Abstract
Toxigenic strains of Clostridium difficile have been reported to produce both toxins A and B nearly always, and nontoxigenic strains have been reported to produce neither of these toxins. Recent studies indicate that it is not always true. We established a PCR assay to differentiate toxin A-negative, toxin B-positive (toxin A−, toxin B+) strains from both toxin-positive (toxin A+, toxin B+) strains and both toxin-negative (toxin A−, toxin B−) strains as an alternative to cell culture assay and enzyme-linked immunosorbent assay (ELISA). By using the PCR primer set NK11 and NK9 derived from the repeating sequences of the toxin A gene, a shorter segment (ca. 700 bp) was amplified from toxin A−, toxin B+ strains compared to the size of the segment amplified from toxin A+, toxin B+ strains (ca. 1,200 bp), and no product was amplified from toxin A−, toxin B− strains. We examined a total of 421 C. difficile isolates by PCR. Of these, 48 strains showed a shorter segment by the PCR, were negative by ELISAs for the detection of toxin A, and were positive by cell culture assay. Although the cytotoxin produced by the toxin A−, toxin B+ strains was neutralized by anti-toxin B serum, the appearance of the cytotoxic effects on Vero cell monolayers was distinguishable from that of toxin A+, toxin B+ strains. By immunoblotting, the 44 toxin A−, toxin B+ strains were typed to serogroup F and the remaining four strains were serogroup X. Pulsed-field gel electrophoresis separated the 48 strains into 19 types. The PCR assay for the detection of the repeating sequences combined with PCR amplification of the nonrepeating sequences of either the toxin A or the toxin B gene is indicated to be useful for differentiating toxin A−, toxin B+ strains from toxin A+, toxin B+ and toxin A−, toxin B− strains and will contribute to elucidation of the precise role of toxin A−, toxin B+ strains in intestinal diseases.
Clostridium difficile is a causative agent of pseudomembranous colitis and a principle pathogen causing antibiotic-associated diarrhea (AAD) and antibiotic-associated colitis (AAC). Strains of C. difficile produce two toxins, toxin A and toxin B, which are involved in the pathogenicity of this organism (3, 15). Toxin A has been referred to as an enterotoxin in view of its fluid accumulation activity in animal intestinal loop tests (17), and toxin B has been referred to as a cytotoxin whose cytopathic effect is considerably much more potent than that of toxin A. Because toxigenic strains of C. difficile are believed to release both toxins A and B and cell culture assay for toxin B is the most sensitive method for the detection of toxin B, the cell culture assay is considered the “gold standard” for the diagnosis of C. difficile-associated diarrhea and colitis.
A strain named C. difficile 8864 has been reported to be toxin A-negative and toxin B-positive (toxin A−, toxin B+) (5, 14). The strain fails to produce toxin A but shows enterotoxic activity in a rabbit ileal loop test. The activity may be due to the fact that toxin B has a potent enterotoxin activity(s) (14) or that this strain produces additional factors unrelated to toxin A (5). Serogroup F strains of C. difficile which were often isolated from asymptomatic infants were toxin A− by enzyme-linked immunosorbent assay (ELISA) and toxin B+ by cell culture assay and generated no diarrhea in hamsters and axenic mice (6, 8). However, little regarding the clinical significance, pathogenicity, prevalence, and genetic backgrounds of the toxin A−, toxin B+ strains of C. difficile is known. A simple and rapid method is needed to identify toxin A−, toxin B+ strains to characterize them.
Here we report the use of PCR to amplify a segment of the repeating sequences of the toxin A gene to distinguish toxin A−, toxin B+ strains from toxin A+, toxin B+ and toxin A−, toxin B− strains as an alternative to cell culture assay for toxin B and ELISA for toxin A. We examined 421 clinically isolated C. difficile strains and found an unexpectedly high incidence of toxin A−, toxin B+ strains not only in children but also in adults.
MATERIALS AND METHODS
Bacterial strains.
The 427 strains of C. difficile used in this study were isolated at the following facilities: Institute of Anaerobic Bacteriology, Gifu University School of Medicine, Gifu, Japan; Gifu University Hospital, Gifu, Japan; Nagoyashi Koseiin Geriatric Hospital, Nagoya, Japan; Department of Pediatrics, Meitetsu Hospital, Nagoya, Japan; Tropical Disease Research Centre, Airlangga University, Surabaya, Indonesia; and Centers for Disease Control and Prevention (CDC), Atlanta, Ga.
