Abstract
Background
The psychological risk factors of premenstrual dysphoric disorder (PMDD) are not fully understood, but initial evidence points to a potential role of unfavorable cognitive emotion regulation (ER-) strategies. Given the symptom cyclicity of PMDD, ambulatory assessment is ideally suited to capture psychological and physiological processes across the menstrual cycle. Our study examines habitual ER-strategies in women with PMDD and their predictive value for the course of mood and basal cortisol across the cycle in affected women.
Methods
Women with and without PMDD (n = 61 each) were compared regarding habitual mindfulness, reappraisal, and repetitive negative thinking (RNT). Momentary affect and cortisol output were assessed over two consecutive days per cycle phase (menstrual, follicular, ovulatory, late luteal).
Results
Women with PMDD reported lower mindfulness, less use of reappraisal and stronger RNT than controls (ps < 0.035). In women with PMDD, higher mindfulness and reappraisal and lower RNT predicted decreased negative and increased positive affect across the menstrual cycle (ps < 0.027). However, women using more favorable ER-strategies displayed stronger mood cyclicity, resulting in stronger mood deterioration in the late luteal phase, thereby resembling women with more unfavorable ER-strategies toward the end of the cycle. Lower mindfulness predicted lower cortisol in the menstrual phase.
Conclusions
Protective ER-strategies seem to be generally linked to better momentary mood in women with PMDD, but do not appear to protect affected women from premenstrual mood deterioration. Habitual mindfulness, in turn, seems to buffer blunted cortisol activity in women with PMDD, especially in the menstrual phase.
Key words: ambulatory assessment, cortisol, emotion regulation, menstrual cycle, premenstrual dysphoric disorder
Premenstrual dysphoric disorder (PMDD) is characterized by key affective and further psychological, behavioral, and physiological symptoms, which occur during the late luteal phase of the menstrual cycle and remit within the week following menses [American Psychiatric Association (APA), 2013]. PMDD affects around 5% of women of fertile age (Beddig & Kuehner, 2017; Eisenlohr-Moul, 2019) and causes clinically significant distress or functional impairment in daily life (APA, 2013) with increased risk of a chronic symptom course (Wittchen, Becker, Lieb, & Krause, 2002) and suicidality (Osborn, Brooks, O'Brien, & Wittkowski, 2020). PMDD must be differentiated from the more frequent and less severe premenstrual syndrome (PMS), which is not uniformly defined and does not necessarily require affective symptoms (Hantsoo & Riddle, 2021).
Research on PMDD risk factors has mainly focused on its pathophysiology, suggesting a hypersensitivity to normal fluctuations of reproductive steroid hormones, which interacts with the GABAergic, serotonergic, and HPA-axis systems (Beddig & Kuehner, 2017; Eisenlohr-Moul, 2019; Hantsoo & Epperson, 2015). Regarding the latter, research on PMDD suggests that higher levels of perceived stress are linked to exacerbations of premenstrual symptoms (e.g. Gollenberg et al., 2010; Namavar Jahromi, Pakmehr, & Hagh-Shenas, 2011; Owens & Eisenlohr-Moul, 2018), pointing to a possible role of HPA-axis dysfunction in PMDD (Kiesner & Granger, 2016; Owens & Eisenlohr-Moul, 2018). However, respective evidence is mixed, ranging from blunted to increased cortisol activity in women with premenstrual disorders (e.g. Huang, Zhou, Wu, Wang, & Zhao, 2015; Kiesner & Granger, 2016; Owens & Eisenlohr-Moul, 2018). The missing differentiation between PMDD and PMS in previous research may be one reason for these inconsistencies. For example, Odber, Cawood, and Bancroft (1998) identified higher premenstrual basal cortisol levels compared to postmenstrual levels in women with mild PMS symptoms, but demonstrated a reverse cortisol pattern with significantly decreased basal cortisol levels in the premenstrual phase for women with severe premenstrual mood changes. Similarly, experimental studies point to hypoactivation of the HPA-axis in response to stress in women with PMDD (Huang et al., 2015; Klatzkin, Lindgren, Forneris, & Girdler, 2010). Reduced HPA-axis activation, in turn, has been shown to be associated with poorer mental and physical health, reflecting a biological mechanism of a spectrum of stress-related disorders (e.g. Adam et al., 2017).
Multifactorial approaches emphasize the additional role of intrapersonal psychological factors, which interact with possible biological characteristics in PMDD (Blake, 1995; Kleinstäuber et al., 2016). In this regard, the high lifetime comorbidity of PMDD with depressive and anxiety disorders (Cohen et al., 2002; Yen et al., 2020) point to the potential role of transdiagnostic psychological risk factors in the development and maintenance of PMDD. In particular, the role of possible cognitive emotion regulation (ER) dysfunction has been discussed (Owens & Eisenlohr-Moul, 2018). According to strategy-based models, ER-strategies can be classified in terms of their formal characteristics, such as being adaptive v. maladaptive (Naragon-Gainey, McMahon, & Chacko, 2017). When used habitually, adaptive and maladaptive ER-strategies have been shown to be linked to favorable v. unfavorable long-term psychological and physiological health outcomes (McRae & Gross, 2020).
Initial evidence indicates that women with PMDD tend to use more maladaptive strategies, such as avoidance coping, catastrophizing, and ruminative strategies (Craner, Sigmon, Martinson, & McGillicuddy, 2014; Craner, Sigmon, & Young, 2016; Eggert, Kleinstäuber, Hiller, & Witthöft, 2017). In particular, habitual rumination has been shown to contribute to larger increases in premenstrual depressive symptoms in women with premenstrual mood disorders (Dawson et al., 2018). In turn, habitual reappraisal (Wu, Liang, Wang, Zhao, & Zhou, 2016) and mindfulness (Lustyk, Gerrish, Douglas, Bowen, & Marlatt, 2011) were linked to less premenstrual symptom severity in nonclinical samples.
Since neuroticism was found to be positively associated with PMDD (Aperribai, Alonso-Arbiol, Balluerka, & Claes, 2016; Miller et al., 2010) and higher neuroticism may foster the use of more maladaptive ER-strategies (Yang et al., 2020), it is essential to take neuroticism scores into account when studying the use of habitual ER-strategies in PMDD. The same applies to clinical symptom levels, such as concurrent depression scores, which may affect the assessment of the habitual use of certain ER-strategies.
Previous PMDD research has mainly studied the role of habitual ER-strategies in cross-sectional study designs (e.g. Craner et al., 2014; Lustyk et al., 2011). However, only few studies have investigated their effects on cycle-related symptom change (e.g. Craner et al., 2016; Dawson et al., 2018). Furthermore, to our knowledge, studies investigating associations of ER-strategies with cortisol activity across the menstrual cycle in women with PMDD are totally lacking.
Given the cyclicity of PMDD symptomatology, ambulatory assessment (AA) designs with multiple real-time assessments during daily life are well-suited (Bosman, Jung, Miloserdov, Schoevers, & Aan Het Rot, 2016; Owens & Eisenlohr-Moul, 2018). AA allows to capture within-person psychological and physiological processes across the menstrual cycle, reduces recall biases and increases ecological validity (Trull & Ebner-Priemer, 2013). In this context, our previous research provided clear evidence of menstrual cycle-related variations of daily life experiences in women with PMDD compared to healthy controls (Beddig, Reinhard, Ebner-Priemer, & Kuehner, 2020; Beddig, Reinhard, & Kuehner, 2019). In particular, women with PMDD showed increased negative and decreased positive affect (PA) (Beddig et al., 2020) and increased psychological stress-reactivity during the late luteal phase as well as a delayed cortisol awakening response and flattened diurnal cortisol slope across the menstrual cycle compared to healthy controls (Beddig et al., 2019). Moreover, high negative affect (NA) and low cortisol output independently predicted a worse clinical course of PMDD symptomatology over a 4-month interval (Beddig & Kuehner, 2020).
The current study
In the present AA study, we aimed to examine the role of habitual ER-strategies for the cyclical course of momentary mood and cortisol activity in women with PMDD. Consistent with previous research, we expected that women with PMDD would report lower levels of mindfulness and reappraisal and higher levels of repetitive negative thinking (RNT) compared to controls. We further expected that in women with PMDD, lower levels of mindfulness and reappraisal and higher levels of RNT would predict (a) increased NA, (b) decreased PA, and (c) decreased cortisol activity, especially in the late luteal phase compared to women with more favorable ER-strategies. Finally, we examined whether observed associations would hold when controlling for levels of neuroticism and depressive symptoms.
