Abstract
The human lymphocyte antigen B27 (HLA B27) is a member of the HLA class I family of genes in the major histocompatibility complex whose name goes back to its discovery in studies of transplanted tissue compatibility. Its prevalence in the mid-European population is about 8%. The association of HLA B27 alleles with ankylosing spondylitis (AS), a highly heritable disease, which is part of the spectrum of axial spondyloarthritis (axSpA), was discovered 50 years ago. HLA B27 explains less than 30% of the total genetic load. About 60%–90% of axSpA patients worldwide carry HLA B27. The prevalence of the disease is linked to the frequency of HLA B27 in the population which implies that there are relevant differences. Among the roughly 200 subtypes known there are two which are not disease associated. The function of HLA class I molecules is to present peptides to the immune system to defend the organism against microbes targeted by CD8+T cells. This is much supported by the role of the endoplasmic reticulum aminopeptidase 1 (ERAP 1) in AS, an enzyme that is responsible for the intracellular trimming of peptides, since polymorphisms of this gene are only associated with HLA-B27+ disease. The arthritogenic peptide hypothesis trying to explain the pathogenesis of AS is based on that very immune function assuming that also self peptides can be presented. HLA-B27 also plays an important role in classification, diagnosis and severitiy of axSpA.
Keywords: Spondylitis, Ankylosing; Arthritis; Inflammation
Part I genetics, structure and function of HLA B27
Introduction
Human lymphocyte antigens (HLAs) have been discovered in the first half of the last century in the context of histocompatibility research. The association of HLA B27 with a common rheumatic disease, ankylosing spondylitis (AS), was discovered by Schlosstein et al and Brewerton et al in parallel in 1973,1 2 now 50 years ago. This is the strongest association of any known association of a common variant with any human disease. The heritability of AS is believed to explain 80% of the pathogenesis of the disease.3 Many susceptibility alleles for the disease have been identified, with 113 established loci contributing roughly 10% of the heritability of AS, over and above the major effect of HLA B27, which determines ~25% of the genetic risk.3 Other related diseases belonging to the spectrum of spondyloarthritis (SpA) such as Reiter’s syndrome* and anterior uveitis (AU) are also associated with HLA B27.4 5
* This term has been abandoned, the disease is now internationally named reactive arthritis.
The canonical function of HLA-B27 is to present peptides to CD8 lymphocytes, leading to adaptive immune responses against foreign and, potentially, self-antigens. The ‘arthritogenic peptide’ theory as a possible mechanism by which HLA-B27 induces AS proposes that HLA-B27 presents peptides from exogenous sources to CD8 lymphocytes, which subsequently cross-react with antigens at certain sites causing inflammation and associated symptoms.6 7
The major histocompatibility complex
HLA B27 is a member of the HLA class I family of genes in the major histocompatibility complex (MHC) whose name goes back to its discovery in studies of transplanted tissue compatibility. Its role in the immune system is to present peptide antigens to CD8 T cells,6 in addition to T-cell receptors (TCR)s7 (see figure 1).
Figure 1.
(A, B): Confirmation of HLA B27. (A) Confirmation of HLA B27 showing 3 alpha chains, ß2 microglobulin and the peptide binding cleft. Sehnal et al.83 (B) Confirmation of HLA-B*2705 with the alpha chains shown in green and ß2 microglobulin in yellow, complexed to a fragment of the influenza nucleoprotein NP383-391 (red).
The MHC gene family is divided into three subgroups: MHC class I (A, B and C), MHC class II (HLA-DP, HLA-DM, HLA-DOA, HLA-DOB, HLA-DQ and HLA-DR) and MHC class III. Among the 224 genes present in the human MHC, there are two types of genes coding for MHC class I or II proteins. These highly polymorphic genes8 encode for more than 19 000 HLA class I (IMGT, http://www.imgt.org). HLA B27 is expressed in nucleated cells and in platelets—in essence in all cells but red blood cells.3 6 Different disease associations may be explained by changes in the peptide-binding repertoire of an HLA molecule.9
Structure of HLA B27
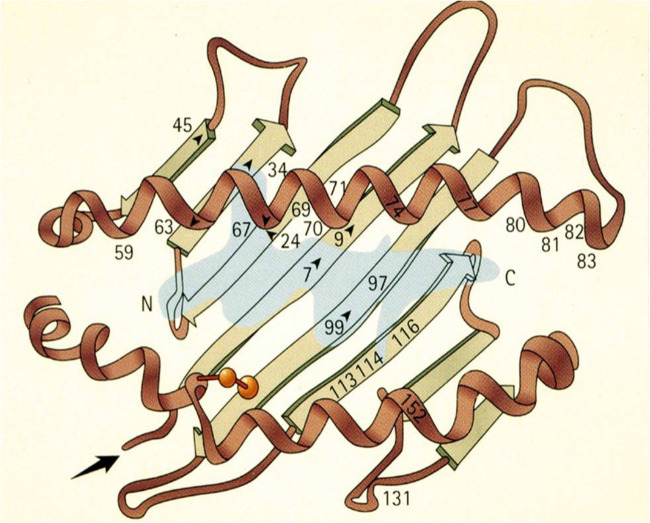
Studies using X-ray crystallography in the 1990ies have revealed that the electron density in the antigen-binding site of HLA-B27 suggests the presence of non-americ peptides in a largely extended conformation.10 Pockets in the antigen-binding cleft of HLA molecules normally bind four side chains and the amino and carboxyl termini of the peptide (figure 2). However, the structure of HLA B27 is different from other HLA molecules, since there are six ‘pockets’ with antigen binding capacities11 (see figure 3). Importantly, a tight hydrogen-bonding network between the HLA-B*2705 B-pocket and the peptide P2 arginine guanidinium anchor has been described.12 The rather unique B pocket contains a glutamic acid residue at position 45 and an unpaired cysteine residue at position 67 (Cys67) that is likely to be involved in the formation of aberrant disulfide-linked heavy chain (HC) complexes which may be pathogenetically relevant as they can form stable homodimers lacking ß2-microglobulin.9 The extensive polymorphism of HLA B 27 results from the considerable variability of the HC component of the protein.8 The positions that have the strongest effects on peptide binding are the most polymorphic ones.3 6 A Lys residue at position 70 increases the reactivity of cysteine at position 67.4 7 This may also explain why HLA-B27 has a stronger tendency to fold slowly or misfold in the endoplasmic reticulum than other HLA antigens, which could induce a proinflammatory response. In addition, involvement of the F pocket,13 and the substitution of an aspartate with a histidine on position 116 seem to also play a role.14 Furthermore, the amino acid sequence of HLA-B 27 at position 97, in the epitope-binding groove, may be relevant for HLA associations with AS.15
Figure 2.