C. difficile was isolated on cycloserine-cefoxitin-mannitol agar (Nissui Pharmaceutical Co. Ltd., Tokyo, Japan) from stool specimens, which were treated with alcohol for spore selection (1). All strains used in this study were identified by standard procedures (1) combined with colony morphology on cycloserine-cefoxitin-mannitol agar and cell morphology after Gram staining. PCR with primer set PG-48 and PG-49 (9) derived from the C. difficile 16S rRNA gene was used to confirm the identification.
Toxin assays.
C. difficile was cultured anaerobically in brain heart infusion broth (Becton Dickinson and Company, Cockeysville, Md.) for 5 to 7 days. Two ELISA kits, Tox-A TEST (TechLab, Blacksburg, Va.) and VIDAS CDA (bioMerieux Vitek, Hazelwood, Mo.), were used to detect toxin. Enterotoxicity was determined by a rabbit intestinal loop assay (5) with 10-week-old female Japanese White rabbits. To detect a low level of toxin A, the culture supernatants of toxin A−, toxin B+ strains were concentrated 10 times by ultrafiltration with Molcut II (NMWL 10,000; Millipore, Marlborough, Mass.). Toxin B was detected by cell culture assay with Vero cells (5). For detection of toxin B, the culture supernatant was filtered through Millex-GP (Millipore) filters before it was applied to cell monolayers. C. difficile goat anti-toxin B serum (TechLab) was used for the cytotoxin neutralization assay.
PCR assay.
PCR assay was performed as described previously (13), with modifications. A single colony was suspended in 50 μl of TES (50 mM Tris hydrochloride [pH 8.0], 5 mM EDTA, 50 mM NaCl), and the suspension was heated at 95°C for 10 min and centrifuged at 15,000 × g for 2 min. One microliter of the resultant supernatant was added to 30 μl of a reaction buffer consisting of 2.5 mM MgCl2, 50 mM KCl, 10 mM Tris-HCl (pH 9.0), the four deoxynucleoside triphosphates (200 μM each), 45 ng of each primer, and 0.75 U of Taq DNA polymerase (Pharmacia Biotech, Uppsala, Sweden). Two primer sets were used to detect the toxin A gene; primers NK3 and NK2 were derived from the nonrepeating portion of the C. difficile toxin A gene, and primers NK11 and NK9 were derived from the repeating portion of the C. difficile toxin A gene (13). A segment of the toxin B gene was amplified by using primer NK104 (sequence, 5′-GTGTAGCAATGAAAGTCCAAGTTTACGC-3′; positions 2945 to 2972) (4) and primer NK105 (sequence, 5′-CACTTAGCTCTTTGATTGCTGCACCT-3′; positions 3123 to 3148) (4), which were derived from the nonrepeating sequence of the C. difficile toxin B gene. The specificity of the PCR product with primer set NK104-NK105 was confirmed by Southern blot analysis as described previously (10). Probe NK106 (sequence, 5′-GACTTACTTCCTACATTATCTGAAGG-3′; positions 3058 to 3083) (4) was used and was 3′ end labeled with digoxigenin with a digoxigenin labeling kit (Boehringer Mannheim, Mannheim, Germany). The thermal profile for primer pairs NK3-NK2 and NK104-NK105 was 35 cycles comprising 95°C for 20 s and 55°C for 120 s. At the conclusion of the PCR cycles, the tubes were incubated at 74°C for 5 min. PCR amplification with primer pair NK11-NK9 was performed for 35 cycles, consisting of 95°C for 20 s and 62°C for 120 s.
Phenotypic and genotypic typings.
The C. difficile strains were analyzed by Western immunoblotting (11) and pulsed-field gel electrophoresis (PFGE) typing (12). Immunoblot typing was performed with EDTA-extracted proteins of bacterial cells, followed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis, as described previously (11), with antisera against reference strains of the 10 different serogroups established by a slide agglutination test (7). For PFGE analysis, the DNA in the inserts was digested with SacII (New England Biolabs, Beverly, Mass.), and the resultant restriction fragments were separated at a constant voltage of 200 V with 25-s pulses for 5 h followed by 50-s pulses for 19 h (12).
