Abstract
Cardiovascular involvement in idiopathic inflammatory myopathies (IIM) is an understudied area which is gaining increasing recognition in recent times. Recent advances in imaging modalities and biomarkers have allowed the detection of subclinical cardiovascular manifestations in IIM. However, despite the availability of these tools, the diagnostic challenges and underestimated prevalence of cardiovascular involvement in these patients remain significant. Notably, cardiovascular involvement remains one of the leading causes of mortality in patients with IIM. In this narrative literature review, we outline the prevalence and characteristics of cardiovascular involvement in IIM. Additionally, we explore investigational modalities for early detection of cardiovascular involvement, as well as newer approaches in screening to facilitate timely management.
|
Key points • Cardiac involvement in IIM in majority cases is subclinical and a major cause of mortality. • Cardiac magnetic resonance imaging is sensitive for detection of subclinical cardiac involvement. |
Supplementary Information
The online version contains supplementary material available at 10.1007/s10067-023-06599-4.
Keywords: Cardiac, Cardiovascular, Dermatomyositis, Heart, Myopathies, Myositis
Introduction
Idiopathic inflammatory myopathies (IIM) are heterogeneous, multisystem diseases with varied phenotype and presentation. Several subtypes are recognized, including dermatomyositis (DM), juvenile DM (JDM), clinically amyopathic DM (CADM), polymyositis (PM), inclusion body myositis (IBM), immune-mediated necrotizing myopathy (IMNM), anti-synthetase syndrome (ASS), and overlap myositis [1]. A diagnosis of IIM is associated with significant morbidity and an increased risk of mortality. Cancer, pulmonary involvement and cardiovascular disease are the most common causes of death worldwide [2–4]. Notably, a significant link between inflammation and cardiovascular involvement in rheumatic diseases has been described [5, 6]. Thus, patients with IIM are at an increased risk of cardiovascular involvement.
Despite advances in the management of patients with IIM, identification of cardiac involvement in these patients is often delayed. Limitations of clinical examination and traditional cardiac investigations have led to recognition of newer imaging modalities for the early detection of cardiac involvement in IIM.
This narrative review focuses on recent literature regarding cardiovascular involvement in patients with IIM, including its manifestation as a result of disease activity, cardiac inflammation and damage or comorbid cardiovascular risk factors. The authors discuss various investigational modalities for evaluating cardiac involvement with emphasis on the newer imaging modalities. This review aims to explore the undiscovered impact of cardiac involvement in patients with IIM, newer modalities for accurate identification and assessment, and the unmet need for standardized investigational protocol.
Methods
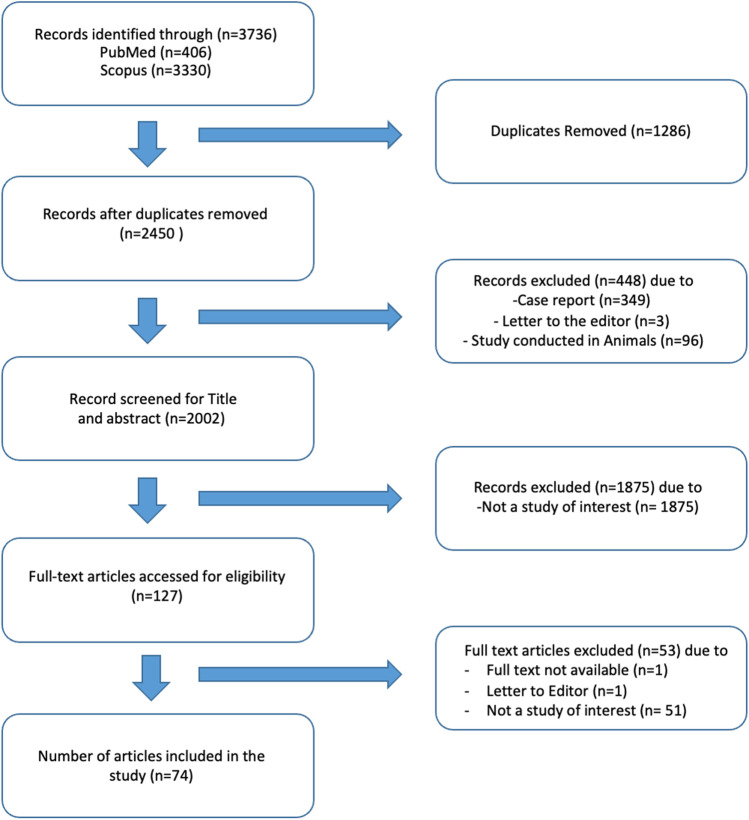
The two largest medical databases, namely Scopus and PubMed were searched on the same day for relevant articles published between 2000 and 2022. The search terms used were [myositis OR dermatomyositis OR polymyositis OR inclusion body myositis OR idiopathic inflammatory myopathy OR autoimmune myopathy OR idiopathic inflammatory myopathies OR autoimmune myopathies] AND [cardiovascular risk OR heart failure OR cardiomyopathy OR cardiovascular disease OR myocarditis OR heart infarction OR heart disease OR heart arrhythmia OR coronary artery disease OR ischemic heart disease OR thromboembolism] as outlined in previously published methods protocols for narrative reviews [7]. Only data published in English were included. Case reports, case series with less than 10 cases, letter to editor, book chapters, and animal studies were excluded. Since most of the literature on myositis was covered in the above-mentioned databases, and Web of Science is rich in older literature and information in regional languages, these were considered beyond the scope of the authors’ current work. The detailed search strategy and results are outlined in Fig. 1. Additional studies were identified from references of the extracted articles and included where relevant.
Fig. 1.
Workflow flowchart
Epidemiology
Although first described in 1899 by Oppenheim, cardiac involvement in myositis was not a well-recognized manifestation until the 1970s [8, 9]. The understanding and definition of cardiac involvement in myositis have significantly evolved in recent years owing to the advent of more sensitive diagnostic techniques [10]. The prevalence of cardiac manifestations in IIM varies widely, ranging from 6 to 75% depending on the patient inclusion criteria, definition of cardiac involvement, and the imaging modalities used. Importantly, a substantial proportion of cardiac involvement in myositis is subclinical, with fewer than 10% being clinically manifest unless investigated [9]. The EuroMyositis registry, comprising 3067 cases across 11 countries, reported that cardiac involvement occurring due to the IIM disease process was most frequent in the connective tissue diseases-overlap myositis group and was least frequent in patients with IBM [11].
Mortality and cardiovascular involvement (Table 1)
Table 1.
Mortality and cardiovascular involvement in patients with IIM
| Author (year of publication) | Study group (n) | Diagnosis | Study design | Key observation | Country |
|---|---|---|---|---|---|
| Dobloug et al. [2] (2017) | 716 | IIM | Cohort study | IIM patients had a higher mortality rate compared to the general population. Malignancies, cardiac diseases and respiratory diseases were the main causes of death. Cardiac disease accounted for 28.1% of all deaths in IIM patients. The mortality rate was highest in the first year after the diagnosis of IIM | Sweden |
| Limaye et al. [12] (2012) | 364 | IIM | Retrospective Cohort study | A standardized mortality ratio (SMR) of 1.75 was reported in IIM, and it was highest in patients with DM (2.40). The major causes of death were cardiovascular disease (31%), infection (22%) and malignancy (11%). Risk factors for death included the absence of autoantibodies | Australia |
| Dankó et al. [13] (2004) | 162 |
DM (n = 42) PM (n = 75) Juvenile (n = 9) Cancer associated myositis (n = 7) Overlap myositis (n = 29) |
Cohort study | The most frequent causes of death were cardiac and pulmonary complications. Cardiovascular manifestations were responsible for 8 out of 18 deaths (two cases of arrhythmia, three of heart failure, two of cardiac arrest and one of myocardial infarction) and were significant prognostic factors for mortality. Deaths from cardiovascular disease occurred a median of 59 months post IIM diagnosis | Hungary |
| Jung et al. [14] (2020) | 3014 |
DM (n = 1860) PM (n = 1154) |
Population based study | Reported a very high SMR in young IIM patients (DM: 15.0, PM: 8.1). No influence of sex on the SMR was observed. Cardiovascular events were reported in 155 (5.1%) patients and 40.6% of patients with cardiovascular disease died | Korea |
Several independent research groups worldwide have reported an increased risk of cardiovascular mortality in patients with IIM. For instance, a Swedish population-based nationwide cohort study by Dobloug et al. found that cardiac disease was one of the most common causes of mortality among IIM patients [2]. In a retrospective cohort study by Limaye et al., a standardized mortality ratio (SMR) of 1.75 was reported in IIM; the ratio was highest in the DM subtype at 2.40. The study also found that cardiovascular disease accounted for nearly one-third of deaths in the cohort of 364 individuals from South Australasia, followed by infection (22%) and malignancy (11%) [12]. Similarly, a large Korean population-based study of 3014 cases confirmed a high SMR and a high incidence of cardiovascular disease, even in young individuals with IIM (DM: 15.0, PM: 8.1) [14]. Dankó et al. conducted a survival analysis of 162 Hungarian patients and found that lung and cardiac complications were the most frequent causes of death. Death attributable to cardiovascular complications occurred late in the course of the disease (median 59 months). Cardiovascular manifestations were responsible for 8 out of 18 deaths (two cases of arrhythmia, three of heart failure, two of cardiac arrest, and one of myocardial infarction) and were identified as significant adverse prognostic factors [13].
Collectively, these studies suggest cardiac complications are a common cause of mortality among IIM patients.
