) axis of the spore, which is 45° ± 10° in A. algerae and 37° ± 12° in Enc. hellem. This handedness may arise from the composition of polar filaments (such as PTPs) or the mechanical asymmetry involved in polar tube assembly [31]. The polar filament is composed of multiple concentric layers of different electron densities and thicknesses, mainly including the outer dense area, middle electron–lucent area, and inner dense area [29, 30, 49, 61]. Chioralia et al. [62] observed up to six concentric layers in the coiled region and three layers in the manubrium of the A. algerae polar filament. Kelley et al. [63] applied the waffle method to reveal the macrostructure of the polar filament in Enc. hellem: concentric circles were observed in axial view and cylinders in side view, and 2.5-nm-diameter bumps on the second cylindrical layer of the polar filament. It is widely accepted that the polar filament is located inside the plasma membrane; however, Cali et al. [64] observed the existence of a continuous network of membranes between the polar filament and spore cytoplasm of A. algerae, suggesting that the polar tube might be located outside the plasma membrane.
Under suitable stimulation, the polar filament instantly everts from the thinnest end of the spore wall, forming a hollow polar tube. The length of the extruded polar tube is generally 50–500 µm, which is more than twice the length of the polar filament [33, 47]. The diameter of the polar tube is about 0.1–0.2 µm [47, 65–67], and is so elastic that it can be enlarged up to 600 nm to facilitate the traversal of various intracellular cargo [33, 48, 67–69]. The polar tube has the following morphological features: tube-within-tube, cylinder, cylinder-within-cylinder, and unassembled PTPs [33, 46, 64]. Cryo-electron microscopy (cryo–EM) was first used to observe the polar tube of A. algerae, demonstrating that the PTPs were regularly arranged to form overlapping sheets, and a multilayer concentric ring structure composed of electron-dense membrane-like materials was found in the polar tube [68]. The surface of the polar tube had a row of ridges at intervals of 5–6 nm, rapidly increasing the diameter of the polar tube to transport the cargo. Some fine fibers were observed on the outermost layer of the polar tubes, which might be the glycosylated PTPs. At the tip of the extruded polar tube of A. algerae, a “J”-shaped hook existed [31, 68], which was also found in the extruded polar tube of N. bombycis [70]. During the process of germination, the polar tube first elongated to its maximum length , and then rapidly shortened once the sporoplasm was released [31].
Composition
The key role of the polar tube in microsporidian infection has led to increasing research interest in its composition. A polar tube is composed of proteins, and many polar tube proteins (PTPs) have been reported from microsporidia to date [35, 37, 71–74]. Taking advantage of the special solubility of the polar tube, Keohane et al. [75] treated the microsporidium Glugea americanus with 1% sodium dodecyl sulfate (SDS), 9 mol/L urea, and 2% dithiothreitol (DTT), and successively isolated four major proteins with molecular weight of 23, 27, 34, and 43 kDa. The monoclonal antibody specifically recognized the 43-kDa protein localized on the microsporidian polar tube [76–79]. Moreover, this protein contained several cysteine residues at the N-terminal and C-terminal, suggesting that the disulfide bond was important for PTP1 function [72, 75, 80]. Homologous proteins with similar solubility and molecular weight as other microsporidia were also identified and designated as polar tube protein 1 (PTP1) [75, 81, 82]. Moreover, PTP1 is an O-mannosylated glycoprotein. Pretreatment of RK13 cells with mannose was found to reduce the infectivity of the microsporidium Enc. hellem, implying that O-mannosylated PTP1 plays an important role in microsporidian infection [83]. The 35-kDa PTP2 protein was identified by a similar method [82]. PTP2 is more conserved than PTP1 among different microsporidian species, which all have the basic isoelectric point, high lysine content, and conserved cysteine residue sites [82, 84]. Interestingly, the ptp1 and ptp2 gene loci of different microsporidia are adjacent, and their neighbor genes are also relatively conserved in N. bombycis, N. ceranae, Enc. hellem, Enc. intestinalis, and so on [82, 84, 85]. PTP3 was screened from a complementary DNA (cDNA) library of Enc. cuniculi. The molecular weight of PTP3 is approximately 150 kDa. Unlike PTP1 and PTP2, PTP3 only dissolves in the presence of SDS [73]. Moreover, EcPTP1, EcPTP2, and EcPTP3 formed a large protein complex by cross-linkers, and EcPTP3 could interact not only with itself but also with EcPTP1 and EcPTP2 [86]. Hence, PTP3 has been hypothesized to act as a scaffolding protein for polar tube formation [73, 86].