RESULTS
First, we examined the specificity of a PCR assay for the detection of a segment of the toxin B gene using primer set NK104-NK105. The specificities of the PCR products were eventually confirmed by Southern hybridization with the digoxigenin-labeled NK106 probe. A total of 273 randomly selected C. difficile strains were tested by both cell culture and PCR. All 204 toxin B+ strains were PCR positive, and the remaining 69 toxin B− strains were PCR negative. Southern hybridization gave results consistent with the PCR results (data not shown).
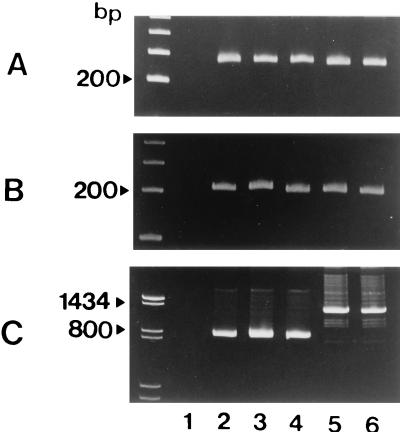
Second, six strains of C. difficile were examined for the production of toxins A and B and the presence of the toxin A and B genes. The results are summarized in Table 1. Five of the six strains tested were PCR positive for the segment of the nonrepeating region of the toxin A gene (Fig. 1A, lanes 2 to 6) and the toxin B gene (Fig. 1B, lanes 2 to 6) and were cell culture positive for toxin B. However, there were three strains from which a shorter segment of approximately 700 bp (Fig. 1C, lanes 2 to 4) was amplified by PCR for the repeating region of the toxin A gene, whereas for toxin A+, toxin B+ a PCR product of approximately 1,200 bp in size (Fig. 1C, lanes 5 and 6) was generated. The three strains were toxin A− by two ELISAs and a rabbit ileal loop test. Thus, we designated them toxin A−, toxin B+ strains.
TABLE 1.
Results of toxin production assays and PCR for the repeating and nonrepeating regions of toxin A and B genes for six strains
| Strain no. | Cell culture assay result for toxin B | Result of the following ELISA for toxin A:
|
PCR result for the following:
|
Product size (bp) from PCR for toxin A genec (NK11-NK9)b | Rabbit ileal loop test result | ||
|---|---|---|---|---|---|---|---|
| Tox-A TEST | VIDAS CDA assay | Toxin B genea (NK104-NK105)b | Toxin A genea (NK3-NK2)b | ||||
| 1 | − | − | − | − | − | − | |
| 2 | + | − | − | + | + | ca. 700 | − |
| 3 | + | − | − | + | + | ca. 700 | − |
| 4 | + | − | − | + | + | ca. 700 | − |
| 5 | + | + | + | + | + | ca. 1,200 | + |
| 6 | + | + | + | + | + | ca. 1,200 | + |
Nonrepeating region.
Primer set.
Repeating region.
FIG. 1.
Polyacrylamide gel electrophoresis of PCR products of six C. difficile strains. Three primer pairs, NK3-NK2 derived from the nonrepeating portion of the toxin A gene (A), NK104-NK105 derived from the nonrepeating portion of the toxin B gene (B), and NK11-NK9 derived from the repeating portion of the toxin A gene (C), were used for PCR. Lane 1, toxin A−, toxin B− strain; lanes 2 to 4, toxin A−, toxin B+ strains; lanes 5 and 6, toxin A+, toxin B+ strains. The size markers used were a 100-bp ladder (A and B) and pBR322 digested with AvaII and AvaII-EcoRI (C).
On the basis of the results that were obtained, a total of 421 C. difficile strains isolated from 190 children ranging in age from 0 and 14 years and 231 adults older than 14 years of age were examined by the PCR assay. Of the 421 strains tested, 246 were PCR positive and 175 were PCR negative for the nonrepeating region of the both the toxin A and the toxin B genes. Of the 246 PCR-positive strains tested, 48 generated a PCR product of approximately 700 bp and 198 yielded a PCR product of approximately 1,200 bp by PCR for the repetitive region of the toxin A gene.