Peptide presented in HLA B27. The figure shows a peptide antigen in blue, usually 8–10 amino acids long, present in the pocket of HLA-B27 molecule for presentation to CD8+cytotoxic T cells specific for that peptide antigen. The letter N denotes the amino terminus (end point) and C stands for the carboxy terminus of the bound peptide. The numbers indicate the locations of important amino acids of HLA-B27 molecules. (From: Khan84 2005).
Figure 3.
Peptide binding sites of HLA B27 subtypes. *Mapping the polymorphic and conserved residues of the HLA-B27 subtypes in terms of the specificity pockets described in the X-ray crystallographic structure of HLA-A*0201. (A) Specificity pockets A-F of HLA- A*0201#. (B) HLA-B27 subtype polymorphisms (solid areas) and B pocket conserved residues (shaded areas) mapped onto the ribbon structure of HLA-A*0201##. *Buxton et al.12 #As described by Saper et al.85 (Copyright permission of Academic Press). ##As described by Bjorkman et al.86
Function of HLA B27
If a cell is infected by viruses or bacteria, fragments of the microbe, for example, peptides from digested proteins which are small polymers of about 8–10 amino acids in length, are transported to the cell surface where they, bound to HLA class I molecules, stimulate CD8-positive T cells to defend the organism.10 Thus, one physiological immune function of HLA-B27 is to protect against infections—for example, against the HIV and hepatitis C (HCV) (see figure 1B).16–18 Thus, it seems likely that HLA B27 confers an evolutionary advantage related to the defence against infections.19
HLA B27 subtypes
In early 2017, a total of 213 alleles of HLA-B*27 were known at the nucleotide sequence level, while, at the translated protein level, there were 160 known subtypes based on one or more amino acid sequence differences.20 Of relevance, some of these subtypes exhibit a differential association with AS. The most common disease-associated subtypes are B*2702 (Mediterranean population), B*2705 (whites and American Indians) and B*2704 (Asians). In contrast, the subtypes HLA-B*2706 and HLA-B*2709 are not associated with AS,21 22 and the HLA B27 association with AS is less strong in Arab countries.23 In contrast, Inuit and native Alaskans have the highest rates of HLA B27 in the world and correspondingly the highest prevalence of AS.24
The frequency of HLA B27 homozygotes among patients with AS was slightly greater than expected indicating that such patients are more susceptible to developing AS than HLA B27 heterozygotes.25 However, the clinical symptoms and the degree of radiographic damage26 was not much different between these two groups.
AS: genetics and HLA-B27
For classification purposes, the term axial SpA (axSpA) covers both patients with definite structural damage in the sacroiliac joints (SIJ) named radiographic axSpA or AS (according to the modified New York (mNY) criteria) and patients without such definite structural changes (non-radiographic axSpA), see also part II of this review. However, most data are available in AS—a highly heritable rheumatic disease characteristically causing chronic inflammation and new bone formation in the axial skeleton, while some patients also show inflammation in peripheral joints and entheses, the anterior uvea, and, less commonly, other organs such as the skin and the gut.27
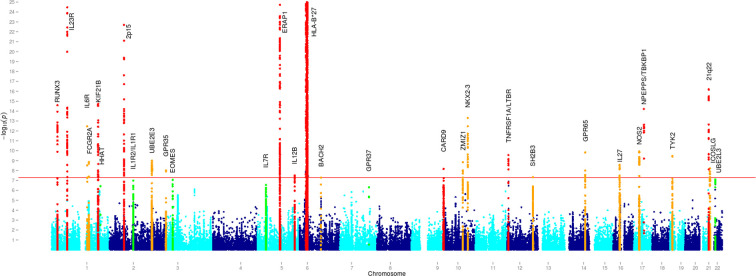
The worldwide distribution of AS is related to the prevalence of HLA B27, but the underlying mechanism remains unclear. While HLA B27 alleles are found in about 85% of patients, there is strong evidence indicating that also other HLA-B alleles and MHC genes are involved, as well as non-MHC loci.3 9 Altogether, 116 genetic associations for AS have now been identified contributing ~30% of the overall genetic risk.7 The HLA associations of AS are complex and multiple HLA alleles are involved. HLA-B27 contributes ~25% of the heritability of AS, and non-MHC loci identified to date contribute another ~10%.3 Key pathways identified include aminopeptidases (ERAP) and interleukin (IL)-23 (figure 4) but multiple other pathways have been reported.3
Figure 4.
Disease associations at genome-wide significance were observed for 25 loci, including the MHC. Manhattan plots in the European Cohort Analysis Association peaks are colour coded in red if previously identified, orange if novel and in green if identified as suggestive association. MHC, major histocompatibility complex. The strongest association found was, expectedly, for HLA B27 but ERAP 1 and the IL-23 receptor were also clearly associated. The significance of other weaker associations has in most cases remained unclear.
