Abstract
Background
Randomized controlled trials (RCTs) reported contrasting results about reverse left ventricular remodeling (LVR) after sodium-glucose co-transporter-2 inhibitors (SGLT2i) therapy in patients with heart failure (HF).
Methods and results
We performed a metanalysis of RCTs of SGLT2i administration in HF outpatients published until June 2022 searching four electronic databases. The protocol has been published in PROSPERO. Primary LVR outcome was change in absolute LV end-diastolic (LVEDV) and end-systolic volume (LVESV) from baseline to study endpoint. Secondary outcomes included changes in LVEDV and LVESV indexed to body surface area, LV Mass index (LVMi), LV ejection fraction (LVEF), and N-terminal pro-B-type natriuretic peptide (NTproBNP). Mean differences (MDs) with 95% CIs were pooled. A total of 9 RCTs (1385 patients) were analyzed. All of them reported data on LVEF. Six trials reported data on LVESV and LVEDV (n = 951); LVMi was available in 640. SGLT2i treatment significantly reduced LVEDV [MD= -10.59 ml (-17.27; -3.91), P = 0.0019], LVESV [MD= -8.80 ml (-16.91; -0.694), P = 0.0334], and LVMI [MD= -5.34 gr/m2 (-9.76; -0.922), P = 0.0178], while LVEF significantly increased [MD = + 1.98% (0.67; 0.306), P = 0.0031]. By subgroup analysis, the beneficial effects of SGLT2i on LVEF did not differ by imaging method used, time to follow-up re-evaluation, or HF phenotype. Reduction in LV volumes tended to be greater in HF with reduced EF (HFrEF) than in those with preserved EF (HFpEF), while the opposite was observed for LVMi.
Conclusions
Treatment with SGLT2i significantly reversed cardiac volumes, improving LV systolic function and LV mass, particularly in HFrEF patients.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12933-023-01970-w.
Keywords: SGLT2i; Cardiac magnetic resonance imaging; Echocardiography, Cardiac remodelling; HFrEF, HFpEF
Introduction
Initially introduced as a major therapeutic advance in the field of type-2 diabetes mellitus (T2DM) to aid glycemic control, sodium-glucose co-transporter-2 inhibitor (SGLT2i) drugs have also demonstrated to strongly reduce the incidence of a composite endpoint of cardiovascular death/HF re-hospitalizations both in heart failure (HF) patients with reduced ejection fraction (HFrEF) [1, 2] and, more recently, in HF patients with preserved EF (HFpEF) [3, 4]. Interestingly, in all these studies the beneficial effects of SGLT2i on outcome were observed in patients regardless of T2DM status [1–4], thus making these drugs a new major weapon in the arsenal of available medications used to fight HF.
How SGLT2-inhibitors exert such striking clinical benefits in HF is not completely known, and their beneficial effects cannot be simply explained by their actions on glycemic control, or as osmotic diuretics [5]. Recently, randomized controlled trials (RCT), enrolling patients with T2DM with or without HF, have explored the capability of these drugs to favorably impact left ventricular remodeling (LVR) indices (LV volumes, myocardial mass, and LV systolic function), either assessed by echocardiography or cardiovascular magnetic resonance (CMR), providing contrasting results [6–18]. Therefore, these aspects remain controversial.
We performed an updated metanalysis of RCTs in patients with HF to evaluate the effects of chronic administration of SGLT2-inhibitors on LV volumes, mass, ejection fraction (EF), and N-terminal pro-B-type natriuretic peptide (NTproBNP).
Methods
Data source
This meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The protocol has been published in the PROSPERO International prospective register of systematic reviews (CRD42023264057). Using the search algorithm detailed in the Supplementary Material Data file, four electronic databases [PUBMED, Web Of Science (WOS), Cochrane Central Register of Controlled Trials, OVID MEDLINE] were searched for RCTs published until June 2022 without any restriction regarding study duration and sample size. Reference lists of all identified studies were also manually searched for further relevant investigations. Reviews, meta-analyses, case reports, summary articles, letters without data, conference abstracts, editorials, posters, nonhuman studies, and non-English articles were excluded.
Study Selection and eligibility criteria
Two investigators (P.B. and A.M.) independently reviewed the titles and abstracts of all citations retrieved, selected eligible studies, and extracted valuable data from downloaded articles, according to the inclusion and exclusion criteria. Discrepancies were resolved by a third author. Studies were eligible for the analysis in the following cases: (1) they were full-text and relevant data could be acquired; (2) they had to provide baseline and follow-up data for LV end-diastolic (LVEDV) and end-systolic volumes (LVESV), or LV ejection fraction, either assessed by CMR or echocardiography; (3) randomized comparison between SGLT2-inhibitors and placebo or active control.
At least two studies reporting outcome variables were required to be eligible for the analysis. Studies that did not provide enough data to analyze the effect on LV remodeling parameters were excluded, as were studies enrolling patients with type-1 diabetes mellitus (T1DM), or patients aged less than 18 years.
Definition of outcomes
Our primary efficacy outcome was the difference in the mean change (baseline versus follow-up evaluation) between the treatment group and control group in LVEF, LVEDV, and LVESV. Secondary endpoints were change in LV volumes indexed by body surface area (BSA), LV mass index (LVMi), and NT-proBNP.
Data extraction and quality assessment
The above reported investigators independently extracted relevant outcome data and any disagreement was resolved by consensus in discussion with all authors. The following data were extracted: first author’s name, study’s acronym, publication year, study location, sample size, patient characteristics (sex, age, previous medication, estimated glomerular filtration rate [eGFR]), setting [HFrEF, HFpEF, T2DM], type of SGLT2-inhibitor, time to follow-up re-evaluation, and method of measurement (echocardiography, CMR). The methodological quality of the included studies was evaluated using the Cochrane Risk of Bias Tool [19] across the domains of sequence generation, allocation concealment, blinding, incomplete outcome data, and selective reporting.
