Abstract
Background
It is unclear whether curative-intent local therapy of metastases is of similar benefit for the biological distinct subgroup of patients with deficient mismatch repair (dMMR) metastatic colorectal cancer (mCRC) compared with proficient mismatch repair (pMMR) mCRC.
Patients and Methods
In this nationwide study, recurrence-free (RFS) and overall survival (OS) were analyzed in patients with dMMR versus pMMR mCRC who underwent curative-intent local treatment of metastases between 2015 and 2018. Subgroup analyses were performed for resection of colorectal liver metastases (CRLM) and cytoreductive surgery ± hyperthermic intraperitoneal chemotherapy (CRS ± HIPEC). Multivariable regression was conducted.
Results
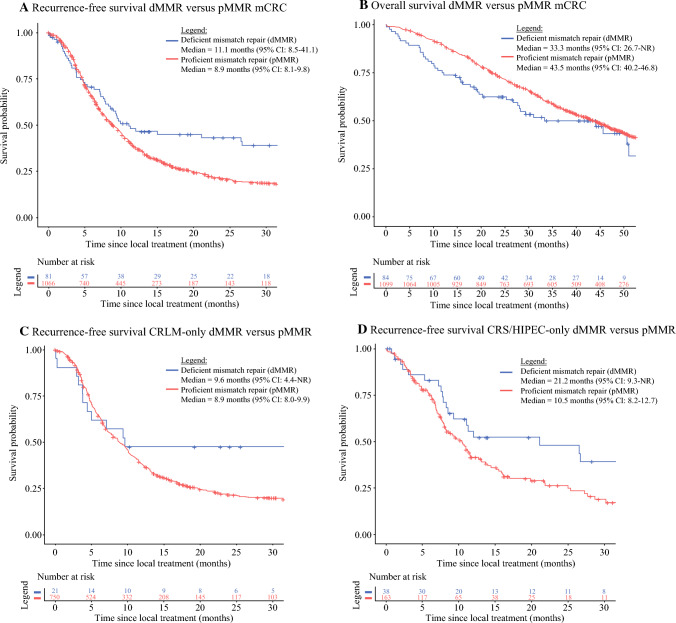
Median RFS was 11.1 months [95% confidence interval (CI) 8.5–41.1 months] for patients with dMMR tumors compared with 8.9 months (95% CI 8.1–9.8 months) for pMMR tumors. Two-year RFS was higher in patients with dMMR versus pMMR (43% vs. 21%). Results were similar within subgroups of local treatment (CRLM and CRS ± HIPEC). Characteristics differed significantly between patients with dMMR and pMMR mCRC; however, multivariable analysis continued to demonstrate dMMR as independent factor for improved RFS [hazard ratio (HR): 0.57, 95% CI 0.38–0.87]. Median OS was 33.3 months for dMMR mCRC compared with 43.5 months for pMMR mCRC, mainly due to poor survival of patients with dMMR in cases of recurrence in the preimmunotherapy era.
Conclusion
Patients with dMMR eligible for curative-intent local treatment of metastases showed a comparable to more favorable RFS compared with patients with pMMR, with a clinically relevant proportion of patients remaining free of recurrence. This supports local treatment as a valuable treatment option in patients with dMMR mCRC and can aid in shared decision-making regarding upfront local therapy versus immunotherapy.
Supplementary Information
The online version contains supplementary material available at 10.1245/s10434-023-13974-7.