Risk of cardiovascular disease
Accumulating evidence suggests an increased incidence of cardiovascular diseases such as acute coronary syndrome, heart failure, and arrhythmias in patients with IIM. Two meta-analyses (Xiong et al. and Ungprasert et al.) have examined the association between IIM and cardiovascular events in patients with PM and DM. The study by Xiong et al. included 25,433 patients with PM/DM and found a 2.3-fold increase in the risk of cardiovascular events within the first 5 years of IIM diagnosis. Moreover, PM/DM was identified as an independent risk factor for the development of cardiovascular events [15]. The study by Ungprasert et al., which comprised 13,201 patients, reported a 2.24-fold increased risk of cardiovascular disease in patients with IIM. Indeed, cardiovascular disease was identified as the leading cause of mortality in this cohort [16]. Prieto et al. conducted a study in the UK comprising 603 patients with IIM and reported a significantly greater rate of cardiovascular events in patients with IIM than in healthy controls, even after adjustment for traditional cardiovascular risk factors. This finding suggests that the increased risk could not be entirely attributed to traditional cardiovascular risk factors and was partly accounted for by the systemic inflammation present in these patients. The excess risk was most apparent in the first 5 years post IIM diagnosis and then appeared to decline [17]. These results are consistent with a case-control study which reported an increased risk of cardiovascular disease in patients with IIM, even in the absence of traditional risk factors [18]. Overall, the evidence suggests that patients with IIM have an increased risk of developing cardiovascular disease, particularly within the first 5 years of IIM diagnosis. This risk cannot be entirely attributed to traditional cardiovascular risk factors and may be partly due to systemic inflammation.
Traditional cardiovascular risk factors (Table 2)
Table 2.
Traditional cardiovascular risk factors in patients with IIM
| Author (year of publication) | Study group (n) | Diagnosis | Study design | Key observation | Country |
|---|---|---|---|---|---|
| Wang et al. [19] (2014) | 60 | PM | Case control study | Duration of PM was less than 6 months and patients were treatment naive. 50% patients had reduced HDL levels and 47% patients had raised triglyceride levels. Serum CRP levels negatively correlated with HDL cholesterol. The inflammatory state in PM could be responsible for the metabolism of HDL cholesterol | China |
| Diederichsen et al. [20] (2015) | 76 |
DM PM |
Cross sectional observational study | Hypertension (71% versus. 42%) and diabetes mellitus (13% versus 0%) were more frequent in patients of DM and PM than in age- and sex-matched healthy controls. These patients also had significantly higher serum triglyceride levels. Presence of coronary artery atherosclerosis as evidenced by calcification on computed tomography scan was more prevalent in IIM patients, however, this was not associated with DM or PM | Denmark |
| Bae et al. [21] (2021) | 95 |
DM (n = 55) PM (n = 30) IBM (n = 10) |
Observational study | Worse HDL function was reported in patients with DM as opposed to patients with PM or IBM. Higher disease activity was associated with impaired antioxidant function of HDL | USA |
| Qin et al. [22] (2022) | - | IIM | Systematic review and Meta-analysis | Patients with IIM were 1.44, 1.67, and 1.48 times more likely to have hypertension, diabetes mellitus, and dyslipidemia respectively, when compared with non-IIM individuals | China |
| Pakhchanian et al. [23] (2021) | 5578 | DM | TriNetX registry | Of 5578 patients with DM, 66.82% of patients had hypertension as opposed to 25.05% of the general population. Similarly, the prevalence of ischemic heart disease (27.18% vs 7.3%) and diabetes mellitus (33.87% vs 12.14%) was higher in DM patients when compared to general population | |
| Oreska et al. [24] (2022) | 39 |
DM-16 PM-7 IMNM-8 ASS-8 |
Cross sectional pilot study | Authors found no significant differences in the prevalence of traditional risk factors between patients with IIM and healthy controls | Czech Republic |
Diabetes mellitus, hypertension, and dyslipidemia are traditional risk factors for coronary heart disease. Several studies have investigated the prevalence of these risk factors in patients with IIM [12, 19, 21–27]. For instance, Diederichsen et al. observed a higher prevalence of hypertension (71% versus 42%) and diabetes mellitus (13% versus 0%) in patients with DM and PM compared to age- and sex-matched healthy controls. Patients with IIM also had significantly higher levels of serum triglycerides. Multivariate analysis revealed that smoking and age were associated with accelerated atherosclerosis in this cohort [20].
This same study showed that the presence of coronary artery atherosclerosis, as evidenced by calcification on computed tomography scan, was more prevalent in IIM patients. However, this was associated with the presence of traditional cardiovascular risk factors rather than IIM-specific factors [20]. Conversely, Prieto et al. reported an increased risk of cardiovascular diseases in patients with IIM even in the absence of traditional cardiovascular risk factors [17]. A systematic review and meta-analysis by Qin et al. also reported that patients with IIM were 1.44, 1.67, and 1.48 times more likely to have hypertension, diabetes mellitus, and dyslipidemia respectively, when compared with non-IIM individuals [22]. In a case-control study by Wang et al., untreated patients with early PM (n = 60) had dyslipidemia characterized by high triglyceride and low high-density lipoprotein cholesterol levels, suggesting a high risk of atherosclerosis [19]. Similarly, a study by Pakhchanian et al. using the TriNetX database (5578 DM patients) found that 66.82% of patients had hypertension as opposed to 25.05% of the general population. The prevalence of ischemic heart disease (27.18% vs 7.3%) and diabetes mellitus (33.87% vs 12.14%) was also higher in DM patients when compared to the general population [23]. In contrast, a single-centre study (n = 39 patients) conducted by Oreska et al. found no significant differences in the prevalence of traditional risk factors between patients with IIM and healthy controls [24]. While mixed results have been reported, large cohort studies and meta-analyses all suggest an increased risk of cardiovascular risk factors in patients with IIM.
A possible mechanism for accelerated cardiovascular disease in IIM was explored in a study evaluating the antioxidant function of high-density lipoprotein (HDL) particles isolated from patient blood. The protective function of HDL in IIM patients was abnormal compared to healthy controls, with patients with DM and those with higher disease activity having particularly impaired HDL function [21]. Together, these studies underscore the need to determine the relative contribution of conventional risk factors vis-à-vis disease-specific inflammation-induced accelerated atherogenesis in the development of cardiovascular disease.
Arrhythmias (Supplementary Table)
Various types of conduction system abnormalities have been described in patients with IIM, including supraventricular and ventricular tachycardia, fascicular blocks, bundle branch blocks, and atrioventricular (AV) blocks. In a large retrospective cohort study of 32,085 patients with DM or PM, Naaraayan et al. reported a significantly higher prevalence of atrial fibrillation, atrial flutter, supraventricular tachycardia, and unclassified arrhythmias in patients with IIMs < 70 years old compared with matched controls. The prevalence of arrhythmias was found to be higher in men than in women. Importantly, the diagnosis of arrythmias was associated with significant in-hospital mortality among patients with PM/DM [28]. A cross-sectional study of 112 IIM patients (78 DM and 34 PM) by Deveza et al. reported a higher frequency of arrhythmia in patients with PM when compared with DM. However, there was no significant difference in the occurrence of arrhythmias between IIM patients and the control group [29]. In a retrospective cohort study of 75 patients with IIM by Huang et al., PM and positive serum anti-mitochondrial antibodies (AMA) were strongly associated with ventricular arrhythmia in PM/DM patients with myocardial involvement [30].
Several mechanisms which may predispose to arrhythmias in patients with chronic inflammatory diseases such as IIM have been hypothesized, including infiltration of atria or ventricle by inflammatory cell infiltrate, autonomic dysfunction with resultant reduced heart rate variability and ‘acquired channelopathies’ due to release of cytokines or autoantibodies [31]. In a clinicopathological study of 16 autopsied patients with PM/DM conducted by Haupt et al., direct involvement of the conduction system by myositis and contraction-band necrosis was described [32].
Thromboembolism (Supplementary Table)
Inflammation is known to play a role in promoting thrombosis by upregulating procoagulant pathways, downregulating natural anticoagulant factors and suppressing fibrinolysis [33]. An increased incidence of thrombosis in patients with chronic inflammatory diseases and autoimmune diseases is well described [34–38]. Patients with IIM are no exception, with several studies having reported a significantly increased risk of venous thromboembolism in patients with DM/PM [34, 39–42]. An enhanced in vitro thrombin generation profile in patients with DM/PM has been demonstrated, which may correlate to an increased risk of thromboembolic events [43]. Carruthers et al. reported an eight- and six-fold higher risk of developing venous thromboembolism in patients with DM and PM respectively. Notably, this risk was highest during the first year for patients with PM and during the initial 2 years after diagnosis in patients with DM, potentially attributable to uncontrolled inflammatory activity [44]. Similarly, in a retrospective analysis of 1144 Chinese patients with IIM, over half of IIM patients with thromboembolic events developed this complication in the 6 months before or after diagnosis of IIM. Increased myositis disease activity, malignancy, infection, and duration of steroid use were significant risk factors for thrombosis in this study [45]. A Swedish population-based cohort study (Antovic et al.), found that the risk of thromboembolic events was eight times higher in patients with IIM than in the general population, with the highest observed in patients with DM and a history of cancer. In line with other reports, the incidence rates were maximum in the first year post diagnosis and decreased thereafter [46]. Conversely, another retrospective cohort study found that age at diagnosis was the only independent risk factor for thrombotic events in patients with IIM. For each year of age, there was a 3.5% increased chance of experiencing a thrombotic event in patients with IIM. In their cohort of 253 patients with IIM, only 6% had anti-phospholipid antibodies, with no significant difference between the patients and comparators [47].
Pregnant women with autoimmune disease, including IIM, are also at an increased risk of thromboembolism. Indeed, Bleau et al. studied 43,523 pregnant women with autoimmune disease and observed that the risk of thromboembolism was particularly high for patients with DM comparted to other autoimmune diseases [48].
Atherosclerotic cardiovascular diseases and acute coronary syndrome (Table 3)
Table 3.
