Abstract
Introduction:
Zebrafish larvae are one of the few vertebrates amenable to large-scale drug discovery screens. Larval swimming behavior is often used as an outcome variable and many fields of study have developed assays for evaluating swimming performance. An unintended consequence of this wide interest is that details related to assay methodology and interpretation become scattered across the literature. The aim of this review is to consolidate this information, particularly as it relates to high-throughput approaches.
Areas covered:
The authors describe larval swimming behaviors as this forms the basis for understanding their experimentally evoked swimming or spontaneous activity. Next, they detail how swimming activity can serve as an outcome variable, particularly in the multi-well formats used in large-scale screening studies. They also highlight biological and technical factors that can impact the sensitivity and variability of these measurements.
Expert opinion:
Careful attention to animal husbandry, experimental design, data acquisition, and interpretation of results can improve screen outcomes by maximizing swimming activity while minimizing intra- and inter-larval variability. The development of more sensitive, quantitative methods of assessing swimming performance that can be incorporated into high-throughput workflows will be important in order to take full advantage of the zebrafish model.
Keywords: zebrafish, Danio rerio, swimming, biomechanics, locomotion, drug development
1. Introduction
The widespread use of zebrafish in the biological sciences dates to the early 1980’s when larvae were used to study molecular aspects of vertebrate development [1]. Zebrafish larvae have several features that made them well suited for developmental studies: they have a relatively short generation time, their eggs are fertilized externally, their development is rapid, and the chorion and bodies of embryos and larvae are transparent. Taken together, these features enabled real-time, in vivo observation of vertebrate developmental processes across an easily manageable time frame [2].
In the mid-1990’s, zebrafish larvae were chosen for the first large-scale studies of vertebrate genotype-phenotype relationships [3]. A few years later, techniques for injecting anti-sense morpholinos into embryos allowed investigators to disrupt the expression of their gene of interest, creating zebrafish knock-downs or morphants [4]. That same year, a chemical genetics approach took advantage of the permeability of embryos and larvae to exogenous chemicals in order to evaluate the effects of over 1,000 small molecules on zebrafish embryo development [5].
These studies paved the way for large-scale screens designed to identify small molecules with therapeutic effects in a living vertebrate model [6–8]. This type of phenotype-driven strategy has several advantages over the target-based approach that currently dominates drug discovery [8–10]. These advantages include the early elimination of unacceptable candidates (due to toxicity, off-target effects, or limited bio-availability), a streamlined method of re-purposing existing drugs for new therapeutic uses, and the discovery of novel disease pathways (because there is no a prior bias as to a candidates mechanism of action). Large-scale phenotyping screens are not limited to drug discovery but have benefits in other health-related fields such as toxicology and environmental science [11].
One of the features that make zebrafish attractive for these screening approaches is the early appearance of robust, reproducible phenotypes that can serve as biomarkers or outcome variables. One of the most widely studied of these is swimming activity. Various aspects of swimming performance have been used in studies probing the sensory-neural control of vertebrate motor activity [12–16], as indicators of complex psycho-social states [17,18], as sensitive markers of environmental stress or chemical toxicity [19–21], and as phenotypic readouts for a variety of neuromuscular disease models [22–27].
This multi-disciplinary interest in zebrafish swimming performance has had the unintended effect of dispersing methodological practices across the literature. In this review we have consolidated this information in order to provide investigators with generalized guidelines that can be applied to their particular field of interest. We first review biomechanical aspects of zebrafish larvae swimming, as this forms the basis of interpreting their activity. We then address methods investigators have used to measure swimming activity and performance, particularly in formats conducive to large scale screening studies.
2. Swimming behavior
2.1. Locomotor development
Neuromuscular development occurs rapidly in zebrafish (Figure 1). As early as 17 hours post-fertilization (hpf), larvae will display a spontaneous, slow, alternating left to right coiling of the tail towards the head [28]. By 24 hpf, all of the 32–34 somites that will eventually differentiate into the adult myotomes have been formed and innervated [2]. From 24–48 hpf, embryos will coil in response to touch or bright light [28–31] and by 48 hpf dechorinated embryos respond to gentile tactile stimulation with a burst of crude cyclical swimming behavior [30].
Figure 1: Locomotor milestones attained during zebrafish larvae development.
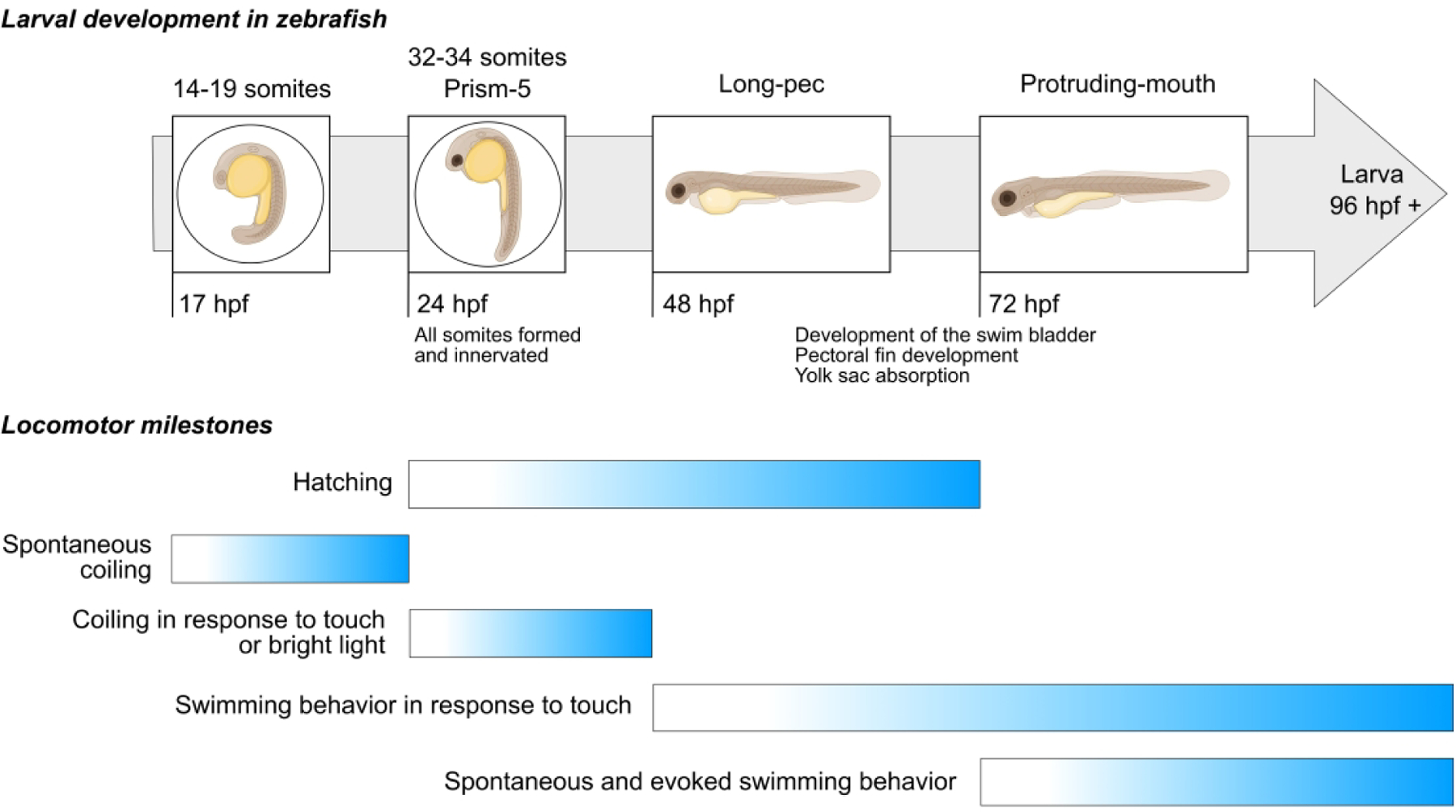
Note that embryos are capable of moving very early in development, well before 24 hpf. By the time they hatch, usually between 48–72 hpf, the neuromuscular system has developed to the point where larvae will swim in response to touch. After another day or two of development, larvae show proficient spontaneous and evoked swimming behaviors. Times are based on an incubation temperature of 28.5°C.
By the time zebrafish hatch they have the ability to swim in circular patterns for periods of up to 1000 ms [32–34]. Over the next 24–36 hours, inflation of the swim bladder, pectoral fin development, and yolk sac absorption contribute to increased locomotor stability, a reduction in drag, and improved swimming efficiency [32,34–36]. By five to seven days post-fertilization (dpf), zebrafish larvae have become adept swimmers and display a variety of spontaneous and evoked locomotor behaviors such as routine exploratory swimming, hunting and prey capture, and escape or startle responses [12,13,16,32,36–39]. These behaviors have been well characterized and form the basis for methods designed to assess their swimming performance.
2.2. High speed data acquisition
Zebrafish larval movement takes place on a millisecond time scale and the mathematical analysis of their swimming behavior, or kinematics, requires rapid rates of data collection. High speed video cameras are therefore essential tools for capturing larval movement for biomechanical analysis. Video is typically collected at rates of 1000 samples/s or faster in arenas of sufficient size so the walls do not restrict movement. The water column is kept shallow so that the movements of interest can be assumed to occur in the two-dimensional plane perpendicular to the camera’s optical axis [40]. Larval movement in three-dimensional space has recently been reported [41–44]. This review focuses on two-dimensional approaches due to its simpler technological and computational demands and wider use (particularly in high-throughput screens).
After movement has been captured on video, the behavior must be quantified. This is accomplished by identifying the x- and y-coordinates of anatomical landmarks, or keypoints, so that the larvae can be modeled biomechanically. Early studies identified keypoints manually [12] but advances in machine vision and tracking algorithms now allow systematic and rapid processing of this step [13,45,46]. Details regarding the digital processing of zebrafish videos are beyond the scope of this review but have been reviewed by others [47].
