Abstract
Background
Rheumatic immune-related adverse events (R-irAEs) occur in 5–15% of patients receiving immune checkpoint inhibitors (ICI) and, unlike other irAEs, tend to be chronic. Herein, we investigate the factors influencing cancer and R-irAEs outcomes with particular focus on adverse effects of anti-inflammatory treatment.
Methods
In this prospective, multicenter, long-term, observational study, R-irAEs were comprehensively analyzed in patients with malignant melanoma (MM, n=50) and non-small cell lung cancer (NSCLC, n=41) receiving ICI therapy who were enrolled in the study between August 1, 2018, and December 11, 2022.
Results
After a median follow-up of 33 months, progressive disease or death occurred in 66.0% and 30.0% of MM and 63.4% and 39.0% of patients with NSCLC. Male sex (progression-free survival (PFS): p=0.013, and overall survival (OS): p=0.009), flare of a pre-existing condition (vs de novo R-irAE, PFS: p=0.010) and in trend maximum glucocorticoid (GC) doses >10 mg and particularly ≥1 mg/kg prednisolone equivalent (sex-adjusted PFS: p=0.056, OS: p=0.051) were associated with worse cancer outcomes. Patients receiving disease-modifying antirheumatic drugs (DMARDs) showed significantly longer PFS (n=14, p=0.011) and OS (n=20, p=0.018). Effects of these variables on PFS and/or OS persisted in adjusted Cox regression models. Additionally, GC treatment negatively correlated with the time from diagnosis of malignancy and the latency from ICI start until R-irAE onset (all p<0.05). R-irAE features and outcomes were independent of other baseline patient characteristics in both studied cancer entities.
Conclusion
Male sex, flare of pre-existing rheumatologic conditions and extensive GC treatment appeared to be linked with unfavorable cancer outcomes, while DMARD use had a favorable impact. These findings challenge the current dogma of restrictive DMARD use for R-irAE and thus may pave the way to better strategies and randomized controlled trials for the growing number of patients with R-irAE.
Keywords: Immune Checkpoint Inhibitors, Immunotherapy, Melanoma, Non-Small Cell Lung Cancer, rheumatic immune-related adverse event
WHAT IS ALREADY KNOWN ON THIS TOPIC
Up to 20% of patients treated with immune-checkpoint inhibitors develop rheumatic immune-related adverse events (R-irAE), which is under-recognized in large registrational trials and appear to be associated with protracted courses, reduced quality of life, but often beneficial oncological outcomes. Strategies to treat R-irAEs need to control musculoskeletal symptoms without compromising antitumor activity of cancer therapy.
WHAT THIS STUDY ADDS
The type of underlying malignancy and most patient baseline characteristics are not related to the development, clinical features and outcome of R-irAEs. Oncological outcomes of patients with R-irAE are influenced by patient sex, emergence of R-irAE de novo versus as flare of a pre-existing condition as well as use of glucocorticoids and/or other immunomodulatory or immunosuppressive treatments.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
Toxicity management of R-irAEs with glucocorticoid-sparing therapies urgently needs to be investigated in randomized controlled trials to determine if these are superior to high-dose of glucocorticoid therapy regarding cancer and R-irAE outcome. Future R-irAE trials could recruit mixed cohorts of patients with melanoma, lung and possibly other cancers in order to overcome the challenge of low patient numbers limiting the quality of obtainable evidence.
Introduction
Immune checkpoint inhibitors (ICIs) have revolutionized the treatment of several cancer entities by targeting immunoregulatory molecules such as cytotoxic T-lymphocyte associated protein 4 (CTLA-4), programmed death-1 (PD-1) and its ligand PD-L1, resulting in enhanced T cell-mediated antitumor responses. However, in most cases, this immunotherapy is associated with off-target inflammation, known as immune-related adverse events (irAEs). These can affect any organ system, thereby providing a novel link between malignancy and autoimmunity. While most irAEs resolve after short-term glucocorticoid (GC) therapy, a hallmark of rheumatic irAEs (R-irAEs), which affect 5–15% of ICI patients, is a persistent course with recurrent flares. This leads to a growing patient population often requiring prolonged anti-inflammatory treatment and long-term interdisciplinary care.1–4 Reliable identification and management of R-irAEs is a major challenge as these mostly only mimic classical rheumatic and musculoskeletal diseases (RMDs) and are therefore referred to as “rheumatoid arthritis (RA)-like” or “spondyloarthritis (SpA)-like”.1 5 Valid data from prospective studies as well as classification and outcome measures are largely missing for R-irAEs.5 However, they cannot be derived from previous large ICI clinical trials due to under-recognition/under-reporting of R-irAEs and shortcomings of the Common Terminology Criteria for Adverse Events for R-irAEs.1 2 6–11 This also leaves unanswered the question of whether R-irAE characteristics differ according to the underlying tumor as described for skin and gastrointestinal irAEs.12 13
Current recommendations for the management of R-irAEs from oncology and rheumatology societies are based on evidence from case reports or series, expert opinion and extrapolation from classical RMDs.1 14–19 To date, due to concerns about compromising antitumor efficacy—in contrast to the treat-to-target remission principles used for classical RMDs—clinicians have prioritized symptomatic relief through a severity-based step-up therapeutic approach using non-steroidal anti-inflammatory drugs (NSAIDs), GCs, disease-modifying antirheumatic drugs (DMARDs) and immunosuppressants. DMARDs can be classified as either conventional synthetic (cs) or biologic (b) DMARDs and are immunomodulatory agents used in arthritis (eg, RA) but also in other diseases such as connective tissue diseases (eg, systemic lupus erythematosus, Sjögren’s syndrome) or vasculitis with the aim of GC sparing. The most commonly used csDMARDs are methotrexate (MTX) and leflunomide with similar efficacy and safety in arthritis, the latter usually used when MTX is contraindicated (eg, renal failure), intolerable or infective. Both have pleiotropic effects on the inflammatory processes and involved immune cell populations, however, the main proposed mechanisms of action for low-dose MTX are interference with nucleoside synthesis (antiproliferative) and elevation of adenosine levels (anti-inflammatory), and for leflunomide inhibition of dihydroorotate dehydrogenase, thus blocking the synthesis of pyrimidine, its effect being particularly toxic to activated lymphocytes which depend on de novo synthesis of uridine. In contrast, the class of bDMARDs (in the context or R-irAEs mainly tumor necrosis factor inhibitors (TNFi, eg, adalimumab) and interleukin (IL)-6R antagonists (IL-6i, eg, tocilizumab)) unfold their effect by targeted protein ligand binding and subsequent inhibiting mechanisms such as the neutralization of key cytokine functions or B/T-cell activation.20 Although infections, mostly non-severe (eg, nasopharyngitis), occur more frequently under csDMARDs, and to a higher degree in bDMARDs (OR 1.0–1.3 per study vs csDMARDs), due to rather low incidence of serious infections under csDMARDs and even under bDMARDs both are considered immunomodulatory rather than immunosuppressive.21
While the occurrence of irAEs has mostly been shown to be associated with improved efficacy of immunotherapy (favorable response rates, prolonged survival3 22–27), detailed data on the impact of R-irAEs on tumor outcome are scarce,3 6 28 29 and the question of whether anti-inflammatory treatment can reverse this benefit remains largely unanswered.1 3 6 28 29 In this context, a recent retrospective multicenter observational study in patients with R-irAE-arthritis suggests that the use of bDMARDs may be inferior to MTX regarding progression-free survival (PFS), but superior in terms of rapid symptom relief.30 However, it does not address some fundamental questions in patient-centered care, particularly, whether DMARDs should be preferred over GC treatment. Our comprehensive analysis of a well-characterized prospective malignant melanoma (MM) and non-small cell lung cancer (NSCLC) cohort with long-term follow-up addresses these questions and shows that clinical management by restrictive GC use influences the characteristics and outcome of R-irAEs.
Patients and methods
Patient recruitment and data collection
A detailed description of the study design, patient recruitment and data collection is available in the online supplemental file 1. Briefly, this prospective, multicenter (two sites), observational study included patients treated with ICI (anti-CTLA-4, anti-PD-1, anti-PD-L-1 therapy or combination) for MM or NSCLC who presented with R-irAEs. R-irAEs were classified as flares of a pre-existing (suspected) condition or de novo appearance. Patients were enrolled between August 1, 2018, and December 11, 2022. Clinical data, including baseline characteristics, time from start of ICI to R-irAE onset, and R-irAE treatments, and patient survival data (169 parameters) were extracted and analyzed.
jitc-2023-007557supp001.pdf (1.5MB, pdf)
Statistical analyses
Statistical analysis was conducted using SPSS V.29.0 (IBM, Ehningen, Germany) and R software V.>4.00.0. Comparisons among categorical variables were assessed by χ2 test. Metric variables were described as mean values±SD for normally distributed values or median (first quartile (Q1); third quartile (Q3)) for not normally distributed values and compared by Student’s t-test or Mann-Whitney U test, as appropriate. Latency (time frame from start of ICI treatment line related to R-irAE until R-irAE onset) was compared by Kruskal-Wallis test and uncorrected Dunn’s test. Progression-free survival (PFS) and overall survival (OS) related to tumor entity and neoadjuvant or adjuvant versus palliative ICI treatment lines was estimated by the Kaplan-Meier method and calculated as time from start of ICI treatment line related to R-irAE onset until progression or death from any cause, respectively. Thereby, estimators were adjusted for right censoring and left truncation (due to immortal time bias between ICI treatment start and R-irAE onset). Further PFS and OS subanalyses focused on patients with palliative (first or any line) ICI treatment at R-irAE onset and separated them according to different variables. For all PFS analyses, patients with oncological progression before R-irAE onset under ICI treatment line related to R-irAE were excluded. In patients with no events of progression or death at cut-off, the date of last contact was used for censored calculation. Univariate comparisons of Kaplan-Meier estimators were done with the log-rank test. To identify confounding effects between variables adjusted Cox regression models were calculated for selected parameters (cancer type, sex, R-irAE subtype, ICI treatment regimen and GC dose), but patient numbers were considered not sufficient for robust multivariate regression analysis of all parameters. Therefore, χ2 test (categorical variables), Student’s t-test or Mann-Whitney U test (metric variables), as appropriate, were used to identify additional confounding effects of sex, R-irAE subtype and ICI treatment regimen on all parameters. Correlation analysis of latencies was conducted using Spearman’s rank correlation. P values in all analyses were considered significant with values of p<0.05 in a descriptive sense.
