Abstract
Eosinophilic, noneosinophilic, or mixed granulocytic inflammations are the hallmarks of asthma heterogeneity. Depending on the priming of lung immune and structural cells, subjects with asthma might generate immune responses that are TH2-prone or TH17-prone immune response. Bacterial infections caused by Haemophilus, Moraxella, or Streptococcus spp. induce the secretion of IL-17, which in turn recruit neutrophils into the airways. Clinical studies and experimental models of asthma indicated that neutrophil infiltration induces a specific phenotype of asthma, characterized by an impaired response to corticosteroid treatment. The understanding of pathways that regulate the TH17-neutrophils axis is critical to delineate and develop host-directed therapies that might control asthma and its exacerbation episodes that course with infectious comorbidities. In this review, we outline clinical and experimental studies on the role of airway epithelial cells, S100A9, and high mobility group box 1, which act in concert with the IL-17-neutrophil axis activated by bacterial infections, and are related with asthma that is difficult to treat. Furthermore, we report critically our view in the light of these findings in an attempt to stimulate further investigations and development of immunotherapies for the control of severe asthma.
Key words: Severe asthma, bacterial infections, neutrophil, IL-17, lung inflammation
Asthma is a chronic disease characterized by airway inflammation, reversible or irreversible airway obstruction, airway hyperresponsiveness (AHR), and lung remodeling. Coughing, wheezing, shortness of breath, and chest tightness are the symptoms of the disease.1,2 Asthma is viewed as a complex and heterogeneous syndrome, which phenotypically may be classified as mild, moderate, and difficult to treat to severe, that is most likely caused by a combination of genetic predisposition, changes in lifestyle, and environmental factors. Despite this heterogeneity and complexity, 2 endotypes of asthma are defined: allergic (or atopic) and nonallergic asthma.3
Asthma affects children, teenagers, and adults. According to the Global Asthma Network, 339 million people are affected worldwide.4,5 The prevalence and mortality associated with asthma changes from one region to another. Indeed, although the prevalence of asthma is elevated in high-income countries, the highest number of deaths is recorded in low- and middle-income countries, which corresponds to more than 80% of asthma-related deaths.2, 6 The global prevalence of asthma also depends on sex and age. The incidence and prevalence of asthma are higher in children, but its morbidity and mortality are higher in adults. Sex prevalence varies along life, with higher prevalence among boys than among girls. However, for all adulthood, females are more affected than males.5
In this review, we will address current knowledge about the mechanisms that modulate neutrophilic inflammation in a specific asthma phenotype, which is difficult to treat, in the context of bacterial infectious comorbidities in an attempt to highlight the challenges and to overcome them for the development of host-directed therapies.
Pathogenesis of asthma
Allergic asthma is defined by eosinophilic infiltrate, mast cell activation, and IgE production.7 Usually, allergic asthma has an early onset and is associated with other allergic conditions such as allergic rhinitis and atopic dermatitis.3 Nonallergic asthma has a late onset and might be characterized by a TH2-high immune response with eosinophilic infiltrate, or TH2-low immune response. TH2-low endotype is characterized by neutrophilic inflammation that could be due to obesity, environmental exposure to pollutants such as diesel particles and cigarette smoke, or infections.3,8,9
In TH2-high asthma, eosinophilic inflammation is one of the main readouts of the disease. Allergens are recognized by PRRs, including Toll-like receptor (TLR)-4, on airway epithelial cells (AECs) and stimulate the secretion of IL-25, IL-33, thymic stromal lymphopoetin, GM-CSF, IL-1, chemokine (C-C motif) ligand (CCL)2, CCL20, and β-defensins. AECs also release ATP, lysophosphatidic acid, uric acid, and reactive oxygen species. Cytokines and chemokines recruit and activate dendritic cells (DCs) and type 2 innate lymphoid cells, which secrete IL-5, IL-9, and IL-13 and stimulate the differentiation of TH2 cells. TH2 cells secrete IL-4, IL-5, and IL-13 and induce allergen-specific IgE production.7,10 IgE binds to high-affinity IgE receptor on mast cells. Subsequent contacts with the allergen induce mast cell degranulation and immediate allergic reaction mediated by histamine, cysteinyl leukotrienes, and cytokines as tumor necrosis factor.11 TH2 cells, eosinophils, and basophils induce late allergic reaction. Although memory TH2 cells recognize antigens that usually are innocuous to the host and mediate eosinophil differentiation and recruitment, and collaborate for mucus production, eosinophils and basophils play a role in the induction of tissue damage mediated by major basic protein, eosinophil cationic protein, eosinophil peroxidase, and eosinophil-derived neurotoxin.12
The identification of additional TH-cell subsets brought new insight about the complexity of asthma pathogenesis. Higher number of circulating TH9 cells and increased levels of IL-9 were detected in allergic asthmatic patients compared with nonallergic subjects and in the asthma murine model.13, 14, 15
TH17 cells also participate in the immunopathology of asthma. Although IL-17 is associated with neutrophilic asthma, high levels of IL-17 in the airways and increased levels of IL-22 in the serum of patients with asthma compared with healthy individuals suggested that these cytokines also participate in the immune response of allergic asthma.16, 17, 18, 19 Fig 1 summarizes the description of immunopathology in the allergic asthma.
FIG 1.
Immunopathology of allergic asthma. Inhaled allergens activate AECs that recruit and activate DCs and ILC2s by production of IL-25, IL-33, TSLP, ATP, uric acid, LPA, and ROS. DCs and ILC2s induce TH2 differentiation and activation. TH9 and TH17 might also undergo differentiation. Eosinophil (EΦ) recruitment and activation followed by activation of TH2, TH9, and TH17 promote AHR and mucus production, AEC damage, and AHR. ILC2, Type 2 innate lymphoid cell; LPA, lysophosphatidic acid; ROS, reactive oxygen species; TSLP, thymic stromal lymphopoetin.
The heterogeneity of asthma endotypes includes eosinophilic (allergic and nonallergic), noneosinophilic (neutrophilic, TH1/TH17, and paucigranulocytic), and mixed granulocytic inflammation.20 Subjects with asthma might generate a TH2-prone and/or TH17-prone immune response depending on the inflammatory milieu. The persistent neutrophilic inflammation, which might be complete or partially mediated by TH17 cells, or mixed eosinophilic/neutrophilic inflammation has been reported as severe asthma.21,22 Subjects with neutrophilic airway inflammation were refractory to inhaled corticosteroid treatment.23
High levels of IL-17 in serum19 and in the lung16,24 were reported in allergic asthmatic patients, but few clinical studies involving TH17-related cytokines (IL-17, IL-23, and IL-22) were performed. In a randomized, double-blind, placebo-controlled study, the blocking of IL-17 signaling using brodalumab (anti–IL-17RA mAb) in subjects with inadequately controlled moderate to severe asthma had no evidence of an effect in the overall study population; however, an increase in symptom-free days was observed in the high-reversibility subgroup treated with brodalumab, encouraging further study of IL-17 blockade in this asthma subgroup.25 Concerning IL-23 signaling, a recent data of a randomized, phase 2a, multicenter, and double-blind clinical trial with risankizumab (anti–IL-23p19 mAb) showed that the treatment was not beneficial in severe asthma.26 Although anti–IL-22 therapies have not been evaluated in asthma yet,27 the use of fezakinumab (anti–IL-22 mAb) was efficient in severe atopic dermatitis, which has similar inflammatory features of asthma.28 A metadata study showed that a subset of subjects with asthma may respond to anti–IL-22 antibody therapy.29 Nevertheless, a deeper investigation is needed to ascertain the role of IL-17 and IL-22 in the modulation of TH2 immune responses. Considering that IL-22 activates DCs,30,31 it will be relevant to investigate whether IL-22 plays a direct or an indirect role in other innate cells, such as eosinophils. Interestingly, the presence of IL-22 was detrimental during the sensitization phase, but it was protective during the allergen challenge,18 suggesting a time-dependent effect of IL-22.
