Abstract
Non-aureus staphylococci (NAS), particularly antimicrobial-resistant NAS, have a substantial impact on human and animal health. In the current study, we investigated (1) the species profiles of NAS isolates collected from healthy broilers, farm environments, and farm workers in Korea, (2) the occurrence of antimicrobial-resistant NAS isolates, especially methicillin resistance, and (3) the genetic factors involved in the methicillin and fluoroquinolone resistance. In total, 216 NAS isolates of 16 different species were collected from healthy broilers (n=178), broiler farm environments (n=18), and farm workers (n=20) of 20 different broiler farms. The two most dominant broiler-associated NAS species were Staphylococcus agnetis (23.6%) and Staphylococcus xylosus (22.9%). Six NAS isolates were mecA-positive carrying staphylococcal cassette chromosome mec (SCCmec) II (n=1), SCCmec IV (n=1), SCCmec V (n=2), or non-typeable SCCmec element (n=2). While two mecA-positive Staphylococcus epidermidis isolates from farm workers had SCCmec II and IV, a mecA-positive S. epidermidis isolate from broiler and a Staphylococcus haemolyticus isolate farm environment carried SCCmec V. The occurrence of multidrug resistance was observed in 48.1% (104/216 isolates) of NAS isolates with high resistance rates to β-lactams (>40%) and fusidic acid (59.7%). Fluoroquinolone resistance was confirmed in 59 NAS isolates (27.3%), and diverse mutations in the quinolone resistance determining regions of gyrA, gyrB, parC, and parE were identified. These findings suggest that NAS in broiler farms may have a potential role in the acquisition, amplification, and transmission of antimicrobial resistance.
Keywords: non-aureus staphylococci, broiler, species profiles, antimicrobial resistance
Introduction
Several recent studies have demonstrated that non-aureus staphylococci (NAS), including coagulase-negative staphylococci (CoNS), have a substantial impact on human and animal health (Fisher et al., 2018). Nosocomial CoNS are particularly well-known for their ability to form biofilms and acquire resistance to multiple antimicrobial agents (Nadell et al., 2009; O’Gara and Humphreys, 2001; Schilcher and Horswill, 2020). It has been suggested that NAS in livestock farm environments represent a significant reservoir of antimicrobial resistance (AMR) genes, facilitating the horizontal transfer of AMR (Feßler et al., 2018; Shen et al., 2013). Methicillin-resistant Staphylococcus aureus (MRSA) has also been proposed to have acquired staphylococcal cassette chromosome mec (SCCmec) from CoNS, which normally colonize various animal hosts.
The high prevalence of antimicrobial-resistant staphylococci, particularly multidrug-resistant (MDR) staphylococci, in livestock animals and farm environments has become a significant hazard to food safety and public health. Although a relatively large number of studies have focused on livestock-associated MRSA (LA-MRSA) in swine and cattle farms, the prevalence of NAS and their AMR profiles in livestock farms, especially broiler farms, are not well established. Only a few recent studies have investigated the prevalence of antimicrobial-resistant S. aureus in broiler farms, poultry slaughterhouses, and retail chicken meat samples in Korea (Kim et al., 2018; Lee et al., 2022). In addition, NAS species, such as Staphylococcus agnetis, Staphylococcus saprophyticus, Staphylococcus xylosus, Staphylococcus simulans, and Staphylococcus lentus, have been reported as opportunistic pathogens in poultry (Boamah et al., 2017; Pyzik et al., 2019; Silva et al., 2022; Szafraniec et al., 2020). However, information on the distribution of NAS species in broiler farms and their AMR profiles is still not available in Korea.
In the present study, we analyzed the species profiles of NAS isolates collected from broiler farms, including healthy broilers, farm environments, and farm workers. In addition, the AMR profiles of the NAS isolates and the major genetic factors involved in the resistance phenotypes, especially methicillin and fluoroquinolone resistance, were examined. To the best of our knowledge, this is the first study to report the species profiles, AMR, and genetic factors associated with fluoroquinolone resistance in NAS isolates from broiler farms in Korea.
Materials and Methods
Collection of samples
All swab samples were obtained from 20 different broiler farms across eight provinces of Korea in 2019 as previously described (Lee et al., 2022). A total of 916 swab samples were collected from healthy broilers (n=782; cloacal and throat swabs), farm facilities (n=71; floor, fence, drinking system, feeder pan, sewage, and ventilators), and farm workers (n=63; hand and nasal swabs). After sampling, all swabs were transported using ice-cooled containers and processed for the isolation of NAS within 36 h of sample collection. The sampling protocol was reviewed and approved by the IRB/IACUC at Chung-Ang University.
Isolation and identification of non-aureus staphylococci (NAS)
Isolation and culture of NAS strains from swab samples were performed as previously described, with minor modifications (Lee et al., 2022). Briefly, all swab samples were inoculated into 4.5 mL of tryptic soy broth (Difco Laboratories, Detroit, MI, USA) containing 10% sodium chloride and incubated at 37°C for 17–24 h for enrichment. Next, 15–20 μL of the pre-enriched cultures were streaked onto Baird-Parker agar (Difco Laboratories) supplemented with egg yolk and potassium tellurite (Becton Dickinson, Sparks, MD, USA) and incubated for 48 h at 37°C. One or two colonies of presumptive staphylococci per sample were then selected for subsequent identification of species. The species of the NAS strains were first identified by using a matrix-assisted laser desorption ionization (MALDI)-Biotyper classification system (Bruker, Bremen, Germany) and then 16S rRNA sequencing for strains with <2.0 scores in the MALDI-Biotyper analysis, as previously described (Yang et al., 2022).