Results
Patient and cancer characteristics
A total of 50 patients with MM and 41 patients with NSCLC who developed any R-irAE under ICI treatment were enrolled in this study. Rheumatological evaluation was prospective and started at a median of ~2 months after R-irAE onset. Baseline characteristics were comparable between MM and NSCLC cohorts with respect to sex distribution and body mass index, but patients with NSCLC were older and more often smokers, consistent with the epidemiology of each disease (table 1). ICI treatment at R-irAE onset comprised mainly PD-(L)1 inhibitors alone or in combination with CTLA-4 inhibitors, the latter significantly more often in patients with MM. Median follow-up was ~33 months (1,000.0 days (610.0; 1,333.0)) after R-irAE onset, during which time progressive disease (related to ICI line at R-irAE onset) or death was observed in 66.0% and 63.4% of patients with MM and patients with NSCLC, respectively, while death was observed in 30.0% versus 39.0% (table 1, online supplemental figure 3). An overview of cancer characteristics is provided in table 1, online supplemental figure 2, 3 and table 1.
Table 1.
Demographic and clinical characteristics
| Variable | MM cohort (n=50) |
NSCLC cohort (n=41) |
P value |
| Demographics | |||
| Females (%) | 26 (52.0) | 21 (51.2) | 0.941 |
| Age (years)* (Q1; Q3) | 56.7 (52.9; 71.3) | 66.0 (59.4; 72.4) | <0.05 |
| BMI (kg/m²)† (Q1; Q3) | 26.3 (24.5; 28.7) | 25.4 (22.6; 30.0) | 0.685 |
| Smoker ever (%) | 22 (44.0) | 38 (92.7) | <0.001 |
| Malignancy characteristics | |||
| Cancer duration* (months)±SD | 35.9±44.9 | 15.8±26.3 | <0.001 |
| Cancer stage III at ICI start (%) | 15 (30.0) | 5 (12.2) | <0.05 |
| Cancer stage IV at ICI start (%) | 35 (70.0) | 36 (87.8) | <0.05 |
| ICI treatment | |||
| Combined anti-CTLA-4 and anti-PD-1 at R-irAE onset (%) | 16 (32.0) | 2 (4.9) | 0.001 |
| Neoadjuvant ICI treatment ever (%) | 0 (0) | 2 (4.9) | 0.114 |
| Adjuvant ICI treatment ever (%) | 19 (38.0) | 1 (2.4) | <0.001 |
| ICI duration until R-irAE onset (days) (Q1; Q3) | 129.5 (42.75; 225.0) | 98.0 (41.0; 224.5) | 0.771 |
|
32 (64.0) | 26 (63.4) | 0.954 |
| Progression and survival status | |||
| Progressive disease since ICI line start related to R-irAE (%) | 33 (66.0) | 26 (63.4) | 0.797 |
|
290.0 (75.5; 721.0) | 257.5 (90.5; 430.5) | 0.557 |
| Death (%) | 15 (30.0) | 16 (39.0) | 0.366 |
|
777.6±367.3 | 798.9±419.2 | 0.882 |
| Autoimmunity characteristics | |||
| Follow-up time since R-irAE onset (days)±SD | 1,062.3±600.0 | 990.5±514.3 | 0.547 |
| R-irAE duration until REV (days) (Q1; Q3) | 64.5 (32.0; 161.5) | 59.0 (15.0; 115.0) | 0.144 |
| R-irAE phenotype‡ | |||
| De-novo overall | 42 (84.0) | 34 (82.9) | 0.891 |
|
7 (14.0) | 8 (19.5) | 0.481 |
|
26 (52.0) | 5 (12.2) | <0.001 |
| Pre-existing (suspected) condition (RMD or paraneoplasia) | 13 (26.0) | 13 (31.7) | 0.549 |
|
10 (20.0) | 7 (17.1) | 0.722 |
| R-irAE remission status at last follow-up: | |||
| R-irAE duration >6 months (%) | 38 (76.0) | 29 (70.7) | 0.570 |
| Complete remission (%) | 31 (62.0) | 17 (43.6) | 0.084 |
|
198.0 (130.0; 418.0) | 165.0 (67.0; 410.0) | 0.311 |
| Ongoing (%): | |||
|
13 (26.0) | 9 (23.1) | 0.751 |
|
3 (6.0) | 8 (20.5) | <0.05 |
|
3 (6.0) | 5 (12.8) | 0.263 |
| History of other organ autoimmune disease: | |||
| Own history: | 8 (16.0) | 7 (17.1) | 0.891 |
| Family history: | 13 (26.0) | 12 (29.3) | 0.728 |
| Other irAEs | |||
| Total (%) | 32 (64.0) | 19 (47.5) | 0.116 |
| Skin (%) | 11 (22.0) | 8 (19.5) | 0.771 |
| Gastrointestinal tract (%) | 9 (18.0) | 3 (7.3) | 0.134 |
| Liver (%) | 7 (14.0) | 1 (2.4) | 0.053 |
| Lung (%) | 6 (12.0) | 4 (9.8) | 0.733 |
| Endocrine (%) | 18 (36.0) | 7 (17.1) | <0.05 |
| Other (%) | 7 (14.0) | 6 (14.6) | 0.931 |
Numbers represent mean values±SD for normally distributed values; median (first quartile (Q1) and third quartile (Q3)) for not normally distributed values or frequencies (%).
*At time of R-irAE onset.
†At time of first rheumatological evaluation.
‡Six patients suffered from more than one distinct R-irAE.
BMI, body mass index; CTLA-4, cytotoxic T-lymphocyte associated protein 4; ICI, immune checkpoint inhibitor(s); MM, malignant melanoma; NSCLC, non-small cell lung cancer; PD-1, programmed death-1; REV, rheumatologic evaluation; R-irAE, rheumatic immune-related adverse event; RMD, rheumatic and musculoskeletal disease.
Autoimmunity characteristics
The R-irAE subtypes observed in the MM and NSCLC cohorts showed strong similarities (online supplemental figure 1) with polyarticular arthritis as the most frequent inflammatory R-irAE manifestation and inflammatory findings on ultrasound in the majority (~67%) of cases (online supplemental table 2, 3). De novo R-irAEs occurred in approximately 83% of patients in both cohorts, with SpA-like, polymyalgia rheumatica (PMR)-like and RA-like being the most common clinically observed subtypes. De novo R-irAEs fulfilled the classification criteria for classical disease in only ~16% of cases. However, suspected de novo classical RMDs were significantly more frequent in patients with MM compared with patients with NSCLC. Most patients with a suspected pre-existing RMD condition (10/13 (76.9%) of MM and 7/13 (53.8%) of NSCLC patients) experienced an R-irAE flare during ICI treatment; these flares accounted for ~19% of patients (online supplemental figure 1). Of note, only three flare patients were actively treated with GCs within the year prior to ICI initiation and previous DMARD expositions (all stopped minimum 5 years prior to ICI start) were reported in only three other cases. Non-rheumatic irAEs were observed in 64.0% versus 47.5% of patients with MM and patients with NSCLC, respectively, with some variability across the different organ systems (table 1).
Evaluation of laboratory findings showed similarly elevated C-reactive protein levels in ~80% of cases, with median concentrations of 24.3 mg/L (8.2; 56.9; (norm≤5)), and elevated mean soluble IL2 receptor (sCD25) levels in ~40% of patients. Higher creatine kinase (CK) levels were significantly more common in patients with MM, but de novo myositis was diagnosed in only 3/16 and 1/2 patients with elevated CK, respectively. More patients with NSCLC than with MM had a neutrophil-to-lymphocyte ratio (NLR)>5 at the start of ICI treatment. Analysis of autoantibodies associated with RMDs showed that a small proportion (<5%) of patients were positive for specific immunoserological findings (online supplemental table 4).
R-irAE management
The vast majority of patients received NSAIDs and/or GC for the treatment of their R-irAE, but GC were also applied for various other reasons (eg, other organ irAE, or as adjunct medication to chemotherapy or radiotherapy) (table 2). Mean cumulative GC doses were generally similar between patients with MM and patients with NSCLC. The percentage of patients receiving >10 mg prednisolone equivalent since R-irAE onset was numerically lower in the MM cohort compared with the NSCLC cohort (64.0% vs 80.5%), but comparable when looking at R-irAE as indication only (~14%). GC treatment longer than 6 months (due to R-irAEs) was observed less frequently in patients with MM versus patients with NSCLC (34.0% vs 46.3%), potentially because GC was successfully tapered more often due to R-irAE remission in patients with MM (41.9% vs 28.6%).
Table 2.