AS is highly familial, with an increased risk in siblings of AS patients 82-fold higher than the disease prevalence in the general community. Twin studies have suggested that even >90% of AS susceptibility is genetic in origin while the environmental trigger is likely ubiquitous. As already mentioned, one of the strongest genetic associations (OR=60) of any polygenic human disease is with HLA B27.3 Genome wide association studies (GWAS) show that AS involves both the adaptive and innate immune systems, and that the IL-23 and TNF pathways are the major effector pathways involved.28 29
The chances of an HLA B27+ individual to develop AS in a lifetime are only 1%–2%. This percentage may increase to 20% for people with a first-degree relative (FDR) with AS.3 In an early study,30 the recurrence risk of AS in different degrees of relatives (independent of HLA B27) was found to be: 63% in monozygotic twins, 8.2% in FDR, 1.0% in second-degree relatives and 0.7% in third-degree relatives. The parent–child recurrence risk was 7.9%, and this was not significantly different from the sibling recurrence risk (8.2%) suggesting the absence of a dominant genetic effect in AS.30
Disease severity as defined by definite radiographic changes in the SIJ according to the mNY criteria is an additional risk factor.31 Affected mothers pass on the disease significantly more often to their offspring than do affected fathers.32
The combination of HLA B60 with HLA B27 increases chances of developing AS by 3–6 times.3 Furthermore, HLA B27 interacting with the endoplasmic reticulum (ER) aminopeptidase 1 (ERAP1) gene contributes to the development of AS (see below).29
The relationship between various ethnicities and the prevalence of HLA B27 is well known, and the prevalence of AS is closely related. Thus, the HLA B27 prevalence is 10%–16% in Scandinavian populations, and 25%–50% in Inuit, Yupik and Indigenous Northern Americans,3 23 while less than 60% of American black patients with AS carry HLA B27, and the allele is extremely rare in African blacks of unmixed ancestry.33
There are additional HLA-B variants associated with AS, and also other HLA class I and II variants.3 For example, HLA-B51 being also a risk variant for AS, is the major risk allele for Behçet’s syndrome.34 The important role of antigen presentation in the onset of HLA-class I-associated diseases is much supported by the fact that at least in AS, Behçet and psoriasis, which are associated with HLA-B*27, HLA-B*51 and HLA-C*06, respectively, share an association with ERAP1 (see below). Interestingly, these alleles were also shown to protect against some viral infections such as HIV and hepatitis C.35
How does HLA-B27 contribute to the susceptibility to AS and axSpA?
There are several hypotheses how to explain the role of HLA-B 27 in disease but none is definitely proven. The two already mentioned are: (1) misfolding of the HLA-B27 molecule inside the cell and (2) free HC of HLA-B27 homodimers form a new MHC class II-like molecule which can stimulate innate and adaptive immune responses.
The third hypothesis is based on the assumption that arthritogenic peptides derived from microbes are presented by HLA-B27 to stimulate CD8+T cells which subsequently interact with HLA-B27-bound self-peptides. GWAS studies have confirmed strong genetic links with ERAP1 polymorphism in HLA-B2705 positive patients with AS, but not in HLA-B27-negative patients.36 Mainly two ERAP proteins are involved in peptide trimming in the ER which hereby determine their amino acid sequence before they are being transported to the cell surface to be presented by HLA molecules. The fact that HLA-B27 subtypes which are associated with AS differ by only one or two amino acids in the antigen binding groove as compared with non-associated subtypes support this hypothesis. ERAP1 variants contribute to pathogenesis of diseases by providing peptides presented by HLA B27.6 9 Interestingly, ERAP1 is only associated with AS in HLA-B27 positive cases, or HLA-B27-negative/HLA-B*4001 positive cases.37 Of interest, the same ERAP1 haplotypes interact with HLA-Cw6 in psoriasis, and HLA-B51 in Behçet’s disease.35 The ERAP2 association is present in both HLA B27+ and HLA B27-negative AS.
Effects on the peptidome presented by HLA B27 may either lead to presentation of arthritogenic peptides or failure to present disease-protective peptides.6 9
Arthritogenic peptides may be similar and cross-reactive to human ‘self’-peptides, and prime CD8+T cells to targets endogenous antigens detected at the site of disease.38 39 T cell expansions of a restricted pool of CD8+T cells using the same TCR expressing a disease-associated public β-chain variable region-complementary-determining region 3β (BV9-CDR3β) motif have been more frequently found in blood and synovial fluid of AS patients but only in a small proportion of healthy HLA B27 carriers. Peptides identified from these TCR were shared self-peptides and microbial peptides that activated AS-derived TCRs.40 These data suggest that there may be several microbial triggers with shared structural features which induce cross-reactive self-peptides. These findings strongly support that AS (axSpA) is driven by presentation of antigenic peptides to the adaptive immune system by HLA-B27.40
The introduction of HLA B27 and human ß2-microglobulin genes into a species of rats known to be quite susceptible to experimentally induced inflammatory disease has drawn a lot of intention more than 30 years ago because the pattern of organ system involvement with the gastrointestinal tract, peripheral and vertebral joints, the male genital tract, skin, nails and the heart showed a striking resemblance to HLA B27-associated human diseases.41 The most interesting result out of many experiments following on that first report was that the B27 transgenic rats raised in a germfree environment did not develop inflammatory intestinal or peripheral joint disease, whereas the skin and genital inflammatory lesions remained unaffected by the germfree state.42
In conclusion of this part, the pathogenetic background of the strong HLA association of AS has not been fully elucidated to date. Recent findings have rather supported the arthritogenic peptide hypothesis.
Part II clinical implications of determining HLA B27 in patients under suspicion of axSpA
The association of HLA B27 with AS discovered in 19731 2 is the strongest association with any human disease known to date. However, the association of HLA-B27 with AS and other SpA is not only interesting from a pathogenetic point of view3 but also from the clinical perspective which implies the usefulness of determining HLA B27 in the process of finding the correct diagnosis of axSpA.
