Abstract
Background
Atherogenic index of plasma (AIP), a marker of atherosclerosis and cardiovascular disease (CVD), was related to the all-cause mortality and CVD-specific mortality in a U-shape in general population respectively. However, no studies have investigated these associations in hypertensive populations. Herein, this study aims to explore the relationship of AIP and all-cause mortality and CVD-specific mortality in patients with hypertension in order to provide some reference for the risk hierarchical management of hypertension.
Methods
Demographic and clinical data of 17,382 adult patients with hypertension were extracted from the National Health and Nutrition Examination Survey (NHANES) database in 2005–2018 in this retrospective cohort study. We used weighted univariate COX regression analysis to screen the covariates, and that weighted univariate and multivariate COX regression analyses to explore the association between AIP and all-cause mortality and CVD-specific mortality with hazard ratios (HRs) and 95% confidence intervals (CIs). Subgroup analyses of age, gender, body mass index (BMI), CVD, diabetes mellitus (DM), antihyperlipidemic agents use, and hypotensive drugs use were also performed for further exploration of these relationships.
Results
The average follow-up time was 97.10 months. A total of 2,844 patients died, and 971 of them died due to CVD. After adjusting for age, race, education level, marital status, poverty-income ratio (PIR), smoking, BMI, physical activity, antihyperlipidemic agents, DM, CVD, hypotensive drugs, estimated glomerular filtration rate (eGFR), and total energy intake, we found that both low [HR = 1.18, 95%CI: (1.07–1.32)] and high [HR = 1.17, 95%CI: (1.03–1.33)] levels of AIP were linked to an increased risk of all-cause mortality, and the U-shape association between AIP and CVD-specific mortality was also found [low AIP level: HR = 1.26, 95%CI: (1.05–1.51); high AIP level: HR = 1.26, 95%CI: (1.06–1.49)]. Furthermore, these relationships were existed in patients whose BMI > 25, were non-Hispanic White, with non-CVD, non-DM, non-antihyperlipidemic agents, and used hypertension drug (all P < 0.05).
Conclusion
AIP was associated with both all-cause mortality and CVD-specific mortality in patients with hypertension, but the specific role of AIP in prognosis in hypertensive populations is needed further exploration.
Keywords: AIP, All-cause mortality, CVD-specific mortality, NHANES
Background
Hypertension is a chronic disease with high prevalence and is the leading preventable risk factor for cardiovascular diseases (CVD) [1, 2]. Globally, more than one billion people are considered hypertensive and the prevalence is expected to reach 29.2% by 2025 [3, 4]. Comparing to the general population, patients with hypertension have increased risks of all-cause mortality [5]. The prevention and management of hypertension has become a major public health challenge.
In addition to hypertension, dyslipidemia is also a risk factor for CVD, and is associated with the risk of hypertension occurrence [4]. Atherogenic index of plasma (AIP), namely total cholesterol (TC) and high-density lipoprotein cholesterol (HDL-C) ratio, is a common indicator of lipid metabolism and a marker of atherosclerosis and CVD [6]. A 10-year prospective cohort study by Dong et al. [7] on the relationships of lipid profiles with CVD and mortality found a U-shape relationship between AIP and the CVD-specific mortality in a community-dwelling population in China. Another study explored the effect of AIP on mortality risk in the general population, the restricted cubic spline (RCS) analysis showed that a U-shape association between AIP and all-cause mortality but the non-linear association between AIP and CVD-specific mortality was not significant [8].
AIP is a novel predictive biomarker for cardiovascular illnesses, and is also a robust biomarker of dyslipidemia and atherosclerosis, has been used to quantify comprehensive lipid levels [9]. Studies have indicated it could be recommended as a potential biomarker in the early diagnosis of CVD events [9, 10]. Nevertheless, no studies have investigated the association of AIP with all-cause and CVD-specific mortality in hypertensive populations. Considering the role of lipid metabolism in hypertension and CVD and the poor prognosis of hypertension, a mature and convenient biomarker for the prognosis of patients with hypertension is in need clinically.
Herein, this study aims to explore the relationship between AIP and all-cause mortality and CVD-specific mortality in patients with hypertension, and further assess these relationships in subgroups of age, gender, body mass index (BMI), and complications. We hope this study can help to provide some references for the risk hierarchical management in hypertensive populations.
Methods
Study design and population
Data in this retrospective cohort study were extracted from the National Health and Nutrition Examination Survey (NHANES) database in 2005–2018. NHANES is a multipurpose research program done by the National Center for Health Statistics (NCHS) that assesses the health and nutritional status of adults and children in the United States [11]. The survey regularly collects data of approximately 5,000 persons from 15 areas since 1999 (datasets of every two years were incorporated into a cycle) that includes a household interview followed by a standardized physical examination in a mobile examination center (MEC). A stratified multistage sampling design with a weighting scheme based on the selection of counties, blocks, households, and persons within households is used by NHANES to represent the civilian, non-institutionalized population in the United States and accurately estimate disease prevalence. Details of study implementation are available for online access: https://wwwn.cdc.gov/nchs/nhanes/tutorials/module2.aspx.
A total of 22,628 patients with hypertension were initially included. The exclusion criteria were (1) aged < 18 years old, and (2) missing information of serum TC or HDL or mortality. We also excluded individuals who missing information of study variables including education level, marital status, physical activity, Dietary Approaches to Stop Hypertension (DASH) score, smoking, BMI, and CVD. After excluding patients who lost to the follow-up, finally, 17,382 of them were eligible.
All methods for this study were performed in accordance with the relevant guidelines and regulations. Written informed consent was obtained from participants by NHANES. Since NHANES is publicly available, and the data of patients were de-identified, the requirement of ethical approval for this study was waived by the Institutional Review Board of People’s Hospital of Xinjiang Uygur Autonomous Region.
Definition of atherogenic index of plasma
Blood sample collection and measurement in NHAENS were conducted according to a standardized protocol from the Centers for Disease Control and Prevention (CDC). Serum HDL-C was measured by direct immunoassay or precipitation [12]. Also, serum TC and HDL-C levels were measured enzymatically with a Hitachi 704 Analyzer (Boehringer Mannheim Diagnostics, Indianapolis, IN, USA) [13]. The AIP was calculated by the serum TC (mmol/L) and HDL-C (mmol/L) ratio. More information of laboratory examination was provided on the website of CDC. We divided the AIP into three levels according to the tertiles in this study, including low level: AIP < 3.25, median level: 3.25 ≤ AIP ≤ 4.34, and high level: AIP > 4.34.
Variables collection
We extracted variables including age, gender, race, marital status, education level, poverty income ratio (PIR), smoking and drinking status, physical activity, BMI, diabetes mellitus (DM), CVD, antihyperlipidemic agents, hypotensive drugs, estimated glomerular filtration rate (eGFR), DASH score, total energy intake, and survival time from the NHANES database.
