Abstract
Background
There is increasing heterogeneity in the clinical phenotype of patients admitted to the intensive care unit (ICU) with coronavirus disease 2019 (COVID-19,) and reasons for mechanical ventilation are not limited to COVID pneumonia. We aimed to compare the characteristics and outcomes of intubated patients admitted to the ICU with the primary diagnosis of acute hypoxemic respiratory failure (AHRF) from COVID-19 pneumonia to those patients admitted for an alternative diagnosis.
Methods
Retrospective cohort study of adults with confirmed SARS-CoV-2 infection admitted to nine ICUs between March 18, 2020, and April 30, 2021, at an urban university institution. We compared characteristics between the two groups using appropriate statistics. We performed logistic regression to identify risk factors for death in the mechanically ventilated COVID-19 population.
Results
After exclusions, the final sample consisted of 319 patients with respiratory failure secondary to COVID pneumonia and 150 patients intubated for alternative diagnoses. The former group had higher ICU and hospital mortality rates (57.7% vs. 36.7%, P<0.001 and 58.9% vs. 39.3%, P<0.001, respectively). Patients with AHRF secondary to COVID-19 pneumonia also had longer ICU and hospital lengths-of-stay (12 vs. 6 days, P<0.001 and 20 vs. 13.5 days, P=0.001). After risk-adjustment, these patients had 2.25 times higher odds of death (95% confidence interval, 1.42–3.56; P=0.001).
Conclusions
Mechanically ventilated COVID-19 patients admitted to the ICU with COVID-19-associated respiratory failure are at higher risk of hospital death and have worse ICU utilization outcomes than those whose reason for admission is unrelated to COVID pneumonia.
Keywords: COVID-19, intensive care unit outcomes, mechanical ventilation
INTRODUCTION
The high prevalence of infection with severe acute respiratory syndrome coronavirus disease 2 (SARS-CoV-2) since the onset of the coronavirus disease 2019 (COVID-19) pandemic in the United States (US) in 2020 has been accompanied by an increase in the heterogeneity of patients admitted to the intensive care unit (ICU) with positive SARS-CoV-2 polymerase chain reaction (PCR) test results. The clinical manifestations of COVID-19 caused by SARS-CoV-2 infection can vary widely, ranging from asymptomatic to severe acute respiratory distress syndrome (ARDS) among those whose infection progresses primarily in the lungs [1].
The bulk of existing literature has focused on the characteristics and outcomes of COVID-19 patients who suffer the hallmark severe respiratory decline that often culminates in ARDS. There are limited data on patients admitted to the ICU with SARS-CoV-2 infection but not necessarily due to severe COVID-19 pneumonia. Therefore, we aim to describe and compare the characteristics and outcomes of mechanically ventilated COVID-19 patients admitted to the ICU with primary diagnosis of acute hypoxemic respiratory failure (AHRF) secondary to COVID-19 pneumonia to those of COVID-19 patients also ultimately requiring intubation but who initially presented to the ICU for a diagnosis other than COVID-19–associated respiratory failure. We anticipated that those patients admitted to the ICU with AHRF secondary to COVID pneumonia would have worse clinical and utilization outcomes.
MATERIALS AND METHODS
We performed a retrospective cohort study of consecutive adult patients ≥18 years of age with COVID-19 who ultimately required invasive mechanical ventilation (IMV) and were admitted to the ICU between March 18, 2020, and April 30, 2021, at Jane and Leonard Korman Respiratory Institute, Thomas Jefferson University. All patients had SARS-CoV-2 infection confirmed by laboratory PCR assay. The study cohort was screened from a larger registry cohort of critically ill patients with COVID-19 who were admitted to one of nine ICUs (99 beds) at three urban university hospitals in a single healthcare system. All ICUs were managed under a high-intensity model of physician staffing [2]. We excluded patients who did not require IMV and those with non-index ICU admissions. Patients were divided into two groups for comparison: those admitted to the ICU with AHRF secondary to COVID pneumonia and those with a positive SARS-CoV-2 PCR result but who were admitted to the ICU for reasons other than COVID-19–associated respiratory failure. To avoid biasing our results, we further excluded patients from the group admitted to the ICU without COVID-19–associated respiratory failure whose ultimate reason for intubation was primary COVID pneumonia, as the outcomes of these patients were more likely to be defined by their phenotype of respiratory failure rather than their initial reason for ICU admission. Trained data abstractors assigned patients into comparator groups based upon detailed chart review. Each case was reviewed by two investigators and screened for discrepancies in designation. In cases where the designation was equivocal, the decision was arbitrated by the most senior physician and primary investigator on the research team (EJY).
We chart-abstracted demographic, clinical, and therapeutic data from the electronic medical records. Data were collected and managed using a secure web-based software platform, Research Electronic Data Capture (REDCap), hosted at our university [3,4] and accessible to key study personnel. To compare values between the two groups with or without COVID-19–associated respiratory failure, we used the chi-square or Fisher’s exact test for categorical variables and Wilcoxon rank-sum test (Mann-Whitney U two-sample test) for continuous variables, as appropriate. To evaluate the impact of the primary predictor of AHRF secondary to COVID pneumonia on mortality in the entire cohort, we conducted logistic regression adjusting for prespecified variables of age, sex, race, body mass index (calculated as weight in kilograms divided by height in meters squared); comorbidity variables of hypertension, diabetes mellitus, congestive heart failure, coronary artery disease, obstructive lung disease (chronic obstructive pulmonary disease or asthma), and cancer; and other clinically relevant variables at ICU admission, such as Acute Physiology and Chronic Health Evaluation (APACHE) II score and post-intubation ratio of PaO2 to the fraction of inspired oxygen (FiO2) [5]. As a subgroup analysis, we performed a logistic regression using characteristics of the cohort without COVID-19–associated respiratory failure to identify risk factors for death. A two-sided P-value <0.05 was considered statistically significant. Statistical analyses were performed with Stata 15.1 (StataCorp.). Collection of ICU COVID-19 registry data was approved by Jefferson’s Office of Human Research Institutional Review Board (20E.414) with a waiver of informed consent.