Statistical analysis
We used mean±standard deviation (SD) change from baseline to calculate the pooled effects. If only the standard error or 95% confidence intervals (CIs) were reported, SD was calculated as described by Altman and Bland [20] and Cochrane Handbook, respectively [19]. If only median values with interquartile range were reported, means and SDs were estimated using the Box-Cox method [21]. For studies reporting only baseline and final measurements separately, but not change-from-baseline mean (SD), a correlation coefficient was calculated according to other included studies which provided mean (SD) for baseline, final, and change values. A conservative estimate (minimum correlation) was used to impute the missing SD of mean changes [22].
The pooled effect size was summarized as mean difference (MD) with corresponding 95% CIs according to the inverse-variance method. A random-effects model was applied to compute the effect size with 95% CIs. Heterogeneity between studies was assessed using the Q statistic, and its extent was calculated by the I [2] test, with an I [2] value > 50% indicating high heterogeneity. Intention-to-treat analysis was used wherever possible.
A predefined subgroup analysis was performed by stratifying according to imaging methods used (echocardiography, CMR), time to follow-up re-assessment (</≥6 months), and HF phenotype. Since the reporting studies used different imaging methods, sensitivity analysis was performed to evaluate the robustness of the effects by using standardized mean difference (SMD) for LV volumes data. Publication bias was tested visually using the funnel plot and quantitatively using the Begg adjusted-rank correlation test [23]. Meta-analyses were performed using R software (version 4.2.1; 2022, The R Foundation for Statistical Computing; https://www.r-project.org) and STATA (version 17, StataCorp, Lakeway Drive, College Station, Texas, USA).
Results
Search results and baseline characteristics
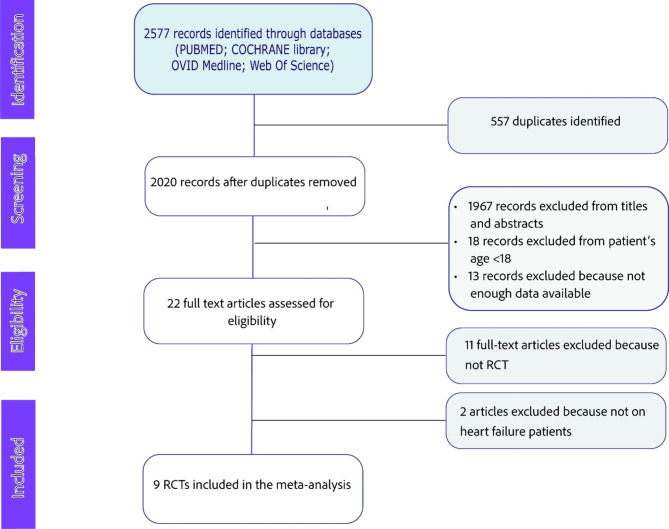
A total of 9 studies met the inclusion criteria and were analyzed in this metanalysis (detailed flowchart in Figure-1). Baseline characteristics of the included studies are summarized in Table 1. The year of publication ranged from 2020 to 2022. A total of 1385 patients were included, 685 of whom had been assigned to SGLT2i (empagliflozin in 3 studies, dapagliflozin in 1 study, canagliflozin in 3 studies, Ipragliflozin in 1 study, and Luseogliflozin in 1 study; Table-1). The mean patient age ranged from 56.2 to 73.2 years, and 89.7% of subjects were male. A total of 987 patients (in 7 studies) had T2DM with HFrEF, while three studies (398 patients) reported data on T2DM with HFpEF. Of the 9 included studies, 8 reported data on concomitant treatment with renin-angiotensin-system (RAS) inhibitors [angiotensin-converting enzyme inhibitors (ACEi), angiotensin-receptor blockers (ARBs), angiotensin-receptor-neprilysin inhibitors (ARNI)], and beta-blockers (Table-1). Use of RAS-inhibitors (89.8% versus 90.4%, P = 0.7086) and beta-blockers (94% versus 94.1%, P = 0.937) did not significantly differ between patients assigned to SGLT2-inhibitors or controls. The follow-up duration ranged from 3 to 12 months (median xxxxxx). LV imaging was performed with CMR in 3 studies [10, 14, 15], and the remaining with echocardiography [6, 8, 9, 12, 16, 18].
Fig. 1.
Flow diagram showing detailed study selection process
Table 1.
Characteristics of Included Studies and Patients of the Meta-Analysis
| Trial, First Author, year | Active treatment | Imaging Modality | Setting | N° Patients | Age | Males (%) | FU (mo) | LV Remodeling Indices | Beta-blockers, (%) | ACEi/ARBs/ARNI, (%) |
|---|---|---|---|---|---|---|---|---|---|---|
|
SUGAR-DM-HF trial Lee Matthew MY, 2021 |
Empagliflozin | CMR | HFrEF | 92 | 69±11 | 73 | 9 | EDV, EDVi, ESV, ESVi, EF, LVMi, | 91 | 100 |
|
EMPA-TROPISM, Santos-Gallego CG et al., 2021 |
Empagliflozin | CMR | HFrEF | 80 | 62±12 | 64 | 6 | EDV, ESV, EF, LVM | 88 | 85 |
|
EMPIRE-HF, Omar Massar et al., 2021 |
Canagliflozin | Echo | HFrEF | 179 | 64±11 | 85 | 3 | EDV, EDVi, ESV, ESVi, EF, LVMi, | 95 | 100 |
|
The REFORM Trial, Singh JSS et al., 2020 |
Dapagliflozin | CMR | HFrEF | 56 | 67±7 | 66 | 12 | EDV, EDVi, ESV, ESVi, EF, LVMi | 82 | 89 |
|
CANA-HF, Carbone S, 2020 |
Canagliflozin | Echo | HFrEF | 36 | 56±7 | 78 | 3 | EDVi, ESVi, EF, NTproBNP | 94 | 86 |
|
EMMY Trial Von Lewinski D et al., 2022 |
Empagliflozin | Echo | HFrEF | 476 | 58±9 | 82 | 6.5 | EDV, EDVi ESV, ESVi, EF, NTproBNP | 96 | 98 |
|
EXCEED Trial, Akasaka H et al., 2020 |
Ipragliflozin | Echo | HFpEF | 68 | 71±8 | 60 | 6 | EDV, ESV, EF, LVMi | NA | NA |
|
CANDLE Trial, Tanaka A. et al., 2020 |
Canagliflozin | Echo | HFrEF, HFpEF | 233 | 69±10 | 75 | 6 | EF, NTproBNP | 70 | 76 |
|
MUSCAT-HF, Ejiri K. et al., 2020 |
Luseogliflozin | Echo | HFpEF | 165 | 73±7 | 62 | 3 | EF, LVMi, NTproBNP | 61 | 59 |
Risk of bias assessment
The risk of bias assessment is reported in detail in the Supplementary material data (Supplemental Tables-S1 and Figure-S1). The overall risk of bias was low in most RCTs.