Metastatic colorectal cancer (mCRC) is a highly heterogeneous disease with approximately 5% of patients having a tumor harboring deficient mismatch repair (dMMR).1,2 dMMR mCRC forms a poorer biological subgroup with distinct prognostic, predictive, and therapeutic implications compared with proficient mismatch repair (pMMR) mCRC.2 Local treatment of metastases prolongs overall survival (OS) and is, up to now, recommended in all patients with resectable mCRC.3 Consensus of curative-intent local therapy of metastases was mainly based on studies including patients with pMMR mCRC, and its benefit is not known for patients with dMMR mCRC.3 However, there are important differences between dMMR and pMMR tumors. Patients with dMMR mCRC obtain a lower response rate to palliative systemic therapy (5% vs. 44%)4,5 and the OS in the preimmunotherapy era is shorter (16 vs. 24 months).6 Furthermore, there are important differences in patterns of metastatic spread; dMMR tumors more often metastasize to the lymph nodes and pMMR tumors to the liver, making patients with dMMR tumors less often amenable to surgery (10% vs. 26%).7
Currently, immunotherapy leads to durable responses as a first-line systemic treatment in patients with dMMR mCRC with a prolonged progression-free survival of 16.5 months in patients receiving monoimmunotherapy compared with 8.2 months in patients receiving systemic palliative therapy in the randomized phase III KEYNOTE-177 trial.8 Around 30% of patients that received immunotherapy had primary progression; however, patients that responded to immunotherapy often had a long duration of response with 48% of patients being progression-free at 2 years. Furthermore, small studies have showed that salvage local treatment after immunotherapy treatment in patients with an irresectable primary tumor or mCRC is effective with a 2-year recurrence-free survival (RFS) of 62–81%, although complete histological responses were already observed in 56–93% of cases.9–11 This setting concerns salvage local treatment after immunotherapy. With the remarkable results of immunotherapy, the value of upfront local therapy treatment in patients with dMMR mCRC is questioned.8 However, there is only limited data regarding local treatment in patients with mCRC and resectable metastases without upfront immunotherapy.
The aim of this study is to describe the characteristics, RFS, and OS after curative-intent local treatment of metastases in patients with dMMR versus pMMR tumors in the period before immunotherapy, which was approved as standard of care. This allows evaluation of clinical outcomes after local treatment for patients with dMMR mCRC without upfront immunotherapy and will provide valuable knowledge for shared decision making regarding immunotherapy or upfront local treatment in case of resectable metastases.
Patients and Methods
Study Design and Data Collection
This is an observational nationwide study using specified individual patient data of The Netherlands Cancer Registry (NCR), collected from medical records of all Dutch hospitals by trained data managers. Race/ethnicity is not registered by the NCR, because this is not allowed in The Netherlands. All patients in The Netherlands receiving curative-intent local treatment of metastases with dMMR mCRC from 2015 to 2018 were selected, as well as those with pMMR mCRC from 2015 to 2016. Curative-intent local treatment of metastases included metastasectomy, radiofrequency ablation, or microwave ablation of all visible metastases.
MMR and/or microsatellite instability (MSI) status was collected from patient records when determined during routine clinical practice. Patients with unknown MMR and/or microsatellite instability status were excluded. MMR expression was defined as deficient when there was a loss in protein expression of either MLH1, PMS2, MSH2, or MSH6. MSI was assessed with the mononucleotide repeat pentaplex panel (BAT-25, BAT-26, NR-21, MONO-27, and NR-24) and determined as MSI high when at least two markers showed MSI. Identification of Lynch syndrome or sporadic dMMR/MSI-H was based on a tailored approach by MMR protein expression, family history, BRAFV600E status, and MLH1 promotor hypermethylation status, as previously described by Parsons et al.12 The NCR was linked with The Dutch Nationwide Pathology Databank (PALGA) to obtain original pathology reports, including BRAFV600E and RAS status, if determined during routine clinical practice. Since RAS and BRAFV600E mutations are considered mutually exclusive, RAS was considered wild-type when RAS mutation status was unknown and a BRAFV600E mutation was present and vice versa.13
Local therapy was categorized into colorectal liver metastasectomy (CRLM), cytoreductive surgery with or without hyperthermic intraperitioneal chemotherapy (CRS ± HIPEC), pulmonary metastasectomy, lymph node metastasectomy, and other/combination local therapy (e.g., cerebral metastasectomy or combination of CRLM and CRS ± HIPEC). CRLM included liver metastasectomy, radiofrequency ablation, and microwave ablation. Sidedness of the primary tumor was defined as right-sided comprising tumors from cecum up to the transverse colon and left-sided from splenic flexure up to sigmoid. World Health Organization (WHO) performance status was measured before the start of treatment. Staging was determined by pathological stage and, when necessary, complemented with clinical stage.
Outcome
RFS was the primary outcome and defined as time from local treatment to recurrence of disease or death, whichever occurred first, and OS as time from local treatment to death or last follow-up alive.
The NCR obtained information on RFS until August 2020 and was linked with the National Municipal Personal Records Database in February 2021 to obtain the most recent information on vital status. Imaging modalities and frequencies were performed according to local clinical practice. The NCR data were pseudomized and consent was obtained by an opt-out approach.