Therapeutic management of rheumatic irAE
| MM cohort (n=50) | NSCLC cohort (n=41)* | P value | |
| NSAID treatment | |||
| NSAIDs for R-irAE ever (%) | 96.0 | 43.9 | <0.001 |
| NSAIDs monotherapy only (%) | 8.0 | 0 | 0.064 |
| Glucocorticoids (GC) treatment | |||
| GC since R-irAE ever (%) | 92.0 | 97.6 | 0.247 |
| monotherapy (±NSAID) only (%) | 64.0 | 73.2 | 0.350 |
| ≤10 mg† only (%) | 28.0 | 17.1 | 0.218 |
| >10 mg† ever (%) | 64.0 | 80.5 | 0.083 |
|
40.0 | 43.9 | 0.707 |
|
16.0 | 12.2 | 0.606 |
|
24.0 | 9.8 | 0.076 |
|
24.0 | 53.7 | <0.01 |
| Cumulative GC dose (mg†) since R-irAE onset (Q1;Q3) | 2,331.5 (840.9; 4452.5) | 2,087.5 (1,250.0; 6815.5) | 0.259 |
|
915.0 (203.1; 2367.5) | 1,160.0 (391.3; 3280.0) | 0.225 |
| Duration of GC treatment (days) (Q1;Q3) | 152.5 (83.8; 438.8) | 201.0 (92.0; 638.5) | 0.243 |
|
92.5 (20.0; 346.3) | 147.0 (21.0; 397.0) | 0.360 |
|
34.0 | 46.3 | 0.231 |
| GC successfully tapered due to R-irAE remission (%) | 41.9 | 28.6 | 0.224 |
| DMARDs | |||
| Any DMARD treatment since ICI line start related to R-irAE (%) | 16 (32.0) | 10 (24.4) | 0.424 |
|
3.0 (1.1; 6.7) | 3.5 (1.1; 5.7) | 0.931 |
| csDMARDs since ICI line start related to R-irAE (%) | 30.0 | 17.1 | 0.152 |
|
8 (16.0) | 6 (14.6) | 0.857 |
|
0 | 1 (2.4) | 0.267 |
|
7§ (14.0) | 0 (0) | <0.05 |
| bDMARDs since ICI line start related to R-irAE (%) | 5 (10.0) | 5 (12.2) | 0.739 |
|
4 (8.0) | 5 (12.2) | 0.505 |
|
2 (4.0) | 1 (2.4) | 0.678 |
|
1†† (2.0) | 0 (0) | 0.363 |
| tsDMARDs since ICI line start related to R-irAE (%) | 0 (0) | 0 (0) | 1.000 |
| ICI | |||
| ICI temporarily discontinued due to any irAE (%) | 23 (46.9) | 8 (20.5) | <0.05 |
|
5 (10.2) | 5 (12.8) | 0.701 |
| ICI permanently discontinued due to any irAE (%) | 21 (42.9) | 19 (46.3) | 0.740 |
|
6 (12.2) | 14 (34.1) | <0.05 |
Numbers represent median (first quartile (Q1) and third quartile (Q3)) (all metric values not normally distributed) or frequencies (%).
*One patient with NSCLC and SpA-like R-irAE has declined any kind of treatment for his symptoms.
†Prednisolone equivalent.
‡Only DMARDs initiated after R-irAE onset are regarded here.
§Mycophenolate (n=4, due to other organ irAE), sulfasalazine (n=2), azathioprine (n=1, due to other organ irAE).
¶Adalimumab and infliximab in sequence in MM (n=1), NSCLC (n=3).
**Tocilizumab, pretreated with TNFi in MM (n=1), NSCLC (n=1).
††Ustekinumab (n=1), pretreated with TNFi.
cs/b/tsDMARD, conventional synthetic/biological/targeted synthetic disease modifying anti-rheumatic drugs; ICI, immune checkpoint inhibitor(s); IL-6i, interleukin 6 inhibitors; irAE, immune-related adverse event; MM, malignant melanoma; NSAID, non-steroidal anti-inflammatory drug; NSCLC, non-small lung cancer; REV, rheumatologic evaluation; R-irAE, rheumatic irAE; SpA, spondyloarthritis; TNFi, tumor necrosis factor inhibitors.
DMARDs were required for the treatment of R-irAE in ~28% of the patient cohorts. MTX was the most commonly used drug (~15%), while bDMARDs (mostly TNFi) were required in only ~11% of cases. Temporary discontinuation of ICI therapy due to any irAE was significantly more frequent in patients with MM (46.9% vs 20.5%), while permanent discontinuation occurred in a comparable proportion of both cohorts (~44%). For management of R-irAEs, ICIs were discontinued temporarily in ~11% and permanently in 12.2% versus 34.1% (p=0.013) of patients with MM and patients with NSCLC, respectively (table 2).
Timing of R-irAE onset
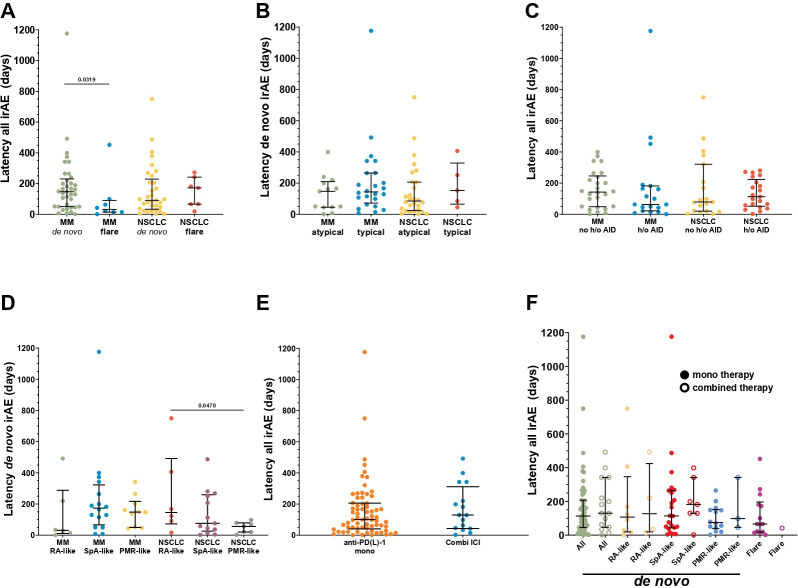
R-irAEs occurred with a median latency of ~4 months (MM: 129.5 days (42.75; 225.0); NSCLC: 98.0 days (41.0; 224.5)) from the start of ICI therapy. Subanalyses showed a significantly earlier onset of flares of pre-existing conditions versus de novo R-irAEs in patients with MM (32.0 days (14.0; 90.8) vs 147.0 days (50.0; 230.0), p=0.032) (figure 1A). Conversely, a trend towards shorter latencies was observed rather in de novo cases with atypical versus typical presentations (clinically suspicious for new-onset of classical RMD) in patients with NSCLC (85.0 days (23.8; 206.5) vs 153.0 days (65.0; 328.5), p=0.338), but not in patients with MM (147.0 days (45.0; 209.5) vs 143.5 days (72.0; 264.8), p=0.353) (figure 1B). Medical and family history of any autoimmune disease (including RMD and psoriasis, table 1) as a potential risk factor for an R-irAE occurrence was not associated with differences in latency times (figure 1C). Notably, within de novo subtypes in patients with NSCLC, PMR-like R-irAEs had a significantly shorter latency than RA-like R-irAEs (56.0 days (20.0; 79.0) vs 145.5 days (72.25; 492.0), p=0.048) with a similar trend compared with SpA-like (76.0 days (25.5; 259.5)). In patients with MM, RA-like R-irAE latencies were the shortest (31.0 days (10.8; 288.0)), while SpA-like (173.5 days (66.3; 322.5)) did not markedly differ from PMR-like R-irAE courses (148.0 days (49.8; 217.0)). The latter was, however, significantly longer compared with PMR-like latencies in patients with NSCLC (p=0.025) (figure 1D). Of note, the latencies of R-irAE onset were not significantly different between anti-PD-1/PD-L1 alone versus combined ICI treatment in our overall cohort (100.0 days (40.5; 206.0) vs 129.5 days (43.0; 311.5), p=0.353) (figure 1E). While there was a trend towards longer latencies for patients with MM on combined ICI treatment (mono (n=31): 107.0 days (28.0; 189.0] vs combined (n=14): 156.0 days (45.0; 342.0), p=0.254), the opposite was true for patients with NSCLC, however, with the limitation of only two patients with NSCLC on combined ICI therapy (mono (n=38): 94.5 days (43.0; 244.3) vs combined (n=2): 50.0 days (2.0; n/a), p=0.310). In addition, when separated by the ICI treatment regimen, the latencies of individual irAE-subtypes also did not differ significantly, although the validity of this finding is also limited by the small number of patients in this subanalysis (figure 1F).
Figure 1.