The VIDAS CDA test was performed for the detection of toxin A with 100 of the 198 strains for which the PCR for the repetitive region of the toxin A gene generated a longer PCR product and with all 48 strains for which the PCR for the repetitive region of the toxin A gene generated a shorter PCR product, resulting in the former strains being positive for toxin A and the latter strains being negative for toxin A. Furthermore, the 48 toxin A−, toxin B+ strains were tested by the Tox-A TEST, with negative results. To examine the strains for a low level of production of toxin A, if any such strains existed, the culture supernatants of the 48 strains were concentrated 10 times and were subjected to the Tox-A TEST, with negative results.
Eventually, the bioassay results completely agreed with those of the PCR assay. Toxin A−, toxin B− strains were negative by PCR with the NK3-NK2 and NK104-NK105 primer sets, toxin A+, toxin B+ strains were positive by PCR with these two primer sets and generated an approximately 1,200-bp product by PCR with the NK11-NK9 primer set, and toxin A−, toxin B+ strains were positive by PCR with these two primer sets and yielded an approximately 700-bp product by PCR with the NK11-NK9 primer set.
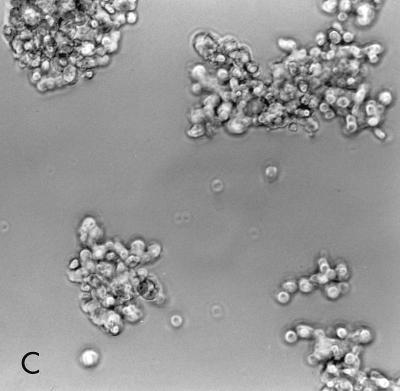
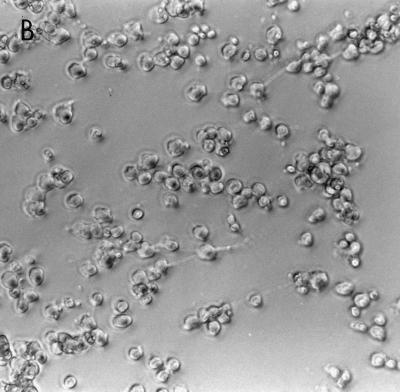
Although the cytotoxin produced by all toxin A−, toxin B+ strains was neutralized by C. difficile anti-toxin B serum, the cytotoxic effect of toxin A−, toxin B+ strains on Vero cell monolayers was apparently distinguishable from that of toxin A+, toxin B+ strains. Culture filtrates of toxin A+, toxin B+ strains caused cell rounding and disrupted cell-to-cell contact; in contrast, Vero cells incubated with culture filtrates of toxin A−, toxin B+ strains appeared as discrete clusters (Fig. 2).
FIG. 2.
Comparison of cytotoxic effects of a toxin A−, toxin B+ strain with those of a toxin A+, toxin B+ strain on a Vero cell sheet. Filtrates of C. difficile strains cultured in brain heart infusion were applied to monolayers of Vero cells. (A) Normal Vero cell monolayer; (B) Vero cells treated with culture filtrate from toxin A+, toxin B+ strain; (C) Vero cells treated with culture filtrate from toxin A−, toxin B+ strain.
Western immunoblot typing with 48 toxin A−, toxin B+ strains showed that 44 strains belonged to serogroup F (subserogroup F-0) and that four strains belonged to serogroup X (subserogroup X-2). All four strains of serogroup X were originally from Indonesia. PFGE analysis after SacII digestion demonstrated a variety of banding patterns among the toxin A−, toxin B+ strains. Overall, PFGE analysis separated the 48 toxin A−, toxin B+ strains into 19 types. No evidence of the monoclonal spread of a toxin A−, toxin B+ strain was found for any cohort studied.
For the 333 subjects whose clinical backgrounds were recorded, we examined whether there was a relation between the symptoms of the diarrheal disorder and the toxigenic patterns of the C. difficile strain (Table 2). Toxin A−, toxin B+ strains were isolated from 6.7 and 12.5% of symptomatic and asymptomatic children, respectively. For adults, no toxin A−, toxin B+ strain was recovered from symptomatic patients, but 12.5% of asymptomatic adults were colonized or infected with toxin A−, toxin B+ strains. Among the 88 subjects whose data are not listed in Table 2 due to their unclear clinical background, 31.8% were positive for toxin A−, toxin B+ strains.