Left ventricular end-diastolic volume and volume-index
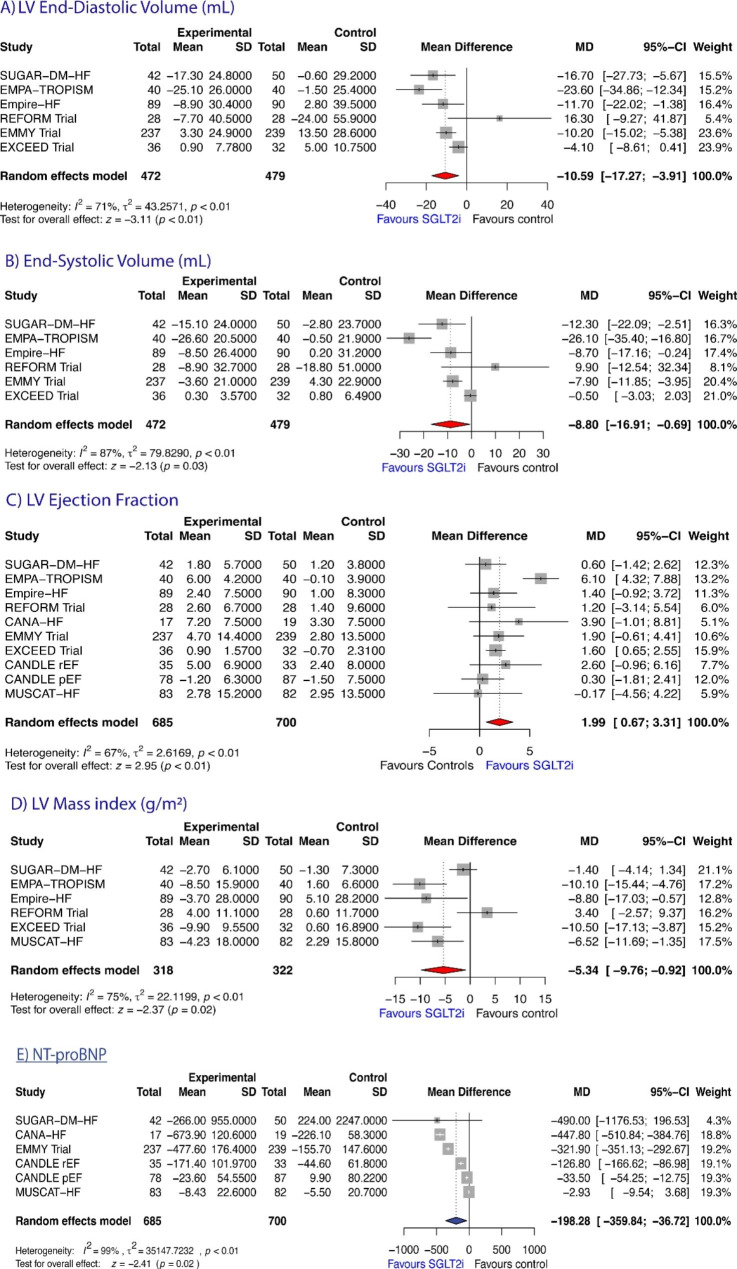
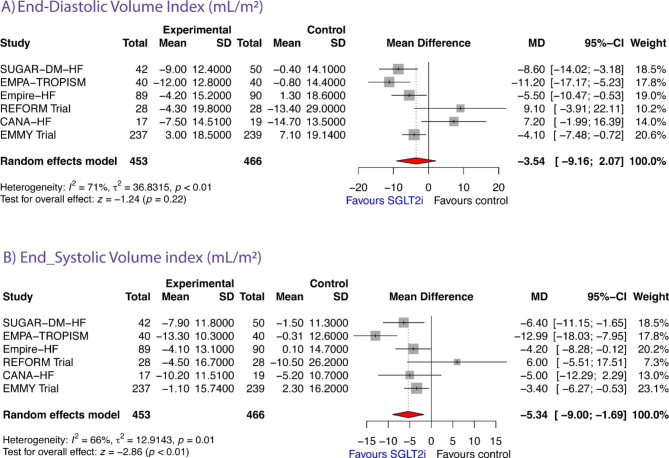
Five studies reported data on both LVEDV and LVEDV-index [10, 12, 14, 15, 18], 1 study reported data only on LVEDV [6], and 1 study reported data only on LVEDV-index [8]. Therefore, a total of 6 studies reported data on LVEDV [6, 10, 12, 14, 15, 18] (n = 951). The pooled data from these studies showed that, compared to controls, LVEDV significantly decreased after SGLT2i by -10.59 mL (95% CI: -17.27 to -3.91; z = -3.11, P = 0.0019, Figure-2A). This effect was not observed when LVEDV was indexed by BSA [MD = 3.54 (95% CI: -9.16 to 2.07; z = -1.24, P = 0.2163, Figure-3A). According to the Q-test, (LVEDV: Q test = 17.10, P = 0.0043, tau² = 43.26, I² = 80.8%; LVEDV-index: Q = 17.44, P = 0.0037, tau² = 36.83, I² = 71.3%) the true outcome appears to be heterogeneous. Subgroup analyses showed no significant difference in the mean reduction of LVEDV measured by either imaging method (CMR versus echocardiographic studies; Q = 0.07, P = 0.7916; Supplemental Figure-S2, top), or time to follow-up re-evaluation (Q = 0.04, P = 0.8329; Supplemental Figure-S2, bottom), while the effect on LVEDV was more evident for HFrEF than for HFpEF patients (Q = 4.65, P = 0.0310; Supplemental Figure-S2, center). Neither the rank correlation nor the regression test indicated any funnel plot asymmetry (P = 0.4655 and P = 0.8121, respectively, Supplemental Table-S1).
