Abstract
Biallelic pathogenic variants in the surfactant protein (SP)-B gene (SFTPB) have been associated with fatal forms of interstitial lung diseases (ILD) in newborns and exceptional survival in young children. We herein report the cases of two related adults with pulmonary fibrosis due to a new homozygous SFTPB pathogenic variant, c.582G>A p.(Gln194=). In vitro transcript studies showed that this SFTPB synonymous pathogenic variant induces aberrant splicing leading to three abnormal transcripts with the preservation of the expression of a small proportion of normal SFTPB transcripts. Immunostainings on lung biopsies of the proband showed an almost complete loss of SP-B expression. This hypomorphic splice variant has thus probably allowed the patients’ survival to adulthood while inducing an epithelial cell dysfunction leading to ILD. Altogether, this report shows that SFTPB pathogenic variants should be considered in atypical presentations and/or early-onset forms of ILD particularly when a family history is identified.
Subject terms: Respiratory tract diseases, Genetics research
Introduction
Surfactant protein (SP)-B deficiency has been associated with fatal forms of respiratory distress in newborns [1–3]. Patients usually present at term, after an unremarkable pregnancy, with immediate respiratory failure, diffuse alveolar or interstitial opacities and a fatal evolution towards refractory hypoxemia in a few days or weeks despite maximal levels of ventilation. In these newborns, most of the reported pathogenic variants are loss-of-function variations leading to premature stop codons, the most frequently reported one being the c.361delinsGAA, p.(Pro121Glufs*95) [4–7]. We herein report a homozygous SFTPB hypomorphic splice pathogenic variant in two related adults with pulmonary fibrosis.
Cases presentation
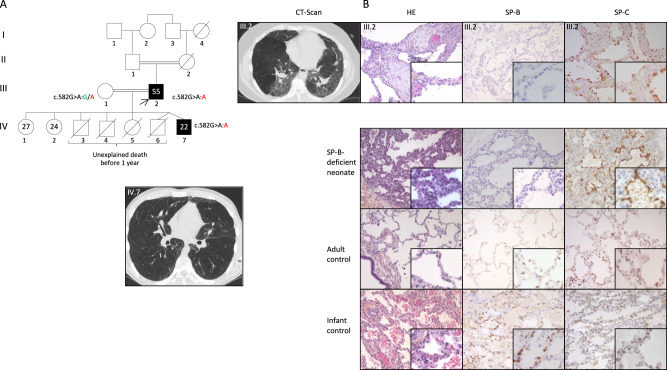
The proband, a non-smoker man born to a consanguineous union, presented with fibrosing interstitial lung disease (ILD) with non-predominant features of alveolar hemorrhage evolving since the age of 34 years. He had no history of neonatal respiratory failure nor childhood respiratory symptoms. He first presented with dyspnea and cough, leading to a thoracic CT scan showing mosaicism with ground glass opacities and emphysema (Fig. 1A). The lung biopsy (unavailable sample) reported lung fibrosis. His respiratory status deteriorated with, at the age of 42 years, an FEV1 of 2.21 L (64%); FVC 3.13 L (74%), TLCO 45% and an impaired 6-min walking test with a nadir of SaO2 at 72% for 304 m (50%). Despite treatments with oral corticosteroids, hydroxychloroquine, azathioprine (quickly stopped because of a lack of efficacy) and azithromycin, he required continuous oxygen therapy since the age of 44 years and benefited from bi-pulmonary lung transplantation at the age of 51 years. The histopathology of the explanted lungs was consistent with a usual interstitial pneumonia pattern (Fig. 1B). The genetic analysis by targeted-capture NGS sequencing of telomerase-related and surfactant-related genes identified no pathogenic variant in telomerase genes but a synonymous homozygous SFTPB variation: c.582G>A, p.(Gln194=). The patient had 7 children, also from a consanguineous union (Fig. 1A, III.1, heterozygous for the reported variant), including 4 who died before one year of age from an unknown cause and a boy who presented neonatal respiratory distress at term with a pneumothorax. At 4 months, this boy was hospitalized for respiratory syncytial virus bronchiolitis. At 10 months, alveolo-interstitial opacities have persisted and a working diagnosis of mild alveolar hemorrhage was first proposed based on a moderated anemia (Hb 11 g/dL) and 30% of hemosiderin-laden macrophages at bronchoalveolar lavage cytology analysis. No CT scan was available and he had no lung biopsy to confirm the diagnosis. After 2 years of corticosteroid treatment, he was stable. At 16 years, the CT scan showed mild ground glass opacities, and large areas of attenuations without emphysema or cysts. At the last evaluation (aged 22), the patient presented a dyspnea on exertion with distension (forced vital capacity 5.2 L, 120% of expected values, FEV1 3.85 L, 101%) and his CT scan showed mild ILD with ground glass opacifications, mosaic attenuation with an area of decreased attenuation and subtle distortion (Fig. 1A). He was also found to be homozygous for the SFTPB c.582G>A variation.