Dot plots of individual latencies from start of (R-irAE-related) ICI therapy until R-irAE onset in patients with MM versus patients with NSCLC. Five patients with MM and one patients with NSCLC experiencing >1 distinct R-irAE were excluded for this analysis. Bars indicate median (first quartile; third quartile). Latency values between different groups were compared by Kruskal-Wallis test and uncorrected Dunn’s test. (A) Latencies in de novo versus flare R-irAE subtypes. (B) Comparison of latencies between clinically classified new onset of classical rheumatic and musculoskeletal disease (“typical”) and atypical clinical presentations (“atypical”) of de novo R-irAEs. (C) Latencies separated according to presence (“history of (h/o)”) or absence (“no history of; no (h/o)”) of patient’s medical and/or family history of any autoimmune disease (AID) in de novo and flare R-irAEs as a potential risk factor for R-irAE occurrence. (D) Latencies of different de novo R-irAE subtypes: RA, rheumatoid arthritis-like; SpA, spondyloarthritis-like; PMR, polymyalgia rheumatica-like. (E) Comparison of latencies between patients treated with anti-PD-1/PD-L1 only versus combination therapy of anti-PD-1/PD-L1 and anti-CTLA-4 at R-irAE onset. MM and NSCLC cohorts were merged for this analysis as combined ICI treatment applies only to two patients with NSCLC. (F) Latencies of de novo and flare R-irAE subtypes of merged MM and NSCLC cohorts separated by anti-PD-1/PD-L1 only versus combination therapy of anti-PD-1/PD-L1 and anti-CTLA-4 at R-irAE onset. CTLA-4, cytotoxic T-lymphocyte associated protein 4; ICI, immune checkpoint inhibitor; irAE, immune-related adverse event; MM, malignant melanoma; NSCLC, non-small cell lung cancer; PD-1/PD-L1, programmed death-1/its ligand; R-irAE, rheumatic irAE.
Oncological outcomes related to R-irAE
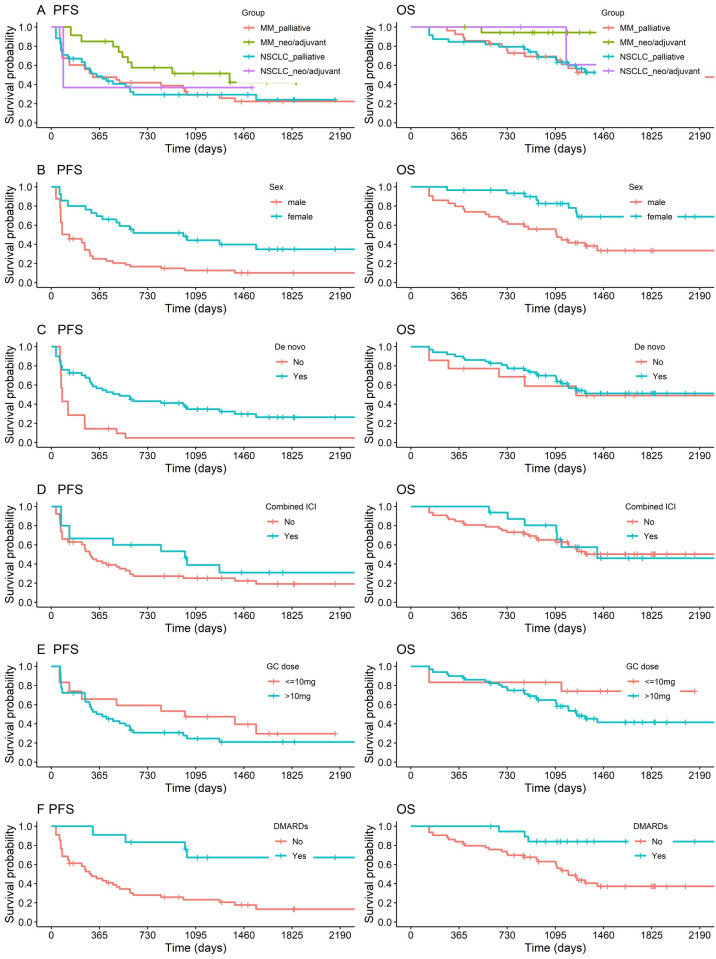
R-irAEs, like irAEs in general, appear to be associated with a favorable tumor outcome.3 6 28 29 However, it is not clear which patients benefit most and whether this beneficial effect is affected by R-irAE treatment. To address this question, we performed Kaplan-Meier survival analyses (figure 2). To adjust for the confounder of significantly better OS (p=0.028) of patients with neoadjuvant/adjuvant ICI treatment and only a small fraction of these in the NSCLC cohort, exclusively patients with palliative ICI treatment at R-irAE onset were included in further subanalyses, resulting in comparable PFS and OS in the remaining patients with MM and patients with NSCLC (figure 2A). PFS and OS of patients with R-irAE analyzed by sex showed a significant survival benefit for female patients (PFS: p=0.013; OS: p=0.009) (figure 2B). De novo R-irAEs were associated with significantly better PFS (p=0.010) but not OS compared with flare R-irAEs (figure 2C). ICI treatment regimen was associated with a trend to better short-term, but not long-term PFS and comparable OS. GC doses >10 mg prednisolone equivalent used for any indication after R-irAE onset appeared to be associated with a negative effect on OS (p=0.100) particularly for doses ≥1 mg/kg (OS: 0.068, sex-adjusted PFS: p=0.056, OS: p=0.051), suggesting a significant effect might be observed in a larger cohort (figure 2E, online supplemental figure 4D, 5E, table 3). The use of GC >10 mg specifically for R-irAE treatment had similar unfavorable effects for PFS as its use as adjunctive medication for chemotherapy or radiotherapy. Strikingly, the use of GC >10 mg specifically for treatment of irAEs other than R-irAEs appeared to affect the PFS adversely to less or no extent (online supplemental figure 4E). Use of DMARDs for any cause was associated with significantly better PFS (n=14, p=0.011) and OS (n=20, p=0.018), and no difference was detectable between patients receiving csDMARDs or bDMARDs in our cohort (figure 2F; online supplemental figure 4F). However, the validity of this finding needs to be verified in a larger cohort. Early R-irAE onset (≤165 days), duration and remission of R-irAEs did not significantly affect PFS or OS (online supplemental figure 4).
Figure 2.
Progression-free (PFS) and overall survival (OS) analyzed by Kaplan–Meier method. For all PFS analyses, patients with oncological progression before R-irAE onset under ICI treatment line related to R-irAE were excluded. Univariate comparisons of Kaplan-Meier estimators were done with the log-rank test. (A) PFS and OS in patients with MM versus patients with NSCLC separated by neoadjuvant or adjuvant (PFS/OS: MM=18/19; NSCLC=3/3) compared with palliative ICI treatment lines (PFS/OS: MM=24/31; NSCLC=32/38) at R-irAE onset (merged MM and NSCLC neo-/adjuvant vs palliative PFS: p=0.187; OS: p=0.028). (B–F) PFS and OS of merged MM and NSCLC cohorts limited to palliative ICI treatment lines at R-irAE onset separated by: (B) sex (PFS/OS: female=28/33 vs male=28/36; PFS: p=0.013; OS: p=0.009); (C) R-irAE subtypes (PFS/OS: de novo=45/56 vs flare=11/12; PFS: p=0.010; OS: p=0.827); (D) anti-PD-1/PD-L1 only versus combination therapy of anti-PD-1/PD-L1 and anti-CTLA-4 at R-irAE onset (PFS/OS: monotherapy=43/53 vs combined ICI treatment=13/16; PFS: p=0.336; OS: p=0.968); (E) GC dose for any indication since R-irAE onset (PFS/OS: ≤10 mg=15/12 vs >10 mg prednisolone equivalent=36/54; PFS: p=0.315; OS: p=0.100); (F) DMARD use (PFS/OS: DMARDs (any kind)=14/20 vs no DMARDs=38/49; PFS: p=0.011; OS: p=0.018; patients with DMARD initiation after progress time point were regarded in OS-subanalysis). CTLA-4, cytotoxic T-lymphocyte associated protein 4; DMARDs, disease modifying anti-rheumatic drugs; GC, glucocorticoid; ICI, immune checkpoint inhibitor; MM, malignant melanoma; NSCLC, non-small cell lung cancer; PD-1/PD-L1, programmed death-1/its ligand; R-irAE, rheumatic immune-related adverse event.
Table 3.