In moderate to severe asthma, AECs secrete neutrophil chemoattractant CXCL8 (IL-8), CCL2, and CCL20, which recruit DCs.32 Activated DCs migrate to the mediastinal lymph node where they prime naive T cells to differentiate into TH17 cells through the expression of IL-1β, TGF-β, IL-6, and IL-23 by DCs. Primed TH17 cells migrate into the lungs and produce IL-17 and IL-22.22 TH17 cells increase mucus production (IL-17) and exacerbate AR (IL-17/IL-22) in severe asthma.33 In addition, IL-17 directly acts on bronchial smooth muscle contraction through the upregulation of Ras homolog family member A protein, associated with actomyosin contractility.34 During severe asthma, type 3 innate lymphoid cells are early providers of IL-17 and IL-22. Type 3 innate lymphoid cells may play a critical role in severe asthma phenotypes by producing IL-17 and IFN-γ, which stimulate neutrophil recruitment,10 besides IL-22. IL-17 sustains neutrophilic inflammation by stimulating CXCL1 and CXCL8 production by AECs. CXCL1 and CXCL8 enhance airway smooth muscle contraction and stimulate collagen synthesis.21,35 Furthermore, TH17 cells and IL-17 induce the expression of polymeric immunoglobulin receptor in the airway epithelium and a subsequent increase in airway IgM and IgA levels in mice.36 Therefore, IL-17 plays a key role in the mucosal immune response by regulating IgA production and transport to the lungs and gut.36, 37, 38 This pathway if unbalanced might be deleterious to the host and cause pathology.
IL-22 enhances airway smooth muscle contraction, promotes collagen deposition and production of S100A9, an alarmin that induces neutrophil influx, by AECs, and is associated with neutrophil survival by inhibiting apoptosis pathway. S100A9 may stimulate AECs to produce neutrophil survival cytokines such as MCP-1, IL-6, and CXCL8.39, 40, 41, 42 Increased levels of S100A9 were reported in the sputum of subjects with severe uncontrolled asthma compared with subjects with controlled asthma,43 suggesting that S100A9 could be a biomarker of neutrophilic inflammation in severe asthma.43,44 In an animal model of neutrophil-dominant asthma generated using ovalbumin (OVA) and complete Freund’s adjuvant, S100A9 was reported to generate and amplify neutrophilic inflammation followed by a high production of IL-1β, IL-17, and IFN-γ in the lungs lysates.44 S100A9 interacts with TLR4 or receptor for advanced glycation end products in granulocytes and induces its own production in these cells. S100A9 may also interact with TLR4 or receptor for advanced glycation end products in monocytes. In both cells, S100A9 induces the production of IL-1β, IL-6, IL-18, and tumor necrosis factor. IL-1β has an autocrine and paracrine effect on both granulocytes and monocytes.45
In severe asthma, S100A8 and S100A9 induce also the production of high mobility group box 1 (HMGB1), a proinflammatory alarmin produced by inflammatory and airway cells.46 Sputum levels of HMGB1 were significantly higher in children with severe asthma compared with children with mild and moderate asthma.47 In addition, in a model of neutrophilic asthma induced by OVA combined with LPS, anti–HMGB1-neutralizing antibody administered intranasally before OVA sensitization reduced neutrophilic inflammation, IL-17 production, and AHR.48 As S100A9, HMGB1 also interacts with receptor for advanced glycation end products and TLR4 and induces the release of neutrophil extracellular traps (NETs).49,50 NETs play a detrimental effect because their compounds histones, neutrophil elastase (NE), myeloperoxidase, cathepsin G, and DNA cause tissue damage.51
The role of NETs in severe asthma is not limited to tissue damage in AECs. NETs might indirectly stimulate the recruitment of neutrophils into the airways by a mechanism dependent on NET-activated AECs, which secrete CXCL1, CXCL2, and CXCL8 and drive neutrophilic inflammation through the TLR4/nuclear factor kappa B (NF-kB) pathway.52 Likewise, another study showed that during severe asthma, NETs induce a second wave of neutrophil infiltration in the airway by stimulating macrophages to produce IL-1β, which intensifies the recruitment of neutrophils and the production of NETs. The accumulation of NETs may amplify tissue damage and aggravate asthma pathology.53 Activated neutrophils produce reactive oxygen species, antimicrobial peptides, elastase, and matrix metalloproteinase-9.54,55 Reactive oxygen species, NE, and matrix metalloproteinase-9 may be involved in tissue damage during severe asthma.56 NE augments CXCL8 production by AECs, promoting a loop in neutrophil recruitment as well as the inactivation of tissue inhibitor of metalloproteinase-1. Increased levels of matrix metalloproteinase-9 and NE and decreased levels of tissue inhibitor of metalloproteinase-1 contribute to bronchoconstriction in asthma.57 Fig 2 summarizes the description of immunopathology in severe asthma.
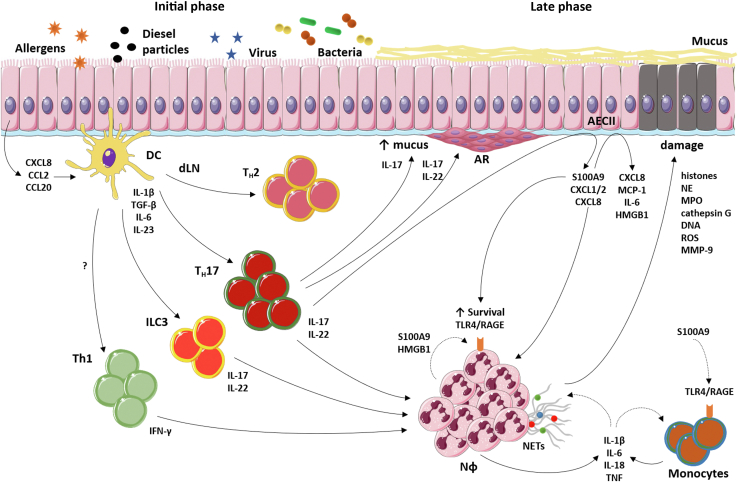
FIG 2.
Immunopathology of severe asthma. Allergens, pollutants, and viral/bacterial infections promote the activation of AECs that recruit and activate DCs by production of IL-1β, IL-6, CXCL8, CCL2, and CCL20. DCs migrate to draining lymph node (dLN) and induce TH1, TH2, and TH17 cell differentiation. ILC3s provide the early sources of IL-17 and IL-22. ILC3, TH17, and TH1 contribute to recruit neutrophils (NΦ). NΦ cause epithelial damage by release of NETs. AEC-derived signals (S100A9, CXCL1, CXCL2, and CXCL8) stimulate the NΦ survival and recruitment, respectively. The interface of neutrophils and AECs amplifies the local inflammation and aggravates asthma immunopathology. ILC3, Type 3 innate lymphoid cell.
Asthma and infectious comorbidities
Viral, bacterial, and fungal infections might accelerate the progression of asthma and induce severe disease.57 Therefore, the therapeutic management of asthma is more difficult during infectious comorbidities. The relationship between asthma and infections is complex and involves the pathogen and factors associated with the host genetic background. Overall, the failure in mechanisms of pathogen tolerance induces host cell death and might cause tissue injury. The recognition of microbial-associated molecular patterns by pattern recognition receptors on host cells activates innate leukocytes that secrete proinflammatory cytokines and regulate positively the expression of MHC and costimulatory and adhesion molecules. Signaling through cytokines and PRRs drives the differentiation of CD4+ T-cell subsets. This is the critical outcome in the landscape of comorbidities that might affect the type and magnitude of inflammation in subjects with asthma. Although the immune response and inflammatory response against the infection is important for pathogen control, they may worsen the episodes of asthma and cause severe disease. The comprehension of the mechanisms by which microbial components are detrimental or protective in asthma might provide the basis for host-directed therapies for severe asthma. In this sense, 2 detrimental possibilities may be considered in asthma and infection comorbidities: (1) asthma impairs and weakens the immune response against pathogens and (2) infections change asthma phenotype and induce a difficult-to-treat or severe asthma endotype.