RESULTS
From March 18, 2020, to April 30, 2021, there were 843 ICU admissions for patients infected with SARS-CoV-2. Of these, 335 (39.6%) were excluded as they did not require IMV, and an additional 32 were excluded as ICU readmissions during the same hospital stay (i.e., non-index ICU admissions). Seven patients admitted to the ICU for reasons other than AHRF secondary to COVID pneumonia whose reason for intubation was ultimately COVID pneumonia were excluded to avoid biasing the results. After exclusions, our final study cohort included 469 intubated patients with COVID-19 stratified as 319 patients with AHRF secondary to COVID pneumonia and 150 patients without this designation (Figure 1).
Figure 1.
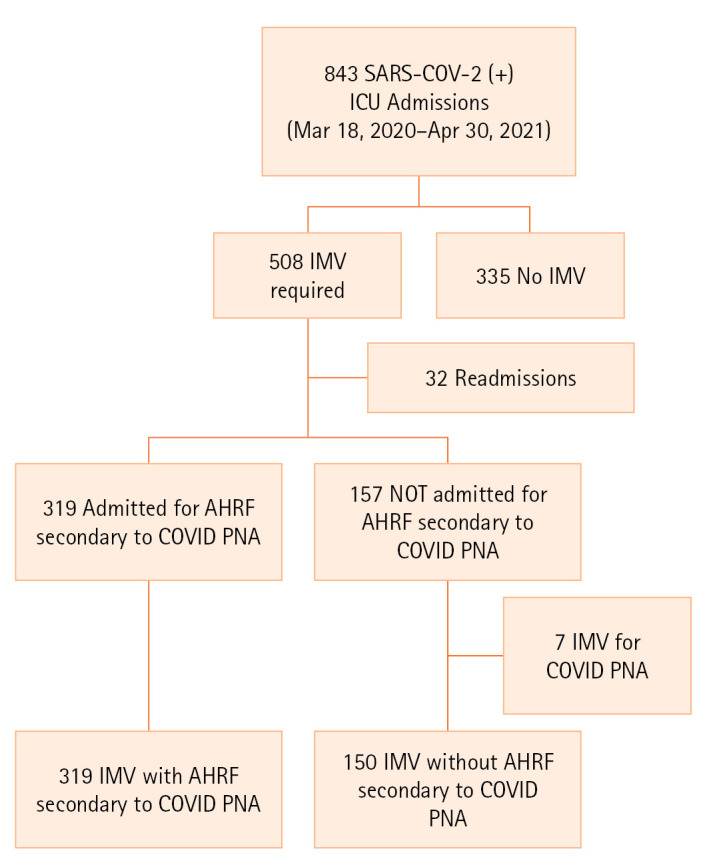
Patient selection. SARS-CoV-2: severe acute respiratory syndrome coronavirus disease 2; ICU: intensive care unit; IMV: invasive mechanical ventilation; AHRF: acute hypoxemic respiratory failure; COVID: coronavirus disease; PNA: pneumonia.
The baseline characteristics of each cohort are summarized in Table 1. Patients were similar in age between the two groups and were predominantly male. Race distribution differed between the two groups, with higher proportions of Asians and Hispanics among patients admitted to the ICU with COVID-19–associated respiratory failure. The median body mass index was significantly higher among patients with COVID-19–associated acute respiratory failure. Hypertension was the most common comorbidity among all patients, while HIV/AIDS and cirrhosis were relatively uncommon comorbidities. The prevalence of cerebrovascular event was significantly higher in the group of patients without AHRF secondary to COVID pneumonia. Patients admitted to the ICU with AHRF secondary to COVID pneumonia were more likely to present to the hospital from home and were also more likely to have spent some time on the general hospital floor prior to ICU admission compared to the comparator group.
Table 1.