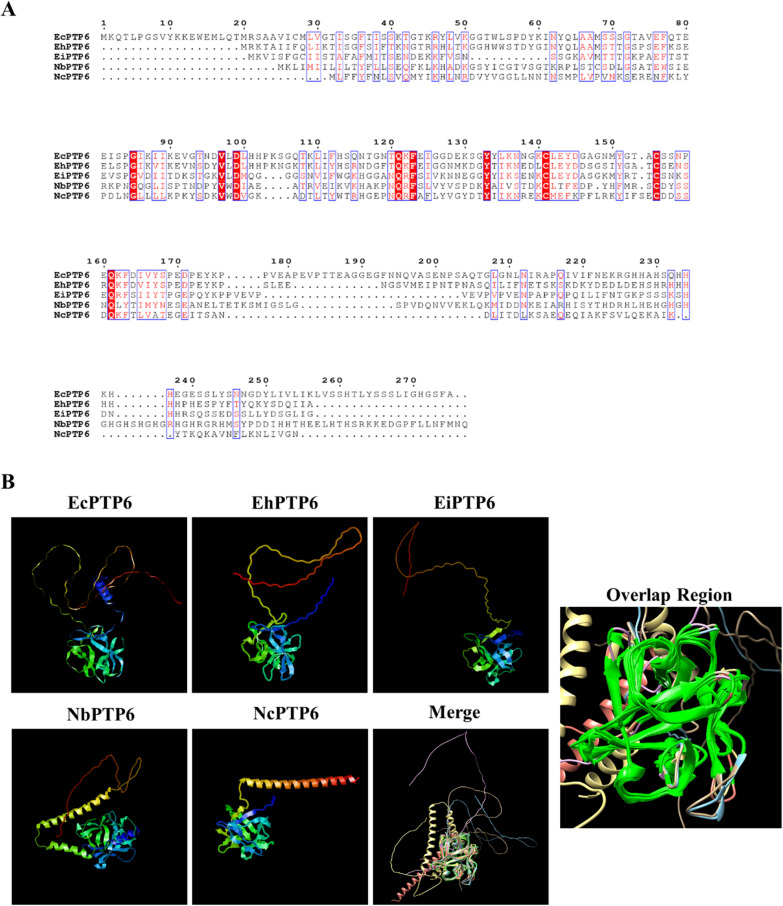
With the development of proteomics technology, novel PTPs have increasingly been screened and identified. In 2017, EhPTP4 was identified from Enc. hellem [35]. Unlike the previous three PTPs, EhPTP4 was specifically localized at the tip of the extruded polar tube and interacted with the special transferrin receptor 1 (TfR1) on the host cell membrane [35]. By comparing the sequence characteristics of PTP1–PTP4 from the genus Encephalitozoon, we found that the homologous PTPs have many common features (Table 1). Lv et al. first identified NbPTP6 from N. bombycis, which was rich in histidine and serine. Similar to EhPTP4, NbPTP6 could bind with the host cell surface, suggesting that the potential interaction receptor of NbPTP6 is present on the host cell membrane to promote microsporidian infection [74]. Recently, the polar filament and polar tube were isolated and purified from N. bombycis. For the analysis of the proteomic composition of these two structures, the candidate PTPs were screened to provide a reference for the identification of novel PTPs [36]. Although the more novel PTPs are screened, the sequence identity of PTPs from different microsporidia is so low that it hinders the finding of homologous proteins by blastp (protein–protein BLAST [Basic Local Alignment Search Tool]) analysis. The development of AlphaFold provides more possibilities for predicting the three-dimensional structure of proteins. Although amino acid sequence identity among the PTP6 homologous proteins from five different microsporidian species is not high (Fig. 2A), they show a highly conservative spatial structure by AlphaFold2 [87, 88]. The overlap region is concentrated at the 10th to 185th amino acid region of PTP6, whose secondary structure is mainly beta-strand and a small amount of random coil (Fig. 2B). In the future, combining the multiple sequence alignment with the protein structural alignment is a recommended method of exploring more novel homologous PTPs with low sequence similarity among different microsporidian species. However, no PTP structure has been resolved until now. Obtaining more information on the structural characteristics of PTPs provides insights into the evolution and function of these proteins, and also for revealing the PTP assembly on the microsporidian polar tube.
Table 1.
Comparison of the sequence characteristics of PTP1–PTP4 from the genus Encephalitozoon
| Protein | Gene symbol | Number of amino acids | pI | Major amino acid | Number of cysteine | Signal peptide | O-glycosylation site | N-glycosylation site | Subcellular location |
|---|---|---|---|---|---|---|---|---|---|
| EcPTP1 | ECU06_0250 | 395 | 4.5 | P | 17 | NF | Y | Y | On the whole PT of Enc. cuniculi [71] |
| EiPTP1 | Eint_060150 | 371 | 4.3 | P | 17 | Y | Y | NF | |
| EhPTP1 | EHEL_060170 | 413 | 4.2 | P | 19 | Y | Y | Y | |
| ErPTP1 | EROM_060160 | 380 | 4.4 | S | 15 | NF | Y | Y | |
| EcPTP2 | ECU06_0240 | 277 | 8.6 | K | 8 | Y | Y | NF | On the whole PT of Enc. cuniculi [82] |
| EiPTP2 | Eint_060140 | 275 | 8.6 | K | 8 | Y | Y | Y | |
| EhPTP2 | EHEL_060160 | 272 | 8.8 | K | 8 | Y | Y | Y | |
| ErPTP2 | EROM_060150 | 274 | 8.8 | K | 8 | Y | Y | Y | |
| EcPTP3 | ECU11_1440 | 1256 | 6.1 | A | 1 | Y | Y | Y | On the whole PT of Enc. cuniculi [73] |
| EiPTP3 | Eint_111330 | 1256 | 5.3 | E | 1 | Y | Y | Y | |
| EhPTP3 | EHEL_111330 | 1284 | 6.1 | A | 1 | Y | Y | Y | |
| ErPTP3 | EROM_111330 | 1254 | 6.5 | A | 1 | Y | Y | Y | |
| EcPTP4 | ECU07_1090 | 276 | 7.1 | E | 6 | Y | Y | Y | On the tip of the PT of Enc. hellem [35] |
| EiPTP4 | Eint_071050 | 279 | 7.3 | E | 6 | NF | Y | Y | |
| EhPTP4 | EHEL_071080 | 278 | 7.9 | E | 6 | Y | Y | Y | |
| ErPTP4 | EROM_071050 | 280 | 8.4 | E | 6 | NF | Y | Y |
“Ec” = Encephalitozoon cuniculi, “Ei” = Enc. intestinalis, “Eh” = Enc. hellem, “Er” = Enc. romalae, PT = polar tube
“Y” = YES, “NF” = not found
The signal peptide was predicted by SignalP 4.1 (https://services.healthtech.dtu.dk/services/SignalP-4.1/)
NETNGLYC (https://services.healthtech.dtu.dk/services/NetNGlyc-1.0/) and NETOGLYC (https://services.healthtech.dtu.dk/services/NetOGlyc-4.0/) were used to analyze N- and O-glycosylation sites
Fig. 2.
Comparison of the predicted PTP6 protein structure among different species of microsporidia by AlphaFold2. A Amino acid sequence alignment was generated with ClustalW (https://www.genome.jp/tools-bin/clustalw) and colored by ESPript 3.0 (https://espript.ibcp.fr/ESPript/cgi-bin/ESPript.cgi). B The PTP6 protein structure model of Enc. cuniculi (GenBank No. AGE95102.1), Enc. hellem (GenBank no. AFM98867.1), Enc. intestinalis (GenBank no. ADM12100.1), N. bombycis (GenBank no. EOB11485.1), and N. ceranae (GenBank no. EEQ82670.1) were predicted by AlphaFold2. The highlighted fluorescent green is the overlap region of PTP6 in different species of microsporidia produced by Chimera 1.16
Function
The polar tube acts as a bridge for transporting the infectious cargo into host cells, which is the key function of the polar tube [30, 33, 36, 37, 58, 89, 90]. Live-cell imaging of nuclei transport through the A. algerae polar tube showed that the nuclei were elongated, then recovered a globular shape after exiting the polar tube [31]. Lv et al. labeled the diplokaryons of N. bombycis and also found that they were elongated for transport in the polar tube [70]. However, Takvorian et al. [68] observed some circular diplokaryons surrounded by a membrane forming the oval or sperm-head-shaped structures in the A. algerae polar tube. Therefore, understanding how the nuclei are transported through the polar tube still requires further evidence. The anterior end of the extruded polar tube would form a “J”-shaped hook [68], where the droplet sporoplasm attached to the polar tube for several minutes [33]. In 2019, the interaction between sporoplasm surface protein 1 (EhSSP1) and EhPTP4 was found, contributing to sporoplasm adhesion at the tip of the polar tube [91].