Cox regression analysis of selected confounders
| Confounder | Adjustment | HR | 95% CI | P value |
| Cancer type | PFS: NSCLC effect (vs MM) | 0.974 | 0.496 to 1.911 | 0.939 |
| Female sex effect | 0.424 | 0.216 to 0.832 | 0.013 | |
| OS: NSCLC effect (vs MM) | 0.957 | 0.459 to 1.996 | 0.906 | |
| Female sex effect | 0.338 | 0.149 to 0.768 | 0.001 | |
| PFS: NSCLC effect (vs MM) | 0.746 | 0.343 to 1.622 | 0.460 | |
| Combined ICI treatment effect | 0.546 | 0.206 to 1.445 | 0.223 | |
| OS: NSCLC effect (vs MM) | 0.759 | 0.321 to 1.794 | 0.529 | |
| Combined ICI treatment effect | 0.823 | 0.301 to 2.248 | 0.704 | |
| R-irAE subtype | PFS: De novo effect (vs flare) | 0.343 | 0.153 to 0.767 | 0.009 |
| NSCLC effect | 1.119 | 0.565 to 2.219 | 0.747 | |
| OS: De novo effect (vs flare) | 0.918 | 0.347 to 2.432 | 0.864 | |
| NSCLC effect | 0.846 | 0.401 to 1.787 | 0.662 | |
| PFS: De novo effect (vs flare) | 0.420 | 0.188 to 0.940 | 0.034 | |
| Female sex effect | 0.475 | 0.239 to 0.945 | 0.034 | |
| OS: De novo effect (vs flare) | 1.218 | 0.453 to 3.277 | 0.696 | |
| Female sex effect | 0.328 | 0.142 to 0.756 | 0.009 | |
| PFS: De novo effect (vs flare) | 0.371 | 0.165 to 0.833 | 0.016 | |
| Combined ICI treatment effect | 0.780 | 0.330 to 1.842 | 0.571 | |
| OS: De novo effect (vs flare) | 0.907 | 0.339 to 2.422 | 0.845 | |
| Combined ICI treatment effect | 0.946 | 0.378 to 2.367 | 0.906 | |
| ICI treatment regimen | PFS: Combined ICI treatment effect (vs ICI monotherapy) | 0.776 | 0.331 to 1.821 | 0.561 |
| Female sex effect | 0.440 | 0.222 to 0.871 | 0.018 | |
| OS: Combined ICI treatment effect (vs ICI monotherapy) | 0.983 | 0.419 to 2.305 | 0.968 | |
| Female sex effect | 0.336 | 0.149 to 0.759 | 0.009 | |
| GC dose | PFS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 1.460 | 0.553 to 3.853 | 0.444 |
|
1.582 | 0.652 to 3.834 | 0.310 | |
| NSCLC effect | 1.036 | 0.500 to 2.143 | 0.924 | |
| OS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 2.947 | 0.628 to 13.823 | 0.171 | |
|
3.901 | 0.897 to 16.974 | 0.070 | |
| NSCLC effect | 0.923 | 0.425 to 2.003 | 0.839 | |
| PFS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 1.677 | 0.636 to 4.420 | 0.296 | |
|
2.525 | 0.976 to 6.532 | 0.056 | |
| Female sex effect | 0.274 | 0.128 to 0.587 | 0.001 | |
| OS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 2.866 | 0.617 to 13.323 | 0.179 | |
|
4.320 | 0.992 to 18.814 | 0.051 | |
| Female sex effect | 0.297 | 0.130 to 0.676 | 0.004 | |
| PFS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 1.152 | 0.427 to 3.112 | 0.780 | |
|
1.639 | 0.681 to 3.948 | 0.270 | |
| De novo effect | 0.323 | 0.133 to 0.783 | 0.012 | |
| OS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 2.820 | 0.604 to 13.161 | 0.187 | |
|
3.858 | 0.875 to 17.005 | 0.075 | |
| De novo effect | 1.065 | 0.400 to 2.841 | 0.899 | |
| PFS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 1.305 | 0.488 to 3.490 | 0.596 | |
|
1.736 | 0.706 to 4.267 | 0.230 | |
| Combined ICI treatment effect | 0.621 | 0.247 to 1.560 | 0.310 | |
| OS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 2.942 | 0.624 to 13.866 | 0.172 | |
|
3.925 | 0.903 to 17.063 | 0.068 | |
| Combined ICI treatment effect | 1.074 | 0.443 to 2.604 | 0.875 | |
| PFS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 1.243 | 0.471 to 3.284 | 0.660 | |
|
1.530 | 0.639 to 3.663 | 0.340 | |
| DMARD (any kind) ever effect | 0.274 | 0.103 to 0.729 | 0.009 | |
| OS: GC dose >10 mg <1 mg/kg * ever (vs ≤10 mg* only) | 2.229 | 0.476 to 10.442 | 0.309 | |
|
3.087 | 0.705 to 13.522 | 0.135 | |
| DMARD (any kind) ever effect | 0.252 | 0.075 to 0.842 | 0.025 | |
| DMARD | PFS: DMARD (any kind) ever (vs no DMARD) | 0.268 | 0.010 to 0.720 | 0.009 |
| NSCLC effect | 0.725 | 0.363 to 1.450 | 0.363 | |
| OS: DMARD (any kind) ever (vs no DMARD) | 0.219 | 0.066 to 0.731 | 0.014 | |
| NSCLC effect | 0.694 | 0.333 to 1.447 | 0.330 | |
| PFS: DMARD (any kind) ever (vs no DMARD) | 0.361 | 0.135 to 0.961 | 0.041 | |
| Female sex effect | 0.517 | 0.259 to 1.030 | 0.061 | |
| OS: DMARD (any kind) ever (vs no DMARD) | 0.270 | 0.081 to 0.897 | 0.033 | |
| Female sex effect | 0.380 | 0.168 to 0.862 | 0.021 | |
| PFS: DMARD (any kind) ever (vs no DMARD) | 0.331 | 0.126 to 0.875 | 0.026 | |
| De novo effect | 0.415 | 0.187 to 0.921 | 0.031 | |
| OS: DMARD (any kind) ever (vs no DMARD) | 0.227 | 0.068 to 0.761 | 0.016 | |
| De novo effect | 0.712 | 0.268 to 1.890 | 0.495 | |
| PFS: DMARD (any kind) ever (vs no DMARD) | 0.290 | 0.104 to 0.811 | 0.018 | |
| Combined ICI treatment effect | 1.073 | 0.436 to 2.639 | 0.878 | |
| OS: DMARD (any kind) ever (vs no DMARD) | 0.221 | 0.066 to 0.745 | 0.015 | |
| Combined ICI treatment effect | 1.306 | 0.550 to 3.100 | 0.545 |
Analyses limited to palliative ICI treatment lines at R-irAE onset.
*Prednisolone equivalent.
DMARD, disease modifying anti-rheumatic drugs; GC, glucocorticoids; ICI, immune checkpoint inhibitor(s); MM, malignant melanoma; NSCLC, non-small cell lung cancer; OS, overall survival; PFS, progression-free survival; R-irAE, rheumatic immune-related adverse event.
Confounder analyses
To identify confounding effects between variables adjusted Cox regression models were calculated (table 3). Sex proved to be the strongest confounding factor with beneficial influence of female sex on all PFS and OS subanalyses depicted in figure 2. However, when adjusted for sex, a significantly favorable effect of de novo R-irAE subtype (vs flare) on PFS, of DMARD use on PFS and OS and in trend of GC >10 mg particularly for doses ≥1 mg/kg mainly on OS was preserved. Cancer type and ICI treatment regimen had no effects on cancer outcome in adjusted Cox regression analyses (table 3).
The distinct sex-dependent differences in survival of patients with R-irAE in our cohort prompted us to compare the sex distribution of all available parameters to rule out confounding and to evaluate sex as a confounder for other observed outcome differences. As possible confounders of sex-dependent outcomes, we identified a higher proportion of additional malignancies in men and the number of ICI treatment lines as the strongest feature, with significantly more females receiving a first-line ICI treatment at the time of R-irAE onset (online supplemental table 5). However, survival analyses of patients receiving only first-line ICIs at R-irAE onset, which had a more balanced distribution of additional malignancies between the sexes, confirmed the previously observed survival trends (online supplemental figure 5). In survival analyses of the overall R-irAE study population, the significantly worse outcomes for male patients with R-irAE were maintained despite the survival benefit for adjuvantly treated patients. Overall, deaths from any cause were significantly more likely in male patients with R-irAE in all subcohorts (online supplemental table 5).
Interestingly, in female patients we observed a strong trend towards more frequent joint involvement and/or back pain, which probably explains why more women received DMARDs. A higher proportion of women in the combined cohort were treated with DMARDs, which may have contributed to the improved PFS and OS in DMARD patients (figure 2F; figure 3F). In contrast, the sex distribution was balanced for GC dose >10 mg and de novo R-irAE type, making it unlikely that sex was a confounder for the differences in outcome related to these parameters (online supplemental table 5).
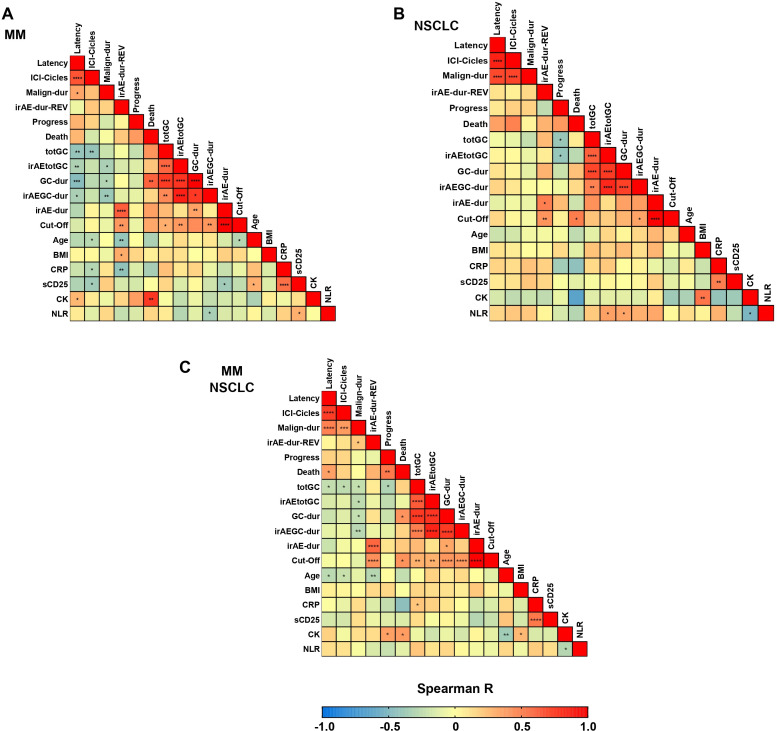
Figure 3.
Spearman correlogram of baseline and outcome related metric variables in (A) MM cohort, (B) NSCLC cohort and (C) merged MM and NSCLC cohorts. *p<0.05; **p<0.01; ***p<0.001. Variables: Age, Age R-irAE onset; BMI, body mass index; CK, maximum value of creatine kinase; CRP, maximum value of c-reactive protein; Cut-Off, R-irAE duration until cut-off (days); Death, time until death (days); GC, glucocorticoid; GC-dur, GC treatment duration (days) since R-irAE onset; ICI, immune checkpoint inhibitor; ICI-Cycles, number of cycles of ICI related to irAE-onset; irAE, immune-related adverse event; irAE-dur; R-irAE duration from onset until remission (days); irAE-dur-REV, R-irAE duration until first rheumatologic evaluation (days); irAEGC-dur, R-irAE related GC treatment duration (days); irAEtotGC, R-irAE related cumulative GC dose (mg prednisolone equivalent); Latency, duration from start of (R-irAE-related) ICI therapy until R-irAE onset; Malign-dur, malignancy duration until R-irAE onset (months); NLR, neutrophil-to-lymphocyte ratio at ICI start; Progress, time until progress (days); REV, rheumatologic evaluation; R-irAE, rheumatic irAE; sCD25, maximum value of soluble IL-2 receptor (sCD25); totGC; Cumulative GC dose (mg prednisolone equivalent) since R-irAE onset.