Once locomotion has been reduced to a time series of x- and y-coordinates, the larvae can be represented as a linked segment model and the movement of each segment as well as the angles between segments tracked across time. This enables quantification of not only changes in larval position, such as distance, displacement, velocity, and acceleration, but also changes in the relationship between body segments, such as head angle, tail angle, body curvature, tail beat frequency, and tail beat amplitude [12,13,32,36,48].
2.3. Slow routine swimming
Zebrafish larvae will slowly explore their environment, presumably searching for prey [49,50]. This spontaneous or routine slow swimming is not continuous but occurs in discrete bouts, sometimes referred to as “scoots”. Each bout is powered by 3–5 low amplitude, 25–40 Hz oscillations of the caudal portion of the tail that propel a larvae 2–3 mm, or ~ 0.50 to 0.75 body lengths, at instantaneous velocities that average about 10 mm/s or ~ 2 body lengths/s [12,13,32,46,51].
At these slow swimming speeds, the hydrodynamic forces opposing movement are mainly viscous [52] and larvae will coast only about 20% of their body length when active propulsion stops [53]. Consequently, routine swimming appears intermittent or sporadic in nature, consisting of short episodes of movement, typically 250 ms or less, that may be repeated every 1–2 s [16,34,46,52,54].
The viscous forces that predominate during slow routine swimming also limit turning [52]. Generally, a single routine turn, or R-turn, changes the direction of travel by < 90° [12]. Routine turns tend to occur in non-random patterns with several turns biased in one direction followed by a sequence biased in the opposite direction [16]. This intermittent zig-zag or slaloming movement pattern may be an effective strategy for exploratory or searching behavior.
The head moves side-to-side in opposition to the tail as the larvae swims forward. This extraneous side-to-side motion can be captured by measuring yaw, defined as the difference between the direction the head is pointing and the direction the body is moving. During slow swimming, yaw is < 3° [12] indicating minimal side-to-side translation as the larvae moves forward.
2.4. Burst swimming
Burst or cyclical swimming is often associated with escape responses (see next section) although it can occur independently of an escape. The speed attained during burst swimming is up to 10-fold faster than routine swimming [12,32]. This greater speed is accomplished by a larger bend of the tail, both in amplitude and it’s more rostral location, and by an elevated tail beat frequency [12,32]. The more powerful tail beat activity results in a greater side-to-side counter movement of the head with yaw exceeding 25° [12].
2.5. Escape responses
Larvae use escape or startle responses to avoid potentially harmful situations and environments. Escape responses are an early behavior, appearing as soon as 2 days post-fertilization [38,55]. While several neurons play a role in the response [56], much attention has focused on two reticulospinal neurons, or Mauthner cells, due to their large size, accessibility, and key role in triggering the escape response. The Mauthner cell bodies synapse with neurons originating in the optic tectum, spinal cord, inner ear, and lateral line and their axons synapse with motor neurons that innervate the trunk and tail muscles [57]. Consequently, tactile, visual, auditory, or vibrational stimuli can depolarize the Mauthner cell and trigger the muscular activity that initiates an all-or-none locomotor response [12,13,15,36,39]. The duration of the latency period between the stimulus and when movement begins is < 10 ms, giving rise to a so-called short-latency escape response [13]. Short-latency escape responses can also be experimentally evoked by the direct depolarization of a Mauthner cell using field stimulation [39,58].
Sometimes a stimulus can be too weak to elicit a short-latency escape response but will still be of sufficient magnitude to startle the larvae. This type of escape response is independent of Mauthner cell activation and is characterized by a long latency period of ~ 30 ms [13]. Long-latency escape responses are a graded response in which the degree of body curvature is dependent on the stimulus intensity, resulting in a response that is more variable in its kinematics [13,36].
An escape response can be broken down into three distinct stages (Figure 2). In the first stage, the larvae bends into a C- or U-shape, usually in a direction that turns the head of the larvae away from the stimuli. This C-start requires ~ 10 ms to complete and serves to re-orientate the direction of the larvae by up to 180° from its initial heading [12,13,36]. The second stage of the escape response is characterized by a rapid (again occurring in roughly 10 ms), powerful counter-bend of the tail. This motion serves to accelerate the larvae away from the perceived threat [12,13,32,36]. The power stroke concludes with a reversal of tail rotation and the start of stage 3. Stage 3 is characterized by repetitive cycles of burst swimming [12]. This stage can last several hundred ms and is where the majority of displacement occurs.
Figure 2: Stages of an escape response.
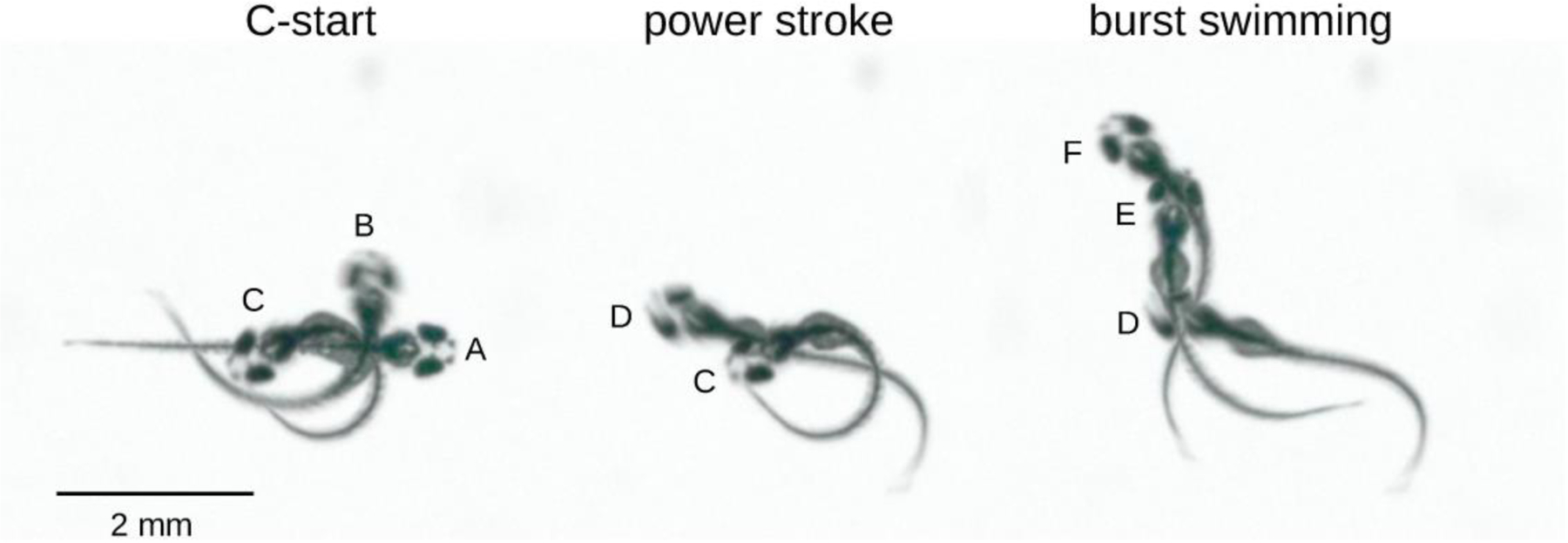
In Stage 1, the C-start, the larvae bends to draw the head and tail together (A to B to C). Stage 2 consists of a rapid counter-bend of the tail (C to D), producing the power stroke. The power stroke is followed by cycles of burst swimming, of which only the initial cycle is illustrated here (D to E to F).
2.6. O-bends
When light adapted larvae are suddenly subjected to darkness, they respond with a unique locomotor behavior termed an O-bend [14]. An O-bend is similar to a C-start in that the larvae curls into a C- or O-shape followed by a powerful counter-bend of the tail. However, in contrast to a C-start, an O-bend has a 25-fold longer latency and consistently re-orientates the larvae by ~ 180°. Following an O-bend, larvae will begin an area-restricted search characterized by a slow, downward helical swimming pattern [44]. After 2–3 minutes, the larvae will return to the water column surface and conduct a wider roaming search strategy [44]. It has been proposed that this behavior represents a light-searching survival response [44], as early zebrafish larvae are dependent on vision for prey detection [59,60].
2.7. Prey capture
Zebrafish larvae must align themselves < 0.5 mm from their prey prior to a predatory strike [50]. This is accomplished using slow swimming coupled with 2–4 unilateral, low angular velocity, J-shaped tail bends which are thought to produce minimal hydrodynamic disturbances that could startle the prey [59]. Pectoral fin activity likely aids in body stabilization in preparation for a strike [36]. After alignment, a more rostral, more severe bend of the tail and a faster tail beat frequency, produces a brief but rapid capture swim towards the prey [37].
3. Experimentally evoked swimming
Prolonged slow swimming and brief high-intensity escape responses represent the two extremes in zebrafish locomotion. In this section we discuss how each of these behaviors can be studied in the laboratory.
3.1. Slow swimming
Continuous swimming has been evaluated in specially-designed swim tunnels [61,62]. A common outcome variable of this approach is the swimming speed at which larvae become exhausted, or the critical swimming speed. The critical swimming speed has been reported to be ~30 mm/s for 5–7 dpf larvae [61]. However, because larvae typically swim in intermittent bursts of activity, very young larvae may attach themselves to the walls of the swim tunnel between bouts of active swimming [62] which can complicate interpretation.
Another approach for studying slow swimming takes advantage of the tendency of 5–7 dpf larvae to response to visual cues. Larvae will attempt to maintain a stable position relative to their visual field. Therefore, if their visual field is moving, for example by projecting moving lines onto the base of the arena, the larvae will move in the same direction as the lines [63]. Zebrafish larvae can attain speeds of up to 30 mm/s for several seconds [64]. Note that this is similar to their critical swimming speed determined in a swim tunnel [61]. While this approach has been used to study the integration of vision and movement, it might serve as a general method of evoking swimming activity provided there are no visual disturbances in the zebrafish strain being studied.