Physical activity was translated into energy expenditure. Metabolic equivalent (MET) was calculated based on the questionnaire of physical activity reports (PAQ) in NHANES using the following formula: Energy expenditure (MET·min) = recommended MET × exercise time of corresponding activity (min). Complications including DM, CVD, and dyslipidemia of individuals were diagnosed according to laboratory examination, self-reports and the medication history. DM was defined as fasting blood glucose ≥ 7.0 mmol/L or Glycosylated Hemoglobin (HbAlc) ≥ 6.5% or self-reported DM or receiving hypoglycemic therapy. CVD diagnosis was according to a positive answer to the NHANES multiple choice question (MCQ): “Have you ever been told you had (congestive) heart failure, coronary heart disease, angina/angina pectoris, heart attack and stroke " or using of cardiovascular drugs. The information of antihyperlipidemic agents was from self-reported, or the records of prescription medications-drug information (serial number 358 − 19). The eGFR was calculated using the following formula: estimated GFR = 175 × standardized serum creatinine (Scr) − 1.154 × age − 0.203 × 1.212 (if black) × 0.742 (if female), where GFR is expressed as mL/min/1.73 m2 of body surface area and Scr is expressed in mg/dL [14]. The DASH dietary score was calculated based on the nine-item, nutrient-based DASH index: protein, fiber, magnesium, calcium, potassium, total fat, saturated fat, cholesterol, and sodium. The optimal micronutrient targets are energy adjusted per 1000 kcal. Meeting the goal for each component provides one point, meeting an intermediate goal between the DASH diet goal and the nutrient content of the DASH control diet provides 0.5 points, and meeting neither goal gives zero points. In addition, this pattern score ranges between 0 and 9 points, and higher scores indicates greater adherence to the DASH dietary pattern [15]. The total energy intake was calculated according to the two 24-hour dietary recalls in the NHANES. The first 24-hour recall interview was conducted in person in the mobile exam centers (MEC) by trained interviewers, and the second interview was performed by telephone or mail three to ten days later. We used the records of first 24-hour recall in this study.
Outcome and follow-up
The study outcomes were all-cause mortality and CVD-specific mortality. The information of mortality in the NHANES in 2005–2018 were linked to mortality data from the National Death Index death certificate records (www.cdc.gov/nchs/data-linkage/mortality-public.htm) until December 31, 2019, matched using a probabilistic matching algorithm to identify mortality status [16]. CVD-specific mortality (codes I00–I09, I11, I13, I20–I51, and I60–I69) was estimated using the International Classification of Disease Tenth Revision (ICD-10). The follow-up ended if the patients died or until the 31st December 2019.
Statistical analysis
Continuous data were described by mean ± standard error (mean ± SE), and weighted t test was used for comparation between groups. Enumeration data were expressed as number with constituent ratio [N (%)], and chi-square test for the comparison. Since we merged seven cycles of NHANES datasets, and to enable more excellent statistical reliability, the MEC sample weights (WTMEC2YR/7) were used for weighted analyses (https://wwwn.cdc.gov/nchs/nhanes/tutorials/default.aspx).
Weighted univariate COX regression analyses were used to screen the covariates that related to all-cause and CVD-specific mortality respectively. Weighted univariate and multivariate COX regression analyses were used to explore the association between AIP and all-cause and CVD-specific mortality. Restricted cubic spline (RCS) curves were used to show the relationships between AIP and the two outcomes. Model 1 adjusted for age (years) and race. Model 2 adjusted for age, race, education level, marital status, PIR, smoking, BMI (kg/m2), physical activity, antihyperlipidemic agents, DM, CVD, hypotensive drugs, eGFR [mL/ (min·1.73 m2)], and energy intake (kcal). We also explored these relationships in age, gender, BMI, CVD, DM, antihyperlipidemic agents use, and hypotensive drugs use subgroups.
The evaluation indexes were hazard ratios (HRs) and 95% confidence intervals (CIs). Two-sided P < 0.05 was considered significant. Statistical analysis was performed using SAS 9.4 (SAS Institute, Cary, NC, USA) and R version 4.2.0 (Institute for Statistics and Mathematics, Vienna, Austria). Missing data were deleted.
Results
Characteristics of hypertension patients
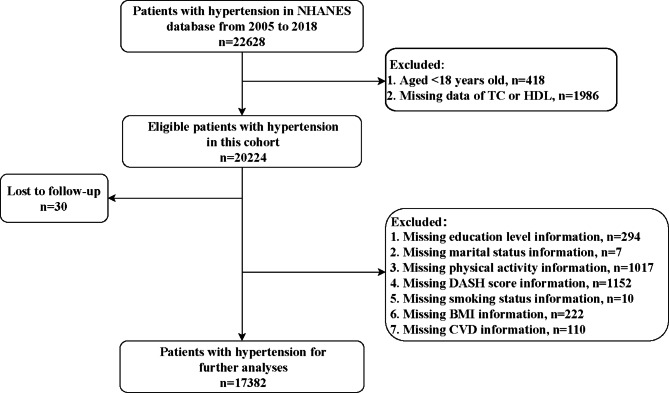
Figure 1 is the flow chart of the participants screening. A total of 22,628 patients with hypertension were initially included. Then we excluded those who aged < 18 years old (n = 418), and without information of serum TC or HDL (n = 1986). In the eligible patients, 30 of them lost to the follow-up. Those who missing information of education level (n = 294), marital status (n = 7), physical activity (n = 1017), DASH score (n = 1152), smoking status (n = 10), BMI (n = 222), and CVD (n = 110) were also excluded. Finally, 17,382 of hypertension patients were included for further analyses.
Fig. 1.
Flow chart of the participants screening
Table 1 shows the characteristics of patients with hypertension. The average follow-up time was approximately 97 months. A total of 2,844 patients died, and 971 of them died due to CVD. Among the patients, 9,050 (52.05%) were males and 8,332 (47.95%) were females. Most of them were non-Hispanic White (70.55%), BMI > 25 (79.98%), and not have CVD (85.37%). Additionally, age, marital status, education level, PIR, antihyperlipidemic agents use, hypotensive drugs use, eGFR, DASH score, and total energy intake were all different among the three AIP level groups (all P < 0.05).
Table 1.