Methods
Participants
The present sample and study design have been previously described in detail in Beddig et al. (2019). Women with and without PMDD (n = 61 each) were recruited at the Central Institute of Mental Health (CIMH), Mannheim, Germany between March 2016 and October 2018. Inclusion criteria included (a) age between 20 and 42, (b) consistent length of menstrual cycle between 22 and 34 days, and (c) fulfillment of diagnostic criteria of a PMDD diagnosis based on DSM-5 criteria (PMDD group) or exemption from any PMDD affective core symptoms (control group). Women were ineligible if they were pregnant or lactating during the last 6 months, taking hormonal contraceptives and pharmaceutical medication during the last 3 months, or if they reported a body mass index <18 or >35, late evening or night shifts, a history of gynecological diseases (e.g. hysterectomy or ovariectomy), psychotic or bipolar disorder, and current substance abuse or dependence. The study protocol was approved by the ethics committee of the Medical Faculty Mannheim, Heidelberg University. All participants gave written informed consent and were paid 100€ for their participation.
Procedure
The procedure consisted of a preliminary telephone screening, a baseline session, and 8 days of subsequent AA. Demographic and clinical characteristics, inclusion and exclusion criteria, as well as psychological traits (i.e., habitual ER-strategies; see below) were assessed during the baseline session at the CIMH in Mannheim (Germany). Eligible participants then received a study smartphone and detailed instructions regarding the AA procedure.
To verify the diagnosis of PMDD, the SCID-PMDD, a reliable Structured Interview for DSM-IV-TR PMDD (κ = 0.96; Accortt, Bismark, Schneider,& Allen, 2011) was administered to both samples during the diagnostic baseline session. The SCID-PMDD covers all symptom criteria of PMDD together with the criterion of relational, occupational, and recreational impairment or distress and the exclusion criterion of a mere exacerbation of symptoms of another disorder (cf. Kuehner & Nayman, 2021). The interview format for 11 symptoms of PMDD is modeled after the Structured Clinical Interview for DSM-IV for Axis I (SCID-I; Wittchen, Zaudig, & Fydrich, 1997), with additional questions on the timing of symptom-on- and offset across the menstrual cycle and the number of symptomatic cycles experienced for each of the 11 symptoms. A diagnosis of PMDD required fulfilling respective criteria within the diagnostic algorithm adapted for DSM-5, according to which functional impairment as a criterion is not mandatory if the premenstrual symptoms are associated with clinically significant distress (APA, 2013). Controls had to be free of any PMDD affective core symptom. In order to keep the compliance rate high and to avoid further participant burden within the AA-design, additional prospective daily symptom ratings during at least two symptomatic cycles before study inclusion were not required. Current and lifetime DSM-IV-TR Axis I psychiatric comorbidities and exclusion criteria were assessed with the SCID-I (Wittchen et al., 1997). All interviews were performed by a trained research psychologist (T.B.).
Ambulatory assessment (AA)
AA was carried out using Motorola Moto G 2nd Generation smartphones with the software movisensXS, version 0.6.3658 (movisens GmbH, Karlsruhe, Germany). AA took place over two consecutive days per menstrual cycle phase (menstrual, follicular, ovulatory, and late luteal phase).
The typical menstrual cycle lasts about 28 (21–35) days and can be divided into four cycle phases with predictable fluctuations of the ovarian hormones progesterone (P4) and estradiol (E2). It starts with the onset of menstruation, which represents the cycle day 1, endures about 5 days and is characterized by low P4 and E2 levels. The follicular phase is marked by consistently low P4 and rising E2 levels with a peak prior to ovulation, which is followed by a rapid E2 decrease after ovulation. The luteal phase covers the days from ovulation until menses during which E2 and P4 gradually rise, reaching their highest levels during the mid-luteal phase, and then show a rapid withdrawal during the late luteal (premenstrual) phase, i.e., the week prior to the next menses (Schmalenberger et al., 2021).
Individual cycle calendars were prepared based on the date of the last menstruation onset and the average cycle length in order to specify the start date of ovulation testing and exact days of the AA. The ovulation phase was identified by a chromatographic ovulation test (gabControl hlH Ovulationsteststreifen, gabmed, Cologne). The testing started a few days before the predicted ovulation and had to be continued until a positive result occurred. If ovulation did not occur, women were asked to repeat the testing in the next menstrual cycle. In order to avoid sequence effects, they started AA in different cycle phases.
The menstrual phase was assessed on the second and third days of menstruation (M = 2.95 days, s.d. = 2.21). The assessments during the follicular phase were examined on the second and third days after the end of menstruation (M = 8.61 days, s.d. = 1.94). The ovulatory phase (M = 17.15 days, s.d. = 2.0) was assessed on the 2 days following a positive ovulation test result. In case of a negative test, participants were asked to repeat the testing during the following menstrual cycle. Assessments of the late luteal phase took place on the fourth and third day before the next expected menstruation (M = 26.38 days, s.d. = 3.02). If the menstruation occurred at least 3 days earlier or later than expected, women were asked to repeat the AA during the next cycle in order to ensure a late luteal-phase assessment.
Women performed eight assessments per day starting exactly at 9:00 h. The remaining seven assessments took place between 10:00 h and 21:30 h at semi-random time points with a completion time of 3–4 min per assessment. Ignored or rejected alarms were coded as missing (for detailed information see Beddig et al., 2019).
Ambulatory assessment (AA) variables
Momentary NA and PA were assessed using 12 items from previous AA studies by our group (e.g. Kuehner, Welz, Reinhard, & Alpers, 2017; Timm et al., 2018). At each assessment, women reported the extent to which they felt several negative (upset, irritated, nervous, listless, down, and bored, α = 0.832†,1) and positive (cheerful, energetic, enthusiastic, satisfied, relaxed, and calm, α = 0.708) emotions on a seven-point Likert scale ranging from 1 (not at all) to 7 (very much).
At the first assessment (09:00 h) of each AA day, women further reported time of awakening and sleep duration (number of hours) as well as sleep quality (‘How did you sleep last night?’) measured on a seven-point Likert scale ranging from 1 (very bad) to 7 (very good) by single items.
Salivary measure of cortisol
Twenty minutes after each subjective AA, women collected saliva cortisol samples with standard salivettes (Sarstedt, Germany), resulting in eight saliva samples per day for analysis. Women were instructed not to eat, drink anything other than water, smoke, physically exercise, and brush their teeth the next 20 min until saliva collection, and also to refrain from strenuous exercise during AA days (cf. Schlotz, 2019). Immediately after collection of each cortisol sample, they indicated whether they had eaten, drunk anything other than water, smoked, or brushed their teeth (0 = no, 1 = yes) and the extent of their physical activity (seven-point Likert scale: 1 = not at all to 7 = very much) during the last 20 min. The smartphone further provided a random three-digit code, which had to be recorded on the respective label of the salivette tube used during each saliva collection (Schlotz, 2019). Until being returned, all samples were stored in the participants' home freezer and were frozen at −20 °C at the laboratory prior to biochemical analysis. At the laboratory of Professor Kirschbaum (Dresden, Germany), salivettes were centrifuged at 3000 rpm for 5 min, resulting in a clear supernatant of low viscosity. Salivary concentrations were measured using commercially available chemiluminescence-immunoassay with high sensitivity (IBL International, Hamburg, Germany). The intra- and interassay coefficients for cortisol were below 8%.
Trait-level measures
Mindfulness
The German version of the 15-item Mindfulness Attention Awareness Scale (MAAS; Brown & Ryan, 2003) was administered to measure participants' habitual tendency to be attentive and aware of present-moment experiences. The items were rated on a six-point Likert scale, with higher scores indicating greater mindfulness (α = 0.891).
Reappraisal
The six-item subscale of the Emotion Regulation Questionnaire (ERQ; Abler & Kessler, 2009) was used to assess habitual usage of reappraisal. All items were rated on a seven-point Likert scale with higher scores indicating higher usage of reappraisal (α = 0.842).