The significantly worse PFS in patients with flares of a pre-existing rheumatic condition prompted us to investigate additional confounding factors. We identified differences in clinical (ie, more frequent (large) joint inflammation and stiffness) and serological (C-reactive protein elevation) parameters suggesting a more inflammatory phenotype in these patients compared with de novo R-irAEs. This may explain why these patients were more likely to receive GC doses >10 mg and reached significantly higher R-irAE-related cumulative GC doses and treatment durations during follow-up, which may have contributed to the worse PFS. Notably, DMARD use was more frequent and started earlier in flare patients (online supplemental table 6).
As differences in regard to latency and severity of irAEs are known to depend on ICI treatment regimen (PD-(L)1 inhibitor monotherapy vs combination with CTLA-4 inhibitors), we additionally investigated this parameter as a possible confounder despite the lack of effect on cancer outcome in Cox regression analyses (table 3, online supplemental table 7). The vast majority of patients receiving combined ICI treatment had an MM, explaining the significantly longer cancer duration observed for patients in this group (table 1). Importantly, sex, death rates and R-irAE related GC treatment durations were evenly distributed in all subcohorts regardless of ICI regimen. In the combined ICI groups we found a small trend to more de novo R-irAEs (~80% vs 93%), higher rates of reported myalgia, myositis and (mostly clinically irrelevant) CK elevation, but lower rates of tenosynovitis. Expectedly, there was a trend for more other organ irAEs (significant only in patients on first-line ICIs at R-irAE onset) and significantly more liver irAEs under combined ICI treatment in all subcohorts. Subsequently, combined ICI treatment led to significantly more frequent GC use >10 mg due to other organ irAE and a trend to higher cumulative GC doses and treatment durations. Oppositely, additional exposure to chemotherapy was more frequent in patients with NSCLC predominant in the monotherapy subgroup and accordingly, GC use >10 mg due to chemotherapy was observed rather in the monotherapy groups. Accordingly, any DMARDs and particularly csDMARDs other than methotrexate or leflunomide due to other organ irAE were significantly more likely to be initiated in patients receiving combined ICIs with a trend to faster first initiation, while rates of bDMARD prescriptions did not differ. Importantly, regardless of ICI regimen, patients were equally likely to reach complete remission of the R-irAE during follow-up (online supplemental table 7).
Correlation between baseline variables, treatment, and patient outcomes
The two measures of time to R-irAE onset, latency time from ICI initiation and time from diagnosis of malignancy, were strongly positively correlated in both the combined and separate MM and NSCLC cohorts (figure 3). Both parameters in the MM cohort (figure 3A) and mainly malignancy duration in the combined cohort (figure 3C) were significantly negatively correlated with almost all GC dose and duration parameters. This may indicate either that a shorter time to R-irAE onset was associated with higher levels of GC, or that the probability of developing R-irAEs or other irAEs requiring GC treatment decreases with the duration of ICI treatment and/or malignancy duration. Consistent with the findings in the Kaplan-Meier analysis, cumulative GC for any cause (combined and NSCLC cohorts) and cumulative R-irAE-related GC (NSCLC cohort) were negatively correlated with time to progression (figure 3B and C), suggesting an unfavorable effect of higher cumulative GC on cancer outcomes. The finding of a significant positive correlation between GC treatment duration and time to death in the combined and MM cohorts is at first counterintuitive in this context, but may be attributed to a higher likelihood of longer GC use in patients who survive longer. Finally, all GC treatment parameters in patients with NSCLC were positively correlated with NLR levels (figure 3B), which have been previously shown to be associated with worse cancer outcomes,31 highlighting the potential prognostic value of NLR levels at ICI initiation in patients with NSCLC. Importantly, initial CRP or sCD25 levels are unlikely to be useful predictors of GC treatment after the onset of R-irAE.
Discussion
The increasing prevalence of chronic or recurrent R-irAEs due to the prolonged survival of ICI-treated patients, as well as the expanding use of ICIs in early-stage indications and different tumors, poses new challenges to rheumatologists, oncologists and general practitioners. Inadequately recognized, diagnosed and managed R-irAEs are frequently associated with functional impairment and chronically debilitating courses. Thus, they often limit continuation of ICI therapy and jeopardize clinical benefit.1 A better understanding of the characteristics and clinical management of (R-)irAE, as well as their impact on individual patient cancer outcomes is of crucial importance for the long-term management of these patients.
Although baseline characteristics were largely comparable between the two patient cohorts, a slightly younger mean age in patients with MM and a higher proportion of smokers were observed in patients with NSCLC which could be expected based on previous data.32 The majority of prospectively recruited patients in our study had de novo R-irAEs, as opposed to flares of pre-existing RMDs, in line with a large retrospective case–control study.13 The most frequent R-irAEs in both cohorts, in descending order, were SpA-like, PMR-like and RA-like subtypes. Fulfillment of classification criteria for classical RMDs in de novo R-irAEs was low, but generally in line with previous findings.1 Despite several baseline differences between our MM and NSCLC cohorts, R-irAE characteristics and outcomes were strikingly similar. This finding is of major importance for future R-irAE-focused clinical trials, as—after validation in further cancer entities—it may allow conclusions to be drawn across different oncological subpopulations and improve the daily oncology practice. The selection of appropriate control patients for these studies will depend on a specific time frame within the ICI treatment. IrAEs in different organ systems show distinct peaks in latency from initiation of ICIs to their onset,33 and R-irAEs appear to be consistent with this observation.34 In this context, we were able to confirm the previously reported shorter latency in PMR-like R-irAEs,34 but conversely, saw a trend to longer latencies in patients with MM with combined ICI therapy. The correlation between latencies and malignancy duration may explain the differences observed between MM and NSCLC cohorts for latencies of PMR-like R-irAEs and of R-irAEs under combined ICI treatment. Interestingly, both observed malignancy duration and latency until R-irAE negatively correlated with GC treatment since R-irAE onset. This may suggest that the longer the cancer has been present, the weaker the inflammatory response, both in terms of timing of R-irAE onset from ICI initiation and the intensity of immunosuppressive treatment required.
Consistent with previously published data, we observed higher mortality rates in the overall NSCLC population compared with patients with MM receiving ICI during our extensive follow-up of ~33 months.35 However, this difference in outcomes may be explained by more adjuvant ICI therapies in the MM group, as cancer type showed no effect on outcome in adjusted Cox regression analyses in the palliative subcohort. The significantly worse oncological outcome in male patients with R-irAE in our study is an interesting finding that adds to the knowledge in this field. In the pre-ICI era, worse survival rates have been reported for men with MM.36 37 However, for patients with cancer undergoing immunotherapy in general—without the specific focus on R-irAE—there are conflicting data from several large meta-analyses regarding outcomes in men versus women with NSCLC or MM, including a better but not significant ICI response38–41 and longer PFS in men,40–42 inconsistent outcomes depending on the type of ICI,43–46 and a better outcome in women on CTLA-4 therapy42. While only minimal sex-associated differences were observed for irAE characteristics,47 including the likelihood for chronicity,48 additional larger studies are required to explore the influence of sex on outcomes of patients who develop R-irAEs during ICI therapy.
Our study provides novel, clinically-relevant evidence that potential superiority of DMARD use to extensive GC treatment for R-irAEs in terms of patient survival urgently needs to be addressed in large randomized controlled trials. GC doses >10 mg specifically for the treatment of R-irAEs, but not for other irAEs, appeared to adversely affect PFS to a similar extent as in patients receiving chemotherapy or radiotherapy and GC as adjunctive medication. This finding challenges the view that GC >10 mg prednisolone equivalent prior to ICI treatment,49 but not after ICI initiation,50 is a negative predictor for optimal outcomes. Further support for our findings comes from recent preclinical evidence that CD8 T cells from patients with R-irAE have a similar metabolic profile to CD8 T cells from patients with autoimmune arthritis and could therefore derive similar benefit from DMARDs typically used in classical RMDs.51 Overall, these findings challenge the current strategy of a restrictive, second-line (b)DMARD use in the management of R-irAE.1 Recent data from an observational study with a different methodological approach showed that the time to cancer progression in patients with de novo ICI arthritis was significantly shorter for patients treated with TNFi, and to lesser extent IL-6 inhibitors, compared with the csDMARD methotrexate.30 Thus, despite a more rapid symptom relief and the hypothesis of possible superiority in pre-existing autoimmune conditions,52 the maintenance of the restrictive use of bDMARDs was favored.30 In line with this status quo, our rather restrictive use of DMARDs limits a reliable discrimination of csDMARD versus bDMARD effects on cancer outcome and the lack of difference observed in our cohort has to be interpreted with great caution. However, our patients overall did appear to benefit when GC >10 mg could be avoided as an alternative to DMARD initiation in more severe cases. Thus, in addition to the issues raised in this recent study,30 our findings pave the way for future large randomized controlled trials addressing the fundamental question of whether and when DMARDs should be initiated in general. In particular, the finding of poorer PFS in patients with flares—a study population largely excluded from previous ICI clinical trials52–54—possibly due to a more extensive GC use owing to a more inflammatory and persistent phenotype compared with de novo R-irAEs, adds further relevance to this matter.52 Collectively, the question of a suitable personalized (pre-)treatment strategy for patients with R-irAE with more severe and/or chronic courses needs to be urgently addressed in future randomized controlled cancer trials.1 2 6–11
A strength of our study is the comprehensive prospective collection of data (169 parameters) on R-irAE and cancer with extensive statistical analysis limited to two cancer subgroups and reporting depth rarely found in other studies according to a recent scoping literature review.5 Limitations of this study include shared decision-making as an important factor in DMARD initiation and limited statistical power for some outcomes due to small numbers of patients for certain subgroups. Furthermore, we chose to focus here specifically on R-irAEs in routine interdisciplinary care without comparison to non-(R-)irAE controls, nor the exploration of novel molecular biomarkers, as we have addressed both in previous studies.3 6 55 However, given the rarity of R-irAE focused trials, our study represents a relatively large prospective data set.