Fig. 2.
Forest plots showing the effects of SGLT2 inhibitors on (A) LV end-diastolic volume; (B) LV end-systolic volume; (C) LV Ejection Fraction; (D) LV Mass Index; and (E) NT-proBNP.
Fig. 3.
Forrest plot showing changes in end-diastolic volume index (A) and end-systolic volume index (B) from baseline to study endpoint in randomized controlled trials of heart failure patients treated with sodium glucose transporter-2 inhibitor therapy versus controls
Left ventricular end-systolic volume and volume-index
A total of 6 studies reported data on LVESV [6, 10, 12, 14, 15, 18] (n = 951). Of them, five studies reported data on both LVESV and LVESV-index [10, 12, 14, 15, 18], 1 study reported data only on LVESV [6], and 1 study reported data only on LVESV-index [8]. The observed MDs ranged from − 0.50 to -26.10. The estimated average MD based on the random-effects model was − 8.80 mL (95% CI: -16.91 to -0.69); Fig. 2B); therefore, the average outcome differed significantly from “zero” hypothesis (z = -2.13, p = 0.0334) in SGLT2i group compared to controls. This effect remained significant when LVESV was indexed by BSA [MD = -5.34 mL (95% CI: -9.00 to -1.69; z = -2.86, P = 0.0042; Figure-3B). According to the Q-test, (LVESV: Q test = 38.11, P < 0.0001, tau² = 79.83, I² = 86.9%; LVESV-index: Q = 14.79, P = 0.0113, tau² = 12.91, I² = 66.2%), once again the true outcomes appear to be heterogeneous.
Subgroup analyses showed no significant difference in the mean reduction of LVESV measured by either imaging method (Q = 0.45, P = 0.5020; Supplemental Figure-S3, top) and time to follow-up re-evaluation (Q = 0.01, P = 0.9994; Supplemental Figure-S3, bottom); however, the effect of SGLT2i on LVESV was more evident for HFrEF than for HFpEF patients (Q = 5.32, P = 0.0211; Supplemental Figure-S2, center). There was no funnel plot asymmetry by both the rank correlation and the regression test (P = 0.3481 and P = 0.6673, respectively, Supplemental Table-S1), suggesting no evidence of publication bias.
Effects of SGLT2i on LVEF
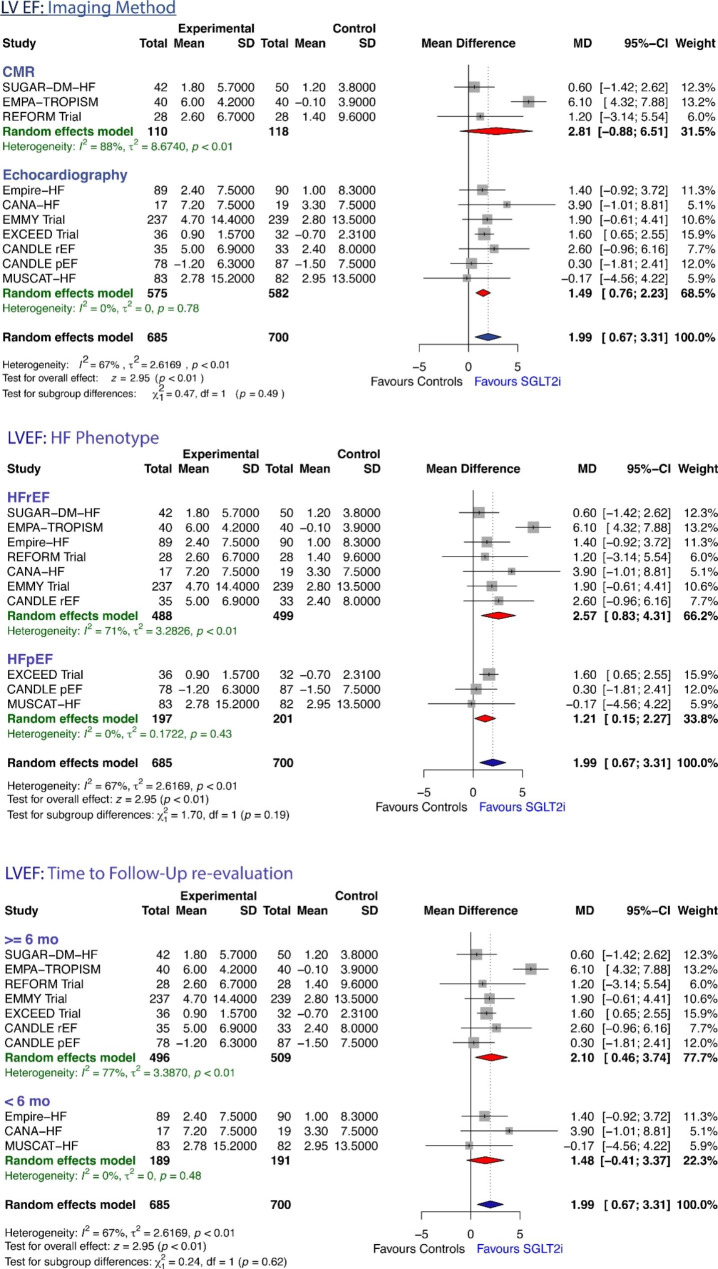
LVEF data were available for all the 9 studies [6, 8–10, 12, 14–16, 18]. The pooled data from these studies showed that, based on the random-effects model, LVEF significantly increased with SGLT2i therapy compared with controls (MD + 1.99%, 95%CI: 0.67 to 3.31, z = 2.95, P = 0.0031; Fig. 1C). The effects on LVEF were not homogeneous across studies (Q test = 27.51, P = 0.0011, tau² = 2.62, I² = 67.3%). No significant difference in the mean increase in LVEF between patients treated with SGLT2i and controls were seen according to subgroup analyses based on imaging method, HF phenotype (HFrEF vs. HFpEF), and time to follow-up re-evaluation (Figure-4). Neither the rank correlation nor the regression test indicated any funnel plot asymmetry (P = 0.3481 and P = 0.6673, respectively, Supplemental Table-S1), suggesting no evidence of publication bias.