Antimicrobial susceptibility tests
To determine the AMR profiles of the NAS strains, standard disc diffusion assays were performed according to the Clinical and Laboratory Standards Institute guidelines (CLSI) (CLSI, 2020a; CLSI, 2020b) and the European Committee on Antimicrobial Susceptibility Testing (EUCAST). The 14 antimicrobial agents included in the disc diffusion assays were ampicillin (AMP, 10 μg), cefoxitin (FOX, 30 μg), penicillin (PEN, 10 units), gentamicin (GEN, 50 μg), chloramphenicol (CHL, 30 μg), ciprofloxacin (CIP, 5 μg), clindamycin (CLI, 2 μg), erythromycin (ERY, 15 μg), fusidic acid (FUS, 50 μg), mupirocin (MUP, 200 μg), rifampin (RIF, 5 μg), sulfamethoxazole-trimethoprim (SXT, 23.73–1.25 μg), quinupristin-dalfopristin (SYN, 15 μg), and tetracycline (TET, 30 μg). All antimicrobial discs were purchased from BD BBLTM (Becton Dickinson, Franklin Lakes, NJ, USA), except for the MUP discs (Oxoid, Hampshire, UK). The minimum inhibitory concentrations of vancomycin (VAN), tigecycline (TGC), linezolid (LZD), and teicoplanin (TEC) were determined using standard E-test strips (bioMérieux, Craponne, France) on Mueller-Hinton agar (Difco Laboratories). Two reference strains, S. aureus ATCC29213 and S. aureus MW2, were used in antimicrobial susceptibility assays.
Detection of mecA and staphylococcal cassette chromosome mec (SCCmec) typing
All NAS strains displaying resistance phenotypes to β-lactam antimicrobial agents were subjected to polymerase chain reaction (PCR) to detect mecA gene, as previously described (Geha et al., 1994). The types of SCCmec were then determined in all mecA-positive NAS strains using previously described multiplex PCR methods that amplify chromosomal cassette recombinase (ccr) genes and mec regulatory elements. The combinations of ccr types and mec complexes were analyzed to assign the SCCmec types of methicillin-resistant NAS strains (Geha et al., 1994; Mendoza et al., 1998).
Mechanisms of fluoroquinolone resistance in the non-aureus staphylococci (NAS) strains
To detect the mutations in fluoroquinolone-resistant strains, quinolone-resistance determining regions (QRDRs) within gyrA, gyrB, parC, and parE were PCR-amplified using specific primer sets as previously described (Lee et al., 2020; Sreedharan et al., 1991; Takahashi et al., 1998; Takahata et al., 1997). The PCR products of the QRDRs were then sequenced (Bionics, Seoul, Korea), and mutations were identified using the Cluster Omega server (http://ebi.ac.uk/Tools/msa/clustalo/). The published sequences of S. agnetis (CP009623.1), Staphylococcus chromogenes (CP046028.1), Staphylococcus arlettae (AP019698.1), Staphylococcus cohnii (CP027422.1), Staphylococcus condimenti (CP018776.1), S. haemolyticus (CP035291.1), Staphylococcus gallinarum (CP086207.1), S. saprophyticus (CP031196.1), S. simulans (LT963435.1), and S. xylosus (CP013922.1) were used to design QRDR-specific primer sets in each species (Supplementary Table S1).
Results
Profiles of non-aureus staphylococci (NAS) from broiler farms
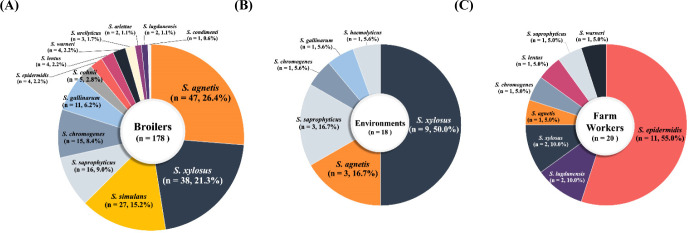
A total of 216 NAS strains of 16 different species were isolated from 916 swab samples (216/916, 23.58%) collected from broiler farms during the study period (Figs. 1A, B, and C). Of the 216 NAS strains, 148 (68.5%) CoNS strains displayed 14 different species diversities, whereas 68 coagulase-variable staphylococci (CoVS) were composed of only two species of staphylococci, S. agnetis and S. chromogenes. As shown in Fig. 1A, 178/216 (82.4%) NAS strains were isolated from healthy broilers, with S. agnetis and S. xylosus being most frequently associated with healthy broilers (Fig. 1A). Similar to the profiles of NAS strains in broilers, 83.3% (15/18) of NAS strains from farm environments were S. xylosus (n=9), S. agnetis (n=3), and S. saprophyticus (n=3) strains (Fig. 1B). NAS strains from farm workers were distinctive from those of broilers and farm environments in that Staphylococcus epidermidis (11/20, 55%) was most prevalent in the nasal cavities of farm workers (Fig. 1C).
Fig. 1. Profiles of non-aureus staphylococci (NAS) isolated from broilers (A), farm environments (B), and farm workers (C) in Korea.
A total of 216 NAS isolates of 16 different staphylococcal species were collected from broiler farms in Korea.
Occurrence of mecA in non-aureus staphylococci (NAS) isolated from broilers
Six methicillin-resistant NAS were identified in 216 NAS strains (3.24%) isolated from broiler farms (Table 1). All six strains were mecA-positive (Table 1) and were resistant to FOX (Table 2). Analysis of SCCmec elements in seven methicillin- resistant NAS revealed that two CoNS (S. epidermidis and S. haemolyticus) carried SCCmec V (Table 1). Two methicillin-resistant S. epidermidis strains isolated from farm workers contained SCCmec II and SCCmec IV. Although two CoNS strains (Staphylococcus sciuri and Staphylococcus ureilyticus) were mecA-positive, the SCCmec types could not be determined using PCR because of unidentifiable ccr or mec types.
