Abstract
This report describes a new statistical method for estimating the MIC of fluconazole for yeasts pathogenic to humans. This method is based on comparison of the colony sizes on solid media containing different concentrations of fluconazole. By this method, the MICs of fluconazole for 10 yeast strains were comparable to results obtained by the standard method recommended by the National Committee for Clinical Laboratory Standards. This method is simple to perform and easy to interpret. The turnaround time is faster than other methods. The method should be applicable to the determination MICs of other antifungal drugs for yeasts.
Because of increases in the number of yeast infections, the drugs available for their management, and the emergence of resistant isolates, antifungal drug susceptibility testing has become an important component of current health care management (1). In 1995, in a landmark publication in the field of antifungal susceptibility testing, the National Committee for Clinical Laboratory Standards (NCCLS) published the Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, which proposed a standard method (M27-T) to determine yeast susceptibilities (2). Over the past 2 years, the application of M27-T has improved the interlaboratory agreement of MIC results (5). However, this macrodilution procedure is laborious and relatively expensive. It has not been widely adopted in clinical laboratories. Several new methods have been developed recently, including various broth microdilution procedures, E tests, and a disk diffusion method (4, 5). In most cases, the results obtained with these new methods were comparable to those obtained by the M27-T. In many of the studies comparing different methods for a given set of strains, MICs for more than 80% of the strains were within a fourfold drug concentration range (5, 6). These new methods are somewhat less laborious, but similar to the NCCLS method, they are affected by the size and age of the inoculum and by the time of incubation.
Despite publication of numerous MIC methods and their widespread use, all lack statistical meaning. The endpoint readings are arbitrary, though by definition, the MIC is the lowest concentration of drug(s) that significantly inhibits microbial growth (statistically) compared to growth in the absence of drug.
Here we evaluate a method that has the potential to overcome the problems associated with the other methods. This method measures and compares colony sizes of individual strains at different drug concentrations on solid agar medium. This colony size method had been used to test the drug susceptibility of phytopathogenic molds (3), but not yeasts pathogenic to humans.
Ten strains, distributed among six pathogenic yeast species, were used in this study. The strain designations and geographic origins are shown in Table 1. A synthetic medium, yeast morphology agar (YMA; Difco, Detroit, Mich.), was used. A typical petri dish (100 by 15 mm) contains 20 to 25 ml of YMA. As in the M27-T method, each strain was tested with a no-drug control and at the following 12 concentrations of fluconazole, which were incorporated into YMA: 0.125, 0.25, 0.5, 1, 2, 4, 8, 16, 32, 64, 128, and 256 μg/ml. For each strain, two types of inocula were compared: one prepared from a fresh culture incubated at 35°C for 2 days and another used after storage at 4°C for a week.
TABLE 1.
Comparison of two methods to determine the MICs of fluconazole for six yeast species
| Species | Straina | Geographic origin | MIC (μg/ml) by method:
|
|||||
|---|---|---|---|---|---|---|---|---|
| Colony size
|
NCCLS | |||||||
| Old inoculum; 24 h | Fresh inoculum
|
|||||||
| 24 h | 48 h | 72 h | 96 h | |||||
| Cryptococcus neoformans var. neoformans | DUMC-3 | North Carolina | 8 | 16 | 16 | 8 | 8 | 16 |
| Cryptococcus neoformans var. gattii | ATCC 32269 | Zaire | 1 | 16 | 4 | 2 | 2 | 4 |
| Candida albicans | MMRL390 | North Carolina | 1 | 1 | 0.5 | 0.25 | 0.25 | 0.5 |
| MMRL419 | North Carolina | 128 | 64 | 64 | 64 | 64 | 64 | |
| Saccharomyces cerevisiae | MMRL323 | Brazil | 32 | 32 | 8 | 8 | 8 | 32 |
| MMRL325 | Brazil | 8 | 8 | 8 | 8 | 8 | 16 | |
| Candida krusei | MMRL310 | Brazil | 4 | 8 | 4 | 4 | 4 | 16 |
| MMRL413 | North Carolina | 1 | 2 | 2 | 2 | 2 | 4 | |
| Candida tropicalis | MMRL319 | Brazil | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 | 1 |
| Candida glabrata | MMRL330 | Brazil | 32 | 64 | 128 | 128 | 128 | 32 |
MMRL, Medical Mycology Research Laboratory at Duke University; DUMC, Duke University Medical Center.