Demographic and admission characteristics of enrolled COVID-19 patients
| Characteristic | Mechanically ventilated without AHRF secondary to COVID PNA (n=150) | Mechanically ventilated with AHRF secondary to COVID PNA (n=319) | P-value |
|---|---|---|---|
| Age (yr) | 64 (55–75) | 65 (55–74) | 0.86 |
| Male | 98 (65.3) | 203 (63.6) | 0.72 |
| Race | 0.03 | ||
| Black | 57 (38.0) | 104 (32.6) | |
| White | 62 (41.3) | 125 (39.2) | |
| Hispanic | 4 (2.7) | 31 (9.7) | |
| Asian/Pacific-islander | 19 (12.7) | 49 (15.4) | |
| Other/unknown | 8 (5.3) | 10 (3.1) | |
| Body mass index (kg/m2) | 27.0 (24.0–32.6) | 30.1 (25.3–36.6) | <0.001 |
| ≥30 kg/m2 | 53 (33.8) | 162 (50.8) | <0.001 |
| Smoking status | 0.05 | ||
| Active | 13 (8.7) | 19 (6.0) | |
| Former | 45 (30.3) | 115 (36.1) | |
| Never | 49 (32.7) | 125 (39.2) | |
| Unknown | 47 (28.7) | 60 (18.8) | |
| Comorbidity | |||
| Diabetes mellitus | 56 (37.3) | 137 (43.0) | 0.25 |
| COPD/asthma | 22 (14.7) | 67 (21.0) | 0.10 |
| Cerebrovascular accident | 23 (15.3) | 22 (6.9) | 0.004 |
| Congestive heart failure | 30 (20.0) | 50 (15.7) | 0.25 |
| Cancer | 25 (16.7) | 36 (11.3) | 0.11 |
| End-stage renal disease | 13 (8.7) | 18 (5.6) | 0.22 |
| Hypertension | 84 (56.0) | 201 (63.0) | 0.15 |
| Coronary artery disease | 31 (20.1) | 55 (17.2) | 0.37 |
| HIV/AIDS | 5 (3.3) | 3 (0.9) | 0.12 |
| Cirrhosis | 7 (4.7) | 8 (2.5) | 0.22 |
| Admission source | <0.001 | ||
| Home | 97 (64.7) | 259 (81.2) | |
| Nursing home | 16 (10.7) | 27 (8.5) | |
| Rehabilitation facility | 9 (6.0) | 5 (1.6) | |
| Other/unknown | 28 (18.7) | 28 (8.8) | |
| Care site immediately prior to ICU | <0.001 | ||
| Emergency room | 55 (36.7) | 111 (34.8) | |
| Floor | 39 (26.0) | 127 (39.8) | |
| Stepdown unit | 2 (1.3) | 28 (8.8) | |
| Another hospital | 42 (28.0) | 52 (16.3) | |
| Other | 12 (8.0) | 1 (0.3) | |
| Full code status at ICU admission | 140 (93.3) | 303 (95.0) | 0.47 |
| Chronic pre-hospital medication | |||
| Corticosteroid | 7 (4.7) | 16 (5.0) | 0.87 |
| Immunomodulator | 4 (2.7) | 14 (4.4) | 0.45 |
| Anticoagulation | 31 (20.7) | 44 (13.8) | 0.06 |
Values are presented as median (interquartile range) or number (%). COVID-19: coronavirus disease 2019; AHRF: acute hypoxemic respiratory failure; PNA: pneumonia; COPD: chronic obstructive pulmonary disease; HIV: human immunodeficiency virus; AIDS: acquired immunodeficiency syndrome; ICU: intensive care unit.
Clinical data are presented in Table 2. The admission severity of illness as measured by APACHE II scores was similar between the two groups. For those patients with available inflammatory markers data, peak values for all markers were significantly higher for those admitted to the ICU for AHRF secondary to COVID pneumonia. These patients were more significantly hypoxemic on initial post-intubation arterial blood gas, with PaO2/FiO2 ratios consistent with moderate-to-severe ARDS (Table 2) [6]. Accordingly, this patient group was also more likely to receive interventions specific to ARDS, such as prone ventilation and inhaled vasodilator therapy. The use of therapeutics like new systemic anticoagulation, systemic steroids, and anti–interleukin-6 therapy with tocilizumab was more common in the group with COVID-19–associated respiratory failure. Renal failure culminating in the initiation of renal replacement therapy was also more common in this group. All patients who were cannulated (n=46, 9.8% of total study population of 469, but 14.5% of the COVID-19 associated respiratory failure group) for extracorporeal membrane oxygenation originated from the group with AHRF secondary to COVID pneumonia.
Table 2.
Clinical characteristics and intensive care unit interventions of enrolled COVID-19 patients
| Characteristic | Mechanically ventilated without AHRF secondary to COVID PNA (n=150) | Mechanically ventilated with AHRF secondary to COVID PNA (n=319) | P-value |
|---|---|---|---|
| APACHE II scorea) | 27 (20.5–32) | 27 (18–34) | 0.81 |
| Symptom duration preceding hospitalization (day) | NA | ||
| <7 | NA | 148 (46.4) | |
| 7–14 | NA | 116 (36.4) | |
| >14 | NA | 19 (6.0) | |
| Unknown | NA | 36 (11.3) | |
| Laboratory value | |||
| Peak D-dimer (ng/ml) | 1,733 (778–7,179) (n=122) | 5,117 (1,851–17,440) (n=313) | <0.001 |
| Peak C-reactive protein (mg/dl) | 11.3 (4.8–19.9) (n=108) | 25.1 (14–33.5) (n=311) | <0.001 |
| Peak ferritin (ng/ml) | 686 (281–1,754,5) (n=80) | 1,548.5 (767.5–3,068.5) (n=280) | <0.001 |
| Initial PaO2/FiO2 post-intubation | 218 (145-304) (n=60) | 108 (82-173) (n=221) | <0.001 |
| Interventions | |||
| High-flow nasal oxygenb) | 8 (5.3) | 124 (38.9) | <0.001 |
| Non-invasive positive pressure ventilationb) | 11 (7.3) | 149 (46.7) | <0.001 |
| Prone ventilation | 3 (2.0) | 174 (54.6) | <0.001 |
| Prone duration (day) | 1 (1–3) | 3 (2–7) (n=167) | 0.15 |
| Inhaled vasodilator | 11 (7.3) | 101 (31.7) | <0.001 |
| New systemic anticoagulation | 34 (22.7) | 135 (42.3) | <0.001 |
| Systemic steroidsc) | 72 (48.0) | 236 (74.0) | <0.001 |
| Tocilizumab | 4 (2.7) | 84 (26.3) | <0.001 |
| Convalescent plasma | 9 (6.0) | 23 (7.2) | 0.63 |
| New initiation RRT | 22 (14.7) | 85 (26.7) | 0.004 |
| ECMO cannulation | 0 | 46 (14.5) | <0.001 |
Values are presented as median (interquartile range) or number (%).