Table 1. SCCmec types of methicillin-resistant NAS strains isolated from broiler farms.
| NAS (n=isolates) | mecA positive (%) | mec gene | ccr gene | SCCmec type | Isolated from |
|---|---|---|---|---|---|
| CoVS (68) | |||||
| Staphylococcus agnetis (51) | - | - | - | - | |
| Staphylococcus chromogenes (17) | - | - | - | - | |
| CoNS (148) | |||||
| Staphylococcus arlettae (2) | - | - | - | - | |
| Staphylococcus cohnii (5) | - | - | - | - | |
| Staphylococcus condimenti (1) | - | - | - | - | |
| Staphylococcus epidermidis (15) | 3 (20) | B | A2B2 | IV | Worker |
| A | A2B2 | II | Worker | ||
| C2 | C | V | Broiler | ||
| Staphylococcus gallinarum (12) | - | - | - | - | |
| Staphylococcus haemolyticus (1) | 1 (100) | C2 | C | V | Environment |
| Staphylococcus lentus (5) | |||||
| Staphylococcus lugdunesis (2) | |||||
| Staphylococcus saprophyticus (20) | - | - | - | - | |
| Staphylococcus sciuri (1) | 1 (100) | A | - | NT | Broiler |
| Staphylococcus simulans (27) | - | - | - | - | |
| Staphylococcus ureilyticus (3) | 1 (33.3) | - | C | NT | Broiler |
| Staphylococcus warneri (5) | - | - | - | - | |
| Staphylococcus xylosus (49) | - | - | - | - |
SCCmec, staphylococcal cassette chromosome mec; NAS, non-aureus staphylococci; ccr, chromosomal cassette recombinase; CoVS, coagulase-variable staphylococci; CoNS, coagulase-negative staphylococci; NT, non-typeable.
Table 2. Antimicrobial resistance profiles of NAS strains isolated from broiler farms.
| Species (No. of isolates) | Number (%) of isolates resistant to: | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AMP | FOX | PEN | CHL | CIP | CLI | ERY | FUS | GEN | MUP | RIF | SXT | SYN | TET | VAN | TEC | LZD | TGC | MDR (%) | |
| CoVS (68) | |||||||||||||||||||
| Staphylococcus agnetis (51) | 29 (56.9) | 0 | 28 (54.9) | 6 (11.8) | 11 (21.6) | 1 (2) | 1 (2) | 47 (90.2) | 28 (54.9) | 1 (2) | 0 | 0 | 0 | 9 (17.6) | 0 | 0 | 0 | 0 | 30 (59) |
| Staphylococcus chromogenes (17) | 5 (29.4) | 0 | 5 (29.4) | 5 (29.4) | 7 (41.2) | 8 (47.1) | 3 (17.6) | 0 | 0 | 0 | 0 | 0 | 0 | 6 (35.3) | 0 | 0 | 0 | 0 | 8 (47) |
| CoVS Total | 34 (50) | 0 | 33 (48.5) | 11 (16.2) | 18 (26.5) | 9 (13.2) | 4 (5.9) | 47 (69.1) | 28 (41.2) | 1 (1.5) | 0 | 0 | 0 | 15 (22.1) | 0 | 0 | 0 | 0 | 38 (55.9) |
| CoNS (148) | |||||||||||||||||||
| Staphylococcus arlettae (2) | 2 (100) | 0 | 2 (100) | 2 (100) | 1 (50) | 1 (50) | 2 (100) | 2 (100) | 0 | 0 | 0 | 0 | 0 | 2 (100) | 0 | 0 | 0 | 0 | 2 (100) |
| Staphylococcus cohnii (5) | 1 (20) | 0 | 2 (40) | 5 (100) | 1 (20) | 3 (60) | 3 (60) | 5 (100) | 0 | 0 | 0 | 0 | 0 | 4 (80) | 0 | 0 | 0 | 0 | 5 (100) |
| Staphylococcus condimenti (1) | 0 | 0 | 0 | 0 | 1 (100) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 (100) | 0 | 0 | 0 | 0 | 0 |
| Staphylococcus epidermidis (15) | 9 (60) | 3 (20) | 9 (60) | 0 | 1 (6.7) | 1 (6.7) | 3 (20) | 6 (40) | 2 (13.3) | 2 (13.3) | 0 | 2 (13.3) | 0 | 3 (20) | 0 | 0 | 0 | 0 | 4 (26.7) |
| Staphylococcus gallinarum (12) | 12 (100) | 0 | 12 (100) | 2 (16.7) | 8 (66.7) | 8 (66.7) | 8 (66.7) | 2 (16.7) | 0 | 0 | 0 | 0 | 0 | 9 (75) | 0 | 0 | 0 | 0 | 8 (66.7) |
| Staphylococcus haemolyticus (1) | 1 (100) | 1 (100) | 1 (100) | 1 (100) | 1 (100) | 1 (100) | 1 (100) | 0 | 0 | 0 | 0 | 0 | 1 (100) | 1 (100) | 0 | 0 | 0 | 0 | 1 (100) |
| Staphylococcus lentus (5) | 0 | 0 | 0 | 3 (60) | 3 (60) | 1 (20) | 3 (60) | 5 (100) | 0 | 0 | 0 | 1 (20) | 0 | 1 (20) | 0 | 0 | 0 | 0 | 4 (80) |
| Staphylococcus lugdunensis (2) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Staphylococcus saprophyticus (20) | 3 (15) | 0 | 12 (60) | 1 (5) | 6 (30) | 3 (15) | 8 (40) | 19 (95) | 0 | 0 | 0 | 1 (5) | 0 | 11 (55) | 0 | 0 | 0 | 0 | 13 (65) |
| Staphylococcus sciuri (1) | 1 (100) | 1 (100) | 1 (100) | 0 | 1 (100) | 0 | 1 (100) | 1 (100) | 0 | 0 | 0 | 1 (100) | 0 | 1 (100) | 0 | 0 | 0 | 0 | 1 (100) |
| Staphylococcus simulans (27) | 3 (11.1) | 0 | 3 (11.1) | 3 (11.1) | 5 (18.5) | 6 (22.2) | 6 (22.2) | 0 | 1 (3.7) | 0 | 0 | 3 (11.1) | 0 | 3 (11.1) | 0 | 0 | 0 | 0 | 5 (19) |
| Staphylococcus ureilyticus (3) | 0 | 1 (33.3) | 0 | 2 (66.7) | 0 | 1 (33.3) | 1 (33.3) | 3 (100) | 0 | 0 | 0 | 0 | 0 | 1 (33.3) | 0 | 0 | 0 | 0 | 2 (66.7) |
| Staphylococcus warneri (5) | 5 (100) | 0 | 5 (100) | 0 | 0 | 0 | 1 (20) | 1 (20) | 0 | 1 (20) | 0 | 0 | 0 | 1 (20) | 0 | 0 | 0 | 0 | 1 (20) |