Fig. 1. Family tree, CT scan and histological features.
A Genealogical tree of the described family. Consanguineous unions are indicated with double lines. Black symbols show subjects with pulmonary fibrosis. The age at the time of the study is mentioned within symbols. The arrow indicates the proband. The genotypes are provided with the reference nucleotide in green and the pathogenic variant in red. The CT scan of the proband (III.2) at 51 years (before lung transplantation) shows parenchymal distortion, ground glass opacities, centrilobular nodules and mosaic attenuation with area of decreased attenuation and pulmonary fibrosis. The CT scan of his son (IV.7) at 16 years shows mild ground glass opacities, large areas of attenuation especially in the lingula and subtle distortion. B The native lung explant examination of the proband (III.2) using Haematoxylin and eosin (HE) staining shows fibroblastic foci alternating with less affected areas consistent with a usual interstitial pneumonia pattern. Pulmonary hemosiderosis was not observed nor pulmonary alveolar proteinosis. The SP-B-deficient neonate showed thickened septa with moderate pulmonary alveolar proteinosis and the adult and infant controls showed a normal lung parenchyma. SP-B antibody targets pro-SP-B as it has been developed against amino acids 121–197. SP-B immunostaining (magnification ×20 and ×40) showed an almost complete loss of SP-B expression in the patient’s type 2 alveolar epithelial cells (AEC)2, a complete loss of SP-B expression in SP-B-deficient neonate and a normal intracytoplasmic granular AEC2 SP-B expression in both adult and infant controls. Note that the anti-SP-B antibody does not target the peptide corresponding to the in-phase deleted transcript (p.(Asp132_Gln194del)) which lacks most of the epitope (Fig. 2). SP-C antibody has been developed against full-length SP-C (amino acids 1-197) and can thus detect both pro-SP-C and mature SP-C. SP-C immunostaining (magnification ×20 and ×40) showed a higher degree of expression in the patient’s and the SP-B-deficient neonate’s type 2 alveolar epithelial cells than in adult and infant controls. HE haematoxylin and eosin, AEC alveolar epithelial cell.
Material and methods
Transcript study
To assess the pathogenicity of the identified c.582G>A variation involving the last nucleotide of exon 5 of SFTPB, we generated SFTPB mini-gene constructs. As the studied variant is synonymous, a cDNA expression vector could not be used for protein functional studies. Moreover, the genomic DNA of SFTPB is long (NM_000542.5, 13,436 bp) and could hardly be used for the construction of a genomic expression vector. Thus, the genomic region spanning exons 4–6 of human SFTPB (NM_000542.5) was cloned into the pcDNA3.1_V5_His_TOPO vector resulting in plasmid pSFTPB_WT. The SFTPB c.582G>A variation and the c.582+1G>T pathogenic variant (used as a positive control fully disrupting the donor splice site of intron 5, MaxEntScan score: 1.56) were introduced into pSFTPB_WT by site-directed mutagenesis and were respectively designated pSFTPB_mut and pSFTPB_ctr.
After FuGene transfection of the plasmids in A549 cells, total RNA was isolated at day 2 (RNeasy mini kit, Qiagen). cDNAs were generated from mRNA using oligodT with the Transcriptor High Fidelity cDNA Synthesis Kit and PCR-amplified before agarose gel electrophoresis and Sanger sequencing.