Characteristics of patients with hypertension
| Variables | Total (n = 17,382) | AIP levels | P | ||
|---|---|---|---|---|---|
| < 3.25 (n = 5736) |
3.25–4.34 (n = 5732) | > 4.34 (n = 5914) |
|||
| Age, years, n (%) | < 0.001 | ||||
| <40 | 2798 (18.54) | 769 (15.59) | 841 (17.00) | 1188 (22.73) | |
| 40–60 | 5937 (40.83) | 1601 (33.86) | 1934 (41.21) | 2402 (46.90) | |
| ≥60 | 8647 (40.63) | 3366 (50.55) | 2957 (41.79) | 2324 (30.37) | |
| Gender, n (%) | < 0.001 | ||||
| Male | 9050 (52.05) | 2449 (40.79) | 2887 (49.63) | 3714 (64.74) | |
| Female | 8332 (47.95) | 3287 (59.21) | 2845 (50.37) | 2200 (35.26) | |
| Race, n (%) | < 0.001 | ||||
| Mexican American | 2250 (6.34) | 551 (4.41) | 766 (6.35) | 933 (8.12) | |
| Other Hispanic | 1568 (4.52) | 404 (3.28) | 525 (4.40) | 639 (5.79) | |
| Non-Hispanic White | 7801 (70.55) | 2526 (70.60) | 2542 (70.77) | 2733 (70.30) | |
| Non-Hispanic Black | 4153 (12.04) | 1724 (15.31) | 1381 (12.02) | 1048 (9.04) | |
| Other race-including multi-racial | 1610 (6.54) | 531 (6.40) | 518 (6.46) | 561 (6.75) | |
| Marital status, n (%) | < 0.001 | ||||
| Married | 9380 (58.82) | 2841 (55.56) | 3199 (60.60) | 3340 (60.13) | |
| Widowed | 2015 (8.86) | 874 (12.04) | 648 (8.91) | 493 (5.87) | |
| Divorced | 2259 (11.98) | 751 (12.04) | 729 (11.42) | 779 (12.47) | |
| Separated | 610 (2.50) | 217 (2.69) | 200 (2.76) | 193 (2.08) | |
| Never married | 2076 (11.64) | 712 (11.69) | 651 (11.00) | 713 (12.20) | |
| Living with partner | 1042 (6.20) | 341 (5.98) | 305 (5.31) | 396 (7.24) | |
| Education level, n (%) | < 0.001 | ||||
| Less Than 9th Grade | 1975 (5.66) | 576 (5.05) | 677 (5.52) | 722 (6.37) | |
| 9-11th Grade | 2464 (10.55) | 791 (10.01) | 810 (10.64) | 863 (10.97) | |
| High School Grad/GED or Equivalent | 4197 (24.61) | 1367 (23.33) | 1363 (24.58) | 1467 (25.81) | |
| Some College or AA degree | 5134 (32.33) | 1668 (31.10) | 1712 (33.00) | 1754 (32.82) | |
| College Graduate or above | 3612 (26.85) | 1334 (30.52) | 1170 (26.27) | 1108 (24.03) | |
| PIR, n (%) | 0.002 | ||||
| ≤1 | 2989 (11.10) | 919 (10.33) | 976 (10.56) | 1094 (12.32) | |
| >1 | 12,806 (81.48) | 4267 (81.33) | 4245 (82.19) | 4294 (80.95) | |
| Unknown | 1587 (7.42) | 550 (8.35) | 511 (7.25) | 526 (6.73) | |
| Smoking, n (%) | 0.230 | ||||
| Yes | 8365 (48.20) | 2696 (47.66) | 2729 (47.57) | 2940 (49.30) | |
| No | 9017 (51.80) | 3040 (52.34) | 3003 (52.43) | 2974 (50.70) | |
| Drinking, n (%) | 0.654 | ||||
| Yes | 16,417 (95.42) | 5393 (95.15) | 5419 (95.54) | 5605 (95.55) | |
| No | 965 (4.58) | 343 (4.85) | 313 (4.46) | 309 (4.45) | |
| Physical activity, MET*min/week, n (%) | 0.761 | ||||
| ≤750 | 7190 (36.99) | 2365 (36.61) | 2407 (36.84) | 2418 (37.47) | |
| >750 | 10,192 (63.01) | 3371 (63.39) | 3325 (63.16) | 3496 (62.53) | |
| BMI, kg/m2, n (%) | < 0.001 | ||||
| ≤25 | 3588 (20.02) | 1896 (34.17) | 1037 (17.61) | 655 (9.27) | |
| >25 | 13,794 (79.98) | 3840 (65.83) | 4695 (82.39) | 5259 (90.73) | |
| DM, n (%) | 0.064 | ||||
| No | 12,746 (78.72) | 4266 (80.15) | 4173 (78.38) | 4307 (77.74) | |
| Yes | 4636 (21.28) | 1470 (19.85) | 1559 (21.62) | 1607 (22.26) | |
| CVD, n (%) | < 0.001 | ||||
| No | 14,355 (85.37) | 4579 (82.76) | 4736 (85.75) | 5040 (87.40) | |
| Yes | 3027 (14.63) | 1157 (17.24) | 996 (14.25) | 874 (12.60) | |
| Antihyperlipidemic agents, n (%) | < 0.001 | ||||
| No | 11,310 (67.79) | 3247 (59.38) | 3670 (66.51) | 4393 (76.77) | |
| Yes | 6072 (32.21) | 2489 (40.62) | 2062 (33.49) | 1521 (23.23) | |
| Hypotensive drugs, n (%) | < 0.001 | ||||
| No | 6300 (39.88) | 1801 (35.27) | 1974 (38.15) | 2525 (45.78) | |
| Yes | 11,082 (60.12) | 3935 (64.73) | 3758 (61.85) | 3389 (54.22) | |
| eGFR, mL/ (min·1.73 m2), Mean (S.E) | 92.19 (0.32) | 90.94 (0.48) | 92.15 (0.42) | 93.38 (0.42) | < 0.001 |
| DASH score, Mean (S.E) | 2.18 (0.02) | 2.31 (0.03) | 2.15 (0.03) | 2.09 (0.03) | < 0.001 |
| Energy intake, kcal, Mean (S.E) | 2073.96 (9.50) | 1972.78 (12.82) | 2052.93 (14.51) | 2187.30 (17.84) | < 0.001 |
| Survival time, months, Mean (S.E) | 97.10 (1.57) | 91.93 (1.91) | 96.70 (2.17) | 102.27 (1.97) | < 0.001 |
| Mortality, n (%) | < 0.001 | ||||
| Survival | 14,538 (86.36) | 4666 (84.12) | 4857 (87.96) | 5015 (86.90) | |
| CVD-specific mortality | 971 (4.65) | 364 (5.51) | 294 (3.94) | 313 (4.54) | |
| Other-cause mortality | 1873 (8.99) | 706 (10.37) | 581 (8.10) | 586 (8.56) | |
AIP: atherogenic index of plasma, PIR: poverty income ratio, MET: metabolic equivalent, BMI: body mass index, DM: diabetes mellitus, CVD: cardiovascular disease, eGFR: estimated glomerular filtration rate, DASH: Dietary Approaches to Stop Hypertension, S.E: standard error
Association between AIP and all-cause and CVD-specific mortality
Table 2 shows the covariates that associated with all-cause mortality and CVD-specific mortality respectively. Age, race, education level, marital status, PIR, smoking, BMI, physical activity, antihyperlipidemic agents, DM, CVD, hypotensive drugs, eGFR, and total energy intake were all significantly associated with both all-cause mortality and CVD-specific mortality, and they were further included in the adjusted model.