| Staphylococcus xylosus (49) | 18 (36.7) | 0 | 35 (71.4) | 12 (24.5) | 11 (22.4) | 2 (4.1) | 4 (8.2) | 39 (79.6) | 3 (6.1) | 0 | 0 | 0 | 0 | 21 (42.9) | 0 | 0 | 0 | 0 | 20 (40.8) |
| CoNS total | 55 (37.2) | 6 (4.1) | 82 (55.4) | 31 (20.9) | 39 (26.4) | 27 (18.2) | 41 (27.7) | 83 (56.1) | 6 (4.1) | 3 (2.0) | 0 | 8 (5.4) | 1 (0.7) | 59 (39.9) | 0 | 0 | 0 | 0 | 66 (44.6) |
| Total (216) | 89 (41.2) | 6 (2.8) | 115 (53.2) | 42 (19.4) | 57 (26.4) | 36 (16.7) | 45 (20.8) | 130 (60.2) | 34 (15.7) | 4 (1.9) | 0 | 8 (3.7) | 1 (0.5) | 74 (34.3) | 0 | 0 | 0 | 0 | 104 (48.1) |
NAS, non-aureus staphylococci; AMP, ampicillin; FOX, cefoxitin; PEN, penicillin; CHL, chloramphenicol; CIP, ciprofloxacin; CLI, clindamycin; ERY, erythromycin; FUS, fusidic acid; GEN, gentamicin; MUP, mupirocin; RIF, rifampin; SXT, trimethoprim-sulfamethoxazole; SYN, quinupristin- dalfopristin; TET, tetracycline; VAN, vancomycin; TEC, teicoplanin; LZD, linezolid; TGC, tigecycline; MDR, multi-drug resistance; CoVS, coagulase-variable staphylococci; CoNS, coagulase-negative staphylococci.
Antimicrobial resistance (AMR) profiles of non-aureus staphylococci (NAS) isolates from broiler farms
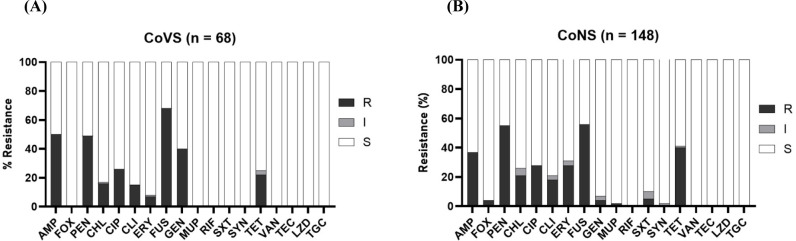
All 216 NAS isolates (both CoVS and CoNS) were susceptible to RIF, VAN, LZD, TEC, and TGC (Figs. 2A and B). CoVS strains (41.2%), particularly S. agnetis strains (54.9%), showed higher levels of resistance to GEN than the CoNS strains (4.1%; Table 2). Both CoVS and CoNS displayed relatively higher levels of resistance to FUS, AMP, PEN, TET, and CIP than to other antimicrobial agents. MDR phenotypes, which show resistance to more than three different classes of antimicrobial agents, were observed in 55.9% and 44.6% of CoVS and CoNS strains, respectively (Table 2). When the two major species of CoVS (S. agnetis and S. chromogenes) were compared for resistance, S. agnetis strains exhibited significantly higher levels of resistance to AMP, PEN, FUS, and GEN, whereas S. chromogenes showed higher resistance to CIP, CLI, and TET (Table 2). Among the four major groups of CoNS strains, S. xylosus and S. saprophyticus strains displayed higher levels of MDR than S. simulans and S. epidermidis strains. S. xylosus and S. saprophyticus strains were highly resistant to PEN (>60%), FUS (>79.6%) and TET (>42.9%).
Fig. 2. Antimicrobial resistance profiles of NAS isolates from broiler farms in Korea.
Antimicrobial resistance phenotypes of CoVS (A) and CoNS (B) isolates are shown. AMP, ampicillin; FOX, cefoxitin; PEN, penicillin; CHL, chloramphenicol; CIP, ciprofloxacin; CLI, clindamycin; ERY, erythromycin; FUS, fusidic acid; GEN, gentamicin; MUP, mupirocin; RIF, rifampicin; SXT, trimethoprim-sulfamethoxazole; SYN, quinupristin-dalfopristin; TET, tetracycline; VAN, vancomycin; TEC, teicoplanin; LZD, linezolid; TGC, tigecycline; R, resistant; I, intermediate; S, susceptible; NAS, non-aureus staphylococci; CoVS, coagulase-variable staphylococci; CoNS, coagulase-negative staphylococci.
Mutations in the quinolone-resistance determining regions (QRDRs) of fluoroquinolone-resistant non-aureus staphylococci (NAS) strains
Eighteen CoVS (18/68 strains, 26.5%) and 39 CoNS (39/148 strains, 26.4%) strains displayed resistance to fluoroquinolones (Table 3). All 57 fluoroquinolone-resistant NAS strains had at least one point mutation in the QRDRs of gyrA, gyrB, parC, or parE genes. As shown in Table 3, all fluoroquinolone-resistant NAS strains had the S84L mutation in gyrA, except for six strains of S. cohnii (n=1), S. condimenti (n=1), S. lentus (n=3) and S. epidermidis (n=1). Mutations in gyrB were identified in only three species of CoNS: S. sciuri, S. simulans, and S. xylosus strains. Among the four point mutations in gyrB (S567A, A512R, E490G, and S494T), the S567A mutation was most frequently identified in fluoroquinolone-resistant CoNS, especially in S. xylosus. Although the S80L mutation was most frequently observed in fluoroquinolone-resistant NAS strains, particularly in S. agnetis and S. saprophyticus strains, high levels of heterogeneity were observed in parC mutations of S. epidermidis, S. gallinarum, and S. xylosus strains (Table 3). In contrast to mutations in gyrA, gyrB, and parC, which were identified in multiple species of fluoroquinolone-resistant NAS, the T379K mutation in parE was detected only in S. simulans strains.