It should be noticed that in opposition to this notion, the hygiene hypothesis elegantly explored and supported the idea that infections might negatively regulate type 2 inflammation, the hallmark of allergic asthma. The hygiene hypothesis might be briefly summarized by stating that the exposure to pathogens, or infectious agents or their molecules, drives the immune response to the TH1 profile or toward regulatory T (Treg) cells, and counteracts or reduces the TH2 profile.58 In the former decade, we evaluated the therapeutic potential of experimental vaccines against tuberculosis in an asthma model. Subunit vaccines and a DNA vaccine with mycobacterial antigens, used as allergen-free immunotherapies in murine asthma model with OVA or Dermatophagoides pteronyssinus protein (Der-p1), reduced airway inflammation and improved pulmonary function.59, 60, 61, 62
Although infections might regulate diversely the immunopathology of asthma, in the following section, we shall discuss how the infections drive the severity of asthma and we will focus on how airway and pulmonary bacterial infections might be detrimental in asthma. This field deserves investigations to advance in the search of targets for immunotherapies. Furthermore, in our critical view, the order of stimuli (allergen-infection or infection-allergen) and the time-dependency of infection are critical factors that drive the exacerbation or reduction of asthma immunopathology.
Bacterial infections and asthma exacerbations
Pathogenic bacteria, atypical bacteria, and lung microbiota might affect the immunopathology of asthma.20,22,63, 64, 65, 66 The association between atypical bacteria, as Mycoplasma pneumoniae and Chlamydia pneumoniae, and asthma exacerbation is strongly associated with the enhancement of allergic asthma phenotype and type 2 inflammation induced by the infection.64
Clinical evidence and experimental studies support a significant association between asthma severity and changes in bacterial community profiles. A recent clinical trial with 214 children showed alterations in the airway microbiota at time of early loss of asthma control. The airway microbiota dominated by Corynebacterium and Dolosigranulum was associated with favorable clinical outcomes compared with microbiota dominated by pathogenic bacteria, including Staphylococcus, Streptococcus, and Moraxella.67 These data reinforce the results obtained in a study with adults classified as adults with mild asthma, which revealed a negative correlation between the relative abundance of Corynebacterium and Moraxella in nasal brush. In addition, this study showed that the relative abundance of Moraxella and Streptococcus correlated positively with systemic eosinophilia and lower airway eosinophils and bronchial levels of tumor necrosis factor and IL-7, respectively.68 In 17 of 28 treatment-resistant patients with severe asthma, the dominant species within the airway bacterial community were Moraxella catarrhalis and members of Haemophilus and Streptococcus genera. Chronic infections with these species were associated with longer asthma duration and positively correlated with higher IL-8 concentrations and neutrophil counts in the sputum.69 If there is such relationship, further studies are necessary to indicate which one comes first, asthma severity or bacterial chronic infections.
Chronic bacterial infections are possible factors that contribute to the development of neutrophilic asthma20,22 and the neutrophil-mediated inflammation could explain the inefficacy of corticosteroids in a TH2-low asthma phenotype.20 Considering this, antibiotic therapy has been suggested. Treatment with macrolide (azithromycin) as an immunomodulator and anti-inflammatory agent for 6 months reduced the exacerbation in those with noneosinophilic asthma, suggesting that chronic bacterial infections in TH2-low patients is a significant contributing factor to exacerbation risk.70
Airway neutrophils from subjects with asthma spontaneously release lower levels of IL-8, IL-1β, and tumor necrosis factor compared with healthy controls, suggesting an impaired role of these cells, which may impact the susceptibility to airway infections.71 Both bacteria from Haemophilus and Streptococcus genera require neutrophils to control chronic infections,72,73 and the function of neutrophils is compromised in asthma milieu.
Haemophilus influenzae
Hinfluenzae is a Gram-negative, nonencapsulated bacterium that chronically infects the airways and is the most common isolated bacteria from patients with asthma, associated with neutrophilic asthma and corticosteroid-reduced response in asthma.74 Accordingly, abundance of H influenzae in the airways is associated with predominance of neutrophils in the sputum of patients with asthma.75
The cell type mostly infected by H influenzae is AECs. Nontypeable H influenzae is recognized in the airways by TLR2, resulting in Myd88 recruitment with NF-κB activation. However, the subsequent effector mechanisms responsible for the clearing of the infection and the control of inflammatory response remain elusive.76 Mice previously infected with H influenzae and submitted to OVA-induced asthma exhibited a higher bacterial load compared with single infected mice. Furthermore, chronic H influenzae infection and OVA exposure induced neutrophilic inflammation and TH17 responses that promoted bacterial persistence, leading to the development of a phenotype similar to steroid-resistant neutrophilic asthma.77 Anti–IL-17 treatment of OVA-sensitized and H influenza–infected mice completely inhibited airway neutrophilic inflammation induced by the infection, suggesting that neutrophilic inflammation is dependent on IL-17.73 In addition, infection by nontypeable H influenzae in mice exposed to allergen promotes increase of AHR, MUC5A, and MUC5B expression and is associated with hyperphosphorylation of p38 mitogen-activated protein kinase. The combined treatment with dexamethasone and SB203580, a specific inhibitor of p38, substantially suppressed the H influenzae–induced asthma exacerbation.78 On the basis of these findings, the authors suggested that cotreatment with dexamethasone and SB203580 could be a novel strategy against steroid-resistant asthma.
Acute H influenzae infection induces TH17 cells, neutrophil influx, increased mucus production, and attenuation of the lung function in mice.79 Long-term exposure to low dose of H influenzae after OVA challenge resulted in increased tissue remodeling and reduced number of Treg cells in the lungs and IL-10 levels in the bronchoalveolar lavage fluid, suggesting an impaired anti-inflammatory response during the comorbidity.79
In a recent randomized clinical trial, azithromycin treatment for 48 weeks of adults with uncontrolled asthma reduced H influenzae load in sputum and efficiently reduced the asthma exacerbations rate.80 However, this long-term treatment favored the antibiotic resistance to Staphylococcus aureus, but not to H influenza and S pneumoniae. These findings show that the comorbidity asthma and bacterial infections must be treated in 2 ways: short-term antibiotic and regulation of inflammatory response. Therefore, the search for new anti-inflammatory targets to control bacterial infection–associated asthma exacerbations is critical.
Moraxella catarrhalis
Mcatarrhalis is a Gram-negative diplococcus, nonencapsulated bacterium that colonizes the mucosal membranes of nasopharynx. M catarrhalis has emerged as a transmittable pathogen, causing infections such as pneumonia, laryngitis, endocarditis, meningitis, and otitis media.81 Particularity, M catarrhalis is associated with chronic obstructive pulmonary disease and asthma exacerbations. Bacteriome characterization of hospitalized children with asthma in consequence of an episode of asthma exacerbation showed the presence of M catarrhalis in the nasopharyngeal swabs.82 Moreover, children with asthma with nasal airway microbiota colonized predominantly by Moraxella genera have an increased risk of asthma exacerbations.83
Epithelial cells from mucosal surface are the main targets of M catarrhalis. In the alveoli, M catarrhalis interacts with TLR2, TLR4, and TLR9 from AECs, resulting in NF-κB activation. Consequently, AECs increase the expression of adhesion molecules (intercellular adhesion molecule 1 and vascular cell adhesion molecule 1), and produce CCL2 and MCP-1, which recruit monocytes and DCs.81 Lipooligosaccharidae from the bacterial membrane activates TLR4 in macrophages by a mechanism dependent on CD14, leading to NF-κB activation and production of IL-6, IL-8, and tumor necrosis factor.84 IL-6, tumor necrosis factor, and IL-8 produced by macrophages result in recruitment and activation of monocytes, and exacerbation of inflammatory response.81,85 The recognition of pathogens by DCs results in an increase in costimulatory molecules (CD80 and CD86), CD44, an adhesion molecule, and MHC-II expression. In addition, activated DC promotes both CD8+ and CD4+ T-lymphocyte activation.81 CD4+ lymphocytes secrete cytokines that induce recruitment and activation of granulocytes. As a consequence, activated neutrophils release elastase, which induces tissue damage.