Atherosclerotic cardiovascular disease and acute coronary syndrome in patients with IIM
| Author (year of publication) | Study group (n) | Diagnosis | Study design | Key observation | Country |
|---|---|---|---|---|---|
| Linos et al. [49] (2013) | 10156 | DM | Case control analyses | 20% of all DM hospitalizations were associated with atherosclerotic cardiovascular diagnosis. Heart failure (12% of DM hospitalizations) was the most common followed by myocardial infarction (4.4% of DM hospitalizations). When compared to DM patients without atherosclerotic heart disease, patients with associated atherosclerotic heart disease were twice more likely to die during hospital stay (OR = 2.0 95% CI 1.7–2.5, p < 0.0001). In those patients with both dermatomyositis and cardiovascular disease the odds ratio for death was 1.98 when compared to controls with only cardiovascular disease | USA |
| Weng et al. [50] (2019) | 1145 |
DM (n = 640) PM (n = 505) |
Retrospective population-based cohort | The authors reported adjusted hazard ratios (aHR) of 2.21 (95% CI 1.64, 2.99) in DM and 3.73 (95% CI 2.83, 4.90) in PM for coronary heart disease. There was a 2- and 3-fold increase in the risks of incident CHD in patients with DM and PM respectively when compared to their respective comparison groups | Taiwan |
| Leclair et al. [51] (2019) | 655 |
DM (n = 218) Other IIM (n = 437) |
Population based cohort study | This population-based cohort study reported that patients with IIM experienced their first ACS episode earlier than general population comparators identified from national registries and matched for follow up period [median (interquartile range) 2.4 (1.0–4.6) vs 3.5 (1.8–6.0) years)]. The hazard ratio was 2.4 for ACS in IIM patients compared to general population, with an increased risk in the first year of diagnosis | Sweden |
| Rai et al. [52] (2016) | 774 |
DM (n = 350) PM (n = 4242) |
Matched cohort analyses | This matched cohort analysis reported a nearly 4-fold increased risk of myocardial infarction in patients with PM and a 3-fold increased risk in DM patients when compared with controls | Canada |
| Lin et al. [53] (2015) | 2029 | DM/PM | Population based retrospective cohort study | The authors reported a 1.74 times greater incidence of ACS in a DM/PM cohort of 2029 patients. This incidence was greater in men than in woman. Patients with hypertension and end stage renal disease had significantly greater risk of developing ACS (aHR 2.48, 95% CI 1.37–4.52; aHR 4.86, 95% CI 1.41–16.88, respectively). The authors postulated that the occurrence of ACS in these patients may be attributed to mechanisms other than traditional cardiovascular risk factors as a higher risk of ACS (HR 1.75) was observed in DM/PM patients without comorbidities when compared with non-DM/PM patients | Taiwan |
| Lai et al. [54] (2013) | 907 | DM | Prospective cohort study | The authors reported an adjusted hazard ratio (3.37) of acute myocardial infarction close to the crude hazard ratio (3.96) in 907 patients of DM, implying that DM plays an independent role in increased cardiovascular events | Taiwan |
| Tisseverasinghe et al. [55] (2009) | 607 | DM/PM | Nested case control analyses | An association between the incidence of myocardial infarction and stroke and the presence of hypertension and lipid abnormalities in patients with DM/PM was observed. Immunomodulators such as methotrexate, azathioprine, antimalarial agents, cyclophosphamide) were negatively associated with these arterial events | Canada |
Numerous studies have investigated the relationship between IIM and cardiovascular events. In a case-control analysis of 10,156 patients with DM conducted in the USA, researchers found that 20% of all DM hospitalizations were associated with an atherosclerotic cardiovascular diagnosis. Patients with DM and atherosclerotic heart disease were twice as likely to die during their hospital stay compared to DM patients who did not suffer from atherosclerotic heart disease (odds ratio (OR) 2.0, 95% confidence interval (CI) 1.7–2.5, p < 0.0001). Furthermore, among patients with both DM and cardiovascular disease, the odds ratio for death was 1.98 when compared to controls with only cardiovascular disease [49]. This suggests an additive effect of DM and cardiovascular disease on risk of death. Similarly, other studies have reported an increased risk of coronary heart disease in IIM patients when compared to controls [50, 56].
Patients with IIM are at a significantly increased risk of experiencing an acute coronary syndrome (ACS) when compared to the general population as evidenced by several studies [51–54]. A study by Lin et al. reported a greater incidence of ACS in men than in women, and in those with hypertension and end stage renal disease. However, the authors also noticed an higher risk of ACS [Hazard ratio (HR) 1.75] in DM/PM patients without comorbidities when compared to the non-DM/PM cohort, leading them to postulate that mechanisms other than traditional cardiovascular risk factors may contribute to the increased ACS risk in these patients [53]. In line with previously mentioned study, Lai et al. reported an adjusted HR (3.37) for acute myocardial infarction close to the crude hazard ratio (3.96) in 907 patients of DM, implying that DM plays an independent role in increased cardiovascular events [54]. Finally, a nested case-control analysis by Tisseverasinghe et al. reported an association between the incidence of myocardial infarction and stroke, hypertension and lipid abnormalities. On the other hand immunomodulators such as methotrexate, azathioprine, antimalarial agents, cyclophosphamide were negatively associated with these arterial events [55].
Thus, factors besides traditional cardiovascular risk factors might contribute to the increased risk of cardiovascular disorders in patients with IIM; a finding which needs to be explored further.
Heart failure (Supplementary Table)
Heart failure is the most frequently reported clinically apparent cardiovascular manifestation in patients with IIM [9, 57]. In a retrospective cohort study in Taiwan, patients with PM/DM showed a significantly increased risk of heart failure as compared to those without PM/DM (HR 2.06, 95%CI 1.36–3.12). The risk of heart failure was found to be highest within first 10 years following diagnosis of PM/DM [58]. In a cross-sectional multicenter study of 108 patients with IIM and with myocardial involvement, 62% of patients presented with heart failure, 56.5% with clinically significant arrhythmia, and 18.5% of patients presented with both. Anti-mitochondrial antibodies and pulmonary hypertension were significant associated factors [59]. In a single center retrospective study of 32 Chinese patients with IIM and heart failure, 13 of the 17 recorded deaths were attributed to cardiogenic shock [60].
Myocarditis (Supplementary Table)
Diagnosing myocarditis in patients with inflammatory myopathies poses a challenge due to its subclinical nature in the majority of cases [9]. The prevalence of myocarditis in IIM has been reported to be 38% on cardiac pathology [61]. Conversely, Dieval et al. reported that the prevalence of clinically apparent was 3.4% in patients with ASSD. Myocarditis was the initial presenting feature in 42% of the ASSD patients with myocarditis in this study. Myocarditis was reported to be associated with active disease but not with any particular antibody specificity [62]. Liu et al. reported a shorter disease duration, more manifestations of heart failure and symptoms of IIMs and increased frequency of AMA-M2 antibody positivity in patients of IIM with myocarditis [63].
Autoantibodies
Emerging data indicate an association between cardiovascular disease and certain myositis-specific antibodies, myositis-associated antibodies and anti-mitochondrial antibodies [64–66]. In a longitudinal cohort study comprising 619 adult and 371 juvenile patients with IIM, 31 adult patients were diagnosed with cardiomyopathy. While only 5% of adult patients in this cohort were AMA-positive, the presence of this antibody was associated with the development of cardiomyopathy and persistent muscle weakness. Notably, this association was not seen in the pediatric population [67]. Additionally, a retrospective case review reported cardiac involvement in five of seven patients with positive anti-mitochondrial antibodies. The authors noted that cardiac function in patients with cardiomyopathy or myocarditis could improve with immunosuppression [68]. In another study of 63 patients with IIM (DM, PM, CADM), those who were anti-MDA5 (melanoma differentiation-associated protein 5) positive (N = 21) showed significantly lower T waves amplitude on the electrocardiogram (ECG) than anti-MDA-5 negative patients. Nonetheless, there were no differences in the contractile function of the heart between the two groups. These low amplitudes were restored during the remission phase, suggesting a possible role for aberrant immune responses associated with active anti-MDA5+ DM [69].
Juvenile dermatomyositis (Table 4)
Table 4.
Cardiovascular manifestations in Juvenile Dermatomyositis
| Author (year of publication) | Study group (n) | Diagnosis | Study design | Key observation | Country |
|---|---|---|---|---|---|
| Coyle et al. [70] (2009) | 17 |
JDM (n = 16) Juvenile Polymyositis [JPM (n = 1)] |
Cohort study | Authors reported a high frequency of metabolic abnormalities and metabolic syndrome in patients with JDM. 71% of patients had a blood pressure greater then 75th percentile, and 47% patients had BMI > 85th percentile. Hypertriglyceridemia was present in 47.1% of patients. Metabolic abnormalities appeared to be linked to disease activity | USA |
| Silverberg et al. [71] (2018) | 1407 | JDM | Hypertension was the most common comorbidity in children with JDM. Even in the absence of traditional cardiovascular risk factors, children with JDM had an increased risk of cardiovascular comorbidities when compared to non-JDM patients. Thus, traditional cardiovascular risk factors may not completely account for the increased rates of cardiovascular comorbidities in JDM | USA | |
| Kozu e al. [72] (2013) | 25 | JDM | Kozu et al studied the lipid profile of 25 patients with JDM. Compared to healthy controls, patients with JDM had significantly higher triglyceride levels and lower median high density lipoprotein levels. The authors reported a positive correlation between dyslipidemia and disease activity | Brazil | |
| Witczak et al. [73] (2022) | 57 | JDM | Cross-sectional study | In this study, 18% of JDM patients had cardiac involvement, which was mostly subclinical. Patients with cardiac involvement showed higher disease activity 1 year after diagnosis and an unfavorable lipid profile | Norway |
| Witczak et al. [74] (2022) | 59 | JDM | Cross-sectional study | This study demonstrated higher body fat percentage and lower appendicular lean mass index in patients with active JDM when compared to children with inactive JDM. Central fat distribution was linked to cardiometabolic alterations, notably left ventricular dysfunction | Norway |
| Barth et al. [75] (2019) | 58 | JDM | The authors did not find any significant differences in the ECG, heart rate variability, and systolic or diastolic function between patients with low and normal NCD. However, they reported an association between lung involvement and low NCD. A plausible explanation for the lack of association between NCD and cardiac dysfunction could be the small sample size of the study, making the study underpowered to demonstrate this association | Norway | |
| Schwartz et al. [76] (2013) | 59 | JDM | Cross-sectional study | In this cross-sectional study of 59 JDM patients examined at a median of 16.8 years after disease onset, both systolic and diastolic dysfunction were reported to be linked to longer disease duration and high disease activity score of skin but not muscle at 1 year. Cumulative prednisolone dose was also a predictor of diastolic dysfunction. Thus, sustained early skin activity could predict cardiac dysfunction in long term | Norway |
| Barth et al. [77] (2016) | 55 | JDM | Barth et al demonstrated lower heart rate variability (HRV) in patients with active JDM than inactive JDM. This reduced HRV was found to be associated with elevated inflammatory markers, reduced myocardial function and active disease | Norway |
JDM, although rare, is the most common childhood inflammatory myopathy. There are several similarities between the clinical features of JDM and those of adult DM [78].