Table 3. Point mutations identified in the QRDRs of gyrA, gyrB, parC, and parE genes in fluoroquinolone-resistant NAS strains.
| NAS species | No. of FQ-resistant isolates (%) | Mutations in QRDRs | Total No. | ||||
|---|---|---|---|---|---|---|---|
| gyrA | gyrB | parC | parE | ||||
| CoVS | Staphylococcus agnetis (51) | 11 (21.6) | S84L | - | S80L | - | 10 |
| S84L | - | S80L, Y56H | - | 1 | |||
| Staphylococcus chromogenes (17) | 7 (41.2) | S84L | - | - | - | 6 | |
| S84L | - | R87I, S89T | - | 1 | |||
| CoNS | Staphylococcus arlettae (2) | 1 (50) | S84L | - | T80I | - | 1 |
| Staphylococcus cohnii (5) | 1 (20) | S84F, V158A | - | - | - | 1 | |
| Staphylococcus condimenti (1) | 1 (100) | S84F | - | - | - | 1 | |
| Staphylococcus epidermidis (15) | 1 (6.7) | S28E, V29C, S84F | - | Q81K, D81H, G107A, S108R, I109L | - | 1 | |
| Staphylococcus gallinarum (12) | 8 (66.7) | S84L | - | Y56F, Y74F, S80I, G92D | 8 | ||
| Staphylococcus haemolyticus (1) | 1 (100) | S84L | - | - | - | 1 | |
| Staphylococcus lentus (5) | 3 (60) | T172A | - | S80V | - | 1 | |
| T172A | - | S80L | - | 2 | |||
| Staphylococcus saprophyticus (20) | 6 (43.8) | S84L | - | S80L, R96C | - | 6 | |
| Staphylococcus sciuri (1) | 1 (100) | S84L, T172A | S567A | S80I | - | 1 | |
| Staphylococcus simulans (27) | 5 (18.5) | S84L | A512R | - | T379K | 1 | |
| S84L, D105N, A119F | - | - | T379K | 1 | |||
| S84L, A132S, A173S | A512R | - | T379K | 1 | |||
| S84L, A132S | E490G, S494T | - | T379K | 1 | |||
| S84L, A132S, A173S | E490G, S494T | - | T379K | 1 | |||
| Staphylococcus xylosus (49) | 11 (22.4) | S84L | S567A | F160L, V165I, Y195F, I209V | - | 10 | |
| S84L | S567A | D148H, F160L, L164W, V165I, S168N, D178H, G185R, V187L, Y194F, K207T, Y208S, I209V, D213G | - | 1 | |||
QRDRs, quinolone resistance determining regions; NAS, non-aureus staphylococci; FQ, fluoroquinolone; CoVS, coagulase-variable staphylococci; CoNS, coagulase-negative staphylococci.
Discussion
Staphylococci are normal flora on the skin and mucous membranes of poultry and other livestock animals. However, some staphylococci can cause opportunistic infections in poultry and food poisoning in humans (Lee et al., 2022; Mekhloufi et al., 2021; Silva et al., 2022). Although recent studies have demonstrated the important role of CoNS and non-aureus CoPS in the transmission of AMR between livestock animals and humans (Huebner and Goldmann, 1999; Silva et al., 2022), limited data are available on the prevalence and species profiles of NAS, as well as their AMR resistance profiles.
Overall, the prevalence rates of CoVS and CoNS among the 216 NAS strains collected from broiler farms were 31.5% (68/216) and 68.5% (148/216), respectively (Table 1). Previous studies conducted in Korea (Lee et al., 2020) and other countries (Marek et al., 2016; Osman et al., 2016; Pyzik et al., 2019) also reported higher prevalence of CoNS compared with CoVS in broiler and retail chicken meat. In a study by Awan and Matsumoto (1998), 77/79 staphylococcal isolates from broilers that showed signs of illness were CoNS, such as S. simulans, S. lentus, and S. cohnii. These results suggest that NAS strains, especially various species of CoNS strains, are colonizing healthy broilers and farm environments, causing opportunistic infections in broilers. In this study, although only two species of CoVS, S. agnetis and S. chromogenes, were detected, 14 different species of CoNS were identified (Table 1 and Figs. 1A, B, and C). The most frequently isolated species of NAS from healthy broilers were S. agnetis (26.4%), S. xylosus (21.3%), and S. simulans (15.2%; Fig. 1A). Frequent occurrence of S. agnetis (3/18, 16.7%) and S. xylosus (9/18, 50.0%) were also observed in the farm environment (Fig. 1B). Recently published studies by our group and others reported that various NAS strains present in retail chicken meat (Martins et al., 2013; Osman et al., 2016; Yurdakul et al., 2013). In particular, the overall detection rate of NAS in retail chicken was 45.3% in Korea, and the most frequently detected NAS species in retail chicken meat samples were S. agnetis (19.4%) and S. saprophyticus (19%; Lee et al., 2020), suggesting S. agnetis may have transmitted from broiler farms to retail chicken meat. Although the most common NAS species in the nasal cavities of farm workers in this study were S. epidermidis (11/20, 55.0%) and Staphylococcus lugdunensis (2/20, 10.0%), a few strains of NAS species identified in broilers, such as S. agnetis (n=1), S. xylosus (n=2), and S. saprophyticus (n=1), were also isolated from the broiler farm workers (Fig. 1C). These NAS species have been shown to be mainly associated with local and chronic infections in broiler chickens and other poultry (Marek et al., 2016; Szafraniec et al., 2020). It has been reported that S. agnetis and S. xylosus have various virulence factors involved in host cell adherence, immune evasion, and toxin biosynthesis, which contribute to the persistence and pathogenesis of staphylococci in poultry (Szafraniec et al., 2020). S. agnetis has been associated with bacterial chondronecrosis, endocarditis, and septicemia in broiler chickens (Szafraniec et al., 2020). Moreover, exfoliative toxin genes, which contribute to the scalded-skin syndrome in humans, have been found in S. agnetis and S. chromogenes strains from poultry (Ladhani et al., 1999). These findings suggest that NAS species in broiler farms, especially CoVS species, may be a potential health hazard for broilers and a potential source of chicken meat contamination in Korea.