COVID-19: coronavirus disease 2019; AHRF: acute hypoxemic respiratory failure; PNA: pneumonia; APACHE: Acute Physiology and Chronic Health Evaluation; NA: not applicable; PaO2/FiO2: arterial partial pressure of oxygen divided by inspired oxygen concentration; RRT: renal replacement therapy; ECMO: extracorporeal membrane oxygenation.
Two uncalculatable APACHE scores;
Noninvasive respiratory support at or near the time of intensive care unit admission;
Patients who received isolated steroid doses as pre-medication for Tocilizumab administration classified as not having received a systemic steroid course.
Further details about the mechanically ventilated COVID-19 patients admitted to the ICU for reasons other than COVID-19–associated AHRF are provided in Tables 3 and 4. The most common causes for ICU admission in this group were neurologic compromise (36%) followed by cardiovascular event or instability (16%) (Table 3). Observed hospital mortality was highest among patients admitted to the ICU for neurologic diagnoses (Table 3). Among these patients, the majority (65 patients, 43.3%) was intubated for airway protection (Table 4).
Table 3.
Reasons for intensive care unit admission in mechanically ventilated COVID-19 patients without acute hypoxemic respiratory failure secondary to COVID pneumonia and mortality
| Diagnostic group | Number (%) (n=150) | Hospital mortality |
|---|---|---|
| Sepsisa) | 22 (14.7) | 9 (6.0) |
| Neurologic | 54 (36) | 20 (13.3) |
| Status epilepticus | 14 | |
| Encephalopathy | 14 | |
| Stroke | 8 | |
| Intracranial hemorrhage | 12 | |
| Otherb) | 6 | |
| Cardiovascular | 24 (16) | 13 (8.7) |
| Cardiac arrest | 8 | |
| Decompensated heart failure | 8 | |
| Aortic diseasec) | 5 | |
| Myocardial infarction | 2 | |
| Conduction disorder | 1 | |
| Pulmonary | 16 (10.7) | 3 (2.0) |
| Multifactorial hypoxiad) | 5 | |
| Tracheostomy complications | 2 | |
| Airway obstruction | 2 | |
| Othere) | 7 | |
| Gastrointestinal | 18 (12) | 10 (6.7) |
| Gastrointestinal bleed | 6 | |
| Decompensated cirrhosis | 4 | |
| Acute liver failure | 2 | |
| Otherf) | 6 | |
| Endocrine | 3 (2) | 1 (0.67) |
| Diabetic ketoacidosis | 2 | |
| Hypoglycemia | 1 | |
| Renal | 1 (0.7) | 1 (0.67) |
| Acute renal failure | 1 | |
| Hematologic | 3 (2) | 2 (1.3) |
| Thrombotic diseaseg) | 2 | |
| Coagulopathic hemorrhage (non-gastrointestinal) | 1 | |
| Musculoskeletal | 8 (5.3) | 0 |
| Trauma from motor vehicle accident, assault, or fall | 5 | |
| Spinal cord injuryh) | 3 | |
| Other | 1 (0.7) | 0 |
| MRI under sedation | 1 |
Values are presented as number (%).
COVID-19: coronavirus disease 2019; MRI: magnetic resonance imaging.
Includes infections from bacteremia of unidentified source, bacterial pneumonia, epidural abscess, viral meningitis, retropharyngeal abscess, necrotizing fasciitis, urinary tract infection, and gastrocutaneous fistula;
Includes intracranial tumor, cerebral venous thrombosis, pneumocephalus, narcotic overdose, and encephalomyelitis;
Includes aortic dissection and aneurysm/pseudoaneurysm;
Includes a combination of chronic obstructive pulmonary disease, heart failure, pulmonary edema, pneumonia, and pulmonary embolism;
Includes chronic obstructive pulmonary disease exacerbation, pulmonary embolism, pneumothorax, tracheal stenosis, inflammatory alveolitis, angioedema, and inhalational injury;
Includes anastomotic leak, perforated colon, acute mesenteric ischemia, pancreatitis, and small bowel obstruction;
Includes multiple thrombi and superior mesenteric artery thrombosis;
Includes cervical fracture and stenosis.
Table 4.
Reasons for intubation in mechanically ventilated COVID-19 patients without acute hypoxemic respiratory failure secondary to COVID pneumonia
| Reason for intubation | Number (%) (n=150) |
|---|---|
| Airway protection | 65 (43.3) |
| Surgery/procedure | 41 (27.3) |
| Aspiration/bacterial pneumonia | 15 (10) |
| Cardiac arrest | 10 (6.7) |
| Decompensated heart failure | 8 (5.3) |
| Multifactorial hypoxia | 5 (3.3) |
| Othera) | 6 (4) |
COVID-19: coronavirus disease 2019.