Immunohistochemistry assay
The lung biopsy from the proband was compared to three controls: a healthy child and adult as well as an SP-B-deficient neonate carrying a biallelic loss-of-function pathogenic variants in SFTPB (c.[75G>A];[361delinsGAA], p.[(Trp25*)];[(Pro121Glufs*95)]). Haematoxylin and eosin stainings were performed. Immunostaining assays were performed on a Leica Bond Platform using an anti-SP-B polyclonal antibody (HPA062148, Sigma, 1/20 targeting the 121–197 amino acids) and an anti-SP-C antibody (SC-13979, 1/100 targeting both SP-C and pro-SP-C). No frozen tissue was available to assess ex vivo RNA expression.
Submission of data to public databases
Variant and phenotypes of the proband and his son have been described in LOVD database under respective URL and https://databases.lovd.nl/shared/individuals/00435123 and https://databases.lovd.nl/shared/individuals/00435124, and accession numbers 00435123 and 00435124.
Results
Functional consequences of the SFTPB c.582G>A p.(Gln194=) variation
The identified SFTPB c.582G>A variation has never been described in patients nor in the gnomAD variation database. In silico analysis predicts it to weaken the splice-donor site of intron 5 (MaxEntScan score: 5.85 vs. 10.07).
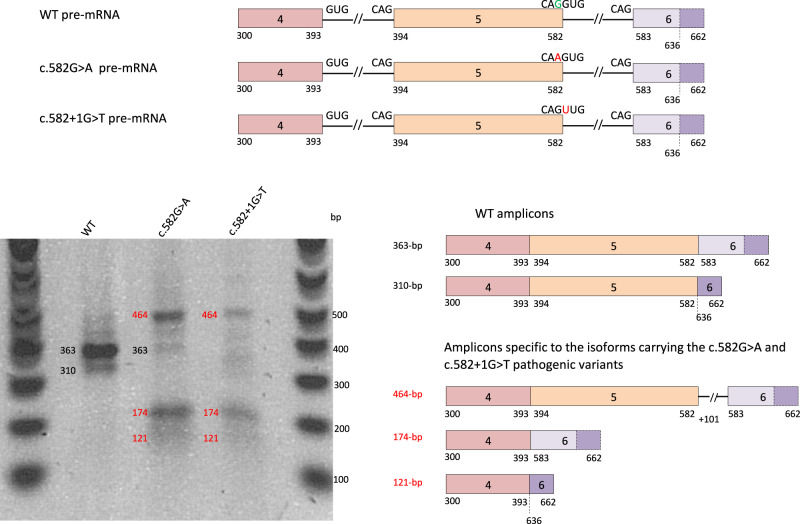
Transcripts isolated from cells transfected with pSFTPB_WT yielded a 363-bp amplicon corresponding to normal splicing and a 310-bp one resulting from the use of a cryptic splice-acceptor site in exon 6 (Fig. 2). A similar assay performed with pSFTPB_mut generated three molecular species in addition to the 363-bp expected SFTPB amplicon. The first two amplicons lead to premature Stop codons: a 464-bp one [p.(Asp195Valfs*54)] resulting from the use of a cryptic splice-donor site located within intron 5 leading to the retention of the first 101-bp of this intron and a 121-bp one [p.(Asp132Glyfs*39)] resulting from the use of the cryptic splice-acceptor site in exon 6. As SFTPB encompasses 11 exons, it is highly likely that, in vivo, these transcripts leading to premature Stop codons trigger the nonsense-mediated mRNA decay pathway and lead to the absence of protein production. The third amplicon, a 174-bp one [p.(Asp132_Gln194del)] lacking exon 5 corresponds to an in-phase deletion located in the pro-protein (SP-B mature protein: amino acids 201–279). This deletion includes two major protein cleavage sites, certainly preventing the production of a mature SP-B protein (Supplementary Fig. 1) [8, 9]. Whether or not this shorter peptide, if not degraded, could trigger unfolded protein response and induce an epithelial cell dysfunction remains to be ascertained.