3.2. Escape responses
As detailed in the previous section, escape responses can be triggered by a broad range of stimuli. Gentle tactile stimulation is easily administered and represents a rapid approach for judging larval motility [48,55]. This is well illustrated by Granato et al. [65] who used touch-evoked escape response to identify 166 mobility mutants in the first large-scale vertebrate genotype-phenotype screen. Several of these mobility mutants were subsequently found to be due to mutations in zebrafish genes orthologous to a number of human neuromuscular disorders [66–68].
The touch-evoked escape response is normally a qualitative method that relies upon human vision and discretion to judge whether larval swimming velocity and/or displacement is impaired. Some investigators have modified the touch-evoked escape response in order to make the approach more quantitative in nature. For example, by placing a transparent plastic sheet printed with concentric rings under the dish holding the larvae, initiating the escape response with the larvae centered in the inner most ring, and recording the response on video, the time required by the larvae to swim to the outermost ring could be determined [69]. This approach was used to evaluate a novel drug treatment for muscular dystrophy. Another method for quantifying escape response swimming is to record the movement using high speed videography to then calculate maximum acceleration of larvae after the touch stimulus [48]. This approach was able to differentiate a nemaline myopathy model from wild type larvae.
Commercial activity monitors often have a mechanism for startling larvae. However, the low sampling rates and spatial resolution typically used in these monitors limits analysis to overall movement after the escape has been completed. Using faster sampling rates would enable investigators not only to assess the distance larvae move but also kinematic properties underlying movement, as has been done in many of the behavioral studies cited above [12,13,15,36]. Capturing escape responses with fast sampling rates so that swimming kinematics could be resolved may hold promise for the evaluation of new therapeutic treatments in future studies.
4. Spontaneous swimming activity
Slow, spontaneous swimming forms the basis for the majority of high throughput approaches to measuring larval swimming performance. Unlike behavioral studies, which tend to focus on motor control and swimming biomechanics, zebrafish activity assays are primarily concerned with global descriptors of locomotion, such as the distance covered, the time engaged in swimming, and the specific location within an arena where the activity took place. These data are often collected using commercial or laboratory constructed activity monitors which consist of a light-proof enclosure with a holder for a multiwell plate at the bottom and a video camera at the top. Various aspects of the environment within the enclosure can be controlled by the investigator, including the temperature of the multiwell plate, the intensity of lighting, and the type of lighting, i.e. either visible or infrared.
With this type of setup activity assays can be conducted relatively rapidly on large numbers of larvae with minimal investigator oversight. This makes them compatible with the high throughput workflows of large-scale screening studies. Consequently, activity assays have taken on increased importance in fields such as drug discovery where swimming behavior is an important biomarker or outcome variable.
Software originally developed for tracking flies [70] has been used to track multiple larvae in a single arena by resolving crossings and collisions [34,71]. However, this approach may not be applicable for screening studies where the identity of each larvae must often be maintained before, during, and after tracking has been completed in order to allow for subsequent genotyping or other phenotyping assays. To preserve larval identity, most high-throughput behavioral studies are conducted with larvae arrayed in multi-well plates, one larva per well. These studies differ in several ways from the behavioral approach detailed previously.
Activity assays typically utilize video that is collected at much slower frame rates than required for biomechanical analysis. Typical rates fall in the 25–30 Hz range. It is then a common practice to bin or average the activity data over time periods ranging from 0.5 second up to 60 seconds (or longer) depending on the overall duration of recording and the research aims. This helps to smooth the swimming data across time, speed the analysis of the data, and facilitate long duration data acquisition sessions.
Note that the temporal resolution of an assay is not necessarily the video frame rate but the rate that data were subsequently analyzed after taking any binning or averaging into account. The higher the rate of analysis, the more detail this type of assay can resolve. For instance, an analysis conducted with 0.5 s bins was found to capture ~ 90% of the displacement measured from video collected at 30 Hz [25]. Thus, binning rates of 1–2 Hz may represent an acceptable compromise between resolution and the practical limitations of collecting longer duration videos.
After video has been collected, there are two general methods available for quantifying movement. The first method is based on identifying changes in pixel intensity from one frame to the next. Summing these frame-to-frame differences produces an estimate of overall movement. This method is most useful when movements of interest are independent of changes in the position of the larval center of mass. For example, when a zebrafish embryo is still in its chorion there may be substantial coiling activity of the tail but minimal displacement of the embryo. Under these conditions, changes in pixel intensity can be used to quantify the extent of tail coiling activity [29,31].
The second method of evaluating movement is conceptually similar to the approach used in many of the behavioral studies described in the previous section. However, instead of modeling the larvae as a series of interconnected body segments, the larvae is usually denoted as a single point that approximates the larval center of mass. The two-dimensional coordinates of this point can then be tracked frame-to-frame to yield measures of time engaged in movement, distance moved, and position within the well. Thus, the emphasis is on the overall movement of the larvae rather than on how various body segments move relative to each other. Both open source and proprietary software packages are available for performing the data processing steps necessary for quantifying center of mass movement in multi-well plate formats. Several of the open-source options have recently been reviewed [72].
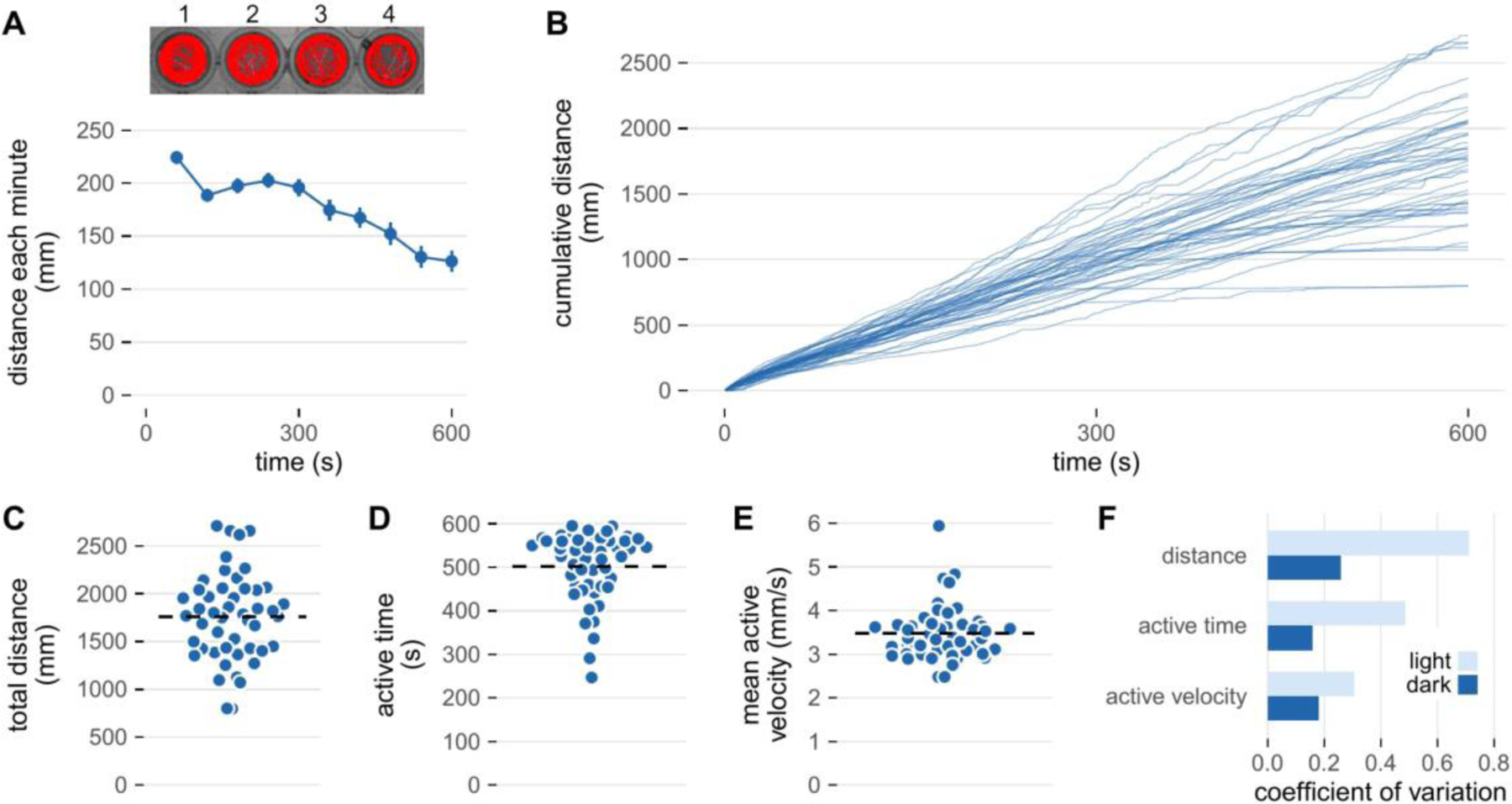
Figure 3 illustrates representative activity data for wild-type larvae collected using a commercial infra-red activity monitor. Larvae were equilibrated 20 minutes in darkness prior to data collection. There is an immediate increase in locomotion during the first minute of data collection due to a spike in activity immediately after lights on. After this initial spike, movement is relatively constant for several minutes but then gradually begins to increase around minute 4–5. Note that there is variation in the distance covered by individual larvae. In this section, we discuss how to interpret this type of data and in the following section ways to minimize intra-larval variability.
Figure 3: Activity of zebrafish larvae in a lighted environment.