Polar tubes may also interact with host surface proteins. Han et al. [35] analyzed the interaction between EhPTP4 and the host TfR1, which was the first identified host receptor for PTPs. TfR1 recombinant protein and anti-TfR1 antibody had an obvious inhibitory effect on the microsporidian infection rate in TfR1 knockout cells [35]. Moreover, it was also found that mannose-pretreated RK13 cells reduced Enc. hellem infection, implying that O-mannosylated PTP1 might play an important role in interacting with mannose receptors on host cells to promote microsporidian infection [83].
As the outermost structure of mature spores, the spore wall plays an important role in maintaining homeostasis and fixing the polar filament in mature spores. At present, studies on the interaction between the spore wall and polar tube are mainly in the microsporidium N. bombycis. As spore wall proteins, NbSWP5 and NbSWP9 were also reported to be localized on the extruded polar tube [92, 93], and NbSWP5 was found to interact with NbPTP2 and NbPTP3 [93]. Meanwhile, NbSWP9 interacted with NbPTP1, NbPTP2, and NbPTP3, which was considered to be a scaffolding protein to anchor the polar tube fixed to the spore wall for protection of the nucleus in mature spores [92].
Application
As a pathogen with such a broad infection range, microsporidia not only threaten human health, but also cause serious economic losses for the aquaculture industry [94]. Therefore, the diagnosis and control of microsporidia have become an important research target in recent years (Table 2).
Table 2.
PTPs as the target application in the diagnosis and control of microsporidia
| Applications | Target | Species | Host | Methods | References |
|---|---|---|---|---|---|
| Diagnosis | ptp2 (MT249228.1) | Enterocytozoon hepatopenaei | Shrimp | RPA and CRISPR–Cas12a | [98] |
| ptp2 (MT249228.1) | Enterocytozoon hepatopenaei | Shrimp | SYBR Green I fluorescence qPCR | [99] | |
| ptp3 (XM_002996713.1) | Nosema ceranae | Honey bee | LAMP | [100] | |
| ptp1 (AJ005666) | Encephalitozoon cuniculi | Human | PCR | [78] | |
| Control |
ptp3 |
Nosema ceranae | Honey bee | Oral administration of dsRNA | [103] |
| PTP1 (XP_003072984.1) | Encephalitozoon intestinalis | Human | Treat with anti-polar tube sera | [104] |
CRISPR, clustered regularly interspaced short palindromic repeats; dsRNA, double-stranded RNA; qPCR, quantitative polymerase chain reaction; RPA, recombinase polymerase amplification
The microscopic examination method for N. bombycis invented by Pasteur is still used in China today, and is a low-cost and simple operation; however, it has great limitations in sensitivity and specificity [95–97]. Therefore, PTPs are unique and conserved to utilize as the microsporidian diagnostic target in modern molecular detection methods. With ptp2 as the detection target, Kanitchinda et al. (2020) used recombinase polymerase amplification (RPA) and clustered regularly interspaced short palindromic repeats (CRISPR)–Cas12a fluorescence methods to detect Ent. hepatopenaei in the hepatopancreas tissue of shrimp [98]. Similarly, a SYBR Green I fluorescence quantitative PCR method targeting the ptp2 gene was established which, combined with Fluorescent Brightener 28 staining, could be applied to detect and analyze the quantity of Ent. hepatopenaei in the field shrimp [99]. Lannutti et al. [100] developed a loop-mediated isothermal amplification (LAMP) assay targeting the ptp3 gene for rapid detection and monitoring of N. ceranae in honey bees. Due to the length polymorphism and sequence diversity, ptp1 genes have gradually become a good candidate target for analysis of the species type of human-infecting microsporidia (such as Ent. bieneusi and Enc. cuniculi), providing a supplementary method for genotyping [77, 78]. According to a large-scale serosurvey reported, an immune response to the polar tube of Enc. intestinalis appeared in 8% of Dutch blood donors and 5% of pregnant women in France [101]. In this case, the serological diagnostic method of targeting PTPs (based on immunofluorescence antibody staining, enzyme-linked immunoassay [ELISA], and western blotting) is an important tool for studying the pathogenicity and epidemiology of human-infecting microsporidia.
In light of the limited drugs available for treatment (only albendazole and fumagillin are currently approved for microsporidiosis) [102], PTPs have potential importance in the control of microsporidia. Rodriguez Garcia et al. [103] silenced the ptp3 gene expression of N. ceranae in bees by oral administration of ptp3 double-stranded RNA (dsRNA) to achieve microsporidian load reduction, which provided a novel idea for the prevention and control of microsporidia in RNA interference (RNAi) therapy. In addition, human anti-polar tube sera could partially reduce the microsporidium Enc. intestinalis infection in vitro, which was also thought to be a potential microsporidiosis treatment [104].