Fig. 2. In vitro assessment of SFTPB splicing defects associated with c.582G>A p.(Gln194=).
SFTPB exons 4–6 pre-mRNA corresponding to plasmids pSFTPB-WT, pSFTPB_mut and pSFTPB_ctr are represented. The reference nucleotide is in green and the pathogenic variant is in red. The dotted line highlights a cryptic acceptor splice site in exon 6 (nucleotide 636). After transfection of A549 cells with the above-mentioned plasmids, the migration on a 1.5% agarose-BET gel of the RT-PCR products obtained from extracted RNAs is shown. The main amplicons characterized by Sanger sequencing and obtained from the normal and mutant constructs are represented on the right of the gel image. Two amplicons were observed for the pSFTPB_WT: a 363 bp (canonical splicing) and a 310 bp (using exon 6 cryptic splice-acceptor site corresponding to putative p.(Asp195Glyfs*39)). Both the c.582G>A (pSFTPB_mut) and the c.582+1G>T (pSFTPB_ctr) pathogenic variants resulted in three specific and aberrant amplicons, i.e., from top to bottom: one 464 bp transcript related to the retention of the first 101 bp of intron 5 and predicting a frameshift with a premature Stop codon in exon 6 (p.(Asp195Valfs*54)), and two 174 and 121 bp amplicons lacking exon 5 and corresponding to the use of intron 5 canonical splice-acceptor site (p.(Asp132_Gln194del) in-phase deletion), or exon 6 cryptic splice-acceptor site (p.(Asp132Glyfs*39)) respectively. The data are representative from 3 independent experiments. WT wild type.
With a positive control of abnormal splicing—the c.582+1G>T variant predicted to fully disrupt the splice-donor site of intron 5—we did not detect any normal transcript, but observed the same three aberrant transcripts.
Altogether, the c.582G>A pathogenic variant is therefore consistent with a drastic alteration of the expression of SP-B with, however, a residual expression of normal SP-B.
Lung tissue expression of SP-B and SP-C
SP-B immunostaining revealed an almost complete loss of SP-B expression in the lung from the proband. A control experiment performed on a lung biopsy sample from a neonate with an SP-B deficiency due to a biallelic loss-of-function pathogenic variants in SFTPB showed a complete loss of SP-B expression (Fig. 1B). The SFTPB c.582G>A pathogenic variant also had an impact on SP-C metabolism, as observed by the higher expression of SP-C and pro-SP-C in both the proband and the SP-B deficient neonate compared to both controls.
Discussion
We herein report for the first time two adult patients from a single family with biallelic SFTPB pathogenic variants. Very rare cases of prolonged survival have been described in SP-B deficiency after infant lung transplantation [10–12]. Only one case has been reported with a delayed presentation in a 4-month infant carrying the homozygous c.706C>T p.(Arg236Cys) missense pathogenic variant [13]. The patient was alive at the age of 8 years with oxygen supplementation. Three other patients with prolonged survival also presented at birth with severe respiratory distress. Two of them were homozygous for the c.465G>T p.(Gly155=) splice variant which creates a strong donor splice site within exon 5 (MaxEntScan score 9.80) and one of them was still alive at school age [14]. The third one was homozygous for the missense c.770T>C, p.Leu257Pro pathogenic variant and died at 13 months from respiratory failure [15]. A mouse model of transgenic mice displaying a reduced expression of SP-B showed a different parenchymal aspect than in the described patient with an enlargement of the tissue airspaces at 6 weeks (corresponding to a young adult in humans) [16]. Like in the family we are currently reporting, hypomorphic splice pathogenic variants may allow the persistence of a variable proportion of normal transcripts leading to a residual SP-B function which could explain the various age at onset (adult in the proband and neonate in his son) and the long-term survival of the patients. The possible epithelial cell dysfunction and the partial SP-B deficiency probably explain the fibrosing ILD in both patients [17]. Interestingly, ILD was initially diagnosed as mild alveolar hemorrhage in both patients. It is likely that the alveolar bleeding could be due either to chronic dysfunction of the alveolar epithelium eroding the alveolar capillaries or to pulmonary hypertension [18].