Table 2.
Screening of the covariates related to all-cause and CVD-specific mortality
| Variables | All-cause mortality | CVD-specific mortality | ||
|---|---|---|---|---|
| HR (95% CI) | P | HR (95% CI) | P | |
| Age | ||||
| <40 | Ref | Ref | ||
| 40–60 | 1.52 (1.23–1.87) | < 0.001 | 1.44 (1.03–2.01) | 0.032 |
| ≥60 | 4.22 (3.52–5.07) | < 0.001 | 4.32 (3.14–5.95) | < 0.001 |
| Gender | ||||
| Male | Ref | Ref | ||
| Female | 0.97 (0.88–1.07) | 0.582 | 0.90 (0.76–1.07) | 0.242 |
| Race | ||||
| Mexican American | Ref | Ref | ||
| Other Hispanic | 1.01 (0.78–1.30) | 0.945 | 1.17 (0.79–1.72) | 0.435 |
| Non-Hispanic White | 1.61 (1.37–1.90) | < 0.001 | 1.57 (1.21–2.03) | < 0.001 |
| Non-Hispanic Black | 1.36 (1.14–1.61) | < 0.001 | 1.39 (1.05–1.84) | 0.023 |
| Other race - including multi-racial | 1.13 (0.89–1.43) | 0.314 | 1.39 (0.97–1.99) | 0.074 |
| Education level | ||||
| Less Than 9th Grade | Ref | Ref | ||
| 9-11th Grade | 0.72 (0.61–0.86) | < 0.001 | 0.59 (0.45–0.78) | < 0.001 |
| High School Grad/GED or Equivalent | 0.68 (0.58–0.79) | < 0.001 | 0.67 (0.51–0.87) | 0.003 |
| Some College or AA degree | 0.53 (0.44–0.63) | < 0.001 | 0.52 (0.40–0.67) | < 0.001 |
| College Graduate or above | 0.44 (0.36–0.54) | < 0.001 | 0.46 (0.34–0.61) | < 0.001 |
| Marital status | ||||
| Married | Ref | Ref | ||
| Widowed | 3.08 (2.76–3.43) | < 0.001 | 3.19 (2.60–3.91) | < 0.001 |
| Divorced | 1.23 (1.05–1.45) | 0.012 | 1.09 (0.83–1.43) | 0.549 |
| Separated | 1.14 (0.90–1.46) | 0.284 | 1.37 (0.90–2.09) | 0.143 |
| Never married | 0.78 (0.64–0.95) | 0.015 | 0.79 (0.61–1.04) | 0.095 |
| Living with partner | 0.75 (0.58–0.96) | 0.022 | 0.44 (0.27–0.71) | < 0.001 |
| PIR | ||||
| ≤1 | Ref | Ref | ||
| >1 | 0.76 (0.66–0.88) | < 0.001 | 0.81 (0.64–1.04) | 0.093 |
| Unknown | 0.78 (0.65–0.95) | 0.011 | 0.55 (0.38–0.79) | 0.001 |
| Smoking | ||||
| Yes | Ref | Ref | ||
| No | 0.68 (0.62–0.75) | < 0.001 | 0.82 (0.70–0.95) | 0.008 |
| Drinking | ||||
| Yes | Ref | Ref | ||
| No | 1.01 (0.80–1.27) | 0.958 | 0.76 (0.48–1.22) | 0.255 |
| BMI | ||||
| ≤25 | Ref | Ref | ||
| >25 | 0.74 (0.66–0.83) | < 0.001 | 0.78 (0.66–0.92) | 0.003 |
| Physical activity | ||||
| ≤750 | Ref | Ref | ||
| >750 | 0.55 (0.50–0.61) | < 0.001 | 0.54 (0.45–0.65) | < 0.001 |
| Antihyperlipidemic agents | ||||
| No | Ref | Ref | ||
| Yes | 1.77 (1.63–1.92) | < 0.001 | 1.99 (1.72–2.31) | < 0.001 |
| DM | ||||
| No | Ref | Ref | ||
| Yes | 1.86 (1.70–2.05) | < 0.001 | 1.90 (1.61–2.23) | < 0.001 |
| CVD | ||||
| No | Ref | Ref | ||
| Yes | 2.81 (2.52–3.14) | < 0.001 | 3.27 (2.68–3.98) | < 0.001 |
| Hypertension treatment | ||||
| No | Ref | Ref | ||
| Yes | 2.04 (1.82–2.28) | < 0.001 | 2.25 (1.86–2.73) | < 0.001 |
| eGFR | 0.56 (0.53–0.58) | < 0.001 | 0.53 (0.48–0.58) | < 0.001 |
| DASH score | 1.01 (0.97–1.06) | 0.643 | 1.05 (0.97–1.14) | 0.267 |
| Energy intake | 0.84 (0.79–0.89) | < 0.001 | 0.84 (0.76–0.92) | < 0.001 |
CVD: cardiovascular disease, HR: hazard ratio, CI: confidence interval, Ref: reference, PIR: poverty income ratio, BMI: body mass index, DM: diabetes mellitus, eGFR: estimated glomerular filtration rate, DASH: Dietary Approaches to Stop Hypertension
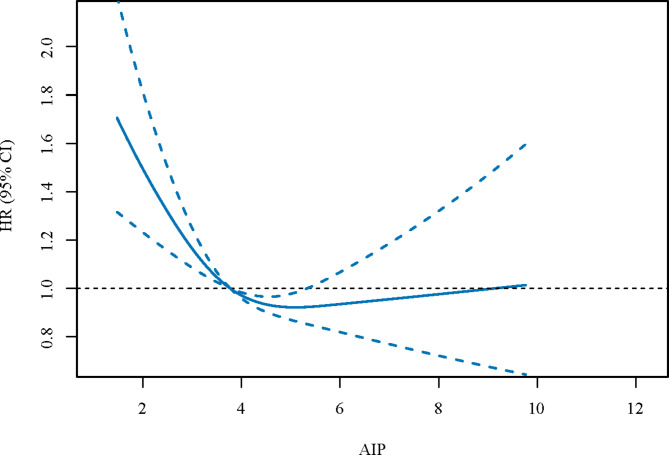
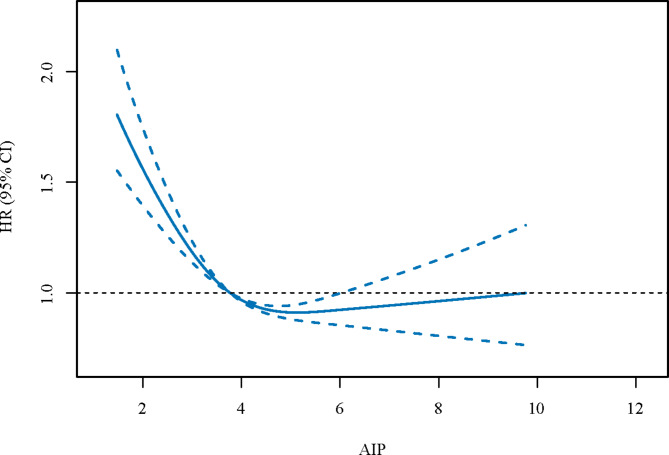
We then explored the relationships between AIP and all-cause mortality, and between AIP and CVD-specific mortality (Table 3). After adjusting for the covariables, we found that both low [HR = 1.18, 95%CI: (1.07–1.32)] and high [HR = 1.17, 95%CI: (1.03–1.33)] levels of AIP were linked to an increased risk of all-cause mortality. Similarly, the U-shape association between AIP and CVD-specific mortality was also found [low AIP level: HR = 1.26, 95%CI: (1.05–1.51); high AIP level: HR = 1.26, 95%CI: (1.06–1.49)]. Figures 2 and 3 were the RCS curves of AIP and all-cause mortality, and AIP and CVD-specific mortality respectively.