Cardiovascular risk factors and metabolic syndrome appear prevalent among patients with JDM. For instance, several studies have observed a high frequency of hypertension [70, 71], dyslipidemia [70–73] and elevated body fat percentage [70, 74] in JDM patients versus controls. In several studies, metabolic abnormalities were found to be correlated with JDM disease activity [70, 72]. Importantly, increased rates of cardiovascular involvement have been observed even in the absence of traditional cardiovascular risk factors, suggesting that such risk factors may not completely account for the increased rates of cardiovascular disease in JDM [71].
Potential cardiac manifestations in JDM include pericarditis, systolic or diastolic dysfunction and electrocardiographic abnormalities [75, 76, 79]. In a cross-sectional study of 57 patients with JDM, 18% of the patients had cardiac involvement- defined as left ventricular systolic or diastolic dysfunction on echocardiography, which was mostly subclinical. Patients with cardiac involvement showed higher disease activity 1 year after diagnosis and an unfavorable lipid profile [73]. In a cross-sectional study of 59 patients with JDM examined at a median of 16.8 years after disease onset, both systolic and diastolic dysfunction were reported to be linked to longer disease duration and high disease activity score of skin but not muscle at 1 year. The cumulative prednisolone dose was also a predictor of diastolic dysfunction. Thus, sustained early skin activity could predict cardiac dysfunction in the long term [76].
A low heart rate variability (HRV) is a strong predictor of cardiac arrest [80]. Barth et al. demonstrated a lower HRV in patients with active JDM than in those with inactive JDM, indicating the presence of autonomic nervous system dysregulation in JDM. This reduced HRV was found to be associated with elevated inflammatory markers, reduced myocardial function, and active disease [77].
Abnormal nailfold capillary density (NCD) is an indicator of systemic vasculopathy and thus may have utility in the evaluation of JDM. Barth et al. did not find any significant differences in the electrocardiography, heart rate variability, and systolic or diastolic function between patients with low and normal NCD. However, these authors reported an association between lung involvement and low NCD. A plausible explanation for the lack of association between NCD and cardiac dysfunction could be the small sample size of the study, making the study underpowered to demonstrate this association [75].
Investigations (Table 5)
Table 5.
Investigational findings in patients with IIM (key studies)
| Author (year of publication) | Study group (n) | Diagnosis | Study design | Key observation | Country |
|---|---|---|---|---|---|
| Electrocardiogram (ECG) | |||||
| Deveza et al. [29] (2014) | 112 |
DM (n = 78) PM (n = 34) |
Cross-sectional study | One-third of patients had ECG abnormalities namely conduction disorders, chamber enlargement and rhythm disturbances. The rhythm disturbances included ventricular extrasystole, atrial fibrillation, first-degree AV block, supraventricular tachycardia, supraventricular extrasystoles. All these abnormalities were more frequent in PM than in DM (50% vs. 24.4%, p = 0.008). The study, however, did not find any significant difference in ECG abnormalities between patients and controls, except for a higher prevalence of left ventricular hypertrophy in the former | Brazil |
| Triplett et al. [81] (2020) | 109 | IMNM | Retrospective study | In patients with IMNM, an abnormal ECG was documented in 55 out of 86 patients. Of the various abnormalities, prolongation of corrected QT interval (QTc) was the most frequent | USA |
| Cox et al. [82] (2010) | 51 | Sporadic IBM | Cross-sectional study | Patients with IBM did not have increased risk of cardiac involvement compared to general population as evidenced by the similar frequencies of ECG abnormalities between the two groups | Netherlands |
| Wang et al. [83] (2014) | 51 | DM | Patients with DM had no clinically evident cardiovascular disease assessed using Doppler ECG. Authors reported a statistically significant association between LVDD and duration of the disease, pointing towards subclinical cardiac involvement with disease advancement | China | |
| Nuclear Cardiac Imaging | |||||
| Diederichsen et al. [84] (2016) | 76 |
DM (n = 24) PM (n = 52) |
Cross-sectional study | Age, disease duration, presence of myositis specific or associated autoantibodies and high cardiac 99m Tc-PYP uptake were found to be associated with LVDD. This association of LVDD with increased cardiac 99m Tc-PYP uptake indicated myocardial inflammation as a primary cause of cardiac involvement in IIM patients | Denmark |
| Echocardiography | |||||
| Plazak et al. [85] (2011) | 15 | DM/PM | Cross-sectional study | This study of patients with autoimmune disease included 15 DM/PM patients. Pathologic valvular leaflet thickening and/or pericardial thickening was found in 46.7% of patients with DM/PM. Pericardial effusion was also reported in 66.7% of DM/PM patients. Patients with DM/PM had dilated right ventricle with elevated right ventricular systolic pressure | Poland |
| Zhong et al. [86] (2017) | 60 | DM/PM | Cross-sectional study | In a study of 60 PM/DM patients with preserved LVEF, speckle tracking echocardiography demonstrated subtle systolic dysfunction. The severity of cardiac involvement was related to the systemic disease burden | China |
| Guerra et al. [87] (2017) | 28 | DM/PM | Case control study | The authors reported a 4.9-fold higher risk of subclinical ventricular systolic dysfunction in IIM patients compared to healthy controls using speckle tracking echocardiography. The basal and mid-segments of the anterior, anterior-septal, and lateral wall of the left ventricle were most frequently involved. The presence of cardiac involvement was not associated with disease duration or disease activity | Italy |
| Liu et al. [88] (2022) | 46 | DM/PM | Observational study | Using speckle tracking echocardiography, it was noted that in patients with PM and DM, the myocardium at the base was more severely affected than at the apex. This pattern of basal weakness accurately differentiated myocardial involvement of DM and PM from acute viral myocarditis | China |
| Cardiac magnetic resonance | |||||
| Khoo et al. [89] (2019) | 19 |
DM (n = 4) PM (n = 4) IBM (n = 2) IMNM (n = 2) Myositis not otherwise specified (n = 4) Overlap myositis (n = 2) DM/SSc overlap (n = 1) |
Cohort study | Despite being asymptomatic for cardiac disease, almost 50% of patients with IIM had apparent cardiac involvement on CMR in the form of late gadolinium enhancement (LGE). Except for one patient, all patients showed varying degrees of cardiac fibrosis | Australia |
| Sun et al. [90] (2021) | 51 |
DM (n = 19) PM (n = 20) NM (n = 12) |
Observational study | This study demonstrated more severe LGE lesions in PM patients when compared to patients with DM. Patient with necrotizing myositis (NM) did not demonstrate late gadolinium enhancement | China |
| Kersten et al. [91] (2021) | 47 |
DM (n = 10) PM (n = 31) Other sub-forms of IIM (n = 6) |
Cohort study | Patients with IIM had lower myocardial deformation parameters (indicative of impaired myocardial function) when compared to healthy volunteers using cardiac magnetic resonance imaging. In this study there was no significant difference in the LVEF between patients and healthy volunteers, suggesting that change in myocardial deformation may precede changes in LVEF | Germany |
| Rosenbohm et al. [92] (2020) | 20 | IBM | Case series of 20 patients | In this case series of 20 patients with sporadic IBM, CMR demonstrated decreased left and right ventricular stroke volumes and an increased early myocardial enhancement when compared to controls. These changes were attributed to the hypertensive heart disease present in these patients. There was no statistical difference in LGE between patients and controls | Germany |
| Cardiac enzymes | |||||
| Lillekar et al. [93] (2018) | 123 |
DM (n = 39) PM (n = 34) ASS (n = 37) IMNM (n = 8) IIM-CTD overlap disease (n = 5) |
Cross sectional study | Cardiac troponin I (cTnI) levels were higher in patients with cardiac involvement, irrespective of disease activity. cTnI had the highest specificity (95%) and positive predictive value (62%) for detecting cardiac involvement, however, it lacked sensitivity. Cardiac troponin T (cTnT) correlated with the patient and evaluator global assessment and the quality of life as assessed by Health Assessment Questionnaire (HAQ) more strongly than cTnI and creatinine kinase (CK) | UK and Denmark |
| Sun et al. [90] (2021) | 51 |
DM (n = 19) PM (n = 20) IMNM (n = 12) |
Observational study | In a study of 51 IIM patients (19 patients with DM, 20 patients with PM and 12 patients with IMNM), patients with IMNM had very high serum markers of cardiac damage (CK-MB and cTnT) as compared to patients with PM and DM. The study showed that serum CK-MB and cTnT did not accurately reflect myocardial involvement in IIM, however, NT-pro BNP correlated positively with CMR finding of LGE. | China |
Given that cardiovascular involvement is one of the most common causes of increased morbidity and mortality in patients with IIM, screening of patients for cardiac involvement is of paramount importance. As most patients have subclinical cardiac involvement, the need for highly sensitive and noninvasive testing methods is gaining importance.