Genetic causes of familial ILD have been better understood over the past decade and are mostly related to telomerase-related genes and surfactant-related genes pathogenic variants [19–21]. Pathogenic variants in surfactant genes that were hitherto associated with ILD in childhood are increasingly linked to young-adult onset ILD, and should be considered in atypical presentations of ILD particularly when a family history is identified [20, 21]. In the former patients, whatever the age of the patient is, a genetic analysis of the genes involved in surfactant metabolism could increase the number of described cases and the awareness of adult forms of surfactant genes pathogenic variants.
Supplementary information
Acknowledgements
We thank the patients and their family. We thank the French national networks for rare lung diseases: Centre de référence des maladies respiratoires rares (RespiRare), Centre de référence des maladies pulmonaires rares (OrphaLung) and Filière de soins pour les maladies respiratoires rares (RespiFIL). The ILD cohort has been developed in collaboration with the Rare Disease Cohort (RaDiCo)-ILD project (ANR-10-COHO-0003), the ERS Clinical research collaboration for chILD-EU and the COST Innovative Grant OpenILD CIG16125.
Author contributions
TD and NN performed the functional studies and wrote the manuscript that was reviewed by all the authors. ML contributed to the data analysis. PD provided his expertise in functional studies. GP and ALB were in charge of the patients. VN and FDL performed the molecular diagnosis under the supervision of ML and SA. AC performed the histological analyses.
Funding
Our work is supported by the Legs Poix from the Chancellerie des Universités (grants 2013 n°1305, 2014 n°1405, 2015 n°1015, 2016 n°2077, 2017 n°DP2017/1860 and 2022 n°2022000594).
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Written informed consents were obtained from the two patients. Ethical approval was obtained from the local authorities under the number 060916.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Marie Legendre, Nadia Nathan.
Supplementary information
The online version contains supplementary material available at 10.1038/s41431-023-01413-w.
References
- 1.Nogee LM, de Mello DE, Dehner LP, Colten HR. Brief report: deficiency of pulmonary surfactant protein B in congenital alveolar proteinosis. N Engl J Med. 1993;328:406–10. doi: 10.1056/NEJM199302113280606. [DOI] [PubMed] [Google Scholar]
- 2.Nogee LM, Garnier G, Dietz HC, Singer L, Murphy AM, deMello DE, et al. A mutation in the surfactant protein B gene responsible for fatal neonatal respiratory disease in multiple kindreds. J Clin Invest. 1994;93:1860–3. doi: 10.1172/JCI117173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nathan N, Borensztajn K, Clement A. Genetic causes and clinical management of pediatric interstitial lung diseases. Curr Opin Pulm Med. 2018;24:253–9. doi: 10.1097/MCP.0000000000000471. [DOI] [PubMed] [Google Scholar]
- 4.Hamvas A. Inherited surfactant protein-B deficiency and surfactant protein-C associated disease: clinical features and evaluation. Semin Perinatol. 2006;30:316–26. doi: 10.1053/j.semperi.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 5.Nathan N, Taam RA, Epaud R, Delacourt C, Deschildre A, Reix P, et al. A national internet-linked based database for pediatric interstitial lung diseases: the French network. Orphanet J Rare Dis. 2012;7:40. doi: 10.1186/1750-1172-7-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kurath-Koller S, Resch B, Kraschl R, Windpassinger C, Eber E. Surfactant protein B deficiency caused by homozygous C248X mutation-a case report and review of the literature. AJP Rep. 2015;5:e53–9. doi: 10.1055/s-0035-1545668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Turcu S, Ashton E, Jenkins L, Gupta A, Mok Q. Genetic testing in children with surfactant dysfunction. Arch Dis Child. 2013;98:490–5. doi: 10.1136/archdischild-2012-303166. [DOI] [PubMed] [Google Scholar]