TABLE 2.
Isolation of three toxin-producing types of C. difficile strains from children and adults
| Source | Subject | Symptom | No. (%) of the following toxin-producing types of C. difficile:
|
Total no. of isolates | ||
|---|---|---|---|---|---|---|
| Toxin A+, toxin B+ | Toxin A−, toxin B+ | Toxin A−, toxin B− | ||||
| Meitetsu Hospital | Child | Symptomatic | 30 (28.6) | 7 (6.7) | 68 (64.8) | 105 |
| Child | Asymptomatic | 11 (24.4) | 3 (6.7) | 31 (68.9) | 45 | |
| TDRCa | Child | Asymptomatic | 2 (5.0) | 5 (12.5) | 33 (82.5) | 40 |
| Nagoyashi-Koseiin Geriatric | Adult | Symptomatic | 3 (100) | 0 | 0 | 3 |
| Hospital | Adult | Asymptomatic | 26 (65.0) | 5 (12.5) | 9 (22.5) | 40 |
| CDC | Adult | Symptomatic | 97 (97.0) | 0 | 3 (3.0) | 100 |
| Total | 169 | 20 | 144 | 333 | ||
TDRC, Tropical Disease Research Centre, Surabaya, Indonesia.
DISCUSSION
In this study, we successfully discriminated toxin A+, toxin B+, toxin A−, toxin B+, and toxin A−, toxin B− strains of C. difficile by PCR using two primer sets derived from the nonrepeating sequence of the toxin A and the toxin B genes and a primer set derived from the repetitive sequence of the toxin A gene. PCR for the nonrepeating sequence of the toxin A gene always gave results that were the same as those of PCR for the nonrepeating sequence of the toxin B gene. PCR for the repetitive sequence of the toxin A gene with primer set NK11-NK9 was able to differentiate toxin A−, toxin B+ strains from toxin A+, toxin B+ strains. Thus, PCR with primer set NK11-NK9 combined with either one of the PCR assays detecting the nonrepeating sequence of the toxin A or the toxin B gene can be used to differentiate three types of toxin production. In a previous investigation, Depitre et al. (8) failed to differentiate toxin A−, toxin B+ strains (serogroup F) from toxin A+, toxin B+ strains using PCR with primer set P31-P60 directed at the repeating region of the toxin A gene. We tried to use several primer sets to amplify a portion of the repetitive sequences of the toxin A gene; however, all but primer set NK11-NK9 failed to provide distinct PCR results.
All except four of the toxin A−, toxin B+ strains tested in this study were typed into serogroup F by immunoblotting; the exceptions were four strains recovered in Indonesia that belonged to serogroup X. These findings are consistent with those presented in a previous report of Depitre et al. (8) that serogroup F strains produce toxin B but not toxin A. These observations indicate that C. difficile strains belonging to serogroup F have a unique feature in their toxigenicity. Interestingly, polyclonal antisera against serogroups F and X had a cross-reaction to each other by a dot blot test (data not shown), suggesting that serogroups F and X share some immunological features. PFGE analysis of the toxin A−, toxin B+ strains tested in this study provided no evidence that a single clone was spread within each hospital.
Depitre et al. (8) reported that C. difficile serogroup F strains in Belgium were often isolated from asymptomatic children and that they were all toxin A−, toxin B+ strains. Valenzuela Montero et al. (18) demonstrated that 7 (12.1%) of the 58 cytotoxic strains from healthy Chilean infants were toxin A− by a commercial ELISA. In this study toxin A−, toxin B+ strains were isolated from 6.7% of symptomatic and asymptomatic children (younger than 15 years old) in Japan and 12.5% of healthy children (younger than 2 years old) in Indonesia. Taken together, the studies indicate that, globally, some children, especially younger ones, carry toxin A−, toxin B+ strains of C. difficile in their intestines.