Repetitive negative thinking (RNT)
Participants completed the German version of the Perseverative Thinking Questionnaire (PTQ; Ehring et al., 2011), a well-validated 15-item scale assessing the habitual tendency to engage in RNT. All items were answered on a five-point Likert scale with higher scores signifying higher levels of RNT (α = 0.957).
Neuroticism
Neuroticism was measured with the 12-item Neuroticism Subscale derived from the NEO Five Factor Inventory (NEO-FFI; Costa & McCrae, 1992; α = 0.879).
Depressive symptoms
The severity of self-rated depressive symptoms during the last 2 weeks was measured with the 21-item Beck Depression Inventory-II (BDI-II; Beck, Steer, & Brown, 1996; α = 0.915).
Statistical analyses
Group differences in ER-strategies
To test group differences in habitual ER-strategies, a one-way multivariate analysis of variance (MANOVA) was conducted with group (PMDD v. controls) as the independent variable and cognitive ER-strategies (mindfulness, reappraisal, and RNT) as dependent variables.
Associations of ER-strategies with daily affect and cortisol in women with PMDD
To examine the predictive value of ER-strategies on the course of momentary affect and cortisol across the menstrual cycle in women with PMDD (N = 61), multilevel models (MLM) were estimated to take into account that assessments (level 1) were nested within participants (level 2) (Nezlek, Schroeder-Abé, & Schuetz, 2006). Prior to the main analyses, random intercept models were fitted for each outcome to calculate intraclass correlation coefficients (ICC). Furthermore, possible confounders of daily affect (time since awakening, time since first assessment, assessment day) were analyzed in separate random intercept models and were retained in the models if significant (p < 0.05). This applied to assessment day. All level 2 predictors were grand-mean-centered within the PMDD-group to improve the interpretability of the resulting MLM parameters.
Cortisol data were log-transformed to adjust for skewness. Then, outliers more than three standard deviations from the group mean were winsorized to three standard deviations (Stalder et al., 2016). Time was centered at waking time. Possible confounders of basal cortisol secretion (age, current medication use, habitual smoking, time, time2, time of awakening, sleep quality, sleep duration, weekday v. weekend, and drinking anything other than water, smoking cigarettes, eating, brushing the teeth, and the level of physical activity during the last 20 min) were analyzed in separate random intercept models, and were retained in the models if significant (p < 0.05). This applied to time, time2, time of awakening, sleep duration as well as recent physical activity and drinking.
Random intercept models, in which the intercept was allowed to vary between individuals, were estimated using restricted maximum likelihood estimation (REML). MLMs were carried out in two steps. First, we estimated the main effects of each habitual ER-strategy (mindfulness, reappraisal, and RNT) on each momentary outcome (NA, PA, cortisol) in separate MLMs and controlled for cycle phase as a categorical level 2 variable. In a second step, these models were expanded by entering the interaction effects of cycle phase with ER-strategies (cycle phase×ER-strategy) on each momentary outcome. In case of a significant interaction effect, we subsequently estimated simple slopes for each ER-strategy on mood and cortisol per cycle phase in post-hoc analyses. Finally, all analyses were repeated by controlling for possible main effects of neuroticism and depressive symptoms.
Statistical analyses were performed using IBM SPSS version 25 with the significance level set at α = 0.05. This value was not adjusted for multiple testing as the tests were based on preplanned hypotheses (Armstrong, 2014).
Results
Sample description
The descriptives on demographics and questionnaire measures are listed in Table 1. Groups did not significantly differ with respect to age, education level, work situation, partner status, and percentage of having children. However, as expected, women with PMDD displayed higher levels of depressive symptoms and neuroticism scores.
Table 1.
Demographic and clinical characteristics of women with PMDD and controls
| PMDD (n = 61) %/M (s.d.) | Controls (n = 61) %/M (s.d.) | Test statistic | p | |
|---|---|---|---|---|
| Demographic variables | ||||
| Age | 29.4 (5.8) | 29.5 (5.1) | t(120) = −0.03 | 0.977 |
| Education (% with high school degree) | 72.1% | 75.4% | χ2(1) = 0.17 | 0.681 |
| Work situation (% in regular job or education) | 80.3% | 90.2% | χ2(1) = 2.35 | 0.126 |
| Partner status (% married or living together) | 60.7% | 59.0% | χ2(1) = 0.03 | 0.853 |
| Children (%) | 24.6% | 26.2% | χ2(1) = 0.04 | 0.835 |
| BMI | 23.6 (4.1) | 23.5 (4.3) | t(120) = 0.12 | 0.903 |
| Clinical variables | ||||
| SCID-I lifetime diagnosis of depression | 54.1% | 21.3% | χ2(1) = 13.96 | <0.001 |
| BDI-II depression score | 10.9 (8.9) | 4.8 (5.6) | t(119) = 4.51 | <0.001 |
| NEO-FFI neuroticism | 2.11 (0.7) | 1.5 (0.7) | t(118) = 4.46 | <0.001 |
BMI, body mass index; SCID-I, Structured Clinical Interview for DSM-IV Axis I; BDI-II, Beck Depression Inventory-II; NEO-FFI, NEO Five Factor Inventory.
Group differences in ER-strategies
The MANOVA yielded a significant effect of group (PMDD v. controls) on ER-strategies [Wilks' λ = 0.852; F(3,116) = 6.71, p < 0.001]. Univariate ANOVAs revealed significant group differences in habitual mindfulness [F(1,3) = 4.55, p = 0.035], reappraisal [F(1,7) = 5.31, p = 0.023], and RNT [F(1,12) = 18.19, p < 0.001]. In particular, women with PMDD reported lower levels of mindfulness (MPMDD = 4.13, s.d.PMDD = 0.96; Mcontrols = 4.45, s.d.controls = 0.69), less use of reappraisal strategies (MPMDD = 4.19, s.d.PMDD = 1.34; Mcontrols = 4.67, s.d.controls = 0.88), and higher levels of RNT (MPMDD = 1.92, s.d.PMDD = 0.91; Mcontrols = 1.29, s.d.controls = 0.70).
Multilevel analyses (MLM)
Compliance
MLMs were based on 61 women with PMDD. Altogether, 3381 of 3904 possible subjective assessments (four menstrual cycle phases × 16 assessments per phase × 61 participants) were recorded, which corresponds to a response rate of 86.6%. This reflects a high level of compliance (cf. Courvoisier, Eid, & Lischetzke, 2012). The compliance rate for cortisol assessments in the PMDD group amounted to 84.5%.
Intra-class correlation (ICC)
ICCs indicated that 22% of variability in NA and 26% of variability in PA of the PMDD sample were attributable to between-person differences. For cortisol assessments, the ICC indicated that 16% of variability in cortisol levels were due to between-person differences.
Associations of ER-strategies with momentary affect across the cycle
The main effect analyses revealed that lower mindfulness [B = −0.15, s.e. = 0.07, t(59) = −2.30, p = 0.025] and reappraisal [B = −0.12, s.e. = 0.05, t(59) = −2.68, p = 0.010], and higher RNT levels [B = 0.16, s.e. = 0.07, t(59) = 2.27, p = 0.027] predicted increased NA across the menstrual cycle (Table 2). In turn, higher mindfulness [B = 0.22, s.e. = 0.08, t(59) = 2.93, p = 0.005] and reappraisal [B = 0.15, s.e. = 0.06, t(59) = 2.81, p = 0.007] and lower RNT levels [B = −0.23, s.e. = 0.08, t(59) = −2.80, p = 0.007] predicted increased PA across the menstrual cycle (Table 2).
Table 2.