Statistical Analysis
Differences between baseline characteristics were analyzed with a t-test for continous variables and chi-squared test for categorical variables. Kaplan–Meier curves were obtained with censoring of patients that were lost to follow-up. Median follow-up was calculated by the reverse Kaplan–Meier approach. Multiple imputation by a substantive model compatible with fully conditional specification was used for missing data.14 A univariable and multivariable Cox regression model was derived with ten preselected factors, based on literature and expert opinion.15,16 Proportional hazards were visually checked with Schoenfeld residuals and statistically tested. A p-value < 0.05 was considered statistically significant. All analyses were performed in R version 3.5.1 (packages ‘gtsummary’, ‘smcfcs’, ‘survminer’, ‘survival’, ‘prodlim,’ and ‘table’ were used).17
Results
Patient Characteristics
A total of 84 of 380 patients with dMMR mCRC (22%) received local treatment versus 1099 of 2319 patients with pMMR mCRC (47%), as expected based on the known different metastatic patterns and consequent lower amenability for local therapy for patients with dMMR mCRC. The mean age and WHO performance status was comparable between patients with dMMR mCRC and pMMR mCRC, while sex (38% vs. 58% male, p-value < 0.01), BRAFV600E mutation status (50% vs. 4%, p-value < 0.01), RAS mutation status (21% vs. 52%, p-value < 0.01), site of primary tumor (78% right-sided vs. 27%, p-value < 0.01), liver-only disease (27% vs. 70%, p-value < 0.01), and peritoneal involvement (44% vs. 18%, p-value < 0.01) differed significantly (Table 1). This also led to significant differences in type of local therapy, whereas CRLM-only resection was performed in 27% of patients with dMMR mCRC compared with 70% of pMMR mCRC and CRS/HIPEC in 48% and 16%, respectively. Neoadjuvant systemic chemotherapy was significantly more often administered in patients with pMMR tumors than dMMR tumors (42% vs. 17%, p-value < 0.01). All patients were included in the period before immunotherapy, which was approved as standard of care.
Table 1.
Patient and treatment characteristics
| pMMR (N = 1099) | dMMR (N = 84) | p-value | |
|---|---|---|---|
| Sex, no. of male | 632 (58%) | 32 (38%) | < 0.01 |
| Age in years, mean (SD) | 61.1 (10.2) | 63.1 (11.7) | 0.14 |
| Sidedness | |||
| Left-sided | 443 (41%) | 14 (17%) | < 0.01 |
| Right-sided | 293 (27%) | 65 (78%) | |
| Rectosigmoid/rectum | 356 (32%) | 4 (5%) | |
| Missing | 7 | 1 | |
| Resection status of primary tumor | |||
| No resection | 57 (5%) | 1 (1%) | 0.17 |
| Resection | 1042 (95%) | 83 (99%) | |
| Differentation grade | |||
| Well | 22 (2%) | 0 (0%) | < 0.01 |
| Moderate | 885 (89%) | 37 (54%) | |
| Poor | 83 (8%) | 31 (46%) | |
| Missing | 109 | 16 | |
| T stage | |||
| T1–3 | 782 (72%) | 39 (47%) | < 0.01 |
| T4 | 301 (28%) | 44 (53%) | |
| Missing | 16 | 1 | |
| N stage | |||
| N0 | 344 (32%) | 28 (33%) | 0.83 |
| N1/2 | 746 (68%) | 56 (67%) | |
| Missing | 9 | 0 | |
| Stage | |||
| Stage I–III | 335 (30%) | 37 (45%) | 0.01 |
| Stage IV | 764 (70%) | 46 (55%) | |
| Missing | 0 | 1 | |
| WHO performance status | |||
| 0 | 331 (68%) | 17 (63%) | 0.88 |
| 1+ | 159 (32%) | 10 (37%) | |
| Missing | 609 | 57 | |
| Metastatic localization | |||
| Liver only | 769 (70%) | 23 (27%) | < 0.01 |
| Peritoneal involvement | 201 (18%) | 37 (44%) | |
| Other | 129 (12%) | 24 (29%) | |
| Number of metastatic sites | |||
| 1 | 878 (80%) | 72 (86%) | 0.41 |
| 2+ | 221 (20%) | 12 (14%) | |
| Molecular status | |||
| BRAFV600E mutation | 22 (4%) | 26 (50%) | < 0.01 |
| RAS mutation | 322 (52%) | 11 (21%) | |
| BRAFV600E and RAS wildtype | 274 (44%) | 15 (29%) | |
| Missing | 481 | 32 | |
| Lynch syndrome status | |||
| Probable Lynch | – | 21 (31%) | – |
| Sporadic | – | 46 (69%) | |
| Missing | – | 17 | |
| Local therapy | |||