Fig. 4.
Forrest plot showing changes in LV Ejection Fraction from baseline to study endpoint in randomized controlled trials of heart failure patients treated with sodium glucose transporter-2 inhibitor therapy versus controls, according to pre-specified subgroups
Effects of SGLT2i on LV mass index
Information on LVM index was available in 6 studies [6, 9, 10, 12, 14, 15] (n = 640). The pooled analysis of these studies according to a random effect model showed that treatment with SGLT2i was associated with a significant decrease in LV mass index (MD = -5.34 g, 95% CI -9.76 to -0.92, z=-2.37, P = 0.0178, Figure-3A), without any significant differences among subgroups analyzed (Supplemental Figure-S4). Even for LVM index heterogeneity of the effect SGLT2i was seen across studies (Q test = 20.28, P = 0.0011, tau² = 22.12, I² = 75.3%).
Effects of SGLT2i on NT-proBNP values
A total of 6 studies reported data on NT-proBNP values [8–10, 16, 18] (n = 1385). The observed MD ranged from − 2.93 to -490.0. The estimated average MD based on the random-effects model was − 198.28 pg/mL (95% CI: -359.84 to -36.71; Figure-3B); therefore, the average outcome differed significantly from zero (z = -2.41, P = 0.0162) in SGLT2i group compared to controls. According to the Q-test, (Q test = 639.23, P < 0.0001, tau² = 35147.7, I² = 99.2%) the true outcome appears highly heterogeneous among the studies. There was no evidence of publication bias as indicated by a non-significant rank correlation and regression test (P = 0.4524 and P = 0.3161, respectively, Supplemental Table-S1).
Sensitivity analysis
To evaluate the robustness of the effect, sensitivity analyses was performed by using standardized mean difference (SMD) for LV volumes, LVEF, and LVM-index. Analyses showed that the effect size remained of similar size and direction, suggesting that the trends identified in the main analysis remained unchanged (LVEDV, SMD: -0.399 mL, 95%CI: -0.67 to -0.128, P = 0.0039; I [2]: 62.2%; LVESV, SMD: -0.38 mL, 95%CI: -0.73 to -0.028, P = 0.0341; I [2]: 73.6%; LVEF, SMD: +0.34 mL, 95%CI: 0.072 to 0.62, P = 0.0134; I [2]: 75.1%; LVM-index, SMD: -0.37 mL, 95%CI: -0.65 to -0.088, P = 0.0102; I [2]: 61.9%) (Supplemental Figure-S5).
Discussion
The present meta-analysis provides updated data on the effects of SGLT2i on LV remodeling indices (LV volumes, mass, EF) based on all available studies involving diabetic patients with HF. The pooled results of this analysis show significant improvement in LV volumes and LVEF, as well as a reduction in LVM index and NTproBNP, following SGLT2i treatment compared with controls. The improvement in LVR indices was evident regardless of the imaging method used (echocardiography or CMR), time to LVR reassessment, and HF phenotype, while LV volumes were more significantly reduced in HFrEF compared to HFpEF patients. A consonant pattern of effects on LVR was seen in all studies examined, with the exception of the REFORM trial [15], which was the only one showing an opposite direction of the effect of SGLT2i on LV volumes. This could be due to the fact that i both types of HF were analyzed in REFORM, without providing separate results.
Improvements in LVRR may be one of the proposed mechanisms by which SGLT2i can exert favorable effects on clinical outcomes. Indeed, in HF patients, drugs with positive effects on LV remodeling in the short term end up improving mortality in the long term [24]. Specifically, a sub-analysis of 30 mortality trials of 25 drug/device therapies and 88 remodeling trials of the same therapies, proved that the odds ratio for death in the mortality trials was significantly correlated with drug/device effects on LVEF (r = -0.51, p < 0.001), EDV (r = 0.44, p = 0.002), and ESV (r = 0.48, p = 0.002)24.
However, although reversal of LV remodeling is an important factor in reducing mortality and morbidity in HFrEF [25], it is unlikely that it could entirely explain all the beneficial prognostic effect observed with this class of drugs. Indeed, the SGLT2i-related outcome improvement reported in RCTs [1–4], as well as improvement in quality of life [26], appears very early after therapy is started, before a significant impact on LV volumes and shape could become appreciable.