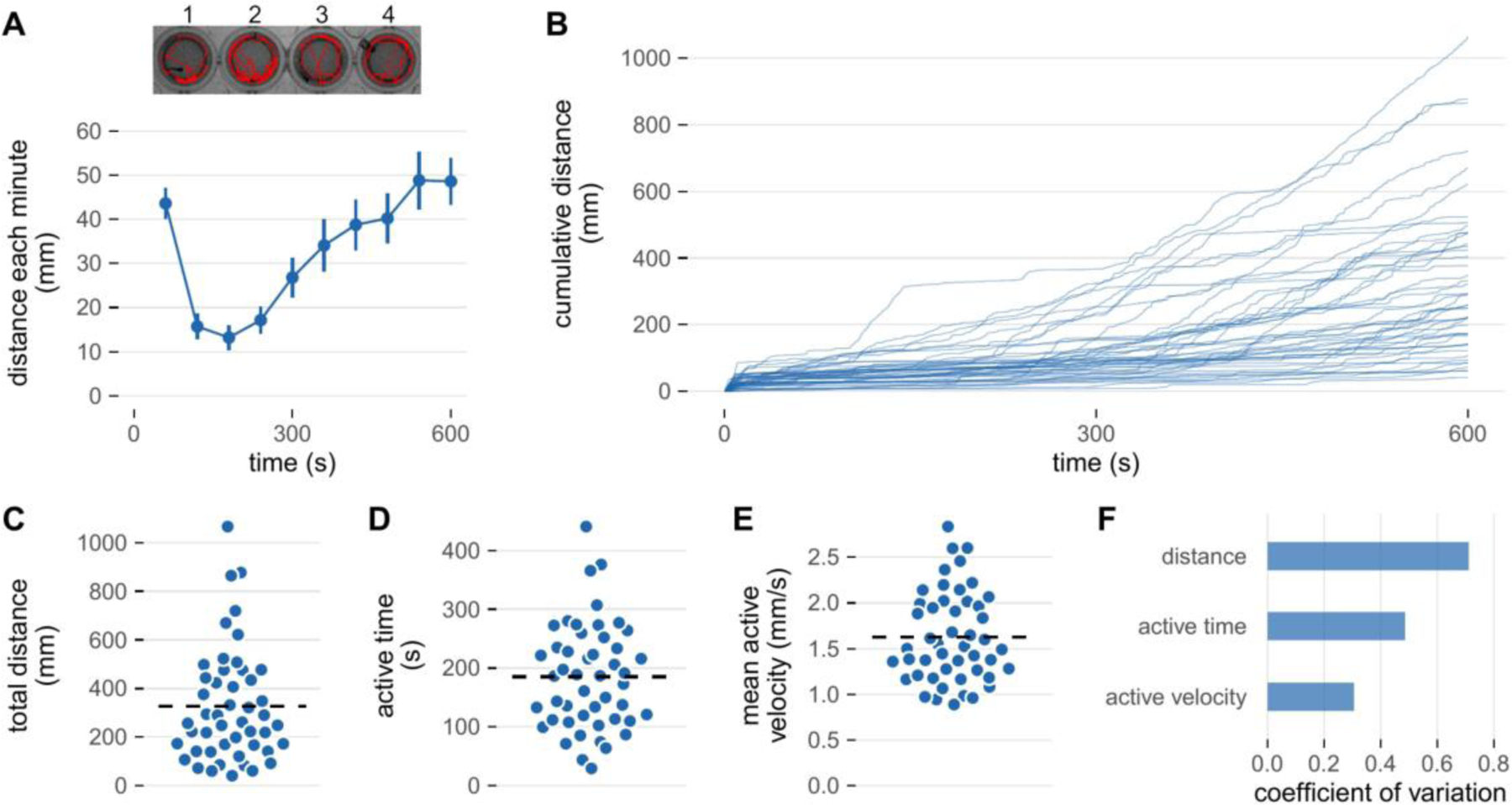
Spontaneous swimming activity was measured in an infra-red activity monitor (DanioVision, Noldus Information Technology, Wageningen, the Netherlands). Larvae (6 days post-fertilization) were studied in 48-well plates at 28°C that were equilibrated in the monitor for 20 minutes in the dark prior to data collection. Following equilibration, the illumination was switched on and the larvae filmed at 25 frames/s for 10 minutes. Two-dimensional coordinates of the larval center of mass were determined using EthoVision software. Dependent variables were derived from this data set using custom scripts written in R. A) Top: Activity tracks of 4 representative larvae during the 10 minute lights on protocol. Bottom: The average distance covered by all 48 larvae for each minute of the protocol. Values are mean ± SE. B) Cumulative distance (calculated every 500 ms) across the protocol. Each line represents an individual larvae. C) The total distance covered by each larva derived from the data in Panel B. The total distance is a function of, D) the time that larvae were engaged in activity (active time), and E) their average velocity while active (mean active velocity). F) The coefficient of variation for cumulative distance, active time, and mean active velocity. In panels C, D, and E the horizontal dashed line indicates the mean.
4.1. Temporal aspects
A key to interpreting swimming activity is to remember that zebrafish larvae do not swim continuously. This intermittent swimming behavior is evident in Figure 3-B where most individuals can be observed to move in stops and starts. This spontaneous swimming activity appears similar to the slow swimming ‘scoots’ discussed previously although it is unclear whether the physical constraints of a small well alter normal swimming behavior or biomechanics.
Because of the intermittent nature of swimming activity, the observation time of an experiment is not equivalent to the time larvae were actually in motion. Swimming time can be calculated by quantifying the time that movement exceeds a baseline threshold. Relatively fast rates of data acquisition and analysis reveal that 7 dpf zebrafish larvae spent only about one-third of the observation time in motion [25]. Analyzing data in longer time bins will likely overestimate the fraction of time larvae spend in motion. This would not affect the quantification of the distance moved but may confound time dependent variables, such as movement velocity. Reliable temporal measures therefore require appropriate sampling and analysis rates.
4.2. Kinematic aspects
The total distance larvae move during a period of observation is perhaps the most widely reported variable describing their activity. Dividing total distance by the observation period yields a mean swimming velocity. However, when comparing two experimental groups of larvae, mean velocity provides no additional information that is not provided by total distance because the former is simply the later divided by a constant (provided observation time is constant across groups). In the remainder of this review, we will refer to this variable as “total distance or mean velocity”.
Because larvae are likely to spend a substantial proportion of the observation time stationary, their mean velocity will greatly underestimate the actual velocity they moved about their arena. This confounding can be reduced by assessing active velocity, defined as the distance moved in a sampling period divided by the duration of the sampling period (Figure 3-E). The active velocity of spontaneously swimming larvae has been reported to be 2–3 times faster than their corresponding mean velocity [25].
The discontinuous nature of larval movement means that the use of total distance or mean velocity as the sole outcome variable of a study may obscure important aspects of movement. For instance, total distance or mean velocity was reported to increase 10-fold as larvae matured from 3 to 9 dpf [25]. If one ignores the discontinuous nature of larval locomotion, then this result would seem to indicate that larvae swam faster as they matured. However, active velocity actually decreased 33% between 3 to 9 dpf as the observed increase in total distance or mean velocity was due to a 15-fold increase in the proportion of time engaged in movement [25].
Conversely, observing that total distance or mean velocity was not affected by an intervention does not necessarily mean that the underlying swimming kinematics were unchanged. As an example, exposing larvae to ethanol was reported to have no effect on total swimming distance [73]. However, larvae exposed to ethanol swam less frequently, from one episode every 1.25 s to one episode every 2 seconds. This was offset by a 50% increase in the duration of each swimming episode resulting in no change in total distance or mean velocity. Thus, larvae swam less frequently but for longer periods of time, a finding obscured by focusing solely on total distance or mean velocity.
Supplementing total distance or mean velocity with additional kinematic variables can also reduce the variability in swimming performance. Total swim distance is the product of the time spent in motion and the active velocity. Time in motion and active velocity both have inherent variability which is then compounded by taking their product. Consequently, total swimming distance can be up to 3-fold more variable than separate measures of active velocity or episode duration [25,73]. This is illustrated by the range of coefficients of variation for distance, active time, and active velocity shown in Figure 3-F. Thus, a straight-forward way to increase the statistical power of an activity assay is to report active velocity or active time in addition to total distance or mean velocity [25].
4.3. Spatial aspects
The spatial distribution of swimming activity within an arena may be an important outcome measurement. There is consensus that larvae spend a disproportionate amount of time moving around the circumference of a small arena rather than moving back and forth across its center [19,74–76]. The tendency of an organism to remain in close physical contact with an object or wall is termed thigmotaxis and is often interpreted as an index of anxiety [17,77]. An additional advantage of collecting positional data is that the intra-individual coefficient of variation across days has been reported to be less than measures of larval movement [76].
In a circular well, place or edge preference has been quantified by defining a virtual smaller diameter inner arena centered within the physical well. Some investigators have made the center area half the diameter of the entire well [19] while others have made the areas enclosed by the inner and outer arenas equal [75]. During data processing, the time the larvae spends within the virtual central arena is compared to the time spent elsewhere in order to create a place preference variable [19,74,75]. An alternative approach is to calculate the radial index, defined as the distance of the larvae from the center of the well divided by the well radius [76]. The radial index for zebrafish larvae has been reported to average about 0.75 indicating a bias towards the edge of the arena [76].
5. Factors influencing swimming behaviors
The distance zebrafish larvae move per unit time can vary considerably from study to study [25,73,75,78–83]. The majority of this variability has been attributed to variability within and between larvae [25]. Accounting for extraneous variables that contribute to this variability may increase the power of a study and facilitate comparisons of results obtained in different laboratories.
Biological sources of variation include the strain studied, the developmental stage of the larvae, and the time of day data were collected. Technical factors, such as fish husbandry and the physical properties of the arena can impact larval biology and consequently swimming performance. One specific technical factor, the lighting condition, has such a strong impact on swimming behavior that it provides a way to experimentally manipulate swimming activity.