Conclusion
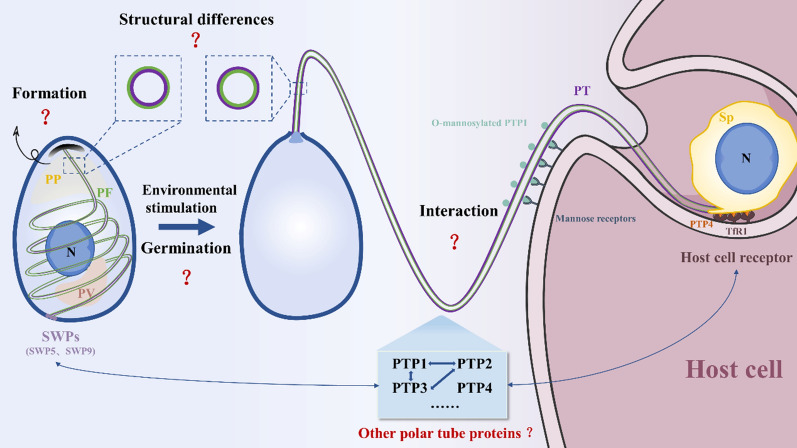
For one and a half centuries, as the special and unique infection organelle, the polar tube has been the research focus of microsporidia. With the continuous development of proteomics and bioinformatics technology, an increasing number of potential PTPs will be screened, laying the foundation for analyzing the composition of the polar tube. In addition, with the help of cryo-EM technology, the structural characteristics of the polar tube and polar filament are gradually becoming clear. However, many aspects of the microsporidian polar tube are not yet understood. In the future, the study and application of the polar tube can be carried out from the following perspectives (Fig. 3): (1) The polar filament is considered to form in the early sporoblast, but the key factor for activation of the polar filament formation is unclear. (2) To clarify the process of the polar filament transformation into the polar tube, the characteristics of PTP assembly on the polar filament need to be demonstrated. (3) The study of the interaction between the polar tube and host is needed in order to reveal the infection mechanism of microsporidia. (4) Finally, additional novel PTPs can be applied for the diagnosis and control of microsporidia.
Fig. 3.
Summary. In response to external environmental stimulation, the spore germinates with the polar filament everted, forming a hollow polar tube, and then the infectious cargo is transported to inject into host cells through the polar tube. During this process, the polar tube interacts with both the microsporidian spore wall proteins and host receptors, and there are still many unknown aspects that need to be solved. PT polar tube, PF polar filament, PP polaroplast, PV posterior vacuole, Sp sporoplasm, TfR1 transferrin receptor 1
Acknowledgements
The authors would like to thank Dr. Youpeng Fan and Mr. Yunlin Tang for their comments on the use of AlphaFold2.
Abbreviations
- PTPs
Polar tube proteins
- GC
Golgi complex
- Cryo-EM
Cryo-electron microscopy
- TfR1
Transferrin receptor 1
Author contributions
YQC designed the project, analyzed the data, and wrote the manuscript. QL and HJL provided help with the figure processing. ZKX and LYH collected the material and information. LQ gave advice on the manuscript. GQP and ZYZ revised the manuscript. MXL designed the project and provided overall study supervision. All authors read and approved the final manuscript.
Funding
This study was supported by grants from the National Natural Science Foundation of China (Grant No. 31402138), the Natural Science Foundation of Chongqing, China (cstc2021jcyj-cxttX0005), and the Opening fund of State Key Laboratory of Silkworm Genome Biology (SKLSGB-ORP202105).
Availability of data and materials
No data were collected for this review. All data and information synthesized in the review are already published and publicly available, and those publications are properly cited in the submission.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Sazmand A, Bahari A, Papi S, Otranto D. Parasitic diseases of equids in Iran (1931–2020): a literature review. Parasit Vectors. 2020;13:586. doi: 10.1186/s13071-020-04472-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Vossbrinck CR, Maddox JV, Friedman S, Debrunner-Vossbrinck BA, Woese CR. Ribosomal RNA sequence suggests microsporidia are extremely ancient eukaryotes. Nature. 1987;326:411–414. doi: 10.1038/326411a0. [DOI] [PubMed] [Google Scholar]
- 3.Lee SC, Corradi N, Byrnes EJ, 3rd, Torres-Martinez S, Dietrich FS, Keeling PJ, et al. Microsporidia evolved from ancestral sexual fungi. Curr Biol. 2008;18:1675–1679. doi: 10.1016/j.cub.2008.09.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Keeling PJ, Doolittle WF. Alpha-tubulin from early-diverging eukaryotic lineages and the evolution of the tubulin family. Mol Biol Evol. 1996;13:1297–1305. doi: 10.1093/oxfordjournals.molbev.a025576. [DOI] [PubMed] [Google Scholar]
- 5.Keeling PJ, Luker MA, Palmer JD. Evidence from beta-tubulin phylogeny that microsporidia evolved from within the fungi. Mol Biol Evol. 2000;17:23–31. doi: 10.1093/oxfordjournals.molbev.a026235. [DOI] [PubMed] [Google Scholar]
- 6.Hirt RP, Logsdon JM, Healy B, Dorey MW, Doolittle WF, Embley TM. Microsporidia are related to fungi: evidence from the largest subunit of RNA polymerase II and other proteins. Proc Natl Acad Sci U S A. 1999;96:580–585. doi: 10.1073/pnas.96.2.580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hirt RP, Healy B, Vossbrinck CR, Canning EU, Embley TM. A mitochondrial Hsp70 orthologue in Vairimorpha necatrix: molecular evidence that microsporidia once contained mitochondria. Curr Biol. 1997;7:995–998. doi: 10.1016/s0960-9822(06)00420-9. [DOI] [PubMed] [Google Scholar]
- 8.Arisue N, Sachez LB, Weiss LM, Muller M, Hashimoto T. Mitochondrial-type hsp70 genes of the amitochondriate protists, Giardia intestinalis, Entamoeba histolytica and two microsporidians. Parasitol Int. 2002;51:9–16. doi: 10.1016/s1383-5769(01)00093-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Germot A, Philippe H, LeGuyader H. Evidence for loss of mitochondria in microsporidia from a mitochondrial-type hsp70 in Nosema locustae. Mol Biochem Parasitol. 1997;87:159–168. doi: 10.1016/s0166-6851(97)00064-9. [DOI] [PubMed] [Google Scholar]
- 10.Germot A, Philippe H. Critical analysis of eukaryotic phylogeny: a case study based on the hsp70 family. J Eukaryot Microbiol. 1999;46:116–124. doi: 10.1111/j.1550-7408.1999.tb04594.x. [DOI] [PubMed] [Google Scholar]