Asthma exacerbations caused by M catarrhalis infection are also associated with recruitment and activation of TH17 cells. In a mouse model of HDM-induced asthma, M catarrhalis infection in different time points of allergic airway inflammation induced severe airway inflammation and mucus production and added to increased concentrations of IL-17 and neutrophils in the bronchoalveolar lavage fluid.86 IL-17 knockout mice exposed to allergen and infected displayed a significant reduction in neutrophils in the bronchoalveolar lavage fluid, in the lung inflammation, and in mucus production, indicating that IL-17–producing CD4+ cells were a key mediator of M catarrhalis–induced asthma exacerbations.86
Streptococcus pneumoniae
Spneumoniae is a Gram-positive diplococcus bacterium that frequently colonizes the human nasopharynx. Pneumococcal infections lead to serious invasive diseases such as meningitis, septicemia, and pneumonia, and are an important cause of morbidity and mortality among children and in older adults.87,88
The susceptibility to Streptococcus infection was already described in patients with asthma. A study with 224 patients with asthma and 668 males without asthma showed that asthma was the only significant risk factor for S pneumoniae carriage.89 A nested case-control study conducted with 635 subjects with invasive pneumococcal disease and 6350 controls showed that subjects with asthma had an increased risk of invasive pneumococcal disease.90
In the alveoli, S pneumoniae interacts with type II AECs, alveolar macrophages, and DCs through the recognition of pathogen-associated molecular patterns (PAMPs) by PRRs; pneumococcal lipoteichoic acid and cell wall peptidoglycanas are recognized by TLR2, pneumolysin is recognized by TLR4, and CpG motifs are recognized by TLR9.91 An important synergy among TLRs has been described in response to S pneumoniae, especially TLR2/4/9 for induction of cytokines and chemokines92 and TLR7/9/13 associated with susceptibility to infection.93 TLR activation induces the production of tumor necrosis factor, CXCL1, and CXCL2, which recruit neutrophils. AEC also release alarmins, as S100A8 and S100A9, in the early phase of S pneumoniae infection that precedes neutrophil recruitment.94 Although the activation of individual TLRs has a limited role in pneumococcal infection, MyD88, the adaptor protein of the signaling pathway of TLR, is crucial for initiating proinflammatory cytokine release and for the enhanced production of antimicrobial peptide to restrict S pneumoniae outgrowth.95 DC recognize and phagocyte pneumococcal mainly by pneumococcal adherence and virulence factor A and release IL-1β, IL-6, IL-8, IL-12, and tumor necrosis factor.96 Airway DC, specially CD103+ subset, might induce the release of IFN-γ and IL-17 by invariant natural killer cells,97 whereas it has been shown that bone marrow–derived DC are important to induce TH17-adaptive immune response,98,99 both important to pneumococcal clearance. During pneumococcal infection, TH17, TH1, and TH2 cells are critical to clear S pneumoniae infection. TH1 and TH17 cells recruit and activate macrophages and neutrophils, and TH2 cells release IL-4 and induce the production of antibodies.72 A potential synergistic effect of TH17 cells and antibodies induces, respectively, increased recruitment of neutrophils and opsonization, which improve the control of chronic bacterial infection.100 Besides antimicrobial defense mechanisms, neutrophils use NETs to restrict bacterial spreading.101 Furthermore, IL-17 amplifies type II AEC activation by increasing the production of antimicrobial peptides and chemokines.102 IL-17 and IL-22 are also produced by type 3 innate lymphoid cells by a mechanism dependent on DC, IL-1β, and IL-23.41,103,104
Although the susceptibility to S pneumoniae infection is increased in patients with asthma, the effect of infection during asthma episodes is still controversial and needs to be clarified. The induction of TH1, TH2, and TH17 cells and the recruitment of neutrophils are potential risks for the development of severe asthma, and as we discussed previously, the exacerbation of pulmonary inflammation generates tissue damage.
Epidemiological and experimental findings showed a protective role for S pneumoniae infection in asthma. Children with asthma treated with sulfisoxazole and pneumococcal vaccine showed 56% reduction in the frequency of acute asthmatic attacks and 90% decrease in hospitalizations associated with otitis media.105 Similarly, a retrospective cohort study, which evaluated the effectiveness of a 23-valent pneumococcal polysaccharide vaccine, showed that the vaccination decreased the risk of hospital admission for asthma compared with nonvaccinated subjects.106 Experimentally, it was demonstrated that killed or live S pneumoniae administered before, during, or after OVA sensitization attenuated allergic inflammation.107,108
The protective role of infection after OVA sensitization increased TH1 immune response in BALB/c mice, whereas S pneumoniae infection during OVA sensitization induced Treg cells and increased IL-10 production.108 In addition, intranasal administration of pneumococcal conjugate vaccine, but not the polysaccharide vaccine, suppressed allergic immune response and increased Treg cells in the draining lymph nodes, lungs, and spleen.109 Later, these authors identified pneumococcal components, 3 polysaccharides and pneumolysoid, as key immunoregulators of S pneumoniae–induced Treg cells. These components of S pneumoniae drove the differentiation of highly suppressive Treg cells, which inhibited TH2 immune response, prevented the induction of TH17 immune response, and disabled DC response, resulting in the effective suppression of OVA-induced inflammation,110 supporting the hygiene hypothesis.58 Experimental S pneumoniae infection in neonatal mice 21 days before OVA sensitization enhanced AHR and increased neutrophil recruitment and TH17 cells into the airways, whereas IL-17 depletion alleviated airway inflammation mediated mostly by neutrophils, and decreased AHR.111
Conclusions
The interplay between asthma and bacterial infections is complex and involves both asthma and pathogen-specific immune responses. The comorbidities might affect the lung milieu and increase a preexisting TH2 profile in subjects with allergic asthma (eosinophil inflammation) or drive the immune response to TH17 profile and generate severe asthma (neutrophil inflammation or granulocytic inflammation). Specific pathogens might activate TH17 cells that mediate neutrophil influx to the airways, which in turn exacerbates asthma (Fig 3).
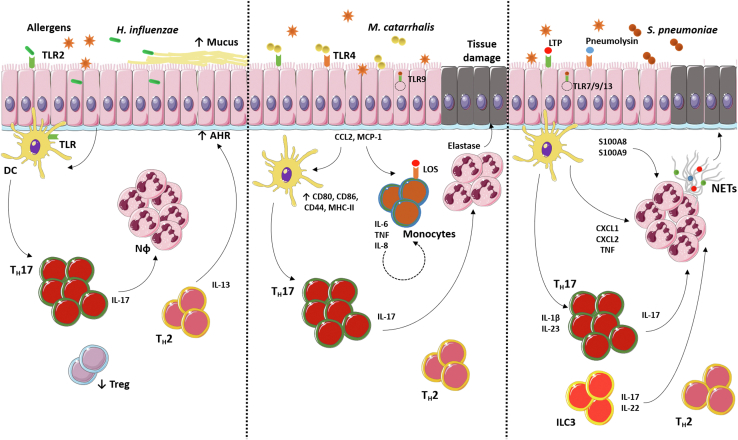
FIG 3.
Bacterial infections and asthma exacerbation. Bacterial infections activate TLRs on DCs and might drive the allergic immune response from TH2 to TH2/TH17 cells. Activated eosinophils and neutrophils produce inflammatory mediators that activate smooth muscle cells and damage epithelial cells, which release S100A8 and S100A9 alarmins. TH17 cells might indirectly aggravate the immunopathology of asthma, inducing the recruitment of neutrophils that produce NETs.
It is likely that exacerbation of airway inflammation might require host-directed therapy, depending on the phenotype of asthma and on the comorbidity. The investigation of mediators and receptors using experimental models might provide new molecular targets. The confirmation of these targets is important to delineate immunotherapies or adjuvant therapies based on the concept of host-directed therapies for neutrophilic asthma endotype.
Footnotes
The study was funded by São Paulo Research Foundation (FAPESP, Brazil) (grant nos. 2017/21629-5, 2019/09881-6, and 2019/18793-3) and the National Council for Scientific and Technological Development (CNPq, Brazil) (grant no. 142139/2019-0).
Disclosure of potential conflict of interest: The authors declare that they have no relevant conflicts of interest.
References
- 1.Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention, 2020. Available from: www.ginasthma.org. GINA; 2020. https://ginasthma.org/wp-content/uploads/2020/06/GINA-2020-report_20_06_04-1-wms.pdf. Accessed August 12, 2020.