5.1. Strain and genetic background
Different zebrafish strains can exhibit substantial differences in swimming performance even when studied under identical experimental conditions. The distance larvae spontaneously swim can vary by more than 2-fold across different strains of fish [25,79,82]. These differences may be an independent strain effect or they may be related to strain specific variation in development as inter-strain differences in mean velocity and time moving were evident at 5 dpf but reduced or absent at later time points [25,82]. Nevertheless, there remains a genetic contribution independent of development as 3 month old adults show considerable inter-strain differences in the distance covered after 1 hour of observation [79].
There are known physiological and morphological differences between zebrafish stains that could impact swimming performance. For instance, AB fish show increased stress-axis activity and related gene expression compared with Tupfel long fin (TL) fish [84]. Steady-state whole blood glucose levels has been shown to be lower in Wild India Kolkata (WIK) strain and higher in Tübingen (TU) strain, which affects myostatin expression and body growth [85]. It has also been reported that TL adult zebrafish have longer caudal fin length, which can govern swimming performance [86]. The AB strain is considerably more likely to respond to an acoustic stimuli with a short-latency escape response than the Tubingen strain, which in turn is more likely to respond with a long-latency response [87]. This indicates that different strains have different genetic traits which may lead to a wide range of swimming performance. This is particularly true for zebrafish models of human diseases in which swimming performance and behavior can be impaired.
Inter-clutches differences are another source of variability for the zebrafish behavior and swimming performance [88]. This source of variability is governed by several factors such as temperature, illumination, and time of day (see below), as well as maternal and/or genetic influences including epigenetic variations [89]. Intra-clutches differences can also be observed and lead to a wide range of swimming behaviors. This can be addressed by considering the genetics of a particular experiment. Setting up heterozygote x heterozygote breeders for recessive traits, and genotyping the fish after measurements have been collected help control for many of these parameters, and provides data for heterozygotes, which may have an intermediate phenotype.
5.2. Developmental stage
Older larvae move greater distances per unit time in comparison to younger larvae [25,81]. The largest increase in distance covered per unit time appears to occur sometime between 4–6 dpf, with lessor increases from 6 out to 9 dpf [25,81]. Intra-larval variability is also reduced between 6–7 dpf in comparison to 5–6 dpf [76] and inter-larval variability of several swimming behaviors (episode frequency and duration, swim time and velocity, and total distance) were all less for 7 dpf vs 4 dpf larvae [73]. Thus, the spontaneous swimming activity of 6–7 dpf larvae appears to be relatively stable compared to earlier time points, with less within and between larvae variability.
5.3. Morphology
Closely related to the developmental stage is larval morphology. Biomechanists typically normalize kinematic properties to fish length. This is routinely done in studies focusing on larval swimming kinematics but rarely in the high-throughput assays of swimming activity. Thus, the impact that differences in larval length may have on intra-larval variability is unclear. Another important morphological characteristic influencing swimming performance is swim bladder inflation. Larvae with under-inflated bladders have a reduced sensitivity to escape response stimuli [87].
5.4. Assay temperature
Zebrafish larvae are typically raised at a temperature of 28.5°C as this was found to be optimal for development [2]. High-throughput activity trials are usually conducted between 25–28.5°C but little is know about whether this temperature range is optimal for assessing swimming activity or for minimizing intra-larval variability. In contrast to activity assays, temperature is known to have a substantial effect on the kinematics of the escape response. For example, a change of only 3°C was found to alter C-start peak amplitude by 25% [87].
5.5. Housing density
Social interactions occur very early in zebrafish development and are important for establishing subsequent adult behavior. Zebrafish larvae (6 dpf) that were raised in isolation covered about 25% less distance per unit time when exposed to acute darkness in comparison to group-raised siblings [83]. In contrast, escape response sensitivity to vibration is enhanced in larvae raised at lower densities [87].
5.6. Nutrition
Even though larvae have the ability to hunt prey by 5 dpf [49,50,90], external feeding can be delayed until larvae are 8 dpf without effecting their survival [91]. However, swimming performance may not be optimal in unfed larvae. Larvae in which feeding was initiated at 5 dpf showed enhanced avoidance of visual stimuli, faster swim velocities, reduced thigmotaxis, and spent more time in motion over the next 1–2 days in comparison to unfed larvae of similar age [92].
Despite the effect of feeding on swimming activity, investigators may chose to study unfed larvae 7 dpf or younger in order to eliminate confounding factors that feeding may introduce into their experimental design [76]. For instance, larvae are most often fed zooplankton, such as Artemia and rotifers, which can vary in terms of their nutritional content and can be a source of inadvertent exposure to bio-contaminants or pathogens [93]. In addition, neuromuscular mutants or larvae exposed to chemicals that impair locomotion may not be as adept at hunting as wild-type or untreated control larvae, adding a confounding variable to the study. Therefore, investigators should carefully consider the pros and cons of using fed larvae for studies of larvae ≤ 7–8 dpf.
5.7. Arena dimensions
Swimming behavior is typically evaluated in large arenas so that movement is unrestricted. However, the wells of a multi-well plate present a much smaller arena than is used in the typical behavioral study. The diameter of wells in a 96 well plate approaches the body length of a 4–7 dpf larvae and there has been concern that the physical dimensions of these small arenas could restrict movement and impact locomotor kinematics. In fact, it has been reported that swim distance (or its equivalent mean velocity) is less in 96 well plates in comparison to 24 well plates [25,81]. We see the same response in our laboratory (Figure 4). This effect has been attributed to a reduction in the time spent in motion rather than any change in active velocity [25].
Figure 4: Activity of zebrafish larvae during a 15:9 hr light-dark cycle in different diameter wells.
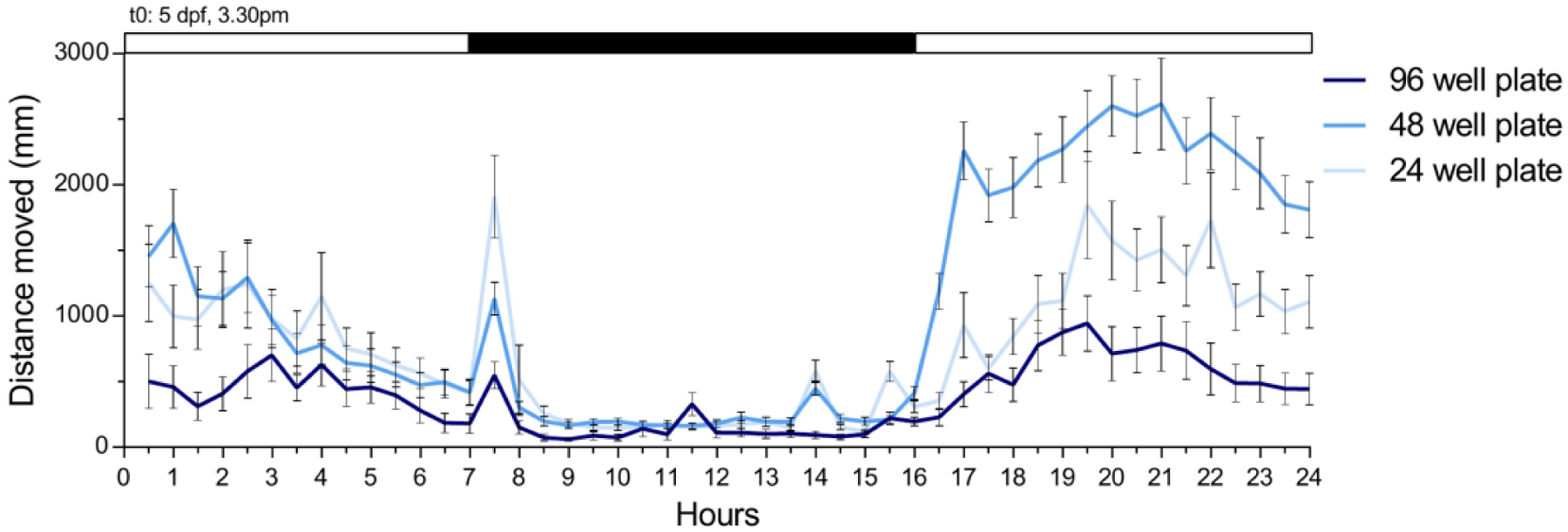
Spontaneous swimming activity was measured in a DanioVision infra-red activity monitor. Wild-type larvae (5 days post-fertilization, dpf) were studied in 24-, 48-, and 96-well plates. At 5 dpf, 3.30 pm (EST) the illumination was switched on for 7 hours, followed by a 9-hour period of darkness, and a 8-hour period of light. Larvae were filmed at 30 frames/s, and the two-dimensional coordinates of the larval center of mass were determined using EthoVision software. The graph represents the average distance covered by all 24 larvae per plate for each hour of the protocol. Values are mean ± SE.
There is less consensus concerning the effect of intermediate well diameters. One group has reported that swimming distance in 48 well plates was similar to that in 96 well plates [81]. In our hands, we find that larvae often swim more in 48 well plates that in 24 well plates (Figure 4).
While this discussion has focused on swimming distance, there may be other considerations when selecting a well size. Small diameter wells, equivalent to the diameter of wells on a 48 well plate, were associated with the least inter-larval variability for a number of swimming behaviors [73].
5.8. Water column depth
The space available for swimming is dependent not only on the diameter of the wells but also on the depth of the water column. Larvae swam more often and covered more total distance in a 5.5 mm water column than in a shallower 1.5 mm column [73]. The deeper column also resulted in less variability for most variables, although not for the total distance covered [73].
5.9. Well-to-well variation
Although all the wells of a multi-well plate are of similar size, they may not necessarily be spatially equivalent. In a clear multi-well plate, larvae placed in wells along the border of the plate may experience different visual stimuli compared to larvae placed in interior wells [94]. This can be avoided by using opaque plates [29] or controlled by plating out larvae so that control and experimental groups are systematically distributed across the plate. There can also be a gradient of illumination from the central wells to the border wells [94], another reason for systematically arranging experimental groups across a plate. Data processing techniques have been developed to normalize movement based on differences in well-to-well illumination [94].