The first reported experience, in 1975, was to study the prevalence of AS in HLA B27+ individuals.43 Based on a survey with a questionnaire and pelvic radiographs of 78 HLA B27+ blood donors a 20% prevalence of AS was found which was a lot lower in later blood donor studies looking for patients with SpA published in 1998.44 In 1977, it was reported for the first time that inflammatory back pain (IBP) was a good discriminating factor for patients with AS.45
The first thought, in 1976, that HLA B27 could be useful for diagnosis was related to sex and especially to women46 because it became clear that it was much more difficult to diagnose AS in women than in men.47 However, an expert review some years later in 1980 concluded ‘that knowledge of the patient’s HLA-B27 status seems to provide only minimal help to the physician’.48
A more sophisticated review in 198249 confirmed that a diagnosis of AS can, in most patients, be made on the basis of the history, physical examination and roentgenographic findings and that HLA-B27 should not be used as a routine diagnostic test. It was also made clear that B27 testing cannot be used to screen an asymptomatic population to detect the disease, and it was recommended not to use it as a routine diagnostic test. However, probability graphs derived from Bayes’ theorem suggested that the proper use of B27 testing is of clinical value as an aid to diagnosis in certain patients. Importantly, the authors also stressed that the test result does not absolutely confirm or exclude the presence of AS; it merely provides a probability statement for the patient. Thus, it was concluded that the test is useful to physicians who understand probability reasoning in clinical decision making.49 This statement is still valid 40 years later.
In a Dutch study published in 198450 on 61 HLA-B27+FDR and 40 HLA-B27− relatives of 20 HLA-B27+ patients with AS only the HLA-B27+ relatives (n=24, 45 years or older) had AS (21%). In a population study with almost 3000 individuals 45 years or older (phenotype frequency of HLA-B27 7.8%), 1.3% of the HLA-B27+ subjects had AS.50 In two family studies of together 50 HLA-B27+ subjects with AS and 201 of their FDR were included.51 Only HLA B27+ but not HLA B27-negative FDR of AS patients reported characteristic symptoms which suggested that the spectrum of clinical manifestations of AS may include individuals with symptomatic disease, but without radiographic evidence of abnormalities of the SIJ or the spine.51 The number of females found in this group was relatively large—an early indication that the prognosis of radiographic progression in AS is better for females than for males.52
In a follow-up study 35 years later,53 a total of 485 individuals, 125 patients with AS and 360 FDR, completed a postal questionnaire. The lifetime risk of axSpA for HLA-B27+ FDR found was substantial: 27.1%, and radiographic changes in the SIJ (as defined by the mNY criteria) came out as an additional risk factor. It was also confirmed that affected mothers pass on the disease more often to their offspring than affected fathers.53
HLA B27 was not part of the modified NY criteria for AS,54 and this was similar in the ESSG criteria for SpA55 published several years later. Although HLA-B27 differentiated well between SpA and non-SpA in the latter study, it was not included because the criteria were designed to be also applied in epidemiological studies all over the world. A ‘unified concept’ for SpA had already been proposed by Moll and Wright in 1974 shortly after the discovery of the HLA B27 association with AS but without yet including it in the concept.56
A higher frequency of HLA B27 was also found in pacemaker patients in whom disturbances of the cardiac conduction system had been diagnosed57 and in patients with complete heart block.58 However, these findings had no influence on clinical practice over time.
In a Belgian study with 440 HLA B27+ patients, 58% had peripheral joint involvement. Three patterns of peripheral joint involvement were described.59 The concept of undifferentiated SpA came up60 61—with varying percentages of HLA B27 positivity.
In the early 1990s, it was recognised that the current criteria for diagnosis of AS are too restricted, and do not recognise the existence of a much wider disease spectrum. As an example, it was noted that radiographic changes in the SIJ are extremely frequent in AS but not in early or atypical forms of the disease.62
In another approach in Germany, early diagnostic criteria were proposed using a combination of the HLA-B27 test with clinical data, ESR and radiological signs, and evaluated in follow-up study of 77 patients with possible AS and controls with other rheumatic diseases.63 After 10 years, 59% had definite AS according to the NY criteria, 19% had possible or undifferentiated SpA, whereas in 22% of patients other diseases were diagnosed.64
The discovery of the usefulness of MRI to detect sacroiliitis early in 199465 and the dramatic effect of anti-TNF therapy in patients with active AS in 200266 had a strong influence on future developments in the field and the role of HLA B27. In 2004, it was calculated that HLA-B27 and MRI had the highest LRs to increase the probability of an axSpA diagnosis.67
In consequence, new classification criteria for axSpA68 and peripheral SpA69 were developed by the Assessment of SpondyloArthritis International Society, and both included HLA B27 testing.
An early review comparing HLA B27+ and HLA B27– patients with AS70 found studies which reported that the latter had an older age of onset, a milder course of radiographic changes (bamboo spine) and a higher frequency of psoriasis and inflammatory bowel disease (IBD), while AU was much more prevalent in HLA B27+AS patients, and familial aggregation was only observed in these patients.70
The percentage of HLA B27+ patients seems to predict efficacy in clinical studies. In the earliest study in terms of disease duration (<3 years) all patients with IBP were HLA B27+ and had remission rates at week 16 exceeding 60%.71 Such a good response rate may be explained by the very short disease duration and the high degree of HLA-B27 positive patients in the study. In addition, the degree of sacroiliac inflammation as assessed by MRI predicted structural damage in HLA B27+ patients after 8 years of follow-up.72
In a cross-sectional analysis of a German cohort study (GESPIC) with 462 patients structural radiographic damage was influenced by male sex and elevated CRP levels, whereas HLA-B27 positivity determined the age at disease onset.73
In a French cohort study on 708 patients with IBP and possible early axial SpA (DESIR), HLA-B27 was associated with an earlier onset of IBP, less delay in diagnosis, axial inflammation (MRI of spine and SIJ), radiographic damage of the SIJ, decreased disease activity and lower frequency of psoriasis.74
In a Dutch 2-year follow-up study with annual MRI of the SIJ conducted in patients with IBP of less than 2 years’ duration, it was found that a positive HLA-B27 result at baseline predicted a positive MRI during follow-up in HLA-B27+ patients, whereas a negative MRI at baseline in HLA-B27-negative patients strongly predicted a negative MRI also at follow-up.75 This is important because it reminds that not only the presence but also the absence of HLA B27 is meaningful in the process of handling patients with or under suspicion of axSpA.