Evidence-based toxicity management is critical, not only for individual cancer outcomes, but also for the overall advancement of oncology practice. In this context, our study adds important new evidence that will improve patient-centered, interdisciplinary care for the growing number of patients with R-irAE.
Acknowledgments
We thank the physicians and nursing teams of the rheumatology and oncology departments at Heidelberg and Mannheim University Hospitals.
For the publication fee we acknowledge financial support by Deutsche Forschungsgemeinschaft (DFG) within the funding programme „Open Access Publikationskosten“ as well as by Heidelberg University.
Footnotes
Contributors: KG, LDi, H-ML, PC, and JL contributed to conception and design of the study. KG and LDi prepared and organized the database. KG, LDi, LDa, JW, VO, JG, H-ML, JCH, PC, and JL contributed cases and/or evaluated cases. KG, JG, H-ML, and JL evaluated patients on-site as rheumatologists. JCH and PC were involved as oncologists in charge of malignancy treatment and immune checkpoint inhibitors toxicity management. MF was involved as a statistician. KG, MF, and MMS-C performed the statistical analysis. KG, LDi, and JL wrote the first draft of the manuscript and drafted tables. MMS-C and MF drafted figures. All authors contributed to manuscript revision, read and approved the submitted version. Guarantor: KG.
Funding: Institutional (unrestricted Olympia Morata Program fellowship by the medical faculty of University of Heidelberg to KG) and partial governmental funding (to PC) for patient with non-small cell lung cancer analysis by the German Center for Lung Research (DZL).
Competing interests: No, there are no competing interests.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. All data relevant to the study are included in the article or uploaded as supplementary information. All cumulative data are presented in the manuscript and appendix. Raw data will be made available upon reasonable request from the corresponding author.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
The study was officially approved by the institutional ethics committees of the Heidelberg University (S-391/2018) and Medical faculty Mannheim (2019-518N), Germany. All patients provided written informed consent for participation in the study.
References
- 1. Kostine M, Finckh A, Bingham CO, et al. EULAR points to consider for the diagnosis and management of rheumatic immune-related adverse events due to cancer Immunotherapy with Checkpoint inhibitors. Ann Rheum Dis 2021;80:36–48. 10.1136/annrheumdis-2020-217139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Benesova K, Lorenz H-M, Leipe J, et al. How I treat cancer: treatment of Rheumatological side effects of Immunotherapy. ESMO Open 2019;4(Suppl 4):e000529. 10.1136/esmoopen-2019-000529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Daniello L, Elshiaty M, Bozorgmehr F, et al. Therapeutic and Prognostic implications of immune-related adverse events in advanced non-small-cell lung cancer. Front Oncol 2021;11:703893. 10.3389/fonc.2021.703893 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Braaten TJ, Brahmer JR, Forde PM, et al. Immune Checkpoint inhibitor-induced inflammatory arthritis persists after Immunotherapy cessation. Ann Rheum Dis 2020;79:332–8. 10.1136/annrheumdis-2019-216109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Ghosh N, Couette N, van Binsbergen WH, et al. Identification of outcome domains in immune Checkpoint inhibitor-induced inflammatory arthritis and Polymyalgia Rheumatica: A Scoping review by the OMERACT irAE working group. Semin Arthritis Rheum 2023;58:152110. 10.1016/j.semarthrit.2022.152110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Buder-Bakhaya K, Benesova K, Schulz C, et al. Characterization of arthralgia induced by PD-1 antibody treatment in patients with Metastasized cutaneous malignancies. Cancer Immunol Immunother 2018;67:175–82. 10.1007/s00262-017-2069-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Suarez-Almazor ME, Kim ST, Abdel-Wahab N, et al. Review: immune-related adverse events with use of Checkpoint inhibitors for Immunotherapy of cancer. Arthritis Rheumatol 2017;69:687–99. 10.1002/art.40043 [DOI] [PubMed] [Google Scholar]
- 8. Cappelli LC, Gutierrez AK, Bingham CO, et al. Rheumatic and musculoskeletal immune-related adverse events due to immune Checkpoint inhibitors: A systematic review of the literature. Arthritis Care Res (Hoboken) 2017;69:1751–63. 10.1002/acr.23177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Hassel JC, Heinzerling L, Aberle J, et al. Combined immune Checkpoint blockade (anti-PD-1/anti-CTLA-4): evaluation and management of adverse drug reactions. Cancer Treat Rev 2017;57:36–49. 10.1016/j.ctrv.2017.05.003 [DOI] [PubMed] [Google Scholar]
- 10. Veccia A, Kostine M, Tison A, et al. Rheumatic immune-and nonimmune-related adverse events in phase 3 clinical trials assessing PD-(L)1 Checkpoint inhibitors for lung cancer: A systematic review and meta-analysis. Joint Bone Spine 2022;89:105403. 10.1016/j.jbspin.2022.105403 [DOI] [PubMed] [Google Scholar]
- 11. Woodworth T, Furst DE, Alten R, Bingham CO, 3rd, Yocum D, Sloan V, et al . Standardizing assessment and reporting of adverse effects in rheumatology clinical trials II: the rheumatology common toxicity criteria V.2.0. J Rheumatol 2007;34:1401–14. [PubMed] [Google Scholar]
- 12. Khoja L, Day D, Wei-Wu Chen T, et al. Tumour- and class-specific patterns of immune-related adverse events of immune Checkpoint inhibitors: a systematic review. Ann Oncol 2017;28:2377–85. 10.1093/annonc/mdx286 [DOI] [PubMed] [Google Scholar]
- 13. Cunningham-Bussel A, Wang J, Prisco LC, et al. Predictors of rheumatic immune-related adverse events and de novo inflammatory arthritis after immune Checkpoint inhibitor treatment for cancer. Arthritis Rheumatol 2022;74:527–40. 10.1002/art.41949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Schneider BJ, Naidoo J, Santomasso BD, et al. Management of immune-related adverse events in patients treated with immune Checkpoint inhibitor therapy: ASCO guideline update. J Clin Oncol 2021;39:4073–126. 10.1200/JCO.21.01440 [DOI] [PubMed] [Google Scholar]
- 15. Haanen JBAG, Carbonnel F, Robert C, et al. Management of toxicities from Immunotherapy: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 2018;29(Suppl 4):iv264–6. 10.1093/annonc/mdy162 [DOI] [PubMed] [Google Scholar]
- 16. on behalf of the Society for Immunotherapy of Cancer Toxicity Management Working Group, Puzanov I, Diab A, et al. Managing toxicities associated with immune Checkpoint inhibitors: consensus recommendations from the society for Immunotherapy of cancer (SITC). J Immunotherapy Cancer 2017;5. 10.1186/s40425-017-0300-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Cappelli LC, Gutierrez AK, Bingham CO, et al. Rheumatic and musculoskeletal immune-related adverse events due to immune Checkpoint inhibitors: A systematic review of the literature. Arthritis Care Res 2017;69:1751–63. 10.1002/acr.23177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Leipe J, Mariette X. Management of rheumatic complications of ICI therapy: a rheumatology viewpoint. Rheumatology (Oxford) 2019;58(Suppl 7):vii49–58. 10.1093/rheumatology/kez360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Chanson N, Ramos-Casals M, Pundole X, et al. Immune Checkpoint inhibitor-associated Sarcoidosis: A usually benign disease that does not require Immunotherapy discontinuation. Eur J Cancer 2021;158:208–16. 10.1016/j.ejca.2021.05.041 [DOI] [PubMed] [Google Scholar]
- 20. Prasad P, Verma S, Surbhi, Ganguly NK, et al. Rheumatoid arthritis: advances in treatment strategies. Mol Cell Biochem 2023;478:69–88. 10.1007/s11010-022-04492-3 [DOI] [PubMed] [Google Scholar]
- 21. Ozen G, Pedro S, England BR, et al. Risk of serious infection in patients with rheumatoid arthritis treated with biologic versus Nonbiologic disease-modifying Antirheumatic drugs. ACR Open Rheumatol 2019;1:424–32. 10.1002/acr2.11064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Foster CC, Couey MA, Kochanny SE, et al. Immune-related adverse events are associated with improved response, progression-free survival, and overall survival for patients with head and neck cancer receiving immune Checkpoint inhibitors. Cancer 2021;127:4565–73. 10.1002/cncr.33780 [DOI] [PubMed] [Google Scholar]
- 23. Zhou Y, Chen H, Tang L, et al. Association of immune-related adverse events and efficacy in advanced non-small-cell lung cancer: a systematic review and meta-analysis. Immunotherapy 2023;15:209–20. 10.2217/imt-2022-0028 [DOI] [PubMed] [Google Scholar]
- 24. Weber JS, Hodi FS, Wolchok JD, et al. Safety profile of Nivolumab monotherapy: A pooled analysis of patients with advanced Melanoma. J Clin Oncol 2017;35:785–92. 10.1200/JCO.2015.66.1389 [DOI] [PubMed] [Google Scholar]