5.10. Time of day
Unlike rodent models, zebrafish larvae are more active during their light period and less active during their dark period, a pattern evident as early as 2 dpf [82,95]. Figure 4 illustrates this effect, with 5 dpf larvae showing little activity during their dark period and a large increase in activity in the light. The figure also shows a gradual increase in activity during the initial hours of the light cycle, a stabilization of activity for several hours, and then a gradual reduction in activity over the last half of the light cycle. This is consistent with findings that for larvae younger than 7 dpf, spontaneous swimming distance did not stabilize until 2–3 hours into the light cycle [96]. In contrast, larvae 7 dpf and older showed stable spontaneous activity regardless of when they were studied during the light cycle [96].
5.11. Illumination
Overlaid on the normal circadian pattern of activity is a masking response in which abrupt transitions from dark to light, and especially light to dark, modulate swimming behavior [14]. Search behaviors that occur when a larvae transitions from a light to dark environment (discussed in ‘O-bends’ section) have been observed in a relatively large three-dimensional arena that did not restrict movement [44]. However, the same research group found that if larvae were restricted to small arenas, a darkness induced O-bend was followed by a period of hyperactivity [14] leading to the conclusion that in small restrictive arenas, like those in a multi-well plate, the hyperactivity that follows an O-bend is due to wall avoidance.
As illustrated in Figure 5, abruptly altering illumination can be used as a method of acutely manipulating zebrafish larvae swimming activity. Light adapted larvae arrayed in multi-well plates and subjected to an abrupt transition to darkness travel 3 to 10-fold greater distances [14,19,20,76,78,80,81]. This increase in activity can be attributed to a substantial increase in the time spent in motion (some larvae are in motion almost continually, (Figure 5-D) and a somewhat smaller increase in active swimming velocity (Figure 5-E).
Figure 5: Activity of zebrafish when exposed to a dark environment.
Immediately upon the conclusion of the protocol outlined in Figure 3, the visible illumination in the activity tracker was switched off and the activity of the larvae recorded for another 10 minutes. Data were processed as described in the legend to Figure 3. A) Top: Activity tracks of 4 larvae during the dark protocol. The larvae shown are the same larvae illustrated at the top of Figure 3-A. Bottom: Mean (±SE) distance covered each minute during the dark protocol. B) Cumulative distance across the 10 minute dark protocol. Each line represents an individual larva. C) Total distance covered by each larva. D) The total time that each larva was active. E) The mean active velocity of each larva. F) The coefficient of variation for total distance, active time, and mean active velocity for the protocol conducted in the light and in the dark. In panels C, D, and E the horizontal dashed line indicates the mean.
At least 20 minutes of prior light adaptation is required to maximize this hyperactivity response to acute darkness [14,80]. This increased activity in darkness peaks around 5 minutes and then gradually decays over the next 20–30 minutes until it reaches a level consistent with the dark-adapted baseline [78,80,81].
Intra-individual variation in activity is lower when larvae are studied in dark vs. lighted environments [76]. Figure 5-F shows how the coefficient of variation for distance, active time, and active velocity are all reduced when larvae are studied in the dark. This response has been interpreted to indicate that the more a larva moves, the less variable its activity becomes [76].
Finally, the light to dark transition is modulated by the level of darkness. A light to complete darkness transition will produce the greatest locomotor response while transitioning to dimly lit conditions will elicit intermediate responses [14,74,81]. This relationship raises the possibility that investigators may be able to exert some control over the degree of hyperactivity by varying light intensity.
6. Expert opinion
Swimming activity can be used as an outcome variable in phenotype-based drug discovery screens. The probability that a screen will identify novel candidate compounds is a function of the dynamic range and the variability of the outcome variable [97]. It is therefore important to consider how dynamic range be maximized and variability minimized in order to facilitate novel drug discovery.
In the near-term, investigators can focus on optimizing their experimental design as well as giving thought to the interpretation of their results. In Table 1 we have compiled a summary of ways in which this can be accomplished when studying larvae in multi-well plate formats. Note that it may not always be possible to maximize dynamic range and minimize variability simultaneously. Investigators therefore need to be ready to make trade-offs, focusing on whichever aspects they judge to be more important. The Table also includes suggestions for interpreting results. A key point when interpreting spontaneous swimming is that it is not continuous and that this feature should be taken into consideration when evaluating swimming performance measurements. Our intent in compiling these recommendations is to stimulate investigators to think about more robust ways of quantifying and interpreting the swimming behavior of their zebrafish.
Table 1:
Recommendations for studying activity of zebrafish larvae in multi-well plates.
| recommendation | maximize activity | minimize variability | facilitate interpretation |
|---|---|---|---|
| 1. study 6–7 dpf larvae | x | x | |
| 2. raise larvae in groups | x | ||
| 3. study fed larvae OR study | x | ||
| non-feed larvae (≤ 7 dpf) | x | ||
| 4. use 24–48 well plates | x | x | |
| 5. fill wells with ~ 5 mm fish water | x | ||
| 6. systematically array larvae in wells | x | ||
| 7. study several hours after lights on | x | ||
| 8. study at a consistent time of day | x | ||
| 9. study at a consistent temperature | x | ||
| 10. record some activity in the dark | x | x | |
| 11. analyze data at fast Hz | x | ||
| 12. report distance | x | ||
| 13. report active velocity | x | x | |
| 14. report active time | x | x | |
| 15. report position in well (radial index) | x | ||
| 16. assess any clutch affects | x |
Long-term advances in measuring zebrafish larvae locomotor performance could focus on how to combine the sensitivity and high temporal resolution currently associated with biomechanical and behavioral studies of locomotion with the rapid and high throughput nature of multi-well assays. This could result in the identification of novel outcome measures that have increased sensitivity and reduced intra- and inter-larval variability while at the same time providing a richer quantitative analysis of movement. The later would be likely to led to a more mechanistic interpretation of the disease under investigation as well as an increased understanding of the role that specific genes have on vertebrate locomotion. However, technical issues related to the small size and rapid movement of zebrafish larvae will need to be addressed for this to become a reality. These issues include, but are not necessarily limited to, 1) minimal contact methods of positioning larvae for video recording without interfering with their swimming behavior, and 2) how to rapidly quantify and model the biomechanics of a 4 mm long organism that entail minimal investigator oversight.
Microfludic approaches have been developed for the handling and manipulation of embryos and larvae [98]. It is possible that these methods could be adapted to facilitate large-scale experiments examining swimming behavior. Advances in machine vision and learning could greatly facilitate quantifying zebrafish swimming behavior and has the potential to have a large impact on the field. Already, automated analysis of zebrafish swimming has been accomplished by tracking key anatomical locations or by geometrical modeling, sometimes in three dimensions, and the resulting kinematic data may be used to automatically identify and classified the type of swimming behavior the fish was engaged in [101]. Taken together, these technical developments indicate that more rapid, higher resolution evaluation of zebrafish larvae swimming behavior may eventually be suitable for use in high-throughput screening. Advances such as this will benefit many investigators who use swimming performance as an outcome variable in their work.
Article highlights.
Zebrafish larvae are precocious and robust swimmers and swimming activity is one of their most widely studied phenotypes.
Larval swimming behavior has been characterized using neural-behavioral and biomechanical-based approaches but the methods employed may not always be applicable to high-throughput experiments.
Activity assays, conducted most often in multi-well plates, enable many larvae to be studied simultaneously but often show considerable intra- and inter-animal variability.
Incorporation of insights about larval swimming behavior may facilitate interpretation of activity measurements, revealing previously unappreciated aspects of swimming performance.
Funding:
This work was supported by the Lee and Penny Anderson Family Foundation (to AH Beggs), by the Muscular Dystrophy Association (grant 962927 to MR Lambert), and by the National Institutes of Health (R01AR064300 to LM Kunkel and 1K99AR080197 to MR Lambert)
Footnotes
Declaration of Interest: The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Reviewer Disclosures: Peer reviewers on this manuscript have no relevant financial or other relationships to disclose.
Papers of special note have been highlighted as either of interest (•) or of considerable interest (••) to readers.
References
- [1].Streisinger G, Walker C, Dower N, et al. Production of clones of homozygous diploid zebra fish (Brachydanio Rerio). Nature 1981;291:293–296. [DOI] [PubMed] [Google Scholar]
- [2].Kimmel CB, Ballard WW, Kimmel SR, et al. Stages of embryonic development of the zebrafish. Dev Dyn 1995;203:253–310. [DOI] [PubMed] [Google Scholar]
- [3].Nüsslein-Volhard C The zebrafish issue of Development. Development. 2012;139:4099–4103. [DOI] [PubMed] [Google Scholar]
- [4].Nasevicius A, Ekker SC. Effective targeted gene ‘knockdown’ in zebrafish. Nat Genet 2000;26:216–220. [DOI] [PubMed] [Google Scholar]
- [5].Peterson RT, Link BA, Dowling JE, et al. Small molecule developmental screens reveal the logic and timing of vertebrate development. Proc Natl Acad Sci USA 2000;97:12965–12969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Rennekamp AJ, Peterson RT. 15 years of zebrafish chemical screening. Curr Opin Chem Biol 2015;24:58–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Tamplin OJ, White RM, Jing L, et al. Small molecule screening in zebrafish: Swimming in potential drug therapies. Wiley Interdiscip Rev Dev Biol 2012;1:459–468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Widrick JJ, Kawahara G, Alexander MS, et al. Discovery of novel therapeutics for muscular dystrophies using zebrafish phenotypic screens. J Neuromuscul Dis 2019;6:271–287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Kell DB. Finding novel pharmaceuticals in the systems biology era using multiple effective drug targets, phenotypic screening and knowledge of transporters: Where drug discovery went wrong and how to fix it. FEBS J 2013;280:5957–5980. [DOI] [PubMed] [Google Scholar]
- [10].Zheng W, Thorne N, McKew JC. Phenotypic screens as a renewed approach for drug discovery. Drug Discov Today 2013;18:1067–1073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Bambino K, Chu J. Zebrafish in toxicology and environmental health. Curr Top Dev Biol 2017;124:331–367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12]. Budick SA, O’Malley DM. Locomotor repertoire of the larval zebrafish: Swimming, turning and prey capture. J Exp Biol 2000;203:2565–2579. •• This paper quantified swimming performance of freely moving larvae and used this information to identify several distinctive locomotor behaviors.