Includes pulmonary embolism, inflammatory alveolitis, chronic obstructive lung disease exacerbation, central line complication, bilateral pneumothoraces, and tracheal stenosis.
Table 5 summarizes patient outcomes. Rates of attempted extubation were higher among patients without AHRF secondary to COVID pneumonia compared to those with COVID-19–associated AHRF (57.3% vs. 32.3%, P<0.001), and patients without COVID-19–associated AHRF required fewer ventilator days prior to extubation. Tracheostomy rates were significantly higher in the group of patients with AHRF due to COVID pneumonia. Both the ICU LOS (12 vs. 6 days, P<0.001) and the hospital LOS (20 vs. 13.5 days, P<0.001) were significantly longer for patients with AHRF secondary to COVID pneumonia compared to those without. Patients who survived to hospital discharge were most likely to be discharged to acute rehabilitation facilities for continued recovery. There was no significant difference between the two study groups in discharge rate to home.
Table 5.
Clinical outcomes and discharge data of enrolled COVID-19 patients
| Outcome | Mechanically ventilated without AHRF secondary to COVID PNA (n=150) | Mechanically ventilated with AHRF secondary to COVID PNA (n=319) | P-value |
|---|---|---|---|
| Venous thromboembolic eventa) | |||
| Pulmonary embolism | 6 (19.4) (n=31) | 24 (23.5) (n=102) | 0.63 |
| Deep vein thrombosis | 3 (10.0) (n=30) | 9 (17.3) (n=52) | 0.29 |
| Time to intubationb) (day) | 2 (1–6) (n=24) | 2 (1–4) (n=128) | 0.64 |
| Extubation | 86 (57.3) | 103 (32.3) | <0.001 |
| Reintubation | 20 (23.3) | 17 (16.5) | 0.24 |
| Ventilator days preceding extubation | 3 (2–5) | 8 (5–14) | <0.001 |
| Tracheostomy | 21 (14.0) | 72 (22.6) | 0.03 |
| LOS outcomes (day) | |||
| ICU | 6 (3–12) | 12 (7–23) | <0.001 |
| Hospital | 13.5 (8–24) | 20 (11–33) | <0.001 |
| Unadjusted mortality outcome | |||
| ICU | 55 (36.7) | 184 (57.7) | <0.001 |
| Hospital | 59 (39.3) | 188 (58.9) | <0.001 |
| Full code status | |||
| At ICU discharge | 92 (61.3) | 158 (49.5) | 0.06 |
| At hospital discharge | 86 (57.3) | 155 (48.6) | 0.28 |
| Hospital discharge dispositionc) | (n=95) | (n=131) | 0.34 |
| Home | 26 (28.6) | 37 (28.2) | |
| Hospice (home or facility) | 5 (5.5) | 2 (1.5) | |
| Long term acute care | 5 (5.5) | 16 (12.2) | |
| Skilled nursing facility | 19 (20.9) | 24 (18.3) | |
| Acute rehabilitation | 28 (30.8) | 43 (32.8) | |
| Other | 8 (8.8) | 9 (6.9) |
Values are presented as number (%) or median (interquartile range).
COVID-19: coronavirus disease 2019; AHRF: acute hypoxemic respiratory failure; PNA: pneumonia; LOS: length of stay; ICU: intensive care unit.
Includes only those with radiographically confirmed venous thromboembolism;
Defined as number of days to intubation from date of ICU admission. Excludes outside hospital transfers and 3 intubated prior to ICU admission;
Denominator includes only survivors to hospital discharge.
Patients with AHRF secondary to COVID pneumonia had significantly higher unadjusted rates of ICU mortality (57.7% vs. 36.7%, P<0.001) and hospital mortality (58.9% vs. 39.3%, P<0.001) compared to those without such pneumonia (Table 5). After risk adjustment, AHRF from COVID pneumonia conferred 2.25 times greater odds of hospital death (95% confidence interval [CI], 1.42–3.56; P=0.001) compared to mechanically ventilated COVID-19 patients intubated for reasons other than COVID pneumonia (Table 6). Additional risk factors for mortality among the entire cohort of mechanically ventilated COVID-19 patients included severity of hypoxemia and APACHE II score upon ICU admission (Table 6). In adjusted analyses isolated to consider only the population of mechanically ventilated COVID-19 patients admitted to the ICU without COVID-19–associated respiratory failure, APACHE illness severity was similarly identified as a risk factor for death (odds ratio [OR], 1.08; 95% CI, 1.03–1.13; P=0.002). Diabetes mellitus also was identified as a mortality risk factor in this cohort (OR, 2.79; 95% CI, 1.22–6.40; P=0.02) (Supplementary Table 1).
Table 6.