The potential effect of SGLT2 inhibitors on LV structure and function is thought to be multifactorial and mediated predominantly by systemic hemodynamic and metabolic effects [27]. The failing heart is characterized by a myocardial metabolic remodeling [28], with a shift from free fatty acids (FFA) utilization of healthy myocardium, toward glucose consumption. From one hand, these adaptations lead to reduced myocardial oxygen requests, but they also decrease the production of adenosine triphosphate (ATP) molecules [29]. This latter effect tends to be compensated by oxidation of ketone bodies that are the most energetically efficient fuel because of the higher number of ATP molecules produced at the lowest oxygen requirements [29, 30]. It has been hypothesized that the positive effects of SGLT2i on LV structural remodeling might be mediated by their capability of improving myocardial energetics via switching myocardial metabolism from glucose to ketone bodies. Santos-Gallego [31] and coworkers, using a nondiabetic porcine model in which HF was induced after 2-hours balloon occlusion of the proximal left anterior descending artery, clearly demonstrated that, compared to control group, empagliflozin markedly improved LV remodeling indices (LV mass, dilatation, and LV sphericity) after 2 months. This effect was associated with a reduced uptake of myocardial glucose and less glucose-related enzymes in empagliflozin-treated pigs, suggesting a shift from glucose toward ketone bodies utilization, with consequent increase in myocardial ATP content and enhanced myocardial work efficiency [31]. This metabolic switch from the energy-inefficient glucose towards consumption of fatty acids and ketone bodies has also been confirmed in humans in HFpEF [32], and they are thought to mediate the improvement in LV remodeling after SGLT2i therapy. However, the recent EMPAVISION trial did not show improvement in myocardial energetics in patients with either HFrEF or HFpEF [33].
It is also possible that an improvement in cardiac microvascular endothelial cells could contribute the positive effects of SGLT2-inhibitors on LV remodeling, as evidenced by experimental data suggesting an enhanced bioavailability of endothelial-derived nitric oxide as a consequence of the capability of SGLT2 inhibitors to counteract the TNF-a-induced increase in the cytoplasmatic reactive oxygen species [34]. Other possible mechanisms of action of SGLT2-inhibitors are related to their capability to directly inhibit cardiac Na+/H+ exchanger (NHE) thus reducing Na+ concentrations in cardiomyocytes [35], which activity is generally increased in diabetic and failing hearts. Furthermore, SGLT2i ameliorate myocardial ischemia-reperfusion injury, reduce infarct size and microvascular obstruction, decrease apoptosis and oxidative stress [36].
The reduction of LVM index in HF patients observed in this meta-analysis represents another important effect of SGLT2i treatment, the mechanisms of which are not fully understood. It may either reflect a decrease in cardiomyocyte mass, or changes in interstitial water content, or both. It has also been supposed that the regression of LVM might be mediated by the hypotensive effect of SGLT2-inhibitors. However, in the EMPA-HEART trial the change in 24-hour ambulatory blood pressure was not related to the change in LV mass over 6-month [17] therapy, suggesting that the effect on LV mass could be at least partly unrelated to blood pressure reduction. Interestingly, in some studies the reduction in LV mass took place in the absence of concurrent reduction in LV volumes, thus reflecting an overall reduction in wall thickness [17]. Further studies focusing on detailed tissue characterization by CMR may contribute to understand which mechanisms is mostly responsible for the observed reduction in LVM. Finally, the dual SGLT1/2 inhbitor sotagliflozin is also being investigated in the SOTA-P-CARDIA trial (NCT05562063).
Conclusions
Sodium-glucose cotransporter-2 inhibitors might play an interesting role in reversing adverse cardiac remodeling and improving LV systolic function in HF patients, mainly in those with HFrEF. Reversed cardiac remodeling may partially explain the favourable effects of SGLT2i on HF. These results should be interpreted in light of limitations including: (a) heterogeneity of the studies; (b) small sample size, in particular of the studies conducted with CMR; (c) different duration of the time to re-evaluation; (d) different types of SGLT2 inhibitors used.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
Not Applicable.
Author contributions
No relationships with industries exist for the Authors, except for Prof. Ambrosio that reports grants from Angelini, Behring, Menarini, and from Merck, outside the submitted work. The paper is not under consideration elsewhere; none of the paper’s contents have been previously published; all authors have read and approved the manuscript; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Erberto Carluccio, conception and design; analysis and interpretation of data; drafting of the manuscript and revising it critically for important intellectual content. Paolo Biagioli, collection of data, interpretation of data; drafting of the manuscript. Gianpaolo Reboldi, drafting of the manuscript, statistical analysis revision, revising it critically for important intellectual content. Anna Mengoni, collection of data, revising and selecting literature, revising the manuscript critically for important intellectual content. Rosanna Lauciello, collection of data, revising and selecting literature, bias evaluation Cinzia Zuchi, collection of data, revising and selecting literature, bias evaluationSandra D’Addario, collection of data, revising and selecting literature, bias evaluation; Giuliana Bardelli, collection of data, revising and selecting literature, bias evaluation; Giuseppe Ambrosio, revising the manuscript critically for important intellectual content and final approval of the manuscript.