| CRLM only | 769 (70%) | 23 (27%) | < 0.01 |
| CRS/HIPEC only | 172 (16%) | 40 (48%) | |
| Pulmonary metastasectomy only | 38 (3%) | 1 (1%) | |
| Lymph node resection only | 19 (2%) | 10 (12%) | |
| Other/combination* | 101 (9%) | 10 (12%) | |
| Systemic therapy | |||
| Neoadjuvant systemic therapy | 457 (42%) | 14 (17%) | < 0 .01 |
| Adjuvant systemic therapy after local therapy | 101 (9%) | 13 (15%) | |
| No perioperative systemic therapy | 541 (49%) | 57 (68%) | |
Bold-values are considered significant (p-value < 0.05)
CRLM colorectal liver metastases, CRS cytoreductive surgery, dMMR deficient mismatch repair, HIPEC hyperthermic intraperitoneal chemotherapy, mCRC metastatic colorectal cancer, pMMR proficient mismatch repair, SD standard deviation, WHO World Health Organization
*Other local treatment: metastasectomy of other organs (e.g., cerebral metastasectomy) or combination treatment (e.g., CRLM and CRS/HIPEC)
Recurrence-Free and Overall Survival
Follow-up for patients with mCRC with a dMMR tumor was median 42.3 months [interquartile range (IQR) 29.5–49.1 months] and for pMMR it was a median of 51.6 months (IQR 45.8–58.6 months). Median RFS was 11.1 months (95% CI 8.5–41.1 months) for patients with a dMMR tumor compared with 8.9 months (95% CI 8.1–9.8 months) for pMMR. A proportion of 43% of patients with dMMR mCRC were recurrence-free 2 years after local treatment, compared with 21% of patients with pMMR. Stratified for type of local treatment, the same trend was shown for CRS ± HIPEC and resection of CRLM (Fig. 1). The improved RFS for patients with dMMR tumors is therefore not a result of differences in type of local therapy. No large differences in RFS were observed when stratifying for molecular groups. Patients with dMMR mCRC and a BRAFV600E-mutated tumor showed a median RFS of 7.1 months (95% CI 3.9–N/R months, n = 26), with a RAS mutation RFS of 7.1 months (95% CI 5.0–N/R months, n = 10) and with a BRAFV600E/RAS wild-type RFS of 9.4 months (95% CI: 7.2–N/R months, n = 13). Comparable outcomes were shown for patients with pMMR mCRC, demonstrating a median RFS of 7.2 months (95% CI: 4.8–11.5 months, n = 22), 7.0 months (95% CI: 6.3–7.8 months, n = 318), and 7.4 months (95% CI 6.6–9.4 months, n = 269), respectively.
Fig. 1.
Recurrence-free and overall survival of deficient mismatch repair (dMMR) versus proficient mismatch repair (pMMR) metastatic colorectal cancer patients (A, B), recurrence-free survival in subgroups of colorectal liver metastases treated patients (C), and cytoreductive surgery/hyperthermic intraperitoneal chemotherapy treated patients (D)
Median OS for patients with dMMR mCRC was 33.3 months (95% CI 26.7–N/R months) compared with 43.5 months for patients with pMMR mCRC (95% CI 40.2–46.8 months), mainly due to poor survival of patients with dMMR mCRC in case of recurrence in the preimmunotherapy era. A total of 822 patients had recurrence of disease and additional follow-up was available for 98% (n = 39/40) for patients with dMMR tumors and 79% (n = 619/782) for patients with pMMR tumors. After recurrence, palliative systemic chemotherapy was administered in 56% of patients with dMMR tumors and 53% of patients with pMMR tumors. Three patients with dMMR tumors received immunotherapy after recurrence in a trial setting. Salvage local therapy occurred in 6 patients with dMMR mCRC (15%, N = 1 hepatectomy, N = 3 CRS ± HIPEC, N = 2 other) and 160 patients with pMMR mCRC (26%, N = 85 hepatectomy, N = 20 CRS ± HIPEC, N = 55 other). The difference in chance of salvage local treatment is mainly due to differences in metastasis pattern with more liver-only recurrences in patients with pMMR tumors (N = 213, 34%) compared with dMMR tumors (N = 3, 8%). In case of recurrence, a median OS of 10.2 months (95% CI 8.0–24.3 months) for dMMR mCRC and 27.4 months (95% CI 25.5–30.6 months) for pMMR mCRC was demonstrated (Supplementary Fig. S1).