- 11.Peyretaillade E, Broussolle V, Peyret P, Metenier G, Gouy M, Vivares CP. Microsporidia, amitochondrial protists, possess a 70-kDa heat shock protein gene of mitochondrial evolutionary origin. Mol Biol Evol. 1998;15:683–689. doi: 10.1093/oxfordjournals.molbev.a025971. [DOI] [PubMed] [Google Scholar]
- 12.James TY, Kauff F, Schoch CL, Matheny PB, Hofstetter V, Cox CJ, et al. Reconstructing the early evolution of fungi using a six-gene phylogeny. Nature. 2006;443:818–822. doi: 10.1038/nature05110. [DOI] [PubMed] [Google Scholar]
- 13.Keeling PJ. Congruent evidence from alpha-tubulin and beta-tubulin gene phylogenies for a zygomycete origin of microsporidia. Fungal Genet Biol. 2003;38:298–309. doi: 10.1016/s1087-1845(02)00537-6. [DOI] [PubMed] [Google Scholar]
- 14.Weiss LM. Clinical syndromes associated with microsporidiosis. In: Weiss LM, Becnell JJ, editors. Microsporidia: pathogens of opportunity. Wiley Blackwell; 2014. pp. 371–401. [Google Scholar]
- 15.Murareanu BM, Sukhdeo R, Qu R, Jiang J, Reinke AW. Generation of a microsporidia species attribute database and analysis of the extensive ecological and phenotypic diversity of microsporidia. MBio. 2021;12:e0149021-e. doi: 10.1128/mBio.01490-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Koehler AV, Springer YP, Randhawa HS, Leung TLF, Keeney DB, Poulin R. Genetic and phenotypic influences on clone-level success and host specialization in a generalist parasite. J Evol Biol. 2012;25:66–79. doi: 10.1111/j.1420-9101.2011.02402.x. [DOI] [PubMed] [Google Scholar]
- 17.Clavel J, Julliard R, Devictor V. Worldwide decline of specialist species: toward a global functional homogenization? Front Ecol Environ. 2011;9:222–228. [Google Scholar]
- 18.Lange B, Kaufmann AP, Ebert D. Genetic, ecological and geographic covariables explaining host range and specificity of a microsporidian parasite. J Anim Ecol. 2015;84:1711–1719. doi: 10.1111/1365-2656.12421. [DOI] [PubMed] [Google Scholar]
- 19.Desportes I, Lecharpentier Y, Galian A, Bernard F, Cochandpriollet B, Lavergne A, et al. Occurrence of a new microsporidan Enterocytozoon bieneusi n. g., n. sp., in the Enterocytes of a human patient with AIDS. J Protozool. 1985;32:250–254. doi: 10.1111/j.1550-7408.1985.tb03046.x. [DOI] [PubMed] [Google Scholar]
- 20.Visvesvara GS. In vitro cultivation of microsporidia of clinical importance. Clin Microbiol Rev. 2002;15:401–413. doi: 10.1128/CMR.15.3.401-413.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Juarez SI, Putaporntip C, Jongwutiwes S, Ichinose A, Yanagi T, Kanbara H. In vitro cultivation and electron microscopy characterization of Trachipleistophora anthropophthera isolated from the cornea of an AIDS patient. J Eukaryot Microbiol. 2005;52:179–190. doi: 10.1111/j.1550-7408.2005.00024.x. [DOI] [PubMed] [Google Scholar]
- 22.Franzen C, Nassonova FS, Scholmerich J, Issi IV. Transfer of the members of the genus Brachiola (microsporidia) to the genus Anncaliia based on ultrastructural and molecular data. J Eukaryot Microbiol. 2006;53:26–35. doi: 10.1111/j.1550-7408.2005.00066.x. [DOI] [PubMed] [Google Scholar]
- 23.Weber R, Bryan RT. Microsporidial infections in immunodeficient and immunocompetent patients. Clin Infect Dis. 1994;19:517–521. doi: 10.1093/clinids/19.3.517. [DOI] [PubMed] [Google Scholar]
- 24.Didier ES, Didier PJ, Friedberg DN, Stenson SM, Orenstein JM, Yee RW, et al. Isolation and characterization of a new human microsporidian, Encephalitozoon hellem (n. sp.), from 3 AIDS patients with keratoconjunctivitis. J Infect Dis. 1991;163:617–621. doi: 10.1093/infdis/163.3.617. [DOI] [PubMed] [Google Scholar]
- 25.Han B, Pan GQ, Weiss LM. Microsporidiosis in humans. Clin Microbiol Rev. 2021;34:e0001020. doi: 10.1128/CMR.00010-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Didier ES, Weiss LM. Microsporidiosis: Not just in aids patients. Curr Opin Infect Dis. 2011;24:490–495. doi: 10.1097/QCO.0b013e32834aa152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lewis NL, Francis IC, Hawkins GS, Coroneo MT. Bilateral microsporidial keratoconjunctivitis in an immunocompetent non-contact lens wearer. Cornea. 2003;22:374–376. doi: 10.1097/00003226-200305000-00018. [DOI] [PubMed] [Google Scholar]
- 28.Metge S, Van Nhieu JT, Dahmane D, Grimbert P, Foulet F, Sarfati C, et al. A case of Enterocytozoon bieneusi infection in an HIV-negative renal transplant recipient. Eur J Clin Microbiol Infect Dis. 2000;19:221–223. doi: 10.1007/s100960050463. [DOI] [PubMed] [Google Scholar]
- 29.Lom J. On the structure of extruded microsporidian polar filament. Z Parasitenk. 1972;38:200–213. [Google Scholar]
- 30.Xu Y, Weiss LM. The microsporidian polar tube: a highly specialised invasion organelle. Int J Parasitol. 2005;35:941–953. doi: 10.1016/j.ijpara.2005.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jaroenlak P, Cammer M, Davydov A, Sall J, Usmani M, Liang FX, et al. 3-dimensional organization and dynamics of the microsporidian polar tube invasion machinery. PLoS Pathog. 2020;16:e1008738. doi: 10.1371/journal.ppat.1008738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Franzen C. How do microsporidia invade cells? Folia Parasitol (Praha) 2005;52:36–40. doi: 10.14411/fp.2005.005. [DOI] [PubMed] [Google Scholar]
- 33.Weidner E. Ultrastructural study of microsporidian invasion into cells. Z Parasitenk. 1972;40:227–242. doi: 10.1007/BF00329623. [DOI] [PubMed] [Google Scholar]
- 34.Takvorian PM, Weiss LM, Cali A. The early events of Brachiola algerae (microsporidia) infection: Spore germination, sporoplasm structure, and development within host cells. Folia Parasitol (Praha) 2005;52:118–129. doi: 10.14411/fp.2005.015. [DOI] [PubMed] [Google Scholar]