- 2.World Health Organization Asthma 2022. https://www.who.int/news-room/fact-sheets/detail/asthma
- 3.Hammad H., Lambrecht B.N. The basic immunology of asthma. Cell. 2021;184:1469–1485. doi: 10.1016/j.cell.2021.02.016. [DOI] [PubMed] [Google Scholar]
- 4.Global Asthma Network The Global Asthma Report 2018. http://globalasthmareport.org/resources/Global_Asthma_Report_2018.pdf
- 5.Dharmage S.C., Perret J.L., Custovic A. Epidemiology of asthma in children and adults. Front Pediatr. 2019;7:1–15. doi: 10.3389/fped.2019.00246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.To T., Stanojevic S., Moores G., Gershon A.S., Bateman E.D., Cruz A.A., et al. Global asthma prevalence in adults: findings from the cross-sectional world health survey. BMC Public Health. 2012;12:204. doi: 10.1186/1471-2458-12-204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lambrecht B.N., Hammad H. The immunology of asthma. Nat Immunol. 2015;16:45–56. doi: 10.1038/ni.3049. [DOI] [PubMed] [Google Scholar]
- 8.Boonpiyathad T., Sözener Z.C., Satitsuksanoa P., Akdis C.A. Immunologic mechanisms in asthma. Semin Immunol. 2019;46 doi: 10.1016/j.smim.2019.101333. [DOI] [PubMed] [Google Scholar]
- 9.Manni M.L., Trudeau J.B., Scheller E.V., Mandalapu S., Elloso M.M., Kolls J.K., et al. The complex relationship between inflammation and lung function in severe asthma. Mucosal Immunol. 2014;7:1186–1198. doi: 10.1038/mi.2014.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Harper R.W., Zeki A.A. Immunobiology of the critical asthma syndrome. Clin Rev Allergy Immunol. 2014;48:54–65. doi: 10.1007/s12016-013-8407-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hall S., Agrawal D.K. Key mediators in the immunopathogenesis of allergic asthma. Int Immunopharmacol. 2014;23:316–329. doi: 10.1016/j.intimp.2014.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Acharya K.R., Ackerman S.J. Eosinophil granule proteins: form and function. J Biol Chem. 2014;289:17406–17415. doi: 10.1074/jbc.R113.546218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jones C.P., Gregory L.G., Causton B., Campbell G.A., Lloyd C.M. Activin A and TGF-β promote TH9 cell-mediated pulmonary allergic pathology. J Allergy Clin Immunol. 2012;129 doi: 10.1016/j.jaci.2011.12.965. 1000-10.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jia L., Wang Y., Li J., Li S., Zhang Y., Shen J., et al. Detection of IL-9 producing T cells in the PBMCs of allergic asthmatic patients. BMC Immunol. 2017;18:1–9. doi: 10.1186/s12865-017-0220-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tong R., Xu L., Liang L., Huang H., Wang R., Zhang Y. Analysis of the levels of Th9 cells and cytokines in the peripheral blood of mice with bronchial asthma. Exp Ther Med. 2018;15:2480–2484. doi: 10.3892/etm.2018.5700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Barczyk A., Pierzcha W., Sozañska E. Interleukin-17 in sputum correlates with airway hyperresponsiveness to methacholine. Respir Med. 2003;97:726–733. doi: 10.1053/rmed.2003.1507. [DOI] [PubMed] [Google Scholar]
- 17.Hashimoto T., Akiyama K., Kobayashi N., Mori A. Comparison of IL-17 production by helper T cells among atopic and nonatopic asthmatics and control subjects. Int Arch Allergy Immunol. 2005;137:51–54. doi: 10.1159/000085432. [DOI] [PubMed] [Google Scholar]
- 18.Besnard A.G., Sabat R., Dumoutier L., Renauld J.C., Willart M., Lambrecht B., et al. Dual role of IL-22 in allergic airway inflammation and its cross-talk with IL-17A. Am J Respir Crit Care Med. 2011;183:1153–1163. doi: 10.1164/rccm.201008-1383OC. [DOI] [PubMed] [Google Scholar]
- 19.Zhao Y., Yang J., Gao Y.D., Guo W. Th17 immunity in patients with allergic asthma. Int Arch Allergy Immunol. 2010;151:297–307. doi: 10.1159/000250438. [DOI] [PubMed] [Google Scholar]
- 20.Russell R.J., Brightling C.E. Pathogenesis of asthma: implications for precision medicine. Clin Sci. 2017;131:1723–1735. doi: 10.1042/CS20160253. [DOI] [PubMed] [Google Scholar]
- 21.Newcomb D.C., Peebles R.S. Th17-mediated inflammation in asthma. Curr Opin Immunol. 2013;25:755–760. doi: 10.1016/j.coi.2013.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vroman H., van den Blink B., Kool M. Mode of dendritic cell activation: the decisive hand in Th2/Th17 cell differentiation. Implications in asthma severity? Immunobiology. 2015;220:254–261. doi: 10.1016/j.imbio.2014.09.016. [DOI] [PubMed] [Google Scholar]
- 23.Green R.H., Brightling C.E., Woltmann G., Parker D., Wardlaw A.J., Pavord I.D. Analysis of induced sputum in adults with asthma: identification of subgroup with isolated sputum neutrophilia and poor response to inhaled corticosteroids. Thorax. 2002;57:875–879. doi: 10.1136/thorax.57.10.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chesné J., Braza F., Mahay G., Brouard S., Aronica M., Magnan A. IL-17 in severe asthma. Where do we stand? Am J Respir Crit Care Med. 2014;190:1094–1101. doi: 10.1164/rccm.201405-0859PP. [DOI] [PubMed] [Google Scholar]
- 25.Busse W.W., Holgate S., Kerwin E., Chon Y., Feng J., Lin J., et al. Randomized, double-blind, placebo-controlled study of brodalumab, a human anti–IL-17 receptor monoclonal antibody, in moderate to severe asthma. Am J Respir Crit Care Med. 2013;188:1294–1302. doi: 10.1164/rccm.201212-2318OC. [DOI] [PubMed] [Google Scholar]
- 26.Brightling C.E., Nair P., Cousins D.J., Louis R., Singh D. Risankizumab in severe asthma—a phase 2a, placebo-controlled trial. N Engl J Med. 2021;385:1669–1679. doi: 10.1056/NEJMoa2030880. [DOI] [PubMed] [Google Scholar]
- 27.Margelidon-Cozzolino V., Tsicopoulos A., Chenivesse C., de Nadai P. Role of Th17 cytokines in airway remodeling in asthma and therapy perspectives. Front Allergy. 2022;3 doi: 10.3389/falgy.2022.806391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Guttman-Yassky E., Brunner P.M., Neumann A.U., Khattri S., Pavel A.B., Malik K., et al. Efficacy and safety of fezakinumab (an IL-22 monoclonal antibody) in adults with moderate-to-severe atopic dermatitis inadequately controlled by conventional treatments: a randomized, double-blind, phase 2a trial. J Am Acad Dermatol. 2018;78 doi: 10.1016/j.jaad.2018.01.016. 872-81.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Badi Y., Pavel A.B., Riley J.H., Chung K.F., Guttman-Yassky E., Adcock I.M. Is fezakinumab, an anti-IL22 antibody, a putative novel therapy for a subset of severe asthma? Eur Respir J. 2019;54:OA1606. [Google Scholar]
- 30.Schnyder-Candrian S., Togbe D., Couillin I., Mercier I., Brombacher F., Quesniaux V., et al. Interleukin-17 is a negative regulator of established allergic asthma. J Exp Med. 2006;203:2715–2725. doi: 10.1084/jem.20061401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schnyder B., Lima C., Schnyder-Candrian S. Interleukin-22 is a negative regulator of the allergic response. Cytokine. 2010;50:220–227. doi: 10.1016/j.cyto.2010.02.003. [DOI] [PubMed] [Google Scholar]