Electrocardiogram (ECG)
Electrocardiographic changes are common in patients with IIM [94], with abnormalities detected in approximately 32.5–72% of patients [9]. In a study by Deveza et al., one-third of patients with DM and PM had electrocardiographic abnormalities including conduction disorders, chamber enlargement, and rhythm disturbances. The various rhythm disturbances observed were ventricular extrasystoles, atrial fibrillation, first-degree AV block, supraventricular tachycardia, and supraventricular extrasystoles. These abnormalities were more frequent in PM than in DM (50% versus 24.4%, p = 0.008]. However, the study did not find any significant difference in electrocardiographic abnormalities between patients and control groups, except for a higher prevalence of left ventricular hypertrophy in the former [29]. In a study of IMNM, 55 out of 86 patients had an abnormal electrocardiography. Of the various abnormalities, prolongation of the corrected QT interval (QTc) was the most frequent [81].
Conversely, patients with sporadic inclusion body myositis do not appear to have increased risk of cardiac involvement compared to the general population, as evidenced by the similar frequencies of ECG abnormalities between the two groups [82].
Echocardiography
Echocardiographic abnormalities are reported in 14–65% of patients with IIM [3]. Left ventricular diastolic dysfunction (LVDD) is the most common echocardiographic abnormality [61, 81, 84, 95, 96]. Several studies have demonstrated that IIM patients have a greater prevalence of LVDD compared to the general population [83, 84, 95]. A study by Wang et al. reported a statistically significant association between LVDD and disease duration, pointing towards increased subclinical cardiac involvement with disease advancement [83]. Diederichsen et al. reported that age, disease duration, presence of myositis-specific or myositis-associated autoantibodies, and high cardiac 99m Tc-PYP uptake are associated with LVDD. This association between LVDD and increased cardiac 99m Tc-PYP uptake indicated myocardial inflammation as a primary cause of cardiac involvement in patients with IIM [84].
In a study of patients with generalized autoimmune disease (comprising 60 patients with scleroderma, 60 patients with systemic lupus erythematosus, and 15 patients with DM/PM) pathologic valvular leaflet thickening and/or pericardial thickening on echocardiography was found in 46.7% of patients with DM/PM. Pericardial effusion was also reported in 66.7% of the patients with DM/PM. All 15 patients with DM/PM had dilated right ventricle with elevated right ventricular systolic pressure [85].
Left ventricular systolic function has long been assessed using left ventricular ejection fraction (LVEF). However, LVEF has limited sensitivity in detecting early subclinical cardiac involvement, as it typically does not worsen until cardiac function is severely compromised. Speckle tracking echocardiography is an emerging modality for detection of subclinical cardiac involvement by directly assessing myocardial wall deformation [97]. Echocardiographic assessment using the long axis strain and not LVEF for evaluating systolic involvement in patients with IIM has been recommended [94].
In a study of 60 patients with PM/DM with preserved LVEF, speckle-tracking echocardiography demonstrated subtle systolic dysfunction. The severity of cardiac involvement was related to the systemic disease burden [86]. Guerra et al. reported a 4.9-fold higher risk of subclinical ventricular systolic dysfunction in patients with IIM than to healthy controls using speckle tracking echocardiography. The basal and mid-segments of the anterior, anteroseptal, and lateral walls of the left ventricle were the most frequently involved. However, in their study, the cardiac involvement was not associated with disease duration or disease activity [87]. In a study by Liu et al. using speckle tracking echocardiography, it was noted that in patients with PM and DM, the myocardium at the base was more severely affected than at the apex. This pattern of basal weakness was successful in accurately differentiating myocardial involvement of DM and PM from acute viral myocarditis [88].
Nuclear cardiac imaging
Nuclear cardiac imaging can be used to detect myocarditis and myocardial damage from past ischaemic insults. A retrospective study by Okada et al. examined the usefulness of 123I-BMIPP, which reflects fatty acid metabolism, and 201TlCl (201 thallium chloride), which reflects myocardial perfusion, scintigraphy for detecting myocarditis in 26 PM or DM patients. Half of the patients demonstrated 123I-BMIPP/ 201TICI mismatch, and these patients had significantly lower LVEF. They suggested 123I-BMIPP and 201TICI nuclide scintigraphy could be contemplated for the evaluation of myocarditis in patients with IIM [98].
Cardiac magnetic resonance
Cardiac magnetic resonance (CMR) imaging is considered the most reliable noninvasive modality for the detection of myocarditis and myocardial fibrosis in patients with IIM [57]. Khoo et al. reported that almost 50% of patients with IIM had apparent cardiac involvement on CMR imaging, as evidenced by late gadolinium enhancement (LGE), despite being asymptomatic for cardiac disease. Except for one patient, all participants demonstrated varying degrees of fibrosis in this study [89]. Huber et al. demonstrated that CMR T1 mapping parameters in myocardial and skeletal muscles can be used to differentiate between acute viral myocarditis and IIM related myocarditis [99]. An observational study by Sun et al. demonstrated more severe LGE lesions in patients with PM than in patients with DM. Conversely, patients with IMNM did not demonstrate late gadolinium enhancement [90]. Kersten et al. reported lower myocardial deformation parameters in patients with IIM than in healthy volunteers. Notably, there was no significant difference in the LVEF between patients and healthy volunteers, suggesting that changes in myocardial deformation may precede changes in LVEF [91].
Cardiac magnetic resonance (CMR) imaging has shown promise in the early detection of cardiac involvement in patients with IIM. A case series of 53 patients with PM, DM or non-specific myositis reported myocardial LGE patterns that were consistent with inflammation. Despite a normal LVEF, 54.5% of the patients had positive LGE. There was no significant correlation between the clinical symptoms and CMR results [92]. Several studies have demonstrated the superiority of multiparametric CMR approaches to conventional methods (ECG and echocardiography) in early detection of cardiac involvement, even in patients with negative LGE [100–102]. Based on their study evaluating CMR in patients of IIM at first diagnosis and after a year of receiving standard treatment, Xu et al. suggested that multiparametric CMR could help monitor response to effective treatment [103]. In a case series of 20 patients with sporadic IBM, CMR demonstrated decreased left and right ventricular stroke volumes and increased early myocardial enhancement compared to controls. However, these changes were attributed to hypertensive heart disease in these patients. Of note, there was no statistical difference in the LGE between patients and controls [104].
Cardiac enzymes
Cardiac troponins are highly sensitive markers of cardiac involvement, particularly infarction. A retrospective study by Aggarwal et al. reported elevated cardiac troponin T (cTnT), but not cardiac troponin I (cTnI), in patients with IIM. cTnT levels correlated with total creatine kinase (CK) levels in this study [105]. Of note, cTnT can arise from the skeletal muscles of patients with IIM, which may explain why this enzyme correlates with muscle disease activity [93, 106]. Conversely, in a study by Lilleker et al., cTnI levels were higher in patients with cardiac involvement, irrespective of IIM disease activity. cTnI had the highest specificity (95%) and positive predictive value (62%) for the detection of cardiac involvement, although this test lacked sensitivity. cTnT correlated more strongly with the patient and evaluator global assessment and the quality of life as assessed by the Health Assessment Questionnaire (HAQ) than cTnI and CK [93].
In a study of 51 patients with IIM (19 patients with DM, 20 patients with PM, 12 patients with IMNM) patients with IMNM had significantly higher cTnT and CK, but lower N-terminal prohormone of brain natriuretic peptide (NT-proBNP) levels and no positive LGE on CMR when compared to patients with PM and DM, again suggesting that serum CK-MB and cTnT do not accurately reflect myocardial involvement in IIM. NT-pro BNP correlated positively with CMR finding of LGE [90].
Although several diagnostic methods are available for investigating cardiac involvement in patients with IIM, traditional methods such as ECG and echocardiography are not sensitive to detect subclinical and early changes. CMR has proven to be sensitive for the detection of subclinical cardiac involvement, the most frequently encountered cardiac abnormality. Data on nuclear cardiac imaging for detection of myocarditis in patients with IIM is very limited, and needs to be studied further.
Conclusion
Cardiovascular risk factors are prevalent in individuals diagnosed with IIM. Although clinically apparent cardiac involvement is uncommon in patients with IIM, cardiovascular involvement is a major contributor to morbidity and mortality. It remains unclear whether the excess cardiovascular morbidity and mortality seen in IIM is a result of increased prevalence of cardiovascular risk factors, chronic systemic inflammation or subclinical cardiac inflammation. Further research may help understand the relative contribution of these mechanisms.
Of concern, subclinical cardiac involvement may not be identified using conventional investigations such as electrocardiography or echocardiography. Advances in highly sensitive imaging platforms such as CMR, coupled with the increasing availability of these modalities has made it easier to detect subclinical cardiac involvement; however, larger studies are needed to assess the feasibility of employing such imaging modalities in the routine screening of patients with IIM. This scoping review identifies a pressing need to develop guidelines for screening cardiac involvement and cardiovascular risk factors in patients with IIM.
Supplementary information
(DOCX 18 kb)
Acknowledgements
JD is the recipient of the Sylvia and Charles Viertel Charitable Foundation Clinical Investigator Award, the John T Reid Charitable Trust Centenary Fellowship and the RACP Australian Rheumatology Association & D.E.V Starr Research Establishment Fellowship. Dr. Prakash Gupta for this excellent contribution in the literature search strategy.
Author contribution
Conceptualization: LG; literature search: MS and LG; writing—original draft: MS; writing—review and editing: all authors.
Compliance with ethical standards
Disclosure
None.
Disclaimer
No part of this review was copied or published elsewhere.