The protocol for the colony size method is as follows. (i) A loopful of cells from a single colony grown on an agar plate is transferred to a 0.5-ml sterile microcentrifuge tube containing 0.1 ml of sterile physiological saline (0.9% NaCl). This is then vortexed at high speed for 5 s or mixed by vigorous finger tapping to separate and suspend cells. (ii) With an inoculation loop, one loopful of the yeast suspension is streaked on each of the 13 plates (50 mm in diameter in the present study) of YMA containing the different drug concentrations. The streaking is done for isolated colonies, since it is essential that the resulting colonies be well separated on each plate. (iii) The plates are incubated at 35°C for up to 96 h. (iv) To determine the MIC, each plate is inverted on the stage of a light microscope. At a magnification of ×50 to ×100, the ocular scale is used to measure the diameters of ≥20 random colonies at each drug concentration.
For plates with the old inoculum, colonies were measured after incubation for 24 h. For plates with a fresh inoculum, colonies were measured at 24, 48, 72, and 96 h. For each isolate, the mean colony size and standard deviation were calculated at each drug concentration. The diameters of colonies on control and drug-containing plates were compared by analysis of variance or Student’s t test (7), with P < 0.01 as the threshold of significance. The MIC for each strain was defined as the lowest drug concentration that significantly decreased the diameter of the colonies compared with the mean colony size in the absence of drug. For each strain, the MIC of fluconazole was also determined by the standard M27-T protocol; strains of Cryptococcus neoformans were assessed after 72 h of incubation, and strains of the other species, were assessed after 48 h of incubation.
The MICs for all 10 strains were determined by both the colony size method and the M27-T method and are presented in Table 1. Overall, the MIC estimates from the colony size method are comparable to those obtained by the standard broth macrodilution method. For nine of the isolates, the MICs determined by the two methods were within a fourfold drug concentration of each other, even when the ages of inocula and lengths of incubations differed. The only exception was the strain of C. neoformans var. gattii, for which MICs varied widely at the different time periods (up to an 8-fold difference [Table 1]) and between the old and the fresh inocula after 24 h of incubation (a 16-fold difference [Table 1]). Nevertheless, each MIC estimate for this strain was within a fourfold concentration of the MIC estimated by the M27-T method (Table 1). The density of colonies on the plates did not affect the size measurements as long as ≥20 colonies were well separated. The same procedure was repeated for these strains after a 3-month period, and the MIC estimates were all within a twofold concentration of the original determinations (data not shown), which suggests that the colony size method has a high degree of reproducibility.
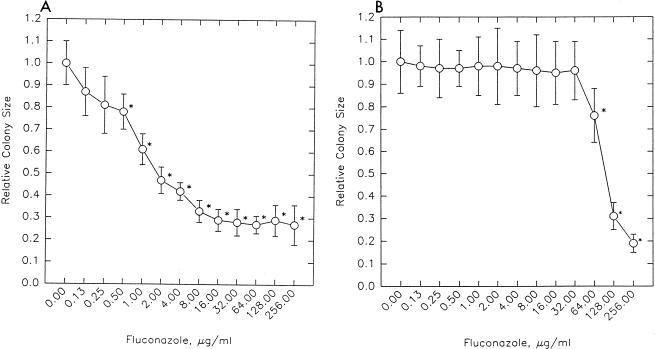
The graphs in Fig. 1 show the relationship between colony size and drug concentration. The figure depicts data after 48 h for two strains of Candida albicans, MMRL390 and MMRL419. The measurements of colony size are presented relative to the average colony diameter without the drug. These strains reflect two different patterns of fluconazole susceptibility. The MIC for strain MMRL390 is low, 0.5 μg/ml. MMRL390’s colony diameter decreased relatively slowly as the drug concentration increased; at the highest concentration of fluconazole, 256 μg/ml, the colony diameter was 27% of the colony diameter in the absence of drug. In contrast, the MIC for strain MMRL419 is high, 64 μg/ml. MMRL419’s colony diameter decreased only slightly from control growth until the concentration of fluconazole was 32 μg/ml, beyond which the colony diameters decreased sharply; at a fluconazole concentration of 256 μg/ml, the relative size was only 19% of the size without fluconazole (Fig. 1).
FIG. 1.
Relative colony sizes of two C. albicans strains at 13 drug concentrations. Drug concentrations are shown on the x axis, and relative average colony sizes and standard deviations are on the y axis. (A) C. albicans MMRL390 (fluconazole MIC, 0.5 μg/ml). (B) C. albicans MMRL419 (fluconazole MIC, 64 μg/ml). An asterisk denotes statistical significance (P < 0.01).