- 35.Han B, Polonais V, Sugi T, Yakubu R, Takvorian PM, Cali A, et al. The role of microsporidian polar tube protein 4 (PTP4) in host cell infection. Plos Pathog. 2017;13:e1006341. doi: 10.1371/journal.ppat.1006341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lv Q, Zhou B, Liao H, He X, Chen Y, Pan G, et al. Proteomic profile of polar filament and polar tube from fungal pathogen microsporidium Nosema bombycis provides new insights into its unique invasion organelle. J Proteomics. 2022;263:104617. doi: 10.1016/j.jprot.2022.104617. [DOI] [PubMed] [Google Scholar]
- 37.Weiss LM, Delbac F, Russell Hayman J, Pan G, Dang X. The microsporidian polar tube and spore wall. In: Weiss LM, Becnell JJ, editors. Microsporidia: pathogens of opportunity. Wiley Blackwell; 2014. pp. 261–306. [Google Scholar]
- 38.Canning EU, Hulls RH. A microsporidan infection of Anopheles gambiae Giles, from Tanzania, interpretation of its mode of transmission and notes on Nosema infections in mosquitoes. J Protozool. 1970;17:531–539. doi: 10.1111/j.1550-7408.1970.tb04723.x. [DOI] [PubMed] [Google Scholar]
- 39.Canning EU, Hollister W. In vitro and in vivo investigations of human microsporidia. J protozool. 1991;38:631–635. [PubMed] [Google Scholar]
- 40.Han B, Takvorian PM, Weiss LM. Invasion of host cells by microsporidia. Front Microbiol. 2020;11:172. doi: 10.3389/fmicb.2020.00172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Undeen A, Avery S. Effect of anions on the germination of Nosema algerae (Microspora: Nosematidae) spores. J Insect Pathol. 1988;52:84–89. [Google Scholar]
- 42.Frixione E, Ruiz L, Cerbon J, Undeen AH. Germination of Nosema algerae (Microspora) spores: Conditional inhibition by D2O, ethanol and Hg2+ suggests dependence of water influx upon membrane hydration and specific transmembrane pathways. J Eukaryot Microbiol. 1997;44:109–116. doi: 10.1111/j.1550-7408.1997.tb05946.x. [DOI] [PubMed] [Google Scholar]
- 43.Frixione E, Ruiz L, Undeen AH. Monovalent cations induce microsporidian spore germination in vitro. J Eukaryot Microbiol. 1994;41:464–468. [Google Scholar]
- 44.Troemel ER, Becnel JJ. Genome analysis and polar tube firing dynamics of mosquito-infecting microsporidia. Fungal Genet Biol. 2015;83:41–44. doi: 10.1016/j.fgb.2015.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Undeen AH, Frixione E. The role of osmotic pressure in the germination of Nosema algerae spores. J Protozool. 1990;37:561–567. doi: 10.1111/j.1550-7408.1990.tb01265.x. [DOI] [PubMed] [Google Scholar]
- 46.Weidner E. The microsporidian spore invasion tube. III. Tube extrusion and assembly. J Cell Biol. 1982;93:976–979. doi: 10.1083/jcb.93.3.976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Frixione E, Ruiz L, Santillan M, Devargas LV, Tejero JM, Undeen AH. Dynamics of polar filament discharge and sporoplasm expulsion by microsporidian spores. Cell Motil Cytoskel. 1992;22:38–50. [Google Scholar]
- 48.Lom J, Vavra J. The mode of sporoplasm extrusion in microsporidian spores. Acta Protozool. 1963;1:81–92. [Google Scholar]
- 49.Huger A. Electron microscope study on the cytology of a microsporidian spore by means of ultrathin sectioning. J Insect Pathol. 1960;2:81–105.
- 50.Wittner M. Structure of the Microsporidia. In: Wittner M, Weiss LM, editors. The microsporidia and microsporidiosis. ASM press; 1999. pp. 7–84. [Google Scholar]
- 51.Sokolova Y, Snigirevskaya E, Morzhina E, Skarlato S, Mironov A, Komissarchik Y. Visualization of early Golgi compartments at proliferate and sporogenic stages of a microsporidian Nosema grylli. J Eukaryot Microbiol. 2001;48:86S–S87. doi: 10.1111/j.1550-7408.2001.tb00464.x. [DOI] [PubMed] [Google Scholar]
- 52.Beznoussenko GV, Dolgikh VV, Seliverstova EV, Semenov PB, Tokarev YS, Trucco A, et al. Analogs of the Golgi complex in microsporidia: structure and avesicular mechanisms of function. J Cell Sci. 2007;120:1288–1298. doi: 10.1242/jcs.03402. [DOI] [PubMed] [Google Scholar]
- 53.Takvorian PM, Cali A. Enzyme histochemical identification of the Golgi apparatus in the microsporidian Glugea stephani. J Eukaryot Microbiol. 1994;41:63S–S64. [PubMed] [Google Scholar]
- 54.Takvorian PM, Cali A. Polar tube formation and nucleoside diphosphatase activity in the microsporidian Glugea stephani. J Eukaryot Microbiol. 1996;43:102S–S103. doi: 10.1111/j.1550-7408.1996.tb05025.x. [DOI] [PubMed] [Google Scholar]
- 55.Weidner E. Ultrastructural study of microsporidian development. Z Zellforsch Mikrosk Anat. 1970;105:33–54. [PubMed] [Google Scholar]
- 56.Morin-Ganet MN, Rambourg A, Deitz SB, Franzusoff A, Kepes F. Morphogenesis and dynamics of the yeast Golgi apparatus. Traffic. 2000;1:56–68. doi: 10.1034/j.1600-0854.2000.010109.x. [DOI] [PubMed] [Google Scholar]
- 57.Sokolova YY, Mironov AA. Structure and function of the Golgi organelle in parasitic protists. In: Mironov AA, Pavelka M, editors. The Golgi apparatus. Springer; 2008. pp. 647–74. [Google Scholar]
- 58.Keohane EM, Weiss LM. The structure, function, and composition of the microsporidian polar tube. In: Wittner M, Weiss LM, editors. The microsporidia and microsporidiosis. ASM press; 1999. pp. 196–224. [Google Scholar]
- 59.Chioralia G, Trammer T, Kampen H, Seitz HM. Relevant criteria for detecting microsporidia in stool specimens. J Clin Microbiol. 1998;36:2279–2283. doi: 10.1128/jcm.36.8.2279-2283.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Cali A, Takvorian PM. The microsporidia: Pathology in man and occurrence in nature. SE Asian J Trop Med. 2004;35:58–64. [Google Scholar]
- 61.Vinckier D, Porchet E, Vivier E, Vávra J, Torpier G. A freeze-fracture study of microsporidia (Protozoa: Microspora): II. The extrusion apparatus: polar filament, polaroplast, posterior vacuole. Eur J Protistol. 1993;29:370–80. doi: 10.1016/S0932-4739(11)80399-8. [DOI] [PubMed] [Google Scholar]