Footnotes
Part of the Topical Collection entitled ‘Cardiovascular Issues in Rheumatic Diseases’
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ashton C, Paramalingam S, Stevenson B, et al. Idiopathic inflammatory myopathies: a review. Intern Med J. 2021;51:845–852. doi: 10.1111/imj.15358. [DOI] [PubMed] [Google Scholar]
- 2.Dobloug GC, Svensson J, Lundberg IE, Holmqvist M. Mortality in idiopathic inflammatory myopathy: results from a Swedish nationwide population-based cohort study. Ann Rheum Dis. 2018;77:40–47. doi: 10.1136/annrheumdis-2017-211402. [DOI] [PubMed] [Google Scholar]
- 3.Marie I. Morbidity and mortality in adult polymyositis and dermatomyositis. Curr Rheumatol Rep. 2012;14:275–285. doi: 10.1007/s11926-012-0249-3. [DOI] [PubMed] [Google Scholar]
- 4.Maldonado F, Patel RR, Iyer VN, et al. Are respiratory complications common causes of death in inflammatory myopathies? An autopsy study. Respirology. 2012;17:455–460. doi: 10.1111/j.1440-1843.2011.02103.x. [DOI] [PubMed] [Google Scholar]
- 5.Raggi P, Genest J, Giles JT, et al. Role of inflammation in the pathogenesis of atherosclerosis and therapeutic interventions. Atherosclerosis. 2018;276:98–108. doi: 10.1016/j.atherosclerosis.2018.07.014. [DOI] [PubMed] [Google Scholar]
- 6.Pedro-Botet J, Climent E, Benaiges D. Atherosclerosis and inflammation. New therapeutic approaches. Med Clin. 2020;155:256–262. doi: 10.1016/j.medcli.2020.04.024. [DOI] [PubMed] [Google Scholar]
- 7.Gasparyan AY, Ayvazyan L, Blackmore H, Kitas GD. Writing a narrative biomedical review: considerations for authors, peer reviewers, and editors. Rheumatol Int. 2011;31:1409–1417. doi: 10.1007/s00296-011-1999-3. [DOI] [PubMed] [Google Scholar]
- 8.Oppenheim H. Zur dermatomyositis. Berl Klin Wochenschr. 1899;36:805–807. [Google Scholar]
- 9.Lundberg IE. The heart in dermatomyositis and polymyositis. Rheumatology. 2006;45:iv18–iv21. doi: 10.1093/rheumatology/kel311. [DOI] [PubMed] [Google Scholar]
- 10.Fairley JL, Wicks I, Peters S, Day J. Defining cardiac involvement in idiopathic inflammatory myopathies: a systematic review. Rheumatology (Oxford) 2021;61:103–120. doi: 10.1093/rheumatology/keab573. [DOI] [PubMed] [Google Scholar]
- 11.Lilleker JB, Vencovsky J, Wang G, et al. The EuroMyositis registry: an international collaborative tool to facilitate myositis research. Ann Rheum Dis. 2018;77:30–39. doi: 10.1136/annrheumdis-2017-211868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Limaye V, Hakendorf P, Woodman RJ, et al. Mortality and its predominant causes in a large cohort of patients with biopsy-determined inflammatory myositis. Intern Med J. 2012;42:191–198. doi: 10.1111/j.1445-5994.2010.02406.x. [DOI] [PubMed] [Google Scholar]
- 13.Dankó K, Ponyi A, Constantin T, et al. Long-term survival of patients with idiopathic inflammatory myopathies according to clinical features: a longitudinal study of 162 cases. Medicine. 2004;83:35–42. doi: 10.1097/01.md.0000109755.65914.5e. [DOI] [PubMed] [Google Scholar]
- 14.Jung K, Kim H, Park W, et al. Incidence, survival, and risk of cardiovascular events in adult inflammatory myopathies in South Korea: a nationwide population-based study. Scand J Rheumatol. 2020;49:323–331. doi: 10.1080/03009742.2019.1707281. [DOI] [PubMed] [Google Scholar]
- 15.Xiong A, Hu Z, Zhou S, et al. Cardiovascular events in adult polymyositis and dermatomyositis: a meta-analysis of observational studies. Rheumatology (Oxford) 2022;61:2728–2739. doi: 10.1093/rheumatology/keab851. [DOI] [PubMed] [Google Scholar]
- 16.Ungprasert P, Suksaranjit P, Spanuchart I, et al. Risk of coronary artery disease in patients with idiopathic inflammatory myopathies: a systematic review and meta-analysis of observational studies. Semin Arthritis Rheum. 2014;44:63–67. doi: 10.1016/j.semarthrit.2014.03.004. [DOI] [PubMed] [Google Scholar]
- 17.Párraga Prieto C, Ibrahim F, Campbell R, et al. Similar risk of cardiovascular events in idiopathic inflammatory myopathy and rheumatoid arthritis in the first 5 years after diagnosis. Clin Rheumatol. 2021;40:231–238. doi: 10.1007/s10067-020-05237-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Barsotti S, Saponaro C, Gaggini M, et al. Cardiometabolic risk and subclinical vascular damage assessment in idiopathic inflammatory myopathies: a challenge for the clinician. Clin Exp Rheumatol. 2019;37:1036–1043. [PubMed] [Google Scholar]
- 19.Wang H, Cai Y, Cai L, et al. Altered lipid levels in untreated patients with early polymyositis. PloS One. 2014;9:e89827. doi: 10.1371/journal.pone.0089827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Diederichsen LP, Diederichsen ACP, Simonsen JA, et al. Traditional cardiovascular risk factors and coronary artery calcification in adults with polymyositis and dermatomyositis: a danish multicenter study. Arthritis Care Res. 2015;67:848–854. doi: 10.1002/acr.22520. [DOI] [PubMed] [Google Scholar]
- 21.Bae SS, Lee YY, Shahbazian A, et al. High-density lipoprotein function is abnormal in idiopathic inflammatory myopathies. Rheumatology. 2020;59:3515–3525. doi: 10.1093/rheumatology/keaa273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Qin L, Li F, Luo Q et al (2022) Coronary heart disease and cardiovascular risk factors in patients with idiopathic inflammatory myopathies: a systemic review and meta-analysis. Front Med 8. 10.3389/fmed.2021.808915 [DOI] [PMC free article] [PubMed]
- 23.Pakhchanian H, Khan H, Raiker R, et al. COVID-19 outcomes in patients with dermatomyositis: a registry-based cohort analysis. Semin Arthritis Rheum. 2022;56:152034. doi: 10.1016/j.semarthrit.2022.152034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Oreska S, Storkanova H, Kudlicka J, et al. Cardiovascular risk in myositis patients compared to the general population: preliminary data from a single-center cross-sectional study. Front Med. 2022;9:861419. doi: 10.3389/fmed.2022.861419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.de Moraes MT, de Souza FHC, de Barros TBM, Shinjo SK. Analysis of metabolic syndrome in adult dermatomyositis with a focus on cardiovascular disease. Arthritis Care Res. 2013;65:793–799. doi: 10.1002/acr.21879. [DOI] [PubMed] [Google Scholar]
- 26.de Souza FHC, Shinjo SK. The high prevalence of metabolic syndrome in polymyositis. Clin Exp Rheumatol. 2014;32:82–87. [PubMed] [Google Scholar]
- 27.Araujo PAO, Silva MG, Borba EF, Shinjo SK. High prevalence of metabolic syndrome in antisynthetase syndrome. Clin Exp Rheumatol. 2018;36:241–247. [PubMed] [Google Scholar]
- 28.Naaraayan A, Meredith A, Nimkar A, et al. Arrhythmia prevalence among patients with polymyositis–dermatomyositis in the United States: an observational study. Heart Rhythm. 2021;18:1516–1523. doi: 10.1016/j.hrthm.2021.05.029. [DOI] [PubMed] [Google Scholar]
- 29.Deveza LMA, Miossi R, de Souza FHC, et al. Electrocardiographic changes in dermatomyositis and polymyositis. Rev Bras Reumatol Engl Ed. 2016;56:95–100. doi: 10.1016/j.rbre.2014.08.012. [DOI] [PubMed] [Google Scholar]
- 30.Huang Y, Liu H, Wu C, et al. Ventricular arrhythmia predicts poor outcome in polymyositis/dermatomyositis with myocardial involvement. Rheumatology (Oxford) 2021;60:3809–3816. doi: 10.1093/rheumatology/keaa872. [DOI] [PubMed] [Google Scholar]
- 31.Patel KHK, Jones TN, Sattler S, et al. Proarrhythmic electrophysiological and structural remodeling in rheumatoid arthritis. Am J Physiol Heart Circ Physiol. 2020;319:H1008–H1020. doi: 10.1152/ajpheart.00401.2020. [DOI] [PubMed] [Google Scholar]
- 32.Haupt HM, Hutchins GM. The heart and cardiac conduction system in polymyositis-dermatomyositis: a clinicopathologic study of 16 autopsied patients. Am J Cardiol. 1982;50:998–1006. doi: 10.1016/0002-9149(82)90408-8. [DOI] [PubMed] [Google Scholar]
- 33.Xu J, Lupu F, Esmon CT. Inflammation, innate immunity and blood coagulation. Hamostaseologie. 2010;30(5–6):8–9. [PubMed] [Google Scholar]
- 34.Ramagopalan SV, Wotton CJ, Handel AE, et al. Risk of venous thromboembolism in people admitted to hospital with selected immune-mediated diseases: record-linkage study. BMC Med. 2011;9:1. doi: 10.1186/1741-7015-9-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Holmqvist ME, Neovius M, Eriksson J, et al. Risk of venous thromboembolism in patients with rheumatoid arthritis and association with disease duration and hospitalization. JAMA. 2012;308:1350–1356. doi: 10.1001/2012.jama.11741. [DOI] [PubMed] [Google Scholar]
- 36.Cervera R, Khamashta MA, Font J, et al. Morbidity and mortality in systemic lupus erythematosus during a 10-year period: a comparison of early and late manifestations in a cohort of 1,000 patients. Medicine. 2003;82:299–308. doi: 10.1097/01.md.0000091181.93122.55. [DOI] [PubMed] [Google Scholar]
- 37.Zöller B, Li X, Sundquist J, Sundquist K. Autoimmune diseases and venous thromboembolism: a review of the literature. Am J Cardiovasc Dis. 2012;2:171–183. [PMC free article] [PubMed] [Google Scholar]