In adults, although the clinical background was unclear for a portion of the subjects studied, toxin A−, toxin B+ strains were recovered at unexpectedly high rates from geographically separate facilities in Japan, indicating that intestinal carriage of these strains is not rare among Japanese adults. In contrast, none of the 100 C. difficile strains recovered from patients with AAD or AAC in the United States were toxin A−, toxin B+. A recent study in the United States by Lyerly et al. (16) showed a low incidence of toxin A−, toxin B+ strains (0.2%). Altaie et al. (2) recovered no toxin A−, toxin B+ strains from 300 stool specimens obtained from patients who were clinically suspected of having AAD. Toxin A−, toxin B+ strains are likely to be of low prevalence in the United States or uncommon, at least in patients with AAD or AAC. So far no information regarding the prevalence of toxin A−, toxin B+ strains in other countries is available.
Although the cytotoxin produced by toxin A−, toxin B+ strains was neutralized by anti-toxin B serum, the appearance of cytotoxic effects on Vero cell monolayers was distinguishable between toxin A+, toxin B+ strains and toxin A−, toxin B+ strains. Interestingly, the toxin B produced by strain 8864 caused a clumping of rounding cells within the cell sheet on Vero cells (5), which was similar to the cytotoxic effects of the toxin A−, toxin B+ strains observed in this study. A difference in the N-terminal region of the toxin B gene between the reference strain of serogroup F (strain 1470) and the toxin A+, toxin B+ strain (strain VPI 10463) has been documented (19). In addition, the morphological response of endothelial cells of the pig pulmonary artery to toxin B of strain 1470 was reported to be different from that to toxin B of toxin A+, toxin B+ strains but indistinguishable from that to the lethal toxin of Clostridium sordellii (19). These results suggest that the structure of toxin B of toxin A−, toxin B+ C. difficile strains may be similar to that of the lethal toxin of C. sordellii and different in part from that of the toxin B of toxin A+, toxin B+ C. difficile strains.
Nevertheless, the enteropathogenicity of toxin A−, toxin B+ strains remains unclear. We tested seven toxin A−, toxin B+ strains (three from children and four from adults) for enterotoxic activities in a rabbit ileal loop assay, but the result was no fluid accumulation. The results suggest that toxin A−, toxin B+ strains may not cause diarrheal disease in humans. Additional experiments including an experiment with a hamster model and a study of the prevalence of toxin A−, toxin B+ strains among symptomatic and asymptomatic subjects will be required to determine the role of toxin A−, toxin B+ strains in intestinal disorders.
In conclusion, PCR assay for the detection of the repeating sequence of the toxin A gene is useful as a tool for discriminating toxin A+, toxin B+, toxin A−, toxin B+, and toxin A−, toxin B− strains. It is a simple and rapid technique for the identification of toxin A−, toxin B+ strains and should be valuable in elucidating the pathogenic and epidemic potentials of these strains.
ACKNOWLEDGMENTS
We thank Kakuyo Sawa of Gifu University Hospital for sample collection and George E. Killgore of the CDC for editing this article.
REFERENCES
- 1.Allen S D, Baron E J. Clostridium. In: Balows A, Hausler W J Jr, Herrmann K L, Isenberg H D, Shadomy H J, editors. Manual of clinical microbiology. 5th ed. Washington, D.C: American Society for Microbiology; 1991. pp. 505–521. [Google Scholar]
- 2.Altaie S S, Penque P H, Mookherjee S, Evans D T. Should laboratories test for toxin A-negative, toxin B-positive Clostridium difficile? Lab Med. 1996;27:468–471. [Google Scholar]
- 3.Banno Y, Kobayashi T, Kono H, Watanabe K, Ueno K, Nozawa Y. Biochemical characterization and biologic actions of two toxins (D-1 and D-2) from Clostridium difficile. Rev Infect Dis. 1984;6:S11–S20. doi: 10.1093/clinids/6.supplement_1.s11. [DOI] [PubMed] [Google Scholar]