- [13]. Burgess HA, Granato M. Sensorimotor gating in larval zebrafish. J Neurosci 2007;27:4984–4994. •• This work developed a machine vision algorithm to classify swimming behaviors of thousands of zebrafish larvae.
- [14]. Burgess HA, Granato M. Modulation of locomotor activity in larval zebrafish during light adaptation. J Exp Biol 2007;210:2526–2539. • This paper showed that different lighting conditions can greatly influence swimming activity of zebrafish larvae.
- [15].Dunn TW, Gebhardt C, Naumann EA, et al. Neural circuits underlying visually evoked escapes in larval zebrafish. Neuron 2016;89:613–628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16]. Dunn TW, Mu Y, Narayan S, et al. Brain-wide mapping of neural activity controlling zebrafish exploratory locomotion. Elife 2016;5:e12741. • This work described the kinematic properties of slow spontaneous swimming behavior, information that is valuable when interpreting studies conducting using fish arrayed in multi-well plates.
- [17].Kalueff AV, Gebhardt M, Stewart AM, et al. Towards a comprehensive catalog of zebrafish behavior 1.0 and beyond. Zebrafish 2013;10:70–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Tierney KB. Behavioural assessments of neurotoxic effects and neurodegeneration in zebrafish. Biochim Biophys Acta 2011;1812:381–389. [DOI] [PubMed] [Google Scholar]
- [19].Colón-Cruz L, Kristofco L, Crooke-Rosado J, et al. Alterations of larval photo-dependent swimming responses (PDR): New endpoints for rapid and diagnostic screening of aquatic contamination. Ecotoxicol Environ Saf 2018;147:670–680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Irons TD, MacPhail RC, Hunter DL, et al. Acute neuroactive drug exposures alter locomotor activity in larval zebrafish. Neurotoxicol Teratol 2010;32:84–90. [DOI] [PubMed] [Google Scholar]
- [21].Selderslaghs IWT, Van Rompay AR, De Coen W, et al. Development of a screening assay to identify teratogenic and embryotoxic chemicals using the zebrafish embryo. Reprod Toxicol 2009;28:308–320. [DOI] [PubMed] [Google Scholar]
- [22].Lambert MR, Spinazzola JM, Widrick JJ, et al. PDE10A inhibition reduces the manifestation of pathology in DMD zebrafish and represses the genetic modifier PITPNA. Mol Ther 2021;29:1086–1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Pakula A, Lek A, Widrick J, et al. Transgenic zebrafish model of DUX4 misexpression reveals a developmental role in FSHD pathogenesis. Hum Mol Genet 2019;28:320–331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Serafini PR, Feyder MJ, Hightower RM, et al. A limb-girdle muscular dystrophy 2I model of muscular dystrophy identifies corrective drug compounds for dystroglycanopathies. JCI Insight 2018;3:e120493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25]. Farrell TC, Cario CL, Milanese C, et al. Evaluation of spontaneous propulsive movement as a screening tool to detect rescue of Parkinsonism phenotypes in zebrafish models. Neurobiol Dis 2011;44:9–18. •• This key paper identified several factors that may confound the interpretation of spontaneous swimming activity data and describe methods for addressing these issues.
- [26].Gros-Louis F, Kriz J, Kabashi E, et al. Als2 mRNA splicing variants detected in KO mice rescue severe motor dysfunction phenotype in Als2 knock-down zebrafish. Hum Mol Genet 2008;17:2691–2702. [DOI] [PubMed] [Google Scholar]
- [27].Dowling JJ, Arbogast S, Hur J, et al. Oxidative stress and successful antioxidant treatment in models of RYR1-related myopathy. Brain 2012;135:1115–1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Saint-Amant L, Drapeau P. Time course of the development of motor behaviors in the zebrafish embryo. J Neurobiol 1998;37:622–632. [DOI] [PubMed] [Google Scholar]
- [29]. Kokel D, Peterson RT. Using the zebrafish photomotor response for psychotropic drug screening. Methods Cell Biol 2011;105:517–524. • This paper demonstrates how changes in pixel intensity can be used to quantify movement of early zebrafish embryos.
- [30].Naganawa Y, Hirata H. Developmental transition of touch response from slow muscle-mediated coilings to fast muscle-mediated burst swimming in zebrafish. Dev Biol 2011;355:194–204. [DOI] [PubMed] [Google Scholar]
- [31].Selderslaghs IWT, Hooyberghs J, Blust R, et al. Assessment of the developmental neurotoxicity of compounds by measuring locomotor activity in zebrafish embryos and larvae. Neurotoxicol Teratol 2013;37:44–56. [DOI] [PubMed] [Google Scholar]
- [32]. Müller UK, van Leeuwen JL. Swimming of larval zebrafish: Ontogeny of body waves and implications for locomotory development. J Exp Biol 2004;207:853–868. • This paper describes biomechanical properties of zebrafish larvae during slow and burst swimming activity.
- [33].Buss RR, Drapeau P. Synaptic drive to motoneurons during fictive swimming in the developing zebrafish. J Neurophysiol 2001;86:197–210. [DOI] [PubMed] [Google Scholar]
- [34].Lambert AM, Bonkowsky JL, Masino MA. The conserved dopaminergic diencephalospinal tract mediates vertebrate locomotor development in zebrafish larvae. J Neurosci 2012;32:13488–13500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Lindsey BW, Smith FM, Croll RP. From inflation to flotation: Contribution of the swimbladder to whole-body density and swimming depth during development of the zebrafish (Danio rerio). Zebrafish 2010;7:85–96. [DOI] [PubMed] [Google Scholar]
- [36].McClenahan P, Troup M, Scott EK. Fin-tail coordination during escape and predatory behavior in larval zebrafish. PLoS One 2012;7:e32295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Borla MA, Palecek B, Budick S, et al. Prey capture by larval zebrafish: Evidence for fine axial motor control. Brain Behav Evol 2002;60:207–229. [DOI] [PubMed] [Google Scholar]
- [38].Kimmel CB, Patterson J, Kimmel RO. The development and behavioral characteristics of the startle response in the zebra fish. Dev Psychobiol 1974;7:47–60. [DOI] [PubMed] [Google Scholar]
- [39].Eaton RC, Farley RD. Mauthner neuron field potential in newly hatched larvae of the zebra fish. J Neurophysiol 1975;38:502–512. [DOI] [PubMed] [Google Scholar]
- [40].Fontaine E, Lentink D, Kranenbarg S, et al. Automated visual tracking for studying the ontogeny of zebrafish swimming. J Exp Biol 2008;211:1305–1316. [DOI] [PubMed] [Google Scholar]
- [41].Voesenek CJ, Pieters RPM, van Leeuwen JL. Automated reconstruction of three-dimensional fish motion, forces, and torques. PLoS One 2016;11:e0146682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Nair A, Azatian G, McHenry MJ. The kinematics of directional control in the fast start of zebrafish larvae. J Exp Biol 2015;218:3996–4004. [DOI] [PubMed] [Google Scholar]
- [43].van Leeuwen JL, Voesenek CJ, Müller UK. How body torque and Strouhal number change with swimming speed and developmental stage in larval zebrafish. J R Soc Interface 2015;12:0479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Horstick EJ, Bayleyen Y, Sinclair JL, et al. Search strategy is regulated by somatostatin signaling and deep brain photoreceptors in zebrafish. BMC Biol 2017;15:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Štih V, Petrucco L, Kist AM, et al. Stytra: An open-source, integrated system for stimulation, tracking and closed-loop behavioral experiments. PLoS Comput Biol 2019;15:e1006699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Mirat O, Sternberg JR, Severi KE, et al. ZebraZoom: An automated program for high-throughput behavioral analysis and categorization. Front Neural Circuits 2013;7:107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Martineau PR, Mourrain P. Tracking zebrafish larvae in group–status and perspectives. Methods 2013;62:292–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Sztal TE, Ruparelia AA, Williams C, et al. Using touch-evoked response and locomotion assays to assess muscle performance and function in zebrafish. J Vis Exp 2016; [DOI] [PMC free article] [PubMed]
- [49].Muto A, Kawakami K. Prey capture in zebrafish larvae serves as a model to study cognitive functions. Front Neural Circuits 2013;7:110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Westphal RE, O’Malley DM. Fusion of locomotor maneuvers, and improving sensory capabilities, give rise to the flexible homing strikes of juvenile zebrafish. Front Neural Circuits 2013;7:108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Thorsen DH, Cassidy JJ, Hale ME. Swimming of larval zebrafish: Fin-axis coordination and implications for function and neural control. J Exp Biol 2004;207:4175–4183. [DOI] [PubMed] [Google Scholar]
- [52].Fuiman LA, Webb PW. Ontogeny of routine swimming activity and performance in zebra danios (Teleostei: Cyprinidae). Animal Behaviour 1988;36:250–261. [Google Scholar]
- [53].McHenry MJ, Lauder GV. The mechanical scaling of coasting in zebrafish (Danio rerio). J Exp Biol 2005;208:2289–2301. [DOI] [PubMed] [Google Scholar]
- [54].Müller UK, Stamhuis EJ, Videler JJ. Hydrodynamics of unsteady fish swimming and the effects of body size: Comparing the flow fields of fish larvae and adults. J Exp Biol 2000;203:193–206. [DOI] [PubMed] [Google Scholar]
- [55].Smith LL, Beggs AH, Gupta VA. Analysis of skeletal muscle defects in larval zebrafish by birefringence and touch-evoke escape response assays. J Vis Exp 2013;82:e50925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Liu KS, Fetcho JR. Laser ablations reveal functional relationships of segmental hindbrain neurons in zebrafish. Neuron 1999;23:325–335. [DOI] [PubMed] [Google Scholar]