Multivariable logistic regression model for hospital death in mechanically ventilated COVID-19 patients
| Characteristic | Adjusted odds ratio (95% CI) | P-value |
|---|---|---|
| Primary COVID | 2.25 (1.42–3.56) | 0.001 |
| Age >65 yr | 1.51 (0.98–2.32) | 0.06 |
| Female sex | 0.86 (0.56–1.31) | 0.48 |
| Race other than white | 0.83 (0.55–1.26) | 0.38 |
| Body mass index ≥30 kg/m2 | 0.84 (0.55–1.27) | 0.40 |
| PaO2/FiO2 ≤150 | 2.03 (1.30–3.17) | 0.002 |
| APACHE II (1-unit increase) | 1.03 (1.01–1.05) | 0.004 |
| Diabetes mellitus | 1.11 (0.73–1.67) | 0.63 |
| COPD/asthma | 1.44 (0.85–2.41) | 0.17 |
| Cerebrovascular accident | 2.04 (1.01–4.13) | 0.05 |
| Congestive heart failure | 0.95 (0.55–1.67) | 0.87 |
| Cancer | 1.66 (0.90–3.09) | 0.11 |
| Hypertension | 0.75 (0.49–1.16) | 0.20 |
| Coronary artery disease | 1.55 (0.88–2.74) | 0.13 |
COVID-19: coronavirus disease 2019; CI: confidence interval; PaO2/FiO2: arterial partial pressure of oxygen divided by inspired oxygen concentration; APACHE: Acute Physiology and Chronic Health Evaluation; COPD: chronic obstructive pulmonary disease.
DISCUSSION
The results of our retrospective cohort comparison demonstrate that, among mechanically ventilated patients admitted to the ICU with SARS-CoV-2 infection, those admitted to the ICU with COVID-19–associated respiratory failure are less likely to survive to hospital discharge compared to those whose initial ICU needs were unrelated to COVID pneumonia. In addition, patients whose ICU needs were defined by AHRF secondary to COVID pneumonia also experience both longer ICU and hospital LOS times and are less likely to undergo an attempt at liberation from IMV in the context of a longer duration of IMV prior to extubation.
The bulk of the literature examining the outcomes of mechanically ventilated patients with COVID-19 is based on the assumption that most patients are intubated due to COVID ARDS [7,8]. The clinical outcomes of those with primary respiratory failure secondary to COVID-19 pneumonia are most often compared to those of patients with comparable critical illnesses but without COVID-19 [9-11]. However, our study shows various reasons for ICU admission and precipitants for IMV among patients infected with SARS-CoV-2 and not only progression of COVID-19 pneumonia to ARDS. To our knowledge, our study is the first to describe the heterogeneity of critical illness presentations among COVID-19 patients and the associated variability in their reasons for intubation. Our study is also the first to characterize intubated patients admitted to the ICU with and not due to COVID-19 and to report comparative outcomes.
Given the high associated mortality of ARDS overall, it is not surprising that the survival of our cohort with AHRF secondary to COVID pneumonia was worse than that of the cohort without, likely as a result of the severity of hypoxemia in this group [12]. Estimates of mortality for COVID-19 patients admitted to the ICU vary widely but have recently been reported to be around 40% [13,14]. When patients admitted to the ICU for COVID-19 require IMV, their mortality rate increases even further to 59% [13]. Yet, in our cohort of intubated patients without COVID-19–associated AHRF, the observed ICU mortality rate was 36.7%, and the observed hospital mortality was 39.3%, suggesting that intubated patients admitted to the ICU for reasons other than COVID pneumonia compose a distinct patient group with likely better survival. Mortality in the typical critically ill patient with COVID-19 can be impacted by demographic, clinical, and hospital-level factors [5]. In our study, critical illness defined by COVID ARDS, its associated hypoxemia, and severity of illness score at ICU presentation were the largest contributors to reduced survival.
Risk factors for death specifically in the mechanically ventilated COVID-19 patient admitted to the ICU for reasons other than severe hypoxemia are less well-defined. In our limited sample of such patients, a higher APACHE score was a risk factor for death, which is similar to results from the entire cohort. Interestingly, among the individual comorbidities explored, diabetes mellitus was a significant predictor of death in this group. Previous studies have identified diabetes mellitus as a highly prevalent contributor to increased risk of death among hospitalized COVID-19 patients, particularly those who are critically ill [15]. The postulated mechanisms may include the impact of hyperglycemia on viral replication or immune dysregulation [16]. However, our results show diabetes mellitus to uniquely impact mortality in the cohort of patients intubated for reasons other than COVID pneumonia. Although limited by a relatively small sample size, identification of this risk factor particular to this group further suggests differing phenotypes of mechanically ventilated COVID-19 patients characterized by unique disease modifiers in addition to differing outcomes.
Shorter ICU, hospital, and pre-extubation ventilator durations were also seen in the group of patients without COVID-19–associated respiratory failure compared to the group with COVID-19–associated respiratory failure. In the first six months of the pandemic in the US, the median hospital LOS for COVID-19 patients admitted to the ICU was 15 days (interquartile range, 6–20 days) using administrative data [17]. In the Netherlands, ICU LOS has been shown to vary between “waves” of the pandemic, with averages ranging from 16.0 to 20.6 days [18]. Among mechanically ventilated COVID-19 patients in particular, the mean hospital LOS was as long as 24.5 days among survivors in the Inova Health System [7]. These previously reported hospital and ICU LOS values are more comparable to the LOS in our cohort of patients with AHRF secondary to COVID pneumonia. This further suggests that the COVID-19 population admitted to the ICU without AHRF secondary to COVID pneumonia has distinct, and likely more favorable, utilization outcomes.