Main and interaction effects of ER-strategies with cycle phase on momentary affect and cortisol activity
| Predictor | Negative affect (NA)a | Positive affect (PA)a | Cortisolb | ||||||
|---|---|---|---|---|---|---|---|---|---|
| df | F | p | df | F | p | df | F | p | |
| Mindfulness | |||||||||
| Step 1 | |||||||||
| Cycle phase | 3, 3320 | 105.30 | <0.001 | 3, 3320 | 91.19 | <0.001 | 3, 3011 | 0.87 | 0.458 |
| Mindfulness | 1, 59 | 5.30 | 0.025 | 1, 59 | 8.60 | 0.005 | 1, 59 | 3.18 | 0.080 |
| Step 2c | |||||||||
| Cycle phase × mindfulness | 3, 3317 | 6.76 | <0.001 | 3, 3317 | 4.03 | 0.007 | 3, 3007 | 3.09 | 0.026 |
| Reappraisal | |||||||||
| Step 1 | |||||||||
| Cycle phase | 3, 3320 | 105.37 | <0.001 | 3, 3320 | 91.27 | <0.001 | 3, 3011 | 0.86 | 0.462 |
| Reappraisal | 1, 58 | 7.17 | 0.010 | 1, 59 | 7.90 | 0.007 | 1, 59 | 0.51 | 0.478 |
| Step 2c | |||||||||
| Cycle phase × reappraisal | 3, 3317 | 5.37 | <0.001 | 3, 3317 | 3.21 | 0.022 | 3, 3007 | 2.58 | 0.052 |
| Repetitive negative thinking (RNT) | |||||||||
| Step 1 | |||||||||
| Cycle phase | 3, 3320 | 105.37 | <0.001 | 3, 3320 | 91.27 | <0.001 | 3, 3010 | 0.86 | 0.461 |
| RNT | 1, 59 | 5.16 | 0.027 | 1, 59 | 7.81 | 0.007 | 1, 59 | 0.14 | 0.712 |
| Step 2c | |||||||||
| Cycle phase × RNT | 3, 3318 | 6.97 | <0.001 | 3, 3317 | 6.17 | <0.001 | 3, 3008 | 2.06 | 0.103 |
ER-strategies, emotion regulation strategies; RNT, repetitive negative thinking.
All models include random intercepts at level 2.
Models include fixed effects of assessment day.
Models include fixed effects of time, time2, time of awakening, sleep duration as well as physical activity and drinking during the past 20 min.
Step 2 models additionally include main effects of cycle phase and ER-strategy.
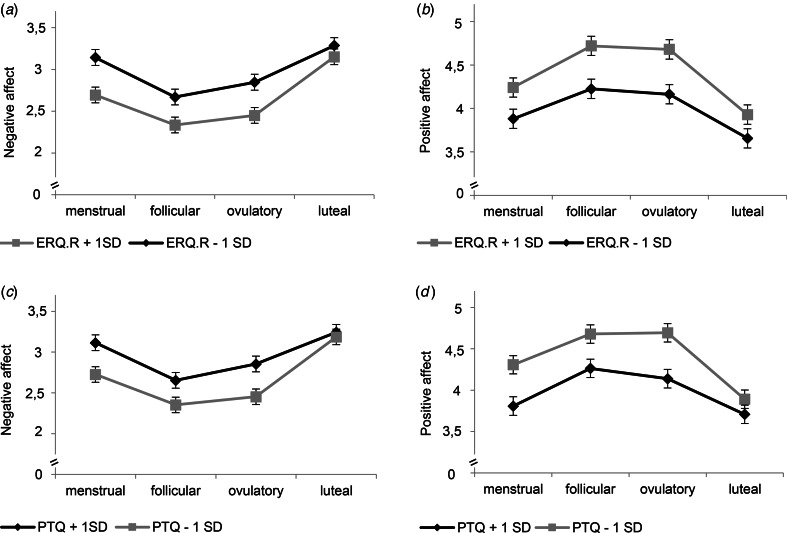
Our second-step analyses identified significant interaction effects of cycle phase by mindfulness [FNA(3,3317) = 6.76, p < 0.001; FPA(3,3317) = 4.03, p = 0.007], reappraisal [FNA(3,3317) = 5.37, p < 0.001; FPA(3,3317) = 3.21, p = 0.022], and RNT [FNA(3,3318) = 6.97, p < 0.001; FPA(3,3317) = 6.17, p < 0.001] in predicting NA and PA (Table 2). As depicted in Figs. 1 and 2 for illustration purposes, post-hoc tests revealed that favorable ER-strategies, i.e. higher mindfulness and reappraisal and lower RNT levels, predicted decreased NA and increased PA only in the menstrual, follicular, and ovulatory phases (all ps < 0.029, see Table 3 for post-hoc test results), while these ER-strategies did not show any effect on momentary affect in the late luteal phase (all ps > 0.05). Thus, contrary to our hypotheses, women with favorable ER-strategies showed stronger mood deterioration toward the late luteal phase, thereby converging with women with unfavorable ER-strategies (see Figs. 1 and 2).
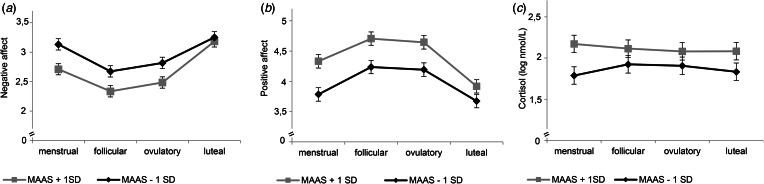
Fig. 1.
Interaction effects of mindfulness and cycle phase on momentary affect and cortisol activity. MAAS, Mindfulness Attention Awareness Scale. Estimated mean values of momentary negative affect (a), positive affect (b), and log-transformed basal cortisol activity (c) per menstrual cycle phase for low and high scores on MAAS (M ± 1s.d.) from multilevel models for illustration purposes. Error bars represent standard error of the estimated mean. All models include random intercepts at level 2. Models in (a) and (b) include fixed effects of assessment day. The model in (c) includes fixed effects of time, time2, time of awakening, sleep duration as well as physical activity and drinking during the past 20 min.
Fig. 2.
Interaction effects of reappraisal and repetitive negative thinking with cycle phase on momentary affect. ERQ.R, Emotion Regulation Questionnaire_reappraisal subscale; PTQ, Perseverative Thinking Questionnaire. Estimated mean values of momentary negative affect and positive affect per menstrual cycle phase for low and high scores (M ± 1s.d.) on ERQ.R (a, b) and PTQ (c, d) for illustration purposes. Error bars represent standard error of the estimated mean. All models include random intercepts at level 2 and fixed effects of assessment day.
Table 3.
Estimated simple slopes of mindfulness, reappraisal, and repetitive negative thinking (RNT) on negative affect, positive affect, and cortisol activity per cycle phase
| ER-strategy | Cycle phase | Negative affect | Positive affect | Cortisola | |||
|---|---|---|---|---|---|---|---|
| B (s.e.) | p | B (s.e.) | p | B (s.e.) | p | ||
| Mindfulness | Menstrual phase | −0.22 (0.07) | 0.002 | 0.29 (0.08) | <0.001 | 0.20 (0.08) | 0.012 |
| Follicular phase | −0.18 (0.07) | 0.014 | 0.25 (0.08) | 0.003 | 0.10 (0.08) | 0.201 | |
| Ovulatory phase | −0.17 (0.07) | 0.017 | 0.24 (0.08) | 0.005 | 0.09 (0.08) | 0.243 | |
| Luteal phase | −0.04 (0.07) | 0.619 | 0.13 (0.08) | 0.120 | 0.13 (0.08) | 0.096 | |
| Reappraisal | Menstrual phase | −0.17 (0.05) | 0.001 | 0.14 (0.06) | 0.024 | – | – |
| Follicular phase | −0.13 (0.05) | 0.014 | 0.19 (0.06) | 0.002 | – | – | |
| Ovulatory phase | −0.15 (0.05) | 0.004 | 0.19 (0.06) | 0.001 | – | – | |
| Luteal phase | −0.05 (0.05) | 0.306 | 0.10 (0.06) | 0.085 | – | – | |
| Repetitive negative thinking | Menstrual phase | 0.22 (0.08) | 0.005 | −0.28 (0.09) | 0.002 | – | – |
| Follicular phase | 0.17 (0.08) | 0.029 | −0.23 (0.09) | 0.010 | – | – | |
| Ovulatory phase | 0.22 (0.08) | 0.004 | −0.31 (0.09) | <0.001 | – | – | |
| Luteal phase | 0.03 (0.07) | 0.675 | −0.10 (0.09) | 0.248 | – | – | |
ER-strategy, emotion regulation strategy.
Log-transformed cortisol values in nmol/l. Simple slope values are presented only for significant interaction terms of cycle phase by ER-strategy.