The relevance of HLA B27 testing in patients with axial involvement in psoriatic arthritis and arthritis associated with chronic IBD has been shown in several studies.76–78
In several studies trying to identify the optimal strategy for referral of patients under suspicion of axSpA from the primary care physician to the rheumatologist HLA B27 came out as an important factor—not necessarily in the first attempt.79 However, HLA B27 alone is never sufficient to establish the diagnosis.49
A German population-based study (SHIP) showed a high frequency of inflammation in the axial skeleton in normal individuals with HLA B27 and, in women, a history of delivery as significant risk factors.80 Of interest, the impact of HLA B27 was only significant in male individuals.81
In conclusion, 50 years after the discovery of the HLA B27 association with AS, HLA B27 testing is now considered an important cornerstone of diagnosis and classification. Recently, polygenic risk scores82 were published which are supposed to have higher discriminatory capacity for a diagnosis of AS than CRP, sacroiliac MRI or HLA-B27 alone. Whether this difference is strong enough to make into clinical practice remains to be seen. As discussed in part I of this review better understanding of the functional role of HLA-B27 in the pathogenesis of SpA is likely to be crucial for the development of curative therapies.
Footnotes
Contributors: All authors have equally contributed.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Not applicable.
References
- 1.Brewerton DA, Hart FD, Nicholls A, et al. Ankylosing spondylitis and HL-A 27. Lancet 1973;1:904–7. 10.1016/s0140-6736(73)91360-3 [DOI] [PubMed] [Google Scholar]
- 2.Schlosstein L, Terasaki PI, Bluestone R, et al. High association of an HL-A antigen, W27, with ankylosing spondylitis. N Engl J Med 1973;288:704–6. 10.1056/NEJM197304052881403 [DOI] [PubMed] [Google Scholar]
- 3.Hanson A, Brown MA. Genetics and the causes of ankylosing spondylitis. Rheum Dis Clin North Am 2017;43:401–14. 10.1016/j.rdc.2017.04.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brewerton DA, Caffrey M, Nicholls A, et al. Reiter’s disease and HL-A 27. Lancet 1973;302:996–8. 10.1016/s0140-6736(73)91091-x [DOI] [PubMed] [Google Scholar]
- 5.Brewerton DA, Caffrey M, Nicholls A, et al. Letter: acute anterior uveitis and HL-A 27. Lancet 1974;1:464. 10.1016/s0140-6736(74)92431-3 [DOI] [PubMed] [Google Scholar]
- 6.Ranganathan V, Gracey E, Brown MA, et al. Pathogenesis of ankylosing spondylitis - recent advances and future directions. Nat Rev Rheumatol 2017;13:359–67. 10.1038/nrrheum.2017.56 [DOI] [PubMed] [Google Scholar]
- 7.Garrido-Mesa J, Brown MA. T cell repertoire profiling and the mechanism by which HLA-B27 causes ankylosing spondylitis. Curr Rheumatol Rep 2022;24:398–410. 10.1007/s11926-022-01090-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Khan MA. Remarkable polymorphism of HLA-B27: an ongoing saga. Curr Rheumatol Rep 2010;12:337–41. 10.1007/s11926-010-0126-x [DOI] [PubMed] [Google Scholar]
- 9.Bowness P. HLA-B27. Annu Rev Immunol 2015;33:29–48. 10.1146/annurev-immunol-032414-112110 [DOI] [PubMed] [Google Scholar]
- 10.Madden DR, Gorga JC, Strominger JL, et al. The structure of HLA-B27 reveals nonamer self-peptides bound in an extended conformation. Nature 1991;353:321–5. 10.1038/353321a0 [DOI] [PubMed] [Google Scholar]
- 11.Jardetzky TS, Lane WS, Robinson RA, et al. Identification of self peptides bound to purified HLA-B27. Nature 1991;353:326–9. 10.1038/353326a0 [DOI] [PubMed] [Google Scholar]
- 12.Buxton SE, Benjamin RJ, Clayberger C, et al. Anchoring pockets in human histocompatibility complex leukocyte antigen (HLA) class I molecules: analysis of the conserved B ("45") pocket of HLA-B27. J Exp Med 1992;175:809–20. 10.1084/jem.175.3.809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Guiliano DB, North H, Panayoitou E, et al. Polymorphisms in the F pocket of HLA-B27 subtypes strongly affect assembly, chaperone interactions, and heavy-chain misfolding. Arthritis Rheumatol 2017;69:610–21. 10.1002/art.39948 [DOI] [PubMed] [Google Scholar]
- 14.Fiorillo MT, Greco G, Maragno M, et al. The naturally occurring polymorphism Asp116-->His116, differentiating the ankylosing spondylitis-associated HLA-B*2705 from the non-associated HLA-B*2709 subtype, influences peptide-specific Cd8 T cell recognition. Eur J Immunol 1998;28:2508–16. [DOI] [PubMed] [Google Scholar]
- 15.Chen L, Shi H, Yuan J, et al. Position 97 of HLA-B, a residue implicated in pathogenesis of ankylosing spondylitis, plays a key role in cell surface free heavy chain expression. Ann Rheum Dis 2017;76:593–601. 10.1136/annrheumdis-2016-209512 [DOI] [PubMed] [Google Scholar]
- 16.Neumann-Haefelin C, McKiernan S, Ward S, et al. Dominant influence of an HLA-B27 restricted Cd8+ T cell response in mediating HCV clearance and evolution. Hepatology 2006;43:563–72. 10.1002/hep.21049 [DOI] [PubMed] [Google Scholar]
- 17.den Uyl D, van der Horst-Bruinsma IE, van Agtmael M. Progression of HIV to AIDS: a protective role for HLA B27 AIDS Rev 2004;6:89–96. [PubMed] [Google Scholar]