There are several limitations to our study. First, patient data were retrospectively collected from hospitals in a single system and may not be reflective of hospitals in non-urban settings that serve patients of differing demographics and socioeconomic vulnerabilities. Second, we did not have data on day-to-day ICU or hospital-level factors (e.g., staffing, equipment, or bed availability) that may have impacted results. Third, we did not record the vaccination status of admitted patients in our cohort, as the U.S. Food and Drug Administration’s approval of the first COVID-19 vaccine did not occur in the US until December 11, 2020. The likelihood of an incidentally COVID-19–positive patient being mechanically ventilated in the ICU anecdotally increased in the months following the availability of vaccines. It is unknown how vaccination status impacts presentation to the ICU with respiratory failure unrelated to COVID-19. However, the majority of patients included in this study was hospitalized before widespread availability of vaccines against COVID-19 for adults in the US, limiting the impact of vaccination status on results. Fourth, although we did have data on specific therapeutics, such as tocilizumab and systemic steroids, which are largely indicated for patients with hypoxemia, we did not extract data on the use of other therapies like baricitinib or patient involvement in clinical trials, which may also have impacted outcomes. Last, as the cohort of COVID-19 patients admitted to the ICU without AHRF secondary to COVID pneumonia is not yet well-defined, it is possible that we are unable to account for unmeasured confounders that may have impacted their outcomes.
In conclusion, in our single-center descriptive cohort study, mechanically ventilated patients admitted for COVID-19–associated respiratory failure are a distinct population with worse outcomes compared to patients who ultimately required intubation but who were admitted to the ICU with a comorbid COVID-19 diagnosis. The multi-wave nature of the COVID-19 pandemic has been shown to result in varying outcomes across time periods in different countries, with a general trend of improvement over time [19-23]. The sociopolitical context in which effective public health measures (e.g., social distancing, hand hygiene, universal masking) are adopted [24] and vaccines are administered has affected the epidemiology of COVID-19. More recently, the rise of the Omicron variant has resulted in more patients being infected with SARS-CoV-2 but overall having less severe symptoms [25,26].
As the pandemic (and the virus) continues to evolve, we anticipate an increase in patients with increasingly milder and/or completely asymptomatic infections who may develop critical illness for other reasons yet whose disease courses are impacted by their comorbid SARS-CoV-2 infection. Recognition of the differing clinical phenotypes of patients in the ICU with and not necessarily due to COVID-19 will stimulate further inquiry into identifying risk factors for deterioration and predictors of survival among variable hosts. Acknowledgment of finer distinctions among intubated COVID-19 patients will allow providers to better understand the role of SARS-CoV-2 infection in the evolution of critical illness such that focused, and perhaps even anticipatory, treatment plans may be tailored to ensure the best possible patient outcomes.
KEY MESSAGES
▪ There is increasing heterogeneity in clinical phenotype among mechanically ventilated patients with coronavirus disease 2019 (COVID-19).
▪ Intubated patients admitted to the intensive care unit (ICU) with acute hypoxemic respiratory failure secondary to COVID pneumonia are a distinct population with worse clinical and utilization outcomes compared to intubated patients whose reason for ICU admission is unrelated to COVID pneumonia.
Acknowledgments
None.
Footnotes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING
None.
AUTHOR CONTRIBUTIONS
Conceptualization: JAJ, KFM, VMP, EJY. Methodology: EJY. Software: EJY. Validation: EJY. Formal analysis: EJY. Investigation: JAJ, KFM, VMP, TT, GM, PK, CM. Resources: EJY. Data curation: JAJ, KFM, VMP, TT, GM, PK, CM, EJY. Visualization: JAJ, VMP, BJG, EJY. Supervision: EJY. Project administration: EJY. Writing–original draft: JAJ, EJY. Writing–review & editing: KFM, VMP, TT, GM, PK, CM, BJG.
SUPPLEMENTARY MATERIALS
Supplementary materials can be found via https://doi.org/10.4266/acc.2022.01123.
Multivariable logistic regression model for hospital death in mechanically ventilated COVID-19 patients without acute hypoxemic respiratory failure secondary to COVID pneumonia
REFERENCES
- 1.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–20. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pronovost PJ, Angus DC, Dorman T, Robinson KA, Dremsizov TT, Young TL. Physician staffing patterns and clinical outcomes in critically ill patients: a systematic review. JAMA. 2002;288:2151–62. doi: 10.1001/jama.288.17.2151. [DOI] [PubMed] [Google Scholar]