Associations of ER-strategies with momentary cortisol activity across the cycle
The main effects of mindfulness [B = 0.13, s.e. = 0.07; t(59) = 1.78, p = 0.080], reappraisal [B = 0.04, s.e. = 0.05; t(59) = 0.72, p = 0.478], and RNT [B = −0.03, s.e. = 0.08; t(59) = −0.37, p = 0.712] on momentary cortisol activity were not significant (Table 2). However, there was a significant interaction effect of cycle phase and mindfulness on momentary cortisol activity [F(3,3007) = 3.09; p = 0.026]. As shown in Fig. 1c, again for illustration purposes, lower mindfulness was associated with decreased basal cortisol activity only in the menstrual phase, with no associations in the follicular, ovulatory, and luteal phase (see Table 3).
Confounder analysis
Neuroticism and severity of depressive symptoms (BDI-II) as covariates did not change the size of any reported ER-strategy × cycle phase interaction effects.
Discussion
ER deficits are transdiagnostic risk factors and important treatment targets for a variety of psychological disorders (Aldao, Nolen-Hoeksema, & Schweizer, 2010). Unfavorable ER-strategies may interact with the menstrual cycle to contribute to cycle-related changes in mood and cortisol activity in women with PMDD. The present study aimed to compare cognitive ER-strategies in women with and without PMDD, and to explore the predictive value of these strategies for momentary affect and basal cortisol activity across the menstrual cycle in women with PMDD. As expected, women with PMDD reported more maladaptive and less adaptive strategies than healthy controls. Favorable strategies were generally linked to better mood in women with PMDD, whereas they did not appear to protect affected women from cycle-dependent mood worsening during the late luteal phase. Habitual mindfulness was associated with higher cortisol levels during the menstrual phase.
Differences in the use of ER-strategies between women with and without PMDD
In our study, women with PMDD showed lower mindfulness and reappraisal and higher RNT levels than controls. This is consistent with previous research (Craner et al., 2014; Dawson et al., 2018; Lustyk et al., 2011; Wu et al., 2016), indicating heightened vulnerability toward unfavorable ER-strategies in affected women.
ER-strategies × cycle phase on momentary affect in women with PMDD
In women with PMDD, higher habitual mindfulness and reappraisal and lower RNT predicted lower NA and higher PA during the menstrual, follicular, and ovulatory phases, but not during the luteal phase. Women reporting more favorable ER-strategies displayed larger mood cyclicity with stronger mood worsening toward the luteal phase, thereby converging with those using more unfavorable strategies. Thus, contrary to our expectations, favorable habitual cognitive ER-strategies seem not to protect affected women from cycle-related mood worsening.
These results may provide first indications of possible limitations of cognitive behavioral interventions (CBI) addressing cognitive ER-strategies in the treatment of PMDD and underscore the need for more nuanced research on possible differential effects of specific interventions. Previous reviews found mixed evidence for CBIs in the treatment of premenstrual dysphoric symptoms (Kleinstäuber, Witthöft, & Hiller, 2012; Lustyk, Gerrish, Shaver, & Keys, 2009). A recent review (Han, Cha, & Kim, 2019) showed that, in particular, the acquisition of active behavioral coping strategies were helpful in premenstrual symptom relief. However, the trials included in these reviews suffer from inadequate study designs and inclusion of women with PMS. A first trial specifically on women with PMDD (Hunter et al., 2002) again showed that the use of active behavioral strategies at the end of a CBI, but not changes in causal attributions, predicted a good clinical outcome after 1 year. A recent 8-week internet-based CBI trial on women with PMDD, which consisted of psychoeducation and cognitive and behavioral strategies, revealed high effect sizes for symptom reduction and psychosocial functioning (Weise et al., 2019). While the effects of specific strategies cannot be extracted from this multimodal intervention, the authors showed that habitual active coping with premenstrual symptoms predicted improved treatment outcomes. Altogether, these results point to potential incremental effects of behavioral over cognitive strategies in PMDD treatment, and may also explain the lack of luteal-phase effects of cognitive ER-strategies on mood in the present study. Evidence-based therapies targeting behavioral skills such as Dialectical-Behavioral-Therapy have already been suggested, but not yet evaluated for PMDD treatment (Eisenlohr-Moul, 2019). However, since we investigated trait characteristics of ER-strategies and not respective interventions, we are aware that these conclusions are highly speculative, but can be tested, for example, in studies with dismantling designs (Papa & Follette, 2015). On the other hand, it is quite conceivable that higher levels of favorable ER-strategies do not necessarily imply that women are able to use these strategies effectively in the premenstrual phase in which mainly biologically determined mood changes occur. The lacking predictive value of habitual ER-strategies for mood-related changes during the luteal phase may also indicate that the unilateral classification of ER in adaptive v. maladaptive may be reductive and may ignore the dynamic nature of ER (Aldao, 2013). Instead, the affective impact of ER-strategies may depend on the flexibility to apply them in accordance with contextual demands and individuals' regulatory goals (Aldao, Sheppes, & Gross, 2015; Mikkelsen, Tramm, Zachariae, Gravholt, & O'Toole, 2021; Wenzel, Rowland, Weber, & Kubiak, 2020). In PMDD, contextual, especially cycle-phase-specific, characteristics may predict the choice and the efficacy of ER-strategies (cf. Aldao, 2013; Sheppes, Scheibe, Suri, & Gross, 2011; Wenzel, Rowland, & Kubiak, 2021). In this context, AA is particularly suited to assess the possible cycle-phase-specific use of certain ER-strategies at the state level. Furthermore, it offers the opportunity to directly induce specific ER-states and to assess their cycle-specific effects in an experimental field design (cf. Huffziger et al., 2013 for a similar approach; Huffziger, Ebner-Priemer, Koudela, Reinhard, & Kuehner, 2012). In this way, future research can gain a deeper understanding of ER-processing and its impact on affect across the menstrual cycle in women with PMDD.
ER-strategies × cycle phase on momentary cortisol activity in women with PMDD
To our knowledge, our study is the first to investigate the impact of ER-strategies on cycle-related variations of basal cortisol activity in women with PMDD. Mindfulness, reappraisal and RNT did not predict overall or luteal-phase-specific cortisol output. In contrast, higher trait mindfulness was linked to higher cortisol levels in the menstrual phase.
The high intra-individual variability of cortisol release in the current study (ICC = 16%) matches with the observation that substantial variance in diurnal cortisol is due to moment-to-moment and day-to-day fluctuations rather than to stable between-person differences (Doane, Chen, Sladek, Van Lenten, & Granger, 2015). In experimental studies inducing mental stress, women with affective disorders and women with PMDD show blunted cortisol activity (Hantsoo & Epperson, 2015; Zorn et al., 2017). In contrast, no clear associations have been identified between induced ER-strategies and cortisol responses in general (Mikkelsen et al., 2021). The effects of trait ER-strategies on diurnal cortisol levels outside the lab have also only been rarely examined (Otto, Sin, Almeida, & Sloan, 2018).
The lacking effect of trait reappraisal on daily life cortisol is in line with results from nonclinical samples (Otto et al., 2018; Rnic, Jopling, Tracy, & LeMoult, 2022) and has been attributed to the low physiological effort required for reappraisal as a more automatic process in the long term (Otto et al., 2018). There is also some indication that reappraisal is more effective in the context of controllable stressors (cf. Mikkelsen et al., 2021). Given that women with PMDD often describe themselves as feeling out of control during the late luteal phase (cf. APA, 2013), this may explain why this ER-strategy may not be able to affect cycle-related cortisol activity. Similarly, the lacking effect of trait RNT on cortisol is in accordance with findings, which show that state measures of RNT are more closely related to cortisol activity than trait measures, particularly if the latter do not specifically measure stress-related RNT (Zoccola & Dickerson, 2012).
High habitual mindfulness appeared to counteract low cortisol secretion particularly in the menstrual phase, but not in the luteal phase (as expected). Blunted presentation of cortisol secretion seems to be associated with a triad of increased pain, stress sensitivity, and fatigue (Fries, Hesse, Hellhammer, & Hellhammer, 2005), and higher cortisol concentrations are commonly related to lower pain intensity (cf. Ubeda-D'Ocasar et al., 2020). In turn, studies have shown beneficial effects of mindfulness-based interventions on menstrual pain and pain perception (Payne, Seidman, Romero, & Sim, 2020; Purnamasari, Rohita, Zen, & Ningrum, 2020), and the activation of cortisol release may constitute one possible mechanism through which habitual mindfulness can dampen respective symptoms. However, these considerations are again speculative because we did not measure cycle-related physical symptoms in our study, which is clearly warranted in future AA studies.