Uni- and Multivariable Analysis
Univariable analyses of ten preselected factors showed a significantly improved RFS for dMMR compared with pMMR (HR 0.62, 95% CI 0.44–0.88). Next to dMMR status, stage of disease (stage IV vs. stage I–III) was a significant predictor in univariable analysis (HR 1.22, 95% CI 1.04–1.43). Multivariable analysis demonstrated dMMR status as independent predictor for improved RFS (HR 0.57, 95% CI 0.38–0.87). Both stage IV disease (HR 1.24, 95% CI 1.03–1.49) and poor differentiation grade of primary tumor (HR 1.38, 95% CI 1.03–1.84) were predictive of a poor RFS (Table 2). Notably, perioperative systemic therapy, BRAF mutation status, sidedness, and type of local therapy were not univariable nor multivariable significant predictors.
Table 2.
Univariable and multivariable regression of preselected factors on recurrence-free survival
| Variable | Category | Univariable regression | Multivariable regression | |||||
|---|---|---|---|---|---|---|---|---|
| HR1 | 95% CI1 | p-value | HR1 | 95% CI1 | p-value | |||
| Age | – | – | 1.00 | 0.99, 1.00 | 0.4 | 1.00 | 0.99, 1.00 | 0.3 |
| Sex | Female | (vs. male) | 0.91 | 0.78, 1.06 | 0.2 | 0.88 | 0.75, 1.03 | 0.1 |
| Sidedness | Right-sided | (vs. left-sided) | 0.95 | 0.79, 1.14 | 0.6 | 0.98 | 0.80, 1.19 | 0.8 |
| Rectosigmoid | (vs. left-sided) | 1.18 | 0.99, 1.40 | 0.1 | 1.18 | 0.98, 1.41 | 0.1 | |
| Differentiation grade | Poor | (vs. moderate/well) | 1.16 | 0.90, 1.49 | 0.2 | 1.38 | 1.03, 1.84 | 0.030 |
| Stage | Stage IV | (vs. stage I–III) | 1.22 | 1.04, 1.43 | 0.017 | 1.24 | 1.03, 1.49 | 0.024 |
| BRAFV600E mutation status | Mutation | (vs. wildtype) | 0.96 | 0.65, 1.43 | 0.9 | 1.41 | 0.82, 2.42 | 0.2 |
| RAS mutation status | Mutation | (vs. wildtype) | 1.11 | 0.93, 1.33 | 0.2 | 1.15 | 0.92, 1.42 | 0.2 |
| Mismatch repair status | dMMR | (vs. pMMR) | 0.62 | 0.44, 0.88 | 0.007 | 0.57 | 0.38, 0.87 | 0.009 |
| Type of local treatment | CRS/HIPEC only | (vs. CRLM only) | 0.84 | 0.69, 1.02 | 0.1 | 0.90 | 0.72, 1.12 | 0.3 |
| Perioperative chemotherapy | Received | (vs. not received) | 1.07 | 0.93, 1.24 | 0.3 | 0.91 | 0.77, 1.08 | 0.3 |
Bold-values are considered significant (p-value < 0.05)
CI confidence interval, CRLM colorectal liver metastasectomy, CRS/HIPEC cytoreductive surgery/hyperthermic intraperitoneal chemoperfusion, dMMR deficient mismatch repair, HR hazard ratio for RFS, mCRC metastatic colorectal cancer
Discussion
We present survival outcomes of the current largest real-world data cohort of patients with dMMR mCRC and pMMR mCRC treated with local therapy in the preimmunotherapy era. Consensus of curative-intent local therapy of metastases was mainly based on studies including patients with pMMR mCRC while its benefit is not known for the distinctive biological subgroup of patients with dMMR mCRC.3 Our results show that median RFS is longer for patients with dMMR mCRC compared with pMMR mCRC with a median RFS of 11.1 months compared with 8.9 months. The proportion of patients who remain free of recurrence is larger in patients with dMMR mCRC; 2-year RFS was 43% in dMMR mCRC and 21% in pMMR mCRC. When correcting for important differences in characteristics between groups, such as sidedness of primary tumor, type of local therapy, and perioperative chemotherapy, dMMR remains a strong independent predictor for improved RFS compared with pMMR with a HR of 0.57 (95% CI 0.38–0.87, p = 0.009).