Funding
Not applicable.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors have read and approved the manuscript.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.McMurray JJV, Solomon SD, Inzucchi SE, Kober L, Kosiborod MN, Martinez FA, Ponikowski P, Sabatine MS, Anand IS, Belohlavek J, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381:1995–2008. doi: 10.1056/NEJMoa1911303. [DOI] [PubMed] [Google Scholar]
- 2.Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, Januzzi J, Verma S, Tsutsui H, Brueckmann M, et al. Cardiovascular and renal outcomes with Empagliflozin in Heart failure. N Engl J Med. 2020;383:1413–24. doi: 10.1056/NEJMoa2022190. [DOI] [PubMed] [Google Scholar]
- 3.Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Bohm M, Brunner-La Rocca HP, Choi DJ, Chopra V, Chuquiure-Valenzuela E, et al. Empagliflozin in Heart failure with a preserved ejection fraction. N Engl J Med. 2021;385:1451–61. doi: 10.1056/NEJMoa2107038. [DOI] [PubMed] [Google Scholar]
- 4.Solomon SD, McMurray JJV, Claggett B, de Boer RA, DeMets D, Hernandez AF, Inzucchi SE, Kosiborod MN, Lam CSP, Martinez F, et al. Dapagliflozin in Heart failure with mildly reduced or preserved ejection fraction. N Engl J Med. 2022;387:1089–98. doi: 10.1056/NEJMoa2206286. [DOI] [PubMed] [Google Scholar]
- 5.Lopaschuk GD, Verma S. Mechanisms of Cardiovascular benefits of Sodium glucose co-transporter 2 (SGLT2) inhibitors: a state-of-the-art review. JACC Basic Transl Sci. 2020;5:632–44. doi: 10.1016/j.jacbts.2020.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Akasaka H, Sugimoto K, Shintani A, Taniuchi S, Yamamoto K, Iwakura K, Okamura A, Takiuchi S, Fukuda M, Kamide K, et al. Effects of ipragliflozin on left ventricular diastolic function in patients with type 2 diabetes and heart failure with preserved ejection fraction: the EXCEED randomized controlled multicenter study. Geriatr Gerontol Int. 2022;22:298–304. doi: 10.1111/ggi.14363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brown AJM, Gandy S, McCrimmon R, Houston JG, Struthers AD, Lang CC. A randomized controlled trial of dapagliflozin on left ventricular hypertrophy in people with type two diabetes: the DAPA-LVH trial. Eur Heart J. 2020;41:3421–32. doi: 10.1093/eurheartj/ehaa419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Carbone S, Billingsley HE, Canada JM, Bressi E, Rotelli B, Kadariya D, Dixon DL, Markley R, Trankle CR, Cooke R, et al. The effects of canagliflozin compared to sitagliptin on cardiorespiratory fitness in type 2 diabetes mellitus and heart failure with reduced ejection fraction: the CANA-HF study. Diabetes Metab Res Rev. 2020;36:e3335. doi: 10.1002/dmrr.3335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ejiri K, Miyoshi T, Kihara H, Hata Y, Nagano T, Takaishi A, Toda H, Nanba S, Nakamura Y, Akagi S, et al. Effect of Luseogliflozin on Heart failure with preserved ejection fraction in patients with diabetes Mellitus. J Am Heart Assoc. 2020;9:e015103. doi: 10.1161/JAHA.119.015103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee MMY, Brooksbank KJM, Wetherall K, Mangion K, Roditi G, Campbell RT, Berry C, Chong V, Coyle L, Docherty KF, et al. Effect of Empagliflozin on Left ventricular volumes in patients with type 2 diabetes, or Prediabetes, and heart failure with reduced ejection fraction (SUGAR-DM-HF) Circulation. 2021;143:516–25. doi: 10.1161/CIRCULATIONAHA.120.052186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Oldgren J, Laurila S, Akerblom A, Latva-Rasku A, Rebelos E, Isackson H, Saarenhovi M, Eriksson O, Heurling K, Johansson E, et al. Effects of 6 weeks of treatment with dapagliflozin, a sodium-glucose co-transporter-2 inhibitor, on myocardial function and metabolism in patients with type 2 diabetes: a randomized, placebo-controlled, exploratory study. Diabetes Obes Metab. 2021;23:1505–17. doi: 10.1111/dom.14363. [DOI] [PubMed] [Google Scholar]
- 12.Omar M, Jensen J, Ali M, Frederiksen PH, Kistorp C, Videbaek L, Poulsen MK, Tuxen CD, Moller S, Gustafsson F, et al. Associations of Empagliflozin with Left ventricular volumes, Mass, and function in patients with heart failure and reduced ejection fraction: a Substudy of the Empire HF Randomized Clinical Trial. JAMA Cardiol. 2021 doi: 10.1001/jamacardio.2020.6827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rau M, Thiele K, Hartmann NK, Schuh A, Altiok E, Mollmann J, Keszei AP, Bohm M, Marx N, Lehrke M. Empagliflozin does not change cardiac index nor systemic vascular resistance but rapidly improves left ventricular filling pressure in patients with type 2 diabetes: a randomized controlled study. Cardiovasc Diabetol. 2021;20:6. doi: 10.1186/s12933-020-01175-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Santos-Gallego CG, Vargas-Delgado AP, Requena-Ibanez JA, Garcia-Ropero A, Mancini D, Pinney S, Macaluso F, Sartori S, Roque M, Sabatel-Perez F, et al. Randomized Trial of Empagliflozin in nondiabetic patients with heart failure and reduced ejection fraction. J Am Coll Cardiol. 2021;77:243–55. doi: 10.1016/j.jacc.2020.11.008. [DOI] [PubMed] [Google Scholar]
- 15.Singh JSS, Mordi IR, Vickneson K, Fathi A, Donnan PT, Mohan M, Choy AMJ, Gandy S, George J, Khan F, et al. Dapagliflozin Versus Placebo on Left ventricular remodeling in patients with diabetes and heart failure: the REFORM Trial. Diabetes Care. 2020;43:1356–9. doi: 10.2337/dc19-2187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tanaka H, Soga F, Tatsumi K, Mochizuki Y, Sano H, Toki H, Matsumoto K, Shite J, Takaoka H, Doi T, et al. Positive effect of dapagliflozin on left ventricular longitudinal function for type 2 diabetic mellitus patients with chronic heart failure. Cardiovasc Diabetol. 2020;19:6. doi: 10.1186/s12933-019-0985-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Verma S, Mazer CD, Yan AT, Mason T, Garg V, Teoh H, Zuo F, Quan A, Farkouh ME, Fitchett DH, et al. Effect of Empagliflozin on Left Ventricular Mass in patients with type 2 diabetes Mellitus and Coronary Artery Disease: the EMPA-HEART CardioLink-6 Randomized Clinical Trial. Circulation. 2019;140:1693–702. doi: 10.1161/CIRCULATIONAHA.119.042375. [DOI] [PubMed] [Google Scholar]