Table 3.
Association between AIP and all-cause and CVD-specific mortality
| AIP levels | Crude model | Model 1 | Model 2 | |||
|---|---|---|---|---|---|---|
| h (95% CI) | P | HR (95% CI) | P | HR (95% CI) | P | |
| All-cause mortality | ||||||
| 3.25–4.34 | Ref | Ref | Ref | |||
| < 3.25 | 1.40 (1.26–1.56) | < 0.001 | 1.23 (1.12–1.37) | < 0.001 | 1.18 (1.07–1.32) | 0.002 |
| > 4.34 | 1.02 (0.89–1.17) | 0.764 | 1.24 (1.08–1.41) | 0.002 | 1.17 (1.03–1.33) | 0.016 |
| CVD-specific mortality | ||||||
| 3.25–4.34 | Ref | Ref | Ref | |||
| < 3.25 | 1.49 (1.23–1.80) | < 0.001 | 1.31 (1.09–1.56) | 0.004 | 1.26 (1.05–1.51) | 0.015 |
| > 4.34 | 1.08 (0.90–1.29) | 0.403 | 1.31 (1.10–1.57) | 0.003 | 1.26 (1.06–1.49) | 0.009 |
AIP: atherogenic index of plasma, CVD: cardiovascular disease, HR: hazard ratio, CI: confidence interval, Ref: reference
Model 1: adjusted for age and gender;
Model 2: adjusted for age, race, education level, marital status, PIR, smoking, BMI, physical activity, antihyperlipidemic agents, DM, CVD, hypotensive drugs, eGFR, and energy intake
Fig. 2.
The RCS curves of AIP and all-cause mortality
Fig. 3.
The RCS curves of AIP and CVD-specific mortality
Relationships of AIP and all-cause mortality and CVD-specific mortality in different subgroups
We further explored these associations in different subgroups (Table 4). After adjusting for covariates, the U-shape relationships between AIP and all-cause mortality, and between AIP and CVD-specific mortality were also found in patients with hypertension whose BMI > 25, were non-Hispanic White, with non-CVD, non-DM, and non-antihyperlipidemic agents, and used hypertension drug (all P < 0.05). Differently, the U-shape association between AIP level and all-cause mortality was found in female patients (HR = 1.16 for low level and HR = 1.20 for high level), while the U-shape association between AIP level and of CVD-specific mortality was found in patients aged ≥ 60 years old (HR = 1.28 for low level and HR = 1.36 for high level).
Table 4.
Relationships of AIP and all-cause and CVD-specific mortality in age, gender, race, BMI, CVD, DM, antihyperlipidemic agents use and hypotensive drugs use subgroups
| Subgroups | AIP levels | All-cause mortality | CVD-specific mortality | ||
|---|---|---|---|---|---|
| HR (95% CI) | P | HR (95% CI) | P | ||
| Age < 40 | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.55 (0.88–2.73) | 0.129 | 0.98 (0.44–2.20) | 0.967 | |
| > 4.34 | 1.37 (0.85–2.21) | 0.190 | 0.70 (0.33–1.49) | 0.355 | |
| 40 ≤ Age < 60 | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.30 (0.98–1.72) | 0.068 | 1.19 (0.74–1.90) | 0.472 | |
| > 4.34 | 1.45 (1.11–1.88) | 0.006 | 1.21 (0.80–1.81) | 0.363 | |
| Age ≥ 60 | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.10 (0.98–1.23) | 0.121 | 1.28 (1.05–1.57) | 0.016 | |
| > 4.34 | 1.05 (0.91–1.21) | 0.486 | 1.36 (1.11–1.68) | 0.004 | |
| Male | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.27 (1.10–1.48) | 0.001 | 1.37 (1.06–1.77) | 0.015 | |
| > 4.34 | 1.12 (0.94–1.32) | 0.196 | 1.17 (0.90–1.53) | 0.232 | |
| Female | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.16 (1.00-1.34) | 0.044 | 1.23 (0.95–1.59) | 0.112 | |
| > 4.34 | 1.20 (1.00-1.44) | 0.046 | 1.29 (1.03–1.63) | 0.028 | |
| BMI ≤ 25 | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.05 (0.87–1.27) | 0.627 | 1.03 (0.72–1.47) | 0.88 | |
| > 4.34 | 0.99 (0.76–1.30) | 0.962 | 1.11 (0.73–1.68) | 0.633 | |
| BMI > 25 | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.26 (1.09–1.45) | 0.001 | 1.40 (1.10–1.78) | 0.007 | |
| > 4.34 | 1.22 (1.04–1.43) | 0.013 | 1.31 (1.05–1.63) | 0.017 | |
| Mexican American | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.39 (0.94–2.05) | 0.097 | 1.16 (0.66–2.03) | 0.611 | |
| > 4.34 | 1.18 (0.84–1.66) | 0.345 | 1.20 (0.69–2.06) | 0.516 | |
| Other Hispanic | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.02 (0.66–1.56) | 0.930 | 1.01 (0.42–2.45) | 0.977 | |
| > 4.34 | 0.67 (0.42–1.08) | 0.100 | 0.63 (0.30–1.31) | 0.214 | |
| Non-Hispanic White | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.22 (1.06–1.41) | 0.005 | 1.34 (1.06–1.69) | 0.013 | |
| > 4.34 | 1.18 (1.00-1.39) | 0.048 | 1.28 (1.03–1.60) | 0.024 | |
| Non-Hispanic Black | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.11 (0.92–1.33) | 0.284 | 1.25 (0.91–1.71) | 0.174 | |
| > 4.34 | 1.38 (1.08–1.77) | 0.010 | 1.36 (0.93-2.00) | 0.110 | |
| Other race-including multi-racial | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 0.72 (0.38–1.36) | 0.312 | 0.75 (0.25–2.25) | 0.608 | |
| > 4.34 | 1.01 (0.61–1.67) | 0.958 | 1.36 (0.53–3.45) | 0.521 | |
| Non-CVD | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.29 (1.14–1.46) | < 0.001 | 1.33 (1.04–1.70) | 0.026 | |
| > 4.34 | 1.25 (1.07–1.46) | 0.005 | 1.29 (1.03–1.61) | 0.026 | |
| CVD | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 0.99 (0.81–1.20) | 0.923 | 1.15 (0.92–1.43) | 0.226 | |
| > 4.34 | 0.94 (0.79–1.13) | 0.512 | 1.10 (0.80–1.50) | 0.573 | |
| Non-DM | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.26 (1.11–1.44) | < 0.001 | 1.26 (1.02–1.57) | 0.033 | |