Limitations and future directions
Our study has some limitations. First, the sample size was modest although exceeding the recommended minimum size for estimating cross-level interactions (Hox, Moerbeek, & Van de Schoot, 2017). Second, in order to keep participants' burden low and compliance rates high within this intensive AA-design, the PMDD diagnosis was assessed with a retrospective, although well-validated, structured interview (SCID-PMDD; Accortt et al., 2011). This in turn can bear the risk of recall-bias toward false-positive symptom reports (Schmalenberger et al., 2021). DSM-5 requires prospective daily symptom ratings for at least two symptomatic cycles for a definite diagnosis. Thus, the present PMDD-diagnoses must be considered provisional (APA, 2013).
Furthermore, we only examined cognitive ER-strategies. The further inclusion of behavioral ER-strategies (e.g. stress-reduction-skills) would allow to assess possible effects of specific classes of strategies. We also focused on measures of self-assessed habitual ER-strategies, which are characterized by possible recall-bias, restriction to conscious ER, and by unclear predictive validity regarding their use in daily life (Naragon-Gainey et al., 2017). In this context, implicit assessments of ER (e.g. Eggert et al., 2017) and AA-designs can help to uncover automatic aspects of ER-processes and possible cycle-specific ER-deficits and, consequently, possible treatment targets.
Conclusions
The present findings suggest that protective habitual cognitive ER-strategies are generally linked to improved momentary mood in women with PMDD but do not appear to protect affected women from cycle-dependent mood deteriorations. Conversely, habitual mindfulness seems to exert a beneficial effect on basal cortisol activity only in the menstrual phase. These findings emphasize the importance of a refined research on ER and its interaction with cycle phases to predict psychological and endocrinological changes in PMDD across the menstrual cycle. This will also help the progression towards an individualization of PMDD treatment targets.
The notes appear after the main text.
Note
All reported Cronbach's alpha values (α) in the paper refer to the present sample.
Financial support
This study was supported by a research grant from the Deutsche Forschungsgemeinschaft (DFG KU1464/6-1,3) to CK.
Conflict of interest
None.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
References
- Abler, B., & Kessler, H. (2009). Emotion regulation questionnaire – Eine deutschsprachige Fassung des ERQ von Gross und John. Diagnostica, 55(3), 144–152. 10.1026/0012-1924.55.3.144. [DOI] [Google Scholar]
- Accortt, E. E., Bismark, A., Schneider, T. R., & Allen, J. J. B. (2011). Diagnosing premenstrual dysphoric disorder: The reliability of a structured clinical interview. Archives of Women's Mental Health, 14(3), 265–267. 10.1007/s00737-011-0209-3. [DOI] [PubMed] [Google Scholar]
- Adam, E. K., Quinn, M. E., Tavernier, R., McQuillan, M. T., Dahlke, K. A., & Gilbert, K. E. (2017). Diurnal cortisol slopes and mental and physical health outcomes: A systematic review and meta-analysis. Psychoneuroendocrinology, 83, 25–41. 10.1016/j.psyneuen.2017.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aldao, A. (2013). The future of emotion regulation research: Capturing context. Perspectives on Psychological Science, 8(2), 155–172. 10.1177/1745691612459518. [DOI] [PubMed] [Google Scholar]
- Aldao, A., Nolen-Hoeksema, S., & Schweizer, S. (2010). Emotion-regulation strategies across psychopathology: A meta-analytic review. Clinical Psychology Review, 30(2), 217–237. 10.1016/j.cpr.2009.11.004. [DOI] [PubMed] [Google Scholar]
- Aldao, A., Sheppes, G., & Gross, J. J. (2015). Emotion regulation flexibility. Cognitive Therapy and Research, 39(3), 263–278. 10.1007/s10608-014-9662-4. [DOI] [Google Scholar]
- American Psychiatric Association. (2013). Diagnostic and statistical manual of mental Disorders (5th ed.). Arlington: American Psychiatric Association. [Google Scholar]
- Aperribai, L., Alonso-Arbiol, I., Balluerka, N., & Claes, L. (2016). Development of a screening instrument to assess premenstrual dysphoric disorder as conceptualized in DSM-5. Journal of Psychosomatic Research, 88, 15–20. 10.1016/j.jpsychores.2016.07.003. [DOI] [PubMed] [Google Scholar]
- Armstrong, R. A. (2014). When to use the Bonferroni correction. Ophthalmic and Physiological Optics, 34(5), 502–508. 10.1111/opo.12131. [DOI] [PubMed] [Google Scholar]
- Beck, A. T., Steer, R. A., & Brown, G. K. (1996). Manual for the beck depression inventory-II. San Antonio, Texas: Psychological Corporation. [Google Scholar]
- Beddig, T., & Kuehner, C. (2017). Aktuelle Aspekte zur Prämenstruellen Dysphorischen Störung – Ein Überblick. [Current aspects of premenstrual dysphoric disorder – A review]. Psychotherapie Psychosomatik Medizinische Psychologie, 67(12), 504–513. 10.1055/s-0043-113816. [DOI] [PubMed] [Google Scholar]
- Beddig, T., & Kuehner, C. (2020). Ambulatory assessment characteristics predict the clinical course of premenstrual dysphoric disorder. Psychotherapy and Psychosomatics, 89(6), 393–395. 10.1159/000505999. [DOI] [PubMed] [Google Scholar]
- Beddig, T., Reinhard, I., Ebner-Priemer, U., & Kuehner, C. (2020). Reciprocal effects between cognitive and affective states in women with premenstrual dysphoric disorder: An ecological momentary assessment study. Behaviour Research and Therapy, 131, 103613. 10.1016/j.brat.2020.103613. [DOI] [PubMed] [Google Scholar]
- Beddig, T., Reinhard, I., & Kuehner, C. (2019). Stress, mood, and cortisol during daily life in women with premenstrual dysphoric disorder (PMDD). Psychoneuroendocrinology, 109, 104372. 10.1016/j.psyneuen.2019.104372. [DOI] [PubMed] [Google Scholar]
- Blake, F. (1995). Cognitive therapy for premenstrual syndrome. Cognitive and Behavioral Practice, 2(1), 167–185. 10.1016/S1077-7229(05)80009-4. [DOI] [Google Scholar]
- Bosman, R. C., Jung, S. E., Miloserdov, K., Schoevers, R. A., & Aan Het Rot, M. (2016). Daily symptom ratings for studying premenstrual dysphoric disorder: A review. Journal of Affective Disorders, 189, 43–53. 10.1016/j.jad.2015.08.063. [DOI] [PubMed] [Google Scholar]
- Brown, K. W., & Ryan, R. M. (2003). The benefits of being present: Mindfulness and its role in psychological well-being. Journal of Personality and Social Psychology, 84(4), 822–848. 10.1037/0022-3514.84.4.822. [DOI] [PubMed] [Google Scholar]
- Cohen, L. S., Soares, C. N., Otto, M. W., Sweeney, B. H., Liberman, R. F., & Harlow, B. L. (2002). Prevalence and predictors of premenstrual dysphoric disorder (PMDD) in older premenopausal women: The Harvard study of moods and cycles. Journal of Affective Disorders, 70(2), 125–132. 10.1016/S0165-0327(01)00458-X. [DOI] [PubMed] [Google Scholar]
- Costa, P. T., & McCrae, R. R. (1992). Revised NEO personality inventory (NEO-PI-R) and NEO five-factor inventory (NEO-FFI) professional manual. Odessa, Florida: Psychological Assessment Resources. [Google Scholar]
- Courvoisier, D. S., Eid, M., & Lischetzke, T. (2012). Compliance to a cell phone-based ecological momentary assessment study: The effect of time and personality characteristics. Psychological Assessment, 24(3), 713. 10.1037/a0026733. [DOI] [PubMed] [Google Scholar]
- Craner, J. R., Sigmon, S. T., Martinson, A. A., & McGillicuddy, M. L. (2014). Premenstrual disorders and rumination. Journal of Clinical Psychology, 70(1), 32–47. 10.1002/jclp.22007. [DOI] [PubMed] [Google Scholar]