- 32.Sozzani S., Luini W., Borsatti A., Polentarutti N., Zhou D., Piemonti L., et al. Receptor expression and responsiveness of human dendritic cells to a defined set of CC and CXC chemokines. J Immunol. 1997;159:1993–2000. [PubMed] [Google Scholar]
- 33.Aujla S.J., Alcorn J.F. TH17 cells in asthma and inflammation. Biochim Biophys Acta Gen Subj. 2011;1810:1066–1079. doi: 10.1016/j.bbagen.2011.02.002. [DOI] [PubMed] [Google Scholar]
- 34.Chiba Y., Tanoue G., Suto R., Suto W., Hanazaki M., Katayama H., et al. Interleukin-17A directly acts on bronchial smooth muscle cells and augments the contractility. Pharmacol Rep. 2017;69:377–385. doi: 10.1016/j.pharep.2016.12.007. [DOI] [PubMed] [Google Scholar]
- 35.Korn T., Bettelli E., Oukka M., Kuchroo V.K. IL-17 and Th17 cells. Annu Rev Immunol. 2009;27:485–517. doi: 10.1146/annurev.immunol.021908.132710. [DOI] [PubMed] [Google Scholar]
- 36.Jaffar Z., Ferrini M.E., Herritt L.A., Roberts K. Cutting edge: lung mucosal Th17-mediated responses induce polymeric Ig receptor expression by the airway epithelium and elevate secretory IgA levels. J Immunol. 2009;182:4507–4511. doi: 10.4049/jimmunol.0900237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dann S.M., Manthey C.F., Le C., Miyamoto Y., Gima L., Abrahim A., et al. IL-17A promotes protective IgA responses and expression of other potential effectors against the lumen-dwelling enteric parasite Giardia. Exp Parasitol. 2015;156:68–78. doi: 10.1016/j.exppara.2015.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Christensen D., Mortensen R., Rosenkrands I., Dietrich J., Andersen P. Vaccine-induced Th17 cells are established as resident memory cells in the lung and promote local IgA responses. Mucosal Immunol. 2017;10:260–270. doi: 10.1038/mi.2016.28. [DOI] [PubMed] [Google Scholar]
- 39.Kim D.H., Gu A., Lee J.S., Yang E.J., Kashif A., Hong M.H., et al. Suppressive effects of s100a8 and s100a9 on neutrophil apoptosis by cytokine release of human bronchial epithelial cells in asthma. Int J Med Sci. 2020;17:498–509. doi: 10.7150/ijms.37833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ryckman C., Vandal K., Rouleau P., Talbot M., Tessier P.A. Proinflammatory activities of S100: proteins S100A8, S100A9, and S100A8/A9 induce neutrophil chemotaxis and adhesion. J Immunol. 2003;170:3233–3242. doi: 10.4049/jimmunol.170.6.3233. [DOI] [PubMed] [Google Scholar]
- 41.Sonnenberg G.F., Fouser L.A., Artis D. Border patrol: regulation of immunity, inflammation and tissue homeostasis at barrier surfaces by IL-22. Nat Immunol. 2011;12:383–390. doi: 10.1038/ni.2025. [DOI] [PubMed] [Google Scholar]
- 42.Witte K., Koch E., Volk H.D., Wolk K., Sabat R. The herbal extract EPsÆ 7630 increases the antimicrobial airway defense through monocyte-dependent induction of IL-22 in T cells. J Mol Med. 2020;98:1493–1503. doi: 10.1007/s00109-020-01970-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lee T.H., Jang A.S., Park J.S., Kim T.H., Choi Y.S., Shin H.R., et al. Elevation of S100 calcium binding protein A9 in sputum of neutrophilic inflammation in severe uncontrolled asthma. Ann Allergy Asthma Immunol. 2013;111 doi: 10.1016/j.anai.2013.06.028. 268-75.e1. [DOI] [PubMed] [Google Scholar]
- 44.Lee T.H., Chang H.S., Bae D.J., Song H.J., Kim M.S., Park J.S., et al. Role of S100A9 in the development of neutrophilic inflammation in asthmatics and in a murine model. Clin Immunol. 2017;183:158–166. doi: 10.1016/j.clim.2017.08.013. [DOI] [PubMed] [Google Scholar]
- 45.Kessel C., Holzinger D., Foell D. Phagocyte-derived S100 proteins in autoinflammation: putative role in pathogenesis and usefulness as biomarkers. Clin Immunol. 2013;147:229–241. doi: 10.1016/j.clim.2012.11.008. [DOI] [PubMed] [Google Scholar]
- 46.Halayko A.J., Ghavami S. S100A8/A9: a mediator of severe asthma pathogenesis and morbidity? Can J Physiol Pharmacol. 2009;87:743–755. doi: 10.1139/Y09-054. [DOI] [PubMed] [Google Scholar]
- 47.Cuppari C., Manti S., Chirico V., Caruso R., Salpietro V., Giacchi V., et al. Sputum high mobility group box-1 in asthmatic children: a noninvasive sensitive biomarker reflecting disease status. Ann Allergy Asthma Immunol. 2015;115:103–107. doi: 10.1016/j.anai.2015.06.008. [DOI] [PubMed] [Google Scholar]
- 48.Zhang F., Huang G., Hu B., Fang L.P., Cao E.H., Xin X.F., et al. Anti-HMGB1 neutralizing antibody ameliorates neutrophilic airway inflammation by suppressing dendritic cell-mediated th17 polarization. Mediators Inflamm. 2014;2014 doi: 10.1155/2014/257930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Papayannopoulos V. Neutrophil extracellular traps in immunity and disease. Nat Rev Immunol. 2018;18:134–147. doi: 10.1038/nri.2017.105. [DOI] [PubMed] [Google Scholar]
- 50.Tadie J.M., Bae H.B., Jiang S., Park D.W., Bell C.P., Yang H., et al. HMGB1 promotes neutrophil extracellular trap formation through interactions with Toll-like receptor 4. Am J Physiol Lung Cell Mol Physiol. 2013;304:8–12. doi: 10.1152/ajplung.00151.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liu T., Wang F.P., Wang G., Mao H. Role of neutrophil extracellular traps in asthma and chronic obstructive pulmonary disease. Chin Med J (Engl) 2017;130:730–736. doi: 10.4103/0366-6999.201608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wan R., Jiang J., Hu C., Chen X., Chen C., Zhao B., et al. Neutrophil extracellular traps amplify neutrophil recruitment and inflammation in neutrophilic asthma by stimulating the airway epithelial cells to activate the TLR4/NF-κB pathway and secrete chemokines. Aging (Albany NY) 2020;12:1–17. doi: 10.18632/aging.103479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chen X., Li Y., Qin L., He R., Hu C. Neutrophil extracellular trapping network promotes the pathogenesis of neutrophil-associated asthma through macrophages. Immunol Invest. 2021;50:544–561. doi: 10.1080/08820139.2020.1778720. [DOI] [PubMed] [Google Scholar]
- 54.Radermecker C., Louis R., Bureau F., Marichal T. Role of neutrophils in allergic asthma. Curr Opin Immunol. 2018;54:28–34. doi: 10.1016/j.coi.2018.05.006. [DOI] [PubMed] [Google Scholar]
- 55.Brinkmann V., Zychlinsky A. Beneficial suicide: why neutrophils die to make NETs. Nat Rev Microbiol. 2007;5:577–582. doi: 10.1038/nrmicro1710. [DOI] [PubMed] [Google Scholar]
- 56.Polosa R., Thomson N.C. Smoking and asthma: dangerous liaisons. Eur Respir J. 2013;41:716–725. doi: 10.1183/09031936.00073312. [DOI] [PubMed] [Google Scholar]
- 57.Ray A., Kolls J.K. Neutrophilic inflammation in asthma and association with disease severity. Trends Immunol. 2017;38:942–954. doi: 10.1016/j.it.2017.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Strachan D. Family size, infection and atopy: the first decade of the “hygiene hypothesis”. Thorax. 2000;55:2S–10. doi: 10.1136/thorax.55.suppl_1.s2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Fonseca D.M., Wowk P.F., Paula M.O., Campos L.W., Gembre A.F., Turato W.M., et al. Recombinant DNA immunotherapy ameliorate established airway allergy in a IL-10 dependent pathway. Clin Exp Allergy. 2012;42:131–143. doi: 10.1111/j.1365-2222.2011.03845.x. [DOI] [PubMed] [Google Scholar]