The median OS of patients with dMMR mCRC is shorter than for patients with pMMR mCRC (33.3 months compared with 43.5 months). This is likely due to reduced survival after disease recurrence for patients with dMMR mCRC, in the era before standard immunotherapy, with a median OS of 10.2 months compared with 27.4 months for patients with pMMR mCRC. This is in line with the lower response rates to systemic chemotherapy in patients with dMMR compared with pMMR mCRC and the generally more favorable prognosis of patients with pMMR mCRC with systemic chemotherapy in later lines of treatment.6,18 Additionally, salvage local therapy occurred more often in patients with pMMR mCRC tumors, potentially due to more frequent liver-only recurrence and possibilities for repeated hepatectomy. This likely contributes to the improved OS after first recurrence for patients with pMMR tumors. Notably, in our cohort, immunotherapy could not have affected median OS, since reimbursement of immunotherapy in The Netherlands is arranged since February 2021 and OS data were retrieved through the Municipal Personal Records Database on 31 January 2021.
As expected, we found that patients with dMMR tumors were less often treated with local therapy (22% dMMR vs. 47% pMMR). There are various causes why patients with dMMR mCRC may receive treatment with local therapy less often. Patients with dMMR mCRC present more frequently with metastatic sites involving lymph nodes, which is less often amenable for curative-intent local treatment compared with liver-only disease, which is often seen with pMMR mCRC.7 Furthermore, despite the subgroup of young patients with dMMR due to Lynch syndrome, in general, the dMMR population is older than the pMMR mCRC population, which could influence decision making regarding local treatment, especially regarding more extensive local treatment such as CRS ± HIPEC.7 In our real-world cohort, patients with dMMR mCRC had more tumors harboring BRAFV600E mutations (50% vs. 4%), more right-sided tumors (78% vs. 27%), less liver-only disease (27% vs.70%), and more peritoneal involvement (44% vs. 18%), resulting in more CRS–HIPEC therapy (48% vs. 16%) and fewer CRLM resections (27% vs. 70%). The lower proportion of curative-intent local therapy of metastases in patients with dMMR mCRC is therefore probably caused by the distinct clinicopathological characteristics between the two groups. As immunotherapy was not the standard available in the inclusion period, this did not influence the decision making for patients with dMMR regarding local treatment and could not contribute to the lower proportion of patients receiving local therapy. Notably, nationwide, all patients receiving local treatment in this time period were included, limiting the risk of selection bias.
The absence of data concerning the tumor load of patients is a limitation of our study, e.g., peritoneal cancer index for CRS ± HIPEC or number of liver metastases for CRLM treatment, while this is recognized to be an important prognostic factor for recurrence.19 Lastly, to increase sample size, patients with dMMR mCRC were from the period 2015 to 2018 while patients with pMMR mCRC were from the period 2015 to 2016. However, we do not expect a better RFS or OS for patients with dMMR as a result of the more recent time period due to no availability of immunotherapy as standard of care at the time and no other important novel systemic treatments that became available in this period.
Scarce data exists for patients with dMMR mCRC treated with local therapy. Two exclusive CRS ± HIPEC studies included 15–44 patients with dMMR, both showing superior RFS and OS compared to pMMR.15,20,21 These results align with our study and show that local therapy of metastases in patients with dMMR mCRC that are not pretreated with immunotherapy can lead to durable RFS and reasonable OS. On the other hand, immunotherapy also shows durable responses and long-term survival benefit in patients with dMMR mCRC. This must be taken into account when determining the position of upfront local therapy in resectable patients with dMMR mCRC.8 Additionally, salvage local treatment following stable disease or partial response in patients treated with immunotherapy demonstrates a high 2-year RFS.9–11 A potential disadvantage of immunotherapy as first-line therapy in patients with dMMR mCRC with resectable disease is the chance of primary progression, which occurs in 30% of patients, potentially resulting in non-resectable disease afterwards.8 Additionally, 10% of patients treated with immunotherapy develop severe immune-related adverse events.22 A substantial subset of patients with dMMR in our study obtained a durable RFS and long OS, with a chance of cure by only local treatment, potentially sparing the treatment of immunotherapy. In clinical decision making, the (dis)advantages of both treatment options should be considered, including toxicity, risk of complications, and the possibilities regarding sequence of treatment.