Recently, methicillin resistance in NAS strains, especially methicillin-resistant CoNS strains, has been reported in livestock farms and in foods of animal origin (Mahato et al., 2017). Analysis of the SCCmec types of the six mecA-positive NAS strains revealed that two CoNS (S. epidermidis and S. haemolyticus) from broilers or the surrounding environment carried SCCmec V, which is typically associated with methicillin-resistant staphylococci originating from livestock and companion animals (Back et al., 2020; Lee and Yang, 2020). In contrast to SCCmec V in broiler-associated NAS and S. aureus (Lee et al., 2022) strains, the two strains of S. epidermidis from farm workers harbored SCCmec II or SCCmec IV, which has been predominantly found in clinical S. epidermidis isolates from human patients (Barbier et al., 2010). Similar to previous studies, which reported heterogeneity of SCCmec elements in methicillin-resistant NAS isolates (Rolo et al., 2017), SCCmec types of the two mecA-positive CoNS strains, one S. sciuri and one S. ureilyticus strain, could not be determined because of non-typeable ccr/mec genes. Although the prevalence of SCCmec elements in NAS isolates in this study was much less than that of S. aureus in a previously reported study (2.8% vs. 21.5%; Lee et al., 2022) these results indicate that diverse species of NAS may contribute to the transmission of SCCmec elements among staphylococci.
It is estimated that approximately 20% of all antibiotics sold in the livestock industry are used for poultry in Korea (Lim et al., 2014). In particular, β-lactams, fluoroquinolones, and tetracyclines have been extensively used to prevent and treat staphylococcal infections in poultry. Correlating to the administration of antibiotics in poultry, relatively high levels of resistance to AMP, PEN, CIP, and TET were identified in both CoVS and CoNS strains (Figs. 2A and B), indicating that the administration of antibiotics facilitated the occurrence of AMR, including the NAS strains in this study. Although FUS has not been listed as a major antibiotic used in poultry farms in Korea, the highest levels of resistance have been identified in several species of CoVS and CoNS, particularly in S. agnetis (90.2%), S. xylosus (79.6%), and S. saprophyticus (95.0%; Table 2). Previously, it was reported that certain species of NAS, such as S. agnetis, S. saprophyticus, S. cohnii, and S. ureilyticus, frequently carry fusD and fusF for intrinsic resistance to FUS (Chen et al., 2015; O’Neill et al., 2007). The frequent occurrence of antimicrobial-resistant NAS strains, especially MDR strains displaying resistance phenotypes to the antibiotics commonly used in poultry farms, indicates that NAS strains from poultry farms can serve as important reservoirs for AMR.
Frequent occurrence of quinolone resistance has been reported in LA-MRSA and NAS isolates through point mutations in the QRDRs of the two essential enzymes, DNA gyrase and topoisomerase IV (Takahashi et al., 1998; Takahata et al., 1997). In this study, all 57 fluoroquinolone-resistant NAS strains possessed one or more point mutations in the QRDRs of gyrA, gyrB, parC, or parE genes (Table 3). In line with previous studies in S. aureus and CoNS (Lee et al., 2022; Takahashi et al., 1998), mutations at codon 84 of gyrA (S84L and S84F) and codon 80 of parC (S80L, T80I, S80I, and S80V) were most frequently found in fluoroquinolone-resistant NAS strains. In addition to the S84L mutation, nine different amino acid substitutions were identified in QRDRs of gyrA in CoNS strains. High level of heterogeneity in QRDRs of parC was also observed in CoNS strains, especially S. epidermidis, S. gallinarum, and S. xylosus strains. Although none of the fluoroquinolone-resistant CoVS strains had mutations in gyrB and parE, S. sciuri, S. simulans, and S. xylosus carried mutations in the QRDRs of gyrB and parE (Table 3). While S. sciuri and S. xylosus strains had S567A mutation in gyrB, S. simulans strains had A512R, E490G, and S494T mutations in gyrB. As shown in Table 3, only S. simulans strains carried the T379K mutation in parE. Although the T379K mutation has not been previously described, several other mutations in parE have been found in S. aureus (Fujimoto-Nakamura et al., 2005) and Streptococcus pneumoniae (Jones et al., 2000) in association with quinolone resistance. Further studies are required to determine whether the newly identified mutations in gyrB and parE can act synergistically with gyrA and parC mutations to further enhance quinolone resistance in CoNS strains.
In conclusion, our results suggest that (1) there is a relatively high degree of species diversity in NAS isolates, especially CoNS isolates, collected from broiler farms; (2) frequent occurrence of MDR in NAS isolates with high resistance to β-lactams, fluoroquinolones, and fusidane was observed; and (3) fluoroquinolone resistance in broiler-associated NAS isolates was mainly caused by various mutations in the QRDRs of gyrA and parC.
Acknowledgements
This study was supported by funding from the Research of Korea Centers for Disease Control and Prevention (Projects No. 2017NER54060 & 2021ER220100). We thank Soo In Lee and Sun Do Kim for excellent technical assistance with antimicrobial susceptibility assays.
Conflicts of Interest
The authors declare no potential conflicts of interest.
Author Contributions
Conceptualization: Kim GB, Park KT, Yang SJ. Formal analysis: Park JH, Lee GY, Lim JH. Methodology: Lee GY, Lim JH. Investigation: Park JH, Park KT, Yang SJ. Writing - original draft: Park JH, Yang SJ. Writing - review & editing: Park JH, Lee GY, Lim JH, Kim GB, Park KT, Yang SJ.
Ethics Approval
The sampling protocol was reviewed and approved by the IRB/IACUC (2018-00112) at Chung-Ang University.