- 60.Fonseca D.M., Wowk P.F., Paula M.O., Gembre A.F., Baruffi M.D., Fermino M.L., et al. Requirement of MyD88 and Fas pathways for the efficacy of allergen-free immunotherapy. Allergy. 2015;70:275–284. doi: 10.1111/all.12555. [DOI] [PubMed] [Google Scholar]
- 61.Fonseca D.M., Paula M.O., Wowk P.F., Campos L.W., Gembre A.F., Turato W.M., et al. IFN-γ-mediated efficacy of allergen-free immunotherapy using mycobacterial antigens and CpG-ODN. Immunol Cell Biol. 2011;89:777–785. doi: 10.1038/icb.2011.9. [DOI] [PubMed] [Google Scholar]
- 62.Prado R.Q., Bertolini T.B., Piñeros A.R., Gembre A.F., Ramos S.G., Silva C.L., et al. Attenuation of experimental asthma by mycobacterial protein combined with CpG requires a TLR9-dependent IFN-γ-CCR2 signalling circuit. Clin Exp Allergy. 2015;45:1459–1471. doi: 10.1111/cea.12564. [DOI] [PubMed] [Google Scholar]
- 63.Boulet L.P. Influence of comorbid conditions on asthma. Eur Respir J. 2009;33:897–906. doi: 10.1183/09031936.00121308. [DOI] [PubMed] [Google Scholar]
- 64.Carr T.F., Kraft M. Chronic infection and severe asthma. Immunol Allergy Clin North Am. 2016;36:483–502. doi: 10.1016/j.iac.2016.03.010. [DOI] [PubMed] [Google Scholar]
- 65.Guilbert T.W., Denlinger L.C. Role of infection in the development and exacerbation of asthma. Expert Rev Respir Med. 2010;4:71–83. doi: 10.1586/ers.09.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lambrecht B.N., Hammad H. The immunology of the allergy epidemic and the hygiene hypothesis. Nat Immunol. 2017;18:1076–1083. doi: 10.1038/ni.3829. [DOI] [PubMed] [Google Scholar]
- 67.Zhou Y., Jackson D., Bacharier L.B., Mauger D., Boushey H., Castro M., et al. The upper-airway microbiota and loss of asthma control among asthmatic children. Nat Commun. 2019;10:1–10. doi: 10.1038/s41467-019-13698-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Durack J., Huang Y.J., Nariya S., Christian L.S., Mark Ansel K., Beigelman A., et al. Bacterial biogeography of adult airways in atopic asthma. Microbiome. 2018;6:1–16. doi: 10.1186/s40168-018-0487-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Green B.J., Wiriyachaiporn S., Grainge C., Rogers G.B., Kehagia V., Lau R., et al. Potentially pathogenic airway bacteria and neutrophilic inflammation in treatment resistant severe asthma. PLoS One. 2014;9:4–10. doi: 10.1371/journal.pone.0100645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Brusselle G.G., VanderStichele C., Jordens P., Deman R., Slabbynck H., Ringoet V., et al. Azithromycin for prevention of exacerbations in severe asthma (AZISAST): a multicentre randomised double-blind placebo-controlled trial. Thorax. 2013;68:322–329. doi: 10.1136/thoraxjnl-2012-202698. [DOI] [PubMed] [Google Scholar]
- 71.Baines K.J., Simpson J.L., Scott R.J., Gibson P.G. Immune responses of airway neutrophils are impaired in asthma. Exp Lung Res. 2009;35:554–569. doi: 10.1080/01902140902777490. [DOI] [PubMed] [Google Scholar]
- 72.Brooks L.R.K., Mias G.I. Streptococcus pneumoniae’s virulence and host immunity: aging, diagnostics, and prevention. Front Immunol. 2018;9:1366. doi: 10.3389/fimmu.2018.01366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Essilfie A.-T., Simpson J.L., Horvat J.C., Preston J.A., Dunkley M.L., Foster P.S., et al. Haemophilus influenzae infection drives IL-17-mediated neutrophilic allergic airways disease. PLoS Pathog. 2011;7 doi: 10.1371/journal.ppat.1002244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhang J., Zhu Z., Zuo X., Pan H., Gu Y., Yuan Y., et al. The role of NTHi colonization and infection in the pathogenesis of neutrophilic asthma. Respir Res. 2020;21:1–12. doi: 10.1186/s12931-020-01438-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Simpson J.L., Daly J., Baines K.J., Yang I.A., Upham J.W., Reynolds P.N., et al. Airway dysbiosis: Haemophilus influenza and Tropheryma in poorly controlled asthma. Eur Respir J. 2016;47:792–800. doi: 10.1183/13993003.00405-2015. [DOI] [PubMed] [Google Scholar]
- 76.Andrews C.S., Miyata M., Susuki-Miyata S., Lee B.C., Komatsu K., Li J.D. Nontypeable Haemophilus influenzae-induced MyD88 short expression is regulated by positive IKKβ and CREB pathways and negative ERK1/2 pathway. PLoS One. 2015;10:1–15. doi: 10.1371/journal.pone.0144840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Essilfie A.T., Simpson J.L., Dunkley M.L., Morgan L.C., Oliver B.G., Gibson P.G., et al. Combined Haemophilus influenzae respiratory infection and allergic airways disease drives chronic infection and features of neutrophilic asthma. Thorax. 2012;67:588–599. doi: 10.1136/thoraxjnl-2011-200160. [DOI] [PubMed] [Google Scholar]
- 78.Wang G., Pang Z., Chen-Yu Hsu A., Guan X., Ran N., Yuan Y., et al. Combined treatment with SB203580 and dexamethasone suppresses non-typeable Haemophilus influenzae-induced Th17 inflammation response in murine allergic asthma. Eur J Pharmacol. 2019;862 doi: 10.1016/j.ejphar.2019.172623. [DOI] [PubMed] [Google Scholar]
- 79.Yang X., Wang Y., Zhao S., Wang R., Wang C. Long-term exposure to low-dose Haemophilus influenzae during allergic airway disease drives a steroid-resistant neutrophilic inflammation and promotes airway remodeling. Oncotarget. 2018;9:24898–24913. doi: 10.18632/oncotarget.24653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Taylor S.L., Leong L.E.X., Mobegi F.M., Choo J.M., Wesselingh S., Yang I.A., et al. Long-term azithromycin reduces Haemophilus influenzae and increases antibiotic resistance in severe asthma. Am J Respir Crit Care Med. 2019;200:309–317. doi: 10.1164/rccm.201809-1739OC. [DOI] [PubMed] [Google Scholar]
- 81.Su Y., Singh B., Riesbeck K. Moraxella catarrhalis: from interactions with the host immune system to vaccine development. Future Microbiol. 2012;7:1073–1100. doi: 10.2217/fmb.12.80. [DOI] [PubMed] [Google Scholar]
- 82.Romero-Espinoza J.A., Moreno-Valencia Y., Coronel-Tellez R.H., Castillejos-Lopez M., Hernandez A., Dominguez A., et al. Virome and bacteriome characterization of children with pneumonia and asthma in Mexico City during winter seasons 2014 and 2015. PLoS One. 2018;13:1–14. doi: 10.1371/journal.pone.0192878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.McCauley K., Durack J., Valladares R., Fadrosh D.W., Lin D.L., Calatroni A., et al. Distinct nasal airway bacterial microbiotas differentially relate to exacerbation in pediatric patients with asthma. J Allergy Clin Immunol. 2019;144:1187–1197. doi: 10.1016/j.jaci.2019.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hassan F. Molecular mechanisms of Moraxella catarrhalis-induced otitis media. Curr Allergy Asthma Rep. 2013;13:512–517. doi: 10.1007/s11882-013-0374-8. [DOI] [PubMed] [Google Scholar]
- 85.Xie H., Gu X.-X. Moraxella catarrhalis lipooligosaccharide selectively upregulates ICAM-1 expression on human monocytes and stimulates adjacent naïve monocytes to produce TNF-α through cellular cross-talk. Cell Microbiol. 2008;10:1453–1467. doi: 10.1111/j.1462-5822.2008.01138.x. [DOI] [PubMed] [Google Scholar]