- 25. Eggermont AMM, Kicinski M, Blank CU, et al. Association between immune-related adverse events and recurrence-free survival among patients with stage III Melanoma randomized to receive Pembrolizumab or placebo: A secondary analysis of a randomized clinical trial. JAMA Oncol 2020;6:519–27. 10.1001/jamaoncol.2019.5570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Akamatsu H, Murakami E, Oyanagi J, et al. Immune-related adverse events by immune Checkpoint inhibitors significantly predict durable efficacy even in responders with advanced non-small cell lung cancer. Oncologist 2020;25:e679–83. 10.1634/theoncologist.2019-0299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Fan Y, Xie W, Huang H, et al. Association of immune related adverse events with efficacy of immune Checkpoint inhibitors and overall survival in cancers: A systemic review and meta-analysis. Front Oncol 2021;11:633032. 10.3389/fonc.2021.633032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Kostine M, Rouxel L, Barnetche T, et al. Rheumatic disorders associated with immune Checkpoint inhibitors in patients with cancer-clinical aspects and relationship with tumour response: a single-centre prospective cohort study. Ann Rheum Dis 2018;77:393–8. 10.1136/annrheumdis-2017-212257 [DOI] [PubMed] [Google Scholar]
- 29. Liew DFL, Leung JLY, Liu B, et al. Association of good Oncological response to therapy with the development of rheumatic immune-related adverse events following PD-1 inhibitor therapy. Int J Rheum Dis 2019;22:297–302. 10.1111/1756-185X.13444 [DOI] [PubMed] [Google Scholar]
- 30. Bass AR, Abdel-Wahab N, Reid PD, et al. Comparative safety and effectiveness of TNF inhibitors, Il6 inhibitors and methotrexate for the treatment of immune Checkpoint inhibitor-associated arthritis. Ann Rheum Dis 2023;82:920–6. 10.1136/ard-2023-223885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Giustini N, Bazhenova L. Recognizing Prognostic and predictive biomarkers in the treatment of non-small cell lung cancer (NSCLC) with immune Checkpoint inhibitors (Icis). Lung Cancer (Auckl) 2021;12:21–34. 10.2147/LCTT.S235102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Weng YM, Peng M, Hu MX, et al. Clinical and molecular characteristics associated with the efficacy of PD-1/PD-L1 inhibitors for solid tumors: a meta-analysis. Onco Targets Ther 2018;11:7529–42. 10.2147/OTT.S167865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Spain L, Diem S, Larkin J. Management of toxicities of immune Checkpoint inhibitors. Cancer Treat Rev 2016;44:51–60. 10.1016/j.ctrv.2016.02.001 [DOI] [PubMed] [Google Scholar]
- 34. Gómez-Puerta JA, Lobo-Prat D, Perez-García C, et al. n.d. Clinical patterns and follow-up of inflammatory arthritis and other immune-related adverse events induced by Checkpoint inhibitors. A multicenter study. Front Med;9. 10.3389/fmed.2022.888377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Topalian SL, Hodi FS, Brahmer JR, et al. Five-year survival and correlates among patients with advanced Melanoma, renal cell carcinoma, or non-small cell lung cancer treated with Nivolumab. JAMA Oncol 2019;5:1411. 10.1001/jamaoncol.2019.2187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Gamba CS, Clarke CA, Keegan THM, et al. Melanoma survival disadvantage in young, non-Hispanic white males compared with females. JAMA Dermatol 2013;149:912–20. 10.1001/jamadermatol.2013.4408 [DOI] [PubMed] [Google Scholar]
- 37. Khosrotehrani K, Dasgupta P, Byrom L, et al. Melanoma survival is superior in females across all tumour stages but is influenced by age. Arch Dermatol Res 2015;307:731–40. 10.1007/s00403-015-1585-8 [DOI] [PubMed] [Google Scholar]
- 38. Botticelli A, Onesti CE, Zizzari I, et al. The Sexist behaviour of immune Checkpoint inhibitors in cancer therapy Oncotarget 2017;8:99336–46. 10.18632/oncotarget.22242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Pinto JA, Vallejos CS, Raez LE, et al. Gender and outcomes in non-small cell lung cancer: an old Prognostic variable comes back for targeted therapy and Immunotherapy ESMO Open 2018;3:e000344. 10.1136/esmoopen-2018-000344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Wu Y, Ju Q, Jia K, et al. Correlation between sex and efficacy of immune Checkpoint inhibitors (PD-1 and CTLA-4 inhibitors). Int J Cancer 2018;143:45–51. 10.1002/ijc.31301 Available: http://doi.wiley.com/10.1002/ijc.v143.1 [DOI] [PubMed] [Google Scholar]
- 41. Conforti F, Pala L, Bagnardi V, et al. Cancer Immunotherapy efficacy and patients' sex: a systematic review and meta-analysis. Lancet Oncol 2018;19:737–46. 10.1016/S1470-2045(18)30261-4 [DOI] [PubMed] [Google Scholar]
- 42. Grassadonia A, Sperduti I, Vici P, et al. Effect of gender on the outcome of patients receiving immune Checkpoint inhibitors for advanced cancer: A systematic review and meta-analysis of phase III randomized clinical trials. J Clin Med 2018;7:542. 10.3390/jcm7120542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Wang C, Qiao W, Jiang Y, et al. Effect of sex on the efficacy of patients receiving immune Checkpoint inhibitors in advanced non-small cell lung cancer. Cancer Med 2019;8:4023–31. 10.1002/cam4.2280 Available: https://onlinelibrary.wiley.com/toc/20457634/8/8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Wallis CJD, Butaney M, Satkunasivam R, et al. Association of patient sex with efficacy of immune Checkpoint inhibitors and overall survival in advanced cancers: A systematic review and meta-analysis. JAMA Oncol 2019;5:529–36. 10.1001/jamaoncol.2018.5904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Madala S, Rasul R, Singla K, et al. Gender differences and their effects on survival outcomes in lung cancer patients treated with PD-1/PD-L1 Checkpoint inhibitors: A systematic review and meta-analysis. Clinical Oncology 2022;34:799–809. 10.1016/j.clon.2022.03.010 [DOI] [PubMed] [Google Scholar]
- 46. Kapoor A, Noronha V, Patil V, et al. Impact of sex of the patient on efficacy and safety of cancer Immunotherapy: A retrospective audit. Cancer Res Stat Treat 2021;4:238. 10.4103/crst.crst_111_21 [DOI] [Google Scholar]
- 47. Jing Y, Zhang Y, Wang J, et al. Association between sex and immune-related adverse events during immune Checkpoint inhibitor therapy. J Natl Cancer Inst 2021;113:1396–404. 10.1093/jnci/djab035 [DOI] [PubMed] [Google Scholar]
- 48. Patrinely JR, Johnson R, Lawless AR, et al. Chronic immune-related adverse events following adjuvant anti-PD-1 therapy for high-risk Resected Melanoma. JAMA Oncol 2021;7:744. 10.1001/jamaoncol.2021.0051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Arbour KC, Mezquita L, Long N, et al. Impact of baseline steroids on efficacy of programmed cell Death-1 and programmed death-ligand 1 blockade in patients with non-small-cell lung cancer. J Clin Oncol 2018;36:2872–8. 10.1200/JCO.2018.79.0006 [DOI] [PubMed] [Google Scholar]
- 50. Horvat TZ, Adel NG, Dang T-O, et al. Immune-related adverse events, need for systemic immunosuppression, and effects on survival and time to treatment failure in patients with Melanoma treated with Ipilimumab at Memorial Sloan Kettering cancer center. J Clin Oncol 2015;33:3193–8. 10.1200/JCO.2015.60.8448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Benesova K, Kraus FV, Carvalho RA, et al. Distinct immune-Effector and metabolic profile of Cd8+ T cells in patients with autoimmune Polyarthritis induced by therapy with immune Checkpoint inhibitors. Ann Rheum Dis 2022;81:1730–41. 10.1136/ard-2022-222451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Haanen J, Ernstoff MS, Wang Y, et al. Autoimmune diseases and immune-Checkpoint inhibitors for cancer therapy: review of the literature and personalized risk-based prevention strategy. Ann Oncol 2020;31:724–44. 10.1016/j.annonc.2020.03.285 [DOI] [PubMed] [Google Scholar]
- 53. Khan SS, Komiya T. Exclusion of patients with autoimmune disease in lung cancer clinical trials. Asian Pac J Cancer Prev 2023;24:331–6. 10.31557/APJCP.2023.24.1.331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Khan SA, Pruitt SL, Xuan L, et al. Prevalence of autoimmune disease among patients with lung cancer: implications for Immunotherapy treatment options. JAMA Oncol 2016;2:1507–8. 10.1001/jamaoncol.2016.2238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Schindler H, Lusky F, Daniello L, et al. Serum Cytokines predict efficacy and toxicity, but are not useful for disease monitoring in lung cancer treated with PD-(L)1 inhibitors. Front Oncol 2022;12:1010660. 10.3389/fonc.2022.1010660 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2023-007557supp001.pdf (1.5MB, pdf)
Data Availability Statement
Data are available upon reasonable request. All data relevant to the study are included in the article or uploaded as supplementary information. All cumulative data are presented in the manuscript and appendix. Raw data will be made available upon reasonable request from the corresponding author.