- 18.Dazert E, Neumann-Haefelin C, Bressanelli S, et al. Loss of viral fitness and cross-recognition by CD8+ T cells limit HCV escape from a protective HLA-B27-restricted human immune response. J Clin Invest 2009;119:376–86. 10.1172/JCI36587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Neumann-Haefelin C. Protektiver Effekt von HLA-B27 auf den Verlauf der HIV- und hepatitis-C-virus-Infektion [Protective role of HLA-B27 in HIV and hepatitis C virus infection]. Dtsch Med Wochenschr 2011;136:320–4. 10.1055/s-0031-1272531 [DOI] [PubMed] [Google Scholar]
- 20.Khan MA. An update on the genetic polymorphism of HLA-B*27 with 213 alleles encompassing 160 subtypes (and still counting). Curr Rheumatol Rep 2017;19. 10.1007/s11926-017-0640-1 [DOI] [PubMed] [Google Scholar]
- 21.López-Larrea C, Sujirachato K, Mehra NK, et al. HLA-B27 subtypes in Asian patients with ankylosing spondylitis. Tissue Antigens 1995;45:169–76. 10.1111/j.1399-0039.1995.tb02436.x [DOI] [PubMed] [Google Scholar]
- 22.Paladini F, Taccari E, Fiorillo MT, et al. Distribution of HLA-B27 subtypes in sardinia and continental Italy and their association with spondylarthropathies. Arthritis Rheum 2005;52:3319–21. 10.1002/art.21308 [DOI] [PubMed] [Google Scholar]
- 23.Ziade NR. HLA B27 antigen in middle Eastern and Arab countries: systematic review of the strength of association with axial spondyloarthritis and methodological gaps. BMC Musculoskelet Disord 2017;18:280. 10.1186/s12891-017-1639-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sheehan NJ. HLA-B27: what’s new Rheumatology (Oxford) 2010;49:621–31. 10.1093/rheumatology/kep450 [DOI] [PubMed] [Google Scholar]
- 25.Jaakkola E, Herzberg I, Laiho K, et al. Finnish HLA studies confirm the increased risk conferred by HLA-B27 homozygosity in ankylosing spondylitis. Ann Rheum Dis 2006;65:775–80. 10.1136/ard.2005.041103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kim T-J, Sung I-H, Lee S, et al. HLA-B27 homozygosity has no influence on radiographic damage in ankylosing spondylitis: observation study of Korean spondyloArthropathy registry (OSKAR) data. Joint Bone Spine 2013;80:488–91. 10.1016/j.jbspin.2012.12.003 [DOI] [PubMed] [Google Scholar]
- 27.Braun J, Sieper J. Ankylosing spondylitis. Lancet 2007;369:1379–90. 10.1016/S0140-6736(07)60635-7 [DOI] [PubMed] [Google Scholar]
- 28.Burton PR, Clayton DG, Cardon LR, et al. Association scan of 14,500 nonsynonymous SNPs in four diseases identifies autoimmunity variants. Nat Genet 2007;39:1329–37. 10.1038/ng.2007.17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Evans DM, Spencer CCA, Pointon JJ, et al. Interaction between Erap1 and HLA-B27 in ankylosing spondylitis implicates peptide handling in the mechanism for HLA-B27 in disease susceptibility. Nat Genet 2011;43:761–7. 10.1038/ng.873 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Brown MA, Laval SH, Brophy S, et al. Recurrence risk modelling of the genetic susceptibility to ankylosing Spondylitis. Ann Rheum Dis 2000;59:883–6. 10.1136/ard.59.11.883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hamersma J, Cardon LR, Bradbury L, et al. Is disease severity in ankylosing spondylitis genetically determined. Arthritis Rheum 2001;44:1396–400. [DOI] [PubMed] [Google Scholar]
- 32.Calin A, Brophy S, Blake D. Impact of sex on inheritance of ankylosing spondylitis: a cohort study. The Lancet 1999;354:1687–90. 10.1016/S0140-6736(99)03219-5 [DOI] [PubMed] [Google Scholar]
- 33.Jamalyaria F, Ward MM, Assassi S, et al. Ethnicity and disease severity in ankylosing spondylitis a cross-sectional analysis of three ethnic groups. Clin Rheumatol 2017;36:2359–64. 10.1007/s10067-017-3767-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Giza M, Koftori D, Chen L, et al. Is Behçet’s disease a 'class 1-Opathy'? The role of HLA-B*51 in the pathogenesis of Behçet’s disease. Clin Exp Immunol 2018;191:11–8. 10.1111/cei.13049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fiorillo MT, Paladini F, Tedeschi V, et al. HLA class I or class II and disease association: catch the difference if you can. Front Immunol 2017;8:1475. 10.3389/fimmu.2017.01475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.The Australo-Anglo-American Spondyloarthritis Consortium (TASC) . Genome-wide association study of ankylosing spondylitis identifies non-MHC susceptibility Loci. Nat Genet 2010;42:123–7. 10.1038/ng.513 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cortes A, Pulit SL, Leo PJ, et al. Major histocompatibility complex associations of ankylosing spondylitis are complex and involve further epistasis with ERAP1. Nat Commun 2015;6:7146. 10.1038/ncomms8146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hermann E, Yu DT, Meyer zum Büschenfelde KH, et al. HLA-B27-restricted CD8 T cells derived from synovial fluids of patients with reactive arthritis and ankylosing spondylitis. Lancet 1993;342:646–50. 10.1016/0140-6736(93)91760-j [DOI] [PubMed] [Google Scholar]
- 39.Atagunduz P, Appel H, Kuon W, et al. HLA-B27-restricted CD8+ T cell response to cartilage-derived self peptides in ankylosing spondylitis. Arthritis Rheum 2005;52:892–901. 10.1002/art.20948 [DOI] [PubMed] [Google Scholar]
- 40.Yang X, Garner LI, Zvyagin IV, et al. Autoimmunity-associated T cell receptors recognize HLA-B*27-bound peptides. Nature 2022;612:771–7. 10.1038/s41586-022-05501-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hammer RE, Maika SD, Richardson JA, et al. Spontaneous inflammatory disease in transgenic rats expressing HLA-B27 and human beta 2m: an animal model of HLA-B27-associated human disorders. Cell 1990;63:1099–112. 10.1016/0092-8674(90)90512-d [DOI] [PubMed] [Google Scholar]