- 62.Chioralia G, Trammer T, Maier WA, Seitz HM. Morphologic changes in Nosema algerae (Microspora) during extrusion. Parasitol Res. 1998;84:123–131. doi: 10.1007/s004360050368. [DOI] [PubMed] [Google Scholar]
- 63.Kelley K, Raczkowski AM, Klykov O, Jaroenlak P, Bobe D, Kopylov M, et al. Waffle method: a general and flexible approach for improving throughput in FIB-milling. Nat Commu. 2022;13:1857. doi: 10.1038/s41467-022-29501-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cali A, Weiss LM, Takvorian PM. Brachiola algerae spore membrane systems, their activity during extrusion, and a new structural entity, the multilayered interlaced network, associated with the polar tube and the sporoplasm. J Eukaryot Microbiol. 2002;49:164–174. doi: 10.1111/j.1550-7408.2002.tb00361.x. [DOI] [PubMed] [Google Scholar]
- 65.Kudo R, Daniels E. An electron microscope study of the spore of a microsporidian Thelohania californica. J Protozool. 1963;10:112–120. doi: 10.1111/j.1550-7408.1963.tb01645.x. [DOI] [PubMed] [Google Scholar]
- 66.Lom J, Corliss JO. Ultrastructural observations on the development of the microsporidian protozoon Plistophora hyphessobryconis schaperclaus. J Protozool. 1967;14:141–152. [Google Scholar]
- 67.Weidner E. Microsporidian spore invasion tube ultrastructure, isolation, and characterization of protein comprising tube. J Cell Biol. 1976;71:23–34. doi: 10.1083/jcb.71.1.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Takvorian PM, Han B, Cali A, Rice WJ, Gunther L, Macaluso F, et al. An ultrastructural study of the extruded polar tube of Anncaliia algerae (microsporidia) J Eukaryot Microbiol. 2020;67:28–44. doi: 10.1111/jeu.12751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Olsen P, Rice W, Liu T. In vitro germination of Nosema apis spores under conditions favorable for the generation and maintenance of sporoplasms. J Invertebr Pathol. 1986;47:65–73. [Google Scholar]
- 70.Lv Q, Chen Y, Qi L, Hong L, Xie Z, Liao H, et al. Microsporidia dressing up: the spore polaroplast transport through the polar tube and transformation into the sporoplasm membrane. bioRxiv. 2023;538940. [DOI] [PMC free article] [PubMed]
- 71.Delbac F, Peyret P, Méténier G, David D, Danchin A, Vivarès CP. On proteins of the microsporidian invasive apparatus: complete sequence of a polar tube protein of Encephalitozoon cuniculi. Mol Microbiol. 1998;29:825–834. doi: 10.1046/j.1365-2958.1998.00975.x. [DOI] [PubMed] [Google Scholar]
- 72.Delbac F, David D, Metenier G, Vivares C. First complete amino acid sequence of a polar tube protein in a microsporidian species Encephalitozoon cuniculi. J Eukaryot Microbiol. 1997;44:77S. doi: 10.1111/j.1550-7408.1997.tb05791.x. [DOI] [PubMed] [Google Scholar]
- 73.Peuvel I, Peyret P, Metenier G, Vivares CP, Delbac F. The microsporidian polar tube: Evidence for a third polar tube protein (PTP3) in Encephalitozoon cuniculi. Mol Biochem Parasitol. 2002;122:69–80. doi: 10.1016/s0166-6851(02)00073-7. [DOI] [PubMed] [Google Scholar]
- 74.Lv Q, Wang LJ, Fan YP, Meng XZ, Liu KK, Zhou BQ, et al. Identification and characterization a novel polar tube protein (NbPTP6) from the microsporidian Nosema bombycis. Parasit Vectors. 2020;13:475. doi: 10.1186/s13071-020-04348-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Keohane EM, Takvorian PM, Cali A, Tanowitz HB, Wittner M, Weiss LM. Identification of a microsporidian polar tube protein reactive monoclonal antibody. J Eukaryot Microbiol. 1996;43:26–31. doi: 10.1111/j.1550-7408.1996.tb02468.x. [DOI] [PubMed] [Google Scholar]
- 76.Peuvel I, Delbac F, Metenier G, Peyret P, Vivares CP. Polymorphism of the gene encoding a major polar tube protein PTP1 in two microsporidia of the genus Encephalitozoon. Parasitology. 2000;121:581–587. doi: 10.1017/s0031182000006910. [DOI] [PubMed] [Google Scholar]
- 77.Haro M, del Aguila C, Fenoy S, Henriques-Gil N. Intraspecies genotype variability of the microsporidian parasite Encephalitozoon hellem. J Clin Microbiol. 2003;41:4166–4171. doi: 10.1128/JCM.41.9.4166-4171.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Xiao L, Li L, Visvesvara GS, Moura H, Didier ES, Lal AA. Genotyping Encephalitozoon cuniculi by multilocus analyses of genes with repetitive sequences. J Clin Microbiol. 2001;39:2248–2253. doi: 10.1128/JCM.39.6.2248-2253.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Weiss LM. Microsporidia: emerging pathogenic protists. Acta Trop. 2001;78:89–102. doi: 10.1016/s0001-706x(00)00178-9. [DOI] [PubMed] [Google Scholar]
- 80.Keohane EM, Orr GA, Zhang HS, Takvorian PM, Cali A, Tanowitz HB, et al. The molecular characterization of the major polar tube protein gene from Encephalitozoon hellem, a microsporidian parasite of humans. Mol Biochem Parasitol. 1998;94:227–236. doi: 10.1016/s0166-6851(98)00071-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Delbac F, Duffieux F, David D, Metenier G, Vivares CP. Immunocytochemical identification of spore proteins in two microsporidia, with emphasis on extrusion apparatus. J Eukaryot Microbiol. 1998;45:224–231. doi: 10.1111/j.1550-7408.1998.tb04529.x. [DOI] [PubMed] [Google Scholar]
- 82.Delbac F, Peuvel I, Metenier G, Peyretaillade E, Vivares CP. Microsporidian invasion apparatus: identification of a novel polar tube protein and evidence for clustering of ptp1 and ptp2 genes in three Encephalitozoon species. Infect Immun. 2001;69:1016–1024. doi: 10.1128/IAI.69.2.1016-1024.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Xu Y, Takvorian PM, Cali A, Orr G, Weiss LM. Glycosylation of the major polar tube protein of Encephalitozoon hellem, a microsporidian parasite that infects humans. Infect Immun. 2004;72:6341–6350. doi: 10.1128/IAI.72.11.6341-6350.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Polonais V, Prensier G, Metenier G, Vivares CP, Delbac F. Microsporidian polar tube proteins: highly divergent but closely linked genes encode PTP1 and PTP2 in members of the evolutionarily distant Antonospora and Encephalitozoon groups. Fungal Genet Biol. 2005;42:791–803. doi: 10.1016/j.fgb.2005.05.005. [DOI] [PubMed] [Google Scholar]