- 38.Lee JJ, Pope JE. A meta-analysis of the risk of venous thromboembolism in inflammatory rheumatic diseases. Arthritis Res Ther. 2014;16:435. doi: 10.1186/s13075-014-0435-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chung W-S, Lin C-L, Sung F-C, et al. Increased risk of venous thromboembolism in patients with dermatomyositis/polymyositis: a nationwide cohort study. Thromb Res. 2014;134:622–626. doi: 10.1016/j.thromres.2014.06.021. [DOI] [PubMed] [Google Scholar]
- 40.Li Y, Wang P, Li L, et al. Increased risk of venous thromboembolism associated with polymyositis and dermatomyositis: a meta-analysis. Ther Clin Risk Manag. 2018;14:157–165. doi: 10.2147/TCRM.S157085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lee YH, Song GG. Idiopathic inflammatory myopathy and the risk of venous thromboembolism: a meta-analysis. Rheumatol Int. 2017;37:1165–1173. doi: 10.1007/s00296-017-3735-0. [DOI] [PubMed] [Google Scholar]
- 42.Ungprasert P, Sanguankeo A. Risk of venous thromboembolism in patients with idiopathic inflammatory myositis: a systematic review and meta-analysis. Rheumatol Int. 2014;34:1455–1458. doi: 10.1007/s00296-014-3023-1. [DOI] [PubMed] [Google Scholar]
- 43.Kuszmiersz P, Siwiec-Koźlik A, Pacholczak-Madej R, et al. Thrombin generation potential is increased in patients with autoimmune inflammatory myopathies. Adv Med Sci. 2022;67:346–352. doi: 10.1016/j.advms.2022.08.005. [DOI] [PubMed] [Google Scholar]
- 44.Carruthers EC, Choi HK, Sayre EC, Aviña-Zubieta JA. Risk of deep venous thrombosis and pulmonary embolism in individuals with polymyositis and dermatomyositis: a general population-based study. Ann Rheum Dis. 2016;75:110–116. doi: 10.1136/annrheumdis-2014-205800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen X, Huang S, Jin Q, et al. Thromboembolic events in idiopathic inflammatory myopathy: a retrospective study in China. Clin Rheumatol. 2022;41:2431–2438. doi: 10.1007/s10067-022-06140-z. [DOI] [PubMed] [Google Scholar]
- 46.Antovic A, Notarnicola A, Svensson J, et al. Venous thromboembolic events in idiopathic inflammatory myopathy: occurrence and relation to disease onset. Arthritis Care Res. 2018;70:1849–1855. doi: 10.1002/acr.23560. [DOI] [PubMed] [Google Scholar]
- 47.Notarnicola A, Barsotti S, Näsman L, et al. Evaluation of risk factors and biomarkers related to arterial and venous thrombotic events in idiopathic inflammatory myopathies. Scand J Rheumatol. 2021;50:390–397. doi: 10.1080/03009742.2020.1861647. [DOI] [PubMed] [Google Scholar]
- 48.Bleau N, Patenaude V, Abenhaim HA. Risk of venous thromboembolic events in pregnant patients with autoimmune diseases: a population-based study. Clin Appl Thromb Hemost. 2016;22:285–291. doi: 10.1177/1076029614553023. [DOI] [PubMed] [Google Scholar]
- 49.Linos E, Fiorentino D, Lingala B et al (2013) Atherosclerotic cardiovascular disease and dermatomyositis: an analysis of the Nationwide Inpatient Sample survey. Arthritis Res Ther 15. 10.1186/ar4135 [DOI] [PMC free article] [PubMed]
- 50.Weng M-Y, Lai EC-C, Yang Y-HK. Increased risk of coronary heart disease among patients with idiopathic inflammatory myositis: a nationwide population study in Taiwan. Rheumatology. 2019;58:1935–1941. doi: 10.1093/rheumatology/kez076. [DOI] [PubMed] [Google Scholar]
- 51.Leclair V, Svensson J, Lundberg IE, Holmqvist M. Acute coronary syndrome in idiopathic inflammatory myopathies: a population-based study. J Rheumatol. 2019;46:1509–1514. doi: 10.3899/jrheum.181248. [DOI] [PubMed] [Google Scholar]
- 52.Rai SK, Choi HK, Sayre EC, Aviña-Zubieta JA. Risk of myocardial infarction and ischaemic stroke in adults with polymyositis and dermatomyositis: a general population-based study. Rheumatology (Oxford) 2016;55:461–469. doi: 10.1093/rheumatology/kev336. [DOI] [PubMed] [Google Scholar]
- 53.Lin Y-N, Lin C-L, Chang K-C, Kao C-H. Increased subsequent risk of acute coronary syndrome for patients with dermatomyositis/polymyositis: a nationwide population-based retrospective cohort study. Scand J Rheumatol. 2015;44:42–47. doi: 10.3109/03009742.2014.918652. [DOI] [PubMed] [Google Scholar]
- 54.Lai Y-T, Dai Y-S, Yen M-F, et al. Dermatomyositis is associated with an increased risk of cardiovascular and cerebrovascular events: a Taiwanese population-based longitudinal follow-up study. Br J Dermatol. 2013;168:1054–1059. doi: 10.1111/bjd.12245. [DOI] [PubMed] [Google Scholar]
- 55.Tisseverasinghe A, Bernatsky S, Pineau CA. Arterial events in persons with dermatomyositis and polymyositis. J Rheumatol. 2009;36:1943–1946. doi: 10.3899/jrheum.090061. [DOI] [PubMed] [Google Scholar]
- 56.Lai EC-C, Huang Y-C, Liao T-C, Weng M-Y (2022) Premature coronary artery disease in patients with immune-mediated inflammatory disease: a population-based study. RMD Open 8. 10.1136/rmdopen-2021-001993 [DOI] [PMC free article] [PubMed]
- 57.Jayakumar D, Zhang R, Wasserman A, Ash J. Cardiac manifestations in idiopathic inflammatory myopathies: an overview. Cardiol Rev. 2019;27:131–137. doi: 10.1097/CRD.0000000000000241. [DOI] [PubMed] [Google Scholar]
- 58.Lin C-Y, Chen H-A, Hsu T-C, et al. Time-dependent analysis of risk of new-onset heart failure among patients with polymyositis and dermatomyositis. Arthritis Rheumatol. 2022;74:140–149. doi: 10.1002/art.41907. [DOI] [PubMed] [Google Scholar]
- 59.Zhang L, Zhu H, Yang P, et al. Myocardial involvement in idiopathic inflammatory myopathies: a multi-center cross-sectional study in the CRDC-MYO Registry. Clin Rheumatol. 2021;40:4597–4608. doi: 10.1007/s10067-021-05828-y. [DOI] [PubMed] [Google Scholar]
- 60.Liu Y, Fang L, Chen W, et al. Clinical characteristics, treatment, and outcomes in patients with idiopathic inflammatory myopathy concomitant with heart failure. Int Heart J. 2020;61:1005–1013. doi: 10.1536/ihj.19-568. [DOI] [PubMed] [Google Scholar]
- 61.Gupta R, Wayangankar SA, Targoff IN, Hennebry TA. Clinical cardiac involvement in idiopathic inflammatory myopathies: a systematic review. Int J Cardiol. 2011;148:261–270. doi: 10.1016/j.ijcard.2010.08.013. [DOI] [PubMed] [Google Scholar]
- 62.Dieval C, Deligny C, Meyer A, et al. Myocarditis in patients with antisynthetase syndrome: prevalence, presentation, and outcomes. Medicine. 2015;94:e798. doi: 10.1097/MD.0000000000000798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Liu Y, Fang L, Chen W, et al. Identification of characteristics of overt myocarditis in adult patients with idiopathic inflammatory myopathies. Cardiovasc Diagn Ther. 2020;10:405–420. doi: 10.21037/cdt.2020.03.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhang L, Yang H, Lei J, et al. Muscle pathological features and extra-muscle involvement in idiopathic inflammatory myopathies with anti-mitochondrial antibody. Semin Arthritis Rheum. 2021;51:741–748. doi: 10.1016/j.semarthrit.2021.05.019. [DOI] [PubMed] [Google Scholar]
- 65.Højgaard P, Witting N, Rossing K, et al. Cardiac arrest in anti-mitochondrial antibody associated inflammatory myopathy. Oxf Med Case Reports. 2021;2021:89–93. doi: 10.1093/omcr/omaa150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Korsholm SS, Andersson DC, Knudsen JB et al (2022) Myositis-specific autoantibodies and QTc changes by ECG in idiopathic inflammatory myopathies. Rheumatology (Oxford) keac013. 10.1093/rheumatology/keac013 [DOI] [PubMed]
- 67.Sabbagh SE, Pinal-Fernandez I, Casal-Dominguez M, et al. Anti-mitochondrial autoantibodies are associated with cardiomyopathy, dysphagia, and features of more severe disease in adult-onset myositis. Clin Rheumatol. 2021;40:4095–4100. doi: 10.1007/s10067-021-05730-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Albayda J, Khan A, Casciola-Rosen L, et al. Inflammatory myopathy associated with anti-mitochondrial antibodies: a distinct phenotype with cardiac involvement. Semin Arthritis Rheum. 2018;47:552–556. doi: 10.1016/j.semarthrit.2017.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Matsuo T, Sasai T, Nakashima R, et al. ECG changes through immunosuppressive therapy indicate cardiac abnormality in anti-MDA5 antibody-positive clinically amyopathic dermatomyositis. Front Immunol. 2021;12:765140. doi: 10.3389/fimmu.2021.765140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Coyle K, Rother KI, Weise M, et al. Metabolic abnormalities and cardiovascular risk factors in children with myositis. J Pediatr. 2009;155:882–887. doi: 10.1016/j.jpeds.2009.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Silverberg JI, Kwa L, Kwa MC, et al. Cardiovascular and cerebrovascular comorbidities of juvenile dermatomyositis in US children: an analysis of the National Inpatient Sample. Rheumatology (Oxford) 2018;57:694–702. doi: 10.1093/rheumatology/kex465. [DOI] [PubMed] [Google Scholar]
- 72.Kozu KT, Silva CA, Bonfá E, et al. Dyslipidaemia in juvenile dermatomyositis: the role of disease activity. Clin Exp Rheumatol. 2013;31:638–644. [PubMed] [Google Scholar]