- 42.Taurog JD, Richardson JA, Croft JT, et al. The germfree state prevents development of gut and joint inflammatory disease in HLA-B27 transgenic rats. J Exp Med 1994;180:2359–64. 10.1084/jem.180.6.2359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Calin A, Fries JF. Striking prevalence of ankylosing spondylitis in "healthy" W27 positive males and females. N Engl J Med 1975;293:835–9. 10.1056/NEJM197510232931701 [DOI] [PubMed] [Google Scholar]
- 44.Braun J, Bollow M, Remlinger G, et al. Prevalence of spondylarthropathies in HLA-B27 positive and negative blood donors. Arthritis Rheum 1998;41:58–67. [DOI] [PubMed] [Google Scholar]
- 45.Calin A, Porta J, Fries JF, et al. Clinical history as a screening test for ankylosing spondylitis. JAMA 1977;237:2613–4. [PubMed] [Google Scholar]
- 46.Levitin PM, Gough WW, Davis JS. HLA-B27 antigen in women with ankylosing spondylitis. JAMA 1976;235:2621–2. 10.1001/jama.1976.03260500037025 [DOI] [PubMed] [Google Scholar]
- 47.Feldtkeller E. Erkrankungsalter und Diagnoseverzögerung BEI Spondylarthropathien [Age at disease onset and delayed diagnosis of Spondyloarthropathies]. Z Rheumatol 1999;58:21–30. 10.1007/s003930050149 [DOI] [PubMed] [Google Scholar]
- 48.Calin A. HLA-B27: to type of not to type? Ann Intern Med 1980;92:208–11. 10.7326/0003-4819-92-2-208 [DOI] [PubMed] [Google Scholar]
- 49.Khan MA, Khan MK. Diagnostic value of HLA-B27 testing ankylosing spondylitis and Reiter’s syndrome. Ann Intern Med 1982;96:70–6. 10.7326/0003-4819-96-1-70 [DOI] [PubMed] [Google Scholar]
- 50.van der Linden SM, Valkenburg HA, de Jongh BM, et al. The risk of developing ankylosing spondylitis in HLA-B27 positive individuals. A comparison of relatives of spondylitis patients with the general population. Arthritis Rheum 1984;27:241–9. 10.1002/art.1780270301 [DOI] [PubMed] [Google Scholar]
- 51.Khan MA, van der Linden SM, Kushner I, et al. Spondylitic disease without radiologic evidence of sacroiliitis in relatives of HLA-B27 positive ankylosing spondylitis patients. Arthritis Rheum 1985;28:40–3. 10.1002/art.1780280107 [DOI] [PubMed] [Google Scholar]
- 52.Ramiro S, Stolwijk C, van Tubergen A, et al. Evolution of radiographic damage in ankylosing spondylitis: a 12 year prospective follow-up of the OASIS study. Ann Rheum Dis 2015;74:52–9. 10.1136/annrheumdis-2013-204055 [DOI] [PubMed] [Google Scholar]
- 53.van der Linden SM, Khan MA, Li Z, et al. Recurrence of axial spondyloarthritis among first-degree relatives in a prospective 35-year-follow-up family study. RMD Open 2022;8:e002208. 10.1136/rmdopen-2022-002208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.van der Linden S, Valkenburg HA, Cats A. Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum 1984;27:361–8. 10.1002/art.1780270401 [DOI] [PubMed] [Google Scholar]
- 55.Dougados M, van der Linden S, Juhlin R, et al. The European Spondylarthropathy study group preliminary criteria for the classification of spondylarthropathy. Arthritis Rheum 1991;34:1218–27. 10.1002/art.1780341003 [DOI] [PubMed] [Google Scholar]
- 56.Moll JM, Haslock I, Macrae IF, et al. Associations between ankylosing spondylitis, psoriatic arthritis, Reiter’s disease, the intestinal arthropathies, and Behcet’s syndrome. Medicine (Baltimore) 1974;53:343–64. 10.1097/00005792-197409000-00002 [DOI] [PubMed] [Google Scholar]
- 57.Bergfeldt L, Edhag O, Vedin L, et al. Ankylosing spondylitis: an important cause of severe disturbances of the cardiac conduction system. Prevalence among 223 pacemaker-treated men. Am J Med 1982;73:187–91. 10.1016/0002-9343(82)90177-2 [DOI] [PubMed] [Google Scholar]
- 58.Bergfeldt L, Möller E. Complete heart block - another HLA B27 associated disease manifestation. Tissue Antigens 1983;21:385–90. 10.1111/j.1399-0039.1983.tb00188.x [DOI] [PubMed] [Google Scholar]
- 59.Joos R, Veys EM, Mielants H, et al. Clinical manifestations in HLA-B27-positive patients. Clin Exp Rheumatol 1987;5 Suppl 1:S41–7. [PubMed] [Google Scholar]
- 60.Prakash S, Mehra NK, Bhargava S, et al. HLA B27 related 'Unclassifiable' seronegative spondyloarthropathies. Ann Rheum Dis 1983;42:640–3. 10.1136/ard.42.6.640 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zeidler H, Werdier D, Klauder A, et al. Undifferentiated arthritis and spondylarthropathy as a challenge for prospective follow-up. Clin Rheumatol 1987;6:112–20. 10.1007/BF02203394 [DOI] [PubMed] [Google Scholar]
- 62.Khan MA, van der Linden SM. A wider spectrum of spondyloarthropathies. Semin Arthritis Rheum 1990;20:107–13. 10.1016/0049-0172(90)90023-9 [DOI] [PubMed] [Google Scholar]
- 63.Zeidler H, Mau R, Mau W, et al. Evaluation of early diagnostic criteria including HLA-B27 for ankylosing spondylitis in a follow-up study. Z Rheumatol 1985;44:249–53. [PubMed] [Google Scholar]
- 64.Mau W, Zeidler H, Mau R, et al. Evaluation of early diagnostic criteria for ankylosing spondylitis in a 10 year follow-up. Z Rheumatol 1990;49:82–7. [PubMed] [Google Scholar]
- 65.Braun J, Bollow M, Eggens U, et al. Use of dynamic magnetic resonance imaging with fast imaging in the detection of early and advanced sacroiliitis in spondylarthropathy patients. Arthritis Rheum 1994;37:1039–45. 10.1002/art.1780370709 [DOI] [PubMed] [Google Scholar]