- 8.Ueno T, Linder S, Na C-L, Rice WR, Johansson J, Weaver TE. Processing of pulmonary surfactant protein B by napsin and cathepsin H. J Biol Chem. 2004;279:16178–84. doi: 10.1074/jbc.M312029200. [DOI] [PubMed] [Google Scholar]
- 9.Brasch F, Ochs M, Kahne T, Guttentag S, Schauer-Vukasinovic V, Derrick M, et al. Involvement of napsin A in the C- and N-terminal processing of surfactant protein B in type-II pneumocytes of the human lung. J Biol Chem. 2003;278:49006–14. doi: 10.1074/jbc.M306844200. [DOI] [PubMed] [Google Scholar]
- 10.Palomar LM, Nogee LM, Sweet SC, Huddleston CB, Cole FS, Hamvas A. Long-term outcomes after infant lung transplantation for surfactant protein B deficiency related to other causes of respiratory failure. J Pediatr. 2006;149:548–53. doi: 10.1016/j.jpeds.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 11.Eldridge WB, Zhang Q, Faro A, Sweet SC, Eghtesady P, Hamvas A, et al. Outcomes of lung transplantation for infants and children with genetic disorders of surfactant metabolism. J Pediatr. 2017;184:157–64.e2. doi: 10.1016/j.jpeds.2017.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ballard PL, Nogee LM, Beers MF, Ballard RA, Planer BC, Polk L, et al. Partial deficiency of surfactant protein B in an infant with chronic lung disease. Pediatrics. 1995;96:1046–52. doi: 10.1542/peds.96.6.1046. [DOI] [PubMed] [Google Scholar]
- 13.López-Andreu JA, Hidalgo-Santos AD, Fuentes-Castelló MA, Mancheño-Franch N, Cerón-Pérez JA, Esteban-Ricós MJ, et al. Delayed presentation and prolonged survival of a child with surfactant protein B deficiency. J Pediatr. 2017;190:268–70.e1. doi: 10.1016/j.jpeds.2017.07.009. [DOI] [PubMed] [Google Scholar]
- 14.Dunbar AE, 3rd, Wert SE, Ikegami M, Whitsett JA, Hamvas A, White FV, et al. Prolonged survival in hereditary surfactant protein B (SP-B) deficiency associated with a novel splicing mutation. Pediatr Res. 2000;48:275–82. doi: 10.1203/00006450-200009000-00003. [DOI] [PubMed] [Google Scholar]
- 15.Hamouda S, Trabelsi I, de Becdelièvre A, Boussetta K. Difficulties in the treatment of an infant survivor with inherited surfactant protein-B deficiency in Tunisia. Ann Thorac Med. 2022;17:132–5. doi: 10.4103/atm.atm_445_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nesslein LL, Melton KR, Ikegami M, Na C-L, Wert SE, Rice WR, et al. Partial SP-B deficiency perturbs lung function and causes air space abnormalities. Am J Physiol Lung Cell Mol Physiol. 2005;288:L1154–61. doi: 10.1152/ajplung.00392.2004. [DOI] [PubMed] [Google Scholar]
- 17.Katzen J, Beers MF. Contributions of alveolar epithelial cell quality control to pulmonary fibrosis. J Clin Invest. 2020;130:5088–99.. doi: 10.1172/JCI139519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang JY, Young LR. Insights into the pathogenesis of pulmonary fibrosis from genetic diseases. Am J Respir Cell Mol Biol. 2022;67:20–35. doi: 10.1165/rcmb.2021-0557TR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Garcia CK. Idiopathic pulmonary fibrosis: update on genetic discoveries. Proc Am Thorac Soc. 2011;8:158–62. doi: 10.1513/pats.201008-056MS. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.van Moorsel CHM, van der Vis JJ, Grutters JC. Genetic disorders of the surfactant system: focus on adult disease. Eur Respir Rev. 2021;30:200085. doi: 10.1183/16000617.0085-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sutton RM, Bittar HT, Sullivan DI, Silva AG, Bahudhanapati H, Parikh AH, et al. Rare surfactant-related variants in familial and sporadic pulmonary fibrosis. Hum Mutat. 2022;43:2091–101. doi: 10.1002/humu.24476. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.