| > 4.34 | 1.19 (1.05–1.35) | 0.005 | 1.27 (1.05–1.55) | 0.015 | |
| DM | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.01 (0.83–1.22) | 0.937 | 1.24 (0.92–1.67) | 0.163 | |
| > 4.34 | 1.09 (0.82–1.46) | 0.534 | 1.20 (0.84–1.72) | 0.319 | |
| Non-antihyperlipidemic agents | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.38 (1.19–1.60) | < 0.001 | 1.37 (1.04–1.81) | 0.026 | |
| > 4.34 | 1.20 (1.00-1.45) | 0.049 | 1.38 (1.12–1.70) | 0.003 | |
| Antihyperlipidemic agents use | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.01 (0.87–1.19) | 0.864 | 1.13 (0.89–1.45) | 0.305 | |
| > 4.34 | 1.18 (0.97–1.44) | 0.095 | 1.14 (0.83–1.56) | 0.430 | |
| Non-hypertension drug | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.22 (0.92–1.61) | 0.159 | 1.05 (0.64–1.71) | 0.854 | |
| > 4.34 | 1.10 (0.85–1.42) | 0.465 | 0.88 (0.56–1.39) | 0.586 | |
| Hypertension drug use | 3.25–4.34 | Ref | Ref | ||
| < 3.25 | 1.18 (1.05–1.32) | 0.007 | 1.34 (1.11–1.61) | 0.002 | |
| > 4.34 | 1.21 (1.04–1.40) | 0.012 | 1.41 (1.17–1.70) | < 0.001 | |
AIP: atherogenic index of plasma, CVD: cardiovascular disease, BMI: body mass index, DM: diabetes mellitus, HR: hazard ratio, CI: confidence interval, Ref: reference
Age subgroup: adjusted for race, education level, marital status, PIR, smoking, BMI, physical activity, antihyperlipidemic agents, DM, CVD, hypotensive drugs, eGFR, and energy intake;
Gender subgroup: adjusted for age, race, education level, marital status, PIR, smoking, BMI, physical activity, antihyperlipidemic agents, DM, CVD, hypotensive drugs, eGFR, and energy intake;
BMI subgroups: adjusted for age, race, education level, marital status, PIR, smoking, physical activity, antihyperlipidemic agents, DM, CVD, hypotensive drugs, eGFR, and energy intake;
Race subgroup: adjusted for age, education level, marital status, PIR, smoking, BMI, physical activity, antihyperlipidemic agents, DM, CVD, hypotensive drugs, eGFR, and energy intake;
CVD subgroup: adjusted for age, race, education level, marital status, PIR, smoking, BMI, physical activity, antihyperlipidemic agents, DM, hypotensive drugs, eGFR, and energy intake;
DM subgroup: adjusted for age, race, education level, marital status, PIR, smoking, BMI, physical activity, antihyperlipidemic agents, CVD, hypotensive drugs, eGFR, and energy intake;
Antihyperlipidemic agents use subgroup: adjusted for age, race, education level, marital status, PIR, smoking, BMI, physical activity, DM, CVD, hypotensive drugs, eGFR, and energy intake;
Hypertension drug use subgroup: adjusted for age, race, education level, marital status, PIR, smoking, BMI, physical activity, antihyperlipidemic agents, DM, CVD, eGFR, and energy intake
Discussion
In this study, we explored the association between AIP and all-cause mortality and CVD-specific mortality in patients with hypertension. The results showed that after adjusting for the covariates, AIP level was related to both all-cause mortality and CVD-specific mortality in a U-shape. These relationships were also found in patients whose BMI > 25, were non-Hispanic White, with non-CVD, non-DM, and non-antihyperlipidemic agents, and used hypertension drug.
To our knowledge, no study has explored the association between AIP and all-cause mortality and CVD-specific mortality in hypertensive population. Results of previous relevant study remained inconsistent. Dong et al. [7] performed a 10-year prospective cohort study on the associations between lipid profiles and CVD and mortality in a Chinese community-dwelling population, which found a U-shape relationship between AIP and the CVD-specific mortality. Zhou et al. [8] searched in health adults from the 1999–2014 NHANES with the aim of exploring the effect of AIP levels (< 2.86, 2.86–3.46, 3.46–4.12, 4.12–5.07, and > 5.07) on the risk of all-cause mortality and CVD-specific mortality using RCS analyses. Their results showed a U-shape association between AIP and all-cause mortality, but this association between AIP and CVD-specific mortality was not significant. In this study, we found both low and high levels of AIP were linked to increased risks of all-cause and CVD mortality (a U-shape relationship) in patients with hypertension. The AIP has been reported to be an independent risk factor for CVD occurrence and mortality [17]. The potential mechanism of relationship between AIP and CVD mortality could be explained by the association of the index with lipoprotein particle size: it is inversely related to LDL cholesterol particle diameter [18]. Results of some studies showed that small dense LDL cholesterol particles were related to higher risk of CVD [19, 20]. Besides, chronic inflammation leads to lipid metabolism derangement, which may also play an important role in the association between AIP and CVD-specific mortality. The specific mechanism of the effect of low or high AIP level on all-cause mortality and CVD-specific mortality in patients with hypertension is needed to be further explored.