- 73.Witczak BN, Schwartz T, Barth Z, et al. Associations between cardiac and pulmonary involvement in patients with juvenile dermatomyositis—a cross-sectional study. Rheumatol Int. 2022;42:1213–1220. doi: 10.1007/s00296-021-05071-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Witczak BN, Bollerslev J, Godang K, et al. Body composition in long-standing juvenile dermatomyositis: associations with disease activity, muscle strength and cardiometabolic measures. Rheumatology (Oxford) 2022;61:2959–2968. doi: 10.1093/rheumatology/keab805. [DOI] [PubMed] [Google Scholar]
- 75.Barth Z, Schwartz T, Flatø B, et al. Association between nailfold capillary density and pulmonary and cardiac involvement in medium to longstanding juvenile dermatomyositis. Arthritis Care Res. 2019;71:492–497. doi: 10.1002/acr.23687. [DOI] [PubMed] [Google Scholar]
- 76.Schwartz T, Sanner H, Gjesdal O, et al. In juvenile dermatomyositis, cardiac systolic dysfunction is present after long-term follow-up and is predicted by sustained early skin activity. Ann Rheum Dis. 2014;73:1805–1810. doi: 10.1136/annrheumdis-2013-203279. [DOI] [PubMed] [Google Scholar]
- 77.Barth Z, Nomeland Witczak B, Schwartz T, et al. In juvenile dermatomyositis, heart rate variability is reduced, and associated with both cardiac dysfunction and markers of inflammation: a cross-sectional study median 13.5 years after symptom onset. Rheumatology. 2016;55:535–543. doi: 10.1093/rheumatology/kev376. [DOI] [PubMed] [Google Scholar]
- 78.Swafford C, Roach ES. Juvenile dermatomyositis and the inflammatory myopathies. Semin Neurol. 2020;40:342–348. doi: 10.1055/s-0040-1705120. [DOI] [PubMed] [Google Scholar]
- 79.Schwartz T, Sanner H, Husebye T, et al. Cardiac dysfunction in juvenile dermatomyositis: a case-control study. Ann Rheum Dis. 2011;70:766–771. doi: 10.1136/ard.2010.137968. [DOI] [PubMed] [Google Scholar]
- 80.Huikuri HV, Stein PK. Heart rate variability in risk stratification of cardiac patients. Prog Cardiovasc Dis. 2013;56:153–159. doi: 10.1016/j.pcad.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 81.Triplett J, Kassardjian CD, Liewluck T, et al. Cardiac and respiratory complications of necrotizing autoimmune myopathy. Mayo Clin Proc. 2020;95:2144–2149. doi: 10.1016/j.mayocp.2020.03.032. [DOI] [PubMed] [Google Scholar]
- 82.Cox FM, Delgado V, Verschuuren JJ, et al. The heart in sporadic inclusion body myositis: a study in 51 patients. J Neurol. 2010;257:447–451. doi: 10.1007/s00415-009-5350-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Wang H, Liu H-X, Wang Y-L, et al. Left ventricular diastolic dysfunction in patients with dermatomyositis without clinically evident cardiovascular disease. J Rheumatol. 2014;41:495–500. doi: 10.3899/jrheum.130346. [DOI] [PubMed] [Google Scholar]
- 84.Diederichsen LP, Simonsen JA, Diederichsen AC, et al. Cardiac abnormalities in adult patients with polymyositis or dermatomyositis as assessed by noninvasive modalities. Arthritis Care Res. 2016;68:1012–1020. doi: 10.1002/acr.22772. [DOI] [PubMed] [Google Scholar]
- 85.Plazak W, Kopec G, Tomkiewicz-Pajak L, et al. Heart structure and function in patients with generalized autoimmune diseases: echocardiography with tissue Doppler study. Acta Cardiol. 2011;66:159–165. doi: 10.2143/AC.66.2.2071246. [DOI] [PubMed] [Google Scholar]
- 86.Zhong Y, Bai W, Xie Q, et al. Cardiac function in patients with polymyositis or dermatomyositis: a three-dimensional speckle-tracking echocardiography study. Int J Cardiovasc Imaging. 2018;34:683–693. doi: 10.1007/s10554-017-1278-9. [DOI] [PubMed] [Google Scholar]
- 87.Guerra F, Gelardi C, Capucci A, et al. Subclinical cardiac dysfunction in polymyositis and dermatomyositis: a speckle-tracking case-control study. J Rheumatol. 2017;44:815–821. doi: 10.3899/jrheum.161311. [DOI] [PubMed] [Google Scholar]
- 88.Liu X, Jin F, Guo T, et al. Relative basal myocardial weakness: differentiating dermatomyositis and polymyositis with cardiac involvement from viral myocarditis using speckle-tracking echocardiography and quantification of myocardial work. Quant Imaging Med Surg. 2022;12:3762–3777. doi: 10.21037/qims-21-1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Khoo T, Stokes MB, Teo K, et al. Cardiac involvement in idiopathic inflammatory myopathies detected by cardiac magnetic resonance imaging. Clin Rheumatol. 2019;38:3471–3476. doi: 10.1007/s10067-019-04678-z. [DOI] [PubMed] [Google Scholar]
- 90.Sun J, Yin G, Xu Y, et al. Phenotyping of myocardial involvement by cardiac magnetic resonance in idiopathic inflammatory myopathies. Eur Radiol. 2021;31:5077–5086. doi: 10.1007/s00330-020-07448-7. [DOI] [PubMed] [Google Scholar]
- 91.Kersten J, Güleroglu AM, Rosenbohm A, et al. Myocardial involvement and deformation abnormalities in idiopathic inflammatory myopathy assessed by CMR feature tracking. Int J Cardiovasc Imaging. 2021;37:597–603. doi: 10.1007/s10554-020-02020-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rosenbohm A, Buckert D, Gerischer N, et al. Early diagnosis of cardiac involvement in idiopathic inflammatory myopathy by cardiac magnetic resonance tomography. J Neurol. 2015;262:949–956. doi: 10.1007/s00415-014-7623-1. [DOI] [PubMed] [Google Scholar]
- 93.Lilleker JB, Diederichsen ACP, Jacobsen S, et al. Using serum troponins to screen for cardiac involvement and assess disease activity in the idiopathic inflammatory myopathies. Rheumatology (Oxford) 2018;57:1041–1046. doi: 10.1093/rheumatology/key031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Schwartz T, Diederichsen LP, Lundberg IE et al (2016) Cardiac involvement in adult and juvenile idiopathic inflammatory myopathies. RMD Open 2. 10.1136/rmdopen-2016-000291 [DOI] [PMC free article] [PubMed]
- 95.Zhang Y, Yang X, Qin L, et al. Left ventricle diastolic dysfunction in idiopathic inflammatory myopathies: a meta-analysis and systematic review. Mod Rheumatol. 2022;32:589–597. doi: 10.1093/mr/roab041. [DOI] [PubMed] [Google Scholar]
- 96.Danieli MG, Gelardi C, Guerra F, et al. Cardiac involvement in polymyositis and dermatomyositis. Autoimmun Rev. 2016;15:462–465. doi: 10.1016/j.autrev.2016.01.015. [DOI] [PubMed] [Google Scholar]
- 97.Johnson C, Kuyt K, Oxborough D, Stout M. Practical tips and tricks in measuring strain, strain rate and twist for the left and right ventricles. Echo Res Pract. 2019;6:R87–R98. doi: 10.1530/ERP-19-0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Okada Y, Takakuwa Y, Ooka S, et al. Usefulness of 123I-BMIPP and 201TlCl nuclide scintigraphy in evaluation of myocarditis in patients with polymyositis or dermatomyositis. Medicine. 2021;100:e27173. doi: 10.1097/MD.0000000000027173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Huber AT, Bravetti M, Lamy J, et al. Non-invasive differentiation of idiopathic inflammatory myopathy with cardiac involvement from acute viral myocarditis using cardiovascular magnetic resonance imaging T1 and T2 mapping. J Cardiovasc Magn Reson. 2018;20:11. doi: 10.1186/s12968-018-0430-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wang Y, Wang Q, Cao J, et al. Cardiovascular magnetic resonance mapping and strain assessment for the diagnosis of cardiac involvement in idiopathic inflammatory myopathy patients with preserved left ventricular ejection fraction. J Thorac Imaging. 2021;36:254–261. doi: 10.1097/RTI.0000000000000578. [DOI] [PubMed] [Google Scholar]
- 101.Zhao P, Huang L, Ran L, et al. CMR T1 mapping and strain analysis in idiopathic inflammatory myopathy: evaluation in patients with negative late gadolinium enhancement and preserved ejection fraction. Eur Radiol. 2021;31:1206–1215. doi: 10.1007/s00330-020-07211-y. [DOI] [PubMed] [Google Scholar]
- 102.Huang L, Tao Q, Zhao P, et al. Using multi-parametric quantitative MRI to screen for cardiac involvement in patients with idiopathic inflammatory myopathy. Sci Rep. 2022;12:9819. doi: 10.1038/s41598-022-13858-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Xu Y, Sun J, Wan K et al (2020) Multiparametric cardiovascular magnetic resonance characteristics and dynamic changes in myocardial and skeletal muscles in idiopathic inflammatory cardiomyopathy. J Cardiovasc Magn Reson 22. 10.1186/s12968-020-00616-0 [DOI] [PMC free article] [PubMed]
- 104.Rosenbohm A, Buckert D, Kassubek J, et al. Sporadic inclusion body myositis: no specific cardiac involvement in cardiac magnetic resonance tomography. J Neurol. 2020;267:1407–1413. doi: 10.1007/s00415-020-09724-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Aggarwal R, Lebiedz-Odrobina D, Sinha A, et al. Serum cardiac troponin T, but not troponin I, is elevated in idiopathic inflammatory myopathies. J Rheumatol. 2009;36:2711–2714. doi: 10.3899/jrheum.090562. [DOI] [PubMed] [Google Scholar]
- 106.Bodor GS, Survant L, Voss EM, et al. Cardiac troponin T composition in normal and regenerating human skeletal muscle. Clin Chem. 1997;43:476–484. doi: 10.1093/clinchem/43.3.476. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX 18 kb)