Conclusions
Patients with dMMR mCRC in the real world benefit from curative-intent local therapy of metastases in the preimmunotherapy era, with a median RFS of 11.1 months compared with 8.9 months for patients with pMMR mCRC. After 2 years, 43% of all dMMR mCRC were free of recurrence after local treatment compared with 21% of the patients with pMMR mCRC. Differences in metastatic pattern led to more CRLM resections in patients with pMMR mCRC and more CRS/HIPEC in patients with dMMR mCRC; however, when stratified for type of local therapy, a comparable to better RFS for dMMR mCRC is still demonstrated per treatment type. Furthermore, when correcting for important differences in patient characteristics, dMMR remains a significant independent predictor for improved RFS. The relatively large proportion of patients with dMMR mCRC who remain free of recurrence after curative-intent local treatment of metastases could aid in the decision of upfront immunotherapy versus local therapy in case of resectable metastases for patients with dMMR mCRC. This should be decided by shared decision making, weighing the chance of potential cure against the chance of adverse events from immunotherapy, and risk of complications of local treatment for every individual patient.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgement
The authors thank the registration team of The Netherlands Comprehensive Cancer Organisation (IKNL) for the collection of data for The Netherlands Cancer Registry, as well as IKNL staff for scientific advice. The authors also thank the registration team of the nationwide network and registry of histo- and cytopathology in The Netherlands (PALGA) for collection of mutation status in the Dutch cohort.
Funding
No funder or sponsor was involved in this study.
Data Availability
J.R. and K.Z. had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Disclosures
C.J.P.: Nordic Pharma. E.W.: None. F.v.d.B.: None. G.V.: BMS, Merck, Servier, Personal Genome Diagnostics, Bayer, Sirtex, Pierre Fabre, Lilly. I.d.H.: ROCHE, RanD Biotech. J.H.: None. J.R.: Bayer, BMS, Merck-Serono, Pierre Fabre, Servier, HUB4organoids, Cleara Biotech. K.B.: None. K.Z.: None. M.K.: Nordic Farma, Merck-Serono, Pierre Fabre, Servier, Bayer, Bristol Myers Squibb, Merck, Roche and Servier. M.L.: None. W.v.G.: None.
Footnotes
Data were presented at ESMO 2022, Paris.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Punt CJA, Koopman M, Vermeulen L. From tumour heterogeneity to advances in precision treatment of colorectal cancer. Nat Rev Clin Oncol. 2017;14(4):235–246. doi: 10.1038/nrclinonc.2016.171. [DOI] [PubMed] [Google Scholar]
- 2.Koopman M, Kortman GAM, Mekenkamp L, et al. Deficient mismatch repair system in patients with sporadic advanced colorectal cancer. Br J Cancer. 2009;100(2):266–273. doi: 10.1038/sj.bjc.6604867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cutsem EV, Cervantes A, Adam R, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol. 2016;27(8):1386–1422. doi: 10.1093/annonc/mdw235. [DOI] [PubMed] [Google Scholar]
- 4.Aasebø KØ, Dragomir A, Sundström M, et al. Consequences of a high incidence of microsatellite instability and BRAF-mutated tumors: a population-based cohort of metastatic colorectal cancer patients. Cancer Med. 2019;8(7):3623–3635. doi: 10.1002/cam4.2205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wensink E, Bond M, Kucukkose E, et al. A review of the sensitivity of metastatic colorectal cancer patients with deficient mismatch repair to standard-of-care chemotherapy and monoclonal antibodies, with recommendations for future research. Cancer Treat Rev. 2021 doi: 10.1016/j.ctrv.2021.102174. [DOI] [PubMed] [Google Scholar]
- 6.Wensink GE, Elferink MAG, May AM, et al. Survival of patients with deficient mismatch repair metastatic colorectal cancer in the pre-immunotherapy era. Br J Cancer. 2021;124(2):399–406. doi: 10.1038/s41416-020-01076-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tran B, Kopetz S, Tie J, et al. Impact of BRAF mutation and microsatellite instability on the pattern of metastatic spread and prognosis in metastatic colorectal cancer. Cancer. 2011;117(20):4623–4632. doi: 10.1002/cncr.26086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.André T, Shiu KK, Kim TW, et al. Pembrolizumab in microsatellite-instability-high advanced colorectal cancer. N Engl J Med. 2020;383(23):2207–2218. doi: 10.1056/NEJMoa2017699. [DOI] [PubMed] [Google Scholar]