- 86.Alnahas S., Hagner S., Raifer H., Kilic A., Gasteiger G., Mutters R., et al. IL-17 and TNF-α are key mediators of Moraxella catarrhalis triggered exacerbation of allergic airway inflammation. Front Immunol. 2017;8:1–11. doi: 10.3389/fimmu.2017.01562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.World Health Organization Weekly epidemiological record. Relevé épidémiologique hebdomadaire. 2019;(8):85–104. [Google Scholar]
- 88.World Health Organization Weekly epidemiological record. Relevé épidémiologique hebdomadaire. 2021;(23):217–228. [Google Scholar]
- 89.Jounio U., Juvonen R., Bloigu A., Silvennoinen-Kassinen S., Kaijalainen T., Kauma H., et al. Pneumococcal carriage is more common in asthmatic than in non-asthmatic young men. Clin Respir J. 2010;4:222–229. doi: 10.1111/j.1752-699X.2009.00179.x. [DOI] [PubMed] [Google Scholar]
- 90.Talbot T.R., Hartert T.V., Mitchel E., Halasa N.B., Arbogast P.G., Poehling K.A., et al. Asthma as a risk factor for invasive pneumococcal disease. N Engl J Med. 2005;352:2082–2090. doi: 10.1056/NEJMoa044113. [DOI] [PubMed] [Google Scholar]
- 91.Paterson G.K., Mitchell T.J. Innate immunity and the pneumococcus. Microbiology. 2006;152:285–293. doi: 10.1099/mic.0.28551-0. [DOI] [PubMed] [Google Scholar]
- 92.Lee K.S., Scanga C.A., Bachelder E.M., Chen Q., Snapper C.M. TLR2 synergizes with both TLR4 and TLR9 for induction of the MyD88-dependent splenic cytokine and chemokine response to Streptococcus pneumoniae. Cell Immunol. 2007;245:103–110. doi: 10.1016/j.cellimm.2007.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Famà A., Midiri A., Mancuso G., Biondo C., Lentini G., Galbo R., et al. Nucleic acid-sensing Toll-like receptors play a dominant role in innate immune recognition of Pneumococci. MBio. 2020;11:e00415–e00420. doi: 10.1128/mBio.00415-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Raquil M.-A., Anceriz N., Rouleau P., Tessier P.A. Blockade of antimicrobial proteins S100A8 and S100A9 inhibits phagocyte migration to the alveoli in Streptococcal pneumonia. J Immunol. 2008;180:3366–3374. doi: 10.4049/jimmunol.180.5.3366. [DOI] [PubMed] [Google Scholar]
- 95.Dudek M., Puttur F., Arnold-Schrauf C., Kühl A.A., Holzmann B., Henriques-Normark B., et al. Lung epithelium and myeloid cells cooperate to clear acute pneumococcal infection. Mucosal Immunol. 2016;9:1288–1302. doi: 10.1038/mi.2015.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Noske N., Kämmerer U., Rohde M., Hammerschmidt S. Pneumococcal interaction with human dendritic cells: phagocytosis, survival, and induced adaptive immune response are manipulated by PavA. J Immunol. 2009;183:1952–1963. doi: 10.4049/jimmunol.0804383. [DOI] [PubMed] [Google Scholar]
- 97.Ivanov S., Fontaine J., Paget C., MacHo Fernandez E., Van Maele L., Renneson J., et al. Key role for respiratory CD103+ dendritic cells, IFN-, and IL-17 in protection against Streptococcus pneumoniae infection in response to-galactosylceramide. J Infect Dis. 2012;206:723–734. doi: 10.1093/infdis/jis413. [DOI] [PubMed] [Google Scholar]
- 98.Hoe E., Anderson J., Nathanielsz J., Toh Z.Q., Marimla R., Balloch A., et al. The contrasting roles of Th17 immunity in human health and disease. Microbiol Immunol. 2017;61:49–56. doi: 10.1111/1348-0421.12471. [DOI] [PubMed] [Google Scholar]
- 99.Kim H.Y., Kim S.K., Seo H.S., Jeong S., Ahn K.B., Yun C.-H., et al. Th17 activation by dendritic cells stimulated with gamma-irradiated Streptococcus pneumoniae. Mol Immunol. 2018;101:344–352. doi: 10.1016/j.molimm.2018.07.023. [DOI] [PubMed] [Google Scholar]
- 100.Wilson R., Cohen J.M., Jose R.J., De Vogel C., Baxendale H., Brown J.S. Protection against Streptococcus pneumoniae lung infection after nasopharyngeal colonization requires both humoral and cellular immune responses. Mucosal Immunol. 2015;8:627–639. doi: 10.1038/mi.2014.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Beiter K., Wartha F., Albiger B., Normark S., Zychlinsky A., Henriques-Normark B. An endonuclease allows Streptococcus pneumoniae to escape from neutrophil extracellular traps. Curr Biol. 2006;16:401–407. doi: 10.1016/j.cub.2006.01.056. [DOI] [PubMed] [Google Scholar]
- 102.Luo J., An X., Yao Y., Erb C., Ferguson A., Kolls J.K., et al. Epigenetic regulation of IL-17-induced chemokines in lung epithelial cells. Mediators Inflamm. 2019;2019 doi: 10.1155/2019/9050965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Van Maele L., Carnoy C., Cayet D., Ivanov S., Porte R., Deruy E., et al. Activation of type 3 innate lymphoid cells and interleukin 22 secretion in the lungs during Streptococcus pneumoniae infection. J Infect Dis. 2014;210:493–503. doi: 10.1093/infdis/jiu106. [DOI] [PubMed] [Google Scholar]
- 104.Spits H., Artis D., Colonna M., Diefenbach A., Di Santo J.P., Eberl G., et al. Innate lymphoid cells—a proposal for uniform nomenclature. Nat Rev Immunol. 2013;13:145–149. doi: 10.1038/nri3365. [DOI] [PubMed] [Google Scholar]
- 105.Schuller D.E. Prophylaxis of otitis media in asthmatic children. Pediatr Infect Dis J. 1983;2:280–283. doi: 10.1097/00006454-198307000-00004. [DOI] [PubMed] [Google Scholar]
- 106.Ansaldi F., Turello V., Lai P., Batone G., De Luca S., Rosselli R., et al. Effectiveness of a 23-valent polysaccharide vaccine in preventing pneumonia and non-invasive pneumococcal infection in elderly people: a large-scale retrospective cohort study. J Int Med Res. 2005;33:490–500. doi: 10.1177/147323000503300503. [DOI] [PubMed] [Google Scholar]
- 107.Hartmann C., Behrendt A.-K., Henken S., Wölbeling F., Maus U.A., Hansen G. Pneumococcal pneumonia suppresses allergy development but preserves respiratory tolerance in mice. Immunol Lett. 2015;164:44–52. doi: 10.1016/j.imlet.2014.12.001. [DOI] [PubMed] [Google Scholar]
- 108.Preston J.A., Thorburn A.N., Starkey M.R., Beckett E.L., Horvat J.C., Wade M.A., et al. Streptococcus pneumoniae infection suppresses allergic airways disease by inducing regulatory T-cells. Eur Respir J. 2011;37:53–64. doi: 10.1183/09031936.00049510. [DOI] [PubMed] [Google Scholar]
- 109.Thorburn A.N., O’Sullivan B.J., Thomas R., Kumar R.K., Foster P.S., Gibson P.G., et al. Pneumococcal conjugate vaccine-induced regulatory T cells suppress the development of allergic airways disease. Thorax. 2010;65:1053–1060. doi: 10.1136/thx.2009.131508. [DOI] [PubMed] [Google Scholar]
- 110.Thorburn A.N., Brown A.C., Nair P.M., Chevalier N., Foster P.S., Gibson P.G., et al. Pneumococcal components induce regulatory T cells that attenuate the development of allergic airways disease by deviating and suppressing the immune response to allergen. J Immunol. 2013;191:4112–4120. doi: 10.4049/jimmunol.1201232. [DOI] [PubMed] [Google Scholar]
- 111.Yang B., Liu R., Yang T., Jiang X., Zhang L., Wang L., et al. Neonatal Streptococcus pneumoniae infection may aggravate adulthood allergic airways disease in association with IL-17A. PLoS One. 2015;10:1–12. doi: 10.1371/journal.pone.0123010. [DOI] [PMC free article] [PubMed] [Google Scholar]