Subgroup analyses of age, gender, race, BMI, DM, CVD, antihyperlipidemic agents use and hypotensive drugs use were also performed for further exploration of this U-shape relationship in different populations. In a study on the relationship between AIP and obesity among adults in Taiwan showed that subjects with high AIP levels exhibited significant differences in systolic blood pressure (SBP), diastolic blood pressure (DBP), waist circumference, fasting plasma glucose, serum HDL-C, serum low-density lipoprotein cholesterol (LDL-C), serum TC and prevalence of DM, hypertension, hyperlipidemia and metabolic syndrome [21]. However, the study in Taiwan was not examine the association of AIP with other parameters in obese and non-obese individuals or in different degrees of obesity. Herein, our study was partly extend exploration of the study in Taiwan, because we found that in obese patients instead of those without obesity, AIP levels were related to both all-cause mortality and CVD-specific mortality. Ethnic difference in serum lipid profile, and may further influence the obesity and even death [22]. We found U-shape relationships between AIP and all-cause mortality, and between AIP and CVD-specific mortality in hypertensive patients who were non-Hispanic White. Obesity has been reported to be a stronger risk factor for all-cause mortality among non-Hispanic Caucasians than for African-Americans, and however, the association between obesity and risks of CHD mortality in different race have not been explored exclusively [23]. Fu et al. [24] confirmed that AIP could be used as an independent predictor for the prognosis of typer 2 diabetes mellitus (T2DM) patients in the long-term follow-up, because comparing with patients without T2DM, patients with T2DM tend to have more cardiovascular risk factors, including hyperlipidemia. In the current study, in hypertensive patients without DM, AIP levels were related to both all-cause mortality and CVD-specific mortality. In addition, we found these associations in patients without CVD, not take antihyperlipidemic agents and used hypertension drug. Actually, hyperglycemia, insulin resistance and hyperinsulinemia can all cause lipid metabolism disorders, oxidative stress and vascular endothelial damage, ultimately leading to the aggravation of coronary atherosclerosis [25]. Due to the AIP is calculated by serum TC and HDL-C ratio, the antihyperlipidemic agents use and hypertension drug use may affect the overall vascular and lipid metabolism levels, the role of AIP in the all-cause and CVD-specific mortality in patients with hypertension who had different medication situations should be further explored. Differently, the U-shape association between AIP levels and all-cause mortality was only found in females. Such gender differences in AIP values have been demonstrated in a previous fifteen-year cohort study, in which Sadeghi et al. [26] conducted on healthy adults aged over 35 years old to assess the value of AIP in the prediction of mortality. Relationship of AIP and CVD-specific mortality was also found in patients aged ≥ 60 years old. A study on predictive performance of AIP of all-cause mortality and CVD-specific mortality in middle-aged and elderly Lithuanian population showed that the risk of CVD mortality significantly increased in males with the highest AIP quintile compared with that for the lowest quintile [27]. In females, all-cause mortality was significantly higher for the 4th quintile of AIP as compared with the 1st quintile [27]. Płaczkowska et al. [28] found that different lipid parameters and AIP are associated with a high concentration of small dense LDL depending on the age group. Generally, a stronger association of small dense LDL with lipid parameters was observed in older age, which may be attributed to a longer duration of metabolic disorders. The strength and direction of these relationships observed in middle-aged and older adults differ in various studies [29–31].
This study population was from the NHANES database, which using a multistage sampling design with a weighting scheme that the sample size was large and representative. To our knowledge, it was the first time to explore the association between AIP and all-cause mortality and CVD-specific mortality in patients with hypertension. Also, we tried the best to adjust for the covariates and performed subgroup analyses to explored these associations in different populations. However, there were still some limitations in this study. The study variables including dietary and complications were collected from NHANES that were self-reported, which may lead to a recall bias. Moreover, we were unable to perform the adjustments of some other covariates due to diseases and interventions that occurred during follow-up were not available in NHANES.
Conclusion
AIP was associated with both all-cause mortality and CVD-specific mortality in patients with hypertension in a U-shape. In clinical, the AIP level in patients with different age, gender, race, BMI, DM, CVD, antihyperlipidemic agents and hypertension drug use should be carefully monitored and controlled, in order to improve their prognosis.
Acknowledgements
Not applicable.
List of abbreviations
- CVD
Cardiovascular diseases
- AIP
Atherogenic index of plasma
- HDL-C
High-density lipoprotein cholesterol
- RCS
Restricted cubic spline
- BMI
Body mass index
- NHANES
National Health and Nutrition Examination Survey
- NCHS
National Center for Health Statistics
- DM
Diabetes mellitus
- eGFR
Estimated glomerular filtration rate
- DASH
Dietary Approaches to Stop Hypertension
- MET
Metabolic equivalent
- PAQ
Physical activity reports
- HbAlc
Glycosylated Hemoglobin
- MCQ
Multiple choice question
Authors’ contributions
GD designed the study and wrote the manuscript. GD and NM collected, analyzed, and interpreted the data. GD critically reviewed, edited, and approved the manuscript. All authors read and approved the final manuscript.
Funding
Not applicable.
Data Availability
The datasets generated and/or analyzed during the current study are available in the NHANES database, https://wwwn.cdc.gov/nchs/nhanes/.
Declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The requirement of ethical approval for this was waived by the Institutional Review Board of People’s Hospital of Xinjiang Uygur Autonomous Region, because the data was accessed from NHANES (a publicly available database). The need for written informed consent was waived by the Institutional Review Board of People’s Hospital of Xinjiang Uygur Autonomous Region due to retrospective nature of the study. All methods were performed in accordance with the relevant guidelines and regulations.
Consent for publication
Not applicable.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Parati G, Lackland DT, Campbell NRC, Owolabi MO, Bavuma C, Mamoun Beheiry H, et al. How to improve awareness, treatment, and control of hypertension in Africa, and how to reduce its consequences: a call to Action from the World Hypertension League. Hypertension. 2022;79:1949–61. doi: 10.1161/HYPERTENSIONAHA.121.18884. [DOI] [PubMed] [Google Scholar]
- 2.Di Palo KE, Barone NJ. Hypertension and heart failure: Prevention, targets, and treatment. Cardiol Clin. 2022;40:237–44. doi: 10.1016/j.ccl.2021.12.011. [DOI] [PubMed] [Google Scholar]
- 3.Hengel FE, Sommer C, Wenzel U. [Arterial hypertension] Dtsch Med Wochenschr. 2022;147:414–28. doi: 10.1055/a-1577-8663. [DOI] [PubMed] [Google Scholar]