- 9.Alexandre C, Medhi K, Christelle DLF, et al. Outcomes of surgical resection in microsatellite instable colorectal cancer after immune checkpoint inhibitor treatment. Br J Surg. 2022 doi: 10.1093/bjs/znac425. [DOI] [PubMed] [Google Scholar]
- 10.Klemen ND, Court CM, Fernandes MC, et al. Local therapy for oligoprogression or consolidation in high mutational burden stage 4 colorectal cancer treated with PD-1 or PD-L1 blockade. Ann Surg Oncol. 2022;29(13):8373–8382. doi: 10.1245/s10434-022-12095-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ludford K, Cohen R, Svrcek M, et al. Pathological tumor response following immune checkpoint blockade for deficient mismatch repair advanced colorectal cancer. J Natl Cancer Inst. 2021;113(2):208–211. doi: 10.1093/jnci/djaa052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Parsons MT, Buchanan DD, Thompson B, Young JP, Spurdle AB. Correlation of tumour BRAF mutations and MLH1 methylation with germline mismatch repair (MMR) gene mutation status: a literature review assessing utility of tumour features for MMR variant classification. J Med Genet. 2012;49(3):151–157. doi: 10.1136/jmedgenet-2011-100714. [DOI] [PubMed] [Google Scholar]
- 13.Roock WD, Claes B, Bernasconi D, et al. Effects of KRAS, BRAF, NRAS, and PIK3CA mutations on the efficacy of cetuximab plus chemotherapy in chemotherapy-refractory metastatic colorectal cancer: a retrospective consortium analysis. Lancet Oncol. 2010;11(8):753–762. doi: 10.1016/S1470-2045(10)70130-3. [DOI] [PubMed] [Google Scholar]
- 14.Bartlett JW, Seaman SR, White IR, Carpenter JR. Multiple imputation of covariates by fully conditional specification: accommodating the substantive model. Stat Methods Med Res. 2015;24(4):462–487. doi: 10.1177/0962280214521348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tonello M, Baratti D, Sammartino P, et al. Microsatellite and RAS/RAF mutational status as prognostic factors in colorectal peritoneal metastases treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy (HIPEC) Ann Surg Oncol. 2022;29(6):3405–3417. doi: 10.1245/s10434-021-11045-3. [DOI] [PubMed] [Google Scholar]
- 16.Goey KKH, Sørbye H, Glimelius B, et al. Consensus statement on essential patient characteristics in systemic treatment trials for metastatic colorectal cancer: supported by the ARCAD Group. Eur J Cancer. 2018;100:35–45. doi: 10.1016/j.ejca.2018.05.010. [DOI] [PubMed] [Google Scholar]
- 17.R: a language and environment for statistical computing. https://www.gbif.org/tool/81287/r-a-language-and-environment-for-statistical-computing. Accessed 15 Feb 2022.
- 18.Tougeron D, Sueur B, Zaanan A, et al. Prognosis and chemosensitivity of deficient MMR phenotype in patients with metastatic colorectal cancer: an AGEO retrospective multicenter study. Int J Cancer. 2020;147(1):285–296. doi: 10.1002/ijc.32879. [DOI] [PubMed] [Google Scholar]
- 19.Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer. Ann Surg. 1999;230(3):309. doi: 10.1097/00000658-199909000-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hamed AB, Shuai Y, Derby J, et al. Impact of primary tumor location and genomic alterations on survival following cytoreductive surgery and hyperthermic intraperitoneal chemoperfusion for colorectal peritoneal metastases. Ann Surg Oncol. 2023;30(7):4459–4470. doi: 10.1245/s10434-023-13463-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Larsen SG, Goscinski MA, Dueland S, et al. Impact of KRAS, BRAF and microsatellite instability status after cytoreductive surgery and HIPEC in a national cohort of colorectal peritoneal metastasis patients. Br J Cancer. 2022;126(5):726–735. doi: 10.1038/s41416-021-01620-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Overman MJ, Ernstoff MS, Morse MA. Where we stand with immunotherapy in colorectal cancer: deficient mismatch repair, proficient mismatch repair, and toxicity management. Am Soc Clin Oncol Educ Book. 2018;38:239–247. doi: 10.1200/EDBK_200821. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
J.R. and K.Z. had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.