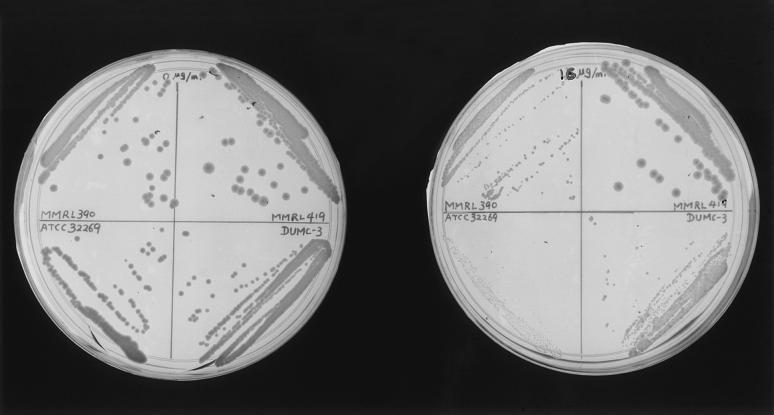
The colony size method is simple, fast, and inexpensive and requires no instrumentation, only a microscope with an ocular scale. MICs for all species tested could be determined after incubation for 24 h. Even without using the microscope, simple comparisons of the colony sizes could usually determine the MICs for strains (Fig. 2). Multiple isolates could be streaked on a single petri dish. Most importantly, the colony size method could provide a statistical meaning to the MIC.
FIG. 2.
Colony size comparisons showing growth on a control plate (without drug) on the left and a plate with 16 μg of fluconazole per ml on the right. Each of the four strains was streaked on a quadrant of a 5-cm plate (reading clockwise from the upper left): C. albicans MMRL390, C. albicans MMRL419, C. neoformans var. neoformans DUMC-3, and C. neoformans var. gattii ATCC 32269. The plates depict growth after incubation at 35°C for 48 h. Compared with growth on the control plate, there are significant decreases in the colony sizes of three of the yeasts at a fluconazole concentration of 16 μg/ml. C. albicans MMRL419 (upper right quadrant) is resistant to this concentration of fluconazole.
This method could be readily and inexpensively incorporated into the routine procedures of the clinical mycology laboratory. An abbreviated protocol could be used to screen relevant clinical isolates for resistance to fluconazole. In this report, we measured colony sizes at all of the twofold concentrations recommended by NCCLS. However, for screening purposes, plates containing only selected drug concentrations (e.g., 1, 8, and 32 μg/ml) could be prepared and stored. Clinical isolates of C. albicans could then be streaked on each plate, as well as on the no-drug control plate. The subsequent comparison of growth would provide tentative evidence of susceptibility or resistance. We are now in the process of testing additional species, more isolates of each species, and more drugs to evaluate the possibility of a broader application of this method.
Acknowledgments
This research was supported by Public Health Service grants AI25783 and AI28836 from the National Institutes of Health.
REFERENCES
- 1.Lederberg J, Shope R E, Oaks S C., Jr . Emerging infections: microbial threats to health in the United States. Washington, D.C: National Academy Press; 1992. [PubMed] [Google Scholar]
- 2.National Committee for Clinical Laboratory Standards. Reference method for broth dilution antifungal susceptibility testing of yeasts: tentative standard M27-T. Wayne, Pa: National Committee for Clinical Laboratory Standards; 1995. [Google Scholar]
- 3.Peever T L, Milgroom M G. Lack of correlation between fitness and resistance to sterol biosynthesis-inhibiting fungicides in Pyrenophora teres. Phytopathology. 1994;84:515–519. [Google Scholar]
- 4.Pfaller M A, Barry A L. Evaluation of a novel colorimetric broth microdilution method for antifungal susceptibility testing of yeast isolates. J Clin Microbiol. 1994;32:1992–1996. doi: 10.1128/jcm.32.8.1992-1996.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pfaller M A, Rex J H, Rinaldi M G. Antifungal susceptibility testing: technical advances and potential clinical applications. Clin Infect Dis. 1997;24:776–784. doi: 10.1093/clinids/24.5.776. [DOI] [PubMed] [Google Scholar]
- 6.Sewell D L, Pfaller M A, Barry A L. Comparison of broth macrodilution, broth microdilution, and E test antifungal susceptibility tests for fluconazole. J Clin Microbiol. 1994;32:2099–2102. doi: 10.1128/jcm.32.9.2099-2102.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sokal R R, Rohlf F J. Biometry: the principles and practices of statistics in biological research. 2nd ed. New York, N.Y: W. H. Freeman & Company; 1981. [Google Scholar]