- Craner, J. R., Sigmon, S. T., & Young, M. A. (2016). Self-focused attention and symptoms across menstrual cycle phases in women with and without premenstrual disorders. Cognitive Therapy and Research, 40(1), 118–127. 10.1007/s10608-015-9721-5. [DOI] [Google Scholar]
- Dawson, D. N., Eisenlohr-Moul, T. A., Paulson, J. L., Peters, J. R., Rubinow, D. R., & Girdler, S. S. (2018). Emotion-related impulsivity and rumination predict the perimenstrual severity and trajectory of symptoms in women with a menstrually related mood disorder. Journal of Clinical Psychology, 74(4), 579–593. 10.1002/jclp.22522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doane, L. D., Chen, F. R., Sladek, M. R., Van Lenten, S. A., & Granger, D. A. (2015). Latent trait cortisol (LTC) levels: Reliability, validity, and stability. Psychoneuroendocrinology, 55, 21–35. 10.1016/j.psyneuen.2015.01.017. [DOI] [PubMed] [Google Scholar]
- Eggert, L., Kleinstäuber, M., Hiller, W., & Witthöft, M. (2017). Emotional interference and attentional processing in premenstrual syndrome. Journal of Behavior Therapy and Experimental Psychiatry, 54(1), 77–87. 10.1016/j.jbtep.2016.07.002. [DOI] [PubMed] [Google Scholar]
- Ehring, T., Zetsche, U., Weidacker, K., Wahl, K., Schönfeld, S., & Ehlers, A. (2011). The perseverative thinking questionnaire (PTQ): Validation of a content-independent measure of repetitive negative thinking. Journal of Behavior Therapy and Experimental Psychiatry, 42(2), 225–232. 10.1016/j.jbtep.2010.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenlohr-Moul, T. (2019). Premenstrual disorders: A primer and research agenda for psychologists. The Clinical Psychologist, 72(1), 5–17. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7193982/. [PMC free article] [PubMed] [Google Scholar]
- Fries, E., Hesse, J., Hellhammer, J., & Hellhammer, D. H. (2005). A new view on hypocortisolism. Psychoneuroendocrinology, 30(10), 1010–1016. 10.1016/j.psyneuen.2005.04.006. [DOI] [PubMed] [Google Scholar]
- Gollenberg, A. L., Hediger, M. L., Mumford, S. L., Whitcomb, B. W., Hovey, K. M., Wactawski-Wende, J., & Schisterman, E. F. (2010). Perceived stress and severity of perimenstrual symptoms: The BioCycle study. Journal of Women's Health, 19(5), 959–967. https://doi.org/10.1089=jwh.2009.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han, J., Cha, Y., & Kim, S. (2019). Effect of psychosocial interventions on the severity of premenstrual syndrome: A meta-analysis. Journal of Psychosomatic Obstetrics & Gynecology, 40(3), 176–184. 10.1080/0167482x.2018.1480606. [DOI] [PubMed] [Google Scholar]
- Hantsoo, L., & Epperson, C. N. (2015). Premenstrual dysphoric disorder: Epidemiology and treatment. Current Psychiatry Reports, 17(11), 87. 10.1007/s11920-015-0628-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hantsoo, L., & Riddle, J. (2021). Treatment of premenstrual dysphoric disorder (PMDD): Advances and challenges. Advances in Psychiatry and Behavioral Health, 1(1), 91–106. 10.1016/j.ypsc.2021.05.009. [DOI] [Google Scholar]
- Hox, J. J., Moerbeek, M., & Van de Schoot, R. (2017). Multilevel analysis: Techniques and applications. New York, New York: Routledge. [Google Scholar]
- Huang, Y., Zhou, R., Wu, M., Wang, Q., & Zhao, Y. (2015). Premenstrual syndrome is associated with blunted cortisol reactivity to the TSST. Stress, 18(2), 160–168. 10.3109/10253890.2014.999234. [DOI] [PubMed] [Google Scholar]
- Huffziger, S., Ebner-Priemer, U., Eisenbach, C., Koudela, S., Reinhard, I., Zamoscik, V. E., … Kuehner, C. (2013). Induced ruminative and mindful attention in everyday life: An experimental ambulatory assessment study. Journal of Behavior Therapy and Experimental Psychiatry, 44(3), 322–328. 10.1016/j.jbtep.2013.01.007. [DOI] [PubMed] [Google Scholar]
- Huffziger, S., Ebner-Priemer, U., Koudela, S., Reinhard, I., & Kuehner, C. (2012). Induced rumination in everyday life: Advancing research approaches to study rumination. Personality and Individual Differences, 53(6), 790–795. 10.1016/j.paid.2012.06.009. [DOI] [Google Scholar]
- Hunter, M. S., Ussher, J. M., Cariss, M., Browne, S., Jelley, R., & Katz, M. (2002). Medical (fluoxetine) and psychological (cognitive–behavioural therapy) treatment for premenstrual dysphoric disorder: A study of treatment processes. Journal of Psychosomatic Research, 53(3), 811–817. 10.1016/S0022-3999(02)00338-0. [DOI] [PubMed] [Google Scholar]
- Kiesner, J., & Granger, D. A. (2016). A lack of consistent evidence for cortisol dysregulation in premenstrual syndrome/premenstrual dysphoric disorder. Psychoneuroendocrinology, 65, 149–164. 10.1016/j.psyneuen.2015.12.009. [DOI] [PubMed] [Google Scholar]
- Klatzkin, R. R., Lindgren, M. E., Forneris, C. A., & Girdler, S. S. (2010). Histories of major depression and premenstrual dysphoric disorder: Evidence for phenotypic differences. Biological Psychology, 84(2), 235–247. 10.1016/j.biopsycho.2010.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinstäuber, M., Schmelzer, K., Ditzen, B., Andersson, G., Hiller, W., & Weise, C. (2016). Psychosocial profile of women with premenstrual syndrome and healthy controls: A comparative study. International Journal of Behavioral Medicine, 23(6), 752–763. 10.1007/s12529-016-9564-9. [DOI] [PubMed] [Google Scholar]
- Kleinstäuber, M., Witthöft, M., & Hiller, W. (2012). Cognitive-behavioral and pharmacological interventions for premenstrual syndrome or premenstrual dysphoric disorder: A meta-analysis. Journal of Clinical Psychology in Medical Settings, 19(3), 308–319. 10.1007/s10880-012-9299-y. [DOI] [PubMed] [Google Scholar]
- Kuehner, C., & Nayman, S. (2021). Premenstrual exacerbations of mood disorders: Findings and knowledge gaps. Current Psychiatry Reports, 23, 78. 10.1007/s11920-021-01286-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuehner, C., Welz, A., Reinhard, I., & Alpers, G. W. (2017). Lab meets real life: A laboratory assessment of spontaneous thought and its ecological validity. PLoS ONE, 12(9), e0184488. 10.1371/journal.pone.0184488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lustyk, M. K., Gerrish, W. G., Douglas, H., Bowen, S., & Marlatt, G. A. (2011). Relationships among premenstrual symptom reports, menstrual attitudes, and mindfulness. Mindfulness, 2(1), 37–48. 10.1007/s12671-011-0041-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lustyk, M. K., Gerrish, W. G., Shaver, S., & Keys, S. L. (2009). Cognitive-behavioral therapy for premenstrual syndrome and premenstrual dysphoric disorder: A systematic review. Archives of Women's Mental Health, 12(2), 85–96. 10.1007/s00737-009-0052-y. [DOI] [PubMed] [Google Scholar]
- McRae, K., & Gross, J. J. (2020). Emotion regulation. Emotion, 20(1), 1–9. 10.1037/emo0000703. [DOI] [PubMed] [Google Scholar]
- Mikkelsen, M. B., Tramm, G., Zachariae, R., Gravholt, C. H., & O'Toole, M. S. (2021). A systematic review and meta-analysis of the effect of emotion regulation on cortisol. Comprehensive Psychoneuroendocrinology, 5, 100020. 10.1016/j.cpnec.2020.100020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller, A., Vo, H., Huo, L., Roca, C., Schmidt, P. J., & Rubinow, D. R. (2010). Estrogen receptor alpha (ESR-1) associations with psychological traits in women with PMDD and controls. Journal of Psychiatric Research, 44(12), 788–794. 10.1016/j.jpsychires.2010.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Namavar Jahromi, B., Pakmehr, S., & & Hagh-Shenas, H. (2011). Work stress, premenstrual syndrome and dysphoric disorder: Are there any associations?. Iranian Red Crescent Medical Journal, 13(3), 199–202. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3371938/. [PMC free article] [PubMed] [Google Scholar]