- 18.von Lewinski D, Kolesnik E, Tripolt NJ, Pferschy PN, Benedikt M, Wallner M, Alber H, Berger R, Lichtenauer M, Saely CH, et al. Empagliflozin in acute myocardial infarction: the EMMY trial. Eur Heart J. 2022;43:4421–32. doi: 10.1093/eurheartj/ehac494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA, et al. The Cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Altman DG, Bland JM. Detecting skewness from summary information. BMJ. 1996;313:1200. doi: 10.1136/bmj.313.7066.1200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.McGrath S, Zhao X, Steele R, Thombs BD, Benedetti A, Collaboration DESD. Estimating the sample mean and standard deviation from commonly reported quantiles in meta-analysis. Stat Methods Med Res. 2020;29:2520–37. doi: 10.1177/0962280219889080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wiebe N, Vandermeer B, Platt RW, Klassen TP, Moher D, Barrowman NJ. A systematic review identifies a lack of standardization in methods for handling missing variance data. J Clin Epidemiol. 2006;59:342–53. doi: 10.1016/j.jclinepi.2005.08.017. [DOI] [PubMed] [Google Scholar]
- 23.Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–101. doi: 10.2307/2533446. [DOI] [PubMed] [Google Scholar]
- 24.Kramer DG, Trikalinos TA, Kent DM, Antonopoulos GV, Konstam MA, Udelson JE. Quantitative evaluation of drug or device effects on ventricular remodeling as predictors of therapeutic effects on mortality in patients with heart failure and reduced ejection fraction: a meta-analytic approach. J Am Coll Cardiol. 2010;56:392–406. doi: 10.1016/j.jacc.2010.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Konstam MA, Kramer DG, Patel AR, Maron MS, Udelson JE. Left ventricular remodeling in heart failure: current concepts in clinical significance and assessment. JACC Cardiovasc Imaging. 2011;4:98–108. doi: 10.1016/j.jcmg.2010.10.008. [DOI] [PubMed] [Google Scholar]
- 26.Requena-Ibanez JA, Santos-Gallego CG, Rodriguez-Cordero A, Vargas-Delgado AP, Badimon JJ. Empagliflozin improves quality of life in nondiabetic HFrEF patients. Sub-analysis of the EMPATROPISM trial. Diabetes Metab Syndr. 2022;16:102417. doi: 10.1016/j.dsx.2022.102417. [DOI] [PubMed] [Google Scholar]
- 27.Novo G, Guarino T, Di Lisi D, Biagioli P, Carluccio E. Effects of SGLT2 inhibitors on cardiac structure and function. Heart Fail Rev. 2022 doi: 10.1007/s10741-022-10256-4. [DOI] [PubMed] [Google Scholar]
- 28.Wende AR, Brahma MK, McGinnis GR, Young ME. Metabolic Origins of Heart failure. JACC Basic Transl Sci. 2017;2:297–310. doi: 10.1016/j.jacbts.2016.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ferrannini E, Mark M, Mayoux E. CV Protection in the EMPA-REG OUTCOME Trial: a thrifty substrate hypothesis. Diabetes Care. 2016;39:1108–14. doi: 10.2337/dc16-0330. [DOI] [PubMed] [Google Scholar]
- 30.Del Franco A, Ambrosio G, Baroncelli L, Pizzorusso T, Barison A, Olivotto I, Recchia FA, Lombardi CM, Metra M, Ferrari Chen YF, et al. Creatine deficiency and heart failure. Heart Fail Rev. 2022;27:1605–16. doi: 10.1007/s10741-021-10173-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Santos-Gallego CG, Requena-Ibanez JA, San Antonio R, Ishikawa K, Watanabe S, Picatoste B, Flores E, Garcia-Ropero A, Sanz J, Hajjar RJ, et al. Empagliflozin ameliorates adverse left ventricular remodeling in nondiabetic heart failure by enhancing myocardial energetics. J Am Coll Cardiol. 2019;73:1931–44. doi: 10.1016/j.jacc.2019.01.056. [DOI] [PubMed] [Google Scholar]
- 32.Santos-Gallego CG, Mayr M, Badimon J. SGLT2 inhibitors in Heart failure: targeted Metabolomics and energetic metabolism. Circulation. 2022;146:819–21. doi: 10.1161/CIRCULATIONAHA.122.060805. [DOI] [PubMed] [Google Scholar]
- 33.Hundertmark MJ, Adler A, Antoniades C, Coleman R, Griffin JL, Holman RR, Lamlum H, Lee J, Massey D, Miller J, et al. Assessment of Cardiac Energy metabolism, function, and physiology in patients with heart failure taking Empagliflozin: the Randomized, controlled EMPA-VISION trial. Circulation. 2023;147:1654–69. doi: 10.1161/CIRCULATIONAHA.122.062021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Juni RP, Kuster DWD, Goebel M, Helmes M, Musters RJP, van der Velden J, Koolwijk P, Paulus WJ, van Hinsbergh VWM. Cardiac microvascular endothelial enhancement of cardiomyocyte function is impaired by inflammation and restored by Empagliflozin. JACC Basic Transl Sci. 2019;4:575–91. doi: 10.1016/j.jacbts.2019.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Uthman L, Baartscheer A, Bleijlevens B, Schumacher CA, Fiolet JWT, Koeman A, Jancev M, Hollmann MW, Weber NC, Coronel R, et al. Class effects of SGLT2 inhibitors in mouse cardiomyocytes and hearts: inhibition of na(+)/H(+) exchanger, lowering of cytosolic na(+) and vasodilation. Diabetologia. 2018;61:722–6. doi: 10.1007/s00125-017-4509-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Santos-Gallego CG, Requena-Ibanez JA, Picatoste B, Fardman B, Ishikawa K, Mazurek R, Pieper M, Sartori S, Rodriguez-Capitan J, Fuster V, et al. Cardioprotective effect of Empagliflozin and circulating ketone bodies during Acute myocardial infarction. Circ Cardiovasc Imaging. 2023;16:e015298. doi: 10.1161/CIRCIMAGING.123.015298. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.