- 3.Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap): a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–81. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L, et al. The REDCap consortium: building an international community of software platform partners. J Biomed Inform. 2019;95:103208. doi: 10.1016/j.jbi.2019.103208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gupta S, Hayek SS, Wang W, Chan L, Mathews KS, Melamed ML, et al. Factors associated with death in critically ill patients with coronavirus disease 2019 in the US. JAMA Intern Med. 2020;180:1436–47. doi: 10.1001/jamainternmed.2020.3596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.ARDS Definition Task Force. Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307:2526–33. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 7.King CS, Sahjwani D, Brown AW, Feroz S, Cameron P, Osborn E, et al. Outcomes of mechanically ventilated patients with COVID-19 associated respiratory failure. PLoS One. 2020;15:e0242651. doi: 10.1371/journal.pone.0242651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Krause M, Douin DJ, Kim KK, Fernandez-Bustamante A, Bartels K. Characteristics and outcomes of mechanically ventilated COVID-19 patients: an observational cohort study. J Intensive Care Med. 2021;36:271–6. doi: 10.1177/0885066620954806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sjoding MW, Admon AJ, Saha AK, Kay SG, Brown CA, Co I, et al. Comparing clinical features and outcomes in mechanically ventilated patients with COVID-19 and acute respiratory distress syndrome. Ann Am Thorac Soc. 2021;18:1876–85. doi: 10.1513/AnnalsATS.202008-1076OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hodgson CL, Higgins AM, Bailey MJ, Mather AM, Beach L, Bellomo R, et al. Comparison of 6-month outcomes of survivors of COVID-19 versus non-COVID-19 critical illness. Am J Respir Crit Care Med. 2022;205:1159–68. doi: 10.1164/rccm.202110-2335OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Todi S, Ghosh S. A comparative study on the outcomes of mechanically ventilated COVID-19 vs non-COVID-19 patients with acute hypoxemic respiratory failure. Indian J Crit Care Med. 2021;25:1377–81. doi: 10.5005/jp-journals-10071-24009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bellani G, Laffey JG, Pham T, Fan E, Brochard L, Esteban A, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 2016;315:788–800. doi: 10.1001/jama.2016.0291. [DOI] [PubMed] [Google Scholar]
- 13.Tzotzos SJ, Fischer B, Fischer H, Zeitlinger M. Incidence of ARDS and outcomes in hospitalized patients with COVID-19: a global literature survey. Crit Care. 2020;24:516. doi: 10.1186/s13054-020-03240-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Armstrong RA, Kane AD, Cook TM. Outcomes from intensive care in patients with COVID-19: a systematic review and meta-analysis of observational studies. Anaesthesia. 2020;75:1340–9. doi: 10.1111/anae.15201. [DOI] [PubMed] [Google Scholar]
- 15.Saha S, Al-Rifai RH, Saha S. Diabetes prevalence and mortality in COVID-19 patients: a systematic review, meta-analysis, and meta-regression. J Diabetes Metab Disord. 2021;20:939–50. doi: 10.1007/s40200-021-00779-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lim S, Bae JH, Kwon HS, Nauck MA. COVID-19 and diabetes mellitus: from pathophysiology to clinical management. Nat Rev Endocrinol. 2021;17:11–30. doi: 10.1038/s41574-020-00435-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nguyen NT, Chinn J, Nahmias J, Yuen S, Kirby KA, Hohmann S, et al. Outcomes and mortality among adults hospitalized with COVID-19 at US medical centers. JAMA Netw Open. 2021;4:e210417. doi: 10.1001/jamanetworkopen.2021.0417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dongelmans DA, Termorshuizen F, Brinkman S, Bakhshi-Raiez F, Arbous MS, de Lange DW, et al. Characteristics and outcome of COVID-19 patients admitted to the ICU: a nationwide cohort study on the comparison between the first and the consecutive upsurges of the second wave of the COVID-19 pandemic in the Netherlands. Ann Intensive Care. 2022;12:5. doi: 10.1186/s13613-021-00978-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lambermont B, Rousseau AF, Seidel L, Thys M, Cavalleri J, Delanaye P, et al. Outcome improvement between the first two waves of the coronavirus disease 2019 pandemic in a single tertiary-care hospital in Belgium. Crit Care Explor. 2021;3:e0438. doi: 10.1097/CCE.0000000000000438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Maslo C, Friedland R, Toubkin M, Laubscher A, Akaloo T, Kama B. Characteristics and outcomes of hospitalized patients in south africa during the COVID-19 Omicron wave compared with previous waves. JAMA. 2022;327:583–4. doi: 10.1001/jama.2021.24868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Carbonell R, Urgelés S, Rodríguez A, Bodí M, Martín-Loeches I, Solé-Violán J, et al. Mortality comparison between the first and second/third waves among 3,795 critical COVID-19 patients with pneumonia admitted to the ICU: a multicentre retrospective cohort study. Lancet Reg Health Eur. 2021;11:100243. doi: 10.1016/j.lanepe.2021.100243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sartini M, Del Puente F, Oliva M, Carbone A, Blasi Vacca E, Parisini A, et al. Riding the COVID waves: clinical trends, outcomes, and remaining pitfalls of the SARS-CoV-2 pandemic: an analysis of two high-incidence periods at a hospital in Northern Italy. J Clin Med. 2021;10:5239. doi: 10.3390/jcm10225239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hoogenboom WS, Pham A, Anand H, Fleysher R, Buczek A, Soby S, et al. Clinical characteristics of the first and second COVID-19 waves in the Bronx, New York: a retrospective cohort study. Lancet Reg Health Am. 2021;3:100041. doi: 10.1016/j.lana.2021.100041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cowling BJ, Aiello AE. Public health measures to slow community spread of coronavirus disease 2019. J Infect Dis. 2020;221:1749–51. doi: 10.1093/infdis/jiaa123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Worldometer . Worldometer; 2022. Coronavirus cases [Internet] [cited 2022 May 20]. Available from: https://www.worldometers.info/coronavirus/#countries. [Google Scholar]
- 26.Ren SY, Wang WB, Gao RD, Zhou AM. Omicron variant (B.1.1.529) of SARS-CoV-2: mutation, infectivity, transmission, and vaccine resistance. World J Clin Cases. 2022;10:1–11. doi: 10.12998/wjcc.v10.i1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Multivariable logistic regression model for hospital death in mechanically ventilated COVID-19 patients without acute hypoxemic respiratory failure secondary to COVID pneumonia