Supplemenrary Materials
Supplementary Materials
References
- Awan MA, Matsumoto M. Heterogeneity of staphylococci and other bacteria isolated from six-week-old broiler chickens. Poult Sci. 1998;77:944–949. doi: 10.1093/ps/77.7.944. [DOI] [PubMed] [Google Scholar]
- Back SH, Eom HS, Lee HH, Lee GY, Park KT, Yang SJ. Livestock-associated methicillin-resistant Staphylococcus aureus in Korea: Antimicrobial resistance and molecular characteristics of LA-MRSA strains isolated from pigs, pig farmers, and farm environment. J Vet Sci. 2020;21:e2. doi: 10.4142/jvs.2020.21.e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbier F, Ruppé E, Hernandez D, Lebeaux D, Francois P, Felix B, Desprez A, Maiga A, Woerther PL, Gaillard K, Jeanrot C, Wolff M, Schrenzel J, Andremont A, Ruimy R. Methicillin-resistant coagulase-negative staphylococci in the community: High homology of SCCmec IVa between Staphylococcus epidermidis and major clones of methicillin-resistant Staphylococcus aureus. J Infect Dis. 2010;202:270–281. doi: 10.1086/653483. [DOI] [PubMed] [Google Scholar]
- Boamah VE, Agyare C, Odoi H, Adu F, Gbedema SY, Dalsgaard A. Prevalence and antibiotic resistance of coagulase-negative Staphylococci isolated from poultry farms in three regions of Ghana. Infect Drug Resist. 2017;10:175–183. doi: 10.2147/IDR.S136349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen HJ, Hung WC, Lin YT, Tsai JC, Chiu HC, Hsueh PR, Teng LJ. A novel fusidic acid resistance determinant, fusF, in Staphylococcus cohnii. J Antimicrob Chemother. 2015;70:416–419. doi: 10.1093/jac/dku408. [DOI] [PubMed] [Google Scholar]
- Clinical and Laboratory Standards Institute [CLSI] Performance standards for antimicrobial disk and dilution susceptibility tests for bacteria isolated from animals. Clinical and Laboratory Standards Institute; Wayne, PA, USA: 2020a. [Google Scholar]
- Clinical and Laboratory Standards Institute [CLSI] Performance standards for antimicrobial susceptibility testing. Clinical and Laboratory Standards Institute; Wayne, PA, USA: 2020b. [Google Scholar]
- Feßler AT, Wang Y, Wu C, Schwarz S. Mobile macrolide resistance genes in staphylococci. Plasmid. 2018;99:2–10. doi: 10.1016/j.plasmid.2018.05.001. [DOI] [PubMed] [Google Scholar]
- Fisher EL, Otto M, Cheung GYC. Basis of virulence in enterotoxin-mediated staphylococcal food poisoning. Front Microbiol. 2018;9:436. doi: 10.3389/fmicb.2018.00436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujimoto-Nakamura M, Ito H, Oyamada Y, Nishino T, Yamagishi J. Accumulation of mutations in both gyrB and parE genes is associated with high-level resistance to novobiocin in Staphylococcus aureus. Antimicrob Agents Chemother. 2005;49:3810–3815. doi: 10.1128/AAC.49.9.3810-3815.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geha DJ, Uhl JR, Gustaferro CA, Persing DH. Multiplex PCR for identification of methicillin-resistant staphylococci in the clinical laboratory. J Clin Microbiol. 1994;32:1768–1772. doi: 10.1128/jcm.32.7.1768-1772.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huebner J, Goldmann DA. Coagulase-negative staphylococci: Role as pathogens. Annu Rev Med. 1999;50:223–236. doi: 10.1146/annurev.med.50.1.223. [DOI] [PubMed] [Google Scholar]
- Jones ME, Sahm DF, Martin N, Scheuring S, Heisig P, Thornsberry C, Köhrer K, Schmitz FJ. Prevalence of gyrA, gyrB, parC, and parE mutations in clinical isolates of Streptococcus pneumoniae with decreased susceptibilities to different fluoroquinolones and originating from worldwide surveillance studies during the 1997-1998 respiratory season. Antimicrob Agents Chemother. 2000;44:462–466. doi: 10.1128/AAC.44.2.462-466.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim YB, Seo KW, Jeon HY, Lim SK, Lee YJ. Characteristics of the antimicrobial resistance of Staphylococcus aureus isolated from chicken meat produced by different integrated broiler operations in Korea. Poult Sci. 2018;97:962–969. doi: 10.3382/ps/pex357. [DOI] [PubMed] [Google Scholar]
- Ladhani S, Joannou CL, Lochrie DP, Evans RW, Poston SM. Clinical, microbial, and biochemical aspects of the exfoliative toxins causing staphylococcal scalded-skin syndrome. Clin Microbiol Rev. 1999;12:224–242. doi: 10.1128/CMR.12.2.224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee GY, Lee SI, Kim SD, Park JH, Kim GB, Yang SJ. Clonal distribution and antimicrobial resistance of methicillin-susceptible and -resistant Staphylococcus aureus strains isolated from broiler farms, slaughterhouses, and retail chicken meat. Poult Sci. 2022;101:102070. doi: 10.1016/j.psj.2022.102070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee GY, Yang SJ. Comparative assessment of genotypic and phenotypic correlates of Staphylococcus pseudintermedius strains isolated from dogs with otitis externa and healthy dogs. Comp Immunol Microbiol Infect Dis. 2020;70:101376. doi: 10.1016/j.cimid.2019.101376. [DOI] [PubMed] [Google Scholar]
- Lee SI, Kim SD, Park JH, Yang SJ. Species distribution, antimicrobial resistance, and enterotoxigenicity of non-aureus staphylococci in retail chicken meat. Antibiotics. 2020;9:809. doi: 10.3390/antibiotics9110809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim SK, Lee JE, Lee HS, Nam HM, Moon DC, Jang GC, Park YJ, Jung YG, Jung SC, Wee SH. Trends in antimicrobial sales for livestock and fisheries in Korea during 2003-2012. Korean J Vet Res. 2014;54:81–86. doi: 10.14405/kjvr.2014.54.2.81. [DOI] [Google Scholar]