- [57].Sillar KT. Mauthner cells. Curr Biol 2009;19:R353–355. [DOI] [PubMed] [Google Scholar]
- [58].Tabor KM, Bergeron SA, Horstick EJ, et al. Direct activation of the Mauthner cell by electric field pulses drives ultrarapid escape responses. J Neurophysiol 2014;112:834–844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].McElligott MB, O’Malley DM. Prey tracking by larval zebrafish: Axial kinematics and visual control. Brain Behav Evol 2005;66:177–196. [DOI] [PubMed] [Google Scholar]
- [60].Gahtan E, Tanger P, Baier H. Visual prey capture in larval zebrafish is controlled by identified reticulospinal neurons downstream of the tectum. J Neurosci 2005;25:9294–9303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Fiaz AW, Léon-Kloosterziel KM, Gort G, et al. Swim-training changes the spatio-temporal dynamics of skeletogenesis in zebrafish larvae (Danio rerio). PLoS ONE 2012;7:e34072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Bagatto B, Pelster B, Burggren WW. Growth and metabolism of larval zebrafish: Effects of swim training. J Exp Biol 2001;204:4335–4343. [DOI] [PubMed] [Google Scholar]
- [63].LeFauve MK, Rowe CJ, Crowley-Perry M, et al. Using a variant of the optomotor response as a visual defect detection assay in zebrafish. J Biol Methods 2021;8:e144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [64].Severi KE, Portugues R, Marques JC, et al. Neural control and modulation of swimming speed in the larval zebrafish. Neuron 2014;83:692–707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [65].Granato M, van Eeden FJ, Schach U, et al. Genes controlling and mediating locomotion behavior of the zebrafish embryo and larva. Development 1996;123:399–413. [DOI] [PubMed] [Google Scholar]
- [66].Bassett D, Currie PD. Identification of a zebrafish model of muscular dystrophy. Clin Exp Pharmacol Physiol 2004;31:537–540. [DOI] [PubMed] [Google Scholar]
- [67].Jacoby AS, Busch-Nentwich E, Bryson-Richardson RJ, et al. The zebrafish dystrophic mutant softy maintains muscle fibre viability despite basement membrane rupture and muscle detachment. Development 2009;136:3367–3376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Steffen LS, Guyon JR, Vogel ED, et al. Zebrafish orthologs of human muscular dystrophy genes. BMC Genomics 2007;8:79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Goody MF, Kelly MW, Reynolds CJ, et al. NAD+ biosynthesis ameliorates a zebrafish model of muscular dystrophy. PLoS Biol 2012;10:e1001409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [70].Branson K, Robie AA, Bender J, et al. High-throughput ethomics in large groups of Drosophila. Nat Methods 2009;6:451–457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [71].Mora-Zamorano FX, Svoboda KR, Carvan MJ. The nicotine-evoked locomotor response: A behavioral paradigm for toxicity screening in zebrafish (Danio rerio) embryos and eleutheroembryos exposed to methylmercury. PLoS ONE 2016;11:e0154570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Franco-Restrepo JE, Forero DA, Vargas RA. A review of freely available, open-source software for the automated analysis of the behavior of adult zebrafish. Zebrafish 2019;16:223–232. [DOI] [PubMed] [Google Scholar]
- [73].Ingebretson JJ, Masino MA. Quantification of locomotor activity in larval zebrafish: Considerations for the design of high-throughput behavioral studies. Front Neural Circuits 2013;7:109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Schnörr SJ, Steenbergen PJ, Richardson MK, et al. Measuring thigmotaxis in larval zebrafish. Behav Brain Res 2012;228:367–374. [DOI] [PubMed] [Google Scholar]
- [75].Colwill RM, Creton R. Locomotor behaviors in zebrafish (Danio rerio) larvae. Behav Processes 2011;86:222–229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Fitzgerald JA, Kirla KT, Zinner CP, et al. Emergence of consistent intra-individual locomotor patterns during zebrafish development. Sci Rep 2019;9:13647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Lundegaard PR, Anastasaki C, Grant NJ, et al. MEK inhibitors reverse cAMP-mediated anxiety in zebrafish. Chem Biol 2015;22:1335–1346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [78].de Esch C, van der Linde H, Slieker R, et al. Locomotor activity assay in zebrafish larvae: Influence of age, strain and ethanol. Neurotoxicol Teratol 2012;34:425–433. [DOI] [PubMed] [Google Scholar]
- [79].Lange M, Neuzeret F, Fabreges B, et al. Inter-individual and inter-strain variations in zebrafish locomotor ontogeny. PLoS ONE 2013;8:e70172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [80].MacPhail RC, Brooks J, Hunter DL, et al. Locomotion in larval zebrafish: Influence of time of day, lighting and ethanol. Neurotoxicology 2009;30:52–58. [DOI] [PubMed] [Google Scholar]
- [81].Padilla S, Hunter DL, Padnos B, et al. Assessing locomotor activity in larval zebrafish: Influence of extrinsic and intrinsic variables. Neurotoxicol Teratol 2011;33:624–630. [DOI] [PubMed] [Google Scholar]
- [82].Vignet C, Bégout M-L, Péan S, et al. Systematic screening of behavioral responses in two zebrafish strains. Zebrafish 2013;10:365–375. [DOI] [PubMed] [Google Scholar]
- [83].Zellner D, Padnos B, Hunter DL, et al. Rearing conditions differentially affect the locomotor behavior of larval zebrafish, but not their response to valproate-induced developmental neurotoxicity. Neurotoxicol Teratol 2011;33:674–679. [DOI] [PubMed] [Google Scholar]
- [84].Gorissen M, Manuel R, Pelgrim TNM, et al. Differences in inhibitory avoidance, cortisol and brain gene expression in TL and AB zebrafish. Genes Brain Behav 2015;14:428–438. [DOI] [PubMed] [Google Scholar]
- [85].Meyer BM, Froehlich JM, Galt NJ, et al. Inbred strains of zebrafish exhibit variation in growth performance and myostatin expression following fasting. Comp Biochem Physiol A Mol Integr Physiol 2013;164:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [86].Wakamatsu Y, Ogino K, Hirata H. Swimming capability of zebrafish is governed by water temperature, caudal fin length and genetic background. Sci Rep 2019;9:16307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [87].Burgess HA, Granato M. The neurogenetic frontier–lessons from misbehaving zebrafish. Brief Funct Genomic Proteomic 2008;7:474–482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [88].Joo W, Vivian MD, Graham BJ, et al. A Customizable Low-Cost System for Massively Parallel Zebrafish Behavioral Phenotyping. Front Behav Neurosci 2020;14:606900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [89].Schwerte T, Voigt S, Pelster B. Epigenetic variations in early cardiovascular performance and hematopoiesis can be explained by maternal and clutch effects in developing zebrafish (Danio rerio). Comp Biochem Physiol A Mol Integr Physiol 2005;141:200–209. [DOI] [PubMed] [Google Scholar]
- [90].Bianco IH, Kampff AR, Engert F. Prey capture behavior evoked by simple visual stimuli in larval zebrafish. Front Syst Neurosci 2011;5:101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [91].Hernandez RE, Galitan L, Cameron J, et al. Delay of initial feeding of zebrafish larvae until 8 days postfertilization has no impact on survival or growth through the juvenile stage. Zebrafish 2018;15:515–518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [92].Clift D, Richendrfer H, Thorn RJ, et al. High-throughput analysis of behavior in zebrafish larvae: Effects of feeding. Zebrafish 2014;11:455–461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [93].Watts SA, Lawrence C, Powell M, et al. The vital relationship between nutrition and health in zebrafish. Zebrafish 2016;13 Suppl 1:S72–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [94].Xie R, Zhang M, Venkatraman P, et al. Normalization of large-scale behavioural data collected from zebrafish. PLoS ONE 2019;14:e0212234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [95].Hurd MW, Cahill GM. Entraining signals initiate behavioral circadian rhythmicity in larval zebrafish. J Biol Rhythms 2002;17:307–314. [DOI] [PubMed] [Google Scholar]
- [96].Kristofco LA, Cruz LC, Haddad SP, et al. Age matters: Developmental stage of Danio rerio larvae influences photomotor response thresholds to diazinion or diphenhydramine. Aquat Toxicol 2016;170:344–354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [97].Zhang J-H, Chung TDY, Oldenburg KR. A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J Biomol Screen 1999;4:67–73. [DOI] [PubMed] [Google Scholar]
- [98].Khalili A, Rezai P. Microfluidic devices for embryonic and larval zebrafish studies. Brief Funct Genomics 2019;18:419–432. [DOI] [PubMed] [Google Scholar]
- [99].Voesenek CJ, Pieters RPM, Muijres FT, et al. Reorientation and propulsion in fast-starting zebrafish larvae: An inverse dynamics analysis. J Exp Biol 2019;222. [DOI] [PubMed]
- [100].Zhao Z, Li G, Xiao Q, et al. Quantification of the influence of drugs on zebrafish larvae swimming kinematics and energetics. PeerJ 2020;8:e8374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [101].Marques JC, Lackner S, Félix R, et al. Structure of the zebrafish locomotor repertoire revealed with unsupervised behavioral clustering. Curr Biol 2018;28:181–195.e5. [DOI] [PubMed] [Google Scholar]