- 66.Braun J, Brandt J, Listing J, et al. Treatment of active ankylosing spondylitis with infliximab: a randomised controlled multicentre trial. Lancet 2002;359:1187–93. 10.1016/s0140-6736(02)08215-6 [DOI] [PubMed] [Google Scholar]
- 67.Rudwaleit M, van der Heijde D, Khan MA, et al. How to diagnose axial spondyloarthritis early. Ann Rheum Dis 2004;63:535–43. 10.1136/ard.2003.011247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Rudwaleit M, van der Heijde D, Landewé R, et al. The development of assessment of Spondyloarthritis international society classification criteria for axial spondyloarthritis (part II): validation and final selection. Ann Rheum Dis 2009;68:777–83. 10.1136/ard.2009.108233 [DOI] [PubMed] [Google Scholar]
- 69.Rudwaleit M, van der Heijde D, Landewé R, et al. The assessment of spondyloarthritis International society classification criteria for peripheral spondyloarthritis and for spondyloarthritis in general. Ann Rheum Dis 2011;70:25–31. 10.1136/ard.2010.133645 [DOI] [PubMed] [Google Scholar]
- 70.Linssen A, Feltkamp TE. B27 positive diseases versus B27 negative diseases. Ann Rheum Dis 1988;47:431–9. 10.1136/ard.47.5.431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Barkham N, Keen HI, Coates LC, et al. Clinical and imaging efficacy of infliximab in HLA-B27-positive patients with magnetic resonance imaging-determined early sacroiliitis. Arthritis Rheum 2009;60:946–54. 10.1002/art.24408 [DOI] [PubMed] [Google Scholar]
- 72.Bennett AN, McGonagle D, O’Connor P, et al. Severity of baseline magnetic resonance imaging-evident sacroiliitis and HLA-B27 status in early inflammatory back pain predict radiographically evident ankylosing spondylitis at eight years. Arthritis Rheum 2008;58:3413–8. 10.1002/art.24024 [DOI] [PubMed] [Google Scholar]
- 73.Rudwaleit M, Haibel H, Baraliakos X, et al. The early disease stage in axial spondylarthritis: results from the German Spondyloarthritis inception cohort. Arthritis Rheum 2009;60:717–27. 10.1002/art.24483 [DOI] [PubMed] [Google Scholar]
- 74.Chung HY, Machado P, van der Heijde D, et al. HLA-B27 positive patients differ from HLA-B27 negative patients in clinical presentation and imaging: results from the DESIR cohort of patients with recent onset axial spondyloarthritis. Ann Rheum Dis 2011;70:1930–6. 10.1136/ard.2011.152975 [DOI] [PubMed] [Google Scholar]
- 75.van Onna M, Jurik AG, van der Heijde D, et al. HLA-B27 and gender independently determine the likelihood of a positive MRI of the sacroiliac joints in patients with early inflammatory back pain: a 2-year MRI follow-up study. Ann Rheum Dis 2011;70:1981–5. 10.1136/annrheumdis-2011-200025 [DOI] [PubMed] [Google Scholar]
- 76.Castillo-Gallego C, Aydin SZ, Emery P, et al. Magnetic resonance imaging assessment of axial psoriatic arthritis: extent of disease relates to HLA-B27. Arthritis Rheum 2013;65:2274–8. 10.1002/art.38050 [DOI] [PubMed] [Google Scholar]
- 77.Coates LC, Baraliakos X, Blanco FJ, et al. The phenotype of axial Spondyloarthritis: is it dependent on HLA-B27 status? Arthritis Care Res (Hoboken) 2021;73:856–60. 10.1002/acr.24174 [DOI] [PubMed] [Google Scholar]
- 78.Rios Rodriguez V, Sonnenberg E, Proft F, et al. Presence of spondyloarthritis associated to higher disease activity and HLA-B27 positivity in patients with early Crohn's disease: clinical and MRI results from a prospective inception cohort. Joint Bone Spine 2022;89:105367. 10.1016/j.jbspin.2022.105367 [DOI] [PubMed] [Google Scholar]
- 79.Braun A, Gnann H, Saracbasi E, et al. Optimizing the identification of patients with axial spondyloarthritis in primary care - the case for a two-step strategy combining the most relevant clinical items with HLA B27. Rheumatology 2013;52:1418–24. 10.1093/rheumatology/ket115 [DOI] [PubMed] [Google Scholar]
- 80.Baraliakos X, Richter A, Feldmann D, et al. Which factors are associated with bone marrow oedema suspicious of axial Spondyloarthritis as detected by MRI in the Sacroiliac joints and the spine in the general population. Ann Rheum Dis 2021;80:469–74. 10.1136/annrheumdis-2020-218669 [DOI] [PubMed] [Google Scholar]
- 81.Braun J, Baraliakos X, Bülow R, et al. Striking sex differences in magnetic resonance imaging findings in the sacroiliac joints in the population. Arthritis Res Ther 2022;24:29. 10.1186/s13075-021-02712-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Li Z, Wu X, Leo PJ, et al. Polygenic risk scores have high diagnostic capacity in ankylosing spondylitis. Ann Rheum Dis 2021;80:1168–74. 10.1136/annrheumdis-2020-219446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sehnal D, Bittrich S, Deshpande M, et al. Mol* viewer: modern web App for 3d visualization and analysis of large Biomolecular structures. Nucleic Acids Res 2021;49:W431–7. 10.1093/nar/gkab314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Khan MA. Spondyloarthropathies. In: Hunder G, ed. Atlas of rheumatology. Philadelphia: Current Medicine, 2005: 151–80. [Google Scholar]
- 85.Saper MA, Bjorkman PJ, Wiley DC. Refined structure of the human Histocompatibility antigen HLA-A2 at 2.6 A resolution. J Mol Biol 1991;219:277–319. 10.1016/0022-2836(91)90567-p [DOI] [PubMed] [Google Scholar]
- 86.Bjorkman PJ, Saper MA, Samraoui B, et al. Structure of the human class I Histocompatibility antigen, HLA-A2. Nature 1987;329:506–12. 10.1038/329506a0 [DOI] [PubMed] [Google Scholar]