- Mahato S, Mistry HU, Chakraborty S, Sharma P, Saravanan R, Bhandari V. Identification of variable traits among the methicillin resistant and sensitive coagulase negative staphylococci in milk samples from mastitic cows in India. Front Microbiol. 2017;8:1446. doi: 10.3389/fmicb.2017.01446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marek A, Stepień-Pyśniak D, Pyzik E, Adaszek Ł, Wilczyński J, Winiarczyk S. Occurrence and characterization of Staphylococcus bacteria isolated from poultry in Western Poland. Berl Munch Tierarztl Wochenschr. 2016;129:147–152. [PubMed] [Google Scholar]
- Martins PD, de Almeida TT, Basso AP, de Moura TM, Frazzon J, Tondo EC, Frazzon APG. Coagulase-positive staphylococci isolated from chicken meat: Pathogenic potential and vancomycin resistance. Foodborne Pathog Dis. 2013;10:771–776. doi: 10.1089/fpd.2013.1492. [DOI] [PubMed] [Google Scholar]
- Mekhloufi OA, Chieffi D, Hammoudi A, Bensefia SA, Fanelli F, Fusco V. Prevalence, enterotoxigenic potential and antimicrobial resistance of Staphylococcus aureus and methicillin-resistant Staphylococcus aureus (MRSA) isolated from algerian ready to eat foods. Toxins. 2021;13:835. doi: 10.3390/toxins13120835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mendoza M, Meugnier H, Bes M, Etienne J, Freney J. Identification of Staphylococcus species by 16S-23S rDNA intergenic spacer PCR analysis. Int J Syst Bacteriol. 1998;48:1049–1055. doi: 10.1099/00207713-48-3-1049. [DOI] [PubMed] [Google Scholar]
- Nadell CD, Xavier JB, Foster KR. The sociobiology of biofilms. FEMS Microbiol Rev. 2009;33:206–224. doi: 10.1111/j.1574-6976.2008.00150.x. [DOI] [PubMed] [Google Scholar]
- O’Gara JP, Humphreys H. Staphylococcus epidermidis biofilms: Importance and implications. J Med Microbiol. 2001;50:582–587. doi: 10.1099/0022-1317-50-7-582. [DOI] [PubMed] [Google Scholar]
- O’Neill AJ, McLaws F, Kahlmeter G, Henriksen AS, Chopra I. Genetic basis of resistance to fusidic acid in staphylococci. Antimicrob Agents Chemother. 2007;51:1737–1740. doi: 10.1128/AAC.01542-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osman K, Badr J, Al-Maary KS, Moussa IMI, Hessain AM, Girah ZMSA, Abo-shama UH, Orabi A, Saad A. Prevalence of the antibiotic resistance genes in coagulase-positive- and negative-Staphylococcus in chicken meat retailed to consumers. Front Microbiol. 2016;7:1846. doi: 10.3389/fmicb.2016.01846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pyzik E, Marek A, Stępień-Pyśniak D, Urban-Chmiel R, Jarosz LS, Jagiełło-Podębska I. Detection of antibiotic resistance and classical enterotoxin genes in coagulase-negative staphylococci isolated from poultry in Poland. J Vet Res. 2019;63:183–190. doi: 10.2478/jvetres-2019-0023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolo J, Worning P, Nielsen JB, Bowden R, Bouchami O, Damborg P, Guardabassi L, Perreten V, Tomasz A, Westh H, de Lencastre H, Miragaia M. Evolutionary origin of the staphylococcal cassette chromosome mec (SCCmec) Antimicrob Agents Chemother. 2017;61:e02302–e02316. doi: 10.1128/AAC.02302-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schilcher K, Horswill AR. Staphylococcal biofilm development: Structure, regulation, and treatment strategies. Microbiol Mol Biol Rev. 2020;84:e00026–19. doi: 10.1128/MMBR.00026-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen J, Wang Y, Schwarz S. Presence and dissemination of the multiresistance gene cfr in Gram-positive and Gram-negative bacteria. J Antimicrob Chemother. 2013;68:1697–1706. doi: 10.1093/jac/dkt092. [DOI] [PubMed] [Google Scholar]
- Silva V, Caniça M, Ferreira E, Vieira-Pinto M, Saraiva C, Pereira JE, Capelo JL, Igrejas G, Poeta P. Multidrug-resistant methicillin-resistant coagulase-negative staphylococci in healthy poultry slaughtered for human consumption. Antibiotics. 2022;11:365. doi: 10.3390/antibiotics11030365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sreedharan S, Peterson LR, Fisher LM. Ciprofloxacin resistance in coagulase-positive and -negative staphylococci: Role of mutations at serine 84 in the DNA gyrase A protein of Staphylococcus aureus and Staphylococcus epidermidis. Antimicrob Agents Chemother. 1991;35:2151–2154. doi: 10.1128/AAC.35.10.2151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szafraniec GM, Szeleszczuk P, Dolka B. A review of current knowledge on Staphylococcus agnetis in poultry. Animals. 2020;10:1421. doi: 10.3390/ani10081421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi H, Kikuchi T, Shoji S, Fujimura S, Lutfor AB, Tokue Y, Nukiwa T, Watanabe A. Characterization of gyrA, gyrB, grlA and grlB mutations in fluoroquinolone-resistant clinical isolates of Staphylococcus aureus. J Antimicrob Chemother. 1998;41:49–57. doi: 10.1093/jac/41.1.49. [DOI] [PubMed] [Google Scholar]
- Takahata M, Yonezawa M, Matsubara N, Watanabe Y, Narita H, Matsunaga T, Igarashi H, Kawahara M, Onodera S, Oishi Y. Antibacterial activity of quinolones against coagulase-negative staphylococci and the quinolone resistance-determining region of the gyrA genes from six species. J Antimicrob Chemother. 1997;40:383–386. doi: 10.1093/jac/40.3.383. [DOI] [PubMed] [Google Scholar]
- Yang YJ, Lee GY, Kim SD, Park JH, Lee SI, Kim GB, Yang SJ. Profiles of non-aureus staphylococci in retail pork and slaughterhouse carcasses: Prevalence, antimicrobial resistance, and genetic determinant of fusidic acid resistance. Food Sci Anim Resour. 2022;42:225–239. doi: 10.5851/kosfa.2021.e74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yurdakul NE, Erginkaya Z, Ünal E. Antibiotic resistance of enterococci, coagulase negative staphylococci and Staphylococcus aureus isolated from chicken meat. Czech J Food Sci. 2013;31:14–19. doi: 10.17221/58/2012-CJFS. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Materials