- 4.Chen S, Cheng W. Relationship between lipid profiles and hypertension: a cross-sectional study of 62,957 chinese adult males. Front Public Health. 2022;10:895499. doi: 10.3389/fpubh.2022.895499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aune D, Huang W, Nie J, Wang Y. Hypertension and the Risk of All-Cause and Cause-Specific Mortality: An Outcome-Wide Association Study of 67 Causes of Death in the National Health Interview Survey. Biomed Res Int. 2021; 2021: 9376134. [DOI] [PMC free article] [PubMed]
- 6.Si Y, Fan W, Han C, Liu J, Sun L. Atherogenic index of plasma, triglyceride-glucose index and monocyte-to-lymphocyte ratio for Predicting Subclinical Coronary Artery Disease. Am J Med Sci. 2021;362:285–90. doi: 10.1016/j.amjms.2021.05.001. [DOI] [PubMed] [Google Scholar]
- 7.Dong J, Yang S, Zhuang Q, Sun J, Wei P, Zhao X, et al. The Associations of lipid profiles with Cardiovascular Diseases and Death in a 10-Year prospective cohort study. Front Cardiovasc Med. 2021;8:745539. doi: 10.3389/fcvm.2021.745539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhou D, Liu X, Lo K, Huang Y, Feng Y. The effect of total cholesterol/high-density lipoprotein cholesterol ratio on mortality risk in the general population. Front Endocrinol (Lausanne) 2022;13:1012383. doi: 10.3389/fendo.2022.1012383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fernández-Macías JC, Ochoa-Martínez AC, Varela-Silva JA, Pérez-Maldonado IN. Atherogenic index of plasma: Novel Predictive Biomarker for Cardiovascular Illnesses. Arch Med Res. 2019;50(5):285–94. doi: 10.1016/j.arcmed.2019.08.009. [DOI] [PubMed] [Google Scholar]
- 10.Si Y, Fan W, Han C, Liu J, Sun L. Atherogenic index of plasma, triglyceride-glucose index and monocyte-to-lymphocyte ratio for Predicting Subclinical Coronary Artery Disease. Am J Med Sci. 2021;362(3):285–90. doi: 10.1016/j.amjms.2021.05.001. [DOI] [PubMed] [Google Scholar]
- 11.Shi YY, Zheng R, Cai JJ, Qian SZ. The association between triglyceride glucose index and depression: data from NHANES 2005–2018. BMC Psychiatry. 2021;21(1):267. doi: 10.1186/s12888-021-03275-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bucholz EM, Rodday AM, Kolor K, Khoury MJ, de Ferranti SD. Prevalence and predictors of cholesterol screening, awareness, and Statin Treatment among US adults with familial hypercholesterolemia or other forms of severe dyslipidemia (1999–2014) Circulation. 2018;137:2218–30. doi: 10.1161/CIRCULATIONAHA.117.032321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Doran B, Guo Y, Xu J, Weintraub H, Mora S, Maron DJ, et al. Prognostic value of fasting versus nonfasting low-density lipoprotein cholesterol levels on long-term mortality: insight from the National Health and Nutrition Examination Survey III (NHANES-III) Circulation. 2014;130:546–53. doi: 10.1161/CIRCULATIONAHA.114.010001. [DOI] [PubMed] [Google Scholar]
- 14.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–12. [DOI] [PMC free article] [PubMed]
- 15.Jayanama K, Theou O, Godin J, Cahill L, Shivappa N, Hébert JR, et al. Relationship between diet quality scores and the risk of frailty and mortality in adults across a wide age spectrum. BMC Med. 2021;19(1):64. doi: 10.1186/s12916-021-01918-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.National Center for Health Statistics. Linked mortality files matching methodology. 2011.
- 17.Onat A, Can G, Kaya H, Hergenc G. Atherogenic index of plasma” (log10 triglyceride/high-density lipoprotein-cholesterol) predicts high blood pressure, diabetes, and vascular events. J Clin Lipidol. 2010;4:89–98. doi: 10.1016/j.jacl.2010.02.005. [DOI] [PubMed] [Google Scholar]
- 18.Dobiasova M, Frohlich J, Sedova M, Cheung MC, Brown BG. Cholesterol esterification and atherogenic index of plasma correlate with lipoprotein size and findings on coronary angiography. J Lipid Res. 2011;52:566–71. doi: 10.1194/jlr.P011668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kokubo Y, Watanabe M, Higashiyama A, Honda-Kohmo K. Small-dense low-density lipoprotein cholesterol: a subclinical marker for the primary Prevention of Coronary Heart Disease. J Atheroscler Thromb. 2020;27:641–3. doi: 10.5551/jat.ED134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Qiao YN, Zou YL, Guo SD. Low-density lipoprotein particles in atherosclerosis. Front Physiol. 2022;13:931931. doi: 10.3389/fphys.2022.931931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang JS, Yeh WC, Tsai YW, Chen JY. The relationship between Atherogenic Index of plasma and obesity among adults in Taiwan. Int J Environ Res Public Health. 2022; 19. [DOI] [PMC free article] [PubMed]
- 22.Stone NJ, Grundy SM. The 2018 AHA/ACC/Multi-Society cholesterol guidelines: looking at past, present and future. Prog Cardiovasc Dis. 2019;62:375–83. doi: 10.1016/j.pcad.2019.11.005. [DOI] [PubMed] [Google Scholar]
- 23.Clark K. The effect of age on the association between body-mass index and mortality. J Insur Med. 1998;30:48–9. [PubMed] [Google Scholar]
- 24.Fu L, Zhou Y, Sun J, Zhu Z, Xing Z, Zhou S, et al. Atherogenic index of plasma is associated with major adverse cardiovascular events in patients with type 2 diabetes mellitus. Cardiovasc Diabetol. 2021;20:201. doi: 10.1186/s12933-021-01393-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Beckman JA, Paneni F, Cosentino F, Creager MA. Diabetes and vascular disease: pathophysiology, clinical consequences, and medical therapy: part II. Eur Heart J. 2013;34:2444–52. doi: 10.1093/eurheartj/eht142. [DOI] [PubMed] [Google Scholar]
- 26.Sadeghi M, Heshmat-Ghahdarijani K, Talaei M, Safaei A, Sarrafzadegan N, Roohafza H. The predictive value of atherogenic index of plasma in the prediction of cardiovascular events; a fifteen-year cohort study. Adv Med Sci. 2021;66(2):418–23. doi: 10.1016/j.advms.2021.09.003. [DOI] [PubMed] [Google Scholar]
- 27.Tamosiunas A, Luksiene D, Kranciukaite-Butylkiniene D, Radisauskas R, Sopagiene D, Bobak M. Predictive importance of the visceral adiposity index and atherogenic index of plasma of all-cause and cardiovascular disease mortality in middle-aged and elderly lithuanian population. Front Public Health. 2023;11:1150563. doi: 10.3389/fpubh.2023.1150563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Placzkowska S, Solkiewicz K, Bednarz-Misa I, Kratz EM. Atherogenic plasma index or non-high-density lipoproteins as markers best reflecting age-related high concentrations of small dense low-density lipoproteins. Int J Mol Sci. 2022; 23. [DOI] [PMC free article] [PubMed]
- 29.Srisawasdi P, Chaloeysup S, Teerajetgul Y, Pocathikorn A, Sukasem C, Vanavanan S, et al. Estimation of plasma small dense LDL cholesterol from classic lipid measures. Am J Clin Pathol. 2011;136:20–9. doi: 10.1309/AJCPLHJBGG9L3ILS. [DOI] [PubMed] [Google Scholar]
- 30.Moriyama K, Takahashi E. Non-HDL cholesterol is a more Superior Predictor of small-dense LDL cholesterol than LDL cholesterol in japanese subjects with TG levels <400 mg/dL. J Atheroscler Thromb. 2016;23:1126–37. doi: 10.5551/jat.33985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hoogeveen RC, Gaubatz JW, Sun W, Dodge RC, Crosby JR, Jiang J, et al. Small dense low-density lipoprotein-cholesterol concentrations predict risk for coronary heart disease: the Atherosclerosis Risk in Communities (ARIC) study. Arterioscler Thromb Vasc Biol. 2014;34:1069–77. doi: 10.1161/ATVBAHA.114.303284. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available in the NHANES database, https://wwwn.cdc.gov/nchs/nhanes/.