- 4.Barroso L A, Wang S Z, Phelps C J, Johnson J L, Wilkins T D. Nucleotide sequence of Clostridium difficile toxin B gene. Nucleic Acids Res. 1990;18:4004. doi: 10.1093/nar/18.13.4004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Borriello S P, Wren B W, Hyde S, Seddon S V, Sibbons P, Krishna M M, Tabaqchali S, Manek S, Price A B. Molecular, immunological, and biological characterization of a toxin A-negative, toxin B-positive strain of Clostridium difficile. Infect Immun. 1992;60:4192–4199. doi: 10.1128/iai.60.10.4192-4199.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Delmee M, Avesani V. Virulence of ten serogroups of Clostridium difficile in hamsters. J Med Microbiol. 1990;33:85–90. doi: 10.1099/00222615-33-2-85. [DOI] [PubMed] [Google Scholar]
- 7.Delmee M, Laroche Y, Avesani V, Cornelis G. Comparison of serogrouping and polyacrylamide gel electrophoresis for typing Clostridium difficile. J Clin Microbiol. 1986;24:991–994. doi: 10.1128/jcm.24.6.991-994.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Depitre C, Delmee M, Avesani V, L’Haridon R, Roels A, Popoff M, Corthier G. Serogroup F strains of Clostridium difficile produce toxin B but not toxin A. J Med Microbiol. 1993;38:434–441. doi: 10.1099/00222615-38-6-434. [DOI] [PubMed] [Google Scholar]
- 9.Gumerlock P H, Tang Y J, Meyers F J, Silva J., Jr Use of the polymerase chain reaction for the specific and direct detection of Clostridium difficile in human feces. Rev Infect Dis. 1991;13:1053–1060. doi: 10.1093/clinids/13.6.1053. [DOI] [PubMed] [Google Scholar]
- 10.Jotwani R, Kato N, Kato H, Watanabe K, Ueno K. Detection of Bacteroides fragilis in clinical specimens by polymerase chain reaction amplification of the neuraminidase gene. Curr Microbiol. 1995;31:215–219. doi: 10.1007/BF00298376. [DOI] [PubMed] [Google Scholar]
- 11.Kato H, Cavallaro J J, Kato N, Bartley S L, Killgore G E, Watanabe K, Ueno K. Typing of Clostridium difficile by Western immunoblotting with 10 different antisera. J Clin Microbiol. 1993;31:413–415. doi: 10.1128/jcm.31.2.413-415.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kato H, Kato N, Watanabe K, Ueno K, Ushijima H, Hashira S, Abe T. Application of typing by pulsed-field gel electrophoresis to the study of Clostridium difficile in a neonatal intensive care unit. J Clin Microbiol. 1994;32:2067–2070. doi: 10.1128/jcm.32.9.2067-2070.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kato N, Ou C Y, Kato H, Bartley S L, Brown V K, Dowell V R, Jr, Ueno K. Identification of toxigenic Clostridium difficile by the polymerase chain reaction. J Clin Microbiol. 1991;29:33–37. doi: 10.1128/jcm.29.1.33-37.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lyerly D M, Barroso L A, Wilkins T D, Depitre C, Corthier G. Characterization of a toxin A-negative, toxin B-positive strain of Clostridium difficile. Infect Immun. 1992;60:4633–4639. doi: 10.1128/iai.60.11.4633-4639.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lyerly D M, Krivan H C, Wilkins T D. Clostridium difficile: its disease and toxins. Clin Microbiol Rev. 1988;1:1–18. doi: 10.1128/cmr.1.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lyerly D M, Neville L M, Evans D T, Fill J, Allen S, Greene W, Sautter R, Hnatuck P, Torpey D J, Schealbe R. Multicenter evaluation of the Clostridium difficile TOX A/B TEST. J Clin Microbiol. 1998;36:184–190. doi: 10.1128/jcm.36.1.184-190.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lyerly D M, Saum K E, MacDonald D K, Wilkins T D. Effects of Clostridium difficile toxins given intragastrically to animals. Infect Immun. 1985;47:349–352. doi: 10.1128/iai.47.2.349-352.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Valenzuela Montero M E, Lobos T, Valenzuela A, Roos O, Rodriguez L, Piemonte P, Soledad Giglio M. Prevalence Clostridium difficile among healthy Chilean infants: evaluation by commercial enzyme immunoassay versus standard cytotoxin assay. Clin Infect Dis. 1995;20:S259–S260. doi: 10.1093/clinids/20.supplement_2.s259. [DOI] [PubMed] [Google Scholar]
- 19.von Eichel-Streiber C, Meyer zu Heringdorf D, Habermann E, Sartingen S. Closing in on the toxic domain through analysis of a variant Clostridium difficile cytotoxin B. Mol Microbiol. 1995;17:313–321. doi: 10.1111/j.1365-2958.1995.mmi_17020313.x. [DOI] [PubMed] [Google Scholar]