- 85.Slamovits CH, Fast NM, Law JS, Keeling PJ. Genome compaction and stability in microsporidian intracellular parasites. Curr Biol. 2004;14:891–896. doi: 10.1016/j.cub.2004.04.041. [DOI] [PubMed] [Google Scholar]
- 86.Bouzahzah B, Nagajyothi F, Ghosh K, Takvorian PM, Cali A, Tanowitz HB, et al. Interactions of Encephalitozoon cuniculi polar tube proteins. Infect Immun. 2010;78:2745–2753. doi: 10.1128/IAI.01205-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Varadi M, Anyango S, Deshpande M, Nair S, Natassia C, Yordanova G, et al. AlphaFold protein structure database: massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 2021;50:D439–D444. doi: 10.1093/nar/gkab1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Jumper J, Evans R, Pritzel A, Green T, Figurnov M, Ronneberger O, et al. Highly accurate protein structure prediction with AlphaFold. Nature. 2021;596:583–589. doi: 10.1038/s41586-021-03819-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Keohane EM, Weiss LM. Characterization and function of the microsporidian polar tube: a review. Folia Parasitol (Praha) 1998;45:117–127. [PubMed] [Google Scholar]
- 90.Kramer JP. Variations among the spores of the microsporidian Perezia pyraustae Palliot. Am Mid Nat. 1960;64:485–487. [Google Scholar]
- 91.Han B, Ma YF, Tu V, Tomita T, Mayoral J, Williams T, et al. Microsporidia interact with host cell mitochondria via voltage-dependent anion channels using sporoplasm surface protein 1. MBio. 2019;10:e01944–e2019. doi: 10.1128/mBio.01944-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Yang D, Pan L, Peng P, Dang X, Li C, Li T, et al. Interaction between SWP9 and polar tube proteins of the microsporidian Nosema bombycis and function of SWP9 as a scaffolding protein contribute to polar tube tethering to the spore wall. Infect Immun. 2017;85:e00872–e916. doi: 10.1128/IAI.00872-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Li Z, Pan G, Li T, Huang W, Chen J, Geng L, et al. SWP5, a spore wall protein, interacts with polar tube proteins in the parasitic microsporidian Nosema bombycis. Eukaryot Cell. 2012;11:229–237. doi: 10.1128/EC.05127-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Stentiford GD, Becnel JJ, Weiss LM, Keeling PJ, Didier ES, Williams BAP, et al. Microsporidia emergent pathogens in the global food chain. Trends Parasitol. 2016;32:336–348. doi: 10.1016/j.pt.2015.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Franzen C, Muller A. Molecular techniques for detection, species differentiation, and phylogenetic analysis of microsporidia. Clin Microbiol Rev. 1999;12:243–285. doi: 10.1128/cmr.12.2.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Procop GW. Molecular diagnostics for the detection and characterization of microbial pathogens. Clin Infect Dis. 2007;45:S99–S111. doi: 10.1086/519259. [DOI] [PubMed] [Google Scholar]
- 97.Field AS. Light microscopic and electron microscopic diagnosis of gastrointestinal opportunistic infections in HIV-positive patients. Pathology. 2002;34:21–35. doi: 10.1080/00313020120111230. [DOI] [PubMed] [Google Scholar]
- 98.Kanitchinda S, Srisala J, Suebsing R, Prachumwat A, Chaijarasphong T. CRISPR–Cas fluorescent cleavage assay coupled with recombinase polymerase amplification for sensitive and specific detection of Enterocytozoon hepatopenaei. Biotechnol Rep (Amst) 2020;27:e00485. doi: 10.1016/j.btre.2020.e00485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wang L, Lv Q, He Y, Gu R, Zhou B, Chen J, et al. Integrated qPCR and staining methods for detection and quantification of Enterocytozoon hepatopenaei in shrimp Litopenaeus vannamei. Microorganisms. 2020;8:1366. doi: 10.3390/microorganisms8091366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Lannutti L, Mira A, Basualdo M, Rodriguez G, Erler S, Silva V, et al. Development of a Loop-mediated isothermal amplification (LAMP) and a direct lamp for the specific detection of Nosema ceranae, a parasite of honey bees. Parasitol Res. 2020;119:3947–3956. doi: 10.1007/s00436-020-06915-w. [DOI] [PubMed] [Google Scholar]
- 101.vanGool T, Vetter JCM, Weinmayr B, VanDam A, Derouin F, Dankert J. High seroprevalence of Encephalitozoon species in immunocompetent subjects. J Infect Dis. 1997;175:1020–1024. doi: 10.1086/513963. [DOI] [PubMed] [Google Scholar]
- 102.Wei J, Fei Z, Pan G, Weiss LM, Zhou Z. Current therapy and therapeutic targets for microsporidiosis. Front Microbiol. 2022;13:835390. doi: 10.3389/fmicb.2022.835390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Rodríguez-García C, Evans JD, Li W, Branchiccela B, Li JH, Heerman MC, et al. Nosemosis control in European honey bees, Apis mellifera, by silencing the gene encoding Nosema ceranae polar tube protein 3. J Exp Biol. 2018;221:jeb184606. doi: 10.1242/jeb.184606. [DOI] [PubMed] [Google Scholar]
- 104.Peek R, Delbac F, Speijer D, Polonais V, Greve S, Wentink-Bonnema E, et al. Carbohydrate moieties of microsporidian polar tube proteins are targeted by immunoglobulin G in immunocompetent individuals. Infect Immuni. 2005;73:7906–7913. doi: 10.1128/IAI.73.12.7906-7913.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data were collected for this review. All data and information synthesized in the review are already published and publicly available, and those publications are properly cited in the submission.