- Naragon-Gainey, K., McMahon, T. P., & Chacko, T. P. (2017). The structure of common emotion regulation strategies: A meta-analytic examination. Psychological Bulletin, 143(4), 384–427. 10.1037/bul0000093. [DOI] [PubMed] [Google Scholar]
- Nezlek, J. B., Schroeder-Abé, M., & Schuetz, A. (2006). Mehrebenenanalysen in der psychologischen Forschung. Vorteile und Möglichkeiten der Mehrebenenmodellierung mit Zufallskoeffizienten. Psychologische Rundschau, 57(4), 213–223. 10.1026/0033-3042.57.4.213. [DOI] [Google Scholar]
- Odber, J., Cawood, E. H., & Bancroft, J. (1998). Salivary cortisol in women with and without perimenstrual mood changes. Journal of Psychosomatic Research, 45(6), 557–568. 10.1016/S0022-3999(98)00061-0. [DOI] [PubMed] [Google Scholar]
- Osborn, E., Brooks, J., O'Brien, P. M. S., & Wittkowski, A. (2020). Suicidality in women with premenstrual dysphoric disorder: A systematic literature review. Archives of Women's Mental Health, 24(2), 173–184. 10.1007/s00737-020-01054-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otto, L. R., Sin, N. L., Almeida, D. M., & Sloan, R. P. (2018). Trait emotion regulation strategies and diurnal cortisol profiles in healthy adults. Health Psychology, 37(3), 301–305. 10.1037/hea0000564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owens, S. A., & Eisenlohr-Moul, T. (2018). Suicide risk and the menstrual cycle: A review of candidate RDoC mechanisms. Current Psychiatry Reports, 20, 106. 10.1007/s11920-018-0962-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papa, A., & Follette, W. C. (2015). Dismantling studies of psychotherapy. The Encyclopedia of Clinical Psychology, 1–6. 10.1002/9781118625392.wbecp523. [DOI] [Google Scholar]
- Payne, L. A., Seidman, L. C., Romero, T., & Sim, M. S. (2020). An open trial of a mind–body intervention for young women with moderate to severe primary dysmenorrhea. Pain Medicine, 21(7), 1385–1392. 10.1093/pm/pnz378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purnamasari, K. D., Rohita, T., Zen, D. N., & Ningrum, W. M. (2020). The effect of deep breathing exercise on menstrual pain perception in adolescents with primary dysmenorrhea. Pertanika Journal, 2(28), 649–657. http://www.pertanika2.upm.edu.my/resources/files/Pertanika%20PAPERS/JST%20Vol.%2028%20(2)%20Apr.%202020/13%20JST-1637-2019.pdf. [Google Scholar]
- Rnic, K., Jopling, E., Tracy, A., & LeMoult, J. (2022). Emotion regulation and diurnal cortisol: A longitudinal study of early adolescents. Biological Psychology, 167, 108212. 10.1016/j.biopsycho.2021.108212. [DOI] [PubMed] [Google Scholar]
- Schlotz, W. (2019). Investigating associations between momentary stress and cortisol in daily life: What have we learned so far? Psychoneuroendocrinology, 105, 105–116. 10.1016/j.psyneuen.2018.11.038. [DOI] [PubMed] [Google Scholar]
- Schmalenberger, K. M., Tauseef, H. A., Barone, J. C., Owens, S. A., Lieberman, L., Jarczok, M. N., … Eisenlohr-Moul, T. A. (2021). How to study the menstrual cycle: Practical tools and recommendations. Psychoneuroendocrinology, 123, 104895. 10.1016/j.psyneuen.2020.104895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheppes, G., Scheibe, S., Suri, G., & Gross, J. J. (2011). Emotion-regulation choice. Psychological Science, 22(11), 1391–1396. 10.1177/0956797611418350. [DOI] [PubMed] [Google Scholar]
- Stalder, T., Kirschbaum, C., Kudielka, B. M., Adam, E. K., Pruessner, J. C., Wüst, S., … Clow, A. (2016). Assessment of the cortisol awakening response: Expert consensus guidelines. Psychoneuroendocrinology, 63, 414–432. 10.1016/j.psyneuen.2015.10.010. [DOI] [PubMed] [Google Scholar]
- Timm, C., Rachota-Ubl, B., Beddig, T., Zamoscik, V. E., Ebner-Priemer, U., Reinhard, I., … Kuehner, C. (2018). Mindfulness-based attention training improves cognitive and affective processes in daily life in remitted patients with recurrent depression: A randomized controlled trial. Psychotherapy and Psychosomatics, 87(3), 184–186. 10.1159/000488862. [DOI] [PubMed] [Google Scholar]
- Trull, T. J., & Ebner-Priemer, U. (2013). Ambulatory assessment. Annual Review of Clinical Psychology, 9, 151–176. 10.1146/annurev-clinpsy-050212-185510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ubeda-D'Ocasar, E., Jimenez Diaz-Benito, V., Gallego-Sendarrubias, G. M., Valera-Calero, J. A., Vicario-Merino, A., & Hervas-Perez, J. P. (2020). Pain and cortisol in patients with fibromyalgia: Systematic review and meta-analysis. Diagnostics, 10, 922. 10.3390/diagnostics10110922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weise, C., Kaiser, G., Janda, C., Kues, J. N., Andersson, G., Strahler, J., & Kleinstäuber, M. (2019). Internet-Based cognitive-behavioural intervention for women with premenstrual dysphoric disorder: A randomized controlled trial. Psychotherapy and Psychosomatics, 88(1), 16–29. 10.1159/000496237. [DOI] [PubMed] [Google Scholar]
- Wenzel, M., Rowland, Z., & Kubiak, T. (2021). How much variance can event intensity and emotion regulation strategies explain in momentary affect in daily life? Emotion. Advance online publication. 10.1037/emo0000816. [DOI] [PubMed] [Google Scholar]
- Wenzel, M., Rowland, Z., Weber, H., & Kubiak, T. (2020). A round peg in a square hole: Strategy-situation fit of intra- and interpersonal emotion regulation strategies and controllability. Cognition and Emotion, 34(5), 1003–1009. 10.1080/02699931.2019.1697209. [DOI] [PubMed] [Google Scholar]
- Wittchen, H. U., Becker, E., Lieb, R., & Krause, P. (2002). Prevalence, incidence and stability of premenstrual dysphoric disorder in the community. Psychological Medicine, 32(1), 119–132. 10.1017/s0033291701004925. [DOI] [PubMed] [Google Scholar]
- Wittchen, H. U., Zaudig, M., & Fydrich, T. (1997). Structured clinical interview for DSM-IV. Göttingen: Hogrefe. [Google Scholar]
- Wu, M., Liang, Y., Wang, Q., Zhao, Y., & Zhou, R. (2016). Emotion dysregulation of women with premenstrual syndrome. Scientific Reports, 6(1), 1–10. 10.1038/srep38501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang, J., Mao, Y., Niu, Y., Wei, D., Wang, X., & Qiu, J. (2020). Individual differences in neuroticism personality trait in emotion regulation. Journal of Affective Disorders, 265, 468–474. 10.1016/j.jad.2020.01.086. [DOI] [PubMed] [Google Scholar]
- Yen, J. Y., Lin, P. C., Huang, M. F., Chou, W. P., Long, C. Y., & Ko, C. H. (2020). Association between generalized anxiety disorder and premenstrual dysphoric disorder in a diagnostic interviewing study. International Journal of Environmental Research and Public Health, 17, 988. 10.3390/ijerph17030988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zoccola, P. M., & Dickerson, S. S. (2012). Assessing the relationship between rumination and cortisol: A review. Journal of Psychosomatic Research, 73(1), 1–9. 10.1016/j.jpsychores.2012.03.007. [DOI] [PubMed] [Google Scholar]
- Zorn, J. V., Schuer, R. R., Boks, M. P., Kahn, R. S., Joëls, M., & Vinkers, C. H. (2017). Cortisol stress reactivity across psychiatric disorders: A systematic review and meta-analysis. Psychoneuroendocrinology, 77, 25–36. 10.1016/j.psyneuen.2016.11.036. [DOI] [PubMed] [Google Scholar]